Abstract
Background: Immunizations have been previously described as potential triggering events for the development of certain glomerular diseases. However, glomerular disease occurrences are being reported after exposure to a severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccine.
Case Report: A 50-year-old male presented to a nephrology clinic for evaluation of persistent proteinuria. Six weeks prior to evaluation, the patient had reported developing a rash 2 weeks after receiving the first dose of a SARS-CoV-2 vaccine (BNT162b2 mRNA, Pfizer, Inc). His primary care provider treated the rash with corticosteroids, leading to partial improvement of the skin lesions. Three weeks after the first vaccine injection, the patient received his scheduled second vaccine injection. Within 2 days, the rash reappeared. This time, the lesions were more severe in nature. Skin biopsy revealed immunoglobulin A (IgA)-dominant leukocytoclastic vasculitis. After the patient completed 2 weeks of oral corticosteroids, urinalysis revealed proteinuria, and consultation with nephrology was requested. On examination, healing papules were noted on his legs. Serum creatinine 2 weeks after the second dose of vaccine was 0.9 mg/dL. Microscopic examination of the urinary sediment revealed acanthocytes. Urine protein to creatinine ratio 3 weeks after the second dose of vaccine was 1.1 g/day. Serum complements were normal, and all pertinent serology was negative. Kidney biopsy findings were consistent with IgA nephropathy.
Conclusion: The clinical presentation and pathologic findings in this case strongly suggest that the Pfizer SARS-CoV-2 vaccine can trigger a clinical syndrome compatible with Henoch-Schönlein purpura. The recurrence of the rash following the second dose argues for a definite causal association by the Naranjo criteria.
Keywords: BNT162b2 vaccine, COVID-19, glomerulonephritis, glomerulonephritis–IgA, purpura–Schonlein-Henoch, vaccines
INTRODUCTION
Individuals affected by coronavirus disease 2019 (COVID-19) may be at risk of acquiring certain forms of glomerular disease.1,2 Reports of acute glomerular syndromes have emerged since the widespread vaccination against infection by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) occurred across the globe between January and June 2021.3,4 Because immunizations have been previously described to trigger certain forms of glomerular disease,5-7 the recent reports of such events occurring after COVID-19 vaccination are not entirely surprising. However, data are still scarce regarding what types of glomerular pathologies can be elicited by COVID-19 vaccines. Reports published in 2021 describe cases of immunoglobulin A (IgA) nephropathy relapsing after exposure to COVID-19 vaccination.8-11 However, to our knowledge, de novo IgA vasculitis in an adult following a COVID-19 vaccine has not been previously reported.
We describe the case of a 50-year-old male who developed dermatologic and renal manifestations of IgA vasculitis after receiving a SARS-CoV-2 vaccine.
CASE REPORT
A 50-year-old male presented to a nephrology clinic for evaluation of persistent proteinuria. His medical history was only pertinent for seasonal allergy and a mild COVID-19 infection that did not require hospital admission 4 months prior to evaluation. He was taking no medications. Six weeks prior to presentation, the patient had developed a skin rash on his lower legs that erupted 2 weeks after he received the first dose of a SARS-CoV-2 mRNA vaccine (BNT162b2 (mRNA), Pfizer, Inc) (Figure 1). His primary care provider treated the rash with fluocinonide 0.05% cream, applied to the affected area twice daily for 1 to 2 weeks. Three weeks after the first vaccine injection, the patient received his scheduled second vaccine injection. Within 2 days, the skin rash reappeared. This time, the lesions were more severe in nature, with violaceous nonblanching papules and blisters involving the same area on his lower legs but also affecting his thighs, the dorsal aspect of his forearms, lower abdomen, upper back, and buttocks. He reported concomitant myalgias. He denied gross hematuria or dark or foamy urine. Skin biopsy revealed IgA-dominant leukocytoclastic vasculitis. The patient was treated with prednisone for 3 weeks: 60 mg daily for the first week, 40 mg daily for the second week, and 20 mg daily for the third week.
Figure 1.
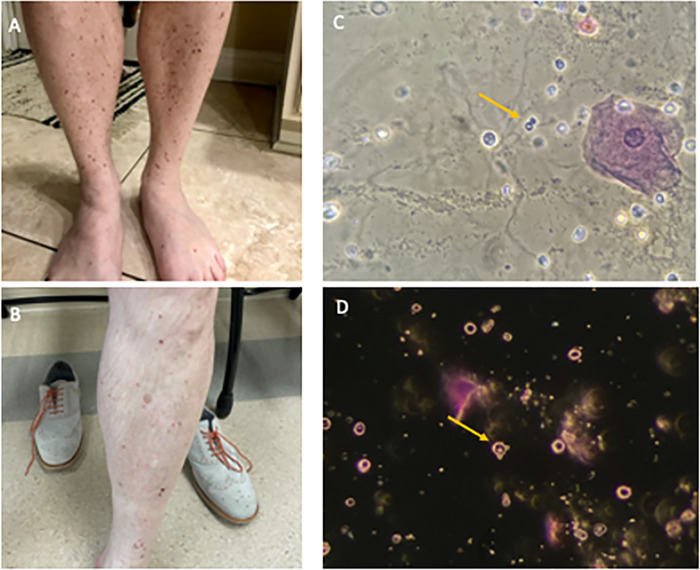
Clinical manifestations of immunoglobulin A vasculitis. (A) Palpable purpuric rash on the lower extremities 2 weeks after the patient received the first dose of the severe acute respiratory syndrome coronavirus 2 mRNA vaccine (BNT162b2, Pfizer, Inc) and (B) after 2 weeks of oral corticosteroids. Microscopic examination of urinary sediment stained with Sternheimer-Malbin stain revealed glomerular hematuria, characterized by acanthocytes (arrows) inspected under (C) phase-contrast microscopy and (D) dark-field microscopy illumination. Original magnification ×400.
After completion of 2 weeks of oral corticosteroids, urinalysis revealed proteinuria, and a nephrology consultation was requested. On examination in the nephrology clinic, the patient's vital signs were normal with blood pressure of 122/81 mmHg. Healing papules were noted on his lower extremities, but otherwise the physical examination was normal. Laboratory data are shown in Table 1. Two weeks after the second dose of vaccine, serum creatinine was normal at 0.9 mg/dL. Microscopic examination of the urinary sediment revealed acanthocytes (Figure 1). Three weeks after the second dose of vaccine, urine protein to creatinine ratio (UPCR) was 1.1 g/day. Serum complements were normal, and all pertinent serology was negative. A second UPCR repeated 1 week later (4 weeks after the second dose of vaccine) was still elevated at 0.8 g/day. Microscopic examination of the urinary sediment was repeated, and acanthocytes were again identified.
Table 1.
Patient's Laboratory Data Before and After Vaccination
| Parameter | Reference Range | 4 Days Before First Dose of Vaccine | 2 Weeks After Second Dose of Vaccine | 3 Weeks After Second Dose of Vaccinea |
|---|---|---|---|---|
| Clinical chemistry | ||||
| Sodium, mmol/L | 136–145 | 138 | 140 | |
| Potassium, mmol/L | 3.5–5.1 | 4.4 | 4.2 | |
| Chloride, mmol/L | 95–110 | 104 | 103 | |
| Bicarbonate, mmol/L | 23–29 | 26 | 27 | |
| Anion gap, mmol/L | 5–15 | 8 | 10 | |
| Blood urea nitrogen, mg/dL | 6–20 | 15 | 11 | |
| Creatinine, mg/dL | 0.5–1.4 | 0.9 | 0.9 | |
| eGFR, mL/min/1.73 m2 | >60 | >60 | >60 | |
| Calcium, mg/dL | 8.7–10.5 | 9.3 | 9.8 | |
| Glucose, mg/dL | 70–110 | 107 | 103 | |
| Alkaline phosphatase, U/L | 55–135 | 149 | 135 | |
| Protein total, g/dL | 6.0–8.4 | 7.5 | 8.3 | |
| Albumin, g/dL | 3.5–5.2 | 3.8 | 3.4 | |
| Bilirubin total, mg/dL | 0.1–1.0 | 0.4 | 0.7 | |
| Aspartate transaminase, U/L | 10–40 | 22 | 13 | |
| Alanine transaminase, U/L | 10–44 | 28 | 25 | |
| C-reactive protein, mg/L | 0.0–8.2 | 100 | ||
| Erythrocyte sedimentation rate, mm/h | 0–10 | 92 | ||
| Complete blood count | ||||
| Hemoglobin, g/dL | 14.0–18.0 | 14.3 | 13.3 | |
| Platelets, K/uL | 150–350 | 339 | 407 | |
| White cell count, K/uL | 3.90–12.70 | 9.4 | 9.2 | |
| Neutrophils, % | 38.0–73.0 | 69.8 | 67.7 | |
| Lymphocytes, % | 18.0–48.0 | 20.7 | 22.3 | |
| Monocytes, % | 4.0–15.0 | 6.6 | 7.1 | |
| Eosinophils, % | 0.0–8.0 | 0.2 | 0.2 | |
| Basophils, % | 0.0–1.9 | 0.04 | 0.02 | |
| Immunology | ||||
| ANA screen | Negative <1:80 | Negative <1:80 | ||
| Complement (C3), mg/dL | 50–180 | N/R | N/R | 148 |
| Complement (C4), mg/dL | 11–44 | N/R | N/R | 25 |
| Antistreptolysin O titer, IU/mL | <200 | 113 | ||
| C-ANCA | <1:20 titer | <1:20 titer | ||
| P-ANCA | <1:20 titer | <1:20 titer | ||
| Infectious disease | ||||
| Hepatitis B surface antigen | Negative | |||
| Urine | ||||
| Color | Yellow, straw, amber | Yellow | Yellow | |
| Appearance | Clear | Cloudy | Hazy | |
| Specific gravity | 1.005–1.030 | ≥1.030 | 1.025 | |
| pH | 5.0–8.0 | 5.0 | 5.0 | |
| Glucose | Negative | Negative | Negative | |
| Protein | Negative | Negative | 2+ | |
| Ketones | Negative | Negative | Negative | |
| Occult blood | Negative | Negative | 2+ | |
| Nitrite | Negative | Negative | Negative | |
| Bilirubin | Negative | Negative | Negative | |
| Leukocytes, hpf | 0–4 | Trace | Trace | |
| Red blood cells, hpf | 0–5 | 10 | ||
| White blood cells, hpf | None-Occ | 3 | 18 | |
| Bacteria | Rare | None | ||
| Protein to creatinine ratio, g/day | 0.00–0.20 | 1.1 |
aClinical chemistry and complete blood count were not done 3 weeks after the second dose of vaccine.
ANA, antinuclear antibody; C-ANCA, cytoplasmic-antineutrophil cytoplasmic antibodies; eGFR, estimated glomerular filtration rate; N/R, not reported; P-ANCA, perinuclear-antineutrophil cytoplasmic antibodies.
Upon discussion of risks and benefits of establishing a diagnosis, the patient agreed to a kidney biopsy. The biopsy was performed without complications and consisted of 16 glomeruli by light and immunofluorescence microscopy with 2 globally sclerosed with diagnostic changes of an IgA nephropathy with dominant polyclonal, lambda slightly more than kappa, mesangial staining by immunofluorescence microscopy, and corresponding scattered mesangial deposits by electron microscopy. Mild mesangial hypercellularity was present in only 1 of the 16 glomeruli in the light microscopy sample, with no endocapillary hypercellularity, no segmental sclerosis, minimal (approximately 5%) interstitial fibrosis and tubular atrophy, and no crescents or necrosis (Figure 2). The findings were diagnosed as IgA nephropathy, which in the context of the skin lesions was consistent with IgA vasculitis. Although the MEST-C score has not been validated in IgA vasculitis, these lesions correspond to M0E0S0T0C0 (M=mesangial proliferation, E=endocapillary proliferation, S=segmental sclerosis, T=tubulointerstitial fibrosis, C=crescents).
Figure 2.
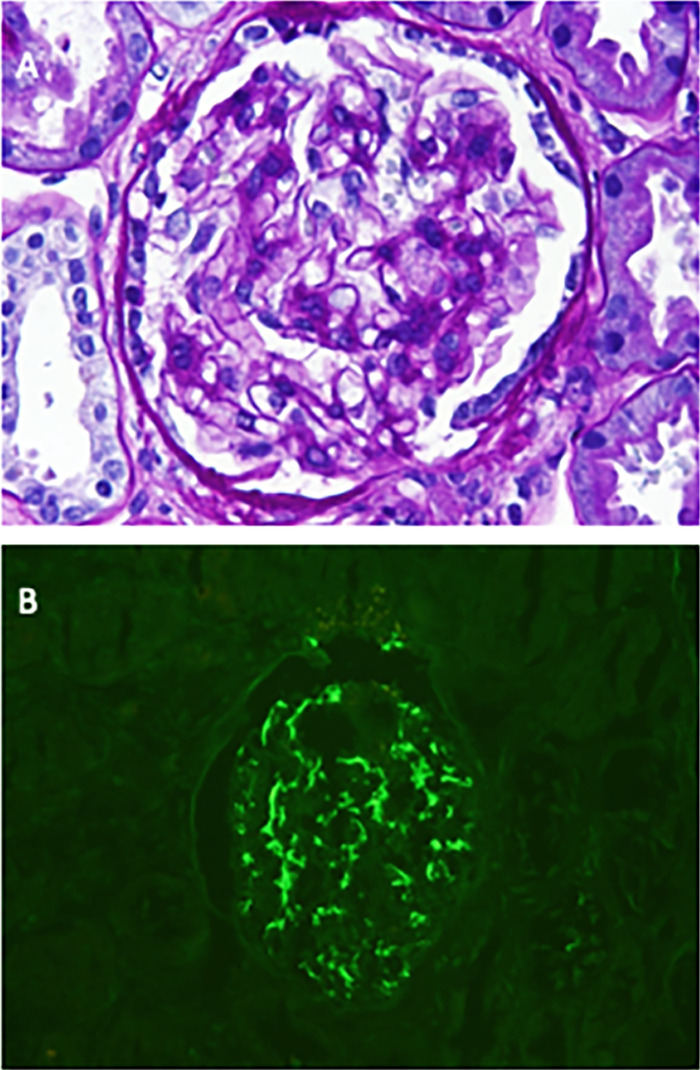
Kidney biopsy specimen showing pathologic features of immunoglobulin A (IgA) nephropathy. (A) Focal mild mesangial hypercellularity was present (periodic acid Schiff stain, original magnification ×400). (B) IgA-dominant granular diffuse global mesangial staining for IgA was present, 3+ on a 0 to 3+ scale (anti-IgA immunofluorescence, original magnification ×400).
After the kidney biopsy, the patient began treatment with lisinopril 10 mg orally daily. No further immunosuppression was given. At a follow-up visit 1 week after the kidney biopsy, the rash had subsided completely. The UPCR 4 weeks after starting the lisinopril was 0.5 g/day. Figure 3 presents a timeline of the case.
Figure 3.

Timeline of case report. Day –4 corresponds to 4 days before the patient received the first dose of vaccine. UA, urinalysis; UPCR, urine protein to creatinine ratio.
DISCUSSION
Emergence of glomerular disease following vaccination against viral infections is a known phenomenon. A T lymphocyte–mediated cellular response appears to be a mechanistic link between the development of antibodies against a virus and the pathogenesis of a glomerular lesion.12,13 Perhaps the glomerular entity best characterized as being associated with immunizations is minimal change disease. Several reports demonstrate that diffuse podocyte effacement and nephrotic syndrome can occur following vaccination for influenza and other viruses.5,6,14-19 The COVID-19 pandemic resulted in an unprecedented volume of vaccinations worldwide. Not surprisingly, some reports of glomerular disease development20,21 or relapse of a preexisting glomerular disease have been published, including some describing IgA nephropathy.8-11 In our case, the clinical presentation and pathologic findings strongly suggest that the SARS-CoV-2 BNT162b2 (mRNA) vaccine from Pfizer can trigger a clinical syndrome compatible with IgA vasculitis (Henoch-Schönlein purpura). The timing of the initial appearance of the rash with respect to the vaccination and the recurrence of the rash following reexposure to the vaccine argue for a “definite” causal association by the Naranjo criteria.22
IgA vasculitis can be triggered by many factors. IgA vasculitis occurrences after immunizations have been reported in children. A case-control study in children found an increased risk of IgA vasculitis within 12 weeks after administration of the measles, mumps, and rubella vaccination (odds ratio 3.4).23 The flu vaccine has been reported to be the vaccine type most commonly associated with vasculitis.24 Cutaneous manifestations after a COVID-19 vaccine have been reported. In a registry-based study of 414 patients who received mRNA COVID-19 vaccines, delayed large local reactions were most common.25 A case reporting Henoch-Schönlein purpura without additional organ involvement after a BNT162b2 (mRNA) vaccine was published in July 2021,26 making our case, to our knowledge, the first de novo Henoch-Schönlein purpura case with both a cutaneous manifestation and kidney involvement.
Published reports describe glomerular diseases after COVID-19 vaccines, including a case of antineutrophil cytoplasmic antibody vasculitis,21 granulomatous vasculitis,27 and 7 cases of relapse of IgA nephropathy following exposure to COVID-19 vaccines (Table 2).8-11
Table 2.
Patient Demographics and Clinical Characteristics of Previously Reported Cases of Post-COVID-19 Vaccine Immunoglobulin A Nephropathy (IgAN)
| Study | Age, Sex, Race | SARS-CoV-2 Vaccine | Time of Flare-Up After COVID-19 Vaccine | Year IgAN First Diagnosed | Treatment | Gross Hematuria Events During Disease Course | Persistent Microscopic Hematuria | Proteinuria in 2020, g/d | Proteinuria Between SARS-CoV-2 Vaccine Doses, g/d | Proteinuria after Last SARS-CoV-2 Vaccine Dose, g/d |
|---|---|---|---|---|---|---|---|---|---|---|
| Rahim et al, 20218 | 52, F, Asian | BNT162b2 (Pfizer) | 24 hours after the second dose | 2017 | RAASi | Yes | N/R | 633.1 mg/ga | N/R | 2,411 mg/g after 48 hours, 1,441 mg/g after 5 daysa |
| Negrea and Rovin, 20219 | 38, F, W | mRNA-1273 (Moderna) | 8 to 24 hours after the second dose | 2005 | RAASi | At presentation; during 1 episode of gastroenteritis; occasionally after yearly influenza vaccine | Yes | 0.63 | 0.82 | 1.40 |
| 38, F, W | mRNA-1273 (Moderna) | 8 to 24 hours after the second dose | 2019 | Cyc + pred for 6 months, followed by RAASi | At presentation only | Yes | 0.43 | 0.59 | 0.4 | |
| Perrin et al, 202110 | 22, M, N/R | mRNA-1273 (Moderna) | Day 2 and day 25 after the first dose; day 2 after the second dose | 2019 | Steroids for 6 months followed by RAASi | No | Yes | 0.20 | 0.34 | 0.40 |
| 41, F, N/R | BNT162b2 (Pfizer) | Day 2 after the first dose (the patient refused the second dose) | 2005 | Tac, MPA, and steroids for kidney transplantation | Yes | Yes | 0 | 0.47 | 0.41 | |
| 27, F, N/R | BNT162b2 (Pfizer) | Day 2 after the second dose | 2020 | Steroid for 1 month followed by RAASi | No | No | 20 | 1.9 | 1.2 | |
| Horino, 202111 | 46, F, Asian | BNT162b2 (Pfizer) | 12 hours after the second dose | 4 years before | Prednisolone | Yes | No | N/R | N/R | Nephrotic range |
| Abramson et al, 202120 | 30, M, W | mRNA-1273 (Moderna) | Day 2 after the second dose | No history of IgAN | RAASi | At presentation | N/R | N/R | N/R | 0.8 |
| Anderegg et al, 202121 | 39, M, N/R | mRNA-1273 (Moderna) | Immediately after the second dose | No history of IgAN | Cyc + steroids | At presentation | Yes | N/R | N/R | Nephrotic range |
aUrine microalbumin/creatinine.
Cyc, cyclophosphamide; F, female; M, male; MPA, mycophenolic acid; N/R, not reported; pred, prednisone; RAASi, renin-angiotensin-aldosterone system inhibitor; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; tac, tacrolimus; W, White.
To our knowledge, only 2 cases of de novo IgA nephropathy after receiving the mRNA-1273 vaccine (Moderna, Inc) have been reported.20,21 Our case is unique in a few aspects. First, the diagnosis of IgA nephropathy was de novo after the BNT162b2 (mRNA) Pfizer vaccine. Second, the patient exhibited simultaneous involvement of skin and kidney consistent with IgA vasculitis.
Confirming the diagnosis of IgA vasculitis requires skin or kidney biopsy. Characteristic findings in a skin biopsy, as observed in our patient, include neutrophilic infiltration of the dermal small blood vessel walls associated with fibrinoid necrosis and disruption of the vessel wall.28 Kidney biopsy is reserved for patients in whom the diagnosis is uncertain or in cases of persistent hematuria, proteinuria, or decreased kidney function. Arguably, the kidney biopsy was not a necessity in our patient given the high clinical suspicion of IgA vasculitis in the context of the dermopathologic diagnosis of IgA vasculitis. However, because of the lack of reports of similar cases of this nature, because the diagnosis had potential long-term implications, and because the patient was at low risk for procedural complications (normal blood pressure, normal serum creatinine, normal hemoglobin, platelets, and coagulation profile), the decision was made to pursue a kidney biopsy.
CONCLUSION
Clinicians need to be aware that IgA vasculitis can develop in susceptible individuals following exposure to a COVID-19 vaccination. Careful monitoring of urinalysis and kidney function by primary care providers may be advised, particularly for patients with preexisting glomerular disease (such as IgA nephropathy or minimal change disease) or patients with a history of atopy or seasonal allergies, such as the patient reported in this case.
ACKNOWLEDGMENTS
The authors have no financial or proprietary interest in the subject matter of this article.
This article meets the Accreditation Council for Graduate Medical Education and the American Board of Medical Specialties Maintenance of Certification competencies for Patient Care and Medical Knowledge.
REFERENCES
- 1.Shimmel A, Shaikhouni S, Mariani L. Current understanding of clinical manifestations of COVID-19 in glomerular disease. Glomerular Dis. 2021;1(4):250-264. doi: 10.1159/000518276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wu H, Larsen CP, Hernandez-Arroyo CF, et al. AKI and collapsing glomerulopathy associated with COVID-19 and APOL1 high-risk genotype. J Am Soc Nephrol. 2020;31(8):1688-1695. doi: 10.1681/ASN.2020050558 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bomback AS, Kudose S, D'Agati VD. De novo and relapsing glomerular diseases after COVID-19 vaccination: what do we know so far? Am J Kidney Dis. 2021;78(4):477-480. doi: 10.1053/j.ajkd.2021.06.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sacker A, Kung V, Andeen N. Anti-GBM nephritis with mesangial IgA deposits after SARS-CoV-2 mRNA vaccination. Kidney Int. 2021;100(2):471-472. doi: 10.1016/j.kint.2021.06.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gutiérrez S, Dotto B, Petiti JP, et al. Minimal change disease following influenza vaccination and acute renal failure: just a coincidence? Nefrologia. 2012;32(3):414-415. doi: 10.3265/Nefrologia.pre2012.Feb.11370 [DOI] [PubMed] [Google Scholar]
- 6.Kikuchi Y, Imakiire T, Hyodo T, et al. Minimal change nephrotic syndrome, lymphadenopathy and hyperimmunoglobulinemia after immunization with a pneumococcal vaccine. Clin Nephrol. 2002;58(1):68-72. doi: 10.5414/cnp58068 [DOI] [PubMed] [Google Scholar]
- 7.Patel C, Shah HH. Membranous nephropathy and severe acute kidney injury following influenza vaccination. Saudi J Kidney Dis Transpl. 2015;26(6):1289-1293. doi: 10.4103/1319-2442.168676 [DOI] [PubMed] [Google Scholar]
- 8.Rahim SEG, Lin JT, Wang JC. A case of gross hematuria and IgA nephropathy flare-up following SARS-CoV-2 vaccination. Kidney Int. 2021;100(1):238. doi: 10.1016/j.kint.2021.04.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Negrea L, Rovin BH. Gross hematuria following vaccination for severe acute respiratory syndrome coronavirus 2 in 2 patients with IgA nephropathy. Kidney Int. 2021;99(6):1487. doi: 10.1016/j.kint.2021.03.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Perrin P, Bassand X, Benotmane I, Bouvier N. Gross hematuria following SARS-CoV-2 vaccination in patients with IgA nephropathy. Kidney Int. 2021;100(2):466-468. doi: 10.1016/j.kint.2021.05.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Horino T. IgA nephropathy flare-up following SARS-CoV-2 vaccination. QJM. 2021:hcab223. doi: 10.1093/qjmed/hcab223 [DOI] [PubMed] [Google Scholar]
- 12.de Wolf ACMT, van Aalst S, Ludwig IS, et al. Regulatory T cell frequencies and phenotypes following anti-viral vaccination. PLoS One. 2017;12(6):e0179942. doi: 10.1371/journal.pone.0179942 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shalhoub RJ. Pathogenesis of lipoid nephrosis: a disorder of T-cell function. Lancet. 1974;304(7880):556-560. doi: 10.1016/s0140-6736(74)91880-7 [DOI] [PubMed] [Google Scholar]
- 14.Kielstein JT, Termühlen L, Sohn J, Kliem V. Minimal change nephrotic syndrome in a 65-year-old patient following influenza vaccination. Clin Nephrol. 2000;54(3):246-248. [PubMed] [Google Scholar]
- 15.Lebedev L, Sapojnikov M, Wechsler A, et al. Minimal change disease following the Pfizer-BioNTech COVID-19 vaccine. Am J Kidney Dis. 2021;78(1):142-145. doi: 10.1053/j.ajkd.2021.03.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Clajus C, Spiegel J, Bröcker V, Chatzikyrkou C, Kielstein JT. Minimal change nephrotic syndrome in an 82 year old patient following a tetanus-diphteria-poliomyelitis-vaccination. BMC Nephrol. 2009;10:21. doi: 10.1186/1471-2369-10-21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Humphreys BD, Vanguri VK, Henderson J, Antin JH. Minimal-change nephrotic syndrome in a hematopoietic stem-cell transplant recipient. Nat Clin Pract Nephrol. 2006;2(9):535-539; quiz 540. doi: 10.1038/ncpneph0271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Işlek I, Cengiz K, Cakir M, Küçüködük S. Nephrotic syndrome following hepatitis B vaccination. Pediatr Nephrol. 2000;14(1):89-90. [PubMed] [Google Scholar]
- 19.Ozdemir S, Bakkaloğlu A, Oran O. Nephrotic syndrome associated with recombinant hepatitis B vaccination: a causal relationship or just a mere association? Nephrol Dial Transplant. 1998;13(7):1888-1889. doi: 10.1093/oxfordjournals.ndt.a027900 [DOI] [PubMed] [Google Scholar]
- 20.Abramson M, Mon-Wei Yu S, Campbell KN, Chung M, Salem F. IgA nephropathy after SARS-CoV-2 vaccination. Kidney Med. 2021;3(5):860-863. doi: 10.1016/j.xkme.2021.05.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Anderegg MA, Liu M, Saganas C, et al. De novo vasculitis after mRNA-1273 (Moderna) vaccination. Kidney Int. 2021;100(2):474-476. doi: 10.1016/j.kint.2021.05.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30(2):239-245. doi: 10.1038/clpt.1981.154 [DOI] [PubMed] [Google Scholar]
- 23.Da Dalt L, Zerbinati C, Strafella MS, et al. ; Italian Multicenter Study Group for Drug and Vaccine Safety in Children. Henoch-Schönlein purpura and drug and vaccine use in childhood: a case-control study. Ital J Pediatr. 2016;42(1):60. doi: 10.1186/s13052-016-0267-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bonetto C, Trotta F, Felicetti P, et al. ; Brighton Collaboration Vasculitis Working Group. Vasculitis as an adverse event following immunization - systematic literature review. Vaccine. 2016;34(51):6641-6651. doi: 10.1016/j.vaccine.2015.09.026 [DOI] [PubMed] [Google Scholar]
- 25.McMahon DE, Amerson E, Rosenbach M, et al. Cutaneous reactions reported after Moderna and Pfizer COVID-19 vaccination: a registry-based study of 414 cases. J Am Acad Dermatol. 2021;85(1):46-55. doi: 10.1016/j.jaad.2021.03.092 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hines AM, Murphy N, Mullin C, Barillas J, Barrientos JC.. Henoch-Schönlein purpura presenting post COVID-19 vaccination. Vaccine. 2021;39(33):4571-4572. doi: 10.1016/j.vaccine.2021.06.079 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gillion V, Jadoul M, Demoulin N, Aydin S, Devresse A. Granulomatous vasculitis after the AstraZeneca anti-SARS-CoV-2 vaccine. Kidney Int. 2021;100(3):706-707. doi: 10.1016/j.kint.2021.06.033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Johnson EF, Wetter DA, Lehman JS, Hand JL, Davis DMR, Tollefson MM. Leukocytoclastic vasculitis in children: clinical characteristics, subtypes, causes and direct immunofluorescence findings of 56 biopsy-confirmed cases. J Eur Acad Dermatol Venereol. 2017;31(3):544-549. doi: 10.1111/jdv.13952 [DOI] [PubMed] [Google Scholar]


