Abstract
Environmental contamination from toxic metals and pesticides is an issue of great concern due to their harmful effects to human health and the ecosystems. In this framework, we assessed the adverse effects when aquatic organisms are exposed to toxicants such as deltamethrin (DM) and lead (Pb), alone or in combination, using zebrafish as a model. Moreover, we likewise evaluated the possible protective effect of vitamin C (VC) supplementation against the combined acute toxic effects of the two toxicants. Juvenile zebrafish were exposed to DM (2 μg L−1) and Pb (60 μg L−1) alone and in combination with VC (100 μg L−1) and responses were assessed by quantifying acetylcholinesterase (AChE) activity, lipid peroxidation (MDA), some antioxidant enzyme activities (SOD and GPx), three-dimension locomotion responses and changes of elements concentrations in the zebrafish body. Our results show that VC has mitigative effects against behavioral and biochemical alterations induced by a mixture of contaminants, demonstrating that it can be used as an effective antioxidant. Moreover, the observations in the study demonstrate zebrafish as a promising in vivo model for assessing the neuroprotective actions of bioactive compounds.
Keywords: deltamethrin, lead, vitamin C, protective role, zebrafish, 3D locomotion analysis
1. Introduction
In recent years, heavy metals and organic compound contamination in aquatic and terrestrial ecosystems has drawn increasing attention due to the detrimental effects on living organisms [1,2,3,4,5]. Environmental surveys have shown the co-occurrence of toxic metals and pyrethroids in water, sediments and fish samples [6,7,8,9]. Lead (Pb) is a toxic metal whose widespread use has resulted in extensive environmental contamination and health problems around the world. The World Health Organization lists Pb in its top ten chemicals of major public health concerns [10]. Although the use of Pb has been significantly reduced in fuel, paint, plumbing and food container solder, resulting in a substantial decline in population exposure to this toxic metal, significant sources of exposure and remanent Pb still exist, especially in developing and poor countries. For example, Pb was found in the bottom sediments of water bodies in the upper Silesia region (southern Poland) in amounts ranging from 32 to 3340 ppm [11]. In water samples from e-waste recycling sites in Guiyu of China, Pb concentration even reached as high as 400 μg L−1 [12]. An independent testing at six Washington DC public schools in 2008 showed that 2–41% of sampled tap water has a Pb contamination problem, with the highest lead detected at 1987 μg L−1, significantly higher than the EPA-recommended maximum level of 20 μg L−1 for schools [13]. As it is described in literature, in addition to its major neurotoxicity [14,15,16,17,18,19] the exposure to Pb has been linked to oxidative stress [20], cardiovascular toxicity [21], as well as gut microbiota dysbiosis and hepatic metabolic disorder [12] in zebrafish. Among the Pb-induced zebrafish neurobehavioral changes identified to date, learning and memory deficits, a decrease in the exploratory activities, altered social and aggressive behavior, color preference or responses to mechanosensory and visual stimuli are included [22,23,24,25,26]. It is very interesting to note that males showed a greater number of attacks on the mirror image versus females at higher exposure regimens (1 and 10 μM Pb), while females displayed a greater number of attacks versus males only at the lower treatment level (0.1 μM Pb) [22]. According to Xu and colleagues, chronic exposure to Pb induced learning impairments that persisted for at least three generations [27]. On the other hand, no significant changes were found in social interactions in adult zebrafish exposed to 50 μg L−1 PbCl2 [25]. Pyrethroids are synthetic insecticides derived from the natural pyrethrins, that are used for controlling various insect pests in agriculture, and are also used as veterinary drugs and domestic insecticides [28]. Due to their low toxicity and short half-life, these insecticides are being used as viable substitutes for other biocides, such as organophosphates and organochlorines. Pyrethroids are divided in two groups, type I (i.e., alletrin, permethrin, piretrin) and type II (deltamethrin, spermethrin) α cyano group. Deltamethrin (DM) is one of the most popular type II synthetic pyrethroids because of its photostability and high activity against a broad spectrum of insect pests [29,30]. As a result of their widespread use and high lipophilicity, DM can easily reach the aquatic ecosystems and cause serious environmental and health problems. For example, DM is found in environmental samples, such as water and sediments, food, organisms and even human samples (e.g., blood and urine) [31,32,33,34,35,36,37]. The DM concentration in water samples ranged from ng L−1 to μg L−1. Previous studies have reported that DM exposure could induce zebrafish developmental delay and malformation [38,39,40], apoptosis [38,40], cardiovascular toxicity [38], gonadotoxicity [41] as well as changes in neurochemical, behavioral and cognitive endpoints [20,38,39,42,43,44]. Hyperactivity and swimming towards the surfaces were observed in adult zebrafish after DM (>0.1 μg L−1) exposure [39,45]. An increase in the thigmotaxis of zebrafish larvae (preference for the walls) has been observed after treatment with low concentrations of DM (0.1, 1 and 10 μg L−1), while at high concentrations (25 and 50 μg L−1) the activity decreased [38]. A similar result was found by Hu et al. [43] where they documented significantly reduced average swimming speed of zebrafish larvae exposed to DM at 25 μg L−1. Moreover, the results showed that the locomotor activity of zebrafish larvae and the transcript levels of the components of estrogenic and dopaminergic pathways were altered by DM exposure. This was corroborated by Kung et al., where DM exposure during the embryonic period (3–72 hpf) causes hyperactivity in larval zebrafish, which is likely mediated by dopaminergic dysfunction [42].
Although multiple contaminants at low or higher levels coexist and may interact in the environment, most of the published results are focusing on the effects of single chemicals. The evaluation of single pollutant acute and chronic effects does not offer a close to reality view of their impact against aqueous ecosystems. The few studies that evaluated the combined toxicity using zebrafish revealed both synergistic and antagonistic interaction between chemicals [46,47,48,49,50,51]. For example, Our group research previously demonstrated that the toxicity of DM when combined with Cd and Ni significantly decreased as shown by some of the behavioral variables and oxidative stress responses [46]. A similar tendency was reported by Tilton et al. [47], when the addition of copper (Cu) to the moderate dose of chlorpyrifos (CPF) partially attenuated the number of fish undergoing freeze responses. Synergistic responses on zebrafish were observed for binary mixtures of iprodione in combination with pyrimethanil or acetamiprid and ternary mixtures of iprodione + pyraclostrobin in combination with pyrimethanil or acetamiprid [51]. Ku et al. found that the combination of NiSO4 and buprofezin formed a complex that facilitated the uptake of compounds by the zebrafish embryos, inducing synergistic effects [50].
Today, zebrafish has become a preferred model organism for ecotoxicological research due to its small size, prolific spawning, rapid life cycle, easy husbandry and low cost, as well because they share many molecular, biochemical, cellular and physiological characteristics with higher vertebrates [52,53,54,55,56]. Additionally, because they exhibit complex behaviors zebrafish may be use in modeling neurobehavioral disorders, to probe the neuroprotective actions of bioactive compounds and to assess the short- or long-term consequences of toxic chemicals exposure [57,58,59]. Although the use of behavioral tests for studying the toxicological effects is relatively a new approach, they have gained significant attention compared with conventional classical methods assessing the growth, lethality and reproductive effects [60,61]. They have attracted interest due to their sensitivity and rapidity, where changes in fish behavioral responses following chemicals exposure may be observed immediately and at relatively low concentrations. The 3D locomotion tracking offers a marked advantage in the existing 2D video tracking, enabling a comprehensive analysis of fish locomotion, interpretation of complex fish behaviors (e.g., anxiogenic vs. anxiolytic manipulations) and detection of subtle behavior alterations [62].
Nowadays there is an increased interest in evaluating the efficiency of dietary products (e.g., vitamin C, vitamin E, quercetin, etc.) in alleviation of pollutants toxicity [63,64,65]. Recent studies have shown that vitamin C (VC, ascorbic acid) co-administration or supplementation in diets of aquatic organisms may reduce the toxicity induced by various pollutants, such as toxic metals, pesticides or pharmaceutical products, acting as a recuperative/protective agent (Table 1). The pretreatment with VC appeared to be a viable option to counter the anxiogenic-like effect and biochemical alterations induced by methylmercury in zebrafish [66]. Similarly, Robea et al. [67] showed that the pretreatment with VC (25 μg L−1) significantly attenuated the neurotoxicity of fipronil (Fip, 600 μg L−1) and pyriproxyfen (Pyr, 600 μg L−1) mixture, as well the oxidative stress. Results revealed that embryonic treatment of zebrafish with VC during embryogenesis enhanced the cell proliferation leading to increased somatic growth in the larval stages, which persisted until the juvenile stage [68]. Butachlor-induced toxicity [69] and neomycin-induced ototoxicity [70] have been ameliorated by VC co-administration, as indicated by decreased mortality, malformation occurrence and the reversion in the response of biomarkers of oxidative stress. VC supplementation was shown to alleviate oxidative stress in tilapia (Oreochromis niloticus) liver and brain tissues induced by chlorpyrifos (CPF) exposure [71]. Only a few species, including humans, guinea pigs, capybara and some birds and fish (i.e.,zebrafish) cannot synthesize VC and this has to be obtained from diet.
Table 1.
Summary of fish studies regarding the protective/recuperative role of vitamin C.
| Chemical Exposure |
Vitamin C | Exposure Period | Fish | Assessments | Refs. | |
|---|---|---|---|---|---|---|
| administration | - | 0.5 and 1.0 g kg−1 VC diets | 6 weeks | zebrafish |
|
[72] |
| pretreatment with VC | 1 μg g−1 MeHg | 2 mg g−1
(intraperitoneal) |
24 h | adult zebrafish |
|
[66] |
| 600 μg L−1 Pyr 600 μg L−1 Fip |
25 μg L−1 | 14 days | zebrafish juvenile |
|
[67] | |
| dietary supplement of VC | 12 and 24 μg L−1 CPF | 200 mg VC 100 g−1 feed | 96 h | Nile tilapia, Oreochromis Niloticus |
|
[71] |
| 5 μg L−1 DM | 100 mg VC 100 g−1 feed supplemented diet for 60 days | 24 h | catfish, Clarias gariepinus |
|
[73] | |
| 0.22 μg L−1 and 0.44 μg L−1
cypermethrin |
300 mg VC 100 g−1 feed supplemented diet for 20 days | 20 days of exposure + 15 days of recuperation | Nile tilapia, Oreochromis niloticus |
|
[74] | |
| co-administration | 200 and 400 μg L−1 CPF | 200 and 400 μg L−1 | 72 h | zebrafish embryos |
|
[75] |
| 1–15 μmol L−1 butachlor | 40 and 80 μg L−1 | 72 h | zebrafish embryos/larvae |
|
[69] | |
| 20 μM neomycin | 0.002–0.02 μg L−1 (14.2–113.6 μM) |
30 min | zebrafish embryos/larvae |
|
[70] | |
| 2 μg L−1 DM and 60 μg L−1 Pb | 100 μg L−1 | 48 h | juvenile zebrafish |
|
Present study |
In the present study, the risk of exposure to lead and deltamethrin at environmental concentration, as well as the ameliorative effect of vitamin C in juvenile zebrafish were assessed by behavioral and biochemical analyses. To the best of our knowledge, no study has earlier reported the protective role of vitamin C against a heavy metal/pyrethroid insecticide mixture induced toxicity in a zebrafish model. In addition, so far there is no report concerning the application of 3D locomotor activity test for studying the efficiency of VC in alleviation of toxicity induced by a mixture of chemicals.
2. Results and Discussion
In order to assess the toxicity of DM and Pb mixtures, as well the protective effects of VC against impairments induced by the pyrethroid insecticide and heavy metal mixture acute exposure against zebrafish juvenile, we performed the three-dimensional (3D) locomotor activity assay. No deaths were found in the control and experimental groups after acute exposure to chemicals. Although variations existed between individuals, distinct spatial differences among the five groups can easily be observed from the 3D swim path reconstructions. As shown in Figure 1, the zebrafish exposed to DM alone displayed an increased top exploration along the perimeter of the tank that turned to normal after 48 h, while the specimens exposed to DM and Pb mixtures exhibited the tendency to swim near the bottom of the tank and gradually started to move to the upper zone after 24 h. Thereby, the time spent in the upper zone indicates an anxiolytic-like behavior and the tendency of zebrafish to swim at the bottom of the tank shows a typical geotaxis, as a result of a high anxiety level [62,76]. Similarly, Egan et al. [77] observed that chronic fluoxetine (100 μg L−1) and 0.3% ethanol acute treatments increased the time spent in the top area and the number of transitions to the upper portion of zebrafish. The acute ibogaine (10 mg L−1) exposure of zebrafish evoked a short initial top exploration followed by a gradual swimming toward the bottom of the tank [78]. The treatment of adult zebrafish with ZnCl2 (0.5–1.5 mg L−1) [79] and PbCl2 (50 μg L−1) [25] for different time periods resulted in reduction in exploratory activity, average speed, entries in the upper zone and elevated freezing duration compared to the control group.
Figure 1.
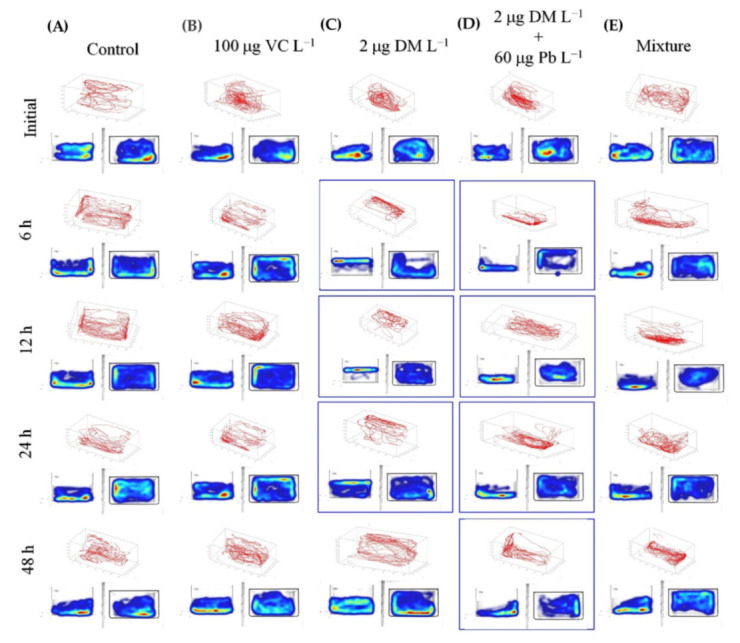
The 3D swim traces results for zebrafish control group (A) and groups treated with (B) 100 μg VC L−1, (C) 2 µg DM L−1, (D) 2 µg DM L−1 + 60 µg Pb L−1 and (E) mixture of VC, Pb and DM at 0, 6, 12, 24 and 48 h (n = 15). An automated integration of traces using Track3D software results in 3D swim tracks (red color). The bottom panels show the side and top view of zebrafish in the 4 min test.
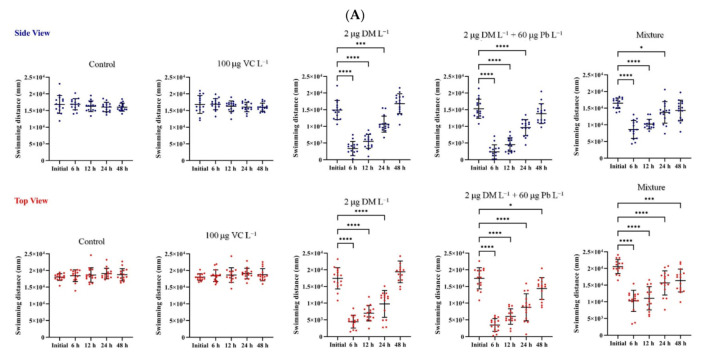
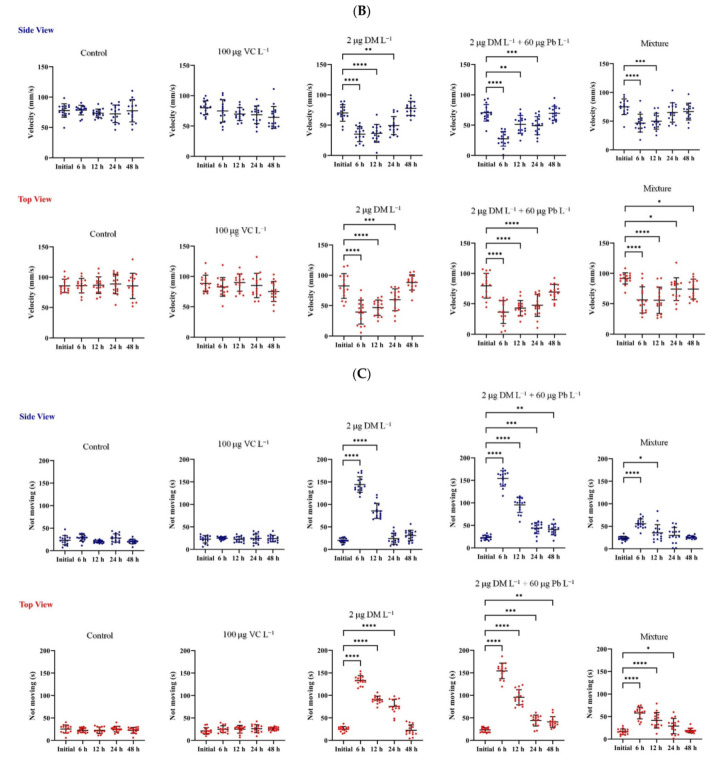
Generally, the time spent as moving/freezing represents the anxiety level. As can be seen in Figure 2A,B, the control and VC groups did not show changes in the total distance traveled during the exposure period, while a significant difference was observed for the total distance swam in the groups exposed to DM alone and in combination with Pb (Figure 2C,D). In addition, both groups presented a significant increasing trend in distance swam and velocity during acute exposure period, as a physiological adaptation of zebrafish to chemicals. The lower value of total distance traveled was measured 6 h after exposure for pyrthroid and heavy metal mixture-treated group. No behavioral change was depicted 48 h after exposure to DM alone. The exposure to DM without and with Pb increased the immobile mean time from an average of 10% of the time for control and VC groups to an average of 60% and 64% of the time, respectively. Based on the zebrafish behavioral endpoints and 3D swim path reconstruction, the DM associated with Pb elicited a slightly more pronounced effects.
Figure 2.
3D locomotor activity assay for control and treated groups with 100 μg VC L−1, 2 µg DM L−1, 2 µg DM L−1 + 60 µg Pb L−1 and mixture of VC, Pb and DM. The graphs show (A) the total distance in the 3D space covered by the zebrafish (mm), (B) the velocity (mm/s) and (C) freezing duration (s) under the condition of no exposure and exposure to chemicals. Each dot represents an individual zebrafish (n = 15 each experimental group). Data were expressed as mean ± SD. Statistically significant differences are denoted by * p< 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001 (one-way ANOVA, followed by a Dunnett’s multiple comparisons test).
Considering the multifaceted role played by vitamin C, we investigated the effectiveness of ascorbic acid in reducing the impairments caused by DM and Pb mixture acute exposure against zebrafish juvenile. As illustrated in Figure 2E, the VC supplementation significantly ameliorated the DM + Pb− induced alteration in zebrafish 3 D locomotion. Similarly to the control and VC-treated groups, the zebrafish covered the entire tank in their exploration. In addition, simultaneous acute exposure to DM + Pb mixture and vitamin C has normalized the values of kinematic parameters, within initial limits. For example, the co-administration of VC increased the moving duration measured at 6 h after exposure of zebrafish juvenile with 41%.
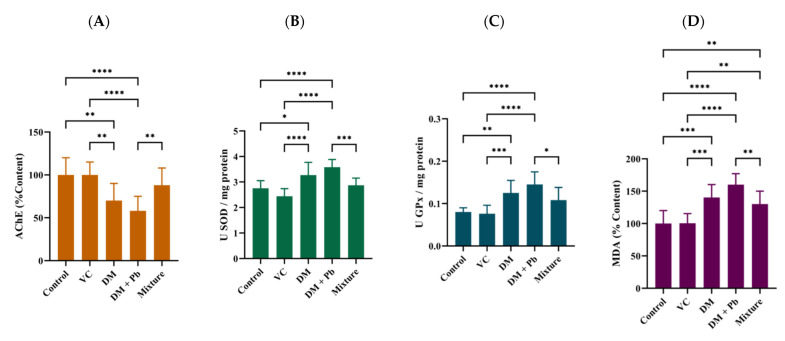
As a result of exposure to environmental pollutants, the fine equilibrium between reactive oxygen species (ROS) production and removal by the antioxidant defense systems may be impaired and oxidative damage may occur in the organism [80]. It is known that fish have an efficient antioxidant defense system, composed of various antioxidant enzymes (e.g., superoxide dismutase (SOD), glutathione peroxidase (GPx), glutathione S-transferase (GST) or catalase (CAT)), as well as other low molecular weight scavengers (e.g., non-protein thiols (NPSH)) [81]. With the aim being to evaluate the potential oxidative stress induced by DM and Pb and the efficiency of VC supplementation to mitigate their harmful effects against zebrafish, the superoxide dismutase (SOD) and glutathione peroxidase (GPx) activities, as well MDA levels were determined. While SOD and GPx constitute the first defense line against the toxic effect of oxygen-derived free radicals, lipid peroxidation is one of the main manifestation of oxidative damage induced by chemical compounds [67]. Interestingly, we found that SOD activity, GPx activity and lipid peroxidation were significantly increased in response to the DM and DM + Pb treatments when compared with the control and vitamin C groups, while the raised enzymes activities and MDA level measured for pyrethroid and heavy metal group were significantly decreased by co-administration of vitamin C (Figure 3B–D). Though the present data could not conclusively deduce whether the ameliorative effect of VC against oxidative stress is through ROS elimination or inhibition of free radical formation, the changes observed indicate that it plays a significant role to the response towards the chemicals. The findings are consistent with previous results, Korkmaz et al. [74] and Datta and Kaviraj [73] evaluating the efficiency of VC supplementation in order to remove harmful effects of pesticides (cypermethrin and deltamethrin) in tissue of Oreochromis niloticus and Clarias gariepinus.
Figure 3.
Activity of (A) AChE, (B) SOD, (C) GPx and (D) MDA levels in zebrafish following treatment with vitamin C (100 μg L−1), DM (2 μg L−1) and Pb (60 μg L−1) alone and in combination with VC (100 μg L−1) for 48 h. Values are expressed as means ± SD (n = 9). Statistically significant differences are denoted by * p < 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001 (one-way ANOVA, followed by a Turkey’s test).
These following studies documented that the repair was not complete and a prolonged recovery period was necessary to return to the basal state. Moreover, it has been found that VC may regulate the production of ROS through Wnt10b signaled in the gill of zebrafish [72]. Wu and colleagues studied the protective role of VC on neomycin-induced ototoxicity in zebrafish and demonstrated that the uptake of neomycin was not influenced by ascorbic acid and they assumed that the inhibition of excessive ROS production contributed to VC protective activity [70].
Acetylcholinesterase (AChE), a key enzyme in cholinergic transmission in the nervous system, is an important biomarker in determining the effects of environmental pollutants in fish. Significant decrease in AChE activity in zebrafish brains exposed to DM with and without Pb were observed relative to control group (Figure 3A), also noted previously by other studies [25,40,77,82]. Parlak reported that AChE activity dramatically decreased in a dose-dependent manner as compared with the control group after exposure of zebafish embryos to detamethrin (2.5–50 μg L−1) [40]. Thi et al. shown a reduction in AChE activity in fish after exposure to PbCl2 (50 μg L−1) [25]. In addition, the neurobehavioral impairment observed in the PbCl2-exposed zebrafish has been shown to not be primarily associated with the cholinergic system. A strong, as well as a poor relationship between the alteration of AChE activity and behavioral impairments upon short- or long-term exposure to toxic chemicals have been reported in the literature [47,83,84]. Similar to those observed previously, the effects of chemicals mixture on AChE activity were reversed by vitamin C supplementation, also providing a possible explanation for the preservation of swimming behavior.
Although in the literature it is indicated that pyrethroids interfere with the functioning of sodium channel [85] and the heavy metals uptake is mediated by Ca2+ transport pathways [86], in our study the acute exposure to DM and Pb alone and in combination with VC have no visible effect on the total sodium or calcium from the zebrafish body. As shown in Table 2, no significant change was measured in element concentrations between the different experimental conditions, possibly due to the short exposure duration. This finding agrees well with our previous results: when the chronic effects of DM and the acute effects of Cd, Ni and DM on zebrafish is studied, the concentration of essential elements involved in biological structure and biochemical processes did not change in zebrafish body mass [44,46].
Table 2.
Total electrolytes and trace metal profiles reported in zebrafish body measured for wet weight (average ± SD).
| Elements (μg g−1) |
Exposure | |||
|---|---|---|---|---|
| Control | 100 μg VC L−1 | 2 μg DM L−1 + 60 μg Pb L−1 | 2 μg DM L−1 + 60 μg Pb L−1
+ 100 μg VC L−1 |
|
| Cu | 1.23 ± 0.14 | 1.14 ± 0.04 | 1.16 ± 0.117 | 1.22 ± 0.05 |
| Fe | 20.5 ± 0.6 | 19.3 ± 1.2 | 17.5 ± 2.2 | 18.7 ± 1.5 |
| Ca | 5586 ± 156 | 5478 ± 325 | 5681 ± 225 | 5521 ± 358 |
| Mg | 390 ± 46 | 402 ± 11.6 | 397 ± 37.6 | 367 ± 52 |
| Na | 708 ± 52 | 644 ± 29 | 717 ± 59 | 679 ± 28 |
| K | 2061 ± 69 | 1993 ± 57 | 2062 ± 269 | 2067 ± 105 |
The results for the fish muscle reference material that was prepared and analyzed were: Na (certified 2.8 g kg−1 and obtained 2.65 ± 1.3 g kg−1), K (certified 21.8 g kg−1 and obtained 21.1 ± 0.65g kg−1), Ca (certified 0.342 g kg−1 and obtained 0.343 ± 0.002 g kg−1), Mg (certified 1.37 g kg−1 and obtained 1.38 ± 0.03 g kg−1), Zn (certified 16 ± 1.1 mg kg−1 and obtained 15.3 ± 1.2 mg kg−1), Fe (certified 9.4 ± 1.4 mg kg−1 and obtained 9.3 ± 0.96 mg kg−1) and Cu (certified 1.67 ± 0.16 mg kg−1 and obtained 1.64 ± 0.32 mg kg−1).
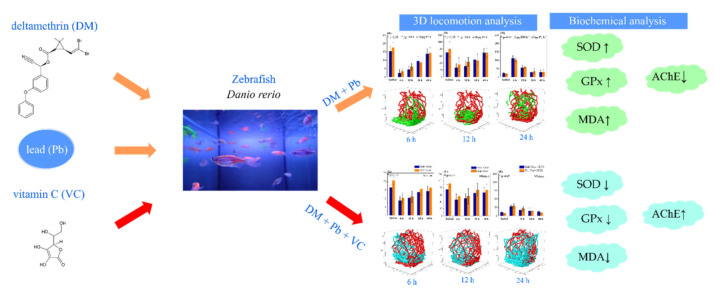
Finally, our results not only advance the understanding of deltamethrin and lead combined toxicity, but also reveal the efficiency of vitamin C co-supplementation in the alleviation of behavioral and biochemical alterations induced by a mixture of pollutants (Figure 4). Although we proved that VC has mitigative effects, there is a shortcoming concerning the quantification of the chemicals in water and fish tissues. In addition, further research is required into molecular mechanisms to establish a link between the molecular events and behavioral effects, as well on the capacity of zebrafish to recover after exposure to contaminants (post-exposure period).
Figure 4.
Schematic presentation of the present study. The adverse effects of exposure to environmental pollutants, such as lead and deltamethrin mixture, and the efficiency of vitamin C in alleviation of their toxicity in juvenile zebrafish were assessed. The exposure to a heavy metal/pyrethroid mixture induced behavioral abnormalities and oxidative stress in fish. In addition, the efficiency of vitamin C in alleviation of toxicity caused by chemicals exposure was demonstrated.
3. Materials and Methods
3.1. Ethical Note
The animals were strictly maintained and treated according to EU Commission Recommendation (2007) on guidelines for the accommodation and care of animals used for experimental and other scientific purposes, Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes. This experiment has been approved by Ethics Commission from Faculty of Biology, Alexandru Ioan Cuza” University of Iasi, with the registration number 2923/05.10.2021.
3.2. Chemicals
Lead standard solution Certipur® (Pb, 1000 mg L−1, 119,776), nitric acid 65% Suprapur® (HNO3, 100,441), hydrogen peroxide 30% Perhydro® (H2O2, 107,210), malonaldehyde bis(diethyl acetal) (T9889), 2-thiobarbituric acid (TBA, T5500), trichloroacetic acid (TCA, 91,230), ethanol EMSURE® (159,010), phosphate buffered salin (P4417), tris hydrochloride solution (T2819), acetylthiocholine iodide (ATCh, A5751), 5, 5-dithio-2, 2-nitrobenzoic acid (DTNB, 322123), Bradford Reagent (B6916), Bovine Serum Albumin (A8022), Superoxide Dismutase Determination Kit (SOD, 19160-1KT-F), Glutathione Peroxidase Cellular Activity Assay Kit (GPx, CGP1-1KT) were purchased from Merck, Darmstadt, Germany. The deltamethrin used in the present study is the active compound of a well-known insecticide (DM, 100 g L−1) purchased from a local market with quality certification. The vitamin C (VC, ascorbic acid) liquid form was purchased from a local pharmacy. To avoid any conflict of interest for these products, the brand name of the producer was kept under anonymity. We chose to use them in the study in order to investigate a scenario similar to a real case.
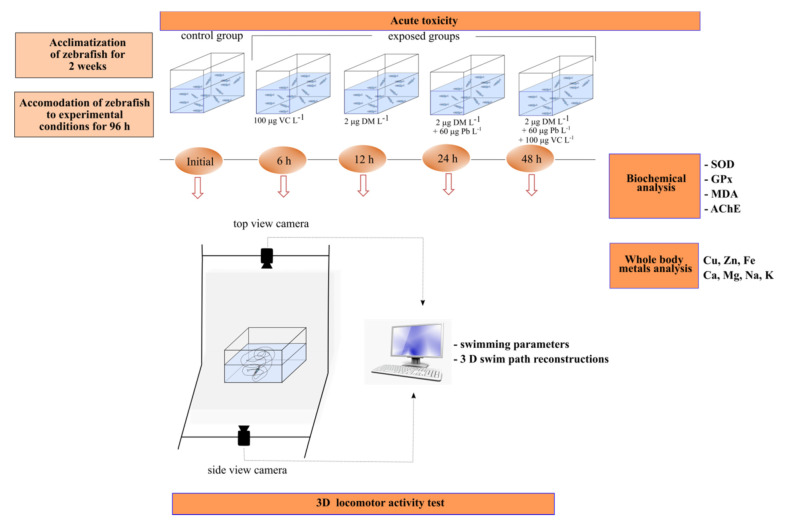
The experimental setup was designed according to OECD Guidelines for the Testing of Chemicals. Section 2: Effects on Biotic Systems. Fish acute toxicity test No. 203. After the acclimatization for 2 weeks, wild-type zebrafish (Dario rerio Hamilton, 1822) (n = 15 per group) were randomly divided into five groups as following: Group I was the control group, while Group II (fish exposed to VC at a concentration of 100 μg L−1), Group III (fish exposed to DM at a concentration of 2 μg L−1), Group IV (fish exposed to 2 μg DM L−1 and 60 μg Pb L−1) and Group V (fish exposed to 2 μg DM L−1 and 60 μg Pb L−1 in combination with 100 μg VC L−1) were the exposed groups housed in 10 L glass tanks with the respective treatment (Figure 5). The tanks had been filled with dechlorinated tap water that was constantly aerated by an air pump. Fish were not fed 24 h prior to and during exposure and the solution was renewed daily to maintain a similar concentration. In addition, before starting the behavioral tests, during 96 h zebrafish were gently hand-netted from their experimental tank to a temporary tank in order to recover from handling stress, and thereafter, transferred to a novel tank for 3D observations. After the testing was completed, fish from the control and the experimental groups were humanely euthanized with cold water between 2 and 4 °C, according to animal welfare regulations [87].
Figure 5.
The schematic representation of the experimental setup used to elucidate the potential toxic effect of DM in combination with Pb on zebrafish and its possible amelioration by vitamin C supplementation.
3.3. 3D locomotor Activity Test
The behavioral measurements were carried out in a 10 L glass trapezoidal tank filled with 6 L of dechlorinated tap water by using the Track3D module of EthoVisionXT 14 video tracking software (Noldus Information Technology, TheNetherlands, Holland). The experimental setup has been described in detail in our previous paper [46]. After the experimental accommodation for 96 h, each experimental group is studied using the 3D approach over a 4-min period to set the baseline behavior, presented in our study as initial behavior. Then the 3D locomotion tracking was acquired at 6 h, 12 h, 24 and 48 h for each individual and each experimental condition (100 μg VC L−1, 2 μg DM L−1 without and with 60 μg Pb L−1 and their mixture) to demonstrate the acute changes caused by chemicals exposure (Figure 5). The discussions on behavioral response results were for each group with comparison between initial behavior and treatments based on the averages resulting after experimental repetition. In order to assess the pollutants’ effects as well as the protective effect of VC against behavioral impairments, we analyzed the following behavioral endpoints: the total distance moved in the 3D space (m), velocity (mm s−1) and freezing duration (s), defined as the absence of the body movements.
3.4. Determination of AChE Activity
Following completion of exposure, whole brains were extracted and homogenized on ice in 60 vol. (v/w) of 0.05 M Tris-HCl, pH 8.0, according to a previously reported protocol [88]. AChE activity in the homogenates was determined according to protocol described by Pan et al. [82]. Briefly, 50 μL sample and 50 μL ATCh (5 mM) were incubated at 30 °C for 15 min to a final volume of 0.1 mL and then the reaction was stopped by 0.125 mM DTNB-phosphate-ethanol reagent inside 0.9 mL (12.4 mg of DTNB dissolved in 125 mL 95% ethanol, 75 mL distilled water and 50 mL 0.1 M PBS, pH 7.5) as the thiol indicator. The color was detected immediately at 412 nm by using the Specord 210 Plus from Analytik Jena, Germany. The results were normalized to the total protein content determined with the Bradford method [89] in which bovine serum albumin was used as standard. The AChE activity (% of control) is used to analyze the effects induced by chemicals. To ensure the consistence of our results, the tests were performed in triplicates.
3.5. Determination of Whole Body MDA Concentration, SOD and GPx Activities
After 48 h exposure period, each fish was homogenized in 10 volumes of ice-cold 0.1 M phosphate buffer (pH 7.4) and then centrifuged at 5500 rpm for 15 min (4 °C). The supernatant fraction was used to determine the SOD and GPx activities, MDA level and the protein concentrations in samples. Lipid peroxidation (LPO) was assessed using thiobarbituric acid reactive substances determination method. Briefly, whole-body homogenates (200 μL) were mixed to trichloroacetic acid (0.92 M in 0.25 N HCl) and thiobarbituric acid (26 mM) and vortexed. Afterwards, a 20 min incubation at 100 °C (boiling water bath) and a 10 min centrifugation (3000 rpm) were performed. The supernatants were read at 532 nm and the samples absorbances were compared with a melon-dialdehyde (MDA, 0.5–20 nmol mL−1) standard curve. Raw data were corrected by the protein content and normalized as percentage of control values. [90] Activities of SOD and GPx were determined colorimetrically using kits (Merck, Darmstadt, Germany), following the manufacturer’s instructions.
3.6. Whole Body Ions and Trace Metals Analysis
For the mineralization process, three fish per experimental group were washed several times with ultrapure water, ground, weighed (~0.3 g) and microwave digested (Berghof speedwave MWS-2) with 4 mL of HNO3 and 2 mL of 30% H2O2. Subsequently, the samples were transferred to 50 mL tubes and diluted with ultrapure water until a 50 mL volume was reached. Copper (Cu) concentrations were measured with HR-CS GF-AAS (ContrAA 700-Analytik, Jena, Germany) following the method described by Plavan et al. [91], while an air-acetylene FL-ASS with hollow-cathode lamps (GBC Avanta, Australia) was used to determine zinc (Zn), iron (Fe), calcium (Ca), sodium (Na), magnesium (Mg) and potassium (K) concentrations. Calibration has been accomplished with certified standard solutions (1000 mg L−1, from Merck, Darmstadt, Germany). The methods’ accuracy were validated by using a reference material for fish muscle (ERM-BB422), certified by the EU Joint Research Center Institute for Reference Materials and Measurements. From the reference material, 6 samples were prepared using the same digestion protocol. All concentrations were reported as µg g−1 wet weight ± SD [46].
3.7. Statistical Analysis
Firstly, the normality and distribution of the data were assessed using Graph Pad Prism software (v 9.0, San Diego, CA, USA), calculating Shapiro-Wilk test. Afterwards, comparisons among treatments were performed using one-way ANOVA followed by Turkey’s or Dunnett’s multiple comparisons tests, as indicated in the figure legends. All experiments were replicated at least three times to ensure the reproducibility of the results, the results being expressed as the mean ± standard deviation (SD). The p-values were shortened with the following symbols: * p < 0.05, ** p < 0.01, *** p < 0.001 and **** p <0.0001 (significance was set at p < 0.05). Plots were generated in Graph Pad Prism v9.0. The video trials resulted from the behavioral tests were sorted in files for each group. These were reused and combined in EthoVision XT 14 to generate average heat maps for experiment with replicas. With the Track3D module were extracted data where the average was calculated and represented in 3D figures (the 3D spatio-temporal reconstruction of the swim paths).
4. Conclusions
The results of behavioral and biochemical assays reveal that acute exposure to deltamethrin without or with lead may elevate the oxidative stress and anxiety level characterized by increased freezing duration and reduced exploratory activity, as well as may cause a significant decrease in the activity of AChE in zebrafish brain. Particular movement patterns have been observed for zebrafish exposed to DM and DM + Pb. The animals exposed to DM displayed significantly more transitions to and time in the upper half, over the 4 min testing period, while the specimens exposed to mixture spent more time at the tank bottom. Zebrafish exposed to DM and Pb gradually started to move to the upper zone after 24 h. It has been observed that VC co-administration can mitigate the behavioral impairments and the negative effect of oxidative stress caused by chemicals exposure in zebrafish. Treatment with VC reduced the elevated lipid peroxidation, as well the increased levels of SOD and GPx, and restored partially the reduced AChE activity. Together, these results indicate that zebrafish is a good model for testing chemicals present in the aquatic environment and similarly, in investigating compounds, such as vitamin C, that can mitigate the consequences from toxicant exposure. Future studies are necessary for the detailed characterization of the mechanisms underlying both the joint toxic effects and protective role observed.
Acknowledgments
This paper is dedicated to the memory of Stefan-Adrian Strungaru, colleague, researcher and friend, who prematurely passed away in April 2021.
Author Contributions
Conceptualization, R.J. and E.P.; methodology, R.J.; software, R.J., E.P., E.-I.F. and I.-A.S.; validation, R.J. and E.P.; formal analysis, A.C. and M.N.; investigation, E.P., E.-I.F. and I.-A.S.; resources, M.N., I.-A.S., C.C.L. and A.C.; data curation, R.J.; writing—original draft preparation, R.J.; writing—review and editing, R.J., C.C.L. and C.F.; visualization, R.J., E.P., C.F. and C.C.L.; supervision, M.N., C.F. and A.C.; C.C.L.; funding acquisition. All authors have read and agreed to the published version of the manuscript.
Funding
This study was partly funded by the Norwegian Seafood Research Fund (ref. 901472) and the Norwegian Research Council (ref. 194050).
Institutional Review Board Statement
The study was conducted according to EU Commission Recommendation (2007) on guidelines for the accommodation and care of animals used for experimental and other scientific purposes, Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes and OECD Guidelines for the Testing of Chemicals. Section 2: Effects on Biotic Systems. Fish acute toxicity test No. 203, and approved by Ethics Commission from Faculty of Biology, Alexandru Ioan Cuza” University of Iasi (2923/05.10.2021).
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Stara A., Pagano M., Albano M., Savoca S., Di Bella G., Albergamo A., Koutkova Z., Sandova M., Velisek J., Fabrello J. Effects of long-term exposure of Mytilus galloprovincialis to thiacloprid: A multibiomarker approach. Environ. Pollut. 2021;289:117892. doi: 10.1016/j.envpol.2021.117892. [DOI] [PubMed] [Google Scholar]
- 2.Sula E., Aliko V., Marku E., Nuro A., Faggio C. Evaluation of kidney histopathological alterations in Crucian Carp, Carassius carassius, from a pesticide and PCB-contaminated freshwater ecosystem, using light microscopy and organ index mathematical model. Int. J. Aquat. Biol. 2020;8:154–165. [Google Scholar]
- 3.Stara A., Pagano M., Capillo G., Fabrello J., Sandova M., Albano M., Zuskova E., Velisek J., Matozzo V., Faggio C. Acute effects of neonicotinoid insecticides on Mytilus galloprovincialis: A case study with the active compound thiacloprid and the commercial formulation calypso 480 SC. Ecotoxicol. Environ. Saf. 2020;203:110980. doi: 10.1016/j.ecoenv.2020.110980. [DOI] [PubMed] [Google Scholar]
- 4.Pagano M., Stara A., Aliko V., Faggio C. Impact of neonicotinoids to aquatic invertebrates—in vitro studies on Mytilus galloprovincialis: A review. J. Mar. Sci. Eng. 2020;8:801. doi: 10.3390/jmse8100801. [DOI] [Google Scholar]
- 5.Stara A., Pagano M., Capillo G., Fabrello J., Sandova M., Vazzana I., Zuskova E., Velisek J., Matozzo V., Faggio C. Assessing the effects of neonicotinoid insecticide on the bivalve mollusc Mytilus galloprovincialis. Sci. Total Environ. 2020;700:134914. doi: 10.1016/j.scitotenv.2019.134914. [DOI] [PubMed] [Google Scholar]
- 6.Salvadó V., Quintana X.D., Hidalgo M. Monitoring of nutrients, pesticides, and metals in waters, sediments, and fish of a wetland. Arch. Environ. Contam. Toxicol. 2006;51:377–386. doi: 10.1007/s00244-005-0107-7. [DOI] [PubMed] [Google Scholar]
- 7.Allinson G., Zhang P., Bui A., Allinson M., Rose G., Marshall S., Pettigrove V. Pesticide and trace metal occurrence and aquatic benchmark exceedances in surface waters and sediments of urban wetlands and retention ponds in Melbourne, Australia. Environ. Sci. Pollut. Res. 2015;22:10214–10226. doi: 10.1007/s11356-015-4206-3. [DOI] [PubMed] [Google Scholar]
- 8.Nasr I., Sallam A., Abd El-Khair A. Monitoring of certain pesticide residues and some heavy metals in fresh cow’s milk at Gharbia Governorate, Egypt. J. Appl. Sci. 2007;7:3038–3044. doi: 10.3923/jas.2007.3038.3044. [DOI] [Google Scholar]
- 9.Kronvang B., Laubel A., Larsen S.E., Friberg N. Pesticides and heavy metals in Danish streambed sediment. Hydrobiologia. 2003;494:93–101. doi: 10.1023/A:1025441610434. [DOI] [Google Scholar]
- 10.WHO Chemicals of Major Public Health Concerns. 2010. [(accessed on 21 August 2021)]. Available online: https://www.who.int/teams/environment-climate-change-and-health/chemical-safety-and-health/health-impacts/chemicals/lead.
- 11.Rzętała M.A. Lead in the bottom sediments of water bodies in the Upper Silesia region (southern Poland) Desalination Water Treat. 2015;54:779–789. doi: 10.1080/19443994.2014.886297. [DOI] [Google Scholar]
- 12.Xia J., Lu L., Jin C., Wang S., Zhou J., Ni Y., Fu Z., Jin Y. Effects of short term lead exposure on gut microbiota and hepatic metabolism in adult zebrafish. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2018;209:1–8. doi: 10.1016/j.cbpc.2018.03.007. [DOI] [PubMed] [Google Scholar]
- 13.Triantafyllidou S., Lambrinidou Y., Edwards M. Lead (Pb) exposure through drinking water: Lessons to be learned from recent US experience. Glob. NEST J. 2009;11:341. [Google Scholar]
- 14.Abdel-Wahab M.S., Jilani A., Yahia I., Al-Ghamdi A.A. Enhanced the photocatalytic activity of Ni-doped ZnO thin films: Morphological, optical and XPS analysis. Superlattices Microstruct. 2016;94:108–118. doi: 10.1016/j.spmi.2016.03.043. [DOI] [Google Scholar]
- 15.Peterson S.M., Zhang J., Freeman J.L. Developmental reelin expression and time point-specific alterations from lead exposure in zebrafish. Neurotoxicol. Teratol. 2013;38:53–60. doi: 10.1016/j.ntt.2013.04.007. [DOI] [PubMed] [Google Scholar]
- 16.Zhang J., Peterson S.M., Weber G.J., Zhu X., Zheng W., Freeman J.L. Decreased axonal density and altered expression profiles of axonal guidance genes underlying lead (Pb) neurodevelopmental toxicity at early embryonic stages in the zebrafish. Neurotoxicol. Teratol. 2011;33:715–720. doi: 10.1016/j.ntt.2011.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lee J., Freeman J.L. Embryonic exposure to 10 μg L− 1 lead results in female-specific expression changes in genes associated with nervous system development and function and Alzheimer’s disease in aged adult zebrafish brain. Metallomics. 2016;8:589–596. doi: 10.1039/C5MT00267B. [DOI] [PubMed] [Google Scholar]
- 18.Roy N.M., DeWolf S., Schutt A., Wright A., Steele L. Neural alterations from lead exposure in zebrafish. Neurotoxicol. Teratol. 2014;46:40–48. doi: 10.1016/j.ntt.2014.08.008. [DOI] [PubMed] [Google Scholar]
- 19.Tu H., Peng T., Liu J., Chen X., Fan C., Huang Z., Zhang Y., Zou F., Meng X. Role of neurexin2a in lead-induced locomotor defect in developing zebrafish. Aquat. Toxicol. 2018;194:167–175. doi: 10.1016/j.aquatox.2017.11.011. [DOI] [PubMed] [Google Scholar]
- 20.Kataba A., Botha T.L., Nakayama S.M., Yohannes Y.B., Ikenaka Y., Wepener V., Ishizuka M. Acute exposure to environmentally relevant lead levels induces oxidative stress and neurobehavioral alterations in larval zebrafish (Danio rerio) Aquat. Toxicol. 2020;227:105607. doi: 10.1016/j.aquatox.2020.105607. [DOI] [PubMed] [Google Scholar]
- 21.Kataba A., Botha T.L., Nakayama S.M.M., Yohannes Y.B., Ikenaka Y., Wepener V., Ishizuka M. Environmentally relevant lead (Pb) water concentration induce toxicity in zebrafish (Danio rerio) larvae. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2022;252:109215. doi: 10.1016/j.cbpc.2021.109215. [DOI] [PubMed] [Google Scholar]
- 22.Weber D.N., Ghorai J.K. Experimental design affects social behavior outcomes in adult zebrafish developmentally exposed to lead. Zebrafish. 2013;10:294–302. doi: 10.1089/zeb.2012.0780. [DOI] [PubMed] [Google Scholar]
- 23.Rice C., Ghorai J.K., Zalewski K., Weber D.N. Developmental lead exposure causes startle response deficits in zebrafish. Aquat. Toxicol. 2011;105:600–608. doi: 10.1016/j.aquatox.2011.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chen J., Chen Y., Liu W., Bai C., Liu X., Liu K., Li R., Zhu J.-H., Huang C. Developmental lead acetate exposure induces embryonic toxicity and memory deficit in adult zebrafish. Neurotoxicol. Teratol. 2012;34:581–586. doi: 10.1016/j.ntt.2012.09.001. [DOI] [PubMed] [Google Scholar]
- 25.Bui Thi N.H., Nguyen Thi N.A., Audira G., Siregar P., Liang S.-T., Huang J.-C., Hsiao C.-D. Chronic exposure to low concentration lead chloride-induced anxiety and loss of aggression and memory in zebrafish. Int. J. Mol. Sci. 2020;21:1844. doi: 10.3390/ijms21051844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bault Z.A., Peterson S.M., Freeman J.L. Directional and color preference in adult zebrafish: Implications in behavioral and learning assays in neurotoxicology studies. J. Appl. Toxicol. 2015;35:1502–1510. doi: 10.1002/jat.3169. [DOI] [PubMed] [Google Scholar]
- 27.Xu X., Weber D., Burge R., VanAmberg K. Neurobehavioral impairments produced by developmental lead exposure persisted for generations in zebrafish (Danio rerio) Neurotoxicology. 2016;52:176–185. doi: 10.1016/j.neuro.2015.12.009. [DOI] [PubMed] [Google Scholar]
- 28.Corcellas C., Eljarrat E., Barceló D. First report of pyrethroid bioaccumulation in wild river fish: A case study in Iberian river basins (Spain) Environ. Int. 2015;75:110–116. doi: 10.1016/j.envint.2014.11.007. [DOI] [PubMed] [Google Scholar]
- 29.Radovanović T.B., Gavrilović B.R., Petrović T.G., Despotović S.G., Gavrić J.P., Kijanović A., Mirč M., Kolarov N.T., Faggio C., Prokić M.D. Impact of desiccation pre-exposure on deltamethrin-induced oxidative stress in Bombina variegata juveniles. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2021;250:109191. doi: 10.1016/j.cbpc.2021.109191. [DOI] [PubMed] [Google Scholar]
- 30.Gharaei A., Karimi M., Mirdar J., Miri M., Faggio C. Population growth of Brachionus calyciflorus affected by deltamethrin and imidacloprid insecticides. Iran. J. Fish. Sci. 2020;19:588–601. [Google Scholar]
- 31.Yáñez L., Ortiz-Pérez D., Batres L.E., Borja-Aburto V.C.H., Díaz-Barriga F. Levels of dichlorodiphenyltrichloroethane and deltamethrin in humans and environmental samples in malarious areas of Mexico. Environ. Res. 2002;88:174–181. doi: 10.1006/enrs.2002.4333. [DOI] [PubMed] [Google Scholar]
- 32.Daka P., Obuseng V., Torto N., Huntsman-Mapila P. Deltamethrin in sediment samples of the Okavango Delta, Botswana. Water Sa. 2006;32:483–488. doi: 10.4314/wsa.v32i4.5288. [DOI] [Google Scholar]
- 33.Pawlisz A., Busnarda J., McLauchlin A., Caux P.Y., Kent R. Canadian water quality guidelines for deltamethrin. Environ. Toxicol. Water Qual. Int. J. 1998;13:175–210. doi: 10.1002/(SICI)1098-2256(1998)13:3<175::AID-TOX1>3.0.CO;2-4. [DOI] [Google Scholar]
- 34.Feo M.L., Ginebreda A., Eljarrat E., Barceló D. Presence of pyrethroid pesticides in water and sediments of Ebro River Delta. J. Hydrol. 2010;393:156–162. doi: 10.1016/j.jhydrol.2010.08.012. [DOI] [Google Scholar]
- 35.Esteve-Turrillas F.A., Pastor A., de la Guardia M. Determination of pyrethroid insecticide residues in vegetable oils by using combined solid-phases extraction and tandem mass spectrometry detection. Anal. Chim. Acta. 2005;553:50–57. doi: 10.1016/j.aca.2005.08.004. [DOI] [Google Scholar]
- 36.Channa K.R., Röllin H.B., Wilson K.S., Nøst T.H., Odland J.Ø., Naik I., Sandanger T.M. Regional variation in pesticide concentrations in plasma of delivering women residing in rural Indian Ocean coastal regions of South Africa. J. Environ. Monit. 2012;14:2952–2960. doi: 10.1039/c2em30264k. [DOI] [PubMed] [Google Scholar]
- 37.Hardt J., Angerer J. Biological monitoring of workers after the application of insecticidal pyrethroids. Int. Arch. Occup. Environ. Health. 2003;76:492–498. doi: 10.1007/s00420-003-0451-8. [DOI] [PubMed] [Google Scholar]
- 38.Li M., Liu X., Feng X. Cardiovascular toxicity and anxiety-like behavior induced by deltamethrin in zebrafish (Danio rerio) larvae. Chemosphere. 2019;219:155–164. doi: 10.1016/j.chemosphere.2018.12.011. [DOI] [PubMed] [Google Scholar]
- 39.Liu X., Zhang Q., Li S., Mi P., Chen D., Zhao X., Feng X. Developmental toxicity and neurotoxicity of synthetic organic insecticides in zebrafish (Danio rerio): A comparative study of deltamethrin, acephate, and thiamethoxam. Chemosphere. 2018;199:16–25. doi: 10.1016/j.chemosphere.2018.01.176. [DOI] [PubMed] [Google Scholar]
- 40.Parlak V. Evaluation of apoptosis, oxidative stress responses, AChE activity and body malformations in zebrafish (Danio rerio) embryos exposed to deltamethrin. Chemosphere. 2018;207:397–403. doi: 10.1016/j.chemosphere.2018.05.112. [DOI] [PubMed] [Google Scholar]
- 41.Petrovici A., Strungaru S.-A., Nicoara M., Robea M.A., Solcan C., Faggio C. Toxicity of deltamethrin to zebrafish gonads revealed by cellular biomarkers. J. Mar. Sci. Eng. 2020;8:73. doi: 10.3390/jmse8020073. [DOI] [Google Scholar]
- 42.Kung T.S., Richardson J.R., Cooper K.R., White L.A. Developmental deltamethrin exposure causes persistent changes in dopaminergic gene expression, neurochemistry, and locomotor activity in zebrafish. Toxicol. Sci. 2015;146:235–243. doi: 10.1093/toxsci/kfv087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hu T., Ma Y., Qiao K., Jiang Y., Li S., Gui W., Zhu G. Estrogen receptor: A potential linker of estrogenic and dopaminergic pathways in zebrafish larvae following deltamethrin exposure. Sci. Total Environ. 2022;802:149938. doi: 10.1016/j.scitotenv.2021.149938. [DOI] [PubMed] [Google Scholar]
- 44.Strungaru S.-A., Plavan G., Ciobica A., Nicoara M., Robea M.A., Solcan C., Petrovici A. Toxicity and chronic effects of deltamethrin exposure on zebrafish (Danio rerio) as a reference model for freshwater fish community. Ecotoxicol. Environ. Saf. 2019;171:854–862. doi: 10.1016/j.ecoenv.2019.01.057. [DOI] [PubMed] [Google Scholar]
- 45.Huang Y., Zhang J., Han X., Huang T. The use of zebrafish (Danio rerio) behavioral responses in identifying sublethal exposures to deltamethrin. Int. J. Environ. Res. Public Health. 2014;11:3650–3660. doi: 10.3390/ijerph110403650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Jijie R., Solcan G., Nicoara M., Micu D., Strungaru S.-A. Antagonistic effects in zebrafish (Danio rerio) behavior and oxidative stress induced by toxic metals and deltamethrin acute exposure. Sci. Total Environ. 2020;698:134299. doi: 10.1016/j.scitotenv.2019.134299. [DOI] [PubMed] [Google Scholar]
- 47.Tilton F.A., Bammler T.K., Gallagher E.P. Swimming impairment and acetylcholinesterase inhibition in zebrafish exposed to copper or chlorpyrifos separately, or as mixtures. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2011;153:9–16. doi: 10.1016/j.cbpc.2010.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kim K., Jeon H.-J., Choi S.-D., Tsang D.C.W., Oleszczuk P., Ok Y.S., Lee H.-S., Lee S.-E. Combined toxicity of endosulfan and phenanthrene mixtures and induced molecular changes in adult Zebrafish (Danio rerio) Chemosphere. 2018;194:30–41. doi: 10.1016/j.chemosphere.2017.11.128. [DOI] [PubMed] [Google Scholar]
- 49.Wu S., Li X., Liu X., Yang G., An X., Wang Q., Wang Y. Joint toxic effects of triazophos and imidacloprid on zebrafish (Danio rerio) Environ. Pollut. 2018;235:470–481. doi: 10.1016/j.envpol.2017.12.120. [DOI] [PubMed] [Google Scholar]
- 50.Ku T., Yan W., Jia W., Yun Y., Zhu N., Li G., Sang N. Characterization of synergistic embryotoxicity of nickel and buprofezin in zebrafish. Environ. Sci. Technol. 2015;49:4600–4608. doi: 10.1021/es506293t. [DOI] [PubMed] [Google Scholar]
- 51.Wang Y., Wu S., Chen J., Zhang C., Xu Z., Li G., Cai L., Shen W., Wang Q. Single and joint toxicity assessment of four currently used pesticides to zebrafish (Danio rerio) using traditional and molecular endpoints. Chemosphere. 2018;192:14–23. doi: 10.1016/j.chemosphere.2017.10.129. [DOI] [PubMed] [Google Scholar]
- 52.Jijie R., Mihalache G., Balmus I.-M., Strungaru S.-A., Baltag E.S., Ciobica A., Nicoara M., Faggio C. Zebrafish as a Screening Model to Study the Single and Joint Effects of Antibiotics. Pharmaceuticals. 2021;14:578. doi: 10.3390/ph14060578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Cahova J., Blahova J., Plhalova L., Svobodova Z., Faggio C. Do Single-Component and Mixtures Selected Organic UV Filters Induce Embryotoxic Effects in Zebrafish (Danio rerio)? Water. 2021;13:2203. doi: 10.3390/w13162203. [DOI] [Google Scholar]
- 54.Faria M., Prats E., Ramírez J.R.R., Bellot M., Bedrossiantz J., Pagano M., Valls A., Gomez-Canela C., Porta J.M., Mestres J. Androgenic activation, impairment of the monoaminergic system and altered behavior in zebrafish larvae exposed to environmental concentrations of fenitrothion. Sci. Total Environ. 2021;775:145671. doi: 10.1016/j.scitotenv.2021.145671. [DOI] [PubMed] [Google Scholar]
- 55.Plhalova L., Sehonova P., Blahova J., Doubkova V., Tichy F., Faggio C., Berankova P., Svobodova Z. Evaluation of tramadol hydrochloride toxicity to juvenile zebrafish—morphological, antioxidant and histological responses. Appl. Sci. 2020;10:2349. doi: 10.3390/app10072349. [DOI] [Google Scholar]
- 56.Blahova J., Cocilovo C., Plhalova L., Svobodova Z., Faggio C. Embryotoxicity of atrazine and its degradation products to early life stages of zebrafish (Danio rerio) Environ. Toxicol. Pharmacol. 2020;77:103370. doi: 10.1016/j.etap.2020.103370. [DOI] [PubMed] [Google Scholar]
- 57.Sehonova P., Tokanova N., Hodkovicova N., Kroupova H.K., Tumova J., Blahova J., Marsalek P., Plhalova L., Doubkova V., Dobsikova R. Oxidative stress induced by fluoroquinolone enrofloxacin in zebrafish (Danio rerio) can be ameliorated after a prolonged exposure. Environ. Toxicol. Pharmacol. 2019;67:87–93. doi: 10.1016/j.etap.2019.02.002. [DOI] [PubMed] [Google Scholar]
- 58.Plhalova L., Blahova J., Divisova L., Enevova V., Casuscelli di Tocco F., Faggio C., Tichy F., Vecerek V., Svobodova Z. The effects of subchronic exposure to NeemAzal T/S on zebrafish (Danio rerio) Chem. Ecol. 2018;34:199–210. doi: 10.1080/02757540.2017.1420176. [DOI] [Google Scholar]
- 59.Bartoskova M., Dobsikova R., Stancova V., Zivna D., Blahova J., Marsalek P., Zelnickova L., Bartos M., Di Tocco F.C., Faggio C. Evaluation of ibuprofen toxicity for zebrafish (Danio rerio) targeting on selected biomarkers of oxidative stress. Neuroendocrinol. Lett. 2013;34:102–108. [PubMed] [Google Scholar]
- 60.Aliko V., Mehmeti E., Qirjo M., Faggio C. ‘Drink and sleep like a fish’: Goldfish as a behavior model to study pharmaceutical effects in freshwater ecosystems. J. Biol. Res.-Boll. Della Soc. Ital. Biol. Sper. 2019;92:1. doi: 10.4081/jbr.2019.7939. [DOI] [Google Scholar]
- 61.Yalsuyi A.M., Vajargah M.F., Hajimoradloo A., Galangash M.M., Prokić M.D., Faggio C. Evaluation of Behavioral Changes and Tissue Damages in Common Carp (Cyprinus carpio) after Exposure to the Herbicide Glyphosate. Vet. Sci. 2021;8:218. doi: 10.3390/vetsci8100218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Cachat J., Stewart A., Utterback E., Hart P., Gaikwad S., Wong K., Kyzar E., Wu N., Kalueff A.V. Three-dimensional neurophenotyping of adult zebrafish behavior. PLoS ONE. 2011;6:e17597. doi: 10.1371/journal.pone.0017597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Zeng X., Du Z., Ding X., Jiang W. Protective effects of dietary flavonoids against pesticide-induced toxicity: A review. Trends Food Sci. Technol. 2021;109:271–279. doi: 10.1016/j.tifs.2021.01.046. [DOI] [Google Scholar]
- 64.Dale K., Rasinger J., Thorstensen K., Penglase S., Ellingsen S. Vitamin E reduces endosulfan-induced toxic effects on morphology and behavior in early development of zebrafish (Danio rerio) Food Chem. Toxicol. 2017;101:84–93. doi: 10.1016/j.fct.2017.01.004. [DOI] [PubMed] [Google Scholar]
- 65.Li S., Cao C., Shi H., Yang S., Qi L., Zhao X., Sun C. Effect of quercetin against mixture of four organophosphate pesticides induced nephrotoxicity in rats. Xenobiotica. 2016;46:225–233. doi: 10.3109/00498254.2015.1070443. [DOI] [PubMed] [Google Scholar]
- 66.Puty B., Maximino C., Brasil A., da Silva W.L.L., Gouveia Jr A., Oliveira K.R.M., Batista E.D.J.O., Crespo-Lopez M.E., Rocha F.A.F., Herculano A.M. Ascorbic acid protects against anxiogenic-like effect induced by methylmercury in zebrafish: Action on the serotonergic system. Zebrafish. 2014;11:365–370. doi: 10.1089/zeb.2013.0947. [DOI] [PubMed] [Google Scholar]
- 67.Robea M.A., Jijie R., Nicoara M., Plavan G., Ciobica A.S., Solcan C., Audira G., Hsiao C.-D., Strungaru S.-A. Vitamin C Attenuates Oxidative Stress and Behavioral Abnormalities Triggered by Fipronil and Pyriproxyfen Insecticide Chronic Exposure on Zebrafish Juvenile. Antioxidants. 2020;9:944. doi: 10.3390/antiox9100944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Francis S., Delgoda R., Young R. Effects of embryonic exposure to α-lipoic acid or ascorbic acid on hatching rate and development of zebrafish (Danio rerio) Aquac. Res. 2012;43:777–788. doi: 10.1111/j.1365-2109.2011.02889.x. [DOI] [Google Scholar]
- 69.Xiang Q., Xu B., Ding Y., Liu X., Zhou Y., Ahmad F. Oxidative stress response induced by butachlor in zebrafish Embryo/Larvae: The protective effect of vitamin C. Bull. Environ. Contam. Toxicol. 2018;100:208–215. doi: 10.1007/s00128-017-2245-9. [DOI] [PubMed] [Google Scholar]
- 70.Wu C.Y., Lee H.J., Liu C.F., Korivi M., Chen H.H., Chan M.H. Protective role of L-ascorbic acid, N-acetylcysteine and apocynin on neomycin-induced hair cell loss in zebrafish. J. Appl. Toxicol. 2015;35:273–279. doi: 10.1002/jat.3043. [DOI] [PubMed] [Google Scholar]
- 71.Özkan F., Gündüz S.G., Berköz M., Hunt A.Ö., Yalın S. The protective role of ascorbic acid (vitamin C) against chlorpyrifos-induced oxidative stress in Oreochromis niloticus. Fish Physiol. Biochem. 2012;38:635–643. doi: 10.1007/s10695-011-9544-6. [DOI] [PubMed] [Google Scholar]
- 72.Liu D., Gu Y., Yu H. Vitamin C regulates the production of reactive oxygen species through Wnt10b signaling in the gill of zebrafish. Fish Physiol. Biochem. 2021;47:1271–1282. doi: 10.1007/s10695-021-00982-1. [DOI] [PubMed] [Google Scholar]
- 73.Datta M., Kaviraj A. Ascorbic acid supplementation of diet for reduction of deltamethrin induced stress in freshwater catfish Clarias gariepinus. Chemosphere. 2003;53:883–888. doi: 10.1016/S0045-6535(03)00557-5. [DOI] [PubMed] [Google Scholar]
- 74.Korkmaz N., Cengiz E., Unlu E., Uysal E., Yanar M. Cypermethrin-induced histopathological and biochemical changes in Nile tilapia (Oreochromis niloticus), and the protective and recuperative effect of ascorbic acid. Environ. Toxicol. Pharmacol. 2009;28:198–205. doi: 10.1016/j.etap.2009.04.004. [DOI] [PubMed] [Google Scholar]
- 75.Rodríguez-Fuentes G., Rubio-Escalante F.J., Noreña-Barroso E., Escalante-Herrera K.S., Schlenk D. Impacts of oxidative stress on acetylcholinesterase transcription, and activity in embryos of zebrafish (Danio rerio) following Chlorpyrifos exposure. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2015;172:19–25. doi: 10.1016/j.cbpc.2015.04.003. [DOI] [PubMed] [Google Scholar]
- 76.Burbano L.D.A., Porfiri M. Modeling Zebrafish Geotaxis as a Feedback Control Process; Proceedings of the 2021 American Control Conference (ACC); New Orleans, LA, USA. 25–28 May 2021; pp. 660–665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Egan R.J., Bergner C.L., Hart P.C., Cachat J.M., Canavello P.R., Elegante M.F., Elkhayat S.I., Bartels B.K., Tien A.K., Tien D.H. Understanding behavioral and physiological phenotypes of stress and anxiety in zebrafish. Behav. Brain Res. 2009;205:38–44. doi: 10.1016/j.bbr.2009.06.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Cachat J., Kyzar E.J., Collins C., Gaikwad S., Green J., Roth A., El-Ounsi M., Davis A., Pham M., Landsman S. Unique and potent effects of acute ibogaine on zebrafish: The developing utility of novel aquatic models for hallucinogenic drug research. Behav. Brain Res. 2013;236:258–269. doi: 10.1016/j.bbr.2012.08.041. [DOI] [PubMed] [Google Scholar]
- 79.Sarasamma S., Audira G., Juniardi S., Sampurna B.P., Liang S.-T., Hao E., Lai Y.-H., Hsiao C.-D. Zinc chloride exposure inhibits brain acetylcholine levels, produces neurotoxic signatures, and diminishes memory and motor activities in adult zebrafish. Int. J. Mol. Sci. 2018;19:3195. doi: 10.3390/ijms19103195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lazado C.C., Voldvik V., Breiland M.W., Osório J., Hansen M.H., Krasnov A. Oxidative Chemical Stressors Alter the Physiological State of the Nasal Olfactory Mucosa of Atlantic Salmon. Antioxidants. 2020;9:1144. doi: 10.3390/antiox9111144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Nunes M.E.M., Müller T.E., Murussi C., do Amaral A.M.B., Gomes J.L.C., Marins A.T., Leitemperger J., Rodrigues C.C.R., Fiuza T.L., Costa M.D., et al. Oxidative effects of the acute exposure to a pesticide mixture of cypermethrin and chlorpyrifos on carp and zebrafish–A comparative study. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2018;206–207:48–53. doi: 10.1016/j.cbpc.2018.03.002. [DOI] [PubMed] [Google Scholar]
- 82.Pan H., Zhang X., Ren B., Yang H., Ren Z., Wang W. Toxic assessment of cadmium based on online swimming behavior and the continuous AChE activity in the gill of zebrafish (Danio rerio) Water Air Soil Pollut. 2017;228:355. doi: 10.1007/s11270-017-3540-0. [DOI] [Google Scholar]
- 83.Chebbi S.G., David M. Neurobehavioral responses of the freshwater teleost, Cyprinus carpio (Linnaeus.) under quinalphos intoxication. Biotechnol. Anim. Husb. 2009;25:241–249. doi: 10.2298/BAH0904241C. [DOI] [Google Scholar]
- 84.Nema S., Bhargava Y. Quantitative assessment of cypermethrin induced behavioural and biochemical anomalies in adult zebrafish. Neurotoxicol. Teratol. 2018;68:57–65. doi: 10.1016/j.ntt.2018.05.003. [DOI] [PubMed] [Google Scholar]
- 85.DeMicco A., Cooper K.R., Richardson J.R., White L.A. Developmental neurotoxicity of pyrethroid insecticides in zebrafish embryos. Toxicol. Sci. 2010;113:177–186. doi: 10.1093/toxsci/kfp258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Komjarova I., Bury N. Evidence of common cadmium and copper uptake routes in zebrafish Danio rerio. Environ. Sci. Technol. 2014;48:12946–12951. doi: 10.1021/es5032272. [DOI] [PubMed] [Google Scholar]
- 87.Valentim A.M., van Eeden F.J., Strähle U., Olsson I.A.S. Euthanizing zebrafish legally in Europe: Are the approved methods of euthanizing zebrafish appropriate to research reality and animal welfare? EMBO Rep. 2016;17:1688–1689. doi: 10.15252/embr.201643153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Richetti S.K., Rosemberg D.B., Ventura-Lima J., Monserrat J.M., Bogo M.R., Bonan C.D. Acetylcholinesterase activity and antioxidant capacity of zebrafish brain is altered by heavy metal exposure. NeuroToxicol. 2011;32:116–122. doi: 10.1016/j.neuro.2010.11.001. [DOI] [PubMed] [Google Scholar]
- 89.Bradford M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- 90.Phelps S., Harris W.S. Garlic supplementation and lipoprotein oxidation susceptibility. Lipids. 1993;28:475–477. doi: 10.1007/BF02535949. [DOI] [PubMed] [Google Scholar]
- 91.Plavan G., Jitar O., Teodosiu C., Nicoara M., Micu D., Strungaru S.-A. Toxic metals in tissues of fishes from the Black Sea and associated human health risk exposure. Environ. Sci. Pollut. Res. 2017;24:7776–7787. doi: 10.1007/s11356-017-8442-6. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.