Highlights
-
•
The endothelial glycocalyx is a ubiquitous intravascular structure essential for vascular homeostasis.
-
•
During sepsis, the glycocalyx is degraded via the collective action of a variety of redundant sheddases, the regulation of which remains the focus of active investigation.
-
•
Septic loss of the glycocalyx imparts both local vascular injury (leading to acute respiratory distress syndrome and acute kidney injury) as well as the systemic consequences of circulating glycosaminoglycan fragments (leading to cognitive dysfunction).
-
•
Glycocalyx degradation during sepsis is potentially shaped by clinically-modifiable factors, suggesting opportunities for therapeutic intervention to mitigate the end-organ consequences of sepsis.
Abbreviations: ARDS, Acute respiratory distress syndrome; LPS, Lipopolysaccharide; GAG, Glycosaminoglycan; PG, Proteoglycan; HPSE-1/2, Heparanase-1/2; ANP, Atrial Natriuretic Peptide; ADAM, A Disintegrin and Metalloproteinase; MMP, Matrix Metalloproteinase; Ang2, Angiopoietin-2; DAMP, Damage-associated Molecular Pattern; TIMP, Tissue inhibitors of matrix metalloproteinase; FFP, Fresh Frozen Plasma
Keywords: Sepsis, Proteoglycans, Glycosaminoglycans, Endothelial glycocalyx
Abstract
The glycocalyx is a ubiquitous structure found on endothelial cells that extends into the vascular lumen. It is enriched in proteoglycans, which are proteins attached to the glycosaminoglycans heparan sulfate, chondroitin sulfate, dermatan sulfate, keratan sulfate, and hyaluronic acid. In health and disease, the endothelial glycocalyx is a central regulator of vascular permeability, inflammation, coagulation, and circulatory tonicity. During sepsis, a life-threatening syndrome seen commonly in hospitalized patients, the endothelial glycocalyx is degraded, significantly contributing to its many clinical manifestations. In this review we discuss the intrinsically linked mechanisms responsible for septic endothelial glycocalyx destruction: glycosaminoglycan degradation and proteoglycan cleavage. We then examine the consequences of local endothelial glycocalyx loss to several organ systems and the systemic consequences of shed glycocalyx constituents. Last, we explore clinically relevant non-modifiable and modifiable factors that exacerbate or protect against endothelial glycocalyx shedding during sepsis.
Introduction
The glycocalyx is a ubiquitous structure on the apical membrane of endothelial cells, extending 0.5 to 4.5 µm into the vascular lumen depending upon the vascular bed [1], [2]. The glycocalyx is enriched in membrane-bound proteoglycans (PGs), which are proteins attached to the glycosaminoglycans (GAGs) heparan sulfate, chondroitin sulfate, dermatan sulfate, keratan sulfate, and hyaluronic acid [3]. First visualized within capillaries of the mouse diaphragm in the 1960s, the endothelial glycocalyx was originally thought to be functionally insignificant [4], however, over the last five decades it has become recognized as a key mediator of endothelial function.
In normal physiologic states, the endothelial glycocalyx has a variety of roles. Many GAG chains within the glycocalyx are highly-sulfated, conferring negative charge and creating an endovascular barrier that limits vascular permeability [3]. This barrier function also restricts circulating cells (e.g., neutrophils and platelets) from interacting with endothelial cells, thus affecting inflammation and coagulation [5], [6]. Additionally, glycocalyx deformation, which can be caused by increased intravascular shear forces, induces endothelial nitric oxide-mediated vasodilation [7]. In states of stress or disease such as sepsis [8], acute lung injury [9], ischemia–reperfusion injury [10], hyperglycemia [11], and hypervolemia [12] the endothelial glycocalyx is degraded, inducing significant pathophysiologic consequences including capillary leakage and edema, progressive inflammation, platelet aggregation, coagulopathy, loss of vascular tone and responsiveness, and ultimately end organ damage and possible death [13].
This review seeks to summarize existing knowledge concerning the mechanisms responsible for sepsis-related endothelial glycocalyx degradation and their subsequent local and systemic consequences. Furthermore, we explore how these mechanisms may be unwittingly amplified by common and potentially modifiable interventions (such as intravenous fluid resuscitation) routinely performed during the care of septic patients.
Endothelial glycocalyx degradation during sepsis
Sepsis has been recognized for thousands of years [14], yet only within the past few decades has there been significant progress in our understanding of its pathophysiology. Sepsis is characterized by life-threatening organ dysfunction caused by a dysregulated host response to infection [15]. While its early recognition and our clinical approach have improved over time, sepsis continues to have a profound global impact [16], [17].
In the last two decades, there have been substantial advances in our understanding of the importance of glycobiology to sepsis. These have been driven by a rapidly growing, highly-collaborative scientific community in the field, the development of innovative methods to measure the endothelial glycocalyx in vivo [9], [18], [19], [20], [21], and a technical revolution in the isolation and measurement of glycosaminoglycans in biologic tissues (for a comprehensive review of these techniques see [22]). Due to these advances, it has recently been discovered that during sepsis the endothelial glycocalyx is degraded. This phenomenon has been observed in numerous human and pre-clinical studies [23]. For example, Nieuwdorp et al. administered low doses of a bacterial endotoxin (Lipopolysaccharide, LPS) to human volunteers and found decreases in the thickness of the sublingual glycocalyx as measured by orthogonal polarization spectroscopy from 0.6 to 0.3 µm [21]. These measurements correlated with increases in plasma levels of glycocalyx-derived GAG constituents, suggesting that luminal glycocalyx degradation, rather than internalization, was responsible for thinning during sepsis [21]. Consistent with these clinical findings, Kataoka et al. reported the glycocalyx in septic mice to be 0.98 nm compared to 70.68 nm in matched controls via electron microscopy [20].
Mechanisms of endothelial glycocalyx degradation during sepsis
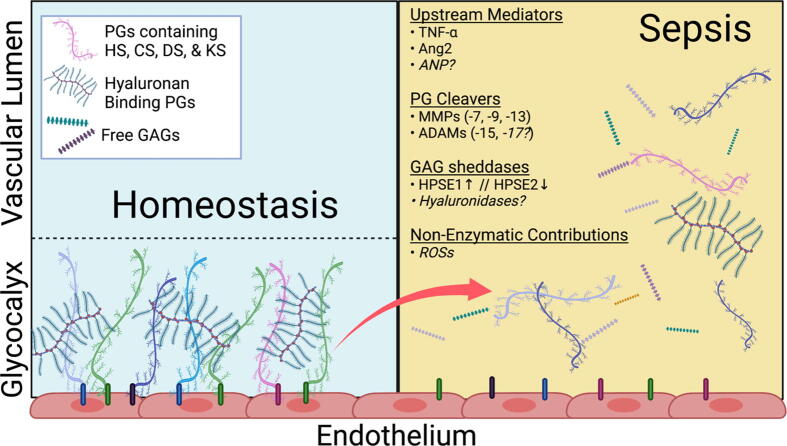
Endothelial glycocalyx degradation during sepsis occurs via two linked “sheddase” mechanisms: GAG degradation and core PG cleavage (see Fig. 1). During sepsis, both GAG oligosaccharides [24] and PG extracellular domains (ectodomains) [25], [26] circulate, but the precise interplay between GAG degradation and PG cleavage underlying endothelial glycocalyx degradation remains incompletely understood.
Fig. 1.
Mechanisms Responsible for Endothelial Glycocalyx Degradation during Sepsis. During normal physiological conditions (left) the endothelial glycocalyx participates in maintenance of vascular homeostasis. During sepsis (right) the glycocalyx is degraded by various enzymatic and non-enzymatic pathways that are upregulated and/or activated by several known upstream mediators leading to local and systemic consequences. PGs proteoglycan, HS heparan sulfate, CS chondroitin sulfate, DS dermatan sulfate, KS keratan sulfate, GAG glycosaminoglycan, HPSE heparanase, ROSs Reactive Oxygen Species. Created with Biorender.com.
Heparanase-1 (HPSE1), the enzymatically active form of the proenzyme proheparanase, is the only identified mammalian enzyme capable of degrading heparan sulfate polysaccharides into shorter chain oligosaccharides [27], [28], [29]. It is also the lone GAG-sheddase known to be activated during sepsis. Prior to recognition of its importance to sepsis, HPSE1 had been found to be a key pathophysiologic mediator in cancer and inflammatory disorders [27], [30]. Both preclinical and clinical studies have established the central role of HPSE1-mediated heparan sulfate degradation to septic endothelial glycocalyx degradation [9], [31], [32], [33]. Heparanase-2 (HPSE2), a molecule with significant homology to HPSE1 that lacks glucuronidase activity, directly inhibits HPSE1 [34], [35]. Since this discovery, it has been hypothesized that the relative expression (and/or activity) of HPSE1 to HPSE2 may determine the degree of heparan sulfate shedding and subsequent glycocalyx collapse during sepsis [36].
Hyaluronan, which can be degraded by six different hyaluronidases, is unique in that it is unsulfated and not covalently bound to proteoglycans [37], [38]. It is an important component of the glycocalyx due to its ability to form complexes with proteins and other sulfated GAGs, helping to maintain structural stability [39]. Patients with sepsis have higher serum levels of hyaluronan than non-septic patients and urine hyaluronan levels predict both renal failure and mortality in patients with sepsis [40], [41]. While increases in circulating hyaluronan during sepsis have been described, upregulation of endogenous hyaluronidases has not been reported. Several pathogenic microorganisms are known to produce hyaluronidases, but these pathogen-derived enzymes have not been shown to directly contribute to glycocalyx degradation nor circulating hyaluronan levels during sepsis [42], [43], [44].
Less is known about the circulation of chondroitin sulfate, dermatan sulfate, and keratan sulfate during sepsis. Urinary chondroitin sulfate levels are elevated and predictive of outcomes [41], but no chondroitin sulfate-directed sheddase has been found to be involved in sepsis. Several chondroitin sulfate PGs (i.e., syndecans, decorin, and thrombomodulin) are elevated, which may account for elevations in circulating chondroitin sulfate as measured by GAG isolation and mass spectrometry, a technique that is unable to routinely distinguish between free and PG-bound GAGs [25], [45]. Dermatan sulfate and keratan sulfate levels have never been directly measured during sepsis, but it is likely that proteoglycans containing these GAGs are shed from the endothelial glycocalyx during sepsis. If these GAGs are indeed freely circulating, candidate sheddases include hyaluronidases, testicular hyaluronidase (SPAM1, [46]), and/or non-specific degradation by reactive oxygen species [47].
Like GAGs, PG ectodomains have also been found to be released from the endothelial glycocalyx during sepsis. Matrix metalloproteinases (MMPs) and members of the A Disintegrin and Metalloproteinase (ADAMs) enzyme family are zinc-dependent endopeptidases that share many characteristics with one another including their roles in inflammation and their ability to cleave PGs from the endothelial glycocalyx [48]. Plasma concentrations of MMPs correlate with severity of sepsis [49], [50] and their inhibition confers protection against sepsis [51], [52]. While 24 mammalian MMPs are known to exist, only MMPs −7, −9, and −13 have been directly implicated in PG shedding during sepsis [53], [54], [55], [56]. Members of the ADAMs family are proteases that are similarly upregulated during sepsis with levels correlating with severity and outcomes [57]. ADAM15 cleaves PGs of the endothelial glycocalyx in pre-clinical models of sepsis and in ex vivo preparations of human lungs perfused with LPS [58]. ADAM17 may also be involved in degradation of the endothelial glycocalyx during sepsis, as it can cleave syndecans [59], [60].
Atrial Natriuretic Peptide (ANP), which is released by cardiac myocytes in response to atrial distention from vascular volume overload, has also been described as a potential endothelial glycocalyx sheddase. ANP is believed to counteract volume overload in part by increasing endothelial microvascular permeability to facilitate fluid extravasation from vasculature beds [61]. Increases in ANP-related microvascular permeability have been posited to be mediated by endothelial glycocalyx degradation. While studies utilizing intravenous administration of ANP to isolated guinea pig hearts demonstrated increased shedding of the PG syndecan-1 [12], subsequent large animal and human studies have not shown this same effect [25], [62]. Therefore, further studies are necessary to fully elucidate ANP’s role in endothelial glycocalyx degradation during sepsis.
These sheddase mechanisms are activated by upstream agonists including proinflammatory cytokines. Sepsis-related activation of the GAG sheddase HPSE1 is dependent upon endothelial-derived TNF-α [9]. The Angiopoietin-2 (Ang2)/Tie2 pathway, which plays a major role in endothelial homeostasis during sepsis, has also been shown to be an upstream regulator of endothelial glycocalyx sheddases. Ang2 is an endogenous antagonist to Tie2 signaling. It is induced during sepsis, which leads to vascular inflammation, thrombosis, and increased permeability [63], [64], [65]. In septic patients, higher levels of circulating Ang2 have been associated with increased circulating markers of glycocalyx degradation [66], [25]. Concordantly, Ang2, in a HPSE1-dependent fashion, is a potent degrader of the glycocalyx both in vivo and in vitro [67], and Ang2 inhibition can reduce endothelial glycocalyx shedding in murine models of sepsis [68]. Many other molecules including macrophage migratory inhibitor factor (MIF1), phorbol esters, and Tissue inhibitors of matrix metalloproteinases (TIMPs) are involved in glycocalyx degradation in other diseases and at non-endothelial surfaces, suggestive of their potential relevance to septic endothelial glycocalyx degradation [55], [69], [70], [71], [72], [73], [74].
The enzymatic destruction of the endothelial glycocalyx through these and other interconnected cellular mechanisms has significant physiologic consequences. Not only are there local effects from the loss of this protective barrier, but glycocalyx degradation products themselves can travel through the bloodstream and affect distant sites (Table 1).
Table 1.
Consequences of Septic Endothelial Glycocalyx Degradation. Disruption of the endothelial glycocalyx during sepsis creates a vulnerable endothelial layer. This leads to local consequences, ultimately contributing to end organ damage. Once degraded, fragments of the glycocalyx also travel though the blood and become systemic effectors.
| Local | Systemic |
|---|---|
| General | General |
| Enhanced leukocyte-endothelial interaction → Local Inflammation | Augmenting inflammatory signaling → Systemic inflammation |
| Platelet adhesion → Development of microthrombi | Binding of coagulation factors → Coagulopathy |
| Nitric oxide-mediated vasodilation → Dysfunction of vascular tone | Inhibition of antimicrobial peptides → Increased susceptibility to infection |
| Increased vascular permeability → Tissue edema | Binding histones → Dampened inflammation |
| Organ Specific | Organ Specific |
| Lung injury and ARDS | Deposition of pathogenic heparan sulfate in the brain → Cognitive dysfunction |
| Kidney injury | |
| Gut barrier breakdown | |
| Increased blood brain barrier permeability |
Abbreviation: ARDS = Acute Respiratory Distress Syndrome
Local consequences of endothelial glycocalyx degradation
The endothelial glycocalyx has multiple roles in physiologic homeostasis including barrier function, mechanotransduction of shear stress, inhibition of leukocyte-endothelial interaction, and dampening of coagulation at the endothelial surface. Disruption of the glycocalyx during sepsis consequently promotes vascular permeability and tissue edema, circulatory tonicity dysfunction, enhanced tissue inflammation, and coagulopathy. The clinical manifestations and severity of these processes, however, varies depending on the organ affected.
In the lungs, loss of the endothelial glycocalyx contributes to lung injury and development of the acute respiratory distress syndrome (ARDS), a common complication of sepsis. In preclinical models of sepsis, pulmonary endothelial glycocalyx degradation increases neutrophil adhesion and propagates lung injury in a HPSE1-dependent fashion [9], [31], [75].
Kidney injury during sepsis is also common and associated with poor clinical outcomes. Early activation of glomerular HPSE1 in the kidneys during experimental sepsis coincides with injury; consistent with lung injury, HPSE1 inhibition attenuates injury [33]. Unlike septic lung injury, activation of glomerular HPSE1 does not alter vascular permeability nor neutrophil influx, suggesting distinct consequences of HPSE1-mediated glycocalyx degradation in the kidneys [33].
Intestinal injury is often overlooked during sepsis, despite gastrointestinal tract inflammation and gut barrier breakdown being hallmarks of the disease [76]. Disruption of this barrier may promote bacterial translocation into the blood stream. Chen et al. demonstrated that HPSE1 inhibition in experimental sepsis attenuates intestinal inflammation [77], indirect evidence of the importance of the endothelial glycocalyx to gastrointestinal integrity.
The endothelial glycocalyx also exists in the brain where it is believed to play an important role in the function of the blood–brain barrier [78]. The endothelium of all cerebral capillaries expresses an endothelial glycocalyx that is significantly denser than the glycocalyx of other organs [79]. In an endotoxemia model of sepsis, the cerebral endothelial glycocalyx, while degraded, is less damaged than in other vascular beds [79]. The underlying mechanism and precise consequences of this destruction have not been fully elucidated, but it is plausible that this may contribute to sepsis-associated brain dysfunction.
Systemic consequences of endothelial glycocalyx degradation
Circulating glycocalyx fragment levels have been extensively studied as biomarkers of sepsis severity. Circulating heparan sulfate levels are substantially higher in critically ill patients with sepsis compared to healthy controls, positively correlate with severity of illness, and predict in-hospital mortality [24]. Urine hyaluronan levels have likewise been shown to predict renal failure and hospital mortality [41]. Intriguingly, it has recently been discovered that circulating glycocalyx fragments (in particular heparan sulfate), beyond being markers of endothelial glycocalyx fragments may mechanistically contribute to the multiorgan consequences of sepsis [80].
The strong negative charge of the soluble GAGs, heparan, chondroitin, dermatan, and keratan sulfate, enables them to bind to proteins and modify downstream function. Accordingly, the large amounts of GAGs released into the bloodstream during sepsis can modulate various signaling cascades central to sepsis pathophysiology. For example, heparan sulfate is known to bind interferon gamma (INFγ). While bound by heparan sulfate, INFγ is protected from normal degradation, thus increasing its half-life and prolonging its pro-inflammatory effects [81]. Additionally, heparan sulfate fragments act as damage-associated molecular patterns (DAMPs) by binding to toll-like receptor 4, similarly augmenting systemic inflammation [82]. GAGs also bind to and neutralize endogenous antimicrobial peptides, increasing host susceptibility to infection [83]. Similarly, GAGs interact strongly with multiple coagulation factors including antithrombin II, Factors Xa, IXa, IIa, and protein C thus contributing to coagulopathy of sepsis [84], [85]. Additionally, proteoglycans released from the endothelial glycocalyx during sepsis have systemic effects. For example, the ectodomain of CD44 released from the glycocalyx during sepsis damages endothelial barrier function through disruption of adherens junctions [58]. While beyond the scope of this review, several other PGs known to be released from the endothelial glycocalyx during sepsis are known to substantially impact molecular pathways important to sepsis pathophysiology (see [86], [87] for excellent reviews on these topics).
Beyond their effects within the circulation, glycocalyx fragments pathologically invade the brain during sepsis. Brain dysfunction is a common and devastating problem after recovery from sepsis [88]. While the precise pathophysiology of these effects is not fully understood, the hippocampus, a brain region essential for memory formation, is central to this common sequela [89]. During sepsis, endothelial glycocalyx-derived heparan sulfate oligosaccharides penetrate the hippocampus with striking specificity, sparing other systemic and neuronal vascular beds [90]. A subset of these hippocampus-penetrating oligosaccharides (those enriched in -NS and -NS2S sulfation) can avidly bind and inhibit brain-derived neurotrophic factor (BDNF), a neurotrophin vital to learning and memory [91], [92]. Higher circulating levels of BDNF-avid heparan sulfate oligosaccharides were found to be associated with persistent cognitive impairment after recovery from sepsis, strongly suggesting a role in its pathophysiology.
Circulating GAGs also have potential beneficial effects during sepsis. Histones are cationic proteins present in nuclei of all eukaryotic cells. During sepsis, histones are released into circulation and act as DAMPs. Their release promotes proinflammatory cytokine pathways, which propagates inflammation and directly contributes to cellular injury [93]. Highly-sulfated heparan sulfate oligosaccharides (fragments ≥ 10 saccharides in length) are capable of binding and inhibiting histones, thus dampening inflammation. The mechanisms underlying the protective effect of glycocalyx fragments on histone-induced endothelial injury are complex, however, as even short heparan sulfate oligosaccharide (incapable of bindings histones) can attenuate histone-induced lung injury [94].
Non-modifiable factors implicated in septic endothelial glycocalyx degradation
Most early descriptions of endothelial glycocalyx degradation during sepsis were investigated in either bacterial infections in humans [24], [83] or pre-clinical bacterial endotoxin models [9], [21], [95]. Similarly, many sepsis-inducing, non-bacterial infectious diseases have been found to cause endothelial glycocalyx degradation including Plasmodium falciparum malaria [96], Crimean-Congo Hemorrhagic Fever [97], Hantavirus [98], Dengue Fever [99], Influenza [100], and COVID-19 [36], [101], [102]. Additionally, several clinical characteristics of septic patients have been associated with greater glycocalyx damage including increased severity of illness [25], [83], [103], non-pulmonary sources of sepsis [24], [103], and occurrence of bacteremia [25].
Beyond infectious etiologies and in-hospital characteristics of septic patients, other factors may affect glycocalyx integrity during illness. Both chronic kidney disease [104] and hemodialysis in patients with end stage renal disease have been associated with endothelial glycocalyx breakdown [105]. Additionally, acute processes such as ischemia [106], trauma [107], burn injury [108], or severe hemorrhage [109], which independently cause endothelial glycocalyx degradation, likely exacerbate damage during sepsis. Less is known about contributions of other patient characteristics such as age and most medical comorbidities to endothelial glycocalyx degradation during sepsis, although some patient characteristics have been associated with baseline endothelial glycocalyx derangement. The endothelial glycocalyx in patients with vascular diseases (including atherosclerosis and diabetes), cirrhosis, and of older age have all been shown to be abnormal at baseline [110], [111], [112], [113]. Whether such pre-existing abnormalities worsen or attenuate septic shedding has not been directly studied.
Modifiable factors implicated in septic endothelial glycocalyx degradation
Intravenous fluids have long been a mainstay in the treatment of sepsis. There is mounting evidence, however, that the beneficial hemodynamic effects of fluid resuscitation may be partially offset by poorly-understood deleterious effects on organ function, based upon associations between aggressive fluid resuscitation practices and worsened disease severity, higher rates of intubation, and increased risk of need for renal replacement therapy [114], as well as increased patient mortality [115]. These harmful effects of aggressive fluid resuscitation may be in part mediated by iatrogenic destruction of the glycocalyx [116]. This is supported by a preclinical investigation in an ovine model of sepsis in which fluid resuscitation increased glycocalyx degradation as measured by circulating hyaluronan levels [117]. This association has also been examined in septic patients. In a retrospective study that included two independent patient cohorts affected by sepsis, a significant association was found between the volume of intravenous fluids administered during sepsis resuscitation and degree of endothelial glycocalyx damage, even when adjusted for age and disease severity [25]. In a prospective clinical study, Saoraya et. al evaluated patients treated with rapid (30 mL/kg/hr) or conservative fluid resuscitation (10 mL/kg/hr). They found a higher reliance on vasopressors and mechanical ventilation in the rapid fluid resuscitation arm with a trend towards higher syndecan-1 levels (a marker of glycocalyx degradation) and worsened mortality [118]. In aggregate, these findings suggest that worse clinical outcomes associated with overly aggressive fluid resuscitation strategies during sepsis may be mediated by endothelial glycocalyx destruction.
The clinical utility of steroids has been extensively studied for the treatment of septic shock, the most severe form of sepsis. Although there have been conflicting results regarding their clinical utility, treatment guidelines continue to recommend the use of steroids in a subset of patients with septic shock [119]. At a cellular level, steroids are known to decrease cellular permeability and transendothelial fluid flow [120], and preservation of the endothelial glycocalyx may underlie these effects. In a study of isolated guinea pig hearts, high dose hydrocortisone prior to induction of ischemia reduced the rate of flow of paracellular fluid into tissue, extravasation of colloid from the vasculature, and serum heparan sulfate and syndecan-1 levels [121]. In a similar study, injection of hydrocortisone prior to infusion of TNF-α attenuated vascular permeability and glycocalyx degradation [122]. In both experiments the glycocalyx was measured via electron microscopy and glycocalyx integrity was maintained in the steroid treatment arm, while the control arm demonstrated substantial glycocalyx loss. This has also been observed in humans undergoing cardiac surgery requiring cardiopulmonary bypass. Those randomized to hydrocortisone when compared to placebo demonstrated significant reductions in plasma heparan sulfate and syndecan-1 during and after surgery [123]. While this evidence suggests beneficial effects of steroids on endothelial glycocalyx stability, these observations have yet to be validated in studies of sepsis.
During infection, activation of the coagulation cascade can be an adaptive process, serving to prevent dissemination of microbes [124]. However, in excess, this can lead to microcirculatory damage, organ failure, and disseminated intravascular coagulopathy [125]. Antithrombin is a known physiologic anticoagulant whose activity is accentuated by binding of heparan sulfate from the endothelial glycocalyx [69]. In a rat model of sepsis, recombinant antithrombin was shown to decrease leukocyte adhesion, reduce parameters of DIC, lower circulating syndecan-1 and hyaluronan levels, and inhibit glycocalyx destruction [126]. In a canine shock model, unfractionated heparin in combination with standard care reduced glycocalyx shedding when compared to standard care alone [127]. In clinical practice anticoagulant therapies are not routinely utilized in sepsis patients due to concerns for increased risk of hemorrhage, but their potential off-target effect of endothelial glycocalyx preservation has not been systematically studied.
Therapeutic plasma exchange with replacement by fresh frozen plasma (FFP) has been posited as a potential therapy for sepsis. As previously mentioned, heparan sulfate, when shed into the blood, may act at distant sites (e.g., brain) and as a pro-inflammatory signal via DAMP signaling pathways [82]. Plasma exchange may reduce circulating pro-inflammatory signals including heparan sulfate or replace components of healthy plasma to restore a balance in regulatory enzymes of the glycocalyx. A series of exploratory studies demonstrated that plasma exchange with FFP as replacement fluid was safe and resulted in favorable hemodynamic improvements, improved cytokine profiles, and improved endothelial barrier function [128], [129]. Additional clinical trials seek to explore if plasma exchange is indeed beneficial for septic patients (ClinicalTrials.gov NCT04057872, NCT03844542). Interestingly, FFP administration alone during sepsis without plasma exchange may also protect and/or restore the glycocalyx via unclear molecular mechanisms [130], [131]. A summary of these non-modifiable and modifiable factors associated with endothelial glycocalyx degradation are included in Table 2.
Table 2.
Factors Associated with Septic Endothelial Glycocalyx Degradation.
| Non-Modifiable Factors |
|---|
| •Infection Characteristics |
| •Severity |
| •Site* |
| •Type |
| •Co-occurring Acute Medical Problems |
| •Ischemia |
| •Hemorrhage |
| •Burn Injury |
| •Trauma |
| •Pre-existing Conditions** |
| •Chronic Kidney Disease |
| •Diabetes Mellitus |
| •Atherosclerosis |
| •Cirrhosis |
| •Older Age |
| Clinically Modifiable Factors |
| •Fluid Resuscitation Strategy (Harm) |
| •Corticosteroids (Benefit) |
| •Anticoagulants (Benefit) |
| •Plasma Exchange (Benefit) |
| •Fresh Frozen Plasma (Benefit) |
*Bacteremia greater than non-bacteremia; non-pulmonary greater than pulmonary.
** These conditions are associated with baseline glycocalyx derangement, but are not proven to modify shedding during sepsis.
Conclusions
The endothelial glycocalyx, once believed to be physiologically irrelevant, is now widely accepted as a crucial mediator of endothelial homeostasis that contributes to sepsis pathophysiology. While there is still much to be elucidated, several direct “sheddases” (e.g., heparanase, MMPs, ADAMs) and upstream pathways (e.g., TNF-α, Ang2) have been convincingly implicated in its degradation during sepsis. Despite clear pathologic consequences, the redundant nature of these pathways suggests physiologic benefits of its destruction and challenges therapies solely focused on normalizing glycocalyx integrity. A better understanding of the mechanisms, adaptive roles, and maladaptive consequences of endothelial glycocalyx degradation during sepsis will likely offer opportunities to refine our clinical approach to caring for patients with sepsis.
Funding Sources
This work was supported by the National Institutes of Health, [Grants R03AG074056 (JAH), K08HL159353 (JAH), T32HL007085 (RCS)]; and the American Thoracic Society Research Program [ATS Unrestricted Research Grant in Critical Care (JAH)].
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgement
The authors would like to acknowledge Christophe J. Langouët-Astrié for assistance with the formulation and creation of Fig. 1 using Biorender.com.
References
- 1.Vink H., Duling B.R. Identification of distinct luminal domains for macromolecules, erythrocytes, and leukocytes within mammalian capillaries. Circ. Res. 1996;79(3):581–589. doi: 10.1161/01.res.79.3.581. [DOI] [PubMed] [Google Scholar]
- 2.Megens R.T.A., Reitsma S., Schiffers P.H.M., Hilgers R.H.P., De Mey J.G.R., Slaaf D.W., oude Egbrink M.G.A., van Zandvoort M.A.M.J. Two-photon microscopy of vital murine elastic and muscular arteries. Combined structural and functional imaging with subcellular resolution. J. Vasc. Res. 2007;44(2):87–98. doi: 10.1159/000098259. [DOI] [PubMed] [Google Scholar]
- 3.Weinbaum S., Tarbell J.M., Damiano E.R. The structure and function of the endothelial glycocalyx layer. Annu. Rev. Biomed. Eng. 2007;9(1):121–167. doi: 10.1146/annurev.bioeng.9.060906.151959. [DOI] [PubMed] [Google Scholar]
- 4.Luft J.H. Fine structures of capillary and endocapillary layer as revealed by ruthenium red. Fed Proc. 1966;25(6):1773–1783. [PubMed] [Google Scholar]
- 5.Mulivor A.W., Lipowsky H.H. Role of glycocalyx in leukocyte-endothelial cell adhesion. Am. J. Physiol. Heart Circ. Physiol. 2002;283(4):H1282–H1291. doi: 10.1152/ajpheart.00117.2002. [DOI] [PubMed] [Google Scholar]
- 6.Vink H., Constantinescu A.A., Spaan J.A.E. Oxidized lipoproteins degrade the endothelial surface layer : implications for platelet-endothelial cell adhesion. Circulation. 2000;101(13):1500–1502. doi: 10.1161/01.cir.101.13.1500. [DOI] [PubMed] [Google Scholar]
- 7.Florian J.A., Kosky J.R., Ainslie K., Pang Z., Dull R.O., Tarbell J.M. Heparan sulfate proteoglycan is a mechanosensor on endothelial cells. Circ. Res. 2003;93(10) doi: 10.1161/01.RES.0000101744.47866.D5. [DOI] [PubMed] [Google Scholar]
- 8.Chelazzi C., Villa G., Mancinelli P., De Gaudio A.R., Adembri C. Glycocalyx and sepsis-induced alterations in vascular permeability. Crit. Care. 2015;19(1) doi: 10.1186/s13054-015-0741-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schmidt E.P., Yang Y., Janssen W.J., Gandjeva A., Perez M.J., Barthel L., Zemans R.L., Bowman J.C., Koyanagi D.E., Yunt Z.X., Smith L.P., Cheng S.S., Overdier K.H., Thompson K.R., Geraci M.W., Douglas I.S., Pearse D.B., Tuder R.M. The pulmonary endothelial glycocalyx regulates neutrophil adhesion and lung injury during experimental sepsis. Nat. Med. 2012;18(8):1217–1223. doi: 10.1038/nm.2843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Abassi Z., Armaly Z., Heyman S.N. Glycocalyx Degradation in Ischemia-Reperfusion Injury. Am. J. Pathol. 2020;190(4):752–767. doi: 10.1016/j.ajpath.2019.08.019. [DOI] [PubMed] [Google Scholar]
- 11.Nieuwdorp M., Mooij H.L., Kroon J., Atasever B., Spaan J.A.E., Ince C., Holleman F., Diamant M., Heine R.J., Hoekstra J.B.L., Kastelein J.J.P., Stroes E.S.G., Vink H. Endothelial glycocalyx damage coincides with microalbuminuria in type 1 diabetes. Diabetes. 2006;55(4):1127–1132. doi: 10.2337/diabetes.55.04.06.db05-1619. [DOI] [PubMed] [Google Scholar]
- 12.Bruegger D., Jacob M., Rehm M., Loetsch M., Welsch U., Conzen P., Becker B.F. Atrial natriuretic peptide induces shedding of endothelial glycocalyx in coronary vascular bed of guinea pig hearts. Am. J. Physiol. Heart Circ. Physiol. 2005;289(5):H1993–H1999. doi: 10.1152/ajpheart.00218.2005. [DOI] [PubMed] [Google Scholar]
- 13.Becker B.F., Chappell D., Bruegger D., Annecke T., Jacob M. Therapeutic strategies targeting the endothelial glycocalyx: acute deficits, but great potential. Cardiovasc. Res. 2010;87(2):300–310. doi: 10.1093/cvr/cvq137. [DOI] [PubMed] [Google Scholar]
- 14.Funk D.J., Parrillo J.E., Kumar A. Sepsis and septic shock: a history. Crit. Care Clin. 2009;25(1):83–101. doi: 10.1016/j.ccc.2008.12.003. [DOI] [PubMed] [Google Scholar]
- 15.Singer M., Deutschman C.S., Seymour C.W., Shankar-Hari M., Annane D., Bauer M., Bellomo R., Bernard G.R., Chiche J.-D., Coopersmith C.M., Hotchkiss R.S., Levy M.M., Marshall J.C., Martin G.S., Opal S.M., Rubenfeld G.D., van der Poll T., Vincent J.-L., Angus D.C. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3) JAMA. 2016;315(8):801. doi: 10.1001/jama.2016.0287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rhee C., Dantes R., Epstein L., Murphy D.J., Seymour C.W., Iwashyna T.J., Kadri S.S., Angus D.C., Danner R.L., Fiore A.E., Jernigan J.A., Martin G.S., Septimus E., Warren D.K., Karcz A., Chan C., Menchaca J.T., Wang R., Gruber S., Klompas M. Incidence and Trends of Sepsis in US Hospitals Using Clinical vs Claims Data, 2009–2014. JAMA. 2017;318(13):1241. doi: 10.1001/jama.2017.13836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Buchman T.G., Simpson S.Q., Sciarretta K.L., Finne K.P., Sowers N., Collier M., Chavan S., Oke I., Pennini M.E., Santhosh A., Wax M., Woodbury R., Chu S., Merkeley T.G., Disbrow G.L., Bright R.A., MaCurdy T.E., Kelman J.A. Sepsis Among Medicare Beneficiaries: 1. The Burdens of Sepsis, 2012–2018*. Crit. Care Med. 2020;48(3):276–288. doi: 10.1097/CCM.0000000000004224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yang Y., Hippensteel J.A., Schmidt E.P. Intravital Microscopy in the Mouse Lung. Methods Mol. Biol. 2018;1809:331–339. doi: 10.1007/978-1-4939-8570-8_21. [DOI] [PubMed] [Google Scholar]
- 19.Nieuwdorp M., Meuwese M.C., Mooij H.L., Ince C., Broekhuizen L.N., Kastelein J.J.P., Stroes E.S.G., Vink H. Measuring endothelial glycocalyx dimensions in humans: a potential novel tool to monitor vascular vulnerability. J. Appl. Physiol. 2008;104(3):845–852. doi: 10.1152/japplphysiol.00440.2007. [DOI] [PubMed] [Google Scholar]
- 20.Kataoka H., Ushiyama A., Akimoto Y., Matsubara S., Kawakami H., Iijima T. Structural Behavior of the Endothelial Glycocalyx Is Associated With Pathophysiologic Status in Septic Mice: An Integrated Approach to Analyzing the Behavior and Function of the Glycocalyx Using Both Electron and Fluorescence Intravital Microscopy. Anesth. Analg. 2017;125(3):874–883. doi: 10.1213/ANE.0000000000002057. [DOI] [PubMed] [Google Scholar]
- 21.Nieuwdorp M., et al. Tumor necrosis factor-alpha inhibition protects against endotoxin-induced endothelial glycocalyx perturbation. Atherosclerosis. 2009;202(1):296–303. doi: 10.1016/j.atherosclerosis.2008.03.024. [DOI] [PubMed] [Google Scholar]
- 22.Khan S.A., Mason R.W., Kobayashi H., Yamaguchi S., Tomatsu S. Advances in glycosaminoglycan detection. Mol. Genet. Metab. 2020;130(2):101–109. doi: 10.1016/j.ymgme.2020.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Colbert J.F., Schmidt E.P. Endothelial and microcirculatory function and dysfunction in sepsis. Clin. Chest Med. 2016;37(2):263–275. doi: 10.1016/j.ccm.2016.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Schmidt E.P., Li G., Li L., Fu L.i., Yang Y., Overdier K.H., Douglas I.S., Linhardt R.J. The circulating glycosaminoglycan signature of respiratory failure in critically ill adults. J. Biol. Chem. 2014;289(12):8194–8202. doi: 10.1074/jbc.M113.539452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hippensteel J.A., Uchimido R., Tyler P.D., Burke R.C., Han X., Zhang F., McMurtry S.A., Colbert J.F., Lindsell C.J., Angus D.C., Kellum J.A., Yealy D.M., Linhardt R.J., Shapiro N.I., Schmidt E.P. Intravenous fluid resuscitation is associated with septic endothelial glycocalyx degradation. Crit. Care. 2019;23(1) doi: 10.1186/s13054-019-2534-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fisher J., Linder A., Bentzer P. Elevated plasma glypicans are associated with organ failure in patients with infection. Intens. Care Med. Exp. 2019;7(1):2. doi: 10.1186/s40635-018-0216-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Vlodavsky I., Friedmann Y., Elkin M., Aingorn H., Atzmon R., Ishai-Michaeli R., Bitan M., Pappo O., Peretz T., Michal I., Spector L., Pecker I. Mammalian heparanase: gene cloning, expression and function in tumor progression and metastasis. Nat. Med. 1999;5(7):793–802. doi: 10.1038/10518. [DOI] [PubMed] [Google Scholar]
- 28.Vreys V., David G. Mammalian heparanase: what is the message? J. Cell Mol. Med. 2007;11(3):427–452. doi: 10.1111/j.1582-4934.2007.00039.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Abboud-Jarrous G., Rangini-Guetta Z., Aingorn H., Atzmon R., Elgavish S., Peretz T., Vlodavsky I. Site-directed mutagenesis, proteolytic cleavage, and activation of human proheparanase. J. Biol. Chem. 2005;280(14):13568–13575. doi: 10.1074/jbc.M413370200. [DOI] [PubMed] [Google Scholar]
- 30.Goldberg R., Meirovitz A., Hirshoren N., Bulvik R., Binder A., Rubinstein A.M., Elkin M. Versatile role of heparanase in inflammation. Matrix Biol. 2013;32(5):234–240. doi: 10.1016/j.matbio.2013.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang L., Huang X., Kong G., Xu H., Li J., Hao D., Wang T., Han S., Han C., Sun Y., Liu X., Wang X. Ulinastatin attenuates pulmonary endothelial glycocalyx damage and inhibits endothelial heparanase activity in LPS-induced ARDS. Biochem. Biophys. Res. Commun. 2016;478(2):669–675. doi: 10.1016/j.bbrc.2016.08.005. [DOI] [PubMed] [Google Scholar]
- 32.Martin L., De Santis R., Koczera P., Simons N., Haase H., Heinbockel L., Brandenburg K., Marx G., Schuerholz T., Stover C.M. The Synthetic Antimicrobial Peptide 19–2.5 Interacts with Heparanase and Heparan Sulfate in Murine and Human Sepsis. PLoS ONE. 2015;10(11):e0143583. doi: 10.1371/journal.pone.0143583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lygizos M.I., Yang Y., Altmann C.J., Okamura K., Hernando A.A., Perez M.J., Smith L.P., Koyanagi D.E., Gandjeva A., Bhargava R., Tuder R.M., Faubel S., Schmidt E.P. Heparanase mediates renal dysfunction during early sepsis in mice. Physiol. Rep. 2013;1(6):e00153. doi: 10.1002/phy2.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Levy-Adam F., Feld S., Cohen-Kaplan V., Shteingauz A., Gross M., Arvatz G., Naroditsky I., Ilan N., Doweck I., Vlodavsky I. Heparanase 2 interacts with heparan sulfate with high affinity and inhibits heparanase activity. J. Biol. Chem. 2010;285(36):28010–28019. doi: 10.1074/jbc.M110.116384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kiyan Y., Tkachuk S., Kurselis K., Shushakova N., Stahl K., Dawodu D., Kiyan R., Chichkov B., Haller H. Heparanase-2 protects from LPS-mediated endothelial injury by inhibiting TLR4 signalling. Sci. Rep. 2019;9(1) doi: 10.1038/s41598-019-50068-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Stahl K., et al. Injury to the Endothelial Glycocalyx in Critically Ill COVID-19 Patients. Am. J. Respir. Crit. Care Med. 2020(ja). doi: 10.1164/rccm.202007-2676LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jung H. Hyaluronidase: An overview of its properties, applications, and side effects. Arch Plast Surg. 2020;47(4):297–300. doi: 10.5999/aps.2020.00752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Fraser J.R.E., Laurent T.C., Laurent U. Hyaluronan: its nature, distribution, functions and turnover. J. Intern. Med. 1997;242(1):27–33. doi: 10.1046/j.1365-2796.1997.00170.x. [DOI] [PubMed] [Google Scholar]
- 39.Broekhuizen L.N., Mooij H.L., Kastelein J.JP., Stroes E.SG., Vink H., Nieuwdorp M. Endothelial glycocalyx as potential diagnostic and therapeutic target in cardiovascular disease. Curr. Opin. Lipidol. 2009;20(1):57–62. doi: 10.1097/MOL.0b013e328321b587. [DOI] [PubMed] [Google Scholar]
- 40.Yagmur E., Koch A., Haumann M., Kramann R., Trautwein C., Tacke F. Hyaluronan serum concentrations are elevated in critically ill patients and associated with disease severity. Clin. Biochem. 2012;45(1-2):82–87. doi: 10.1016/j.clinbiochem.2011.10.016. [DOI] [PubMed] [Google Scholar]
- 41.Schmidt E.P., Overdier K.H., Sun X., Lin L., Liu X., Yang Y., Ammons L.A., Hiller T.D., Suflita M.A., Yu Y., Chen Y., Zhang F., Cothren Burlew C., Edelstein C.L., Douglas I.S., Linhardt R.J. Urinary Glycosaminoglycans Predict Outcomes in Septic Shock and Acute Respiratory Distress Syndrome. Am. J. Respir. Crit. Care Med. 2016;194(4):439–449. doi: 10.1164/rccm.201511-2281OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Shimizu M.T., Almeida N.Q., Fantinato V., Unterkircher C.S. Studies on hyaluronidase, chondroitin sulphatase, proteinase and phospholipase secreted by Candida species. Mycoses. 1996;39(5-6):161–167. doi: 10.1111/j.1439-0507.1996.tb00120.x. [DOI] [PubMed] [Google Scholar]
- 43.Rivera Starr C., Engleberg N.C. Role of hyaluronidase in subcutaneous spread and growth of group A streptococcus. Infect. Immun. 2006;74(1):40–48. doi: 10.1128/IAI.74.1.40-48.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hynes W.L., Walton S.L. Hyaluronidases of Gram-positive bacteria. FEMS Microbiol. Lett. 2000;183(2):201–207. doi: 10.1111/j.1574-6968.2000.tb08958.x. [DOI] [PubMed] [Google Scholar]
- 45.Merline R., Moreth K., Beckmann J., Nastase M.V., Zeng-Brouwers J., Tralhão J.G., Lemarchand P., Pfeilschifter J., Schaefer R.M., Iozzo R.V., Schaefer L. Signaling by the matrix proteoglycan decorin controls inflammation and cancer through PDCD4 and MicroRNA-21. Sci. Signal. 2011;4(199) doi: 10.1126/scisignal.2001868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Honda T., Kaneiwa T., Mizumoto S., Sugahara K., Yamada S. Hyaluronidases Have Strong Hydrolytic Activity toward Chondroitin 4-Sulfate Comparable to that for Hyaluronan. Biomolecules. 2012;2(4):549–563. doi: 10.3390/biom2040549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Moseley R., Waddington R.J., Embery G. Degradation of glycosaminoglycans by reactive oxygen species derived from stimulated polymorphonuclear leukocytes. BBA. 1997;1362(2-3):221–231. doi: 10.1016/s0925-4439(97)00083-5. [DOI] [PubMed] [Google Scholar]
- 48.Bode W., Gomis-Rüth F.X., Stöckler W. Astacins, serralysins, snake venom and matrix metalloproteinases exhibit identical zinc-binding environments (HEXXHXXGXXH and Met-turn) and topologies and should be grouped into a common family, the 'metzincins'. FEBS Lett. 1993;331(1–2):134–140. doi: 10.1016/0014-5793(93)80312-i. [DOI] [PubMed] [Google Scholar]
- 49.Pugin J., Widmer M.-C., Kossodo S., Liang C.-M., Preas H.L., Suffredini A.F. Human neutrophils secrete gelatinase B in vitro and in vivo in response to endotoxin and proinflammatory mediators. Am. J. Respir. Cell Mol. Biol. 1999;20(3):458–464. doi: 10.1165/ajrcmb.20.3.3311. [DOI] [PubMed] [Google Scholar]
- 50.Nakamura T., et al. Modulation of plasma metalloproteinase-9 concentrations and peripheral blood monocyte mRNA levels in patients with septic shock: effect of fiber-immobilized polymyxin B treatment. Am. J. Med. Sci. 1998;316(6):355–360. doi: 10.1097/00000441-199812000-00001. [DOI] [PubMed] [Google Scholar]
- 51.Shapira L., Soskolne W.A., Houri Y., Barak V., Halabi A., Stabholz A. Protection against endotoxic shock and lipopolysaccharide-induced local inflammation by tetracycline: correlation with inhibition of cytokine secretion. Infect. Immun. 1996;64(3):825–828. doi: 10.1128/iai.64.3.825-828.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Maitra S.R., Bhaduri S., Valane P.D., Tervahartiala T., Sorsa T., Ramamurthy N. Inhibition of matrix metalloproteinases by chemically modified tetracyclines in sepsis. Shock. 2003;20(3):280–285. doi: 10.1097/00024382-200309000-00014. [DOI] [PubMed] [Google Scholar]
- 53.Albert J., Radomski A., Soop A., Sollevi A., Frostell C., Radomski M.W. Differential release of matrix metalloproteinase-9 and nitric oxide following infusion of endotoxin to human volunteers. Acta Anaesthesiol. Scand. 2003;47(4):407–410. doi: 10.1034/j.1399-6576.2003.00059.x. [DOI] [PubMed] [Google Scholar]
- 54.Li Q., Park P.W., Wilson C.L., Parks W.C. Matrilysin shedding of syndecan-1 regulates chemokine mobilization and transepithelial efflux of neutrophils in acute lung injury. Cell. 2002;111(5):635–646. doi: 10.1016/s0092-8674(02)01079-6. [DOI] [PubMed] [Google Scholar]
- 55.Nam E.J., Park P.W. Shedding of cell membrane-bound proteoglycans. Methods Mol. Biol. 2012;836:291–305. doi: 10.1007/978-1-61779-498-8_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Vandenbroucke R.E., Libert C. Is there new hope for therapeutic matrix metalloproteinase inhibition? Nat. Rev. Drug Discov. 2014;13(12):904–927. doi: 10.1038/nrd4390. [DOI] [PubMed] [Google Scholar]
- 57.Kermarrec N., et al. Regulation of peritoneal and systemic neutrophil-derived tumor necrosis factor-alpha release in patients with severe peritonitis: role of tumor necrosis factor-alpha converting enzyme cleavage. Crit. Care Med. 2005;33(6):1359–1364. doi: 10.1097/01.ccm.0000166359.47577.57. [DOI] [PubMed] [Google Scholar]
- 58.Yang, X., et al., A disintegrin and metalloproteinase 15-mediated glycocalyx shedding contributes to vascular leakage during inflammation. Cardiovasc Res, 2018. 114(13): p. 1752-1763. [DOI] [PMC free article] [PubMed]
- 59.Dreymueller D., Martin C., Kogel T., Pruessmeyer J., Hess F.M., Horiuchi K., Uhlig S., Ludwig A. Lung endothelial ADAM17 regulates the acute inflammatory response to lipopolysaccharide. EMBO Mol. Med. 2012;4(5):412–423. doi: 10.1002/emmm.201200217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Pruessmeyer J., Martin C., Hess F.M., Schwarz N., Schmidt S., Kogel T., Hoettecke N., Schmidt B., Sechi A., Uhlig S., Ludwig A. A disintegrin and metalloproteinase 17 (ADAM17) mediates inflammation-induced shedding of syndecan-1 and -4 by lung epithelial cells. J. Biol. Chem. 2010;285(1):555–564. doi: 10.1074/jbc.M109.059394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kuhn M. Endothelial actions of atrial and B-type natriuretic peptides. Br. J. Pharmacol. 2012;166(2):522–531. doi: 10.1111/j.1476-5381.2012.01827.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Damén T., Kolsrud O., Dellgren G., Hesse C., Ricksten S.-E., Nygren A. Atrial natriuretic peptide does not degrade the endothelial glycocalyx: A secondary analysis of a randomized porcine model. Acta Anaesthesiol. Scand. 2021;65(9):1305–1312. doi: 10.1111/aas.13853. [DOI] [PubMed] [Google Scholar]
- 63.Higgins S.J., De Ceunynck K., Kellum J.A., Chen X., Gu X., Chaudhry S.A., Schulman S., Libermann T.A., Lu S., Shapiro N.I., Christiani D.C., Flaumenhaft R., Parikh S.M. Tie2 protects the vasculature against thrombus formation in systemic inflammation. J. Clin. Invest. 2018;128(4):1471–1484. doi: 10.1172/JCI97488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Thamm K., Schrimpf C., Retzlaff J., Idowu T.O., van Meurs M., Zijlstra J.G., Ghosh C.C., Zeitvogel J., Werfel T.A., Haller H., Parikh S.M., David S. Molecular Regulation of Acute Tie2 Suppression in Sepsis. Crit. Care Med. 2018;46(9):e928–e936. doi: 10.1097/CCM.0000000000003269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Parikh S.M., Mammoto T., Schultz A., Yuan H.-T., Christiani D., Karumanchi S.A., Sukhatme V.P., Barnes P.J. Excess circulating angiopoietin-2 may contribute to pulmonary vascular leak in sepsis in humans. PLoS Med. 2006;3(3):e46. doi: 10.1371/journal.pmed.0030046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Inkinen N., Pettilä V., Lakkisto P., Kuitunen A., Jukarainen S., Bendel S., Inkinen O., Ala-Kokko T., Vaara S.T. Association of endothelial and glycocalyx injury biomarkers with fluid administration, development of acute kidney injury, and 90-day mortality: data from the FINNAKI observational study. Ann Intensive Care. 2019;9(1) doi: 10.1186/s13613-019-0575-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Lukasz, A., et al., Endothelial glycocalyx breakdown is mediated by angiopoietin-2. Cardiovasc Res, 2017. 113(6): p. 671-680. [DOI] [PubMed]
- 68.Han S., et al. Amelioration of sepsis by TIE2 activation-induced vascular protection. Sci. Transl. Med. 2016;8(335):p. 335ra55. doi: 10.1126/scitranslmed.aad9260. [DOI] [PubMed] [Google Scholar]
- 69.Becker B.F., Jacob M., Leipert S., Salmon A.H.J., Chappell D. Degradation of the endothelial glycocalyx in clinical settings: searching for the sheddases. Br. J. Clin. Pharmacol. 2015;80(3):389–402. doi: 10.1111/bcp.12629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Lipowsky H.H., Lescanic A. The effect of doxycycline on shedding of the glycocalyx due to reactive oxygen species. Microvasc. Res. 2013;90:80–85. doi: 10.1016/j.mvr.2013.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Manon-Jensen T., Multhaupt H.A.B., Couchman J.R. Mapping of matrix metalloproteinase cleavage sites on syndecan-1 and syndecan-4 ectodomains. FEBS J. 2013;280(10):2320–2331. doi: 10.1111/febs.12174. [DOI] [PubMed] [Google Scholar]
- 72.Rubio-Gayosso I., Platts S.H., Duling B.R. Reactive oxygen species mediate modification of glycocalyx during ischemia-reperfusion injury. Am J. Physiol. Heart Circ. Physiol. 2006;290(6):H2247–H2256. doi: 10.1152/ajpheart.00796.2005. [DOI] [PubMed] [Google Scholar]
- 73.Chen H.-R., Chao C.-H., Liu C.-C., Ho T.-S., Tsai H.-P., Perng G.-C., Lin Y.-S., Wang J.-R., Yeh T.-M., Fernandez-Sesma A. Macrophage migration inhibitory factor is critical for dengue NS1-induced endothelial glycocalyx degradation and hyperpermeability. PLoS Pathog. 2018;14(4):e1007033. doi: 10.1371/journal.ppat.1007033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Fitzgerald M.L., Wang Z., Park P.W., Murphy G., Bernfield M. Shedding of syndecan-1 and -4 ectodomains is regulated by multiple signaling pathways and mediated by a TIMP-3-sensitive metalloproteinase. J. Cell Biol. 2000;148(4):811–824. doi: 10.1083/jcb.148.4.811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Bashenko Y., Ilan N., Krausz M.M., Vlodavsky I., Hirsh M.I. Heparanase pretreatment attenuates endotoxin-induced acute lung injury in rats. Shock. 2007;28(2):207–212. doi: 10.1097/shk.0b013e3180311d84. [DOI] [PubMed] [Google Scholar]
- 76.Kwak Y.-K., Vikström E., Magnusson K.-E., Vécsey-Semjén B., Colque-Navarro P., Möllby R., Blanke S.R. The Staphylococcus aureus alpha-toxin perturbs the barrier function in Caco-2 epithelial cell monolayers by altering junctional integrity. Infect. Immun. 2012;80(5):1670–1680. doi: 10.1128/IAI.00001-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Chen S., et al. Unfractionated heparin attenuates intestinal injury in mouse model of sepsis by inhibiting heparanase. Int. J. Clin. Exp. Pathol. 2015;8(5):4903–4912. [PMC free article] [PubMed] [Google Scholar]
- 78.Kutuzov N., Flyvbjerg H., Lauritzen M. Contributions of the glycocalyx, endothelium, and extravascular compartment to the blood-brain barrier. Proc. Natl. Acad. Sci. U.S.A. 2018;115(40):E9429–E9438. doi: 10.1073/pnas.1802155115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Ando Y., Okada H., Takemura G., Suzuki K., Takada C., Tomita H., Zaikokuji R., Hotta Y., Miyazaki N., Yano H., Muraki I., Kuroda A., Fukuda H., Kawasaki Y., Okamoto H., Kawaguchi T., Watanabe T., Doi T., Yoshida T., Ushikoshi H., Yoshida S., Ogura S. Brain-Specific Ultrastructure of Capillary Endothelial Glycocalyx and Its Possible Contribution for Blood Brain Barrier. Sci. Rep. 2018;8(1) doi: 10.1038/s41598-018-35976-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Oshima, K., et al., More than a biomarker: the systemic consequences of heparan sulfate fragments released during endothelial surface layer degradation (2017 Grover Conference Series). Pulmonary circulation, 2017. 8(1): p. 2045893217745786. [DOI] [PMC free article] [PubMed]
- 81.Lortat-Jacob H., Baltzer F., Grimaud J.A. Heparin decreases the blood clearance of interferon-gamma and increases its activity by limiting the processing of its carboxyl-terminal sequence. J. Biol. Chem. 1996;271(27):16139–16143. doi: 10.1074/jbc.271.27.16139. [DOI] [PubMed] [Google Scholar]
- 82.Goodall K.J., Poon I.K.H., Phipps S., Hulett M.D., Srinivasula S.M. Soluble heparan sulfate fragments generated by heparanase trigger the release of pro-inflammatory cytokines through TLR-4. PLoS ONE. 2014;9(10):e109596. doi: 10.1371/journal.pone.0109596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Nelson A., Berkestedt I., Schmidtchen A., Ljunggren L., Bodelsson M. Increased levels of glycosaminoglycans during septic shock: relation to mortality and the antibacterial actions of plasma. Shock. 2008;30(6):623–627. doi: 10.1097/SHK.0b013e3181777da3. [DOI] [PubMed] [Google Scholar]
- 84.Muñoz E.M., Linhardt R.J. Heparin-binding domains in vascular biology. Arterioscler. Thromb. Vasc. Biol. 2004;24(9):1549–1557. doi: 10.1161/01.ATV.0000137189.22999.3f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Levi M., van der Poll T. Coagulation and sepsis. Thromb. Res. 2017;149:38–44. doi: 10.1016/j.thromres.2016.11.007. [DOI] [PubMed] [Google Scholar]
- 86.Gill S., Wight T.N., Frevert C.W. Proteoglycans: key regulators of pulmonary inflammation and the innate immune response to lung infection. Anat Rec (Hoboken) 2010;293(6):968–981. doi: 10.1002/ar.21094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Frey H., Schroeder N., Manon-Jensen T., Iozzo R.V., Schaefer L. Biological interplay between proteoglycans and their innate immune receptors in inflammation. FEBS J. 2013;280(10):2165–2179. doi: 10.1111/febs.12145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Calsavara A.J.C., Costa P.A., Nobre V., Teixeira A.L. Factors Associated With Short and Long Term Cognitive Changes in Patients With Sepsis. Sci. Rep. 2018;8(1) doi: 10.1038/s41598-018-22754-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Semmler A., Widmann C.N., Okulla T., Urbach H., Kaiser M., Widman G., Mormann F., Weide J., Fliessbach K., Hoeft A., Jessen F., Putensen C., Heneka M.T. Persistent cognitive impairment, hippocampal atrophy and EEG changes in sepsis survivors. J. Neurol. Neurosurg. Psychiatry. 2013;84(1):62–69. doi: 10.1136/jnnp-2012-302883. [DOI] [PubMed] [Google Scholar]
- 90.Zhang X., Han X., Xia K.e., Xu Y., Yang Y., Oshima K., Haeger S.M., Perez M.J., McMurtry S.A., Hippensteel J.A., Ford J.A., Herson P.S., Liu J., Schmidt E.P., Linhardt R.J. Circulating heparin oligosaccharides rapidly target the hippocampus in sepsis, potentially impacting cognitive functions. Proc Natl Acad Sci U S A. 2019;116(19):9208–9213. doi: 10.1073/pnas.1902227116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Leal G., Afonso P.M., Salazar I.L., Duarte C.B. Regulation of hippocampal synaptic plasticity by BDNF. Brain Res. 2015;1621:82–101. doi: 10.1016/j.brainres.2014.10.019. [DOI] [PubMed] [Google Scholar]
- 92.Hippensteel, J.A., et al., Circulating heparan sulfate fragments mediate septic cognitive dysfunction. J. Clin. Invest., 2019. [DOI] [PMC free article] [PubMed]
- 93.Silk, E., et al., The role of extracellular histone in organ injury. Cell Death Dis, 2017. 8(5): p. e2812. [DOI] [PMC free article] [PubMed]
- 94.Zhang Y., Haeger S.M., Yang Y., Dailey K.L., Ford J.A., Schmidt E.P. Circulating Heparan Sulfate Fragments Attenuate Histone-Induced Lung Injury Independently of Histone Binding. Shock. 2017;48(6):666–673. doi: 10.1097/SHK.0000000000000907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Marechal X., Favory R., Joulin O., Montaigne D., Hassoun S., Decoster B., Zerimech F., Neviere R. Endothelial glycocalyx damage during endotoxemia coincides with microcirculatory dysfunction and vascular oxidative stress. Shock. 2008;29(5):572–576. doi: 10.1097/SHK.0b013e318157e926. [DOI] [PubMed] [Google Scholar]
- 96.Yeo T.W., Bush P.A., Chen Y., Young S.P., Zhang H., Millington D.S., Granger D.L., Mwaikambo E.D., Anstey N.M., Weinberg J.B. Glycocalyx breakdown is increased in African children with cerebral and uncomplicated falciparum malaria. FASEB J. 2019;33(12):14185–14193. doi: 10.1096/fj.201901048RR. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Ozturk B., Kuscu F., Tutuncu E., Sencan I., Gurbuz Y., Tuzun H. Evaluation of the association of serum levels of hyaluronic acid, sICAM-1, sVCAM-1, and VEGF-A with mortality and prognosis in patients with Crimean-Congo hemorrhagic fever. J. Clin. Virol. 2010;47(2):115–119. doi: 10.1016/j.jcv.2009.10.015. [DOI] [PubMed] [Google Scholar]
- 98.Connolly-Andersen A.M., Thunberg T., Ahlm C. Endothelial activation and repair during hantavirus infection: association with disease outcome. Open Forum. Infect Dis. 2014;1(1):p. ofu027. doi: 10.1093/ofid/ofu027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Tang T.H., et al. Increased Serum Hyaluronic Acid and Heparan Sulfate in Dengue Fever: Association with Plasma Leakage and Disease Severity. Sci. Rep. 2017;7:46191. doi: 10.1038/srep46191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Benatti M.N., Fabro A.T., Miranda C.H. Endothelial glycocalyx shedding in the acute respiratory distress syndrome after flu syndrome. J Intensive Care. 2020;8:72. doi: 10.1186/s40560-020-00488-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Buijsers B., et al. Increased Plasma Heparanase Activity in COVID-19 Patients. Front. Immunol. 2020;11 doi: 10.3389/fimmu.2020.575047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Queisser K.A., Mellema R.A., Middleton E.A., Portier I., Manne B.K., Denorme F., Beswick E.J., Rondina M.T., Campbell R.A., Petrey A.C. COVID-19 generates hyaluronan fragments that directly induce endothelial barrier dysfunction. JCI. Insight. 2021;6(17) doi: 10.1172/jci.insight.14747210.1172/jci.insight.147472DS1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Murphy L.S., Wickersham N., McNeil J.B., Shaver C.M., May A.K., Bastarache J.A., Ware L.B. Endothelial glycocalyx degradation is more severe in patients with non-pulmonary sepsis compared to pulmonary sepsis and associates with risk of ARDS and other organ dysfunction. Ann Intensive Care. 2017;7(1) doi: 10.1186/s13613-017-0325-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Padberg J.-S., Wiesinger A., di Marco G.S., Reuter S., Grabner A., Kentrup D., Lukasz A., Oberleithner H., Pavenstädt H., Brand M., Kümpers P. Damage of the endothelial glycocalyx in chronic kidney disease. Atherosclerosis. 2014;234(2):335–343. doi: 10.1016/j.atherosclerosis.2014.03.016. [DOI] [PubMed] [Google Scholar]
- 105.Vlahu C.A., Lemkes B.A., Struijk D.G., Koopman M.G., Krediet R.T., Vink H. Damage of the endothelial glycocalyx in dialysis patients. J. Am. Soc. Nephrol. 2012;23(11):1900–1908. doi: 10.1681/ASN.2011121181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Rehm M., Bruegger D., Christ F., Conzen P., Thiel M., Jacob M., Chappell D., Stoeckelhuber M., Welsch U., Reichart B., Peter K., Becker B.F. Shedding of the endothelial glycocalyx in patients undergoing major vascular surgery with global and regional ischemia. Circulation. 2007;116(17):1896–1906. doi: 10.1161/CIRCULATIONAHA.106.684852. [DOI] [PubMed] [Google Scholar]
- 107.Ostrowski S.R., Johansson P.I. Endothelial glycocalyx degradation induces endogenous heparinization in patients with severe injury and early traumatic coagulopathy. J Trauma Acute Care Surg. 2012;73(1):60–66. doi: 10.1097/TA.0b013e31825b5c10. [DOI] [PubMed] [Google Scholar]
- 108.Tapking C., Hernekamp J.F., Horter J., Kneser U., Haug V., Vogelpohl J., Schulte M., Kremer T., Hundeshagen G. Influence of burn severity on endothelial glycocalyx shedding following thermal trauma: A prospective observational study. Burns. 2021;47(3):621–627. doi: 10.1016/j.burns.2020.07.021. [DOI] [PubMed] [Google Scholar]
- 109.Tamura T., Sano M., Matsuoka T., Yoshizawa J., Yamamoto R., Katsumata Y., Endo J., Homma K., Kajimura M., Suzuki M., Kobayashi E., Sasaki J. Hydrogen Gas Inhalation Attenuates Endothelial Glycocalyx Damage and Stabilizes Hemodynamics in a Rat Hemorrhagic Shock Model. Shock. 2020;54(3):377–385. doi: 10.1097/SHK.0000000000001459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Machin D.R., Bloom S.I., Campbell R.A., Phuong T.T.T., Gates P.E., Lesniewski L.A., Rondina M.T., Donato A.J. Advanced age results in a diminished endothelial glycocalyx. Am J Physiol Heart Circ Physiol. 2018;315(3):H531–H539. doi: 10.1152/ajpheart.00104.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Vlodavsky I., Blich M., Li J.-P., Sanderson R.D., Ilan N. Involvement of heparanase in atherosclerosis and other vessel wall pathologies. Matrix Biol. 2013;32(5):241–251. doi: 10.1016/j.matbio.2013.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Schiefer J., Lebherz-Eichinger D., Erdoes G., Berlakovich G., Bacher A., Krenn C.G., Faybik P. Alterations of Endothelial Glycocalyx During Orthotopic Liver Transplantation in Patients With End-Stage Liver Disease. Transplantation. 2015;99(10):2118–2123. doi: 10.1097/TP.0000000000000680. [DOI] [PubMed] [Google Scholar]
- 113.Dogné S., Flamion B., Caron N. Endothelial Glycocalyx as a Shield Against Diabetic Vascular Complications: Involvement of Hyaluronan and Hyaluronidases. Arterioscler. Thromb. Vasc. Biol. 2018;38(7):1427–1439. doi: 10.1161/ATVBAHA.118.310839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Saoraya J., Wongsamita L., Srisawat N., Musikatavorn K. Plasma syndecan-1 is associated with fluid requirements and clinical outcomes in emergency department patients with sepsis. Am. J. Emerg. Med. 2021;42:83–89. doi: 10.1016/j.ajem.2021.01.019. [DOI] [PubMed] [Google Scholar]
- 115.Andrews B., Semler M.W., Muchemwa L., Kelly P., Lakhi S., Heimburger D.C., Mabula C., Bwalya M., Bernard G.R. Effect of an Early Resuscitation Protocol on In-hospital Mortality Among Adults With Sepsis and Hypotension: A Randomized Clinical Trial. JAMA. 2017;318(13):1233. doi: 10.1001/jama.2017.10913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Chappell D., Bruegger D., Potzel J., Jacob M., Brettner F., Vogeser M., Conzen P., Becker B.F., Rehm M. Hypervolemia increases release of atrial natriuretic peptide and shedding of the endothelial glycocalyx. Crit. Care. 2014;18(5) doi: 10.1186/s13054-014-0538-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Byrne L., Obonyo N.G., Diab S.D., Dunster K.R., Passmore M.R., Boon A.-C., Hoe L.S., Pedersen S., Fauzi M.H., Pimenta L.P., Van Haren F., Anstey C.M., Cullen L., Tung J.-P., Shekar K., Maitland K., Fraser J.F. Unintended Consequences; Fluid Resuscitation Worsens Shock in an Ovine Model of Endotoxemia. Am. J. Respir. Crit. Care Med. 2018;198(8):1043–1054. doi: 10.1164/rccm.201801-0064OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Saoraya J., Wongsamita L., Srisawat N., Musikatavorn K. The effects of a limited infusion rate of fluid in the early resuscitation of sepsis on glycocalyx shedding measured by plasma syndecan-1: a randomized controlled trial. J Intensive Care. 2021;9(1) doi: 10.1186/s40560-020-00515-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Evans L., et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock 2021. Crit. Care Med. 2021;49(11):e1063–e1143. doi: 10.1097/CCM.0000000000005337. [DOI] [PubMed] [Google Scholar]
- 120.Underwood J.L., Murphy C.G., Chen J., Franse-Carman L., Wood I., Epstein D.L., Alvarado J.A. Glucocorticoids regulate transendothelial fluid flow resistance and formation of intercellular junctions. Am. J. Physiol. 1999;277(2):C330–C342. doi: 10.1152/ajpcell.1999.277.2.C330. [DOI] [PubMed] [Google Scholar]
- 121.Chappell D., Jacob M., Hofmann-Kiefer K., Bruegger D., Rehm M., Conzen P., Welsch U., Becker B. Hydrocortisone preserves the vascular barrier by protecting the endothelial glycocalyx. Anesthesiology. 2007;107(5):776–784. doi: 10.1097/01.anes.0000286984.39328.96. [DOI] [PubMed] [Google Scholar]
- 122.Chappell D., et al. TNF-alpha induced shedding of the endothelial glycocalyx is prevented by hydrocortisone and antithrombin. Basic Res. Cardiol. 2009;104(1):78–89. doi: 10.1007/s00395-008-0749-5. [DOI] [PubMed] [Google Scholar]
- 123.Brettner F., Chappell D., Nebelsiek T., Hauer D., Schelling G., Becker B.F., Rehm M., Weis F. Preinterventional hydrocortisone sustains the endothelial glycocalyx in cardiac surgery. Clin. Hemorheol. Microcirc. 2019;71(1):59–70. doi: 10.3233/CH-180384. [DOI] [PubMed] [Google Scholar]
- 124.Antoniak S. The coagulation system in host defense. Res Pract Thromb Haemost. 2018;2(3):549–557. doi: 10.1002/rth2.12109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Simmons J., Pittet J.F. The coagulopathy of acute sepsis. Curr Opin Anaesthesiol. 2015;28(2):227–236. doi: 10.1097/ACO.0000000000000163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Iba T., Levy J.H., Aihara K., Kadota K., Tanaka H., Sato K., Nagaoka I. Newly Developed Recombinant Antithrombin Protects the Endothelial Glycocalyx in an Endotoxin-Induced Rat Model of Sepsis. Int. J. Mol. Sci. 2020;22(1):176. doi: 10.3390/ijms22010176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Yini S., Heng Z., Xin A., Xiaochun M. Effect of unfractionated heparin on endothelial glycocalyx in a septic shock model. Acta Anaesthesiol. Scand. 2015;59(2):160–169. doi: 10.1111/aas.12418. [DOI] [PubMed] [Google Scholar]
- 128.Knaup H., Stahl K., Schmidt B.M.W., Idowu T.O., Busch M., Wiesner O., Welte T., Haller H., Kielstein J.T., Hoeper M.M., David S. Early therapeutic plasma exchange in septic shock: a prospective open-label nonrandomized pilot study focusing on safety, hemodynamics, vascular barrier function, and biologic markers. Crit. Care. 2018;22(1) doi: 10.1186/s13054-018-2220-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.David S., Bode C., Putensen C., Welte T., Stahl K., Busch M., Haller H., Hoeper M.M., Lehmann F., Schmidt J.J., Schmidt B.M.W., Seeliger B., Sauer A., Wand P., Wiesner O., Wedemeyer H. Adjuvant therapeutic plasma exchange in septic shock. Intensive Care Med. 2021;47(3):352–354. doi: 10.1007/s00134-020-06339-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Straat M., Müller M.CA., Meijers J.CM., Arbous M.S., Spoelstra - de Man A.ME., Beurskens C.JP., Vroom M.B., Juffermans N.P. Effect of transfusion of fresh frozen plasma on parameters of endothelial condition and inflammatory status in non-bleeding critically ill patients: a prospective substudy of a randomized trial. Crit. Care. 2015;19(1) doi: 10.1186/s13054-015-0828-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Torres L.N., Sondeen J.L., Ji L., Dubick M.A., Filho I.T. Evaluation of resuscitation fluids on endothelial glycocalyx, venular blood flow, and coagulation function after hemorrhagic shock in rats. J Trauma Acute Care Surg. 2013;75(5):759–766. doi: 10.1097/TA.0b013e3182a92514. [DOI] [PubMed] [Google Scholar]