Abstract
Background and Aim
Because acute infectious gastroenteritis may cause post‐infection irritable bowel syndrome and functional dyspepsia and the severe acute respiratory syndrome coronavirus‐2 affects gastrointestinal (GI) tract, coronavirus disease‐19 (COVID‐19) may cause post‐infection‐functional GI disorders (FGIDs). We prospectively studied the frequency and spectrum of post‐infection‐FGIDs among COVID‐19 and historical healthy controls and the risk factors for its development.
Methods
Two hundred eighty patients with COVID‐19 and 264 historical healthy controls were followed up at 1 and 3 months using translated validated Rome Questionnaires for the development of chronic bowel dysfunction (CBD), dyspeptic symptoms, and their overlap and at 6‐month for IBS, uninvestigated dyspepsia (UD) and their overlap. Psychological comorbidity was studied using Rome III Psychosocial Alarm Questionnaire.
Results
At 1 and 3 months, 16 (5.7%), 16 (5.7%), 11 (3.9%), and 24 (8.6%), 6 (2.1%), 9 (3.2%) of COVID‐19 patients developed CBD, dyspeptic symptoms, and their overlap, respectively; among healthy controls, none developed dyspeptic symptoms and one developed CBD at 3 months (P < 0.05). At 6 months, 15 (5.3%), 6 (2.1%), and 5 (1.8%) of the 280 COVID‐19 patients developed IBS, UD, and IBS‐UD overlap, respectively, and one healthy control developed IBS at 6 months (P < 0.05 for all except IBS‐UD overlap). The risk factors for post‐COVID‐19 FGIDs at 6 months included symptoms (particularly GI), anosmia, ageusia, and presence of CBD, dyspeptic symptoms, or their overlap at 1 and 3 months and the psychological comorbidity.
Conclusions
This is the first study showing COVID‐19 led to post‐COVID‐19 FGIDs. Post‐COVID‐19 FGIDs may pose a significant economic, social, and healthcare burden to the world.
Keywords: Beta coronavirus, COVID‐19, Functional dyspepsia, Gastrointestinal symptoms, Gut–brain axis disorders, Irritable bowel syndrome, Post‐infection IBS
Introduction
Infection with severe acute respiratory syndrome coronavirus‐2 (SARS‐CoV‐2), a ribonucleic acid (RNA) virus, originating from Wuhan, China, in December 2019, led to a devastating pandemic of the century, termed coronavirus disease‐19 (COVID‐19). 1 , 2 About 80–85% of patients infected with SARS‐CoV‐2 may remain asymptomatic, and others may develop respiratory and gastrointestinal (GI) illnesses. 1 , 3 Though chronic respiratory consequences of SARS‐CoV‐2 infection are being recognized increasingly, long‐term GI illness following COVID‐19, although quite likely, has not yet been reported. 4 , 5 Functional GI disorders (FGIDs) are renamed currently as disorders of the gut–brain interaction. 6 Post‐infection FGIDs (PI‐FGIDs), including PI‐irritable bowel syndrome (IBS) and PI‐functional dyspepsia (FD), are known to develop following bacterial, protozoal, and viral infection of the GI tract. 7 , 8 Factors associated with a higher frequency of development of PI‐FGIDs include protozoal and bacterial (particularly invasive) pathogens causing acute GI infection, severe and prolonged illness, antibiotic use, female gender, and younger age. 7 , 8 Moreover, several studies showed that the presence of psychological factors such as anxiety and depression are the risk factors for the development of PI‐FGIDs 7 , 8 , 9 ; such psychological issues are common among people during the current COVID‐19 pandemic. 10 Therefore, it is worthwhile to study the factors, including the psychosocial factors associated with the development of PI‐FGIDs, if any, among COVID‐19 patients.
As the angiotensin‐converting enzyme‐2 (ACE‐2) receptor, the SARS‐CoV‐2 virus entry site, is present in the epithelium of the digestive tract, COVID‐19 also affects the GI tract. 1 We previously hypothesized that COVID‐19 is expected to be followed by the development of PI‐FGIDs. 5 The presence of digestive symptoms in about one‐fifth of the patients, 1 , 3 , 11 , 12 detection of viral RNA in the feces of the half of patients, 11 increased fecal calprotectin, 13 , 14 altered gut microbiota, 15 abnormal intestinal permeability, 5 , 16 and increased serotonin 17 may also suggest that the SARS‐CoV‐2 virus not only infects the GI tract but may also lead to long‐term GI consequences such as FGIDs. 5 However, there has been no study yet to evaluate the frequency and spectrum of FGIDs, if any, following COVID‐19. Accordingly, we undertook a prospective case–control study with the following aims, (i) to evaluate the frequency and the spectrum of PI‐FGID following COVID‐19 and historical healthy controls, (ii) comparison of the frequency of post‐COVID‐19‐FGIDs among symptomatic and asymptomatic patients with COVID‐19, and (iii) factors associated with the development of post‐COVID‐19 FGIDs. Because IBS and FD are the most prevalent forms of FGIDs and most previous studies on PI‐FGID reported on these two subtypes of FGIDs, 7 , 8 , 9 we aimed to evaluate these two disorders. Whereas IBS is characterized by recurrent hypogastric pain during last 3 months with onset at least 6 months ago associated altered stool form and passage, FD denotes upper abdominal pain (epigastric pain syndrome [EPS]) or post‐prandial fullness (post‐prandial distress syndrome [PDS]) of similar duration. 6 , 9
Method
Study design
This study included two cohorts of patients with COVID‐19, one recruited consecutively from the admitted patients in three teaching hospitals (Sheikh Russel National Gastroliver Institute and Hospital, Dhaka Medical College Hospital, and Mugda Medical College Hospital) during 4 months (April to August 2020) in Dhaka, Bangladesh, and the other diagnosed in a referral laboratory in northern India from out‐patient samples obtained from various districts of the State of Uttar Pradesh and the Institute recruited during three weeks period (April to May 2020). These data were compared with the 6‐month follow‐up data for IBS and UD development on a cohort of healthy subjects published earlier from whom 264 were included carefully matching for age and gender. 9 Hindi and Bengali translated validated Enhanced Asian Rome III Questionnaires were used to collect data from the Indian and the Bangladeshi study cohorts, respectively, at inclusion and during follow‐up at 1, 3, and 6 months. 18 , 19 Data on alarm symptoms were also collected. 18 , 19 The questionnaire used to collect the data from SARS‐CoV‐2 infected patients also included data related to COVID‐19, including its severity and comorbidity. Besides, Rome III Psychosocial Alarm Questionnaire was also used. 20 Patients with prior history of FGIDs by Rome III criteria (assessed by self‐reporting by the patients using the translated‐validated enhanced Asian Rome III questionnaire), 18 , 19 history of abdominal surgery, major psychiatric illness, inflammatory bowel disease, and GI cancer were excluded. Patients with critical COVID‐19 were excluded. Patients were followed up either physically or over the telephone at 1, 3, and 6 months using translated and validated Hindi (for Indian patients) and Bengali (Bangladeshi patients) enhanced Asian Rome III questionnaire. 18 , 19 The data obtained were entered electronically in Survey Monkey (SurveyMonkey Enterprise, San Mateo, CA, USA). The study protocol was approved by the respective Institutional Ethics Committees in Bangladesh (SRGl&H/Admin/2020‐2021/2279) and India (2020‐117‐EMP‐EXP‐17). Written or electronic informed consent was obtained from each study participant.
Diagnosis of COVID‐19, assessment of its severity and GI symptoms
The diagnosis of COVID‐19 was based on the presence of SARS‐CoV‐2 RNA in naso‐pharyngeal and oro‐pharyngeal samples using reverse transcriptase‐polymerase chain reaction (RT‐PCR) by standard technique. 21 The severity of the COVID‐19 was assessed as described previously 3 , 22 : (i) critical (required ventilator), (ii) severe (needed oxygen), (iii) moderate (though pneumonia present, did not require oxygen), and (iv) mild (only upper respiratory symptoms). 21 Those without symptoms were classified as asymptomatic. GI symptoms were assessed at the time of diagnosis of COVID‐19 as categorical variables (yes or no); symptom such as diarrhea was further verified by the stool forms and frequency as reported by the patients.
Definitions
Diagnoses of FGIDs were made using Rome III criteria as Rome IV criteria are less sensitive to diagnose IBS. 23 However, the frequency of IBS development by the Rome IV criteria was compared with that by the Rome III criteria. 23 Patients fulfilling the symptom criteria both for FD and IBS were considered as having uninvestigated dyspepsia (UD)‐IBS overlap disorders. If the symptoms of IBS or dyspepsia were present during the 3‐month follow‐up, but the subjects did not fulfill the 6‐month duration criterion as proposed in various iterations of Rome criteria, the terms chronic bowel dysfunction (CBD) and dyspeptic symptoms, respectively, were used as suggested by us earlier and accepted by the Rome Foundation Working Team on Post‐infection IBS. 9 , 24 For Kaplan–Meier analysis, the time to develop CBD, dyspeptic symptoms, IBS, and UD were calculated from the earliest time to fulfill the symptom criteria for these disorders. However, for calculation time of onset of the overlap disorder, the time point of fulfilling the criteria for the second of the overlap disorders was considered as the time of its commencement. IBS and UD subtypes were defined by the diagnostic Rome III criteria. 6 , 23
Statistical analysis
Sample size calculation
Based on the data from the earlier studies that showed an average frequency of occurrence of 21% of PI‐IBS following acute gastroenteritis and 12 studies that showed the frequency to be 8% among controls, 8 the sample size was calculated for 90% power, 99% confidence interval (two‐sided), the ratio of cases to control of one. 216 patients with COVID‐19 and 216 healthy controls were considered necessary for the study. Considering the possibility of 10–20% loss to follow‐up among cases, 280 COVID‐19 patients and 264 controls were included.
Data collection and analysis
The Indian data were entered into Survey Monkey by one, and Bangladeshi data by another investigator; the data were cross‐checked randomly by three investigators. The Shapiro–Wilk test was used to check the normal distribution of the data. The categorical data were presented as proportion. The continuous data were presented as mean ± standard deviation (SD) or median and range or inter‐quartile range (IQR). Categorical variables were analyzed by the χ 2 test with Yates correction as applicable. Parametric and non‐parametric continuous data were analyzed using unpaired t and Mann–Whitney U tests, respectively. Longitudinal data were analyzed by Kaplan–Meier survival analysis and log–rank test. Subgroup analysis was also performed to compare PI‐IBS and PI‐UD development frequency during the follow‐up among the symptomatic and asymptomatic patients with COVID‐19 and those with and without GI symptoms. Multivariable analysis was performed using a step‐wise logistic regression method. A P value of less than 0.05 was considered significant. Because anorexia is a common and non‐specific symptom during any viral illness, those who had anorexia as the only GI symptom were grouped separately for the purpose of the analysis. Statistical analysis was performed using R, Epicalc, and R‐studio software (R development core team, Vienna, Austria), MedCalc (Warandeberg 3, 1000 Brussels, Belgium), and Epi Info (Centers for Disease Control and Prevention [CDC], Atlanta, Georgia, USA).
Results
Of 293 (109 from India and 184 from Bangladesh) patients with COVID‐19, 13 from Bangladesh were excluded from the final analysis due to inadequate records or loss to follow‐up. The demographic and clinical parameters of the patients from the two countries are presented in Supporting information Table S1. Of the 280 patients finally included in the analysis, 164 (58.6%) were symptomatic and 116 (41.4%) asymptomatic. None of these patients had previous FD or IBS by the Rome criteria based on patient reporting using translated‐validated questionnaire, 18 , 19 history of abdominal surgery, major psychiatric illness, inflammatory bowel disease, and GI cancer in the past. Five patients with critical COVID‐19 based on a classification system reported earlier 3 , 22 were initially excluded, all of whom died. The COVID‐19 patients were comparable with the healthy controls in age (39.5 ± 15.4 years vs 36.8 ± 11.6 years, respectively; P = NS) and gender (204/280 [72.9%] vs 193/264 [73%] male; P = NS).
Baseline demographic and clinical parameters of the patients
At the time of inclusion, of the 164 symptomatic patients, 119 (42.5%), 33 (11.8%), and 12 (4.3%), patients with COVID‐19 had a mild, moderate, and severe illness, respectively. One hundred sixteen (41.4%) had asymptomatic COVID‐19.
Symptomatic as compared to asymptomatic patients were older in age (median 42.5 years [range 31–55] vs 29.5 years [23–45]; P < 0.001) and were less often male (105/164 [64%] vs 99/116 [85.3%]; P = <0.001), and more often had comorbidities (90/164 [54.9%] vs 8/116 [6.9%]; P < 0.001).
Baseline GI symptoms
The GI symptoms included nausea (n = 53. 18.9%), vomiting (n = 29, 10.4%), diarrhea (n = 58, 20.7%), and abdominal pain (n = 31, 11.1%). Although anorexia was a common GI symptom, this was often associated with the other symptoms (n = 71, 25%) except in 48 (17%) patients in whom anorexia was the only GI symptom. The other 17 (6%) patients with GI symptoms did not experience anorexia. Ageusia and anosmia were present in 99 (35.4%) and 87 (31.1%) patients, respectively.
PI‐FGID among COVID‐19 patients and healthy controls
All the patients were followed‐up telephonically due to travel restriction due to lock‐down except the staff members of the hospitals contacting COVID‐19 and being included in this study who were followed‐up physically. Of the 280 COVID‐19 patients, at 1‐month follow‐up, 16 (5.7%), 16 (5.7%), and 11 (3.9%) developed CBD, dyspeptic symptoms, and their overlap, respectively (Fig. 1); in contrast, none of the 264 healthy controls developed CBD or dyspeptic symptoms at the 1‐month follow‐up (P < 0.05). At 3 months, 24 (8.6%), 6 (2.1%), and 9 (3.2%) of the 280 COVID‐19 patients developed CBD, dyspeptic symptoms, and their overlap, respectively, in contrast to one CBD and no dyspeptic symptom or their overlap among healthy controls (P < 0.05) (Fig. 1). At 6‐month follow‐up, 15 (5.3%), 6 (2.1%), and 5 (1.8%) of the 280 COVID‐19 patients developed IBS (Rome III criteria), UD, and IBS‐UD overlap, respectively. Of these 15 IBS patients diagnosed using Rome III criteria, 14 fulfilled Rome IV criteria. None of them had specific alarm symptoms. In contrast, one of the 264 controls developed IBS using Rome III criteria, and none developed UD (P < 0.05 for all except IBS‐UD overlap). Kaplan–Meier analysis showed that the cumulative probability of FGIDs (CBD, dyspeptic symptoms, IBS, UD) was higher among COVID‐19 patients than healthy controls during 6‐month follow‐up (Fig. 2a–f). Of the 16 patients with CBD at 1 month, in one the symptoms resolved, and in nine CBD developed de novo at 3 months. Of the 16 patients with dyspeptic symptoms only at 1 month, symptoms disappeared in 14, and it appeared afresh in 4 at 3 months. Of the 11 patients with CBD‐dyspeptic symptoms overlap at 1 month, in three symptoms resolved, and in one these developed de novo at 3 months. There was no patient in whom symptoms appeared de novo during the follow‐up period from 3 to 6 months, although the symptoms did resolve in some (Fig. 1).
Figure 1.
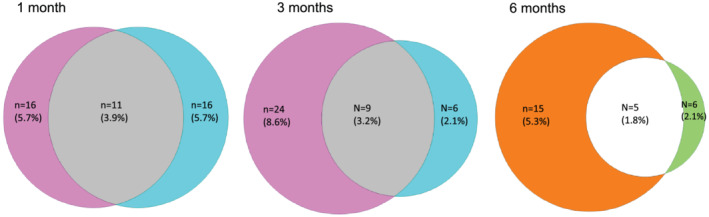
Venn diagrams showing chronic bowel dysfunction (CBD), dyspeptic symptoms, and their overlap at 1 and 3 months and irritable bowel syndrome (IBS), uninvestigated dyspepsia (UD) and their overlap at 6 months follow‐up in patients with coronavirus disease‐19 (COVID‐19).  , CBD;
, CBD;  , Dyspeptic symptoms;
, Dyspeptic symptoms;  , Overlap;
, Overlap;  , IBS;
, IBS;  , UD;
, UD;  , IBS‐UD overlap.
, IBS‐UD overlap.
Figure 2.
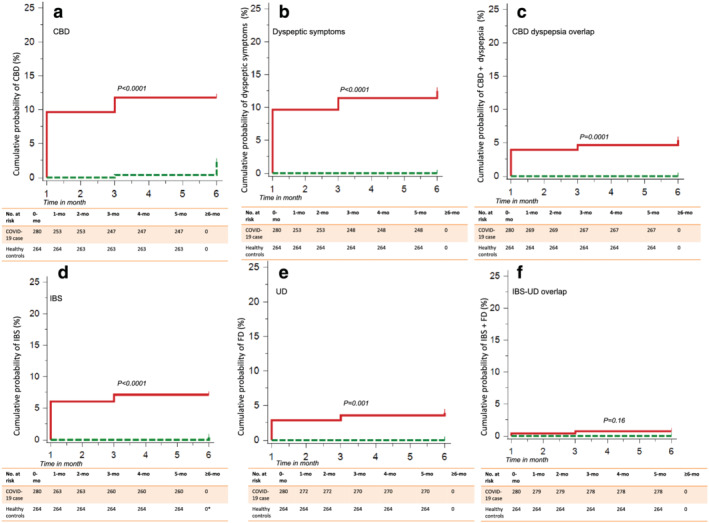
Kaplan–Meier curves showing the development of (a) chronic bowel dysfunction (CBD), (b) dyspeptic symptoms, and (c) their overlap and (d) irritable bowel syndrome (IBS), (e) uninvestigated dyspepsia (UD), and (f) their overlap during six month follow‐up among patients with coronavirus disease‐19 (COVID‐19) as compared with healthy controls.  , Case;
, Case;  , Control.
, Control.
Of the 20 patients with IBS (including 5 with UD overlap), 12 (60%) had diarrhea‐predominant, 4 (20%) had constipation‐predominant, and the other 4 (20%) had unclassified IBS. Of the 11 UD patients (including 5 with IBS overlap), nine (82%) had PDS and two (18%) had EPS.
Post‐COVID‐19‐FGID among symptomatic and asymptomatic patients
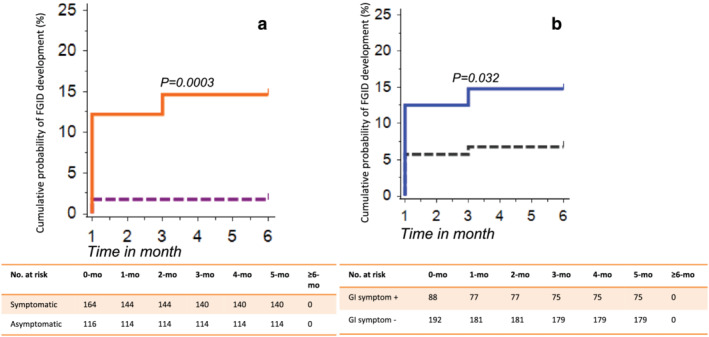
At 1‐month follow‐up, symptomatic compared with asymptomatic patients with COVID‐19, more often developed CBD (14/164 [8.5%] vs 2/116 [1.7%]; P = 0.017), dyspeptic symptoms (15/164 [9.1%] vs 1/116 [0.9%; P = 0.002), and their overlap (11/164 [6.7%] vs 0/116 [0%]; P = 0.003), respectively. However, the frequency of GI symptom development was comparable among asymptomatic COVID‐19 patients as compared with controls at 1 month (2/116 vs 0/264; P = ns) and at 3 months (22/164 [13.4%] vs 2/116 [1.7%]; P = 0.0003; 6/164 [3.7%] vs 0/116 [0%]; P = 0.04; and 9/164 [5.5%] vs 0/116 [0%]; P = 0.01, respectively) (Table 1). At 6‐month follow‐up, symptomatic patients with COVID‐19 more often developed FGIDs (IBS 13/164 [7.8%] vs 2/116 [1.7%]; P = 0.03; UD 6/164 [3.7%] vs 0/116 [0%]; P = 0.05; and IBS‐UD overlap 5/164 [3%] vs 0/116 [0%]; P = 0.07). Kaplan–Meier analysis revealed a higher cumulative probability of development of IBS, UD, and IBS‐UD overlap among symptomatic patients with COVID‐19 than those without symptoms (Fig. 3a).
Table 1.
Relationship between presence of GI symptoms and development of post‐COVID‐19 GI consequences (CBD, dyspeptic symptoms, IBS, UD and their overlap)
| Parameters | GI symptoms with or without anorexia (n = 88) | Anorexia alone † (n = 48) | GI symptoms absent (n = 144) | P values |
|---|---|---|---|---|
| Presence of CBD, dyspeptic symptoms, and their overlap at 1 month | ||||
| QCBD only | 8 (9.1%) | 6 (12.5%) | 2 (1.4%) | |
| Dyspeptic symptoms only | 10 (11.4%) | 4 (8.3%) | 2 (1.4%) | <0.001 |
| Overlap | 9 (10.2%) | 1 (2.1%) | 1(0.7%) | |
| None | 61 (69.3%) | 37 (77.1%) | 139 (96.5%) | |
| Presence of CBD, dyspeptic symptoms, and their overlap at 3 months | ||||
| CBD only | 14 (15.9%) | 8 (16.6%) | 2 (1.4%) | |
| Dyspeptic symptoms only | 2 (2.3%) | 1 (2.1%) | 3 (2.1%) | <0.001 |
| Overlap | 7 (8%) | 1 (2.1%) | 1 (0.7%) | |
| None | 65 (73.9%) | 38 (79.2%) | 138 (95.8%) | |
| Presence of IBS, UD, and their overlap at 6 months | ||||
| IBS | 6 (6.8%) | 7 (14.6%) | 2 (1.4%) | |
| UD | 2 (2.3%) | 2 (4.2%) | 2 (1.4%) | 0.0002 |
| IBS‐UD overlap | 5 (5.7%) | 0 (0%) | 0 (0%) | |
| None | 75 (85.2%) | 39 (81.2%) | 140 (97.2%) | |
COVID‐19, coronavirus disease‐19; CBD, chronic bowel dysfunction; FGID, functional gastrointestinal disorders; GI, gastrointestinal; IBS, irritable bowel syndrome; UD, uninvestigated dyspepsia.
Because anorexia is a non‐specific symptoms for viral illness, patients with anorexia alone were grouped separately.
Figure 3.
Kaplan–Meier curves showing the development of functional gastrointestinal disorders (e.g. irritable bowel syndrome [IBS], uninvestigated dyspepsia [UD], and their overlap) at 6 month follow‐up (a) among symptomatic as compared to the asymptomatic patients with coronavirus disease‐19 (COVID‐19), and (b) among those with and without gastrointestinal (GI) symptoms. (a)  , Asymptomatic;
, Asymptomatic;  , Symptomatic. (b)
, Symptomatic. (b)  , No GI symptoms;
, No GI symptoms;  , GI symptoms +.
, GI symptoms +.
Relationship between GI symptoms during the COVID‐19 and post‐COVID‐19 FGID
As shown in Table 2 and Figure 3b, the presence of GI symptoms during the baseline COVID‐19 was more often associated with the development of FGIDs (either IBS, or UD, or their overlap) during 6‐month follow‐up. As shown in Table 2, 48 (17%) patients with anorexia alone were grouped separately for the analysis.
Table 2.
Factors associated with post‐COVID‐19 FGIDs (including irritable bowel syndrome, uninvestigated dyspepsia, and their overlap)
| Parameters | FGID present (n = 26) | No FGID (n = 254) | P values |
|---|---|---|---|
| Age (year, median, IQR) | 38 (31.8–45) | 35 (26–52) | 0.69 |
| Gender (male) | 19 (73.1%) | 185 (72.8%) | 1 |
| Diet (vegetarian) | 0 (0%) | 22 (8.7%) | 0.24 |
| Presence of comorbidity | 12 (46.2%) | 86 (33.9%) | 0.3 |
| Addiction (alcohol, tobacco) | 5 (20%) | 33 (15.1%) | 0.55 |
| Severity of COVID‐19 | |||
| Mild | 16 (61.5%) | 103 (40.6%) | |
| Moderate | 8 (30.8%) | 25 (9.8%) | <0.001 |
| Severe | 0 (0%) | 12 (4.7%) | |
| Asymptomatic | 2 (7.7%) | 114 (44.9%) | |
| GI symptoms † | |||
| Absent | 4 (15.4%) | 140 (55.1%) | <0.001 |
| Anorexia with other GI symptoms | 13 (50%) | 58 (22.8%) | |
| Anorexia only | 9 (34.6%) | 39 (15.4%) | |
| Other GI symptoms only | 0 (0%) | 17 (6.7%) | |
| Presence of anosmia | 15 (57.7%) | 72 (28.6%) | 0.005 |
| Presence of ageusia | 15 (57.7%) | 84 (33.3%) | 0.024 |
| Presence of CBD, dyspepsia at 1 month | |||
| No | 4 (15.4%) | 233 (91.7%) | |
| CBD | 14 (53.8%) | 2 (0.8%) | <0.001 |
| Dyspeptic symptoms | 5 (19.2%) | 11 (4.3%) | |
| Overlap | 3 (11.5%) | 8 (3.1%) | |
| Presence of CBD, dyspepsia at 3 months | |||
| No | 4 (15.4%) | 237 (93.3%) | |
| CBD | 19 (73.1%) | 5 (2%) | <0.001 |
| Dyspeptic symptoms | 1 (3.8%) | 5 (2%) | |
| Overlap | 2 (7.7%) | 7 (2.8%) | |
CBD, chronic bowel dysfunction; COVID‐19, coronavirus disease‐19; FGID, functional gastrointestinal disorders; GI, gastrointestinal.
Because anorexia is a common and non‐specific symptom during any viral illness, those who had anorexia as the only GI symptom (n = 48, 17%) were separately grouped.
Other risk factors of post‐COVID‐19 FGID
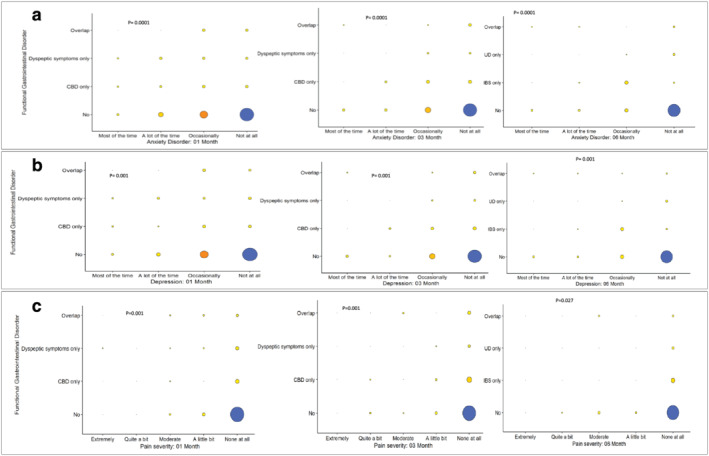
The other risk factors for the development of post‐COVID‐19 FGIDs such as IBS, UD, and IBS‐UD overlap at 6‐month follow‐up on univariate analysis were the presence of symptoms, particularly GI symptoms, anosmia, ageusia, and presence of CBD, dyspeptic symptoms or their overlap at 1 and 3 months and the psychological comorbidity (Tables 2, and 3 and Fig. 4). On multivariate analysis, the presence of GI symptoms, and CBD, dyspeptic symptoms or their overlap at 1and 3 months were the independent predictors of the development of post‐COVID‐19 FGIDs (Table 4).
Table 3.
Relationship between the psychological factors (according to Rome III Psychosocial Alarm Questionnaire) at one‐month follow‐up and the development of post‐COVID‐19 FGIDs (DGIBs) at 6 months
| Parameters | FGID present (n = 26) | No FGID (n = 254) † | P values |
|---|---|---|---|
| Anxiety question: In the last week, have you felt tense or wound up? | |||
| Most of the time | 3 (11.5%) | 5 (2.0%) | |
| A lot of time | 3 (11.5%) | 13 (5.3%) | 0.009 |
| Occasionally | 9 (34.6%) | 59 (24.3%) | |
| Not at all | 11 (42.3%) | 166 (68.3%) | |
| Depression question: In the last week, have you felt downhearted or low? | |||
| Most of the time | 3 (11.5%) | 5 (2.0%) | |
| A good bit of time | 3 (11.5%) | 13 (5.3%) | 0.007 |
| Some of the time | 9 (34.6%) | 59 (24.3%) | |
| Not at all | 11 (42.3%) | 166 (68.3%) | |
| Suicidal ideation question: Have you felt recently so low that you felt like hearting or killing yourself? | |||
| Often | 0 (0%) | 0 (0%) | |
| Occasionally | 1 (3.8%) | 1 (0.4%) | 0.4 |
| Not at all | 25 (96.4%) | 238 (99.6%) | |
| Pain severity question: During the last 4‐week, how much bodily pain have you had? | |||
| Extremely | 0 (0%) | 0 (0%) | |
| Quite a bit | 1 (3.8%) | 7 (2.8%) | |
| Moderately | 5 (19.2%) | 14 (5.8%) | 0.04 |
| A little bit | 3 (11.5%) | 24 (9.9%) | |
| Not at all | 17 (65.4%) | 198 (81.5%) | |
| Impairment question: During the last 4‐week, how much did pain/other symptoms interfere with your normal activities? | |||
| Extremely | 0 (0%) | 1 (0.4%) | |
| Moderate | 3 (11.5%) | 4 (1.6%) | 0.03 |
| A little bit | 2 (7.7%) | 13 (5.3%) | |
| Not at all | 21 (80.8%) | 225 (92.6%) | |
| Coping impairment question: When I have pain/other symptoms, these appear to be terrible and never get better | |||
| Always | 1 (3.8%) | 1 (0.4%) | |
| Sometimes | 7 (26.9%) | 16 (6.6%) | <0.0001 |
| Never | 18 (69.2%) | 226 (93%) | |
| Abuse question: Have you been physically, emotionally, sexually victimized any time? | |||
| Yes | 0 (0%) | 1 (0.4%) | 1 |
| Never | 26 (100%) | 242 (99.6%) | |
COVID‐19, coronavirus disease‐19; FGID, functional gastrointestinal disorders.
These parameters were available for 243 of 254 patients without FGID. For suicidal idea question, data were available for 239 patients.
For the purpose of the comparison, occasionally, sometimes, a little bit were grouped with not all.
Figure 4.
Balloon plots showing that some of the psychological factors (as per Rome III Psychosocial Alarm Questionnaire) initially and during follow‐up were associated with the development functional gastrointestinal disorders (FGIDs) among the patients with coronavirus disease‐19 (COVID‐19). CBD, chronic bowel dysfunction; IBS, irritable bowel syndrome; UD, uninvestigated dyspepsia. Figures in row (a) present data on anxiety questions, row (b) depression questions, and row (c) body pain questions at 1, 3, and 6 months, respectively. For P values, refer to Table 3. (a)  , 150;
, 150;  , 100;
, 100;  , 50;
, 50;  , 0;
, 0;  . (b)
. (b)  , 150;
, 150;  , 100;
, 100;  , 50;
, 50;  , 0;
, 0;  . (c)
. (c)  , 150;
, 150;  , 100;
, 100;  , 50;
, 50;  , 0;
, 0;  .
.
Table 4.
Multivariate analysis of relationship between presence of GI symptoms † and various parameters associated with FGIDs
| Parameters | Crude odds ratio (95% CI) | Crude P values | Adjusted odds ratio (95% CI) | Adjusted P values (LR test) |
|---|---|---|---|---|
| Presence of GI symptoms (reference: no GI symptom) | ||||
| GI symptoms with or without anorexia | 6.0 (1.9–19.1) | 0.002 | 0.2 (0.03–1.8) | 0.01 |
| Anorexia only | 8.4 (2.5–28.9) | <0.001 | 2.8 (0.4–21.1) | 0.6 ‡ |
| Anosmia (yes vs no) | 3.5 (1.5–7.9) | 0.003 | 2.4 (0.6–10.5) | 0.09 |
| Ageusia (yes vs no) | 2.7 (1.2–6.2) | 0.01 | 2.1 (0.4–10.2) | 0.09 |
| Severity of COVID‐19 (reference, asymptomatic and mild) § | 19 (3.79,95.13) | <0.001 | 12.18 (0.29,506.72) | 0.02 |
| Either CBD, dyspeptic symptoms or both at 1‐mo (yes vs no) | 67.2 (20.9–214.9) | <0.001 | 18.4 (3.8–88.7) | <0.001 |
| Either CBD, dyspeptic symptoms or both at 3‐mo (yes vs no) | 86.5 (26.4–283.3) | <0.001 | 36.6 (6.5–206.2) | <0.001 |
CBD, chronic bowel dysfunction; CI, confidence interval; COVID‐19, coronavirus disease‐19; FGID, functional gastrointestinal disorders; GI, gastrointestinal; LR, likelihood ratio.
Because anorexia is a common and non‐specific symptom during any viral illness, those who had anorexia as the only GI symptom (n = 48, 17%) were separately grouped.
Wald's test.
For the purpose of comparison, asymptomatic and mild were grouped together; moderate and severe were grouped together.
There was no relationship between treatment received for COVID‐19 during the index illness and development of UD after 6 months. Of the 11 patients having symptoms of UD (including five with IBS overlap), nine received oral azithromycin, two received other intravenous antibiotics, one received corticosteroid, and all received paracetamol for fever for 1 to 2 weeks. None has been receiving non‐steroidal anti‐inflammatory drugs, which might cause dyspeptic symptoms, during or after recovery from COVID‐19.
Discussion
The current study shows that (i) patients with COVID‐19 had a higher probability of development of CBD, dyspeptic symptoms, and their overlap during the first 3‐month follow‐up and IBS (particularly IBS‐D), UD (particularly PDS), and IBS‐UD overlap during a 6‐month follow‐up compared with the healthy controls, (ii) symptomatic patients with COVID‐19 had a greater risk for the development of the above conditions than the asymptomatic patients, (iii) presence of GI symptoms at baseline during COVID‐19 was associated with a higher frequency of development of FGIDs, and (iv) the other risk factors of post‐COVID‐19 FGIDs included the presence of anosmia, ageusia, the psychological comorbidity and presence of CBD, dyspeptic symptoms, or their overlap during the early follow‐up period.
The PI‐FGIDs have been reported following bacterial, viral, and protozoal infection of the GI tract. 7 , 8 Earlier, based on mechanistic evidence, we hypothesized that COVID‐19 might be followed by the development of post‐COVID‐19 FGIDs. 5 , 25 This is the first study showing that 5.3%, 2.1%, and 1.3% of patients with COVID‐19 developed IBS, UD, and IBS‐UD overlap during 6‐month follow‐up; these figures were higher than those among healthy controls. This is not unexpected as infection with SARS‐CoV‐2 is known to result in progressive alveolar damage and fibrosis during long‐term follow‐up in the respiratory tract, 4 and GI tract involvement with the virus is well‐known. 1 , 2 The evidence of infection and invasion of the GI tract by the virus include the presence of ACE‐2 receptors in GI mucosa, 1 , 2 the occurrence of digestive manifestations in about 18% of patients, 11 the presence of viral RNA in feces in 50% of patients, 11 rise in fecal calprotectin, 14 gut microbiota dysbiosis among them, 15 increase in serotonin in the gut mucosa, 17 altered intestinal permeability, 16 the affinity of SARS‐CoV‐2 virus to involve the central and peripheral nervous systems (and hence the enteric nervous system), 26 , 27 and findings of mucosal damage on GI endoscopy and autopsy studies. 28 This study for the first time proves the hypothesis proposed by us earlier that COVID‐19 may lead to post‐COVID‐19 FGIDs.
This study also showed that symptomatic patients more often developed IBS, UD, and IBS‐UD overlap than asymptomatic COVID‐19 patients (7.8%, 3.7%, and 3% vs 1.7%, 0%, and 0%, respectively). Diarrhea and vomiting are the two defining parameters for PI‐FGIDs following acute GI infection. 8 , 24 The GI manifestations in patients with symptomatic COVID‐19 include diarrhea and vomiting. 1 , 3 , 7 , 11 Patients with diarrhea are known to have higher fecal calprotectin and mucosal serotonin levels than those without diarrhea following SARS‐CoV‐2 infection, suggesting a possibility of gut mucosal inflammation to be a mechanism for diarrhea in these patients. 5 , 14 , 17 Because gut mucosal inflammation caused by invasive pathogens causing acute infectious gastroenteritis is a predictor of PI‐FGIDs and SARS‐CoV‐2 is known to cause chronic lung damage, chronic inflammation of the gut is not unexpected in patients with COVID‐19. 4 , 8 Compared with the frequency of occurrence of PI‐FGIDs following bacterial and even viral pathogens infecting the GI tract, the incidence of post‐COVID‐19 FGID appears to be less. 7 , 8 This is not unexpected. The PI‐FGIDs, by definition, meant the occurrence of these disorders following acute infectious gastroenteritis. 24 In an earlier study from Canada, PI‐IBS's frequency following viral gastroenteritis was 12.5% at 6‐month follow‐up. 29 Because SARS‐CoV‐2 is primarily a respiratory pathogen and infects the GI tract only in half of the patients with COVID‐19, the development of post‐COVID‐19 FGIDs is expected to be lesser than that following the viral gastroenteritis. The results of our study support this possibility. Interestingly, the frequency of post‐COVID‐19 FGID development among patients with asymptomatic COVID‐19 was comparable with healthy controls. Our study shows that only symptomatic COVID‐19 poses a risk of development of post‐COVID‐19 FGIDs.
What could be the other mechanisms of development of post‐COVID‐19 FGIDs? Mechanisms of PI‐IBS development following acute gastroenteritis may be classified into agent, host, and host–agent interaction‐related factors. 7 , 8 Specific pathogens, particularly the invasive ones such as Shigella dysenteriae , Campylobacter jejuni , and protozoa such as Giardia lamblia are more likely to be followed by PI‐IBS than viruses. 7 , 8 Host factors include the host's genetic make‐up and degree of T regulatory response. The host–agent interaction‐related factors include the balance between the inflammatory (e.g., interleukin [IL]‐8) and anti‐inflammatory (e.g., IL‐10) cytokine responses. 7 , 8 Other infective agents such as helminthes may induce a Th2‐response reducing the inflammatory response, and hence, the development of PI‐IBS. 7 , 8 Several altered intestinal microenvironment‐related factors are important in the pathogenesis of IBS in general, such as gut microbiota dysbiosis, altered mucosal serotonin, intestinal permeability, bile acid abnormality, and mucosal inflammation, some of which have been shown in patients with COVID‐19 as well. 5 , 25 , 30 Mechanistic studies on post‐COVID‐19 FGIDs are needed.
The development of post‐COVID‐19 FGIDs among patients with GI symptoms during the initial illness compared with those without is interesting. It has been shown earlier that the development of PI‐IBS had a relationship with the severity of diarrhea during the index episode of acute gastroenteritis (AGE). 7 , 8 As severe diarrhea might suggest a greater degree of injury to the GI tract, a relationship between PI‐IBS development and severity of diarrhea during the inciting episode of AGE can be easily explained. Similarly, GI symptoms during the initial attack of COVID‐19 might indicate that the virus involved the GI tract. Hence, a protracted long‐term GI inflammation leading to post‐COVID‐19 FGID is not unexpected. In fact, earlier studies on PI‐IBS following AGE did show that the severity of the initial insult, the degree of acute gut inflammation, and psychological issues originating from brain during the initial episode of AGE did persist for months to years resulting in protracted inflammation that may result from inadequate T regulatory response and hypervigilence leading to PI‐FGIDs. 8 Our results showed a comparable frequency of dyspeptic symptoms and CBD during the initial 1‐month follow‐up after COVID‐19. The upper GI symptoms tended to resolve during a longer‐term follow‐up of 6 months. This might suggest that though the initial dyspeptic symptoms might be non‐specific due to systemic viral illness and drugs, the later lower GI symptoms fulfilling CBD and IBS's criteria might indicate more specific pathology related to protracted mucosal inflammation of the GI tract and altered gut–brain interaction. The presence of GI symptoms during the initial COVID‐19 episode poses the greater risk of persistent GI symptoms fulfilling the criteria for post‐COVID‐19 FGIDs.
A relationship between the development of post‐COVID‐19 FGID and anosmia, ageusia, during the initial episode of COVID‐19, and psychological comorbidity, might indicate a connection between peripheral (enteric) and central nervous involvement systems in its pathogenesis. Guillain‐Barre and Miller‐Fisher syndromes have been described in patients with COVID‐19 indicating involvement of the peripheral nervous system. 26 An earlier autopsy study on 21 patients with fatal COVID‐19 showed marked inflammatory changes in the central nervous system, including the olfactory bulb and brainstem, particularly medulla oblongata. 27 Because smell and taste sensations are carried by the olfactory nerves directly arising from the brain and the facial and glossopharyngeal nerves from the pons and medulla oblongata, respectively, a relationship between loss of these sensations and the development of post‐COVID‐19 FGIDs can be easily explained. Because the hypothalamus controls the autonomic nervous system and the parasympathetic supply for most of the GI tract is by the vagus nerves, which arise from the medulla oblongata, a possible involvement of the autonomic nervous system due to the SARS‐CoV‐2 infection altering the gut–brain axis leading to the development of post‐COVID‐19 FGID cannot be excluded. In fact, several studies showed involvement of autonomic nervous system by the SARS‐CoV‐2 resulting in autonomic dysfunction that may persist up to 1 year after recovery from COVID‐19. 31
Our study is not without limitations. We used Rome III rather than the most recently described Rome IV criteria as the latter is 50% less sensitive to diagnose IBS. 23 Interestingly, 14/15 (93%) post‐COVID‐19 IBS patients diagnosed using Rome III criteria fulfilled the Rome IV criteria as well. Because this is the first study on post‐COVID‐19 FGIDs, we had to calculate the sample size based on previous studies on PI‐IBS following AGE. The follow‐up period was relatively short (only 6 months). We did not study the mechanisms of post‐COVID‐19 FGIDs. In fact, it is possible that detailed investigations of our patients with post‐COVID‐19 FGIDs might reveal micro‐organic abnormalities as have been shown among patients with PI‐IBS earlier. 8 , 9 , 30 , 32 However, the current Rome algorithm does not necessitate that. 24 A diagnosis of FD requires a normal esophagogastroduodenoscopy and negative tests for Helicobacter pylori as per Rome IV criteria. 33 Therefore, we used the term UD rather than FD though in the earlier studies, most patients with UD turned out to be FD rather than organic dyspepsia. 34 Accordingly, earlier studies did use the term FD even without undertaking an endoscopic examination of the upper GI tract. 7 , 23 Hence, the frequency of UD showed in our study may be considered to reflect the incidence of FD. None of our patients diagnosed as IBS had alarm features. 33 Inclusion of historical controls is a limitation of the study. However, considering a high rate of asymptomatic COVID‐19 in the community during the current pandemic, it is perhaps strength rather than limitation as they were recruited long before COVID‐19 pandemic began mitigating the possibility of asymptomatic infection. Moreover, as anxiety among people during the pandemic is increased, 10 which might lead to higher frequency of FGIDs among them, it is possible that the frequency of development of FGIDs might have been under‐estimated among our historical healthy controls. Moreover, more patients with COVID‐19 than healthy controls had comorbidity, which might explain lesser frequency of development of GI symptoms indicative of FGIDs among the controls. We designed to assess the psychological issues at one month rather than at baseline. Due to exaggerated media campaign, public awareness programs, lock‐down and its implementation, quarantine, fear about COVID‐19 was too high in the population. Hence, we believed that psychological assessment of a subject immediately after being diagnosed having COVID‐19 would over‐estimate the degree of psychological abnormalities. Our data showing persistent psychological issues among patients with post‐COVID‐19 FGID compared to those who did not have persistent GI symptoms perhaps support our hypothesis. Whether the results of the current study are applicable to the other population remain to be seen. For example, 73% of our patients who developed FGIDs were male (Table 2) in contrast to the studies from other population in which female patients outnumber male patients. 23 , 32 However, a few recent studies from India also showed female preponderance among patients with FGIDs. 32 , 35 Anorexia is not a symptom specific to the GI tract but may occur due to any systemic illness and drugs. However, because most of the earlier studies on COVID‐19 also listed anorexia as a GI symptom, 1 , 3 , 11 we followed the same paradigm. Considering the fact that anorexia is not specific to GI tract, we analyzed the data of the patients who had anorexia alone separately. Because critical COVID‐19 patients are less likely to survive, we did not include them according to our study design. Hence, our study cannot provide the estimates of frequency of post‐COVID‐19 FGID development among patients with critical COVID‐19 if they survive. Moreover, none of the 12 patients with severe COVID‐19 developed FGIDs. However, the sample size of severe COVID‐19 was too small to draw any meaningful conclusion on development of post‐COVID‐19 FGIDs in patients with severe COVID‐19. Some studies reported that moderate to severe patients with COVID‐19 tend to have more GI symptoms. 1 , 2 , 3 , 11 , 12 According to the results of this study, more of them may go on to develop post‐COVID‐19‐FGID. But it could also be argued that the global distribution of COVID‐19 cases were as such with more asymptomatic and mild disease than the severe manifestations. 1 , 2 , 3 Although the two groups of patients included from Bangladesh and India were not comparable as the former group of patients was mostly hospitalized and the latter group was mostly from the community sample, they were pooled together to provide a mix of asymptomatic, mild with the more severe disease.
Despite the above‐mentioned limitations, our study is important as it's the first study showing about 9% of patients with COVID‐19 may develop FGIDs during the 6‐month follow‐up. This may pose a significant economic, social, and healthcare burden to the world, considering the massive load of COVID‐19 during this pandemic. Until June 16, 2021, 177 511 047 people have contracted this highly infectious virus, and 3 840 361 died globally. Moreover, considering the inevitable surge of post‐COVID‐19 FGIDs, clinicians must be aware of this possibility in their clinics.
In conclusion, symptomatic COVID‐19, particularly those with GI symptoms, may develop FGIDs during a 6‐month follow‐up. The risk factors of post‐COVID‐19 FGIDs included GI symptoms, presence of anosmia, ageusia, psychological comorbidity, and presence of CBD, dyspeptic symptoms, or their overlap during the initial follow‐up period.
Supporting information
Table S1. Demographic and clinical parameters of the patients with COVID‐19 included from India and Bangladesh.
Acknowledgments
U. C. G. and U. G. thank the Department of Biotechnology, Government of India, for the funding for the Indian part of the study (project No. BT/PR40311/COD/139/9/2020).
Ghoshal, U. C. , Ghoshal, U. , Rahman, M. M. , Mathur, A. , Rai, S. , Akhter, M. , Mostafa, T. , Islam, M. S. , Haque, S. A. , Pandey, A. , Kibria, M. G. , and Ahmed, F. (2022) Post‐infection functional gastrointestinal disorders following coronavirus disease‐19: A case–control study. Journal of Gastroenterology and Hepatology, 37: 489–498. 10.1111/jgh.15717.
Declaration of conflict of interest: None of the authors has any conflict of interest to declare in relation to this paper.
Author contribution: U. C. G. conceptualized and designed the study, supervised the study conduct, analyzed the data, and wrote the first draft of the paper. U. G. designed the study, performed laboratory works, supervised the study, and contributed to manuscript writing. M. M. R. contributed to the study design, supervised the study conduct and data collection, and approved the final version of the paper. A. M., S. R., M. A., T. M, M. S. I., S. A. H., M. G. K., and F. A. contributed to the data collection and study conduct. A. P. performed laboratory work. All the authors approved the final version of the paper.
References
- 1. Ghoshal UC, Ghoshal U, Dhiman RK. Gastrointestinal and hepatic involvement in severe acute respiratory syndrome coronavirus 2 infection: a review. J. Clin. Exp. Hepatol. 2020; 10: 622–628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Dhar J, Samanta J, Kochhar R. Corona virus disease‐19 pandemic: the gastroenterologists' perspective. Indian J. Gastroenterol. 2020; 39: 220–231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Ghoshal UC, Ghoshal U, Mathur A et al. The spectrum of gastrointestinal symptoms in patients with coronavirus disease‐19: predictors, relationship with disease severity, and outcome. Clin. Transl. Gastroenterol. 2020; 11: e00259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Li Y, Wu J, Wang S et al. Progression to fibrosing diffuse alveolar damage in a series of 30 minimally invasive autopsies with COVID‐19 pneumonia in Wuhan. China. Histopathology 2020; 78: 542–555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Schmulson M, Ghoshal UC, Barbara G. Managing the inevitable surge of post‐COVID‐19 functional gastrointestinal disorders. Am. J. Gastroenterol. 2021; 116: 4–7. [DOI] [PubMed] [Google Scholar]
- 6. Drossman DA, Hasler WL. Rome IV‐functional GI disorders: disorders of gut–brain interaction. Gastroenterology 2016; 150: 1257–1261. [DOI] [PubMed] [Google Scholar]
- 7. Klem F, Wadhwa A, Prokop LJ et al. Prevalence, risk factors, and outcomes of irritable bowel syndrome after infectious enteritis: a systematic review and meta‐analysis. Gastroenterology 2017; 152: 1042–1054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Ghoshal UC, Rahman MM. Post‐infection irritable bowel syndrome in the tropical and subtropical regions: Vibrio cholerae is a new cause of this well‐known condition. Indian J. Gastroenterol. 2019; 38: 87–94. [DOI] [PubMed] [Google Scholar]
- 9. Rahman MM, Ghoshal UC, Sultana S et al. Long‐term gastrointestinal consequences are frequent following sporadic acute infectious diarrhea in a tropical country: a prospective cohort study. Am. J. Gastroenterol. 2018; 113: 1363–1375. [DOI] [PubMed] [Google Scholar]
- 10. Rudenstine S, McNeal K, Schulder T et al. Depression and anxiety during the COVID‐19 pandemic in an urban, low‐income public university sample. J. Trauma. Stress 2021; 34: 12–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Cheung KS, Hung IFN, Chan PPY et al. Gastrointestinal manifestations of SARS‐CoV‐2 infection and virus load in fecal samples from a Hong Kong cohort: systematic review and meta‐analysis. Gastroenterology 2020; 159: 81–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Kumar A, Arora A, Sharma P et al. gastrointestinal and hepatic manifestations of corona virus disease‐19 and their relationship to severe clinical course: a systematic review and meta‐analysis. Indian J. Gastroenterol. 2020; 39: 268–284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Britton GJ, Chen‐Liaw A, Cossarini F et al. SARS‐CoV‐2‐specific IgA and limited inflammatory cytokines are present in the stool of select patients with acute COVID‐19. Sci. Rep. 2021; 11: 13308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Effenberger M, Grabherr F, Mayr L et al. Faecal calprotectin indicates intestinal inflammation in COVID‐19. Gut 2020; 69: 1543–1544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Zuo T, Zhang F, Lui GCY et al. Alterations in gut microbiota of patients with COVID‐19 during time of hospitalization. Gastroenterology 2020; 159: 944–955 e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Cardinale V, Capurso G, Ianiro G et al. Intestinal permeability changes with bacterial translocation as key events modulating systemic host immune response to SARS‐CoV‐2: a working hypothesis. Dig. Liver Dis. 2020; 52: 1383–1389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Ha S, Jin B, Clemmensen B et al. Serotonin is elevated in COVID‐19‐associated diarrhoea. Gut 2021; 70: 2015–2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Ghoshal UC, Gwee KA, Chen M et al. Development, translation and validation of enhanced Asian Rome III questionnaires for diagnosis of functional bowel diseases in major Asian languages: a Rome Foundation‐Asian Neurogastroenterology and Motility Association Working Team Report. J Neurogastroenterol Motil 2015; 21: 83–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Rahman MM, Ghoshal UC, Rowshon AH et al. Translation and validation of enhanced asian Rome III questionnaires in Bengali language for diagnosis of functional gastrointestinal disorders. J Neurogastroenterol Motil 2016; 22: 240–247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. American College of Gastroenterology . Rome III psychosocial alarm questionnaire. The American Journal of Gastroenterology 2010; 105: 796–797. [Google Scholar]
- 21. Ghoshal U, Vasanth S, Tejan N. A guide to laboratory diagnosis of corona virus disease‐19 for the gastroenterologists. Indian J. Gastroenterol. 2020; 39: 236–242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Wallis LA. COVID‐19 Severity Scoring Tool for low resourced settings. Afr J Emerg Med 2020. 10.1016/j.afjem.2020.03.002 Online ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Sperber AD, Bangdiwala SI, Drossman DA et al. Worldwide prevalence and burden of functional gastrointestinal disorders, results of Rome Foundation global study. Gastroenterology 2021; 160: 99–114. [DOI] [PubMed] [Google Scholar]
- 24. Barbara G, Grover M, Bercik P et al. Rome Foundation working team report on post‐infection irritable bowel syndrome. Gastroenterology 2019; 156: 46–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Ghoshal UC. Post‐infection irritable bowel syndrome. Gut Liver 2021. (in press). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Reza‐Zaldivar EE, Hernandez‐Sapiens MA, Minjarez B et al. Infection mechanism of SARS‐COV‐2 and its implication on the nervous system. Front. Immunol. 2020; 11: 621735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Schurink B, Roos E, Radonic T et al. Viral presence and immunopathology in patients with lethal COVID‐19: a prospective autopsy cohort study. Lancet Microbe 2020; 1: e290–e299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Lin L, Jiang X, Zhang Z et al. Gastrointestinal symptoms of 95 cases with SARS‐CoV‐2 infection. Gut 2020; 69: 997–1001. [DOI] [PubMed] [Google Scholar]
- 29. Marshall JK, Thabane M, Borgaonkar MR et al. Postinfectious irritable bowel syndrome after a food‐borne outbreak of acute gastroenteritis attributed to a viral pathogen. Clin. Gastroenterol. Hepatol. 2007; 5: 457–460. [DOI] [PubMed] [Google Scholar]
- 30. Barbara G, Feinle‐Bisset C, Ghoshal UC et al. The intestinal microenvironment and functional gastrointestinal disorders. Gastroenterology 2016; 18: S0016–5085(16) 00219‐5. [DOI] [PubMed] [Google Scholar]
- 31. Becker RC. Autonomic dysfunction in SARS‐COV‐2 infection acute and long‐term implications COVID‐19 editor's page series. J. Thromb. Thrombolysis 2021; 17: 1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Ghoshal UC. Marshall and Warren Lecture 2019: a paradigm shift in pathophysiological basis of irritable bowel syndrome and its implication on treatment. J. Gastroenterol. Hepatol. 2020; 35: 712–721. [DOI] [PubMed] [Google Scholar]
- 33. Stanghellini V, Chan FKL, Hasler WL, Malagelada JR, Suzuki H, Tack J, Talley NJ. Gastroduodenal disorders. Gastroenterology 2016; 150: 1380–1392. [DOI] [PubMed] [Google Scholar]
- 34. Rahman MM, Ghoshal UC, Kibria MG et al. Functional dyspepsia, peptic ulcer, and Helicobacter pylori infection in a rural community of South Asia: an endoscopy‐assisted household survey. Clin Transl Gastroenterol 2021; 12: e00334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Goyal O, Nohria S, Dhaliwal AS, Goyal P, Soni RK, Chhina RS, Sood A. Prevalence, overlap and risk factors for Rome IV functional gastrointestinal disorders among college students in northern India. Indian J. Gastroenterol. 2021; 40: 144–153. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1. Demographic and clinical parameters of the patients with COVID‐19 included from India and Bangladesh.


