Abstract
The diverse populations of tissue-resident and transitory T cells present in the skin share a common functional need to enter, traverse, and interact with their environment. These processes are largely dependent on the regulated expression of adhesion molecules, such as selectins and integrins, which mediate bidirectional interactions between immune cells and skin stroma. Dysregulation and engagement of adhesion pathways contribute to ectopic T-cell activity in tissues, leading to the initiation and/or exacerbation of chronic inflammation. In this paper, we review how the molecular interactions supported by adhesion pathways contribute to T-cell dynamics and function in the skin. A comprehensive understanding of the molecular mechanisms underpinning T-cell adhesion in inflammatory skin disorders will facilitate the development of novel tissue-specific therapeutic strategies.
Abbreviations: AD, atopic dermatitis; BM, basement membrane; DC, dendritic cell; DETC, dendritic epidermal γδ T cell; ECM, extracellular matrix; HF, hair follicle; JC, John Cunningham; LAD, leukocyte adhesion deficiency; PML, progressive multifocal leukoencephalopathy; Th, T helper; Treg, regulatory T cell; Trm, tissue-resident memory
Overview
Adhesion is a fundamental process in immune cell function. This is definitively demonstrated by leukocyte adhesion deficiency (LAD) syndrome, a rare genetic disorder caused by defects in a select number of adhesion genes, especially in ITGB2. Without bone marrow transplantation, patients with severe disease are prone to recurrent infections, demonstrate impaired wound healing, and are unlikely to survive beyond 2 years of life (Almarza Novoa et al., 2018). Immune responses in peripheral tissues require adhesion pathways to mediate cell recruitment, tissue immunosurveillance, and intercellular interaction between different cell types. These activities are dependent on a large assemblage of adhesion proteins, including integrins, selectins, and cadherins that mediate cell‒cell and cell‒extracellular matrix (ECM) interactions. Contact between adhesion molecules and their ligands provides a means for force transmission and signal transduction that enables cellular movement, positioning, and the tight interactions required for immune synapse formation (Basu and Huse, 2017; Cavallaro and Dejana, 2011; Hammer et al., 2019). Adhesive function is directly driven by adhesion proteins but also encompasses the layers of regulatory mechanisms that create cell lineage (i.e., CD4+ T cell vs. CD8+ T cell), cell state (i.e., quiescence vs. activation), and tissue (i.e., skin vs. colon) specificity.
Mammalian skin contains large populations of tissue-resident and recirculating T cells. These cells protect against infection and also play an important role in maintaining skin barrier integrity and tissue homeostasis. Regulatory T cells (Tregs) interact with epithelial stem cells to facilitate differentiation during hair follicle (HF) cycling (Ali et al., 2017b), whereas Tregs, subsets of CD8+ T cells, MAIT cells, and γδ T cells contribute to wound repair and re-epithelialization after skin injury (Constantinides et al., 2019; Harrison et al., 2019; Jameson et al., 2002; Nosbaum et al., 2016). In addition, Tregs keep local immune responses in balance by tightly regulating inflammatory signaling (Kalekar and Rosenblum, 2019; Kalekar et al., 2019). Performing these functions in the skin requires adhesion-mediated recruitment, interstitial migration, and cell‒cell interaction. In this study, we follow T cells on a journey as they enter and traverse the skin, interact with antigen-presenting cells or malignant cells, and eventually emigrate to draining lymph nodes. We review how the outcome of this journey is determined by the composition of adhesion molecules expressed on various skin T-cell populations and describe how inflammation and immune activation shape adhesive capacity.
Not surprisingly, dysregulation of T cells in the skin is a prominent feature of many skin disorders, and either augmentation or suppression of their activity provides a compelling strategy toward treating human disease (Ho and Kupper, 2019). Given the importance of cellular adhesion in immune cell function, adhesion molecules have long been considered promising therapeutic targets (Ley et al., 2016). Research and clinical efforts in this area have produced several therapies currently approved for the treatment of autoimmune diseases, including multiple sclerosis and Crohn’s disease (Ley et al., 2016). Unfortunately, these successes have yet to be realized for the treatment of skin disorders. To date, Efalizumab, the only adhesion-targeting therapy approved for a skin disease (psoriasis) was withdrawn from clinical use secondary to severe and potentially life-threatening side effects (Pugashetti and Koo, 2009). Nonetheless, rapid progress in understanding the nuances surrounding T-cell adhesion in tissues provides the justification for continued exploration of these pathways as targets for therapeutic manipulation. In this paper, we discuss the attempts to develop therapies modulating T-cell adhesion and examine the opportunities for applying new research findings toward developing treatments for skin disease.
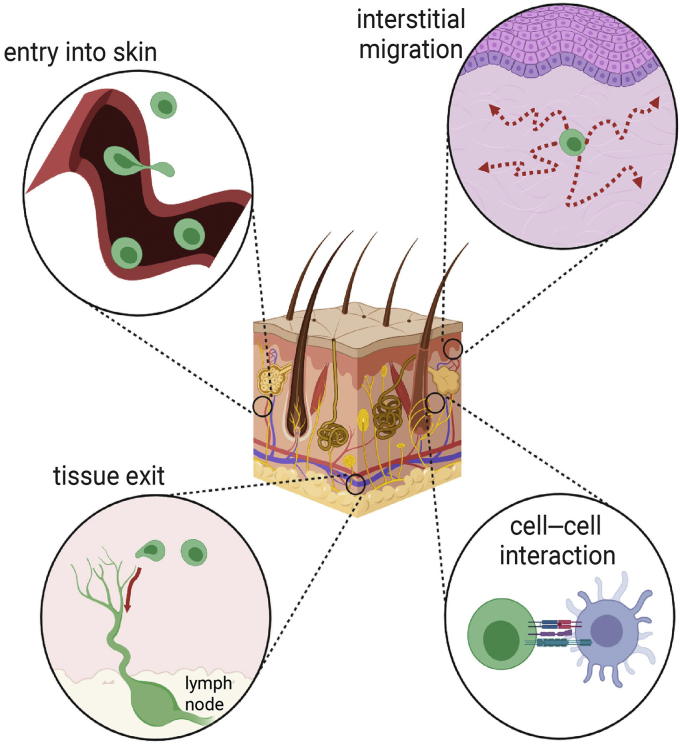
A T cell’s guide to skin adhesion
In the absence of inflammatory signals, tissue-resident T cells, including tissue-resident memory (Trm) cells and dendritic epidermal γδ T cells (DETCs) (in mice), continually survey the skin while other subsets circulate between the tissue, draining lymph nodes, and peripheral blood (Brown et al., 2010; Collins et al., 2016; Gebhardt et al., 2011). Despite the significant differences in the ontogeny and transcriptional signature of tissue-resident and recirculating T cells (Szabo et al., 2019), adhesion molecules are a shared molecular requirement for movement into and through the skin. However, differential induction or activation of adhesion pathways contributes to nuances in the functional behaviors of each population. Broadly, there are four phases of T-cell behavior dependent on adhesion: entry into the skin, interstitial migration, interactions with other cells, and egress from the skin (Figure 1). An individual T cell may not be primed to perform each step sequentially, but it will need to express a specific set of molecules to accomplish any given task. In the following sections, we discuss how T cells use adhesion molecules in the context of each process. However, it should be noted that the majority of research in this field to date has focused on proinflammatory T cells (i.e., conventional CD4+ and CD8+ T cells) and that much less is known about how Tregs utilize adhesion and adhesion-associated molecules to regulate their functions in the skin.
Figure 1.
Cellular adhesion is required for T-cell function in the skin. To extend immune protection to peripheral tissues, T cells must actively leave circulation, migrate through interstitial spaces, interact with other cells, and eventually return to circulation by lymphatic vessels. Each of these processes is dependent on adhesive interactions between T cells and other cells or between T cells and the extracellular matrix. Expression patterns of adhesion molecules temporally and spatially regulate T-cell dynamics by selectively assisting specific movements. Only those T cells expressing the correct collection of adhesion receptors will be able to traverse the tissue environment. Inflammation controls T-cell access to the tissue by modulating this adhesion molecule signature.
A sticky toolkit: regulation of T-cell adhesion
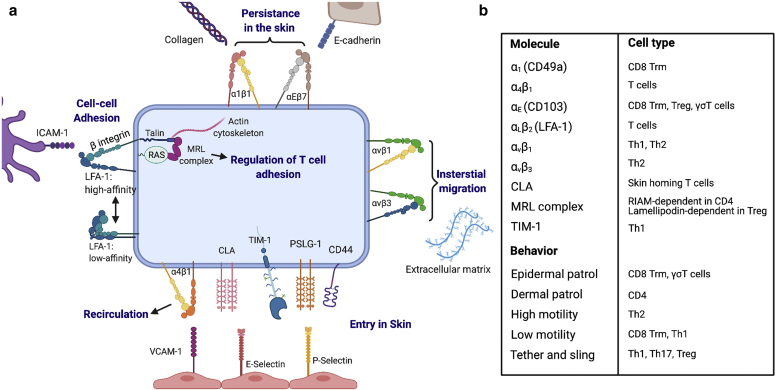
Cellular adhesion is principally driven by several groups of molecules expressed on T cells, including integrins, selectins, cadherins, and Ig superfamily members (Figure 2) (Harjunpää et al., 2019). T cells also express a number of other glycoproteins, such as CD44, that contribute to mediating adhesion (Baaten et al., 2012). Many additional proteins indirectly influence cellular behavior by regulating the ability of expressed adhesion molecules to bind their ligand. For example, most integrins are present on the cell surface in a relatively closed conformational state with a low affinity for ligand binding. TCR engagement or chemokine exposure produces signaling cascades that induce a conformational change in both integrin subunits. This extends the integrin into an active form and substantially increases ligand affinity (Harjunpää et al., 2019). Transformation into an active conformation is dependent on the integrin-binding proteins, talin and kindlin, which mediate force transmission through connections to the cytoskeleton (Harjunpää et al., 2019; Sun et al., 2019). Through these cytoskeletal interactions, integrins and other adhesion molecules impart T cells with mechanosensitivity that is essential for navigating the mechanically dynamic tissue environment of the skin (Biggs et al., 2020; Sun et al., 2019).
Figure 2.
A diverse set of molecules drive T-cell adhesion. (a) To enter the skin, T cells must interact with blood vessel endothelial cells (a, bottom) before crossing the endothelium into the tissue parenchyma. Initial tethering and/or rolling interactions are mediated by low-affinity binding between the T-cell glycoproteins PSLG-1, CLA (a carbohydrate variant of PSLG-1), and CD44, with E- and P-selectin expressed on the endothelium. Firm adhesion and transendothelial migration require high-affinity ligation of the integrins LFA-1 and VLA-4 with the Ig superfamily members ICAM-1 and VCAM-1. Once in the skin, interactions between T cells and skin epithelium as well as the interaction of T cells with infected or cancerous target cells are also integrin dependent (a, top and left). Integrin αEβ7 binding to E-cadherin is especially important for association with the epithelium. (b) Although the expression of cell adhesion molecules is highly sensitive to the inflammatory milieu, several differences in adhesion activity have been observed across T-cell subsets. Heterogeneity in the levels and timing of adhesion molecule expression will directly influence T-cell behavior in the skin, including tissue entry, immunosurveillance, and local migration patterns. Th, T helper; Treg, regulatory T cell; Trm, tissue-resident memory.
Interestingly, additional adaptor proteins modulate talin‒integrin interactions leading to cell lineage and cell state‒dependent augmentation of adhesion. Recently, we found that the talin-binding C-type lectin, layilin, is preferentially expressed on highly activated CD8+ T cells infiltrating melanoma tumors but is nearly absent on T cells in peripheral blood (Borowsky and Hynes, 1998; Mahuron et al., 2020). Layilin enhances integrin αLβ2 (LFA-1) activation and binding to its ligand ICAM-1 to promote antitumor immunity (Mahuron et al., 2020). CD8+ T cells expressing high levels of IL-17 (Tc17 cells) infiltrating psoriatic lesions are also enriched for layilin expression compared with T cells from healthy skin, suggesting that although layilin may protect against cancer, it could contribute to autoimmunity (Liu et al., 2020). Differential regulation of integrin activation was also demonstrated between Tregs and CD4+ T-conventional cells. Whereas most CD4+ T cells are entirely dependent on RIAM to mediate β integrin binding to talin, this protein is dispensable in Tregs. Deletion of RIAM protected mice in a spontaneous colitis model owing to a reduced accumulation of effector T cells in the gut, whereas Treg homing to this tissue was normal (Sun et al., 2021). Lamellipodin, a RIAM paralog preferentially expressed in Tregs, is able to facilitate integrin‒talin binding in these cells (Sun et al., 2021).
The expression pattern of adhesion molecules themselves also has importance in dictating T-cell function. For T cells to gain access to peripheral tissues, they must express an adhesion signature appropriate for that tissue. Both γδ T cells and αβ T cells require the expression of the glycoprotein CLA to facilitate skin entry by binding to E-selectin expressed on skin endothelial cells (Berg et al., 1991; Biedermann et al., 2002; Fuhlbrigge et al., 1997; Jiang et al., 2010). Studies examining T-cell recruitment under inflammatory conditions have implicated CD43 and CD44 as additional E-selectin ligands contributing to skin entry (Ali et al., 2017a; Nácher et al., 2011). Integrin expression is also critical because CD18−/− (the β2 subunit of LFA-1) knockout mice exhibited reduced inflammation in allergic contact dermatitis and delayed-type hypersensitivity reactions owing to impaired T-cell accumulation in skin lesions. Notably, Langerhans cell precursors and dendritic cells (DCs) displayed normal migratory behavior (Grabbe et al., 2002). Conversely, mice with a hypomorphic mutation that reduces CD18 levels to 2–16% of wild type spontaneously develop skin lesions mirroring human psoriasis (Bullard et al., 1996). In this context, both CD4+ and CD8+ T cells efficiently enter the skin, and tissue pathology is potentiated by a defective Treg compartment (Kess et al., 2003; Singh et al., 2013; Wang et al., 2008). Reduced CD18 was observed to hinder Treg interaction with DCs, impair their suppressive capability, and promote lineage instability by converting Tregs to IL-17‒producing effector T helper (Th)17 cells (Singh et al., 2013; Wang et al., 2008). Another integrin important for skin-specific function (although not for entry into the skin) is αEβ7. CD103 (the αE subunit of αEβ7) is a marker of Trm cells and is widely expressed among T cells in the skin (Hardenberg et al., 2018). αEβ7 interacts with E-cadherin and likely has a role in positioning T cells within the tissue by allowing an interaction with the epithelium (discussed in detail in the following section) (Hardenberg et al., 2018). Mice deficient in CD103 develop severe skin lesions at least partially owing to a defective Treg compartment (Braun et al., 2015; Schön et al., 2000). Collectively, these observations begin to define an adhesome signature intrinsically connected to T-cell function in the skin.
Entry into the skin
T-cell entry into the skin is initiated by tethering and rolling interactions with blood vessel endothelial cells (Garrood et al., 2006; Schön et al., 2003). CLA and a related glycoprotein, PSLG-1 (CD162), expressed on T cells bind endothelial E- and P-selectin. In addition, CD44 coassociation with integrin α4β1 (VLA-4) has been found to facilitate early binding to the endothelial ligand VCAM-1 (Issekutz and Issekutz, 2002; Nandi et al., 2004; Schön et al., 2003; Siegelman et al., 2000). These relatively low-affinity interactions act to slow down the T cells caught in high-speed blood flow (Garrood et al., 2006; Schön et al., 2003). Some nuance has been reported in the mechanisms utilized by different T-cell subsets to initiate tethering. Th1, Th17, and Tregs were found to use a tether and sling behavior to improve adherence efficiency. These subsets were markedly better at forming these structures than Th2 cells, which directly translated into superior attachment to the vessel wall (Abadier et al., 2017). Similarly, in Th1 and Th17 but not in Th2 cells, TIM-1 binds P-selectin to promote skin entry (Angiari et al., 2014). Interestingly, this may explain why tissue-resident Th2 cells are absent in healthy skin of C57BL/6 mice, whereas Tregs and Th1 cells are readily observed (Nussbaum et al., 2013).
Low-velocity travel along vessel walls stimulates integrin activation leading to firm adhesion primarily through LFA-1/ICAM-1 and VLA-4/VCAM-1 binding (Garrood et al., 2006; Schön et al., 2003). Firm adhesion fully arrests the T cells and allows cytoskeleton reorganization and cellular polarization to form a leading-edge lamellipodium. Lymphocytes then traverse the endothelium primarily through endothelial cell junctions (Garrood et al., 2006; Sumagin and Sarelius, 2010). Integrins facilitate transendothelial migration through bidirectional signaling that both increases endothelial permissiveness and directly prepares T cells for tissue entry. LFA-1 engagement with ICAM-1 on endothelial cells induces a MAPK signaling cascade that in turn promotes vascular permeability and induces cytokine and chemokine production (Dragoni et al., 2017; Sumagin and Sarelius, 2010). In T cells, LFA-1 ligation during transendothelial migration was recently found to drive the synthesis of intracellular complement C3, which was among the most enriched molecular signature of tissue lymphocytes. Examining the T cells from patients with LAD syndrome, the study authors observed reduced C3 mRNA and impaired effector cytokine production (Kolev et al., 2020). Other integrins besides LFA-1 likely contribute to transendothelial migration because the double deletion of the cytoskeletal effector proteins, VASP and EVL, impaired T-cell trafficking into lipopolysaccharide-treated ear skin owing to defective integrin α4 function. Strikingly, T cells deficient in both VASP and EVL exhibited normal crawling and adhesion under shear flow but defective transendothelial migration (Estin et al., 2017).
Because T-cell entry into tissue is a critical point in their functional capability, the processes of rolling, firm adhesion, and transendothelial migration are highly influenced by inflammatory and pathologic conditions. Such stimuli modify adhesion molecule expression on both T cells and the endothelium to regulate lymphocyte entry into the skin (Capece et al., 2017; DeNucci et al., 2010; Ley and Kansas, 2004; Mahuron et al., 2020). For example, analysis of peripheral blood from both adult and pediatric patients with atopic dermatitis (AD) revealed the expansion of CLA+ Th2 cells, consistent with the type-2 inflammatory skew of this disease (Czarnowicki et al., 2015a, 2015b). In ex vivo transendothelial migration experiments, blood-derived T cells from patients with scleroderma exhibited significantly enhanced migratory ability compared with healthy control cells (Stummvoll et al., 2004). In support of these findings, CD4+ T cells from patients with scleroderma not only expressed elevated levels of CD11a (the αL subunit of LFA-1) but also reduced the methylation of the CD11a promoter, indicating an epigenetically encoded propensity for increased integrin expression (Wang et al., 2014). These results suggest that in patients with skin disease, circulating T cells are likely ectopically primed for tissue entry.
Interstitial migration
After passing through the endothelium, T cells enter into a complex 3‒dimensional interstitial space comprising tissue cells, soluble signaling mediators, and the ECM. T cells must integrate cues from this environment to patrol the tissue and position themselves within the skin microarchitecture (Gaylo et al., 2016). Cutaneous tissue is a dynamic and high-force environment that experiences differential levels of mechanical stress across the epidermis and dermis and during tissue renewal (Biggs et al., 2020; Hsu et al., 2018). Notably, injury and inflammation precipitate the remodeling of the ECM, which influence the signals T cells receive through their adhesion receptors (Gaylo et al., 2016; Hsu et al., 2018). Consequently, T-cell motility behaviors (i.e., travel speed and area of immunosurveillance) are directly dictated by the mechanical landscape within the tissue (Gaylo et al., 2016). An abnormal distribution of tissue forces is a feature of several skin disorders (Hsu et al., 2018).
T-cell subsets appear to differentially segregate in the skin. A large subset of Tregs localizes to HFs (Ali et al., 2017b; Sanchez Rodriguez et al., 2014). Effector and memory CD4+ T cells are also commonly observed around the HFs; however, these cells also patrol within the dermis (Collins et al., 2016; Gaylo et al., 2016; Gebhardt et al., 2011). In contrast, CD8+ T cells and DETCs preferentially associate with the epidermal basement membrane (BM), although in healthy human skin, a relatively large proportion of CD8s inhabit the epidermis and are capable of migrating back and forth between the dermis and epidermis (Cheuk et al., 2014; Dijkgraaf et al., 2019; Gebhardt et al., 2011; Thelen and Witherden, 2020). Interstitial motility of skin T cells was presumed to be through integrin-independent amoeboid crawling, as has been described in lymph nodes and for myeloid cells (Gaylo et al., 2016; Jacobelli et al., 2009; Lämmermann et al., 2008; Weninger et al., 2014). However, evidence suggests that at least in the context of inflammation, effector CD4+ T cells require integrin αv to move within the dermis (Fernandes et al., 2020; Overstreet et al., 2013). Patrolling T cells use the ECM as a scaffold for interstitial migration, relying on αvβ1 and αvβ3 interactions with fibronectin-decorating collagen fibers (Fernandes et al., 2020; Overstreet et al., 2013). The differences between the motility observed during steady state and inflammation are likely determined by structural remodeling of the ECM (Fernandes et al., 2020; Gaylo et al., 2016; Overstreet et al., 2013; Sorokin, 2010). In this process there is also evidence of subset-driven variation because Th2 differentiation increases αvβ3 expression compared with Th1 cells. Functionally, Th2 cells were observed to patrol a larger area of inflamed dermis than Th1 cells, and manipulation of αvβ3 levels was sufficient to reverse this behavior (Gaylo-Moynihan et al., 2019).
Transit between the dermis and epidermis in healthy skin is likely facilitated by pores in the epithelial BM, although this has not been studied in detail (Oakford et al., 2011). Leukocyte trafficking through such structures is presumably an integrin-independent process owing to their dependency on protrusion-based contractile motility when passing through tight spaces (Jacobelli et al., 2010; Kelley et al., 2014; Lämmermann et al., 2008). Nonetheless, integrin α1β1 blockade with a mAb against α1 (CD49a) markedly reduced the accumulation of T cells in the epidermis of human psoriatic skin xenografts. Treatment with this antibody also mitigated otherwise spontaneous progression of asymptotic transplants toward pathology, implicating epidermal T cells as mediators of psoriasis (Conrad et al., 2007). However, a more recent study examining α1 knockout in murine T-cell responses to Herpes simplex virus infection found no requirement for this integrin in epidermal localization (Bromley et al., 2020). Instead, α1 supported CD8+ Trm cell persistence in the epidermis, promoted the formation of dendrite protrusions in these cells, and was directly linked to effector function through the induction of IFN-γ (Bromley et al., 2020; Cheuk et al., 2017). Given that the original blocking experiments were performed in the context of transplantation and relied on the skin from patients with psoriasis, it is possible that specific inflammatory signals lead to reorganization of the ECM and precipitate a requirement for α1β1 in dermal to epidermal transit. Integrin αEβ7 is another adhesion molecule implicated in T-cell function at the epithelium. Induced on T cells by TGFβ signaling, αEβ7 binds to E-cadherin, which is highly expressed on epithelial cells (Hardenberg et al., 2018; Sivasankar, 2013). Although CD103 (αE) was found to influence DETC dendrite formation, it did not contribute to the cellular morphology of CD8+ Trm cells (Schlickum et al., 2008; Zaid et al., 2017). Trm cells deficient in CD103 demonstrated increased motility within the epidermis, suggesting that this integrin acts to locally restrain (or guide) T-cell migration (Zaid et al., 2017). Consistent with this possibility, deletion of CD103 did not impact the entry of CD8+ Trm cells in the epidermis but was required for their continued persistence at this skin compartment (MacKay et al., 2013).
Cell‒cell interactions
In addition to meditating motility, adhesion molecules are inextricably linked to T-cell activation and differentiation. LFA-1, VLA-4, and αEβ7 are participants in immunological synapse formation and, through engagement with ligands on antigen-presenting or target cells, modulate TCR signaling (Basu and Huse, 2017; Hammer et al., 2019; Hardenberg et al., 2018; Jankowska et al., 2018). This contribution to cell‒cell interaction is directly pertinent to CD8+ effector function because either LFA-1 or αEβ7 is required for efficient cytotoxicity toward cancer cells (Franciszkiewicz et al. 2013; Le Floc’h et al. 2007). A B16 melanoma cell line engineered to overexpress E-cadherin was more easily eliminated by immune cells, suggesting that the stoichiometry of integrin‒cadherin interactions between T cells and their targets regulates effector activity (Shields et al., 2019). In agreement with these data, culturing CD8+ Trm cells on the α1 ligand, collagen IV, augments IFN-γ production (Cheuk et al., 2017). Skin epithelium may also be able to provide T-costimulatory signals by LFA-1. Primary human keratinocyte cultures pretreated with IFN-γ were able to activate naïve peripheral blood T cells and induce differentiation toward a Th1 and Th17 phenotype (Orlik et al., 2020). Whereas CD2 ligation is necessary for full T-cell activation, LFA-1 binding to ICAM-1 facilitates cell‒cell contact and promotes productive signaling (Orlik et al., 2020; Varga et al., 2010; Verma et al., 2016). Targeting molecular regulators of LFA-1 activation, including its inhibitor MAP4K4 and layilin, has shown potential as a strategy for the treatment of tumors and viral infection (Esen et al., 2020; Mahuron et al., 2020).
Recirculation
A large proportion of antigen-experienced skin T cells eventually exit the tissue through draining lymphatics to reenter blood circulation (Bromley et al., 2013; Brown et al., 2010; Collins et al., 2016; Hunter et al., 2016). Even human CD4+CD103+ Trm cells have been identified in circulation, indicating the potential for this population to be mobilized from tissues (Klicznik et al., 2019). T-cell emigration from the skin appears to be at least partially dependent on CCR7/CCL21 and appears to be restrained by S1P signaling (Bromley et al., 2013; Laidlaw et al., 2019; Ledgerwood et al., 2008). Notably, S1P receptor antagonism was found to halt lymphocyte egress by inhibiting migration across lymphatic endothelium in an LFA-1/ICAM-1– and VLA-4/VCAM-1–dependent manner (Ledgerwood et al., 2008). Tregs but not other T cells have been observed to facilitate their own egress by directly promoting the interactions with endothelial VCAM-1 (Brinkman et al., 2016). Presentation of lymphotoxin by Tregs to lymphatic endothelial cells induces VCAM-1 expression and is associated with Treg emigration to the draining lymph node (Brinkman et al., 2016; Piao et al., 2018). More recent intravital microscopy experiments revealed that T cells actively crawl through the skin afferent lymphatic capillaries before reaching larger collector vessels. This crawling is dependent on LFA-1 and is modulated by inflammation. Contact hypersensitivity increases T-cell velocity through the upregulation of ICAM-1 on lymphatic endothelial cells. Whereas VLA-4 was dispensable for T-cell movement within the lymphatics, its blockade reduced migration from inflamed skin to the draining lymph node, suggesting a contribution for this integrin to initial lymphatic entry (Teijeira et al., 2017).
A new horizon in T-cell adhesion
Although there is increasing evidence pointing to tissue and cellular specificity as a major driver of adhesion dynamics, many of the molecular pathways discussed earlier are broadly conserved across leukocyte lineages. Transformational advances in understanding T-cell function will come through linking cellular behavior to the biochemistry of adhesion molecules dissected in other leukocytes. For example, although we have highlighted several emerging examples of tissue specificity in T-cell migration, there is significantly stronger evidence for this concept in neutrophil tissue recruitment (Margraf et al., 2019). Contributing mechanisms, such as the existence of a β2 integrin bent-open headpiece conformation that impairs neutrophil adhesion as well as the reverse transendothelial migratory capability of these cells, have not been deeply examined in T cells (Burn and Alvarez, 2017; Fan et al., 2016; Margraf et al., 2019). In addition, focused efforts to understand T-cell adhesion in the context of their natural three-dimensional environment will be of the highest yield. Advances in intravital microscopy as well as organoid culture systems provide promise toward illuminating human T-cell behavior in the skin.
Adhesion-based therapeutics in skin disease: promises and challenges
Because T-cell biology is inextricably linked to cellular adhesion, manipulating these pathways in inflammatory disorders, including in skin diseases, has long been recognized as a compelling therapeutic strategy (Figure 3). However, the fundamental importance and complexity of adhesion biology present an enormous challenge. Designing a drug to target molecules such as integrins requires a deep understanding of molecular mechanisms across multiple biological states, tissues, and cell types and against a backdrop of intrinsic regulatory layers. Nonetheless, overcoming these difficulties presents an exceptional opportunity for creating highly efficacious precision medicines.
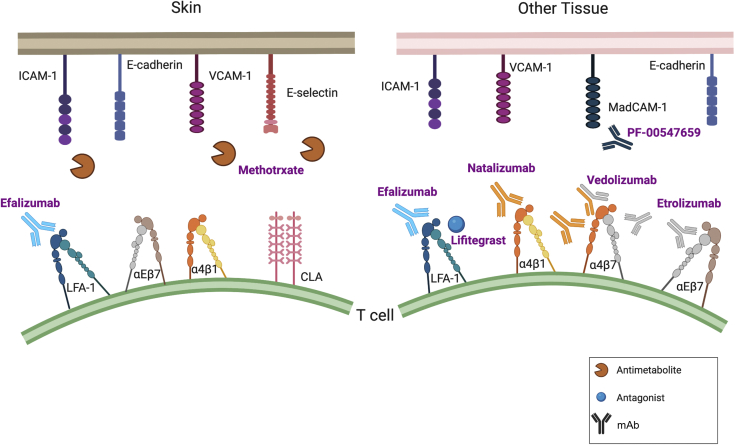
Figure 3.
Strategies to target adhesion molecules in disease. Several adhesion molecule inhibitors (both small molecules and mAbs) have been developed to treat inflammatory disorders. To date, efalizumab is the only integrin-blocking therapy to have been approved for use in skin diseases, although it was subsequently withdrawn owing to severe complications. Nonetheless, these drugs illustrate potential opportunities and pitfalls in designing adhesion-based therapeutics. Whereas vedolizumab exhibits exquisite specificity by binding to a conformational epitope unique to the heterodimerization of α4 and β7 integrins, natalizumab and efalizumab encountered problems owing to the promiscuity of their integrin targets (α4 and αL, respectively). Etrolizumab, a β7 integrin‒blocking monoclonal in phase III clinical trials for the treatment of IBD may have relevance to skin disease given the importance of αEβ7 on skin-resident T cells (Sandborn et al., 2020). One of methotrexate’s proposed mechanisms is the downregulation of selectin and immunoglobulin superfamily molecules expressed on endothelial cells. However, only limited success has been observed with a monoclonal (PF-00547659) targeting MAdCAM-1 on mucosal endothelium (Sandborn et al., 2018). IBD, inflammatory bowel disease.
Therapies targeting adhesion pathways are currently approved for the treatment of autoimmune and inflammatory diseases, including multiple sclerosis, Crohn’s disease, and UC (Alsoud et al., 2020; Ley et al., 2016). Predominantly, these approaches rely on a mAb-based blockade of integrin function, although a small molecule LFA-1 antagonist, lifitegrast, is currently approved for dry eye disease (Haber et al., 2019). Vedolizumab, which possesses a safe and effective track record in the treatment of Crohn’s disease and UC (Alsoud et al., 2020; Feagan et al., 2013), provides an illustrative example for the successful development of tissue-specific adhesion‒based therapeutics. Vedolizumab binds to a conformational epitope unique to the heterodimerization of human α4 and β7 integrins. This allows precise antagonism of α4β7 interactions with MAdCAM-1 on mucosal endothelial cells (Soler et al., 2009). Similar to the role of CLA in skin entry, MAdCAM-1 binding mediates the transmigration of T cells across the intestinal mucosa (Fedyk et al., 2012; Soler et al., 2009). Clinical studies and nonhuman primate experiments demonstrate that vedolizumab’s molecular specificity translates into targeted immunomodulation. Healthy subjects and animals treated with vedolizumab exhibited significant loss of β7+ leukocytes from the intestinal tract but not from other tissues (Fedyk et al., 2012; Milch et al., 2013).
Vedolizumab’s success in achieving a tissue-specific response contrasts with that of natalizumab, another approved anti-α4β7 monoclonal. Binding an α4 epitope, natalizumab, blocks both α4β7 and α4β1 (VLA-1), which is more widely expressed, including on skin-homing T cells (discussed earlier) (Fedyk et al., 2012; Ley et al., 2016). Unfortunately, this comparatively reduced specificity manifests in the potential for developing progressive multifocal leukoencephalopathy (PML), a life-threatening CNS side effect due to reactivation of the polyoma John Cunningham (JC) virus (Cortese et al., 2021; Ley et al., 2016; Milch et al., 2013). Although the exact pathogenesis of this complication remains unclear, blockade of α4β1‒VCAM-1 interaction is a likely culprit. JC virus‒reactive T cells are important contributors to viral control and disease prevention, but in contrast to vedolizumab, natalizumab treatment is associated with a reduction in CNS T-cell populations. (Cortese et al., 2021; Ley et al., 2016; Milch et al., 2013).
Efalizumab, which was briefly approved for treating psoriasis before being removed from the market owing to incidences of PML (complication risk of 1 in 400 compared with that of 1 in 1,000 for natalizumab), illustrates an additional complexity in developing integrin-based drugs (Cortese et al., 2021; Ley et al., 2016). An anti-αL monoclonal, efalizumab, was expected to be highly specific for blocking LFA-1 (αLβ2) because the αL integrin chain only pairs with β2 (Ley et al., 2016; Mancuso et al., 2020). However, although efalizumab effectively reduces LFA-1 at the T-cell surface, it was also observed to cause the downregulation of α4β1 (Grönholm et al., 2016; Guttman-Yassky et al., 2008; Mancuso et al., 2020). The mechanisms underlying this response are unclear; however, outside-in signaling through LFA-1 is known to influence α4β1, and crosstalk between these integrins is a feature of their molecular function (Grönholm et al., 2016; Kim and Hammer, 2019; Mancuso et al., 2020). In vitro experiments indicate that efalizumab can induce α4β1 without LFA-1 activation, which is a requirement for integrin crosstalk (Mancuso et al., 2020). Therefore, α4β1 downregulation may occur through additional biochemical interactions.
Although efalizumab ultimately proved disappointing, its effectiveness at reducing cutaneous inflammation is clear. A phase 3 clinical trial testing efalizumab in the treatment of psoriasis found that the majority of patients experienced at least a 50% reduction in lesion severity and area (Lebwohl et al., 2003). Several case reports and small trials have also reported success in using efalizumab to treat hypertrophic lupus erythematosus, lichen planus, and AD (Böhm and Luger, 2007; Navarini et al., 2010; Takiguchi et al., 2007). Follow-up analysis on patients with relapsing psoriasis after cessation of efalizumab observed renewed T-cell and myeloid skin infiltration in skin lesions (Johnson-Huang et al., 2012). In addition to reducing skin homing of pathogenic T-cell clones, efalizumab induces T-cell hyporesponsiveness characterized by attenuated T-cell activation and downregulation of costimulatory and TCR complex molecules (Guttman-Yassky et al., 2008; Kuschei et al., 2011). Together, these findings support the potential for targeting LFA-1‒mediated adhesion in skin disease.
An alternative approach to integrin blockade is to target selectins and their ligands. Modulation of selectin interactions required for T-cell entry into the skin may improve selectivity toward inflamed tissue because the expression of this molecule is highly sensitive to inflammatory stimuli (Ley and Kansas, 2004). Methotrexate is a synthetic folic acid analog and anti-inflammatory agent that has long been used in the treatment of inflammatory skin disorders (Shen et al., 2012). Although methotrexate has broad effects, it is interesting that at least some of its anti-inflammatory properties are due to the regulation of adhesion molecules and prevention of lymphocyte accumulation in the skin. Specifically, methotrexate treatment reduces endothelial expression of E-selectin and VCAM-1 while also downregulating CLA on T cells (Dahlman-Ghozlan et al., 2004; Johnston et al., 2005; Sigmundsdottir et al., 2004). Preclinical experiments attempting to directly target E-selectin with a blocking mAb successfully prevented human Th2-cell infiltration into human skin xenografts (Biedermann et al., 2002). Likewise, administration of a sugar derivative pan-selectin inhibitor reduced inflammation in a mouse model of allergic dermatitis (Ikegami-Kuzuhara et al., 2001). However, a clinical trial treating patients with psoriasis observed no therapeutic benefit of E-selectin blockade (Bhushan et al., 2002).
A potentially attractive therapeutic approach is targeting integrin modulating proteins. This may have the advantage of inhibiting or augmenting adhesion of specific lymphocyte subsets in specific tissues only in inflammatory or malignant contexts. As mentioned previously, the C-type lectin, layilin, modulates LFA-1 activation on CD8+ T cells and is only expressed on a limited number of lymphocyte populations in inflamed or malignant skin (Liu et al., 2020; Mahuron et al., 2020). Thus, selective modulation of proteins such as layilin may have a beneficial therapeutic effect with fewer adverse reactions than directly targeting integrins or their ligands.
Conclusions and outlook
Adhesion molecules are critical mediators of T-cell accumulation and function in nonlymphoid tissues. These molecules exert functional effects almost continuously as T cells enter and traverse the skin. This breadth of activity means that adhesion mechanisms are highly complex and exhibit multiple layers of regulation and molecular redundancy. Nevertheless, recent research has uncovered several examples of tissue and cell-type specificities, such as between Th1 and Th2 cells or between Tregs and effector T cells. Small variations in how T-cell populations use adhesion molecules to interact with their local environment can result in a large influence on immune response and disease pathology. Because many inflammatory skin disorders have a strong T-cell component, modulating adhesive interactions has long been considered an attractive therapeutic strategy; yet, this approach has most likely not realized its full potential. Despite the current absence of an approved adhesion-targeting therapy for skin diseases, a detailed understanding of relevant molecular pathways has grown significantly since efalizumab was first developed. Developing drugs to target T-cell adhesion presents a formidable challenge in overcoming intrinsic molecular redundancy and functional complexity. The severe off-target complications associated with efalizumab and natalizumab reinforce this difficulty and emphasize a need to comprehensively consider epitope specificity, expression profile, and molecular regulation in designing therapeutics to modulate cellular adhesion. At the same time, doing so opens the possibility for harnessing exquisite biological precision.
ORCIDs
Joshua M. Moreau: http://orcid.org/0000-0002-1227-1508
Victoire Gouirand: http://orcid.org/0000-0002-2666-0061
Michael D. Rosenblum: http://orcid.org/0000-0002-0462-5732
Author Contributions
Conceptualization: JMM, VG, MDR; Investigation: JMM, VG, MDR; Methodology: JMM, VG, MDR; Project Administration: JMM, VG, MDR; Resources: JMM, VG, MDR; Software: JMM, VG, MDR; Supervision: MDR; Validation: JMM, VG, MDR; Visualization: JMM, VG, MDR; Writing - Original Draft Preparation: JMM, VG, MDR; Writing - Review and Editing: JMM, VG, MDR
Acknowledgments
This work was primarily supported by grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institute of Health (R01AR071944 and DP2-AR068130) to MDR. JMM is supported by the Human Frontier Science Program Long-term Fellowship (LT000183/2018-L). Illustrations were created, in part, with BioRender.com.
Conflict of Interest
MDR is a consultant and cofounder of Treg Bio., a company focused on regulatory T-cell therapeutics. The remaining authors declare no conflict of interest.
accepted 30 April 2021; corrected proof published online 29 May 2021
Footnotes
Cite this article as: JID Innovations 2021;X:100014
References
- Abadier M., Pramod A.B., McArdle S., Marki A., Fan Z., Gutierrez E., et al. Effector and regulatory T cells roll at high shear stress by inducible tether and sling formation. Cell Rep. 2017;21:3885–3899. doi: 10.1016/j.celrep.2017.11.099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ali A.J., Abuelela A.F., Merzaban J.S. An analysis of trafficking receptors shows that CD44 and P-selectin glycoprotein ligand-1 collectively control the migration of activated human T-cells. Front Immunol. 2017;8:492. doi: 10.3389/fimmu.2017.00492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ali N., Zirak B., Rodriguez R.S., Pauli M.L., Truong H.A., Lai K., et al. Regulatory T cells in skin facilitate epithelial stem cell differentiation. Cell. 2017;169:1119–1129.e11. doi: 10.1016/j.cell.2017.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Almarza Novoa E., Kasbekar S., Thrasher A.J., Kohn D.B., Sevilla J., Nguyen T., et al. Leukocyte adhesion deficiency-I: a comprehensive review of all published cases. J Allergy Clin Immunol Pract. 2018;6:1418–1420.e10. doi: 10.1016/j.jaip.2017.12.008. [DOI] [PubMed] [Google Scholar]
- Alsoud D., Vermeire S., Verstockt B. Monitoring vedolizumab and ustekinumab drug levels in patients with inflammatory bowel disease: hype or hope? Curr Opin Pharmacol. 2020;55:17–30. doi: 10.1016/j.coph.2020.09.002. [DOI] [PubMed] [Google Scholar]
- Angiari S., Donnarumma T., Rossi B., Dusi S., Pietronigro E., Zenaro E., et al. TIM-1 glycoprotein binds the adhesion receptor P-selectin and mediates T cell trafficking during inflammation and autoimmunity. Immunity. 2014;40:542–553. doi: 10.1016/j.immuni.2014.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baaten B.J., Tinoco R., Chen A.T., Bradley L.M. Regulation of antigen-experienced T cells: lessons from the quintessential memory marker CD44. Front Immunol. 2012;3:23. doi: 10.3389/fimmu.2012.00023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Basu R., Huse M. Mechanical communication at the immunological synapse. Trends Cell Biol. 2017;27:241–254. doi: 10.1016/j.tcb.2016.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berg E.L., Yoshino T., Rott L.S., Robinson M.K., Warnock R.A., Kishimoto T.K., et al. The cutaneous lymphocyte antigen is a skin lymphocyte homing receptor for the vascular lectin endothelial cell-leukocyte adhesion molecule 1. J Exp Med. 1991;174:1461–1466. doi: 10.1084/jem.174.6.1461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhushan M., Bleiker T.O., Ballsdon A.E., Allen M.H., Sopwith M., Robinson M.K., et al. Anti-e-selectin is ineffective in the treatment of psoriasis: a randomized trial. Br J Dermatol. 2002;146:824–831. doi: 10.1046/j.1365-2133.2002.04743.x. [DOI] [PubMed] [Google Scholar]
- Biedermann T., Schwärzler C., Lametschwandtner G., Thoma G., Carballido-Perrig N., Kund J., et al. Targeting CLA/E-selectin interactions prevents CCR4-mediated recruitment of human Th2 memory cells to human skin in vivo. Eur J Immunol. 2002;32:3171–3180. doi: 10.1002/1521-4141(200211)32:11<3171::AID-IMMU3171>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- Biggs L.C., Kim C.S., Miroshnikova Y.A., Wickström S.A. Mechanical forces in the skin: roles in tissue architecture, stability, and function. J Invest Dermatol. 2020;140:284–290. doi: 10.1016/j.jid.2019.06.137. [DOI] [PubMed] [Google Scholar]
- Böhm M., Luger T.A. Lichen planus responding to efalizumab. J Am Acad Dermatol. 2007;56(Suppl.):S92–S93. doi: 10.1016/j.jaad.2006.10.957. [DOI] [PubMed] [Google Scholar]
- Borowsky M.L., Hynes R.O. Layilin, a novel talin-binding transmembrane protein homologous with C- type lectins, is localized in membrane ruffles. J Cell Biol. 1998;143:429–442. doi: 10.1083/jcb.143.2.429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braun A., Dewert N., Brunnert F., Schnabel V., Hardenberg J.H., Richter B., et al. Integrin α E (CD103) Is involved in regulatory T-cell function in allergic contact hypersensitivity. J Invest Dermatol. 2015;135:2982–2991. doi: 10.1038/jid.2015.287. [DOI] [PubMed] [Google Scholar]
- Brinkman C.C., Iwami D., Hritzo M.K., Xiong Y., Ahmad S., Simon T., et al. Treg engage lymphotoxin beta receptor for afferent lymphatic transendothelial migration. Nat Commun. 2016;7:12021. doi: 10.1038/ncomms12021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bromley S.K., Akbaba H., Mani V., Mora-Buch R., Chasse A.Y., Sama A., et al. CD49a regulates cutaneous resident memory CD8+ T cell persistence and response. Cell Rep. 2020;32:108085. doi: 10.1016/j.celrep.2020.108085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bromley S.K., Yan S., Tomura M., Kanagawa O., Luster A.D. Recirculating memory T cells are a unique subset of CD4 + T cells with a distinct phenotype and migratory pattern. J Immunol. 2013;190:970–976. doi: 10.4049/jimmunol.1202805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown M.N., Fintushel S.R., Lee M.H., Jennrich S., Geherin S.A., Hay J.B., et al. Chemoattractant receptors and lymphocyte egress from extralymphoid tissue: changing requirements during the course of inflammation. J Immunol. 2010;185:4873–4882. doi: 10.4049/jimmunol.1000676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bullard D.C., Scharffetter-Kochanek K., Mcarthur M.J., Chosay J.G., Mcbride M.E., Montgomery C.A., et al. A polygenic mouse model of psoriasiform skin disease in CD18-deficient mice. Proc Natl Acad Sci USA. 1996;93:2116–2121. doi: 10.1073/pnas.93.5.2116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burn T., Alvarez J.I. Reverse transendothelial cell migration in inflammation: to help or to hinder? Cell Mol Life Sci. 2017;74:1871–1881. doi: 10.1007/s00018-016-2444-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Capece T., Walling B.L., Lim K., Kim KD Do, Bae S., Chung H.L., et al. A novel intracellular pool of LFA-1 is critical for asymmetric CD8+ T cell activation and differentiation. J Cell Biol. 2017;216:3817–3829. doi: 10.1083/jcb.201609072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cavallaro U., Dejana E. Adhesion molecule signalling: not always a sticky business. Nat Rev Mol Cell Biol. 2011;12:189–197. doi: 10.1038/nrm3068. [DOI] [PubMed] [Google Scholar]
- Cheuk S., Schlums H., Gallais Sérézal I., Martini E., Chiang S.C., Marquardt N., et al. CD49a expression defines tissue-resident CD8+ T cells poised for cytotoxic function in human skin. Immunity. 2017;46:287–300. doi: 10.1016/j.immuni.2017.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheuk S., Wikén M., Blomqvist L., Nylén S., Talme T., Ståhle M., et al. Epidermal Th22 and Tc17 cells form a localized disease memory in clinically healed psoriasis. J Immunol. 2014;192:3111–3120. doi: 10.4049/jimmunol.1302313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins N., Jiang X., Zaid A., Macleod B.L., Li J., Park C.O., et al. Skin CD4(+)memory T cells exhibit combined cluster-mediated retention and equilibration with the circulation. Nat Commun. 2016;7:11514. doi: 10.1038/ncomms11514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conrad C., Boyman O., Tonel G., Tun-Kyi A., Laggner U., De Fougerolles A., et al. Alphabeta1 integrin is crucial for accumulation of epidermal T cells and the development of psoriasis. Nat Med. 2007;13:836–842. doi: 10.1038/nm1605. [DOI] [PubMed] [Google Scholar]
- Constantinides M.G., Link V.M., Tamoutounour S., Wong A.C., Perez-Chaparro P.J., Han S.J., et al. MAIT cells are imprinted by the microbiota in early life and promote tissue repair. Science. 2019;366 doi: 10.1126/science.aax6624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cortese I., Reich D.S., Nath A. Progressive multifocal leukoencephalopathy and the spectrum of JC virus-related disease. Nat Rev Neurol. 2021;17:37–51. doi: 10.1038/s41582-020-00427-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Czarnowicki T., Esaki H., Gonzalez J., Malajian D., Shemer A., Noda S., et al. Early pediatric atopic dermatitis shows only a cutaneous lymphocyte antigen (CLA)(+) TH2/TH1 cell imbalance, whereas adults acquire CLA(+) TH22/TC22 cell subsets. J Allergy Clin Immunol. 2015;136:941–951.e3. doi: 10.1016/j.jaci.2015.05.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Czarnowicki T., Gonzalez J., Shemer A., Malajian D., Xu H., Zheng X., et al. Severe atopic dermatitis is characterized by selective expansion of circulating TH2/TC2 and TH22/TC22, but not TH17/TC17, cells within the skin-homing T-cell population. J Allergy Clin Immunol. 2015;136:104–115.e7. doi: 10.1016/j.jaci.2015.01.020. [DOI] [PubMed] [Google Scholar]
- Dahlman-Ghozlan K., Ortonne J.P., Heilborn J.D., Stephansson E. Altered tissue expression pattern of cell adhesion molecules, ICAM-1, E-selectin and VCAM-1, in bullous pemphigoid during methotrexate therapy. Exp Dermatol. 2004;13:65–69. doi: 10.1111/j.0906-6705.2004.00113.x. [DOI] [PubMed] [Google Scholar]
- DeNucci C.C., Pagán A.J., Mitchell J.S., Shimizu Y. Control of alpha4beta7 integrin Expression and CD4 T cell Homing by the beta1 integrin Subunit. J Immunol. 2010;184:2458–2467. doi: 10.4049/jimmunol.0902407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dijkgraaf F.E., Matos T.R., Hoogenboezem M., Toebes M., Vredevoogd D.W., Mertz M., et al. Tissue patrol by resident memory CD8+ T cells in human skin [published correction appears in Nat Immunol 2020;21:696] Nat Immunol. 2019;20:756–764. doi: 10.1038/s41590-019-0404-3. [DOI] [PubMed] [Google Scholar]
- Dragoni S., Hudson N., Kenny B.A., Burgoyne T., McKenzie J.A., Gill Y., et al. Endothelial MAPKs direct ICAM-1 signaling to divergent inflammatory functions. J Immunol. 2017;198:4074–4085. doi: 10.4049/jimmunol.1600823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Esen E., Sergin I., Jesudason R., Himmels P., Webster J.D., Zhang H., et al. MAP4K4 negatively regulates CD8 T cell-mediated antitumor and antiviral immunity. Sci Immunol. 2020;5 doi: 10.1126/sciimmunol.aay2245. [DOI] [PubMed] [Google Scholar]
- Estin M.L., Thompson S.B., Traxinger B., Fisher M.H., Friedman R.S., Jacobelli J. Ena/VASP proteins regulate activated T-cell trafficking by promoting diapedesis during transendothelial migration. Proc Natl Acad Sci USA. 2017;114:E2901–E2910. doi: 10.1073/pnas.1701886114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fan Z., McArdle S., Marki A., Mikulski Z., Gutierrez E., Engelhardt B., et al. Neutrophil recruitment limited by high-affinity bent β2 integrin binding ligand in cis. Nat Commun. 2016;7:12658. doi: 10.1038/ncomms12658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feagan B.G., Rutgeerts P., Sands B.E., Hanauer S., Colombel J.F., Sandborn W.J., et al. Vedolizumab as induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2013;369:699–710. doi: 10.1056/NEJMoa1215734. [DOI] [PubMed] [Google Scholar]
- Fedyk E.R., Wyant T., Yang L.L., Csizmadia V., Burke K., Yang H., et al. Exclusive antagonism of the α4β7 integrin by vedolizumab confirms the gut-selectivity of this pathway in primates. Inflamm Bowel Dis. 2012;18:2107–2119. doi: 10.1002/ibd.22940. [DOI] [PubMed] [Google Scholar]
- Fernandes N.R.J., Reilly N.S., Schrock D.C., Hocking D.C., Oakes P.W., Fowell D.J. CD4+ T cell interstitial migration controlled by fibronectin in the inflamed skin. Front Immunol. 2020;11:1501. doi: 10.3389/fimmu.2020.01501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Floc’h A., Jalil A., Vergnon I., Le Maux Chansac B., Lazar V., Bismuth G., et al. Alpha E beta 7 integrin interaction with E-cadherin promotes antitumor CTL activity by triggering lytic granule polarization and exocytosis. J Exp Med. 2007;204:559–570. doi: 10.1084/jem.20061524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franciszkiewicz K., Le Floc’h A., Boutet M., Vergnon I., Schmitt A., Mami-Chouaib F. CD103 or LFA-1 engagement at the immune synapse between cytotoxic T cells and tumor cells promotes maturation and regulates T-cell effector functions. Cancer Res. 2013;73:617–628. doi: 10.1158/0008-5472.CAN-12-2569. [DOI] [PubMed] [Google Scholar]
- Fuhlbrigge R.C., Kieffer J.D., Armerding D., Kupper T.S. Cutaneous lymphocyte antigen is a specialized form of PSGL-1 expressed on skin-homing T cells. Nature. 1997;389:978–981. doi: 10.1038/40166. [DOI] [PubMed] [Google Scholar]
- Garrood T., Lee L., Pitzalis C. Molecular mechanisms of cell recruitment to inflammatory sites: general and tissue-specific pathways. Rheumatology (Oxford) 2006;45:250–260. doi: 10.1093/rheumatology/kei207. [DOI] [PubMed] [Google Scholar]
- Gaylo A., Schrock D.C., Fernandes N.R.J., Fowell D.J. T cell interstitial migration: motility cues from the inflamed tissue for micro- and macro-positioning. Front Immunol. 2016;7:428. doi: 10.3389/fimmu.2016.00428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaylo-Moynihan A., Prizant H., Popović M., Fernandes N.R.J., Anderson C.S., Chiou K.K., et al. Programming of distinct chemokine-dependent and -independent search strategies for Th1 and Th2 cells optimizes function at inflamed sites. Immunity. 2019;51:298–309.e6. doi: 10.1016/j.immuni.2019.06.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gebhardt T., Whitney P.G., Zaid A., MacKay L.K., Brooks A.G., Heath W.R., et al. Different patterns of peripheral migration by memory CD4+ and CD8+ T cells. Nature. 2011;477:216–219. doi: 10.1038/nature10339. [DOI] [PubMed] [Google Scholar]
- Grabbe S., Varga G., Beissert S., Steinert M., Pendl G., Seeliger S., et al. β2 integrins are required for skin homing of primed T cells but not for priming naive T cells. J Clin Invest. 2002;109:183–192. doi: 10.1172/JCI11703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grönholm M., Jahan F., Bryushkova E.A., Madhavan S., Aglialoro F., Soto Hinojosa L.S., et al. LFA-1 integrin antibodies inhibit leukocyte α4β1-mediated adhesion by intracellular signaling. Blood. 2016;128:1270–1281. doi: 10.1182/blood-2016-03-705160. [DOI] [PubMed] [Google Scholar]
- Guttman-Yassky E., Vugmeyster Y., Lowes M.A., Chamian F., Kikuchi T., Kagen M., et al. Blockade of CD11a by efalizumab in psoriasis patients induces a unique state of T-cell hyporesponsiveness. J Invest Dermatol. 2008;128:1182–1191. doi: 10.1038/jid.2008.4. [DOI] [PubMed] [Google Scholar]
- Haber S.L., Benson V., Buckway C.J., Gonzales J.M., Romanet D., Scholes B. Lifitegrast: a novel drug for patients with dry eye disease. Ther Adv Ophthalmol. 2019;11 doi: 10.1177/2515841419870366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammer J.A., Wang J.C., Saeed M., Pedrosa A.T. Origin, organization, dynamics, and function of actin and actomyosin networks at the T cell immunological synapse. Annu Rev Immunol. 2019;37:201–224. doi: 10.1146/annurev-immunol-042718-041341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hardenberg J.B., Braun A., Schön M.P. A Yin and Yang in epithelial immunology: the roles of the αE(CD103)β7 integrin in T cells. J Invest Dermatol. 2018;138:23–31. doi: 10.1016/j.jid.2017.05.026. [DOI] [PubMed] [Google Scholar]
- Harjunpää H., Llort Asens M.L., Guenther C., Fagerholm S.C. Cell adhesion molecules and their roles and regulation in the immune and tumor microenvironment. Front Immunol. 2019;10:1078. doi: 10.3389/fimmu.2019.01078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrison O.J., Linehan J.L., Shih H.Y., Bouladoux N., Han S.J., Smelkinson M., et al. Commensal-specific T cell plasticity promotes rapid tissue adaptation to injury. Science. 2019;363 doi: 10.1126/science.aat6280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho A.W., Kupper T.S. T cells and the skin: from protective immunity to inflammatory skin disorders. Nat Rev Immunol. 2019;19:490–502. doi: 10.1038/s41577-019-0162-3. [DOI] [PubMed] [Google Scholar]
- Hsu C.K., Lin H.H., Harn H.I.C., Hughes M.W., Tang M.J., Yang C.C. Mechanical forces in skin disorders. J Dermatol Sci. 2018;90:232–240. doi: 10.1016/j.jdermsci.2018.03.004. [DOI] [PubMed] [Google Scholar]
- Hunter M.C., Teijeira A., Halin C. T cell trafficking through lymphatic vessels. Front Immunol. 2016;7:613. doi: 10.3389/fimmu.2016.00613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikegami-Kuzuhara A., Yoshinaka T., Ohmoto H., Inoue Y., Saito T. Therapeutic potential of a novel synthetic selectin blocker, OJ-R9188, in allergic dermatitis. Br J Pharmacol. 2001;134:1498–1504. doi: 10.1038/sj.bjp.0704397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Issekutz A.C., Issekutz T.B. The role of E-selectin, P-selectin, and very late activation Antigen-4 in T lymphocyte migration to dermal inflammation. J Immunol. 2002;168:1934–1939. doi: 10.4049/jimmunol.168.4.1934. [DOI] [PubMed] [Google Scholar]
- Jacobelli J., Friedman R.S., Conti M.A., Lennon-Dumenil A.M., Piel M., Sorensen C.M., et al. Confinement-optimized three-dimensional T cell amoeboid motility is modulated via myosin IIA–regulated adhesions. Nat Immunol. 2010;11:953–961. doi: 10.1038/ni.1936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobelli J., Bennett F.C., Pandurangi P., Tooley A.J., Krummel M.F. Myosin-IIA and ICAM-1 regulate the interchange between two distinct modes of T cell migration. J Immunol. 2009;182:2041–2050. doi: 10.4049/jimmunol.0803267. [DOI] [PubMed] [Google Scholar]
- Jameson J., Ugarte K., Chen N., Yachi P., Fuchs E., Boismenu R., et al. A role for skin gammadelta T cells in wound repair. Science. 2002;296:747–749. doi: 10.1126/science.1069639. [DOI] [PubMed] [Google Scholar]
- Jankowska K.I., Williamson E.K., Roy N.H., Blumenthal D., Chandra V., Baumgart T., et al. Integrins modulate T cell receptor signaling by constraining actin flow at the immunological synapse. Front Immunol. 2018;9:25. doi: 10.3389/fimmu.2018.00025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang X., Campbell J.J., Kupper T.S. Embryonic trafficking of gammadelta T cells to skin is dependent on E/P selectin ligands and CCR4. Proc Natl Acad Sci USA. 2010;107:7443–7448. doi: 10.1073/pnas.0912943107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson-Huang L.M., Pensabene C.A., Shah K.R., Pierson K.C., Kikuchi T., Lentini T., et al. Post-therapeutic relapse of psoriasis after CD11a blockade is associated with T cells and inflammatory myeloid DCs. PLoS One. 2012;7:1–9. doi: 10.1371/journal.pone.0030308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnston A., Gudjonsson J.E., Sigmundsdottir H., Ludviksson B.R., Valdimarsson H. The anti-inflammatory action of methotrexate is not mediated by lymphocyte apoptosis, but by the suppression of activation and adhesion molecules. Clin Immunol. 2005;114:154–163. doi: 10.1016/j.clim.2004.09.001. [DOI] [PubMed] [Google Scholar]
- Kalekar L.A., Cohen J.N., Prevel N., Sandoval P.M., Mathur A.N., Moreau J.M., et al. Regulatory T cells in skin are uniquely poised to suppress profibrotic immune responses. Sci Immunol. 2019;4 doi: 10.1126/sciimmunol.aaw2910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalekar L.A., Rosenblum M.D. Regulatory T cells in inflammatory skin disease: from mice to humans. Int Immunol. 2019;31:457–463. doi: 10.1093/intimm/dxz020. [DOI] [PubMed] [Google Scholar]
- Kelley L.C., Lohmer L.L., Hagedorn E.J., Sherwood D.R. Traversing the basement membrane in vivo: a diversity of strategies. J Cell Biol. 2014;204:291–302. doi: 10.1083/jcb.201311112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kess D., Peters T., Zamek J., Wickenhauser C., Tawadros S., Loser K., et al. CD4 + T cell-associated pathophysiology critically depends on CD18 gene dose effects in a murine model of psoriasis. J Immunol. 2003;171:5697–5706. doi: 10.4049/jimmunol.171.11.5697. [DOI] [PubMed] [Google Scholar]
- Kim S.H.J., Hammer D.A. Integrin crosstalk allows CD4+ T lymphocytes to continue migrating in the upstream direction after flow. Integr Biol (Camb) 2019;11:384–393. doi: 10.1093/intbio/zyz034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klicznik M.M., Morawski P.A., Höllbacher B., Varkhande S.R., Motley S.J., Kuri-Cervantes L., et al. Human CD4 + CD103 + cutaneous resident memory T cells are found in the circulation of healthy individuals. Sci Immunol. 2019;4 doi: 10.1126/sciimmunol.aav8995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kolev M., West E.E., Kunz N., Chauss D., Moseman E.A., Rahman J., et al. Diapedesis-induced integrin signaling via LFA-1 facilitates tissue immunity by inducing intrinsic complement C3 expression in immune cells. Immunity. 2020;52:513–527.e8. doi: 10.1016/j.immuni.2020.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuschei W.M., Leitner J., Majdic O., Pickl W.F., Zlabinger G.J., Grabmeier-Pfistershammer K., et al. Costimulatory signals potently modulate the T cell inhibitory capacity of the therapeutic CD11a antibody efalizumab. Clin Immunol. 2011;139:199–207. doi: 10.1016/j.clim.2011.02.007. [DOI] [PubMed] [Google Scholar]
- Laidlaw B.J., Gray E.E., Zhang Y., Ramírez-Valle F., Cyster J.G. Sphingosine-1-phosphate receptor 2 restrains egress of γδ T cells from the skin. J Exp Med. 2019;216:1487–1496. doi: 10.1084/jem.20190114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lämmermann T., Bader B.L., Monkley S.J., Worbs T., Wedlich-Söldner R., Hirsch K., et al. Rapid leukocyte migration by integrin-independent flowing and squeezing. Nature. 2008;453:51–55. doi: 10.1038/nature06887. [DOI] [PubMed] [Google Scholar]
- Lebwohl M., Tyring S.K., Hamilton T.K., Toth D., Glazer S., Tawfik N.H., et al. A novel targeted T-cell modulator, efalizumab, for plaque psoriasis. N Engl J Med. 2003;349:2004–2013. doi: 10.1056/NEJMoa030002. [DOI] [PubMed] [Google Scholar]
- Ledgerwood L.G., Lal G., Zhang N., Garin A., Esses S.J., Ginhoux F., et al. The sphingosine 1-phosphate receptor 1 causes tissue retention by inhibiting the entry of peripheral tissue T lymphocytes into afferent lymphatics. Nat Immunol. 2008;9:42–53. doi: 10.1038/ni1534. [DOI] [PubMed] [Google Scholar]
- Ley K., Kansas G.S. Selectins in T-cell recruitment to non-lymphoid tissues and sites of inflammation. Nat Rev Immunol. 2004;4:325–335. doi: 10.1038/nri1351. [DOI] [PubMed] [Google Scholar]
- Ley K., Rivera-Nieves J., Sandborn W.J., Shattil S. Integrin-based therapeutics: biological basis, clinical use and new drugs. Nat Rev Drug Discov. 2016;15:173–183. doi: 10.1038/nrd.2015.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J., Chang H.W., Huang Z.M., Nakamura M., Sekhon S., Ahn R., et al. Single-cell RNA sequencing of psoriatic skin identifies pathogenic Tc17 cell subsets and reveals distinctions between CD8+ T cells in autoimmunity and cancer [e-pub ahead of print] J Allergy Clin Immunol. 2020 doi: 10.1016/j.jaci.2020.11.028. (accessed 24 Feburary 2021) [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacKay L.K., Rahimpour A., Ma J.Z., Collins N., Stock A.T., Hafon M.L., et al. The developmental pathway for CD103(+)CD8+ tissue-resident memory T cells of skin. Nat Immunol. 2013;14:1294–1301. doi: 10.1038/ni.2744. [DOI] [PubMed] [Google Scholar]
- Mahuron K.M., Moreau J.M., Glasgow J.E., Boda D.P., Pauli M.L., Gouirand V., et al. Layilin augments integrin activation to promote antitumor immunity. J Exp Med. 2020;217 doi: 10.1084/jem.20192080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mancuso R.V., Casper J., Schmidt A.G., Krähenbühl S., Weitz-Schmidt G. Anti-αLβ2 antibodies reveal novel endocytotic cross-modulatory functionality. Br J Pharmacol. 2020;177:2696–2711. doi: 10.1111/bph.14996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Margraf A., Ley K., Zarbock A. Neutrophil recruitment: from model systems to tissue-specific patterns. Trends Immunol. 2019;40:613–634. doi: 10.1016/j.it.2019.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milch C., Wyant T., Xu J., Parikh A., Kent W., Fox I., et al. Vedolizumab, a monoclonal antibody to the gut homing α4β7 integrin, does not affect cerebrospinal fluid T-lymphocyte immunophenotype. J Neuroimmunol. 2013;264:123–126. doi: 10.1016/j.jneuroim.2013.08.011. [DOI] [PubMed] [Google Scholar]
- Nácher M., Blázquez A.B., Shao B., Matesanz A., Prophete C., Berin M.C., et al. Physiological contribution of CD44 as a ligand for E-selectin during inflammatory T-cell recruitment. Am J Pathol. 2011;178:2437–2446. doi: 10.1016/j.ajpath.2011.01.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nandi A., Estess P., Siegelman M. Bimolecular complex between rolling and firm adhesion receptors required for cell arrest; CD44 association with VLA-4 in T cell extravasation. Immunity. 2004;20:455–465. doi: 10.1016/s1074-7613(04)00077-9. [DOI] [PubMed] [Google Scholar]
- Navarini A.A., Kerl K., French L.E., Trüeb R.M. Control of widespread hypertrophic lupus erythematosus with T-cell-directed biologic efalizumab. Dermatology. 2010;220:249–253. doi: 10.1159/000277614. [DOI] [PubMed] [Google Scholar]
- Nosbaum A., Prevel N., Truong H.A., Mehta P., Ettinger M., Scharschmidt T.C., et al. Cutting Edge: Regulatory T cells facilitate cutaneous wound healing. J Immunol. 2016;196:2010–2014. doi: 10.4049/jimmunol.1502139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nussbaum J.C., Van Dyken S.J., von Moltke J., Cheng L.E., Mohapatra A., Molofsky A.B., et al. Type 2 innate lymphoid cells control eosinophil homeostasis. Nature. 2013;502:245–248. doi: 10.1038/nature12526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oakford M.E., Dixon S.V., August S., Pickard C., Ardern-Jones M., Lackie P., et al. Migration of immunocytes across the basement membrane in skin: the role of basement membrane pores. J Invest Dermatol. 2011;131:1950–1953. doi: 10.1038/jid.2011.146. [DOI] [PubMed] [Google Scholar]
- Orlik C., Deibel D., Küblbeck J., Balta E., Ganskih S., Habicht J., et al. Keratinocytes costimulate naive human T cells via CD2: a potential target to prevent the development of proinflammatory Th1 cells in the skin. Cell Mol Immunol. 2020;17:380–394. doi: 10.1038/s41423-019-0261-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Overstreet M.G., Gaylo A., Angermann B.R., Hughson A., Hyun Y.M., Lambert K., et al. Inflammation-induced interstitial migration of effector CD4+ T cells is dependent on integrin αV. Nat Immunol. 2013;14:949–958. doi: 10.1038/ni.2682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piao W., Xiong Y., Famulski K., Brinkman C.C., Li L., Toney N., et al. Regulation of T cell afferent lymphatic migration by targeting LTβR-mediated non-classical NFκB signaling. Nat Commun. 2018;9:3020. doi: 10.1038/s41467-018-05412-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pugashetti R., Koo J. Efalizumab discontinuation: a practical strategy. J Dermatolog Treat. 2009;20:132–136. doi: 10.1080/09546630902984596. [DOI] [PubMed] [Google Scholar]
- Sanchez Rodriguez R., Pauli M.L., Neuhaus I.M., Yu S.S., Arron S.T., Harris H.W., et al. Memory regulatory T cells reside in human skin. J Clin Invest. 2014;124:1027–1036. doi: 10.1172/JCI72932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandborn W.J., Lee S.D., Tarabar D., Louis E., Klopocka M., Klaus J., et al. Phase II evaluation of anti-MAdCAM antibody PF-00547659 in the treatment of Crohn’s disease: report of the OPERA study. Gut. 2018;67:1824–1835. doi: 10.1136/gutjnl-2016-313457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandborn W.J., Vermeire S., Tyrrell H., Hassanali A., Lacey S., Tole S., et al. Etrolizumab for the treatment of ulcerative colitis and Crohn’s disease: an overview of the Phase 3 Clinical Program. Adv Ther. 2020;37:3417–3431. doi: 10.1007/s12325-020-01366-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlickum S., Sennefelder H., Friedrich M., Harms G., Lohse M.J., Kilshaw P., et al. Integrin αE(CD103)β7 influences cellular shape and motility in a ligand-dependent fashion. Blood. 2008;112:619–625. doi: 10.1182/blood-2008-01-134833. [DOI] [PubMed] [Google Scholar]
- Schön M.P., Schön M., Warren H.B., Donohue J.P., Parker C.M. Cutaneous inflammatory disorder in integrin alphaE (CD103)-deficient mice. J Immunol. 2000;165:6583–6589. doi: 10.4049/jimmunol.165.11.6583. [DOI] [PubMed] [Google Scholar]
- Schön M.P., Zollner T.M., Boehncke W.H. The molecular basis of lymphocyte recruitment to the skin: clues for pathogenesis and selective therapies of inflammatory disorders. J Invest Dermatol. 2003;121:951–962. doi: 10.1046/j.1523-1747.2003.12563.x. [DOI] [PubMed] [Google Scholar]
- Shen S., O’Brien T., Yap L.M., Prince H.M., McCormack C.J. The use of methotrexate in dermatology: a review. Australas J Dermatol. 2012;53:1–18. doi: 10.1111/j.1440-0960.2011.00839.x. [DOI] [PubMed] [Google Scholar]
- Shields B.D., Koss B., Taylor E.M., Storey A.J., West K.L., Byrum S.D., et al. Loss of E-cadherin inhibits CD103 antitumor activity and reduces checkpoint blockade responsiveness in melanoma. Cancer Res. 2019;79:1113–1123. doi: 10.1158/0008-5472.CAN-18-1722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siegelman M.H., Stanescu D., Estess P. The CD44-initiated pathway of T-cell extravasation uses VLA-4 but not LFA-1 for firm adhesion. J Clin Invest. 2000;105:683–691. doi: 10.1172/JCI8692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sigmundsdottir H., Johnston A., Gudjonsson J.E., Bjarnason B., Valdimarsson H. Methotrexate markedly reduces the expression of vascular E-selectin, cutaneous lymphocyte-associated antigen and the numbers of mononuclear leucocytes in psoriatic skin. Exp Dermatol. 2004;13:426–434. doi: 10.1111/j.0906-6705.2004.00177.x. [DOI] [PubMed] [Google Scholar]
- Singh K., Gatzka M., Peters T., Borkner L., Hainzl A., Wang H., et al. Reduced CD18 Levels Drive Regulatory T cell Conversion into Th17 Cells in the CD18 hypo PL/J Mouse Model of Psoriasis. J Immunol. 2013;190:2544–2553. doi: 10.4049/jimmunol.1202399. [DOI] [PubMed] [Google Scholar]
- Sivasankar S. Tuning the kinetics of cadherin adhesion. J Invest Dermatol. 2013;133:2318–2323. doi: 10.1038/jid.2013.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soler D., Chapman T., Yang L.L., Wyant T., Egan R., Fedyk E.R. The binding specificity and selective antagonism of vedolizumab, an anti-alpha4beta7 integrin therapeutic antibody in development for inflammatory bowel diseases. J Pharmacol Exp Ther. 2009;330:864–875. doi: 10.1124/jpet.109.153973. [DOI] [PubMed] [Google Scholar]
- Sorokin L. The impact of the extracellular matrix on inflammation. Nat Rev Immunol. 2010;10:712–723. doi: 10.1038/nri2852. [DOI] [PubMed] [Google Scholar]
- Stummvoll G.H., Aringer M., Grisar J., Steiner C.W., Smolen J.S., Knobler R., et al. Increased transendothelial migration of scleroderma lymphocytes. Ann Rheum Dis. 2004;63:569–574. doi: 10.1136/ard.2002.004838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sumagin R., Sarelius I.H. Intercellular adhesion molecule-1 enrichment near tricellular endothelial junctions is preferentially associated with leukocyte transmigration and signals for reorganization of these junctions to accommodate leukocyte passage. J Immunol. 2010;184:5242–5252. doi: 10.4049/jimmunol.0903319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun H., Lagarrigue F., Wang H., Fan Z., Lopez-Ramirez M.A., Chang J.T., et al. Distinct integrin activation pathways for effector and regulatory T cell trafficking and function. J Exp Med. 2021;218 doi: 10.1084/jem.20201524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun Z., Costell M., Fässler R. Integrin activation by talin, kindlin and mechanical forces. Nat Cell Biol. 2019;21:25–31. doi: 10.1038/s41556-018-0234-9. [DOI] [PubMed] [Google Scholar]
- Szabo P.A., Miron M., Farber D.L. Location, location, location: tissue resident memory T cells in mice and humans. Sci Immunol. 2019;4:1–12. doi: 10.1126/sciimmunol.aas9673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takiguchi R., Tofte S., Simpson B., Harper E., Blauvelt A., Hanifin J., et al. Efalizumab for severe atopic dermatitis: a pilot study in adults. J Am Acad Dermatol. 2007;56:222–227. doi: 10.1016/j.jaad.2006.08.031. [DOI] [PubMed] [Google Scholar]
- Teijeira A., Hunter M.C., Russo E., Proulx S.T., Frei T., Debes G.F., et al. T cell migration from inflamed skin to draining lymph nodes requires intralymphatic crawling supported by ICAM-1/LFA-1 interactions. Cell Rep. 2017;18:857–865. doi: 10.1016/j.celrep.2016.12.078. [DOI] [PubMed] [Google Scholar]
- Thelen F., Witherden D.A. Get in touch with dendritic epithelial T cells! Front Immunol. 2020;11:1656. doi: 10.3389/fimmu.2020.01656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varga G., Nippe N., Balkow S., Peters T., Wild M.K., Seeliger S., et al. LFA-1 contributes to signal i of T-cell activation and to the production of T(h)1 cytokines. J Invest Dermatol. 2010;130:1005–1012. doi: 10.1038/jid.2009.398. [DOI] [PubMed] [Google Scholar]
- Verma N.K., Fazil M.H.U.T., Ong S.T., Chalasani M.L.S., Low J.H., Kottaiswamy A., et al. LFA-1/ICAM-1 ligation in human T cells promotes Th1 polarization through a GSK3β signaling–dependent Notch pathway. J Immunol. 2016;197:108–118. doi: 10.4049/jimmunol.1501264. [DOI] [PubMed] [Google Scholar]
- Wang H., Peters T., Sindrilaru A., Kess D., Oreshkova T., Yu X.Z., et al. TGF-β-dependent suppressive function of Tregs requires wild-type levels of CD18 in a mouse model of psoriasis. J Clin Invest. 2008;118:2629–2639. doi: 10.1172/JCI34916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y., Shu Y., Xiao Y.F., Wang Q., Kanekura T., Li Y., et al. Hypomethylation and overexpression of ITGAL (CD11a) in CD4(+) T cells in systemic sclerosis. Clin Epigenetics. 2014;6:25. doi: 10.1186/1868-7083-6-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weninger W., Biro M., Jain R. Leukocyte migration in the interstitial space of non-lymphoid organs. Nat Rev Immunol. 2014;14:232–246. doi: 10.1038/nri3641. [DOI] [PubMed] [Google Scholar]
- Zaid A., Hor J.L., Christo S.N., Groom J.R., Heath W.R., Mackay L.K., et al. Chemokine receptor–dependent control of skin tissue–resident memory T cell formation. J Immunol. 2017;199:2451–2459. doi: 10.4049/jimmunol.1700571. [DOI] [PubMed] [Google Scholar]