Abstract
To compare the effects of two nap opportunities (20 and 90 min) to countermeasure the transient naturally occurring increased sleepiness and decreased performances during the post-lunch dip (PLD). Fourteen highly trained judokas completed in a counterbalanced and randomized order three test sessions (control (No-nap), 20- (N20) and 90-min (N90) nap opportunities). Test sessions consisted of the running-based anaerobic sprint test (RAST), simple and multiple-choice reaction times (MCRT) and the Epworth sleepiness scale (ESS). From the RAST, the maximum (Pmax), mean (Pmean) and minimum (Pmin) powers were calculated. Blood samples were taken before and after the RAST to measure the effect of pre-exercise napping on energetic and muscle damage biomarkers and antioxidant defense. N20 increased Pmax and Pmean compared to No-nap (p < 0.001, d = 0.59; d = 0.66) and N90 (p < 0.001, d = 0.98; d = 0.72), respectively. Besides, plasma lactate and creatinine increased only when the exercise was performed after N20. Both N20 (p < 0.001, d = 1.18) and N90 (p < 0.01, d = 0.78) enhanced post-exercise superoxide dismutase activity compared to No-nap. However, only N20 enhanced post-exercise glutathione peroxidase activity (p < 0.001, d = 1.01) compared to pre-nap. Further, MCRT performance was higher after N20 compared to No-nap and N90 (p < 0.001, d = 1.15; d = 0.81, respectively). Subjective sleepiness was lower after N20 compared to No-nap (p < 0.05, d = 0.92) and N90 (p < 0.01, d = 0.89). The opportunity to nap for 20 min in the PLD enhanced RAST, MCRT performances, and antioxidant defense, and decreased sleepiness. However, the opportunity of 90 min nap was associated with decreased repeated sprint performances and increased sleepiness, probably because of the sleep inertia.
Keywords: Midday sleep, Short-term Exercise, Biomarkers of Muscle Damage, Antioxidant Status, Ergogenic aid, Cognitive Performance
INTRODUCTION
Human physical and cognitive performances are not stable throughout the waking day. Most performances (e.g., attention, short-term high-intensity exercise, etc.) fluctuate between a bathyphase in the morning and an acrophase in the afternoon [1]. However, for some individuals and some variables, a dip in the early afternoon is observed [2, 3]. The post-lunch dip (PLD) is the time of day where sleepiness increases and attention falls, which make it a suitable time for rest rather than activity [3]. Although its name is linked with lunch, which could be somehow misleading, the PLD is a part of human biological rhythms and it still occurs even without having lunch and/or under constant routine [2]. However, this is subject to an interpersonal variability, where some people could be more affected than others.
Elite athletes are recognized for having poorer sleep quality and quantity than the general population [4]. This is due to the higher need for recovery, the stress of competitions, and for some of them to the higher frequency of traveling [5]. As sleep is the major mean for athletes’ recovery [5], strategies such as nocturnal sleep extension and napping could benefit exercise performance. Horne et al. [6] reported that 90 min nocturnal sleep extension had little effects compared to 15–20 min nap on PLD’s sleepiness and psychomotor vigilance. Consequently, a huge number of studies focused on the effects of napping on physical performances in the last few years, though, reporting conflicting results [1, 7–13]. Petit et al. [7] reported a neutral effect of napping on Wingate test performance in physical education students after normal sleep night (NSN) and after simulated jet lag. Likewise, Suppiah et al. [9] showed that 20 min nap after NSN had no effect on sprinting performance but declined shooting performance in highly trained adolescent athletes. Studies that failed to report beneficial effects of a short nap on physical performances, suggested that a longer nap might be more beneficial [7, 9]. Indeed, it has been reported that 45 min nap opportunity after NSN enhanced 5 jump test [12] and 5 m shuttle run [13] performances more than 25 or 35 min nap opportunities. Also, it was recently shown that 90 min nap opportunity after partial sleep deprivation (PSD) was more efficient than 20 min nap in enhancing repeated sprint performance [11]. However, there is no information on the effects of a relatively longer nap (e.g., 90 min) on repeated sprint performances after NSN. It has been suggested that the long nap produces more enhancing effects because it contains slow wave sleep (SWS) [14]. While the amount of SWS in a long nap is associated with a higher recuperative value [14], it is also associated with the severity of sleep inertia [15]. Sleep inertia refers to the drowsy feeling and the transient impaired cognitive performance immediately at the awakening [15–17]. Surprisingly, Wertz et al. [18] showed that the effect of sleep inertia could be more severe than 26 h of sleep deprivation on a short-term memory task. The intensity and the duration of sleep inertia depend on the time of the day, the awakening duration before the nap, and also the duration of SWS during the nap [15–17]. It has been shown that sleep inertia lasts between 10–15 min after a 50 min nap [19] and no more than 15 min after napping for 60 min or less [20].
On the other hand, it was reported that repeated sprint exercise caused an increase in biomarkers of muscle and cardiac damage and a lower antioxidant defense after NSN [21] and after PSD [22]. Interestingly, napping was associated with lower levels of biomarkers of muscle damage and higher antioxidant defense after PSD [11]. However, to our knowledge, there is no existing information on the effect of napping after NSN on the exercise induced increase in muscle damage and oxidative stress. Therefore, we aimed to evaluate the effects of two naps lengths during the PLD on sleepiness, biochemical responses and reaction time to repeated sprint exercise. We hypothesize that napping will enhance repeated sprint and reaction time performances and reduce sleepiness and biomarkers of muscle damage. Further, we expect that the longer nap opportunity will result in better enhancement than the short one.
MATERIALS AND METHODS
Ethics
The present study was approved by the local Institutional Review Board (P-SC N° 009/15) and conducted according to the ethical guidelines of the Declaration of Helsinki (64th World Medical Association General Assembly, Fortaleza, Brazil, October 2013). All participants provided an informed consent form after being informed about the study design and risks. They were also informed about their right to withdraw from the study without any penalty.
Participants
The sample size was a priori calculated using the G*power software [23], and following the suggested procedure by Beck [24]. The probability of type I (p ≤ 0.05) and Type II (1 - β ≥ 0.95) errors were both fixed at 0.05. Based on an earlier study with a similar paradigm [22], the smallest Cohen’s d (i.e., Pmax) was retained (d = 0.54). Further, the lowest correlation between repeated measure was retained (GPx; r = 0.44) [22]. The G*power software indicated a minimal required sample size of 12 participants. With the abovementioned considerations, the actual power of the study design was 0.96.
At first, twenty volunteers were screened, from them, sixteen were involved and completed the protocol. It was expected that not all the participants will finish the protocol appropriately [11]. Indeed, only fourteen participants’ data are included in the statistical analysis. They were all good sleepers, non-habitual nappers, non-smokers and free of drugs (20.43 ± 1.22 years, 174.86 ± 8.77 cm, 73.07 ± 11.72 kg, BMI = 23.85 ± 3.12 kg·m-2). They were all major males, highly trained judokas (at least 1st Dan black-belt), regularly engaged in at least ~2 hours/day, 5 days/week of training for at least 5 years and competing at the international level. All of them were moderate or intermediate chronotype (scored between 31 and 69) according to the Horne and Östberg morningness/eveningness questionnaire [25]. Participants provided their last month’s routine sleep schedule and participants who scored > 5 according to the Pittsburg Sleep Quality Index [26] were excluded.
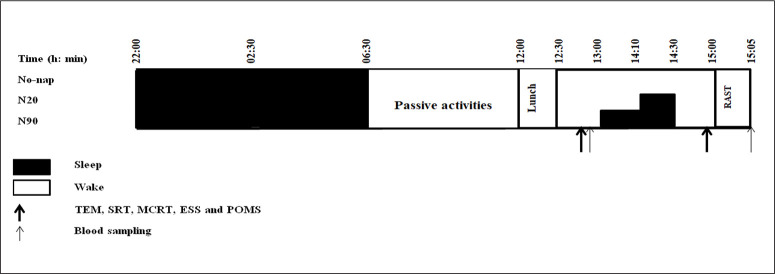
Experimental design (Figure 1)
FIG. 1.
Simplified experimental protocol. N20: 20 min nap opportunity, N90: 90 min nap opportunity, RAST; the running-based anaerobic sprint test, h; hour, min; minute, TEM: temperature, SRT; Simple Reaction Time, MCRT; Multiple Choice Reaction Time, ESS; Epworth Sleepiness Scale and POMS; the Profile of Mood State.
Before starting the main experiment, participants underwent two habitational sessions. In which, they were familiarized with the experimenters, laboratory, used material/devices, the sleeping room, tests, and questionnaires.
Three test sessions after normal sleep night (NSN) were accomplished at least one week apart in a counterbalanced and randomized order (No-nap, ~20 min (N20) and ~90 min (N90) nap opportunities). During different protocol sessions, participants showed-up at the laboratory at ~20h00, consumed a standardized dinner at ~20h30. After that, they were free to watch television, play videogames or surf on the internet, until 22h00 when they were sent to bed to fall asleep (all lights and devices off). Participants were aroused at 06h30 (~08h30 of time in bed “TIB”), which corresponds to their daily routines. After a qualitatively and quantitatively standardized breakfast at 07h00, they stayed awake until 12h00 doing the same passive activities as in the previous evening (no food allowed, drinking water ad libitum). The Iso-caloric lunch at 12h00 was followed by 40 min of rest. After this, pre-nap blood samples were collected and oral temperature (TEM; micro life, France with precision ~0.05°C) was recorded. Then, simple (SRT) and multiple-choice (MCRT) reaction time, the Profile of Mood State (POMS) and the Epworth Sleepiness Scale (ESS) were administered. In the N90 condition, participants entered the sleeping room (i.e., comfortable temperature, entirely dark and quiet) and went to bed at ~12h50. They were free to use a blanket. After being given ~10 min to get used to their surroundings, they wore earplugs and eye-masks and the 90 min naptime started (from ~13h00 to ~14h30). During the N20 condition, participants were watching a neutral documentary between 12h50 and 14h00. They were sitting on comfortable armchairs and got in the napping room at ~14h00, acclimatized for ~10 min, then wore earplugs and eye-masks and the 20 min naptime started (from ~14h10 to ~14h30). Participants’ activities was monitored by an infrared camera connected to the experimenter’s computer. At awakening, participants subjectively rated their sleep quality during the nap on a 100 mm analog scale; ranging from 0 “no sleep at all” to 100 “deep, uninterrupted sleep”. Then, a period of ~30 min separated wake-up from testing to overcome any sleep inertia that might have existed [1, 11]. After that, TEM, SRT, and MCRT were recorded and questionnaires were filled at ~14h50 in the same pre-nap/rest order. Participants spent the period between 13h00 and 15h00 during the No-nap condition watching a neutral documentary, in semi-recumbent position. Afterwards, each participant performed a 5 min warm-up followed by a recovery period of 3 min. Finally, the Running-based anaerobic sprint test (RAST) started at ~15h00, followed by a blood sampling after 5 min of post-exercise passive recovery.
The laboratory conditions were fixed during all experimental days; temperature ~25°C (± 1.8°C), humidity ~35% (± 3.2%), and luminosity (i) ~2000 lux during tests, and (ii) < 5 lux during sleep.
Protocols The Running-based Anaerobic Sprint Test (RAST)
RAST (six 35 m straight-line sprints with 10 s recovery in between for the turnaround) was performed [27].
Simple and multiple-choice reaction times
Participants performed simple (SRT) and multiple-choice (MCRT) reaction times using REACT V0.9 software (Claude Bernard Lyon 1 University, Lyon, France).
Profile of Mood State (POMS)
POMS standard validated psychological test formulated by McNair [28] was administered.
Epworth Sleepiness Scale (ESS)
ESS score defines subjective daytime sleepiness. Its score was correlated with Multiple-Sleep Latency Tests during overnight polysomnography [29].
Rating of perceived exertion (RPE)
The CR-10 psycho-physiological scale given score assessed the exertion which the athlete experience during the exercise [30].
Blood sampling and analysis
Blood samples were collected and analyzed according to Romdhani et al [11, 22]. Table 1 presents all the methods used in the sample analysis.
TABLE 1.
Different methods used in blood analysis
| Biochemical variables | Method |
|---|---|
| Plasma Lactate [La] | Lactate oxidase peroxidase method (intra and inter-assay CV were: 0.91% and 1.9%, respectively) |
| Plasma Glucose (GLC) | Glucose hexokinase method (intra and inter-assay CVs were: 0.94% and 1.31%, respectively) |
| Creatinine (CRE) | Colorimetric (intra and inter-assay CV were 2.75% and 2.93%, respectively). |
| Urea (URE) | Kinetic enzymatic method (intra and inter-assay CV were: 0.33% and 5.66%, respectively). |
| Lactate Dehydrogenase (LDH) | NADH consumption method (intra and inter-assay CV were: 3.72% and 4.31%, respectively). |
| Glutathione Peroxidase (GPx) | Spectrophotometric method based on Paglia and Valentine method (1967; with kit from Randox Lab; Ransel RS. 505). The intra and inter assays CV% were: 7.31 and 4.86, respectively. |
| Superoxide Dismutase (SOD) | SOD activity in erythrocytes was measured by the rate of inhibition of 2-(4-iodophenyl)-3-(4-nitrophenol)-5-phenyltetrazolium chloride (INT) reduction. The kit used in this method was from Randox Lab (Ransod, RX MONZA). 0.5 ml of whole blood was centrifuged and then separated from the plasma. Erythrocytes were washed four times with 3 ml of 0.9% NaCl solution and centrifuged after each wash. 2.0 ml with cold redistilled water was added to the resulting erythrocytes, mixed and left to stand at +4°C for 15 minutes. A 25 fold dilution of lysate was then added. The intra and inter assays CV% were: 5.96 and 4.64, respectively. |
Statistical analyses
The statistical tests were processed using GraphPad Prism 6 (GraphPad Software, San Diego, CA, USA). The Shapiro-Wilks revealed that data were normally distributed. Hence, parametric tests were used. For RAST and [La], a One-Way analysis of variance (ANOVA) with repeated measures (3 napping conditions) was used. For TEM, SRT, MCRT, and self-administered questionnaires, Two-Way repeated measures ANOVA was used (before/after the nap/rest × 3 napping conditions). For biochemical parameters, Two-Way repeated measures ANOVA was used (before the nap/after the exercise × 3 napping conditions). To assess the ANOVA practical significance, Eta-squared (η2) was calculated. Once the ANOVA indicated a significant main effect or interaction effect, the Bonferroni post-hoc test was used to check differences. Furthermore, the Effect Size (d) was calculated according to Cohen [31] to determine the amplitude of the difference in pairwise comparisons. The magnitude of d was classified as small (0.2 < d < 0.5), moderate (0.5 < d < 0.8) and large (d ≥ 0.8). Further, Mean Difference (MD) and the 95% Confidence Interval (95% CI) were provided for pairwise comparison. All values within the text, figures, and tables are reported as mean ± standard deviation (SD). The level of significance was set at p < 0.05.
RESULTS
Sleep quality during the nap
Participants rated their sleep quality during the nap immediately at the awakening. The one-way ANOVA showed a significant effect of napping (F(2,13) = 60.48, p < 0.001, η2 = 0.82). Sleep quality was higher after N20 (p < 0.001, d = 3.21, MD = -3.71, 95% CI = -4.81 to -2.62) and N90 (p < 0.001, d = 5.43, MD = -5.01, 95% CI = -5.95 to -4.04) compared to No-nap, with no difference between N20 and N90.
The running-based anaerobic sprint test and Plasma lactate
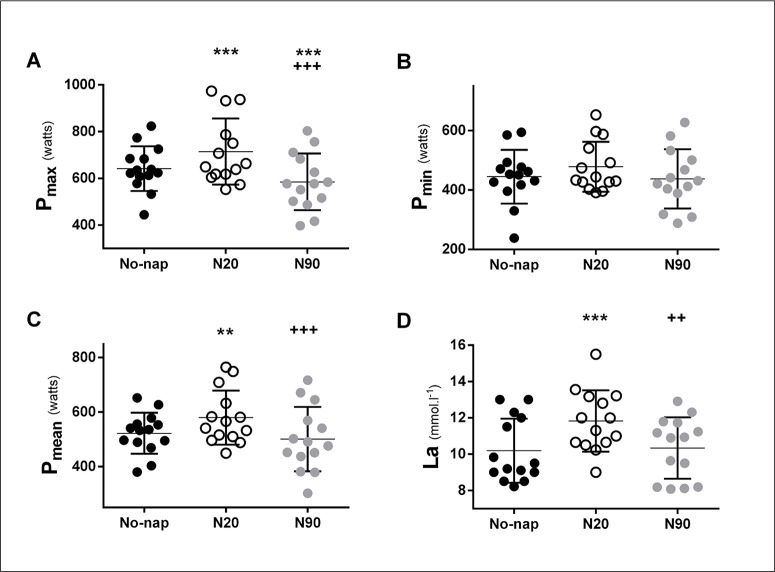
Pmax, Pmean and [La] were higher after N20 compared to No-nap and N90. However, Pmax was lower after N90 compared to No-nap. Pmax, Pmean and [La]’s ANOVA and pairwise comparison are presented in Figure 2 and Table 2a. Pmin, RPE and FI were unchanged across different nap conditions.
FIG. 2.
Individual maximum (Pmax; A), minimum (Pmin; B) and mean (Pmean; C) powers, and [La]: Plasma lactate (D) after control (No-nap ●), 20 min nap (N20 ○) and 90 min nap (N90 ●). Significance is assessed with a one-way ANOVA and Bonferroni multiple comparison tests. *, **and *** presents a significant difference in comparison with No-nap values at p < 0.05, p < 0.01 and p < 0.001, respectively; +, ++ and +++ presents a significant difference in comparison with N20 values at p < 0.05, p < 0.01 and p < 0.001, respectively. Horizontal and vertical bars represent the group means and standard deviations, respectively.
TABLE 2a.
RAST and [La]’s ANOVA output and pairwise comparison
| ANOVA (One-way) | N20 vs No-Nap | N90 vs No-Nap | N20 vs N90 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| F(2, 13) | p | η2 | p | d | MD | 95% CI | p | d | MD | 95% CI | p | d | MD | 95% CI | |
| Pmax (W) | 34.4 | < 0.001 | 0.78 | < 0.001 | 0.59 | -72.9 | -120 to -25.4 | < 0.001 | 0.52 | 56.9 | 30.3 to 83.4 | < 0.001 | 0.98 | 130 | 98.9 to 161 |
| Pmean (W) | 14.7 | < 0.001 | 0.65 | < 0.01 | 0.66 | -57.7 | -106 to -9.44 | NS | 0.21 | 21.1 | -25.8 to 67.9 | < 0.001 | 0.72 | 78.8 | 44.9 to 113 |
| [La] (mmol·l-1) | 8.48 | < 0.001 | 0.54 | < 0.001 | 0.92 | -1.64 | -2.87 to -0.39 | NS | 0.05 | -0.14 | -1.54 to 1.25 | < 0.01 | 0.88 | 1.49 | 0.27 to 2.71 |
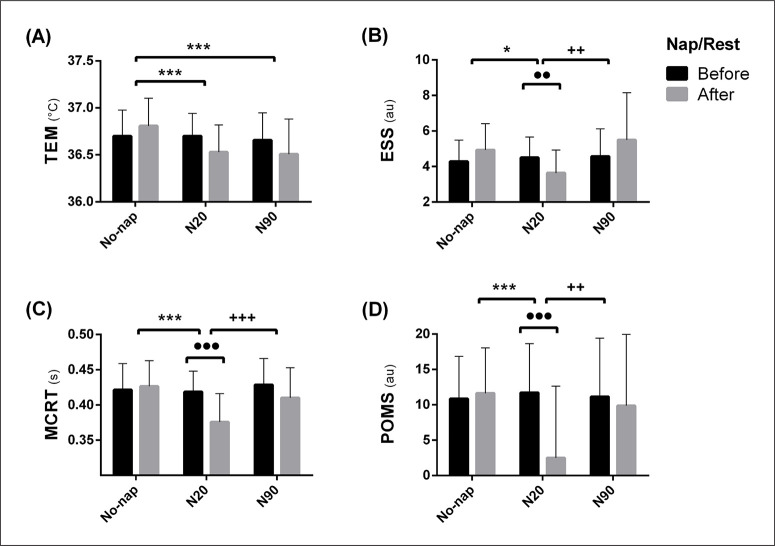
Oral temperature and psycho-cognitive parameters
There was no difference between pre- and post-nap during all conditions in TEM. However, TEM was lower after N20 and N90 compared to No-nap. MCRT was lower after N20 compared to pre-nap (p < 0.001, d = 1.25, MD = 0.04 s, 95% CI = 0.02 to 0.06 s) indicating a better performance. Also, MCRT was lower after N20 compared to No-nap and N90. The number of errors during MCRT was lower after N20 (p < 0.001, d = 1.31, MD = 1.36, 95% CI = 0.29 to 2.27) and N90 (p < 0.001, d = 1.03, MD = 1.14, 95% CI = 0.08 to 2.21) compared to pre-nap. ESS scores decreased after N20 compared to pre-nap (p < 0.01, d = 0.71, MD = 0.86, 95% CI = -0.59 to 2.23) indicating a lower sleepiness. Further, ESS (SOD: D). For legend, please see Fig. 3. scores were significantly lower after N20 compared to No-nap and N90. N20 decreased POMS score compared to pre-nap (p < 0.001, d = 1.05, MD = 9.21, 95% CI = 2.41 to 16.01), indicating a better mood. The SRT was unaffected by neither napping nor the timing of testing. TEM, POMS, ESS and MCRT’s ANOVA and pairwise comparison are presented in Figure 3 and Table 2b.
FIG. 3.
Means ± SD of Temperature (TEM; A), Epworth Sleepiness Scale (ESS; B), Multiple Choice Reaction Time (MCRT; C) and the Profile of Mood State (POMS; D), before and after the nap/rest during different protocol conditions; (No-nap), 20 min nap opportunity (N20) and 90 min nap opportunity (N90). Significance is assessed with the Two-way repeated measure ANOVA and Bonferroni post hoc tests. ●, ●● and ●●● presents a significant difference in comparison with pre-nap/rest values at p < 0.05, p < 0.01 and p < 0.001 respectively; *, **and *** presents a significant difference in comparison with No-nap values at p < 0.05, p < 0.01 and p < 0.001 respectively; +, ++ and +++ presents a significant difference in comparison with N20 values at p < 0.05, p < 0.01 and p < 0.001 respectively. s; second, °C: Celsius, au; arbitrary unit.
TABLE 2b.
TEM and psycho-cognitive parameters’ ANOVA output and pairwise comparison
| ANOVA (interaction) | N20 vs No-Nap | N90 vs No-Nap | N20 vs N90 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| F(2, 26) | p | η2 | p | d | MD | 95% CI | p | d | MD | 95% CI | p | d | MD | 95% CI | |
| TEM (C°) | 4.09 | < 0.01 | 0.41 | < 0.001 | 0.81 | 0.27 | 0.08 to 0.47 | < 0.001 | 0.92 | 0.31 | 0.11 to 0.49 | NS | 0.03 | 0.02 | -0.17 to 0.21 |
| MCRT (s) | 8.77 | < 0.001 | 0.52 | < 0.001 | 1.15 | 0.05 | 0.03 to 0.07 | NS | 0.37 | 0.01 | -0.01 to 0.03 | < 0.001 | 0.81 | -0.03 | -0.05 to -0.01 |
| POMS (au) | 10.4 | < 0.01 | 0.34 | < 0.001 | 1.07 | 9.14 | 2.33 to 15.91 | NS | 0.21 | -0.28 | -7.09 to 6.52 | < 0.01 | 0.72 | -7.36 | -14.2 to -0.51 |
| ESS (au) | 3.59 | < 0.05 | 0.37 | < 0.05 | 0.92 | 1.29 | -0.08 to 2.66 | NS | 0.26 | -0.57 | -1.95 to 0.81 | < 0.01 | 0.89 | -1.86 | -3.23 to -0.48 |
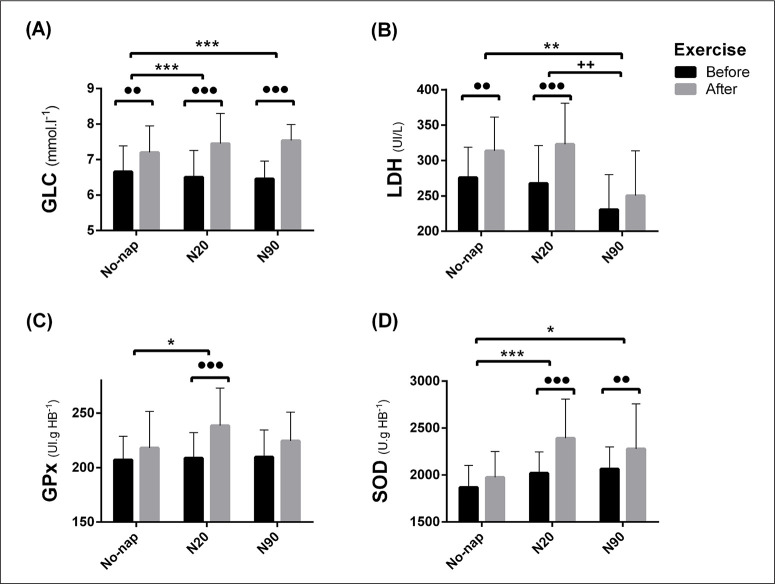
Biochemical parameters
Post-exercise urea (URE) increased only after No-nap compared to pre-exercise (p < 0.001, d = 0.65, MD = -0.53 mmol·l-1, 95% CI = -0.95 to -0.12 mmol·l-1) and both N20 and N90 blunted this increase. Post-exercise lactate dehydrogenase (LDH) was lower after N90 compared to No-nap and N20 (Figure 4 and Table 2c). Post-exercise glutathione peroxidase (GPx) increased only after N20 (p < 0.001, d = 1.01, MD = -29.8 UI.gHB-1, 95% CI = -50.1 to -9.4 UI.gHB-1) compared to pre-nap. Both N20 and N90 enhanced post-exercise superoxide dismutase (SOD) compared to No-nap (Figure 4 and Table 2c). Post-exercise plasma glucose (GLC) increased regardless of nap condition. However, this increase was more important after N20 and N90 than No-nap (Figure 4 and Table 2c). Post-exercise Creatinine (CRE) increased significantly only after N20 (p < 0.001, d = 2.03, MD = -30.9 mmol·l-1, 95% CI = -46.4 to -15.4 mmol·l-1) compared to pre-nap.
FIG. 4.
Plasma Glucose (GLC: A), lactate dehydrogenase (LDH: B), Glutathione Peroxidase (GPx: C) and Superoxide Dismutase (SOD: D). For legend, please see Fig. 3.
TABLE 2c.
Biochemical parameters’ ANOVA output and pairwise comparison
| ANOVA (interaction) | N20 vs No-Nap | N90 vs No-Nap | N20 vs N90 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| F(2, 26) | p | η2 | p | d | MD | 95% CI | p | d | MD | 95% CI | p | d | MD | 95% CI | |
| LDH (UI/l-1) | 5.53 | < 0.01 | 0.38 | NS | 0.17 | -9.21 | -45.1 to 26.5 | < 0.001 | 1.13 | 63.3 | 27.5 to 99.3 | < 0.001 | 1.19 | 72.5 | 36.7 to 108.3 |
| SOD (U.gHB-1) | 5.19 | < 0.05 | 0.32 | < 0.001 | 1.18 | -417 | -680 to -155 | < 0.01 | 0.78 | -305 | -567 to -41 | NS | 0.25 | 113 | -150 to 375 |
| GLC (mmol·l-1) | 3.85 | < 0.05 | 0.23 | < 0.001 | 0.31 | -0.24 | -0.6 to 0.1 | < 0.001 | 0.53 | -0.33 | -0.69 to 0.02 | NS | -0.11 | -0.08 | -0.44 to 0.27 |
RAST; the Running-based Anaerobic test, [La]: Plasma Lactate, Pmax: maximum power during the RAST, Pmean; mean power during the RAST, W; watts, ANOVA; analysis of variance, N20; 20 min nap opportunity, N90; 90 min nap opportunity, F; F de Fisher, p; probability, η2; Eta-squared, d; Cohen’s effect size, MD; Mean difference, 95% CI; 95% confidence Interval, au; arbitrary unit, NS; Non-significant, TEM; Oral Temperature, C°; Celsius, s; second, MCRT; Multiple Choice Reaction Time, POMS; Profile Of Mood State, ESS; Epworth Sleepiness Scale, LDH; Lactate Dehydrogenase, SOD; Superoxide Dismutase and GLC; Plasma Glucose.
DISCUSSION
We showed that a short 20-min nap opportunity (N20) enhanced repeated sprint, reaction time performance, mood, and antioxidant status and reduced sleepiness and biomarkers of muscle damage compared to No-nap and N90. However, contrary to our second hypothesis, the longer nap (N90) resulted in a decreased repeated sprint performance, increased sleepiness and had no effects on reaction time compared to N20.
The current data showed that RAST’s Pmax and Pmean were higher after N20 compared to No-nap. This finding is in accordance with earlier studies that reported an increased short intensive intermittent physical performance after a brief daytime nap the day after NSN [12, 13]. Contrarily, several studies reported a neutral effect of a brief nap on all-out short-term exercise after NSN [7, 9, 10]. The duration and the nature of the exercise could explain this discrepancy. In the current study, participants performed a discontinued repeated sprint of less than 100 s. However, participants in other studies performed a continuous effort (i.e., 20 m straight-line sprint in the study of Suppiah et al. [9] and 30 s Wingate test in the study of Petit et al. [7]. Moreover, Daaloul et al. [10] participants’ performed a specific karate test that lasts more than 13 min. It could be possible that the nap enhances repeated sprint performances by enhancing between sprint recovery [11].
The current study is the first to examine the effect of a relatively long nap opportunity (90 min) on repeated sprint performances after NSN. N90 decreased RAST’s Pmax. The current results could be explained by the severity of sleep inertia after N90 compared to N20. Lovato and Lack [14] reported that longer naps tend to result in greater sleep inertia. Besides, Tietzel and Lack [32] reported that brief naps were associated with shorter periods, and sometimes, did not cause sleep inertia. The higher subjective sleepiness after N90 compared to N20 in the present study confirms this suggestion. Consequently, we suggest that after NSN, athletes should avoid long naps if they have to perform an intermittent exercise at the awakening.
The current results also showed an enhancement of mood and MCRT performance and a lower subjective sleepiness after N20 compared to No-nap. This is in accordance with earlier reports and confirms the beneficial effects of a short nap on psycho-cognitive performances and sleepiness [1, 11, 14, 32]. Contrarily, N90 increased subjective sleepiness, assessed 30 min after awakening. This finding indicates that the 30 min that separated awakening from starting the assessment was not long enough to dissipate sleep residues after the long nap. Indeed, Takahashi et al. [33] reported that sleep inertia could last as long as 2 h after a 90 min nap. It has been also reported that after NSN, naps of 20 min or less do not contain slow wave sleep (SWS), and therefore most likely avoid sleep inertia [17]. Similarly to our current finding, a 10 min nap after NSN produced immediate enhancement of sleepiness and mood [34]. Nonetheless, sleepiness was still high 35 min after awakening from a 30 min nap [34]. This suggests that the short nap in the current study probably have not caused sleep inertia. It could also be possible that the short nap caused sleep inertia that was dissipated during the 30 min that separated awakening from assessments. However, gains obtained from the long nap opportunity -if they exist- require more than 30 min to become tangible, as far as the present study variables are concerned. Hence, further studies are warranted to inspect the effects of a similar nap length on repeated sprint and the time course of sleep inertia.
The nap opportunities per se did not decrease oral temperature (TEM). However, post-napping TEM was lower compared to control. It was suggested that the drop of TEM after N90 contributed to repeated sprint performance enhancement [11]. Although, this does not apply to the current findings since TEM decreased and so did repeated sprint performances.
Post-exercise [La] and CRE were higher after N20 compared to N90, probably because of the higher power output performance after the short nap. Moreover, GLC levels were higher after napping compared to No-nap. These results are in line with earlier findings showing that napping enhances energy availability during the exercise [11, 35]. Both nap opportunities decreased post-exercise URE. Higher URE is an indicator of higher ammonia, which is a toxic waste for the brain and muscles. This confirms earlier suggestions that napping decreases muscle damage [11, 35]. Also, post-exercise LDH was lower after N90 compared to N20. This enzyme interconverts pyruvate and lactate. Thus, the decreased post-exercise LDH levels after N90 could be explained by the decreased lactate production and muscle damage. These results confirm early suggestions, that LDH levels are tributary to the power output and independent of napping and PSD [11, 22].
The current results showed an enhancement of the antioxidant response to the exercise when it was performed after napping, which is in line with previous results [11]. This implies that even a short sleep episode enhances the defense toward the exercise-induced oxidative stress. It was suggested that the increase in enzymatic antioxidant defense after N90 contributes to enhancing repeated sprint performances after PSD [11]. Nevertheless, this does not apply to daytime napping after NSN, since enzymatic antioxidant concentration increased after N90 while performance decreased.
The present study is the first to compare the effects of a short nap with the effects of a long nap opportunity after NSN on mood, reaction time and the biochemical responses to repeated sprint exercise. It has been previously shown that both nap opportunities enhanced repeated sprint performance and decreased subjective sleepiness after PSD, with N90 having a stronger enhancing effect than N20 [11]. For instance, sleep deprivation results in substantial homeostatic disruptions [36–38] that are exacerbated during the PLD [22]. Probably for this reason, the long nap was more beneficial for physical and cognitive performances than the short one [11]. However, the current results showed that the short nap was more beneficial than the long one after NSN. In fact, the PLD is a transient naturally occurring decline in performances even when athletes obtain a sufficient amount of nocturnal sleep [2, 3]. Therefore, a short nap could be scheduled when athletes feel sleepy during the day, despite having a normal sleep during the previous night. Indeed, a brief nap could be beneficial, particularly during the home-confinement period [39, 40], and during rehabilitation programs post-injuries (e.g., post ACL reconstruction) [41].
The current results could be consequent to the chosen protocol. First, the selected participants were non-habitual nappers. It was reported that non-habitual nappers displayed a heavier sleep inertia at the awakening compared to habitual nappers [42]. Thus, results could be different if habitual nappers were included instead of non-habitual nappers. Second, only one test session took place 30 min after the nap. It could be possible that a delayed start of the evaluation might result in different outcomes. Therefore, more studies are needed on this specific point.
CONCLUSIONS
After a previous normal sleep night, the short nap opportunity in the post-lunch dip enhanced repeated sprint and multiple-choice reaction time performances, and antioxidant defense while decreasing sleepiness compared to No-nap and the long nap opportunity. However, despite the enhanced antioxidant defense, the long nap opportunity decreased repeated sprint performances and increased sleepiness compared to No-nap.
Acknowledgment
We express our sincere gratitude to the participants for their maximal efforts and availability during the experiment. We also thank Mr. Firas Ben Slama for his great help in blood collection and sample analysis.
Disclosure of interest
The authors report no conflict of interest.
REFERENCES
- 1.Waterhouse J, Atkinson G, Edwards B, Reilly T. The role of a short post-lunch nap in improving cognitive, motor, and sprint performance in participants with partial sleep deprivation. J Sports Sci. 2007;25(14):1557–1566. doi: 10.1080/02640410701244983. [DOI] [PubMed] [Google Scholar]
- 2.Monk TH. The post-lunch dip in performance. Clin Sports Med. 2005;24(2):15–23. doi: 10.1016/j.csm.2004.12.002. [DOI] [PubMed] [Google Scholar]
- 3.Bes F, Jobert M, Schulz H. Modeling Napping, Post-Lunch Dip, and Other Variations in Human Sleep Propensity. Sleep. 2009;32(3):292–298. doi: 10.1093/sleep/32.3.392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Halson SL. Sleep in Elite Athletes and Nutritional Interventions to Enhance Sleep. Sport Med. 2014;44(S1):13–23. doi: 10.1007/s40279-014-0147-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Juliff LE, Halson SL, Peiffer JJ. Understanding sleep disturbance in athletes prior to important competitions. J Sci Med Sport. 2015;18(1):13–18. doi: 10.1016/j.jsams.2014.02.007. [DOI] [PubMed] [Google Scholar]
- 6.Horne, Anderson C, Platten C. Sleep extension versus nap or coffee, within the context of “sleep debt.”. J Sleep Res. 2008;17(4):432–436. doi: 10.1111/j.1365-2869.2008.00680.x. [DOI] [PubMed] [Google Scholar]
- 7.Petit E, Mougin F, Bourdin H, Tio GG, Haffen E. A 20-min nap in athletes changes subsequent sleep architecture but does not alter physical performances after normal sleep or 5-h phase-advance conditions. Eur J Appl Physiol. 2014;114(2):305–315. doi: 10.1007/s00421-013-2776-7. [DOI] [PubMed] [Google Scholar]
- 8.Blanchfield AW, Lewis-Jones TM, Wignall JR, Roberts JB, Oliver SJ. The influence of an afternoon nap on the endurance performance of trained runners. Eur J Sport Sci. 2018;18(9):1177–1184. doi: 10.1080/17461391.2018.1477180. [DOI] [PubMed] [Google Scholar]
- 9.Suppiah HT, Yong LC, Choong G, Chia M. Effects of a Short Daytime Nap on Shooting and Sprint Performance in High-level Adolescent Athletes. Int J Sports Physiol Perform. 2018:1–25. doi: 10.1123/ijspp.2018-0107. [DOI] [PubMed] [Google Scholar]
- 10.Daaloul H, Souissi N, Davenne D. Effects of Napping on Alertness, Cognitive, and Physical Outcomes of Karate Athletes. Med Sci Sports Exerc. 2019;51(2):338–345. doi: 10.1249/MSS.0000000000001786. [DOI] [PubMed] [Google Scholar]
- 11.Romdhani M, Souissi N, Chaabouni Y, et al. Improved physical performance and decreased muscular and oxidative damage with postlunch napping after partial sleep deprivation in Athletes. Int J Sports Physiol Perform. 2020;15(6):874–883. doi: 10.1123/ijspp.2019-0308. [DOI] [PubMed] [Google Scholar]
- 12.Abdessalem R, Boukhris O, Hsouna H, et al. Effect of napping opportunity at different times of day on vigilance and shuttle run performance. Chronobiol Int. 2019;(10):1334–1342. doi: 10.1080/07420528.2019.1642908. In press. [DOI] [PubMed] [Google Scholar]
- 13.Boukhris O, Abdessalem R, Ammar A, et al. Nap Opportunity During the Daytime Affects Performance and Perceived Exertion in 5-m Shuttle Run Test. Front Physiol. 2019;10:1–8. doi: 10.3389/fphys.2019.00779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lovato N, Lack L. The effects of napping on cognitive functioning. Prog Brain Res. 2010;185:155–166. doi: 10.1016/B978-0-444-53702-7.00009-9. [DOI] [PubMed] [Google Scholar]
- 15.Tassi P, Muzet A. Sleep inertia. Sleep Med Rev. 2000;4(4):341–353. doi: 10.1053/smrv.2000.0098. [DOI] [PubMed] [Google Scholar]
- 16.Ferrara M, De Gennaro L. The sleep inertia phenomenon during the sleep-wake transition: theoretical and operational issues. Aviat Space Environ Med. 2000;71(8):843–848. [PubMed] [Google Scholar]
- 17.Hilditch CJ, Dorrian J, Banks S. A review of short naps and sleep inertia: Do naps of 30 min or less really avoid sleep inertia and slow wave sleep? Sleep Med. 2017;(32):176–190. doi: 10.1016/j.sleep.2016.12.016. [DOI] [PubMed] [Google Scholar]
- 18.Wertz AT, Ronda JM, Czeisler CA, et al. Effects of Sleep Inertia on Cognition. JAMA. 2006;295(2):159. doi: 10.1001/jama.295.2.163. [DOI] [PubMed] [Google Scholar]
- 19.Sallinen M, Härmä M, Akerstedt T, Rosa R, Lillqvist O. Promoting alertness with a short nap during a night shift. J Sleep Res. 1998;7(4):240–247. doi: 10.1046/j.1365-2869.1998.00121.x. [DOI] [PubMed] [Google Scholar]
- 20.Signal TL, van den Berg MJ, Mulrine HM, Gander PH. Duration of Sleep Inertia after Napping during Simulated Night Work and in Extended Operations. Chronobiol Int. 2012;29(6):769–779. doi: 10.3109/07420528.2012.686547. [DOI] [PubMed] [Google Scholar]
- 21.Hammouda O, Chtourou H, Chaouachi A, et al. Effect of short-term maximal exercise on biochemical markers of muscle damage, total antioxidant status, and homocysteine levels in football players. Asian J Sports Med. 2012;3(4):239–246. doi: 10.5812/asjsm.34544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Romdhani M, Hammouda O, Chaabouni Y, et al. Sleep deprivation affects post-lunch dip performances, biomarkers of muscle damage and antioxidant status. Biol Sport. 2019;36(1):55–65. doi: 10.5114/biolsport.2018.78907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Faul F, Erdfelder E, Lang AG, Buchner A. Behavior Research Methods. Vol. 39. Psychonomic Society Inc.; 2007. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences; pp. 175–191. [DOI] [PubMed] [Google Scholar]
- 24.Beck TW. The Importance of A Priori Sample Size Estimation in Strength and Conditioning Research. J Strength Cond Res. 2013;27(8):2323–2337. doi: 10.1519/JSC.0b013e318278eea0. [DOI] [PubMed] [Google Scholar]
- 25.Horne JA, Ostberg O. A self-assessment questionnaire to determine morningness-eveningness in human circadian rhythms. Int J Chronobiol. 1976;4(2):97–110. [PubMed] [Google Scholar]
- 26.Buysse DJ, Reynolds CF, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28(2):193–213. doi: 10.1016/0165-1781(89)90047-4. [DOI] [PubMed] [Google Scholar]
- 27.Draper PN, Whyte G. Anaerobic performance testing. Peak Perform. 1997;96:3–5. [Google Scholar]
- 28.McNair DM. Manual profile of mood state. Educ Ind Test Serv. journal Published online 1971. [Google Scholar]
- 29.Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep. 1991;14(6):540–545. doi: 10.1093/sleep/14.6.540. [DOI] [PubMed] [Google Scholar]
- 30.Foster C. Monitoring training in athletes with reference to overtraining syndrome. Med Sci Sports Exerc. 1998;30(7):1164–1168. doi: 10.1097/00005768-199807000-00023. [DOI] [PubMed] [Google Scholar]
- 31.Cohen J. A power primer. Psychol Bull. 1992;112(1):155–159. doi: 10.1037//0033-2909.112.1.155. [DOI] [PubMed] [Google Scholar]
- 32.Tietzel AJ, Lack LC. The recuperative value of brief and ultra-brief naps on alertness and cognitive performance. J Sleep Res. 2002;11(3):213–218. doi: 10.1046/j.1365-2869.2002.00299.x. [DOI] [PubMed] [Google Scholar]
- 33.Takahashi M, Arito H, Fukuda H. Nurses’ workload associated with 16-h night shifts. II: Effects of a nap taken during the shifts. Psychiatry Clin Neurosci. 1999;53(2):223–225. doi: 10.1046/j.1440-1819.1999.00545.x. [DOI] [PubMed] [Google Scholar]
- 34.Brooks A, Lack L. A Brief Afternoon Nap Following Nocturnal Sleep Restriction: Which Nap Duration is Most Recuperative? Sleep. 2006;29(6):831–840. doi: 10.1093/sleep/29.6.831. [DOI] [PubMed] [Google Scholar]
- 35.Hammouda O, Romdhani M, Chaabouni Y, Mahdouani K, Driss T, Souissi N. Diurnal napping after partial sleep deprivation affected hematological and biochemical responses during repeated sprint. Biol Rhythm Res. 2018;1016:1–13. [Google Scholar]
- 36.Romdhani M, Hammouda O, Smari K, et al. Total Sleep Deprivation and Recovery Sleep Affect the Diurnal Variation of Agility Performance. J Strength Cond Res. 2021;35(1):132–140. doi: 10.1519/JSC.0000000000002614. [DOI] [PubMed] [Google Scholar]
- 37.Skurvydas A, Zlibinaite L, Solianik R, et al. One night of sleep deprivation impairs executive function but does not affect psychomotor or motor performance. Biol Sport. 2020;37(1):7–14. doi: 10.5114/biolsport.2020.89936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tomczak A, Gajewski J, Mazur- Rózycka J. Changes in physiological tremor resulting from sleep deprivation under conditions of increasing fatigue during prolonged military training. Biol Sport. 2014;31(4):303–308. doi: 10.5604/20831862.1127343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Trabelsi K, Ammar A, Liwa M, et al. Globally altered sleep patterns and physical activity levels by confinement in 5056 individuals: ECLB COVID-19 international online survey. Biol Sport. 2021;38(4):495–506. doi: 10.5114/biolsport.2021.101605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yousfi N, Bragazzi NL, Briki W, Zmijewski P, Chamari K. The COVID-19 pandemic: How to maintain a healthy immune system during the lockdown – A multidisciplinary approach with special focus on athletes. Biol Sport. 2020;37(3):211–216. doi: 10.5114/biolsport.2020.95125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Khalladi K, Farooq A, Sas B, et al. Sleep and psychological factors are associated with meeting discharge criteria to return to sport following ACL reconstruction in athletes. Biol Sport. 2020;38(3):305–313. doi: 10.5114/biolsport.2021.99704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Dinges DF. Why We Nap. Boston: Birkhäuser; 1992. Adult Napping and Its Effects on Ability to Function; pp. 118–134. [Google Scholar]