Abstract
Brain metastases (BrM) are treated with multimodality therapy, however the optimal combination and timing of modalities in the setting of recurrent tumours that have failed prior treatments remain poorly defined. We present a case of a patient with biopsy-confirmed renal cell carcinoma BrM with good performance status initially treated with laser interstitial thermal ablation therapy (LITT) followed by stereotactic radiosurgery and dual checkpoint inhibitor immunotherapy. He subsequently developed rapid in-field recurrence which was treated with salvage surgical resection and implantation of intracavitary cesium-131 brachytherapy. The patient’s disease remained stable through 18 months postoperatively. This case illustrates the range of options available and provides a combination salvage therapy strategy in a select group of locally recurrent patients who have exhausted conventional treatment options.
Keywords: cancer intervention, neurooncology, CNS cancer, neurosurgery
Background
Brain metastases (BrM) affect 30% of cancer patients and constitute an important patient population given improving survival rates in common cancers including lung and breast carcinomas, and melanoma.1 2 Current treatment paradigms focus on local disease control through radiotherapy for small or polymetastatic tumours; and surgical resection plus adjuvant radiotherapy for large oligometastatic lesions, with local control for these approaches ranging from 62% to 94%.2–5 In rare cases, particularly for tumours where open resection carries significant morbidity, early data suggests a role for laser interstitial thermal therapy (LITT) ablation.6 In addition, an increasing number of systemic cancer-directed therapies have central nervous system (CNS) activity, including receptor tyrosine kinase inhibitors and immunotherapies.7–9 More recently, the use of intracavitary cesium-131 (Cs-131) brachytherapy has studied in BrM with promising results,10 however the timing and indication for its use remain undefined.
Despite the range of therapeutic options, disease can still recur, necessitating salvage strategies. However, salvage treatments often harbour a trade-off between therapeutic benefit and compounded toxicities.11 For example, BrM re-irradiation carries increased risk of radionecrosis (RN), and resection following radiation or previous resection carries risks of wound compromise and neurologic morbidity.12 13 Moreover, local control after resection without adjuvant radiotherapy is poor for tumours that have recurred following first-line therapies.14
Here we describe a multimodality treatment strategy for a young patient whose BrM recurred despite two local therapies (LITT followed by stereotactic radiosurgery (SRS)) plus dual CNS-active systemic immunotherapy (nivolumab plus ipilimumab). He had excellent performance status with well-controlled extracranial disease and was offered open salvage resection with intracavitary Cs-131 brachytherapy implantation to reduce mass effect and provide local control. At 18 months postoperatively, there is no evidence of recurrence.
Case presentation
A 53-year-old man with a history of previously stage one renal cell carcinoma (RCC) treated with definitive left nephrectomy 14 years prior without adjuvant therapy presented with 1 month of left hand paresthesias and weakness. Brain imaging demonstrated a right parietal mass (figure 1A), and systemic re-staging did not show any extracranial disease. Functional imaging showed the lesion to be in close apposition to both thalamocortical and corticospinal tracts. Given the tumour’s eloquent location and the need for tissue diagnosis, he underwent magnetic resonance imaging-guided stereotactic biopsy and ablative LITT shortly after initial presentation (figure 1B isodose line equivalent to 43°C for 10 min). The biopsy pathology returned as clear cell RCC metastasis, and targeted tumour sequencing panel (MSK-IMPACT) demonstrated mutation in the Von Hippel Lindau tumour suppressor gene and no other currently targetable mutations. He subsequently underwent serial imaging surveillance, which showed rapid marginal recurrence in the posterior aspect of the tumour shortly afterwards. He underwent adjuvant SRS 1 month thereafter (27 Gy in three fractions; figure 1C). Concurrent re-staging scans also identified lung metastases, and he was started on dual checkpoint blockade immunotherapy with nivolumab and ipilimumab (270 mg/90 mg every 4 weeks for four cycles) followed by maintenance nivolumab (480 mg every 4 weeks for 12 months). Although his extracranial disease remained stable, he re-presented with left lower limb weakness 3 months later (grade 3/5 power) and interval brain imaging showed haemorrhagic changes in the treated index tumour in the corona radiata, now 2.4 cm in maximal diameter, with enhancement along the previous biopsy and laser therapy tract concerning for recurrence.15
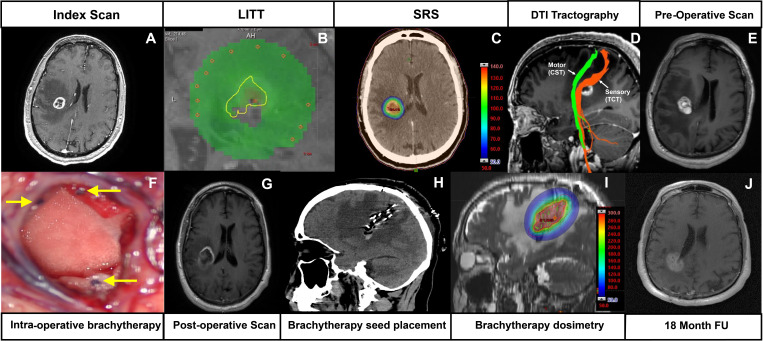
Figure 1.
Sequence of imaging findings. (A) Initial scan at diagnosis shows a ring-enhancing lesion in the right parietal centrum semiovale. (B) Laser interstitial thermal therapy isothermal lines during treatment: the yellow isodose line represents 43°C for 2 min equivalent. (C) Stereotactic radiosurgery dosimetry scan: the colour wash scale bar represents radiation dose proportion, where 100% (red) represents total radiation dose given: 9×3 Gy=27 Gy. (D) Diffusion tensor tractography demonstrating location of corticospinal tract and thalamocortical tract in relation to lesion. (E) Preoperative MRI scan with gadolinium demonstrating recurrence and peri-lesional oedema. (F) Representative intraoperative picture of Cs-131 seed impregnated tiles within a resection cavity. Filament-embedded seeds are seen in cross-section (yellow arrows). (G) Axial T1-weighted MRI with gadolinium showing postoperative resection cavity. (H) Postoperative CT demonstrating the brachytherapy tile placement and orientation of Cs-131 seeds. (I) Brachytherapy dosimetry scan demonstrating estimated radiation coverage. The outer red line represents 5 mm expansion margin estimated to represent a dose of 60 Gy. (J) follow-up T1-weighted MRI scan with gadolinium at 18 months.
Investigations and differential diagnosis
Initial gadolinium-enhanced brain MRI at first presentation demonstrated a right parietal lesion measuring 2.1×1.6×1.6 cm within the centrum semiovale with extensive vasogenic oedema. Concurrent systemic re-staging positron emission tomography (PET) was not suspicious for new extracranial disease. Therefore, the differential diagnosis included metastatic RCC, but in the setting of stable treated and low-stage RCC, a primary brain malignancy such as glioma was also possible. Functional MRI with diffusion tensor tractography was used to identify functional white matter tracts, to rule out impingement, and to define potential surgical corridors to the lesion. The lesion was found to be closely related to both the corticospinal and thalamocortical tracts. LITT was performed as it permitted concurrent diagnostic biopsy and treatment. The patient subsequently was treated with SRS following LITT. In the setting of post-SRS BrM, PET imaging or MR perfusion imaging can often be helpful in the setting of differentiating between post-SRS radionecrosis and tumour recurrence, which are managed differently.16 17 Pure radiation necrosis is often observed and medically managed, for example, with corticosteroids or bevacizumab; extreme cases are resected and in select cases LITT is offered while re-irradiation is avoided where possible.12 Recurrent disease generally requires local therapy, for example resection or re-irradiation.
In this case both imaging modalities suggested tumour rather than RN, which was supported by the lesion’s rapid growth and enhancement along the previous LITT tract.
Treatment
Following multidisciplinary discussion, in which it was determined that he was an appropriate candidate for salvage therapy, he underwent right-sided parietal craniotomy with intraoperative subcortical motor mapping to help inform thalamocortical tract location. The recurrences at the original site and tract were removed, and Cs-131 brachytherapy was placed at a prescription dose of 60 Gy to 5 mm depth from the cavity edge (GammaTile, GT Medical Technologies, USA) 5 months after BrM diagnosis (figure 1E, F).
Outcome and follow-up
The patient had mild improvement in left lower limb power to 4/5 immediately postoperatively which subsequently improved to near normal function, and he remains clinically and radiographically stable at 18 month follow-up (figure 2).
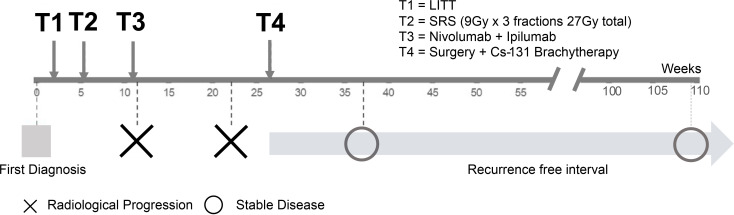
Figure 2.
Timeline in weeks showing treatment interventions above time axis and disease course below time axis.
Discussion
We report the case of durable treatment effect with brachytherapy as a salvage therapy modality after previous treatment with LITT, SRS and CNS-active dual immunotherapy. Although clinical trial data exists for all these modalities, descriptions of combination usage of modalities in the setting of multiply recurrent and highly pretreated BrM are sparse.
Postoperative SRS for surgically resected BrM has become the predominant treatment paradigm with the local control rate at 1 year in prospective studies ranging between 62% and 78%.4 18 Recent large-scale single-institutional and multi-institutional retrospective cohort studies in patients who underwent cavity SRS post-resection have found local control rates of 85%–93% at 1 year, falling to 75% and 71% at 2 and 3 years, respectively, which is also likely influenced by the timing and technique of SRS administration.19 Therefore a substantial minority of patients require the attention of neurosurgical oncology post-surgery, with or without SRS. Salvage treatment options for this group of patients may have diminishing returns,20 21 and although salvage surgical resection is safe and feasible, local recurrence rates in retrospective series have reported to be in the range of 31% and as high as 44% in our experience (unpublished).18 22 23 This high failure rate is in part explained by the constraints on re-irradiation postoperatively, as repeat SRS has been shown to increase the risk of RN, with the 1 year cumulative risk of RN reported to be 20%.21 24
Despite the lack of consensus and poor salvage efficacy in this group of patients, several strategies have been described. LITT has gained popularity in specialised centres as it obviates radiation dose considerations by delivering targeted thermal energy to lesions and also affords the advantages of tissue diagnosis at the time of surgery, through a minimally invasive percutaneous approach and with shortened length of stay compared with craniotomy.25 However, similar to radiotherapy, adjacency to sensitive structures and eloquent tracts can limit the amount of thermal dose delivery, with concomitant recurrence in underdosed regions as was seen in this case. Long-term efficacy has also yet to be demonstrated.26
One strategy to avoid the risk of repeat SRS while potentially conveying local disease control is surgical resection (with or without intraoperative neurophysiological monitoring of eloquent structures) with brachytherapy implantation.10 27 28 In this patient we implanted Cs-131 seeds embedded within foam tiles. Cs-131 is a brachytherapy radioligand with a favourable dosimetric profile.29 In one of the largest published series using this isotope for BrM, Wernicke et al performed brachytherapy with Cs-131 in 24 patients with 100% local control (defined as recurrence within 5 mm of the resection margin), though with median survival of only 9.9 months in a spectrum of BrM histologies.30 The risk of RN and neurocognitive defects was reported to be low, with only one case reported in 42 study patients.10
In summary, the treatment of recurrent BrM remains challenging due to toxicity and efficacy concerns. The decision to pursue salvage therapies aimed at local control and/or palliation should be carefully considered and discussed with patients. In a select group of patients with well-controlled extracranial disease and high performance status, there is a growing palette of treatment options, though these may have complex interactions which can influence decision-making. Management decisions in this context should therefore be made in a multidisciplinary manner, with expertise on the range of options and their interplay in order to maximise patient benefits, limit treatment delays and act in accordance with patient goals in the setting of at-times limited survival. In the presented case, the patient received surgery and Cs-131 brachytherapy treatment and has remained stable 18 months later without recurrence or radiation toxicity despite multiple prior treatments including SRS and immunotherapy, in a highly treatment-refractory tumour. Future trials to further refine treatment pathways and indications for brachytherapy in the setting of recurrent BrM are needed and are currently underway (NCT04690348).
Learning points.
Brain metastases are becoming more common, and first line therapeutic options are well established.
Patients with stable extracranial disease and who have failed first-line treatment with recurrent brain metastases are a clinical challenge.
Beyond standard treatment modalities such as surgery, chemotherapy and radiotherapy, the suite of treatment modalities now also includes laser interstitial thermal therapy, immunotherapies and brachytherapy.
A multidisciplinary approach should be taken with neurosurgeons, medical oncologists, neuro-radiologists and radiation oncologists who are familiar with these modalities in order to propose customised treatment plans to achieve durable local disease control.
Acknowledgments
We would like to thank Dr Mehrnaz Jenabi for providing assistance with tractography images.
Footnotes
Twitter: @Dr_N_Moss
Contributors: KKHY collected data, assembled figures and wrote manuscript. BI supplied radiation dosimetry data and reviewed manuscript. NSM conceived paper, wrote and edited manuscript. All figures were illustrated and assembled by KY. Intraoperative images were provided by NM, brachytherapy dosimetry plan was provided by BI.
Funding: This study was funded by National Cancer Institute, USA (Cancer Center Support Grant P30 CA008748).
Case reports provide a valuable learning resource for the scientific community and can indicate areas of interest for future research. They should not be used in isolation to guide treatment choices or public health policy.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Ethics statements
Patient consent for publication
Consent obtained directly from patient(s).
References
- 1.Achrol AS, Rennert RC, Anders C, et al. Brain metastases. Nat Rev Dis Primers 2019;5:5. 10.1038/s41572-018-0055-y [DOI] [PubMed] [Google Scholar]
- 2.Bander ED, Yuan M, Reiner AS, et al. Durable 5-year local control for resected brain metastases with early adjuvant SRS: the effect of timing on intended-field control. Neurooncol Pract 2021;8:278–89. 10.1093/nop/npab005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brennan C, Yang TJ, Hilden P, et al. A phase 2 trial of stereotactic radiosurgery boost after surgical resection for brain metastases. Int J Radiat Oncol Biol Phys 2014;88:130–6. 10.1016/j.ijrobp.2013.09.051 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brown PD, Ballman KV, Cerhan JH, et al. Postoperative stereotactic radiosurgery compared with whole brain radiotherapy for resected metastatic brain disease (NCCTG N107C/CEC·3): a multicentre, randomised, controlled, phase 3 trial. Lancet Oncol 2017;18:1049–60. 10.1016/S1470-2045(17)30441-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Soltys SG, Adler JR, Lipani JD, et al. Stereotactic radiosurgery of the postoperative resection cavity for brain metastases. Int J Radiat Oncol Biol Phys 2008;70:187–93. 10.1016/j.ijrobp.2007.06.068 [DOI] [PubMed] [Google Scholar]
- 6.Holste KG, Orringer DA. Laser interstitial thermal therapy. Neurooncol Adv 2020;2:vdz035. 10.1093/noajnl/vdz035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zeeshan Ozair M, Giantini Larsen AM, Eng J, et al. Exceptional response of a large and symptomatic EGFR-mutant brain metastasis to osimertinib: case report and review of the literature. JCO Precis Oncol 2021;5:PO.20.00485:585–8. 10.1200/PO.20.00485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bander ED, Yuan M, Carnevale JA, et al. Melanoma brain metastasis presentation, treatment, and outcomes in the age of targeted and immunotherapies. Cancer 2021;127:2062–73. 10.1002/cncr.33459 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tawbi HA, Chung C, Margolin K. Nivolumab and ipilimumab in melanoma metastatic to the brain. N Engl J Med 2018;379:722–30. 10.1056/NEJMoa1805453 [DOI] [PubMed] [Google Scholar]
- 10.Wernicke AG, Hirschfeld CB, Smith AW, et al. Clinical outcomes of large brain metastases treated with neurosurgical resection and intraoperative Cesium-131 brachytherapy: results of a prospective trial. Int J Radiat Oncol Biol Phys 2017;98:1059–68. 10.1016/j.ijrobp.2017.03.044 [DOI] [PubMed] [Google Scholar]
- 11.Chidambaram S, Pannullo SC, Schwartz TH, et al. Reirradiation of recurrent brain metastases: where do we stand? World Neurosurg 2019;125:156–63. 10.1016/j.wneu.2019.01.182 [DOI] [PubMed] [Google Scholar]
- 12.Newman WC, Goldberg J, Guadix SW, et al. The effect of surgery on radiation necrosis in irradiated brain metastases: extent of resection and long-term clinical and radiographic outcomes. J Neurooncol 2021;153:507–18. 10.1007/s11060-021-03790-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.McKay WH, McTyre ER, Okoukoni C, et al. Repeat stereotactic radiosurgery as salvage therapy for locally recurrent brain metastases previously treated with radiosurgery. J Neurosurg 2017;127:148–56. 10.3171/2016.5.JNS153051 [DOI] [PubMed] [Google Scholar]
- 14.Bindal RK, Sawaya R, Leavens ME, et al. Reoperation for recurrent metastatic brain tumors. J Neurosurg 1995;83:600–4. 10.3171/jns.1995.83.4.0600 [DOI] [PubMed] [Google Scholar]
- 15.Carnevale JA, Imber BS, Winston GM, et al. Risk of tract recurrence with stereotactic biopsy of brain metastases: an 18-year cancer center experience. J Neurosurg 2021:1–7. 10.3171/2021.3.JNS204347 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Grkovski M, Kohutek ZA, Schöder H, et al. 18F-Fluorocholine PET uptake correlates with pathologic evidence of recurrent tumor after stereotactic radiosurgery for brain metastases. Eur J Nucl Med Mol Imaging 2020;47:1446–57. 10.1007/s00259-019-04628-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hatzoglou V, Yang TJ, Omuro A, et al. A prospective trial of dynamic contrast-enhanced MRI perfusion and fluorine-18 FDG PET-CT in differentiating brain tumor progression from radiation injury after cranial irradiation. Neuro Oncol 2016;18:873–80. 10.1093/neuonc/nov301 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mahajan A, Ahmed S, McAleer MF, et al. Post-operative stereotactic radiosurgery versus observation for completely resected brain metastases: a single-centre, randomised, controlled, phase 3 trial. Lancet Oncol 2017;18:1040–8. 10.1016/S1470-2045(17)30414-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Eitz KA, Lo SS, Soliman H, et al. Multi-Institutional analysis of prognostic factors and outcomes after Hypofractionated stereotactic radiotherapy to the resection cavity in patients with brain metastases. JAMA Oncol 2020;6:1901. 10.1001/jamaoncol.2020.4630 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ammirati M, Cobbs CS, Linskey ME, et al. The role of retreatment in the management of recurrent/progressive brain metastases: a systematic review and evidence-based clinical practice guideline. J Neurooncol 2010;96:85–96. 10.1007/s11060-009-0055-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rae A, Gorovets D, Rava P, et al. Management approach for recurrent brain metastases following upfront radiosurgery may affect risk of subsequent radiation necrosis. Adv Radiat Oncol 2016;1:294–9. 10.1016/j.adro.2016.08.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mitsuya K, Nakasu Y, Hayashi N, et al. Retrospective analysis of salvage surgery for local progression of brain metastasis previously treated with stereotactic irradiation: diagnostic contribution, functional outcome, and prognostic factors. BMC Cancer 2020;20:331. 10.1186/s12885-020-06800-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Schackert G, Schmiedel K, Lindner C, et al. Surgery of recurrent brain metastases: retrospective analysis of 67 patients. Acta Neurochir 2013;155:1823–32. 10.1007/s00701-013-1821-y [DOI] [PubMed] [Google Scholar]
- 24.Kohutek ZA, Yamada Y, Chan TA, et al. Long-term risk of radionecrosis and imaging changes after stereotactic radiosurgery for brain metastases. J Neurooncol 2015;125:149–56. 10.1007/s11060-015-1881-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hong CS, Deng D, Vera A, et al. Laser-interstitial thermal therapy compared to craniotomy for treatment of radiation necrosis or recurrent tumor in brain metastases failing radiosurgery. J Neurooncol 2019;142:309–17. 10.1007/s11060-019-03097-z [DOI] [PubMed] [Google Scholar]
- 26.Shao J, Radakovich NR, Grabowski M, et al. Lessons learned in using laser interstitial thermal therapy for treatment of brain tumors: a case series of 238 patients from a single institution. World Neurosurg 2020;139:e345–54. 10.1016/j.wneu.2020.03.213 [DOI] [PubMed] [Google Scholar]
- 27.Mahase SS, Navrazhina K, Schwartz TH, et al. Intraoperative brachytherapy for resected brain metastases. Brachytherapy 2019;18:258–70. 10.1016/j.brachy.2019.01.011 [DOI] [PubMed] [Google Scholar]
- 28.Wernicke AG, Smith AW, Taube S, et al. Cesium-131 brachytherapy for recurrent brain metastases: durable salvage treatment for previously irradiated metastatic disease. J Neurosurg 2017;126:1212–9. 10.3171/2016.3.JNS152836 [DOI] [PubMed] [Google Scholar]
- 29.Yang R, Wang J, Zhang H. Dosimetric study of Cs-131, I-125, and Pd-103 seeds for permanent prostate brachytherapy. Cancer Biother Radiopharm 2009;24:701–5. 10.1089/cbr.2009.0648 [DOI] [PubMed] [Google Scholar]
- 30.Wernicke AG, Yondorf MZ, Peng L, et al. Phase I/II study of resection and intraoperative cesium-131 radioisotope brachytherapy in patients with newly diagnosed brain metastases. J Neurosurg 2014;121:338–48. 10.3171/2014.3.JNS131140 [DOI] [PMC free article] [PubMed] [Google Scholar]