Abstract
An ecogenomic analysis of the methanogenic microbial community in a laboratory-scale up-flow anaerobic sludge blanket (UASB) reactor treating soy sauce-processing wastewater revealed a synergistic metabolic network. Granular sludge samples were collected from the UASB reactor operated under psychrophilic (20°C) conditions with a COD removal rate >75%. A 16S rRNA gene amplicon sequencing-based microbial community analysis classified the major microbial taxa as Methanothrix, Methanobacterium, Pelotomaculaceae, Syntrophomonadaceae, Solidesulfovibrio, and members of the phyla Synergistota and Bacteroidota. Draft genomes of dominant microbial populations were recovered by metagenomic shotgun sequencing. Metagenomic- and metatranscriptomic-assisted metabolic reconstructions indicated that Synergistota- and Bacteroidota-related organisms play major roles in the degradation of amino acids. A metagenomic bin of the uncultured Bacteroidales 4484-276 clade encodes genes for proteins that may function in the catabolism of phenylalanine and tyrosine under microaerobic conditions. Syntrophomonadaceae and Pelotomaculaceae oxidize fatty acid byproducts presumably derived from the degradation of amino acids in syntrophic association with aceticlastic and hydrogenotrophic methanogen populations. Solidesulfovibrio organisms are responsible for the reduction of sulfite and may support the activity of hydrogenotrophic methanogens and other microbial populations by providing hydrogen and ammonia using nitrogen fixation-related proteins. Overall, functionally diverse anaerobic organisms unite to form a metabolic network that performs the complete degradation of amino acids in the psychrophilic methanogenic microbiota.
Keywords: wastewater treatment, psychrophilic, methanogenic ecosystem, ecogenomics, amino acid degradation
Soy sauce is a fermented product of soybean that is used as a seasoning for cooking (Lioe et al., 2010). In Japan, there are more than 1,100 soy sauce-processing manufacturing plants and annual production is approximately 744 million liters (Soy Sauce Information Center, 2021, https://www.soysauce.or.jp/). Soy sauce production incurs the discharge of wastewater containing various amino acids with high chemical oxygen demand (COD) concentrations (~150,000 mg COD L–1) at psychrophilic temperatures (e.g., 16 to 20°C) (Kuroda et al., 2017). To treat this high-strength organic wastewater at a low temperature, an anaerobic treatment system, such as up-flow anaerobic sludge blanket (UASB)- and expanded granular sludge bed (EGSB)-type methanogenic bioreactors, is a promising technology due to its high capacity to degrade concentrated substrates and low energy requirements (Lettinga, 1995; Lettinga et al., 1999, 2001). Although growth and methane production activities are lower than those under mesophilic or thermophilic conditions (Lettinga et al., 2001), previous studies using anaerobic bioreactors operated under psychrophilic conditions showed good efficiency for the treatment of various organic wastewater, including citric acid-processing wastewater (Collins et al., 2006), brewery wastewater (Xing et al., 2009), synthetic fatty acid-based wastewater (Collins et al., 2003; Gunnigle et al., 2015), dairy wastewater (McAteer et al., 2020; Paulo et al., 2020), and low-strength sewage wastewater (Keating et al., 2016, 2018; Petropoulos et al., 2017; Ribera-Pi et al., 2020).
In the methanogenic wastewater treatment system, even if temperature conditions vary between psychrophilic, mesophilic, and thermophilic, the three major trophic groups basically form a synergistic microbial interaction to degrade organic compounds in wastewater into methane and carbon dioxide; fermenters degrade organic compounds (e.g., carbohydrates, proteins, amino acids, and lipids) to fatty acids (e.g., propionate, butyrate, and acetate as volatile fatty acids [VFA]; isovalerate, 2-methylbutyrate, and isobutyrate as branched-chain fatty acids [BCFA]) and hydrogen; syntrophic substrate oxidizers (syntrophs) degrade fatty acids to acetate and hydrogen; and methanogenic archaea (methanogens) further utilize acetate and hydrogen with the production of methane and carbon dioxide (Schink and Stams, 2013). To date, major microbial constituents inhabiting psychrophilic methanogenic bioreactors have been identified by 16S rRNA-targeting molecular approaches (Collins et al., 2006; Xing et al., 2009; Gunnigle et al., 2015; Petropoulos et al., 2017; Keating et al., 2018; Paulo et al., 2020; Ribera-Pi et al., 2020). However, there is currently no information on the metabolic functions of the methanogenic microbiota that proliferates in psychrophilic bioreactors treating high-concentration organic wastewater. The metabolic strategies and interactions employed by microorganisms to accomplish anaerobic amino acid degradation under psychrophilic conditions remain poorly understood. As temperature decreases, methanogens may consume hydrogen to lower concentrations (Conrad and Wetter, 1990), potentially influencing how syntrophic organotrophy is accomplished. Wastewater treatment bioreactors represent rare amino acid-rich ecosystems that have sufficient metabolic activity and biomass to allow omics-based investigations of the above behavior.
We herein attempted to clarify the diversity and metabolic functions of dominant microorganisms in an UASB-type methanogenic bioreactor treating synthetic soy sauce-processing wastewater operated at a psychrophilic temperature (20°C) using an ecogenomic approach with a 16S rRNA gene amplicon, shotgun metagenomic, and metatranscriptomic sequence dataset. Ecogenomics reveals the microorganisms involved in the decomposition of each amino acid, those that may utilize fatty acid byproducts in association with methanogens, and those that may have the ability to support the entire microbial interaction, thereby contributing towards a more detailed understanding of methanogenic amino acid degradation under psychrophilic conditions.
Materials and Methods
Reactor operation
The specifications and operation conditions of an UASB-type anaerobic bioreactor were previously described (Kuroda et al., 2017). Briefly, a lab-scale UASB reactor (working volume of 7.0 L, H×L×W; 0.7×0.1×0.1 m) was operated at 20°C using a water jacket (Fig. S1). Reactors were operated under different hydraulic retention time (HRT) and volumetric COD loading rates ranging between 6 and 8.6 days and between 3.6 and 24 kg COD m–3 d–1, respectively (Table S1 and Fig. S2). Seed sludge samples were taken from an UASB reactor treating synthetic soy sauce wastewater at 35°C. The reactor was fed with synthetic wastewater that mimicked the composition of wastewater discharged from a soy sauce-processing factory (Table S2). Synthetic wastewater was prepared by the dilution of soy sauce (Koikuchi shoyu, Kikkoman) with tap water. Trace elements of CoCl2·6H2O (0.17 mg L–1) and NiCl2·6H2O (0.04 mg L–1) were added to synthetic wastewater.
Analytical methods
Dichromate chemical oxygen demand (CODCr), nitrate, and nitrite were analyzed by a DR3900 spectrophotometer using the COD2 HR kit, NITRA Ver kit, and NITRI Ver3 kit, respectively (Hach). Sulfite and sodium sulfite were analyzed using the Sulfite Test Kit Model SU-5 (Hach). Ammonia and amino acid concentrations were measured by the ACQUITY UPLC H-Class system using the AccQ•Tag Ultra kit according to the manufacturer’s instructions (Waters).
Granule sludge sampling and DNA/RNA extraction
Sludge samples were collected in triplicate from the middle of the reactor (Port 3, Fig. S1) after 905 days of operation at the maximum COD loading rate (6,000 mg COD L–1, Table S1). Samples were stored at –80°C prior to use for DNA/RNA extraction. After centrifugation (8,500×g, 3 min), precipitated sludge samples were collected and stored in a –80°C freezer until DNA extraction. Total DNA and RNA were separately extracted from sludge samples using the FastDNA SPIN Kit for Soil kit (MP Biomedicals) and acid-phenol/beads-beating extraction method (Uyeno et al., 2004), respectively.
16S rRNA gene-based microbial community profiling
PCR and amplicon sequencing were performed as previously described (Kuroda et al., 2015, 2016). Briefly, the PCR amplification of 16S rRNA genes was performed with the universal primer set Univ515F/Univ806R. The PCR reaction mixture (20 μL) contained 2.0 μL of template DNA (10 ng μL–1), 0.5 μM of forward and reverse primers, and 10 μL of the Premix Ex Taq Hot Start Version (TaKaRa Bio). PCR was conducted using a thermal cycler (Veriti200, Applied Biosystems) with the following conditions: initial denaturation at 94°C for 3 min, denaturation at 94°C for 45 s, annealing at 50°C for 60 s, elongation at 72°C for 90 s, and a final extension at 72°C for 10 min. The number of PCR cycles was 25. The purification of PCR products was conducted using a QIAquick PCR purification kit (QIAGEN) following the manufacturer’s protocol. Amplicon sequencing was performed on MiSeq with the MiSeq Reagent v2 kit (Illumina).
Raw 16S rRNA gene reads were processed and trimmed with QIIME 1.9.1 (Caporaso et al., 2010) using sequence length (≥150 nt) and quality score (≥25) cut-offs. Screened sequence data were grouped into operational taxonomic units (OTUs) with the UCLUST algorithm using a 97% sequence identity cut-off (Edgar, 2010). Chimeric sequences for each OTU were removed using ChimeraSlayer (Haas et al., 2011). Taxonomy was assigned using classify-sklearn retained on SILVA database version 138 (Yilmaz et al., 2014).
Metagenomic shotgun sequencing, assembly, and binning
Libraries were prepared with the Nextera XT Library Prep kit (Illumina) with a genomic DNA fragment size ranging between 200 and 1,000 bp. Prepared libraries were sequenced on MiSeq with the MiSeq Reagent v3 kit (Illumina), generating paired-end reads up to 300 bp each, at FASMAC. Assembly and binning were performed as previously described (Nobu et al., 2015, 2016). Briefly, the reads obtained were trimmed using Trimmomatic v0.33 (Bolger et al., 2014); digitally normalized and partitioned using the khmer package (Pell et al., 2012); and assembled using SPAdes v.3.5.0 (Bankevich et al., 2012). The assembled contigs were binned using MetaBAT (Kang et al., 2015) and MaxBin 2.0 (Wu et al., 2014, 2016) with manual curation to eliminate contaminated contigs based on rRNA genes as phylogenetic markers. Short contigs (<1,000 bp) were removed. Genes were predicted using Prodigal v2.5 (Hyatt et al., 2010) and annotated using KEGG (Ogata et al., 1999), BLAST KOALA (Altschul et al., 1990; Kanehisa et al., 2016), IMG/M (Markowitz et al., 2014), and Prokka (Seemann, 2014). The phylogeny and taxonomy of each bin were estimated using PhyloPhlAn software (Segata et al., 2013) and GTDBtk v1.4.1 (GTDB release95; default parameters) (Parks et al., 2018), respectively.
Metatranscriptomic sequencing and mapping
Ribosomal RNA was removed from extracted total RNA by Ribo-Zero Magnetic Kit Bacteria (Illumina). Libraries were prepared with the KAPA RNA HyperPrep Kit (Thermo Fisher Scientific). Prepared libraries were sequenced on NextSeq 500 (Illumina), generating paired-end reads up to 75 bp each, at the Bioengineering Lab. The reads obtained were trimmed using Sickle v1.33 (Joshi and Fass, 2011); mapped using bowtie2 (Langmead and Salzberg, 2012); converted from SAM to BAM formats using Samtools v1.5 (Li et al., 2009); and RPKM were counted using featureCounts v1.5.3 (Liao et al., 2014).
Nucleotide sequence accession numbers
The sequence data obtained in the present study have been deposited under DDBJ/EMBL/GenBank accession no. DRA012164.
Results and Discussion
Reactor operation
The operational performance of the UASB reactor treating synthetic soy sauce-processing wastewater is shown in Table S1 and Fig. S2. Influent synthetic soy sauce wastewater contained VFA, amino acids, and sulfite (Table S2). The reactor was continuously operated for more than 1,000 days. After the start-up period (Phase 1), the removal efficiencies of COD and VFA were consistently maintained at 60–93%. Granular sludge samples were collected in triplicate for technical replicates after 905 days of operation, at which point operation achieved the maximum COD loading rate (6,000 mg COD L–1) and a ≥75% soluble COD removal rate, suggesting that major organic compounds in the wastewater were mostly degraded.
Major microbial constituents and their draft genomes
An overview of the 16S rRNA gene amplicon, shotgun metagenomic, and metatranscriptomic sequencing of sludge samples taken from the UASB reactor treating soy sauce wastewater is shown in Table S3. 16S rRNA gene-based microbial community profiling indicated that the OTUs classified as Firmicutes (28.6%), Synergistota (14.2%), Bacteroidota (12.1%), Halobacteriota (14.6%), and Methanobacteriota (15.5%) predominated in the reactor (Fig. S3 and Table S4). These dominant microbial populations were previously observed in psychrophilic methanogenic bioreactors treating various types of organic wastewater (Collins et al., 2003, 2006; Xing et al., 2009; Gunnigle et al., 2015; Keating et al., 2016, 2018; Petropoulos et al., 2017; McAteer et al., 2020; Paulo et al., 2020; Ribera-Pi et al., 2020).
We successfully obtained 24 high quality (i.e., >80% completeness) draft genomes of major microbial taxa from shotgun metagenomic sequence data (Table S5); i.e., Syntrophomonadaceae (bin23), unclassified Firmicutes_B clade (bin36), Pelotomaculaceae (bin7), Ruminococcaceae (bin28), and Lutisporaceae (bin46) for Firmicutes; Aminobacteriaceae (bin48), Pyramidobacter (bin67), Synergistaceae (bin11m), and Thermovirgaceae (bin39) for Synergistota; Petrimonas (bin12), Mangrovibacterium (bin31m), and uncultured Bacteroidales clades (bin11, bin17, and bin44) for Bacteroidota; Methanothrix (bin1), Methanosarcina (bin13), and Methanospirillum (bin29m) for Halobacteriota; and Methanobacterium (bin3) for Methanobacteriota. Draft genomes associated with Desulfobacterota (Solidesulfovibrio bin8 and Syntrophorhabdaceae bin22), Chloroflexota (Anaerolineaceae bin27), Spirochaetota (Treponematales bin28m), Thermotogota (Mesotoga bin20), and Actinobacteriota (Propionibacteriaceae bin9) were also recovered from the metagenomic sequence pool. The phylogenetic relationship of the metagenomic bins and previously isolated representatives is shown in Fig. 1.
Fig. 1.
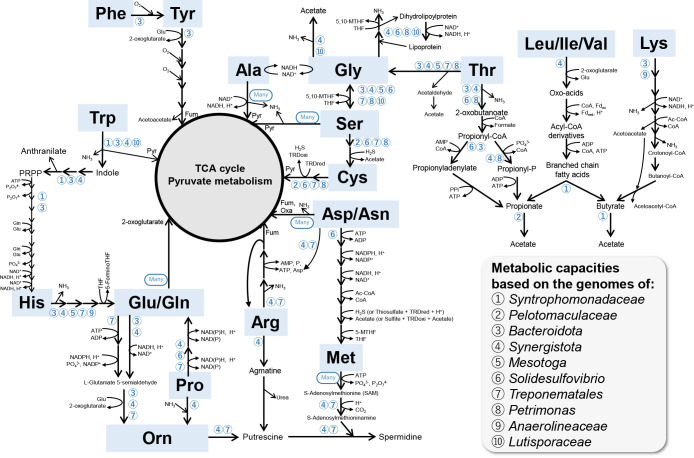
Phylogenetic affiliation and metabolic functions of major microbial constituents in a psychrophilic up-flow anaerobic sludge blanket (UASB) reactor treating synthetic soy sauce wastewater. A distance matrix tree was constructed based on the amino acid sequences of 400 conserved proteins using PhyloPhlAn software. Boldface in the tree indicates the bins obtained in the present study. The averaged RNA expression level (RPKM in triplicate) of each bin is shown as a grayscale heatmap. The metabolic functions of each bin are shown as follows: closed circle, the bin has a complete gene set for each function, and corresponding RNAs were expressed; gray circle, the bin has a partial gene set for each function, and corresponding RNAs were expressed; open circle, although the bin has a complete or partial gene set for each function, corresponding RNAs were not expressed. Amino acids are shown as common three-letter abbreviations according to the International Union of Pure and Applied Chemistry (IUPAC). Other abbreviations: Sec, bacterial secretion system; Pyr, pyruvate; GCS, glycine cleavage system; GRD, glycine reductase; Put, putrescine; Spe, spermidine; SAM, S-adenosyl-l-methionine; Pot, ABC-type polyamine transporter; OpuA, osmotically regulated binding protein-dependent transport system for the osmoprotectant glycine betaine; LIV, branched-chain amino acid transport system; Fum, fumarate; Prop, propionate; BCFA, branched-chain fatty acid; Cyt, cytochrome; Nif, nitrogen fixation complex; ETF, electron transfer flavoprotein; Fix, ETF-oxidizing hydrogenase complex; Fe-S red, Fe-S reductase; Hdr-Flox, heterodisulphide reductase-flavin oxidoreductase complex, Rnf, Rhodobacter nitrogen fixation complex; Eb hyd, electron-bifurcating hydrogenase; Fd hyd, ferredoxin-dependent [NiFe]-hydrogenase; Hya, NADH/quinone-dependent [NiFe]-hydrogenase; Ech, energy-conserving hydrogenase; Mvh, methyl-viologen-reducing hydrogenase; Hox, bidirectional soluble hydrogenase; Eb fdh, electron-bifurcating formate dehydrogenase; Fdo, formate dehydrogenase O.
Transcript levels of annotated genes encoded in each bin are shown in Supplemental Table S5–S30. Among these bins, aceticlastic (Methanothrix bin1), hydrogenotrophic (Methanobacterium bin3 and Methanospirillum bin29m), and methylotrophic (Methanosarcina bin13) methanogens showed relatively high RNA expression levels in methanogenesis-related protein complexes, such as methyl-coenzyme M reductase (Mcr), tetrahydromethanopterin S-methyltransferase (Mtr), CoB--CoM heterodisulfide reductase (Hdr), coenzyme F420-reducing hydrogenase, and formate dehydrogenase (Table S7–S10), indicating stable methanogenic bioconversion. Within the domain Bacteria, Synergistota-related bins (bin11m, bin48, and bin67), Solidesulfovibrio bin8, Pelotomaculaceae bin7, and Syntrophomonadaceae bin23 recorded relatively high transcript levels. They have been suggested to play a role in amino acid degradation (Bhandari and Gupta, 2012), sulfite reduction (Baffert et al., 2019), syntrophic propionate oxidation (Hidalgo-Ahumada et al., 2018), and syntrophic butyrate oxidation (Sieber et al., 2010; Narihiro et al., 2016), respectively, which were inferred from the major components of soy sauce wastewater (Table S1). The potential metabolic functions of dominant bacteria are shown in Fig. 1. Details with a special emphasis on the degradation of amino acids are described in the following sections according to metagenome- and metatranscriptome-informed metabolic reconstruction (Fig. 2, Table S11–S30).
Fig. 2.
Proposed scheme for the methanogenic degradation of amino acids in a psychrophilic up-flow anaerobic sludge blanket (UASB) reactor treating synthetic soy sauce wastewater. In each pathway, representative microbial population(s) are shown as a number in a panel. “Many” indicates that more than 7 representative populations have the corresponding metabolic capacities. Amino acids are shown as a common three-letter abbreviation according to the International Union of Pure and Applied Chemistry (IUPAC). Other abbreviations: PRPP, 5-phospho-alpha-d-ribose 1-diphosphate; THF, tetrahydrofolate; 5-MTHF, 5-methyltetrahydrofolate TRDred, reduced thioredoxin; TRDoxi, oxidized thioredoxin; Fum, fumarate; Pyr, pyruvate; Oxa, oxaloacetate.
Metabolic capacities of dominant organisms
Glutamine, glutamic acid, asparagine, aspartic acid, and alanine
Many microbial constituents utilized Glu/Gln, Asp/Asn, and Ala and produced pyruvate, fumarate, oxaloacetate, and 2-oxoglutarate, which were then used in the citric acid cycle. Gln was generated from Glu and 2-oxoglutarate by glutamate synthase using NAD(P)H as an electron donor. Glutamate dehydrogenase catalyzed the production of 2-oxoglutarate from Gln. Asp was converted to Asn by asparaginase, and aspartate transaminase catalyzed the conversion of Asn to oxaloacetate via an amino-group transfer reaction using 2-oxoglutarate as the substrate and glutamate as the byproduct. Fumarate was produced from Asn by a two-step reaction catalyzed by adenylosuccinate synthetase and adenylosuccinate lyase. Ala may be converted to pyruvate by NADH-dependent alanine dehydrogenase. Glutamate-pyruvate aminotransferase also catalyzed the conversion of Ala to pyruvate via an amino-group transfer reaction via a similar mechanism to aspartate transaminase.
Serine, glycine, threonine, and cysteine
Many bacterial bins harbor a l-serine dehydratase gene cluster that is responsible for the conversion of Ser to pyruvate and NH3 and a serine hydroxymethyltransferase gene for the reversible conversion of Ser and Gly with the co-factor tetrahydrofolate (THF). Gly is further degraded through the glycine cleavage system (GCS) or Stickland reaction with a glycine reductase (GRD) complex. Solidesulfovibrio bin8, Aminobacteriaceae bin48, Lutisporaceae bin46, and Propionibacteriaceae bin9 possess a gene set for GCS (i.e., glycine dehydrogenase, aminomethyltransferase, dihydrolipoyl dehydrogenase, and glycine cleavage system H protein) that catalyzes Gly to NH3 and CO2. The draft genomes of four Synergistota bins and Lutisporaceae bin46 encode a GRD gene cluster (GrdABC) for the conversion of Gly to acetate with the coupled oxidation reaction of some amino acids (e.g., Ala). The partial selenoprotein biosynthetic gene cluster selABCD (l-seryl-tRNA[Sec] selenium transferase, selA; selenocysteine-specific elongation factor, selB; tRNA-Sec, selC selenide, water dikinase, selD) was detected in these GRD-encoded bins because the GrdA subunit contains selenocysteine. Gly was also derived from Thr by l-threonine aldolase found in some of the bacterial bins. Thr may be converted to 2-oxobutanoate and NH3 by threonine ammonia-lyase encoded in the genomes of Bacteroidota, Synergistota, and Propionibacteriaceae, and propionate is then produced via the fermentative degradation of 2-oxobutanoate.
Serine o-acetyltransferase and cysteine synthase are responsible for the conversion of Ser to Cys. Furthermore, l-cysteine:2-oxoglutarate aminotransferase may catalyze Cys and 2-oxoglutarate to 3-mercaptopyruvate and Glu, and 3-mercaptopyruvate may then be transformed to pyruvate by 3-mercaptopyruvate sulfurtransferase with reduced thioredoxin. Solidesulfovibrio bin8, Propionibacteriaceae bin9, Treponematales bin28m, Syntrophorhabdaceae bin22, and Pelotomaculaceae bin7 possess these Ser/Cys utilization enzymes.
Lysine, branched-chain amino acids (BCAA), and fatty acids
The Lys fermentation pathway comprises five enzymes: l-lysine-2,3-aminomutase, β-l-lysine-5,6-aminomutase, 3,5-diaminohexanoate dehydrogenase, 3-keto-5-aminohexanoate cleavage enzyme, and 3-aminobutyryl-CoA ammonia lyase. Through this pathway, Lys was fermented to crotonyl-CoA, and may then be further degraded to acetate via the β-oxidation pathway. Among these enzymes, 3,5-diaminohexanoate dehydrogenase requires NAD+ as a co-factor. Metagenomic bins of Petrimonas bin12, Bacteroidales UBA5261 bin44, and Anaerolineaceae bin27 contain a complete gene set for Lys fermentation, implying their involvement in Lys degradation and butyrate production in this reactor.
Four bins of Synergistota organisms encode BCAA aminotransferase and ferredoxin (Fd)-dependent branched-chain oxo-acid reductase for the degradation of Leu, Ile, and Val to correspond to BCFA: isovalerate, 2-methylbutyrate, and isobutyrate, respectively. The gene cassette for the BCAA transporter (LivFGHKM) was also found in these Synergistota metagenomes. They have no ability to oxidize BCFA due to the lack of 2-methylbutyryl-CoA dehydrogenase, 3-methylbutyryl-CoA dehydrogenase, or isobutyryl-CoA mutase. Multiple active homologs of acyl-CoA dehydrogenase and a gene cassette of isobutyryl-CoA mutase were instead found in Syntrophomonadaceae bin23. In addition, other key enzymes for β-oxidation (i.e., enoyl-CoA hydratase, 3-hydroxybutyryl-CoA dehydrogenase, acetyl-CoA acetyltransferase, phosphotransacetylase, and acetate kinase), the electron transfer flavoprotein (ETF)-oxidizing hydrogenase complex (FixABCX), the heterodisulphide reductase-flavin oxidoreductase (Hdr-Flox) complex, Rhodobacter nitrogen fixation (Rnf) complex, hydrogenases, and formate dehydrogenases were found in Syntrophomonadaceae organisms (Table S11), which are in accordance with the genomic traits of Syntrophomonas strains (Sieber et al., 2010; Narihiro et al., 2016), suggesting its role in the syntrophic oxidation of BCFA and butyrate.
Propionate may be produced by the degradation of 2-methylbutyrate from Ile as well as the degradation of 2-oxobutanoate from Thr, and propionate is then oxidized to acetate via the methylmaronyl-CoA pathway encoded by Pelotomaculaceae bin7. Pelotomaculaceae organisms encode Hdr-Flox, FixABCX, ETF-linked acyl-CoA dehydrogenase, hydrogenases, and formate dehydrogenases (Table S13). Among these energy-conserving systems, the activities of cytoplasmic and membrane-bound formate dehydrogenases were higher than those of hydrogenases. Formate-dominated interspecies electron transfer was demonstrated in our previous ecogenomic studies using a co-culture of Pelotomaculum strains with hydrogenotrophic methanogen (Hidalgo-Ahumada et al., 2018), a lab-scale propionate-fed chemostat (Chen et al., 2020), and full-scale anaerobic digesters (Nobu et al., 2020). In addition to hydrogen/formate-mediated interspecies electron transfer, direct interspecies electron transfer (DIET) employing conductive pili (e-pili) has been investigated (Summers et al., 2010). Pelotomaculaceae bin7 possesses a pilus assembly gene cassette with PilA, which meets the criteria for the e-pili protein (SOY3_bin007_00676, Fig. S4) (Holmes et al., 2017; Walker et al., 2018); however, the transcript level of this cassette is low. In a previous study, the effects of granular-activated carbon (GAC) on pilA gene expression in a psychrophilic (20°C) UASB reactor treating municipal sewage was estimated using a RT-qPCR approach (Zhang et al., 2020). The findings obtained confirmed that the addition of GAC up-regulated pilA gene expression, while the expression levels of pilA were 36-fold lower in non-GAC-amended UASB granular sludge. Therefore, we presume that Pelotomaculaceae bin7 in our psychrophilic UASB reactor has the potential for an e-pili-mediated DIET reaction; however, the transcription level without mediator material is not high. Therefore, the genomic features of Pelotomaculaceae bin7 indicate its versatile syntrophic lifestyle for propionate oxidation in the reactor.
Tryptophan and histidine
The draft genomes of Lutisporaceae bin46, Aminobacteriaceae bin48, Thermovirgaceae bin39, and five Bacteroidota bins harbored tryptophanase, which may catalyze Try to indole, pyruvate, and NH3. Some of these bins possess the ability to convert indole to 5-phospho-alpha-d-ribose 1-diphosphate (PRPP) and anthranilate (2-aminobenzoate). PRPP is a common compound for nucleotide metabolism. Indole and anthranilate are important intermediates for the biosynthesis of alkaloid compounds. Although the absolute abundance and ecophysiological role of indole and its derivatives in methanogenic ecosystems, including our UASB reactor, remain unknown, previous studies reported that these compounds are associated with bacterial signaling in Escherichia coli and Ralstonia solanacearum (Pinero-Fernandez et al., 2011; Song et al., 2020). PRPP may be further converted to His through a 10-step reaction employing ATP, Glu, and NAD+. His may be converted to Glu via a four-step reaction employing histidine ammonia-lyase, urocanate hydratase, imidazolonepropionase, and glutamate formimidoyltransferase by organisms associated with Synergistota, Lutisporaceae, Mesotoga, Bacteroidota, Treponematales, and Anaerolineaceae.
Arginine, methionine, proline, and ornithine
Solidesulfovibrio, Thermovirgaceae, and Treponematales bins harbor two NAD(P)H-dependent enzymes, pyrroline-5-carboxylate reductase and l-glutamate gamma-semialdehyde dehydrogenase, to produce the anaplerotic amino acid Glu from Pro. Ornithine, which is a non-proteinogenic amino acid, may be generated from Glu via a three-step reaction using glutamate 5-kinase, glutamate-5-semialdehyde dehydrogenase, and ornithine aminotransferase, which are encoded within Treponematales bin28m. Three bins of Synergistota organisms (bin48, bin67, and bin39) possess ornithine cyclodeaminase, which catalyzes Pro to Orn. Orn may be converted to putrescine by ornithine decarboxylase encoded in Aminobacteriaceae bin48 and Treponematales bin28m. Putrescine was also produced from Arg by arginine decarboxylase and agmatinase, which are encoded within Thermovirgaceae bin39 and unclassified Firmicutes_B bin36. Four draft genomes (Aminobacteriaceae bin48, Synergistaceae bin11m, Treponematales bin28m, and unclassified Firmicutes_B bin36) encode spermidine synthase, which may catalyze putrescine with S-adenosyl-l-methionine (SAM) to spermidine. Most of the metagenomic draft genomes harbor S-adenosylmethionine synthase to produce SAM from Met. Several organisms possess a gene cassette encoding potential ABC-type polyamine transporter subunits (Pot). Spermidine is a polyamine that is involved in the anaerobic growth of E. coli (Chattopadhyay et al., 2009) and Anaerovibrio lipolytica (Hirao et al., 2000), the microaerobic growth of Borrelia burgdorferi (Bontemps-Gallo et al., 2018), biofilm formation by Yersinia pestis (Patel et al., 2006) and Bacillus subtilis (Burrell et al., 2010), and other physiological functions (Michael, 2018). Zhu et al. (2015) reported that spermidine promoted gene expression and the replacement of damaged proteins in cyanobacteria under cold stress conditions. Some psychrophilic bacteria were shown to accumulate spermidine as a predominant polyamine within their cells (Margesin et al., 2012; Zhang et al., 2012; Dahal et al., 2019; Kumar et al., 2020). These findings imply that spermidine acts as a supportive agent for microbial metabolic activities in methanogenic microbiota in our UASB reactor operated under psychrophilic conditions. Further studies employing a community-level exometabolome approach (Kell et al., 2005; Douglas, 2020) are needed to clarify this assumption.
Phenylalanine and tyrosine
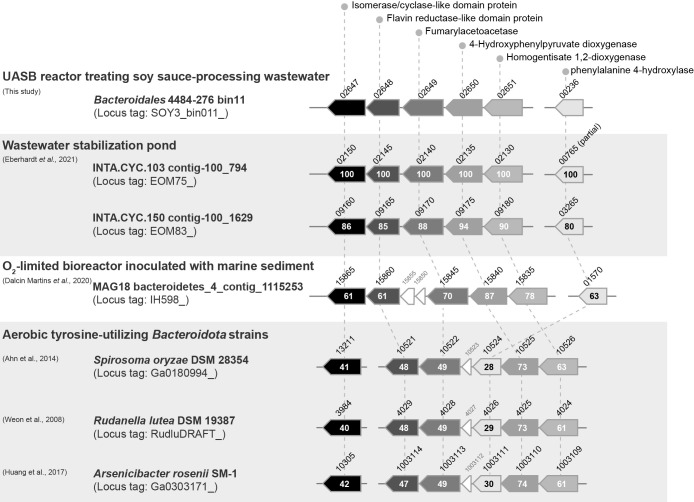
Although the degradation pathway of Phe and Tyr has not yet been elucidated in detail in a methanogenic environment, a bin of uncultured Bacteroidales 4484-276 (bin11) encodes some genes for the aerobic utilization of Phe and Tyr. Phe is degraded to fumarate and acetoacetate by a six-step reaction catalyzed by phenylalanine 4-hydroxylase, l-tyrosine:2-oxoglutarate aminotransferase, 4-hydroxyphenylpyruvate dioxygenase, homogentisate 1,2-dioxygenase, maleylacetoacetate isomerase, and fumarylacetoacetase (Arias-Barrau et al., 2004). Despite rigorous explorations of the gene repertory of all metagenomic sequence data retrieved from UASB granular sludge samples, we did not identify any maleylacetoacetate isomerase gene with significant amino acid similarity to previously reported genes. We found the active RNA expression of a gene encoding the putative isomerase/cyclase-like domain-containing protein (SOY3_bin011_02647) along with a flavin reductase gene (SOY3_bin011_02648), which are adjacent to the genes for homogentisate 1,2-dioxygenase (SOY3_bin011_02651), 4-hydroxyphenylpyruvate dioxygenase (SOY3_bin011_02650), and fumarylacetoacetase (SOY3_bin011_02649) (Fig. 3). The amino acid sequence of the protein SOY3_bin011_02647 showed relatively low identity (~40%) with the proteins of tyrosine-utilizing Bacteroidota isolates, which possess a gene cassette encoding phenylalanine 4-hydroxylase, 4-hydroxyphenylpyruvate dioxygenase, homogentisate 1,2-dioxygenase, and fumarylacetoacetase (Weon et al., 2008; Ahn et al., 2014; Huang et al., 2017). By expanding to the public environmental metagenomic database, three similar gene clusters consisting of homologs of putative isomerase/cyclase-like domain-containing proteins, flavin reductase, homogentisate 1,2-dioxygenase, 4-hydroxyphenylpyruvate dioxygenase, and fumarylacetoacetase, were found in contigs associated with Bacteroidota populations inhabiting ecosystems at low oxygen levels (Eberhardt et al., 2020; Dalcin Martins et al., 2021) (Fig. 3). Among these enzymes, phenylalanine 4-hydroxylase, 4-hydroxyphenylpyruvate dioxygenase, and homogentisate 1,2-dioxygenase require molecular oxygen for reactions to proceed. In our previous ecogenomic study, we revealed that bacterial populations in an anaerobic digester decomposing excess sludge derived from a municipal sewage treatment process survived microaerobic conditions by employing cytochrome bd oxidase, which is a terminal oxidase for aerobic respiration (Nobu et al., 2020). Uncultured Bacteroidales 4484-276 bin11 as well as Syntrophomonadaceae bin23, Solidesulfovibrio bin8, Propionibacteriaceae bin9, and other Bacteroidota bins encode a cytochrome bd oxidase complex that utilizes nanomolar-level oxygen as an electron acceptor to promote oxygen-dependent respiration. Although the exact function of the putative isomerase/cyclase-like domain-containing protein remains unclear, uncultured Bacteroidales 4484-276 bin11 organisms may be responsible for the degradation of Phe and Tyr in the UASB reactor.
Fig. 3.
Comparison of gene cassettes associated with the bioconversion of 4-hydroxyphenylpyruvate to acetoacetate/fumarate found in uncultured Bacteroidales 4484-276 bin11, previously reported metagenomic contigs derived from probable microaerobic ecosystems (Eberhardt et al., 2020; Dalcin Martins et al., 2021), and aerobic Bacteroidota isolates (Weon et al., 2008; Ahn et al., 2014; Huang et al., 2017). An uncultured Bacteroidales 4484-276 bin11-associated cassette encodes homogentisate 1,2-dioxygenase, 4-hydroxyphenylpyruvate dioxygenase, fumarylacetoacetase, flavin reductase-like domain protein, and putative isomerase/cyclase-like domain protein. Phenylalanine 4-hydroxylase is also shown. Abbreviated locus tags are shown (e.g., ‘SOY3_bin011_02651’ as ‘02651’ in the row of Bacteroidales 4484-276 bin11). Numbers in gene boxes indicate amino acid sequence identity (%) to the corresponding gene of uncultured Bacteroidales 4484-276 bin11. The contigs INTA.CYC.103 contig-100_794 (37,009 bp), INTA.CYC.150 contig-100_1629 (24,086 bp), and MAG18 bacteroidetes_4_contig_1115253 (29,273 bp) were originally named as Candidatus Falkowbacteria bacterium (accession no. SABQ01000012), Clostridia bacterium (SABY01000055), and Bacteroidales bacterium (JACZJL010000162), respectively, in the NCBI database. We checked the phylogenetic assignment of these contigs by a blastp search (Altschul et al., 1990) for some functionally important enzymes; i.e., RNA polymerase (locus tag: EOM75_02175), DNA repair protein (EOM75_02190), and chromosomal replication initiator protein (EOM75_02085) for INTA.CYC.103 contig-100_794; RNA polymerase (EOM83_09135), DNA repair protein (EOM83_09120), and aminotransferase (EOM83_09145) for INTA.CYC.150 contig-100_1629; 5′/3′-nucleotidase (IH598_15930), transketolase (IH598_15890), and catalase (IH598_15870) for MAG18 bacteroidetes_4_contig_1115253. Since all enzymes are closely related to Bacteroidota organisms, we temporarily concluded that these contigs may be derived from Bacteroidota-related microorganisms.
Sulfite reduction and possible nitrogen fixation
Soy sauce-processing wastewater contains a high concentration of sodium sulfite (Table S2). Among the major bacterial constituents, only Solidesulfovibrio bin8 encodes the complete gene set for sulfate/sulfite reduction comprising sulfate adenylyltransferase (sat), adenylyl-sulfate reductase (aprAB), the dissimilatory sulfite reductase catalytic subunit (dsrABCD), and membrane-bound sulfite reductase-associated electron transfer protein subunit (dsrMKJOP) with relatively high transcript levels. According to its genome information, this Solidesulfovibrio-related organism may utilize Gly, Ser, Cys, Pro, Thr, Asp/Asn, and Glu/Gln, but does not oxidize fatty acids (e.g., propionate and butyrate). The feature of amino acid utilization is in partial agreement with the ability of previously known amino acid-utilizing Solidesulfovibrio (formerly known as Desulfovibrio) spp. isolated from anaerobic wastewater treatment systems (Baena et al., 1998; Hernandez-Eugenio et al., 2000) and a natural psychrophilic environment (Pecheritsyna et al., 2012). The presence of a Desulfovibrionaceae-related population was previously reported in the 16S rRNA-based microbial community profiling of psychrophilic anaerobic bioreactor ecosystems (McKeown et al., 2009; Keating et al., 2016, 2018; McAteer et al., 2020; Ribera-Pi et al., 2020); however, their ecophysiological function has yet to be clarified. Sulfate-reducing bacteria and methanogenic archaea compete for a carbon source and/or H2 in an anaerobic bioreactor treating sulfate-/sulfite-rich wastewater (Raskin et al., 1996; Omil et al., 1998; Lu et al., 2017; Wu et al., 2018). Through a genome analysis of Solidesulfovibrio bin8, we found the Hdr-Flox complex, an energy-conserving hydrogenase (Ech), membrane-bound cytochrome, and carbon monoxide oxidation (Coo)-hydrogenase with carbon monoxide dehydrogenase, which may catalyze the reversible conversion between CO and CO2, flavoredoxin, and NADH dehydrogenase, all of which are responsible for acquiring ATP coupling with sulfate/sulfite reduction, in members of Desulfovibrionaceae (Chen et al., 1993; Voordouw, 2002; Meyer et al., 2014; Hadj-Said et al., 2015). A nitrogenase-like gene cluster was detected in the genome along with molybdate and ammonia transporters (Table S25). Previous microbial genome analyses indicated that nitrogen fixation-related proteins (Nif) are distributed in functionally diverse microorganisms including fermentative bacteria, syntrophic substrate oxidizers, methanogenic archaea, and sulfate-reducing bacteria (Dos Santos et al., 2012; Narihiro et al., 2016). The transcript levels of nitrogenase-like gene clusters were abundant in Solidesulfovibrio bin8 (Table S25) as well as in the methanogenic archaea (Methanothrix bin1, Methanobacterium bin3, Methanospirillum bin29m, and Methanosarcina bin13) (Table S7–10). We speculated that nitrogen fixation in some syntrophs having Nif-related genes may serve as a mechanism to tolerate acidification by providing hydrogen and ammonia for partner hydrogenotrophic methanogens to survive under hydrogen/ammonia-limited conditions (Narihiro et al., 2016). Sayavedra et al. (2021) recently reported that the sulfate-reducing bacterium Desulfovibrio diazotrophicus strain QI0027 isolated from the human gut exhibited nitrogen fixation activity. These findings suggest that Desulfovibrionaceae organisms interact with other trophic populations in the UASB reactor by providing hydrogen and ammonia, particularly under psychrophilic conditions in which the growth rate and metabolic activity of methanogens are lower than those under mesophilic and thermophilic conditions (Lettinga et al., 2001).
In summary, microbial metabolic functions associated with the methanogenic degradation of amino acids in a psychrophilic UASB reactor treating soy sauce-processing wastewater were investigated using an ecogenomic approach. The results obtained demonstrated that members of Bacteroidota, Synergistota, Treponematales, and Propionibacteriaceae were the major active populations for the anaerobic fermentative degradation of amino acids. Exceptionally, Phe and Tyr were degraded by the unique enzymes encoded in the uncultured Bacteroidota-related organism that may function under microaerobic conditions. Fatty acids resulting from the degradation of the BCAA, Lys and Thr were further oxidized by Pelotomaculaceae- and Syntrophomonadaceae-related bacteria in syntrophic association with partner hydrogenotrophic methanogens. Solidesulfovibrio organisms play a major role in the reduction of sulfite, which is abundant in soy sauce wastewater, and may support the activity of hydrogenotrophic methanogens and other microbial populations by providing hydrogen and ammonia via the Nif-like complex. We consider the maintenance of this sophisticated metabolic network of functionally diverse microbes to be essential for the methanogenic wastewater treatment process under low temperature conditions.
Citation
Kuroda, K., Narihiro, T., Nobu, M. K.., Tobo, A., Yamauchi, M., and Yamada, M. (2021) Ecogenomics Reveals Microbial Metabolic Networks in a Psychrophilic Methanogenic Bioreactor Treating Soy Sauce Production Wastewater. Microbes Environ 36: ME21045.
https://doi.org/10.1264/jsme2.ME21045
Supplementary Material
Acknowledgements
The present study was supported by The Japan Society for the Promotion of Science with Grants-in-Aid for Scientific Research No. 18H01576 and 20K04763. Sequence data submission was supported by the D-way Submission Portal provided by the DNA Data Bank of Japan (DDBJ). We thank Ms. Hazuki Kurashita (Nagaoka University of Technology, Japan) for her technical assistance.
References
- Ahn, J.H., Weon, H.Y., Kim, S.J., Hong, S.B., Seok, S.J., and Kwon, S.W. (2014) Spirosoma oryzae sp. nov., isolated from rice soil and emended description of the genus Spirosoma. Int J Syst Evol Microbiol 64: 3230–3234. [DOI] [PubMed] [Google Scholar]
- Altschul, S.F., Gish, W., Miller, W., Myers, E.W., and Lipman, D.J. (1990) Basic local alignment search tool. J Mol Biol 215: 403–410. [DOI] [PubMed] [Google Scholar]
- Arias-Barrau, E., Olivera, E.R., Luengo, J.M., Fernandez, C., Galan, B., Garcia, J.L., et al. (2004) The homogentisate pathway: a central catabolic pathway involved in the degradation of l-phenylalanine, l-tyrosine, and 3-hydroxyphenylacetate in Pseudomonas putida. J Bacteriol 186: 5062–5077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baena, S., Fardeau, M.L., Labat, M., Ollivier, B., Garcia, J.L., and Patel, B.K. (1998) Desulfovibrio aminophilus sp. nov., a novel amino acid degrading and sulfate reducing bacterium from an anaerobic dairy wastewater lagoon. Syst Appl Microbiol 21: 498–504. [DOI] [PubMed] [Google Scholar]
- Baffert, C., Kpebe, A., Avilan, L., and Brugna, M. (2019) Hydrogenases and H2 metabolism in sulfate-reducing bacteria of the Desulfovibrio genus. Adv Microb Physiol 74: 143–189. [DOI] [PubMed] [Google Scholar]
- Bankevich, A., Nurk, S., Antipov, D., Gurevich, A.A., Dvorkin, M., Kulikov, A.S.,et al. (2012) SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J Comput Biol 19: 455–477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhandari, V., and Gupta, R.S. (2012) Molecular signatures for the phylum Synergistetes and some of its subclades. Antonie van Leeuwenhoek 102: 517–540. [DOI] [PubMed] [Google Scholar]
- Bolger, A.M., Lohse, M., and Usadel, B. (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30: 2114–2120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bontemps-Gallo, S., Lawrence, K.A., Richards, C.L., and Gherardini, F.C. (2018) Borrelia burgdorferi genes, bb0639-0642, encode a putative putrescine/spermidine transport system, PotABCD, that is spermidine specific and essential for cell survival. Mol Microbiol 108: 350–360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burrell, M., Hanfrey, C.C., Murray, E.J., Stanley-Wall, N.R., and Michael, A.J. (2010) Evolution and multiplicity of arginine decarboxylases in polyamine biosynthesis and essential role in Bacillus subtilis biofilm formation. J Biol Chem 285: 39224–39238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caporaso, J.G., Kuczynski, J., Stombaugh, J., Bittinger, K., Bushman, F.D., Costello, E.K., et al. (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7: 335–336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chattopadhyay, M.K., Tabor, C.W., and Tabor, H. (2009) Polyamines are not required for aerobic growth of Escherichia coli: preparation of a strain with deletions in all of the genes for polyamine biosynthesis. J Bacteriol 191: 5549–5552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen, L., Liu, M.Y., and LeGall, J. (1993) Isolation and characterization of flavoredoxin, a new flavoprotein that permits in vitro reconstitution of an electron transfer chain from molecular hydrogen to sulfite reduction in the bacterium Desulfovibrio gigas. Arch Biochem Biophys 303: 44–50. [DOI] [PubMed] [Google Scholar]
- Chen, Y.T., Zeng, Y., Wang, H.Z., Zheng, D., Kamagata, Y., Narihiro, T., et al. (2020) Different interspecies electron transfer patterns during mesophilic and thermophilic syntrophic propionate degradation in chemostats. Microb Ecol 80: 120–132. [DOI] [PubMed] [Google Scholar]
- Collins, G., Woods, A., McHugh, S., Carton, M.W., and O’Flaherty, V. (2003) Microbial community structure and methanogenic activity during start-up of psychrophilic anaerobic digesters treating synthetic industrial wastewaters. FEMS Microbiol Ecol 46: 159–170. [DOI] [PubMed] [Google Scholar]
- Collins, G., Mahony, T., and O’Flaherty, V. (2006) Stability and reproducibility of low-temperature anaerobic biological wastewater treatment. FEMS Microbiol Ecol 55: 449–458. [DOI] [PubMed] [Google Scholar]
- Conrad, R., and Wetter, B. (1990) Influence of temperature on energetics of hydrogen metabolism in homoacetogenic, methanogenic, and other anaerobic-bacteria. Arch Microbiol 155: 94–98. [Google Scholar]
- Dahal, R.H., Chaudhary, D.K., Kim, D.U., and Kim, J. (2019) Description of Sphingobium psychrophilum sp. nov., a cold-adapted bacterium isolated from Arctic soil. Int J Syst Evol Microbiol 71: doi: 10.1099/ijsem.0.004705. [DOI] [PubMed]
- Dalcin Martins, P., de Jong, A., Lenstra, W.K., van Helmond, N., Slomp, C.P., Jetten, M.S.M., et al. (2021) Enrichment of novel Verrucomicrobia, Bacteroidetes, and Krumholzibacteria in an oxygen-limited methane- and iron-fed bioreactor inoculated with Bothnian Sea sediments. MicrobiologyOpen 10: e1175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dos Santos, P.C., Fang, Z., Mason, S.W., Setubal, J.C., and Dixon, R. (2012) Distribution of nitrogen fixation and nitrogenase-like sequences amongst microbial genomes. BMC Genomics 13: 162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Douglas, A.E. (2020) The microbial exometabolome: ecological resource and architect of microbial communities. Philos Trans R Soc Lond B Biol Sci 375: 20190250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eberhardt, M.F., Irazoqui, J.M., and Amadio, A.F. (2020) Beta-galactosidases from a sequence-based metagenome: Cloning, expression, purification and characterization. Microorganisms 9: doi: 10.3390/microorganisms9010055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edgar, R.C. (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26: 2460–2461. [DOI] [PubMed] [Google Scholar]
- Gunnigle, E., Nielsen, J.L., Fuszard, M., Botting, C.H., Sheahan, J., O’Flaherty, V., and Abram, F. (2015) Functional responses and adaptation of mesophilic microbial communities to psychrophilic anaerobic digestion. FEMS Microbiol Ecol 91: fiv132. [DOI] [PubMed] [Google Scholar]
- Haas, B.J., Gevers, D., Earl, A.M., Feldgarden, M., Ward, D.V., Giannoukos, G., et al. (2011) Chimeric 16S rRNA sequence formation and detection in Sanger and 454-pyrosequenced PCR amplicons. Genome Res 21: 494–504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hadj-Said, J., Pandelia, M.E., Leger, C., Fourmond, V., and Dementin, S. (2015) The carbon monoxide dehydrogenase from Desulfovibrio vulgaris. Biochim Biophys Acta 1847: 1574–1583. [DOI] [PubMed] [Google Scholar]
- Hernandez-Eugenio, G., Fardeau, M.L., Patel, B.K.C., Macarie, H., Garcia, J.L., and Ollivier, B. (2000) Desulfovibrio mexicanus sp. nov., a sulfate-reducing bacterium isolated from an upflow anaerobic sludge blanket (UASB) reactor treating cheese wastewaters. Anaerobe 6: 305–312. [Google Scholar]
- Hidalgo-Ahumada, C.A.P., Nobu, M.K., Narihiro, T., Tamaki, H., Liu, W.T., Kamagata, Y., et al. (2018) Novel energy conservation strategies and behaviour of Pelotomaculum schinkii driving syntrophic propionate catabolism. Environ Microbiol 20: 4503–4511. [DOI] [PubMed] [Google Scholar]
- Hirao, T., Sato, M., Shirahata, A., and Kamio, Y. (2000) Covalent linkage of polyamines to peptidoglycan in Anaerovibrio lipolytica. J Bacteriol 182: 1154–1157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holmes, D.E., Shrestha, P.M., Walker, D.J.F., Dang, Y., Nevin, K.P., Woodard, T.L., and Lovley, D.R. (2017) Metatranscriptomic evidence for direct interspecies electron transfer between Geobacter and Methanothrix species in methanogenic rice paddy soils. Appl Environ Microbiol 83: e00223-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang, K., Xu, Y., Zhang, J., Chen, C., Gao, F., and Zhao, F.J. (2017) Arsenicibacter rosenii gen. nov., sp. nov., an efficient arsenic methylating and volatilizing bacterium isolated from an arsenic-contaminated paddy soil. Int J Syst Evol Microbiol 67: 3186–3191. [DOI] [PubMed] [Google Scholar]
- Hyatt, D., Chen, G.L., Locascio, P.F., Land, M.L., Larimer, F.W., and Hauser, L.J. (2010) Prodigal: prokaryotic gene recognition and translation initiation site identification. BMC Bioinf 11: 119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joshi, N.A., and Fass, J.N. (2011) Sickle: A sliding-window, adaptive, quality-based trimming tool for FastQ files (Version 1.33). URL https://github.com/najoshi/sickle [Google Scholar]
- Kanehisa, M., Sato, Y., and Morishima, K. (2016) BlastKOALA and GhostKOALA: KEGG tools for functional characterization of genome and metagenome sequences. J Mol Biol 428: 726–731. [DOI] [PubMed] [Google Scholar]
- Kang, D.D., Froula, J., Egan, R., and Wang, Z. (2015) MetaBAT, an efficient tool for accurately reconstructing single genomes from complex microbial communities. PeerJ 3: e1165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keating, C., Chin, J.P., Hughes, D., Manesiotis, P., Cysneiros, D., Mahony, T., et al. (2016) Biological phosphorus removal during high-rate, low-temperature, anaerobic digestion of wastewater. Front Microbiol 7: 226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keating, C., Hughes, D., Mahony, T., Cysneiros, D., Ijaz, U.Z., Smith, C.J., and O’Flaherty, V. (2018) Cold adaptation and replicable microbial community development during long-term low-temperature anaerobic digestion treatment of synthetic sewage. FEMS Microbiol Ecol 94: fiy095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kell, D.B., Brown, M., Davey, H.M., Dunn, W.B., Spasic, I., and Oliver, S.G. (2005) Metabolic footprinting and systems biology: the medium is the message. Nat Rev Microbiol 3: 557–565. [DOI] [PubMed] [Google Scholar]
- Kumar, S., Suyal, D.C., Yadav, A., Shouche, Y., and Goel, R. (2020) Psychrophilic Pseudomonas helmanticensis proteome under simulated cold stress. Cell Stress Chaperones 25: 1025–1032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuroda, K., Chosei, T., Nakahara, N., Hatamoto, M., Wakabayashi, T., Kawai, T., et al. (2015) High organic loading treatment for industrial molasses wastewater and microbial community shifts corresponding to system development. Bioresour Technol 196: 225–234. [DOI] [PubMed] [Google Scholar]
- Kuroda, K., Nobu, M., Mei, R., Narihiro, T., Bocher, B., Yamaguchi, T., and Liu, W. (2016) A single-granule-level approach reveals ecological heterogeneity in an upflow anaerobic sludge blanket reactor. PLoS One 11: e0167788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuroda, K., Tobo, A., Hatamoto, M., Yamaguchi, T., Watanabe, N., Nanjo, T., et al. (2017) High organic loading treatment of synthetic soy-sauce production wastewater using a combined system consisting of a psychrophilic (20°C) UASB reactor and a DHS reactor at ambient temperature. J Jpn Soc Water Environ 40: 67–75. [Google Scholar]
- Langmead, B., and Salzberg, S.L. (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9: 357–359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lettinga, G. (1995) Anaerobic digestion and wastewater treatment systems. Antonie van Leeuwenhoek 67: 3–28. [DOI] [PubMed] [Google Scholar]
- Lettinga, G., Rebac, S., Parshina, S., Nozhevnikova, A., van Lier, J.B., and Stams, A.J. (1999) High-rate anaerobic treatment of wastewater at low temperatures. Appl Environ Microbiol 65: 1696–1702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lettinga, G., Rebac, S., and Zeeman, G. (2001) Challenge of psychrophilic anaerobic wastewater treatment. Trends Biotechnol 19: 363–370. [DOI] [PubMed] [Google Scholar]
- Li, H., Handsaker, B., Wysoker, A., Fennell, T., Ruan, J., Homer, N., et al. (2009) The Sequence Alignment/Map format and SAMtools. Bioinformatics 25: 2078–2079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liao, Y., Smyth, G.K., and Shi, W. (2014) featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30: 923–930. [DOI] [PubMed] [Google Scholar]
- Lioe, H.N., Selamat, J., and Yasuda, M. (2010) Soy sauce and its umami taste: a link from the past to current situation. J Food Sci 75: R71–76. [DOI] [PubMed] [Google Scholar]
- Lu, X., Zhen, G., Ni, J., Kubota, K., and Li, Y.Y. (2017) Sulfidogenesis process to strengthen re-granulation for biodegradation of methanolic wastewater and microorganisms evolution in an UASB reactor. Water Res 108: 137–150. [DOI] [PubMed] [Google Scholar]
- Margesin, R., Zhang, D.C., and Busse, H.J. (2012) Sphingomonas alpina sp. nov., a psychrophilic bacterium isolated from alpine soil. Int J Syst Evol Microbiol 62: 1558–1563. [DOI] [PubMed] [Google Scholar]
- Markowitz, V.M., Chen, I.M.A., Chu, K., Szeto, E., Palaniappan, K., Pillay, M., et al. (2014) IMG/M 4 version of the integrated metagenome comparative analysis system. Nucleic Acids Res 42: D568–D573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McAteer, P.G., Christine Trego, A., Thorn, C., Mahony, T., Abram, F., and O’Flaherty, V. (2020) Reactor configuration influences microbial community structure during high-rate, low-temperature anaerobic treatment of dairy wastewater. Bioresour Technol 307: 123221. [DOI] [PubMed] [Google Scholar]
- McKeown, R.M., Scully, C., Enright, A.M., Chinalia, F.A., Lee, C., Mahony, T., et al. (2009) Psychrophilic methanogenic community development during long-term cultivation of anaerobic granular biofilms. ISME J 3: 1231–1242. [DOI] [PubMed] [Google Scholar]
- Meyer, B., Kuehl, J.V., Price, M.N., Ray, J., Deutschbauer, A.M., Arkin, A.P., and Stahl, D.A. (2014) The energy-conserving electron transfer system used by Desulfovibrio alaskensis strain G20 during pyruvate fermentation involves reduction of endogenously formed fumarate and cytoplasmic and membrane-bound complexes, Hdr-Flox and Rnf. Environ Microbiol 16: 3463–3486. [DOI] [PubMed] [Google Scholar]
- Michael, A.J. (2018) Polyamine function in archaea and bacteria. J Biol Chem 293: 18693–18701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Narihiro, T., Nobu, M.K., Tamaki, H., Kamagata, Y., Sekiguchi, Y., and Liu, W.T. (2016) Comparative genomics of syntrophic branched-chain fatty acid degrading bacteria. Microbes Environ 31: 288–292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nobu, M.K., Narihiro, T., Rinke, C., Kamagata, Y., Tringe, S.G., Woyke, T., and Liu, W.T. (2015) Microbial dark matter ecogenomics reveals complex synergistic networks in a methanogenic bioreactor. ISME J 9: 1710–1722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nobu, M., Narihiro, T., Kuroda, K., Mei, R., and Liu, W. (2016) Chasing the elusive Euryarchaeota class WSA2: genomes reveal a uniquely fastidious methylreducing methanogen. ISME J 10: 2478–2487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nobu, M.K., Narihiro, T., Mei, R., Kamagata, Y., Lee, P., Lee, P., et al. (2020) Catabolism and interactions of uncultured organisms shaped by eco-thermodynamics in methanogenic bioprocesses. Microbiome 8: 111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogata, H., Goto, S., Sato, K., Fujibuchi, W., Bono, H., and Kanehisa, M. (1999) KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res 27: 29–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Omil, F., Lens, P., Visser, A., Hulshoff Pol, L.W., and Lettinga, G. (1998) Long-term competition between sulfate reducing and methanogenic bacteria in UASB reactors treating volatile fatty acids. Biotechnol Bioeng 57: 676–685. [PubMed] [Google Scholar]
- Parks, D.H., Chuvochina, M., Waite, D.W., Rinke, C., Skarshewski, A., Chaumeil, P.A., and Hugenholtz, P. (2018) A standardized bacterial taxonomy based on genome phylogeny substantially revises the tree of life. Nat Biotechnol 36: 996–1004. [DOI] [PubMed] [Google Scholar]
- Patel, C.N., Wortham, B.W., Lines, J.L., Fetherston, J.D., Perry, R.D., and Oliveira, M.A. (2006) Polyamines are essential for the formation of plague biofilm. J Bacteriol 188: 2355–2363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paulo, L.M., Castilla-Archilla, J., Ramiro-Garcia, J., Escamez-Picon, J.A., Hughes, D., Mahony, T., et al. (2020) Microbial community redundancy and resilience underpins high-rate anaerobic treatment of dairy-processing wastewater at ambient temperatures. Front Bioeng Biotechnol 8: 192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pecheritsyna, S.A., Rivkina, E.M., Akimov, V.N., and Shcherbakova, V.A. (2012) Desulfovibrio arcticus sp. nov., a psychrotolerant sulfate-reducing bacterium from a cryopeg. Int J Syst Evol Microbiol 62: 33–37. [DOI] [PubMed] [Google Scholar]
- Pell, J., Hintze, A., Canino-Koning, R., Howe, A., Tiedje, J.M., and Brown, C.T. (2012) Scaling metagenome sequence assembly with probabilistic de Bruijn graphs. Proc Natl Acad Sci U S A 109: 13272–13277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petropoulos, E., Dolfing, J., Davenport, R.J., Bowen, E.J., and Curtis, T.P. (2017) Developing cold-adapted biomass for the anaerobic treatment of domestic wastewater at low temperatures (4, 8 and 15 degrees C) with inocula from cold environments. Water Res 112: 100–109. [DOI] [PubMed] [Google Scholar]
- Pinero-Fernandez, S., Chimerel, C., Keyser, U.F., and Summers, D.K. (2011) Indole transport across Escherichia coli membranes. J Bacteriol 193: 1793–1798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raskin, L., Rittmann, B.E., and Stahl, D.A. (1996) Competition and coexistence of sulfate-reducing and methanogenic populations in anaerobic biofilms. Appl Environ Microbiol 62: 3847–3857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ribera-Pi, J., Campitelli, A., Badia-Fabregat, M., Jubany, I., Martinez-Llado, X., McAdam, E., et al. (2020) Hydrolysis and methanogenesis in UASB-AnMBR treating municipal wastewater under psychrophilic conditions: Importance of reactor configuration and inoculum. Front Bioeng Biotechnol 8: 567695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sayavedra, L., Li, T., Bueno Batista, M., Seah, B.K.B., Booth, C., Zhai, Q., et al. (2021) Desulfovibrio diazotrophicus sp. nov., a sulfate-reducing bacterium from the human gut capable of nitrogen fixation. Environ Microbiol 23: 3164–3181. [DOI] [PubMed] [Google Scholar]
- Schink, B., and Stams, A.J.M. (2013) Syntrophism among prokaryotes. In The Prokaryotes, 4th edn. Rosenberg, E., DeLong, E., Lory, S., Stackebrandt, E., and Thompson, F. (eds). Berlin Heidelberg: Springer-Verlag, pp. 471–493. [Google Scholar]
- Seemann, T. (2014) Prokka: rapid prokaryotic genome annotation. Bioinformatics 30: 2068–2069. [DOI] [PubMed] [Google Scholar]
- Segata, N., Börnigen, D., Morgan, X.C., and Huttenhower, C. (2013) PhyloPhlAn is a new method for improved phylogenetic and taxonomic placement of microbes. Nat Commun 4: 2304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sieber, J.R., Sims, D.R., Han, C., Kim, E., Lykidis, A., Lapidus, A.L., et al. (2010) The genome of Syntrophomonas wolfei: new insights into syntrophic metabolism and biohydrogen production. Environ Microbiol 12: 2289–2301. [DOI] [PubMed] [Google Scholar]
- Song, S., Yin, W., Sun, X., Cui, B., Huang, L., Li, P., et al. (2020) Anthranilic acid from Ralstonia solanacearum plays dual roles in intraspecies signalling and inter-kingdom communication. ISME J 14: 2248–2260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Summers, Z.M., Fogarty, H.E., Leang, C., Franks, A.E., Malvankar, N.S., and Lovley, D.R. (2010) Direct exchange of electrons within aggregates of an evolved syntrophic coculture of anaerobic bacteria. Science 330: 1413–1415. [DOI] [PubMed] [Google Scholar]
- Uyeno, Y., Sekiguchi, Y., Sunaga, A., Yoshida, H., and Kamagata, Y. (2004) Sequence-specific cleavage of small-subunit (SSU) rRNA with oligonucleotides and RNase H: a rapid and simple approach to SSU rRNA-based quantitative detection of microorganisms. Appl Environ Microbiol 70: 3650–3663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voordouw, G. (2002) Carbon monoxide cycling by Desulfovibrio vulgaris Hildenborough. J Bacteriol 184: 5903–5911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker, D.J., Adhikari, R.Y., Holmes, D.E., Ward, J.E., Woodard, T.L., Nevin, K.P., and Lovley, D.R. (2018) Electrically conductive pili from pilin genes of phylogenetically diverse microorganisms. ISME J 12: 48–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weon, H.Y., Noh, H.J., Son, J.A., Jang, H.B., Kim, B.Y., Kwon, S.W., and Stackebrandt, E. (2008) Rudanella lutea gen. nov., sp. nov., isolated from an air sample in Korea. Int J Syst Evol Microbiol 58: 474–478. [DOI] [PubMed] [Google Scholar]
- Wu, J., Niu, Q., Li, L., Hu, Y., Mribet, C., Hojo, T., and Li, Y.Y. (2018) A gradual change between methanogenesis and sulfidogenesis during a long-term UASB treatment of sulfate-rich chemical wastewater. Sci Total Environ 636: 168–176. [DOI] [PubMed] [Google Scholar]
- Wu, Y.W., Tang, Y.H., Tringe, S.G., Simmons, B.A., and Singer, S.W. (2014) MaxBin: an automated binning method to recover individual genomes from metagenomes using an expectation-maximization algorithm. Microbiome 2: 26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu, Y.W., Simmons, B.A., and Singer, S.W. (2016) MaxBin 2.0: an automated binning algorithm to recover genomes from multiple metagenomic datasets. Bioinformatics 32: 605–607. [DOI] [PubMed] [Google Scholar]
- Xing, W., Zuo, J.E., Dai, N., Cheng, J., and Li, J. (2009) Reactor performance and microbial community of an EGSB reactor operated at 20 and 15 degrees C. J Appl Microbiol 107: 848–857. [DOI] [PubMed] [Google Scholar]
- Yilmaz, P., Parfrey, L.W., Yarza, P., Gerken, J., Pruesse, E., Quast, C., et al. (2014) The SILVA and “All-species Living Tree Project (LTP)” taxonomic frameworks. Nucleic Acids Res 42: D643–D648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang, D.C., Busse, H.J., Wieser, C., Liu, H.C., Zhou, Y.G., Schinner, F., and Margesin, R. (2012) Candidimonas bauzanensis sp. nov., isolated from soil, and emended description of the genus Candidimonas Vaz-Moreira et al. 2011. Int J Syst Evol Microbiol 62: 2084–2089. [DOI] [PubMed] [Google Scholar]
- Zhang, Y.D., Zhang, L., Guo, B., Zhou, Y., Gao, M.J., Sharaf, A., and Liu, Y. (2020) Granular activated carbon stimulated microbial physiological changes for enhanced anaerobic digestion of municipal sewage. Chem Eng J 400: 125838. [Google Scholar]
- Zhu, X., Li, Q., Yin, C., Fang, X., and Xu, X. (2015) Role of spermidine in overwintering of cyanobacteria. J Bacteriol 197: 2325–2334. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.