Abstract
Symptoms and complications associated with severe SARS‐CoV‐2 infection such as acute respiratory distress syndrome (ARDS) and organ damage have been linked to SARS‐CoV‐2 spike protein S1‐induced increased production of pro‐inflammatory cytokines by immune cells. In this study, the effects of an extract of Garcinia kola seeds and garcinoic acid were investigated in SARS‐CoV‐2 spike protein S1‐stimulated human PBMCs. Results of ELISA experiments revealed that Garcinia kola extract (6.25, 12.5, and 25 μg/ml) and garcinoic acid (1.25, 2.5, and 5 μM) significantly reduced SARS‐CoV‐2 spike protein S1‐induced secretion of TNFα, IL‐6, IL‐1β, and IL‐8 in PBMCs. In‐cell western assays showed that pre‐treatment with Garcinia kola extract and garcinoic acid reduced expressions of both phospho‐p65 and phospho‐IκBα proteins, as well as NF‐κB DNA binding capacity and NF‐κB‐driven luciferase expression following stimulation of PBMCs with spike protein S1. Furthermore, pre‐treatment of PBMCs with Garcinia kola extract prior to stimulation with SARS‐CoV‐2 spike protein S1 resulted in reduced damage to adjacent A549 lung epithelial cells. These results suggest that the seed of Garcinia kola and garcinoic acid are natural products which may possess pharmacological/therapeutic benefits in reducing cytokine storm in severe SARS‐CoV‐2 and other coronavirus infections.
Keywords: cytokine storm, Garcinia kola, garcinoic acid, hPBMCs, SARS‐CoV‐2 spike protein S1
1. INTRODUCTION
Since the first report of the emergence of the severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2), there has been a global spread of the infection accompanied by widespread appearance of coronavirus disease 2019 (COVID‐19) (Zhou et al., 2020; Zhu et al., 2020). Among the symptoms and complications associated with SARS‐CoV‐2 infection, acute respiratory distress syndrome (ARDS) and organ damage have been strongly linked to disease severity and death (Moradian et al., 2020).
ARDS and multi‐organ damage in SARS‐CoV‐2 infection have been linked to SARS‐CoV‐2 cytokine storm and the accompanying exaggerated release of inflammatory cytokines (Hirawat, Saifi, & Godugu, 2021; Lee & Choi, 2021; Yang et al., 2020). In SARS‐CoV‐2 infection, cytokine storm involves a vicious cycle of inflammation involving excessive release of cytokines such as interleukin‐1 (IL‐1), IL‐6, IL‐12, and tumour necrosis factor (TNF‐α), which damage the lungs and other organs (Hirawat et al., 2021; Huang et al., 2020). A review by Roshanravan, Seif, Ostadrahimi, Pouraghaei, and Ghaffari (2020) suggested the roles of IL‐2, IL‐7, interferon‐gamma (IFN‐γ), granulocyte‐colony stimulating factor (GCSF), interferon γ‐induced protein 10 kDa (IP‐10), monocyte chemo‐attractant protein 1 (MCP1), macrophage inflammatory proteins (MIP) in SARS‐CoV‐2 cytokine storm.
SARS‐CoV‐2 viral attachment and fusion to the host's cells are facilitated by spike glycoproteins which protrude from the surface through binding to the host ACE2 receptor (Freeman & Swartz, 2020; Merad & Martin, 2020). Interestingly, studies have shown that the spike glycoproteins, especially the S1 sub‐unit are also a target for the host immune responses (Merad & Martin, 2020). Recent studies have confirmed that the S1 sub‐unit induced inflammation in immune cells through activation of cellular inflammatory signalling pathways (Olajide, Iwuanyanwu, Lepiarz‐Raba, & Hindawi, 2021; Shirato & Kizaki, 2021).
Natural products from plants have been widely suggested as potential sources of pharmacological substances for combating different components of COVID‐19 pathogenesis. A molecular docking and cell‐based experiments reported by Kumar et al. (2020) revealed that withanone and withaferin‐A from Withania sominifera have the potential to interact with transmembrane protease serine 2 (TMPRSS2) and block entry of SARS‐CoV‐2 into cells. Also, the flavonoid naringenin was reported to be effective in inhibiting human coronaviruses infection in Vero E6 cells through targeting of the endo‐lysosomal two‐pore channels (Clementi et al., 2021). Furthermore, results of a study by Sagar et al. (2021) showed that bromelain (a cysteine protease found in the pineapple stem) inhibited SARS‐CoV‐2 infection via targeting ACE‐2, TMPRSS2, and SARS‐CoV‐2 S‐protein. In another study, an extract of Scutellaria barbata was shown to inhibit the enzyme activities of Mpro and TMPRSS2, as well as inhibiting SARS‐CoV‐2 infection through a TMPRSS2‐dependent mechanism (Huang et al., 2021).
However, there are currently no scientific reports on natural products which reduce cytokine storm in COVID‐19.
The edible seed of the West African plant Garcinia kola (bitter kola) (Figure 1) has been widely reported to have a wide range of medicinal benefits. Extracts from Garcinia kola have been reported to produce antiinflammatory activity in a number of cellular and animal models (Awogbindin, Olaleye, & Farombi, 2015; Farombi, Shrotriya, & Surh, 2009; Olaleye, Onasanwo, Ige, Wu, & Cho, 2010; Onasanwo, Velagapudi, El‐Bakoush, & Olajide, 2016). Garcinia kola has also been shown to produce anti‐thrombotic activity against adrenaline/ADP‐induced thrombosis in mice (Olajide, 1999). Garcinoic acid (Figure 1B) is an analogue of vitamin E, which has been isolated from Garcinia kola seeds (Mazzini, Betti, Netscher, Galli, & Salvadori, 2009; Wallert et al., 2019). Garcinoic acid was reported to produce antiinflammatory effects through inhibition of cyclo‐oxygenase 2 (COX‐2) and inducible nitric oxide synthase (iNOS) expressions in lipopolysaccharide (LPS)‐stimulated RAW264.7 cells (Wallert et al., 2019), as well as in other cell‐free and cell‐based assays (Alsabil et al., 2016; Dinh et al., 2020).
FIGURE 1.
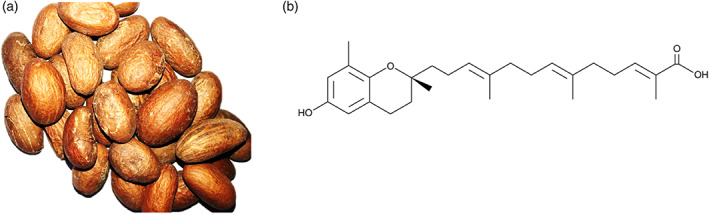
(a) Garcinia kola seeds. (b) Structure of garcinoic acid
Considering the critical role of cytokine storm in the pathogenesis of severe COVID‐19, it is important to investigate natural products that could reduce exaggerated and organ damaging‐inflammation of the disease. Consequently, this study was aimed at investigating the effects of an extract of Garcinia kola and one of its bioactive components (garcinoic acid) on SARS‐CoV‐2 spike protein S1‐induced hyper‐inflammation in PBMCs.
2. MATERIALS AND METHODS
2.1. Materials
Recombinant human coronavirus SARS‐CoV‐2 spike glycoprotein S1 (ab273068; Lots GR3356031‐1 and 3353172‐2; Accession MN908947) was purchased from Abcam (UK). The protein was suspended in sterile water for functional studies. Garcinoic acid, dexamethasone and BAY11‐7082 were purchased from Sigma (UK).
2.2. Extraction of Garcinia kola
Garcinia kola seeds were purchased from a local herbal market in Ibadan, Nigeria and were authenticated in the herbarium of the Forestry Research Institute of Nigeria, Ibadan (Voucher Number FH1‐113029). Dried seeds of Garcinia kola (10 g) were reduced to a fine powder in a blender and were exhaustively extracted with ethanol (300 ml). The liquid extract was filtered and dried in a petri dish at room temperature to yield the crude extract (2.6 g). This was transferred to a glass container and stored at 4°C. The extract was suspended in dimethyl sulphoxide (DMSO) for pharmacological studies. In all cases, the final concentration of DMSO in cell culture medium was 0.2%. DMSO (0.2%) was also used as control treatment in all experiments.
These studies were carried out in accordance with the recent guidelines for best practice in natural products pharmacological research (Heinrich et al., 2018; Izzo et al., 2020).
2.3. Analysis of the extract of Garcinia kola
Confirmation of the presence of garcinoic acid as a major component in the extract used in the assays was achieved by Gas Chromatography–Mass Spectrometry (GCMS) using pertrimethylsilyation using Pierce Tri‐Sil with a characteristic molecular ion seen 571 amu (60%) (2× tms groups) with fragmentation showing also 209 amu (100%). The extract was also analysed by HPLC‐PDA using a HiChrom ACE C18 column (250 mm × 4.6 mm id × 3.5 um) with a flow rate of 1 ml/min. The linear gradient started at 90% water and 10% acetonitrile (containing 0.01% TFA) held for 4 min, rising to 100% acetonitrile over 16 minutes and held for a further 5 min. Garcinoic acid was observed at a retention time of 8.42 min. With UV maxima at 203 and 299 nm (Data S1). Partial purification revealed that the garcinoic acid content of the G. kola extract was approximately 25%.
2.4. Cell culture
Human peripheral blood mononuclear cells (hPBMCs) (Lonza Biosciences; Catalogue #: 4W‐270; Batch: 3038013) were isolated from peripheral blood by apheresis and density gradient separation. Frozen cells were thawed, and transferred to a sterile centrifuge tube. Thereafter, warmed RPMI medium was added to the cells slowly, allowing gentle mixing. The cell suspension was then centrifuged at 400× g for 10 min. After centrifugation, the supernatant was discarded and fresh warmed RPMI was added to the pellet. This was followed by another centrifugation at 400× g for 10 min. Supernatant was removed and cells were suspended in RPMI, counted and allowed to rest overnight. Culture plates were coated with 0.01% poly‐l‐lysine (Sigma) to enhance the attachment of PBMCs.
2.5. Cell viability experiments
The CellTiter 96 AQueous One Solution Cell Proliferation Assay (Promega) is a colorimetric method for determining the number of viable cells in cytotoxicity experiments. The reagent contains a tetrazolium compound [3‐(4,5‐dimethylthiazol‐2‐yl)‐5‐(3‐carboxymethoxyphenyl)‐2‐(4‐sulphophenyl)‐2H‐tetrazolium, inner salt; MTS] and an electron coupling reagent (phenazine ethosulphate; PES).
This assay was used to assess the viability of PBMCs following treatment with either G. kola extract (6.25, 12.5 and 25 μg/ml) or garcinoic acid (1.25, 2.5 and 5 μM), followed by stimulation with spike protein S1 (100 ng/ml) for 24 hr. At the end of the experiment, 20 μl of CellTiter 96® AQueous One solution was added to cells in a 96‐well plate containing 100 μl culture medium, and incubated at 37°C for 2 hr. Absorbance was read at 490 nm in a Tecan Infinite M Nano microplate reader. In all subsequent pharmacological experiments 6.25, 12.5 and 25 μg/ml of Garcinia kola extract and 1.25, 2.5 and 5 μM of garcinoic acid were used.
2.6. Production of pro‐inflammatory cytokines
Human PBMCs were seeded out in 24‐well plate at 5 × 104 cells/ml and treated with G. kola extract or garcinoic acid for 1 hr prior to stimulation with spike protein S1 (100 ng/ml) for a further 24 hr. Dexamethasone (100 nM) was used as a reference drug. Thereafter, medium was collected and centrifuged to obtain culture supernatants. Levels of TNFα in the supernatants were determined using human ELISA™ kit (R and D Systems). Concentrations of TNFα in supernatants were calculated from a mouse TNFα standard curve. Levels of IL‐6 in supernatants were determined using human IL‐6 ELISA kit (R and D Sytems). Similarly, IL‐8 and IL‐1β production were evaluated using human IL‐8 and IL‐1β ELISA kits (R and D Systems). Absorbance measurements were carried out in a Tecan Infinite M Nano microplate reader.
2.7. In cell western (cytoblot) analyses
PBMCs were seeded into a 96‐well plate at 5 × 104 cells/ml and allowed to settle overnight. Cells were then treated with either G. kola extract or garcinoic acid and incubated for 1 hr prior to stimulation with spike protein S1 (100 ng/ml) for a further 15 min. At the end of each experiment, cells were fixed with 8% paraformaldehyde solution (100 μl) for 15 min., followed by washing with PBS. The cells were then incubated with primary antibodies overnight at 4°C. The following antibodies were used: rabbit anti‐phospho‐p65 (Cell Signalling Technology), rabbit anti‐IκBα (Cell Signalling Technology), and rabbit anti‐phospho‐IκBα (Santa Cruz Biotechnology) antibodies. Thereafter, cells were washed with PBS and incubated with anti‐rabbit HRP secondary antibody for 2 hr at room temperature. Then, 100 μl HRP substrate was added to each well and absorbance measured at 450 nm with a Tecan Infinite M Nano microplate reader. Readings were normalised with Janus Green normalisation stain (Abcam).
2.8. NF‐κB p65 transcription factor binding assay
The NF‐κB p65 transcription factor assay is a non‐radioactive ELISA‐based assay for evaluating DNA binding activity of NF‐κB in nuclear extracts. PBMCs were seeded in a 6‐well plate at a density of 5 × 104 cells/ml. The cells were then incubated with G. kola extract, garcinoic acid, dexamethasone (100 nM) or BAY11‐7082 (1 μM) for 1 hr, followed by stimulation with spike protein S1 (100 ng/ml) for another 1 hr. At the end of the incubation, nuclear extracts were prepared from the cells and subjected to NF‐κB transcription factor binding assay according to the instructions of the manufacturer (Abcam).
2.9. Transient transfection and NF‐κB reporter gene assay
Transfection of PBMCs was performed using magnetofection, a method used for transfecting primary and hard‐to‐transfect cells. The cells were seeded in 24‐well plate at a density of 4 × 104 cells/ml and allowed to settle overnight. Thereafter, culture media were replaced with Opti‐MEM, with a further incubation for 2 hr at 37°C. Transfection was performed by preparing a magnetofectamine O2 transfection reagent (OZ Biosciences) and Cignal NF‐κB luciferase reporter (Qiagen) complex at a ratio 3:1 in 50 μl Opti‐MEM. The complex was added to the 24‐well plate, and the plate placed on a magnetic plate (Oz Biosciences) and incubated at 37°C for 30 min, followed by a further magnetic plate‐free incubation for 18 hr. Thereafter, medium was changed to culture media and cells treated with G. kola extract, garcinoic acid, dexamethasone (100 nM) or BAY11‐7082 (1 μM) for 1 hr prior to stimulation with spike protein S1 (100 ng/ml) for a further 3 hr. This was followed by a Dual‐Glo® reporter assay (Promega). Firefly and renilla luminescence intensities were measured using a FLUOstar OPTIMA microplate reader (BMG Labtech).
2.10. Co‐culture of PBMCs with human A549 lung epithelial cells
Human A549 lung epithelial cells were co‐cultured with PBMCs using the transwell system (0.4 μm porous membrane; Corning). Overnight‐rested PBMCs cells were cultured in inserts that constituted the upper chamber. Thereafter, they were pre‐treated with G. kola, garcinoic acid or dexamethasone (100 nM) for 1 hr, and then incubated with spike protein S1 (100 ng/ml). One hour after the initiation of stimulation with spike protein S1, the inserts were placed on A549 cells in the lower chamber for a further 48 hr. At the end of the experiment, supernatants were collected from the PBMC layer and analysed for levels of TNFα and IL‐6 using human ELISA kits as described. Viability of A549 cells were also determined using the LDH assay. Briefly, 50 μl of supernatants from the A549 layer were transferred into a 96‐well plate, followed by the addition of 50 μl of CytoTox 96® reagent (Promega) and incubation in the dark for 30 min at room temperature. The reaction was stopped with 50 μl stop solution and absorbance read at 490 nm.
2.11. Statistical analysis
Data are expressed as mean ± SEM for at least three independent experiments (n = 3) and analysed using one‐way analysis of variance (ANOVA) with post hoc Dunnett's multiple comparison test. Statistical analysis was conducted using the GraphPad Prism software.
3. RESULTS
3.1. Treatment of spike protein S1‐stimulated PBMCs with extract of Garcinia kola and garcinoic acid did not reduce the viability
PBMCs were incubated with either Garcinia kola extract (6.25, 12.5 and 25 μg/ml) or garcinoic acid (1.25, 2.5 and 5 μM) for 60 min and then stimulated with spike protein S1 (100 μg/ml) for a further 24 hr. Results of MTS viability assays revealed that these treatments did not produce significant reduction in cell viability, in comparison with untreated cells (Figure 2a,b).
FIGURE 2.
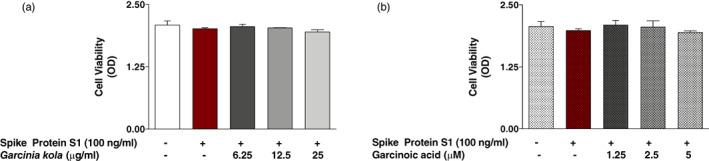
(a) MTS assay showing effects of Garcinia kola extract (6.25, 12.5 and 25 μg/ml) on the viability of SARS‐CoV‐2 spike protein S1‐stimulated PBMCs. Control cells were treated with DMSO (0.2%). (b) MTS assay showing effects of garcinoic acid (1.25, 2.5 and 5 μM) on the viability of SARS‐CoV‐2 spike protein S1‐stimulated PBMCs. Control cells were treated with DMSO (0.2%)
3.2. Garcinia kola extract reduced spike protein S1‐induced increase in cytokine production
Stimulation of PBMCs with spike protein S1 (100 μg/ml) for 24 hr resulted in significant (p < .001) increase in the release of TNFα into culture supernatants, when compared with unstimulated cells. However, pre‐treatment with G. kola (6.25, 12.5 and 25 μg/ml) for 60 min prior to spike protein S1 stimulation resulted in significant (p < .05) and concentration‐dependent reduction in TNFα production (Figure 3a). The highest concentration of G. kola investigated (25 μg/ml) reduced spike protein S1‐induced increased TNFα production by ~47%, while dexamethasone (100 nM) treatment caused a reduction by ~75% (Figure 3a). Similarly, spike protein S1‐induced increased release of IL‐6 was significantly reduced (p < .01) when cells were pre‐treated with G. kola (12.5 and 25 μg/ml), and dexamethasone (Figure 3b). Analyses of culture supernatants further revealed that spike protein S1 stimulation of PBMCs resulted in significant elevation of IL‐1β (Figure 3c) and IL‐8 (Figure 3d), which were reduced in the presence of G. kola extract (6.25–25 μg/ml).
FIGURE 3.
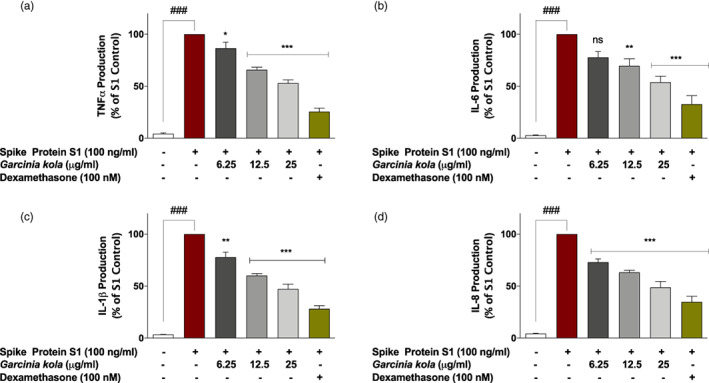
Pre‐treatment of SARS‐CoV‐2 spike protein S1‐stimulated PBMCs with Garcinia kola extract (6.25, 12.5 and 25 μg/ml) and dexamethasone (100 nM) reduced increased production of TNFα (a), IL‐6 (b), IL‐1β (c), and IL‐8 (d). Levels of cytokines in cell supernatants were detected using human TNFα, IL‐6, IL‐1β, and IL‐8 ELISA kits. Values are mean ± SEM for at least 3 independent experiments (ns: not significant; ### p < .001 unstimulated control versus SARS‐CoV‐2 spike protein S1 stimulation. *p < .05; **p < .01; ***p < .001, treatments versus SARS‐CoV‐2 spike protein S1 stimulation; one‐way ANOVA with post‐hoc Dunnett's multiple comparison test)
3.3. Effects of garcinoic acid pre‐treatment on spike protein S1‐induced increased production of pro‐inflammatory cytokines
Experiments on garcinoic acid showed that spike protein S1‐induced increased production of TNFα was significantly reduced by ~22%, 35% and 53% in cells pre‐treated with 1.25, 2.5 and 5 μM of the compound, respectively (Figure 4a). S1‐induced increased production of IL‐6, IL‐1β and IL‐8 were also significantly (p < .01) reduced when cells were pre‐treated with 1.25, 2.5 and 5 μM of garcinoic acid (Figure 4b,c,d). At 5 μM, garcinoic acid reduced the increased production of IL‐6, IL‐1β and IL‐8 by ~45%, ~53% and ~52%, respectively. However, dexamethasone (100 nM) reduced their levels by ~67%, ~75% and ~70%, respectively.
FIGURE 4.
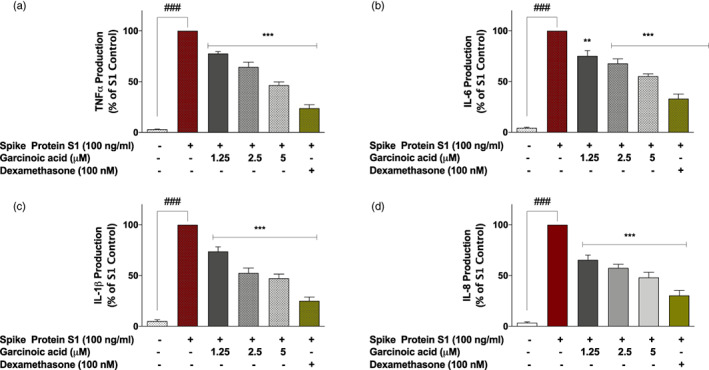
Pre‐treatment of SARS‐CoV‐2 spike protein S1‐stimulated PBMCs with garcinoic acid (1.25, 2.5, and 5 μM) and dexamethasone (100 nM) reduced increased production of TNFα (a), IL‐6 (b), IL‐1β (c), and IL‐8 (d). Levels of cytokines in cell supernatants were detected using human TNFα, IL‐6, IL‐1β, and IL‐8 ELISA kits. Values are mean ± SEM for at least 3 independent experiments (### p < .001 unstimulated control versus SARS‐CoV‐2 spike protein S1 stimulation. **p < .01; ***p < .001, treatments versus SARS‐CoV‐2 spike protein S1 stimulation; one‐way ANOVA with post‐hoc Dunnett's multiple comparison test)
3.4. Garcinia kola extract and garcinoic acid inhibit spike protein S1‐induced inflammation by targeting NF‐κB activation
Encouraged by results showing reduction in spike protein S1‐induced increase in pro‐inflammatory cytokine production by both G. kola extract and garcinoic acid, experiments were conducted to determine whether inhibition of NF‐κB activation contributed to their effects. Stimulation of PBMCs with (100 ng/ml) spike protein S1 resulted in ~8.8‐fold and ~8.2‐fold increases in protein expressions of phoshpho‐p65, and phospho‐IκBα, respectively (Figure 5a,b). However, pre‐treating the cells with G. kola extract (6.25, 12.5 and 25 μg/ml) for 60 min prior to spike protein S1 stimulation resulted in significant (p < .01) reduction in protein levels of both phospho‐p65 and phospho‐IκBα (Figure 5a,b). Also, expressions of both proteins were significantly reduced in cells pre‐treated with dexamethasone (100 nM) and the NF‐κB inhibitor, BAY11‐7082 (1 μM), in comparison with cells stimulated with spike protein S1 alone.
FIGURE 5.
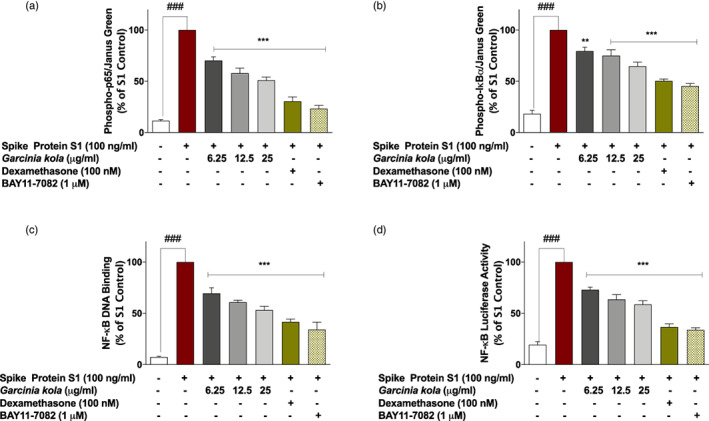
SARS‐CoV‐2 spike protein S1‐induced increased expression of phospho‐p65 (a), phospho‐IκBα (b) and NF‐κB p65 DNA binding (c) in PBMCs was inhibited in the presence of Garcinia kola extract (6.25, 12.5 and 25 μg/ml), dexamethasone (100 nM) and BAY11‐7082 (1 μM). Cells were stimulated for 15 min, followed by in cell western analysis with anti‐phospho‐p65 and anti‐phospho‐IκBα antibodies. DNA binding was evaluated using NF‐κB transcription factor binding assay kit. (d) Effects of Garcinia kola extract (6.25, 12.5 and 25 μg/ml), dexamethasone (100 nM) and BAY11‐7082 (1 μM) on NF‐κB‐driven luciferase activity in SARS‐CoV‐2 spike protein S1‐stimulated PBMCs. Values are mean ± SEM for at least 3 independent experiments (### p < .001 unstimulated control versus SARS‐CoV‐2 spike protein S1 stimulation. **p < .01; ***p < .001, treatments versus SARS‐CoV‐2 spike protein S1 stimulation; one‐way ANOVA with post‐hoc Dunnett's multiple comparison test)
Further experiments to evaluate effects of G. kola extract on spike protein S1‐induced increased activation of NF‐κB signalling revealed that in the presence of 6.25, 12.5 and 25 μg/ml of the extract, DNA binding capacity of p65 sub‐unit was reduced by ~31%, ~39% and ~47%, respectively when compared with S1‐stimulated PBMCs. Results also showed that spike protein S1‐induced DNA binding capacity was reduced by ~58% and ~ 66% when PBMCs were pre‐treated with dexamethasone (100 nM) and BAY11‐7082 (1 μM), respectively (Figure 5c). Results of reporter gene assays in Figure 5d show that stimulation of NF‐κB luciferase reporter transfected cells with spike protein S1 resulted in activation of NF‐κB‐driven luciferase expression, which was significantly (p < .001) reduced by G. kola extract (6.25, 12.5 and 25 μg/ml), dexamethasone (100 nM) and BAY11‐7082 (1 μM).
Investigations to determine the role of NF‐κB inhibition in the effects of garcinoic acid on spike protein S1‐induced hyper‐inflammation in PBMCs revealed that the compound produced significant (p < .01) inhibition of p65 and IκBα phosphorylation (Figure 6a,b). Experiments also showed that garcinoic acid (5 μM) inhibited spike protein S1‐induced NF‐κB DNA binding capacity and NF‐κB‐driven luciferase expression by ~51% and ~42%, respectively. Dexamethasone (100 nM) reduced these events by ~64% and ~61%, respectively (Figure 6c,d).
FIGURE 6.
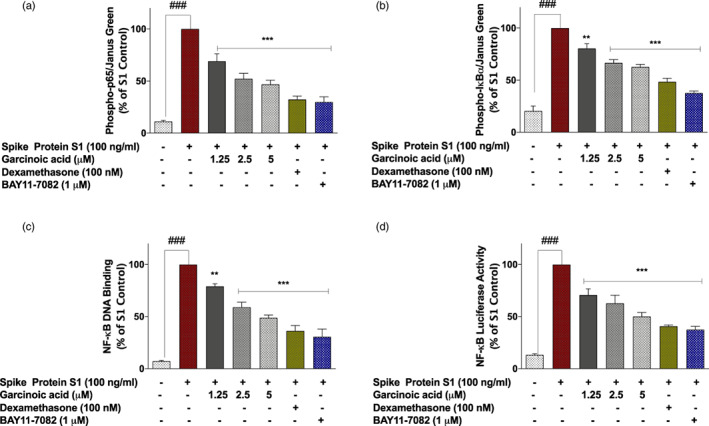
SARS‐CoV‐2 spike protein S1‐induced increased expression of phospho‐p65 (a), phospho‐IκBα (b) and NF‐κB p65 DNA binding (c) in PBMCs was inhibited in the presence of garcinoic acid (1.25, 2.5, and 5 μM), dexamethasone (100 nM) and BAY11‐7082 (1 μM). Cells were stimulated for 15 min, followed by in cell western analysis with anti‐phospho‐p65 and anti‐phospho‐IκBα antibodies. DNA binding was evaluated using NF‐κB transcription factor binding assay kit. (d) Effects of garcinoic acid (1.25, 2.5, and 5 μM), dexamethasone (100 nM) and BAY11‐7082 (1 μM) on NF‐κB‐driven luciferase activity in SARS‐CoV‐2 spike protein S1‐stimulated PBMCs. Values are mean ± SEM for at least 3 independent experiments (### p < .001 unstimulated control versus SARS‐CoV‐2 spike protein S1 stimulation. **p < .01; ***p < .001, treatments versus SARS‐CoV‐2 spike protein S1 stimulation; one‐way ANOVA with post‐hoc Dunnett's multiple comparison test)
3.5. Effects of Garcinia kola extract and garcinoic acid on the viability of A549 human lung alveolar epithelial cells co‐cultured with spike protein S1‐stimulated PBMCs
As shown in Figures 7a and 8a, stimulation of PBMCs with spike protein S1 (100 ng/ml) for 48 hr resulted in a significant (p < .001) increase in the release of LDH by adjacent A549 lung epithelial cells. This was accompanied by significant increases in the levels of both TNFα (Figures 7b and 8b) and IL‐6 (Figures 7c and 8c) production by the PBMCs. Pre‐treatment of PBMCs with 6.25 μg/ml of G. kola extract prior to stimulation with spike protein S1 did not result in significant (p < .05) reduction in cytotoxicity to co‐cultured A549 lung epithelial cells. However, on increasing the concentrations of G. kola extract to 12.5 and 25 μg/ml, significant (p < .05) reduction in damage to A549 cells was observed (Figure 7a). Interestingly, levels of TNFα and IL‐6 were significantly (p < .001) reduced in the presence of all concentrations of the extract tested (Figure 7b,c).
FIGURE 7.
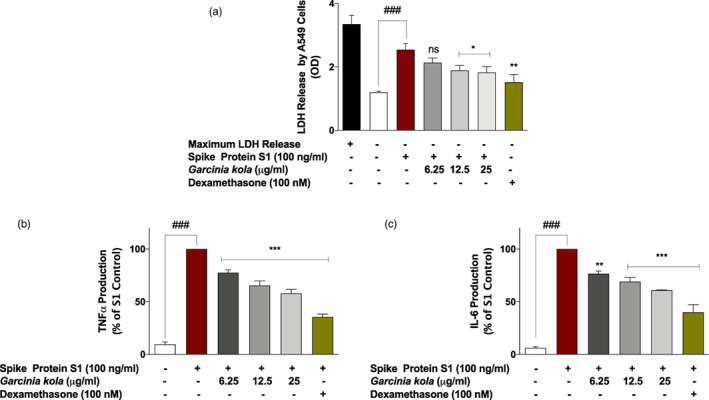
Garcinia kola extract (12.5 and 25 μg/ml) and dexamethasone (100 nM) prevented reduced viability of A549 lung epithelial cells co‐cultured with SARS‐CoV‐2 spike protein S1‐stimulated PBMCs (a), accompanied by reduction in TNFα and IL‐6 levels in the PBMC layer of the co‐culture (b and c). Values are mean ± SEM for at least 3 independent experiments (ns: not significant; ### p < .001 unstimulated control versus SARS‐CoV‐2 spike protein S1 stimulation. *p < .05; **p < .01; ***p < .001, treatments versus SARS‐CoV‐2 spike protein S1 stimulation; one‐way ANOVA with post‐hoc Dunnett's multiple comparison test)
FIGURE 8.
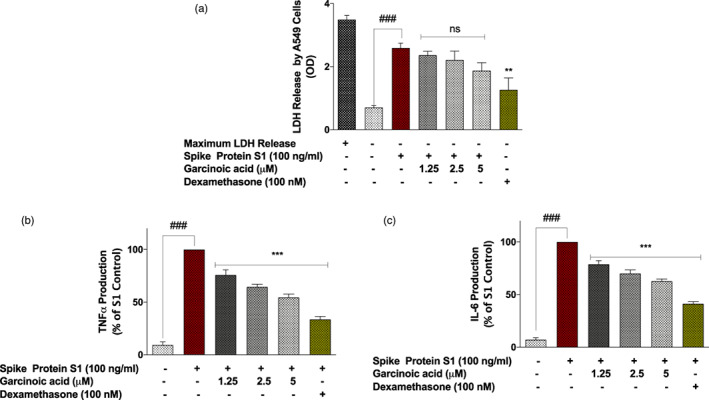
Dexamethasone (100 nM), but not garcinoic acid (1.25, 2.5, and 5 μM) significantly prevented reduced viability of A549 lung epithelial cells co‐cultured with SARS‐CoV‐2 spike protein S1‐stimulated PBMCs (a), accompanied by reduction in TNFα and IL‐6 levels in the PBMC layer of the co‐culture (b and c). Values are mean ± SEM for at least 3 independent experiments (ns: not significant; ### p < .001 unstimulated control versus SARS‐CoV‐2 spike protein S1 stimulation. **p < .01; ***p < .001, treatments versus SARS‐CoV‐2 spike protein S1 stimulation; one‐way ANOVA with post‐hoc Dunnett's multiple comparison test
Pre‐treatment of spike protein S1‐stimulated PBMCs with garcinoic acid (1.25, 2.5, and 5 μM) did not significantly (p < .05) prevent toxicity to adjacent A549 cells after 48 hr (Figure 8a). However, there were significant (p < .001) reductions in the levels of both TNFα and IL‐6 in the PBMC layer of the co‐culture in the presence of all the concentrations of the compound (Figure 8b,c).
4. DISCUSSION
Therapeutic strategies have been proposed for COVID‐19, the disease caused by infection with the SARS‐CoV‐2 virus. Among these are potential treatments for COVID‐19 cytokine storm (Tang et al., 2020). In this regard, antiinflammatory steroids such as dexamethasone have been shown to provide some benefits in the treatment of COVID‐19, based on results of RECOVERY and CoDEX trials (Ledford, 2020; RECOVERY Collaborative Group, 2021; Tomazini et al., 2020). Dexamethasone and other corticosteroids were also reported to provide benefits in previous coronavirus outbreaks, such as Middle East respiratory syndrome (MERS) and severe acute respiratory syndrome (SARS) (Arabi et al., 2018; Chen et al., 2006; Stockman, Bellamy, & Garner, 2006). The benefits of dexamethasone and other corticosteroids in the treatment of coronavirus infections are linked to their antiinflammatory activity in suppressing cytokine storms. Despite the potential benefits of dexamethasone in the treatment of severe COVID‐19 symptoms, concerns have been raised about enhancement of viral replication due to the immunosuppressive property of the drug. Therefore, the use of alternative antiinflammatory modalities, including natural products could provide an alternative therapeutic strategy in treating COVID‐19 cytokine storm.
This study showed that an extract from Garcinia kola seeds reduced the production of pro‐inflammatory cytokines TNFα, IL‐6, IL‐1β and IL‐8 in human PBMCs stimulated with SARS‐CoV‐2 spike protein S1. These are significant observations as clinical evidence has shown that the cytokine storm in COVID‐19 involves a vicious cycle of inflammatory responses which are characterised by excessive release of IL‐1β, IL‐6, TNFα, which then target lung and other tissues (Aziz, Fatima, & Assaly, 2020; Grifoni et al., 2020; Hirawat et al., 2021). The effects of Garcinia kola extract against SARS‐CoV‐2 spike protein S1‐induced increased production of pro‐inflammatory cytokines in PBMCs are consistent with previous reports of similar antiinflammatory activities in vivo and in vitro (Farombi et al., 2009; Olaleye et al., 2010; Onasanwo et al., 2016). Furthermore, a study reported by Awogbindin et al. (2015), shows that a seed extract of Garcinia kola attenuated inflammatory cell infiltration in a mouse model of influenza A virus.
Garcinoic acid is a δ‐tocotrienol derivative, and one of the major bioactive components in the seeds of Garcinia kola. Similar to activities shown by the seed extract of Garcinia kola, garcinoic acid significantly inhibited SARS‐CoV‐2 spike protein S1‐induced exaggerated production of TNFα, IL‐6, IL‐1β and IL‐8 in PBMCs. It therefore appears that garcinoic acid may be contributing to the antiinflammatory effects shown by Garcinia kola extract, as the compound was previously shown to produce antiinflammatory effects in LPS‐stimulated RAW264.7 cells (Wallert et al., 2019). Further phytochemical analyses are required to identify other bioactive components of Garcinia kola with similar pharmacological profiles.
Activation of the NF‐κB transcription factor signalling pathway has been linked to the pathogenesis of severe COVID‐19 (Hirano & Murakami, 2020). Furthermore, a review by Hariharan, Hakeem, Radhakrishnan, Reddy, and Rela (2021) suggests that inhibition of NF‐κB activation, with the resultant reduction in exaggerated production of pro‐inflammatory cytokines like TNFα will potentially result in a reduction in cytokine storm in severe COVID‐19. Our recent studies also showed that NF‐κB activation is a mechanism for SARS‐CoV‐2 spike protein S1‐induced exaggerated production of TNFα, IL‐6, IL‐1β and IL‐8 in PBMCs (Olajide et al., 2021). The seed extract of Garcinia kola and garcinoic acid inhibited both cytoplasmic activation, DNA binding and transcriptional activity of NF‐κB in SARS‐CoV‐2 spike protein S1‐simulated PBMCs, suggesting a role for the transcription factor in their antiinflammatory activities. The effects of Garcinia kola on NF‐κB activation in this study appear to be similar to previously observed activities in RAW264 macrophages and BV‐2 microglia stimulated with bacterial lipopolysaccharide (Abarikwu, 2014; Onasanwo et al., 2016). Similarly, inhibition of NF‐κB activation in LPS‐stimulated macrophages has been reported for garcinoic acid (Schmölz et al., 2014). Further investigations are required to identify specific molecular targets involved in NF‐κB‐mediated inhibition of SARS‐CoV‐2 spike protein S1‐induced inflammation by Garcinia kola and garcinoic acid.
Several studies have implicated COVID‐19 cytokine storm in lung damage and ARDS (Bahrami, Kamalinejad, Latifi, Seif, & Dadmehr, 2020; Seif et al., 2020; Wang et al., 2020). In this regard, SARS‐CoV‐2 is known to activate immune cells leading to the production of a large number of pro‐inflammatory cytokines which cause lung damage. Results of this study showed that Garcinia kola seed extract but not garcinoic acid prevented toxicity to A549 lung epithelial cells which were co‐cultured with SARS‐CoV‐2 spike protein S1‐stimulated PBMCs, suggesting that the antiinflammatory activity of Garcinia kola resulted in the prevention of cytokine‐mediated damage to A549 lung epithelial cells. It is proposed that the antiinsflammatory effects of Garcinia kola seed extract could be an important pharmacological attribute for the treatment of ARDS and lung damage in patients with severe COVID‐19.
Antiinflammatory strategies have shown promising results in the alleviation of ARDS and lung damage resulting from COVID‐19. Tocilizumab (a potential recombinant monoclonal antibody against IL‐6) is currently under investigation for the management of ARDS in patients with COVID‐19 (Khiali, Khani, & Entezari‐Maleki, 2020; Nasir, Mahmood, Habib, Khanum, & Jamil, 2020; Toniati et al., 2020). Similarly antiinflammatory drugs bazedoxifene and raloxifene, used in arthritis have been proposed as treatments for preventing cytokine storm, ARDS and mortality in severe COVID‐19 patients (Smetana Jr, Rosel, & BrÁbek, 2020).
While these results are based on the outcome of in vitro experiments on human PBMCs, they suggest that the seed of Garcinia kola and garcinoic acid are natural products which may possess pharmacological/therapeutic benefits in reducing cytokine storm during the late stage of severe SARS‐CoV‐2 and other coronavirus infections.
Supporting information
Data S1. Supplementary information.
ACKNOWLEDGEMENTS
We thank Tobi Olajide for providing technical assistance with the processing of Garcinia kola seeds.
Olajide, O. A. , Iwuanyanwu, V. U. , Lepiarz‐Raba, I. , Al‐Hindawi, A. A. , Aderogba, M. A. , Sharp, H. L. , & Nash, R. J. (2021). Garcinia kola and garcinoic acid suppress SARS‐CoV‐2 spike glycoprotein S1‐induced hyper‐inflammation in human PBMCs through inhibition of NF‐κB activation. Phytotherapy Research, 35(12), 6963–6973. 10.1002/ptr.7315
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- Abarikwu, S. O. (2014). Kolaviron, a natural flavonoid from the seeds of Garcinia kola, reduces LPS‐induced inflammation in macrophages by combined inhibition of IL‐6 secretion, and inflammatory transcription factors, ERK1/2, NF‐κB, p38, Akt, p‐c‐JUN and JNK. Biochimica et Biophysica Acta, 1840, 2373–2381. 10.1016/j.bbagen.2014.03.006 [DOI] [PubMed] [Google Scholar]
- Alsabil, K. , Suor‐Cherer, S. , Koeberle, A. , Viault, G. , Lavaud, A. , Temml, V. , … Richomme, P. (2016). Semisynthetic and natural garcinoic acid isoforms as new mPGES‐1 inhibitors. Planta Medica, 82, 1110–1116. 10.1055/s-0042-108739 [DOI] [PubMed] [Google Scholar]
- Arabi, Y. M. , Mandourah, Y. , Al‐Hameed, F. , Sindi, A. A. , Almekhlafi, G. A. , Hussein, M. A. , … Saudi Critical Care Trial Group . (2018). Corticosteroid therapy for critically ill patients with Middle East respiratory syndrome. American Journal of Respiratory and Critical Care Medicine, 197, 757–767. 10.1164/rccm.201706-1172OC [DOI] [PubMed] [Google Scholar]
- Awogbindin, I. O. , Olaleye, D. O. , & Farombi, E. O. (2015). Kolaviron improves morbidity and suppresses mortality by mitigating oxido‐inflammation in BALB/c mice infected with influenza virus. Viral Immunology, 28, 367–377. 10.1089/vim.2015.0013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aziz, M. , Fatima, R. , & Assaly, R. (2020). Elevated interleukin‐6 and severe COVID‐19: A meta‐analysis. Journal of Medical Virology, 92, 2283–2285. 10.1002/jmv.25948 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bahrami, M. , Kamalinejad, M. , Latifi, S. A. , Seif, F. , & Dadmehr, M. (2020). Cytokine storm in COVID‐19 and parthenolide: Preclinical evidence. Phytotherapy Research, 34, 2429–2430. 10.1002/ptr.6776 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen, R. C. , Tang, X. P. , Tan, S. Y. , Liang, B. L. , Wan, Z. Y. , Fang, J. Q. , & Zhong, N. (2006). Treatment of severe acute respiratory syndrome with glucosteroids: The Guangzhou experience. Chest, 129, 1441–1452. 10.1378/chest.129.6.1441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clementi, N. , Scagnolari, C. , D'Amore, A. , Palombi, F. , Criscuolo, E. , Frasca, F. , … Filippini, A. (2021). Naringenin is a powerful inhibitor of SARS‐CoV‐2 infection in vitro. Pharmacological Research, 163, 105255. 10.1016/j.phrs.2020.105255 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dinh, C. P. , Ville, A. , Neukirch, K. , Viault, G. , Temml, V. , Koeberle, A. , … Séraphin, D. (2020). Structure‐based design, semi‐synthesis and anti‐inflammatory activity of tocotrienolic amides as 5‐lipoxygenase inhibitors. European Journal of Medicinal Chemistry, 202, 112518. 10.1016/j.ejmech.2020.112518 [DOI] [PubMed] [Google Scholar]
- Farombi, E. O. , Shrotriya, S. , & Surh, Y. J. (2009). Kolaviron inhibits dimethyl nitrosamine‐induced liver injury by suppressing COX‐2 and iNOS expression via NF‐kappaB and AP‐1. Life Sciences, 84, 149–155. 10.1016/j.lfs.2008.11.012 [DOI] [PubMed] [Google Scholar]
- Freeman, T. L. , & Swartz, T. H. (2020). Targeting the NLRP3 inflammasome in severe COVID‐19. Frontiers in Immunology, 11, 1518. 10.3389/fimmu.2020.01518 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grifoni, E. , Valoriani, A. , Cei, F. , Lamanna, R. , Gelli, A. , Ciambotti, B. , … Masotti, L. (2020). Interleukin‐6 as prognosticator in patients with COVID‐19. The Journal of Infection, 81, 452–482. 10.1016/j.jinf.2020.06.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hariharan, A. , Hakeem, A. R. , Radhakrishnan, S. , Reddy, M. S. , & Rela, M. (2021). The role and therapeutic potential of NF‐kappa‐B pathway in severe COVID‐19 patients. Inflammopharmacology, 29, 91–100. 10.1007/s10787-020-00773-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heinrich, M. , Lardos, A. , Leonti, M. , Weckerle, C. , Willcox, M. , with the ConSEFS advisory group, Based on a consultative process of researchers active in ethnopharmacology and with particular input by the ConSEFS Advisory group , … Stafford, G. (2018). Best practice in research: Consensus statement on ethnopharmacological field studies – ConSEFS. Journal of Ethnopharmacology, 211, 329–339. 10.1016/j.jep.2017.08.015 [DOI] [PubMed] [Google Scholar]
- Hirano, T. , & Murakami, M. (2020). COVID‐19: A new virus, but a familiar receptor and cytokine release syndrome. Immunity, 52, 731–733. 10.1016/j.immuni.2020.04.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirawat, R. , Saifi, M. A. , & Godugu, C. (2021). Targeting inflammatory cytokine storm to fight against COVID‐19 associated severe complications. Life Sciences, 267, 118923. 10.1016/j.lfs.2020.118923 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang, C. , Wang, Y. , Li, X. , Ren, L. , Zhao, J. , Hu, Y. , … Cao, B. (2020). Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet (London, England), 395, 497–506. 10.1016/S0140-6736(20)30183-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang, S. T. , Chen, Y. , Chang, W. C. , Chen, H. F. , Lai, H. C. , Lin, Y. C. , … Hung, M. C. (2021). Scutellaria barbata D. Don inhibits the main proteases (Mpro and TMPRSS2) of severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) infection. Viruses, 13, 826. 10.3390/v13050826 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Izzo, A. A. , Teixeira, M. , Alexander, S. , Cirino, G. , Docherty, J. R. , George, C. H. , … Ahluwalia, A. (2020). A practical guide for transparent reporting of research on natural products in the British Journal of pharmacology: Reproducibility of natural product research. British Journal of Pharmacology, 177, 2169–2178. 10.1111/bph.15054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khiali, S. , Khani, E. , & Entezari‐Maleki, T. (2020). A comprehensive review of tocilizumab in COVID‐19 acute respiratory distress syndrome. Journal of Clinical Pharmacology, 60, 1131–1146. 10.1002/jcph.1693 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar, V. , Dhanjal, J. K. , Bhargava, P. , Kaul, A. , Wang, J. , Zhang, H. , … Sundar, D. (2020). Withanone and Withaferin‐a are predicted to interact with transmembrane protease serine 2 (TMPRSS2) and block entry of SARS‐CoV‐2 into cells. Journal of Biomolecular Structure & Dynamics, 1–13. 10.1080/07391102.2020.1775704 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ledford, H. (2020). Coronavirus breakthrough: Dexamethasone is first drug shown to save lives. Nature, 582, 469. 10.1038/d41586-020-01824-5 [DOI] [PubMed] [Google Scholar]
- Lee, C. , & Choi, W. J. (2021). Overview of COVID‐19 inflammatory pathogenesis from the therapeutic perspective. Archives of Pharmacal Research, 44, 99–116. 10.1007/s12272-020-01301-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazzini, F. , Betti, M. , Netscher, T. , Galli, F. , & Salvadori, P. (2009). Configuration of the vitamin E analogue garcinoic acid extracted from Garcinia Kola seeds. Chirality, 21, 519–524. 10.1002/chir.20630 [DOI] [PubMed] [Google Scholar]
- Merad, M. , & Martin, J. C. (2020). Pathological inflammation in patients with COVID‐19: A key role for monocytes and macrophages. Nature Reviews. Immunology, 20, 355–362. 10.1038/s41577-020-0331-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moradian, N. , Gouravani, M. , Salehi, M. A. , Heidari, A. , Shafeghat, M. , Hamblin, M. R. , & Rezaei, N. (2020). Cytokine release syndrome: Inhibition of pro‐inflammatory cytokines as a solution for reducing COVID‐19 mortality. European Cytokine Network, 31, 81–93. 10.1684/ecn.2020.0451 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nasir, N. , Mahmood, F. , Habib, K. , Khanum, I. , & Jamil, B. (2020). Tocilizumab for COVID‐19 acute respiratory distress syndrome: Outcomes assessment using the WHO ordinal scale. Cureus, 12, e12290. 10.7759/cureus.12290 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olajide, O. A. (1999). Investigation of the effects of selected medicinal plants on experimental thrombosis. Phytotherapy Research, 13, 231–232. [DOI] [PubMed] [Google Scholar]
- Olajide, O. A. , Iwuanyanwu, V. U. , Lepiarz‐Raba, I. , & Hindawi, A. A. (2021). Induction of exaggerated cytokine production in human peripheral blood mononuclear cells by a recombinant SARS‐CoV‐2 spike glycoprotein S1 and its inhibition by dexamethasone. Inflammation. 44, 1865–1877. 10.1007/s10753-021-01464-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olaleye, S. B. , Onasanwo, S. A. , Ige, A. O. , Wu, K. K. , & Cho, C. H. (2010). Anti‐inflammatory activities of a kolaviron‐inhibition of nitric oxide, prostaglandin E2 and tumor necrosis factor‐alpha production in activated macrophage‐like cell line. African Journal of Medicine and Medical Sciences, 39, 41–46. [PubMed] [Google Scholar]
- Onasanwo, S. A. , Velagapudi, R. , El‐Bakoush, A. , & Olajide, O. A. (2016). Inhibition of neuroinflammation in BV2 microglia by the biflavonoid kolaviron is dependent on the Nrf2/ARE antioxidant protective mechanism. Molecular and Cellular Biochemistry, 414, 23–36. 10.1007/s11010-016-2655-8 [DOI] [PubMed] [Google Scholar]
- RECOVERY Collaborative Group , Horby, P. , Lim, W. S. , Emberson, J. R. , Mafham, M. , Bell, J. L. , … Landray, M. J. (2021). Dexamethasone in hospitalized patients with Covid‐19. The New England Journal of Medicine, 384, 693–704. 10.1056/NEJMoa2021436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roshanravan, N. , Seif, F. , Ostadrahimi, A. , Pouraghaei, M. , & Ghaffari, S. (2020). Targeting cytokine storm to manage patients with COVID‐19: A mini‐review. Archives of Medical Research, 51, 608–612. 10.1016/j.arcmed.2020.06.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sagar, S. , Rathinavel, A. K. , Lutz, W. E. , Struble, L. R. , Khurana, S. , Schnaubelt, A. T. , … Radhakrishnan, P. (2021). Bromelain inhibits SARS‐CoV‐2 infection via targeting ACE‐2, TMPRSS2, and spike protein. Clinical and Translational Medicine, 11, e281. 10.1002/ctm2.281 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmölz, L. , Wallert, M. , Heise, J. , Galli, F. , Werz, O. , Birringer, M. , & Lorkowski, S. (2014). Regulation of inflammatory pathways by an α‐tocopherol long‐chain metabolite and a d‐tocotrienol‐related natural compound. Free Radical Biology & Medicine, 75(Suppl. 1), S48. 10.1016/j.freeradbiomed.2014.10.813 [DOI] [PubMed] [Google Scholar]
- Seif, F. , Aazami, H. , Khoshmirsafa, M. , Kamali, M. , Mohsenzadegan, M. , Pornour, M. , & Mansouri, D. (2020). JAK inhibition as a new treatment strategy for patients with COVID‐19. International Archives of Allergy and Immunology, 181, 467–475. 10.1159/000508247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirato, K. , & Kizaki, T. (2021). SARS‐CoV‐2 spike protein S1 subunit induces pro‐inflammatory responses via toll‐like receptor 4 signaling in murine and human macrophages. Heliyon, 7, e06187. 10.1016/j.heliyon.2021.e06187 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smetana, K., Jr. , Rosel, D. , & BrÁbek, J. (2020). Raloxifene and bazedoxifene could be promising candidates for preventing the COVID‐19 related cytokine storm, ARDS and mortality. In Vivo (Athens, Greece), 34, 3027–3028. 10.21873/invivo.12135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stockman, L. J. , Bellamy, R. , & Garner, P. (2006). SARS: Systematic review of treatment effects. PLoS Medicine, 3(9), e343. 10.1371/journal.pmed.0030343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang, Y. , Liu, J. , Zhang, D. , Xu, Z. , Ji, J. , & Wen, C. (2020). Cytokine storm in COVID‐19: The current evidence and treatment strategies. Frontiers in Immunology, 11, 1708. 10.3389/fimmu.2020.01708 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomazini, B. M. , Maia, I. S. , Cavalcanti, A. B. , Berwanger, O. , Rosa, R. G. , Veiga, V. C. , … COALITION COVID‐19 Brazil III Investigators . (2020). Effect of dexamethasone on days alive and ventilator‐free in patients with moderate or severe acute respiratory distress syndrome and COVID‐19: The CoDEX randomized clinical trial. JAMA, 324, 1307–1316. 10.1001/jama.2020.17021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toniati, P. , Piva, S. , Cattalini, M. , Garrafa, E. , Regola, F. , Castelli, F. , … Latronico, N. (2020). Tocilizumab for the treatment of severe COVID‐19 pneumonia with hyperinflammatory syndrome and acute respiratory failure: A single center study of 100 patients in Brescia, Italy. Autoimmunity Reviews, 19(7), 102568. 10.1016/j.autrev.2020.102568 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallert, M. , Bauer, J. , Kluge, S. , Schmölz, L. , Chen, Y. C. , Ziegler, M. , … Lorkowski, S. (2019). The vitamin E derivative garcinoic acid from Garcinia kola nut seeds attenuates the inflammatory response. Redox Biology, 24, 101166. 10.1016/j.redox.2019.101166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, C. , Xie, J. , Zhao, L. , Fei, X. , Zhang, H. , Tan, Y. , … Bian, X. W. (2020). Alveolar macrophage dysfunction and cytokine storm in the pathogenesis of two severe COVID‐19 patients. eBioMedicine, 57, 102833. 10.1016/j.ebiom.2020.102833 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang, L. , Liu, S. , Liu, J. , Zhang, Z. , Wan, X. , Huang, B. , … Zhang, Y. (2020). COVID‐19: Immunopathogenesis and Immunotherapeutics. Signal Transduction and Targeted Therapy, 5, 128. 10.1038/s41392-020-00243-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou, P. , Yang, X. L. , Wang, X. G. , Hu, B. , Zhang, L. , Zhang, W. , … Shi, Z. L. (2020). A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature, 579, 270–273. 10.1038/s41586-020-2012-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu, N. , Zhang, D. , Wang, W. , Li, X. , Yang, B. , Song, J. , … China Novel Coronavirus Investigating and Research Team . (2020). A novel coronavirus from patients with pneumonia in China, 2019. The New England Journal of Medicine, 382, 727–733. 10.1056/NEJMoa2001017 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data S1. Supplementary information.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.


