Abstract
Rhino-orbito-cerebral mucormycosis (ROCM) is a life-threatening addition to the COVID-19 disease spectrum and is caused by an angioinvasive saprophytic opportunistic fungus. Early diagnosis is important to avoid disease spread and mortality. Contrast-enhanced magnetic resonance imaging plays a major role in detection of intraorbital and intracranial extension. We present imaging findings of 15 patients with post-COVID-19 rhino-orbito-cerebral mucormycosis who were diagnosed with invasive sinus mucormycosis at our institution and are currently undergoing treatment. All patients were diabetics, and 80% had a history of steroid intake during the course of COVID-19 treatment. There was a male preponderance (73.3%). The maxillary sinus was most commonly involved (86.7%). Orbital and intracranial invasion was seen in 73.3% and 60% of patients, respectively. The presence of retroantral, facial, infratemporal, and orbital fat stranding was an early sign of extrasinus spread. Other common sites of extrasinus involvement were the orbit and face, followed by the orbital apex, masticator space, pterygopalatine fossa, bone, skull base, cavernous sinus, brain, and internal carotid artery. In conclusion, early detection of extrasinus spread of mucormycosis by imaging is important so that aggressive treatment can be given and mortality can be reduced.
Keywords: COVID-19, extrasinus spread, mucormycosis
Mucormycosis is an opportunistic, aggressive, and angioinvasive fungal infection, and there has been a significant increase in rhino-orbito-cerebral mucormycosis (ROCM) infections in post-COVID patients. Hypoxia, hyperglycemia, an acidic medium, and high iron levels serve as an ideal environment for fungal growth. In addition, immunosuppression causes decreased phagocytic activity of white blood cells.1 Imaging helps to confirm the clinical suspicion, assessing the extent of disease and complications, and helps in follow-up. Contrast-enhanced magnetic resonance imaging (MRI) is the imaging investigation of choice. In this article, we present a series of 15 patients who were diagnosed with ROCM after COVID-19 and are undergoing treatment at our tertiary center.
METHODS
We conducted a prospective observational study of all patients who came to the radiology department for assessment of ROCM from May 15, 2021, to July 15, 2021. All the patients had current or past COVID-19 infection and underwent contrast-enhanced MRI of the brain, orbits, and paranasal sinuses.
Among the 15 cases, 11 (73%) were men and 4 (27%) were women, and they ranged in age from 38 to 62 years. All the patients were diabetic, 12 (80%) had a history of steroid intake, and 5 (33%) were hypertensive. The patients presented with pain and heaviness over the cheek; facial pain, numbness, and swelling; nasal congestion and itching; and headache. The most common sinus involved was the maxillary sinus, seen in 13 (87%) patients, followed by the ethmoidal sinus in 11 (73%) patients. Multiple sinus involvement was seen in all patients; none had single sinus involvement. The nasal cavity and nasal turbinate were involved in 11 (73%) patients (Figure 1). Orbital invasion was seen in 11 (73%) of 15 patients, and all showed involvement of orbital fat and extraocular muscles (Table 1, Figure 1). Periantral fat stranding was seen in 10 patients, and infratemporal fossa fat stranding was present in 7 patients; however, extension into the pterygopalatine fossa was seen in only 2 cases. Intracranial involvement was seen in 9 (60%) patients (Table 1, Figure 1). Only 1 patient had hard palate erosions.
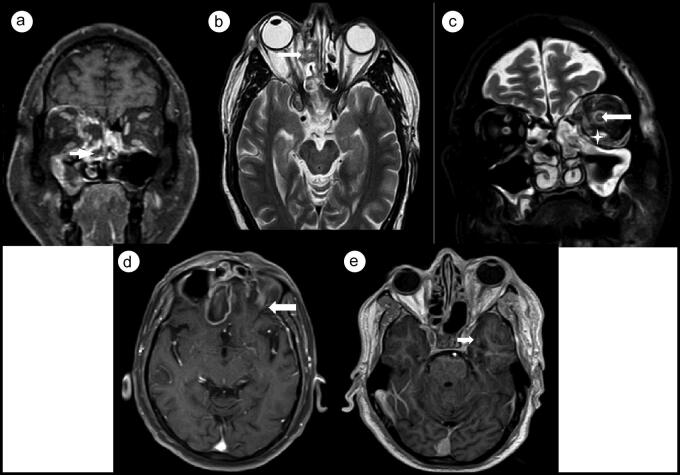
Figure 1.
(a) Post-gadolinium T1-weighted image, with the right middle turbinate showing reduced enhancement, suggestive of the black turbinate sign (arrow). (b) T2-weighted axial image showing soft tissue in the medial extraconal space (arrow), with lateral displacement of the medial rectus. (c) T2-weighted coronal image depicting bulky left extraocular muscles with increased signal (star). Diffuse thickening of the left optic nerve sheath with a hyperintense signal in the left optic nerve suggests optic neuritis (arrow). (d) Post-gadolinium T1-weighted image showing a right inferior frontal gyrus abscess with thin peripheral enhancement (arrow). (e) Post-gadolinium T1-weighted image showing lack of contrast enhancement in the right internal carotid artery (arrow).
Table 1.
MRI findings in 15 patients with intraorbital and intracranial extension of mucormycosis
| Extension | n (%) |
|---|---|
| Intraorbital | |
| Bony erosions of lamina papyracea | 6 (40%) |
| Soft tissue and fat stranding (intraconal and extraconal) | 11 (73%) |
| Involvement of extraocular muscles | 11 (73%) |
| Involvement of optic nerve | 7 (47%) |
| Intracranial | |
| Cavernous sinus | 9 (60%) |
| Internal carotid artery | 2 (13%) |
| Dural enhancement | 3 (20%) |
| Brain abscess | 1 (7%) |
| Brain infarct | 7 (47%) |
| Intracerebral edema | 3 (20%) |
| Skull base | 2 (13%) |
Functional endoscopic sinus surgery and debridement was done in 12 (80%) patients; orbital exenteration, in 8 (53%) patients; and maxillectomy, in 3 patients. Out of the 15 patients, 1 patient died due to severe pneumonitis. Based on a potassium hydroxide mount, mucormycosis was diagnosed in all patients.
DISCUSSION
Mucormycosis is caused by a life-threatening saprophytic fungus normally present in the environment, such as in the skin, soil, air, dust, and spoiled food. It mainly affects the immunocompromised population as well as patients with poorly controlled diabetes mellitus. With COVID-19, a significant number of cases have been reported. Mucormycosis can affect numerous bones and organs of the body, but ROCM is the most common (68%) variety observed in patients globally, followed by pulmonary (13%) and cutaneous (11%).2
Notably, ROCM is a wide-ranging term signifying limited sino-nasal disease, limited rhino-orbital disease, and/or rhino-orbital-cerebral disease. The disease can present with headache, nasal discharge, facial pain, facial swelling, decreased vision, fever, and epistaxis primarily. Other common sites of extrasinus involvement are orbit and face, followed by orbital apex, masticator space, pterygopalatine fossa, bone, skull base, cavernous sinus, brain, and internal carotid artery.3 Patients with cerebral involvement can even present with single cranial nerve palsy or multiple cranial nerve palsies depending on the magnitude of involvement.
The cases included in our study had a male predominance, as was observed by Awadesh Kumar Singh et al. In addition, all cases in our study were diabetic, and 80% had a history of steroid intake. This observation is in line with the conclusions by Awadesh Kumar Singh et al, with the most common risk factors of preexisting diabetes mellitus and use of steroids.4 A study by Patel et al also concluded that in the Indian population, preexisting diabetes mellitus is the most frequent predisposing factor (73.5%), followed by malignancy (9%) and transplantation (7.7%). Due to the angioinvasive nature of the disease, skull base osteomyelitis and bone involvement is usually not seen or is seen only late in the disease. Nine of our cases showed intracranial involvement, 2 showed skull base invasion, and only 1 had hard palate erosions.
On imaging, aggressive sinonasal and orbital changes caused by the disease have been seen by Mnif et al and Herrera et al.5,6 Many studies have shown that cavernous sinus thrombosis and vascular complications of the disease can be detected by MRI. Cavernous sinus involvement appears hypointense on T1 and T2 with intense inhomogeneous postcontrast enhancement. Contrast-enhanced computed tomography and MRI are the best imaging modalities for the detection of Mucor.7 In our study, contrast-enhanced MRI of the brain, orbits, and paranasal sinuses was done for all 15 cases. As Silverman et al described, the presence of retroantral, facial, and orbital fat stranding indicates the aggressive nature of the infection. Periantral fat stranding was present in 10 patients, and 7 patients showed infratemporal fossa fat stranding; only 2 patients showed extension into the pterygopalatine fossa.8
Most authors recommend prompt surgical debridement and long-term antifungal therapy. Surgery is used to remove necrotic tissue, reduce the concentration of fungal spores, and facilitate the effect of antifungal drugs. For treatment purposes, functional endoscopic sinus surgery and debridement were performed in 12 patients, orbital exenteration in 8, and maxillectomy in 3 patients. Nevertheless, reversal of predisposing factors is an indispensable part of treatment. In conclusion, early detection of extrasinus spread of mucormycosis by imaging is important so that aggressive treatment can be given and mortality can be reduced.
References
- 1.Hingad N, Kumar G, Deshmukh R.. Oral mucormycosis causing necrotizing lesion in a diabetic patient: a case report. Int J Oral Maxillofac Pathol. 2012;3(3):8–12. [Google Scholar]
- 2.Patel A, Kaur H, Xess I, et al. A multicentre observational study on the epidemiology, risk factors, management and outcomes of mucormycosis in India. Clin Microbiol Infect. 2020;26(7):944.e9–e15. doi: 10.1016/j.cmi.2019.11.021. [DOI] [PubMed] [Google Scholar]
- 3.Therakathu J, Prabhu S, Irodi A, Sudhakar SV, Yadav VK, Rupa V.. Imaging features of rhinocerebral mucormycosis: a study of 43 patients. Egypt J Radiol Nucl Med. 2018;49(2):447–452. doi: 10.1016/j.ejrnm.2018.01.001. [DOI] [Google Scholar]
- 4.Singh AK, Singh R, Joshi SR, Misra A.. Mucormycosis in COVID-19: a systematic review of cases reported worldwide and in India. Diabetes Metab Syndr. 2021;15(4):102146. doi: 10.1016/j.dsx.2021.05.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mnif N, Hmaied E, Oueslati S, et al. L'imagerie dans la mucormycose rhinocérébrale [Imaging of rhinocerebral mucormycosis]. J Radiol. 2005;86(9):1017–1020. doi: 10.1016/S0221-0363(05)81485-4. [DOI] [PubMed] [Google Scholar]
- 6.Herrera DA, Dublin AB, Ormsby EL, Aminpour S, Howell LP.. Imaging findings of rhinocerebral mucormycosis. Skull Base. 2009;19(2):117–125. doi: 10.1055/s-0028-1096209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Prabhu RM, Patel R.. Mucormycosis and entomophthoramycosis: a review of the clinical manifestations, diagnosis and treatment. Clin Microbiol Infect. 2004;10(Suppl 1):31–47. doi: 10.1111/j.1470-9465.2004.00843.x. [DOI] [PubMed] [Google Scholar]
- 8.Silverman CS, Mancuso AA.. Periantral soft-tissue infiltration and its relevance to the early detection of invasive fungal sinusitis: CT and MR findings. AJNR Am J Neuroradiol. 1998;19(2):321–325. [PMC free article] [PubMed] [Google Scholar]