Abstract
Severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) is the etiology of coronavirus disease 2019 (COVID‐19) pandemic. Current variants including Alpha, Beta, Gamma, Delta, and Lambda increase the capacity of infection and transmission of SARS‐CoV‐2, which might disable the in‐used therapies and vaccines. The COVID‐19 has now put an enormous strain on health care system all over the world. Therefore, the development of animal models that can capture characteristics and immune responses observed in COVID‐19 patients is urgently needed. Appropriate models could accelerate the testing of therapeutic drugs and vaccines against SARS‐CoV‐2. In this review, we aim to summarize the current animal models for SARS‐CoV‐2 infection, including mice, hamsters, nonhuman primates, and ferrets, and discuss the details of transmission, pathology, and immunology induced by SARS‐CoV‐2 in these animal models. We hope this could throw light to the increased usefulness in fundamental studies of COVID‐19 and the preclinical analysis of vaccines and therapeutic agents.
Keywords: animal models, COVID‐19, immunology, pathology, SARS‐CoV‐2, transmission
Animal models that recapitulate characteristics and immune responses observed in COVID‐19 patients are urgently needed. These animal models such as mouse, hamster, nonhuman primate, and ferret, have provided robust platforms for studying the transmission, pathogenesis, and immunology induced by SARS‐CoV‐2, and for evaluating the immunomodulatory and antiviral drugs and vaccines against COVID‐19.
1. INTRODUCTION
Severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) with zoonotic origin emerges in 2019 and causes mild to severe respiratory illness in humans. 1 The high transmission and mutation endow the ability of the novel coronavirus to develop unprecedentedly in the spatial range of epidemic areas. At the time of writing, the ongoing outbreak has caused over 200 million cases and more than 4 million deaths (https://covid19.who.int/). Clinical features are always initially characterized by fever, fatigue, dry cough, diarrhea, and chest pain. 1 , 2 The disease may progress to a severe SARS‐CoV‐2‐induced acute lung injury (ALI) and acute respiratory distress syndrome (ARDS), which causes the majority death in COVID‐19 patients. 3 The lethal features include hyperinflammatory response, lymphopenia and microthrombosis, and so forth, leading to the diffuse alveolar damage and extensive pulmonary injury. 4 , 5 In addition, factors of age, cardiovascular disease, obesity, hypertension, and diabetes may advance the individual's risk when infected with SARS‐CoV‐2. 6 , 7 , 8 , 9
Testing medical countermeasures, including therapeutic agents and vaccines that reduce COVID‐19 morbidity and mortality, becomes an emergency scientific event. 10 , 11 Advancing studies on different animal models for COVID‐19 are imperative for this effort, and provide measurable readouts for the pathogenic mechanisms and potential interventions. To this end, several animal species, including mice, nonhuman primates (NHPs), hamsters, ferrets, and cats, have been well utilized in the studies for SARS‐CoV‐2 (Figure 1). In this review, we provide an overview of the current literatures on animal models for SARS‐CoV‐2 infection and summarize the features for harmonizing and improving the use of current models to investigate COVID‐19. We hope this will throw light to the preclinical analysis of vaccines and therapeutic agents.
FIGURE 1.
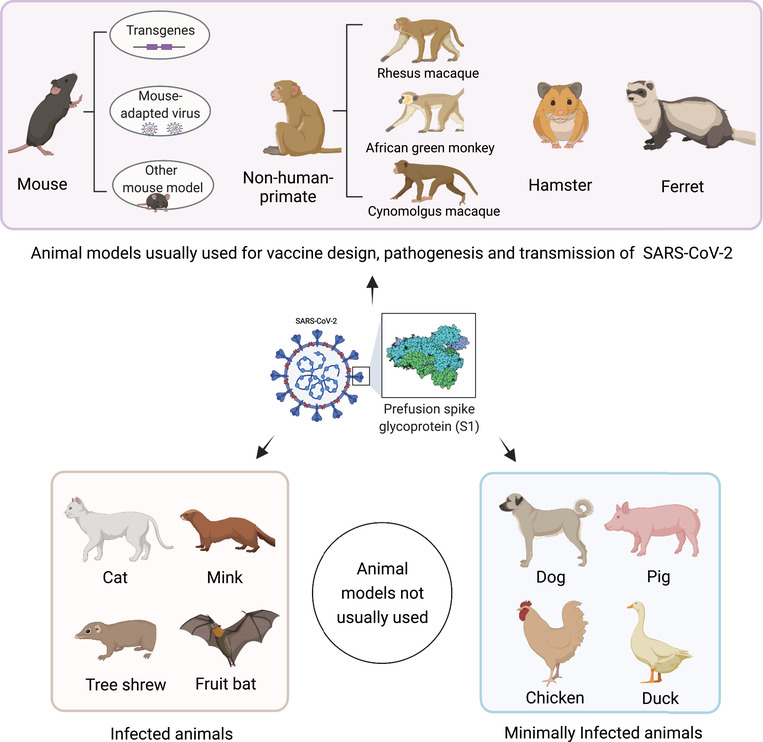
Animal models for SARS‐CoV‐2. Mouse, nonhuman primates (rhesus macaque, African green monkey, and cynomolgus macaque), hamster, and ferret are most used for studying the transmission, pathogenesis, and medical countermeasures. Mouse requires genetically modification for introducing hACE2 or adapted virus by serial passaging for SARS‐CoV‐2 infection. Animals of cat, mink, fruit bat, and Chinese tree shrew are susceptible to the virus infection, while dog, pig, chicken, and duck have the minimal infections; these animals are not usually used as models for SARS‐CoV‐2. The figure is created with BioRender.com
2. MOUSE MODELS
Mouse (Mus musculus) model has been well used for many viral investigations, such as Middle East respiratory syndrome coronavirus (MERS‐CoV) 12 , 13 and SARS‐CoV. 14 , 15 The main impediment for SARS‐CoV‐2 infection in mice is the lack of proper receptors, because SARS‐CoV‐2 entering host cells mainly relies on the human angiotensin‐converting enzyme 2 (ACE2) receptor not mouse ACE2. 16 Therefore, several strategies have been applied to resolve the problem, such as development of genetical modification and mouse adapted virus (Figure 2). We will detail the current mouse models used in SARS‐CoV‐2 investigation and discuss the pathogenic features below.
FIGURE 2.
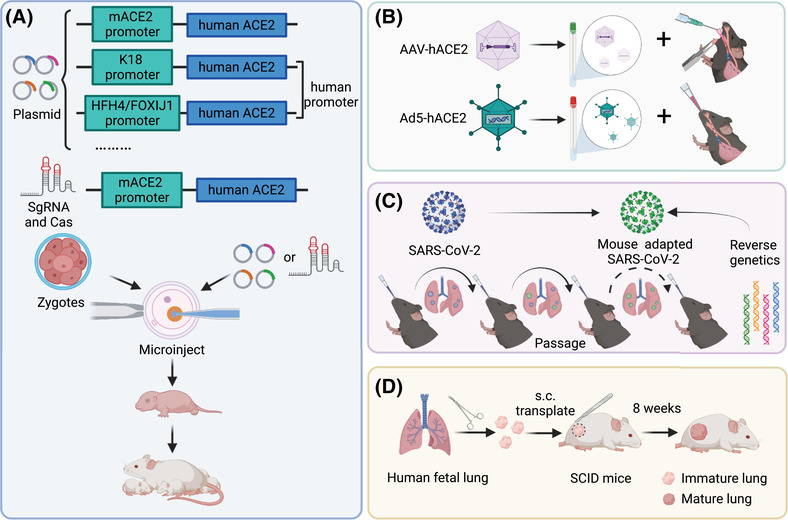
Strategies applied to the infection of mice. (A) Permanent genetic modifications are performed to introduce hACE2 to mice via CRISPR/Cas9 knock‐in technology or recombinant plasmid. The hACE2 expression is under the control of mouse ACE2 (mACE2) promoter, human epithelial cell cytokeratin‐18 (K18) promoter, or human lung ciliated epithelial cell‐specific HFH4/FOXJ1 promoter. (B) Ad5‐hACE2 or AAV‐hACE2 which expresses hACE2 is introduced to mice and sensitizes the respiratory tract of mice to SARS‐CoV‐2 replication. (C) Viruses are adapted to infect wide‐type mice by serial passaging or directly using reverse genetics. (D) SCID mouse‐human lung xenograft model of SARS‐CoV‐2 by surgically grafting human fetal lung tissue under the dorsal skin of SCID mice. The human lung xenografts can develop mature structures closely resembling the normal human lung and show a high viral replication with spreading to the whole lung tissue when infected with SARS‐CoV‐2. The figure is created with BioRender.com
2.1. Genetically modified mice
Generation of human ACE2 (hACE2) receptor expressing mouse is considered as a forthright strategy for SARS‐CoV‐2 infection in mice. To date, several methods including introducing hACE2 into mice via adenoviral/adenoviral‐associated vectors, expressing hACE2 by mouse ACE2 promoter or heterologous gene promoters and clustered regularly interspaced short palindromic repeats (CRISPR) systems, have been used to develop genetically modified mice. Notably, the varied expression of hACE2 among different organs in mice has led to the mild to severe disease in these mouse models, adding further complexity as factor of investigation of SARS‐CoV‐2‐induced pathogenesis and evaluation of therapeutic agents and vaccines.
The first SARS‐CoV‐2 infected transgenic mice are Institute of Cancer Research (ICR) background and express hACE2 under the control of mouse ACE2 promoter. 17 The mice intranasally infected with SARS‐CoV‐2 result in the weight loss, high viral replication in lung tissues, and exhibit interstitial pneumonia with infiltration of macrophages and lymphocytes. 17 The humoral immunity is induced on 21 days postinfection (dpi). Neurological symptoms such as anosmia, confusion and encephalopathy, 18 , 19 and cardiac injury and myocarditis 20 , 21 , 22 are common features that have been reported in COVID‐19 patients. However, the virus is not observed in other organs in this mouse model, such as the heart and brain, except for the intestine. 17 Using CRISPR/Cas9 knock‐in technology, hACE2 linking to tdTomato gene is inserted to the first coding exon of mouse ACE2 by homologous recombination, and the expression is driven by the mouse ACE2 promoter. 23 The hACE2 mice are susceptive to the intranasal infection of SARS‐CoV‐2 and present robust viral replications in organs of lung, trachea, and brain, as well as the olfactory epithelium, and develop the interstitial pneumonia and olfactory dysfunction accompanied by elevated cytokines. 23 , 24 Notably, the aged mice show marked decrease in weight loss and more severe pathology with increased neutrophil infiltration than those in the young ones, consistent with the clinical symptoms observed in COVID‐19 patients. 23 In one study, researchers intratracheally instill SARS‐CoV‐2 to the mice with CRISPR/Cas9 knock‐in of hACE2. 25 The hACE2 mice develop severe pulmonary pathologies with hemorrhage, inflammatory cell infiltration and hyaline membrane formation, and so forth, recapitulating many features observed in COVID‐19 patients with ARDS. 25 Recently, a strategy of rapid generation of hACE2 mice has been developed via combining CRISPR‐Cas9 system with tetraploid complementation. 26 The hACE2 gene is inserted into exon 2 and the expression is driven by the mouse ACE2 promoter. The mice show high viral replication in lung, airway, and small intestine. Pulmonary injury, such as alveolar septal thickening, inflammatory cell infiltration, hemorrhage, and hyaline membrane formation, are observed in mice with background of C57BL/6 or BALB/c, but the severest injury occurs earlier in the BALB/c mice, 26 indicating an earlier inflammatory response.
K18‐hACE2 transgenic mice, in which hACE2 expression is driven by the human epithelial cell cytokeratin‐18 (K18) promoter, 27 has been used for studying the pathogenesis of SARS‐CoV 28 and SARS‐CoV‐2 29 and can cause lethal infection. K18‐hACE2 mice exhibits high susceptible to SARS‐CoV‐2 infection. Intranasal inoculation of SARS‐CoV‐2 results in an approximate 25% of weight loss of mice along with virus spreading to other organs of heart, brain, kidney, spleen, duodenum, and colon. 30 , 31 , 32 , 33 High levels of viral infection in lung tissues severely impair the pulmonary structure and function, and cause thrombosis and vasculitis. 30 , 31 , 34 Severe infiltration of activated neutrophils, monocytes and lymphocytes to lungs, and dysregulated type I/ II IFN response are observed during SARS‐CoV‐2 infection in K18‐hACE2 mice. 30 , 35 Cytokine storm directly correlates with pulmonary injury, multiple organ failure, and an unfavorable prognosis, resulting in ARDS and systemic inflammatory response syndrome (SIRS). 36 Cytokine storm has been found in the lung, spleen, and even in the brain of K18‐hACE2 mice after intranasal inoculation with SARS‐CoV‐2, leading to multiple organ injury. 32 , 37 K18‐hACE2 mice could develop anosmia at early time points after infection, 34 followed by virus neuroinvasion that occurs in mice through the olfactory neuroepithelium or eye in a manner that virus transports only partially depending on hACE2, 32 , 38 a mechanism observed in COVID‐19 patients. 39 Interestingly, the male mice show a higher lethality compared to the female ones, perhaps because of the association with the higher level of inflammatory cytokines. 31 Notably, isolations of SARS‐CoV‐2 in lung and brain tissues from infected K18‐hACE2 mice show differences in genetics and plaque morphology; inoculation of these of lung and brain SARS‐CoV‐2 isolations into new batches of K18‐hACE2 mice results in the lethal pulmonary and central nervous system infection, respectively. 33 The highly tendentious pathogenicity of genetic variants of SARS‐CoV‐2 in different organs may provide an alternative approach to investigate the specific features observed in COVID‐19 patients. Recently, lethal diseases have also been reported among K18‐hACE2 mice infected with SARS‐CoV‐2 variants (B.1.1.7 and B.1.351), 40 suggesting that K18‐hACE2 mice can be used as a robust mouse model against the rapid evolution of SARS‐CoV‐2. Therefore, K18‐hACE2 transgenic mice have been widely used in the preclinical evaluation of vaccines 41 , 42 , 43 , 44 and antiviral drugs 45 , 46 , 47 , 48 against severe SARS‐CoV‐2 infection and COVID‐19 disease.
Another genetically modified mice, that is, HFH4‐hACE2 mice, express hACE2 under the control of a lung ciliated epithelial cell‐specific HFH4/FOXJ1 promoter. 49 , 50 HFH4‐hACE2 mice have been used for investigating the SARS‐CoV 50 , 51 and SARS‐CoV‐2 pathogenesis. 52 , 53 The hACE2 expression level is varied among different organs and is higher in lung tissues. 54 Intranasal infection of SARS‐CoV‐2 induces high viral replication in lung tissues of HFH4‐hACE2 mice, and causes interstitial pneumonia accompanied by elevated cytokines and infiltration of macrophages and lymphocytes and fibrin exudation. 54 The HFH4‐hACE2 mice are also susceptive to the infection of SARS‐CoV‐2 with D614G mutation. 55 Notably, two infection outcomes have been observed: (1) those that lost more than 20% of body weight often accompanied by noticeable neurological symptoms and died eventually; (2) those that lost less than 10% of body weight and survived. Moreover, the HFH4‐hACE2 mice exhibit a gender‐dependent progression that the males develop more lethal disease, 54 sharing some features observed in K18‐hACE2 mice. Besides, the preexposure to SARS‐CoV‐2 protects HFH4‐hACE2 mice from severe pneumonia, 54 indicating an adaptive immunity response in mice. Nevertheless, only some limited studies evaluate the therapeutic agents and vaccines using the HFH4‐hACE2 transgenic mice. 53 , 56
The CAG‐hACE2 transgenic mice have ever been used for investigating the SARS‐CoV infection. 57 , 58 A recent study reports the generation of CAG‐hACE2 mice, which express HA‐tagged hACE2 drove by the CAG promoter and is flanked by HS4 insulators. 59 High level of hACE2 expression in organs of lung, brain, and kidney endows the CAG‐hACE2 mice the susceptibility to the SARS‐CoV‐2 infection. 59 Intranasal infection with low dose virus (1000 plaque‐forming units, PFU) results in an absolute mortality with severe pulmonary pathologies, and the male CAG‐hACE2 mice exposed to SARS‐CoV‐2 infection exhibit much more weight loss than the female mice, 59 which is consistent with the sex‐biased response observed in K18‐hACE2 mice and HFH4‐hACE2 mice as described above. In addition, another study reports the C57BL/6‐Tgtn Smoc mice, which express hACE2 that links to luciferase driven by the CAG promoter (CAG‐human ACE2‐IRES‐Luciferase‐WPRE‐polyA), as models for SARS‐CoV‐2 investigation. The hACE2 expression is high in organs of heart, kidney, stomach, lung, spleen, and liver. Intranasal inoculation with 3 × 104 PFU of SARS‐CoV‐2 induces viral replication in lung and intestine tissues, and causes the interstitial pneumonia characterized by alveolar septa thickening and interstitial infiltrates. 60 Some medical countermeasures such as vaccines and antiviral drugs are under evaluation using the CAG‐hACE2 mice. 60 , 61
Finally, instead of permanent genetic modification, adenovirus or adeno‐associated virus that expresses hACE2, that is, Ad5‐hACE2 or adeno‐associated virus (AAV)‐hACE2 which can transduce a large proportion of pulmonary epithelial cells, is introduced to mice to sensitize the respiratory tract for SARS‐CoV‐2 replication. The wide‐type mice intranasally 62 or oropharyngeally 63 inoculated with Ad5‐hACE2 show high hACE2 expression confining to the alveolar epithelium and occasional expression in the airway epithelium. The transient effect of hACE2 expression can sustain for several days for SARS‐CoV‐2 infection and induces high viral replication in lung tissues, which terminally results in a rapid 20% of weight loss and impairs the pulmonary functions. 62 , 63 A variety of lesions including inflammatory infiltrates, hemorrhage, alveolar edema, and necrotic cell debris with markedly elevated cytokines such as TNF‐α and IL‐6, are observed. 62 , 63 , 64 Type I IFN response is observed during SARS‐CoV‐2 infection in Ad5‐hACE2‐transduced mice, accompanied by upregulation of signal transducer and activator of transcription 1 (STAT1) signaling, which is critical in protection against SARS‐CoV‐2. 62 , 63 Interestingly, pulmonary endothelial cells show viral replication and are induced endotheliopathy in Ad5‐hACE2 mice 65 ; other organs of liver and intestine, and so forth, do not exhibit viral replication. 62 , 63 The responses between C57BL/6 mice and BALB/c mice, such as the weight loss, viral replication, and the degree of pulmonary lesson, are slightly different. 62 In a recent study, intranasal inoculation of Ad5‐hACE2 causes the SARS‐CoV‐2 infection in peripheric leukocytes of mice as early as 1 dpi, which contributes to the systemic viral dissemination. 66 AAV‐mediated expression of hACE2 in mice also endows the viral replication in lung tissues, and increases the pulmonary infiltration of monocytes, neutrophils and lymphocytes, and Type I IFN response and humoral immunity. 67 , 68 However, adenovirus‐mediated delivery results in viral replication to lower titers and mild clinical signs of infection compared to those of K18‐hACE2 mice. 69 The adenovirus or adeno‐associated system circumvents the problem of time‐consuming process of breeding multiple generations and represents an easily and rapidly strategy for the currently urgent need of small animal models for COVID‐19. Countermeasures of new therapies and vaccines against SARS‐CoV‐2 have been under evaluation using the Ad5‐hACE2‐ or AAV‐hACE2‐based mouse model. 62 , 70 , 71 , 72 , 73 A limitation of these mice is that hACE2 is expressed ectopically, which may change the tissue or cellular tropism of the virus.
2.2. Mouse‐adapted viruses
Virus can be adapted to infect wide‐type mice by serial passaging, which may more closely resemble the natural host‐pathogen interactions in mice. This strategy is successful because of a swarm of related viral quasi species in the populations of RNA viruses. The rare viruses in the swarm that contain mutations in the key protein that increase their binding affinity to entering receptors of mice are expected to be selected, owing to their higher levels of replication in lung tissues. This strategy has been successfully applied to investigate the SARS‐CoV 14 , 74 and MERS‐CoV, 12 and has generated highly infective viruses and caused lethal pulmonary diseases.
Recently, evolution in vivo via serial passage of SARS‐CoV‐2 in the mouse lung tissue generates highly infective virus, that is, MASCp6, with increased virulence in both young and aged mice. 75 The mice intranasally inoculated with MASCp6 develop the interstitial pneumonia and inflammatory response. MASCp6 infection causes severe pulmonary pathology and higher inflammation in the aged mice than those in young ones. 75 Also, MASCp36, derived from MASCp6 virus by serial passaging again, obtains robust affinity to mouse ACE2 and exhibits age and gender‐related skewed distribution of mortality with 50% of mortality in aged, male mice when inoculation with only 100 PFU of MASCp36. 76 Another mouse‐adapted virus, that is, SARS‐CoV‐2 MA10, recapitulates the age‐related disease severity observed in humans as well, and causes lethal features of ALI and ARDS, and death in wild‐type mice. 77 Interestingly, BALB/c mice suffer more sorrows compared with C57BL/6J mice, which may because of the different immune paradigms induced by the virus in these two background mice. Other mouse‐adapted SARS‐CoV‐2 viruses, such as HRB26M, 78 MACo3, 79 and rSARS2‐N501YP30, 80 have been reported the caused moderate to lethal disease.
In addition, some researchers have directly developed mouse‐adapted virus using reverse genetics, including SARS‐CoV‐2 MA (precursor of SARS‐CoV‐2 MA10) 81 and rSARS2‐N501YP0 (precursor of rSARS2‐N501YP30). 80 These mouse‐adapted viruses can replicate in the upper and lower airways of mice; however, the viruses are only able to cause mild disease in mice even intranasal inoculation with a high dose of the adapted viruses. Therefore, evolution in vivo via serial passaging of modified SARS‐CoV‐2 using reverse genetics may be an ideal approach to develop SARS‐CoV‐2 strain with high infective and lethal to the general laboratory mice.
Some trials including potential medical countermeasures against SARS‐CoV‐2, have been in progress or done using the mouse model with mouse‐adapted virus. 82 , 83 , 84 However, the mouse model has limitations: (1) The mouse‐adapted virus may cause the infection that does not recapitulate many aspects of human disease and may induce injury via unique pathogenesis; (2) The occurrence of key mutations in immunogen such as receptor‐binding domain (RBD) which is the primary target for the neutralizing antibody response, may lead to the noneffective neutralizations for the mouse‐adapted virus.
2.3. Other mouse models
Several studies have applied the immunodeficient mouse to investigate the pathogenesis in human‐specific viral infections, such as SARS‐CoV. 85 , 86 Recently, researchers have developed a severe combined immunodeficiency (SCID) mouse‐human lung xenograft model for SARS‐CoV‐2 by surgically grafting human fetal lung tissue under the dorsal skin of SCID mice. 87 The human lung xenografts can develop mature structures closely resembling the normal human lung and show a high viral replication with spreading to the whole lung tissue when injected with SARS‐CoV‐2. 87 The SARS‐COV‐2 infection in this mouse model confines to epithelial cells and causes a severe pulmonary damage with elevated pro‐inflammatory response. 87 The immunodeficient mouse can also be used for studying the role of immune effectors combining the human immune system and ACE2 expression; however, this strives has not been used for evaluating the vaccines and therapies.
The high mortality of SARS‐CoV‐2 infection closely associates with the disease progression to a severe COVID‐19‐induced ARDS. 88 The mortality rate in COVID‐19 patients with ARDS is up to 50% and reaches 94% in those who received mechanical ventilation. 9 To this end, mouse models that recapitulate the features of COVID‐19 patients with ARDS should be highlighted. We have ever established a mouse model via intratracheal instillation of SARS‐CoV‐2 (4 × 10 5 PFU) to hACE2 mice (Figure 3). 25 The transgenic mice are generated using CRISPR/Cas9 knock‐in technology. Severe gross lesions involve the bigger size with the bilateral congestion and edema, and patched hemorrhage of lung on 5 dpi. The infected mice develop severe bronchopneumonia, diffuse interstitial pneumonia, and vasculitis. Histopathological changes in lung tissue include reduction of airway space, thickened alveolar septa, pulmonary edema, necrosis, proteinaceous debris in the alveolar space, formation of hyaline membrane, occasional thrombus in pulmonary capillary lumen, consolidation, diffuse hemorrhage, and alveolar damage. Inflammatory infiltrates include massive neutrophils, and scattered monocytes, macrophages, and lymphocytes throughout the lung tissue. Notably, the neutrophils are infiltrated to lung as early as 6 h post inoculation of SARS‐CoV‐2, and present the predominant cell infiltration until 3 dpi. The dead cells are mostly shown in the background of the inflammatory cell infiltrates. In addition, a recent study has reported an animal model for SARS‐CoV‐2‐induced ARDS in genetically unaltered CD‐1 mice. 89 Bleomycin or ricin‐pretreated mice show highly susceptible to SARS‐CoV‐2 infection, accompanied by sustained body weight loss and more than 50% of mortality rates. Notably, the viral replication is observed in the upper and lower respiratory tracts and in the organs of lung and heart, and in serum samples. The deleterious effects induced by SARS‐CoV‐2 infection include severe pulmonary damage characterized by extensive peribronchial and perivascular inflammatory cell infiltrates. This genetically unaltered mouse might be infected via a novel mechanism other than the canonical ACE2‐mediated uptake route. Besides, some strategies, such as intratracheal instillation of Poly I:C and SARS‐CoV‐2 spike protein 90 and intravenous injection of SARS‐CoV‐2 envelope (2‐E) protein, 91 have also been reported to induce ARDS‐like pathological damages in genetically unaltered mice. Finally, the K18‐hACE2 mice can develop lethal disease after SARS‐CoV‐2 infection as describe previously, and may be an alternative model for the ARDS‐like pathological investigation.
FIGURE 3.
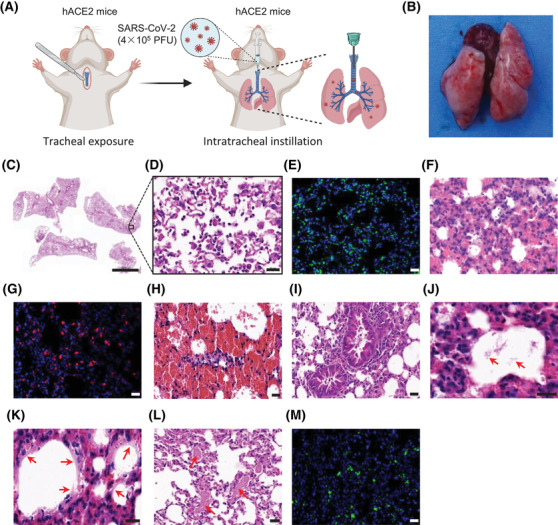
The pulmonary features of a mouse model for SARS‐CoV‐2‐induced ARDS. (A) An illustration for route of the establishment of a mouse model for ARDS. (B) The gross observation for pulmonary morphology on 5 dpi. (C–D) Images of H&E staining for pulmonary pathological changes of mice on 5 dpi. (E) Images of TUNEL staining for cell apoptosis in lung tissue on 5 dpi. (F–G) Images of H&E staining (F) and immunofluorescence staining for Ly6G (G) indicate the neutrophils as the predominant cell infiltration with 60% of the lung tissue. (H–L) SARS‐CoV‐2 infection induces apparent hemorrhage (H), inflammatory cells (I), proteinaceous debris (J, red arrow), hyaline membranes‐like changes (K, red arrow), and thrombi (L, red arrow) in the lung tissue of the mouse model. (M) Images of immunofluorescence staining for SARS‐CoV‐2 spike protein in the lung section of a mouse with SARS‐CoV‐2‐induced ARDS. Scale bars represent 2000 μm (C) or 20 μm (D‐M). Reprinted with the permission from a published reference 25
Chronic diseases, such as obesity, cardiovascular disease, hypertension, and diabetes, are closely correlated to the severe COVID‐19 disease and advance the SARS‐COV‐2‐induced mortality. 7 , 8 , 9 Genetic or dietary modification that induces features of these human chronic diseases has been implemented in mice. 92 , 93 , 94 Efforts of developing the risk‐related models for studying SARS‐CoV‐2 infection may involve mouse‐adapted SARS‐CoV‐2 virus or mice humanized with hACE2 combining related chronic diseases. For example, low dose of mouse‐adapted SARS‐CoV‐2 which is generated through serial passing in the mouse lung, has been reported to cause the enhanced morbidity in aged and diabetic obese mice (generated by dietary modification), resembling the clinical features observed in COVID‐19 patients. 95 Nevertheless, these chronic disease‐related mouse models for SARS‐CoV‐2 infection need to be fully explored, which may provide a robust platform for the efficiency evaluation of potential vaccines and drugs to combat with SARS‐CoV‐2 infection.
Although appropriate mouse models for studying SARS‐CoV‐2 transmission have not been reported, the emerged variants might impart cross‐species transmission of SARS‐CoV‐2 to naïve mice by enhancing receptor binding, such as the currently reported N501Y, E484K, and K417N variants. 96 , 97 , 98 The mutations render effective binding of the RBD to mouse ACE2 and lead to fusion of neighboring membranes and effective infection, 97 which have been found in the mouse‐adapted viruses as well, especially the N501Y mutation. 75 , 95 Further investigations on the potential implications for SARS‐CoV‐2 transmission, such as mouse‐to‐mouse, or mouse as an intermediate host, need to be made to clarify the possible routes whereby viruses are transmitted and limit the mouse‐mediated transmission.
In summary, several mouse models have been used or under development for investigating the pathogenesis of SARS‐CoV‐2 infections. These mouse models develop mild to lethal diseases; however, none of these models can replicate all features observed in COVID‐19 patients, especially the unusual aspects such as the pulmonary vascular disease. Furthermore, more efforts on using mouse models need to be made to demonstrate the roles of risk‐related factors, such as cardiovascular disease and obesity in SARS‐CoV‐2 infection.
3. SYRIAN HAMSTER MODELS
Syrian golden hamsters (Mesocricetus auratus) are small mammals that have been used for decades to study the infections of human respiratory viruses, including influenza virus, SARS‐CoV, 99 , 100 , 101 and genetically modified animals generated for MERS‐CoV. 102 The ACE2 domain of human that interacts with SARS‐CoV‐2 RBD of S protein closely resembles that of hamsters, 103 suggesting that hamsters could be suitable as a natural model for studying the SARS‐CoV‐2 transmission and infection. As expectedly, intranasal inoculation with 5 × 104 TCID50 (median tissue culture infective dose) of SARS‐CoV‐2 induces approximately 15% of weight loss and viral replication in upper and lower respiratory tracts and organs of lung, heart, brain, spleen, and liver of hamsters. 104 , 105 The virus in the lung tissue is mainly presented in the bronchial epithelium, macrophages, and alveolar epithelial cells (type I and II), followed by rapid viral clearance between 6 and 7 dpi. 105 , 106 The hamsters develop mild clinical scores and recover on 14 dpi. 105 , 106 However, intranasal inoculation with high dose of SARS‐CoV‐2 (5 × 105 TCID50) induces a severe pulmonary injury in hamsters that shares many features with COVID‐19 patients, including bilateral and multilobular ground glass opacity, severe weight loss, and increased mortality. 104 , 107 , 108 Multiple organ damages are also observed in the severe SARS‐CoV‐2 infected hamster model, accompanied by higher level of viral replication in tissues of lung, heart, brain spleen, liver, lymph node, kidney, adrenal gland, different segments of alimentary tract, ovary, prostate, and vesicular gland, which eventually leads to the systemic injury. 108 Notably, the focal lesions still exist in part tissues of liver, lymph node, and heart after prolonged infection, 108 suggesting a sustaining damage. The diseases of genital system and digestive system induced by SARS‐CoV‐2 in hamsters would provide a suitable aspect in related human diseases. Besides, olfactory epithelium is also damaged by SARS‐CoV‐2 infection, resulting in the olfactory dysfunction in hamsters as early as 2 dpi. 109 , 110 , 111 In one study, hamsters are orally inoculated with SARS‐CoV‐2 (1 × 105 PFU) and develop no sign of disease compared to the equal viral quantity of intranasal inoculation. 112 The viral replication in organs of lung and small intestine are much lower and the body weight shows no change, 112 suggesting the respiratory infection with less efficiency when oral acquisition of SARS‐CoV‐2 in hamsters.
In hamsters, SARS‐CoV‐2 infection causes mild to lethal pathologic changes. Multifocal bronchiolar epithelial necrosis and inflammatory infiltrates of lung occur as early as 2 dpi, 104 consisting of many neutrophils, macrophages, monocytes, and a few lymphocytes, and develop the most severe on 4 dpi. 105 , 106 , 113 The infection can progress to interstitial pneumonia characterized by massive pulmonary consolidation after infection. 104 , 105 , 106 In addition, endothelialitis, alveolar hemorrhage, and pulmonary edema are also observed in severe infected hamsters. 105 , 114 Infiltration of lymphocytes, pneumocyte hyperplasi, and fibrosis are main features in the later progression. 105 , 114
SARS‐CoV‐2 infection in the hamster model is a closely gender‐ and age‐dependent progression. The old hamsters show more pronounced weight loss and severe pulmonary disease, such as alveolar and perivascular edema, more pulmonary inflammatory infiltrates, and consolidation, compared to the young ones when infected with SARS‐CoV‐2. 106 However, the response to the infection is quite different, that the young hamsters exhibit earlier viral replication and spreading and immune cell influx to lung, while the old hamsters show a delayed and more adverse response to the SARS‐CoV‐2 infection. 106 Resembling the age‐dependent progression, male hamsters show a high viral replication, weight loss, and severe pulmonary disease. 114 The manifestations are coincident with those of COVID‐19 patients.
Importantly, the hamsters are suitable as models to investigate the transmission of SARS‐CoV‐2. SARS‐CoV‐2 can be transmitted by direct contact or via aerosols (respiratory droplets or airborne droplet nuclei) between humans. To date, several studies have launched this animal model for the efforts to prevent the SARS‐CoV‐2 transmission. For example, SARS‐CoV‐2 can be transmitted efficiently from infected hamsters to naive hamsters via aerosols or by direct contact; using surgical mask could well reduce the infected proportion of hamsters (from 66.7% to 16.7%), 115 suggesting the potential benefit of using surgical mask against the SARS‐CoV‐2 transmission. Intranasal inoculation of SARS‐CoV‐2 causes the viral replication in epithelial cells of the duodenum and viruses in faeces, suggesting the hamsters as models for studying the oral‐faecal route of transmission of SARS‐CoV‐2. 105 Notably, transmission via fomites such as soiled cages is not as efficient and the communicable period is short and closely correlates with the infectious virus. 105 However, unlike the challenged hamsters, infected naive hamsters show minimal clinical symptoms, lower viral replication in respiratory tract, and milder disease, and can recover to the original condition. Therefore, the infected naive hamsters may not fully recapitulate the features observed in SARS‐CoV‐2‐infected humans.
Expression levels of chemokines and cytokines, which link to COVID‐19 in humans including IL‐6, IL‐10, IFN‐λ, IFN‐γ, CXCL‐10, MX‐2, and TNF‐α, and so forth, are robustly increased in hamsters on 4 dpi, and are gradually resolved on 7 dpi. 116 , 117 , 118 Type I and III IFN signaling has been involved in the restricting infection of many respiratory viruses including SARS‐CoV and SARS‐CoV‐2. 118 , 119 In STAT2‐knockout hamsters, the immune responses including Type I and III IFN signaling are decreased with a reduced injury of immune pathology; however, the viral replication is increased and disseminate to several peripheral tissues without the keeper of STAT2. 118 Besides, a study on RAG2‐knockout hamsters shows that the absence of the functional B and/or T cells can exacerbate pathology at the early stage of SARS‐CoV‐2 infection. 120 IL‐2 receptor knockout in hamsters results in the prolonged viral persistence, suggesting the susceptibility of SARS‐CoV‐2 to adaptive immune control. 113 Like the observations in COVID‐19 patients, adaptive response is induced and neutralizing antibodies are produced as early as 7 dpi in hamsters. 105 , 121 Passive immunization or antibody therapy shows benefits of reduction of viral dissemination and progression in hamsters. 107 , 120 , 122 , 123 Therefore, the hamster model may be used as an alternative platform to investigate the immune response, therapies, and vaccines.
4. NHP MODELS
NHPs are desirable models with similar anatomy, phylogeny, and immunology to humans. There are a number of researches that have applied those NHP modes, including rhesus macaques (Macaca mulatta), African green monkeys (Chlorocebus aethiops), and cynomolgus macaques (Macaca fascicularis), for investigating the pathogenesis involved in SARS‐CoV‐2 infection and also for developing the medical countermeasures (Figure 4). The ACE2 protein in NHP especially in rhesus macaques gives an overall homology of 91% compared with hACE2. 124 Analysis of NHP single‐cell RNA‐sequencing data sets reveals that the cell subtypes of type II pneumocytes, nasal goblet secretory cells, and absorptive enterocytes co‐express ACE2/type II transmembrane serine protease 2 (TMPRSS2), 125 suggesting the potential as cellular targets of SARS‐CoV‐2. Previous studies have demonstrated that African green monkeys are more susceptible to the SARS‐CoV infection than rhesus and cynomolgus macaques that cause severe disease. 126 The species differences are slightly varied when infected with SARS‐CoV‐2. For example, SARS‐CoV‐2 infection have been reported to cause only minimal to mild clinical features, such as reduced appetite, dehydration, transient fever (occurs between 1 and 2 dpi), mild weight loss, and occasional coughing, with peaking on 7 days in rhesus macaques and recovery between 9 and 17 days responding to SARS‐CoV‐2 infection 127 , 128 ; the African green monkeys have a spectrum of responses from minimal to severe disease, 129 , 130 , 131 while the cynomolgus macaques have a milder decreased physical activity or asymptom. 132 , 133 , 134 Rhesus macaques, African green monkeys, and cynomolgus macaques are all susceptible to the SARS‐CoV‐2 infection, but differences in time‐shifted viral replication and clearance are found among the three kinds of NHPs. 132 , 133 , 134 Intranasal or combined intratracheal and intranasal inoculation of SARS‐CoV‐2 induces the robust viral replication in the upper respiratory tract including nasal turbinate and throat, with peaking between 1 and 3 dpi and persisting until 5–7 days before dropping below detection limits between 14 and 18 dpi. 135 In two studies, SARS‐CoV‐2 infection causes a second wave of viral replication in nasal swab samples on 5 dpi, with prolonged viral shedding period in nasal tissues that can persist for 4 weeks. 128 , 136 The viral replication in the lower respiratory tract such as trachea and in lung tissues increases on 3 dpi and peaks on 9 dpi in one study. 128 Extrapulmonary organs including brain, spleen, liver, heart, kidney, eye, gastrointestinal tract, uterus, bladder, central nervous system, urogenital tract, and lymph node, are also observed viral replication, suggesting the potential of systemic injury. 128 , 134 , 135 , 137 , 138 , 139 , 140 However, some studies negate the viral replication in extrapulmonary organs, except for gastrointestinal tract and lymph node. 127 Notably, only intratracheal inoculation of SARS‐CoV‐2 does not induce the viral replication in nasal tissue and much lower viral loads are observed in the nose and throat swab and in lung tissue compared to the route of intranasal or combined intratracheal and intranasal inoculation. 141 The difference suggests that nasal infection congregates more virus in the upper respiratory tract and aggravates the viral transmission and infection in the lower respiratory tract that may cause more severe disease. Besides, investigators also use the LMA mucosal atomization device that resembles the human infection; this route causes the similar viral replication expect for the prolonged viral loads compared to the nasal infection. 142 In aged NHPs, the viral replication in the upper respiratory tract persists for 10 days with peaking loads and is much higher in lung tissues compared to the young ones 143 ; while the gender might be not the risk factor in severe disease. 134
FIGURE 4.
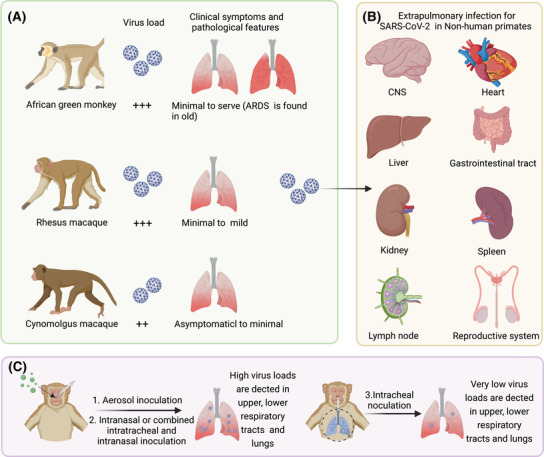
SARS‐CoV‐2 infection in nonhuman primates. (A) Rhesus macaques, African green monkeys, and cynomolgus macaques are all susceptive to SARS‐CoV‐2 infection. Asymptomatic to minimal clinical features and minimal to mild clinical features are observed in cynomolgus macaques and rhesus macaques, respectively. The African green monkeys have a spectrum of responses from minimal to severe disease, in which the aged could progress to ARDS. (B) Viral replication is observed in extrapulmonary organs including brain, spleen, liver, heart, kidney, gastrointestinal tract, uterus, urogenital tract, and lymph node. (C) The route of aerosol, intranasal, or combined intratracheal and intranasal inoculation of SARS‐CoV‐2 causes high viral loads in the upper and lower respiratory tracts and lungs in nonhuman primates. Only intratracheal inoculation causes much lower viral loads in the upper and lower respiratory tracts and lungs. The figure is created with BioRender.com
In NHPs, SARS‐CoV‐2 infection causes minimal to severe pathologic changes. Inflammation in the upper airways is characterized by multifocal squamous metaplasia of the respiratory epithelium and infiltration of a small number of neutrophils. Severe gross lesions involve pulmonary punctate hemorrhage and red lesion. 128 , 134 , 142 Bronchopneumonia and interstitial pneumonia occur as early as 3 dpi that frequently center on small bronchus and terminal bronchioles, respectively. 127 , 136 Multifocal clusters of virus‐infected cells are presented throughout the pulmonary parenchyma. 144 Histopathological changes in lung tissues include thickened alveolar septa, diffuse alveolar damage, formation of hyaline membrane, occasional diffuse hemorrhage, collagen deposition, pulmonary edema, consolidation, minimal necrosis, and pneumocyte hyperplasia. One study has shown the thrombus in pulmonary capillary lumen accompanied by endothelial cell swelling and perivasculitis. 136 Inflammatory infiltration of monocytes, macrophages, neutrophils and eosinophils aggregates in alveoli, pulmonary parenchyma and in the respiratory epithelial submucosa of larger airways, and a small number of lymphocytes are confined predominantly to perivascular sites. 142 SARS‐CoV‐2 antigen is presented in NHPs associated with pulmonary lesions, especially in the alveolar epithelial cells and macrophages. 142 , 143 The NHPs develop the most severe pathology between 4 and 5 dpi, and the diseases of those alleviate gradually but display moderate to mild alveolar inflammation. 128 , 133 The infection causes long‐standing pulmonary injury accompanied by multifocal chronic interstitial pneumonia even on 34 dpi. 142 However, some in vivo studies demonstrate the recovery undergo SARS‐CoV‐2 infection in NHPs. 127 , 137 Extrapulmonary tissues, such as brain, heart, liver, spleen, kidney, stomach, ileum, colon, testis, and lymph node, have been reported to present mild histopathological lesions. 128 , 134 , 136 Those organs show varied levels of edema, infiltration of inflammatory cells on the early stage of infection, while the sustained systemic inflammatory response is devoid. For example, microhemorrhages and neuropathology that is consistent with hypoxic injury are presented in SARS‐CoV‐2‐infected NHPs. 145 Besides, lymph nodes of mesenteries and lung, and spleen display increased immune activity by enlarging germinal centers in response to viral infection. 128 , 134 Like the observations in COVID‐19 patients, the aged NHPs develop more severe diffuse interstitial pneumonia compared to the young ones, 130 , 143 and the aged African green monkeys are reported to progress to ARDS, 130 which has been noted as the main cause of mortality induced by SARS‐CoV‐2. These findings highlight the importance of including age in the selection of animals.
Involved chemokines and cytokines including IL‐1Ra, IL‐1β, IFN‐γ, IFN‐α TNF‐α, IL‐6, IL‐2, IL‐4, IL‐5, RANTES, G‐CSF, GM‐CSF, CCL2, and IP‐10, and so forth, are increased at a range scale of time and space. 128 , 130 , 133 , 134 , 135 Activation of innate immune response occurs between 1 and 3 dpi accompanied by type I IFN response; then the Th1/Th2 response and adaptive response are induced at the middle and late stage of infection. Neutralizing antibodies are produced as early as 5 days after SARS‐CoV‐2 infection in NHPs and peak between 15 and 21 dpi. 127 , 135 The induced immune response protects those of NHPs from adverse effects when rechallenged the SARS‐CoV‐2, including lower viral replication, no remarkable lesion in the upper and lower respiratory tracts and no significant bronchopneumonia, and produces significantly higher neutralizing antibody titers. 136 , 137 , 144 Notably, the aged NHPs present a delayed immune response with more severe cytokines and chemotaxis, but lower titers of SARS‐CoV‐2‐specific IgG antibody levels and weaker antiviral functions compared to the young ones. 146 , 147 Taken together, although the NHPs have failed to recapitulate the severe diseases such as ARDS (only observed in a few aged African green monkeys) and SIRS, these animals still present the priority as models for studying the immunopathology and evaluating the protective efficacy of vaccines and drugs to combat with SARS‐CoV‐2 infection. 148 , 149 , 150 , 151 , 152
5. FERRET MODELS
Ferrets (Mustela putorius furo) are mammals that have been well used as models to study the infection and transmission of human respiratory viruses, including influenza virus, SARS‐CoV. 153 , 154 , 155 The respiratory tract of ferrets anatomically resembles that of humans, such as the glandular density in the bronchial wall, terminal bronchioles, and proportions of the upper and lower respiratory tract. 156 , 157 ACE2 is mainly expressed on glandular and type‐II alveolar epithelial cells of ferrets and shows affinity to the SARS‐CoV infection. 158 , 159 Intranasal inoculation of SARS‐CoV‐2 induces the clinical symptoms that observed in COVID‐19 patients, including reduced activity, nasal discharge, sneezing, wheezing, occasional cough, and increased body temperature in infected ferrets. 160 , 161 The clinical symptoms are manifested on 2 dpi and recover to the initial state on 12 dpi. The body weight loss is controversial in several studies on SARS‐CoV‐2‐infected ferrets. 162 , 163 , 164 High levels of SARS‐CoV‐2 are detected in the upper respiratory tract, such as nasal turbinate and throat, with peaking between 3 and 4 days and persisting until 8 days before dropping below detection limits on 10 days after inoculation. 161 , 162 , 163 The viral replication is also observed in the trachea, lung, intestine, and brain, 161 , 164 suggesting the possibilities of central nervous system injury and oral‐faecal route of transmission. Interestingly, intratracheal inoculation of SARS‐CoV‐2, a route which is reported to cause more severe disease in ferrets when infected with influenza virus 165 , 166 , 167 and in hACE2 mice 25 when infected with SARS‐CoV‐2, shows a low efficient in establishing an infection with milder diseases in ferrets compared to the route of intranasal inoculation. 164 The poor outcome may be caused by the relative lower expression of receptor in lower respiratory tract of ferrets for SARS‐CoV‐2 entering. Furthermore, studies on gender and age‐related SARS‐CoV‐2 infection suggest that the aged ferrets, especially the males, suffer more illnesses, such as higher viral loads, longer viral replication, more severe pulmonary disease, and clinical features. 160 , 168 , 169 The skewed distribution captures some features observed in COVID‐19 patients.
In ferrets, SARS‐CoV‐2 infection causes minimal to mild pathologic changes. The nasal cavity develops a mild inflammation with few necrosis of epithelial cells and occasional hemorrhage. 162 , 167 Moderate bronchopneumonia occurs as early as 3 dpi, accompanied by infiltration of neutrophils, mononuclear cells, macrophages and lymphocytes into the bronchiolar lumina, and mild necrosis of the bronchiolar epithelial cells. 162 , 167 The bronchopneumonia only affects no more than 15% of the lung section on 3 dpi and decreases to less than 5% on 7 dpi. 162 Thickness of the interalveolar septa and alveolar hyperplasias are observed during SARS‐CoV‐2 infection. 162 , 164 Moreover, multifocal hepatitis occurs after inoculation, suggesting the liver damage. 162
The ferrets are suitable to study the transmission of SARS‐CoV‐2. 170 Efficient viral transmission between ferrets agrees with recent SARS‐CoV‐2 outbreaks in mink farms. Direct contact with SARS‐CoV‐2‐infected ferrets can induce the transmission of virus to the naïve ferrets, which occurs as early as 2 dpi. 161 , 171 , 172 Transmission via respiratory droplets and/or aerosols is efficient between ferrets over more than 1 m distance, 173 but shows a delayed infection for 3–7 days 172 compared to the direct contact. However, in one study, the respiratory droplet shows a low efficiency with only 25% of infection between naïve ferrets. 171 The pattern of clinical features, viral replication, pathology, and immune response of the infected naïve ferrets resemble those of inoculated ferrets. 161 , 172 To date, the ferrets are well used as models to measure the countermeasures against SARS‐CoV‐2 transmission. For example, treatment with a lipopeptide fusion inhibitor, which blocks membrane fusion between the virus and host cell, completely prevents the direct contact of transmission of SARS‐CoV‐2 between ferrets, 174 suggesting the potential to reduce community transmission of SARS‐CoV‐2. Besides, MK‐4482/EIDD‐2801, a ribonucleoside analog inhibitor, also mitigates SARS‐CoV‐2 infection and blocks transmission in the ferret model. 175
Involved immune responses include activated T cells, macrophages, and Type I IFN signaling. 160 , 168 , 176 The adaptive response is induced and neutralizing antibodies are produced as early as 8 dpi in ferrets. 162 The induced immune response protects ferrets from adverse effects when rechallenged the SARS‐CoV‐2, including lower viral replication, no remarkable lesion in the upper and lower respiratory tracts, and no significant bronchopneumonia. 162 , 163 , 171 However, lack of ferret‐specific immunological reagents severely impedes the further investigation of immunological mechanisms in this animal model. Some new technologies such as single‐cell RNA (scRNA) sequencing, 176 have been employed to resolve this dilemma. The ferret model has been used in the preclinical evaluation of vaccines 177 , 178 and antiviral drugs. 179
6. OTHER SPECIES
6.1. Cats
Several studies have demonstrated the transmission from human to domestic cats (Felis catus). 180 , 181 , 182 , 183 , 184 Cats are highly susceptible to airborne transmission and SARS‐CoV‐2 infection, and those who infected by their human owners could transmit the virus to the naïve cats. 185 , 186 However, the efficiency of transmission from infected cats to the naïve ones could be attenuated by serial passaging of the virus between cats, 187 , 188 and reinfected cats also show no transmissibility. 189 Intranasal inoculation of SARS‐CoV‐2 induces the viral replication in upper and lower respiratory tract with peaking on 3 dpi and persisting until 5–7 dpi before dropping below detection limits on 14 dpi. 185 , 188 , 190 , 191 The viral replication is also observed in the spleen, lymph node, liver, heart, olfactory bulb, and gastrointestinal tract. 189 , 191 None of clinical feature of disease is found during the infectious period in the current studies, except for one reported loss of body weight, 188 suggesting the minimal to mild disease in infected cats. Pathological analysis shows that inflammation in nasal turbinate and trachea and moderate interstitial pneumonia accompanied by diffuse alveolar damage occur during the SARS‐CoV‐2 infection. 184 , 188 , 189 , 190 , 191 Characteristic changes in lung tissue include swelling and degeneration of bronchiolar epithelial cell, cellular debris, thickened alveolar septa, collagen deposition, mild pulmonary edema, mild‐to‐moderate bronchiolitis with infiltration of monocytes, neutrophils and lymphocytes which are confined predominantly to perivascular sites. The thrombosis is not the common feature in the infected cats. 184 In addition, SARS‐CoV‐2 infection also causes the gastrointestinal tract injury, such as multifocal inflammation with infiltration of neutrophils and lymphocytes in the intestinal submucosa and muscularis. 188 The viral antigen expression is observed mainly in epithelial cells of the alveolar and nasal turbinate, enterocytes of the small intestine and submucosal gland of the trachea. 188 , 189 The adaptive response is induced and neutralizing antibodies are produced as early as 7 dpi and maintained or increased in titer even on 28 dpi in cats. 190 Rechallenged cats exhibit faster and more robust humoral immunity against the viral infection. 189 Although cats may be a potential candidate for studying the asymptomatic to moderate COVID‐19, we should take care of the feasibilities of cats as models for demonstrating the respiratory disease of humans. Additional studies on transmission efficiency need to be further explored, especially cat as an intermediate host between SARS‐CoV‐2 and humans, inflammation and how this mirrors human disease.
6.2. Mink
The mink (Neovison vison) are mammals that present highly susceptive to the infection of SARS‐CoV 192 and SARS‐CoV‐2. 193 The first outbreak of SARS‐CoV‐2 in the mink farm occurs in the Netherlands, 194 and then infections are reported in more countries. 193 , 195 , 196 , 197 Studies have demonstrated the high transmission among mink through direct contact or airborne, accompanied by adapted mutations. 198 , 199 , 200 , 201 , 202 Notably, the transmission can cause the “back to human” infection and subsequently leads to the prevalence among humans. 198 , 202 , 203 Therefore, measures of prevention and control should be tailored to avoid the potential large‐scale community transmission, regarding the impact of mutants on viral fitness pathogenicity and contagiousness between mink and humans. Intranasal inoculation of SARS‐CoV‐2 induces robust replication of virus in upper and lower respiratory tract, including nasal turbinate, tonsil, soft palate, and trachea, and in lung tissue with peaking on 2 dpi and persisting until 8 dpi before dropping below detection limits on 14 dpi. 204 The viral replication is also observed in gastrointestinal tract, but not in other organs. Infected mink show mild to severe clinical features, such as decreased feed intake and a maximum 20% of body weight loss. 204 Pathological analysis shows that inflammation in nasal turbinate is characterized by infiltration of neutrophils, epithelial degeneration, and necrosis. 194 , 204 , 205 Mucus is the main character that enhances the transmission and causes the olfactory function impairment. 206 Viral infection results in the severe diffuse interstitial pneumonia as well. Characteristic changes in lung tissue include swelling and degeneration of bronchial epithelial cells, thickening of alveolar septa, cellular debris, collagen deposition, hemorrhages, pulmonary edema, pneumocyte proliferation, consolidation and multifocal to diffuse alveolar damage with infiltration of macrophages, monocytes and neutrophils throughout the lungs, and lymphocytes around the vessels. 194 , 204 , 205 The viral infection can cause lethal disease in mink with no gender‐related skewed distribution; however, the naïve mink transmitted by the inoculated ones show alleviated clinical features. 194 , 204 Nevertheless, there is scarcely any study on mink as models to investigate the pathogenesis, therapies, and vaccines evaluation.
6.3. Fruit bats
The bats are mammals that have been shown as natural reservoirs for a variety of RNA viruses, including SARS‐CoV and MERS‐CoV. 207 , 208 , 209 , 210 , 211 These viruses have caused serious transmission between animals and humans and led to the lethal disease. The currently emerged SARS‐CoV‐2 among humans is closely related to β‐coronaviruses which is found in bats, 212 , 213 suggesting that SARS‐CoV‐2 might originate in bats. Intranasally inoculated fruit bats (Rousettus aegyptiacus) with SARS‐CoV‐2 induces viral replication in upper and lower respiratory tract including nasal epithelium, trachea, and in lung and lung‐associated lymphatic tissue with no clinical feature. 170 The viral replication in other organs of duodenum, skin, heart, and adrenal gland tissue is at low levels. 170 However, the transmission can only be found in part of bats at nasal epithelium and trachea. Pathological analysis shows minimal to mild pathological lesions accompanied by cellular debris, edema, and infiltration of neutrophils and lymphocytes, and no pathological change is observed in other organs. 170 The adaptive response is induced by SARS‐CoV‐2 and neutralizing antibodies are produced as early as 8 dpi in fruit bats. 170 Although fruit bats are not used in the therapies and vaccines of countermeasures against SARS‐CoV‐2, studies on the bat model may be required for the pathogenesis and transmission between bats and the potential animals else, or humans, and the evolution.
6.4. Chinese tree shrews
The Chinese tree shrews (Tupaia belangeri chinensis) are mammals that are genetically closer to humans than to rodents, and have been used as models for viral infections, including hepatitis viruses (e.g., HBV and HCV), 214 , 215 , 216 arboviruses (Zika virus and dengue virus) 217 , 218 and respiratory viruses (e.g., influenza viruses and human adenovirus B), 219 , 220 and so forth. The receptor ACE2 gives an overall homology of 85.47% compared with that of humans and shows affinity to the SARS‐CoV‐2, and is highly expressed in the organs of kidney, lung, liver, spleen, and spinal marrow. 221 The TMPRSS2and type II transmembrane serine protease 4 (TMPRSS4), two receptors for SARS‐CoV‐2 infection, are also expressed in lung, liver, esophagus, intestine, and kidney. 221 Combined oral, intranasal, and ocular inoculation of SARS‐CoV‐2 induces viral replication in lung tissues, especially in pneumocytes of young tree shrews (1 year old) with peaking on 3 dpi and gradually dropping below detection limits on 14 dpi; the aged tree shrews (5 to 6 years old) are less susceptible to SARS‐CoV‐2 infection compared to the young ones and show a longer duration of virus shedding in lung tissues with peaking on 7 dpi. 222 Interestingly, the male and aged tree shrews have more efficiency in virus shedding than the females. 223 Viral loads in extrapulmonary organs and sera are low or below detection limits. 222 , 223 No clinical features are observed expect the increased body temperature particularly in female animals. 223 Pathological analysis shows the thickened alveolar septa and interstitial hemorrhage in infected tree shrews, accompanied by mild inflammatory infiltrates. 222 , 223 However, some observations such as the injury of extrapulmonary organs (brain, heart, liver, kidney, and spleen) are inconsistent, perhaps because of the difference in inoculation route and challenge stocks. 222 , 223 Although there is scarcely any study on tree shrews as models to investigate the pathogenesis, therapies, and vaccines evaluation against SARS‐CoV‐2 infection because of their disability to recapitulate the most features observed in COVID‐19 patients, more efforts on tree shrews need to be made to demonstrate the potential intermediate host between animals or humans.
6.5. Dogs, pigs, chickens, and ducks
Several studies have shown that animal models including dogs (Canis lupus familiaris), 224 pigs (Sus scrofa domesticus), 170 , 224 chickens (Gallus gallus domesticus), 170 , 224 , 225 , 226 and ducks (Anatinae), 226 are not susceptible to the SARS‐CoV‐2 infection, despite of in silico dock of affinities to SARS‐CoV‐2 for pigs, dogs, 227 , 228 and chickens. 229 Inoculation of SARS‐CoV‐2 induces no clinical features or pathologic changes in these animal models, and the viral replication in chickens, ducks, or pigs which has been reported the infection with SARS‐CoV, 230 is not observed. Therefore, these animal models may be not suitable for investigating the pathogenesis, therapies, and vaccines evaluation against SARS‐CoV‐2.
7. CONCLUSIONS
There are a variety of animal models that have been developed to demonstrate the transmission, pathogenesis, and immunology induced by SARS‐CoV‐2, and to evaluate the immunomodulatory and antiviral drugs and vaccines against COVID‐19. The most important criterion for animal models is to well recapitulate the features that observed in COVID‐19 patients. Genetically modified mice that express hACE2, especially the K18‐hACE2 and CAG‐hACE2 transgenic mice which characterize the severe disease of COVID‐19 patients with ARDS or SIRS, and Syrian hamsters are widely used as rapid models. These animal models are easy to obtain, and have good operability and reproductivity. However, some limitations must be highlighted, such as the different expression of hACE2 among organs in hACE2 transgenic mice, ectopically expressed hACE2 via adenoviral/adenoviral‐associated vectors that may change the tissue or cellular tropism of the virus, the occurrence of key mutations in immunogen such as RBD domain that may lead to the noneffective neutralizations for the mouse‐adapted virus, and the minimal disease in infected naive hamsters. Moreover, NHPs have similar phylogeny and immunology to humans and are widely used for evaluating the protective efficacy of vaccines and drugs; the receptors for SRAS‐CoV‐2 shows different expression in upper and lower respiratory tract, which affects the viral transmission and infection. The association between the expression manner of ACE2 receptors and disease phenotypes need to be considered. Further studies should be required to standardize the challenge stocks, route, and method of viral inoculation, which may lead to the various of diversities in clinical feature, pathology, immunology, and caused mortality.
Understanding the transmission of SARS‐CoV‐2 is vitally important for control of the epidemic disease. At present, Syrian hamsters and ferrets are well characterized as models to study the transmission prevention of SARS‐CoV‐2, because of the comparability of transmission to humans. The cats, mink, fruit bats, and tree shrews are also susceptive to the SARS‐CoV‐2 infection. Moreover, the emerged variants might impart cross‐species transmission of SARS‐CoV‐2 to naïve mice by enhancing receptor binding. Therefore, the potential implications in SARS‐CoV‐2 (naïveté and variants) transmission, such as animal‐to‐animal or animal‐to‐human, and the evolution should be warranted.
To data, all reported animal models cannot completely recapitulate the characters, especially the immunology changes in COVID‐19 patients. It may be beneficial for investigators to apply appropriate options of animal models depending on the scientific goals. Although several models such as mouse and hamster, show age and gender‐related skewed distribution when infected with SARS‐CoV‐2, other risk factors of chronic disease‐related models that correlate to the severe COVID‐19 disease and SARS‐CoV‐2‐induced mortality, including hypertension, diabetes, obesity, and cardiovascular disease, should be further tailored to explore the advanced pathogenesis and the prevention against SARS‐COV‐2. Taken together, animal models provide a fundamental platform to investigate the transmission, pathogenesis, and countermeasures against SARS‐CoV‐2.
CONFLICTS OF INTEREST
All authors declare no competing interests.
AUTHOR CONTRIBUTIONS
Z.B., S.L. and X.P. conceived the manuscript. Z.B. collected references and wrote the manuscript. H.Q. and J.Y. designed the figures. S.L. and X.P. revised the manuscript. All authors commented on the manuscript.
ETHICS APPROVAL
Not applicable.
ACKNOWLEDGMENTS
We thank the reprinted permission of the picture of Figure 3 from the journal of Signal Transduction and Targeted Therapy. This work is supported by the National Natural Science Foundation Regional Innovation and Development (No. U19A2003), National Major Scientific and Technological Special Project for “Significant New Drugs Development” (No. 2018ZX09733001), the Excellent Youth Foundation of Sichuan Scientific Committee Grant in China (No. 2019JDJQ008) and by the Development Program of China (No. 2016YFA0201402).
Bi Z, Hong W, Yang J, Lu S, Peng X. Animal models for SARS‐CoV‐2 infection and pathology. MedComm. 2021;2:548–568. 10.1002/mco2.98
Contributor Information
Shuaiyao Lu, Email: lushuaiyao-km@163.com.
Xiaozhong Peng, Email: pengxiaozhong@pumc.edu.cn.
DATA AVAILABILITY STATEMENT
Not applicable.
REFERENCES
- 1. Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Tang D, Comish P, Kang R. The hallmarks of COVID‐19 disease. PLoS Pathog. 2020;16(5):e1008536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382(18):1708–1720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Xu Z, Shi L, Wang Y, et al. Pathological findings of COVID‐19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8(4):420–422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Mehta P, McAuley DF, Brown M, et al. COVID‐19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395(10229):1033–1034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Verity R, Okell LC, Dorigatti I, et al. Estimates of the severity of coronavirus disease 2019: a model‐based analysis. Lancet Infect Dis. 2020;20(6):669–677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Mehra MR, Desai SS, Kuy S, Henry TD, Patel AN. Cardiovascular disease, drug therapy, and mortality in Covid‐19. N Engl J Med. 2020;382(25):e102. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 8. Rubino F, Amiel SA, Zimmet P, et al. New‐onset diabetes in Covid‐19. N Engl J Med. 2020;383(8):789–790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Wu C, Chen X, Cai Y, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020;180(7):934–943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Kumari P, Rawat K, Saha L. Pipeline pharmacological therapies in clinical trial for COVID‐19 pandemic: a recent update. Curr Pharmacol Rep. 2020:1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Hodgson J. The pandemic pipeline. Nat Biotechnol. 2020;38(5):523–532. [DOI] [PubMed] [Google Scholar]
- 12. Li K, Wohlford‐Lenane CL, Channappanavar R, et al. Mouse‐adapted MERS coronavirus causes lethal lung disease in human DPP4 knockin mice. Proc Natl Acad Sci U S A. 2017;114(15):E3119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Cockrell AS, Yount BL, Scobey T, et al. A mouse model for MERS coronavirus‐induced acute respiratory distress syndrome. Nat Microbiol. 2016;2(2):16226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Day CW, Baric R, Cai SX, et al. A new mouse‐adapted strain of SARS‐CoV as a lethal model for evaluating antiviral agents in vitro and in vivo. Virology. 2009;395(2):210–222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Channappanavar R, Fehr AR, Vijay R, et al. Dysregulated type I interferon and inflammatory monocyte‐macrophage responses cause lethal pneumonia in SARS‐CoV‐infected mice. Cell Host Microbe. 2016;19(2):181–193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Hoffmann M, Kleine‐Weber H, Schroeder S, et al. SARS‐CoV‐2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(2):271–280. e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Bao L, Deng W, Huang B, et al. The pathogenicity of SARS‐CoV‐2 in hACE2 transgenic mice. Nature. 2020;583(7818):830–833. [DOI] [PubMed] [Google Scholar]
- 18. Larvie M, Lev MH, Hess CP. More on neurologic features in severe SARS‐CoV‐2 infection. N Engl J Med. 2020;382(26):e110. [DOI] [PubMed] [Google Scholar]
- 19. Helms J, Kremer S, Merdji H, et al. Neurologic features in severe SARS‐CoV‐2 infection. N Engl J Med. 2020;382(23):2268–2270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Belhadjer Z, Meot M, Bajolle F, et al. Acute heart failure in multisystem inflammatory syndrome in children in the context of global SARS‐CoV‐2 pandemic. Circulation. 2020;142(5):429–436. [DOI] [PubMed] [Google Scholar]
- 21. Lindner D, Fitzek A, Brauninger H, et al. Association of cardiac infection with SARS‐CoV‐2 in confirmed COVID‐19 autopsy cases. JAMA Cardiol. 2020;5(11):1281–1285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Cardenas Suri H, Jimomila Bening D. Letter by Cardenas Suri and Jimomila Bening regarding article, “Acute heart failure in multisystem inflammatory syndrome in children in the context of global SARS‐CoV‐2 pandemic”. Circulation. 2021;143(10):e757‐e758. [DOI] [PubMed] [Google Scholar]
- 23. Sun SH, Chen Q, Gu HJ, et al. A mouse model of SARS‐CoV‐2 infection and pathogenesis. Cell Host Microbe. 2020;28(1):124–133. e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Ye Q, Zhou J, He Q, et al. SARS‐CoV‐2 infection in the mouse olfactory system. Cell Discov. 2021;7(1):49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Hong W, Yang J, Bi Z, et al. A mouse model for SARS‐CoV‐2‐induced acute respiratory distress syndrome. Signal Transduct Target Ther. 2021;6(1):1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Liu FL, Wu KX, Sun JY, et al. Rapid generation of ACE2 humanized inbred mouse model for COVID‐19 with tetraploid complementation. Natl Sci Rev. 2021;8(2):nwaa285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Yang X, Pabon L, Murry CE. Engineering adolescence: maturation of human pluripotent stem cell‐derived cardiomyocytes. Circ Res. 2014;114(3):511–523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. McCray PB Jr, Pewe L, Wohlford‐Lenane C, et al. Lethal infection of K18‐hACE2 mice infected with severe acute respiratory syndrome coronavirus. J Virol. 2007;81(2):813–821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Silvas JA, Morales Vasquez D, Park JG, et al. Contribution of SARS‐CoV‐2 accessory proteins to viral pathogenicity in K18 hACE2 transgenic mice. J Virol. 2021;95(17):e0040221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Winkler ES, Bailey AL, Kafai NM, et al. SARS‐CoV‐2 infection of human ACE2‐transgenic mice causes severe lung inflammation and impaired function. Nat Immunol. 2020;21(11):1327–1335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Golden JW, Cline CR, Zeng X, et al. Human angiotensin‐converting enzyme 2 transgenic mice infected with SARS‐CoV‐2 develop severe and fatal respiratory disease. JCI Insight. 2020;5(19):e142032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Kumari P, Rothan HA, Natekar JP, et al. Neuroinvasion and encephalitis following intranasal inoculation of SARS‐CoV‐2 in K18‐hACE2 mice. Viruses. 2021;13(1):132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Gan ES, Syenina A, Linster M, et al. A mouse model of lethal respiratory dysfunction for SARS‐CoV‐2 infection. Antiviral Res. 2021;193:105138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Zheng J, Wong LR, Li K, et al. COVID‐19 treatments and pathogenesis including anosmia in K18‐hACE2 mice. Nature. 2021;589(7843):603–607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Moreau GB, Burgess SL, Sturek JM, Donlan AN, Petri WA, Mann BJ. Evaluation of K18‐hACE2 mice as a model of SARS‐CoV‐2 infection. Am J Trop Med Hyg. 2020;103(3):1215–1219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Karki R, Sharma BR, Tuladhar S, et al. Synergism of TNF‐alpha and IFN‐gamma triggers inflammatory cell death, tissue damage, and mortality in SARS‐CoV‐2 infection and cytokine shock syndromes. Cell. 2021;184(1):149–168. e17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Oladunni FS, Park JG, Pino PA, et al. Lethality of SARS‐CoV‐2 infection in K18 human angiotensin‐converting enzyme 2 transgenic mice. Nat Commun. 2020;11(1):6122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Carossino M, Montanaro P, O'Connell A, et al. Fatal neuroinvasion of SARS‐CoV‐2 in K18‐hACE2 mice is partially dependent on hACE2 expression. bioRxiv. 2021. [Google Scholar]
- 39. Li Z, Liu T, Yang N, et al. Neurological manifestations of patients with COVID‐19: potential routes of SARS‐CoV‐2 neuroinvasion from the periphery to the brain. Front Med. 2020;14(5):533–541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Horspool AM, Ye C, Wong TY, et al. SARS‐CoV‐2 B.1.1.7 and B.1.351 variants of concern induce lethal disease in K18‐hACE2 transgenic mice despite convalescent plasma therapy. bioRxiv. 2021. [Google Scholar]
- 41. Seo SH, Jang Y. Cold‐adapted live attenuated SARS‐Cov‐2 vaccine completely protects human ACE2 transgenic mice from SARS‐Cov‐2 infection. Vaccines (Basel). 2020;8(4):584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Elia U, Rotem S, Bar‐Haim E, et al. Lipid nanoparticle RBD‐hFc mRNA vaccine protects hACE2 transgenic mice against a lethal SARS‐CoV‐2 infection. Nano Lett. 2021;21(11):4774–4779. [DOI] [PubMed] [Google Scholar]
- 43. Liu R, Americo JL, Cotter CA, et al. One or two injections of MVA‐vectored vaccine shields hACE2 transgenic mice from SARS‐CoV‐2 upper and lower respiratory tract infection. Proc Natl Acad Sci U S A. 2021;118(12):e2026785118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. An D, Li K, Rowe DK, et al. Protection of K18‐hACE2 mice and ferrets against SARS‐CoV‐2 challenge by a single‐dose mucosal immunization with a parainfluenza virus 5‐based COVID‐19 vaccine. Sci Adv. 2021;7(27):eabi5246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Rosenfeld R, Noy‐Porat T, Mechaly A, et al. Post‐exposure protection of SARS‐CoV‐2 lethal infected K18‐hACE2 transgenic mice by neutralizing human monoclonal antibody. Nat Commun. 2021;12(1):944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Noy‐Porat T, Mechaly A, Levy Y, et al. Therapeutic antibodies, targeting the SARS‐CoV‐2 spike N‐terminal domain, protect lethally infected K18‐hACE2 mice. iScience. 2021;24(5):102479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Dampalla CS, Zheng J, Perera KD, et al. Postinfection treatment with a protease inhibitor increases survival of mice with a fatal SARS‐CoV‐2 infection. Proc Natl Acad Sci U S A. 2021;118(29):e2101555118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Caceres CJ, Cardenas‐Garcia S, Carnaccini S, et al. Efficacy of GC‐376 against SARS‐CoV‐2 virus infection in the K18 hACE2 transgenic mouse model. Sci Rep. 2021;11(1):9609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Ostrowski LE, Hutchins JR, Zakel K, O'Neal WK. Targeting expression of a transgene to the airway surface epithelium using a ciliated cell‐specific promoter. Mol Ther. 2003;8(4):637–645. [DOI] [PubMed] [Google Scholar]
- 50. Menachery VD, Yount BL Jr, Sims AC, et al. SARS‐like WIV1‐CoV poised for human emergence. Proc Natl Acad Sci U S A. 2016;113(11):3048–3053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Netland J, Meyerholz DK, Moore S, Cassell M, Perlman S. Severe acute respiratory syndrome coronavirus infection causes neuronal death in the absence of encephalitis in mice transgenic for human ACE2. J Virol. 2008;82(15):7264–7275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Li S, Zhang Y, Guan Z, et al. SARS‐CoV‐2 triggers inflammatory responses and cell death through caspase‐8 activation. Signal Transduct Target Ther. 2020;5(1):235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Musiu C, Caligola S, Fiore A, et al. Fatal cytokine release syndrome by an aberrant FLIP/STAT3 axis. Cell Death Differ. 2021:1–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Jiang RD, Liu MQ, Chen Y, et al. Pathogenesis of SARS‐CoV‐2 in transgenic mice expressing human angiotensin‐converting enzyme 2. Cell. 2020;182(1):50–58. e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Hou YJ, Chiba S, Halfmann P, et al. SARS‐CoV‐2 D614G variant exhibits enhanced replication ex vivo and earlier transmission in vivo. bioRxiv. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Ku M‐W, Authié P, Bourgine M, et al. Full brain and lung prophylaxis against SARS‐CoV‐2 by intranasal lentiviral vaccination in a new hACE2 transgenic mouse model or golden hamsters. bioRxiv. 2021. [Google Scholar]
- 57. Yoshikawa N, Yoshikawa T, Hill T, et al. Differential virological and immunological outcome of severe acute respiratory syndrome coronavirus infection in susceptible and resistant transgenic mice expressing human angiotensin‐converting enzyme 2. J Virol. 2009;83(11):5451–5465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Tseng CT, Huang C, Newman P, et al. Severe acute respiratory syndrome coronavirus infection of mice transgenic for the human angiotensin‐converting enzyme 2 virus receptor. J Virol. 2007;81(3):1162–1173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Tsai C‐Y, Chen C‐Y, Jan J‐T, et al. Sex‐biased response to and brain cell infection by SARS‐CoV‐2 in a highly susceptible human ACE2 transgenic model. bioRxiv. 2021. [Google Scholar]
- 60. Xia S, Lan Q, Zhu Y, et al. Structural and functional basis for pan‐CoV fusion inhibitors against SARS‐CoV‐2 and its variants with preclinical evaluation. Signal Transduct Target Ther. 2021;6(1):288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Liao H‐Y, Huang H‐Y, Chen X, et al. Impact of glycosylation on SARS‐CoV‐2 infection and broadly protective vaccine design. bioRxiv. 2021. [Google Scholar]
- 62. Sun J, Zhuang Z, Zheng J, et al. Generation of a broadly useful model for COVID‐19 pathogenesis, vaccination, and treatment. Cell. 2020;182(3):734–743. e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Han K, Blair RV, Iwanaga N, et al. Lung expression of human angiotensin‐converting enzyme 2 sensitizes the mouse to SARS‐CoV‐2 infection. Am J Respir Cell Mol Biol. 2021;64(1):79–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Gu T, Zhao S, Jin G, et al. Cytokine signature induced by SARS‐CoV‐2 spike protein in a mouse model. Front Immunol. 2020;11:621441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Liu F, Han K, Blair R, et al. SARS‐CoV‐2 infects endothelial cells in vivo and in vitro. Front Cell Infect Microbiol. 2021;11:701278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Geng T, Keilich S, Tafas T, Wang P. Early detection of SARS‐CoV‐2 in circulating immune cells in a mouse model. bioRxiv. 2021. [Google Scholar]
- 67. Israelow B, Song E, Mao T, et al. Mouse model of SARS‐CoV‐2 reveals inflammatory role of type I interferon signaling. J Exp Med. 2020;217(12):e20201241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Sefik E, Israelow B, Zhao J, et al. A humanized mouse model of chronic COVID‐19 to evaluate disease mechanisms and treatment options. Res Sq. 2021. rs.3.rs‐279341. [Google Scholar]
- 69. Rathnasinghe R, Strohmeier S, Amanat F, et al. Comparison of transgenic and adenovirus hACE2 mouse models for SARS‐CoV‐2 infection. Emerg Microbes Infect. 2020;9(1):2433–2445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Gu C, Wu Y, Guo H, et al. Protoporphyrin IX and verteporfin potently inhibit SARS‐CoV‐2 infection in vitro and in a mouse model expressing human ACE2. Sci Bull (Beijing). 2021;66(9):925–936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Fu D, Zhang G, Wang Y, et al. Structural basis for SARS‐CoV‐2 neutralizing antibodies with novel binding epitopes. PLoS Biol. 2021;19(5):e3001209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Guirakhoo F, Kuo L, Peng J, et al. A novel SARS‐CoV‐2 multitope protein/peptide vaccine candidate is highly immunogenic and prevents lung infection in an adeno associated virus human angiotensin‐converting enzyme 2 (AAV hACE2) mouse model. bioRxiv. 2020. [Google Scholar]
- 73. Li Y, Cao L, Li G, et al. Remdesivir metabolite GS‐441524 effectively inhibits SARS‐CoV‐2 infection in mouse models. J Med Chem. 2021. acs.jmedchem.0c01929. [DOI] [PubMed] [Google Scholar]
- 74. Roberts A, Deming D, Paddock CD, et al. A mouse‐adapted SARS‐coronavirus causes disease and mortality in BALB/c mice. PLoS Pathog. 2007;3(1):e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Gu H, Chen Q, Yang G, et al. Adaptation of SARS‐CoV‐2 in BALB/c mice for testing vaccine efficacy. Science. 2020;369(6511):1603–1607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Sun S, Gu H, Cao L, et al. Characterization and structural basis of a lethal mouse‐adapted SARS‐CoV‐2. Nat Commun. 2021;12(1):5654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Leist SR, Dinnon KH Schafer A III, et al. A mouse‐adapted SARS‐CoV‐2 induces acute lung injury and mortality in standard laboratory mice. Cell. 2020;183(4):1070–1085. e12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Wang J, Shuai L, Wang C, et al. Mouse‐adapted SARS‐CoV‐2 replicates efficiently in the upper and lower respiratory tract of BALB/c and C57BL/6J mice. Protein Cell. 2020;11(10):776–782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Montagutelli X, Prot M, Jouvion G, et al. A mouse‐adapted SARS‐CoV‐2 strain replicating in standard laboratory mice. bioRxiv. 2021. [Google Scholar]
- 80.Wong LYR, Zheng J, Wilhelmsen K, et al. Eicosanoid signaling as a therapeutic target in middle‐aged mice with severe COVID‐19. bioRxiv. 2021. [Google Scholar]
- 81. Dinnon KH III, Leist SR, Schafer A, et al. A mouse‐adapted model of SARS‐CoV‐2 to test COVID‐19 countermeasures. Nature. 2020;586(7830):560–566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Zhang NN, Li XF, Deng YQ, et al. A thermostable mRNA vaccine against COVID‐19. Cell. 2020;182(5):1271–1283. e16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Weston S, Coleman CM, Haupt R, et al. Broad anti‐coronavirus activity of Food and Drug Administration‐approved drugs against SARS‐CoV‐2 in vitro and SARS‐CoV in vivo. J Virol. 2020;94(21):e01218‐20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. DiPiazza AT, Leist SR, Abiona OM, et al. COVID‐19 vaccine mRNA‐1273 elicits a protective immune profile in mice that is not associated with vaccine‐enhanced disease upon SARS‐CoV‐2 challenge. Immunity. 2021;54(8):1869–1882. e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Gralinski LE, Ferris MT, Aylor DL, et al. Genome wide identification of SARS‐CoV susceptibility loci using the collaborative cross. PLoS Genet. 2015;11(10):e1005504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Graham RL, Becker MM, Eckerle LD, Bolles M, Denison MR, Baric RS. A live, impaired‐fidelity coronavirus vaccine protects in an aged, immunocompromised mouse model of lethal disease. Nat Med. 2012;18(12):1820–1826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Fu W, Wang W, Yuan L, et al. A SCID mouse‐human lung xenograft model of SARS‐CoV‐2 infection. Theranostics. 2021;11(13):6607–6615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Torres Acosta MA, Singer BD. Pathogenesis of COVID‐19‐induced ARDS: implications for an ageing population. Eur Respir J. 2020;56(3):2002049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Falach R, Bar‐On L, Lazar S, et al. Mice with induced pulmonary morbidities display severe lung inflammation and mortality following exposure to SARS‐CoV‐2. JCI Insight. 2021;6(12):e145916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Gu T, Zhao S, Jin G, et al. Molecular mechanism of SARS‐CoV‐2 components caused ARDS in murine model. bioRxiv. 2020. [Google Scholar]
- 91. Xia B, Shen X, He Y, et al. SARS‐CoV‐2 envelope protein causes acute respiratory distress syndrome (ARDS)‐like pathological damages and constitutes an antiviral target. Cell Res. 2021;31(8):847–860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Skapare E, Konrade I, Liepinsh E, et al. Glyoxalase 1 and glyoxalase 2 activities in blood and neuronal tissue samples from experimental animal models of obesity and type 2 diabetes mellitus. J Physiol Sci. 2012;62(6):469–478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Kleinert M, Clemmensen C, Hofmann SM, et al. Animal models of obesity and diabetes mellitus. Nat Rev Endocrinol. 2018;14(3):140–162. [DOI] [PubMed] [Google Scholar]
- 94. Camacho P, Fan H, Liu Z, He JQ. Small mammalian animal models of heart disease. Am J Cardiovasc Dis. 2016;6(3):70–80. [PMC free article] [PubMed] [Google Scholar]
- 95. Rathnasinghe R, Jangra S, Cupic A, et al. The N501Y mutation in SARS‐CoV‐2 spike leads to morbidity in obese and aged mice and is neutralized by convalescent and post‐vaccination human sera. medRxiv. 2021. [Google Scholar]
- 96. Niu Z, Zhang Z, Gao X, et al. N501Y mutation imparts cross‐species transmission of SARS‐CoV‐2 to mice by enhancing receptor binding. Signal Transduct Target Ther. 2021;6(1):284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Huang H, Zhu Y, Niu Z, Zhou L, Sun Q. SARS‐CoV‐2 N501Y variants of concern and their potential transmission by mouse. Cell Death Differ. 2021;28(10):2840–2842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Li Q, Nie J, Wu J, et al. SARS‐CoV‐2 501Y.V2 variants lack higher infectivity but do have immune escape. Cell. 2021;184(9):2362–2371. e9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Roberts A, Vogel L, Guarner J, et al. Severe acute respiratory syndrome coronavirus infection of golden Syrian hamsters. J Virol. 2005;79(1):503–511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Miao J, Chard LS, Wang Z, Wang Y. Syrian hamster as an animal model for the study on infectious diseases. Front Immunol. 2019;10:2329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Iwatsuki‐Horimoto K, Nakajima N, Ichiko Y, et al. Syrian hamster as an animal model for the study of human influenza virus infection. J Virol. 2018;92(4):e01693‐17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. van Doremalen N, Miazgowicz KL, Munster VJ. Mapping the specific amino acid residues that make hamster DPP4 functional as a receptor for middle east respiratory syndrome coronavirus. J Virol. 2016;90(11):5499–5502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Chan JF, Zhang AJ, Yuan S, et al. Simulation of the clinical and pathological manifestations of Coronavirus Disease 2019 (COVID‐19) in a golden Syrian hamster model: implications for disease pathogenesis and transmissibility. Clin Infect Dis. 2020;71(9):2428–2446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Tostanoski LH, Wegmann F, Martinot AJ, et al. Ad26 vaccine protects against SARS‐CoV‐2 severe clinical disease in hamsters. Nat Med. 2020;26(11):1694–1700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Sia SF, Yan LM, Chin AWH, et al. Pathogenesis and transmission of SARS‐CoV‐2 in golden hamsters. Nature. 2020;583(7818):834–838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Osterrieder N, Bertzbach LD, Dietert K, et al. Age‐dependent progression of SARS‐CoV‐2 infection in Syrian hamsters. Viruses. 2020;12(7):779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Imai M, Iwatsuki‐Horimoto K, Hatta M, et al. Syrian hamsters as a small animal model for SARS‐CoV‐2 infection and countermeasure development. Proc Natl Acad Sci U S A. 2020;117(28):16587–16595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Song Z, Bao L, Yu P, et al. SARS‐CoV‐2 causes a systemically multiple organs damages and dissemination in hamsters. Front Microbiol. 2020;11:618891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Zhang AJ, Lee AC, Chu H, et al. Severe acute respiratory syndrome coronavirus 2 infects and damages the mature and immature olfactory sensory neurons of hamsters. Clin Infect Dis. 2021;73(2):e503‐e512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Urata S, Maruyama J, Kishimoto‐Urata M, et al. Regeneration profiles of olfactory epithelium after SARS‐CoV‐2 infection in golden Syrian hamsters. ACS Chem Neurosci. 2021;12(4):589–595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Bryche B, St Albin A, Murri S, et al. Massive transient damage of the olfactory epithelium associated with infection of sustentacular cells by SARS‐CoV‐2 in golden Syrian hamsters. Brain Behav Immun. 2020;89:579–586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Lee AC, Zhang AJ, Chan JF, et al. Oral SARS‐CoV‐2 inoculation establishes subclinical respiratory infection with virus shedding in golden Syrian hamsters. Cell Rep Med. 2020;1(7):100121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Rosenke K, Meade‐White K, Letko M, et al. Defining the Syrian hamster as a highly susceptible preclinical model for SARS‐CoV‐2 infection. Emerg Microbes Infect. 2020;9(1):2673–2684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Yuan L, Zhu H, Zhou M, et al. Gender associates with both susceptibility to infection and pathogenesis of SARS‐CoV‐2 in Syrian hamster. Signal Transduct Target Ther. 2021;6(1):136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. Chan JF, Yuan S, Zhang AJ, et al. Surgical mask partition reduces the risk of noncontact transmission in a golden Syrian hamster model for Coronavirus Disease 2019 (COVID‐19). Clin Infect Dis. 2020;71(16):2139–2149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Abdelnabi R, Boudewijns R, Foo CS, et al. Comparing infectivity and virulence of emerging SARS‐CoV‐2 variants in Syrian hamsters. EBioMedicine. 2021;68:103403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117. Ye ZW, Yuan S, Chan JF, et al. Beneficial effect of combinational methylprednisolone and remdesivir in hamster model of SARS‐CoV‐2 infection. Emerg Microbes Infect. 2021;10(1):291–304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Boudewijns R, Thibaut HJ, Kaptein SJF, et al. STAT2 signaling restricts viral dissemination but drives severe pneumonia in SARS‐CoV‐2 infected hamsters. Nat Commun. 2020;11(1):5838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Mahlakoiv T, Ritz D, Mordstein M, et al. Combined action of type I and type III interferon restricts initial replication of severe acute respiratory syndrome coronavirus in the lung but fails to inhibit systemic virus spread. J Gen Virol. 2012;93(12):2601–2605. [DOI] [PubMed] [Google Scholar]
- 120. Brocato RL, Principe LM, Kim RK, et al. Disruption of adaptive immunity enhances disease in SARS‐CoV‐2‐infected Syrian hamsters. J Virol. 2020;94(22):e01683‐20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Selvaraj P, Lien CZ, Liu S, et al. SARS‐CoV‐2 infection induces protective immunity and limits transmission in Syrian hamsters. Life Sci Alliance. 2021;4(4):e202000886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Haagmans BL, Noack D, Okba NMA, et al. SARS‐CoV‐2 neutralizing human antibodies protect against lower respiratory tract disease in a hamster model. J Infect Dis. 2021;223(12):2020–2028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123. Kreye J, Reincke SM, Kornau HC, et al. A therapeutic non‐self‐reactive SARS‐CoV‐2 antibody protects from lung pathology in a COVID‐19 hamster model. Cell. 2020;183(4):1058–1069. e19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Chen Y, Liu L, Wei Q, et al. Rhesus angiotensin converting enzyme 2 supports entry of severe acute respiratory syndrome coronavirus in Chinese macaques. Virology. 2008;381(1):89–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Ziegler CGK, Allon SJ, Nyquist SK, et al. SARS‐CoV‐2 receptor ACE2 is an interferon‐stimulated gene in human airway epithelial cells and is detected in specific cell subsets across tissues. Cell. 2020;181(5):1016–1035. e19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. McAuliffe J, Vogel L, Roberts A, et al. Replication of SARS coronavirus administered into the respiratory tract of African Green, rhesus and cynomolgus monkeys. Virology. 2004;330(1):8–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Munster VJ, Feldmann F, Williamson BN, et al. Respiratory disease in rhesus macaques inoculated with SARS‐CoV‐2. Nature. 2020;585(7824):268–272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Zheng H, Li H, Guo L, et al. Virulence and pathogenesis of SARS‐CoV‐2 infection in rhesus macaques: a nonhuman primate model of COVID‐19 progression. PLoS Pathog. 2020;16(11):e1008949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129. Chang MC, Hild S, Grieder F. Nonhuman primate models for SARS‐CoV‐2 research: consider alternatives to macaques. Lab Anim (NY). 2021;50(5):113–114. [DOI] [PubMed] [Google Scholar]
- 130. Blair RV, Vaccari M, Doyle‐Meyers LA, et al. Acute respiratory distress in aged, SARS‐CoV‐2‐infected African Green monkeys but not rhesus macaques. Am J Pathol. 2021;191(2):274–282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131. Fahlberg MD, Blair RV, Doyle‐Meyers LA, et al. Cellular events of acute, resolving or progressive COVID‐19 in SARS‐CoV‐2 infected non‐human primates. Nat Commun. 2020;11(1):6078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132. Böszörményi KP, Stammes MA, Fagrouch ZC, et al. The post‐acute phase of SARS‐CoV‐2 infection in two macaques species is associated with signs of ongoing virus replication and pathology in pulmonary and extrapulmonary tissues. Viruses. 2021;13(8):1673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Salguero FJ, White AD, Slack GS, et al. Comparison of rhesus and cynomolgus macaques as an infection model for COVID‐19. Nat Commun. 2021;12(1):1260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134. Lu S, Zhao Y, Yu W, et al. Comparison of nonhuman primates identified the suitable model for COVID‐19. Signal Transduct Target Ther. 2020;5(1):157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135. Woolsey C, Borisevich V, Prasad AN, et al. Establishment of an African green monkey model for COVID‐19 and protection against re‐infection. Nat Immunol. 2021;22(1):86–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136. Shan C, Yao YF, Yang XL, et al. Infection with novel coronavirus (SARS‐CoV‐2) causes pneumonia in rhesus macaques. Cell Res. 2020;30(8):670–677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137. Deng W, Bao L, Liu J, et al. Primary exposure to SARS‐CoV‐2 protects against reinfection in rhesus macaques. Science. 2020;369(6505):818–823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Jiao L, Yang Y, Yu W, et al. The olfactory route is a potential way for SARS‐CoV‐2 to invade the central nervous system of rhesus monkeys. Signal Transduct Target Ther. 2021;6(1):169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139. Jiao L, Li H, Xu J, et al. The gastrointestinal tract is an alternative route for SARS‐CoV‐2 infection in a nonhuman primate model. Gastroenterology. 2021;160(5):1647–1661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Brann DH, Tsukahara T, Weinreb C, et al. Non‐neuronal expression of SARS‐CoV‐2 entry genes in the olfactory system suggests mechanisms underlying COVID‐19‐associated anosmia. Sci Adv. 2020;6(31):eabc5801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141. Song TZ, Han JB, Yang X, Li MH, Zheng YT. Tissue distribution of SARS‐CoV‐2 in non‐human primates after combined intratracheal and intranasal inoculation. Sci China Life Sci. 2021;64(10):1792–1794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142. Cross RW, Agans KN, Prasad AN, et al. Intranasal exposure of African green monkeys to SARS‐CoV‐2 results in acute phase pneumonia with shedding and lung injury still present in the early convalescence phase. Virol J. 2020;17(1):125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143. Yu P, Qi F, Xu Y, et al. Age‐related rhesus macaque models of COVID‐19. Animal Model Exp Med. 2020;3(1):93–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144. Chandrashekar A, Liu J, Martinot AJ, et al. SARS‐CoV‐2 infection protects against rechallenge in rhesus macaques. Science. 2020;369(6505):812–817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145. Fischer T, Rutkai I, Mayer M, et al. Neuropathology and virus in brain of SARS‐CoV‐2 infected non‐human primates. Res Sq. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146. Singh DK, Singh B, Ganatra SR, et al. Responses to acute infection with SARS‐CoV‐2 in the lungs of rhesus macaques, baboons and marmosets. Nat Microbiol. 2021;6(1):73–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147. Song TZ, Zheng HY, Han JB, et al. Delayed severe cytokine storm and immune cell infiltration in SARS‐CoV‐2‐infected aged Chinese rhesus macaques. Zool Res. 2020;41(5):503–516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148. Mercado NB, Zahn R, Wegmann F, et al. Single‐shot Ad26 vaccine protects against SARS‐CoV‐2 in rhesus macaques. Nature. 2020;586(7830):583–588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149. Yu J, Tostanoski LH, Peter L, et al. DNA vaccine protection against SARS‐CoV‐2 in rhesus macaques. Science. 2020;369(6505):806–811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150. Williamson BN, Feldmann F, Schwarz B, et al. Clinical benefit of remdesivir in rhesus macaques infected with SARS‐CoV‐2. Nature. 2020;585(7824):273–276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151. van Doremalen N, Lambe T, Spencer A, et al. ChAdOx1 nCoV‐19 vaccine prevents SARS‐CoV‐2 pneumonia in rhesus macaques. Nature. 2020;586(7830):578–582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152. Li Y, Bi Y, Xiao H, et al. A novel DNA and protein combination COVID‐19 vaccine formulation provides full protection against SARS‐CoV‐2 in rhesus macaques. Emerg Microbes Infect. 2021;10(1):342–355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153. Maher JA, DeStefano J. The ferret: an animal model to study influenza virus. Lab Anim (NY). 2004;33(9):50–53. [DOI] [PubMed] [Google Scholar]
- 154. Karlsson EA, Meliopoulos VA, Savage C, Livingston B, Mehle A, Schultz‐Cherry S. Visualizing real‐time influenza virus infection, transmission and protection in ferrets. Nat Commun. 2015;6:6378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155. Chu YK, Ali GD, Jia F, et al. The SARS‐CoV ferret model in an infection‐challenge study. Virology. 2008;374(1):151–163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156. Johnson‐Delaney CA, Orosz SE. Ferret respiratory system: clinical anatomy, physiology, and disease. Vet Clin North Am Exot Anim Pract. 2011;14(2):357–367, vii. [DOI] [PubMed] [Google Scholar]
- 157. Enkirch T, von Messling V. Ferret models of viral pathogenesis. Virology. 2015;479‐480:259–270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158. van den Brand JM, Haagmans BL, Leijten L, et al. Pathology of experimental SARS coronavirus infection in cats and ferrets. Vet Pathol. 2008;45(4):551–562. [DOI] [PubMed] [Google Scholar]
- 159. Martina BE, Haagmans BL, Kuiken T, et al. Virology: sARS virus infection of cats and ferrets. Nature. 2003;425(6961):915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160. Kim YI, Yu KM, Koh JY, et al. Age‐dependent pathogenic characteristics of SARS‐CoV‐2 infection in ferrets. Res Sq. 2021. rs.3.rs‐131380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161. Kim YI, Kim SG, Kim SM, et al. Infection and rapid transmission of SARS‐CoV‐2 in ferrets. Cell Host Microbe. 2020;27(5):704–709. e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162. Ryan KA, Bewley KR, Fotheringham SA, et al. Dose‐dependent response to infection with SARS‐CoV‐2 in the ferret model and evidence of protective immunity. Nat Commun. 2021;12(1):81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163. Everett HE, Lean FZX, Byrne AMP, et al. Intranasal infection of ferrets with SARS‐CoV‐2 as a model for asymptomatic human infection. Viruses. 2021;13(1):113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164. van de Ven K, van Dijken H, Wijsman L, et al. Pathology and immunity after SARS‐CoV‐2 infection in male ferrets is affected by age and inoculation route. bioRxiv. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165. Kreijtz JH, Kroeze EJ, Stittelaar KJ, et al. Low pathogenic avian influenza A(H7N9) virus causes high mortality in ferrets upon intratracheal challenge: a model to study intervention strategies. Vaccine. 2013;31(43):4995–4999. [DOI] [PubMed] [Google Scholar]
- 166. de Jonge J, Isakova‐Sivak I, van Dijken H, et al. H7N9 live attenuated influenza vaccine is highly immunogenic, prevents virus replication, and protects against severe bronchopneumonia in ferrets. Mol Ther. 2016;24(5):991–1002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167. Bodewes R, Kreijtz JH, van Amerongen G, et al. Pathogenesis of influenza A/H5N1 virus infection in ferrets differs between intranasal and intratracheal routes of inoculation. Am J Pathol. 2011;179(1):30–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168. Francis ME, Richardson B, McNeil M, et al. Male sex and age biases viral burden, viral shedding, and type 1 and 2 interferon responses during SARS‐CoV‐2 infection in ferrets. bioRxiv. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169. Francis ME, Richardson B, Goncin U, et al. Sex and age bias viral burden and interferon responses during SARS‐CoV‐2 infection in ferrets. Sci Rep. 2021;11(1):14536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170. Schlottau K, Rissmann M, Graaf A, et al. SARS‐CoV‐2 in fruit bats, ferrets, pigs, and chickens: an experimental transmission study. Lancet Microbe. 2020;1(5):E218‐E225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171. Patel DR, Field CJ, Septer KM, et al. Transmission and protection against reinfection in the ferret model with the SARS‐CoV‐2 USA‐WA1/2020 reference isolate. J Virol. 2021;95(13):e0223220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172. Richard M, Kok A, de Meulder D, et al. SARS‐CoV‐2 is transmitted via contact and via the air between ferrets. Nat Commun. 2020;11(1):3496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173. Kutter JS, de Meulder D, Bestebroer TM, et al. SARS‐CoV and SARS‐CoV‐2 are transmitted through the air between ferrets over more than one meter distance. Nat Commun. 2021;12(1):1653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174. de Vries RD, Schmitz KS, Bovier FT, et al. Intranasal fusion inhibitory lipopeptide prevents direct‐contact SARS‐CoV‐2 transmission in ferrets. Science. 2021;371(6536):1379–1382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175. Cox RM, Wolf JD, Plemper RK. Therapeutically administered ribonucleoside analogue MK‐4482/EIDD‐2801 blocks SARS‐CoV‐2 transmission in ferrets. Nat Microbiol. 2021;6(1):11–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176. Lee JS, Koh JY, Yi K, et al. Single‐cell transcriptome of bronchoalveolar lavage fluid reveals sequential change of macrophages during SARS‐CoV‐2 infection in ferrets. Nat Commun. 2021;12(1):4567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177. Marsh GA, McAuley AJ, Au GG, et al. ChAdOx1 nCoV‐19 (AZD1222) vaccine candidate significantly reduces SARS‐CoV‐2 shedding in ferrets. NPJ Vaccines. 2021;6(1):67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178. Kim YI, Kim D, Yu KM, et al. Development of spike receptor‐binding domain nanoparticles as a vaccine candidate against SARS‐CoV‐2 infection in ferrets. mBio. 2021;12(2):e00230‐21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179. Park SJ, Yu KM, Kim YI, et al. Antiviral efficacies of FDA‐approved drugs against SARS‐CoV‐2 infection in ferrets. mBio. 2020;11(3):e01114‐20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180. Patterson EI, Elia G, Grassi A, et al. Evidence of exposure to SARS‐CoV‐2 in cats and dogs from households in Italy. Nat Commun. 2020;11(1):6231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181. Neira V, Brito B, Aguero B, et al. A household case evidences shorter shedding of SARS‐CoV‐2 in naturally infected cats compared to their human owners. Emerg Microbes Infect. 2021;10(1):376–383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182. Fuentealba NA, More G, Bravi ME, et al. First detection and molecular analysis of SARS‐CoV‐2 from a naturally infected cat from Argentina. Vet Microbiol. 2021;260:109179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183. Garigliany M, Van Laere AS, Clercx C, et al. SARS‐CoV‐2 natural transmission from human to cat, Belgium, March 2020. Emerg Infect Dis. 2020;26(12):3069–3071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184. Hosie MJ, Epifano I, Herder V, et al. Detection of SARS‐CoV‐2 in respiratory samples from cats in the UK associated with human‐to‐cat transmission. Vet Rec. 2021;188(8):e247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185. Yang J, Wang W, Chen Z, et al. A vaccine targeting the RBD of the S protein of SARS‐CoV‐2 induces protective immunity. Nature. 2020;586(7830):572–577. [DOI] [PubMed] [Google Scholar]
- 186. Halfmann PJ, Hatta M, Chiba S, et al. Transmission of SARS‐CoV‐2 in domestic cats. N Engl J Med. 2020;383(6):592–594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187. Braun KM, Moreno GK, Halfmann PJ, et al. Transmission of SARS‐CoV‐2 in domestic cats imposes a narrow bottleneck. PLoS Pathog. 2021;17(2):e1009373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188. Bao L, Song Z, Xue J, et al. Susceptibility and attenuated transmissibility of SARS‐CoV‐2 in domestic cats. J Infect Dis. 2021;223(8):1313–1321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189. Gaudreault NN, Carossino M, Morozov I, et al. Experimental re‐infected cats do not transmit SARS‐CoV‐2. Emerg Microbes Infect. 2021;10(1):638–650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190. Bosco‐Lauth AM, Hartwig AE, Porter SM, et al. Experimental infection of domestic dogs and cats with SARS‐CoV‐2: pathogenesis, transmission, and response to reexposure in cats. Proc Natl Acad Sci U S A. 2020;117(42):26382–26388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191. Gaudreault NN, Trujillo JD, Carossino M, et al. SARS‐CoV‐2 infection, disease and transmission in domestic cats. Emerg Microbes Infect. 2020;9(1):2322–2332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192. Shi Z, Hu Z. A review of studies on animal reservoirs of the SARS coronavirus. Virus Res. 2008;133(1):74–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193. Boklund A, Hammer AS, Quaade ML, et al. SARS‐CoV‐2 in Danish mink farms: course of the epidemic and a descriptive analysis of the outbreaks in 2020. Animals (Basel). 2021;11(1):164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194. Oreshkova N, Molenaar RJ, Vreman S, et al. SARS‐CoV‐2 infection in farmed minks, the Netherlands, April and May 2020. Eurosurveillance. 2020;25(23):2–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195. Koopmans M. SARS‐CoV‐2 and the human‐animal interface: outbreaks on mink farms. Lancet Infect Dis. 2021;21(1):18–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196. Aguilo‐Gisbert J, Padilla‐Blanco M, Lizana V, et al. First description of SARS‐CoV‐2 infection in two feral American mink (Neovison vison) caught in the wild. Animals (Basel). 2021;11(5):1422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197. Fenollar F, Mediannikov O, Maurin M, et al. Mink, SARS‐CoV‐2, and the human‐animal interface. Front Microbiol. 2021;12:663815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198. Wang L, Didelot X, Bi Y, Gao GF. Assessing the extent of community spread caused by mink‐derived SARS‐CoV‐2 variants. Innovation (N Y). 2021;2(3):100128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199. Sharun K, Tiwari R, Natesan S, Dhama K. SARS‐CoV‐2 infection in farmed minks, associated zoonotic concerns, and importance of the One Health approach during the ongoing COVID‐19 pandemic. Vet Q. 2021;41(1):50–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200. Hammer AS, Quaade ML, Rasmussen TB, et al. SARS‐CoV‐2 transmission between mink (Neovison vison) and humans, Denmark. Emerg Infect Dis. 2021;27(2):547–551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201. de Rooij MMT, Hakze‐Van der Honing RW, Hulst MM, et al. Occupational and environmental exposure to SARS‐CoV‐2 in and around infected mink farms. Occup Environ Med. 2021. oemed‐2021‐107443. [DOI] [PubMed] [Google Scholar]
- 202. Burkholz S, Pokhrel S, Kraemer BR, et al. Paired SARS‐CoV‐2 spike protein mutations observed during ongoing SARS‐CoV‐2 viral transfer from humans to minks and back to humans. Infect Genet Evol. 2021;93:104897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203. Oude Munnink BB, Sikkema RS, Nieuwenhuijse DF, et al. Transmission of SARS‐CoV‐2 on mink farms between humans and mink and back to humans. Science. 2021;371(6525):172–177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204. Shuai L, Zhong G, Yuan Q, et al. Replication, pathogenicity, and transmission of SARS‐CoV‐2 in minks. Natl Sci Rev. 2020;8(3):nwaa291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205. Molenaar RJ, Vreman S, Hakze‐van der Honing RW, et al. Clinical and pathological findings in SARS‐CoV‐2 disease outbreaks in farmed mink (Neovison vison). Vet Pathol. 2020;57(5):653–657. [DOI] [PubMed] [Google Scholar]
- 206. Chung TW, Sridhar S, Zhang AJ, et al. Olfactory dysfunction in Coronavirus Disease 2019 patients: observational cohort study and systematic review. Open Forum Infect Dis. 2020;7(6):ofaa199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207. Munster VJ, Adney DR, van Doremalen N, et al. Replication and shedding of MERS‐CoV in Jamaican fruit bats (Artibeus jamaicensis). Sci Rep. 2016;6:21878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208. Ge XY, Li JL, Yang XL, et al. Isolation and characterization of a bat SARS‐like coronavirus that uses the ACE2 receptor. Nature. 2013;503(7477):535–538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209. Lau SK, Woo PC, Li KS, et al. Severe acute respiratory syndrome coronavirus‐like virus in Chinese horseshoe bats. Proc Natl Acad Sci U S A. 2005;102(39):14040–14045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210. Drosten C, Meyer B, Muller MA, et al. Transmission of MERS‐coronavirus in household contacts. N Engl J Med. 2014;371(9):828–835. [DOI] [PubMed] [Google Scholar]
- 211. Li W, Shi Z, Yu M, et al. Bats are natural reservoirs of SARS‐like coronaviruses. Science. 2005;310(5748):676–679. [DOI] [PubMed] [Google Scholar]
- 212. Wu F, Zhao S, Yu B, et al. A new coronavirus associated with human respiratory disease in China. Nature. 2020;579(7798):265–269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213. Zhou P, Yang XL, Wang XG, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579(7798):270–273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214. Yang C, Ruan P, Ou C, et al. Chronic hepatitis B virus infection and occurrence of hepatocellular carcinoma in tree shrews (Tupaia belangeri chinensis). Virol J. 2015;12:26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215. Kayesh MEH, Ezzikouri S, Chi H, et al. Interferon‐beta response is impaired by hepatitis B virus infection in Tupaia belangeri. Virus Res. 2017;237:47–57. [DOI] [PubMed] [Google Scholar]
- 216. Amako Y, Tsukiyama‐Kohara K, Katsume A, et al. Pathogenesis of hepatitis C virus infection in Tupaia belangeri. J Virol. 2010;84(1):303–311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217. Jiang L, Lu C, Sun Q. Tree shrew as a new animal model for the study of dengue virus. Front Immunol. 2021;12:621164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218. Zhang NN, Zhang L, Deng YQ, et al. Zika virus infection in Tupaia belangeri causes dermatological manifestations and confers protection against secondary infection. J Virol. 2019;93(8):e01982‐18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219. Li X, Zhou Z, Liu W, et al. Chinese tree shrew: a permissive model for in vitro and in vivo replication of human adenovirus species B. Emerg Microbes Infect. 2021;10(1):424–438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220. Yang ZF, Zhao J, Zhu YT, et al. The tree shrew provides a useful alternative model for the study of influenza H1N1 virus. Virol J. 2013;10:111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221. Li N, Gu W, Lu C, et al. Characteristics of angiotensin I‐converting enzyme 2, type II transmembrane serine protease 2 and 4 in tree shrew indicate it as a potential animal model for SARS‐CoV‐2 infection. Bioengineered. 2021;12(1):2836–2850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222. Xu L, Yu DD, Ma YH, et al. COVID‐19‐like symptoms observed in Chinese tree shrews infected with SARS‐CoV‐2. Zool Res. 2020;41(5):517–526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223. Zhao Y, Wang J, Kuang D, et al. Susceptibility of tree shrew to SARS‐CoV‐2 infection. Sci Rep. 2020;10(1):16007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224. Shi J, Wen Z, Zhong G, et al. Susceptibility of ferrets, cats, dogs, and other domesticated animals to SARS‐coronavirus 2. Science. 2020;368(6494):1016–1020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225. Barr IG, Rynehart C, Whitney P, Druce J. SARS‐CoV‐2 does not replicate in embryonated hen's eggs or in MDCK cell lines. Euro Surveill. 2020;25(25):2001122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226. Suarez DL, Pantin‐Jackwood MJ, Swayne DE, Lee SA, DeBlois SM, Spackman E. Lack of susceptibility to SARS‐CoV‐2 and MERS‐CoV in poultry. Emerg Infect Dis. 2020;26(12):3074–3076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227. Zhai X, Sun J, Yan Z, et al. Comparison of severe acute respiratory syndrome Coronavirus 2 spike protein binding to ACE2 receptors from human, pets, farm animals, and putative intermediate hosts. J Virol. 2020;94(15):e00831‐20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228. Piplani S, Singh PK, Winkler DA, Petrovsky N. In silico comparison of SARS‐CoV‐2 spike protein‐ACE2 binding affinities across species and implications for virus origin. Sci Rep. 2021;11(1):13063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229. Veljkovic V, Vergara‐Alert J, Segales J, Paessler S. Use of the informational spectrum methodology for rapid biological analysis of the novel coronavirus 2019‐nCoV: prediction of potential receptor, natural reservoir, tropism and therapeutic/vaccine target. F1000Res. 2020;9:52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230. Chen W, Yan M, Yang L, et al. SARS‐associated coronavirus transmitted from human to pig. Emerg Infect Dis. 2005;11(3):446–448. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.


