Abstract
Introduction
Thoraco-abdominal duplication is rare congenital malformations of the notochord that occurs in only 2% of cases of alimentary tract duplications.
We report two rare cases of thoraco-abdominal duplication, emphasizing the value of radiological assessment and discussing the place of diagnostic and therapeutic laparoscopy.
Presentation of case
It was a 12-year-old girl and an 8-month-old boy, admitted for epigastralgia and dysphagia with respiratory distress respectively. Imaging was in favor of pancreatic duplication with intra-thoracic extension for the first patient and gastro-esophageal duplication for the second. A mass excision was done laparoscopically for the first and by a thoracotomy for the second. The aftermath of the surgery was simple in both cases.
Clinical discussion
Thoraco-abdominal duplications are rare congenital malformations that account for only 2% of cases of gastrointestinal duplications. Their diagnosis is difficult since the revealing symptomatology is not common. The treatment is only surgical is facilitated by the laparoscopy which has a diagnostic and therapeutic interest.
Conclusion
Our case reports focused on the difficulty of the diagnosis that is done by imaging and is confirmed by surgery with anatomopathological examination of the excised mass. Diagnostic and therapeutic minimally invasive approach should be used whenever possible.
Keywords: Thoracoabdominal duplication, Congenital malformation, Pancreatic duplication, Gastro-esophageal duplication, Minimally invasive, Case report
Highlights
-
•
Thoraco-abdominal duplications account for only 2% of cases of gastrointestinal duplications.
-
•
Their diagnosis is difficult since the revealing symptomatology is not common.
-
•
The treatment depends on the type of duplication.
-
•
This management is facilitated by the laparoscopy.
1. Introduction
Duplication of the alimentary tract is rare congenital malformations that occur in 1 out of every 4500 live births [1]. Chest extension through the diaphragm was described for the first time in 1946 by Valle and White and accounts for only 2% of cases of gastrointestinal duplications [2]. These abnormalities usually manifest early in the first year of life and its diagnosis is difficult since the revealing symptomatology is not common [3].
2. Methods
We report two rare cases of thoraco-abdominal duplication, emphasizing the value of radiological assessment and discussing the place of diagnostic and therapeutic laparoscopy.
This work has been reported in line with the SCARE and the PROCESS criteria [4], [5].
3. Presentation of case
3.1. Case 1
An otherwise healthy 12-year-old girl, was admitted in our department for severe and sudden onset epigastralgia associated with vomiting. Physical examination revealed generalized abdominal tenderness with sus-umbilical contracture. Blood analysis was normal with normal rate of amylasemia (74 IU/l).
Abdominal ultrasound with computerized tomography supplement revealed a bilobed cystic formation (Fig. 1) measuring 80 mm of large axis, appearing to develop at the expense of the head of the pancreas, having a digestive wall, in relation with a thoraco-abdominal duplication, emerging from the second duodenum and extending into the post-inferior mediastinum (Fig. 2).
Fig. 1.

Bilobed cystic intra-pancreatic formation at abdominal ultrasound (red arrow).
Fig. 2.
Thoraco-abdominal computerized tomography showing pancreatic duplication (red arrow).
In order to find a communication between duplication and the digestive tract, an eso-gastro-duodenal transit was performed. It showed the lack of communication (Fig. 3).
Fig. 3.
Eso-gastro-duodenal transit showing lack of communication between the pancreatic duplication and the digestive tract: Red arrow: Digestive tract; yellow arrow: Pancreatic duplication.
Magnetic resonance imaging has eliminated the possibility of communication with the Wirsung duct.
Diagnostic and therapeutic laparoscopy confirmed the diagnosis with evidence of a mass of 7 cm in diameter at the head of the pancreas extended to the posterior mediastinum. Subtotal exeresis with cauterization of the hull bottom was performed without incidents.
The aftermath of the surgery was simple and the patient was discharged five days post-operatively. Anatomopathological examination found gastric heterotopy within the duplication. Currently we are at 6 years' post-operative. The patient is seen every 6 months at the outpatient clinic and she has never developed a pancreatitis.
3.2. Case 2
It was an 8-month-old male infant, followed for gastroesophageal reflux complicated by a low body weight (−3 standard deviation), admitted in our department for dysphagia and respiratory distress.
Blood analysis showed anemia (hemoglobin at 8 g/dl) and hypoproteinemia at 55 g/l. Chest X-ray showed chest opacity extending to the abdomen (Fig. 4). Abdominal ultrasound and thoraco-abdominal computerized tomography concluded to a gastro-esophageal duplication (Fig. 5). Eso-gastro-duodenal transit did not found communication between the duplication and the rest of the digestive tract (Fig. 6).
Fig. 4.

Opacity of the posterior mediastinum extending into the abdomen at chest X-ray (red arrow).
Fig. 5.
Gastro-esophageal duplication at computerized tomography (red arrow).
Fig. 6.
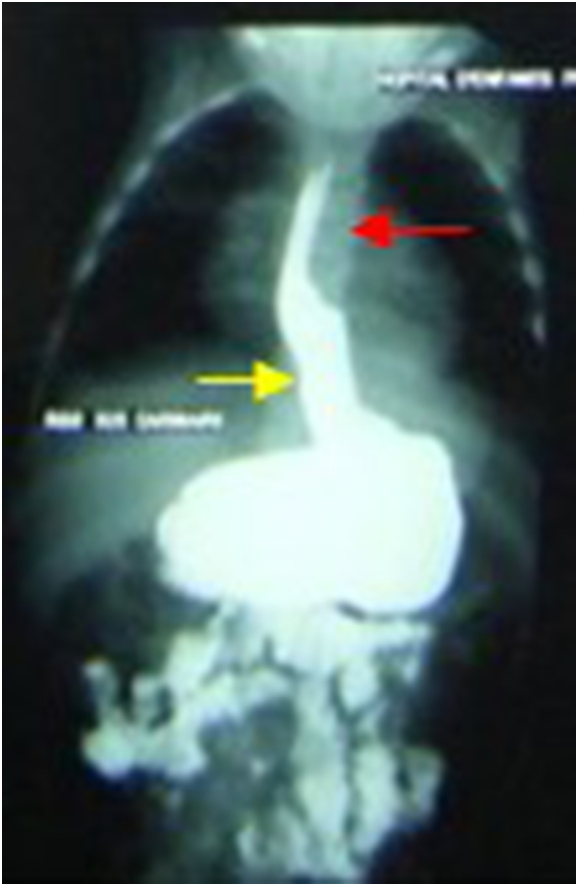
Non communicative duplication at eso-gastro-duodenal opacification: Red arrow: Duplication; yellow arrow: Digestive tract.
The patient underwent a left post-lateral thoracotomy that confirmed the diagnosis. It was an intramuscular duplication, developed at the expense of the thoracic esophagus and extending down to the large gastric tuberosity.
A total exeresis was carried out. The aftermath of the surgery was simple. The follow-up is 4 years. The patient is seen every 6 months at the outpatient department and he is doing well.
4. Discussion
Digestive duplications were first time defined by Ladd and Gross in 1950 as “spherical or tubular formations that can occur from the mouth to the anus, having contact with the normal digestive tract, communicative or not, with a smooth musculoskeletal and a digestive-type mucosa” [6]. Thoraco-abdominal duplications account for 2% of all of them with possible communication with the digestive tube in 60% of cases [7]. They represent an abnormality of the notochord with possible association with other abnormalities especially vertebral ones (hemi vertebrae, anterior spina, meningocele) [7]. Most studies report male predominance with sex-ratio of 1.2 [8], [9], [10].
The original digestive segment may be the esophagus [11], the stomach, or both [9] but also the duodenum [10] or hail. This was an intra-pancreatic duodenal duplication in our first case and gastro-esophageal duplication in the second.
The chest segment often ends at the level of a cervicothoracic spine malformation. Diaphragmatic crossing is done in more than 2/3 of cases by a deep orifice, in the vicinity or through the right pillar, more rarely through the esophageal or aortic orifice [11].
The antenatal discovery is increasingly being described [12]. Elsewhere, they are usually diagnosed during the first year of life in ¾ of cases by respiratory signs due to compression of the airways by chest mass or digestive signs following secretion by the atopic mucosa [8], [9]. Discovery in the older child is rare [10]. This was the case in one of our patients. In these cases, symptoms may include weight stagnation, fever, minor chest symptomatology or signs of compression [10]. Abdominal symptomatology may be made up of paroxysmal pain, digestive hemorrhage or peritonitis secondary to perforation [9]. Digestive symptomatology was the mode of revelation in one of our patients and a combination of digestive and respiratory symptomatology in the second.
Diagnosis is difficult and based on radiological data [11]. The standard abdominal and chest X-ray often reveal a homogeneous liquid opacity in the posterior mediastinum. This opacity is often right-sided, retro cardiac, low located with possible repulsing of the mediastinum and associated cervico-thoracic vertebral abnormalities [13]. Chest and abdominal ultrasound often confirms the cystic nature of the mass, specifies its seat and extent, data that will be further studied by a thoraco-abdominal computerized tomography, which exclude the different differential diagnosis and research a communication with neighborhood structures [14]. The presence of communication can be formally eliminated by the esogastro-duodenal transit [14].
Treatment of thoraco-abdominal duplications is also a second problem to be confronted in the absence of consensus. Although the malignant change is rare, most thoraco-abdominal duplications should be excised on in view of the clinical signs it may give, as in our cases, and the risk of potential complications [15], [16]. Surgery is the only therapeutic and diagnostic of certainty mean by the histological examination of the resected specimen [3]. This surgical treatment is essential in order to avoid secondary complications due to the presence of heterotopic gastric mucosa causing acid secretion and perforation peritonitis [15]. Therefore, all the duplication must be excised otherwise complications due to a heterotopic mucosa can be seen [16].
All thoracoabdominal duplications since first reported by Valle and Wite, have been treated with a thoraco-phreno-laparotomy or thoracotomy associated to laparotomy [17]. It is only in the course of the recent years that laparoscopic treatment associated or not with a thoracoscopic approach was used. In fact combined thoraco-laparoscopy was described to be a successful surgical approach in case of gastro-intestinal duplication with large chest extension [17]. Apart from the possibility of a rapid and precise exploration of the whole abdominal and thoracic cavities, this mini-invasive approach allows to excise the entire mass, close communication with the rest of the digestive tract if it exists and control the hemostasis. It also has good esthetic results and a possibility of rapid recovery so that patients may have a “fast-track surgery” [18]. This approach allowed us to achieve subtotal exeresis of intra-pancreatic thoraco-abdominal duplication and thoracotomy was sufficient for complete exeresis of esogastric duplication.
5. Conclusion
Thoraco-abdominal duplication as extremely rare in children and cases reported in the literature are lacking. Clinical manifestations, most often non-specific, are the most common mode of revelation. Complete surgical resection, at best performed in a single time, is imperative and can be done by mini-invasive approach. Prognosis remains dependent on the nature of the malformations associated.
Consent for publication
Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
Provenance and peer review
Not commissioned, externally peer-reviewed.
Ethical approval
Ethical approval was obtained from the legal guardians of the children.
Funding
None.
Guarantor
Fatma Thamri.
Research registration number
None.
CRediT authorship contribution statement
-
⁎
Fatma Thamri: Drafted the article
-
⁎
Sondes Sahli: Drafted the article and revised it critically
-
⁎
Senda Houidi: Acquisition of data
-
⁎
Bochra Aziza: Conception of the study
-
⁎
Riadh Jouini: Supervision and Final approval of the version to be submitted.
Declaration of competing interest
Authors don't have any conflicts of interests in relation to the work described.
Acknowledgments
Acknowledgments
The authors acknowledge anyone who contributed toward the article including everyone who provided professional writing services or materials.
Submission declaration and verification
The authors declare that the work described has not been published previously, that it is not under consideration for publication elsewhere, that its publication is approved by all authors and tacitly or explicitly by the responsible authorities where the work was carried out, and that, if accepted, it will not be published elsewhere in the same form, in English or in any other language, including electronically without the written consent of the copyright holder.
References
- 1.Gálvez Y., Skába R., Kalousová J., Rousková B., Hríbal Z., Snajdauf J. Alimentary tract duplications in children: high incidence of associated anomalies. Eur. J. Pediatr. Surg. 2004;14(2):79–84. doi: 10.1055/s-2004-815852. avr. [DOI] [PubMed] [Google Scholar]
- 2.Valle A.R., White M.L. Thoracic gastric cyst. Ann. Surg. 1946;123:377–383. mars. [PubMed] [Google Scholar]
- 3.Miller J.M., Ginsberg M. Congenital duplication of the stomach. Arch. Surg. Chic. Ill. 1920;60(5):995–1001. doi: 10.1001/archsurg.1950.01250011018013. mai 1950. [DOI] [PubMed] [Google Scholar]
- 4.Agha R.A., Franchi T., Sohrabi C., Mathew G., Kerwan A., SCARE Group The SCARE 2020 guideline: updating consensus Surgical CAse REport (SCARE) guidelines. Int. J. Surg. Lond. Engl. 2020;84:226–230. doi: 10.1016/j.ijsu.2020.10.034. déc. [DOI] [PubMed] [Google Scholar]
- 5.Agha R.A., Borrelli M.R., Farwana R., Koshy K., Fowler A.J., Orgill D.P., et al. The PROCESS 2018 statement: updating consensus Preferred Reporting Of CasE Series in Surgery (PROCESS) guidelines. Int. J. Surg. Lond. Engl. 2018;60:279–282. doi: 10.1016/j.ijsu.2018.10.031. déc. [DOI] [PubMed] [Google Scholar]
- 6.Fisher H.C. Duplications of the intestinal tract in infants. AMA Arch. Surg. 1950;61(5):957–974. doi: 10.1001/archsurg.1950.01250020965018. Nov. [DOI] [PubMed] [Google Scholar]
- 7.Bhat N.A., Agarwala S., Wadhwa S., Gupta A.K., Bhatnagar V. Thoracoabdominal intestinal duplication with absent inferior vena cava. Pediatr. Surg. Int. 2001;17(7):540–542. doi: 10.1007/s003830000569. 1 sept. [DOI] [PubMed] [Google Scholar]
- 8.Pokorny W.J., Goldstein I.R. Enteric thoracoabdominal duplications in children. J. Thorac. Cardiovasc. Surg. 1984;87(6):821–824. juin. [PubMed] [Google Scholar]
- 9.Mazziotti M.V., Ternberg J.L. Continuous communicating esophageal and gastric duplication. J. Pediatr. Surg. 1997;32(5):775–778. doi: 10.1016/s0022-3468(97)90033-6. mai. [DOI] [PubMed] [Google Scholar]
- 10.Sonoda N., Takaya J., Okamoto K., Taniuchi S., Iwase S., Kobayashi Y., et al. Transdiaphragmatic duodenal duplication in a premature infant. J. Pediatr. Surg. 1987;22(4):372–373. doi: 10.1016/s0022-3468(87)80247-6. avr. [DOI] [PubMed] [Google Scholar]
- 11.Bratu I., Laberge J.-M., Flageole H., Bouchard S. Foregut duplications: is there an advantage to thoracoscopic resection? J. Pediatr. Surg. 2005;40(1):138–141. doi: 10.1016/j.jpedsurg.2004.09.025. janv. [DOI] [PubMed] [Google Scholar]
- 12.Shinnick J.K., Jain S., Alazraki A.L., Durham M.M. Ruptured thoracic and abdominal gastrointestinal duplication cysts presenting with failure to thrive. Pediatr. Emerg. Care. 2018;34(7):e128–e130. doi: 10.1097/PEC.0000000000001532. juill. [DOI] [PubMed] [Google Scholar]
- 13.Blanchet A.S., Depagne C., Nesme P., Perol M., Marchand B., de la Fouchardiere A. Une cause rare d’opacité thoracoabdominale. Rev. Mal. Respir. 2004;21(6):1171–1173. doi: 10.1016/s0761-8425(04)71595-8. 1 déc. [DOI] [PubMed] [Google Scholar]
- 14.Afzal N., Adil S.E.-R., Mushtaq A., Rahman A., Amanullah M. Oesophageal duplication cyst presenting as haemoptysis. J. Pak. Med. Assoc. 2013;63(5):633–634. mai. [PubMed] [Google Scholar]
- 15.Aubrespy P., Boureau M., Derlon S., Weisgerber M., Alessandrini P. Abdomino-thoracic digestive duplications. General review apropos of 2 cases. J. Chir. (Paris) 1984;121(2):85–95. févr. [PubMed] [Google Scholar]
- 16.Bui T., Bankhart M.F., Mandell G.A., Dickman P.S., Bae J.-O. Thoraco-abdominal enteric duplication cyst in association with neurenteric cyst, axial skeletal anomalies, and malrotation. Radiol. Case Rep. 2013;8(1):779. doi: 10.2484/rcr.v8i1.779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Martinez-Ferro M., Laje P., Piaggio L. Combined thoraco-laparoscopy for trans-diaphragmatic thoraco-abdominal enteric duplications. J. Pediatr. Surg. 2005;40(9):e37–e40. doi: 10.1016/j.jpedsurg.2005.05.080. sept. [DOI] [PubMed] [Google Scholar]
- 18.Azzie G., Beasley S. Diagnosis and treatment of foregut duplications. Semin. Pediatr. Surg. 2003;12(1):46–54. doi: 10.1053/spsu.2003.50000. févr. [DOI] [PubMed] [Google Scholar]





