Abstract
Introduction:
Acute kidney injury (AKI) affects 20% of hospitalized patients and worsens outcomes. To limit complications, post-discharge follow-up and kidney function testing are advised. The objective of this study was to evaluate the frequency of follow-up after discharge among AKI survivors.
Methods:
This was a population-based cohort study of adult Olmsted County residents hospitalized with an episode of stage II or III AKI between 2006 and 2014. Those dismissed from the hospital on dialysis, hospice, or who died within 30-days after discharge were excluded. The frequency and predictors of follow-up, defined as an outpatient serum creatinine (SCr) level or an in-person healthcare visit after discharge were described.
Results:
In the 627 included AKI survivors, the 30-day cumulative incidence of a follow-up outpatient SCr was 80% (95% CI: 76%, 83%), a healthcare visit was 82% (95% CI: 79%, 85%), or both was 70% (95% CI: 66%, 73%). At 90-days and 1-year after discharge, the cumulative incidences of meeting both follow-up criteria rose to 82% and 91%, respectively. Independent predictors of receiving both an outpatient SCr assessment and healthcare visit within 30-days included lower estimated glomerular filtration rate at discharge, higher comorbidity burden, longer length of hospitalization, and greater maximum AKI severity. Age, sex, race/ethnicity, education level, and socioeconomic status did not predict follow-up.
Discussion/Conclusions:
Among patients with moderate to severe AKI, 30% did not have follow-up with a SCr and a healthcare visit in the 30-day post-discharge interval. Follow-up was associated with higher acuity of illness rather than demographic or socioeconomic factors.
Keywords: Acute renal failure, creatinine, healthcare utilization, quality improvement, referral, consultation
Introduction
Acute kidney injury (AKI) affects 20% of hospitalized patients and is associated with poor health outcomes.[1–3] Even among the 80–90% of hospitalized patients who survive an AKI episode, long-term morbidity is increased. Chronic kidney disease (CKD), end-stage kidney disease,[4–7] and cardiovascular diseases[8] are more common in AKI survivors than in comparable patients without AKI. To limit the burden of long-term complications in AKI survivors, follow-up care is recommended.[9,10]
Epidemiologic studies of AKI follow-up have focused on select sub-populations, including Medicare and Medicaid patients and those affiliated with a Veteran’s Affairs program; however, these findings may not be broadly generalizable.[11,12] For example, the United States Renal Data System data suggest that 85% of Medicare patients have a follow-up creatinine test in the 6-months after discharge. Insurance claims data from a large United States health care company indicate the frequency is only 54%.[11] A population-based study in a geographically defined region may provide additional insights into AKI follow-up.
Determining factors that predict follow-up for AKI survivors is also needed to identify individuals potentially vulnerable to gaps in care. High-risk conditions such as diabetes and/or CKD may lead to differences in involvement with follow-up care.[11,12] In contexts unrelated to AKI, social and behavioral determinants of health influence follow-up.[13,14] It is unclear whether factors related to acuity of illness or sociodemographic factors predominately influence AKI follow-up. Once identified, these factors may be used to target patient subgroups likely to benefit from systems-based interventions to enhance kidney care follow-up.[15,16]
Thus, the purpose of this study was to use the robust population-based longitudinal data available in the Rochester Epidemiology Project (REP) to characterize the follow-up care of nearly all local residents with stage II or III AKI dismissed from the hospital, not on dialysis who survived for at least 30-days. We described the cumulative incidence of an outpatient serum creatinine check and/or an in-person healthcare visit in the 1-year after discharge and predictors of these core elements of follow-up care.
Methods
Design and setting
This was a population-based cohort study of adults (≥18 years of age) hospitalized at Mayo Clinic in Rochester, MN, who experienced an episode of AKI. Mayo Clinic is the only provider of Nephrology subspecialty care (including dialysis) in Olmsted County. Patients were identified and followed using the REP. The REP is a medical record linkage system that amasses data from virtually all patients and healthcare providers (Mayo Clinic and non-Mayo Clinic including county medical facilities, private practices, regional hospitals, and nursing homes) in Olmsted County, Minnesota. While local residents may seek care outside of data captured by the REP, primary care, specialty care, and acute care services are available in Olmsted County which decreases the likelihood. Additional detail on the REP infrastructure is available in the detailed overview from St. Sauver, et al.[17] Women comprise approximately 50% of the population of Olmsted County, and approximately 17% of Olmsted County residents are non-Caucasian. The REP includes demographic data and comprehensive information about medical diagnoses, hospital admissions, and outpatient follow-up care regardless of where that care was delivered or patient insurance status.[17–19]
Cohort identification
Included individuals were Olmsted County residents admitted to Mayo Clinic Hospital between 01/02/2006 and 12/31/2014 who experienced stage II or stage III AKI.[20] The cohort was created as part of development and validation of an AKI electronic surveillance tool based on KDIGO AKI criteria.[20,21] Briefly, the surveillance tool screened electronic patient health records at 15-minute intervals from ICU admission to hospital discharge. The tool was optimized to detect any rise in serum creatinine or decline in urine output during that timeframe. During tool development, cases of AKI were adjudicated independently in duplicate using manual chart review. The cohort remains open for longitudinal clinical outcomes follow-up and was queried secondarily for this evaluation of the short-term processes of care. For the present study only those identified with stage II or III AKI (i.e., at least a doubling of serum creatinine from baseline or a urine output <0.5mL/kg/hr for ≥ 12-hours) were included. The preadmission baseline creatinine for this tool was the median of all values in the 6-months to −7 days before the index admission. If unavailable, this value was estimated using an assumed estimated glomerular filtration rate (eGFR) of 60 mL/min/1.73m2.[22] Time 0 for this evaluation was the date of hospital discharge. Patients were followed forward in time for up to one year using longitudinal follow-up data from the REP to capture post-discharge care.[23] Excluded individuals were those on dialysis at admission, dismissed on dialysis, or who required dialysis in the 30-days after discharge. Individuals with a kidney transplant before, during, or in the 30-days after the index hospital admission, who were discharged on hospice care, who died within 30-days, or who declined the use of their medical record for research were also excluded.[24] Institutional Review Board approval was obtained from both Mayo Clinic (IRB 18–007889) and Olmsted Medical Center (032-OMC-18) and the need for written informed consent was waived.
Sociodemographic and medical characteristics
Preadmission data captured included age, sex, race/ethnicity, and education level. Socioeconomic status was determined using the previously validated HOUsing-based SocioEconomic Status index (HOUSES index).[25,26] The HOUSES index uses publically available housing data (e.g., square footage, building value, ownership of the housing unit) to construct an individual- rather than an area-based measure of socioeconomic status. Comorbid conditions were identified using ICD 9/10 codes (Table S1). Preadmission and dismissal eGFRs were calculated with the CKD EPI eGFRcreatinine equation (mL/min/1.73m2).[27] Hospital length of stay, maximum AKI severity (stage II or III), and degree of recovery by discharge were collected. Degree of recovery was classified as recovered or stage 0 (serum creatinine <1.5x baseline, but not back to baseline level), stage 1 (1.5–1.9x baseline), stage 2 (2–2.9x baseline), stage 3 (3x baseline or ≥4 mg/dL).[28]
Outcomes
The primary outcome of interest was the cumulative incidence of both an outpatient serum creatinine level and an in-person healthcare visit with a physician or advanced practice provider within 30-days, 90-days, or 1-year after discharge. Secondarily, we described the cumulative incidence of either a serum creatinine level or an in-person healthcare visit at these time points. To identify completion of an outpatient visit, the date of the first outpatient diagnosis code(s) billed after discharge was identified. Manual validation of 10% of the records was performed to ensure the accuracy of this method and confirm that the codes corresponded to the visit date of interest. An additional analysis was performed to evaluate the healthcare visit type. We captured any nephrology visit within the follow-up period. Patients were censored at the time of their last clinical encounter (recorded in the REP) or 1-year (the end of the follow-up period).
Data analysis
Cumulative incidence of follow-up with the 95% confidence interval (CI) was estimated using the Aalen-Johansen method, where death, end-stage kidney disease, or kidney transplantation were considered competing risks. Rates were described at 30-days, 90-days, and 1-year after discharge. Cox proportional hazards models were used to assess the association of preadmission or hospitalization variables and follow-up care. Predictors of interest included age, sex, race/ethnicity, self-reported education level, socioeconomic status, body mass index, comorbidity burden described with the Charlson Comorbidity index, preadmission kidney function, AKI stage (II or III), and discharge eGFR. The occurrence of a hospital readmission during the follow-up interval could increase the likelihood of the follow-up outcome. Thus, hospital readmission was explored as a time-dependent variable, where all patients started in the no readmission group, and at the time of readmission (assuming this occurred prior to the outpatient follow-up event) the patient was moved to the readmission group. Variables satisfied the proportional hazards assumption. Multivariable models were constructed via LASSO (least absolute shrinkage and selection operator) regression. Appreciating that the degree and intensity of follow-up may differ depending on the discharge disposition, a sensitivity analysis excluded individuals discharged to a skilled nursing facility. A sensitivity analysis was also performed for individuals where the pre-admission serum creatinine was observed not estimated. All analyses were performed using SAS version 9.4 software (SAS Institute, Inc.; Cary, NC) and R version 3.6.2 (R Core Team, R Foundation for Statistical Computing, Vienna, Austria).
Results
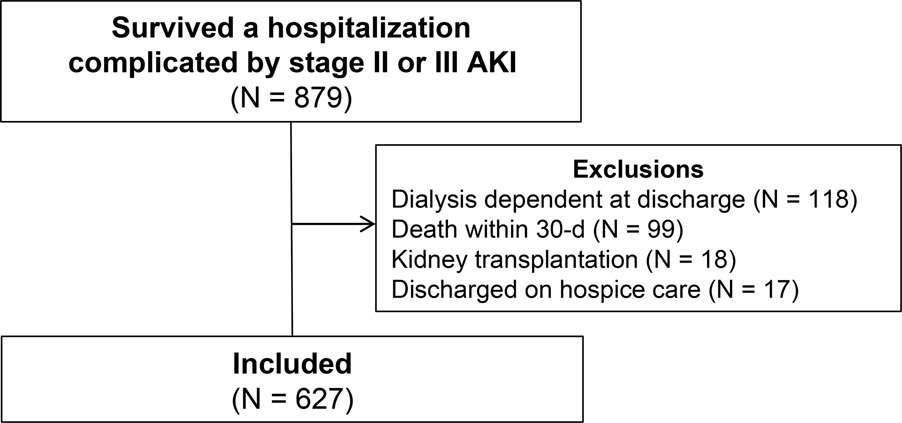
Of the 879 individuals who experienced an episode of stage II or III AKI during their hospitalization and survived to hospital discharge, 627 were included in the study cohort [Figure 1; Stage II: N=400 (64%); Stage III: N=227 (36%)]. The primary reason for exclusion was the use of dialysis at hospital discharge (N = 118). Included individuals were a mean age of 68 years, 46% were men, and 89% self-identified as non-Hispanic Caucasian (Table 1). The mean ± SD pre-admission eGFR was 63 ± 24 mL/min/1.73m2 and 379 (61%) had CKD. The median change in eGFR from pre-admission to discharge was −31% (IQR, −56% to 0%). Patients with stage III AKI during hospitalization were generally similar to those with stage II AKI with the exception of a lower discharge eGFR in patients with stage III AKI (37 ± 27 versus 51 ± 27, respectively; P = <0.001; Table S2). No patients experienced end-stage kidney disease or kidney transplanation before the endpoints of interest, thus these were not ultimately included as competing risks in endpoint analyses.
Figure 1.
Study flow diagram. AKI: Acute kidney injury
Table 1.
Characteristics of included patients before admission and during hospitalization in the overall cohort and according to degree of follow-up within 30-days
| Characteristic | Overalla (N = 627) | Both SCr and Visit within 30-daysa (N = 434) | Only SCr within 30-daysa (N = 64) | Only Visit within 30-daysa (N = 81) | Neither SCr nor visit within 30-daysa (N = 48) |
|---|---|---|---|---|---|
| Age, y | 68 ± 18 | 69 ± 18 | 70 ± 18 | 70 ± 18 | 61 ± 20 |
| Male Sex | 291 (46) | 208 (48) | 33 (52) | 31 (38) | 19 (40) |
| Non-Hispanic Caucasian | 556 (89) | 385 (89) | 56 (88) | 76 (94) | 39 (81) |
| Education levelb | |||||
| 8th grade or less | 58 (10) | 47 (11) | 3 (5) | 3 (4) | 5 (12) |
| Some or completed high-school or GED | 76 (13) | 53 (13) | 8 (13) | 8 (10) | 7 (17) |
| Some or completed college | 219 (36) | 160 (38) | 16 (26) | 29 (37) | 14 (34) |
| Post-graduate studies | 252 (42) | 163 (39) | 35 (57) | 39 (49) | 15 (37) |
| HOUSES z-scorec | −1.2 ± 3.0 | −1.3 ± 2.9 | −1.5 ± 3.5 | −1.2 ± 2.9 | −0.6 ± 3.7 |
| Body mass index, kg/m2 | 29 ± 9 | 29 ± 9 | 30 ± 9 | 28 ± 7 | 27 ± 9 |
| Pre-admission SCr, mg/dLd | 1.2 ± 1.1 | 1.3 ± 1.3 | 1.2 ± 0.7 | 1.0 ± 0.3 | 1.0 ± 0.3 |
| Pre-admission eGFR, mL/min/1.73 m2 | 63 ± 24 | 62 ± 24 | 63 ± 19 | 68 ± 24 | 71 ± 26 |
| eGFR < 60mL/min/1.73 m2 | 335 (53) | 231 (53) | 37 (58) | 43 (53) | 24 (50) |
| Comorbidity burden based on CCI | 5.2 ± 3.8 | 5.4 ± 3.7 | 5.1 ± 3.7 | 5.1 ± 4.2 | 3.8 ± 3.4 |
| Cardiovascular disease | 366 (58) | 263 (61) | 31 (48) | 51 (63) | 21 (44) |
| Heart failure | 288 (46) | 205 (47) | 34 (53) | 34 (42) | 15 (31) |
| Peripheral vascular disease | 342 (55) | 246 (57) | 34 (53) | 41 (51) | 21 (44) |
| CKD | 379 (61) | 278 (64) | 36 (56) | 42 (52) | 23 (48) |
| Diabetes | 271 (43) | 192 (44) | 33 (52) | 28 (35) | 18 (38) |
| ICU during hospitalization | 269 (43) | 189 (44) | 28 (44) | 36 (44) | 16 (33) |
| Maximum AKI severity | |||||
| Stage 2 | 400 (64) | 249 (57) | 41 (64) | 71 (88) | 39 (81) |
| Stage 3 | 227 (36) | 185 (43) | 23 (36) | 10 (12) | 9 (19) |
| Dialysis during hospitalization | 26 (4) | 20 (5) | 4 (6) | 1 (1) | 1 (2) |
| Discharge SCr, mg/dL | 1.9 ± 1.2 | 2.0 ± 1.3 | 1.8 ± 1.1 | 1.3 ± 0.6 | 1.5 ± 0.8 |
| AKI recovery by discharge | |||||
| Stage 0 (SCr 1–1.5x) | 333 (53) e | 215 (50) | 34 (53) | 56 (69) | 28 (58) |
| Stage 1 (SCr 1.5–1.9x) | 120 (19) | 86 (20) | 12 (19) | 14 (17) | 8 (17) |
| Stage 2 (SCr 2–2.9x) | 118 (19) | 84 (19) | 12 (19) | 11 (14) | 11 (23) |
| Stage 3 (SCr >3x or >4 mg/dL) | 56 (9) | 49 (11) | 6 (9) | 0 (0) | 1 (2) |
| Discharge eGFR, mL/min/1.73 m2 | 46 ± 28 | 42 ± 26 | 47 ± 29 | 59 ± 28 | 57 ± 31 |
| Hospital length of stay, days | 9.0 ± 12.1 | 9.5 ± 13.6 | 6.9 ± 6.0 | 8.9 ± 9.7 | 7.2 ± 7.1 |
| Discharge Disposition | |||||
| Home/Self-Care | 351 (56) | 242 (56) | 29 (45) | 44 (54) | 36 (75) |
| Acute Care Hospital | 3 (0.5) | 1 (0.2) | 1 (1.6) | 0 (0.0) | 1 (2.1) |
| SNF/Long Term Care Facility | 186 (30) | 127 (29) | 26 (41) | 26 (32) | 7 (15) |
| Left against medical advice/ discontinued care | 2 (0.3) | 1 (0.2) | 1 (1.6) | 0 (0.0) | 0 (0.0) |
| Home with Home Health Care services | 65 (10) | 50 (12) | 3 (5) | 10 (12) | 2 (4) |
| Rehab facility | 13 (2) | 10 (2) | 2 (3) | 1 (1) | 0 (0) |
| Inpatient Psychiatry | 7 (1) | 3 (0.7) | 2 (3) | 0 (0) | 2 (4) |
AKI: Acute kidney injury; CCI: Charlson Comorbidity Index; eGFR: Estimated glomerular filtration rate; ICU: Intensive care unit; SCr: Serum creatinine; SNF: Skilled nursing facility
Reported as count (percent) for categorical variables and mean ± standard deviation for continuous variables
Data available for 605 of the overall cohort, 423 in the both SCr and visit group, 62 in the only SCr group, 79 in the only visit group, and 41 in the neither SCr nor visit group
The HOUSES index is an individual-level socioeconomic status measure that includes four items of publically available property data based on someone's address (number of bedrooms, number of bathrooms, square footage of the unit, & estimated building value of the unit).[26] Results of the four items are aggregated and standardized into an overall Z-score for the individual. Higher Z-scores reflect a higher socioeconomic status.
Preadmission serum creatinine was available for 416 individuals in the overall cohort, 297 in the both SCr and visit group, 45 in the only SCr group, 47 in the only visit group, and 27 in the neither SCr nor visit group. For the remainder of individuals the baseline serum creatinine was estimated.
Of the patients with Stage 0 AKI recovery, 158 (47%) individuals returned to baseline or better kidney function whereas 175 (53%) individuals had a SCr between >1- and <1.5-times baseline.
Endpoints
For the primary endpoint, the cumulative incidence of follow-up with an outpatient serum creatinine level and a healthcare visit at 30-days was 70% (95% CI 66%, 73%) (Table 2). Incidence of follow-up increased over the 1-year study time period to 91%. Forty-eight individuals had no outpatient serum creatinine or healthcare visit within 30 days. Within 30-days, those with both an outpatient serum creatinine and a healthcare visit had a median of 2 (IQR 1–5) outpatient serum-creatinine assessments and 3 (IQR 2–4) visits.
Table 2.
Cumulative incidence of serum creatinine and a healthcare visit during follow-up at 30-days, 90-days and 1-year in the overall cohort (primary endpoint), as well as in sensitivity analyses.
| Cumulative incidence (95% confidence interval) of both a serum creatinine and a healthcare visit | |||
|---|---|---|---|
|
| |||
| Group | 30-days | 90-days | 1-year |
|
| |||
|
Primary endpoint
| |||
| Serum creatinine and healthcare visit (N = 627) | 70% (66%, 73%) | 82% (79%, 85%) | 91% (89%, 93%) |
|
Sensitivity analyses | |||
| Excluding individuals discharged to a SNF or acute care hospital (N = 438) | 70% (65%, 74%) | 82% (78%, 85%) | 92% (89%, 94%) |
|
| |||
| Excluding individuals with an estimated baseline serum creatinine (N = 416) | 72% (67%, 76%) | 85% (81%, 88%) | 93% (90%, 95%) |
|
| |||
| Maximum AKI severity | |||
| Stage II AKI (N = 400) | 63% (57%, 67%) | 77% (73%, 81%) | 89% (85%, 92%) |
| Stage III AKI (N = 227) | 82% (76%, 86%) | 91% (86%, 94%) | 95% (91%, 97%) |
|
| |||
| Degree of recovery by discharge | |||
| Stage 0 (N = 333) | 65% (60%, 70%) | 78% (73%, 82%) | 89% (85%, 92%) |
| Stage 1 (N = 120) | 72% (63%, 79%) | 89% (81%, 93%) | 95% (88%, 98%) |
| Stage 2 (N = 118) | 71% (62%, 78%) | 83% (74%, 89%) | 91% (83%, 95%) |
| Stage 3 (N = 56) | 87% (74%, 94%) | 93% (81%, 97%) | 96% (86%, 99%) |
|
| |||
| Baseline kidney disease | |||
| CKD (N = 379) | 74% (69%, 78%) | 87% (83%, 90%) | 93% (90%, 95%) |
| No CKD (N = 248) | 63% (56%, 69%) | 75% (69%, 80%) | 88% (83%, 92%) |
|
| |||
| Healthcare visit redefined to exclude non-nephrology specialty care (N = 627) | 59% (54%, 63%) | 73% (69%, 77%) | 89% (85%, 92%) |
AKI: Acute kidney injury; CKD: Chronic kidney disease; SNF: Skilled nursing facility
Several factors significantly predicted the completion of an outpatient serum creatinine and a healthcare visit in univariate analyses (Table S3). In multivariable models, lower discharge eGFR, higher Charlson comorbidity index, longer length of hospitalization, and greater maximum severity of AKI predicted completion of both follow-up care elements (Table 3, Figure 2). None of the sociodemographic factors in Table 1 independently predicted follow-up.
Table 3.
Multivariable model of factors associated with the outcome of serum creatinine and/or a healthcare visit during at 30-days, 90-days, and 1-year
| Outpatient serum creatinine AND a healthcare visit | ||||||
| 30-day | 90-day | 1-year | ||||
| Hazard Ratio (95% CI) | P-value | Hazard Ratio (95% CI) | P-value | Hazard Ratio (95% CI) | P-value | |
| Age (per decade) | 0.97 (0.92–1.03) | 0.38 | 0.98 (0.92–1.03) | 0.45 | 0.97 (0.92–1.02) | 0.23 |
| Male sex | 1.14 (0.95–1.39) | 0.16 | 1.16 (0.97–1.39) | 0.099 | 1.17 (0.99–1.39) | 0.072 |
| Discharge eGFR (per 10 mL/min/1.73m2) | 0.91 (0.87–0.95) | <0.001 | 0.91 (0.88–0.95) | <0.001 | 0.92 (0.89–0.95) | <0.001 |
| Charlson comorbidity index (per 1 unit) | 1.03 (1.01–1.06) | 0.014 | 1.04 (1.01–1.06) | 0.004 | 1.04 (1.02–1.07) | 0.001 |
| ICU stay during hospitalization | 1.08 (0.88–1.32) | 0.45 | 1.09 (0.90–1.31) | 0.39 | 1.06 (0.88–1.27) | 0.53 |
| Length of hospitalization (per week) | 1.07 (1.02–1.11) | 0.005 | 1.06 (1.02–1.11) | 0.007 | 1.06 (1.02–1.11) | 0.006 |
| Maximum AKI severity (III vs II) | 1.55 (1.27–1.89) | <0.001 | 1.52 (1.26–1.84) | <0.001 | 1.47 (1.23–1.76) | <0.001 |
| Hospital Readmission (time-dependent) | 1.17 (0.74–1.86) | 0.50 | 1.41 (0.99–2.01) | 0.060 | 1.46 (1.04–2.03) | 0.027 |
| Outpatient serum creatinine | ||||||
| 30-day | 90-day | 1-year | ||||
| Hazard Ratio (95% CI) | P-value | Hazard Ratio (95% CI) | P-value | Hazard Ratio (95% CI) | P-value | |
| Age (per decade) | 0.94 (0.89–1.00) | 0.035 | 0.95 (0.90–1.01) | 0.076 | 0.95 (0.90–1.00) | 0.051 |
| Male sex | 1.17 (0.97–1.40) | 0.096 | 1.21 (1.02–1.43) | 0.032 | 1.22 (1.03–1.44) | 0.019 |
| Discharge eGFR (per 10 mL/min/1.73m2) | 0.89 (0.86–0.93) | <0.001 | 0.90 (0.86–0.93) | <0.001 | 0.90 (0.87–0.93) | <0.001 |
| Charlson comorbidity index (per 1 unit) | 1.04 (1.01–1.07) | 0.002 | 1.04 (1.02–1.07) | 0.001 | 1.04 (1.02–1.07) | <0.001 |
| ICU stay during Hospitalization | 1.24 (1.03–1.50) | 0.022 | 1.25 (1.04–1.49) | 0.015 | 1.23 (1.04–1.46) | 0.015 |
| Length of hospitalization (per week) | 1.06 (1.02–1.10) | 0.007 | 1.06 (1.01–1.10) | 0.010 | 1.05 (1.01–1.10) | 0.016 |
| Maximum AKI severity (III vs II) | 1.61 (1.33–1.94) | <0.001 | 1.60 (1.33–1.92) | <0.001 | 1.59 (1.33–1.90) | <0.001 |
| Hospital Readmission (time-dependent) | 1.12 (0.46–2.73) | 0.80 | 1.64 (0.81–3.33) | 0.17 | 1.80 (0.92–3.51) | 0.084 |
| Healthcare visit | ||||||
| 30-day | 90-day | 1-year | ||||
| Hazard Ratio (95% CI) | P-value | Hazard Ratio (95% CI) | P-value | Hazard Ratio (95% CI) | P-value | |
| Age (per decade) | 1.04 (0.98–1.10) | 0.16 | 1.03 (0.97–1.08) | 0.37 | 1.02 (0.97–1.07) | 0.49 |
| Male sex | 1.03 (0.87–1.24) | 0.71 | 1.02 (0.86–1.21) | 0.81 | 1.04 (0.88–1.23) | 0.63 |
| Discharge eGFR (per 10 mL/min/1.73m2) | 0.97 (0.94–1.01) | 0.15 | 0.98 (0.94–1.01) | 0.18 | 0.98 (0.94–1.01) | 0.14 |
| Charlson comorbidity index (per 1 unit) | 1.03 (1.00–1.06) | 0.023 | 1.04 (1.01–1.06) | 0.003 | 1.04 (1.01–1.06) | 0.003 |
| ICU stay during Hospitalization | 1.16 (0.96–1.39) | 0.12 | 1.13 (0.94–1.35) | 0.18 | 1.11 (0.93–1.33) | 0.23 |
| Length of hospitalization (per week) | 1.06 (1.02–1.10) | 0.002 | 1.06 (1.02–1.10) | 0.003 | 1.06 (1.02–1.10) | 0.003 |
| Maximum AKI severity (III vs II) | 1.21 (1.00–1.46) | 0.051 | 1.22 (1.02–1.46) | 0.032 | 1.19 (0.99–1.42) | 0.057 |
| Hospital Readmission (time-dependent) | 0.78 (0.47–1.29) | 0.33 | 0.98 (0.65–1.47) | 0.91 | 0.97 (0.66–1.44) | 0.89 |
AKI: Acute kidney injury; eGFR: Estimated glomerular filtration rate; ICU: Intensive care unit; CI: Confidence Interval
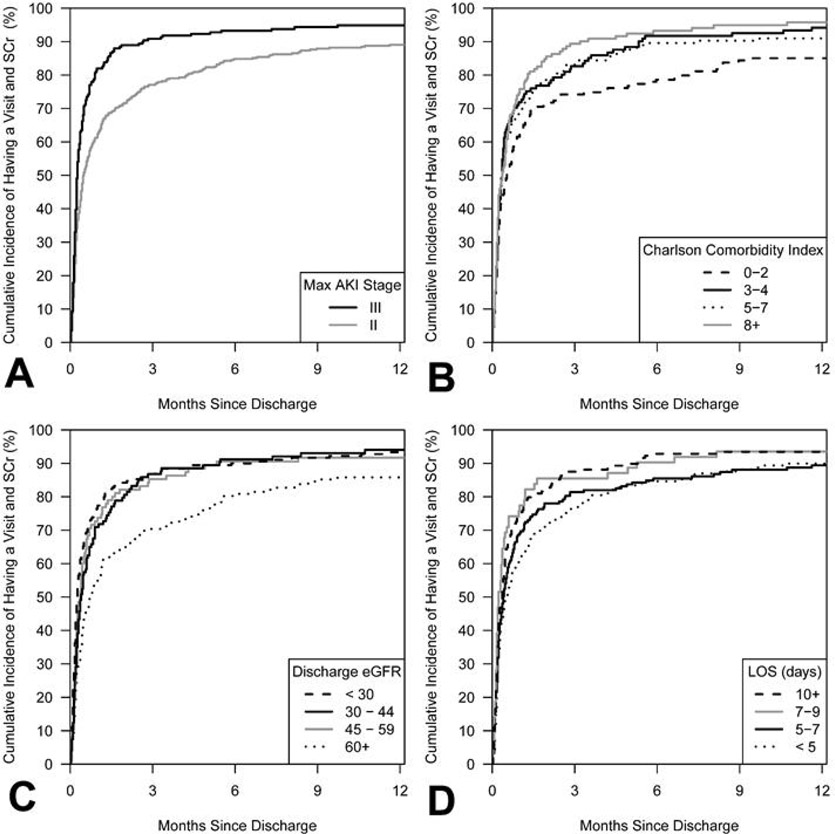
Figure 2.
Cumulative incidence of the primary outcome of an outpatient serum creatinine and an outpatient follow-up visit with a health care provider at one year according to four key predictors. Panel A: Severity of acute kidney injury - Individuals with a maximum severity of AKI stage III were 1.47 (95% CI 1.23–1.76) times more likely to have follow-up within one year compared to those with a maximum of stage II AKI (P < 0.001). Panel B: Charlson Comorbidity Index – Individuals with a greater comorbidity burden were more likely to receive follow-up (HR 1.04 for each point increase, 95% CI 1.02, 1.07; P = 0.001). Panel C: Discharge eGFR– Individuals with worse kidney function at discharge were more likely to receive follow-up (HR 0.92 per 10mL/min/1.73m2 increase in eGFR, 95% CI 0.89, 0.95; P < 0.001). Panel D: Length of hospitalization – Individuals with longer hospitalizations were more likely to have follow-up (HR 1.06 per week increase in length of stay, 95% CI 1.02, 1.11; P = 0.006). SCr: Serum creatinine; AKI: Acute kidney injury; eGFR: estimated glomerular filtration rate; LOS: Length of stay.
In secondary analyses, the cumulative incidence of an outpatient serum creatinine level was 80% (95% CI 76%, 83%) at 30-days. For provider visits, the cumulative incidence was 82% (95% CI: 79%, 85%) at 30-days. Predictive models for follow-up serum creatinine or follow-up provider visit were generally similar to when both follow-up elements were combined, but sample sizes were more limited (Table 3, Figures S1 and S2). An ICU stay during hospitalization significantly predicted creatinine evaluation at 30-days (P = 0.022), 90-days (P = 0.015), and 1-year (P = 0.015). Within 1-year, 23% of individuals with a follow-up healthcare visit (n/N = 133/578) saw a nephrologist (Table S4). 132 (23%) individuals completed both an outpatient serum creatinine and saw a nephrologist within 1-year, of which 113 (86%) had CKD. The median time to outpatient nephrology consultation was 21 (IQR 7–54) days. Among patients with pre-admission diabetes or CKD, 29% saw a nephrologist within 1-year.
A series of sensitivity analyses were performed including an evaluation of a serum creatinine and a healthcare visit where the visit was only counted if it was with a primary care provider or nephrologist, an analysis excluding the 189 individuals discharged to a skilled nursing facility or an acute care hospital, an analysis excluding the 211 individuals with an estimated baseline creatinine value, and stratified evaluations according to AKI severity, degree of recovery by discharge, and baseline CKD. The findings were generally similar in these analyses to the overall group. The frequency of follow-up was higher in patients with more severe AKI, non-recovery, and baseline CKD. When non-nephrology specialty care visits were not included in the follow-up visit definition, incidence of a healthcare visit (primary care/nephrology) and serum creatinine fell from 70% to 59% at 30-days (Table 2).
Discussion
In this population-based cohort study, one-third of AKI survivors lacked follow-up in the 30-days after hospital discharge. Among those who only had one element of follow-up care, the serum creatinine was more often missing than the healthcare visit. By 1-year, 88% of individuals experienced an outpatient serum creatinine check and a healthcare visit. Non-nephrologists oversaw the majority of healthcare visits (90%). Sociodemographic factors did not predict follow-up care. Instead, factors related to illness severity, including a worse severity of AKI, lower discharge eGFR, longer length of stay, and greater comorbidity burden independently predicted a higher likelihood of follow-up.
There is increased recognition of the importance of AKI as a contributor to long-term kidney and cardiovascular outcomes.[29] Kidney follow-up care is recommended during the period of vulnerability after an episode of AKI.[10,28] Delivery of this follow-up care for AKI survivors remains suboptimal. The United States Renal Data System data suggest that follow-up laboratory testing in AKI survivors, including a serum creatinine or urine albumin evaluation within 6-months of discharge, only occurs in 54% and 14% of patients, respectively.[11] Primary care visits occur in 75% of Medicare and Medicaid patients within 3-months, but in other cohorts, the frequency is much lower. In Medicare and Medicaid patients and those affiliated with a Veteran’s Affairs program, estimates indicate that only 4–17% of AKI survivors see nephrologists by one year.[11,12] In the present study, we comprehensively characterized the adequacy of follow-up using a population-based cohort from Olmsted County, MN. Our cohort is slightly younger, with better baseline kidney function and fewer comorbidities than previous patient subsets.[11,12] AKI in this study was defined by biochemical markers, serum creatinine, and urine output, rather than diagnosis codes, which facilitates the detection and inclusion of survivors with less severe AKI during hospitalization.[30] We demonstrated that one-third of AKI survivors lacked an outpatient serum creatinine assessment and a healthcare visit in the 1-month after follow-up. This improved over time, but even out to 1-year after the episode of moderate to severe AKI, 12% of patients lacked both an outpatient serum creatinine evaluation and a follow-up healthcare visit.
An important finding in the present study was the infrequency with which nephrology follow-up occurred for AKI survivors. We demonstrated that only 23% of individuals with a follow-up healthcare visit within 1-year saw a nephrologist. If a patient was to see the nephrologist, it was relatively close to the time of discharge [median 21 days (IQR 7–54)]. Among patients with diabetes or CKD, only one-third of our cohort saw a nephrologist within 1-year. These data are similar to the 8.5% cumulative incidence of nephrology referral in 1-year observed in AKI survivors in a Department of Veterans Affairs dataset.[12] Even in AKI survivors who required dialysis during hospitalization, population-level data from Taiwan indicated follow-up by a nephrologist occurred in only 37% of patients.[31] This gap in follow-up care is despite evidence that nephrologists would like to see these high-risk patients in the clinic[32] and when they do, there is an association with better outcomes.[31,33] Several factors likely contribute to the low frequency of nephrology follow-up even among very high-risk AKI survivors. AKI is often underdiagnosed in hospitalized patients and underreported on dismissal summaries.[34] Interviews with AKI survivors indicate that many factors limit engagement with post-AKI care, including prioritizing other health concerns, lack of education about the significance of AKI, anxiety from competing health demands, and complex discharge plans.[35] A proportion of patients may be referred for specialty follow-up care, but cancel for these or other reasons. Further, nephrology follow-up is a sparse resource that may not be widely accessible to all patients, especially those in rural communities.[36,37] Although the observational data suggest a health benefit for AKI survivors when seen by nephrology, it remains to be determined whether structured kidney health follow-up delivered by trained individuals in primary care could reach patients not seen by nephrologists with a similar health benefit.[9]
To identify individuals at high-risk for gaps in care, we conducted a multivariable analysis of factors that might predict patient follow-up. These data reveal that a greater comorbidity burden was associated with a higher incidence of a serum creatinine assessment and a healthcare visit within one year. This reinforces the hypothesis that greater medical complexity portends clinical follow-up. Among the other factors evaluated, increased severity of AKI during the hospitalization, longer length of stay, and lower discharge eGFR also predicted follow-up. Multivariable risk factors were generally consistent at 30-days, 90-days and 1-year. We did not observe any association between age, sex, race/ethnicity, education level, or socioeconomic status with post-hospital follow-up among AKI survivors. Our sample was comprised of 89% non-Hispanic Caucasian individuals, with a relatively high baseline education level and socioeconomic status, which likely affected the observed follow-up patterns. In previous prognostic studies, non-Caucasian race/ethnicity and worse socioeconomic status were associated with a higher AKI incidence and worse long-term outcomes[38–40].
Nephrology consensus groups recommend follow-up for all AKI survivors but indicate that it should be graded in urgency and intensity based on the severity of AKI, duration of injury, degree of comorbidities, and magnitude of renal recovery by discharge.[10] Our data suggest existing approaches to care delivery moderately achieve these follow-up goals. Based on these findings, individuals at high risk for experiencing gaps in kidney care follow-up are those with less severe AKI (stage II), fewer comorbidities, short hospital stays, and improved GFR by discharge. While this lower risk risk group likely would benefit from post-discharge monitoring of kidney health (i.e., serum creatinine, medication reconciliation and counseling, blood pressure evaluation), these care elements may be able to be delivered by a primary care provider rather than a specialist.
This study has several limitations that must be accounted for when interpreting these findings. Consensus recommendations suggest AKI follow-up should generally include kidney function assessment, patient education, medication review and reconciliation, fluid status and blood pressure evaluation, and self-management counseling during acute illness (i.e., sick day counseling).[10] The focus of this evaluation was on the serum creatinine assessment and healthcare visit components. Other laboratory tests beyond serum creatinine may be warranted for AKI follow-up, but data suggests these are performed even less frequently than the serum creatinine.[11] Moreover, we cannot assess the interpretation of and reaction to the results of kidney monitoring tests. Access to the serum creatinine data does not necessarily result in a change to the care provided, especially if clinicians were unaware of the patient’s AKI episode. A creatinine evaluation may be included as part of a broader panel of laboratory data, but evaluation of its results may not be the care team’s focus. Not all healthcare visits likely provide the degree of kidney care evaluation (nephrology services, non-nephrology specialty referral, primary care). Still, we feel that at least one encounter with a healthcare provider lends an opportunity for a kidney health review. In a sensitivity analysis where visits were defined only as with a primary care provider or a nephrologist, the cumulative incidence of follow-up with a serum creatinine and a visit within 30 days fell from 70% in the main analysis to 59%. In the six years that have passed since the end of the study follow-up period, an increased emphasis has been placed on the importance of kidney follow-up care.[10] Further the advent of telehealth has increased access to follow-up care.[37] It is possible that a more contemporary cohort might reveal an improved incidence of follow-up. We recognize that 30% of our patients were dismissed to skilled nursing facilities or acute care hospitals where nursing care and clinical evaluations that constitute follow-up may have gone undocumented in the electronic health record. A subgroup analysis which excluded these individuals the results were similar. While the included individuals represent the demographics of southeast Minnesota, the sample was mostly non-Hispanic Caucasians with a higher baseline education level which could affect generalizability of these findings. One challenge in epidemiologic studies of this nature is how to handle patients who die or are rehospitalized during the follow-up period. Those that die within 30-days often have a care plan designed to focus on comfort and quality of life. Laboratory monitoring or healthcare visits may not be consistent with these goals of care. These individuals were thus excluded. Death in the included cohort was otherwise modeled as a competing risk. At a rehospitalization, the potential for a future healthcare visit or creatinine assessment could be affected. To address this limitation, we modeled rehospitalization during the follow-up window as a time-dependent variable and restricted the endpoint to include only outpatient creatinine values and outpatient visits. Finally, further studies are needed to determine the impact of post-AKI follow-up on clinical outcomes such as CKD.
Conclusions
In this population-based cohort study, we demonstrated that 30% of patients with moderate to severe AKI failed to receive follow-up in the 30-day post-discharge interval. Medical risk factors, rather than social/demographic characteristics, were the primary determinants of follow-up. More work is needed to enhance care delivery in AKI survivors.
Supplementary Material
Funding:
This project was supported in part by the Mayo Clinic Department of Pharmacy, the National Institutes of Health National Center for Advancing Translational Sciences (NCATS) under Award Number UL1 TR002377, the National Institute on Aging under Award Number R01AG034676, the National Institute of Allergy and Infectious Diseases of the under Award Number K23AI143882 (PI; EFB), and the Agency for Healthcare Research and Quality HS028060–01 (PI; EFB). The funding sources had no role in study design; data collection, analysis, or interpretation; writing the report; or the decision to submit the report for publication. Its contents are solely the authors’ responsibility and do not necessarily represent the official views of the NIH.
Footnotes
Conflict of interest statement: Dr. Barreto reports consultancy for FAST Biomedical, unrelated.
Statement of Ethics: This study protocol was reviewed and approved by Institutional Review Boards from both Mayo Clinic (#18-007889) and Olmsted Medical Center (#032-OMC-18). All included individuals consented to the use of their medical records for research.
Data availability:
The data that support the findings of this study are not publicly available due to their containing information that could compromise the privacy of research participants, but are available from the corresponding author EFB upon reasonable request.
References
- 1.Wang HE, Muntner P, Chertow GM, Warnock DG. Acute kidney injury and mortality in hospitalized patients. Am J Nephrol. 2012;35(4):349–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Susantitaphong P, Cruz DN, Cerda J, Abulfaraj M, Alqahtani F, Koulouridis I, et al. World incidence of AKI: A meta-analysis. Clin J Am Soc Nephrol. 2013;8(9):1482–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chertow GM, Burdick E, Honour M, Bonventre JV, Bates DW. Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol. 2005. November;16(11):3365–70. [DOI] [PubMed] [Google Scholar]
- 4.James MT, Pannu N, Hemmelgarn BR, Austin PC, Tan Z, McArthur E, et al. Derivation and External Validation of Prediction Models for Advanced Chronic Kidney Disease Following Acute Kidney Injury. JAMA. 2017;318(18):1787–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sawhney S, Marks A, Fluck N, Levin A, McLernon D, Prescott G, et al. Post-discharge kidney function is associated with subsequent ten-year renal progression risk among survivors of acute kidney injury. Kidney Int. 2017;92(2):440–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jones J, Holmen J, De Graauw J, Jovanovich A, Thornton S, Chonchol M. Association of complete recovery from acute kidney injury with incident CKD stage 3 and all-cause mortality. Am J Kidney Dis. 2012;60(3):402–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Heung M, Steffick DE, Zivin K, Gillespie BW, Banerjee T, Hsu CY, et al. Acute Kidney Injury Recovery Pattern and Subsequent Risk of CKD: An Analysis of Veterans Health Administration Data. Am J Kidney Dis. 2016;67(5):742–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Odutayo A, Wong CX, Farkouh M, Altman DG, Hopewell S, Emdin CA, et al. AKI and long-term risk for cardiovascular events and mortality. J Am Soc Nephrol. 2017;28(1):377–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Siew ED, Liu KD, Bonn J, Chinchilli V, Dember LM, Girard TD, et al. Improving Care for Patients after Hospitalization with AKI. J Am Soc Nephrol. 2020. October;31(10):2237–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kashani K, Rosner MH, Haase M, Lewington AJPP, O’Donoghue DJ, Wilson FP, et al. Quality Improvement Goals for Acute Kidney Injury. Clin J Am Soc Nephrol. 2019. June;14(6):941–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Saran R, Robinson B, Abbott KC, Agodoa LYC, Albertus P, Ayanian J, et al. US Renal Data System 2016 Annual Data Report: Epidemiology of Kidney Disease in the United States. Am J Kidney Dis. 2017. March;69(3 Suppl 1):A1-A8,S1–S688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Siew ED, Peterson JF, Eden SK, Hung AM, Speroff T, Ikizler TA, et al. Outpatient nephrology referral rates after acute kidney injury. J Am Soc Nephrol. 2012;23(2):305–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Phillips D, Holmes J, Davies R, Geen J, Williams JD, Phillips AO. The influence of socioeconomic status on presentation and outcome of acute kidney injury. Qjm. 2018;111(12):849–57. [DOI] [PubMed] [Google Scholar]
- 14.Barwise A, Juhn YJ, Wi C Il, Novotny P, Jaramillo C, Gajic O, et al. An Individual Housing-Based Socioeconomic Status Measure Predicts Advance Care Planning and Nursing Home Utilization. Am J Hosp Palliat Med. 2019;36(5):362–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Singh G, Hu Y, Jacobs S, Brown J, George J, Bermudez M, et al. Post-Discharge Mortality and Rehospitalization among participants in a Comprehensive Acute Kidney Injury Rehabilitation Program. Kidney 360. 2021. July; 10.34067/KID.0003672021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Silver SA, Adhikari NK, Bell CM, Chan CT, Harel Z, Kitchlu A, et al. Nephrologist Follow-Up versus Usual Care after an Acute Kidney Injury Hospitalization (FUSION): A Randomized Controlled Trial. Clin J Am Soc Nephrol. 2021. July;16(7):1005–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.St Sauver JL, Grossardt BR, Yawn BP, Melton LJ, Rocca WA. Use of a medical records linkage system to enumerate a dynamic population over time: the Rochester epidemiology project. Am J Epidemiol. 2011. May;173(9):1059–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rocca WA, Yawn BP, St Sauver JL, Grossardt BR, Melton LJ. History of the Rochester Epidemiology Project: half a century of medical records linkage in a US population. Mayo Clin Proc. 2012. December;87(12):1202–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.St Sauver JL, Grossardt BR, Leibson CL, Yawn BP, Melton LJ, Rocca WA. Generalizability of epidemiological findings and public health decisions: An illustration from the Rochester Epidemiology Project. Mayo Clin Proc. 2012;87(2):151–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kashani K, Shao M, Li G, Williams AW, Rule AD, Kremers WK, et al. No increase in the incidence of acute kidney injury in a population-based annual temporal trends epidemiology study. Kidney Int. 2017;92(3):721–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ahmed A, Vairavan S, Akhoundi A, Wilson G, Chiofolo C, Chbat N, et al. Development and validation of electronic surveillance tool for acute kidney injury: A retrospective analysis. J Crit Care. 2015;30(5):988–93. [DOI] [PubMed] [Google Scholar]
- 22.Thongprayoon C, Cheungpasitporn W, Harrison AM, Kittanamongkolchai W, Ungprasert P, Srivali N, et al. The comparison of the commonly used surrogates for baseline renal function in acute kidney injury diagnosis and staging Clinical Research. BMC Nephrol. 2016;17(1):1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rule AD, Bergstralh EJ, Melton LJ, Li X, Weaver AL, Lieske JC. Kidney stones and the risk for chronic kidney disease. Clin J Am Soc Nephrol. 2009. April;4(4):804–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Melton LJ. The threat to medical-records research. N Engl J Med. 1997. November;337(20):1466–70. [DOI] [PubMed] [Google Scholar]
- 25.Canney M, Induruwage D, Sahota A, McCrory C, Hladunewich MA, Gill J, et al. Socioeconomic Position and Incidence of Glomerular Diseases. Clin J Am Soc Nephrol. 2020. March;15(3):367–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Juhn YJ, Beebe TJ, Finnie DM, Sloan J, Wheeler PH, Yawn B, et al. Development and initial testing of a new socioeconomic status measure based on housing data. J Urban Health. 2011. October;88(5):933–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Inker LA, Schmid CH, Tighiouart H, Eckfeldt JH, Feldman HI, Greene T, et al. Estimating glomerular filtration rate from serum creatinine and cystatin C. N Engl J Med. 2012;367(1):20–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chawla LS, Bellomo R, Bihorac A, Goldstein SL, Siew ED, Bagshaw SM, et al. Acute kidney disease and renal recovery: Consensus report of the Acute Disease Quality Initiative (ADQI) 16 Workgroup. Nat Rev Nephrol. 2017;13(4):241–57. [DOI] [PubMed] [Google Scholar]
- 29.Silver SA, Gerarduzzi C. Found in Translation: Reasons for Optimism in the Pursuit to Prevent Chronic Kidney Disease After Acute Kidney Injury. Can J Kidney Heal Dis. 2019. January;6(8):205435811986874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Grams ME, Waikar SS, MacMahon B, Whelton S, Ballew SH, Coresh J. Performance and Limitations of Administrative Data in the Identification of AKI. Clin J Am Soc Nephrol. 2014. April;9(4):682–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wu VC, Chueh JS, Chen L, Huang TM, Lai TS, Wang CY, et al. Nephrologist Follow-Up Care of Patients With Acute Kidney Disease Improves Outcomes: Taiwan Experience. Value Heal. 2020;23(9):1225–34. [DOI] [PubMed] [Google Scholar]
- 32.Karsanji DJ, Pannu N, Manns BJ, Hemmelgarn BR, Tan Z, Jindal K, et al. Disparity between nephrologists’ opinions and contemporary practices for community follow-up after AKI hospitalization. Clin J Am Soc Nephrol. 2017;12(11):1753–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Harel Z, Wald R, Bargman JM, Mamdani M, Etchells E, Garg AX, et al. Nephrologist follow-up improves all-cause mortality of severe acute kidney injury survivors. Kidney Int. 2013;83(5):901–8. [DOI] [PubMed] [Google Scholar]
- 34.Greer RC, Liu Y, Crews DC, Jaar BG, Rabb H, Boulware LE. Hospital discharge communications during care transitions for patients with acute kidney injury: A cross-sectional study. BMC Health Serv Res. 2016;16(1):1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Silver SA, Saragosa M, Adhikari NK, Bell CM, Harel Z, Harvey A, et al. What insights do patients and caregivers have on acute kidney injury and posthospitalisation care? A single-centre qualitative study from Toronto, Canada. BMJ Open. 2018;8(6). DOI: 10.1136/bmjopen-2017-021418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Parker MG, Pivert KA, Ibrahim T, Molitoris BA. Recruiting the next generation of nephrologists. Adv Chronic Kidney Dis. 2013. July;20(4):326–35. [DOI] [PubMed] [Google Scholar]
- 37.Tan J, Mehrotra A, Nadkarni GN, He JC, Langhoff E, Post J, et al. Telenephrology: Providing Healthcare to Remotely Located Patients with Chronic Kidney Disease. Am J Nephrol. 2018;47(3):200–7. [DOI] [PubMed] [Google Scholar]
- 38.Hounkpatin HO, Fraser SDS, Johnson MJ, Harris S, Uniacke M, Roderick PJ. The association of socioeconomic status with incidence and outcomes of acute kidney injury. Clin Kidney J. 2020. April;13(2):245–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bjornstad EC, Marshall SW, Mottl AK, Gibson K, Golightly YM, Charles A, et al. Racial and health insurance disparities in pediatric acute kidney injury in the USA. Pediatr Nephrol. 2020;35(6):1085–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Grams ME, Matsushita K, Sang Y, Estrella MM, Foster MC, Tin A, et al. Explaining the racial difference in AKI incidence. J Am Soc Nephrol. 2014;25(8):1834–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data that support the findings of this study are not publicly available due to their containing information that could compromise the privacy of research participants, but are available from the corresponding author EFB upon reasonable request.