Abstract
J Clin Hypertens (Greenwich).
The unique pathophysiology of heart failure with a preserved ejection fraction (HF‐PEF) and the involvement of hypertension in its development are only poorly understood. The upregulation of the renin‐angiotensin‐aldosterone system (RAAS) has been identified as a key pathologic pathway contributing to fibrosis, cardiomyocyte abnormalities, inflammation, and endothelial dysfunction, all of which have been implicated in the progression of hypertension to HF‐PEF. In addition, pharmacologic inhibition of the RAAS has been shown in animal models of diastolic dysfunction and in clinical trials to reduce these deleterious processes and to improve diastolic function. Despite these data, clinical trials performed with RAAS inhibitors in patients with HF‐PEF have failed to demonstrate morbidity and mortality benefits. To date, there is no proven effective therapy specifically for HF‐PEF. The deleterious effects of hypertension on mechanisms underlying the development of HF‐PEF underscore the importance of effective and early control of hypertension for the prevention of HF‐PEF.
Heart failure (HF) is a prevalent condition associated with substantial morbidity and mortality. 1 , 2 Approximately half of all patients with HF have a reduced ejection fraction (REF) and the other half have a preserved ejection fraction (PEF). Although cutoffs vary between studies, a left ventricular (LV) ejection fraction (LVEF) ≥45% or 50% is generally considered to indicate HF with PEF (HF‐PEF). The relationship between REF and PEF in HF is complex and, in fact, diastolic dysfunction may be present in variable degrees in patients with systolic dysfunction and vice versa. Diastolic dysfunction might conceivably represent a precursor of systolic dysfunction in a subset of patients with HF. The unique pathophysiology of HF‐PEF and the involvement of hypertension in its development are only poorly understood. In this review, we will focus on the central roles of hypertension and the renin‐angiotensin‐aldosterone system (RAAS) in its development.
Definition of HF‐PEF
HF‐PEF refers to a clinical status of chronic HF in patients with normal or mildly depressed LVEF. Based on a recent consensus statement from the Heart Failure and Echocardiography Associations of the European Society of Cardiology, HF‐PEF can be diagnosed if the following 3 conditions are present: (1) signs or symptoms of HF; (2) normal or mildly abnormal LVEF (LVEF >50% and an LV end‐diastolic volume index <97 mL/m2); and (3) evidence of diastolic LV dysfunction such as elevated LV filling pressures, LV relaxation abnormalities as assessed by tissue Doppler measurements, or structural abnormalities associated with high levels of brain natriuretic peptide. 3
Several terminologies are interchangeably used in the literature, HF‐PEF, HF with preserved systolic function, HF with normal LVEF, and diastolic HF. We will use the term diastolic dysfunction to describe cardiac function and the term HF‐PEF to describe the clinical syndrome of chronic HF with normal or near‐normal LVEF.
Prevalence and Prognosis of HF‐PEF
Epidemiologic studies in the Western world have shown that 30% to 60% of HF patients have a normal LVEF (≥50%). 1 , 4 , 5 , 6 , 7 , 8 In a recent French study of 660 patients hospitalized for a first episode of HF, 55% of patients had HF‐PEF. 8 This rate increased to 61% when only patients older than 75 years were considered. Similarly, in the large, hospital‐based, EuroHeart Failure study, HF‐PEF (≥40%) was present in 55% of patients with a diagnosis of HF, corresponding to 49% of men and 72% of women. 2
As with HF‐REF, the prognosis for patients with HF‐PEF is poor, with high morbidity and mortality rates. In French hospitals, for example, patients with HF‐PEF had a 5‐year survival rate after a first hospitalization for HF of 43%. 8 This rate is not surprising considering the previously reported 1‐year mortality rates of approximately 25% in patients with HF‐PEF. 5 , 6 Poor prognosis has also been described in a multivariate analysis that controlled for age, sex, and EF. 1 Both mild diastolic dysfunction (hazard ratio [HR], 8.3; P<.001) and moderate or severe diastolic dysfunction (HR, 10.2; P<.001) were significantly predictive of all‐cause mortality.
Hypertension is Strongly Associated With HF‐PEF and Diastolic Dysfunction
The most common characteristics of patients with HF‐PEF are increasingly being recognized (Table). 4 , 5 , 6 , 7 , 8 , 9 Patients with HF‐PEF tend to be elderly and female. 2 , 4 , 7 , 8 , 10 Comorbid conditions include prior coronary artery disease, myocardial infarction, and atrial fibrillation. Cardiovascular (CV) risk factors, which are prevalent as expected, include hypertension, smoking, diabetes, hyperlipidemia, and obesity.
Table.
Characteristics of Patients With HF‐PEF in Epidemiological Studies
| Tribouilloy et al,8 2008 | Bursi et al,4 2006 | Owan et al,5 2006 | Bhatia et al,6 2006 | Masoudi et al,7 2003 | Lenzen et al, 9 2004 | |
|---|---|---|---|---|---|---|
| Country | France | United Statesa | United Statesa | Canada | United States | Europeb |
| PEF definition | ≥50% | ≥50% | ≥50% | >50% | ≥50% | ≥40% |
| No. of patients | 368 | 308 | 2167 | 880 | 6754 | 3148 |
| Age, y | 76 | 77 | 74 | 75 | 80 | 71 |
| Male sex, % | 47 | 43 | 44 | 34 | 29 | 45 |
| LVEF, % | 63 | – | 61 | 62 | – | 56 |
| Comorbidities, % | ||||||
| Hypertension | 74 | 86 | 63 | 55 | 69 | 59 |
| Diabetes | 26 | 36 | 33 | 32 | 37 | 26 |
| Myocardial infarction | 9 | 36 | – | 17 | 21 | – |
| CAD or ischaemia | 28 | – | 53 | 36 | 46 | 59 |
| Stroke/TIA | 5 | – | – | 15 | 17 | 16 |
| Atrial fibrillation | 36 | 31 | 41 | 32 | 36 | 25 |
Abbreviations: CAD, coronary artery disease; HF‐PEF, heart failure with a preserved ejection fraction (PEF); LVEF, left ventricular ejection fraction; TIA, transient ischemic attack. aOlmsted county. b24 countries.
Hypertension, in particular, which is present in 55% to 86% of patients in epidemiologic trials and 60% to 88% of patients in HF‐PEF clinical trials, 4 , 5 , 6 , 7 , 8 , 9 , 11 , 12 , 13 is strongly associated with the development of HF. The risk of developing HF after adjusting for age and other risk factors is approximately 2‐fold higher in hypertensive men and 3‐fold higher in hypertensive women than in normotensive persons. 14 In a large systematic review of the recent trials, the authors showed that the incidence of chronic HF was high in hypertensive patients and comparable with that of stroke. 15 This relationship seems to hold true for patients with HF‐PEF. In a multivariate analysis of data from a US‐based study (N=300), for example, hypertension at presentation was significantly associated with HF‐PEF. 16 In a population‐based European sample (N=1274), arterial hypertension was shown to be an independent predictor of diastolic abnormalities. 17
Furthermore, diastolic dysfunction was found in patients with essential hypertension long before the appearance of overt HF symptoms. In the observational Assessment of Prevalence Observational Study of Diastolic Dysfunction (APROS‐diadys) study, 26% of elderly hypertensive patients without HF symptoms and with an LVEF ≥45% showed some level of diastolic dysfunction on echocardiographic Doppler examination. 10 This rate of diastolic dysfunction was significantly higher in those who had uncontrolled hypertension than in those with controlled hypertension.
Progression From Hypertension to HF‐PEF
The overall progression from hypertension through the CV continuum to HF‐PEF is outlined in the Figure. Hypertension clearly damages the cardiac and vascular systems over time, causing LV hypertrophy (LVH), a decrease in renal function, and vascular changes. The resulting increase in LV afterload and excess myocardial oxygen consumption contributes to cardiac structural abnormalities, promoting the development of HF.
Figure.
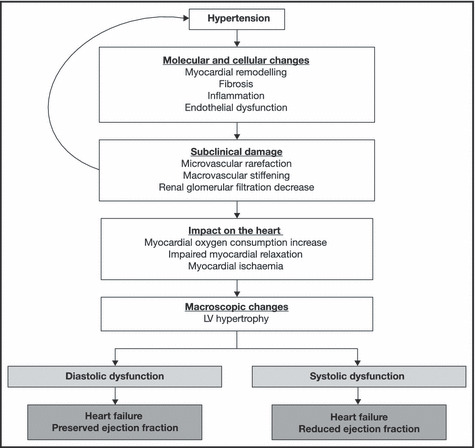
The cardiovascular disease continuum. LV indicates left ventricular.
Indeed, the microvasculature, which mediates the delivery of nutrients and oxygen to tissues and controls vascular resistance, is both a target and a determinant of hypertension. 18 In response to hypertension, small artery and arteriole rarefaction occurs, causing an increase in vascular resistance, a rearrangement of the extracellular matrix, smooth muscle cell proliferation, and migration, and eventually an increase in arterial wall stiffness. As the arterial impedance increases, central systolic blood pressure (BP) and pulse pressure increase, thereby leading to greater LV afterload, myocardial hypertrophy, and myocardial oxygen consumption. Myocardial hypertrophy increases myocardial oxygen demand while high LV filling pressures and LVH impair subendocardial perfusion, particularly when a decrease in central diastolic BP emerges. All of these events may lead to myocardial ischaemia and impaired myocardial relaxation. Notably, myocardial relaxation is an energy‐dependent process, relying heavily on adenosine triphosphate–dependent calcium cycling.
The kidney is also both a target and determinant of BP. 18 In hypertensive patients in whom pulse pressure and arterial stiffness are increased, glomerular filtration is decreased. This causes tubular damage and a progressive degradation of renal function. Renal dysfunction causes a further increase in BP, which leads to increased LV afterload and myocardial oxygen consumption.
Molecular and Cellular Mechanisms
Although extracellular matrix alterations, cardiomyocyte abnormalities, endothelial dysfunction, and inflammation are believed to be the main mechanisms that underlie macroscopic damage to the heart, much remains to be elucidated. 18 , 19 , 20 For example, the importance of each of these molecular and cellular processes in the development of systolic dysfunction vs diastolic dysfunction remains to be clarified. The following discussion will describe what is currently known about the mechanisms surrounding the development of diastolic dysfunction since it is a main contributor to HF‐PEF.
Markers of fibrosis have been shown to be associated with asymptomatic diastolic dysfunction in patients with essential hypertension. 21 More specifically, compromised filling due to increased ventricular stiffness may be due to fibrosis. In a study of 86 hypertensive patients with evidence of diastolic dysfunction, 22 collagen turnover was greater in patients with more severe diastolic dysfunction. Both fibrogenesis, as seen by increased levels of carboxy‐terminal telopeptide of procollagen type I (PICP) and amino‐terminal propeptide of procollagen type III (PIIINP), and collagen degradation, as seen by levels of serum carboxy‐terminal telopeptide of procollagen type I (ICTP), matrix metalloproteinase (MMP)‐2, and MMP‐9, were increased.
Cardiomyocyte abnormalities develop in response to the mechanical stress associated with hypertension and contribute to the development of diastolic dysfunction. 19 , 23 In a study of patients hospitalized for worsening HF (N=44), greater cardiomyocyte diameter and myocyte passive force was noted in patients with diastolic dysfunction than in patients with systolic dysfunction. 23 Such an increase in myocyte width, for example, can be seen as the basis of the development of concentric hypertrophy, a characteristic of diastolic dysfunction. In addition, systolic dysfunction of the left ventricle is more likely to occur as the degree of diastolic dysfunction augments. 24
Inflammation and endothelial dysfunction are also pivotal players in the progression of hypertension to HF. Monocyte‐derived macrophages and T lymphocytes secrete mediator molecules such as cytokines, chemokines, and growth factors, which lead to the activation of endothelial cells, proliferation of smooth muscle cells, and vascular lesions. 20 In rat models of hypertension, macrophages have been shown to accumulate in the perivascular spaces and colocalize with fibroblasts, producing collagen during cardiac hypertrophy. 25 Furthermore, inhibition of myocyte chemoattractant protein‐1 in a rat model of developing diastolic dysfunction inhibited macrophage accumulation, transforming growth factor β induction, fibroblast proliferation, and myocardial fibrosis and ameliorated diastolic dysfunction. 26 Such findings have been confirmed in clinical studies. In a study of CV damage in hypertensive patients, markers of inflammation (urinary albumin, C‐reactive protein, tumor necrosis factor α, and transforming growth factor β) were independently associated with asymptomatic diastolic dysfunction. 27 In studies of patients with stable coronary artery disease, diastolic dysfunction was closely associated with increased platelet activation, inflammation, and endothelial dysfunction. 28 , 29 , 30 C‐reactive protein, for example, was predictive of abnormal diastolic function in patients with coronary artery disease, 29 and endothelial dysfunction was associated with the progression of diastolic dysfunction in patients with coronary artery disease. 31 Taken together, many abnormalities have been identified in patients with HF‐PEF; however, these observations remain descriptive and it is unclear why some patients predominantly develop diastolic dysfunction in response to long‐standing arterial hypertension while others respond with increased LV volumes and a decrease in LVEF. Brutsaert and Keulenear 32 proposed that diastolic dysfunction may proceed to systolic dysfunction over time and suggested that both entities are the extreme manifestations of one syndrome. In any case, the underlying driving pathophysiologic process for the development of diastolic vs systolic HF remains to be elucidated.
The RAAS
As the RAAS regulates renal perfusion and salt and water homoeostasis, as well as many of the pathways that govern CV and renal structure and function, its up‐regulation in hypertension and at all subsequent stages of hypertensive CV disease, including HF, is believed to be the basis for much of the renovascular and CV damage associated with deteriorating heart function such as diastolic dysfunction. 33 , 34 , 35 , 36
RAAS Inhibition: Microscopic and Macroscopic Effects
The pharmacologic inhibition of these RAAS pathways by RAAS inhibitors (angiotensin‐converting enzyme [ACE] inhibitors, angiotensin receptor blockers [ARBs], aldosterone blockers, and renin inhibitors) not only lowers BP, but also reduces extracellular matrix deposition, myocyte hypertrophy, cardiac inflammation, and endothelial damage. 33 Although a direct correlation between the molecular and cellular effects of RAAS inhibition and macroscopic properties of the heart have not been systematically established, it is noteworthy that in some animal and clinical trials, RAAS inhibition improves parameters of diastolic function.
Clinical Trials in HF‐PEF: Effects of RAAS Inhibition
Although HF‐PEF is associated with substantial mortality and morbidity, the treatment modalities for this condition have not been well investigated. There are many reviews and smaller studies in the literature outlining potential strategies for HF‐PEF. 37 In contrast to HF‐REF, however, there are relatively few published large, randomized, controlled clinical trials that evaluate the treatment of HF‐PEF.
ACE Inhibitors
Perindopril. Perindopril has been shown to have cardiac remodelling benefits in post–myocardial infarction patients with preserved systolic function. 38 In the large, randomized, controlled Perindopril in Elderly People With Chronic Heart Failure (PEP‐CHF) study (N=850), however, the primary end point (a composite of all‐cause mortality and unplanned HF‐related hospitalization) was not significantly different between the perindopril and the placebo groups. 11
Angiotensin Receptor Blockers
Candesartan. In the large 3‐year, randomized, controlled Candesartan in Heart Failure: Assessment of Reduction in Mortality and Morbidity (CHARM‐Preserved) trial (N=4500), candesartan was compared with placebo in 3023 patients with chronic HF and an LVEF >40%. 12 No significant difference between groups in the primary outcome (CV death or unplanned admission to hospital for worsening HF) was detected (unadjusted HR 0.89, 95% confidence interval [CI], 0.77–1.03 [P=.118]; covariate adjusted HR 0.86, 95% CI, 0.74–1.0 [P=.051]), however, there were fewer HF hospitalizations in the candesartan group than in the placebo group [P<.02].
Irbesartan. In the double‐blind, placebo‐controlled Irbesartan in Patients With Heart Failure and Preserved Ejection Fraction (I‐PRESERVE) trial, the largest HF‐PEF study to date, the efficacy of irbesartan therapy was evaluated in 4128 chronic HF patients with LVEF ≥45%. Their characteristics were similar to those observed in HF‐PEF epidemiologic studies. 13 The mean follow‐up was 49.5 months. The primary combined outcome of all‐cause mortality and protocol‐specified CV hospitalizations (for HF, myocardial infarction, unstable angina, stroke, or ventricular or atrial arrhythmia) was not significantly reduced by irbesartan compared with placebo (HR, 0.95; 95% CI, 0.86–1.05; P=.35). 39 Importantly, a large proportion of patients were receiving background medication, including ACE inhibitors, aldosterone blockers, and β‐blockers, as well as antiplatelet and lipid‐lowering therapy, 13 possibly leaving little room for incremental benefit with the addition of an ARB.
Aldosterone Blockers
Spironolactone. The Treatment of Preserved Cardiac Function Heart Failure With an Aldosterone Antagonist (TOPCAT) study is an ongoing large, double‐blind, randomized, placebo‐controlled trial, in which 4500 patients diagnosed with HF with an LVEF ≥45% and at least one hospital admission for HF or elevated brain natriuretic peptide levels, will be enrolled to take spironolactone or placebo for 2 years. 40 The primary end point is a composite of CV mortality, aborted cardiac arrest, or hospitalization for the management of HF.
Renin Inhibitors
To date, no large clinical trials in HF‐PEF have been performed with renin inhibitors. The published clinical trials performed so far in the HF‐PEF patient population have essentially been neutral. The guidelines from various countries deal with the management of HF‐PEF patients by recommending the treatment of comorbidities (eg, hypertension) that exist in these patients. In order for research in this field to move forward, we need a better understanding of the mechanisms underlying this syndrome to gain additional potential targets for treatment.
Conclusions
Hypertension is an underlying factor in HF‐PEF. Although the RAAS is a key player in the development of HF‐PEF and, although several classes of RAAS inhibitors are available, therapeutic clinical trials have not shown morbidity and mortality benefits in patients who present with this syndrome. To date, there is no proven effective therapy for HF‐PEF. It is as yet unclear whether the RAAS is not an appropriate target for therapy, or whether current trial designs, allowing extensive use of background therapy, hinder the demonstration of clinical benefit in recent trials. The deleterious effects of hypertension on mechanisms responsible for the development of HF‐PEF underscore the importance of effective and early control of hypertension to prevent the development of HF‐PEF.
Acknowledgments and disclosures:
Sadly, since writing this article, Helmut Drexler has passed away. The other authors would like to take this opportunity to acknowledge his vast contribution to advancement of research in the area of prevention of heart failure. Editorial support for this article was provided by PPSI, Bristol‐Myers Squibb, and Sanofi‐Aventis. Massimo Volpe has been on advisory boards and speakers’ bureaus for Bristol‐Myers Squibb and sanofi‐aventis. Robert McKelvie has received honoraria and consulting fees from Bristol‐Myers Squibb and Sanofi‐Aventis. Both authors were investigators for I‐PRESERVE. Helmut Drexler received honoraria, was on the speakers’ bureau, and was a primary investigator on a clinical trial for Medtronic; received honoraria and was on a steering committee for Pfizer; received honoraria from Novartis; received honoraria and was on advisory boards for Vifor; and was on advisory boards for Amgen.
References
- 1. Redfield MM, Jacobsen SJ, Burnett JC Jr, et al. Burden of systolic and diastolic ventricular dysfunction in the community: appreciating the scope of the heart failure epidemic. JAMA. 2003;289:194–202. [DOI] [PubMed] [Google Scholar]
- 2. Cleland JG, Swedberg K, Follath F, et al. The EuroHeart Failure survey programme – a survey on the quality of care among patients with heart failure in Europe. Part 1: patient characteristics and diagnosis. Eur Heart J. 2003;24:442–463. [DOI] [PubMed] [Google Scholar]
- 3. Paulus WJ, Tschope C, Sanderson JE, et al. How to diagnose diastolic heart failure: a consensus statement on the diagnosis of heart failure with normal left ventricular ejection fraction by the Heart Failure and Echocardiography Associations of the European Society of Cardiology. Eur Heart J. 2007;28:2539–2550. [DOI] [PubMed] [Google Scholar]
- 4. Bursi F, Weston SA, Redfield MM, et al. Systolic and diastolic heart failure in the community. JAMA. 2006;296:2209–2216. [DOI] [PubMed] [Google Scholar]
- 5. Owan TE, Hodge DO, Herges RM, et al. Trends in prevalence and outcome of heart failure with preserved ejection fraction. N Engl J Med. 2006;355:251–259. [DOI] [PubMed] [Google Scholar]
- 6. Bhatia RS, Tu JV, Lee DS, et al. Outcome of heart failure with preserved ejection fraction in a population‐based study. N Engl J Med. 2006;355:260–269. [DOI] [PubMed] [Google Scholar]
- 7. Masoudi FA, Havranek EP, Smith G, et al. Gender, age, and heart failure with preserved left ventricular systolic function. J Am Coll Cardiol. 2003;41:217–223. [DOI] [PubMed] [Google Scholar]
- 8. Tribouilloy C, Rusinaru D, Mahjoub H, et al. Prognosis of heart failure with preserved ejection fraction: a 5 year prospective population‐based study. Eur Heart J. 2008;29:339–347. [DOI] [PubMed] [Google Scholar]
- 9. Lenzen MJ, Scholte op Reimer WJ, Boersma E, et al. Differences between patients with a preserved and a depressed left ventricular function: a report from the EuroHeart Failure Survey. Eur Heart J. 2004;25:1214–1220. [DOI] [PubMed] [Google Scholar]
- 10. Zanchetti A, Cuspidi C, Comarella L, et al. Left ventricular diastolic dysfunction in elderly hypertensives: results of the APROS‐diadys study. J Hypertens. 2007;25:2158–2167. [DOI] [PubMed] [Google Scholar]
- 11. Cleland JG, Tendera M, Adamus J, et al. The perindopril in elderly people with chronic heart failure (PEP‐CHF) study. Eur Heart J. 2006;27:2338–2345. [DOI] [PubMed] [Google Scholar]
- 12. Yusuf S, Pfeffer MA, Swedberg K, et al. Effects of candesartan in patients with chronic heart failure and preserved left‐ventricular ejection fraction: the CHARM‐Preserved Trial. Lancet. 2003;362:777–781. [DOI] [PubMed] [Google Scholar]
- 13. McMurray JJ, Carson PE, Komajda M, et al. Heart failure with preserved ejection fraction: clinical characteristics of 4133 patients enrolled in the I‐PRESERVE trial. Eur J Heart Fail. 2008;10:149–156. [DOI] [PubMed] [Google Scholar]
- 14. Levy D, Larson MG, Vasan RS, et al. The progression from hypertension to congestive heart failure. JAMA. 1996;275:1557–1562. [PubMed] [Google Scholar]
- 15. Tocci G, Sciarretta S, Volpe M. Development of heart failure in recent hypertension trials. J Hypertens. 2008;26:1477–1486. [DOI] [PubMed] [Google Scholar]
- 16. Peyster E, Norman J, Domanski M. Prevalence and predictors of heart failure with preserved systolic function: community hospital admissions of a racially and gender diverse elderly population. J Card Fail. 2004;10:49–54. [DOI] [PubMed] [Google Scholar]
- 17. Fischer M, Baessler A, Hense HW, et al. Prevalence of left ventricular diastolic dysfunction in the community. Results from a Doppler echocardiographic‐based survey of a population sample. Eur Heart J. 2003;24:320–328. [DOI] [PubMed] [Google Scholar]
- 18. Cohuet G, Struijker‐Boudier H. Mechanisms of target organ damage caused by hypertension: therapeutic potential. Pharmacol Ther. 2006;111:81–98. [DOI] [PubMed] [Google Scholar]
- 19. Kass DA, Bronzwaer JG, Paulus WJ. What mechanisms underlie diastolic dysfunction in heart failure? Circ Res. 2004;94:1533–1542. [DOI] [PubMed] [Google Scholar]
- 20. Koenig W, Khuseyinova N. Biomarkers of atherosclerotic plaque instability and rupture. Arterioscler Thromb Vasc Biol. 2007;27:15–26. [DOI] [PubMed] [Google Scholar]
- 21. Muller‐Brunotte R, Kahan T, Lopez B, et al. Myocardial fibrosis and diastolic dysfunction in patients with hypertension: results from the Swedish Irbesartan Left Ventricular Hypertrophy Investigation versus Atenolol (SILVHIA). J Hypertens. 2007;25:1958–1966. [DOI] [PubMed] [Google Scholar]
- 22. Martos R, Baugh J, Ledwidge M, et al. Diastolic heart failure: evidence of increased myocardial collagen turnover linked to diastolic dysfunction. Circulation. 2007;115:888–895. [DOI] [PubMed] [Google Scholar]
- 23. Van Heerebeek L, Borbely A, Niessen HW, et al. Myocardial structure and function differ in systolic and diastolic heart failure. Circulation. 2006;113:1966–1973. [DOI] [PubMed] [Google Scholar]
- 24. Sciarretta S, Paneni F, Ciavarella GM, et al. Evaluation of systolic properties in hypertensive patients with different degrees of diastolic dysfunction and normal ejection fraction. Am J Hypertens. 2009;22:437–443. [DOI] [PubMed] [Google Scholar]
- 25. Nicoletti A, Heudes D, Mandet C, et al. Inflammatory cells and myocardial fibrosis: spatial and temporal distribution in renovascular hypertensive rats. Cardiovasc Res. 1996;32:1096–1107. [DOI] [PubMed] [Google Scholar]
- 26. Kuwahara F, Kai H, Tokuda K, et al. Hypertensive myocardial fibrosis and diastolic dysfunction: another model of inflammation? Hypertension. 2004;43:739–745. [DOI] [PubMed] [Google Scholar]
- 27. Sciarretta S, Ferrucci A, Ciavarella GM, et al. Markers of inflammation and fibrosis are related to cardiovascular damage in hypertensive patients with metabolic syndrome. Am J Hypertens. 2007;20:784–791. [DOI] [PubMed] [Google Scholar]
- 28. Lee KW, Blann AD, Lip GY. Impaired tissue Doppler diastolic function in patients with coronary artery disease: relationship to endothelial damage/dysfunction and platelet activation. Am Heart J. 2005;150:756–766. [DOI] [PubMed] [Google Scholar]
- 29. Williams ES, Shah SJ, Ali S, et al. C‐reactive protein, diastolic dysfunction, and risk of heart failure in patients with coronary disease: heart and Soul Study. Eur J Heart Fail. 2008;10:63–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Elesber AA, Redfield MM, Rihal CS, et al. Coronary endothelial dysfunction and hyperlipidemia are independently associated with diastolic dysfunction in humans. Am Heart J. 2007;153:1081–1087. [DOI] [PubMed] [Google Scholar]
- 31. Ma LN, Zhao SP, Gao M, et al. Endothelial dysfunction associated with left ventricular diastolic dysfunction in patients with coronary heart disease. Int J Cardiol. 2000;72:275–279. [DOI] [PubMed] [Google Scholar]
- 32. Brutsaert DL, De Keulenaer GW. Diastolic heart failure: a myth. Curr Opin Cardiol. 2006;21:240–248. [DOI] [PubMed] [Google Scholar]
- 33. Sciarretta S, Paneni F, Palano F, et al. Role of the renin‐angiotensin‐aldosterone system and inflammatory processes in the development and progression of diastolic dysfunction. Clin Sci (Lond). 2009;116:467–477. [DOI] [PubMed] [Google Scholar]
- 34. Bernal J, Pitta SR, Thatai D. Role of the renin‐angiotensin‐aldosterone system in diastolic heart failure: potential for pharmacologic intervention. Am J Cardiovasc Drugs. 2006;6:373–381. [DOI] [PubMed] [Google Scholar]
- 35. Chrysant SG, Chrysant GS. Clinical experience with angiotensin receptor blockers with particular reference to valsartan. J Clin Hypertens (Greenwich). 2004;6:441–445. [DOI] [PubMed] [Google Scholar]
- 36. Ram CV. Angiotensin receptor blockers: current status and future prospects. Am J Med. 2008;121:656–663. [DOI] [PubMed] [Google Scholar]
- 37. Hogg K, McMurray J. The treatment of heart failure with preserved ejection fraction (“diastolic heart failure”). Heart Fail Rev. 2006;11:141–146. [DOI] [PubMed] [Google Scholar]
- 38. Ferrari R. Effects of angiotensin‐converting enzyme inhibition with perindopril on left ventricular remodeling and clinical outcome: results of the randomized Perindopril and Remodeling in Elderly with Acute Myocardial Infarction (PREAMI) Study. Arch Intern Med. 2006;166:659–666. [DOI] [PubMed] [Google Scholar]
- 39. Massie BM, Carson PE, McMurray JJ, et al. Irbesartan in patients with heart failure and preserved ejection fraction. N Engl J Med. 2008;359:2456–2467. [DOI] [PubMed] [Google Scholar]
- 40. National Heart L, and Blood Institute (NHLBI) . Aldosterone Antagonist Therapy for Adults With Heart Failure and Preserved Systolic Function (TOPCAT). http://www.clinicaltrials.gov/show/NCT00094302. Accessed October 20, 2008.


