Abstract
Background
Autism Spectrum Disorder (ASD) is a neurodevelopmental disorder characterised by repetitive behaviours, cognitive rigidity/inflexibility, and social-affective impairment. Unfortunately, no gold-standard treatments exist to alleviate the core socio-behavioural impairments of ASD. Meanwhile, the prosocial empathogen/entactogen 3,4-methylene-dioxy-methamphetamine (MDMA) is known to enhance sociability and empathy in both humans and animal models of psychological disorders.
Objective
We review the evidence obtained from behavioural tests across the current literature, showing how MDMA can induce prosocial effects in animals and humans, where controlled experiments were able to be performed.
Methods
Six electronic databases were consulted. The search strategy was tailored to each database. Only English-language papers were reviewed. Behaviours not screened in this review may have affected the core ASD behaviours studied. Molecular analogues of MDMA have not been investigated.
Results
We find that the social impairments may potentially be alleviated by postnatal administration of MDMA producing prosocial behaviours in mostly the animal model.
Conclusion
MDMA and/or MDMA-like molecules appear to be an effective pharmacological treatment for the social impairments of autism, at least in animal models. Notably, clinical trials based on MDMA use are now in progress. Nevertheless, larger and more extended clinical studies are warranted to prove the assumption that MDMA and MDMA-like molecules have a role in the management of the social impairments of autism.
Keywords: 3,4-methylenedioxymethamphetamine; animal; autism spectrum disorder; human; MDMA; social behavior
1. INTRODUCTION
Autism Spectrum Disorder (ASD) is a neurodevelopmental disorder manifesting in early childhood [1, 2]. It is characterised by a triad of behavioural symptoms. These include social reciprocal communication and interaction impairment, motor stereotypies (repetitive behaviours), and cognitive rigidity. Each symptom appears to different extents in different individuals [1, 2]. They are the outcome of abnormal brain physiology developed as early as in utero [3]. Briefly, they involve both genetic and environmental risk factors, affecting synaptogenesis and axon motility downstream [4]. The amygdala and nucleus accumbens are purportedly involved [4].
ASD symptoms result in variable impacts on functioning across all domains of life [5-7]. 85% of children with ASD report difficulties at school, with 63% of those having social difficulties, and 52% of those having communication difficulties [8]. These difficulties continue into adulthood, with the majority of individuals with ASD being unemployed or underemployed [9], predictably leading to a considerably lower quality of life [10, 11]. The reported prevalence rates of ASD are rising worldwide [12], implicating an impending increase in disease burden, necessitating the need to develop more effective interventions, as well as treatments to support this population. While it remains unclear what is driving this increased prevalence in ASD [13], factors such as changes in reporting [14], increased awareness and changes in diagnostic criteria [15], among other theories [13], may be contributing factors. Nevertheless, it is imperative to develop more effective interventions and treatments to support this population.
There are currently no FDA-approved pharmacological treatments for the core impairments that define ASD [16]. Two medications are currently FDA-approved for the treatment of accessory traits, irritability and aggressiveness, in ASD: aripiprazole and risperidone. These are controversial for use, due to their adverse effects [16, 17].
A significant number of off-label medications have also been utilised, such as alpha-2 agonists, mood stabilisers, norepinephrine-reuptake inhibitors, serotonin-reuptake inhibitors, antipsychotics and opioid-receptor antagonists [18, 19]. The latter have either insufficient evidence of efficacy, or are only able to address different accessory symptoms of ASD, such as irritability and hyperactivity [18, 19]. In addition, addressing ASD comorbidities can be fraught with complexities. This includes the need to obtain prescriptions for these comorbid affective disorders (such as anxiety and depression) or comorbid neurological disorders (such as epilepsy) [20, 21].
No drugs have been approved for the core impairments in ASD [22, 23]. However, several drugs have shown early-stage evidence for the treatment of these core behaviours, such as bumetanide, pregnenolone, suramin, sulforaphane, folinic acid, propranolol, oxytocin, vasopressin antagonists, and arbaclofen [22, 23]. Furthermore, many non-pharmacological treatments for ASD also exist, though these fail to target core behavioural traits, or do not yet have a sufficient evidence base to be recommended for widespread clinical use [24]. Psychotherapeutic methods include applied behaviour analysis [25-27], meditation/mindfulness [28], early-childhood and parent education [29], cognitive behaviour therapy [30], music therapy [31], and social-skills training [32, 33]. However, these interventions represent a significant financial and time burden for the individuals with ASD and their families, and only address the accompanying traits of ASD (e.g. anxiety).
There have been numerous human studies undertaken on MDMA intake and social interaction, though most of them have been uncontrolled [34]. In contrast, animal models provide a controlled means to examine the potential impact of pharmacological treatments [35]. This means that the model chosen must hold an empirical and theoretical relationship to autism. The behaviours must be unambiguous and homologous between species (construct validity), the model must resemble autism in its clinical features (face validity), and the model must correctly predict clinical treatments for autism (predictive validity) [36].
An animal model of human psychiatric disease is a more ethically accepted way of exploring neurological aberrational mechanisms and drug treatments, where those drugs have not been approved for human use [37-39]. Rodents (mice and rats) have most commonly been used to model ASD pre-clinically, due to construct and face validity, as well as convenience of use in the laboratory [40, 41]. ASD can be environmentally or genetically induced in them, and treatments are then provided to assess alleviation of those induced ASD traits via specially tailored behavioural assays [42].
As far as we are aware, this would be the first systematic review of the studies encompassing animal and human behavioural effects of MDMA, which are relevant to ASD. We conclude that the prosocial behaviours induced by MDMA may counteract at least the social impairments in autism, at least based on support from animal models.
1.1. 3,4-Methylenedioxymethamphetamine (MDMA)
MDMA is more commonly appreciated as the intended-for-consumption agent in ‘ecstasy’, and is known to have prosocial effects in humans [43-46]. In fact, historically it was used in psychotherapy since the early 1960s [47]. Specifically, MDMA increases emotional empathy and sociability [43, 45]. We stipulate, therefore, that various studies in animal models (and also in humans) may provide insights as to the role of MDMA in ameliorating the pathological lack of sociability featured in some individuals on the autism spectrum. MDMA rodent studies have indeed demonstrated that MDMA increases prosocial behaviour and decreases asocial behaviour, similar to MDMA’s effects on humans [43]. Moreover, a recent pilot trial on humans was fruitful in finding social anxiolytic effects of MDMA [48].
The prosocial effect of MDMA is considered to be mediated primarily by serotonin and oxytocin [44, 49-52]. The primary mechanism of action of MDMA is to act as a substrate for serotonin, norepinephrine, and dopamine monoamine transporters, such that their transport is reversed, thereby their respective synapses are saturated [53]. MDMA’s effects on serotonin release seem most relevant to the oxytocin release and prosocial effects [52] because serotoninergic neurons are known to stimulate oxytocin release [54]. MDMA is, however, also a direct agonist at some serotonin receptors. For example, MDMA has a relatively strong affinity for 5-HT1 and 5-HT2 receptors [55], which may also explain its effects on oxytocin and prosocial behaviour, as oxytocin secretion is mediated mostly by 5-HT1A [52, 56], 5-HT2C, and 5-HT4 receptors [56]. In rat models, MDMA activates oxytocinergic neurons in the supraoptic and paraventricular nuclei of the hypothalamus [52]. This induces oxytocin to be synthesised in these areas and released from the posterior pituitary gland into the peripheral blood [56]. Oxytocin, in turn, is likely to decrease amygdala activation and coupling, which normally triggers fear responses, thereby providing a mechanism for reduced social anxiety [57]. This response can vary between individuals, as one study found that variants of the OXTR gene influences the function/structure of oxytocin receptors on the amygdala, thereby modulating downstream impulses to the brainstem to regulate sympathetic and behavioural fear responses [58]. Single-nucleotide polymorphisms in the OXTR gene have even been detected in autistic individuals) [59-61], further supporting the hypothesis that ASD may be at least in part an issue of decreased empathy and increased anxiety (via oxytocin effects on the amygdala and downstream) [59-61]. Whilst MDMA has not been tested on autistic brains, MDMA has shown decreased amygdalar activity in healthy brains [62-64]. Autism is dependent on synaptic plasticity, and there is also plenty of evidence that MDMA has effects on synaptic plasticity [65-67]. We direct the reader to an excellent review on the molecular effects of MDMA [68], as going more into detail is beyond the scope of this RCT-focused review.
1.2. Aims/rationale
We aim to show that, throughout the animal and human literature, MDMA has been shown to have prosocial effects at certain doses (subject to mode of administration and timing of dosing). Hence, clinical studies using MDMA to alleviate the social impairment in ASD are warranted to help establish MDMA as a clinically approved drug in ASD patients. Of note, MDMA could be able to manage a core impairment constituting ASD, which has never been directly addressed by an approved drug before. We also aim to investigate the animal and human literature to see whether MDMA has had effects on the other two core impairments in ASD, namely stereotypy and cognitive rigidity. This latter goal could shed light on the dose adjustments required to optimally reduce these other impairments in future human clinical studies, as well as acting on the social impairment we focus on in this review. We refer to both acute and chronic administration. Acute means a singular dose that is administered to the test animal or human, whereas chronic means several doses administered to the test animal or human over time. The exact timings of these doses are specified in the tables, alongside their respective study. Testing itself may occur between or after the chronic doses, and this is also specified for each study.
2. METHODS
2.1. Study Design
A systematic review of the literature exploring how MDMA influences the presentation of ASD-like characteristics, in particular social behaviours, in animals and humans, was conducted. Specifically, these are studies in rodent strains without autism-salient mutations or exposures, and humans without autism, as no preceding papers have tested MDMA in organisms with ASD.
2.2. Search Strategy
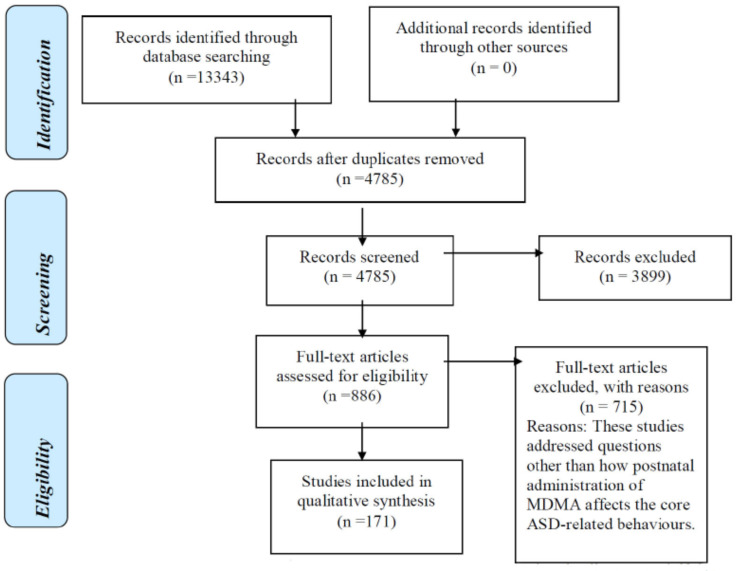
Six electronic databases including ProQuest, PsychInfo, Scopus, Medline, Web of Science and PubMed (search cut-off date: 21/05/20 inclusive) were consulted. The search strategy, tailored to each database, is detailed in Fig. (2). Only English-language studies were used. The study process is outlined via the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, illustrated in Fig. (1).
Fig. (2).
Search strategy, in each database, for both animal and human studies pertaining to the effects of MDMA on ASD-related behaviour.
Fig. (1).
The study numbers used in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines used for this study, for the animal behavioural tests.
2.3. Study Inclusion and Exclusion Criteria
This systematic review identifies literature examining the effects of postnatal MDMA administrations on the core behaviours affected in ASD, in rats and mice, other animals and humans. This includes MDMA treatment at any age past birth. These include social impairment, repetitive behaviour and cognitive rigidity [1, 2]. These diagnostic criteria are taken from DSM-5 and ICD-11, the current gold-standard reference texts for psychological disorders [1, 2] . To this end, the following behavioural tests have been included for rodents: 1) ultrasonic-vocalisation; 2) social-preference; 3) social-novelty-preference; 4) social-interaction; 5) open-field; 6) T/Y-maze; 7) marble-burying; and 8) novel-object-recognition. This review places no limits on age, species or cognitive abilities of the subjects. The inclusion criteria were behaviours core to ASD, behavioural experiments on animals, the use of MDMA to affect the behavioural results, and placebo-controlled studies. The exclusion criteria were applied to papers that only assessed the accessory behaviours to ASD and behaviours not related to ASD, drug self-administration or reinforcement studies, prenatal MDMA administration, or behaviours associated with disorders other than ASD (including Rett syndrome), drugs other than MDMA, uncontrolled studies, and serotonin syndrome. With rodents, approximately 3573 studies were screened, and 127 studies finally reviewed. With other animals, approximately 3456 studies were screened, and 16 studies finally reviewed. With humans, approximately 6205 studies were screened, and 49 studies finally reviewed. “Healthy” human subjects refer to non-autistic patients, whether MDMA-experienced or -naïve, and whether diagnosed with other psychological conditions or not. All were given singular or chronic MDMA doses.
2.4. Data Extraction and Synthesis
2.4.1. Rodent and Non-rodent Animals
From animal studies, the data extracted were as follows: species and sex tested, sample size, dosage timing and frequency, dosage mass and route, treatment and testing ages, ambient temperature (as temperature was shown to influence behavioural outcomes with MDMA) [69], and results of the experimental animals as compared to the control group. The findings are summarised in Tables 1-9. In this review, dosage routes have been categorised as singular (only one dose for the subjects) and chronic (multiple doses for a subject). It is worth noting that some papers use the term, “sub-chronic”, to indicate multiple doses. Initial extraction of the data revealed that MDMA generally serves to increase intra-species sociable behaviours in rodents.
2.4.2. Humans
From human studies, the data extracted were as follows: the method of recruitment of the participants, the population type the participants were selected from, number of participants, sex ratio, mean age of the participants, the oral dose of MDMA administered (this was the only administration route used in humans), timing of doses administered, the time between MDMA intake and the first core-ASD-relevant measurement made, the relevant tests undertaken, the relevant results obtained, and the exact parameters taken into account for each relevant test. The findings are summarised in Tables 10 and 11. Initial extraction of the data revealed that MDMA generally serves to increase social behaviour and altruistic feelings in humans.
2.4.3. Interspecies Focus
We take an inter-species perspective in investigating these behavioural effects of MDMA, in the form of a systematic literature review. This is because it is our intention to attempt to translate laboratory-controlled animal studies to their potential impact in humans, where such rigorous techniques cannot be trialled in humans directly [70].
3. RESULTS
Below, we summarise the general trends of behavioural effect that MDMA has had on animals and humans in the laboratory (placebo-controlled) experiments. Where we state “chronic” doses, these are doses delivered more than once. The timings of these dosages are specified for each study in the tables below. The act of repetitive grooming, whether self- or allo-grooming, is classed as repetitive behaviour in these studies, both by the respective study authors and ourselves, but we consider them also as possible signs of cognitive rigidity.
3.1. Core ASD Behaviour 1: Social Impairment
3.1.1. Rodents
There are 4 major tests, currently in the literature, testing social and communication behaviour among mice and rats of different strains. These are the ultrasonic-vocalisation test, the social-preference test, the social-novelty-preference test and the social-interaction test [71-73]. We have also included a novel-object-recognition test here, to see whether the behavioural effects seen in the social-novelty-preference test are dependent on social or general novelty. In the studies reviewed, a “conspecific” will be mentioned, which is a member of the same species (in these cases, a mouse or rat). Below, we summarise each of these tests along with the MDMA-induced alterations on the respective induced social impairments in rodents.
Each study is shown alongside the species, sex and sample size of the rodents tested (control and relevant treatment groups), whether the study used singular or chronic (multiple) dosing, and if they used chronic dosing, the timing of the doses; the dose and route (mode of injection) used, the ages the rodents were treated and tested at, the temperature of the testing environment, and the results obtained from the experiment in the treated rodents (where it is different from the results in the corresponding control rodents). s.c. = subcutaneous mode of injection.
Each study is shown alongside the species, sex and sample size of the rodents tested (control and relevant treatment groups), whether the study used singular or chronic (multiple) dosing, and if they used chronic dosing, the timing of the doses; the dose and route (mode of injection) used, the ages the rodents were treated and tested at, the temperature of the testing environment, and the results obtained from the experiment in the treated rodents (where it is different from the results in the corresponding control rodents). The studies used singular dosing. i.p. = intraperitoneal mode of injection.
Each study is shown alongside the species, sex and sample size of the rodents tested (control and relevant treatment groups), whether the study used singular or chronic (multiple) dosing, and since they used chronic dosing, the timing of the doses; the dose and route (mode of injection) used, the ages the rodents were treated and tested at, the temperature of the testing environment, and the results obtained from the experiment in the treated rodents (where it is different from the results in the corresponding control rodents). i.p. = intraperitoneal mode of injection.
3.1.1.1. Ultrasonic-vocalisation Tests
The ultrasonic-vocalisation test is performed prior to weaning of the pup, and is a measure of the pup’s ultrasonic vocalisation (USV) ability in response to being separated from its mother [74]. It is believed that the purpose of these distress calls is to induce the mother to retrieve the pup back to its home litter [75]. The normal USV emission by maternally separated rodents is shown to be increased in the rate of calls on PD 6-12, which persists till PD 15 and then declines [76]. Teratogenic agents administered in utero may result in deviations from this pattern as well as the sounds therein. In ASD rodents, MDMA caused the abnormal calls to return to having the normal decrease and subsequent increase in call rate that is observed in healthy rodents within a day [76].
We found one study where MDMA was given post-natally, and the pups were measured for vocalisation ability upon maternal separation (Table 1). The study shows that when 10 mg/kg MDMA is injected subcutaneously singularly to pups pre-weaning, call frequency (the number of calls emitted) decreases initially, then increases later within a day [76]. When the same is given multiple times at an earlier age, later ages show decreased frequency in proportion to dose [76].
Table 1.
Ultrasonic vocalisations (upon maternal separation) from postnatally MDMA-treated rodents, compared with control rodents.
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size |
Singular/
Chronic Dosing |
Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Males | Females | Both | No. Pups per Mother |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Winslow 1990 [76] | Rat | Both | Combined | 42 | Singular | NA | 0.5-10 | s.c. | 9-11 | 9-11 | 24 | NA | NA | Decreased rate of pup calls, increased locomotion. 10 mg/kg: call frequency decreased at 0.5 and 3 hours post-injection, increased at 10 and 24 hours post-injection. | 9-12 |
| Winslow 1990 [76] | Rat | Both | Combined | 36 | Chronic | 7 injections in total | 10 | s.c. | 1-4 | 6, 9, 12, 15 | 24 | NA | NA | PD 9-15: dose-dependent long-lasting decreased call rate |
9-12 |
3.1.1.2. Social-preference Tests
The social-preference test assesses a rodent’s preference to spend time with a conspecific vs. an inanimate object. Thus, the apparatus for the test consists of three chambers: a “home base” and two flanking chambers containing either a caged unknown sex- and age-matched conspecific or an “object”. Control rodents usually spend more time in and make more entries into, the chamber with the caged conspecific than the chamber with the empty cage, indicating social preference. Studies directly take this as a social-preference index, being the ratio of time spent in the conspecific chamber over either the time spent in the object chamber or the total time spent in all chambers. We found three studies where 3-15 mg/kg MDMA, given once intra-peritoneally approximately on PD 56-84, acutely increased rodent social preference (Table 2).
Table 2.
Social preferences in postnatally MDMA-treated rodents, compared with control rodents.
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size | Singular/Chronic Dosing | Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Males | Females | Both |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Heifets 2019 [187] | Mouse | Both | NA | 36-80 | Singular | NA | 3, 7.5, 15 | i.p. | NA | NA | NA | NA | NA | 7.5 and 15 mg/kg: dose-dependently increased |
| Kuteykin-Teplyakov 2014 [188] | Mouse | Males | NA | 59 | Singular | NA | 2, 3, 4, 6 | i.p. | 56-84 | 56-84 | 21 | 3 mg/kg: increased | NA | NA |
| Ramos 2016 [189] | Rat | Males | NA | 64 | Singular | NA | 2.5, 5 | i.p. | “Adult” (250-300g) | “Adult” (250-300g) | 21 | Well-handled, 5 mg/kg: increased. Minimally handled, 5 mg/kg: no effect. 2.5 mg/kg: no effect. | NA | NA |
3.1.1.3. Social-novelty-preference Tests
The social-novelty-preference test assesses a rodent’s preference to spend time with an unfamiliar conspecific over a familiar conspecific. The test uses the same set-up as the social-preference test, but the object (empty cage) is replaced by a new sex- and age-matched conspecific (the unfamiliar). The familiar is the previously encountered conspecific in the opposite flanking chamber. Studies directly take this as a social-novelty-preference index, being the ratio of time spent in the unfamiliar chamber over either the time spent in the familiar chamber or the total time spent in all chambers. We found one study where 5 or 10 mg/kg MDMA, given chronically intraperitoneally on PD 28-52, increased mouse social-novelty preference when tested later on PD 120 (Table 3). This therefore, also shows a long-lasting prosocial effect of the MDMA, which would increase the value of MDMA as a treatment.
Table 3.
Social-novelty preferences in postnatally MDMA-treated rodents, compared with control rodents.
| Study | Species | Sexes |
Sexes
Combined or Separated |
Sample Size |
Singular/
Chronic Dosing |
Chronic Timing |
Dose
(mg/kg) |
Route |
Treatment
Age (PD) |
Testing
Age (PD) |
Tempera-
ture (°C) |
Males | Females | Both |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Morley-Fletcher 2002 [190] | Mouse | Both | Separated | NA | Chronic | Once on each of 3 days, 2 days apart for each |
5, 10 | i.p. | 28, 38, 52 | 120 | 21 | Increased | Increased | NA |
3.1.1.4. Novel-object-recognition Tests
The novel-object-recognition test assesses novelty preference and memory for a new object replacing an existing familiar object. We have classed this in the “social impairment” tests in order to compare with the “social-novelty preference” tests. In control rodents, the norm is to spend more time with the unfamiliar than the familiar object, indicating a natural curiosity to explore the unexplored, and is indicative of an intact representation that the existing environment has changed.
We found 33 studies where MDMA was given mostly chronically postnatally, and the rodent offspring were measured for their tendency to spend more time exploring the unfamiliar/novel object than the familiar (Table 4). The studies show that giving mostly chronic 5-10 mg/kg MDMA, intraperitoneally or subcutaneously, actually exacerbates this loss in the rodent’s exploratory discrimination between the two objects, which is in contrast to our expected effect on social impairment (see social-novelty-preference test). This may confirm that if there is disinterest in both social and non-social novelty, this may indicate cognitive rigidity as opposed to social impairment, because MDMA appears to exacerbate cognitive rigidity (see below). Interestingly, one study shows that a higher temperature (28°C) exacerbates this supposed memory deficit more than a cooler temperature (16oC) [77].
Table 4.
Novel-object preferences in postnatally MDMA-treated rodents, compared with control rodents.
| Study | Species | Sexes |
Sexes Combined
or Separated |
Sample Size |
Singular/
Chronic Dosing |
Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Males | Females | Both |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Abad 2014 [191] | Rat | Males | NA | 14-18 | Chronic | Twice daily for 4 days | 20 | s.c. | 35 | 7 days after treatment ended | 26 | No effect | NA | NA |
| Abad 2016 [192] | Rat | Males | NA | 14-18 | Chronic | 3 times a day, every 3 hours, once a week for 2 weeks | 5 | NA | 28-49 | 28-49 | 26 | Reduced | NA | NA |
| Abad 2016 [192] | Rat | Males | NA | 14-18 | Chronic | 3 times a day, every 3 hours, once a week for 2 weeks for another 3 weeks | 7.5 | NA | 49-70 | 49-70 | 26 | Reduced | NA | NA |
| Abad 2016 [192] | Rat | Males | NA | 14-18 | Chronic | 3 times a day, every 3 hours, once a week for 2 weeks for another 3 weeks | 10 | NA | 70-91 | 70-91 | 26 | Reduced | NA | NA |
| Able 2006 [193] | Rat | Males | NA | 40 | Chronic | Once every 2 hours, for 4 doses on 1 day | 15 | s.c. | 225-250g | 5 days later | 22 | No effect | NA | NA |
| Adeniyi 2016 [194] | Mouse | Males | NA | 10 | Chronic | 5 times over 10 days, at 2-day intervals | 2 | s.c. | 21 | 31, 32 | NA | Reduced | NA | NA |
| Bubenikova-Valesova 2010 [195] | Rat | Males | NA | NA | Chronic | 4 days | 2.5, 5 | s.c. | NA | NA | NA | No effect | NA | NA |
| Clemens 2007 [136] | Rat | Females | NA | 16 | Chronic | 1 injection per week, for 16 weeks | 8 | i.p. | 238g | 120 days later | 28 | NA | No effect | NA |
| Cohen 2005 [196] | Rat | Males | NA | 30 | Chronic | Twice a day (8 hours apart) | 20 | s.c. | 11-20 | 40-49 | 21 | Reduced | NA | NA |
| Cohen 2005 [196] | Rat | Males | NA | 30 | Chronic | 4 times, at 2 intervals | 15 | s.c. | 82-100 | 131-129 | 21 | No effect | NA | NA |
| Edut 2011 [197] | Mouse | Males | NA | 34 | Singular | NA | 10 | i.p. | 25-30g | “Juvenile” | 23 | No effect | NA | NA |
| Edut 2014 [198] | Mouse | Males | NA | 18-26 | Singular | NA | 10 | i.p. | 25-30g | 7, 30 days later | 23 | No effect | NA | NA |
| García-Pardo 2017 [94] | Mouse | Males | NA | 45 | Singular | NA | 5, 10 | i.p. | 42 | 63, 64 | 35-37 | Reduced | NA | NA |
| Llorente-Berzal 2013 [199] | Rat | Both | Separated | 55 | Chronic | Every 5 days, twice daily (4 hours apart) | 10 | s.c. | 30-45 | 75 | 22 | No effect | No effect | NA |
| Ludwig 2008 [200] | Rat | Males | NA | 39+ | Singular | Single injection | 5 | s.c. | “Adult” (242-275g) | 7 days later | NA | No effect | NA | NA |
| Ludwig 2008 [200] | Rat | Males | NA | 39+ | Chronic | 5 daily injections | 5 | s.c. | “Adult” (242-275g) | 7 days later | NA | No effect. Multiple doses: increased | NA | NA |
| Study | Species | Sexes |
Sexes Combined
or Separated |
Sample Size |
Singular/
Chronic Dosing |
Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Males | Females | Both |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ludwig 2008 [200] | Rat | Males | NA | 39+ | Both | Chronic, then single | 5 | s.c. | “Adult” (242-275g) | 7 days later | NA | Multiple doses: increased | NA | NA |
| McGregor 2003 [77] | Rat | Males | NA | 32 | Chronic | 2 consecutive days, every hour for 4 hours | 5 | i.p. | 60-75 | 70-84 days later | 28, 16 | 28°C: reduced | NA | NA |
| Meyer 2008 [201] | Rat | Males | NA | NA | Chronic | 2 doses, 4 hours apart | 10 | s.c. | 35, 40, 45, 50, 55, 60 | 35, 40, 45, 50, 55, 60 | 22-23 | Reduced | NA | NA |
| Nawata 2010 [202] | Mouse | Males | NA | 10-30 | Singular | NA | 10 | i.p. | 30-35g | 30-35g | 23 | No effect | NA | NA |
| Nawata 2010 [202] | Mouse | Males | NA | 10-30 | Chronic | Once daily for 7 days | 10 | i.p. | 30-35g | 30-35g | 23 | Reduced | NA | NA |
| Piper 2004 [203] | Rat | Males | NA | 16 | Chronic | On every 5th day, twice daily, intervals of 4 hours | 10 | s.c. | 35-60 | 65 | 22 | Reduced | NA | NA |
| Piper 2005 [204] | Rat | Males | NA | 20 | Chronic | Hourly intervals over 4 hours, once every 5 days | 5 | s.c. | 35-60 | 67, 68, 69 | 22 | Reduced | NA | NA |
| Piper 2008 [205] | Rat | Males | NA | 20-24 | Chronic | 4 doses, 1 each hour | 10 | s.c. | Young adult (307.7g) | Young adult | 23 | Reduced | NA | NA |
| Rodsiri 2011 [206] | Rat | Males | NA | 21-24 | Chronic | Every 2 hours over 6 hours (3 injections) | 3, 6 | i.p. | 100-130g | 14 days later | 21 | 3 × 6 mg/kg: reduced. 6 mg/kg: no effect | NA | NA |
| Ros-Simo 2013 [207] | Mouse | Males | NA | 20-24 | Chronic | Twice, 6 hours apart | 20 | i.p. | 25 | 28 | 22 | Reduced, long-term | NA | NA |
| Schulz 2013 [208] | Rat | Males | NA | 24 | Chronic | Once daily for 10 days, twice daily (4 hours apart) for 5 days | 7.5 | s.c. | “Adult” (230-300g) | 40-65, 80-105 | 22 | Reduced | NA | NA |
| Shortall 2012 [209] | Rat | Both | Combined | 16 | Chronic | Once daily for 7 days | 5 | i.p. | 170-205g | 7 days later | NA | NA | NA | No effect |
| Shortall 2013 [210] | Rat | Males | NA | 12-16 | Chronic | 2 consecutive days a week, for 3 weeks | 10 | i.p. | Young adult | Young adult | 21 | Reduced | NA | NA |
| Skelton 2008 [211] | Rat | Males | NA | 63 | Chronic | 4 per day (2 hours apart), 1 day per week, 5 weeks (1-week intervals) | 15 | s.c. | 225-250g | 35-39 days later | 22 | No effect | NA | NA |
| van Nieuwenhuijzen 2010 [212] | Rat | Males | NA | 24 | Chronic | Daily over 10 days | 5 | i.p. | 220-300g | 220-300g | 21 | Reduced | NA | NA |
| Vorhees 2007 [213] | Rat | Both | Separated | 160 | Chronic | Every 2 hours, each day | 40 once a day, 20 twice a day, or 10 four times a day | s.c. | 11-20 | 64-68 | 21 | No effect | No effect | NA |
| Vorhees 2009 [214] | Rat | Both | Separated | NA | Chronic | 4 doses, every 2 hours each day | 10, 15, 20, 25 | s.c. | 1-20 | 60 | 21 | No effect | No effect | NA |
3.1.1.5. Social-interaction Tests
The social-interaction test is a simple and direct test of social interaction. The test rodent and a conspecific of the same treatment group are placed in an open arena and their behaviour monitored. Different behavioural acts are measured, and specialist terms are listed in Table 5.1.
We found 59 studies where MDMA was given singularly or chronically postnatally, and the rodents observed for their inter-conspecific social interactions (Table 5). The studies show that singular doses of 5-10 mg/kg MDMA, given intraperitoneally, increases prosocial behaviour and decreases asocial behaviour. Interestingly, at these same doses, chronic dosing has the opposite effect: decreasing prosocial behaviour and increasing asocial behaviour (Table 5). One study was an exception to this [78], where 5-20 mg/kg MDMA given twice daily over 3 days actually increased the duration of social investigation. On either side of the dosage window, 5-10 mg/kg MDMA, this opposite effect is also seen: decreased prosocial behaviour and increased asocial behaviour (Table 5).
Table 5.
Social-interaction behaviours in postnatally MDMA-treated rodents, compared with control rodents.
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size |
Singular/
Chronic Dosing |
Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Social Restriction | Males | Females | Both |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ando 2006 [87] | Rat | Males | NA | 34 | Either | 1 or 2 days (49 days apart) | 15 | i.p. | 49-56 | 49-56 | 21 | Nil | Decreased regular total social-interaction time, locomotion, self-grooming, rearing. Increased adjacent-lying time (more than social-interaction decrease). | NA | NA |
| Ando 2006 [87] | Rat | Males | NA | 34 | Either | 1 or 2 days (49 days apart) | 15 | i.p. | 49-56 | 70-77 | 21 | Nil | 3 weeks later, dosed on 1st day: increased aggression, rearing and line crossings. Acute, dosed on 2nd day: increased total time of social behaviour (adjacent lying), total number of non-social activities and line crossings. |
NA | NA |
| Ando 2010 [215] | Rat | Males | NA | 24 | Singular | NA | 15, 30 | i.p. | 42-49 | 222-229 | 21 | Nil | Fewer line crossings. No change in duration or number of social interactions, aggressive behaviours. | NA | NA |
| Bull 2003 [133] | Rat | Males | NA | 24 | Chronic | Twice daily | 15 | i.p. | 28-30 | 50 | 21 | Nil | Reduced total social interaction. | NA | NA |
| Bull 2003 [216] | Rat | Both | NA | NA | Chronic | Four times daily, for 2 consecutive days | 5 | i.p. | 28 | 84 | NA | Nil | After 54 days' abstinence, social interaction decreased by 27% | NA | NA |
| Bull 2004 [132] | Rat | Males | NA | 32 | Chronic | Each of 4 hours | 5 | i.p. | 28 | 84 | 21 | Nil | Increased social anxiety, decreased total social interaction. | NA | NA |
| Cagiano 2008 [84] | Rat | Males | NA | 90 | Singular | NA | 0.3, 1, 3 | i.p. | “Adult” | “Adult” | 20-22 | Nil | 3 mg/kg: increased time till intromission and ejaculation, decreased copulatory activity. | NA | NA |
| Clemens 2004 [217] | Rat | Males | NA | 24-30 | Chronic | 4 injections in 1 day, 2 hours apart | 2.5, 5 | i.p. | 376g | 28 days later | 28 | Nil | Increased total locomotor activity. 5 mg/kg: reduced social-interaction time. | NA | NA |
| Clemens 2005 [218] | Rat | Females | NA | 16-18 | Chronic | Every 2 hours in 1 day, 4 times | 4 | i.p. | 281g | 42 days later | 28 | Nil | NA | Less social interaction. | NA |
| Clemens 2007 [136] | Rat | Females | NA | 32 | Chronic | 1 injection per week for 16 weeks | 8 | i.p. | 238g | 49 days later | 28 | Nil | NA | Decreased total social-interaction time. | NA |
| Clemens 2007 [136] | Rat | Females | NA | 16 | Chronic | 1 injection per week for 16 weeks | 8 | i.p. | 238g | 113 days later | 28 | Nil | NA | Reduced social-interaction time. | NA |
Interestingly, one study saw a difference in MDMA’s effects on aggressive vs. timid mice: whilst MDMA increased timidity in both types of mice, MDMA increased social behaviour in aggressive mice, and decreased social behaviour in timid mice [79]. Timidity was determined in a preliminary interaction test where mice would be defined as “timid” if they showed no attack, but significant defensive-escape behavior, even in the absence of partner aggression, when placed in a neutral arena with a conspecific [79]. If timidity is a factor in influencing sociability, this finding would make sense, since excessively timid mice would not be inclined to socialise, and aggressive mice without tempering with timidity would not be inclined to socialise. However, timid rats were more likely to become aggressive when a young intruder is introduced into the cage, when given MDMA chronically at 6 mg/kg [80].
Each study is shown alongside the species, sex and sample size of the rodents tested (control and relevant treatment groups), whether the study used singular or chronic (multiple) dosing, and if they used chronic dosing, the timing of the doses; the dose and route (mode of injection) used, the ages the rodents were treated and tested at, the temperature of the testing environment, and the results obtained from the experiment in the treated rodents (where it is different from the results in the corresponding control rodents). i.p. = intraperitoneal mode of injection; s.c. = subcutaneous mode of injection.
Each study is shown alongside the species, sex and sample size of the rodents tested (control and relevant treatment groups), whether the study used singular or chronic (multiple) dosing, and if they used chronic dosing, the timing of the doses; the dose and route (mode of injection) used, the ages the rodents were treated and tested at, the temperature of the testing environment, the duration of social restriction (isolation) enforced before testing, and the results obtained from the experiment in the treated rodents (where it is different from the results in the corresponding control rodents). i.p. = intraperitoneal mode of injection; s.c. = subcutaneous mode of injection.
There have also been temperature, inter-sex-interaction and group effects seen. Similar to human studies with MDMA, a study found that the higher temperature increased social interaction more than the lower temperature [69]. In fact, some studies deliberately increased the testing temperature to 28oC, in order to amplify these social effects [81-83]. Only one study investigated inter-sex interactions, and found that singular 3 mg/kg MDMA decreased the inclination to copulate [84], similar to the doses below 5 mg/kg MDMA, which decreased prosocial behaviour. A study also found that group-housed mice became more physically active, more so than single-housed mice, when given MDMA; and that social interaction duration was increased in the group-housed and not the single-housed group [85]. This is similar to human studies where prosocial effects of MDMA appear enhanced when taken in groups of people [86].
Some interesting effects are noted as follows: one study found a difference between acute and chronic effects of MDMA: when tested on the same day, prosocial behaviour was increased in MDMA-treated rats; when tested 3 weeks later, asocial behaviour was increased a day later, but when dosed again, it acutely increased prosocial behaviour [87]. One study’s singular 8 mg/kg dose caused decreased social exploration [88], another study’s singular 5-10 mg/kg dose also decreased social exploration [89], and another’s singular 5 mg/kg increased asocial behaviours [90]. Notably, these studies socially isolated their animals for 30 days prior to testing, to increase aggressive behaviour [88-90]. Perhaps, in these cases, MDMA’s effects could not entirely overcome that increased aggressive behaviour. Some studies had a significant time delay between testing and treating: 65, 70 and 21 days [91-93], with surprising results of decreased prosocial behaviour [91-93] and increased asocial behavior [93]. One study used social defeat as a variable to influence social interaction, where they used 30 mins. of isolating the test rodent before testing, to induce aggression during testing [94]. Three other studies gave surprising results: social and prosocial behaviours were decreased when MDMA was given at a singular 5-10 mg/kg [95-97].
To summarise the social behavioural tests in rodents, USV calls increased gradually in emission rate when 10 mg/kg MDMA is given singularly or chronically postnatally (Table 1). Rodents’ preference to spend time with a conspecific over an object, or an unfamiliar conspecific over a familiar, increases when given 5-10 mg/kg MDMA singularly or chronically postnatally (Tables 2, 3). Social behavioural results are mixed between the studies, when 5-10 mg/kg MDMA is given singularly or chronically postnatally (Table 5). The literature also shows reports of chronic effects, whereby MDMA-treated rodents retain these prosocial effects (Tables 3, 4, 5).
3.1.2 Other Animals
Most of the non-rodent animal studies focused on social behaviour after MDMA treatment. Since dosage effects are different between species, we will observe each animal model in turn. Four studies tested the behavioural effects of MDMA on monkeys [98-101]. They found that chronic administration of 1.5 mg/kg s.c. [98], or singular dose of 0.03-3 mg/kg i.m. [100] increased prosocial behaviour. Interestingly, Pitts et al. (2017) showed that with lower doses, it was the S(+) MDMA enantiomer that increased this gregariousness more, and at higher doses, the R(−) MDMA enantiomer increased affiliative behaviour more. Chronic 1.5 mg/kg (p.o. and especially i.m.) increased vigilance, indicating perhaps decreased social trust [101]. But the studies also found that chronic 12 mg/kg p.o. decreased vocalisations [99], and that both 12 mg/kg p.o. and 20 mg/kg i.g. [102] induced serotonin behaviour, indicating this range was an unnecessarily high dose.
Four studies examined ASD-specific behaviour in MDMA-treated fish, in particular social behaviour [103-106]. They found that when 1 or 5 mg/kg MDMA was singularly intramuscularly injected into electric fish, asocial behaviour decreased and prosocial behaviour increased [103]. Interestingly, in zebrafish, singularly immersing the animals in 80 mg/L actually increased the distance between adjacent fish and shortened the duration spent near each other [104]. This may be explained by the fact that in one study, which tested for general anxiety in zebrafish, immersing the animals in 0.25-120 mg/L increased the chance of the fish entering and dwelling at the top of the fish tank [107], indicating reduced anxiety. This is because fish tend to become anxious towards the water’s surface as there is a greater risk of predation there [108, 109], and shoaling behaviour is generally a result of alarm pheromones dissipated among the fish [104, 110]. Ponzoni et al. (2016, 2017) found that a singular intramuscular injection of 0.1-10 mg/kg MDMA increased social preference [105, 106]. Two studies looked at social behaviour in singularly MDMA-immersed octopodes [111, 112]. They found that when octopodes were doused in 0.5-0.005 mg/kg MDMA solution, not only did social preference increase [111, 112], but so did voluntary body contact between the animals [112].
3.1.3 Humans
The human studies tested MDMA effects on non-autistic subjects, except for one study which looked specifically at the effects of MDMA on social anxiety in autistic adults [48]. In this study, autism was diagnosed if the subject fulfilled Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders- Fourth Edition Axis I Research Version (SCID-I-RV) or Autism Diagnostic Observation Schedule (ADOS-2 Module 4) criteria. The assessments in all studies were between subjects, where one group were given MDMA and the other placebo. Most studies assessed mostly chronically given 1.5 mg/kg (often by an absolute dose of 100 mg) which may increase the likelihood of finding positive results. We assume a linear positive relationship (gradient: 1.5 mg/kg per 100 mg; intersect: 0 mg/kg = 0 mg) between mg/kg and mg, and convert absolute doses to per kg of participant body weight accordingly. Each study had its own method of measuring social outcomes, therefore classifying into types of tests (as able to be done for the rodent model) would be inaccurate. Specific details of these tests are therefore listed in the tables for each respective study.
As shown in Table 10, all doses increased prosocial behaviour. This includes mostly self-perceptions of extroversion, openness, sociability, talkativeness, thoughtfulness or caring, sensitivity, friendliness, insightfulness of others, gregariousness or desire to be with others, empathy, lovingness, playfulness, closeness to others. Interestingly, social interaction increased more at 0.5 mg/kg than 1 mg/kg in one study [86], and playfulness and lovingness increased more at 1.5 than 0.75 mg/kg (chronic dosing).
Table 10.
Social behaviour in postnatally MDMA-treated subjects, compared with placebo-treated subjects.
| Study | Recruitment | Population Sampled | No. Participants | Sexes | Mean Ages (years) | Oral MDMA Dose | Dosage Timing | Time till First Test (after baseline) | Relevant Tests | Relevant Results | Relevant Parameters Tested | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Baggott 2016 [284] | Newspaper, online adverts, word of mouth | Healthy, MDMA-experienced | 12 | 6 female, 6 male | 29 | 1.5 mg/kg | Singular | 1.5 hours | Self-reports of social anxiety and interpersonal functioning | Decreased social anxiety; increased affiliative (nurturance/communion) feelings | Apprehension towards being judged; dominance/agency and nurturance/communion | ||||||||
| Baggott 2015 [285] | Newspaper, community bulletin board, online adverts | Healthy, MDMA-experienced, Caucasian | 35 | 12 female, 23 male | 24.3 | 1.5 mg/kg | Singular | 4 hours | Verbal recounts of significant others and own emotions | Increased words with social/sexual content. No sex differences. Increased words with factual, decreased words with psychological, content about target person. No changes in phrases referring to relationship to target person, decreased words relating to body of target person and increased words related to cognition and insight | Names of 3 personally important people, altered use of words, discussion topics, number of words spoken, isolated descriptions of target person, proportion and number of phrases describing relationship to target person, words relating to target person's body vs. cognition and insight, drug-related emotions (including social); words with social, negative and positive valence | ||||||||
| Bajger 2015 [286] | Word of mouth, online and newspaper adverts, poster flyers | Black, Hispanic, white | 12 | 3 female, 9 male | 28.9 | 50, 100 mg (~0.75, 1.5 mg/kg) | Twice daily over 5 days | 60 mins. | Self-reports of affect | Repeated dose: increased gregariousness, decreased preference for solitude, 100 mg: increased time spent verbally communicating with others | Private and social (talking and silence) time, mood, preference for solitude or gregariousness, effects of singular vs. repeated doses | ||||||||
| Bedi 2014 [287] | Adverts | Healthy, ecstasy-experienced, mostly Caucasian | 13 | 4 female, 9 male | 24.5 | 0.75, 1.5 mg/kg | Singular | Mood: 65 mins.; speech: 130 mins. | Verbal recounts of significant others | 1.5 mg/kg: words had increased proximity to “friend”, “support”, “intimacy”, “rapport”. 0.75 mg/kg: words had increased proximity to “empathy” | Degrees of “compassion”, “empathy”, “forgive”, “friend”, “intimacy”, “love”, “rapport”, “support”, “talk”, “confident” | ||||||||
| Bedi 2010 [288] | Internet adverts, word of mouth | Healthy, ecstasy-experienced, mostly Caucasian | 21 | 9 female, 12 male | 24.4 | 0.75, 1.5 mg/kg | 4 times, 5/+-day intervals | Mood: 0; emotion recognition: 65 mins. | Self-reports of mood, emotion-recognition tasks | 1.5 mg/kg: increased ratings of “loving”, “friendly”, “playfulness”, decreased accuracy of facial fear recognition in others, increased likelihood of misclassifying emotional expressions as neutral. 0.75 mg/kg: increased ratings of “loneliness” | Moods: “Sociable”, “Playful”, “Loving”, “Lonely”. Friendliness: “‘friendly”, “agreeable”, “helpful”, “forgiving”, “good-natured”, “warm-hearted”, “good-tempered”, “kindly”. Facial cues: anger, fear, happiness, sadness Vocal tones: happy, sad, angry, fearful; with high/low emotional intensities |
||||||||
| Bedi 2009 [64] | Online adverts, word of mouth | Healthy, right-handed, ecstasy-experienced, mostly Caucasian | 9 | 2 female, 7 male | 24 | 0.75, 1.5 mg/kg | 3 sessions, 6/+-day intervals, doses in ascending order | Mood: 0; emotion recognition: 45 mins. | Mood, facial emotion recognition | Increased ratings of sociability, no effect on accuracy of emotion recognition | Moods: sociability and friendliness. Facial emotion recognition (happy, neutral, angry, fearful) for “pleasant”, “neutral”, “unpleasant” | ||||||||
| Study | Recruitment | Population Sampled | No. Participants | Sexes | Mean Ages (years) | Oral MDMA Dose | Dosage Timing | Time till First Test (after baseline) | Relevant Tests | Relevant Results | Relevant Parameters Tested | ||||||||
| Bershad 2017 [289] | Newspaper, bulletin board, online adverts | Healthy, ecstasy-experienced | 39 | 9 female, 30 male | 24.1 | 0.5, 1 mg/kg | Twice (same dose), 7 days apart | 60 mins. | Self-reports of moods, public-speaking task | 1 mg/kg: increased stress, tension, insecurity. 0.5, 1 mg/kg: increased perceptions of public speaking being challenging and threatening | Self-ratings of stress, tension, insecurity; stressfulness and challenge of public speaking | ||||||||
| Bershad 2016 [290] | Flyers, online adverts, word of mouth | Healthy, Caucasian, occasional MDMA users | 68 | 29 female, 39 male | 23.8 | 0.75, 1.5 mg/kg | 3 sessions, 5/+-day intervals | 30 mins. | Self-reports of mood | Increased sociability | Degrees of “lonely”, “sociable,” “loving,” “playful,” “friendly”, “confident” | ||||||||
| Bershad 2019 [291] | University campus and surrounds | Healthy, MDMA-experienced | 36 | 18 female, 18 male | 24.8 | 0.75, 1.5 mg/kg | 4 sessions, 2/+ days apart | 30 mins. | Subjective ratings of mood states and touch stimuli, psychophysiological responses, visual attention to emotional faces | Increased feelings of being “insightful”, “playful”; dose-dependently increased ratings of pleasantness of experienced affective touch; 1.5mg/kg: MDMA increased total number of times participants looked toward happy faces; increased zygomatic activity (smiling when looking at others affectively touching) |
“Sociable,” “Confident,” “Lonely,” “Playful,” “Loving,” “Friendly,” and ‘Restless”; “pleasantness” upon being physically touched or observation of physical touching between others; activity of zygomatic (smiling) and corrugator (frowning) muscles; eye movements for attention bias | ||||||||
| Cami 2000 [292] | Word of mouth | MDMA-experienced, healthy, mostly smokers | 8 | Male | 26.5 | 75, 125 mg (~ 1.1, 1.9 mg/kg) | 4 sessions, 1/+-week intervals | 1 hour | Mood states | No effect on friendliness | Self-ratings of friendliness | ||||||||
| Clark 2015 [148] | Flyers, posters, word of mouth | Mostly Caucasian | 33 | 16 female, 17 male | 24.5 | 0.75, 1.5 mg/kg | Singular | 30 mins. | Subjective drug effects | Increased prosocial feelings of “loving” and “insightful” | Self-ratings of “playful,” “loving”, “insightful” | ||||||||
| Corey 2016 [114] | Psychotherapy patients | Caucasian, PTSD patients | 20 | 18 female, 5 male | 40.5 | 125 mg (~1.9 mg/kg), 62.5 mg/kg | 1-3 times | Large dose, then smaller dose after 2 hours | Talk sessions | Increased ensuic, empathic, entactic utterances | Utterances where patients initiated empathic, entactic or ensuic topics | ||||||||
| Danforth 2018 [48] | Internet adverts, word of mouth, clinician referrals | Healthy, MDMA-naïve | 12 | 16.7% female (all MDMA), 83.3% male | 31.3 | 75, 100, 125 mg (~ 1.1, 1.5, 1.9 mg/kg) | Twice, 1-month interval | 1 day | Social anxiety | Decreased social anxiety, also at 6-month follow-up | Change from baseline, of social anxiety scores, over time | ||||||||
| de Wit 2011 [293] | Online adverts, word of mouth | MDMA-experienced polydrug users, mostly Caucasian smokers | 9 | 2 female, 7 male | 24 | 0.75, 1.5 mg/kg | Ascending order, 6 days apart | 0 | Mood states, emotion recognition | 1.5 mg/kg: increased sociability (and insignificant trend towards friendliness). No effect on emotion-recognition time or accuracy | Sociability and friendliness, emotion-recognition time and accuracy | ||||||||
| Dolder 2018 [294] | University | Mostly MDMA-naïve | 24 | 12 female, 12 male | 22.6 | 125 mg (~ 1.9 mg/kg) | 4 sessions, 7/+-day intervals | Mood 0; emotion recognition: 2.5 hours | Mood states, facial emotion recognition | Increased “happiness”, “open”, “trust”, “feeling close to others”, “I want to be with other people”, “I want to hug someone”, “well-being”, “emotional | “Happy,” “concentration,” “open,” “trust,” “feeling close to others,” “I want to be with other people,” “I want to hug someone”, | ||||||||
| Study | Recruitment | Population Sampled | No. Participants | Sexes | Mean Ages (years) | Oral MDMA Dose | Dosage Timing | Time till First Test (after baseline) | Relevant Tests | Relevant Results | Relevant Parameters Tested | ||||||||
| - | - | - | - | - | - | - | - | - | - | excitation”, “extraversion”, “introversion”, impaired recognition of fearful faces, increased misclassification of emotions as happy, increased sexual measures - “tingly all over,” “sensitive to touch,” “enthusiastic,” “warm all over,” “flushed,” “heart beats faster,” “seductive”, “enthusiastic,” “warm all over,” “passionate,” “sensual,” “pleasure,” “heart beats faster,” “happy,” “powerful,” “forget about all else”, “anticipatory” | sexual - “tempted”, “passionate”, “seductive”, “attractive”, “sensitive to touch”, “stimulated”, “excited”, “heart beats faster”, “anxious”, “displeasure”, “repulsion”, “angry”, “driven”, “urge to satisfy”, “horny”, “impatient”. Accuracy for degrees of happiness, sadness, anger, and fear facial expressions, misclassification of expressions as neutral | ||||||||
| Dumont 2009 [44] | Internet and drug-testing service adverts | Healthy, MDMA-experienced | 14 | 3 female, 12 male | 21.1 | 100 mg (~1.5 mg/kg) | 7-day interval | 0 | Subjective amicability and gregariousness | Subjective amicability were positively correlated with MDMA concentrations, but subjective gregariousness was not. (NB: Both subjective amicability and subjective gregariousness were positively correlated with oxytocin concentrations; measures had stronger correlations with oxytocin than MDMA). | Measures of antagonistic/amicable, withdrawn/gregarious | ||||||||
| Frye 2014 [145] | Flyers, online adverts | Healthy, MDMA-experienced, mostly Caucasian | 36 | 18 female, 18 male | 24.6 | 0.75, 1.5 mg/kg | 3 sessions, 96/+-hour intervals | Mood: 30 mins.; social rejection: 2.75 hours | Mood effects, reactions to social-rejection simulation, correct perceptions of social-exclusion manipulations | Increased “loving” (before and after social-rejection paradigm). Reduced decreasing effect of rejection on mood and self-esteem. Increased perceived percentage of inclusive throws under rejection condition | Self-ratings of ‘Insightful’, ‘Sociable’, ‘Confident’, ‘Lonely’, ‘Playful’, ‘Loving’, and ‘Friendly’, special emphasis on ‘Loving’. Reactions to social rejection: “I felt sad”, “I felt somewhat inadequate during the game”, “I felt like an outsider during the game”, with recollection of number of ball throws (social inclusions) received | ||||||||
| Gabay 2018 [146] | Community | MDMA-experienced | 20 | Male | 24.8 | 100 mg (~ 1.5 mg/kg) | 2 sessions, 1/+ weeks apart | 95 mins. | Preferences of resource distribution amongst others, social-reward measures | Lower probability of rejecting unfair offers in the first-person condition, but not the third-party condition. Increased average percentage offer, prosocial interaction | Preference measures: “unfair” (selfish), “fair” (equal), “hyper-fair” (altruistic) offers. Social-reward measures: admiration, negative social potency, passivity, prosocial interactions, sexual relationships, sociability. | ||||||||
| Gabay 2019 [147] | Community | MDMA-experienced | 20 | Male | 24.8 | 100 mg (~ 1.5 mg/kg) | 2 sessions, 1/+ weeks apart | 95 mins. | Mood states, Prisoner's Dilemma (cooperation vs. | Reduced accuracy in identifying fear and anger. Increased cooperation on the second run of the Prisoner's Dilemma. | Social decision-making and changes in trust (during social interaction), facial emotion recognition (happy, sad, fear, or anger - with intensities; identifying and degree of empathising with emotion) | ||||||||
| Study | Recruitment | Population Sampled | No. Participants | Sexes | Mean Ages (years) | Oral MDMA Dose | Dosage Timing | Time till First Test (after baseline) | Relevant Tests | Relevant Results | Relevant Parameters Tested | ||||||||
| - | - | - | - | - | - | - | - | - | betrayal, when forced to choose), emotion recognition, cognitive and affective empathy | Increased probability of a cooperative decision with trustworthy opponent, but not untrustworthy opponent or game server (neutral). Increased proportion of participants continuing to cooperate after first decision to compete by opponent. Maintained overall level of cooperation (declined with placebo) | |||||||||
| Harris 2002 [295] | NA | Healthy, MDMA-experienced, Caucasian | 8 | 3 female, 5 male | 24-39 | 0.5, 1.5 mg/kg | 3 sessions, 7/+days apart | 30 mins. | Mood states | 1.5 mg/kg: increased yes responses to “Have you had a greater feeling of love for others?” and “Have you liked having people around more?”, insignificant trend towards increased “friendly” self-ratings and “closeness to others” |
Degrees of Closeness to Others, Energetic, Talkative, Friendly, Confident, Insightful, Anxious. “Have you had a greater feeling of love for others?” and “Have you liked having people around more?” |
||||||||
| Holze 2020 [296] | University campus | Healthy, over-25-year-olds | 28 | 14 females, 14 males | 28 | 125 mg | Singular | 30 mins. | Mood states | Increased prosocial feelings of “talkative”, “open”, “extraversion”, and general positive feelings of “well-being”, “blissful state”, “positive mood”, “ineffability” | “Talkative”, “open”, “ego dissolution” | ||||||||
| Hysek 2012 [51] | University campus | Healthy, mostly MDMA-naïve | 48 | 24 female, 24 male | 26 | 125 mg (~ 1.9 mg/kg) | 3 sessions, 10/+ days apart | Eye-reading: 90 mins.; mood: 0 | Identifying emotions/thoughts from eye regions of others; mood states | Increased accuracy in reading positive emotions from the eye region, decreased accuracy in reading negative. No change in neutral emotions or total score. Increased “closeness,” “open,” and “talkative” mood self-reports | Total number of correct discriminations of eye-reading test; subscores computed for positive, negative, neutral emotional valences. Prosocial effects: degrees of “closeness to others,” “open,” and “talkative.” | ||||||||
| Hysek 2014 [45] | University campus | Healthy, mostly MDMA-naïve | 32 | 16 female, 16 male | 25 | 125 mg (~ 1.9 mg/kg) | 10/+ days apart | Empathy test: 3 hours | Mood states, cognitive and emotional empathy, interpersonal reactivity, social value orientation, facial affect recognition | Increased self-ratings of “happy”, “open” and “close to others”, implicit and explicit emotional empathy for positive emotions in men only, increased empathy and prosocial behaviour in men to become comparable to that in women. | Prosociality: ‘happy’, ‘open’, ‘close to others’, inferring mental states (cognitive empathy) degree to which participant felt for individual in picture (explicit emotional empathy), and degree to which participant was aroused by the scene (implicit empathy), trait empathy, social behaviour by resource allocation (degree of maximising allocation for other person) with inequality aversion and joint gain maximisation, accuracy in identifying emotions from facial expressions | ||||||||
| Study | Recruitment | Population Sampled | No. Participants | Sexes | Mean Ages (years) | Oral MDMA Dose | Dosage Timing | Time till First Test (after baseline) | Relevant Tests | Relevant Results | Relevant Parameters Tested | ||||||||
| - | - | - | - | - | - | - | - | - | - | Increased prosociality in men but not women (resource allocation). Promoted shift from joint gain maximization to inequality aversion. Impaired accuracy of emotion recognition, especially in women (insignificant in men), impaired recognition of fearful, angry, disgusted and surprised faces. Impaired recognition accuracy for fearful, angry and sad faces, only in women. Increased detection threshold for fearful faces | |||||||||
| Hysek 2012 [297] | NA | Healthy, ecstasy-naïve | 16 | 8 female, 8 male | 26.1 | 125 mg (~ 1.9 mg/kg) |
10/+ days apart | 0 | Mood states | Increased “open,” “closer to others,” and more “talkative”, “extroversion” | Degrees of “closeness to others,” “talkative,” and “open”, “extroversion” and “introversion” | ||||||||
| Kirkpatrick 2015 [46] | Newspaper, community bulletin board, online adverts | Healthy, MDMA-experienced, mostly Caucasian | 32 | 9 female, 23 male, mostly Caucasian | 24.9 | 0.5, 1 mg/kg |
3 sessions, 5/+ days apart | 60 mins. | Generosity towards stranger | 1.0 mg/kg: increased generosity toward friend, but not stranger. 0.5 mg/kg: increased generosity toward a stranger, in females | Point at which participant switches from decision to monetarily benefit selves vs. others | ||||||||
| Kirkpatrick 2014 [298] | Different institutes | Healthy, mostly MDMA-experienced | 220 | 44% female, 66% male | 25.4 | 1.5 mg/kg, 125 mg (~ 1.9 mg/kg) |
NA | NA | Mood states | Increased closeness to others | Degrees of “closeness to others” | ||||||||
| Kirkpatrick 2015 [86] | Posters, print, internet adverts, word of mouth | Healthy, MDMA-experienced | 33 | 9 female, 24 male | Early 20s | 0.5, 1 mg/kg |
3 sessions | 1.5 mins. | Mood states, social interaction, perceptions of others and selves | Increased social interaction (mostly by talking), especially 0.5 mg/kg. 1 mg/kg: rated another as more socially attractive | Degrees of prosocial effects (“confident,” “friendly,” “insightful,” “loving,” “lonely,” “playful,” “sociable”). Proportion of 1.5 min intervals interacting or talking. Level of erceived social attractiveness and physical attractiveness of another person. Level of attention, interest, understanding, and empathy of another person, participant's own level of responsiveness toward another. Perception of their own levels of social affiliation and social power or status. | ||||||||
| Kirkpatrick 2012 [299] | Word of mouth, newspaper and online adverts | Healthy, MDMA-experienced, mostly polydrug users | 11 | 2 female, 9 male | 29.3 | 100 mg (~1.5 mg/kg) | 4 sessions, 2 days' washout period | 7.5 hours | Video recordings of social interaction, with 2 films played | No effect on social interaction | Private (time spent in bathroom/bedroom) and social (time spent in the recreational area). Social time: time spent talking and time spent in silence. Total minutes spent engaging in each behavior per day. | ||||||||
| Study | Recruitment | Population Sampled | No. Participants | Sexes | Mean Ages (years) | Oral MDMA Dose | Dosage Timing | Time till First Test (after baseline) | Relevant Tests | Relevant Results | Relevant Parameters Tested | ||||||||
| Kirkpatrick 2014 [300] | Newspaper, community bulletin board, online adverts | Healthy, MDMA-experienced, Caucasian | 65 | 25 female, 40 male | 23.8 | 0.75, 1.5 mg/kg | 4 sessions, 5/+ days apart | 25 mins. | Mood states, social and emotional processing | Increased “friendly” (dose-dependently) and “lonely”. 1.5 mg/kg: reduced accuracy in identifying angry and fearful faces. Increased likelihood rating socialising as more desirable | Prosocial effects (‘Confident,' ‘Friendly,' ‘Insightful,' ‘Loving', ‘Lonely,' ‘Playful,' ‘Sociable'). Accuracy of identifying facial expressions of anger, fear, happiness, sadness. Perceived degrees of attractiveness, friendliness, and trustworthiness of facial pictures. Desire to engage in chatting with another, solving word probems or sitting quietly alone | ||||||||
| Kolbrich 2008 [301] | TV, radio, newspaper adverts, flyers, word of mouth | MDMA-experienced, mostly African-Americans | 8 | 2 females, 6 males | 21.1 | 1, 1.6 mg/kg | 3 sessions, 7 days between sessions | 0 | Mood states | Insignificantly increased feelings of closeness to others | Degrees of feelings of closeness to others | ||||||||
| Kuypers 2018 [153] | University and website adverts, word of mouth | Healthy, MDMA-experienced polydrug users | 20 | 8 female, 12 male | 21.2 | 75 mg (~1.1 mg/kg) | 7-day washouts | 90 mins. | Mood states; processing of affective sounds (recorded from strangers); approach-avoidance behaviour to social, threat and trust stimuli | Equally aroused by positive and negative sounds (controls: negative sounds produced more arousal), insignificant avoidance bias to threat faces, insignificant approach bias to trust faces; increased friendliness | Friendliness scale; sounds of happy, sad, fear, disgust, anger; joystick manipulation to indicate approach/avoidance | ||||||||
| Kuypers 2014 [154] | University and website adverts, word of mouth | Healthy, MDMA-experienced polydrug users | 20 | NA | 18-26 | 75 mg (~1.1 mg/kg) | 4 sessions, 7-day washouts | 1 hour, 25 mins. | Empathy: reading emotions from eye region, cognitive and emotional empathy, interpersonal reactivity. Social interaction: trust, social ball-tossing. Mood states | More concerned and more aroused by the emotional content of the pictures | Empathy: identifying emotions as “negative”, “positive”, “neutral”; identifying emotion, rating how concerned (explicit emotional empathy) and aroused (implicit emotional empathy) for the person; tendency to imaginatively transpose oneself into fictional social situations, tendency to spontaneously adopt the psychological viewpoint of others, feelings of warmth, compassion and concern for others, self-oriented feelings of anxiety and discomfort resulting from tense interpersonal settings. Social interaction: inferring mental state and choosing to cooperate, social reciprocation. Mood state: friendliness scale |
||||||||
| Study | Recruitment | Population Sampled | No. Participants | Sexes | Mean Ages (years) | Oral MDMA Dose | Dosage Timing | Time till First Test (after baseline) | Relevant Tests | Relevant Results | Relevant Parameters Tested | ||||||||
| Kuypers 2017 [155] | NA | Healthy | 118 | 55 female, 63 male | 21.2-25.75 | 75, 125 mg (~ 1.1, 1.9 mg/kg) | 7/+-day washouts | 120 mins. | Cognitive, implict and explicit emotional empathy; interpersonal reactivity | More concern (especially for positive stimuli) and arousal (for both positive and negative (as opposed to just positive, as in controls) stimuli) for people depicting emotions | Inferring emotional state, rating how aroused/concerned they felt for the other person; tendency to imaginatively transpose oneself into fictional social situations, tendency to spontaneously adopt the psychological viewpoint of others, feelings of warmth, compassion and concern for others, self-oriented feelings of anxiety and discomfort resulting from tense interpersonal settings. | ||||||||
| Kuypers 2008 [302] | Newspaper adverts, snowballing | Polydrug users, MDMA-experienced, healthy | 14 | 7 female, 7 male | 22.93 | 50, 75 mg (~ 0.75, 1.1 mg/kg) | 7/+-day washouts | 30 mins. | Mood states | Increased friendliness | Friendliness scale | ||||||||
| Liechti 2000 [303] | University hospital, medical school | Healthy, mostly unversity students and physicians | 16 | 4 female, 12 male | 27.4 | 1.5 mg/kg | 4 sessions, 14/+-day intervals | 120 mins. | Mood states | Increased “self-confidence”, “extroversion”, “introversion” | Scales of “extroversion”, “introversion”, “aggression-anger”, “self-confidence” | ||||||||
| Liechti 2001 [115] | University hospital staff, medical school | Healthy, mostly unversity students and physicians, mostly MDMA-naive | 74 | 20 female, 54 male | 27 | 10, 50 mg (~0.2, 0.75 mg/kg) | 2 sessions, 2/+-week interval | 30 mins. | Mood and consciousness states | Increased “comprehensive love”, self-confidence, extroversion, openness, sociability, talkativeness. Increased thoughtfulness and sensitivity in women | Self-confidence, extroversion, introversion, aggression-anger, thoughtfulness, sensitivity. Changes in mood, perception, experience of the self and of the environment | ||||||||
| Liechti 2000 [149] | University hospital, medical school | Healthy, mostly MDMA-naïve | 14 | 1 female, 13 male | 26 | 1.5 mg/kg | 4 sessions, 10/+-day intervals | 75 mins. | Mood states | Decreased vigilance; increased self-confidence, sensitivity and extroversion | Self-confidence, extroversion, introversion, aggression-anger, vigilance | ||||||||
| Liechti 2000 [304] | University hospital, medical school | Healthy, mostly unversity students and physicians, mostly MDMA-naïve, mostly non-smokers | 14 | 5 female, 9 male | 26 | 1.5 mg/kg | 4 sessions, 10/+-day intervals | 75 mins. | Mood states | Increased extroversion and sociability | Introversion, extroversion, sociability | ||||||||
| Liechti 2001 [55] | University hospital, medical school | Mostly university students or physicians, healthy, mostly MDMA-naïve | 44 | 10 female, 34 male | 26, 27 | 1.5 mg/kg | 4 sessions | 120 mins. | Mood states | Increased self-confidence, extraversion | Self-confidence, extroversion, introversion, sensitivity, aggression/anger | ||||||||
| Study | Recruitment | Population Sampled | No. Participants | Sexes | Mean Ages (years) | Oral MDMA Dose | Dosage Timing | Time till First Test (after baseline) | Relevant Tests | Relevant Results | Relevant Parameters Tested | ||||||||
| Schmid 2014 [150] | University | Mostly MDMA-naïve, healthy | 30 | 15 female, 15 male | 24 | 75 mg (~ 1.1 mg/kg) | 7/+-day intervals | Emotion recognition: 75 mins.; mood: 90 mins.; moral judgement: 2 hours; cognitive and emotional empathy: 3 hours; prosociality: 4 hours; mood: 0, 1.25, 5 hours | Facial emotion recognition; cognitive (also in social scenarios) and emotional empathy; prosociality; social decision-making (moral judgement); mood states | Impaired identification of sad, angry and fearful faces; increased misclassification of emotions as neutral; increased both explicit and implicit emotional empathy scores for positive emotional stimuli; increased openness, trust and closeness |
Identifying and misclassifying happiness, sadness, anger and fear; inferring mental state of another, how concerned (explicit emotional empathy) they were for them, how aroused (implicit emotional empathy) they were by the scene; identifying false belief, persuasion, faux pas, metaphor and sarcasm in social contexts; maximising resources for self and others, minimising difference between the two; judging between utilitarian outcomes involving aversive effects to others | ||||||||
| Schmid 2018 [305] | Adverts, word of mouth | Healthy | 24 | 12 female, 12 male | 22.6 | 125 mg (~ 1.9 mg/kg) | 4 sessions, 7/+-day intervals | Mood: 75 mins.; emotion recognition: 150 mins. | Negative emotional states, facial emotion recognition, fearful-face processing | Trend for impaired facial emotion recognition | Emotion recognition: happiness, sadness, anger, fear | ||||||||
| Tancer 2007 [306] | NA | MDMA-experienced, healthy, Caucasian, polydrug users | 8 | 2 female, 6 male | 23.9 | 1.5 mg/kg | 6 sessions, 2/+-day intervals | 60 mins. | Mood states | Increased friendly, talkative scores | Friendliness scale; friendly, self-conscious, social, talkative scales | ||||||||
| Tancer 2003 [113] | NA | MDMA-experienced, mostly smokers, mostly marijuana users | 12 | 6 female, 6 male | 22.3 | 1, 2 mg/kg | 7/+-day intervals | 60 mins. | Mood states | 2 mg/kg: increased friendly, social, talkative scores | Friendliness scale; friendly, self-conscious, social, talkative scales | ||||||||
| Tancer 2001 [307] | NA | MDMA-experienced, mostly Caucasian | 22 | 14 female, 8 male | 23.6 | 1.1-2.1 mg/kg | 2 sessions, 7/+ days apart | 60 mins. | Mood states | Increased friendly score | Friendly scale | ||||||||
| van Wel 2012 [127] | Newpaper adverts, word of mouth | Healthy, MDMA-experienced | 17 | 8 female, 9 male | 22.76 | 75 mg (~ 1.1 mg/kg) | 7/+ days apart | 1.5 hours | Mood states | Increased friendliness | Friendliness score | ||||||||
| Vizeli 2018 [156] | University campus | Healthy, Caucasian, mostly non-drug users | 124 | 64 female, 60 male | 24.8 | 125 mg (~ 1.9 mg/kg) | 7/+ days apart | Mood: 0; emotion recognition, empathy: 90 mins. | Mood states, facial emotion recognition, cognitive and emotional empathy | Increased closeness to others, talkativeness, trust, wanting to be hugged and to hug; impaired recognition of fearful, sad, angry faces; decreased cognitive empathy for all emotions, increased explicit emotional empathy for positive emotions | Mood scales: “closeness to others,” trust,” “want to be hugged,” “want to hug,”“want to be alone,” “want to be with others”, “talkative”. Facial expressions: happiness, sadness, anger, and fear. Empathy: cognitive (inferring mental state of another), implicit emotional (arousal by another's emotional state), explicit emotional (concern for another) | ||||||||
| Study | Recruitment | Population Sampled | No. Participants | Sexes | Mean Ages (years) | Oral MDMA Dose | Dosage Timing | Time till First Test (after baseline) | Relevant Tests | Relevant Results | Relevant Parameters Tested | ||||||||
| Vollenweider 2005 [123] | University hospital staff, medical school | Healthy, mostly MDMA-naïve | 42 | 10 female, 32 male | 25.4-27 | 1.5 mg/kg | NA | 45 mins. | Mood states | Increased self-confidence and extroversion | Self-confidence, extroversion, introversion | ||||||||
| Wardle 2014 [116] | Flyers, online adverts | Healthy, MDMA-experienced, mostly Caucasian | 36 | 18 female, 18 male | 24.6 | 0.75, 1.5 mg/kg | 3 sessions, 7/+ days apart | Mood: 30 mins. Emotion recognition: 70 mins. Conversation: 2 hrs., 20 mins. | Mood states, facial emotion recognition; interpersonal perception | Mood: increased loving, insignificantly increased playful. Emotion recognition: 1.5 mg/kg: impaired recognition of angry expression; decreased frown response to happy expressions, only in females; increased smile response to happy expressions. Increased positive emotion words (both doses). 1.5 mg/kg: insignificantly increased perceived regard, increased empathy, insignificantly increased percepton of empathy from others | Mood: playful, loving, lonely. Emotion recognition: angry, fearful, sad, happy faces. Interpersonal perception: picked 3 personally important people; percentage of positive and negative emotion words to describe each person; participant's perceptions of investigator and scales of regard (“S/he was truly interested in me”), empathy (e.g., “S/he understood me”), and congruence (“I felt that s/he was real and genuine with me”) | ||||||||
| Wardle 2014 [308] | Flyers, online adverts | MDMA-experienced, healthy, mostly Caucasian | 101 | 43 female, 58 male | 24.1 | 0.75, 1.5 mg/kg | 3 or 4, separated by 5/+ days | Mood: 30 mins. Picture ratings: 1 hr., 10 mins. | Mood states, responses to emotional pictures, identifying emotional facial expressions | Dose-dependently increased playfulness, lovingness. 1.5 mg/kg: increased positivity of positive social pictures; 0.75 mg/kg: decreased the positivity of positive non-social pictures (1.5 mg/kg insignificantly had this effect) | Mood: playful’ and ‘loving’. Pictures: social vs. non-social, yielding participant-rated scores on scales of positive/negative and arousal | ||||||||
Between 0.2 and 0.75 mg/kg, recognition of emotions from others’ facial expressions is unaffected. A dose of 1.5 mg/kg and above shows impaired recognition of negative emotions and a tendency to misclassify emotional expressions as neutral or happy (although the studies using 2 and 2.1 mg/kg did not test for facial emotion recognition). 1.9 mg/kg shows increased accuracy in identifying positive emotions (chronic or singular dosing). Likewise, relevant tests show social anxiety increased at 0.5-1 mg/kg, but decreased at 1.1-1.9 mg/kg (including higher self-confidence) (chronic or singular dosing). At a chronic 2 mg/kg, however, one study showed self-consciousness not being affected [113], but this was a self-rating component as part of a wider questionnaire as opposed to a behavioural activity testing for social anxiety. This suggests that perhaps 0.75-1.5 mg/kg MDMA taken orally and chronically may be best to alleviate social impairments in humans, without impairing emotion recognition in others.
There was insufficient variation in age between the studies, to assess age effects. All studies tested young adults in their 20s-30s, with the exception of Corey et al. (2016) whose mean participant age was 40.5 years old. Here, they also chronically used the unusual dose of 62.5 mg/kg [114]. Therefore, the effects of MDMA found in these participants are difficult to attribute a certain cause to, although the common theme of increased empathic utterances continues at this dose.
Sex effects were apparent in some studies. In Liechti et al. (2001), chronic 0.2 and 0.75 mg/kg only increased thoughtfulness and sensitivity in women, but not men [115]. At a chronic 0.5 mg/kg, Kirkpatrick et al. (2015) found that only in females was generosity towards stranger persons increased [46]. At a chronic 1.5 mg/kg, Wardle et al. (2014) found decreased frowns in response to others’ happy faces only in women [116]. At a chronic 1.9 mg/kg, Hysek et al. (2014) found that openness, closeness to others and empathy were increased for positive emotions in men specifically, but only to levels comparable to those of placebo women [45]. Also at this dose, the resource-allocation task showed increased prosocial behaviour only in men. Impaired accuracy of facial emotion recognition occurred more in women than men, with impairments specific to negative expressions only seen in women [45]. Hence, lower MDMA doses seemed to have prosocial effects in females exclusively, whereas higher doses were required for men to attain the same empathy levels as women. Interestingly, however, the higher chronic dose of 1.9 mg/kg impaired recognition of facial expressions more in women than in men [45]. Altogether, these results may indicate a lower tolerance for, or a higher sensitivity to, MDMA in women than in men. This suggests that men may require a higher clinical dose, such as chronic oral 1.9 mg/kg MDMA, to improve social impairments.
3.2 Core ASD Behaviour 2: Repetitive Behaviours
3.2.1 Rodents
3.2.1.1 Open-field Tests
The open-field test places a rodent in the centre of an open apparatus and observes its solitary behaviours. With respect to ASD-specific behaviours, investigators are able to observe motor stereotypies (repetitive purposeless movements), including compulsive self-grooming or repetitive routes travelled. A hole-board can be included to provide holes into which the rodent is allowed to poke its nose or dip its head, as an exploratory behaviour. Whether it tends to poke in the same or different holes, can be an indicator of repetitive stereotypy.
We found 72 studies giving MDMA singularly or chronically postnatally to rodents, and observing them for solitary behaviours. The studies show that stereotypies (such as self-grooming, head dipping or hole-pokings, headshakes, digging and repetitive locomotive patterns) are actually increased at 5-20 mg/kg, and there were fewer varied holepokes at chronic 10 mg/kg (Table 6). At a singular 0.63 and 0.94 mg/kg, there were perseverative locomotor patterns (Table 6). However, stereotypies are decreased at 1-20 mg/kg in other studies (singular or chronic dosing) (Table 6). At 1.25-10 mg/kg, there were dose-dependently decreased repeated, as opposed to varied, holepokes (Table 6).
Table 6.
Solitary behaviours in postnatally MDMA-treated rodents, compared with control rodents.
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size | Singular/Chronic Dosing | Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Males | Females | Both | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Abad 2014 [191] | Rat | Males | NA | 16-24 | Chronic | Twice daily for 4 days | 20 | s.c. | 35 | 46 | 26 | More time spent in centre. | NA | NA | |||||||||||
| Ball 2011 [238] | Rat | Males | NA | 64 | Chronic | 2 daily for 5 consecutive days (6 hours apart) | 5 | i.p. | “Adult” (300-400g) | Same days | NA | Paired MDMA: increased locomotor activity (15 days post-injection). 100 days post-injection: only homecage-treated had hyperlocomotion. | NA | NA | |||||||||||
| Ball 2011 [238] | Rat | Males | NA | 63 | Singular | 15 or 100 days later | 2.5 | i.p. | “Adult” (300-400g) | Same day | NA | Hyperlocomotion in unpaired, 100 days post-injection = hyperlocomotion in paired, 15 days post-injection. 100 days post-injection: paired had higher stereotypy (at 15 days post-injection, higher than MDMA-treated unpaired). | NA | NA | |||||||||||
| Biezonski 2009 [239] | Rat | Males | NA | 16 | Chronic | Twice daily, 4 hours apart, every 5th day | 10 | s.c. | 35-60 | 67 | 23-24 | No effect. | NA | NA | |||||||||||
| Braida 2002 [240] | Rat | Males | NA | 40 | Chronic | Daily | 1, 2, 3, 4 | i.p. | 90 | 104 | 22 | 4 mg/kg: increased line crossings and stereotypy | NA | NA | |||||||||||
| Bull 2003 [133] | Rat | Males | NA | 24 | Chronic | Twice daily | 15 | i.p. | 28-30 | 28 | 21 | 15 mg/kg: higher sustained locomotion (controls: low decreasing locomotion), over 30 mins. Fewer rears, decreased to low plateau (controls: rearing progressively declines). Serotonin syndrome: reciprocal forepaw treading, lateral head weaving, flat body posture. | NA | NA | |||||||||||
| Bull 2004 [132] | Rat | Males | NA | 32 | Chronic | Each of 4 hours | 5 | i.p. | 28 | 84 | 21 | Sustained hyperlocomotion, fewer rearings. | NA | NA | |||||||||||
| Callaway 1990 [241] | Rat | Males | NA | 55 | Singular | NA | 0.3, 1, 3, 10 | s.c. | “Adult” (300-400g) | Same day | NA | 1, 3, 10 mg/kg: decreased exploration (holepokes, rearings). 10 mg/kg: increase of routes taken repetitively. 3, 10 mg/kg: increased sustained hyperlocomotion. Centre avoidance, direction alternation. | NA | NA | |||||||||||
| Callaway 1992 [242] | Rat | Males | NA | 32 | Chronic | 1-week intervals | 10, 30 microg. | i.c. | 300-400g | Same day | NA | Hyperactivity (30 microg. slightly later than 10 microg.). 30 microg.: hypolocomotion in 1st 10 mins., increased rearings. 10 microg.: decreased holepokes. Results dependent on cerebral area injected. | NA | NA | |||||||||||
| Clemens 2004 [217] | Rat | Males | NA | 63 | Chronic | 4 injections in 1 day, one every 2 hours | 2.5 | i.p. | 376g | Same time | 28 | Higher total locomotion. | NA | NA | |||||||||||
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size | Singular/Chronic Dosing | Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Males | Females | Both | |||||||||||
| Clemens 2005 [218] | Rat | Females | NA | 56 | Chronic | Every 2 hours in one day (4 times) | 4 | i.p. | 281g | Same day | 28 | NA | Hyperactivity. | NA | |||||||||||
| Clemens 2007 [136] | Rat | Females | NA | 16 | Chronic | 1 injection per week for 16 weeks | 8 | i.p. | 238g | 7, 56, 112 days later | 28 | NA | Constant hyperactivity between weeks. | NA | |||||||||||
| Cohen 2005 [196] | Rat | Males | NA | 30 | Chronic | Twice a day (8 hours apart) | 20 | s.c. | 11-20 | 5 weeks, 1 day later | 21 | Neonatal: less total and central distance travelled, increased thigmotaxis. | NA | NA | |||||||||||
| Cohen 2005 [196] | Rat | Males | NA | 30 | Chronic | 4 times, at 2 intervals | 15 | s.c. | 82-100 | 5 weeks, 1 day later | 21 | Neonatal and adult: increased thigmotaxis. | NA | NA | |||||||||||
| Colussi-Mas 2008 [243] | Rat | Males | NA | 21 | Chronic | Once daily for 5 consecutive days | 10 | i.p. | “Adult” (250–300g) | Same ages | 21 | No effect. | NA | NA | |||||||||||
| Colussi-Mas 2008 [243] | Rat | Males | NA | 21 | Singular | NA | 5 | i.p. | “Adult” (250–300g) | Same ages | 21 | 5, 10 mg/kg: increased locomotion graph. 5 mg/kg: increased central time. | NA | NA | |||||||||||
| Colussi-Mas 2008 [243] | Rat | Males | NA | 21 | Chronic | Once daily for 5 consecutive days + this one after 2 days | 5 | i.p. | “Adult” (250–300g) | Same ages | 21 | Hyperlocomotion in, and time spent in, central zone | NA | NA | |||||||||||
| Cox 2014 [244] | Rat | Males | NA | 26 | Chronic | Daily for 4 days | 5, 10 | i.p. | 38-41 | 46 | NA | 10 mg/kg: decreased centre time. | NA | NA | |||||||||||
| Daza-Losada 2009 [221] | Mouse | Males | NA | 315 | Singular | NA | 5, 10, 20 | i.p. | 28 | 28 | 21 | 10, 20 mg/kg: increased hyperlocomotion. | NA | NA | |||||||||||
| Ebrahimian 2017 [245] | Mouse | Males | NA | 40 | Both | Single dose, twice | 20, 60 | p.o. | NA | 0 and 9 days later | 22-24 | 20 and 60 mg/kg: total distance travelled dose-dependently increased, rearing decreased. | NA | NA | |||||||||||
| Fantegrossi 2004 [246] | Mouse | Males | NA | 12 | Singular | NA | 32 | i.p. | 20-30g | Same age | 22 | Long-lasting hyperlocomotion (3 hours post-injection). | NA | NA | |||||||||||
| Ferraz-de-Paula 2011 [247] | Mouse | Males | NA | 14-16 | Singular | NA | 0.2-20 | i.p. | 60 | 67 | 22-26 | 5, 8, 10, 20 mg/kg: increased total distance travelled, peripheral time, locomotor activity; decreased frequency of rearing and grooming. 8, 10, 20 mg/kg: increased peripheral distance travelled. 1, 5, 20 mg/kg: increased central distance travelled. 1, 5, 10, 20 mg/kg: increased central time. 5, 10 mg/kg: increased distance travelled, mean locomotor activity; decreased number and time of head-dipping (hole-board). |
NA | NA | |||||||||||
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size | Singular/Chronic Dosing | Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Males | Females | Both | |||||||||||
| Fone 2002 [131] | Rat | Males | NA | 36 | Chronic | Twice daily | 7.5 | i.p. | 39-41 | 39 | 21 | 7.5 mg/kg: sustained hyperlocomotion. Serotonin syndrome: extensive reciprocal forepaw treading, lateral head weaving, flat body posture. | NA | NA | |||||||||||
| García-Pardo 2017 [94] | Mouse | Males | NA | 14-16 | Singular | NA | 5, 10 | i.p. | 42 | 70, 71 | 35-37 | More centre entries. | NA | NA | |||||||||||
| Gold 1988 [248] | Rat | Males | NA | 60 | Singular | NA | 1.25, 2.5, 5, 10 | s.c. | “Adult” (250-400g) | Same day | NA | Hyperlocomotion in first 10 mins. 2.5-10 mg/kg: hypolocomotion for rest of 2 hours. Initially decreased, then increased, holepokes. Dose-dependent sustained decreased repeated:varied holepokes. Decreased, then increased, rearings. Dose-dependent centre avoidance, hyperlocomotion. 10 mg/kg: increased rearings in 2nd hour. 10 mg/kg: hyperlocomotion. | NA | NA | |||||||||||
| Gold 1988 [249] | Rat | Males | NA | 60 | Singular | NA | 0.63, 0.94 | s.c. | “Adult” (250-400g) | Same day | NA | More perseverative locomotion patterns. 0.63 mg/kg: decreased repeated:varied nosepokes. 0.94 mg/kg: decreased rearing. | NA | NA | |||||||||||
| Gold 1988 [249] | Rat | Males | NA | 60 | Singular | NA | 10 | s.c. | “Adult” (250-400g) | Same day | NA | Hyperlocomotion. | NA | NA | |||||||||||
| Gurtman 2002 [134] | Rat | Males | NA | 26 | Chronic | Each of 4 hours | 5 | i.p. | 90 | 118 | 22 | Hyperlocomotion. | NA | NA | |||||||||||
| Hegadoren 1995 [250] | Rat | Males | NA | 24 | Singular | NA | NA | i.p. | 250-400g | Same day | NA | 1st 30 mins.: rapid locomotor increase. Decreased yawning, mouth movements, lying down, stretching. Increased body adjustments, clockwise rotations. Complete insomnia. | NA | NA | |||||||||||
| Hiramatsu 1989 [251] | Rat | Males | NA | 21-27 | Singular | NA | 5, 10, 20 | s.c. | 200-300g | Same ages | 23 | Stereotyped sniffing, head-weaving, backpedalling, turning. NB: S(+) MDMA: more head-weaving, backpedalling, turning than R(-). | NA | NA | |||||||||||
| Ho 2004 [252] | Rat | Males | NA | 65 | Singular | NA | 7.5, 15 | i.p. | 271g | 9-10 days later | 22-24 | No effect. | NA | NA | |||||||||||
| Kalivas 1998 [253] | Rat | Males | NA | 24 | Chronic | 5 days: 5 mg/kg once daily, or 20 mg/kg twice daily | 5, 20 | s.c. | NA | NA | NA | Acute, 5 mg/kg, Day 2: increased horizontal activity, distance travelled, stereotypy. Chronic 5, 20 mg/kg for 4 days: increased activity. Day 19: more increase in horizontal activity, distance travelled, response greater. 2 days washout, daily 5 mg/kg: more horizontal activity, distance travelled. More activity at 30-90 mins. post-injection. | NA | NA | |||||||||||
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size |
Singular/Chronic
Dosing |
Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Males | Females | Both | |||||||||||
| Kindlundh-Högberg 2009 [254] | Mouse | Males | NA | 12 | Chronic | 3 injections every 7th day for 4 days | 5 | i.p. | 120-180 | Same ages | 20 | Increased horizontal activity. 1st week: increased locomotor:horizontal ratio. | NA | NA | |||||||||||
| Kurling 2008 [225] | Rat | Males | NA | 12 | Singular | NA | 5 | i.p. | “Adult” (300-380g) | Same day | 21 | Increased locomotion, burst of rearings, slight agitation, stereotypies (intensive sniffing, head/body weaving, head-bobbing). |
NA | NA | |||||||||||
| Lebsanft 2005 [255] | Rat | Males | NA | 20 | Singular | NA | 5 | s.c. | 47-56 | Same day | 22.5 | No effect. | NA | NA | |||||||||||
| Llorente-Berzal 2013 [199] | Rat | Both | Separated | 60 | Chronic | Every 5 days, twice daily (4 hours apart) | 10 | s.c. | 30-45 | 71 | 22 | Increased thigmotaxis. | Increased thigmotaxis. | NA | |||||||||||
| Lorens 1990 [256] | Rat | Males | NA | 36-48 | Chronic | Every 12 hours for 4 consecutive days | 10, 20, 40 | s.c. | 225-250g | 2-3 weeks later | NA | No effect. | NA | NA | |||||||||||
| Ludwig 2008 [200] | Rat | Males | NA | 39+ | Singular | Single injection | 5 | s.c. | “Adult” (242-275g) | “Adult” (242-275g) | 26 | 5-10 mins.: increased total locomotion. 10-20 mins.: decreased rearing | NA | NA | |||||||||||
| Ludwig 2008 [200] | Rat | Males | NA | 39+ | Chronic | 5 daily injections | 5 | s.c. | “Adult” (242-275g) | “Adult” (242-275g) | 26 | After 10 mins.: increased locomotion. Multiple MDMA injections: increased central and total locomotion, decreased peripheral locomotion. | NA | NA | |||||||||||
| Ludwig 2008 [200] | Rat | Males | NA | 39+ | Both | Chronic, then single | 5 | s.c. | “Adult” (242-275g) | “Adult” (242-275g) | 26 | 1st 10 mins., multiple MDMA injections: decreased rearing. Multiple MDMA injections: increased central and total locomotion, decreased peripheral locomotion. Multiple doses: increased rearing. Check - Low doses had higher locomotor activity than high doses. - check. | NA | NA | |||||||||||
| McNamara 1995 [257] | Rat | Males | NA | 24 | Chronic | Twice daily for 4 days | 5, 10, 20 | i.p. | 200-250g | 7 days later | 22-24 | 10 and 20 mg/kg increased locomotor activity (experimental day 1, 2, 4). 20 mg/kg: increased locomotion (experimental day 3). | NA | NA | |||||||||||
| Mechan 2002 [258] | Rat | Males | NA | 16 | Singular | NA | 12.5 | i.p. | “Adult” (160-200g) | 9–11, 30–32, 71–73 days post-injection | 21 | White light: no effect. Red light: hyperlocomotion till significant on PD 73. | NA | NA | |||||||||||
| Miczek 1994 [227] | Mouse | Males | NA | 29 | Singular | NA | 0.3-10 | NA | “Adult” | Same day | 21 | 10 mg/kg: decreased rearing. | NA | NA | |||||||||||
| Morley 2000 [228] | Rat | Males | NA | 48 | Singular | NA | 1.25, 2.5, 5 | i.p. | 85-95 | 99-109 | 22 | Decreased centre time, increased defaecation. 5 mg/kg: hyperlocomotion. | NA | NA | |||||||||||
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size | Singular/Chronic Dosing | Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Males | Females | Both | |||||||||||
| Morley 2001 [91] | Rat | Males | NA | 48 | Chronic | Each of 4 hours over 2 days | 5 | i.p. | 75-95 | 160-180 | 28 | Hyperlocomotion. | NA | NA | |||||||||||
| O'Loinsigh 2001 [259] | Rat | Males | NA | 12-14 | Chronic | Twice daily for 4 consecutive days | 20 | i.p. | 200-300g | 200-300g | 20-22 | Increased locomotion and stereotypy, decreased rearing | NA | NA | |||||||||||
| Olsen 2016 [260] | Rat | Males | NA | NA | Chronic | Twice | 0.75, 1.5, 3 | i.p. | 105 | 105 | NA | 1.5, 3 mg/kg: dose-dependently decreased response rate for repeated, as opposed to varied, tasks | NA | NA | |||||||||||
| Palenicek 2005 [261] | Rat | Both | Separated | 79 | Singular | NA | 2.5, 5, 10 | s.c. | 50 | 50 | 22-24 | Dose-dependently increased locomotion | Dose-dependently increased locomotion. 2.5 and 5 mg/kg: longer trajectory than males, increased thigmotaxis. | NA | |||||||||||
| Paleníček 2007 [262] | Rat | Both | NA | 88 | Singular | NA | 2.5, 5, 10 | s.c. | 150-220g | 150-220g | 22-24 | Females, 5 mg/kg: sniffing stereotypy. 10 mg/kg: sniffing stereotypy (only males in 2nd session). 2.5, 10 mg/kg in 1st session; 10 mg/kg in 2nd session: males > females. Females, 10 mg/kg: decreased stereotypy | |||||||||||||
| Paulus 1992 [263] | Rat | Males | NA | 47 | Singular | NA | MDMA: 1.25, 2.5, 5, 10 | s.c. | 250-300g | 250-300g | NA | Increased ratio of short:long microevent durations. 5, 10 mg/kg: straighter paths travelled | NA | NA | |||||||||||
| Paulus 1992 [263] | Rat | Males | NA | 61 | Singular | NA | (+)MDMA: 0.3, 1, 3, 10 | s.c. | 250-300g | 250-300g | NA | 3, 10 mg/kg: increased thigmotaxis. 10 mg/kg: straighter paths travelled. Hyperlocomotion | NA | NA | |||||||||||
| Paulus 1992 [263] | Rat | Males | NA | 62 | Singular | NA | (-)MDMA: 1, 3, 10, 30 | s.c. | 250-300g | 250-300g | NA | Long straight paths with thigmotaxis. 10, 30 mg/kg: straighter paths. 30 mg/kg: hyperactivity | NA | NA | |||||||||||
| Piper 2004 [203] | Rat | Males | NA | 16 | Chronic | On every 5th day on PD 35-60, twice daily, 4 hours apart each | 10 | s.c. | 35-60 | 65 | 22 | No effect. | NA | NA | |||||||||||
| Piper 2005 [204] | Rat | Males | NA | 20 | Chronic | Hourly intervals over 4 hours, once every 5 days | 5 | s.c. | 35-70 | 35, 45, 60, 65 | 22 | PD 35: increased headweaving. Hyperlocomotion at 2-4 mins., hypolocomotion at 7-10 mins. |
NA | NA | |||||||||||
| Piper 2008 [205] | Rat | Males | NA | 20-24 | Chronic | 4 doses, 1 each hour | 10 | s.c. | “Young adult” (307.7g) | Young adult | 23 | Hole-board: fewer hole entries after MDMA on 2nd injection. |
NA | NA | |||||||||||
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size | Singular/Chronic Dosing | Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Males | Females | Both | |||||||||||
| Powell 2004 [264] | Mouse | Both | Combined | 15-33 | Singular | NA | 10, 20, 30 | i.p. | 150 | 150 | NA | NA | NA | Hyperlocomotion, straight perseverative movements | |||||||||||
| Procopio-Souza 2011 [85] | Mouse | Males | NA | 19 | Singular | NA | 10 | i.p. | 90 | Same day | 22-23 | Increased bouts of locomotion. | NA | NA | |||||||||||
| Procopio-Souza 2011 [85] | Mouse | Males | NA | 19 | Singular | NA | +10 | i.p. | 97 | Same day | 22-23 | More bouts of hyperlocomotion with MDMA pretreatment. | NA | NA | |||||||||||
| Quinteros-Munoz 2010 [265] | Rat | Males | NA | 56-77 | Singular | NA | 0.25, 0.5, 1, 3, 5, 10 | i.p. | 200-230g | 200-230g | NA | 1 mg/kg: increased grooming; declined after higher doses. 10 mg/kg: decreased head shakes | NA | NA | |||||||||||
| Shen 2011 [233] | Rat | Males | NA | 60 | Chronic | 2, every 5th day | 10 | s.c. | 50-60 | 70 | 26 | No effect. | NA | NA | |||||||||||
| Shen 2011 [233] | Rat | Males | NA | 19 | Chronic | 2, every 5th day | 10 | s.c. | 50-60 | 74 | 26 | Holeboard: decreased varied entries. | NA | NA | |||||||||||
| Shen 2011 [233] | Rat | Males | NA | 19 | Chronic | 2, every 5th day | 10 | s.c. | 48-58 | 68 | 26 | No effect. | NA | NA | |||||||||||
| Slamberova 2018 [266] | Mouse | Males | NA | 16 | Singular | NA | 5 | s.c. | 77-97 | 77-97 | 22-24 | Increased locomotion, distance travelled, average speed. No effect on grooming duration. | NA | NA | |||||||||||
| Stanley 2007 [267] | Rat | Males | NA | 8 | Singular | NA | 10 | i.p. | 250-300g | 250-300g | 22 | Stereotypic-included behaviour increased. | NA | NA | |||||||||||
| Thompson 2009 [82] | Rat | Males | NA | 36 | Singular | NA | 5 | i.p. | “Adult” (345g) | Same day | 28 | Hyperlocomotion, more central time. | NA | NA | |||||||||||
| Walker 2010 [268] | Rat | Males | NA | 30-34 | Singular | NA | 5 | i.p. | 28, 42, 65 | 28, 42, 65 | NA | Early hyperlocomotion (PD 28 < 42, 65). No stereotypy effects. | NA | NA | |||||||||||
| Yamamotová 2012 [269] | Rat | Males | NA | 20 | Singular | NA | 5 | s.c. | 85-90 | 85-90 | NA | Increased stereotypy. | NA | NA | |||||||||||
| Yang 2011 [270] | Rat | Females | NA | 26 | Singular | NA | 5 | i.p. | “Adult” (180-190g) | “Adult” (180-190g) | 21 | NA | Increased horizontal activity and total distance travelled (experimental day 2 > day 1). |
NA | |||||||||||
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size | Singular/Chronic Dosing | Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Males | Females | Both | |||||||||||
| - | - | - | - | - | - | - | - | - | - | - | - | - | Decreased vertical activity (experimental day 2 > day 1). 5 mg/kg: increased total distance travelled, horizontal activity, stereotypies; decreased vertical acitivity (10-110 mins. post injection). | ||||||||||||
| Yang 2011 [270] | Rat | Females | NA | 26 | Singular | 6 consecutive days | 5 | i.p. | 85-87 | 85-87 | 21 | NA | Increased stereotypies, decreased vertical activity (0-20 mins. post-injection). Increased horizontal activity (30 mins.-2 hours post-injection). 5 mg/kg: increased total distance travelled, horizontal activity, stereotypies; decreased vertical acitivity (10-110 mins. post injection). Chronic dosing had no effect. 5 mg/kg: increased/decreased locomotion (experimental day 11 > day 2). | NA | |||||||||||
| Study | Species | Sexes |
Sexes
Combined or Separated |
Sample Size |
Singular/
Chronic Dosing |
Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Males | Females | Both | |||||||||||
| Young 2008 [271] | Mouse | Males | NA | 48 | Singular | NA | (±)-MDMA: 0.3, 1, 3, 10, 30 | i.p. | 27-34g | 27-34g | 22 | Hyperlocomotion. 30 mg/kg: decreased thiogmotaxis. | NA | NA | |||||||||||
| Young 2008 [271] | Mouse | Males | NA | 48 | Singular | NA | S(+)-MDMA: 0.3, 1, 3, 10, 30 | 27-34g | 27-34g | 22 | Hyperlocomotion. | NA | NA | ||||||||||||
| Young 2008 [271] | Mouse | Males | NA | 64 | Singular | NA | R(-)-MDMA: 0.3, 1, 3, 10, 17, 30, 50 | 27-34g | 27-34g | 22 | Hyperlocomotion. | NA | NA | ||||||||||||
Each study is shown alongside the species, sex and sample size of the rodents tested (control and relevant treatment groups), whether the study used singular or chronic (multiple) dosing, and if they used chronic dosing, the timing of the doses; the dose and route (mode of injection) used, the ages the rodents were treated and tested at, the temperature of the testing environment, and the results obtained from the experiment in the treated rodents (where it is different from the results in the corresponding control rodents). i.p. = intraperitoneal mode of injection; s.c. = subcutaneous mode of injection; p.o. = oral route of ingestion; i.c. = intracerebral mode of injection.
Therefore, singular or chronic 1-5 mg/kg MDMA may be able to reduce stereotypies in these rodents.
Thigmotaxis (the anxious tendency to remain close to the walls) decreased when singularly given 1-20 mg/kg, and even chronically 5-10 mg/kg. When rodents were chronically given 10 mg/kg, however, it gave mixed results. When the chronic dose increased even more to 15-20 mg/kg, it increased thigmotaxis with higher sustained hyperlocomotion (possibly indicating anxiety). It also increased at 1.25-5 mg/kg, with accompanying increased defaecation (also taken as an indicator of anxiety), further lending support to the increased-anxiety theory. Interestingly, there seemed to be a sex effect in one study, where females showed a greater thigmotaxic increase than males, at 2.5 and 5 mg/kg MDMA [117]. Hyper-locomotion tended to occur at 5 and 10 mg/kg, as well as decreased exploration (holepokes and rearings). This hyper-locomotion was increased at 3-20 mg/kg, and also when chronically given. At lower doses, hyper-locomotion appeared early on (1.25-10 mg/kg). At 2.5-10 mg/kg, hypo-locomotion followed. Holepokes decreased first, then increased. At 10 mg/kg, rearings also increased, later. There were fewer rearings, but dose-dependently increased locomotion at 20 and 60 mg/kg. When 5 or 20 mg/kg is chronically given, there is also hyper-locomotion. Interestingly, under red light (which triggers less anxiety in rodents), there was increasing hyper-locomotion at 12.5 mg/kg, whereas the same dose under white light had no effect. When chronically given 10 or 15 mg/kg, there was less exploration (rears and holepokes). When 7.5-30 mg/kg was chronically given, symptoms of serotonin syndrome also appeared in the rodents, namely, reciprocal forepaw treading, lateral head weaving, flat body posture.
3.2.1.2. Marble-burying Tests
Testing for repetitive/compulsive behaviours can be undertaken via the marble-burying test in rodents [118, 119]. Here, 16 or 20 marbles are embedded in a 4x4 or 4x5 pattern in a corncob bedding inside a cage [118, 119]. The test rodent is free to roam in the cage for 15 mins., and the number of marbles buried by the rodent is then counted [118, 119].
We found 3 studies which delivered MDMA to rodents chronically postnatally, and tested for marble-burying behaviour (Table 7). The results show that fewer marbles are buried when 2.5 mg/kg MDMA is given intraperitoneally thrice, 3 hours apart, possibly providing a means of decreasing compulsion/stereotypy. At higher doses, however, compulsivity/stereotypy was either increased (10 mg/kg x 4, for 10 days) or was not affected (15 mg/kg x 4 weekly over 5 weeks).
Table 7.
Marble-burying propensity in postnatally MDMA-treated rodents, compared with control rodents.
| Study | Species | Sexes |
Sexes
Combined or Separated |
Sample Size | Singular/Chronic Dosing | Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Males | Females | Both |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Saadat 2006 [272] | Mouse | Males | NA | 16 | Chronic | 3 times at 3-hour intervals | 2.5 | i.p. | 25-30g | Same day | 20 | Reduced | NA | NA |
| Skelton 2008 [211] | Rat | Males | NA | 21 | Chronic | 4 times on 1 day a week, over 5 weeks | 15 | s.c. | 225-250g | 5 days later | 22 | No effect | NA | NA |
| Skelton 2009 [273] | Rat | Both | Combined | 80 | Chronic | 4 times daily | 10 | s.c. | 11-20 | 29 | 21 | NA | NA | Increased |
In terms of repetitive behaviour or stereotypies in rodents, the open-field and marble-burying tests show that when 10-5 mg/kg MDMA is given acutely, it may be possible for repetitive and compulsive behaviours to subside.
3.2.2. Other Animals
Three studies assessed stereotypies in non-rodent animals, specifically monkeys, after chronic MDMA administration. All of them found no effect on stereotypy levels [120-122]. All studies tested only males, however, with singular and chronic doses of 0.32-7.8 mg/kg by intramuscular, oral or intragastric administration [120-122].
3.2.3. Humans
There were no studies specifically investigating the effect of MDMA on repetitive behaviours in human subjects. This indicates a gap in the literature, which may be an opportunity to test potential treatment drugs (such as MDMA) on this core impairment of ASD. However, MDMA may increase stereotyped behaviour in humans, as seen in one study where when given singularly at a dose of 1.5 mg/kg orally [123].
3.3. Core ASD Behaviour 3: Cognitive Rigidity
3.3.1. Rodents
3.3.1.1. T/Y-maze Tests
The Y/T-maze can be used to test cognitive rigidity, or insistence on sameness in routine, in rodents via the spontaneous-alternation task or the food-reward task [124]. Spontaneous alternation monitors whether rodents will
Each study is shown alongside the species, sex and sample size of the rodents tested (control and relevant treatment groups), whether the study used singular or chronic (multiple) dosing, and if they used chronic dosing, the timing of the doses; the dose and route (mode of injection) used, the ages the rodents were treated and tested at, the temperature of the testing environment, and the results obtained from the experiment in the treated rodents (where it is different from the results in the corresponding control rodents). i.p. = intraperitoneal mode of injection; s.c. = subcutaneous mode of injection.
spontaneously alternate arms in successive trials [124], capitalising on natural exploratory behaviour. Animals that persist in selecting the same arm to travel down suggests cognitive rigidity or memory impairment [124]. Thus, this test is also confounded by working spatial memory, which is independent of ASD [124]. One study stated that statistically, it was more likely due to a perseverative tendency than cognitive impairment [125]. Similarly, in a trained version (which either uses a food reward, or blocks off one arm to train, before testing), rodents are explicitly reinforced to travel down one arm [124]. Once trained (training/acquisition phase), the rewarded arm is switched, and the rodent is monitored to see how quickly it explores the previously non-rewarded/non-forced arm (testing/reversal phase) [124]. Frustrated responses (such as bouts of stereotypy) to going towards the previous/wrong arm could also be monitored.
We found 21 studies where MDMA was given mostly chronically postnatally, and the rodents were assessed for their inclinations to spontaneously alternate arms, or to change habit for food (Table 8). The results show that 10-20 mg/kg MDMA seems to have less influence on cognitive rigidity than do doses either side of this range (less than 10 or more than 20 mg/kg), but this may have to do with technique inconsistencies between studies. It did not seem to make a difference whether investigators used Y- or T-shaped mazes. In those using radial mazes, we wonder if their results were confounded by spatial learning and memory as factors influencing spontaneous alternation.
Table 8.
Resistance to change travel direction in postnatally MDMA-treated rodents, compared with control rodents.
| Study | Y-/T-maze | Species | Sexes | Sexes Combined or Separated | Sample Size | Singular/Chronic Dosing | Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Food Restriction | Food | Males | Females | Both |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adeniyi 2016 [194] | Y | Mouse | Males | NA | 10 | Chronic | 5 times over 10 days, at 2-day intervals | 2 | s.c. | 21 | 31, 32 | NA | Nil | NA | No effect | NA | NA |
| Cassel 2005 [274] | T | Rat | Males | NA | 20 | Chronic | 4 times, 24 hours apart | 10 | i.p. | “Adult” | 4 days after last injection | 23 | Nil | NA | No effect | NA | NA |
| Edut 2011 [197] | Y | Mouse | Males | NA | 37 | Singular | NA | 10 | i.p. | 25-30g | “Juvenile” | 23 | Nil | NA | No effect | NA | NA |
| Edut 2014 [198] | Y | Mouse | Males | NA | NA | Singular | NA | 10 | i.p. | 25-30g | Same age | 23 | Nil | Visual cues (shapes) | No effect | NA | NA |
| Kolyaduke 2013 [157] | Y | Rat | Both | Separated | 80 | Chronic | 10 days, once a day | 10 | i.p. | 35, 45 | 90 | 22 | Nil | NA | No effect | No effect | NA |
| Odland 2019 [275] | Y | Mouse | Males | NA | 32 | Singular | NA | 1,3,10 | i.p. | 8-16 weeks | 8-16 weeks | 20-24 | Nil | NA | No effect | NA | NA |
| Ricaurte 1993 [276] | T | Rat | Males | NA | 21 | Chronic | Twice daily for 4 days | 20 | s.c. | 300g | 84 days later | NA | Not specified | Chocolate milk | No effect. | NA | NA |
| Schwarting 2005 [277] | T | Rat | Males | NA | 40 | Singular | NA | 7.5, 15 | i.p. | 409.7g | Same day | 22-24 | 3 days before, for 23 hours each | NA | NA | NA | |
| Schulz 2013 [208] | T | Rat | Males | NA | 24 | Chronic | Once daily for 10 days, twice daily (4 hours apart) for 5 days | 7.5 | s.c. | “Adult” (230-300g) | 40-65, 80-105 | 22 | Not specified | Food pellets | Increased | NA | NA |
| Young 2005 [278] | Y | Rat | Males | NA | 40+ | Singular | NA | 1.5, 2.5, 5 | i.p. | “Adult” | “Adult” | 21 | 4 days | Froot loops (cereal) | 5 mg/kg: increased | NA | NA |
| Reversal learning through odour-span task: | |||||||||||||||||
| Hawkey 2014 [279] | NA | Rat | Males | NA | 6 | Singular | NA | 0.3, 1.0, 1.8, 3.0 | i.p. | 90-150 | 90-150 | NA | Not specified | Sugar pellets | No effect | NA | NA |
| Study | Y/T maze | Species | Sexes | Sexes Combined or Separated | Sample Size | Singular/Chronic Dosing | Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Food Restriction | Food | Males | Females | Both |
| Hawkey 2014 [279] | NA | Rat | Males | NA | 6 | Chronic | Twice daily over 4 days | 10 | i.p. | 90-150 | 90-150 | NA | Not specified | Sugar pellets | Increased | NA | NA |
| Acquired operant-alternation task: | |||||||||||||||||
| Vinals 2013 [280] | NA | Mouse | Males | NA | 19-21 | Chronic | Twice daily over 4 days | 3, 30 | i.p. | 24g | 24g | 21 | Not specified | Chocolate pellets | 30 mg/kg: increased | NA | NA |
| Radial-arm maze: | |||||||||||||||||
| Braida 2002 [240] | 8-arm | Rat | Males | NA | 32 | Chronic | Daily | 1, 2, 3 | i.p. | 90 | 104 | 22 | Restricted (by 15% free-feeding weight) for 2 weeks before | Food pellets | Increased | NA | NA |
| Canales 2014 [159] | 8-arm | Rat | Females | NA | 12 | Chronic | Twice daily, 8 hours apart | 10 | NA | 13-15 | 97 | 21 | Nil | Noyes pellets | NA | No effect | NA |
| Compton 2016 [158] | Plus | Rat | Both | Separated | 26 | Chronic | 2 injections 5 times, with 5-day intervals. | 5 | NA | 35-57 | 131 | 24 | Nil | NA | Increased | Increased | NA |
| Harper 2013 [281] | 8-arm | Rat | Males | NA | 13 | Chronic | 4 times, 2-hour intervals + once a week | 10 + 4 | i.p. | 90-120 | 90-120 | NA | Nil | Chocolate chips | No effect | NA | NA |
| Harper 2013 [281] | 8-arm | Rat | Males | NA | 13 | Chronic | Once a week | 4 | i.p. | 90-120 | 90-120 | NA | Nil | Chocolate chips | No effect | NA | NA |
| Kay 2011 [282] | NA | Rat | Males | NA | 19 | Chronic | 4 times, 1 every 2 hours, for 1 day | 10 | i.p. | 90-120 | 90-120 | NA | Not specified | Chocolate chips | No effect | NA | NA |
| Kay 2011 [282] | NA | Rat | Males | NA | 19 | Chronic | Twice, 24 hours apart | 4 | i.p. | 90-120 | 90-120 | NA | Not specified | Chocolate chips | Increased | NA | NA |
| Ros-Simo 2013 [207] | 8-arm | Mouse | Males | NA | 24-30 | Chronic | Twice, 6 hours apart | 20 | i.p. | 25 | 28 | 22 | Not specified | Chocolate pellets | Increased | NA | NA |
3.3.2. Other Animals
Studies using other animals are explored in Table 9. One study assessed cognitive rigidity in non-rodent animals given MDMA, specifically cynomolgus monkeys. Using only males and chronic dosing of 1.5 mg/kg p.o., they found that when given orally, MDMA animals tended to have more reversal-learning errors, predominated by persevering at the same option [101], indicating that MDMA at this dose and route may even exacerbate cognitive inflexibility. When the dose was given intramuscularly, the same study found no such effect [101], indicating perhaps that the intramuscular route may not be as bioavailable (as the oral route) to create this exacerbating effect.
Table 9.
Non-rodent animal studies testing core ASD behaviours.
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size | Singular/Chronic Dosing | Chronic Timing | Dose (mg/kg) | Route | Treatment Age | Testing Age | Temperature (°C) | Males | Females | Both |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ali 1993 [102] | Rhesus monkey | Females | NA | 14 | Chronic | Twice daily for 4 consecutive days | 1.25, 2.5, 20 | i.g. | Varying weights | Same day | NA | NA | 20 mg/kg: serotonin behaviour | NA |
| Ballesta 2016 [98] | Long-tailed macaque | Males | NA | 3 | Chronic | 3 doses at 1-week intervals | 1, 1.5, 2 | s.c. | 3 years | 3 years | NA | 1 mg/kg: nil. 1.5 mg/kg: increased time spent being groomed | NA | NA |
| Capurro 1997 [103] | Electric fish | Both | Combined | 100 | Singular | NA | 1, 5 | i.m. | 5-50g | 3 days later | 18 | NA | NA | Decreased aggression, increased adjacent swimming, longer latency to first (aggressive) bite |
| Crean 2006 [120] | Rhesus macaque | Males | NA | 6 | Singular | NA | 0.56, 1, 1.78, 2.4 | i.m. | 6-10 years | 6-10 years | 23, 27 | No effect on stereotypy | NA | NA |
| Crean 2007 [121] | Rhesus macaque | Males | NA | 10 | Chronic | Intervals of at least 1 week | 1.78, 5 | i.m., p.o. | 6-10 years | 6-11 years | 23-27 | No effect on stereotypy | NA | NA |
| Edsinger 2018 [111] | Octopus | Both | Separated | NA | Singular | NA | 0.5-0.005 | Immersion | 9 months | 9 months | 22 | Increased social preference | Increased social preference | NA |
| Edsinger 2018 [112] | Octopus | Both | Separated | 7 | Singular | NA | 0.5-0.005 | Immersion | 9 months | 9 months | 20-23 | Increased social preference and exploratory body contact | Increased social preference and exploratory body contact | NA |
| Goodwin 2013 [122] | Baboon | Males | NA | 4 | Chronic | 5-day intervals | 0.32–7.8 | i.g. | “Adult” (18.4-33.2kg) | “Adult” (18.4-33.2kg) | NA | No effect on stereotypy | NA | NA |
| Green 2012 [104] | Zebrafish | Both | Combined | 16-24 | Singular | NA | 80 mg/L | Immersion | 5-8 months | 5-8 months | 25-27 | NA | NA | Increased inter-fish distance, decreased duration of proximity |
| Insel 1989 [283] | Rhesus monkey | Males | NA | 9-12 | Chronic | Twice daily for 4 consecutive days | 2.5, 10 | i.m. | “Adult” (7-12kg) | 1 hour later | 24 | Decreased exploration, increased passivity. 10 mg/kg may induce serotonin-syndrome-like features, upright posture and later continuous activity | NA | NA |
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size | Singular/Chronic Dosing | Chronic Timing | Dose (mg/kg) | Route | Treatment Age | Testing Age | Temperature (°C) | Males | Females | Both |
| Iravani 2003 [99] | Common marmoset | Both | Combined | 26 | Chronic | 4-7-day intervals | 12 | p.o. | 280-330g | 280-330g | 25 | NA | NA | Decreased motor activity and exploration, vocalisation. Serotonin behaviour |
| Pitts 2017 [100] | Squirrel monkey | Males | NA | 4 | Singular | NA | 0.03–3.0 | i.m. | 11-16 years | 11-16 years | 23 | Both enantiomers: increased huddling, decreased activity, increased number of affiliative vocalisations. Lower doses: S(+) MDMA increased huddling, decreased activity, more than did R(−) MDMA. R(−) enantiomer: more affiliative calls at higher doses, than S(+) enantiomer | NA | NA |
| Ponzoni 2016 [105] | Zebrafish | Both | Combined | 20 | Singular | NA | 0.1–20 | i.m. | 6-12 months | 6-12 months | 28 | NA | NA | 0.5-7.5 mg/kg: increased social preference |
| Ponzoni 2017 [106] | Zebrafish | Both | Combined | 40 | Singular | NA | 10, 0.5, 0.1 | i.m. | 6-12 months | 6-12 months | 28 | NA | NA | Increased social preference |
| Stewart 2011 [107] | Zebrafish | Both | Combined | 142 | Singular | NA | 0.25, 40, 80, 120 mg/L | Immersion | 5-7 months | 5-7 months | 25-27 | NA | NA | Dose-dependently decreased time taken to travel to the top of the tank, increased time spent up there, as well as the total number of fish swimming at the top. 40, 80, 120 mg/L: impaired zebrafish habituation |
| Verrico 2008 [101] | Cynomolgus monkey | Males | NA | 4 | Chronic | Twice, 1 week apart, over 3 weeks | 1.5 | p.o. | 4-6 years | 4-6 years | NA | Increased cognitive rigidity and social vigilance | NA | NA |
| Verrico 2008 [101] | Cynomolgus monkey | Males | NA | 5 | Chronic | Once a week for 5 weeks | 1.5 | i.m. | 4-6 years | 4-6 years | NA | No effect on cognitive rigidity, increased social vigilance. | NA | NA |
3.3.3. Humans
As shown in Table 11, there were only two placebo-controlled studies showing how MDMA affects cognitive rigidity in humans, and only one showed some effect. Given that 1.5 mg/kg MDMA is comparable to 100 mg [126], 75 mg should be comparable to approximately 1.1 mg/kg, based on the assumption of a linear positive correlation of drug mass to human body weight. Therefore, the studies compare the effects of chronic 1.1 mg/kg [127] and singular 1.5 mg/kg [123] MDMA in humans. The first study shows no effect of chronically given 1.1 mg/kg [127], but this was tested in MDMA-experienced users who may have higher tolerance levels to MDMA. Alternatively, it may be an effect of the different tests used, despite both testings for the attribute of cognitive rigidity. Van Wel et al. (2012) tested a response to changing rules in a visual game, where latency to detection of the change in rule could be a confounder, and found no effect [127]. However, in a second study which showed an inflexibility-exacerbating effect at 1.5 mg/kg, Vollenweider et al. (2005) tested the pattern of unchanging responses which may also be more subtle and subconscious, thereby less consciously manipulatable [123]. In the former study, a “cue-dependent reversal-learning task” has subjects respond to target stimuli and not respond to non-target stimuli. The target and non-target stimuli are reversed several times throughout the task, and the acquisition and reversal learning rates are measured in each subject [127]. In the latter study, a “two-choice prediction task” has subjects predict where a stimulus will be presented on a computer screen, based on previous outcomes or responses. Here, stay/switch responses are measured, as well as how much the response is influenced by the previous outcome or response, and the predictability of subject responses [123]. Notably, Vollenweider et al. (2005) had a male-biased sample, which may explain the increased vulnerability to stereotypy, as shown in males in the rodent studies [123]. There were too few studies to determine age or sex effects.
Table 11.
Resistance to change in behaviour, in postnatally MDMA-treated subjects, compared with placebo-treated subjects.
| Study | Recruitment | Population Sampled | No. Participants | Sexes | Ages | Oral MDMA Dose | Dosage Timing | Time till First Test | Relevant Tests | Relevant Results | Relevant Parameters Tested |
|---|---|---|---|---|---|---|---|---|---|---|---|
| van Wel 2012 [127] | Newspaper adverts, word of mouth | Healthy, MDMA-experienced | 17 | 8 female, 9 male | 22.76 | 75 mg (~1.1 mg/kg) | 7/+ days apart | 105 mins. | Reversal learning | No effect | Response to changing rules of shapes presented |
| Vollenweider 2005 [123] | University hospital staff, medical school | Healthy, mostly MDMA-naïve | 42 | 10 female, 32 male | 25.4-27 | 1.5 mg/kg | Singular | 120 mins. | Decision-making rigidity | Increased likelihood of previous response predicting current response; fewer different strategies used, but frequency of switching unchanged | Degree to which previous response determines next response; changing of choice strategies, and the frequency of this change |
4. DISCUSSION
We will focus our discussion on rodent and human studies only, as the other animal studies are too sparse and inconsistent for deriving substantial conclusions from.
Each study is shown alongside whether a T- or Y-shaped maze was used, species, sex and sample size of the rodents tested (control and relevant treatment groups), whether the study used singular or chronic (multiple) dosing, and if they used chronic dosing, the timing of the doses; the dose and route (mode of injection) used, the ages the rodents were treated and tested at, the temperature of the testing environment, the duration of food-restriction (deprivation) enforced before testing, the food used, and the results obtained from the experiment in the treated rodents (where it is different from the results in the corresponding control rodents). i.p. = intraperitoneal mode of injection; s.c. = subcutaneous mode of injection.
Each study is shown alongside the species, sex and sample size (control and relevant MDMA groups) of the animals tested, whether the study used singular or chronic (multiple) dosing, and if they used chronic dosing, the timing of the doses; the dose and route (mode of administration) used, the ages the animals were treated and tested at, the temperature of the testing environment, and the results obtained from the experiment in the treated animals (where it is different from the results in the corresponding control rodents). i.p. = intraperitoneal mode of administration; s.c. = subcutaneous mode of administration; p.o.: oral mode of administration; i.m.: intramuscular mode of administration; i.g.: intragastric mode of injection.
Each study is shown alongside how the participants were recruited, the type of population sampled, the number of participants, the sex ratio of the participants, the ages of the participants, the amount of MDMA consumed orally, the dosing schedule, the time from MDMA administration to the first relevant test, the relevant tests used in the study, the relevant results from those tests, and the relevant parameters measured in the relevant tests.
4.1. Social Impairment
4.1.1. Rodent Studies
The outstanding result of this positional systematic review lies in the social-impairment experiments relating to autism. The set of studies show that social behavioural results are mixed between the studies, when 5-10 mg/kg MDMA is given singularly or chronically postnatally (Table 5), with these effects persisting over hours and days (Table 5). Adding value to this treatment, since MDMA has prosocial effects chronically in rodents, humans treated with MDMA would hopefully also benefit chronically beyond the timeframe of therapy sessions.
Given the widely used face and construct validity of any rodent model to represent autistic-like features in humans [128], further investigation needs to be undertaken in rodent ASD models to explore and fine-tune possible effects of MDMA as a treatment for autism-related social impairments in future clinical research. In 2016, there was a paper considering MDMA as a potential social anxiolytic in people with ASD [129]. In fact, a recent study published by Danforth et al. (2018) showed decreased social anxiety in autistic humans who were treated with MDMA [48], further adding substance to our argument. In addition, in this study, sustained long-term effects were also present [48]. However, limitations of this study include small sample size, a wide range of results, some insignificant differences between groups, potentially confounding comorbidities unaccounted for, uncertain autism diagnostic methods, observable MDMA effects interfering with blinding (especially with inability to verify prior drug abstinence), and recruitment challenges [48]. The author follows up with a subsequent observational study of autistic MDMA users, finding both short- and long-term alleviating effects of even non-clinical use of the drug on trauma and social anxiety, with no adverse effects [130].
However, chronic/high dosing or late testing had the opposite effects in rodents. Studies that tested 12-70 days after administering the MDMA, for example evidenced decreased prosocial [91-93] and increased asocial [93] behaviour. Two authors attributed this behaviour to increased anxiety [91, 92], and one author attributed it to either a change in serotonin-receptor function (namely, especially increased 5-HT1A receptor function in the dorsal hippocampus, basolateral amygdala or lateral septum), or a reorganisation of developing serotonergic innervation (in particular the dorsal raphe neurons, notably critical to social interaction) [131]. Morley et al. (2001) support their claim by quoting increased anxiety shown by the same rodents in other experiments in their study [91]. They also provide the alternative explanation that
Each study is shown alongside how the participants were recruited, the type of population sampled, the number of participants, the sex ratio of the participants, the ages of the participants, the amount of MDMA consumed orally, the dosing schedule, the time from MDMA administration to the first relevant test, the relevant tests used in the study, the relevant results from those tests, and the relevant parameters measured in the relevant tests.
it might be due to decreased environmental exploration (which would include the conspecific, in a social test), as a high dose caused decreased locomotion in this test [91]. So although both low and high doses of MDMA were anxiogenic, low or acute doses increased social interaction (with only low doses showing upregulation of the SERT receptor), and high or chronic dosing caused decreased social interaction [91]. The lattermost, they attribute to MDMA-induced neurotoxicity or adaptation to a neurotoxic effect, creating long-lasting effects in their rats, although they concede that from other studies, this probably improved over the last 3 months [91]. Fone et al. (2002), however, who tested only 12 days after treatment, claimed there was no serotonergic neurotoxicity involved in their decreased social interaction [131].
In addition, with the same doses, chronic dosing has the opposite effect: decreasing prosocial behaviour and increasing asocial behaviour. One study was an exception to this [78], where 5-20 mg/kg MDMA given twice daily over 3 days actually increased the duration of social investigation. This may have been anomalous; the only differences the authors highlight are that their studies used a control (instead of treatment-matched) conspecific, and that asocial and exploratory behaviours are not included in that statistic [78].
We found one study where 5 or 10 mg/kg MDMA, given chronically intra-peritoneally on PD 28-52, increased mouse social novelty preference when tested later on PD 120 (Table 3). This study also shows a long-lasting prosocial effect of the MDMA, which would increase the value of MDMA as a treatment. Interestingly, one study showed subsequent MDMA administration to cause a decrease in sensitivity to MDMA-increased social behaviour in rats [83]. Here, MDMA caused decreased social interaction in the long run in rats, and also inhibited prosocial effects of subsequent MDMA doses [83].
Other studies gave surprising results, too: social and prosocial behaviours were decreased when MDMA was given at a singular 5-10 mg/kg [95-97]. Navarro et al. (2004) attributed this to an anxiogenic effect, with c-fos as a molecular marker to support this (heightened in central amygdala, associated with increased anxiety) [96]. Bull et al. (2004) found anxiogenic effects with their chronic dosing, accompanied by a modest serotonin depletion [132]. They attributed their fewer rears to an acute efflux of vesicular serotonin, and decreased 5-HT2A receptor function in the cortex/brainstem [132]. Bull et al. (2003) also found an anxiogenic effect, with reduced social interaction 20 days later, accompanied by 20-40% decreased serotonin in the hippocampus and frontal cortex [133]. They suggest that high-dose MDMA could more directly cause compensatory re-innervation via long-term amygdalar/hippocampal hyperinnervation, causing these effects, which is significant because these are the areas which modulate serotonin receptor function and/or response to negative stimuli [133]. Gurtman et al. (2002) also noticed increased anxiety, with only minimal tissue serotonin concentrations and axon density being recovered, but a large amygdalar serotonin depletion, weeks to years after MDMA administration in rats [134]. The amygdala, of course, is strongly associated with anxiety. Having said that, Welsh et al. (2005) assert that autism may be due to a disruption of the inferior olive which hinders the processing of stimuli, because its neurons are not electrically synchronised, which jeopardises rhythmic output [135]. One study used a moderate chronic dose, but found no significant serotonin depletion (unlike in high acute doses) [136]. According to Clemens et al. (2007), with an acute high MDMA dose, endogenous free-radical-scavenging is exhausted, leading to free-radical-induced damage. With chronic moderate doses, more prefrontal-cortical serotonin depletion still occurs than single-dose, as the recovery time between doses does not seem fast enough [136]. It is unclear whether this is due to neurotoxicity or natural neuroplasticity over time [136].
Some authors attribute these results to specific parts of social interaction. Thompson et al. (2008) specified that this decrease was more applicable to the general-investigation part of social behavior [83]. In fact, prior MDMA exposure’s acute effects of decreased adjacent lying (involving 5-HT1A receptor) can be rescued by increasing the MDMA dosing even more [83]. Adjacent lying is the social passive behaviour of lying side-by-side with another rodent. Thus, the authors propose that the decrease in social behaviour involves non-5-HT1A receptors, such as persistent changes in receptor densities or the serotonin transporter in the amygdala and hypothalamus, mediating social behavior [83]. Thompson et al. (2008) assert that previous MDMA dosing reduces the organism’s sensitivity to future acute MDMA doses, partly by eliciting serotonin release less, and there may also be persistent desensitisation of the oxytocinergic network [83]. Homberg et al. (2007) found these effects specific to playful social behaviours and not confounded by effects on locomotion, with accompanying increased extraneuronal serotonin [95]. They state that it could also be due to increased anxiety, as reflected by their other, anxiety-specific tests. Their increased non-social exploration could just be an indication of the rodent attempting to escape from the arena. This does not explain why only playful social behaviours are affected, however [95]. Whether social interaction can be used as a measure of social anxiety is debated [137]. They used defaecation and freezing behaviours, as well as an unfamiliar and lit environment, as indicators of how anxious the test rats must be. They found that as these variables increased, rat social interaction decreased [137]. Hence, it is possible that social interaction can indicate decreased anxiety in a rodent. Another point of note is that Kang et al. (2015) found that a novel bright environment induced stereotypies, whereas a dark familiar environment did not, in a valproic-acid-induced rodent model of autism [138].
A glaring question may be why oxytocin should not be used directly, as MDMA acts to ultimately induce oxytocin release contributing to its prosocial effects. One such study used a “cyberball” task, whereby each subject was tested on their prosocial helping behaviour towards a known socially excluded person. This study found that with intranasally administered oxytocin, subjects compensated for other players’ ostracism by throwing the ball more often towards the excluded player [139]. Another study gave intranasal oxytocin to autistic adults, and found that there was improved social cognition after 6 weeks’ treatment [140]. But in fact, one paper says that, when intra-nasally administered, very little oxytocin enters the central nervous system, and most accumulates in the peripheral bloodstream causing a myriad of adverse effects such as cardiac arrhythmias [141]. Another study directly administered intranasal oxytocin to autistic pre-teens and teenagers, and found no effects on social behavior [142]. Yet another found no increase in empathic concern after subjects were given intranasal oxytocin [143].
We also examined sex and age effects. There were some sex differences, in studies where both female and male test subjects were employed. In terms of male behaviour, a study found that gonadotrophin-releasing hormone and testosterone were suppressed in MDMA-treated rats [144], which might explain the result found above that MDMA decreased copulating behaviour in rats [84]. Females were more affected with investigation (sniffing) behaviours, whereas males were more affected with other social-interaction behaviours.
4.1.2. Human Studies
The studies suggest that a chronic oral dose of 0.75-1.5 mg/kg might be optimal to address social impairments, depending on the sex and metabolism of the individual. Caution is warranted to ensure the dose minimally affects the ability to recognise emotions in others.
For social interaction, a few specialist tests are worth mentioning to inform future clinical studies, namely a virtual ball-throwing task [145], an ultimatum game [146], the Prisoner’s Dilemma [147], and a welfare trade-off task [46]. The ball-throwing task simulates social rejection and tests for the participant’s self-ratings of self-esteem and mood, whereby a virtual “cyberball” is tossed between the participant and two experimenter-controlled virtual “players”. For the first part, social inclusion was simulated by having the ball tossed to each player equally; the second part simulated social exclusion, by tossing the ball mostly between the two virtual players only. After each part, participants self-rate positive mood and self-esteem. With MDMA, chronic 0.75 and 1.5 mg/kg reduced the decreasing effect of rejection on mood and self-esteem, and increased perceived percentage of inclusive throws under rejection condition [145]. The ultimatum game assesses social decision-making (trust and cooperation), whereby participants are paid based on their acceptance of first-person, third-person and computer-generated monetary offers [146]. The study found that chronic 1.5 mg/kg MDMA decreased the probability of rejecting unfair offers in the first-person condition, increased average percentage offer from the participants themselves, and increased prosocial interaction [146]. The Prisoner’s Dilemma also assesses choice to trust and cooperate, and the study found that chronic 1.5 mg/kg MDMA increased cooperation, especially with a trustworthy opponent, that is maintained over time [147]. Interestingly, chronic 1.5 mg/kg did not increase probability of cooperating with an untrustworthy opponent or game server (neutral), possibly indicating retained interpersonal insight [147]. Indeed, self-ratings of insightfulness have been shown to increase with singular dosings of 0.75 and 1.5 mg/kg [148], although the reliability of self-ratings needs to be checked The same dose given chronically also decreased vigilance [149].
Likewise, in another study, self-reported trust increased at a chronic 1.1 mg/kg [150]. The welfare trade-off task assesses generosity, whereby the participant chooses when to switch between monetarily benefiting themselves to benefitting others [86]. The study found that chronic 1 mg/kg increased generosity to a friend, and that chronic 1.5 mg/kg increased average percentage offer [46]. Hence, it seems that chronic 0.75-1.5 mg/kg MDMA given orally to humans is effective in increasing prosocial feelings towards another person.
A prominent advantage of using human subjects is that they are articulate, so their vocabulary under different treatments can be analysed. 0.75 and 1.5 mg/kg MDMA increased positive emotion words, whereas 1.5 g/kg increased words describing another that were social and reflective of theory of mind (insight into another person’s mental state, which is often considered lacking in ASD [151]); increased time verbally interacting, words closer to “intimacy”, “friend”, “rapport” and “support”. One study administered an oral dose of 62.5 mg/kg with subsequent top-ups of 1.9 mg/kg, and held talking sessions, where they found that those participants exhibited increased ensuic (describing a change in their sense of self), empathic (regarding others' emotions) and entactic (inclination towards physical touch) utterances [114].
Empathy has been suspected to be lacking in ASD individuals [152]. In terms of empathy, these studies showed that at a chronic 1.1 mg/kg MDMA, participants were equally aroused by positive and negative sounds, whereas in control participants, negative sounds produced more arousal than did positive [153]. Chronic 1.1 mg/kg MDMA also increased both explicit and implicit emotional empathy scores for positive emotional stimuli [150]. There was also more concern (especially for positive stimuli) and arousal (for both positive and negative (as opposed to just positive, as in controls) stimuli) for people depicting emotions, at chronic 1.1 mg/kg [154] but also at chronic 1.9 mg/kg [155]. In the middle, chronic 1.5 mg/kg MDMA decreased frown response, and increased smile response, to happy expressions [116]. And at chronic 1.9 mg/kg, there was decreased cognitive empathy for all emotions, but increased explicit emotional empathy for positive emotions [156]. Chronic 1.9 mg/kg MDMA promoted shift from joint gain maximization to inequality aversion, as well as increased prosociality via a resource-allocation task [45]. As a cautionary note, increased trust due to MDMA may even make autistic individuals more prone to be swindled, especially given that they are already thus susceptible if they lack theory of mind.
4.2. Repetitive Behaviours (Rodent and Human Studies)
The studies indicate that singular 10-5 mg/kg MDMA may reduce repetitive and compulsive behaviours in rodents, but more studies need to be conducted specifically on this, as the open-field test is unspecific for stereotypy and the marble-burying test may include compulsive behaviour unaccounted for. Only one study found significant results of stereotypy in humans [123], and this is not enough to draw conclusions from. Further controlled studies in humans are warranted, to look at the effects of MDMA on stereotypy as a core impairment in ASD.
4.3. Cognitive Rigidity (Rodent and Human Studies)
Rodent studies indicate that mostly chronic 10-20 mg/kg MDMA seems to have the least aggravating effect on cognitive rigidity. Older rodents tended to have more cognitive rigidity, but were more likely to investigate their surroundings and perform self-grooming. Younger rodents made fewer pinnings, but more head-weavings as a serotonin behaviour. For cognitive rigidity, the radial-arm maze suggested that older rodents were not as impaired compared to their placebo counterparts, as were younger rodents which showed more working-memory impairments (Table 8). This may be because older rodents naturally have impaired memory, so the difference between MDMA-treated and placebo rats may not be so significant. Only three studies tested females [157-159], so there is insufficient data to properly assess sex effects here. There were too few human studies measuring cognitive rigidity as a parameter to conclude the optimal dose at which this core impairment is most alleviated, but there is a possibility that a singular 1.5 mg/kg p.o. may actually exacerbate an inflexibility, as shown by the indirect measure of the likelihood of changing choices [123].
4.4. Accessory ASD Behaviours (Rodent and Human Studies)
Accessory ASD traits (impairments not included in the 3 core symptoms we have explored in this review) are also worth investigating. For example, because an empathy deficiency is considered to be characteristic of some ASD individuals, we will also introduce an empathy test which could, in future, be used to test behavioural impairment in autistic-like rodents. Conveniently, Bartal et al. (2011) have developed a means to assess empathic tendency in a rodent, by setting up an apparatus with a restrained conspecific inside. The test subject is placed inside the chamber, and is timed to see how quickly they open the restrainer door deliberately in order to free the conspecific [160]. Other infrequent impairments in autism include motor coordination [8]. Since some studies already use variants of motor function tests like the vermicelli-handling task or the sunflower-seed-eating task [161], we wonder whether the MDMA tests could also be extended to accessory behaviours of ASD. Anxiety is also a common comorbidity in ASD individuals [162], and an elevated plus-maze could be used to test MDMA effects in rodents for generalized anxiety [163]. For treatment purposes, however, it would be essential to look at the core impairments first, since these are by definition shared by all autistic individuals. Future directions could also include tracking for sleep or gastrointestinal disturbances, as well as seizures, as other ASD comorbidities [164].
4.5. Recent and Ongoing Clinical Trials
The first randomised double–blind placebo-controlled clinical trial being performed to test MDMA’s prosocial effects on humans with autism has yielded encouraging results [48]: improvement on the Leibowitz Social Anxiety Scale (LSAS) was substantial and statistically significant in the MDMA–treated group, and positive scores were maintained in the follow-up at 6 months. The small sample size of the study (n = 12) limits firm conclusions in regard to the potential impact of the MDMA treatment [48]. Nevertheless, the solid basis of this randomised double-blind placebo-controlled study justifies larger randomised trials to assess MDMA effects on social anxiety in subjects with autism. Of note, another trial is ongoing (ClinicalTrials.gov #NCT04053036) in which the relevant parameter being tested is change in responses to affective touch in autistic adults. No final results are reported at the time of our writing.
Based on MDMA’s well-established prosocial effects, the studies reviewed and the results obtained from the completed trial mentioned above, for this new ongoing trial, we are expecting decreased social anxiety, increased empathy/perception/response to positive emotions/touch, and decreased empathy/perception/response to negative emotions/touch. These trials are based on MDMA’s known effects of increasing oxytocin release, via serotonergic pathways, such that oxytocin deactivates amygdalar alarm signals to the brainstem which then no longer induces behavioural and sympathetic fear responses.
4.6. MDMA Neurotoxicity
The potential toxicology for new pharmacotherapeutic agents is a necessary consideration for adoption. Helpfully, MDMA has been in clinical and recreational use for several decades, and there is now considerable data describing its adverse effects [165]. The most prominent toxicological finding in the literature arises from the excessive administration of MDMA which can cause structural and physiological changes in serotonin neurons. For example, administration of 20 mg/kg MDMA (s.c.) twice daily for 4 days causes decreased brain serotonin in the forebrain in rats by selectively degenerating serotonergic axons [166, 167]. The serotonin axon terminals are selectively ablated, resulting in fragmented projections and causing the serotonin to stay in the pre-terminal fibres, in rats [166]. Serotonin and dopamine are deaminated via monoamine-oxidase (MAO) enzymes (subtypes A and B), forming hydrogen peroxide in serotonergic nerve terminals in vitro, which seems to contribute to serotonergic neurotoxicity [168]. This is substantiated by a 26% decreased 5-HTIAA concentration in the cerebrospinal fluid of individuals with previous MDMA use, suggesting impaired central serotonergic activity [169]. It has therefore, been argued that these adaptive changes in serotonin neurons cause long-term neuropsychological and affective impairments, as has been shown in recreational MDMA users [170, 171].
However, recreational MDMA users also tend to use other drugs in addition to ecstasy [172-174], which could also be the reason for these adverse effects. In fact, one study showed that people who attempt to restrict their drug intake exclusively to ecstasy seem to have negligible adverse neuropsychological sequelae from repeated use [175]. Other potential adverse effects arising from acute ingestion of MDMA in uncontrolled settings include mild serotonin-syndrome symptoms including hyperthermia and resultant hyponatremia, particularly in warm crowded active environments [172, 176]. Further consideration of putative MDMA toxicity also requires consideration of toxic synthetic by-products produced during the illicit synthesis of MDMA. In particular, paramethoxyamphetamine (PMA) is a highly toxic potential by-product of illicit MDMA synthesis that targets serotonin neurons [177]. However, irrespective of potential toxicity from recreational doses and uncontrolled ecstasy manufacture, the clinical use of MDMA has not been shown to produce serious persistent adverse effects in any of the clinical trials reported to date [178-180]. Functional adverse effects are reflective of these serotonin-neuron structural changes, and there are both short- and long-term effects of MDMA use in humans [44, 51, 127, 181, 182].
Hyperthermia has also been associated with worsening neurotoxicity, thus less hyperthermia may mean less serotonin depletion, with their weekly doses [136]. The hyperthermic effects of MDMA have been seen in both humans and rodents. In fact, rodent prosocial behaviour is amplified when the temperature is increased from the normal ~21-220C to ~280C during testing [69]. According to Clemens et al. (2007), repeated MDMA doses decrease the hyperthermic effect (measuring at 8-16 weeks of dosing). This may produce tolerance against serotonin depletion [136]. In terms of its neurotoxicity, MDMA is known to deplete serotonergic terminals in rodents, as well as cortical, hippocampal and striatal neurodegeneration [183]. In addition, MDMA-induced hyperthermia, monoamine oxidation (of dopamine and serotonin), dopamine oxidation, serotonin transports nitric oxide, neurotoxin and peroxinitrite formation, glutamate excitotoxicity, 5-HT2A agonism all contribute to this neurotoxicity [183]. Therefore, its dose should be monitored.
4.7. Dosing Regimen
We are aware that there is a narrow range of doses testable for MDMA. One of the studies found that when MDMA was given on PD 1-5 to rodents, there was increased offspring mortality [184]. Therefore, it would be inadvisable on ethical grounds to test MDMA given within this timeframe.
Previous studies have used interspecies scaling to convert animal drug doses to human drug doses (Fig. 3), but Green et al. (2009) emphasise that bioavailability, active metabolites, plasma-protein binding and systemic exposure patterns all differ between species, and would all affect onset, intensity and duration of the dug’s effect on the subject [185]. Green et al. (2009) compare MDMA dose-plasma concentration response curves between humans and rats, and highlight that an autoinhibition of MDMA metabolism occurs in humans that is unseen in rats, implicating that rats would require 4 times the dose given to humans, to produce similar peak concentrations in the bloodstream [185]. This would need to be taken into account when transitioning from pre-clinical to clinical studies.
Fig. (3).
Proposed equation to calculate equivalent dosages between animals and humans, where D = dose (mg) and W = weight (kg) [185].
There are limitations to administering MDMA in healthy animals and humans, also. MDMA may have unseen spurious effects around the body, which may modulate the parameters measured. Further, the ethics of giving an active drug to already healthy organisms are questionable.
4.8. Summary
In rodents, administering the investigated 5-10 mg/kg MDMA, singularly/chronically intra-peritoneally/ subcutaneously, seems to ameliorate only social impairments significantly in some studies, whereas its effects on cognitive inflexibility and motor stereotypies are either exacerbated or have no effect. Therefore, MDMA’s dose-response curve needs to be assessed, through varying dose, route, temperature and timing, in an effort to find a way to reduce these core impairments in ASD. In any particular autism patient, clinicians may then prescribe a dosing regimen according to their unique proportions of social impairment, cognitive rigidity and motor stereotypies. This is only if more definitive clinical trials support its use, and if the agent can then be legally prescribed.
In humans, the optimal dose at which maximal social impairment alleviation and minimal emotion recognition impairment seems to be 0.75 mg/kg in women and 1.9 mg/kg in men, taken orally and chronically. At 1.5 mg/kg, taken orally chronically, repetitive stereotypy may increase, so doses will have to be adjusted to the individual accordingly. Again, this is only if more definitive clinical trials support its use, and if the agent can then be legally prescribed.
In non-rodent animals, different species have different doses and routes having the same effect of social impairment alleviation, no effect on stereotypy, and similarly to humans, a chronic 1.5 mg/kg p.o. dose causing cognitive rigidity in monkeys. However, sample sizes for non-rodent animal studies are low, especially as the size of the animals is bigger (costs of upkeep), so further pre-clinical studies are likely warranted before making further conclusions.
4.9. Limitations
Our review has several limitations. Firstly, only English-language papers were reviewed. Thus, there is a significant gap in the literature for the MDMA treatment of ASD behaviours. Furthermore, whilst the authors have attempted to provide the most up-to-date evidence possible, there is still this temporal limitation that precludes papers published after the time of study identification from being included in this review. In addition, only six databases (Google Scholar, NCBI, ProQuest, Scopus, Web of Science and Wiley Online Library) have been screened, given the time constraints of this project. Behaviours not screened in this review may have affected the core ASD behaviours studied. In addition, molecular analogues of MDMA may be worth investigating, to optimise treatment drugs to maximise alleviations to the addressed impairments, and minimise the adverse effects and exacerbations of impairments. The use of molecular MDMA analogues to affect human behavioural effects has been explored in a recent review [186]. Methylenedioxymethamphetamine (MDA), 3,4-methylenedio-xyethylamphetamine (MDE) and N-methyl-1,3-benzodioxolbutanamine (MBDB), in particular, have also been proposed as potential psychotherapeutic agents due to their entactogenic properties [186]. Since there are also a wide variety of rodent strains, each of which may have different responses, this would need to be monitored in the future, and may help explain some of the mixed results in the literature. There are also limitations of construct and face validity in mapping healthy rodents to ASD humans: healthy rodents would not have the neurological wiring aberrations present in ASD humans, on top of their differences in species. Hence, their respective MDMA effects may not be so comparable.
CONCLUSION
Whilst social impairment may possibly be addressed in this way, we have surveyed the effects of MDMA on the other core autism-related impairments in rodent studies too, and we find we cannot apply MDMA to reverse these other core behavioural impairments. There are gaps in the current knowledge in this area, as well. Stereotypy and cognitive rigidity in humans and animals have not been studied adequately. Overall, the clinical implications of these studies are that MDMA may be a cost-effective therapeutic for the social disability experienced by ASD individuals, but more pre-clinical trials are needed to establish MDMA as an effective medication for this impairment; and that MDMA may have persistent effects on the prosocial behaviour induced, aiding long-term therapy for autistic individuals. The latest results from recent clinical trials corroborate the hypothesis of MDMA as a valuable tool in addressing social impairment in subjects with autism, but this is far from proven yet.
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size |
Singular/
Chronic Dosing |
Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Social Restriction | Males | Females | Both |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cornish 2003 [69] | Rat | Males | NA | 24 | Singular | NA | 5 | i.p. | NA | NA | 21, 30 | Nil | Increased social interaction, more so at 30°C than 20°C. | NA | NA |
| Curry 2018 [219] | Mouse | Males | NA | 20 | Singular | NA | 3-30 | i.p. | 49-70 | 47-70 | 22 | 30 mins. | 7.8 mg/kg SR-MDMA increased social interaction. 17 mg/kg R-MDMA increased social interaction. S-MDMA had trend towards significance at 7.8 mg/kg | NA | NA |
| Curry 2019 [220] | Mouse | Males | NA | 20 | Chronic | Every alternate day, 4 sessions | 7.8 | i.p. | 49-70 | 49-70 | 22 | 25 mins. | Dose-dependently increased duration of social interaction (non-anogenital sniffing and adjacent lying) | NA | NA |
| Daza-Losada 2008 [78] | Mouse | Males | NA | 43-64 | Chronic | Twice daily | 5, 10, 20 | NA | 28-30 | 51 | 21 | Nil | Increased social-investigation time. | NA | NA |
| Daza-Losada 2009 [221] | Mouse | Males | NA | 315 | Singular | NA | 5, 10, 20 | i.p. | 28 | 28 | 21 | Nil | 5 mg/kg: increased social investigation and contact time. 20 mg/kg: decreased social interaction and contact time time. 10 mg/kg: increased distant exploring. 5, 10 mg/kg: decreased non-social exploration. | NA | NA |
| Dornan 1991 [222] | Rat | Males | NA | 19 | Chronic | Every 12 hours, for 4 consecutive days | 40 | s.c. | 350-475g | 7, 14 days later | 21 | NA | After 7 days: smaller percentage of MDMA rats ejaculated; post-ejaculatory interval increased after repeated injections. After 14 days: no effect. | NA | NA |
| Fone 2002 [131] | Rat | Males | NA | 36 | Chronic | Twice daily | 7.5 | i.p. | 39-41 | 51-53 | 21 | Nil | Decreased social exploration (sniffing, following conspecific). | NA | NA |
| Garcia-Pardo 2015 [93] | Mouse | Males | NA | 30 | Singular | NA | 1.25, 10 | i.p. | 60 | 81 | NA | 1 min. | Less time in attack, social contact. More time in avoidance/flee, defence/submission, distant exploration of conspecific. | NA | NA |
| Gurtman 2002 [134] | Rat | Males | NA | 26 | Chronic | Each of 4 hours | 5 | i.p. | 90 | 132 | 22 | Nil | Decreased time and frequency of social interaction. | NA | NA |
| Homberg 2007 [95] | Rat | Males | NA | NA | Singular | NA | 0.5, 2, 5 | s.c. | 28-35 | Same day | 21 | 3.5 hours | Dose-dependently decreased pinning, pouncing, boxing. Decreased following/chasing. 2, 5 mg/kg: decreased social exploration, social grooming. | NA | NA |
| Kirilly 2006 [223] | Rat | Males | NA | 16 | Singular | NA | 15 | i.p. | 49 | 70 | 21 | 14 days | Increased kicking, decreased grooming and social behaviour. Acute decreased biting, boxing, kicking, wrestling time, increased time to start wrestling. Decreased social behaviour and grooming. When young intruders introduced, increased locomotion and general exploration. 2nd MDMA dose increased locomotion and general exploration more than first dose. | NA | NA |
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size |
Singular/
Chronic Dosing |
Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Social Restriction | Males | Females | Both |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Kirilly 2010 [224] | Rat | Males | NA | NA | Chronic | Twice | 15 | i.p. | 42-49 | 21 days later, and once on testing day | 21 | 14 days | Acute: decreased aggressive-type behaviour. 1st dose: decreased biting, boxing. | NA | NA |
| Kurling 2008 [225] | Rat | Males | NA | 12 | Singular | NA | 1, 5 | i.p. | “Adult” (300–380g) | Same day | 21 | 6 days | Increased locomotor activity, rearing bouts, agitation, stereotyped sniffing, head/body weaving, head bobbing. | NA | NA |
| Machalova 2012 [79] | Mouse | Males | NA | 98 | Singular | NA | 2.5, 10, 30 | i.p. | “Juvenile” (18-20g) | Same day | 22 | Not specified | Aggressive mice: decreased aggression, increased timidity (defensive posture; 30 mg/kg: escape; 10, 30 mg/kg: alert posture); 2.5, 10 mg/kg: increased social behaviour (2.5 mg/kg: social sniffing; 2.5, 10 mg/kg: conspecific-following); 30 mg/kg: walking. Timid mice: 10, 30 mg/kg: increased timidity (increased alter- posture frequency); 30 mg/kg: increased escape frequency. Decreased social behaviour frequency (social sniffing; 10, 30 mg/kg: conspecific-following). 30 mg/kg: walking. |
NA | NA |
| Maldonado 2001 [226] | Mouse | Males | NA | NA | Singular | NA | 0.5, 1.25, 2.5 | i.p. | NA | Same day | NA | NA | 0.5, 1.25 mg/kg: decreased shorter inter-attack intervals | NA | NA |
| Maldonado 2001 [88] | Mouse | Males | NA | 60 | Singular | NA | 1, 8, 15 | i.p. | “Juvenile” (25-30g) | Same day | 21 | 30 days (for aggression) | 8, 15 mg/kg: decreased grooming, digging, social exploration, threat/attack time. 8, 15 mg/kg: increased non-social exploration, defence/submission, distance exploration, avoidance. 1 mg/kg: increased distant exploration, avoidance. |
NA | NA |
| McGregor 2003 [92] | Rat | Males | NA | 24 | Chronic | Each of 4 hours over 2 consecutive days | 5 | i.p. | 90 | 160 | 28 | Nil | Decreased social interaction time and frequency. | NA | NA |
| McGregor 2003 [92] | Rat | Males | NA | 24 | Singular | NA | 5 | i.p. | 90 | 160 | 28 | Nil | Decreased social interaction time. | NA | NA |
| McGregor 2003 [77] | Rat | Males | NA | 32 | Chronic | Each of 4 hours over 2 consecutive days | 5 | i.p. | 60-75 | 56-70 days later | 28, 16 | Nil | Less social interaction, fewer social-interaction bouts. | NA | NA |
| Miczek 1994 [227] | Mouse | Males | NA | 29 | Singular | NA | 0.3, 3, 6, 10 | NA | “Adult” | “Adult” | 21 | Nil | 3-10 mg/kg: decreased frequency of attacks > decreased dose-dependent sideways threats. |
NA | NA |
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size |
Singular/
Chronic Dosing |
Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Social Restriction | Males | Females | Both |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Morley 2000 [228] | Rat | Males | NA | 56 | Singular | NA | 1.25, 2.5, 5 | i.p. | 85-95 | 99-109 | 22 | Nil | Decreased aggression time. 5 mg/kg: increased social interaction time, locomotion. 1.25 and 2.5 mg/kg had no effect on locomotion or social interaction duration | NA | NA |
| Morley 2001 [91] | Rat | Males | NA | 32 | Chronic | Each of 4 hours on 2 consecutive days | 5 | i.p. | 75-95 | 160-180 | 22 | Nil | Decreased total interaction time and frequency, rearing frequency, locomotion. | NA | NA |
| Morley 2001 [91] | Rat | Males | NA | 32 | Singular | NA | 5 | i.p. | 75-95 | 160-180 | 22 | Nil | Decreased total interaction time and frequency, rearing frequency, locomotion, social interaction. | NA | NA |
| Morley 2005 [81] | Rat | Males | NA | 112 | Singular | NA | 5 | i.p. | 50-90 | 50-90 | 28 | 5 mins. | Increased total social interaction time (adjacent lying, approach), decreased anogenital sniffing and rearing. Serotonin syndrome: greater turning, low body posture, piloerection. | NA | NA |
| Morley-Fletcher 2002 [190] | Mouse | Males | NA | 30 | Chronic | Once on each of 3 days, 2 days apart | 5, 10 | i.p. | 28, 38, 52 | 80 | 21 | Nil | Altered environmental exploration time (dosed at early and late adolescence: dose-dependent increase; dosed at middle-adolescence: decrease). Decreased self-grooming. Decreased freezing time (dosed at early adolescence). | NA | NA |
| Navarro 1998 [89] | Mouse | Males | NA | 96 | Singular | NA | 0.5, 1.25, 2.5, 5, 10, 15, 20 | i.p. | 42 | 42 | 20 | 30 days (for aggression) | Decreased aggression (threat, attack) and social exploration; increased distant exploration, avoidance and defence. 5-20 mg/kg: decreased threat/attack time. 2.5-20 mg/kg: increased distant exploration. 5-20 mg/kg: increased non-social exploration, avoidance/defence; decreased social exploration. 1.25-20 mg/kg: decreased digging. 2.5, 20 mg/kg: decreased grooming. | NA | NA |
| Navarro 2004 [96] | Mouse | Males | NA | 40 | Singular | NA | 1, 8, 15 | i.p. | 25-30g | 30 mins. later | 20 | Nil | 1, 8, 15 mg/kg: decreased social-investigation time. 8, 15 mg/kg: decreased digging, increased avoidance/flee. | NA | NA |
| Navarro 2004 [90] | Mouse | Males | NA | 120 | Singular | Experimental day 7 | 1.25, 2.5, 5 | i.p. | 25-30g | 30 mins. after last injection | 21 | 30 days | Increased non-social exploration. 2.5, 5 mg/kg: decreased digging time, threat, attack; increased distant exploration. 5 mg/kg: increased avoidance/flee, defence/submission. | NA | NA |
| Navarro 2004 [90] | Mouse | Males | NA | 120 | Chronic | Experimental days 1-7, daily | 1.25, 2.5, 5 | i.p. | 25-30g | 31 mins. after last injection | 21 | 30 days | 1.25 mg/kg: increased digging. 2.5 mg/kg: increased exploration from distance, decreased non-social exploration behaviors. | NA | NA |
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size |
Singular/
Chronic Dosing |
Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Social Restriction | Males | Females | Both |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Piper 2008 [205] | Rat | Males | NA | 20-24 | Chronic | 4 doses, 1 each hour | 10 | s.c. | “Young adult” (307.7g) | “Young adult” | 23 | Nil | No effect. | NA | NA |
| Procopio-Souza 2011 [85] | Mouse | Males | NA | 24 | Chronic | Acute dose, then repeat 10 days later | 10 | i.p. | 90 | 90, 100 | 22-23 | NA | Increased locomotor frequency (housing: group > individual). Group-housed: increased social-interaction time. 2nd dose: higher locomotion frequency; group-housed: repeated dosing did not modify social behavior; but increased body contact, peaceful following; decreased anogenital sniffing. | NA | NA |
| Ramos 2013 [229] | Rat | Males | NA | 32 | Singular | NA | 2.5, 5 | i.p. | “Adult” (250-300g) | Same day | 23 | 1 hour | 5 mg/kg: increased adjacent-lying time, decreased anogenital sniffing and rearing. | NA | NA |
| Ramos 2015 [230] | Rat | Males | NA | 10 | Singular | NA | 5 | i.p. | “Adult” (250-300g) | “Adult” (250-300g) | 21 | Nil | More time spent in physical contact with a conspecific (but this also occurred with an inanimate object). | NA | NA |
| Rodriguez-Arias 2011 [231] | Mouse | Males | NA | 40 | Chronic | Twice, 4 hours apart, on each of 2 days, 6 days apart | 10, 20 | i.p. | 41, 42 | 64 | 21 | Nil | 20 mg/kg: less social-investigation time, less threat and attack times, more non-social exploration time. | NA | NA |
| Rodriguez-Arias 2015 [232] | Mouse | Males | NA | 179 | Chronic | Twice daily (4-hour intervals), over 2 consecutive days a week | 10, 20 | i.p. | 33, 34, 40, 41 | 75 | 21 | Nil | More aggression (10, 20 mg/kg: more attacks; 10 mg/kg: more threats). 10 mg/kg: more time in social investigation, less time in non-social investigation. | NA | NA |
| Shen 2011 [233] | Rat | Males | NA | 60 | Chronic | Twice every 5th day | 10 | s.c. | 52-62 | 72 | 26 | 7 days | Reduced crawling frequency and time. | NA | NA |
| Slais 2005 [234] | Mouse | Both | NA | NA | Chronic | 2.5 mg/kg: once daily for 5 consecutive days; 30 mg/kg: 8 days later | 2.5, 30 | p.o. | NA | NA | NA | Nil | Decreased aggression in aggressive mice, decreased prosocial behaviour in timid mice. Increased defence-escape behaviour. MDMA pre-treatment (2.5 mg/kg) had no effect. | - | - |
| Slais 2005 [235] | Mouse | Males | NA | NA | Singular | NA | 2.5, 10, 30 | p.o. | NA | NA | NA | Always | Decreased aggression, increased sociability and defence-escape behaviours in aggressive mice. Increased defence-escape behaviour and decreased sociability in timid mice. |
NA | NA |
| Study | Species | Sexes | Sexes Combined or Separated | Sample Size |
Singular/
Chronic Dosing |
Chronic Timing | Dose (mg/kg) | Route | Treatment Age (PD) | Testing Age (PD) | Temperature (°C) | Social Restriction | Males | Females | Both |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Slais 2009 [236] | Mouse | NA | NA | 49 | Chronic | Twice, 1-week interval | 2.5, 10, 30 | p.o. | NA | NA | NA | Nil | Aggressive mice: decreased aggression and sociability, increased defence-escape behaviour; 30 mg/kg: increased walking. Timid mice: increased defence-escape behaviour, decreased prosociality; 30 mg/kg: increased walking | - | - |
| Slamberova 2015 [97] | Rat | Males | NA | 246 | Singular | NA | 2.5, 5.0, 10 | s.c. | “Adult” | Same day | 22-24 | Nil | Decreased time and frequency of mutual sniffing. 10 mg/kg: decreased frequency of mutual sniffing and following. Decreased time and frequency of climbing over conspecific. 5, 10 mg/kg: decreased rearing frequency, time and frequency of allogrooming. | NA | NA |
| Thompson 2004 [237] | Rat | Males | NA | 25 | Chronic | Each of 4 hours on 2 consecutive days | 5 | i.p. | 332g | 84-105 days later | 28 | Nil | Less time in social interaction. | NA | NA |
| Thompson 2007 [52] | Rat | Males | NA | 16 | Singular | NA | 5 | i.p. | 403g | 403g | 28 | Nil | Increased adjacent lying; decreased general investigation, anogenital investigation and rearing. | NA | NA |
| Thompson 2008 [83] | Rat | Males | NA | 48 | Chronic | Each of 4 hours over 2 consecutive days | 5 | i.p. | 6-16 | 90-100 | 28 | 20 mins. | Chronically decreased total social interaction. | NA | NA |
| Thompson 2008 [83] | Rat | Males | NA | 48 | Chronic | Each of 4 hours over 2 consecutive days | 2.5 | i.p. | 9-19 | 90-100 | 28 | 20 mins. | Decreased investigation. | NA | NA |
| Thompson 2008 [83] | Rat | Males | NA | 48 | Singular | NA | 5 | i.p. | 8-18 | 90-100 | 28 | 20 mins. | 2.5 mg/kg: decreased total social interaction, adjacent lying. | NA | NA |
| Thompson 2009 [82] | Rat | Males | NA | 36 | Singular | NA | 5 | i.p. | “Adult” (345g) | Same day | 28 | 20 mins. | Increased total social interaction and general exploration; decreased anogenital sniffing and rearing. | NA | NA |
| van Nieuwenhuijzen 2010 [212] | Rat | Males | NA | 24 | Chronic | Daily over 10 days | 5 | i.p. | 220-300g | 220-300g | 21 | Nil | Decreased general-investigation time. | NA | NA |
| Wallinga 2009 [80] | Rat | Males | NA | 21 | Chronic | Thrice, 3-hour intervals | 6 | i.p. | 425g | 133 | 21 | 1 day | Low-aggressive: 4-month-old intruder caused increased aggressiveness. | NA | NA |
Table 5.
Definitions of specific physical interactions.
| Behaviour | Definition | Type |
|---|---|---|
| Anogenital sniffing | The rodent sniffing the anogenital areas of the conspecific | Social investigative (neither friendly nor hostile) |
| Crawling/mounting | The rodent crawling over the conspecific | Prosocial |
| Digging | The rodent digging a hole into the bedding of the apparatus | Compulsive/repetitive/anxious |
| Fighting | Fighting in adults (approximately PD 90-120) | Hostile |
| Non-anogenital sniffing | The rodent sniffing the non-anogenital areas of the conspecific | Prosocial investigative (friendly) |
| Pinning | The rodent standing over the conspecific which lies flat on its back | Prosocial |
| Play fighting | Fighting in juveniles (approximately PD 30-50) | Prosocial |
| Play responsiveness | The probability of being pinned in response to being pounced on | Prosocial |
| Rearing | The rodent standing up on its hind legs | Environmentally investigative |
ACKNOWLEDGEMENTS
Special thanks to Melissa Black who checked the search protocol for all relevant studies.
LIST OF ABBREVIATIONS
- 5-HT
serotonin
- ASD
autism spectrum disorder
- DSM
Diagnostic and statistical manual (of mental disorders)
- FDA
Food and drug administration
- i.g.
Intragastric (mode of administration)
- i.m.
Intramuscular (mode of administration)
- i.p.
intraperitoneal (mode of administration)
- ICD
International classification of diseases
- MAO
Monoamine oxidase
- MBDB
N-methyl-1,3-benzodioxolbutanamine
- MDA
Methylenedioxymethamphetamine
- MDE
3,4-methylenedio-xyethylamphetamine
- MDMA
3,4-methylene-dioxy-methamphetamine
- OXTR
Oxytocin receptor
- p.o.
Per os (mode of administration)
- PD
Postnatal day
- PMA
Paramethoxyamphetamine
- PRISMA
Preferred Reporting Items for Systematic Reviews and Meta-Analyses
- s.c.
Subcutaneous (mode of administration)
- SERT
Serotonin
- USV
Ultrasonic vocalisation
CONSENT FOR PUBLICATION
Not applicable.
STANDARD OF REPORTING
PRISMA guidelines and methodology were followed.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
REFERENCES
- 1.World Health Organization . International Classification of Diseases. 11th ed. Geneva: 2018. [Google Scholar]
- 2.American Psychiatric Association . Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Washington, DC: 2013. [Google Scholar]
- 3.Anderson M.P., Hooker B.S., Herbert M.R. Bridging from cells to cognition in autism pathophysiology: biological pathways to defective brain function and plasticity. Am. J. Biochem. Biotechnol. 2008 [Google Scholar]
- 4.Park H.R., Lee J.M., Moon H.E., Lee D.S., Kim B-N., Kim J., Kim D.G., Paek S.H. A short review on the current understanding of autism spectrum disorders. Exp. Neurobiol. 2016;25(1):1–13. doi: 10.5607/en.2016.25.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lee S.J., Levounis P. Gamma hydroxybutyrate: an ethnographic study of recreational use and abuse. J. Psychoactive Drugs. 2008;40(3):245–253. doi: 10.1080/02791072.2008.10400639. [DOI] [PubMed] [Google Scholar]
- 6.Renty J.O., Roeyers H. Quality of life in high-functioning adults with autism spectrum disorder: The predictive value of disability and support characteristics. Autism. 2006;10(5):511–524. doi: 10.1177/1362361306066604. [DOI] [PubMed] [Google Scholar]
- 7.Anckarsäter H., Stahlberg O., Larson T., Hakansson C., Jutblad S-B., Niklasson L., Nydén A., Wentz E., Westergren S., Cloninger C.R., Gillberg C., Rastam M. The impact of ADHD and autism spectrum disorders on temperament, character, and personality development. Am. J. Psychiatry. 2006;163(7):1239–1244. doi: 10.1176/ajp.2006.163.7.1239. [DOI] [PubMed] [Google Scholar]
- 8.AIHW . 2020. [Google Scholar]
- 9.Mulder A.M., Cashin A. The need to support students with autism at university. Issues Ment. Health Nurs. 2014;35(9):664–671. doi: 10.3109/01612840.2014.894158. [DOI] [PubMed] [Google Scholar]
- 10.Magiati I., Tay X.W., Howlin P. Cognitive, language, social and behavioural outcomes in adults with autism spectrum disorders: a systematic review of longitudinal follow-up studies in adulthood. Clin. Psychol. Rev. 2014;34(1):73–86. doi: 10.1016/j.cpr.2013.11.002. [DOI] [PubMed] [Google Scholar]
- 11.Howlin P., Goode S., Hutton J., Rutter M. Adult outcome for children with autism. J. Child Psychol. Psychiatry. 2004;45(2):212–229. doi: 10.1111/j.1469-7610.2004.00215.x. [DOI] [PubMed] [Google Scholar]
- 12.Kopetz P.B., Endowed E. Autism worldwide: Prevalence, perceptions, acceptance, action. J. Soc. Sci. 2012;8(2):196. doi: 10.3844/jssp.2012.196.201. [DOI] [Google Scholar]
- 13.Waterhouse L. Autism overflows: increasing prevalence and proliferating theories. Neuropsychol. Rev. 2008;18(4):273–286. doi: 10.1007/s11065-008-9074-x. [DOI] [PubMed] [Google Scholar]
- 14.Hansen S.N., Schendel D.E., Parner E.T. Explaining the increase in the prevalence of autism spectrum disorders: the proportion attributable to changes in reporting practices. JAMA Pediatr. 2015;169(1):56–62. doi: 10.1001/jamapediatrics.2014.1893. [DOI] [PubMed] [Google Scholar]
- 15.King M., Bearman P. Diagnostic change and the increased prevalence of autism. Int. J. Epidemiol. 2009;38(5):1224–1234. doi: 10.1093/ije/dyp261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wink L.K., Erickson C.A., McDougle C.J. Pharmacologic treatment of behavioral symptoms associated with autism and other pervasive developmental disorders. Curr. Treat. Options Neurol. 2010;12(6):529–538. doi: 10.1007/s11940-010-0091-8. [DOI] [PubMed] [Google Scholar]
- 17.McPheeters M.L., Warren Z., Sathe N., Bruzek J.L., Krishnaswami S., Jerome R.N., Veenstra-Vanderweele J. A systematic review of medical treatments for children with autism spectrum disorders. Pediatrics. 2011;127(5):e1312–e1321. doi: 10.1542/peds.2011-0427. [DOI] [PubMed] [Google Scholar]
- 18.Siegel M., Beaulieu A.A. Psychotropic medications in children with autism spectrum disorders: a systematic review and synthesis for evidence-based practice. J. Autism Dev. Disord. 2012;42(8):1592–1605. doi: 10.1007/s10803-011-1399-2. [DOI] [PubMed] [Google Scholar]
- 19.Dove D., Warren Z., McPheeters M.L., Taylor J.L., Sathe N.A., Veenstra-VanderWeele J. Medications for adolescents and young adults with autism spectrum disorders: a systematic review. Pediatrics. 2012;130(4):717–726. doi: 10.1542/peds.2012-0683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bauman M.L. Medical comorbidities in autism: challenges to diagnosis and treatment. Neurotherapeutics. 2010;7(3):320–327. doi: 10.1016/j.nurt.2010.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Morgan C.N., Roy M., Chance P. Psychiatric comorbidity and medication use in autism: A community survey. Psychiatr. Bull. 2003;27(10):378–381. doi: 10.1017/S0955603600003160. [DOI] [Google Scholar]
- 22.Chadman K.K., Fernandes S., DiLiberto E., Feingold R. Do animal models hold value in Autism spectrum disorder (ASD) drug discovery? Expert Opin. Drug Discov. 2019;14(8):727–734. doi: 10.1080/17460441.2019.1621285. [DOI] [PubMed] [Google Scholar]
- 23.Hong M.P., Erickson C.A. Investigational drugs in early-stage clinical trials for autism spectrum disorder. Expert Opin. Investig. Drugs. 2019;28(8):709–718. doi: 10.1080/13543784.2019.1649656. [DOI] [PubMed] [Google Scholar]
- 24.Seida J.K., Ospina M.B., Karkhaneh M., Hartling L., Smith V., Clark B. Systematic reviews of psychosocial interventions for autism: an umbrella review. Dev. Med. Child Neurol. 2009;51(2):95–104. doi: 10.1111/j.1469-8749.2008.03211.x. [DOI] [PubMed] [Google Scholar]
- 25.McPhilemy C., Dillenburger K. Parents’ experiences of applied behaviour analysis (ABA)‐based interventions for children diagnosed with autistic spectrum disorder. Br. J. Spec. Educ. 2013;40(4):154–161. doi: 10.1111/1467-8578.12038. [DOI] [Google Scholar]
- 26.Kirkham P. ‘The line between intervention and abuse’–autism and applied behaviour analysis. Hist. Human Sci. 2017;30(2):107–126. doi: 10.1177/0952695117702571. [DOI] [Google Scholar]
- 27.Rodgers M., Marshall D., Simmonds M., Le Couteur A., Biswas M., Wright K., Rai D., Palmer S., Stewart L., Hodgson R. Interventions based on early intensive applied behaviour analysis for autistic children: a systematic review and cost-effectiveness analysis. Health Technol. Assess. 2020;24(35):1–306. doi: 10.3310/hta24350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sequeira S., Ahmed M. Meditation as a potential therapy for autism: A review. Autism Res. Treat. 2012;2012 doi: 10.1155/2012/835847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hume K., Bellini S., Pratt C. The usage and perceived outcomes of early intervention and early childhood programs for young children with autism spectrum disorder. Top. Early Child. Spec. Educ. 2005;25(4):195–207. doi: 10.1177/02711214050250040101. [DOI] [Google Scholar]
- 30.Spain D., Sin J., Chalder T., Murphy D., Happe F. Cognitive behaviour therapy for adults with autism spectrum disorders and psychiatric co-morbidity: A review. Res. Autism Spectr. Disord. 2015;9:151–162. doi: 10.1016/j.rasd.2014.10.019. [DOI] [Google Scholar]
- 31.Gold C., Wigram T., Elefant C. Music therapy for autistic spectrum disorder. Cochrane Database Syst. Rev. 2006;2 doi: 10.1002/14651858.CD004381.pub2. [DOI] [PubMed] [Google Scholar]
- 32.Laugeson E.A., Frankel F., Gantman A., Dillon A.R., Mogil C. Evidence-based social skills training for adolescents with autism spectrum disorders: the UCLA PEERS program. J. Autism Dev. Disord. 2012;42(6):1025–1036. doi: 10.1007/s10803-011-1339-1. [DOI] [PubMed] [Google Scholar]
- 33.Rao P.A., Beidel D.C., Murray M.J. Social skills interventions for children with Asperger’s syndrome or high-functioning autism: a review and recommendations. J. Autism Dev. Disord. 2008;38(2):353–361. doi: 10.1007/s10803-007-0402-4. [DOI] [PubMed] [Google Scholar]
- 34.Parrott A.C. Human psychopharmacology of Ecstasy (MDMA): a review of 15 years of empirical research. Hum. Psychopharmacol. 2001;16(8):557–577. doi: 10.1002/hup.351. [DOI] [PubMed] [Google Scholar]
- 35.Belzung C., Lemoine M. Criteria of validity for animal models of psychiatric disorders: focus on anxiety disorders and depression. Biol. Mood Anxiety Disord. 2011;1(1):9. doi: 10.1186/2045-5380-1-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Willner P. 1991. [Google Scholar]
- 37.Flint J., Shifman S. Animal models of psychiatric disease. Curr. Opin. Genet. Dev. 2008;18(3):235–240. doi: 10.1016/j.gde.2008.07.002. [DOI] [PubMed] [Google Scholar]
- 38.Theyel B. Animal models in psychiatric disease: A circuit-search approach. Harv. Rev. Psychiatry. 2018;26(5):298–303. doi: 10.1097/HRP.0000000000000193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Edwards S., Koob G.F. Experimental psychiatric illness and drug abuse models: from human to animal, an overview. Methods Mol. Biol. 2012;829:31–48. doi: 10.1007/978-1-61779-458-2_2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Williams S.C.P. New mouse models of autism highlight need for standardized tests. Nat. Med. 2011;17(11):1324–1324. doi: 10.1038/nm1111-1324. [DOI] [PubMed] [Google Scholar]
- 41.Callaway E. Rat models on the rise in autism research. NATNEWS. 2011;23 doi: 10.1038/nature.2011.9415. [DOI] [Google Scholar]
- 42.Ergaz Z., Weinstein-Fudim L., Ornoy A. Genetic and non-genetic animal models for autism spectrum disorders (ASD). Reprod. Toxicol. 2016;64:116–140. doi: 10.1016/j.reprotox.2016.04.024. [DOI] [PubMed] [Google Scholar]
- 43.Kamilar-Britt P., Bedi G. The prosocial effects of 3,4-methylenedioxymethamphetamine (MDMA): Controlled studies in humans and laboratory animals. Neurosci. Biobehav. Rev. 2015;57:433–446. doi: 10.1016/j.neubiorev.2015.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dumont G.J., Sweep F.C., van der Steen R., Hermsen R., Donders A.R., Touw D.J., van Gerven J.M., Buitelaar J.K., Verkes R.J. Increased oxytocin concentrations and prosocial feelings in humans after ecstasy (3,4-methylenedioxymethamphetamine) administration. Soc. Neurosci. 2009;4(4):359–366. doi: 10.1080/17470910802649470. [DOI] [PubMed] [Google Scholar]
- 45.Hysek C.M., Schmid Y., Simmler L.D., Domes G., Heinrichs M., Eisenegger C., Preller K.H., Quednow B.B., Liechti M.E. MDMA enhances emotional empathy and prosocial behavior. Soc. Cogn. Affect. Neurosci. 2014;9(11):1645–1652. doi: 10.1093/scan/nst161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kirkpatrick M., Delton A.W., Robertson T.E., de Wit H. Prosocial effects of MDMA: A measure of generosity. J. Psychopharmacol. 2015;29(6):661–668. doi: 10.1177/0269881115573806. [DOI] [PubMed] [Google Scholar]
- 47.Passie T. The early use of MDMA (‘Ecstasy’) in psychotherapy (1977–1985). Drug Sci. Policy Law. 2018;4:2050324518767442 [Google Scholar]
- 48.Danforth A.L., Grob C.S., Struble C., Feduccia A.A., Walker N., Jerome L., Yazar-Klosinski B., Emerson A. Reduction in social anxiety after MDMA-assisted psychotherapy with autistic adults: a randomized, double-blind, placebo-controlled pilot study. Psychopharmacology (Berl.) 2018;235(11):3137–3148. doi: 10.1007/s00213-018-5010-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.ter Bogt M. T.F.; Engels, R.C. “Partying” hard: party style, motives for and effects of MDMA use at rave parties. Subst. Use Misuse. 2005;40(9-10):1479–1502. doi: 10.1081/JA-200066822. [DOI] [PubMed] [Google Scholar]
- 50.Kalant H. The pharmacology and toxicology of “ecstasy” (MDMA) and related drugs. CMAJ. 2001;165(7):917–928. [PMC free article] [PubMed] [Google Scholar]
- 51.Hysek C.M., Domes G., Liechti M.E. MDMA enhances “mind reading” of positive emotions and impairs “mind reading” of negative emotions. Psychopharmacology (Berl.) 2012;222(2):293–302. doi: 10.1007/s00213-012-2645-9. [DOI] [PubMed] [Google Scholar]
- 52.Thompson M.R., Callaghan P.D., Hunt G.E., Cornish J.L., McGregor I.S. A role for oxytocin and 5-HT(1A) receptors in the prosocial effects of 3,4 methylenedioxymethamphetamine (“ecstasy”). Neuroscience. 2007;146(2):509–514. doi: 10.1016/j.neuroscience.2007.02.032. [DOI] [PubMed] [Google Scholar]
- 53.Verrico C.D., Miller G.M., Madras B.K. MDMA (Ecstasy) and human dopamine, norepinephrine, and serotonin transporters: implications for MDMA-induced neurotoxicity and treatment. Psychopharmacology (Berl.) 2007;189(4):489–503. doi: 10.1007/s00213-005-0174-5. [DOI] [PubMed] [Google Scholar]
- 54.Van de Kar L.D. Neuroendocrine pharmacology of serotonergic (5-HT) neurons. Annu. Rev. Pharmacol. Toxicol. 1991;31(1):289–320. doi: 10.1146/annurev.pa.31.040191.001445. [DOI] [PubMed] [Google Scholar]
- 55.Liechti M.E., Vollenweider F.X. Which neuroreceptors mediate the subjective effects of MDMA in humans? A summary of mechanistic studies. Hum. Psychopharmacol. 2001;16(8):589–598. doi: 10.1002/hup.348. [DOI] [PubMed] [Google Scholar]
- 56.Jørgensen H., Riis M., Knigge U., Kjaer A., Warberg J. Serotonin receptors involved in vasopressin and oxytocin secretion. J. Neuroendocrinol. 2003;15(3):242–249. doi: 10.1046/j.1365-2826.2003.00978.x. [DOI] [PubMed] [Google Scholar]
- 57.Meyer-Lindenberg A. 2008. [Google Scholar]
- 58.Tost H., Kolachana B., Hakimi S., Lemaitre H., Verchinski B.A., Mattay V.S., Weinberger D.R., Meyer-Lindenberg A. A common allele in the oxytocin receptor gene (OXTR) impacts prosocial temperament and human hypothalamic-limbic structure and function. Proc. Natl. Acad. Sci. USA. 2010;107(31):13936–13941. doi: 10.1073/pnas.1003296107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Wu S., Jia M., Ruan Y., Liu J., Guo Y., Shuang M., Gong X., Zhang Y., Yang X., Zhang D. Positive association of the oxytocin receptor gene (OXTR) with autism in the Chinese Han population. Biol. Psychiatry. 2005;58(1):74–77. doi: 10.1016/j.biopsych.2005.03.013. [DOI] [PubMed] [Google Scholar]
- 60.Ylisaukko-oja T., Alarcón M., Cantor R.M., Auranen M., Vanhala R., Kempas E., von Wendt L., Järvelä I., Geschwind D.H., Peltonen L. Search for autism loci by combined analysis of autism genetic resource exchange and finnish families. Ann. Neurol. 2006;59(1):145–155. doi: 10.1002/ana.20722. [DOI] [PubMed] [Google Scholar]
- 61.Jacob S., Brune C.W., Carter C.S., Leventhal B.L., Lord C., Cook E.H., Jr Association of the oxytocin receptor gene (OXTR) in Caucasian children and adolescents with autism. Neurosci. Lett. 2007;417(1):6–9. doi: 10.1016/j.neulet.2007.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Carhart-Harris R.L., Murphy K., Leech R., Erritzoe D., Wall M.B., Ferguson B., Williams L.T., Roseman L., Brugger S., De Meer I., Tanner M., Tyacke R., Wolff K., Sethi A., Bloomfield M.A., Williams T.M., Bolstridge M., Stewart L., Morgan C., Newbould R.D., Feilding A., Curran H.V., Nutt D.J. The effects of acutely administered 3,4-methylenedioxymethamphetamine on spontaneous brain function in healthy volunteers measured with arterial spin labeling and blood oxygen level-dependent resting state functional connectivity. Biol. Psychiatry. 2015;78(8):554–562. doi: 10.1016/j.biopsych.2013.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Gamma A., Buck A., Berthold T., Liechti M.E., Vollenweider F.X. 3,4-Methylenedioxymethamphetamine (MDMA) modulates cortical and limbic brain activity as measured by [H(2)(15)O]-PET in healthy humans. Neuropsychopharmacology. 2000;23(4):388–395. doi: 10.1016/S0893-133X(00)00130-5. [DOI] [PubMed] [Google Scholar]
- 64.Bedi G., Phan K.L., Angstadt M., de Wit H. Effects of MDMA on sociability and neural response to social threat and social reward. Psychopharmacology (Berl.) 2009;207(1):73–83. doi: 10.1007/s00213-009-1635-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Nardou R., Lewis E.M., Rothhaas R., Xu R., Yang A., Boyden E., Dölen G. Oxytocin-dependent reopening of a social reward learning critical period with MDMA. Nature. 2019;569(7754):116–120. doi: 10.1038/s41586-019-1075-9. [DOI] [PubMed] [Google Scholar]
- 66.Rozas C., Loyola S., Ugarte G., Zeise M.L., Reyes-Parada M., Pancetti F., Rojas P., Morales B. Acutely applied MDMA enhances long-term potentiation in rat hippocampus involving D1/D5 and 5-HT2 receptors through a polysynaptic mechanism. Eur. Neuropsychopharmacol. 2012;22(8):584–595. doi: 10.1016/j.euroneuro.2011.11.010. [DOI] [PubMed] [Google Scholar]
- 67.Rozas C., Encina M., Reyes-Parada M., Pancetti F., Cassels B., Morales B. Effects of MDMA on the induction of synaptic plasticity in visual cortex and hippocampus. J Physiol P; 2005. p. 565. [Google Scholar]
- 68.Sessa B., Higbed L., Nutt D. A review of 3, 4-methylenedioxymethamphetamine (MDMA)-assisted psychotherapy. Front. Psychiatry. 2019;10:138. doi: 10.3389/fpsyt.2019.00138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Cornish J.L., Shahnawaz Z., Thompson M.R., Wong S., Morley K.C., Hunt G.E., McGregor I.S. Heat increases 3,4-methylenedioxymethamphetamine self-administration and social effects in rats. Eur. J. Pharmacol. 2003;482(1-3):339–341. doi: 10.1016/j.ejphar.2003.09.060. [DOI] [PubMed] [Google Scholar]
- 70.Mignini L.E., Khan K.S. Methodological quality of systematic reviews of animal studies: a survey of reviews of basic research. BMC Med. Res. Methodol. 2006;6(1):10. doi: 10.1186/1471-2288-6-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Crawley J.N. Mouse behavioral assays relevant to the symptoms of autism. Brain Pathol. 2007;17(4):448–459. doi: 10.1111/j.1750-3639.2007.00096.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Moy S.S., Nadler J.J., Perez A., Barbaro R.P., Johns J.M., Magnuson T.R., Piven J., Crawley J.N. Sociability and preference for social novelty in five inbred strains: an approach to assess autistic-like behavior in mice. Genes Brain Behav. 2004;3(5):287–302. doi: 10.1111/j.1601-1848.2004.00076.x. [DOI] [PubMed] [Google Scholar]
- 73.File S.E., Seth P. A review of 25 years of the social interaction test. Eur. J. Pharmacol. 2003;463(1-3):35–53. doi: 10.1016/S0014-2999(03)01273-1. [DOI] [PubMed] [Google Scholar]
- 74.Scattoni M.L., Gandhy S.U., Ricceri L., Crawley J.N. Unusual repertoire of vocalizations in the BTBR T+tf/J mouse model of autism. PLoS One. 2008;3(8):e3067. doi: 10.1371/journal.pone.0003067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Branchi I., Santucci D., Alleva E. Ultrasonic vocalisation emitted by infant rodents: a tool for assessment of neurobehavioural development. Behav. Brain Res. 2001;125(1-2):49–56. doi: 10.1016/S0166-4328(01)00277-7. [DOI] [PubMed] [Google Scholar]
- 76.Winslow J.T., Insel T.R. Serotonergic modulation of rat pup ultrasonic vocal development: studies with 3,4-methylenedioxymethamphetamine. J. Pharmacol. Exp. Ther. 1990;254(1):212–220. [PubMed] [Google Scholar]
- 77.McGregor I.S., Gurtman C.G., Morley K.C., Clemens K.J., Blokland A., Li K.M., Cornish J.L., Hunt G.E. Increased anxiety and “depressive” symptoms months after MDMA (“ecstasy”) in rats: drug-induced hyperthermia does not predict long-term outcomes. Psychopharmacology (Berl.) 2003;168(4):465–474. doi: 10.1007/s00213-003-1452-8. [DOI] [PubMed] [Google Scholar]
- 78.Daza-Losada M., Rodríguez-Arias M., Maldonado C., Aguilar M.A., Miñarro J. Behavioural and neurotoxic long-lasting effects of MDMA plus cocaine in adolescent mice. Eur. J. Pharmacol. 2008;590(1-3):204–211. doi: 10.1016/j.ejphar.2008.06.025. [DOI] [PubMed] [Google Scholar]
- 79.Machalova A., Slais K., Vrskova D., Sulcova A. Differential effects of modafinil, methamphetamine, and MDMA on agonistic behavior in male mice. Pharmacol. Biochem. Behav. 2012;102(2):215–223. doi: 10.1016/j.pbb.2012.04.013. [DOI] [PubMed] [Google Scholar]
- 80.Wallinga A.E., ten Voorde A.M., de Boer S.F., Koolhaas J.M., Buwalda B. MDMA-induced serotonergic neurotoxicity enhances aggressiveness in low- but not high-aggressive rats. Eur. J. Pharmacol. 2009;618(1-3):22–27. doi: 10.1016/j.ejphar.2009.07.006. [DOI] [PubMed] [Google Scholar]
- 81.Morley K.C., Arnold J.C., McGregor I.S. Serotonin (1A) receptor involvement in acute 3,4-methylenedioxymethamphetamine (MDMA) facilitation of social interaction in the rat. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2005;29(5):648–657. doi: 10.1016/j.pnpbp.2005.04.009. [DOI] [PubMed] [Google Scholar]
- 82.Thompson M.R., Hunt G.E., McGregor I.S. Neural correlates of MDMA (“Ecstasy”)-induced social interaction in rats. Soc. Neurosci. 2009;4(1):60–72. doi: 10.1080/17470910802045042. [DOI] [PubMed] [Google Scholar]
- 83.Thompson M.R., Callaghan P.D., Hunt G.E., McGregor I.S. Reduced sensitivity to MDMA-induced facilitation of social behaviour in MDMA pre-exposed rats. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2008;32(4):1013–1021. doi: 10.1016/j.pnpbp.2008.01.014. [DOI] [PubMed] [Google Scholar]
- 84.Cagiano R., Bera I., Sabatini R., Flace P., Vermesan D., Vermesan H., Dragulescu S.I., Bottalico L., Santacroce L. Effects on rat sexual behaviour of acute MDMA (ecstasy) alone or in combination with loud music. Eur. Rev. Med. Pharmacol. Sci. 2008;12(5):285–292. [PubMed] [Google Scholar]
- 85.Procópio-Souza R., Fukushiro D.F., Trombin T.F., Wuo-Silva R., Zanlorenci L.H., Lima A.J., Ribeiro L.T., Corrêa J.M., Marinho E.A., Kameda S.R., Andersen M.L., Tufik S., Frussa-Filho R. Effects of group exposure on single injection-induced behavioral sensitization to drugs of abuse in mice. Drug Alcohol Depend. 2011;118(2-3):349–359. doi: 10.1016/j.drugalcdep.2011.04.017. [DOI] [PubMed] [Google Scholar]
- 86.Kirkpatrick M.G., de Wit H. MDMA: a social drug in a social context. Psychopharmacology (Berl.) 2015;232(6):1155–1163. doi: 10.1007/s00213-014-3752-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Ando R.D., Benko A., Ferrington L., Kirilly E., Kelly P.A.T., Bagdy G. Partial lesion of the serotonergic system by a single dose of MDMA results in behavioural disinhibition and enhances acute MDMA-induced social behaviour on the social interaction test. Neuropharmacology. 2006;50(7):884–896. doi: 10.1016/j.neuropharm.2005.12.010. [DOI] [PubMed] [Google Scholar]
- 88.Maldonado E., Navarro J.F. MDMA (“ecstasy”) exhibits an anxiogenic-like activity in social encounters between male mice. Pharmacol. Res. 2001;44(1):27–31. doi: 10.1006/phrs.2001.0824. [DOI] [PubMed] [Google Scholar]
- 89.Navarro J.F., Maldonado E. Behavioral profile of 3,4-methylenedioxy-methamphetamine (MDMA) in agonistic encounters between male mice. Prog. Neuropsychopharmacol. Biol. Psychiatry. 1999;23(2):327–334. doi: 10.1016/S0278-5846(98)00100-6. [DOI] [PubMed] [Google Scholar]
- 90.Navarro J.F., Maldonado E. Effects of Acute, Subchronic and Intermittent MDMA (‘ECSTASY’) administration on agonistic interactions between male mice. Aggress. Behav. 2004;30(1):71–83. doi: 10.1002/ab.20004. [DOI] [Google Scholar]
- 91.Morley K.C., Gallate J.E., Hunt G.E., Mallet P.E., McGregor I.S. Increased anxiety and impaired memory in rats 3 months after administration of 3,4-methylenedioxymethamphetamine (“ecstasy”). Eur. J. Pharmacol. 2001;433(1):91–99. doi: 10.1016/S0014-2999(01)01512-6. [DOI] [PubMed] [Google Scholar]
- 92.McGregor I.S., Clemens K.J., Van der Plasse G., Li K.M., Hunt G.E., Chen F., Lawrence A.J. Increased anxiety 3 months after brief exposure to MDMA (“Ecstasy”) in rats: association with altered 5-HT transporter and receptor density. Neuropsychopharmacology. 2003;28(8):1472–1484. doi: 10.1038/sj.npp.1300185. [DOI] [PubMed] [Google Scholar]
- 93.García-Pardo M.P., Blanco-Gandía M.C., Valiente-Lluch M., Rodríguez-Arias M., Miñarro J., Aguilar M.A. Long-term effects of repeated social stress on the conditioned place preference induced by MDMA in mice. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2015;63:98–109. doi: 10.1016/j.pnpbp.2015.06.006. [DOI] [PubMed] [Google Scholar]
- 94.García-Pardo M.P., Roger-Sánchez C., Rodríguez-Arias M., Miñarro J., Aguilar M.A. Cognitive and behavioural effects induced by social stress plus MDMA administration in mice. Behav. Brain Res. 2017;319:63–72. doi: 10.1016/j.bbr.2016.11.012. [DOI] [PubMed] [Google Scholar]
- 95.Homberg J.R., Schiepers O.J.G., Schoffelmeer A.N.M., Cuppen E., Vanderschuren L.J.M.J. Acute and constitutive increases in central serotonin levels reduce social play behaviour in peri-adolescent rats. Psychopharmacology (Berl.) 2007;195(2):175–182. doi: 10.1007/s00213-007-0895-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Navarro J.F., Rivera A., Maldonado E., Cavas M., de la Calle A. Anxiogenic-like activity of 3,4-methylenedioxy-methamphetamine (“Ecstasy”) in the social interaction test is accompanied by an increase of c-fos expression in mice amygdala. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2004;28(2):249–254. doi: 10.1016/j.pnpbp.2003.10.016. [DOI] [PubMed] [Google Scholar]
- 97.Slamberova R., Mikulecka A., Macuchova E., Hrebickova I., Sevcikova M., Nohejlova K., Pometlova M. Effects of psychostimulants on social interaction in adult male rats. 2015. [DOI] [PubMed]
- 98.Ballesta S., Reymond G., Pozzobon M., Duhamel J-R. Effects of MDMA injections on the behavior of socially-housed long-tailed macaques (Macaca fascicularis). PLoS One. 2016;11(2):e0147136. doi: 10.1371/journal.pone.0147136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Iravani M.M., Jackson M.J., Kuoppamäki M., Smith L.A., Jenner P. 3,4-methylenedioxymethamphetamine (ecstasy) inhibits dyskinesia expression and normalizes motor activity in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-treated primates. J. Neurosci. 2003;23(27):9107–9115. doi: 10.1523/JNEUROSCI.23-27-09107.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Pitts E.G., Minerva A.R., Chandler E.B., Kohn J.N., Logun M.T., Sulima A., Rice K.C., Howell L.L. 3,4-Methylenedioxymethamphetamine increases affiliative behaviors in squirrel monkeys in a serotonin 2a receptor-dependent manner. Neuropsychopharmacology. 2017;42(10):1962–1971. doi: 10.1038/npp.2017.80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Verrico C.D., Lynch L., Fahey M.A., Fryer A.K., Miller G.M., Madras B.K. MDMA-induced impairment in primates: antagonism by a selective norepinephrine or serotonin, but not by a dopamine/norepinephrine transport inhibitor. J. Psychopharmacol. 2008;22(2):187–202. doi: 10.1177/0269881107083639. [DOI] [PubMed] [Google Scholar]
- 102.Ali S.F., Newport G.D., Scallet A.C., Binienda Z., Ferguson S.A., Bailey J.R., Paule M.G., Slikker W. Jr Oral administration of 3,4-methylenedioxymethamphetamine (MDMA) produces selective serotonergic depletion in the nonhuman primate. Neurotoxicol. Teratol. 1993;15(2):91–96. doi: 10.1016/0892-0362(93)90067-X. [DOI] [PubMed] [Google Scholar]
- 103.Capurro A., Reyes-Parada M., Olazabal D., Perrone R., Silveira R., Macadar O. Aggressive behavior and jamming avoidance response in the weakly electric fish Gymnotus carapo: Effects of 3,4-Methylenedioxymethamphetamine (MDMA). Comparative Biochemistry and Physiology -. Physiology (Bethesda) 1997;118(3):831–840. [Google Scholar]
- 104.Green J., Collins C., Kyzar E.J., Pham M., Roth A., Gaikwad S., Cachat J., Stewart A.M., Landsman S., Grieco F., Tegelenbosch R., Noldus L.P., Kalueff A.V. Automated high-throughput neurophenotyping of zebrafish social behavior. J. Neurosci. Methods. 2012;210(2):266–271. doi: 10.1016/j.jneumeth.2012.07.017. [DOI] [PubMed] [Google Scholar]
- 105.Ponzoni L., Sala M., Braida D. Ritanserin-sensitive receptors modulate the prosocial and the anxiolytic effect of MDMA derivatives, DOB and PMA, in zebrafish. Behav. Brain Res. 2016;314:181–189. doi: 10.1016/j.bbr.2016.08.009. [DOI] [PubMed] [Google Scholar]
- 106.Ponzoni L., Braida D., Bondiolotti G., Sala M. The Non-Peptide Arginine-Vasopressin v1a Selective Receptor Antagonist, SR49059, Blocks the Rewarding, Prosocial, and Anxiolytic Effects of 3,4-Methylenedioxymethamphetamine and Its Derivatives in Zebra Fish. Front. Psychiatry. 2017;8:146. doi: 10.3389/fpsyt.2017.00146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Stewart A., Riehl R., Wong K., Green J., Cosgrove J., Vollmer K., Kyzar E., Hart P., Allain A., Cachat J., Gaikwad S., Hook M., Rhymes K., Newman A., Utterback E., Chang K., Kalueff A.V. Behavioral effects of MDMA (‘ecstasy’) on adult zebrafish. Behav. Pharmacol. 2011;22(3):275–280. doi: 10.1097/FBP.0b013e328345f758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Egan R.J., Bergner C.L., Hart P.C., Cachat J.M., Canavello P.R., Elegante M.F., Elkhayat S.I., Bartels B.K., Tien A.K., Tien D.H., Mohnot S., Beeson E., Glasgow E., Amri H., Zukowska Z., Kalueff A.V. Understanding behavioral and physiological phenotypes of stress and anxiety in zebrafish. Behav. Brain Res. 2009;205(1):38–44. doi: 10.1016/j.bbr.2009.06.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Stewart A., Kadri F., DiLeo J., Min Chung K., Cachat J., Goodspeed J., Suciu C., Roy S., Gaikwad S., Wong K. The developing utility of zebrafish in modeling neurobehavioral disorders. Int. J. Comp. Psychol. 2010;23(1) [Google Scholar]
- 110.Speedie N., Gerlai R. Alarm substance induced behavioral responses in zebrafish (Danio rerio). Behav. Brain Res. 2008;188(1):168–177. doi: 10.1016/j.bbr.2007.10.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Edsinger E., Dolen G. SLC6A4 binding site and acute prosocial effects of (+/-)-3,4-methylendioxymethamphetamine (MDMA) are evolutionarily conserved in Octopus bimaculoides. Cold Spring Harbor: Cold Spring Harbor Laboratory Press; 2018. [Google Scholar]
- 112.Edsinger E., Dölen G. A Conserved Role for Serotonergic Neurotransmission in Mediating Social Behavior in Octopus. Curr. Biol. 2018;28(19):3136–3142.e4. doi: 10.1016/j.cub.2018.07.061. [DOI] [PubMed] [Google Scholar]
- 113.Tancer M., Johanson C.E. Reinforcing, subjective, and physiological effects of MDMA in humans: a comparison with d-amphetamine and mCPP. Drug Alcohol Depend. 2003;72(1):33–44. doi: 10.1016/S0376-8716(03)00172-8. [DOI] [PubMed] [Google Scholar]
- 114.Corey V.R., Pisano V.D., Halpern J.H. Effects of 3,4-Methylenedioxymethamphetamine on Patient Utterances in a Psychotherapeutic Setting. J. Nerv. Ment. Dis. 2016;204(7):519–523. doi: 10.1097/NMD.0000000000000499. [DOI] [PubMed] [Google Scholar]
- 115.Liechti M.E., Gamma A., Vollenweider F.X. Gender differences in the subjective effects of MDMA. Psychopharmacology (Berl.) 2001;154(2):161–168. doi: 10.1007/s002130000648. [DOI] [PubMed] [Google Scholar]
- 116.Wardle M.C., de Wit H. MDMA alters emotional processing and facilitates positive social interaction. Psychopharmacology (Berl.) 2014;231(21):4219–4229. doi: 10.1007/s00213-014-3570-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Palenicek T., Votava M., Bubenikova V., Horacek J. Increased sensitivity to the acute effects of MDMA (“ecstasy”) in female rats. Physiol. Behav. 2005;86(4):546–553. doi: 10.1016/j.physbeh.2005.08.043. [DOI] [PubMed] [Google Scholar]
- 118.Thomas A., Burant A., Bui N., Graham D., Yuva-Paylor L.A., Paylor R. Marble burying reflects a repetitive and perseverative behavior more than novelty-induced anxiety. Psychopharmacology (Berl.) 2009;204(2):361–373. doi: 10.1007/s00213-009-1466-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Angoa-Pérez M., Kane M.J., Briggs D.I., Francescutti D.M., Kuhn D.M. Marble burying and nestlet shredding as tests of repetitive, compulsive-like behaviors in mice. JoVE. 2013;82:e50978. doi: 10.3791/50978. [Journal of Visualized Experiments]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Crean R.D., Davis S.A., Von Huben S.N., Lay C.C., Katner S.N., Taffe M.A. Effects of (+/-)3,4-methylenedioxymethamphetamine, (+/-)3,4-methylenedioxyamphetamine and methamphetamine on temperature and activity in rhesus macaques. Neuroscience. 2006;142(2):515–525. doi: 10.1016/j.neuroscience.2006.06.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Crean R.D., Davis S.A., Taffe M.A. Oral administration of (+/-) 3,4-methylenedioxymethamphetamine and (+)methamphetamine alters temperature and activity in rhesus macaques. Pharmacol. Biochem. Behav. 2007;87(1):11–19. doi: 10.1016/j.pbb.2007.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Goodwin A.K., Mueller M., Shell C.D., Ricaurte G.A., Ator N.A. Behavioral effects and pharmacokinetics of (±)-3,4-methylenedioxymethamphetamine (MDMA, Ecstasy) after intragastric administration to baboons. J. Pharmacol. Exp. Ther. 2013;345(3):342–353. doi: 10.1124/jpet.113.203729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Vollenweider F.X., Liechti M.E., Paulus M.P. MDMA affects both error-rate dependent and independent aspects of decision-making in a two-choice prediction task. J. Psychopharmacol. 2005;19(4):366–374. doi: 10.1177/0269881105053287. [DOI] [PubMed] [Google Scholar]
- 124.Deacon R.M., Rawlins J.N.P. T-maze alternation in the rodent. Nat. Protoc. 2006;1(1):7–12. doi: 10.1038/nprot.2006.2. [DOI] [PubMed] [Google Scholar]
- 125.Mahmood U., Ahn S., Yang E-J., Choi M., Kim H., Regan P., Cho K., Kim H-S. Dendritic spine anomalies and PTEN alterations in a mouse model of VPA-induced autism spectrum disorder. Pharmacol. Res. 2018;128:110–121. doi: 10.1016/j.phrs.2017.08.006. [DOI] [PubMed] [Google Scholar]
- 126.Shulgin A.T. The background and chemistry of MDMA. J. Psychoactive Drugs. 1986;18(4):291–304. doi: 10.1080/02791072.1986.10472361. [DOI] [PubMed] [Google Scholar]
- 127.van Wel J.H.P., Kuypers K.P.C., Theunissen E.L., Bosker W.M., Bakker K., Ramaekers J.G. Effects of acute MDMA intoxication on mood and impulsivity: role of the 5-HT2 and 5-HT1 receptors. PLoS One. 2012;7(7):e40187. doi: 10.1371/journal.pone.0040187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Patterson P.H. Modeling autistic features in animals. Pediatr. Res. 2011;69(5 Pt 2):34R–40R. doi: 10.1203/PDR.0b013e318212b80f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Danforth A.L., Struble C.M., Yazar-Klosinski B., Grob C.S. MDMA-assisted therapy: A new treatment model for social anxiety in autistic adults. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2016;64:237–249. doi: 10.1016/j.pnpbp.2015.03.011. [DOI] [PubMed] [Google Scholar]
- 130.Danforth A.L. Embracing neurodiversity in psychedelic science: A mixed-methods inquiry into the MDMA experiences of autistic adults. J. Psychoactive Drugs. 2019;51(2):146–154. doi: 10.1080/02791072.2019.1587116. [DOI] [PubMed] [Google Scholar]
- 131.Fone K.C., Beckett S.R., Topham I.A., Swettenham J., Ball M., Maddocks L. Long-term changes in social interaction and reward following repeated MDMA administration to adolescent rats without accompanying serotonergic neurotoxicity. Psychopharmacology (Berl.) 2002;159(4):437–444. doi: 10.1007/s00213-001-0931-z. [DOI] [PubMed] [Google Scholar]
- 132.Bull E.J., Hutson P.H., Fone K.C. Decreased social behaviour following 3,4-methylenedioxymethamphetamine (MDMA) is accompanied by changes in 5-HT2A receptor responsivity. Neuropharmacology. 2004;46(2):202–210. doi: 10.1016/j.neuropharm.2003.08.004. [DOI] [PubMed] [Google Scholar]
- 133.Bull E.J., Hutson P.H., Fone K.C. Reduced social interaction following 3,4-methylenedioxymethamphetamine is not associated with enhanced 5-HT 2C receptor responsivity. Neuropharmacology. 2003;44(4):439–448. doi: 10.1016/S0028-3908(02)00407-0. [DOI] [PubMed] [Google Scholar]
- 134.Gurtman C.G., Morley K.C., Li K.M., Hunt G.E., McGregor I.S. Increased anxiety in rats after 3,4-methylenedioxymethamphetamine: association with serotonin depletion. Eur. J. Pharmacol. 2002;446(1-3):89–96. doi: 10.1016/S0014-2999(02)01820-4. [DOI] [PubMed] [Google Scholar]
- 135.Welsh J.P., Ahn E.S., Placantonakis D.G. Is autism due to brain desynchronization? Int. J. Dev. Neurosci. 2005;23(2-3):253–263. doi: 10.1016/j.ijdevneu.2004.09.002. [DOI] [PubMed] [Google Scholar]
- 136.Clemens K.J., Cornish J.L., Hunt G.E., McGregor I.S. Repeated weekly exposure to MDMA, methamphetamine or their combination: long-term behavioural and neurochemical effects in rats. Drug Alcohol Depend. 2007;86(2-3):183–190. doi: 10.1016/j.drugalcdep.2006.06.004. [DOI] [PubMed] [Google Scholar]
- 137.File S.E., Hyde J.R. Can social interaction be used to measure anxiety? Br. J. Pharmacol. 1978;62(1):19–24. doi: 10.1111/j.1476-5381.1978.tb07001.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Kang J., Kim E. Suppression of NMDA receptor function in mice prenatally exposed to valproic acid improves social deficits and repetitive behaviors. Front. Mol. Neurosci. 2015;8:17. doi: 10.3389/fnmol.2015.00017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Riem M.M.E., Bakermans-Kranenburg M.J., Huffmeijer R., van Ijzendoorn M.H. Does intranasal oxytocin promote prosocial behavior to an excluded fellow player? A randomized-controlled trial with Cyberball. Psychoneuroendocrinology. 2013;38(8):1418–1425. doi: 10.1016/j.psyneuen.2012.12.023. [DOI] [PubMed] [Google Scholar]
- 140.Anagnostou E., Soorya L., Chaplin W., Bartz J., Halpern D., Wasserman S., Wang A.T., Pepa L., Tanel N., Kushki A., Hollander E. Intranasal oxytocin versus placebo in the treatment of adults with autism spectrum disorders: a randomized controlled trial. Mol. Autism. 2012;3(1):16. doi: 10.1186/2040-2392-3-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Leng G., Ludwig M. Intranasal oxytocin: myths and delusions. Biol. Psychiatry. 2016;79(3):243–250. doi: 10.1016/j.biopsych.2015.05.003. [DOI] [PubMed] [Google Scholar]
- 142.Guastella A.J., Gray K.M., Rinehart N.J., Alvares G.A., Tonge B.J., Hickie I.B., Keating C.M., Cacciotti-Saija C., Einfeld S.L. The effects of a course of intranasal oxytocin on social behaviors in youth diagnosed with autism spectrum disorders: a randomized controlled trial. J. Child Psychol. Psychiatry. 2015;56(4):444–452. doi: 10.1111/jcpp.12305. [DOI] [PubMed] [Google Scholar]
- 143.Tabak B.A., Meyer M.L., Castle E., Dutcher J.M., Irwin M.R., Han J.H., Lieberman M.D., Eisenberger N.I. Vasopressin, but not oxytocin, increases empathic concern among individuals who received higher levels of paternal warmth: A randomized controlled trial. Psychoneuroendocrinology. 2015;51:253–261. doi: 10.1016/j.psyneuen.2014.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Dickerson S.M., Walker D.M., Reveron M.E., Duvauchelle C.L., Gore A.C. The recreational drug ecstasy disrupts the hypothalamic-pituitary-gonadal reproductive axis in adult male rats. Neuroendocrinology. 2008;88(2):95–102. doi: 10.1159/000119691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Frye C.G., Wardle M.C., Norman G.J., de Wit H. MDMA decreases the effects of simulated social rejection. Pharmacol. Biochem. Behav. 2014;117:1–6. doi: 10.1016/j.pbb.2013.11.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Gabay A.S., Carhart-Harris R.L., Mazibuko N., Kempton M.J., Morrison P.D., Nutt D.J., Mehta M.A. Psilocybin and MDMA reduce costly punishment in the Ultimatum Game. Sci. Rep. 2018;8(1):8236. doi: 10.1038/s41598-018-26656-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Gabay A.S., Kempton M.J., Gilleen J., Mehta M.A. MDMA increases cooperation and recruitment of social brain areas when playing trustworthy players in an Iterated Prisoner’s Dilemma. J. Neurosci. 2019;39(2):307–320. doi: 10.1523/JNEUROSCI.1276-18.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Clark C.M., Frye C.G., Wardle M.C., Norman G.J., de Wit H. Acute effects of MDMA on autonomic cardiac activity and their relation to subjective prosocial and stimulant effects. Psychophysiology. 2015;52(3):429–435. doi: 10.1111/psyp.12327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Liechti M.E., Saur M.R., Gamma A., Hell D., Vollenweider F.X. Psychological and physiological effects of MDMA (“Ecstasy”) after pretreatment with the 5-HT(2) antagonist ketanserin in healthy humans. Neuropsychopharmacology. 2000;23(4):396–404. doi: 10.1016/S0893-133X(00)00126-3. [DOI] [PubMed] [Google Scholar]
- 150.Schmid Y., Hysek C.M., Simmler L.D., Crockett M.J., Quednow B.B., Liechti M.E. Differential effects of MDMA and methylphenidate on social cognition. J. Psychopharmacol. 2014;28(9):847–856. doi: 10.1177/0269881114542454. [DOI] [PubMed] [Google Scholar]
- 151.Baron-Cohen S. 2000.
- 152.Baron-Cohen S., Wheelwright S. The empathy quotient: an investigation of adults with Asperger syndrome or high functioning autism, and normal sex differences. J. Autism Dev. Disord. 2004;34(2):163–175. doi: 10.1023/B:JADD.0000022607.19833.00. [DOI] [PubMed] [Google Scholar]
- 153.Kuypers K.P.C., de la Torre R., Farre M., Pizarro N., Xicota L., Ramaekers J.G. MDMA-induced indifference to negative sounds is mediated by the 5-HT2A receptor. Psychopharmacology (Berl.) 2018;235(2):481–490. doi: 10.1007/s00213-017-4699-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Kuypers K.P.C., de la Torre R., Farre M., Yubero-Lahoz S., Dziobek I., Van den Bos W., Ramaekers J.G. No evidence that MDMA-induced enhancement of emotional empathy is related to peripheral oxytocin levels or 5-HT1a receptor activation. PLoS One. 2014;9(6):e100719. doi: 10.1371/journal.pone.0100719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Kuypers K.P.C., Dolder P.C., Ramaekers J.G., Liechti M.E. Multifaceted empathy of healthy volunteers after single doses of MDMA: A pooled sample of placebo-controlled studies. J. Psychopharmacol. 2017;31(5):589–598. doi: 10.1177/0269881117699617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Vizeli P., Meyer Zu Schwabedissen H.E., Liechti M.E. Role of serotonin transporter and receptor gene variations in the acute effects of MDMA in healthy subjects. ACS Chem. Neurosci. 2018;10(7):3120–3131. doi: 10.1021/acschemneuro.8b00590. [DOI] [PubMed] [Google Scholar]
- 157.Kolyaduke O.V., Hughes R.N. Increased anxiety-related behavior in male and female adult rats following early and late adolescent exposure to 3,4-methylenedioxymethamphetamine (MDMA). Pharmacol. Biochem. Behav. 2013;103(4):742–749. doi: 10.1016/j.pbb.2012.12.004. [DOI] [PubMed] [Google Scholar]
- 158.Compton D.M., Luetzenberg F.S., Watkins E. Tripping the light fantastic: Modeling the consequences of recreational use of MDMA or 5-MeO-DIPT in humans using weekend “rave” exposures in rat. Psychol. Neurosci. 2016;9(1):105–124. doi: 10.1037/pne0000043. [DOI] [Google Scholar]
- 159.Canales J.J., Ferrer-Donato A. Prenatal exposure to alcohol and 3,4-methylenedioxymethamphetamine (ecstasy) alters adult hippocampal neurogenesis and causes enduring memory deficits. Dev. Neurosci. 2014;36(1):10–17. doi: 10.1159/000356820. [DOI] [PubMed] [Google Scholar]
- 160.Ben-Ami Bartal I., Decety J., Mason P. Empathy and pro-social behavior in rats. Science. 2011;334(6061):1427–1430. doi: 10.1126/science.1210789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Reynolds S., Millette A., Devine D.P. Sensory and motor characterization in the postnatal valproate rat model of autism. Dev. Neurosci. 2012;34(2-3):258–267. doi: 10.1159/000336646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Kerns C.M., Kendall P.C. The presentation and classification of anxiety in autism spectrum disorder. Clin. Psychol. Sci. Pract. 2012;19(4):323–347. doi: 10.1111/cpsp.12009. [DOI] [Google Scholar]
- 163.Rodgers R.J., Johnson N.J.T. Factor analysis of spatiotemporal and ethological measures in the murine elevated plus-maze test of anxiety. Pharmacol. Biochem. Behav. 1995;52(2):297–303. doi: 10.1016/0091-3057(95)00138-M. [DOI] [PubMed] [Google Scholar]
- 164.Mannion A., Leader G., Healy O. An investigation of comorbid psychological disorders, sleep problems, gastrointestinal symptoms and epilepsy in children and adolescents with Autism Spectrum Disorder. Res. Autism Spectr. Disord. 2013;7(1):35–42. doi: 10.1016/j.rasd.2012.05.002. [DOI] [Google Scholar]
- 165.McCann U.D., Slate S.O., Ricaurte G.A. Adverse reactions with 3,4-methylenedioxymethamphetamine (MDMA; ‘ecstasy’). Drug Saf. 1996;15(2):107–115. doi: 10.2165/00002018-199615020-00003. [DOI] [PubMed] [Google Scholar]
- 166.O’Hearn E., Battaglia G., De Souza E.B., Kuhar M.J., Molliver M.E. Methylenedioxyamphetamine (MDA) and methylenedioxymethamphetamine (MDMA) cause selective ablation of serotonergic axon terminals in forebrain: immunocytochemical evidence for neurotoxicity. J. Neurosci. 1988;8(8):2788–2803. doi: 10.1523/JNEUROSCI.08-08-02788.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Ricaurte G.A., McCann U.D., Szabo Z., Scheffel U. Toxicodynamics and long-term toxicity of the recreational drug, 3, 4-methylenedioxymethamphetamine (MDMA, ‘Ecstasy’). Toxicol. Lett. 2000;112-113:143–146. doi: 10.1016/S0378-4274(99)00216-7. [DOI] [PubMed] [Google Scholar]
- 168.Hrometz S.L., Brown A.W., Nichols D.E., Sprague J.E. 3,4-methylenedioxymethamphetamine (MDMA, ecstasy)-mediated production of hydrogen peroxide in an in vitro model: the role of dopamine, the serotonin-reuptake transporter, and monoamine oxidase-B. Neurosci. Lett. 2004;367(1):56–59. doi: 10.1016/j.neulet.2004.05.075. [DOI] [PubMed] [Google Scholar]
- 169.Ricaurte G.A., Finnegan K.T., Irwin I., Langston J.W. Aminergic metabolites in cerebrospinal fluid of humans previously exposed to MDMA: preliminary observations. Ann. N. Y. Acad. Sci. 1990;600(1):699–708. doi: 10.1111/j.1749-6632.1990.tb16919.x. [DOI] [PubMed] [Google Scholar]
- 170.Verheyden S.L., Henry J.A., Curran H.V. Acute, sub-acute and long-term subjective consequences of ‘ecstasy’ (MDMA) consumption in 430 regular users. Hum. Psychopharmacol. 2003;18(7):507–517. doi: 10.1002/hup.529. [DOI] [PubMed] [Google Scholar]
- 171.Montoya A.G., Sorrentino R., Lukas S.E., Price B.H. Long-term neuropsychiatric consequences of “ecstasy” (MDMA): a review. Harv. Rev. Psychiatry. 2002;10(4):212–220. doi: 10.1080/10673220216223. [DOI] [PubMed] [Google Scholar]
- 172.Halpern P., Moskovich J., Avrahami B., Bentur Y., Soffer D., Peleg K. Morbidity associated with MDMA (ecstasy) abuse: a survey of emergency department admissions. Hum. Exp. Toxicol. 2011;30(4):259–266. doi: 10.1177/0960327110370984. [DOI] [PubMed] [Google Scholar]
- 173.Parrott A.C., Sisk E., Turner J.J. Psychobiological problems in heavy ‘ecstasy’ (MDMA) polydrug users. Drug Alcohol Depend. 2000;60(1):105–110. doi: 10.1016/s0376-8716(99)00146-5. [DOI] [PubMed] [Google Scholar]
- 174.Parrott A.C. MDMA in humans: factors which affect the neuropsychobiological profiles of recreational ecstasy users, the integrative role of bioenergetic stress. J. Psychopharmacol. 2006;20(2):147–163. doi: 10.1177/0269881106063268. [DOI] [PubMed] [Google Scholar]
- 175.Halpern J.H., Sherwood A.R., Hudson J.I., Gruber S., Kozin D., Pope H.G., Jr Residual neurocognitive features of long-term ecstasy users with minimal exposure to other drugs. Addiction. 2011;106(4):777–786. doi: 10.1111/j.1360-0443.2010.03252.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Schwartz R.H., Miller N.S. MDMA (ecstasy) and the rave: a review. Pediatrics. 1997;100(4):705–708. doi: 10.1542/peds.100.4.705. [DOI] [PubMed] [Google Scholar]
- 177.Kraner J.C., McCoy D.J., Evans M.A., Evans L.E., Sweeney B.J. Fatalities caused by the MDMA-related drug paramethoxyamphetamine (PMA). J. Anal. Toxicol. 2001;25(7):645–648. doi: 10.1093/jat/25.7.645. [DOI] [PubMed] [Google Scholar]
- 178.Oehen P., Traber R., Widmer V., Schnyder U. A randomized, controlled pilot study of MDMA (± 3,4-Methylenedioxymethamphetamine)-assisted psychotherapy for treatment of resistant, chronic Post-Traumatic Stress Disorder (PTSD). J. Psychopharmacol. 2013;27(1):40–52. doi: 10.1177/0269881112464827. [DOI] [PubMed] [Google Scholar]
- 179.Bouso J.C., Doblin R., Farré M., Alcázar M.A., Gómez-Jarabo G. MDMA-assisted psychotherapy using low doses in a small sample of women with chronic posttraumatic stress disorder. J. Psychoactive Drugs. 2008;40(3):225–236. doi: 10.1080/02791072.2008.10400637. [DOI] [PubMed] [Google Scholar]
- 180.Mithoefer M.C., Wagner M.T., Mithoefer A.T., Jerome L., Martin S.F., Yazar-Klosinski B., Michel Y., Brewerton T.D., Doblin R. Durability of improvement in post-traumatic stress disorder symptoms and absence of harmful effects or drug dependency after 3,4-methylenedioxymethamphetamine-assisted psychotherapy: a prospective long-term follow-up study. J. Psychopharmacol. 2013;27(1):28–39. doi: 10.1177/0269881112456611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Parrott A.C., Lees A., Garnham N.J., Jones M., Wesnes K. Cognitive performance in recreational users of MDMA of ‘ecstasy’: evidence for memory deficits. J. Psychopharmacol. 1998;12(1):79–83. doi: 10.1177/026988119801200110. [DOI] [PubMed] [Google Scholar]
- 182.Vollenweider F.X., Gamma A., Liechti M., Huber T. Psychological and cardiovascular effects and short-term sequelae of MDMA (“ecstasy”) in MDMA-naïve healthy volunteers. Neuropsychopharmacology. 1998;19(4):241–251. doi: 10.1038/sj.npp.1395197. [DOI] [PubMed] [Google Scholar]
- 183.Capela J.P., Carmo H., Remião F., Bastos M.L., Meisel A., Carvalho F. Molecular and cellular mechanisms of ecstasy-induced neurotoxicity: an overview. Mol. Neurobiol. 2009;39(3):210–271. doi: 10.1007/s12035-009-8064-1. [DOI] [PubMed] [Google Scholar]
- 184.Vorhees C.V., Schaefer T.L., Skelton M.R., Grace C.E., Herring N.R., Williams M.T. (+/-)3,4-Methylenedioxymethamphetamine (MDMA) dose-dependently impairs spatial learning in the morris water maze after exposure of rats to different five-day intervals from birth to postnatal day twenty. Dev. Neurosci. 2009;31(1-2):107–120. doi: 10.1159/000207499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Green A.R., Gabrielsson J., Marsden C.A., Fone K.C.F. MDMA: on the translation from rodent to human dosing. Psychopharmacology (Berl.) 2009;204(2):375–378. doi: 10.1007/s00213-008-1453-8. [DOI] [PubMed] [Google Scholar]
- 186.Sáez-Briones P., Hernández A. MDMA (3, 4-methylenedioxymethamphetamine) analogues as tools to characterize MDMA-like effects: an approach to understand entactogen pharmacology. Curr. Neuropharmacol. 2013;11(5):521–534. doi: 10.2174/1570159X11311050007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Heifets B.D., Salgado J.S., Taylor M.D., Hoerbelt P., Cardozo Pinto D.F., Steinberg E.E., Walsh J.J., Sze J.Y., Malenka R.C. Distinct neural mechanisms for the prosocial and rewarding properties of MDMA. Sci. Transl. Med. 2019;11(522):eaaw6435. doi: 10.1126/scitranslmed.aaw6435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Kuteykin-Teplyakov K., Maldonado R. Looking for prosocial genes: ITRAQ analysis of proteins involved in MDMA-induced sociability in mice. Eur. Neuropsychopharmacol. 2014;24(11):1773–1783. doi: 10.1016/j.euroneuro.2014.08.007. [DOI] [PubMed] [Google Scholar]
- 189.Ramos L., Hicks C., Caminer A., Couto K., Narlawar R., Kassiou M., McGregor I.S. MDMA (‘Ecstasy’), oxytocin and vasopressin modulate social preference in rats: A role for handling and oxytocin receptors. Pharmacol. Biochem. Behav. 2016;150-151:115–123. doi: 10.1016/j.pbb.2016.10.002. [DOI] [PubMed] [Google Scholar]
- 190.Morley-Fletcher S., Bianchi M., Gerra G., Laviola G. Acute and carryover effects in mice of MDMA (“ecstasy”) administration during periadolescence. Eur. J. Pharmacol. 2002;448(1):31–38. doi: 10.1016/S0014-2999(02)01904-0. [DOI] [PubMed] [Google Scholar]
- 191.Abad S., Fole A., del Olmo N., Pubill D., Pallàs M., Junyent F., Camarasa J., Camins A., Escubedo E. MDMA enhances hippocampal-dependent learning and memory under restrictive conditions, and modifies hippocampal spine density. Psychopharmacology (Berl.) 2014;231(5):863–874. doi: 10.1007/s00213-013-3304-5. [DOI] [PubMed] [Google Scholar]
- 192.Abad S., Camarasa J., Pubill D., Camins A., Escubedo E. Adaptive plasticity in the hippocampus of young mice intermittently exposed to MDMA could be the origin of memory deficits. Mol. Neurobiol. 2016;53(10):7271–7283. doi: 10.1007/s12035-015-9618-z. [DOI] [PubMed] [Google Scholar]
- 193.Able J.A., Gudelsky G.A., Vorhees C.V., Williams M.T. 3,4-Methylenedioxymethamphetamine in adult rats produces deficits in path integration and spatial reference memory. Biol. Psychiatry. 2006;59(12):1219–1226. doi: 10.1016/j.biopsych.2005.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Adeniyi P.A., Ishola A.O., Laoye B.J., Olatunji B.P., Bankole O.O., Shallie P.D., Ogundele O.M. Neural and behavioural changes in male periadolescent mice after prolonged nicotine-MDMA treatment. Metab. Brain Dis. 2016;31(1):93–107. doi: 10.1007/s11011-015-9691-z. [DOI] [PubMed] [Google Scholar]
- 195.Bubenikova-Valesova V. The effect of estrogen on sensorimotor processing in MDMA-treated ovariectomized female rats. Behav. Pharmacol. 2010;21(5-6):593–593. [Google Scholar]
- 196.Cohen M.A., Skelton M.R., Schaefer T.L., Gudelsky G.A., Vorhees C.V., Williams M.T. Learning and memory after neonatal exposure to 3,4-methylenedioxymethamphetamine (ecstasy) in rats: interaction with exposure in adulthood. Synapse. 2005;57(3):148–159. doi: 10.1002/syn.20166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Edut S., Rubovitch V., Schreiber S., Pick C.G. The intriguing effects of ecstasy (MDMA) on cognitive function in mice subjected to a minimal traumatic brain injury (mTBI). Psychopharmacology (Berl.) 2011;214(4):877–889. doi: 10.1007/s00213-010-2098-y. [DOI] [PubMed] [Google Scholar]
- 198.Edut S., Rubovitch V., Rehavi M., Schreiber S., Pick C.G. A study on the mechanism by which MDMA protects against dopaminergic dysfunction after minimal traumatic brain injury (mTBI) in mice. J. Mol. Neurosci. 2014;54(4):684–697. doi: 10.1007/s12031-014-0399-z. [DOI] [PubMed] [Google Scholar]
- 199.Llorente-Berzal A., Manzanedo C., Daza-Losada M., Valero M., López-Gallardo M., Aguilar M.A., Rodríguez-Arias M., Miñarro J., Viveros M.P. Sex-dependent effects of early maternal deprivation on MDMA-induced conditioned place preference in adolescent rats: possible neurochemical correlates. Toxicology. 2013;311(1-2):78–86. doi: 10.1016/j.tox.2012.12.003. [DOI] [PubMed] [Google Scholar]
- 200.Ludwig V., Mihov Y., Schwarting R.K.W. Behavioral and neurochemical consequences of multiple MDMA administrations in the rat: role of individual differences in anxiety-related behavior. Behav. Brain Res. 2008;189(1):52–64. doi: 10.1016/j.bbr.2007.12.008. [DOI] [PubMed] [Google Scholar]
- 201.Meyer J.S., Piper B.J., Vancollie V.E. 2008. [DOI] [PubMed] [Google Scholar]
- 202.Nawata Y., Hiranita T., Yamamoto T. A cannabinoid CB(1) receptor antagonist ameliorates impairment of recognition memory on withdrawal from MDMA (Ecstasy). Neuropsychopharmacology. 2010;35(2):515–520. doi: 10.1038/npp.2009.158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Piper B.J., Meyer J.S. Memory deficit and reduced anxiety in young adult rats given repeated intermittent MDMA treatment during the periadolescent period. Pharmacol. Biochem. Behav. 2004;79(4):723–731. doi: 10.1016/j.pbb.2004.10.001. [DOI] [PubMed] [Google Scholar]
- 204.Piper B.J., Fraiman J.B., Meyer J.S. Repeated MDMA (“Ecstasy”) exposure in adolescent male rats alters temperature regulation, spontaneous motor activity, attention, and serotonin transporter binding. Dev. Psychobiol. 2005;47(2):145–157. doi: 10.1002/dev.20085. [DOI] [PubMed] [Google Scholar]
- 205.Piper B.J., Fraiman J.B., Owens C.B., Ali S.F., Meyer J.S. Dissociation of the neurochemical and behavioral toxicology of MDMA (‘Ecstasy’) by citalopram. Neuropsychopharmacology. 2008;33(5):1192–1205. doi: 10.1038/sj.npp.1301491. [DOI] [PubMed] [Google Scholar]
- 206.Rodsiri R., Spicer C., Green A.R., Marsden C.A., Fone K.C. Acute concomitant effects of MDMA binge dosing on extracellular 5-HT, locomotion and body temperature and the long-term effect on novel object discrimination in rats. Psychopharmacology (Berl.) 2011;213(2-3):365–376. doi: 10.1007/s00213-010-1921-9. [DOI] [PubMed] [Google Scholar]
- 207.Ros-Simó C., Moscoso-Castro M., Ruiz-Medina J., Ros J., Valverde O. Memory impairment and hippocampus specific protein oxidation induced by ethanol intake and 3, 4-methylenedioxymethamphetamine (MDMA) in mice. J. Neurochem. 2013;125(5):736–746. doi: 10.1111/jnc.12247. [DOI] [PubMed] [Google Scholar]
- 208.Schulz S., Becker T., Nagel U., von Ameln-Mayerhofer A., Koch M. Chronic co-administration of the cannabinoid receptor agonist WIN55,212-2 during puberty or adulthood reverses 3,4 methylenedioxymetamphetamine (MDMA)-induced deficits in recognition memory but not in effort-based decision making. Pharmacol. Biochem. Behav. 2013;106:91–100. doi: 10.1016/j.pbb.2013.03.011. [DOI] [PubMed] [Google Scholar]
- 209.Shortall S.E., Garner R., Worth P., Green A.R., Fone K.C.F., King M.V. The effects of MDMA pre-treatment on behavioural responses to mephedrone in the rat. Eur. Neuropsychopharmacol. 2012;22:S405–S406. doi: 10.1016/S0924-977X(12)70633-8. [DOI] [Google Scholar]
- 210.Shortall S.E., Macerola A.E., Swaby R.T., Jayson R., Korsah C., Pillidge K.E., Wigmore P.M., Ebling F.J., Richard Green A., Fone K.C., King M.V. Behavioural and neurochemical comparison of chronic intermittent cathinone, mephedrone and MDMA administration to the rat. Eur. Neuropsychopharmacol. 2013;23(9):1085–1095. doi: 10.1016/j.euroneuro.2012.09.005. [DOI] [PubMed] [Google Scholar]
- 211.Skelton M.R., Able J.A., Grace C.E., Herring N.R., Schaefer T.L., Gudelsky G.A., Vorhees C.V., Williams M.T. (+/-)-3,4-Methylenedioxymethamphetamine treatment in adult rats impairs path integration learning: a comparison of single vs once per week treatment for 5 weeks. Neuropharmacology. 2008;55(7):1121–1130. doi: 10.1016/j.neuropharm.2008.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.van Nieuwenhuijzen P.S., Long L.E., Hunt G.E., Arnold J.C., McGregor I.S. Residual social, memory and oxytocin-related changes in rats following repeated exposure to γ-hydroxybutyrate (GHB), 3,4-methylenedioxymethamphetamine (MDMA) or their combination. Psychopharmacology (Berl.) 2010;212(4):663–674. doi: 10.1007/s00213-010-1986-5. [DOI] [PubMed] [Google Scholar]
- 213.Vorhees C.V., Schaefer T.L., Williams M.T. Developmental effects of +/-3,4-methylenedioxymethamphetamine on spatial versus path integration learning: effects of dose distribution. Synapse. 2007;61(7):488–499. doi: 10.1002/syn.20379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Vorhees C.V., Johnson H.L., Burns L.N., Williams M.T. Developmental treatment with the dopamine D2/3 agonist quinpirole selectively impairs spatial learning in the Morris water maze. Neurotoxicol. Teratol. 2009;31(1):1–10. doi: 10.1016/j.ntt.2008.09.003. [DOI] [PubMed] [Google Scholar]
- 215.Andó R.D., Adori C., Kirilly E., Molnár E., Kovács G.G., Ferrington L., Kelly P.A., Bagdy G. Acute SSRI-induced anxiogenic and brain metabolic effects are attenuated 6 months after initial MDMA-induced depletion. Behav. Brain Res. 2010;207(2):280–289. doi: 10.1016/j.bbr.2009.10.011. [DOI] [PubMed] [Google Scholar]
- 216.Bull E.J., Hutson P.H., Fone K.C.F. Increased anxiety following 3,4-methylenedioxymethamphetamine (MDMA) is accompanied by attenuation of 5-HT2A receptor responsivity to a 5-HT agonist. J. Psychopharmacol. 2003;17(3):A35–A35. [Google Scholar]
- 217.Clemens K.J., Van Nieuwenhuyzen P.S., Li K.M., Cornish J.L., Hunt G.E., McGregor I.S. MDMA (“ecstasy”), methamphetamine and their combination: long-term changes in social interaction and neurochemistry in the rat. Psychopharmacology (Berl.) 2004;173(3-4):318–325. doi: 10.1007/s00213-004-1786-x. [DOI] [PubMed] [Google Scholar]
- 218.Clemens K.J., Cornish J.L., Li K.M., Hunt G.E., McGregor I.S. MDMA (‘Ecstasy’) and methamphetamine combined: order of administration influences hyperthermic and long-term adverse effects in female rats. Neuropharmacology. 2005;49(2):195–207. doi: 10.1016/j.neuropharm.2005.03.002. [DOI] [PubMed] [Google Scholar]
- 219.Curry D.W. Stereospecificity of the prosocial and neurotoxic effects of 3,4-methylenedioxymethamphetamine (MDMA) in mice. Diss. Abstr. Int. B. Sci. Eng. 2018;78(11-B(E)) [Google Scholar]
- 220.Curry D.W., Berro L.F., Belkoff A.R., Sulima A., Rice K.C., Howell L.L. Sensitization to the prosocial effects of 3,4-methylenedioxymethamphetamine (MDMA). Neuropharmacology. 2019;151:13–20. doi: 10.1016/j.neuropharm.2019.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Daza-Losada M., Rodríguez-Arias M., Maldonado C., Aguilar M.A., Guerri C., Miñarro J. Acute behavioural and neurotoxic effects of MDMA plus cocaine in adolescent mice. Neurotoxicol. Teratol. 2009;31(1):49–59. doi: 10.1016/j.ntt.2008.07.005. [DOI] [PubMed] [Google Scholar]
- 222.Dornan W.A., Katz J.L., Ricaurte G.A. The effects of repeated administration of MDMA on the expression of sexual behavior in the male rat. Pharmacol. Biochem. Behav. 1991;39(3):813–816. doi: 10.1016/0091-3057(91)90171-W. [DOI] [PubMed] [Google Scholar]
- 223.Kirilly E., Benko A., Ferrington L., Ando R.D., Kelly P.A.T., Bagdy G. Acute and long-term effects of a single dose of MDMA on aggression in Dark Agouti rats. Int. J. Neuropsychopharmacol. 2006;9(1):63–76. doi: 10.1017/S146114570500581X. [DOI] [PubMed] [Google Scholar]
- 224.Kirilly E. 2010.
- 225.Kurling S., Kankaanpää A., Seppälä T. Sub-chronic nandrolone treatment modifies neurochemical and behavioral effects of amphetamine and 3,4-methylenedioxymethamphetamine (MDMA) in rats. Behav. Brain Res. 2008;189(1):191–201. doi: 10.1016/j.bbr.2007.12.021. [DOI] [PubMed] [Google Scholar]
- 226.Maldonado E., Davila G., Navarro J.F. MDMA (“ecstasy”) administration changes the temporal and sequential structure of agonistic behavior in male mice. Aggress. Behav. 2001;27(3):197–197. [Google Scholar]
- 227.Miczek K.A., Haney M. Psychomotor stimulant effects of d-amphetamine, MDMA and PCP: aggressive and schedule-controlled behavior in mice. Psychopharmacology (Berl.) 1994;115(3):358–365. doi: 10.1007/BF02245077. [DOI] [PubMed] [Google Scholar]
- 228.Morley K.C., McGregor I.S. (+/-)-3,4-methylenedioxymethamphetamine (MDMA, ‘Ecstasy’) increases social interaction in rats. Eur. J. Pharmacol. 2000;408(1):41–49. doi: 10.1016/S0014-2999(00)00749-4. [DOI] [PubMed] [Google Scholar]
- 229.Ramos L., Hicks C., Kevin R., Caminer A., Narlawar R., Kassiou M., McGregor I.S. Acute prosocial effects of oxytocin and vasopressin when given alone or in combination with 3,4-methylenedioxymethamphetamine in rats: involvement of the V1A receptor. Neuropsychopharmacology. 2013;38(11):2249–2259. doi: 10.1038/npp.2013.125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Ramos L., Hicks C., Caminer A., Goodwin J., McGregor I.S. Oxytocin and MDMA (‘Ecstasy’) enhance social reward in rats. Psychopharmacology (Berl.) 2015;232(14):2631–2641. doi: 10.1007/s00213-015-3899-9. [DOI] [PubMed] [Google Scholar]
- 231.Rodríguez-Arias M., Maldonado C., Vidal-Infer A., Guerri C., Aguilar M.A., Miñarro J. Intermittent ethanol exposure increases long-lasting behavioral and neurochemical effects of MDMA in adolescent mice. Psychopharmacology (Berl.) 2011;218(2):429–442. doi: 10.1007/s00213-011-2329-x. [DOI] [PubMed] [Google Scholar]
- 232.Rodríguez-Arias M., Vaccaro S., Arenas M.C., Aguilar M.A., Miñarro J. The novelty-seeking phenotype modulates the long-lasting effects of adolescent MDMA exposure. Physiol. Behav. 2015;141:190–198. doi: 10.1016/j.physbeh.2015.01.023. [DOI] [PubMed] [Google Scholar]
- 233.Shen E.Y., Ali S.F., Meyer J.S. Chronic administration of THC prevents the behavioral effects of intermittent adolescent MDMA administration and attenuates MDMA-induced hyperthermia and neurotoxicity in rats. Neuropharmacology. 2011;61(8):1183–1192. doi: 10.1016/j.neuropharm.2011.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Slais K., Landa L., Sulcova A. Does repeated treatment with MDMA sensitize to its effects on agonistic behaviour in mice? Eur. Neuropsychopharmacol. 2005;15:S584–S585. doi: 10.1016/S0924-977X(05)81231-3. [DOI] [Google Scholar]
- 235.Slais K., Landa L., Sulcova A. Anti-aggressive and anxiogenic effects of MDMA in aggressive and timid singly housed mice. Behav. Pharmacol. 2005;16:S56–S56. doi: 10.1097/00008877-200509001-00177. [DOI] [Google Scholar]
- 236.Slais K., Landa L., Sulcova A. MDMA decreases sociability and aggression and increases anxiety in mouse model of agonistic behaviour. Activitas Nervosa Superior Rediviva. 2009;51(1-2):79–80. [Google Scholar]
- 237.Thompson M.R., Li K.M., Clemens K.J., Gurtman C.G., Hunt G.E., Cornish J.L., McGregor I.S. Chronic fluoxetine treatment partly attenuates the long-term anxiety and depressive symptoms induced by MDMA (‘Ecstasy’) in rats. Neuropsychopharmacology. 2004;29(4):694–704. doi: 10.1038/sj.npp.1300347. [DOI] [PubMed] [Google Scholar]
- 238.Ball K.T., Klein J.E., Plocinski J.A., Slack R. Behavioral sensitization to 3,4-methylenedioxymethamphetamine is long-lasting and modulated by the context of drug administration. Behav. Pharmacol. 2011;22(8):847–850. doi: 10.1097/FBP.0b013e32834d13b4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Biezonski D.K., Courtemanche A.B., Hong S.B., Piper B.J., Meyer J.S. Repeated adolescent MDMA (“Ecstasy”) exposure in rats increases behavioral and neuroendocrine responses to a 5-HT2A/2C agonist. Brain Res. 2009;1252:87–93. doi: 10.1016/j.brainres.2008.11.045. [DOI] [PubMed] [Google Scholar]
- 240.Braida D., Pozzi M., Cavallini R., Sala M. 3,4 methylenedioxymethamphetamine (ecstasy) impairs eight-arm radial maze performance and arm entry pattern in rats. Behav. Neurosci. 2002;116(2):298–304. doi: 10.1037/0735-7044.116.2.298. [DOI] [PubMed] [Google Scholar]
- 241.Callaway C.W., Wing L.L., Geyer M.A. Serotonin release contributes to the locomotor stimulant effects of 3,4-methylenedioxymethamphetamine in rats. J. Pharmacol. Exp. Ther. 1990;254(2):456–464. [PubMed] [Google Scholar]
- 242.Callaway C.W., Geyer M.A. Stimulant effects of 3,4-methylenedioxymethamphetamine in the nucleus accumbens of rat. Eur. J. Pharmacol. 1992;214(1):45–51. doi: 10.1016/0014-2999(92)90094-K. [DOI] [PubMed] [Google Scholar]
- 243.Colussi-Mas J., Schenk S. Acute and sensitized response to 3,4-methylenedioxymethamphetamine in rats: different behavioral profiles reflected in different patterns of Fos expression. Eur. J. Neurosci. 2008;28(9):1895–1910. doi: 10.1111/j.1460-9568.2008.06467.x. [DOI] [PubMed] [Google Scholar]
- 244.Cox B.M., Shah M.M., Cichon T., Tancer M.E., Galloway M.P., Thomas D.M., Perrine S.A. Behavioral and neurochemical effects of repeated MDMA administration during late adolescence in the rat. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2014;48:229–235. doi: 10.1016/j.pnpbp.2013.09.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Ebrahimian Z., Karimi Z., Khoshnoud M.J., Namavar M.R., Daraei B., Haidari M.R. Behavioral and stereological analysis of the effects of intermittent feeding diet on the orally administrated MDMA (“ecstasy”) in mice. Innov. Clin. Neurosci. 2017;14(1-2):40–52. [PMC free article] [PubMed] [Google Scholar]
- 246.Fantegrossi W.E., Kiessel C.L., Leach P.T., Van Martin C., Karabenick R.L., Chen X., Ohizumi Y., Ullrich T., Rice K.C., Woods J.H. Nantenine: an antagonist of the behavioral and physiological effects of MDMA in mice. Psychopharmacology (Berl.) 2004;173(3-4):270–277. doi: 10.1007/s00213-003-1741-2. [DOI] [PubMed] [Google Scholar]
- 247.Ferraz-de-Paula V., Stankevicius D., Ribeiro A., Pinheiro M.L., Rodrigues-Costa E.C., Florio J.C., Lapachinske S.F., Moreau R.L., Palermo-Neto J. Differential behavioral outcomes of 3,4-methylenedioxymethamphetamine (MDMA-ecstasy) in anxiety-like responses in mice. Braz. J. Med. Biol. Res. 2011;44(5):428–437. doi: 10.1590/S0100-879X2011007500046. [DOI] [PubMed] [Google Scholar]
- 248.Gold L.H., Koob G.F. Methysergide potentiates the hyperactivity produced by MDMA in rats. Pharmacol. Biochem. Behav. 1988;29(3):645–648. doi: 10.1016/0091-3057(88)90034-2. [DOI] [PubMed] [Google Scholar]
- 249.Gold L.H., Koob G.F., Geyer M.A. Stimulant and hallucinogenic behavioral profiles of 3,4-methylenedioxymethamphetamine and N-ethyl-3,4-methylenedioxyamphetamine in rats. J. Pharmacol. Exp. Ther. 1988;247(2):547–555. [PubMed] [Google Scholar]
- 250.Hegadoren K.M., Martin-Iverson M.T., Baker G.B. Comparative behavioural and neurochemical studies with a psychomotor stimulant, an hallucinogen and 3,4-methylenedioxy analogues of amphetamine. Psychopharmacology (Berl.) 1995;118(3):295–304. doi: 10.1007/BF02245958. [DOI] [PubMed] [Google Scholar]
- 251.Hiramatsu M., Nabeshima T., Kameyama T., Maeda Y., Cho A.K. The effect of optical isomers of 3,4-methylenedioxymethamphetamine (MDMA) on stereotyped behavior in rats. Pharmacol. Biochem. Behav. 1989;33(2):343–347. doi: 10.1016/0091-3057(89)90511-X. [DOI] [PubMed] [Google Scholar]
- 252.Ho Y-J., Pawlak C.R., Guo L., Schwarting R.K. Acute and long-term consequences of single MDMA administration in relation to individual anxiety levels in the rat. Behav. Brain Res. 2004;149(2):135–144. doi: 10.1016/S0166-4328(03)00220-1. [DOI] [PubMed] [Google Scholar]
- 253.Kalivas P.W., Duffy P., White S.R. MDMA elicits behavioral and neurochemical sensitization in rats. Neuropsychopharmacology. 1998;18(6):469–479. doi: 10.1016/S0893-133X(97)00195-4. [DOI] [PubMed] [Google Scholar]
- 254.Kindlundh-Högberg A.M.S., Zhang X., Svenningsson P. S100B overexpressing mutant mice exhibit prolonged behavioural and biochemical responses towards repeated intermittent binge treatments with MDMA. Int. J. Neuropsychopharmacol. 2009;12(2):201–215. doi: 10.1017/S1461145708009437. [DOI] [PubMed] [Google Scholar]
- 255.Lebsanft H.B., Kovar K.A., Schmidt W.J. 3,4-Methylenedioxymethamphetamine and naloxone in rat rotational behaviour and open field. Eur. J. Pharmacol. 2005;516(1):34–39. doi: 10.1016/j.ejphar.2005.04.020. [DOI] [PubMed] [Google Scholar]
- 256.Lorens S.A., Hata N., Cabrera T., Hamilton M.E. Behavioural effects of 5,7-DHT and MDMA treatment in rats. Serotonin; 1990. pp. 615–623. [Google Scholar]
- 257.McNamara M.G., Kelly J.P., Leonard B.E. Some behavioural and neurochemical aspects of subacute (+/-)3,4-methylenedioxymethamphetamine administration in rats. Pharmacol. Biochem. Behav. 1995;52(3):479–484. doi: 10.1016/0091-3057(95)00206-C. [DOI] [PubMed] [Google Scholar]
- 258.Mechan A.O., Moran P.M., Elliott M., Young A.J., Joseph M.H., Green R. A study of the effect of a single neurotoxic dose of 3,4-methylenedioxymethamphetamine (MDMA; “ecstasy”) on the subsequent long-term behaviour of rats in the plus maze and open field. Psychopharmacology (Berl.) 2002;159(2):167–175. doi: 10.1007/s002130100900. [DOI] [PubMed] [Google Scholar]
- 259.O’Loinsigh E. The acute and long-term effects of MDMA (Ecstasy) in the rat. Ann Arbor: Ph.D., University College Dublin (Ireland); 2001. [Google Scholar]
- 260.Olsen R.A., Macaskill A.C., Harper D.N. Differential effects of 3,4-methylenedioxymethamphetamine, methamphetamine, meta-Chlorophenylpiperazine, and scopolamine on behavioral repetition versus variation in rats. Pharmacol. Biochem. Behav. 2016;150-151:170–181. doi: 10.1016/j.pbb.2016.11.001. [DOI] [PubMed] [Google Scholar]
- 261.Palenicek T., Bubenikova V., Votava M. 3,4-methylenedioxymethapmhetamine(MDMA) and 4-OH-dimethyltryptamine (psilocin) interaction in rats: Behavioral study on prepulse inhibition of acoustic startle reaction and on locomotion. Behav. Pharmacol. 2005;16:S75–S76. doi: 10.1097/00008877-200509001-00236. [DOI] [Google Scholar]
- 262.Pálenícek T., Hlinák Z., Bubeníková-Valesová V., Votava M., Horácek J. An analysis of spontaneous behavior following acute MDMA treatment in male and female rats. Neuroendocrinol. Lett. 2007;28(6):781–788. [PubMed] [Google Scholar]
- 263.Paulus M.P., Geyer M.A. The effects of MDMA and other methylenedioxy-substituted phenylalkylamines on the structure of rat locomotor activity. Neuropsychopharmacology. 1992;7(1):15–31. [PubMed] [Google Scholar]
- 264.Powell S.B., Lehmann-Masten V.D., Paulus M.P., Gainetdinov R.R., Caron M.G., Geyer M.A. MDMA “ecstasy” alters hyperactive and perseverative behaviors in dopamine transporter knockout mice. Psychopharmacology (Berl.) 2004;173(3-4):310–317. doi: 10.1007/s00213-003-1765-7. [DOI] [PubMed] [Google Scholar]
- 265.Quinteros-Muñoz D., Sáez-Briones P., Díaz-Véliz G., Mora-Gutiérrez S., Rebolledo-Fuentes M., Cassels B.K. Behavioral profiles in rats distinguish among “ecstasy,” methamphetamine and 2,5-dimethoxy-4-iodoamphetamine: Mixed effects for “ecstasy” analogues. Behav. Neurosci. 2010;124(5):662–676. doi: 10.1037/a0020827. [DOI] [PubMed] [Google Scholar]
- 266.Šlamberová R., Nohejlová K., Ochozková A., Mihalčíková L. What is the role of subcutaneous single injections on the behavior of adult male rats exposed to drugs? Physiol. Res. 2018;67(Suppl. 4):S665–S672. doi: 10.33549/physiolres.934053. [DOI] [PubMed] [Google Scholar]
- 267.Stanley N., Salem A., Irvine R.J. The effects of co-administration of 3,4-methylenedioxymethamphetamine (“ecstasy”) or para-methoxyamphetamine and moclobemide at elevated ambient temperatures on striatal 5-HT, body temperature and behavior in rats. Neuroscience. 2007;146(1):321–329. doi: 10.1016/j.neuroscience.2007.01.012. [DOI] [PubMed] [Google Scholar]
- 268.Walker Q.D., Morris S.E., Arrant A.E., Nagel J.M., Parylak S., Zhou G., Caster J.M., Kuhn C.M. Dopamine uptake inhibitors but not dopamine releasers induce greater increases in motor behavior and extracellular dopamine in adolescent rats than in adult male rats. J. Pharmacol. Exp. Ther. 2010;335(1):124–132. doi: 10.1124/jpet.110.167320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Yamamotová A., Šlamberová R. Behavioral and antinociceptive effects of different psychostimulant drugs in prenatally methamphetamine-exposed rats. Physiol. Res. 2012;61(Suppl. 2):S139–S147. doi: 10.33549/physiolres.932428. [DOI] [PubMed] [Google Scholar]
- 270.Yang P.B., Atkins K.D., Dafny N. Behavioral sensitization and cross-sensitization between methylphenidate amphetamine, and 3,4-methylenedioxymethamphetamine (MDMA) in female SD rats. Eur. J. Pharmacol. 2011;661(1-3):72–85. doi: 10.1016/j.ejphar.2011.04.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Young R., Glennon R.A. MDMA (N-methyl-3,4-methylenedioxyamphetamine) and its stereoisomers: Similarities and differences in behavioral effects in an automated activity apparatus in mice. Pharmacol. Biochem. Behav. 2008;88(3):318–331. doi: 10.1016/j.pbb.2007.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Saadat K.S., Elliott J.M., Colado M.I., Green A.R. The acute and long-term neurotoxic effects of MDMA on marble burying behaviour in mice. J. Psychopharmacol. 2006;20(2):264–271. doi: 10.1177/0269881106058022. [DOI] [PubMed] [Google Scholar]
- 273.Skelton M.R., Schaefer T.L., Herring N.R., Grace C.E., Vorhees C.V., Williams M.T. Comparison of the developmental effects of 5-methoxy-N,N-diisopropyltryptamine (Foxy) to (+/-)-3,4-methylenedioxymethamphetamine (ecstasy) in rats. Psychopharmacology (Berl.) 2009;204(2):287–297. doi: 10.1007/s00213-009-1459-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Cassel J.C., Riegert C., Rutz S., Koenig J., Rothmaier K., Cosquer B., Lazarus C., Birthelmer A., Jeltsch H., Jones B.C., Jackisch R. Ethanol, 3,4-methylenedioxymethamphetamine (MDMA, ecstasy) and their combination: Long-term behavioral, neurochemical and neuropharmacological effects in the rat. Naunyn Schmiedebergs Arch. Pharmacol. 2005;371:R75–R75. doi: 10.1038/sj.npp.1300714. [DOI] [PubMed] [Google Scholar]
- 275.Odland A.U., Jessen L., Fitzpatrick C.M., Andreasen J.T. 8-OH-DPAT Induces Compulsive-like Deficit in Spontaneous Alternation Behavior: Reversal by MDMA but Not Citalopram. ACS Chem. Neurosci. 2019;10(7):3094–3100. doi: 10.1021/acschemneuro.8b00593. [DOI] [PubMed] [Google Scholar]
- 276.Ricaurte G.A., Markowska A.L., Wenk G.L., Hatzidimitriou G., Wlos J., Olton D.S. 3,4-Methylenedioxymethamphetamine, serotonin and memory. J. Pharmacol. Exp. Ther. 1993;266(2):1097–1105. [PubMed] [Google Scholar]
- 277.Schwarting R.K.W., Borta A. Analysis of behavioral asymmetries in the elevated plus-maze and in the T-maze. J. Neurosci. Methods. 2005;141(2):251–260. doi: 10.1016/j.jneumeth.2004.06.013. [DOI] [PubMed] [Google Scholar]
- 278.Young J.M., McGregor I.S., Mallet P.E. Co-administration of THC and MDMA (‘ecstasy’) synergistically disrupts memory in rats. Neuropsychopharmacology. 2005;30(8):1475–1482. doi: 10.1038/sj.npp.1300692. [DOI] [PubMed] [Google Scholar]
- 279.Hawkey A., April L.B., Galizio M. Effects of MDMA on olfactory memory and reversal learning in rats. Neurobiol. Learn. Mem. 2014;114:209–216. doi: 10.1016/j.nlm.2014.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Viñals X., Maldonado R., Robledo P. Effects of repeated treatment with MDMA on working memory and behavioural flexibility in mice. Addict. Biol. 2013;18(2):263–273. doi: 10.1111/j.1369-1600.2011.00421.x. [DOI] [PubMed] [Google Scholar]
- 281.Harper D.N., Kay C., Hunt M. Prior MDMA exposure inhibits learning and produces both tolerance and sensitization in the radial-arm maze. Pharmacol. Biochem. Behav. 2013;105:34–40. doi: 10.1016/j.pbb.2013.01.018. [DOI] [PubMed] [Google Scholar]
- 282.Kay C., Harper D.N., Hunt M. The effects of binge MDMA on acquisition and reversal learning in a radial-arm maze task. Neurobiol. Learn. Mem. 2011;95(4):473–483. doi: 10.1016/j.nlm.2011.02.010. [DOI] [PubMed] [Google Scholar]
- 283.Insel T.R., Battaglia G., Johannessen J.N., Marra S., De Souza E.B. 3,4-Methylenedioxymethamphetamine (“ecstasy”) selectively destroys brain serotonin terminals in rhesus monkeys. J. Pharmacol. Exp. Ther. 1989;249(3):713–720. [PubMed] [Google Scholar]
- 284.Baggott M.J., Coyle J.R., Siegrist J.D., Garrison K.J., Galloway G.P., Mendelson J.E. Effects of 3,4-methylenedioxymethamphetamine on socioemotional feelings, authenticity, and autobiographical disclosure in healthy volunteers in a controlled setting. J. Psychopharmacol. 2016;30(4):378–387. doi: 10.1177/0269881115626348. [DOI] [PubMed] [Google Scholar]
- 285.Baggott M.J., Kirkpatrick M.G., Bedi G., de Wit H. Intimate insight: MDMA changes how people talk about significant others. J. Psychopharmacol. 2015;29(6):669–677. doi: 10.1177/0269881115581962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 286.Bajger A.T. Acute, repeated-dose and residual effects of amphetamines on psychological measures in humans. Diss. Abstr. Int. B. Sci. Eng. 2015;75(8-B(E)) [Google Scholar]
- 287.Bedi G., Cecchi G.A., Slezak D.F., Carrillo F., Sigman M., de Wit H. A window into the intoxicated mind? Speech as an index of psychoactive drug effects. Neuropsychopharmacology. 2014;39(10):2340–2348. doi: 10.1038/npp.2014.80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Bedi G., Hyman D., de Wit H. Is ecstasy an “empathogen”? Effects of ±3,4-methylenedioxymethamphetamine on prosocial feelings and identification of emotional states in others. Biol. Psychiatry. 2010;68(12):1134–1140. doi: 10.1016/j.biopsych.2010.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Bershad A.K., Miller M.A., de Wit H. MDMA does not alter responses to the Trier Social Stress Test in humans. Psychopharmacology (Berl.) 2017;234(14):2159–2166. doi: 10.1007/s00213-017-4621-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Bershad A.K., Weafer J.J., Kirkpatrick M.G., Wardle M.C., Miller M.A., de Wit H. Oxytocin receptor gene variation predicts subjective responses to MDMA. Soc. Neurosci. 2016;11(6):592–599. doi: 10.1080/17470919.2016.1143026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Bershad A.K., Mayo L.M., Van Hedger K., McGlone F., Walker S.C., de Wit H. Effects of MDMA on attention to positive social cues and pleasantness of affective touch. Neuropsychopharmacology. 2019;44(10):1698–1705. doi: 10.1038/s41386-019-0402-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Cami J., Farré M., Mas M., Roset P.N., Poudevida S., Mas A., San L., de la Torre R. Human pharmacology of 3,4-methylenedioxymethamphetamine (“ecstasy”): psychomotor performance and subjective effects. J. Clin. Psychopharmacol. 2000;20(4):455–466. doi: 10.1097/00004714-200008000-00010. [DOI] [PubMed] [Google Scholar]
- 293.de Wit H., Bedi G., Phan L., Kirkpatrick M. Effects of MDMA on sociability and neural response to social threat and social reward. Eur. Neuropsychopharmacol. 2011;21:S222–S222. doi: 10.1016/S0924-977X(11)70331-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Dolder P.C., Müller F., Schmid Y., Borgwardt S.J., Liechti M.E. Direct comparison of the acute subjective, emotional, autonomic, and endocrine effects of MDMA, methylphenidate, and modafinil in healthy subjects. Psychopharmacology (Berl.) 2018;235(2):467–479. doi: 10.1007/s00213-017-4650-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Harris D.S., Baggott M., Mendelson J.H., Mendelson J.E., Jones R.T. Subjective and hormonal effects of 3,4-methylenedioxymethamphetamine (MDMA) in humans. Psychopharmacology (Berl.) 2002;162(4):396–405. doi: 10.1007/s00213-002-1131-1. [DOI] [PubMed] [Google Scholar]
- 296.Holze F., Vizeli P., Müller F., Ley L., Duerig R., Varghese N., Eckert A., Borgwardt S., Liechti M.E. Distinct acute effects of LSD, MDMA, and D-amphetamine in healthy subjects. Neuropsychopharmacology. 2020;45(3):462–471. doi: 10.1038/s41386-019-0569-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Hysek C.M., Simmler L.D., Nicola V.G., Vischer N., Donzelli M., Krahenbuhl S., Grouzmann E., Huwyler J., Hoener M.C., Liechti M.E. Duloxetine inhibits effects of MDMA (“ecstasy”) in vitro and in humans in a randomized placebo-controlled laboratory study. PLoS One. 2012;7(5):e36476. doi: 10.1371/journal.pone.0036476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298.Kirkpatrick M.G., Baggott M.J., Mendelson J.E., Galloway G.P., Liechti M.E., Hysek C.M., de Wit H. MDMA effects consistent across laboratories. Psychopharmacology (Berl.) 2014;231(19):3899–3905. doi: 10.1007/s00213-014-3528-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Kirkpatrick M.G., Gunderson E.W., Perez A.Y., Haney M., Foltin R.W., Hart C.L. A direct comparison of the behavioral and physiological effects of methamphetamine and 3,4-methylenedioxymethamphetamine (MDMA) in humans. Psychopharmacology (Berl.) 2012;219(1):109–122. doi: 10.1007/s00213-011-2383-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.Kirkpatrick M.G., Lee R., Wardle M.C., Jacob S., de Wit H. Effects of MDMA and Intranasal oxytocin on social and emotional processing. Neuropsychopharmacology. 2014;39(7):1654–1663. doi: 10.1038/npp.2014.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Kolbrich E.A., Goodwin R.S., Gorelick D.A., Hayes R.J., Stein E.A., Huestis M.A. Physiological and subjective responses to controlled oral 3,4-methylenedioxymethamphetamine administration. J. Clin. Psychopharmacol. 2008;28(4):432–440. doi: 10.1097/JCP.0b013e31817ef470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 302.Kuypers K.P.C., Wingen M., Ramaekers J.G. Memory and mood during the night and in the morning after repeated evening doses of MDMA. J. Psychopharmacol. 2008;22(8):895–903. doi: 10.1177/02698811080220081401. [DOI] [PubMed] [Google Scholar]
- 303.Liechti M.E., Baumann C., Gamma A., Vollenweider F.X. Acute psychological effects of 3,4-methylenedioxymethamphetamine (MDMA, “Ecstasy”) are attenuated by the serotonin uptake inhibitor citalopram. Neuropsychopharmacology. 2000;22(5):513–521. doi: 10.1016/S0893-133X(99)00148-7. [DOI] [PubMed] [Google Scholar]
- 304.Liechti M.E., Vollenweider F.X. Acute psychological and physiological effects of MDMA (“Ecstasy”) after haloperidol pretreatment in healthy humans. Eur. Neuropsychopharmacol. 2000;10(4):289–295. doi: 10.1016/S0924-977X(00)00086-9. [DOI] [PubMed] [Google Scholar]
- 305.Schmidt A., Müller F., Dolder P.C., Schmid Y., Zanchi D., Egloff L., Liechti M.E., Borgwardt S. Acute effects of methylphenidate, modafinil, and MDMA on negative emotion processing. Int. J. Neuropsychopharmacol. 2018;21(4):345–354. doi: 10.1093/ijnp/pyx112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Tancer M., Johanson C-E. The effects of fluoxetine on the subjective and physiological effects of 3,4-methylenedioxymethamphetamine (MDMA) in humans. Psychopharmacology (Berl.) 2007;189(4):565–573. doi: 10.1007/s00213-006-0576-z. [DOI] [PubMed] [Google Scholar]
- 307.Tancer M.E., Johanson C.E. The subjective effects of MDMA and mCPP in moderate MDMA users. Drug Alcohol Depend. 2001;65(1):97–101. doi: 10.1016/S0376-8716(01)00146-6. [DOI] [PubMed] [Google Scholar]
- 308.Wardle M.C., Kirkpatrick M.G., de Wit H. ‘Ecstasy’ as a social drug: MDMA preferentially affects responses to emotional stimuli with social content. Soc. Cogn. Affect. Neurosci. 2014;9(8):1076–1081. doi: 10.1093/scan/nsu035. [DOI] [PMC free article] [PubMed] [Google Scholar]