Abstract
Whole-cell protein electrophoresis and biochemical examination by means of a panel of 64 tests were used to identify 14 putative helicobacters to the species level. The results were confirmed by means of DNA-DNA hybridization experiments and were used to discuss misidentification of helicobacters based on 16S rRNA gene sequence data. The data indicated that comparison of near-complete 16S ribosomal DNA sequences does not always provide conclusive evidence for species level identification and may prove highly misleading. The data also indicated that “Helicobacter westmeadii” is a junior synonym of Helicobacter cinaedi and that Helicobacter sp. strain Mainz belongs to the same species. H. cinaedi occurs in various animal reservoirs, including hamsters, dogs, cats, rats, and foxes. Appropriate growth conditions and identification strategies will be required to establish the genuine significance of this widely distributed Helicobacter species.
The cultivation of Helicobacter pylori from the human gastric mucosa in the early 1980s (33) and the demonstration of its relationship to gastritis, peptic ulcer disease, and gastric neoplasia triggered a series of studies of the ecology and role of Helicobacter-like organisms in a variety of hosts. At present, 18 validly named species and two candidate species (3, 4) have been isolated from gastric or enteric samples of a variety of hosts, including humans, hamsters, rats, mice, ferrets, pigs, poultry, wild birds, cats, dogs, monkeys, and cheetahs. Several other Helicobacter taxa remain unnamed or have not been properly described in accordance with internationally accepted rules of nomenclature, and therefore their names have not been validated (6, 9, 13, 29, 30). The biochemical inertness of all Campylobacter-like organisms, including helicobacters, plays a major role in influencing the identification strategies of clinical laboratories. Classical phenotypic tests routinely used for the identification of clinical bacteria often yield negative or variable results within species. Problems associated with phenotypic identification have led to sequence analysis of rRNA genes (in particular 16S) as an increasingly popular alternative approach for identification of new isolates. Strains have been identified as novel species, primarily because of supposedly sufficient differences in 16S ribosomal DNA (rDNA) sequence similarity to known species (for example, reference 13), or as well-established species, again primarily based on the percentage of similarity of 16S rDNA sequence (for example, references 7 and 28).
In the present study, we describe the identification and characterization of 14 Helicobacter cinaedi isolates obtained from various hosts, including humans, dogs, foxes, and a rat, by using different phenotypic and genotypic approaches and comment on the pitfalls of the 16S rDNA sequence approach. These 14 strains included isolates described in this journal as “Helicobacter westmeadii” sp. nov. (30) and as Helicobacter sp. nov. strain Mainz (13).
MATERIALS AND METHODS
Strains.
Three H. cinaedi reference strains (LMG 7543T, LMG 8770, and LMG 9071) from the original study by Totten et al. (29) and five additional strains (LMG 8558, LMG 8559, LMG 9072, LMG 9153, and LMG 9357), characterized in a previous polyphasic taxonomic study (31), were used as references (Table 1). Fourteen recent isolates obtained from the feces of dogs, foxes, and a rat and from human blood or feces were studied; the isolates were from Finland (n = 6), Belgium (n = 2), Sweden (n = 2), Australia (n = 2), Scotland (n = 1), and Germany (n = 1) (Table 1).
TABLE 1.
H. cinaedi strains examined
| Our no. | Other strain no. | Depositora | Source |
|---|---|---|---|
| R-2971 | KJ457 | Blue fox, feces (Finland) | |
| R-2977 | KJ459 | Blue fox, feces (Finland) | |
| R-2981 | KJ465A | Blue fox, feces (Finland) | |
| R-2982 | KJ465B | Blue fox, feces (Finland) | |
| R-2983 | KJ412 | Dog, feces (Finland) | |
| R-2991 | KJ425 | Dog, feces (Finland) | |
| R-3026 | CCUG 36876 | CCUG | Rat, feces (Sweden) |
| R-4792 | ADN 0413 | M. MacLennan | Dog, feces (Scotland) |
| R-915 | SL6001 | S. Lauwers | Human, feces (Belgium) |
| R-927 | CCUG 33804 | CCUG | Human, septic arthritis (Germany) |
| R-5758 | Strain Bower | M. Yuen | Human, blood (Australia) |
| R-5759 | Strain Flint | M. Yuen | Human, blood (Australia) |
| LMG 8559 | CCUG 15432 | CCUG | Human, blood (Sweden) |
| LMG 8558 | CCUG 17733 | CCUG | Human, feces (Sweden) |
| LMG 7543T | CCUG 18818 | CCUG | Homosexual man, rectal swab (United States) |
| LMG 9071 | CCUG 18819 | CCUG | Homosexual man, blood (United States) |
| LMG 8770 | CCUG 19218 | CCUG | Homosexual man, rectal swab (United States) |
| LMG 9357 | CCUG 19503 | CCUG | Human, blood (Canada) |
| LMG 9072 | CCUG 19504 | CCUG | Human, blood (Canada) |
| LMG 9153 | CCUG 20698 | CCUG | Human, feces (United Kingdom) |
| LMG 16312 | CCUG 33887 | CCUG | Dog, feces (Sweden) |
| LMG 13991 | M2-08-4658 | H. Goossens | Human, feces (Belgium) |
Own isolate if not specified.
Representative strains of all presently named Helicobacter species were obtained from the Culture Collection, University of Göteborg, Department of Clinical Bacteriology, Göteborg, Sweden (CCUG) or the Bacteria Collection, Laboratorium voor Microbiologie Gent, Universiteit Gent, Ghent, Belgium (LMG) and were included as references.
PAGE of whole-cell proteins.
All 14 isolates were grown on Mueller-Hinton agar (catalog no. CM 337; Oxoid, Ltd., Basingstoke, United Kingdom) supplemented with 5% (vol/vol) horse blood and were incubated at 36 to 37°C in a microaerobic atmosphere containing approximately 5% O2, 3.5% CO2, 7.5% H2, and 84% N2. Whole-cell protein extracts were prepared, and sodium dodecyl sulfate-polyacrylamide gel electrophoresis (PAGE) was performed as described before (26). Whole-cell protein profiles of H. cinaedi reference strains and of type and reference strains of other Helicobacter species were available from previous studies. The densitometric analysis, normalization, and interpolation of the protein profiles and numerical analysis were performed with the GelCompar software package version 4.2 (Applied Maths, Kortrijk, Belgium). The profiles were recorded and stored on an IBM PC-compatible computer. The similarities between all pairs of traces were expressed by the Pearson product moment correlation coefficient presented below as percentages of similarity for convenience.
Phenotypic analysis.
All 14 isolates and reference strain LMG 9357 (not examined before by this scheme) were grown on 5% (vol/vol) calf blood agar for 3 days under microaerobic conditions, as described previously (19). A total of 64 phenotypic characters were determined using methods described previously (19–22). The results were compared with data for 37 Campylobacter taxa in a probability matrix using computer-assisted methods.
Dot blot DNA-DNA hybridizations.
DNA was isolated by the method of Pitcher et al. (25), with modifications described before (14). Dot blot DNA-DNA hybridizations were performed as described before (10, 14). The DNA of H. cinaedi LMG 7543T was used as a probe. DNAs of the following strains were hybridized with the LMG 7543T probe: LMG 7543T, LMG 8559, and LMG 8558 (the three reference strains) and R-927, R-2971, R-2977, R-2981, R-2983, R-2991, and R-5759. “Flexispira rappini” CCUG 23435 and Helicobacter canis CCUG 19561 served as negative controls. Aliquots of 0.5, 5, and 50 ng of chromosomal DNA were spotted onto the membranes for each hybridization experiment.
16S rDNA sequencing.
The primers and methods used for DNA extraction, PCR amplification, and direct, automated sequencing of 16S rRNA genes were as described previously (18), except that primer 1492r (5′-TACGGYTACCTTGTTACGACTT) was used in place of 1392r for the initial PCR amplification and subsequent sequencing of the PCR product. This allowed sequences to be obtained (both strands) over ∼95% (compared to ∼90%) of the 16S rRNA gene. In addition, the Taq polymerase and PCR buffer (1.5 mM final MgCl2 concentration) were from Boehringer (Mannheim, Germany). The consensus sequence and the sequences of strains belonging to the same phylogenetic group (retrieved from the EMBL data library) were aligned, and a phylogenetic tree was constructed based on the neighbor-joining method by using the GeneCompar version 2.0 software package (Applied Maths).
The strain numbers and GenBank accession numbers of the strains of the reference species used in the phylogenetic analysis are as follows: “F. rappini”, CCUG 23435 and M88138; Helicobacter acinonychis, LMG 12684T and M88148; Helicobacter bilis, LMG 18386T and U18766; Helicobacter bizzozeronii, R-1051T and Y09404; Helicobacter canis, LMG 18086T and L13464; H. cinaedi, LMG 7543T and M88150; Helicobacter cholecystus, R-3555T and U46129; Helicobacter felis, LMG 11750T and M37642; Helicobacter fennelliae, LMG 7546T and M88154; Helicobacter hepaticus, LMG 16316T and U07574; Helicobacter muridarum, LMG 13646T and M80205; Helicobacter mustelae, LMG 18044T and M35048; Helicobacter nemestrinae, LMG 14378T and X67854; Helicobacter pametensis, LMG 12678T and M88147; Helicobacter pullorum, LMG 16317T and L36141; Helicobacter pylori, LMG 7539T and M88157; Helicobacter rodentium, ATCC 700285T and U96296; Helicobacter salomonis, InkinenT and U89351; Helicobacter sp. strain Mainz, R-927 and X81028; Helicobacter trogontum, R-5081T and U65103; and Sulfurospirillum sp. strain CCUG 13942, L14632.
Nucleotide sequence accession numbers.
The nucleotide sequence accession numbers for the 16S rDNA sequences of strains R-4792, LMG 16312, and R-927 are AF207737, AF207738, and AF207739, respectively.
RESULTS
Whole-cell protein electrophoresis.
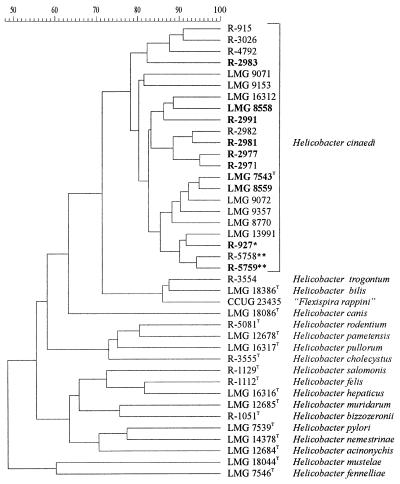
All 14 isolates were identified as H. cinaedi by comparison of their whole-cell protein profiles with a database comprising patterns of over 1,000 Campylobacter, Arcobacter, Helicobacter, and Wolinella strains. Figure 1 shows the result of the numerical comparison of the protein patterns of the 14 isolates, the eight H. cinaedi reference strains, and the type or reference strains of other Helicobacter species. All 22 H. cinaedi strains form a single protein electrophoretic cluster above a similarity level of 80%.
FIG. 1.
Dendrogram expressing similarity of whole-cell protein patterns of all 22 H. cinaedi isolates and of Helicobacter reference strains. The strain numbers in boldface indicate those strains that were included in DNA-DNA hybridization experiments. The strain marked with an asterisk was originally described as Helicobacter sp. strain Mainz; the strains marked with double asterisks were originally described as “H. westmeadii”.
Phenotypic analysis.
Computer-assisted comparison of the biochemical profiles identified 14 of the 15 strains examined (the 14 novel isolates and reference strain LMG 9537) as H. cinaedi with a Willcox probability identification score (IDS) of 0.99 (i.e., 99% probable) or more. Field strain LMG 16312 was not confidently identified by this method (IDS, <0.95). Several atypical features (notably results in nitrate reduction, alkaline phosphatase production, indoxyl acetate hydrolysis, tolerance and reduction of triphenyl tetrazolium chloride, growth at 42°C, and resistance to cephalothin and cefoperazone) were noted in the phenotype of LMG 16312 compared with those of type and reference strains determined previously (23). Table 2 summarizes the key phenotypic features of the H. cinaedi strains examined here and previously (23) that are especially useful for discriminating among other enteric helicobacters associated with human gastroenteritis or septicemia.
TABLE 2.
Selected phenotypic test results of the 15 field and reference strains of H. cinaedi examined in the present study compared with data obtained previously for other human enteric Helicobacter species (23)
| Test | No. of strains giving positive result
|
|||||
|---|---|---|---|---|---|---|
|
H. cinaedi
|
H. canis (n = 11) | H. fennelliae (n = 12) | H. pullorum (n = 16) | “F. rappini” (n = 3)a | ||
| Present study (n = 15) | Previous study (n = 12) | |||||
| Catalase | 7 | 10 | 0 | 11 | 14 | 1 |
| Urease | 0 | 0 | 0 | 0 | 0 | 3 |
| Nitrate reduction | 11 | 12 | 0 | 0 | 16 | 2 |
| Indoxyl acetate hydrolysis | 1 | 1 | 11 | 12 | 0 | 1 |
| Cephalothin | 14 | 12 | 2 | 1 | 16 | 3 |
| Carbenicillin | 0 | 1 | 1 | 0 | 14 | 0 |
| Methyl orange | 8 | 12 | 0 | 5 | 16 | 1 |
| Sodium fluoride | 1 | 0 | 0 | 7 | 16 | 0 |
Data include unpublished results of S. L. W. On for two strains of canine origin.
Dot blot DNA-DNA hybridizations.
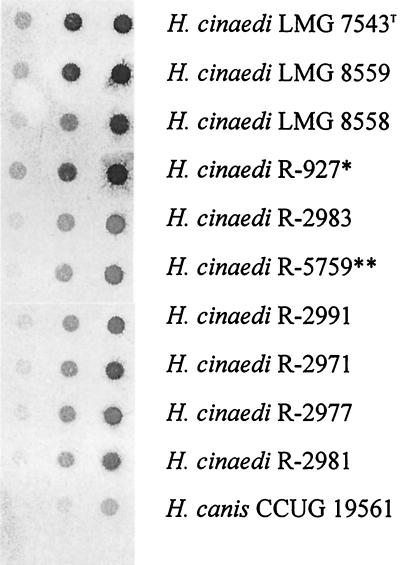
Dot blot DNA-DNA hybridization experiments revealed strong hybridization reactions between the H. cinaedi LMG 7543T probe and all of the H. cinaedi strains tested. Weak or no hybridization was obtained to “F. rappini” or H. canis DNA (the nearest phylogenetic neighbors of H. cinaedi) or to any other Helicobacter species (Fig. 2).
FIG. 2.
Dot blot DNA-DNA hybridization result obtained with the labeled H. cinaedi LMG 7543T probe. The strain marked with an asterisk was originally described as Helicobacter sp. strain Mainz; the strain marked with double asterisks was originally described as “H. westmeadii”.
16S rDNA sequencing and phylogenetic analysis.
The expected (∼1,500-bp) amplicon was obtained from both R-4792 and R-927, whereas strain LMG 16312 yielded an enlarged amplicon of ∼1,700 bp, later found to be due to the presence of an intervening sequence (IVS) of ∼200 bp that started at base 168 of the submitted sequence (AF207738) for this strain. The 16S rDNA sequence similarity levels between the different H. cinaedi isolates varied considerably. Figure 3 summarizes the 62 polymorphic sites (4.3%) identified after alignment of our H. cinaedi sequences with an existing sequence for strain R-927 (accession number X81028) and the 1,444-bp sequence of the H. cinaedi type strain (accession number M88150). The majority of these polymorphic sites were accounted for by the two sequences representing strain R-927. The positions where alignment gaps were introduced into each sequence are also shown in Fig. 3. Three of these seven alignment gaps occurred at the two termini of the IVS for strain LMG 16312 (the IVS sequence itself is not shown), and a single-base deletion was also found in this region for strain R-927 (both sequences). The remaining alignment gaps seem likely to be errors in downloaded EMBL sequences, since the 1-base deletion (position 184) and 2-base insertion (position 826) were not found in any of our submitted sequences. The repeat sequence for R-927 was highly similar (99.9%) to the sequence originally described (13), except that a 2-base insertion (detailed above) was seen in the original sequence and a further four polymorphisms were identified (positions 928 and 1419 to 1421 [Fig. 3]), possibly as a result of nucleotide misincorporation during sequencing procedures in either of the two investigations.
FIG. 3.
Comparison of H. cinaedi 16S rRNA gene sequences with the sequence for the type strain (M88150), showing only polymorphic sites and those sites where gaps (−) were introduced during alignment. X81028 represents a previously published sequence for strain R-927 (13). The numbers (in vertical format) above the sequences correspond to the nucleotide numbering of the M88150 sequence, including three alignment gaps. Bases identical to the M88150 sequence are represented by periods.
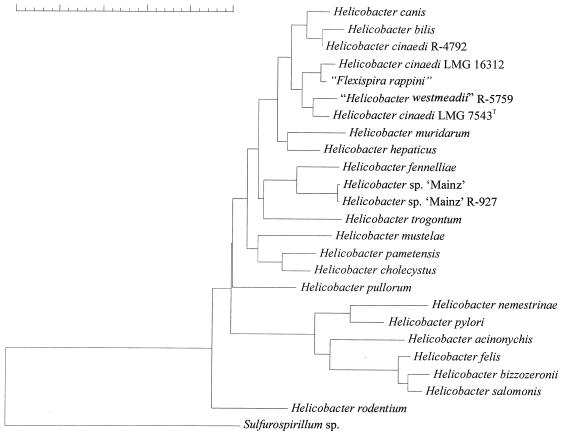
Figure 4 shows the result of neighbor-joining cluster analysis of H. cinaedi strains and related bacteria, which was based upon a comparison of 1,375 nucleotides of the 16S rRNA gene. Similarity values between the sequences of the H. cinaedi type strain (accession no. M88150), strain R-5759 (“H. westmeadii”, accession no. U44756; identical 16S rDNA sequences were reported for strains R-5758 and R-5759), strain R-4792, and strain LMG 16312 varied between 99.7 and 98.5%. The levels of 16S rDNA sequence similarity of these H. cinaedi isolates to the “F. rappini” (accession no. M88138) reference strain were in the same range of 99.5 to 98.3%, and similarities to the H. canis (accession no. L13464) and H. bilis (accession no. U18766) reference strains were only slightly lower (between 97.9 and 98.7%). Not surprisingly, the H. cinaedi strains do not form a distinct cluster in the dendrogram but cluster together with the “F. rappini,” H. canis, and H. bilis reference strains (Fig. 4). The levels of 16S rDNA sequence similarity of strain R-927 (both sequences) to the other H. cinaedi isolates examined were between 97.1 and 96.5% only. The two sequences for this strain were most similar (98%) to that of the H. fennelliae reference strain (accession no. M88154), thereby confirming the observations reported by Husmann et al. (13).
FIG. 4.
Neighbor-joining phylogenetic tree of H. cinaedi strains and related bacteria based on 16S rRNA sequence comparisons. The scale bar indicates 5% sequence dissimilarity.
DISCUSSION
It is well known and widely accepted that the biochemical identification of Helicobacter strains by means of a limited number of commonly used tests is extremely difficult. In the present study, we used two conceptually different approaches to identify 14 putative helicobacters to the species level. The accuracy of the identifications suggested by these methods was checked by DNA-DNA hybridization, which is generally considered the standard for species discrimination (34). Previous studies with a range of different bacteria have demonstrated a correlation between the degree of whole-cell protein pattern similarity as revealed by one-dimensional sodium dodecyl sulfate-PAGE and the level of DNA-DNA hybridization. Vandamme et al. (31) and Costas et al. (2) previously demonstrated that protein pattern analysis is most useful to distinguish Helicobacter species. This was recently confirmed by Jalava et al. (15), who compared DNA-DNA hybridization, whole-cell protein electrophoresis, and biochemical analysis for the differentiation of several gastric Helicobacter species. Similarly, the application of biochemical examination by means of a panel of 64 tests for the identification of Helicobacter strains was validated by the examination of a large collection of well-characterized strains (1, 23).
In the present study, whole-cell protein pattern analysis identified all 14 field isolates unambiguously as H. cinaedi, while the probabilistic analysis of over 60 biochemical test results successfully identified 13 of these strains, as well as a reference strain not examined before. A qualitative DNA-DNA hybridization assay of a selection of 10 strains confirmed that all of the strains belonged to a single species. Previous comparative studies of this qualitative DNA-DNA hybridization assay and the quantitative optical renaturation rates method (5) revealed that the former procedure allowed correct species level classification of strains of various Helicobacter species (14).
In contrast, identification of several of these isolates based on a restricted set of biochemical tests and their levels of 16S rRNA sequence similarity to known Helicobacter species was not straightforward. Husmann et al. (13) reported that strain R-927 represented a novel Helicobacter species referred to as Helicobacter sp. strain Mainz. In our study, its biochemical reactivity pattern and whole-cell protein profile conformed to those of the other H. cinaedi strains (Fig. 1), and DNA-DNA hybridization to the type strain of H. cinaedi revealed a very strong signal (Fig. 2). The main reason for the misidentification of this strain was an overreliance on its unique position in the 16S rDNA sequence-based phylogenetic tree (13) (Fig. 4). This strain indeed has a 16S rRNA gene sequence that is strikingly different from those of other H. cinaedi strains and is in fact more similar to that of the H. fennelliae type strain (Fig. 4). It must be emphasized here that divergence in 16S rRNA gene sequences of up to 4.3% have been reported in another bacterium belonging to the epsilon subdivision of the division Proteobacteria, Campylobacter hyointestinalis (11). For that species, too, DNA-DNA hybridizations confirmed that these strains represented a single genomic species as defined in taxonomic practice (34). For species like this, it is not surprising to find that not all strains cluster together in a phylogenetic tree (Fig. 4).
The classification of strains R-5758 and R-5759 as a novel Helicobacter species, “H. westmeadii” (30), was probably primarily due to failure to grow the isolates under optimal conditions, leading to at least a few equivocal biochemical test results. “H. westmeadii” was reported to be an anaerobic helicobacter, thereby differentiating it from H. cinaedi, its nearest phylogenetic neighbor (Fig. 4). However, like many other helicobacters, both isolates indeed grew very poorly in a microaerobic environment without hydrogen but grew abundantly when hydrogen was supplemented. Incubation of the strains in an incubator routinely used to work with strict anaerobes (atmospheric composition, 5% CO2, 10% H2, and 85% N2) did not yield visible growth. Further biochemical tests performed under these optimal conditions failed to reproduce hippurate activity, and the overall reactivity pattern corresponded to that of typical H. cinaedi strains. Again, DNA-DNA hybridization to the type strain of H. cinaedi revealed a very strong signal (Fig. 2), confirming the identification of these isolates as H. cinaedi. Therefore, the combined evidence from biochemical, protein electrophoretic, and DNA-DNA hybridization analyses indicate unambiguously that “H. westmeadii” is a junior synonym of H. cinaedi.
Undoubtedly, comparison of (nearly) entire 16S rDNA sequences is one of the most powerful tools for establishing the phylogenetic neighborhood of an unknown organism. However, many taxonomic studies have revealed that this approach is often not sensitive enough to identify strains to the species level. Indeed, strains belonging to different species may have identical 16S rRNA gene sequences, and strains of one species may have 16S rRNA genes that differ by up to 3% (27) and even over 4% (11) of the total 16S rRNA gene sequence. There is clearly a lack of knowledge, not only of the strain-to-strain variation within a species, but also of the interoperon variation within a single strain. Therefore, concluding that an unidentified isolate belongs to a particular Helicobacter species because it shares a high percentage of its 16S rRNA gene sequence or concluding that it represents a novel species because it occupies a unique position in the phylogenetic tree or because it shares only 97% of its 16S rRNA gene sequence with its closest neighbor is premature in the absence of appropriate complementary data.
The identification of the present collection of strains as H. cinaedi expands the number of potential reservoirs for infection. H. cinaedi was first described by Totten et al. (29) to delineate a group of Campylobacter-like organisms isolated from homosexual men suffering from enteritis, proctitis, or proctocolitis and has subsequently been isolated in cases of meningitis, bacteremia, and enteritis in humans, mainly those with immature or compromised immune systems (24, 31, 32). Recently, Weir et al. (35) described another helicobacter isolated from the blood of a patient with AIDS. All available information suggests that this isolate is H. cinaedi too. The whole-cell fatty acid and biochemical profiles conform with those of typical H. cinaedi strains (note that Trivett-Moore et al. [30] reported the “H. westmeadii” strains to be indistinguishable from H. cinaedi when examined by means of cellular fatty acid analysis). Indeed, the authors reported the absence of nitrate reduction as a criterion to separate their strain from H. cinaedi, but of a total of 26 H. cinaedi strains present in our biochemical database (data from the present study and from reference 2), 4 did not reduce nitrate. Absence of nitrate reduction is clearly not uncommon in H. cinaedi, and as discussed above, from the perspective of species level identification, the reliability of using 16S rDNA sequence similarity levels between 98.7 and 99.2% (35) is suspect, since intraspecies variation greater than this has been reported on more than one occasion. Our growing knowledge on the multiple reservoirs of H. cinaedi supports the hypothesis of Weir and coworkers that H. cinaedi (including “H. westmeadii” and Helicobacter sp. strain Mainz) may represent a Helicobacter species that is prone to cause sepsis in immunocompromised patients, such as those with AIDS. We now know that this bacterium occurs in hamsters (8), cats and dogs (16) (Table 1), and foxes and rats (Table 1). Its role in these animal hosts is not known. Good incubation and identification strategies will be required to establish the genuine significance of this Helicobacter species in bacteremic and enteric disease. Identification strategies by means of whole-cell protein or fatty acid analysis, extended biochemical testing, or restriction profile analysis of PCR amplicons derived from the 23S rRNA gene (12, 17) should be considered to support tentative identification results obtained by comparison of complete 16S rRNA genes. As useful as the latter method is, present data clearly indicate that it cannot be regarded as the “gold standard” for species-level identification of many members of the epsilon subdivision of the division Proteobacteria (Helicobacter, Campylobacter, Arcobacter, and related bacteria).
ACKNOWLEDGMENTS
P.V. is indebted to the Fund for Scientific Research—Flanders (Belgium) for a position as postdoctoral fellow. C.S.H. thanks the European Molecular Biology Organisation (EMBO) for a short-term research fellowship award, during which some of the work described here was performed.
We thank E. Falsen (CCUG Culture Collection, University of Göteborg, Göteborg, Sweden) and F. Thomson-Carter (Scottish Campylobacter Reference Laboratory, Aberdeen, Scotland) for strains and P. Jordan for technical assistance. We thank L. Gilbert and M. Yuen (Institute of Clinical Pathology and Medical Research, Centre for Infectious Diseases & Microbiology Laboratory Services, Westmead Hospital, Westmead, Australia) for sharing the “Helicobacter westmeadii” strains and for their willingness to help in solving the questions concerning the taxonomic status of this species.
REFERENCES
- 1.Atabay H I, Corry J E L, On S L W. Identification of unusual Campylobacter-like organisms in poultry products as Helicobacter pullorum. J Appl Microbiol. 1998;84:1017–1024. doi: 10.1046/j.1365-2672.1998.00438.x. [DOI] [PubMed] [Google Scholar]
- 2.Costas M, On S L W, Owen R J, Lopez-Urquijo B, Lastovica A J. Differentiation of Helicobacter species by numerical analysis of their one-dimensional electrophoretic protein patterns. Syst Appl Microbiol. 1993;16:396–404. [Google Scholar]
- 3.De Groote D, van Doorn L-J, Ducatelle R, Verschuuren A, Haesebrouck F, Quint W G V, Jalava K, Vandamme P. ‘Candidatus Helicobacter suis’, a gastric helicobacter from pigs, and its phylogenetic relatedness to other gastrospirilla. Int J Syst Bacteriol. 1999;49:1769–1777. doi: 10.1099/00207713-49-4-1769. [DOI] [PubMed] [Google Scholar]
- 4.De Groote D, van Doorn L-J, Ducatelle R, Verschuuren A, Tilmant K, Quint W G V, Haesebrouck F, Vandamme P. Phylogenetic characterization of ‘Candidatus Helicobacter bovis’, a new gastric helicobacter in cattle. Int J Syst Bacteriol. 1999;49:1707–1715. doi: 10.1099/00207713-49-4-1707. [DOI] [PubMed] [Google Scholar]
- 5.De Ley J. Reexamination of the association between melting point, buoyant density, and chemical base composition of deoxyribonucleic acid. J Bacteriol. 1970;101:738–754. doi: 10.1128/jb.101.3.738-754.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dewhirst F E, Seymour C, Fraser G J, Paster B J, Fox J G. Phylogeny of Helicobacter isolates from bird and swine feces and description of Helicobacter pametensis sp. nov. Int J Syst Bacteriol. 1994;44:553–560. doi: 10.1099/00207713-44-3-553. [DOI] [PubMed] [Google Scholar]
- 7.Eaton K A, Dewhirst F E, Paster B J, Tzellas N, Coleman B E, Paola J, Sherding R. Prevalence and varieties of Helicobacter species in dogs from random sources and pet dogs: animal and public health implications. J Clin Microbiol. 1996;34:3165–3170. doi: 10.1128/jcm.34.12.3165-3170.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gebhart C J, Fennell C L, Murtaugh M P, Stamm W E. Campylobacter cinaedi is normal intestinal flora in hamsters. J Clin Microbiol. 1989;27:1692–1694. doi: 10.1128/jcm.27.7.1692-1694.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Goto K, Ohashi H, Ebukuro S, Itoh K, Tohma Y, Takakura A, Wakana S, Ito M, Itoh T. Isolation and characterization of Helicobacter species from the stomach of the house musk shrew (Suncus murinus) with chronic gastritis. Curr Microbiol. 1998;37:44–51. doi: 10.1007/s002849900335. [DOI] [PubMed] [Google Scholar]
- 10.Hänninen M-L, Happonen I, Saari S, Jalava K. Culture and characteristics of Helicobacter bizzozeronii, a new canine gastric Helicobacter sp. Int J Syst Bacteriol. 1996;46:160–166. doi: 10.1099/00207713-46-1-160. [DOI] [PubMed] [Google Scholar]
- 11.Harrington C S, On S L W. Extensive 16S ribosomal RNA gene sequence diversity in Campylobacter hyointestinalis strains: taxonomic, and applied implications. Int J Syst Bacteriol. 1999;49:1171–1175. doi: 10.1099/00207713-49-3-1171. [DOI] [PubMed] [Google Scholar]
- 12.Hurtado A, Owen R J. A rapid identification scheme for Helicobacter pylori and other species of Helicobacter based on 23S rRNA gene polymorphisms. Syst Appl Microbiol. 1997;20:222–231. [Google Scholar]
- 13.Husmann M, Gries C, Jenichen P, Woelfel T, Gerken G, Ludwig W, Bhakdi S. Helicobacter sp. strain Mainz isolated from an AIDS patient with septic arthritis: case report and nonradioactive analysis of 16S rRNA sequence. J Clin Microbiol. 1994;32:3037–3039. doi: 10.1128/jcm.32.12.3037-3039.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jalava K, Kaartinen M, Utriainen M, Happonen I, Hänninen M-L. Helicobacter salomonis sp. nov., a canine gastric Helicobacter sp. related to Helicobacter felis and Helicobacter bizzozeronii. Int J Syst Bacteriol. 1997;48:975–982. doi: 10.1099/00207713-47-4-975. [DOI] [PubMed] [Google Scholar]
- 15.Jalava K, On S L W, Vandamme P A R, Happonen I, Sukura A, Hänninen M-L. Isolation and identification of Helicobacter spp. from canine and feline gastric mucosa. Appl Environ Microbiol. 1998;64:3998–4006. doi: 10.1128/aem.64.10.3998-4006.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kiehlbauch J A, Brenner D J, Cameron D N, Steigerwalt A G, Makowski J M, Baker C N, Patton C M, Wachsmuth I K. Genotypic and phenotypic characterization of Helicobacter cinaedi and Helicobacter fennelliae strains isolated from humans and animals. J Clin Microbiol. 1995;33:2940–2947. doi: 10.1128/jcm.33.11.2940-2947.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Marshall S M, Melito P L, Woodward D L, Johnson W M, Rodgers F G, Mulvey M R. Rapid identification of Campylobacter, Arcobacter, and Helicobacter isolates by PCR-restriction fragment length polymorphism analysis of the 16S rRNA gene. J Clin Microbiol. 1999;37:4158–4160. doi: 10.1128/jcm.37.12.4158-4160.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.On S L W, Atabay H I, Corry J E L, Harrington C S, Vandamme P. Emended description of Campylobacter sputorum and revision of its infrasubspecific (biovar) divisions, including C. sputorum bv. paraureolyticus, a urease-producing variant from cattle and humans. Int J Syst Bacteriol. 1998;48:195–206. doi: 10.1099/00207713-48-1-195. [DOI] [PubMed] [Google Scholar]
- 19.On S L W, Holmes B. Effect of inoculum size on the phenotypic characterisation of Campylobacter species. J Clin Microbiol. 1991;29:923–926. doi: 10.1128/jcm.29.5.923-926.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.On S L W, Holmes B. Reproducibility of tolerance tests that are useful in the identification of campylobacteria. J Clin Microbiol. 1991;29:1785–1788. doi: 10.1128/jcm.29.9.1785-1788.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.On S L W, Holmes B. Assessment of enzyme detection tests useful in identification of campylobacteria. J Clin Microbiol. 1992;30:746–749. doi: 10.1128/jcm.30.3.746-749.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.On S L W, Holmes B. Classification and identification of campylobacters, helicobacters and allied taxa by numerical analysis of phenotypic characters. Syst Appl Microbiol. 1995;18:374–390. [Google Scholar]
- 23.On S L W, Holmes B, Sackin M. A probability matrix for the identification of campylobacters, helicobacters, and allied taxa. J Appl Bacteriol. 1996;81:425–432. doi: 10.1111/j.1365-2672.1996.tb03529.x. [DOI] [PubMed] [Google Scholar]
- 24.Orlicek S L, Welc D, Kuhls T L. Septicemia and meningitis caused by Helicobacter cinaedi in a neonate. J Clin Microbiol. 1993;31:569–571. doi: 10.1128/jcm.31.3.569-571.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pitcher D G, Saunders N A, Owen R J. Rapid extraction of bacterial genomic DNA with guanidium thiocyanate. Lett Appl Microbiol. 1989;8:151–156. [Google Scholar]
- 26.Pot B, Vandamme P, Kersters K. Analysis of electrophoretic whole-organism protein fingerprints. In: Goodfellow M, O'Donnell A G, editors. Modern microbial methods. Chemical methods in prokaryotic systematics. Chichester, United Kingdom: J. Wiley and Sons, Ltd.; 1994. pp. 493–521. [Google Scholar]
- 27.Stackebrandt E, Goebel B M. Taxonomic note: a place for DNA-DNA reassociation and 16S rRNA sequence analysis in the present species definition in bacteriology. Int J Syst Bacteriol. 1994;44:846–849. [Google Scholar]
- 28.Tee W, Leder K, Karroum E, Dyall-Smith M. “Flexispira rappini” bacteremia in a child with pneumonia. J Clin Microbiol. 1998;36:1679–1682. doi: 10.1128/jcm.36.6.1679-1682.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Totten P A, Fennell C L, Tenover F C, Wezenberg J M, Perine P L, Stamm W E, Holmes K K. Campylobacter cinaedi (sp. nov.) and Campylobacter fennelliae (sp. nov.): two new Campylobacter species associated with enteric disease in homosexual men. J Infect Dis. 1985;151:131–139. doi: 10.1093/infdis/151.1.131. [DOI] [PubMed] [Google Scholar]
- 30.Trivett-Moore N L, Rawlinson W D, Yuen M, Gilbert G L. Helicobacter westmeadii sp. nov., a new species isolated from blood cultures of two AIDS patients. J Clin Microbiol. 1997;35:1144–1150. doi: 10.1128/jcm.35.5.1144-1150.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vandamme P, Falsen E, Pot B, Kersters K, De Ley J. Identification of Campylobacter cinaedi isolated from blood and feces of children and adult females. J Clin Microbiol. 1990;28:1016–1020. doi: 10.1128/jcm.28.5.1016-1020.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.van der Ven A J A M, Kulberg B J, Vandamme P, Meis J F G M. Helicobacter cinaedi bacteremia associated with localized pain but not with cellulitis. Clin Infect Dis. 1996;22:710–712. doi: 10.1093/clinids/22.4.710. [DOI] [PubMed] [Google Scholar]
- 33.Warren J R, Marshall B. Unidentified curved bacilli on gastric epithelium in active chronic gastritis. Lancet. 1983;i:1273–1275. [PubMed] [Google Scholar]
- 34.Wayne L G, Brenner D J, Colwell R R, Grimont P A D, Kandler P, Krichevsky M I, Moore L H, Moore W E C, Murray R G E, Stackebrandt E, Starr M P, Trüper H G. Report of the ad hoc committee on reconciliation of approaches to bacterial systematics. Int J Syst Bacteriol. 1987;37:463–464. [Google Scholar]
- 35.Weir S C, Gibert C L, Gordin F M, Fischer S H, Gill V J. An uncommon Helicobacter isolate from blood: evidence of a group of Helicobacter spp. pathogenic in AIDS patients. J Clin Microbiol. 1999;37:2729–2733. doi: 10.1128/jcm.37.8.2729-2733.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]