Highlights
-
•
Outstanding precision was achieved using an Orbitrap for TDM.
-
•
TurboFlow technology enables excellent sample clean-up.
-
•
HRMS is applicable for reliable quantification in TDM.
Abbreviations: CV, coefficient of variation; EMA, European Medicines Agency; ESI, electrospray ionization; HRMS, high-resolution mass spectrometry; ISTD, internal standard; LC-MS/MS, liquid chromatography tandem mass spectrometry; LLOD, lower limit of detection; LLOQ, lower limit of quantification; MS, mass spectrometry; QC, quality control sample; TDM, therapeutic drug monitoring; UHPLC, ultra high-performance liquid chromatography
Keywords: High-resolution mass spectrometry, TurboFlow technology, Therapeutic drug monitoring (TDM), Antidepressants
Abstract
Background
Therapeutic drug monitoring (TDM) of antidepressants is important to ensure compliance and to rule out pharmacokinetic abnormalities. Therefore, reliable methods for quantification are important for clinical laboratories. Most of the currently used mass spectrometry methods use triple quadrupoles as mass analyzers. We aimed to develop a method using high-resolution mass spectrometry (HRMS) and wanted to test the suitability of this analyzer for quantitative TDM assays. This would be beneficial since HRMS instruments can also be used for metabolomics and protein analysis and, thus, many different analyses could be run on one instrument.
Methods
After manual protein precipitation of serum samples, further sample clean-up was achieved using a Turbo Flow column preconnected to the analytical LC column. Stable isotope-labelled counterparts of the target analytes were used as internal standards. For detection, we used a Q Exactive Focus Orbitrap mass spectrometer operating in full-scan mode. Ionization was performed in positive ESI.
Results
Accuracy, recovery, and matrix effect were acceptable for all analytes. The method demonstrated outstanding precision (within-run imprecision <4.5%, between-run imprecision <7.5%). The selectivity of the method was ensured by chromatographic separation of all isobaric compounds. Close agreement between Orbitrap and triple stage based quantification was observed in a comparison measurement of leftover patient samples.
Conclusions
We have established a selective method for the quantification of antidepressants with outstanding precision using a high-resolution Orbitrap mass spectrometer. The applicability of HRMS instruments to TDM was demonstrated.
1. Introduction
Therapeutic drug monitoring (TDM), the measurement of drug concentrations in human blood samples with the aim of obtaining optimal, near-constant drug levels with the goal of achieving treatment efficacy while avoiding side effects and resistance development, is a valuable tool for personalizing therapeutic regimes and optimizing patient benefit [1], [2], [3]. TDM is equally applicable to drugs with either narrow therapeutic ranges or pharmacokinetic variabilities. Additional reasons to monitor drug levels include: toxicity avoidance, evaluation of the pharmacokinetic impact of co-medication, and compliance verification [1]. For the latter, it is important to quantify not only the parent drug, but also its metabolites. This approach provides insights into the timing of drug intake in relation to sampling, which is especially important for antidepressant compliance monitoring. Detailed information concerning TDM of drugs used in psychiatry and neurology are available from the updated AGNP Consensus Guidelines [2].
To avoid ambiguous or misleading results that could negatively influence therapeutic outcome, reliable, sensitive, and selective analytical quantification methods are essential [4]. Methods based on liquid chromatography coupled to mass spectrometry are becoming increasingly popular [3], [4] due to their high specificity, sensitivity, and the potential to quantify different drugs and metabolites simultaneously [5]. Since laboratories providing TDM analyses for psychiatric units are faced with a wide and growing spectrum of drug compounds, it is highly desirable to have multi-analyte panel methods available.
Most mass spectrometric methods for TDM use triple quadrupoles as the mass analyzer [6], [7]. The aim of our work was to establish a reliable method for the quantification of 31 psychoactive drugs, including antidepressants, using a Q Exactive Focus hybrid quadrupole Orbitrap mass spectrometer. This rather new mass spectrometric technology allows scan acquisition over wide mass ranges at high mass resolution and accurate mass [8]. High resolution mass spectrometry has been the gold standard in mass spectrometric analysis of proteins as of the recent few years [9]. In the lower molecular mass range (m/z 50–3000), which is addressed in our approach, the feature of scanning at highest mass resolution, generating huge amounts of data, makes this technology most attractive for metabolomic-type of clinical testing [10].
Orbitrap high resolution mass spectrometry could become an important tool for clinical laboratories [11] since it offers the potential to perform quantitative small molecule analyses, metabolomic-type investigations, as well as protein analyses on a single instrument.
In this work, we were interested in establishing the suitability of a high-resolution accurate mass spectrometer for quantitative TDM assays. Our aim was to show that HRMS is as equally suited for TDM as triple quadrupoles. If reliable quantification measurements are possible with HRMS instruments, it then follows that HRMS instruments are suitable for clinical laboratories, since TDM is one of the main application areas of mass spectrometers in clinical laboratories. Rather than demonstrating superior performance on TDM measurements, the expected advantage of HRMS in TDM is the opportunity to have an instrument in the laboratory which can perform different types of analyses, including TDM. There are only few publications that address the application of the Orbitrap technology with regard to TDM. Table 1 summarizes the reports on Q Exactive (Thermo Scientific) applications for TDM of small molecules using serum or plasma as sample material [12], [13], [14], [15], [16], [17], [18], [19], [20]. Only two of the described methods use a Q Exactive Focus and none of them quantify antidepressants.
Table 1.
Summary of methods applying Orbitrap technology for TDM.
| Publication | Analyte (group) | Device |
|---|---|---|
|
Schuster C et al.[12]: Isotope dilution LC-orbitrap-HRMS with automated sample preparation for the simultaneous quantification of 11 antimycotics in human serum. J Pharm Biomed Anal. 2019 Jan 25;166:398–405. |
Antimycotics | Q Exactive Focus (Thermo Scientific) |
|
Lefeuvre S et al.[13]: A simple ultra-high-performance liquid chromatography-high resolution mass spectrometry assay for the simultaneous quantification of 15 antibiotics in plasma. J Chromatogr B Analyt Technol Biomed Life Sci. 2017 Oct 15;1065–1066:50–58. |
Antibiotics | Q Exactive Focus (Thermo Scientific) |
|
Deltombe O et al.[14]: Development and validation of an ultra-high performance liquid chromatography – high resolution mass spectrometry method for the quantification of total and free teicoplanin in human plasma. Clin Biochem. 2018 Dec 25. |
Teicoplanin | Q Exactive (Thermo Scientific) |
|
Bhatnagar A et al.[15]: Quantitation of the anticancer drug abiraterone and its metabolite Δ(4)-abiraterone in human plasma using high-resolution mass spectrometry. J Pharm Biomed Anal. 2018 May 30;154:66–74 |
Abiraterone + metabolite | Q Exactive Plus Orbitrap (Thermo Scientific) |
|
Qu L et al.[16]: Utilizing online-dual-SPE-LC with HRMS for the simultaneous quantification of amphotericin B, fluconazole, and fluorocytosine in human plasma and cerebrospinal fluid. Talanta. 2017 Apr 1;165:449–457. |
Antimycotics | Q Exactive (Thermo Scientific) |
|
Qu L et al.[17]: Development, validation and clinical application of an online-SPE-LC-HRMS/MS for simultaneous quantification of phenobarbital, phenytoin, carbamazepine, and its active metabolite carbamazepine 10,11-epoxide. Talanta. 2016 Sep 1;158:77–88 |
Antiepileptics | Q Exactive (Thermo Scientific) |
|
Qua L et al.[18]: Quantitative performance of online SPE-LC coupled to Q-Exactive for the analysis of sofosbuvir in human plasma. RSC Adv., 2015, 5, 98269–98277. |
Sofosbuvir | Q Exactive (Thermo Scientific) |
|
Favretto D et al.[19]: Monitoring haloperidol exposure in body fluids and hair of children by liquid chromatography-high-resolution mass spectrometry. Ther Drug Monit. 2013 Aug;35(4):493–501. |
Haloperidol | LTQ-Orbitrap (Thermo Scientific) |
|
Dahmane E et al.[20]: Quantitative monitoring of tamoxifen in human plasma extended to 40 metabolites using liquid-chromatography high-resolution mass spectrometry: new investigation capabilities for clinical pharmacology. Anal Bioanal Chem. 2014 Apr;406(11):2627–40. |
Tamoxifen + metabolites | Exactive Plus Orbitrap (Thermo Scientific) |
Additionally, we worked with the TurboFlow online sample preparation technology to minimize manual sample processing steps thereby shortening the total time to result.
2. Materials and methods
2.1. Chemicals and reagents
Water, methanol, acetonitrile and formic acid were obtained in UHPLC grade from Biosolve (Valkenswaard, the Netherlands). Amitriptyline-HCl, atomoxetine-HCl, citalopram-HBr, clomipramine-HCl, clozapine, desipramine-HCl, desmethylfluoxetine-oxalate, doxepin-HCl, duloxetine-HCl, fluoxetine-HCl, fluvoxamine-maleate, imipramine-HCl, maprotiline-HCl, mianserin-HCl, mirtazapine, N-desmethylcitalopram-HCl, N-desmethylmirtazapine, norclomipramine-HCl, norclozapine, nordoxepin-HCl, normaprotiline, nortrimipramine-maleate, nortriptyline-HCl, O-desmethylvenlafaxine, paroxetine-HCl hemihydrate, protriptyline-HCl, reboxetine-HCl, sertraline-HCl, trazodone-HCl, trimipramine-maleate, venlafaxine-HCl and ammonium formate were from Sigma-Aldrich (Steinheim, Germany). Two different internal standard mixes were obtained from RECIPE (Munich, Germany): ClinMass Internal Standard for Antidepressants lyophilized (d3-atomoxetine, d6-citalopram, d5-desmethylfluoxetine, d7-duloxetine, d5-fluoxetine, d3-fluvoxamine, d3-mianserin, d3-mirtazapine, d4-paroxetine, d5-reboxetine, d3-sertraline, and d6-venlafaxine) and ClinMass Internal Standard for Tricyclic Antidepressants lyophilized (d3-amitriptyline, d3-clomipramine, d4-clozapine, d3-desipramine, d3-doxepin, d3-imipramine, d5-maprotiline, d3-norclomipramine, d8-norclozapine, d3-nordoxepin, d3-nortriptyline, and d3-trimipramine).
2.2. Calibrators and quality control samples (QC)
Calibrators at seven concentration levels (five for norclozapine) and internal QCs at three levels (low, medium, high) were established in water/methanol 50/50 (v/v). In addition to these, commercial serum-based QCs at two different levels each were purchased from RECIPE (Munich, Germany) and Bio-Rad (Munich, Germany). Individual concentrations for each analyte of the calibrators and QCs are shown in Table 2, Table 3, respectively.
Table 2.
Concentrations [ng/mL] of the calibrators.
| Compound | Cal 1 | Cal 2 | Cal 3 | Cal 4 | Cal 5 | Cal 6 | Cal 7 |
|---|---|---|---|---|---|---|---|
| Amitryptiline | 7.07 | 21.2 | 70.7 | 98.2 | 133 | 177 | 354 |
| Atomoxetine | 20.0 | 60.0 | 200 | 333 | 667 | 1000 | 2000 |
| Citalopram | 4.01 | 12.0 | 40.1 | 50.1 | 70.6 | 88.2 | 177 |
| Clomipramine | 20.6 | 61.8 | 206 | 258 | 323 | 403 | 806 |
| Clozapine | 35.0 | 105 | 350 | 438 | 480 | 600 | 1200 |
| Descitalopram | 4.48 | 13.4 | 44.8 | 55.9 | 78.8 | 98.5 | 197 |
| Desfluoxetine | 12.0 | 36.0 | 120 | 167 | 333 | 500 | 1000 |
| Desipramine | 10.0 | 30.0 | 100 | 167 | 225 | 300 | 600 |
| Desmirtazapine | 3.00 | 9.00 | 30.0 | 44.4 | 60.0 | 80.0 | 160 |
| Doxepine | 4.43 | 13.3 | 44.3 | 73.7 | 99.6 | 133 | 266 |
| Duloxetine | 3.00 | 9.00 | 30.0 | 66.7 | 90.0 | 120 | 240 |
| Fluoxetine | 10.7 | 32.2 | 107 | 149 | 298 | 448 | 895 |
| Fluvoxamine | 6.00 | 18.0 | 60.0 | 128 | 173 | 230 | 460 |
| Imipramine | 15.5 | 46.5 | 155 | 194 | 212 | 266 | 531 |
| Maprotiline | 6.63 | 19.9 | 66.3 | 82.1 | 91.9 | 115 | 230 |
| Mianserin | 1.50 | 4.50 | 15.0 | 23.3 | 46.7 | 70.0 | 140 |
| Mirtazapine | 3.00 | 9.00 | 30.0 | 44.4 | 60.0 | 80.0 | 160 |
| Norclomipramine | 23.0 | 69.0 | 230 | 288 | 360 | 450 | 900 |
| Norclozapine | 100 | 200 | 400 | 600 | 1200 | ||
| Nordoxepine | 5.00 | 15.0 | 50.0 | 83.3 | 113 | 150 | 300 |
| Normaprotiline | 10.0 | 30.0 | 100 | 125 | 160 | 200 | 400 |
| Nortryptiline | 7.02 | 21.1 | 70.2 | 97.6 | 132 | 176 | 351 |
| Nortrimipramine | 2.84 | 8.52 | 28.4 | 63.2 | 85.2 | 114 | 227 |
| Norvenlafaxine | 10.0 | 30.0 | 100 | 222 | 300 | 400 | 800 |
| Paroxetine | 2.64 | 7.91 | 26.4 | 33.0 | 42.2 | 52.7 | 105 |
| Protryptiline | 7.00 | 21.0 | 70.0 | 94.4 | 128 | 170 | 340 |
| Reboxetine | 5.38 | 16.1 | 53.8 | 105 | 209 | 314 | 627 |
| Sertraline | 0.894 | 2.68 | 8.94 | 44.7 | 89.4 | 134 | 268 |
| Trazodone | 70.0 | 210 | 700 | 556 | 750 | 1000 | 2000 |
| Trimipramine | 10.8 | 32.4 | 108 | 135 | 173 | 216 | 431 |
| Venlafaxine | 8.84 | 26.5 | 88.4 | 196 | 265 | 354 | 707 |
Table 3.
Concentrations [ng/mL] of quality control samples.
| Compound | QC 1 | QC 2 | QC 3 | QC 1 Recipe | QC 2 Recipe | QC 1 Biorad | QC 2 Biorad |
|---|---|---|---|---|---|---|---|
| Amitryptiline | 21.2 | 88.4 | 177 | 85.6 | 298 | ||
| Atomoxetine | 60.0 | 500 | 1000 | 404 | 969 | ||
| Citalopram | 12.0 | 44.1 | 88.2 | 43.5 | 102 | ||
| Clomipramine | 61.8 | 202 | 403 | ||||
| Clozapine | 150 | 300 | 600 | ||||
| Descitalopram | 13.4 | 49.2 | 98.5 | ||||
| Desfluoxetine | 36.0 | 250 | 500 | 107 | 256 | ||
| Desipramine | 30.0 | 150 | 300 | 89.6 | 302 | ||
| Desmirtazapine | 9.00 | 40.0 | 80.0 | 32.7 | 78.3 | ||
| Doxepine | 13.3 | 66.4 | 133 | ||||
| Duloxetine | 9.00 | 60.0 | 120 | 47.3 | 111 | ||
| Fluoxetine | 32.2 | 224 | 448 | 102 | 247 | ||
| Fluvoxamine | 18.0 | 115 | 230 | 98.2 | 229 | ||
| Imipramine | 46.5 | 133 | 266 | 86.2 | 300 | ||
| Maprotiline | 19.9 | 57.5 | 115 | ||||
| Mianserin | 4.50 | 35.0 | 70.0 | 29.5 | 70.9 | ||
| Mirtazapine | 9.00 | 40.0 | 80.0 | 33.1 | 79.1 | ||
| Norclomipramine | 69.0 | 225 | 450 | ||||
| Norclozapine | 150 | 300 | 600 | ||||
| Nordoxepine | 15.0 | 75.0 | 150 | 94.5 | 313 | ||
| Normaprotiline | 30.0 | 100 | 200 | ||||
| Nortryptiline | 21.1 | 87.8 | 176 | 91.7 | 311 | ||
| Nortrimipramine | 8.52 | 56.8 | 114 | ||||
| Norvenlafaxine | 30.0 | 200 | 400 | 102 | 238 | ||
| Paroxetine | 7.91 | 26.4 | 52.7 | 46.8 | 111 | ||
| Protryptiline | 21.0 | 85.0 | 170 | ||||
| Reboxetine | 16.1 | 157 | 314 | 143 | 338 | ||
| Sertraline | 2.68 | 67.1 | 134 | 26 | 62 | ||
| Trazodone | 210 | 500 | 1000 | 534 | 1284 | ||
| Trimipramine | 32.4 | 108 | 216 | ||||
| Venlafaxine | 26.5 | 177 | 354 | 61.7 | 146 |
2.3. Sample preparation
150 µL of precipitation agent (acetonitrile containing the internal standards, stored at −20 °C) were added to 50 µL of serum, calibrator or quality control sample. After vortex-mixing and 10 min of incubation at room temperature, a centrifugation step followed (10 min, 4 °C, 14000 rpm). The supernatants were transferred into HPLC vials. Further sample clean-up was performed using TurboFlow technology.
2.4. UHPLC, TurboFlow and MS parameters
80 µL of the supernatant were injected onto a Transcend II TLX-1 LC system from Thermo Scientific (Franklin, MA, USA). Online sample clean-up and enrichment was performed using a 0.5 × 50 mm Thermo Scientific TurboFlow Cyclone column. Chromatographic separation was achieved on a 100 × 2.1 mm, 1.9 µm Thermo Scientific Hypersil GOLD Phenyl column kept at 25 °C. The same mobile phases were used for TurboFlow extraction and chromatographic separation. Mobile phase A consisted of 0.1% formic acid in 10 mM ammonium formate in water and mobile phase B consisted of 0.1% formic acid in 10 mM ammonium formate in methanol/water (98/2, v/v). Detailed information about the gradient and flow rate of the TurboFlow and analytical column is given in Table 4. The total run time amounted to 9.5 min.
Table 4.
LC parameters.
| Start | Seconds | TurboFlow column |
Analytical column |
||||||
|---|---|---|---|---|---|---|---|---|---|
| Flow [mL/min] | Gradient | A [%] | B [%] | Flow [mL/min] | Gradient | A [%] | B [%] | ||
| 00:00 | 30 | 2 | step | 100 | 0 | 0.5 | step | 90 | 10 |
| 00:30 | 60 | 0.1 | step | 100 | 0 | 0.4 | step | 90 | 10 |
| 01:30 | 60 | 0.4 | step | 0 | 100 | 0.5 | ramp | 50 | 50 |
| 02:30 | 330 | 0.4 | step | 0 | 100 | 0.5 | step | 50 | 50 |
| 08:00 | 70 | 2 | step | 10 | 90 | 0.5 | step | 0 | 100 |
| 09:10 | 20 | 2 | step | 100 | 0 | 0.5 | step | 90 | 10 |
The analytes were detected with a Q Exactive Focus Orbitrap mass spectrometer (Thermo Scientific) using a resolution of 70,000 (FWHM) at m/z 200. Heated electrospray ionization in positive mode was performed. The vaporizer and the capillary temperature was 320 °C, spray voltage was set to 2500 V, sheath gas was 45 AU, sweep gas 1 AU, and auxiliary gas 10 AU. Data were acquired in full MS mode. The scan range was set to m/z 100–500.
2.5. Data analysis
Data were acquired and processed with Thermo Scientific TraceFinder 3.3 software. An extraction window of 5 ppm was used to extract the individual chromatograms.
2.6. Method validation
Method validation was performed according to a validation scheme which was oriented towards the Guideline of bioanalytical method validation by the European Medicines Agency (EMA) [21]. The method performance was evaluated in terms of precision, accuracy, recovery, linearity, selectivity, matrix effect for each analyte on five different validation runs, and lower limit of quantification (LLOQ). The LLOQ was defined as an appropriate concentration regarding the therapeutic ranges of the measured analytes combined with a signal-to-noise ratio of at least 5:1 and an inaccuracy and imprecision ≤20%. In each run, a blank sample, a zero sample (containing the internal standards, but no analytes), the calibrators, and QCs were measured. The stability of the extracted samples was also evaluated. The selectivity was determined by measuring 6 individual blank serum lots. These matrices were evaluated for possible endogenous interferences (peaks at the retention times of the analytes). Analytical within-run precision was evaluated for each validation run in terms of coefficient of variation (% CV) for each analyte on each control sample and the LLOQ, prepared and analyzed in replicates of five. Between-run precision was evaluated on the same controls including all 25 replicates of the five runs. Within-run accuracy was evaluated for each run as the percentage ratio between average experimental and nominal concentrations at each level using the same set of controls (five replicates per run). Between-run accuracy was evaluated on the same controls, including all 25 replicates of the five runs. In addition to the EMA guideline, the recovery was calculated for each analyte in terms of percentage ratio between the concentration when spiked in serum and extracted, and the concentration when spiked in extracted serum. Matrix effect was evaluated according to the procedure given by Matuszewski [22] for each analyte on six different serum lots as the percentage ratio between the concentration when spiked in extracted serum and the concentration when spiked in solvent and processed following the extraction procedure (matrix effect: 100% × cmatrix/csolvent). Additionally, the matrix effect was evaluated for all analytes by determination of the variation coefficient [%] of the internal standard normalized matrix factor of the six matrix lots (matrix factor = areamatrix/areasolvent) [21]. Each analyte was spiked and analyzed in replicates of five using a mid-range concentration to evaluate recovery and matrix effect. Extracted sample stability was evaluated for each analyte by comparison between freshly-prepared control samples and the same samples kept for 48 h in the autosampler tray at 10⁰C. The lower limit of detection (LLOD) was not determined since it does not contribute any added value that would be relevant to the practical use of the method in a clinical setting.
2.7. Method comparison
The results of the developed method were compared with the values of a validated triple quadrupole-based quantification method used in a routine clinical laboratory. The routine method is based on a protein precipitation with a methanol zinc sulfate solution (50 µL serum + 25 µL ISTD + 125 µL precipitation agent). After incubation, the samples were centrifuged and the supernatants were transferred into HPLC vials. As mobile phases, 10 mM ammonium formate, 0.1% formic acid in water and acetonitrile were used. The HPLC column used was a 100 × 2.1 mm PerfectSil Target ODS-3 HD 5 µm (MZ Analysetechnik GmbH, Mainz, Germany). The flow rate was 0.75 mL/min, a gradient was used, the column temperature was set to 50 °C, and the run time was ∼7 min. Detection was performed with a Xevo TQD (Waters, Milford, Massachusetts, USA) in ESI positive mode.
The patients’ samples were prepared according to our workflow and measured. Our results were then compared to the results of the triple quadrupole measurement. This was performed by a Passing-Bablok regression using Excel and XLSTAT.
2.8. Origin of the patients’ samples
All samples were leftover serum samples from the routine laboratory of our hospital, which were stored at 8 °C until measurement. The samples were from inpatients and outpatients of the psychiatric clinic. For the method comparison, the samples were completely anonymized. Therefore, the method comparison was exempt from a study approval by an appropriate ethical committee.
3. Results and discussion
The main issue in method development was the chromatographic separation of the isobaric compounds. Since the method was designed to be run in Full MS mode, analytes with the same molecular mass (i.e., imipramine/nortrimipramine, maprotiline/amitryptiline, protryptiline/normaprotiline/nortyptiline) had to be chromatographically separated. Therefore, different columns, mobile phases, and gradients were tested to achieve baseline separation of these compounds. A further important step in method development was the establishment of the TurboFlow method. Besides the choice of the right TurboFlow column, there are several steps (e.g., loading step, transfer step, washing step, loop filling step, and equilibrating step) that need to be optimized (e.g., appropriate mobile phase compositions and flow rates). Although the extraction of serum using the TurboFlow technique is supposed to achieve sufficient sample clean-up, substantially extended column lifetime can be expected when performing simple protein precipitation upstream. Since this procedure is straightforward and does not take long, we decided to perform a precipitation step to extended column lifetime and to reduce costs. Besides the described development steps, the high number of analytes with different chemical properties was challenging.
Baseline chromatographic separation was achieved for the 3 sets of isobaric compounds (i.e., imipramine/nortrimipramine, maprotiline/amitryptiline, protryptiline/normaprotiline/ nortyptiline) that could not otherwise be distinguished and properly quantified using a full MS approach. Representative chromatograms are reported in Fig. 1. By using a MS/MS approach, the chromatographic separation of these compounds would not have been necessary because the analytes could be distinguished due to their different fragmentation patterns. Unfortunately, we couldn’t combine the two acquisition modes, but chromatographic separation could be achieved and, thus, selectivity was assured. The selectivity of the method was further confirmed by a lack of interference observed when measuring the six different serum lots.
Fig. 1.
Chromatograms of the 3 sets of isobaric compounds. A: Protryptiline, 8.04 min; B: Normaprotiline, 8.43 min; C: Nortryptiline, 8.76 min; D: Imipramine, 8.17 min; E Nortrimipramine, 8.85 min; F: Maprotiline, 8.40 min; G: Amitryptiline, 8.72 min.
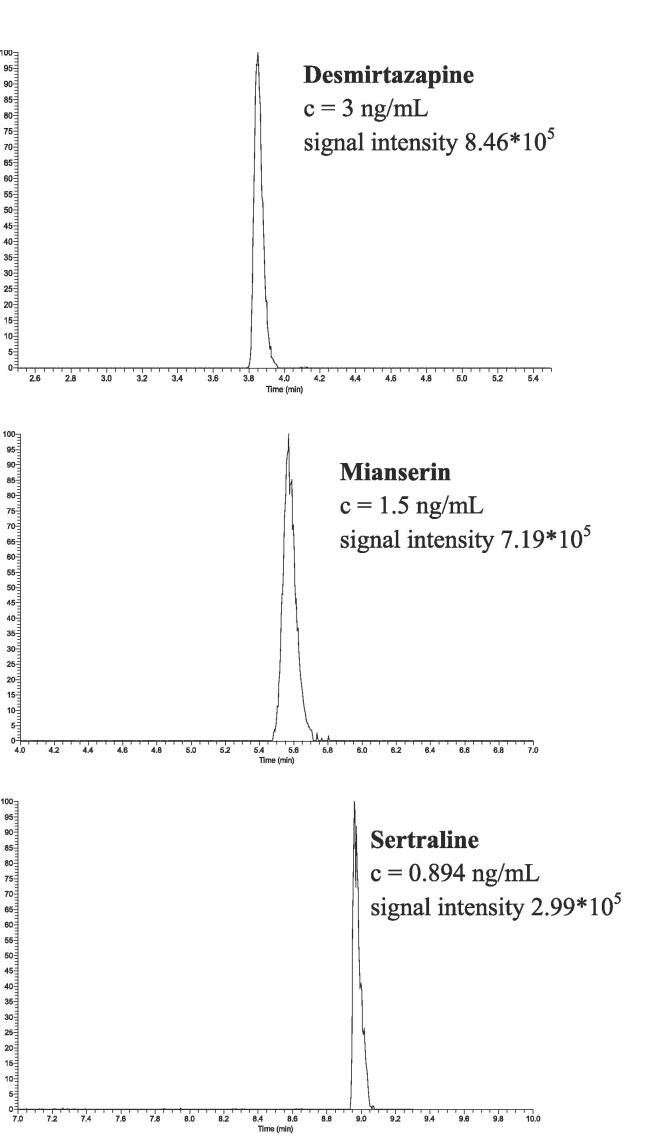
The method proved to be linear for each analyte in the calibration range covered by the calibrators. Representative chromatograms of the lowest calibrator for desmirtazapine, mianserin, and sertraline are reported in Fig. 2. According to the signal intensities of the lowest calibrators it would be possible to measure even lower concentrations. However, from a medical point of view lower concentrations are not relevant and thus we didn’t extend the calibration range.
Fig. 2.
Exemplarily shown chromatograms of the lowest calibrator for desmirtazapine, mianserin and sertraline.
Acceptable results were obtained for both within- and between-run accuracy according to EMA requirements as well as clinical requirements. Results are reported in Table 5. All control samples were within the 85%−115% range, except the Recipe controls for desfluoxetine and fluvoxamine; for these samples the measured values were outside (too low) the given accepted range of the manufacturer.
Table 5.
Accuracy values of all analytes for all QCs and the LLOQs. The within-run accuracy is exemplarily given for run 4 of the validation.
| Compound | LLOQ | QC 1 | QC 2 | QC 3 | Recipe QC 1 | Recipe QC 2 | Biorad QC 1 | Biorad QC 1 |
|---|---|---|---|---|---|---|---|---|
| Amitryptiline | ||||||||
| Within-run [%] | 98.98 | 101.6 | 98.22 | 99.22 | 101.8 | 100.1 | ||
| Between-run [%] | 99.40 | 101.6 | 96.84 | 99.23 | 101.4 | 100.1 | ||
| Atomoxetine | ||||||||
| Within-run [%] | 97.83 | 106.0 | 95.14 | 93.78 | 93.66 | 89.50 | ||
| Between-run [%] | 97.77 | 105.7 | 94.55 | 93.67 | 91.79 | 87.69 | ||
| Citalopram | ||||||||
| Within-run [%] | 99.13 | 100.4 | 96.12 | 97.18 | 113.4 | 107.5 | ||
| Between-run [%] | 99.34 | 100.7 | 95.48 | 96.62 | 112.1 | 107.6 | ||
| Clomipramine | ||||||||
| Within-run [%] | 100.3 | 97.90 | 101.6 | 103.2 | ||||
| Between-run [%] | 100.2 | 98.88 | 101.5 | 103.1 | ||||
| Clozapine | ||||||||
| Within-run [%] | 97.63 | 103.9 | 106.9 | 99.04 | ||||
| Between-run [%] | 97.52 | 103.8 | 105.8 | 98.60 | ||||
| Descitalopram | ||||||||
| within-run [%] | 98.52 | 104.0 | 97.42 | 94.56 | ||||
| between-run [%] | 98.53 | 104.0 | 96.95 | 94.54 | ||||
| Desfluoxetine | ||||||||
| Within-run [%] | 91.92 | 102.3 | 99.46 | 100.2 | 73.55 | 70.25 | ||
| Between-run [%] | 88.65 | 103.0 | 100.5 | 100.7 | 72.69 | 70.28 | ||
| Desipramine | ||||||||
| Within-run [%] | 98.62 | 103.2 | 93.10 | 96.26 | 89.55 | 92.52 | ||
| Between-run [%] | 98.30 | 104.7 | 93.17 | 95.97 | 89.50 | 92.52 | ||
| Desmirtazapine | ||||||||
| Within-run [%] | 98.70 | 104.5 | 98.57 | 97.09 | 114.4 | 98.88 | ||
| Between-run [%] | 98.32 | 106.2 | 98.12 | 95.40 | 112.0 | 98.95 | ||
| Doxepine | ||||||||
| Within-run [%] | 100.4 | 98.53 | 100.4 | 102.5 | ||||
| Between-run [%] | 100.4 | 99.20 | 100.0 | 102.4 | ||||
| Duloxetine | ||||||||
| Within-run [%] | 101.4 | 94.76 | 93.50 | 101.5 | 86.79 | 84.13 | ||
| Between-run [%] | 101.7 | 94.99 | 93.29 | 101.8 | 86.66 | 84.40 | ||
| Fluoxetine | ||||||||
| Within-run [%] | 98.53 | 103.0 | 98.19 | 98.38 | 93.34 | 85.85 | ||
| Between-run [%] | 98.54 | 103.8 | 99.26 | 98.89 | 93.37 | 87.39 | ||
| Fluvoxamine | ||||||||
| Within-run [%] | 99.20 | 102.7 | 95.93 | 97.82 | 73.85 | 72.09 | ||
| between-run [%] | 98.95 | 102.8 | 96.99 | 97.27 | 74.38 | 71.77 | ||
| Imipramine | ||||||||
| within-run [%] | 99.39 | 99.63 | 94.87 | 94.89 | 91.15 | 90.75 | ||
| Between-run [%] | 99.36 | 101.1 | 94.56 | 95.22 | 90.92 | 90.61 | ||
| Maprotiline | ||||||||
| Within-run [%] | 98.33 | 101.9 | 90.67 | 90.17 | ||||
| Between-run [%] | 98.45 | 103.0 | 89.01 | 89.29 | ||||
| Mianserin | ||||||||
| Within-run [%] | 99.80 | 99.25 | 99.41 | 100.0 | 90.52 | 86.23 | ||
| Between-run [%] | 99.88 | 100.2 | 99.24 | 99.75 | 90.57 | 86.29 | ||
| Mirtazapine | ||||||||
| Within-run [%] | 99.83 | 99.68 | 98.91 | 100.4 | 106.2 | 101.3 | ||
| Between-run [%] | 99.83 | 100.8 | 98.63 | 100.3 | 105.5 | 101.5 | ||
| Norclomipramine | ||||||||
| Within-run [%] | 100.0 | 98.74 | 98.14 | 99.86 | ||||
| Between-run [%] | 99.89 | 99.89 | 98.45 | 99.65 | ||||
| Norclozapine | ||||||||
| Within-run [%] | 98.53 | 102.2 | 105.7 | 97.24 | ||||
| Between-run [%] | 93.52 | 101.7 | 108.7 | 103.3 | ||||
| Nordoxepine | ||||||||
| Within-run [%] | 100.6 | 97.19 | 95.79 | 101.5 | 93.74 | 99.17 | ||
| Between-run [%] | 100.6 | 97.55 | 95.65 | 100.9 | 93.62 | 98.74 | ||
| Normaprotiline | ||||||||
| Within-run [%] | 101.3 | 88.69 | 86.87 | 100.0 | ||||
| Between-run [%] | 101.5 | 92.87 | 90.29 | 102.8 | ||||
| Nortryptiline | ||||||||
| Within-run [%] | 98.93 | 102.9 | 95.97 | 97.55 | 98.45 | 99.78 | ||
| Between-run [%] | 98.85 | 103.8 | 94.96 | 97.02 | 97.75 | 98.89 | ||
| Nortrimipramine | ||||||||
| Within-run [%] | 98.37 | 105.2 | 99.35 | 96.93 | ||||
| Between-run [%] | 98.55 | 104.9 | 99.41 | 96.60 | ||||
| Norvenlafaxine | ||||||||
| Within-run [%] | 97.18 | 107.4 | 96.73 | 95.14 | 111.6 | 102.0 | ||
| Between-run [%] | 97.63 | 107.0 | 98.58 | 97.00 | 110.7 | 101.4 | ||
| Paroxetine | ||||||||
| Within-run [%] | 98.79 | 100.9 | 94.76 | 95.77 | 93.60 | 88.38 | ||
| Between-run [%] | 99.15 | 101.5 | 94.93 | 96.03 | 93.15 | 88.92 | ||
| Protryptiline | ||||||||
| Within-run [%] | 99.31 | 94.35 | 95.23 | 107.8 | ||||
| Between-run [%] | 100.3 | 95.91 | 96.12 | 106.6 | ||||
| Reboxetine | ||||||||
| Within-run [%] | 99.27 | 101.8 | 97.99 | 94.94 | 109.5 | 101.3 | ||
| Between-run [%] | 98.84 | 102.2 | 97.87 | 96.03 | 109.9 | 103.0 | ||
| Sertraline | ||||||||
| Within-run [%] | 99.57 | 97.11 | 101.9 | 102.4 | 103.2 | 97.69 | ||
| Between-run [%] | 100.3 | 98.15 | 101.0 | 101.9 | 102.2 | 98.65 | ||
| Trazodone | ||||||||
| Within-run [%] | 98.13 | 108.8 | 101.5 | 101.2 | 112.8 | 100.9 | ||
| Between-run [%] | 98.03 | 109.4 | 100.8 | 100.5 | 112.4 | 100.1 | ||
| Trimipramine | ||||||||
| Within-run [%] | 99.04 | 100.9 | 93.97 | 95.26 | ||||
| Between-run [%] | 99.10 | 101.4 | 94.31 | 95.33 | ||||
| Venlafaxine | ||||||||
| Within-run [%] | 98.51 | 105.3 | 95.47 | 95.79 | 106.2 | 100.2 | ||
| Between-run [%] | 98.22 | 105.4 | 96.39 | 96.04 | 106.0 | 100.8 | ||
The data demonstrated outstanding within- and between-run precision for the method. The maximum % CV for within-run precision was 4.45%. The % CV for between-run precision was below 7.50%. This also indicates, as discussed above, that lower limits of quantification would have been possible, but are not clinically relevant for this analyte set. It is worth noting that, during method development, extraordinary precision values were found for peak areas without correction using internal standard areas (i.e., area ratios), which we didńt expect since good precision can often only be achieved by means of internal standardization when using triple quadrupoles. The within- and between-run precision values for all compounds and QC levels is shown in Table 6.
Table 6.
Precision values of all analytes for all QCs and the LLOQs. The given within-run precision is the average of the within-run precision of all 5 runs of the validation.
| Compound | LLOQ | QC 1 | QC 2 | QC 3 | Recipe QC 1 | Recipe QC 2 | Biorad QC 1 | Biorad QC 1 |
|---|---|---|---|---|---|---|---|---|
| Amitryptiline | ||||||||
| Within-run [%] | 1.57 | 1.07 | 0.911 | 0.806 | 1.72 | 1.46 | ||
| Between-run [%] | 1.68 | 1.10 | 1.22 | 1.29 | 2.77 | 2.07 | ||
| Atomoxetine | ||||||||
| Within-run [%] | 0.959 | 0.808 | 0.580 | 1.07 | 1.78 | 1.60 | ||
| Between-run [%] | 0.938 | 0.964 | 0.703 | 1.25 | 2.13 | 2.20 | ||
| Citalopram | ||||||||
| Within-run [%] | 1.24 | 0.874 | 0.762 | 0.828 | 1.18 | 1.18 | ||
| Between-run [%] | 1.24 | 0.988 | 0.918 | 1.02 | 1.47 | 1.37 | ||
| Clomipramine | ||||||||
| Within-run [%] | 0.606 | 0.696 | 0.834 | 1.07 | ||||
| Between-run [%] | 0.585 | 1.10 | 1.09 | 1.11 | ||||
| Clozapine | ||||||||
| Within-run [%] | 2.29 | 2.51 | 1.33 | 2.25 | ||||
| Between-run [%] | 2.51 | 3.08 | 1.47 | 2.34 | ||||
| Descitalopram | ||||||||
| Within-run [%] | 1.18 | 0.933 | 1.08 | 1.04 | ||||
| Between-run [%] | 1.16 | 1.36 | 1.18 | 1.34 | ||||
| Desfluoxetine | ||||||||
| Within-run [%] | 1.94 | 1.73 | 1.72 | 1.80 | 1.42 | 1.50 | ||
| Between-run [%] | 3.14 | 1.70 | 2.09 | 2.04 | 2.91 | 2.51 | ||
| Desipramine | ||||||||
| Within-run [%] | 0.752 | 0.666 | 1.16 | 1.11 | 1.59 | 0.908 | ||
| Between-run [%] | 0.761 | 1.47 | 1.19 | 1.23 | 2.52 | 1.05 | ||
| Desmirtazapine | ||||||||
| Within-run [%] | 2.06 | 1.33 | 1.78 | 1.51 | 2.21 | 2.04 | ||
| Between-run [%] | 2.00 | 1.71 | 1.81 | 1.70 | 2.55 | 2.57 | ||
| Doxepine | ||||||||
| Within-run [%] | 0.866 | 0.838 | 0.694 | 0.885 | ||||
| Between-run [%] | 0.914 | 1.24 | 0.901 | 1.10 | ||||
| Duloxetine | ||||||||
| Within-run [%] | 2.56 | 1.93 | 2.10 | 1.95 | 1.21 | 2.00 | ||
| Between-run [%] | 2.45 | 2.16 | 2.34 | 2.09 | 1.42 | 2.59 | ||
| Fluoxetine | ||||||||
| Within-run [%] | 1.37 | 1.14 | 1.19 | 1.46 | 1.53 | 1.40 | ||
| Between-run [%] | 1.46 | 1.48 | 1.95 | 2.41 | 1.91 | 2.87 | ||
| Fluvoxamine | ||||||||
| Within-run [%] | 2.16 | 1.22 | 1.44 | 2.18 | 1.53 | 1.84 | ||
| Between-run [%] | 2.05 | 1.38 | 1.98 | 2.16 | 2.64 | 3.36 | ||
| Imipramine | ||||||||
| Within-run [%] | 0.623 | 1.92 | 1.13 | 0.763 | 2.23 | 0.990 | ||
| Between-run [%] | 0.601 | 2.37 | 1.26 | 0.867 | 3.02 | 1.04 | ||
| Maprotiline | ||||||||
| Within-run [%] | 1.08 | 1.44 | 1.39 | 1.64 | ||||
| Between-run [%] | 1.73 | 1.75 | 2.39 | 2.30 | ||||
| Mianserin | ||||||||
| Within-run [%] | 0.882 | 1.73 | 0.933 | 1.00 | 1.44 | 1.14 | ||
| Between-run [%] | 0.902 | 1.72 | 1.01 | 1.08 | 1.80 | 1.42 | ||
| Mirtazapine | ||||||||
| Within-run [%] | 0.970 | 0.668 | 0.956 | 0.746 | 1.23 | 1.09 | ||
| Between-run [%] | 0.935 | 1.39 | 1.01 | 1.02 | 1.32 | 1.36 | ||
| Norclomipramine | ||||||||
| Within-run [%] | 0.643 | 0.701 | 1.33 | 1.26 | ||||
| Between-run [%] | 0.678 | 1.24 | 1.32 | 1.36 | ||||
| Norclozapine | ||||||||
| Within-run [%] | 4.20 | 3.16 | 2.74 | 1.75 | ||||
| Between-run [%] | 4.96 | 3.01 | 3.07 | 3.26 | ||||
| Nordoxepine | ||||||||
| Within-run [%] | 1.23 | 1.15 | 1.01 | 1.02 | 1.57 | 1.19 | ||
| Between-run [%] | 1.26 | 1.58 | 1.10 | 1.20 | 2.94 | 1.49 | ||
| Normaprotiline | ||||||||
| Within-run [%] | 1.83 | 2.45 | 1.74 | 2.95 | ||||
| Between-run [%] | 1.82 | 7.47 | 4.73 | 5.33 | ||||
| Nortryptiline | ||||||||
| Within-run [%] | 1.03 | 0.868 | 0.769 | 0.785 | 1.69 | 0.962 | ||
| Between-run [%] | 1.10 | 1.13 | 1.02 | 1.12 | 2.44 | 1.13 | ||
| Nortrimipramine | ||||||||
| Within-run [%] | 1.99 | 1.87 | 1.48 | 1.32 | ||||
| Between-run [%] | 2.00 | 2.20 | 1.41 | 1.77 | ||||
| Norvenlafaxine | ||||||||
| Within-run [%] | 3.48 | 3.96 | 2.45 | 4.45 | 2.45 | 2.60 | ||
| Between-run [%] | 3.47 | 4.07 | 2.79 | 4.44 | 3.80 | 3.22 | ||
| Paroxetine | ||||||||
| Within-run [%] | 1.54 | 1.25 | 1.18 | 1.23 | 1.41 | 1.49 | ||
| Between-run [%] | 1.49 | 1.44 | 1.34 | 1.45 | 1.64 | 1.97 | ||
| Protryptiline | ||||||||
| Within-run [%] | 2.40 | 2.10 | 2.23 | 3.35 | ||||
| Between-run [%] | 2.56 | 3.64 | 3.72 | 4.34 | ||||
| Reboxetine | ||||||||
| Within-run [%] | 1.28 | 1.60 | 1.65 | 1.80 | 1.71 | 1.33 | ||
| Between-run [%] | 1.37 | 1.74 | 1.94 | 2.61 | 1.77 | 2.05 | ||
| Sertraline | ||||||||
| Within-run [%] | 3.09 | 2.40 | 1.14 | 0.986 | 1.12 | 1.58 | ||
| Between-run [%] | 2.87 | 2.59 | 1.37 | 1.16 | 1.42 | 2.21 | ||
| Trazodone | ||||||||
| Within-run [%] | 2.64 | 2.06 | 1.98 | 2.88 | 1.82 | 2.55 | ||
| Between-run [%] | 2.54 | 2.44 | 2.37 | 3.07 | 1.94 | 2.50 | ||
| Trimipramine | ||||||||
| Within-run [%] | 1.05 | 1.14 | 1.26 | 1.31 | ||||
| Between-run [%] | 1.01 | 1.38 | 1.24 | 1.44 | ||||
| Venlafaxine | ||||||||
| Within-run [%] | 1.72 | 1.73 | 1.61 | 2.05 | 1.63 | 1.48 | ||
| Between-run [%] | 1.73 | 1.82 | 1.64 | 2.12 | 1.73 | 1.63 | ||
Non-significant matrix effect was found for all analytes, with values between 85.8% and 108% according to Matuszewski et al. [22] and variation coefficients of the internal standard normalized matrix factor of the six matrix lots varying between 1.83% and 8.11% (see Table 7). Automated solid phase extraction using the TurboFlow technology was found to be convenient and robust for our method. Efficient removal of serum constituents remaining after protein precipitation by this approach likely had a substantial impact on the excellent precision and reproducibility observed for our method. The applied TurboFlow technology is an online sample clean-up technique that combines, in contrast to the widely used online SPE, two modes of action for removing sample components: size exclusion and the selectivity of the column chemistry [23]. Another advantage is that high injection volumes are possible (in our method 80 µL) even with a relatively high organic amount (in our method 75% acetonitrile), which can increase the sensitivity without overloading the column.
Table 7.
Matrix effect [%], recovery [%], and 48 h stability [%] of the extracted samples of all analytes.
| Matrix effect [%] according to Matuszewski et al. | Matrix effect CV [%] of the ISTD-normalized matrix factor |
Recovery [%] | 48 h stability [%] | |
|---|---|---|---|---|
| Amitryptiline | 97.5 | 4.77 | 91.1 | 99.1 |
| Atomoxetine | 98.5 | 2.26 | 92.5 | 100 |
| Citalopram | 97.3 | 3.37 | 91.9 | 100 |
| Clomipramine | 98.2 | 4.59 | 90.4 | 99.4 |
| Clozapine | 87.8 | 3.86 | 91.5 | 99.4 |
| Descitalopram | 98.1 | 2.88 | 90.9 | 97.7 |
| Desfluoxetine | 88.4 | 3.06 | 88.9 | 95.0 |
| Desipramine | 98.2 | 2.24 | 90.4 | 101 |
| Desmirtazapine | 94.7 | 3.78 | 89.9 | 96.0 |
| Doxepine | 98.6 | 2.02 | 91.0 | 99.2 |
| Duloxetine | 97.7 | 3.78 | 91.6 | 100 |
| Fluoxetine | 96.6 | 3.55 | 90.3 | 99.3 |
| Fluvoxamine | 85.8 | 3.00 | 89.2 | 92.6 |
| Imipramine | 98.6 | 3.30 | 90.6 | 99.0 |
| Maprotiline | 99.1 | 1.83 | 90.5 | 99.5 |
| Mianserin | 97.4 | 2.59 | 90.7 | 99.8 |
| Mirtazapine | 97.6 | 2.31 | 91.8 | 100 |
| Norclomipramine | 96.5 | 8.11 | 89.0 | 99.6 |
| Norclozapine | 99.1 | 2.98 | 89.3 | 99.4 |
| Nordoxepine | 98.6 | 3.06 | 90.3 | 102 |
| Normaprotiline | 99.8 | 5.26 | 89.9 | 97.5 |
| Nortryptiline | 98.3 | 4.08 | 89.7 | 99.7 |
| Nortrimipramine | 96.2 | 4.48 | 90.1 | 100 |
| Norvenlafaxine | 102 | 4.97 | 89.2 | 101 |
| Paroxetine | 96.4 | 4.92 | 89.2 | 99.5 |
| Protryptiline | 99.3 | 6.51 | 90.1 | 104 |
| Reboxetine | 99.0 | 3.26 | 91.6 | 102 |
| Sertraline | 98.7 | 4.20 | 89.0 | 101 |
| Trazodone | 108 | 5.72 | 91.0 | 93.1 |
| Trimipramine | 97.1 | 4.74 | 90.4 | 100 |
| Venlafaxine | 98.3 | 2.84 | 90.0 | 99.5 |
Acceptable recovery values were obtained for all analytes with a minimum value of 88.9% for desmethylfluoxetine. All analytes proved to be stable when the extracted samples were left for 48 h at 10 ⁰C in the autosampler tray, with a maximum loss of signal of 7.4% for fluvoxamine. Recovery and stability values for all analytes are shown in Table 7.
Compared to the EMA guideline, there were some deviations to our validation protocol that should be mentioned. Our calibrator and QC samples were not matrix based. This was due to the large number of analytes that were dissolved in organic stock solutions. If we had added all analytes to blank serum to prepare the calibrators, the serum would have precipitated due to the high organic content. We could not reduce the added amount of organic solvent by using higher concentrated stock solutions since some analytes were purchased already dissolved. This necessitated that we work with solvent calibrators. Furthermore, the stability of the samples was not addressed in the manner that the EMA suggests; we only assessed the stability in extracted samples. We did not collect further stability data, such as freeze-thaw, since our aim was not to establish a routinely used assay (for which these data would be of great importance), but to prove the suitability of HRMS for TDM measurements and, as such, stability data do not play a considerable role. For the same reason, we evaluated the matrix effect only at one concentration and not at two, as suggested by the EMA.
A possible drawback of the described method is the addition of internal standards in one step, dissolved in the precipitation agent. This may be questionable from a purely analytical perspective. However, due to workflow considerations, this approach is a widely used standard in clinical TDM assays, as indicated by various published methods for different analytes (e.g., [24], [25], [26]). Due to the high organic content in the precipitation agent, it can be expected that all protein bonds of analytes are cleaved and equilibration of internal standards up-stream is not required.
For confirmation reasons, we measured leftover patients’ samples and compared the results with the results of a validated LC-MS/MS method from a routine laboratory. In Table 8, the measured values for the mirtazapine samples are shown. To assess the correlation of both methods, a Passing-Bablok-regression [27] was performed. The result is shown in Fig. 3. Since the 95% confidence interval for the intercept includes 0 it can be assumed that there is no systematic difference between the two methods. Since the 95% confidence interval for the slope includes 1 it can be concluded that there is no proportional difference between the compared methods [28]. Thus, it can be assumed, that there is a good agreement between the two methods.
Table 8.
Mirtazapine concentrations [ng/mL].
| Sample | Orbitrap | Triple Quadrupol |
|---|---|---|
| 1 | 31.6 | 30.5 |
| 2 | 26.6 | 25.3 |
| 3 | 57.8 | 53.2 |
| 4 | 20.3 | 19.3 |
| 5 | 35.5 | 33.8 |
| 6 | 41.6 | 39.6 |
| 7 | 30.1 | 30.1 |
| 8 | 32.3 | 28.5 |
| 9 | 59.8 | 58.2 |
| 10 | 25.4 | 23.2 |
| 11 | 78.7 | 76.9 |
| 12 | 30.8 | 29.9 |
| 13 | 78.9 | 77.7 |
| 14 | 8.4 | 8.5 |
| 15 | 60.0 | 60.4 |
| 16 | 16.7 | 16.2 |
| 17 | 29.9 | 28.7 |
| 18 | 63.2 | 66.9 |
| 19 | 54.3 | 56.3 |
| 20 | 75.5 | 70.5 |
Fig. 3.
Passing-Bablok regression analyses of the two methods of mirtazapine measurement, n = 20. Scatter diagram with regression line (black solid line), confidence interval for the regression line (grey solid lines), and identity line (x = y, dashed line). Equation of the regression line: y = 1,0145x + 0,7031. 95% confidence interval for intercept −0.647–2.170 and for slope 0.966–1.064. The Cusum test for linearity shows no significant deviation from linearity (p > 0.05). These results indicate good agreement of the two methods.
4. Conclusion
A liquid chromatography high-resolution mass spectrometry method for the quantification of 31 antidepressive or psychoactive drugs, and some of their metabolites, in human serum was successfully implemented. The described method meets the requirements of our validation protocol, which was oriented towards the Guideline of bioanalytical method validation by the European Medicines Agency in terms of sensitivity, selectivity, linearity of response, precision, and accuracy. Of note was the outstanding precision achieved with the Orbitrap. We demonstrated that high-resolution accurate mass spectrometers are appropriate for accurate and reliable quantification of analytes for TDM measurements. In contrast to quadrupoles, there is also the possibility to use these instruments in the field of metabolomics, which is becoming increasingly important with regard to personalized medicine, for example. Versatility is a major benefit of this instrument type.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Declaration of Competing Interest
The work was supported by Thermo Fisher Scientific providing the instrument for the duration of the study and by RECIPE providing the internal standard mixes; no financial grants or honoraria were provided.
Acknowledgement
The authors would like to thank Thermo Fisher Scientific, especially Claudio DeNardi, for the very kind and helpful support.
References
- 1.Kang J.S., Lee M.H. Overview of therapeutic drug monitoring. Korean J. Internal Med. 2009;24(1):1–10. doi: 10.3904/kjim.2009.24.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hiemke C., Bergemann N., Clement H.W., Conca A., Deckert J., Domschke K., Eckermann G., Egberts K., Gerlach M., Greiner C., et al. Consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology: update 2017. Pharmacopsychiatry. 2018;51(1–02):9–62. doi: 10.1055/s-0043-116492. [DOI] [PubMed] [Google Scholar]
- 3.Adaway J.E., Keevil B.G. Therapeutic drug monitoring and LC-MS/MS. J. Chromatogr., B: Anal. Technol. Biomed. Life Sci. 2012;883–884:33–49. doi: 10.1016/j.jchromb.2011.09.041. [DOI] [PubMed] [Google Scholar]
- 4.Saint-Marcoux F., Sauvage F.L., Marquet P. Current role of LC-MS in therapeutic drug monitoring. Anal. Bioanal. Chem. 2007;388(7):1327–1349. doi: 10.1007/s00216-007-1320-1. [DOI] [PubMed] [Google Scholar]
- 5.Adaway J.E., Keevil B.G., Owen L.J. Liquid chromatography tandem mass spectrometry in the clinical laboratory. Ann. Clin. Biochem. 2015;52(Pt 1):18–38. doi: 10.1177/0004563214557678. [DOI] [PubMed] [Google Scholar]
- 6.Shipkova M., Svinarov D. LC-MS/MS as a tool for TDM services: where are we? Clin. Biochem. 2016;49(13–14):1009–1023. doi: 10.1016/j.clinbiochem.2016.05.001. [DOI] [PubMed] [Google Scholar]
- 7.Maurer H.H. Mass spectrometry for research and application in therapeutic drug monitoring or clinical and forensic toxicology. Ther. Drug Monit. 2018 doi: 10.1097/FTD.0000000000000525. [DOI] [PubMed] [Google Scholar]
- 8.Hu Q., Noll R.J., Li H., Makarov A., Hardman M., Graham Cooks R. The Orbitrap: a new mass spectrometer. J. Mass Spectrom.: JMS. 2005;40(4):430–443. doi: 10.1002/jms.856. [DOI] [PubMed] [Google Scholar]
- 9.Mann M., Kelleher NL. Precision proteomics: the case for high resolution and high mass accuracy. Proc. Natl. Acad. Sci. U.S.A. 2008;105(47):18132–18138. doi: 10.1073/pnas.0800788105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Junot C., Fenaille F., Colsch B., Becher F. High resolution mass spectrometry based techniques at the crossroads of metabolic pathways. Mass Spectrom. Rev. 2014;33(6):471–500. doi: 10.1002/mas.21401. [DOI] [PubMed] [Google Scholar]
- 11.Rochat B., Kottelat E., McMullen J. The future key role of LC-high-resolution-MS analyses in clinical laboratories: a focus on quantification. Bioanalysis. 2012;4(24):2939–2958. doi: 10.4155/bio.12.243. [DOI] [PubMed] [Google Scholar]
- 12.Schuster C., Paal M., Lindner J., Zoller M., Liebchen U., Scharf C., Vogeser M. Isotope dilution LC-orbitrap-HRMS with automated sample preparation for the simultaneous quantification of 11 antimycotics in human serum. J. Pharm. Biomed. Anal. 2019;166:398–405. doi: 10.1016/j.jpba.2019.01.038. [DOI] [PubMed] [Google Scholar]
- 13.Lefeuvre S., Bois-Maublanc J., Hocqueloux L., Bret L., Francia T., Eleout-Da Violante C., Billaud E.M., Barbier F., Got L. A simple ultra-high-performance liquid chromatography-high resolution mass spectrometry assay for the simultaneous quantification of 15 antibiotics in plasma. J. Chromatogr., B: Anal. Technol. Biomed. Life Sci. 2017;1065–1066:50–58. doi: 10.1016/j.jchromb.2017.09.014. [DOI] [PubMed] [Google Scholar]
- 14.Deltombe O., Mertens T., Eloot S., Verstraete A.G. Development and validation of an ultra-high performance liquid chromatography – high resolution mass spectrometry method for the quantification of total and free teicoplanin in human plasma. Clin. Biochem. 2018:30990–30991. doi: 10.1016/j.clinbiochem.2018.12.010. pii: S0009-9120(18)30990-1. [DOI] [PubMed] [Google Scholar]
- 15.Bhatnagar A., McKay M.J., Crumbaker M., Ahire K., Karuso P., Gurney H., Molloy M.P. Quantitation of the anticancer drug abiraterone and its metabolite Δ(4)-abiraterone in human plasma using high-resolution mass spectrometry. J. Pharm. Biomed. Anal. 2018;154:66–74. doi: 10.1016/j.jpba.2018.03.012. [DOI] [PubMed] [Google Scholar]
- 16.Qu L., Qian J., Ma P., Yin Z. Utilizing online-dual-SPE-LC with HRMS for the simultaneous quantification of amphotericin B, fluconazole, and fluorocytosine in human plasma and cerebrospinal fluid. Talanta. 2017;165:449–457. doi: 10.1016/j.talanta.2016.12.052. [DOI] [PubMed] [Google Scholar]
- 17.Qu L., Fan Y., Wang W., Ma K., Yin Z. Development, validation and clinical application of an online-SPE-LC-HRMS/MS for simultaneous quantification of phenobarbital, phenytoin, carbamazepine, and its active metabolite carbamazepine 10,11-epoxide. Talanta. 2016;158:77–88. doi: 10.1016/j.talanta.2016.05.036. [DOI] [PubMed] [Google Scholar]
- 18.Qua L., Wanga W., Zengb D., Lub Y., Yin Z. Quantitative performance of online SPE-LC coupled to Q-Exactive for the analysis of sofosbuvir in human plasma. RSC Adv. 2015;5:98269–98277. [Google Scholar]
- 19.Favretto D., Stocchero G., Nalesso A., Vogliardi S., Boscolo-Berto R., Montisci M., Ferrara S.D. Monitoring haloperidol exposure in body fluids and hair of children by liquid chromatography-high-resolution mass spectrometry. Ther. Drug Monit. 2013;35(4):493–501. doi: 10.1097/FTD.0b013e3182892d11. [DOI] [PubMed] [Google Scholar]
- 20.Dahmane E., Boccard J., Csajka C., Rudaz S., Décosterd L., Genin E., Duretz B., Bromirski M., Zaman K., Testa B., Rochat B. Quantitative monitoring of tamoxifen in human plasma extended to 40 metabolites using liquid-chromatography high-resolution mass spectrometry: new investigation capabilities for clinical pharmacology. Anal. Bioanal. Chem. 2014;406(11):2627–2640. doi: 10.1007/s00216-014-7682-2. [DOI] [PubMed] [Google Scholar]
- 21.European Medicine Agency, Guideline on Bioanalytical Method Validation, 2011. http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2011/08/WC500109686.pdf, (accessed November 2015).
- 22.Matuszewski B.K., Constanzer M.L., Chavez-Eng C.M. Strategies for the assessment of matrix effect in quantitative bioanalytical methods based on HPLC-MS/MS. Anal. Chem. 2003;75(13):3019–3030. doi: 10.1021/ac020361s. [DOI] [PubMed] [Google Scholar]
- 23.Rudewicz P.J. Turbulent flow bioanalysis in drug metabolism and pharmacokinetics. Bioanalysis. 2011;3(14):1663–1671. doi: 10.4155/bio.11.137. [DOI] [PubMed] [Google Scholar]
- 24.Seger C., Tentschert K., Stoggl W., Griesmacher A., Ramsay S.L. A rapid HPLC-MS/MS method for the simultaneous quantification of cyclosporine A, tacrolimus, sirolimus and everolimus in human blood samples. Nat. Protoc. 2009;4(4):526–534. doi: 10.1038/nprot.2009.25. [DOI] [PubMed] [Google Scholar]
- 25.Zander J., Maier B., Zoller M., Teupser D., Vogeser M. Quantification of linezolid in serum by LC-MS/MS using semi-automated sample preparation and isotope dilution internal standardization. Clin. Chem. Lab. Med.: CCLM/FESCC. 2014;52(3):381–389. doi: 10.1515/cclm-2013-0594. [DOI] [PubMed] [Google Scholar]
- 26.Hasselstrom J. Quantification of antidepressants and antipsychotics in human serum by precipitation and ultra high pressure liquid chromatography-tandem mass spectrometry. J. Chromatogr., B: Anal. Technol. Biomed. Life Sci. 2011;879(1):123–128. doi: 10.1016/j.jchromb.2010.11.024. [DOI] [PubMed] [Google Scholar]
- 27.Passing H., Bablok A new biometrical procedure for testing the equality of measurements from two different analytical methods. Application of linear regression procedures for method comparison studies in clinical chemistry, Part I. J. Clin. Chem. Clin. Biochem. Zeitschrift fur klinische Chemie und klinische Biochemie. 1983;21(11):709–720. doi: 10.1515/cclm.1983.21.11.709. [DOI] [PubMed] [Google Scholar]
- 28.Bilic-Zulle L. Comparison of methods: passing and Bablok regression. Biochemia Medica. 2011;21(1):49–52. doi: 10.11613/bm.2011.010. [DOI] [PubMed] [Google Scholar]