Abstract
Background: The prevalence of hyponatremia is highly variable among patients with lung cancer. However, its prevalence and prognostic significance in subgroups of patients with lung cancer have not yet been evaluated in a meta-analysis.
Methods: We have registered our meta-analysis and review protocol to the PROSPERO International Prospective Register of Systematic Reviews, with the following registration number: CRD42020167013. A systematic search was done in the following sources: MEDLINE, Embase, CENTRAL, Web of Science, ClinicalTrials.gov, a WHO Global Health Library.
Results: We identified a total of 8,962 potentially eligible studies, and we included 31 articles in our evaluation. The prevalence of hyponatremia in patients with lung cancer varied between 3 and 94.8% with an average of 25% without any significant differences between the following subgroups: histotype, gender, age, Eastern Cooperative Oncology Group (ECOG) state, and the extent of disease. The overall survival (OS) was significantly lower in hyponatremic compared to normonatremic patients at 10 months [RR.59 (95% CI.47–0.74), p < 0.001] and at 20 months [RR.44 (95% CI.33–0.59), p < 0.001], with worse survival rates in non-small cell lung cancer (NSCLC) [RR.27 (95% CI.12–0.44), p < 0.001] than in small cell lung cancer (SCLC) [RR.42 (95% CI.27–0.57), p < 0.001]. If hyponatremia was corrected, OS at 10 months was significantly higher than in the uncorrected hyponatremia group [RR 1.83 (95% CI 1.37–2.44), p < 0.001], but, at 20 months, no statistically significant difference could be found between these subgroups [RR 2.65 (95% CI.94–7.50), p = 0.067].
Conclusions: Patients with lung cancer diagnosed with hyponatremia, especially patients with NSCLC, seem to have significantly lower survival rates than normonatremic patients. If hyponatremia remains uncorrected, the mortality rates might be even higher.
Keywords: lung cancer, NSCLC, SCLC, hyponatremia, SIADH
Background
Lung cancer is one of the most frequent malignancies worldwide in both sexes, and 75% of patients are diagnosed at an advanced stage. According to the reports of the WHO in 2018, it is the leading cause of mortality among cancers (1). Moreover, based on the WHO's Global Cancer Observatory's report, the incidence of lung cancer is predicted to grow by 72.5% by 2040 (2).
The reported prevalence values of hyponatremia (<135 mmol/L) among patients with lung cancer are highly discordant in the literature, varying between 3.7 and 75% (3–8). Although inappropriate antidiuretic hormone secretion (SIADH) is the most common underlying cause (4, 5), the prevalence of SIADH seems to be lower among patients with lung cancer compared to hyponatremia, varying from 9.1 to 39% in small cell lung cancer (SCLC) (6, 9–11) and 7–4% in non-small cell lung cancer (NSCLC) (6, 12). SIADH was diagnosed even less frequently in the general cancer population (4, 13, 14). This discrepancy may be partially explained by insufficient diagnostic work-up and the presence of other comorbidities that also affect the sodium level (15–17).
Based on literature reports, hyponatremia and SIADH appear to be important negative prognostic factors of mortality in numerous medical conditions, including lung cancer (4, 9, 12, 18–22), but not every study reached this conclusion (6, 10). However, a recent meta-analysis of 15 studies confirmed the prognostic significance of serum sodium levels by demonstrating that the correction of hyponatremia is associated with a reduced risk of mortality in hospitalized patients with oncology (23).
Following a preliminary search of the literature, we found discordant results regarding the prevalence and prognostic significance of hyponatremia in patients with lung cancer, and the findings considerably differ from the studies and reviews cited above. This study aims to make a reappraisal of the occurrence and prognostic impact of pre and posttreatment (oncological, symptomatic, and supportive) hyponatremia, specifically in patients with lung cancer.
Methods
Protocol and Registration
We report this systematic review and meta-analysis according to the preferred reporting items for systematic reviews and meta-analyses (PRISMA, 2009) Statement (24) described in Supplementary File 1. We have registered our meta-analysis and review protocol to the PROSPERO International Prospective Register of Systematic Reviews, with the following registration number: CRD42020167013. There were no deviations from the study protocol.
Search Strategy
The first step was to create a PECO (patient-exposure/prognostic factor-comparison-outcome): “P,” patients with lung cancer; “E,” hyponatremia at the time of the diagnosis/before treatment; “C,” normonatremia at the time of the diagnosis/before treatment; “O,” overall survival time and the rate at 10 and 20 months. Then, we created a search key based on our PECO, which included search terms related to the group of the patients and the prognostic factor: (lung cancer OR SCLC OR NSCLC OR carcinoid) AND (SIADH OR the sodium level OR the Na level OR hyponatremia OR hyponatremia OR syndrome of inappropriate ADH OR antidiuretic hormone OR hypotonicity).
We searched the following sources: MEDLINE (via PubMed), Embase, Cochrane Central Register of Controlled Trials (CENTRAL), Web of Science, ClinicalTrials.gov, and WHO Global Health Library. The “human” filter was applied in the case of MEDLINE, Embase, and the WHO Global Health Library. The “trials” and “completed” filters were utilized when searching in the Cochrane Library or ClinicalTrials.gov, respectively. We did not apply any language or date restriction to our search. We also performed a search on the bibliography of the eligible articles. The date of the search was 12.03.2019. Then, we updated our systematic search with the same search strategy used before on 05.07.2021.
Eligibility and Exclusion Criteria
Studies qualified our systematic review after fulfilling the following requirements: (1) observational or interventional cohort studies, case-control studies, randomized-controlled trials, and case series. Besides full-text articles, conference abstracts were also eligible for inclusion; (2) trials enrolling adult patients (18 years or above) with lung cancer with available information of characteristics of patients with lung cancer (number of patients, age, sex, histological type of cancer, extent of disease, and performance status) and pretreatment serum sodium levels; (3) trials reporting data on overall survival (OS) time and the rate at 10 and 20 months, or with available information about multivariate analysis of the prognostic significance of hyponatremia.
The exclusion criteria were the following: (1) case studies, case reports, reviews, comments, and letters; (2) studies investigating other lung pathologies or solid cancers; (3) lack of data on serum sodium levels before treatment; (4) papers not reporting outcomes mentioned before.
Study Selection and Data Extraction
After the initial search, all records from each database were imported into a reference management program (EndNote X7, Clarivate Analytics, Philadelphia, PA, USA). This software was used to remove duplicates. After the removal of duplicates, the authors screened the remaining articles against the predefined eligibility criteria first by title, abstract, and then full text. Two researchers conducted each step independently, and any disagreements were resolved by consensus.
Numerical data were extracted by two independent investigators, any disagreements were resolved by consensus, and data were manually entered on an Excel 2018 sheet (Office 365, Microsoft, Redmond, WA, USA). These were collected on the first author, year of publication, study design, geographical location, number of patients, and basic demographics (age and sex ratio), in each group. Additional information on the histological type of the tumor, disease extent, Eastern Cooperative Oncology Group (ECOG) Performance status, the grade of hyponatremia, and the length of follow-up was also obtained. We also collected information about hyponatremia improvement that was achieved by oncologic, symptomatic, and/or supportive treatment, including at least two cycles of chemotherapy, sodium supplementation, or fluid restriction.
Outcomes
Finally, data were collected on the primary and secondary outcomes of interest, such as the prevalence of hyponatremia, median OS time, and the OS rate at 10 and 20 months of patients with normonatremia and hyponatremia and results of multivariate analyses of lung cancer patients with hyponatremia.
Risk of Bias Assessment
We used the Quality in Prognostic Studies (QUIPS) tool (25) for assessing the risk of bias, which is described in Supplementary File 2. Any disagreements between the two independent researchers were solved by consensus.
Statistical Analysis
The effect measure of dichotomous variables was reported for each outcome as the relative risk (RR) with the related 95% CI. All tests were two-sided, and p < 0.05 was considered statistically significant (except for heterogeneity, for which a p < 0.10 was considered significant). The random-effects model was used because of the minor differences between individual studies.
Heterogeneity was tested by performing both Cochran's Q test and calculating Higgins' I∧2 indicator. p < 0.10 was considered suggestive of significant heterogeneity. The I2 index corresponds to the percentage of the total variability across studies, which is due to heterogeneity. Based on the Cochrane Handbook for Systematic Reviews of Interventions (26), a rough classification of its value is the following: low (0–40%), moderate (30–60%), substantial (50–90%), and considerable (75–100%). All the statistical analyses were performed using Stata IC (version 15.1).
Results
The selection process is summarized in Figure 1. We identified a total of 8,962 potentially eligible studies in the literature, 1,858 articles in MEDLINE, 5,201 articles in Embase, 315 records in CENTRAL, 1,440 articles in Web of Science, 33 in ClinicalTrials.gov, and 115 in WHO Global Health Library. After screening by title, abstract, and full text, we chose 31 studies for our evaluation based on our eligibility criteria. Since not all studies reported on every parameter, we could include only a limited number of studies in each sub-analysis.
Figure 1.
A preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow diagram representing the process of study search and selection.
Systematic Review
The characteristics of the studies are summarized in a table included in Table 1. All selected reports were published in peer-reviewed journals, including four prospective and 27 retrospective cohort or case-control studies. The number of participants in the trials varied from 40 to 1,083 patients. Every selected article reported demographic information of the investigated population and data on the serum sodium levels at admission or before starting oncological treatment. In all of the 31 articles selected, we analyzed the prevalence of hyponatremia. However, the analysis of the prognostic significance was performed only in 26 studies due to the lack of data in some articles. Furthermore, only 15 studies reported on median OS times, and the OS rates at 10 and 20 months were available in even fewer studies (n = 8).
Table 1.
Characteristics of the included studies in the systematic review.
| Study | Country | Study design | Nr. of patients | Histotype | Follow-up time | Male N (%) | Age cut-off (years) |
Older
N (%) |
Na level cut-off (mmol/L) | Hyponatremia N (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| Yang et al. (27) | China | Retrospective | 320 | SCLC | 40 weeks | 219 (68.4%) | 60 | 222 (69.4%) | 135 | 149 (46.6%) |
| Osterlind et al. (28) | Denmark | Retrospective | 815 | SCLC | 18–54 weeks | 584 (71.7%) | 60 | 428 (52.5%) | 136 | 204 (25.0%) |
| Zarzecka et al. (29) | Poland | Retrospective | 290 | NSCLC | NR | 205 (70.7%) | NR | NR | 135 | 47 (16.2%) |
| Hermes et al. (30) | Germany | Retrospective | 395 | SCLC | NR | 240 (60.8%) | 60 | 260 (65.8%) | 135 | 75 (19.0%) |
| Wang et al. (31) | China | Retrospective | 631 | SCLC | NR | 475 (75.3%) | 65 | 91 (14.4%) | 135 | 66 (10.5%) |
| Berardi et al. (32) | Italy | Retrospective | 433 | NSCLC | NR | 299 (69.1%) | NR | NR | 135 | 69 (15.9%) |
| Sengupta et al. (33) | India | Retrospective | 116 | NSCLC +SCLC |
NR | 83 (71.6%) | NR | NR | 135 |
45 (45.5%)
7 (41.2%) |
| Kobayashi et al. (34) | Japan | Retrospective | 386 | NSCLC | 41.2 months | 259 (67.1%) | NR | NR | 139 | 123 (31.9%) |
| Fucá et al. (35) | Italy | Prospective | 197 | NSCLC | 25.7 months | 120 (60.9%) | 65 | 113 (57.4%) | 135 | 26 (13.2%) |
| Alamoudi (36) | Saudi Arabia | Prospective | 114 | SCLC +NSCLC |
NR | 89 (78.1%) | NR | NR | 130 | 64 (56.1%) |
| Hansen et al. (37) | Denmark | Retrospective | 453 | SCLC | 60 months | 243 (53.6%) | 60 | 323 (71.3%) | 135 | 198 (44%) |
| Svaton et al. (38) | Czech Republic | Retrospective | 544 | NSCLC | 84 months | 343 (63.1%) | 65 | 242 (44.5%) | 136 | 117 (21.5%) |
| Allan et al. (39) | U.K. | Retrospective | 411 | SCLC | NR | NR | 65 | 120 (29.2%) | 135 | 111 (27.0%) |
| Li et al. (40) | China | Retrospective | 1083 | NSCLC | 40.84 months | 757 (69.9%) | 65 | 295 (27.2%) | 141,9 | 165 (15.2%) |
| Osterlind et al. (41) | Denmark | Retrospective | 874 | SCLC | 18 months | 631 (72.2%) | 60 | 450 (51.5%) | 136 | 213 (24.4%) |
| Doshi et al. (42) | India | Retrospective | 257 | NSCLC | NR | 180 (70.0%) | NR | NR | 136 | 120 (46.7%) |
| Johnson et al. (43) | U.S.A. | Prospective | 50 | SCLC +NSCLC |
NR | 33 (66.0%) | NR | NR | 130 |
10 (32.3%)
1 (5.3%) |
| Sagman et al. (44) | Canada | Retrospective | 614 | SCLC | NR | 436 (71.0%) | 70 | 88 (14.3%) | 135 | 142 (23.1%) |
| Jacot et al. (45) | France | Retrospective | 301 | NSCLC | 20.8 months | 242 (80.4%) | NR | NR | NR | 24 (8.0%) |
| Hong et al. (46) | China | Retrospective | 999 | SCLC | 10.6 months | 692 (69.3%) | 60 | 387 (38.7%) | 135 | 163 (29.5%) |
| Maestu et al. (47) | Spain | Retrospective | 341 | SCLC | NR | 336 (98.5%) | 65 | 88 (25.8%) | 135 | 10 (3%) |
| Kawahara et al. (48) | Japan | Retrospective | 286 | SCLC | NR | 233 (81.5%) | 66 | 113 (39.5%) | 136 | 40 (14.0%) |
| Cerny et al. (49) | U.K. | Retrospective | 407 | SCLC | NR | 262 (64.4%) | NR | NR | 132 | 53 (13.0%) |
| Ma et al. (50) | China | Retrospective | 158 | SCLC | NR | 135 (85.4%) | 60 | 61 (38.6%) | 135 | 67 (42.4%) |
| Umemura et al. (51) | Japan | Retrospective | 163 | SCLC | 36 months | 129 (79.1%) | 70 | 69 (42.3%) | 135 | 22 (13.5%) |
| Jacot et al. (52) | France | Retrospective | 231 | NSCLC | 35 months | 194 (84,0%) | 60 | 104 (45%) | 132 | 219 (94.8%) |
| Rechnitzer et al. (53) | Germany | Prospective | 229 | NSCLC | 42 months | 185 (80.8%) | 63 | 69 (30.1%) | 137.5 | 59 (25,8%) |
| Bose et al. (54) | India | Retrospective | 40 | NSCLC | 6 months | 38 (95%) | NR | NR | 125 | 8 (20%) |
| Rinaldi et al. (55) | Italy and UK | Retrospective | 647 | NSCLC | NR | 440 (68%) | 65 | NR | 135 | 105 (16.2%) |
| Chan et al. (56) | Ireland | Retrospective | 624 | LC | NR | 370 (59.3%) | NR | NR | 135 | 197 (31.6%) |
| Huang et al. (57) | China | Retrospective | 358 | SCLC | NR | 286 (79.9%) | 70 | 49 (13.7%) | 137 | 54 (15.1%) |
SCLC, Small cell lung cancer; NSCLC, Non-small cell lung cancer; RCT, Randomized control trial; U.S.A, United States of America; U.K., United Kingdom; NR, Not reported.
The main clinical characteristics of patients in individual articles are shown in Tables 1, 2). Out of the 12,767 patients included, 57.8% (N = 7,379) were diagnosed with SCLC, 41.2% (N = 5,255) with NSCLC, 65% (8,298) were men and 27.3% were women. The reported median age ranged from 49 to 68 years. With a cut-off value of 60 years (eight studies), 52.6% of the studied population was older than 60 years, while, with a cut-off value of 65 years (seven studies), 70.4% was younger. In total, 2,973 patients had hyponatremia at admission or before starting oncological treatment. The applied cut-off values varied between 132 and 139 mmol/L across studies. Hyponatremia was diagnosed in 1,499 of 6,792 patients (22.1%) using a 135 mmol/L cut-off value in most of the included articles (16 studies), while, in five studies, where the cut-off value was 136 mmol/L, the number of patients with hyponatremia was 694 out of 2,776 patients (25%). In most cases, the oncological treatment for SCLC consisted of platinum-based preparations and etoposide, and, for NSCLC, were platinum-based preparations and/or surgery (Table 2).
Table 2.
Different oncological treatment protocols in the included studies.
| Studies | Study interval | Histotype | Number of patients | Oncological treatment protocols | |
|---|---|---|---|---|---|
| Yang et al. (27) | 2006 | 2012 | SCLC | 320 | PB+E |
| Osterlind et al. (28) | 1973 | 1981 | SCLC | 815 | L+C+M, L+C+M+V, L+C+M+V+D+E alternating, L+C+E+V +/- radiotherapy |
| Zarzecka et al. (29) | 2010 | 2012 | NSCLC | 290 | NSCLC – PB and third generation drugs, in SCLC with PB+E |
| Hermes et al. (30) | 2004 | 2008 | SCLC | 395 | PB+E |
| Wang et al. (31) | 2006 | 2013 | SCLC | 631 | PB+E |
| Berardi et al. (32) | 2006 | 2015 | NSCLC | 433 | PB, Non-PB, EGFR- TKI |
| Sengupta et al. (33) | 2011 | 2012 | NSCLC | 116 | unknown |
| Kobayashi et al. (34) | 2000 | 2009 | NSCLC | 386 | resection+ postoperative adjuvant treatment |
| Fucá et al. (35) | 2013 | 2018 | NSCLC | 197 | PD-1/PDL-1 inhibitors |
| Alamoudi (36) | 2004 | 2008 | LC | 114 | unkown |
| Hansen et al. (37) | 1995 | 2005 | SCLC | 453 | PB+E |
| Svaton et al. (38) | 2006 | 2013 | NSCLC | 544 | erlotinib |
| Allan et al. (39) | 1982 | 1988 | SCLC | 411 | M+CCVVP+/-D,+/-radiotherapy and PCI or VI+E |
| Li et al. (40) | 2007 | 2014 | NSCLC | 1083 | resection |
| Osterlind et al. (41) | 1973 | 1981 | SCLC | 874 | L+C+M,L+C+M+V, L+C+M+V+D+E, +/- radiotherapy |
| Doshi et al. (42) | 2010 | 2014 | NSCLC | 257 | PB+PM |
| Johnson et al. (43) | 1989 | 1992 | LC | 50 | PB+E |
| Sagman et al. (44) | 1976 | 1986 | SCLC | 614 | Different protocols: C+D+V, L+P+M, PB+E |
| Jacot et al. (45) | 2003 | 2006 | NSCLC | 301 | surgery, PB, second line, palliative |
| Hong et al. (46) | 2000 | 2009 | SCLC | 999 | chemotherapy(etoposide)+/-radiotherapy+/-surgery |
| Maestu et al. (47) | 1981 | 1993 | SCLC | 341 | different protocols:C+D+V, PB and others+/- radio |
| Kawahara et al. (48) | 1985 | 1988 | SCLC | 286 | different protocols: C+D+V, P+E, or C+D+V+P+E |
| Cerny et al. (49) | 1979 | 1985 | SCLC | 407 | different protocols:C+M+E+/-radiotherapy |
| Ma et al. (50) | 2000 | 2007 | SCLC | 158 | unknown |
| Umemura et al. (51) | 1981 | 2001 | SCLC | 163 | PB or alternating chemotherapy |
| Jacot et al. (52) | 1992 | 1998 | NSCLC | 231 | radiotherapy, chemotherapy, surgery and their combinations |
| Rechnitzer et al. (53) | 1987 | 1990 | NSCLC | 229 | chemotherapy+radiotherapy |
| Bose et al. (54) | 2008 | 2008 | NSCLC | 40 | PB+PC for NSCLC, PB+E for SCLC |
| Rinaldi et al. (55) | 2006 | 2017 | NSCLC | 647 | PB or EGFR-TKI |
| Chan et al. (56) | 2011 | 2016 | LC | 624 | unknown |
| Huang et al. (57) | 2011 | 2018 | SCLC | 358 | PB+ E and others |
SCLC, small cell lung cancer, NSCLC, Non-small cell lung cancer, LC, every lung cancer histotype, PB, Platinum-based preparations, PC, paclitaxel, PM, pemetrexed, L, lomustine, C, cyclophosphamide, M, methotrexate, V, vincristine, D, doxorubicine, E, etoposide, P, procarbazine, VI, vindesine, PCI, prophilactic cranial irradiation, EGFR, epidermal growth factor receptor, TKI, tyrosine kinase inhibitors.
The results of various outcomes among the selected studies are shown in Table 3. The prognostic significance of hyponatremia was evaluated in 26 studies and was found to be an independent prognostic factor in 15 studies, whereas, in eight studies, it was not.
Table 3A.
The outcomes in lung cancer patients among the included studies.
| Study | Median OS times | p-value | Survival rates, mortality | p-value | Multivariate analysis | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| NN patients | HN patients | NN patients | HN patients | HR | 95% CI | p-value | ||||
| Yang et al. (27) | 1.1 ± 0.42 year | 0.83 ± 0.35 year | <0.05 | 62.26% 1-year SR 1.89% 5-year SR |
30.36% 1-year SR 0% 5-year SR |
0.045 0.15 |
NR | - | - | - |
| Osterlind et al. (28) | NR | NR | - | NR | NR | - | NR | - | - | - |
| Zarzecka et al. (29) | NR | NR | - | 7.8% mortality | 28.7% mortality | 0.0001 | NR | - | - | - |
| Hermes et al. (30) | 13 months | 9 months | <0.001 | NR | NR | - | I | - | - | 0.025 LD 0.038 ED |
| Wang et al. (31) | 14.5 months | 11.4 months | <0.001 | NR | NR | - | I | 1.82 | 1.34–2.47 | 0.007 |
| Berardi et al. (32) | 15.5 months | 8.78 months | <0.001 | 57.06% SR | 33.33% SR | 0.19 | I | 1.59 | 1.14–2.21 | 0.006 |
| Sengupta et al. (33) | NR | NR | - | NR | NR | - | NI | - | - | NS |
| Kobayashi et al. (34) | NR | NR | - | 74.8% 5-year SR | 59.7% 5-year SR | 0.002 | I | 1.53 | 1.01–2.32 | 0.047 |
| Fucá et al. (35) | 11.6 months | 2.8 months | <0.001 | 39.76% SR at 10 months | 15.38% SR at 10 months | - | I | 3.11 | 1.91–5.05 | <0.001 |
| Alamoudi (36) | NR | NR | - | NR | NR | - | NR | - | - | - |
| Hansen et al. (37) | 11.2 months | 7.1 months | 0.0001 | 57.25% SR at 10 months | 27.27% SR at 10 months | - | I | 1.6 | 1.27–2.01 | <0.001 |
| Svaton et al. (38) | 10.9 months | 4.6 months | <0.001 | 54.33% SR at 10 months | 21.36% SR at 10 months | <0.001 | I | 1.87 | 1.47–2.39 | <0.001 |
OS, overall survival; NN, normonatremic; HN, hyponatremic; NR, Not reported; S-significant; NS, not significant; I, Independent factor; N.I., Not independent factor; HR, hazard ratio; RR, risk ratio; SR, Survival rate; CI, confidence interval; LD, Limited Disease; ED, Extensive Disease.
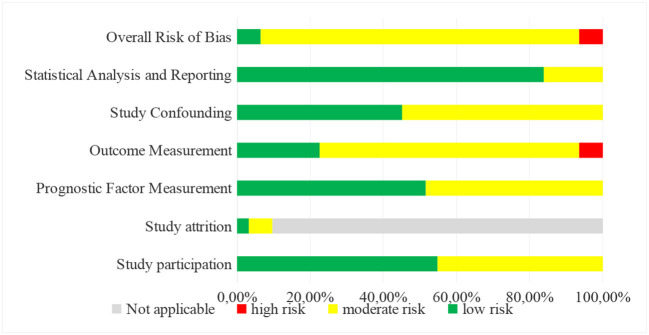
After evaluating the risk of bias in each study using the QUIPS tool, the included reports had a low, high, and most medium risk of bias (Table 4).
Table 3B.
The outcomes in lung cancer patients among the included studies.
| Study | Median OS times | p-value | Survival rates, mortality | p-value | Outcome in multivariate analysis | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| NN patients | HN patients | NN patients | HN patients | HR | 95% CI | p-value | ||||
| Allan et al. (39) | 7 months | 7 months | 0.06 | NR | NR | - | NI | - | - | NS |
| Li et al. (40) | NR | NR | - | 79.6% SR at 25 months | 73.83% SR at 25 months | - | NR | - | - | - |
| Osterlind et al. (41) | 40 weeks | 34 weeks | - | NR | NR | - | I | - | - | <0.05 |
| Doshi et al. (42) | 16 months | 11 months | <0.05 | 61.31% SR at 10 months | 54.16% SR at 10 months | - | I | 2.07 | 1.11–3.84 | <0.05 |
| Johnson et al. (43) | NR | NR | - | NR | NR | - | NI | - | - | NS |
| Sagman et al. (44) | 45 weeks | 42 weeks | 0.006 | NR | NR | - | NI | - | - | NS |
| Jacot et al. (45) | 18.7 months | 4.1 months | <0.0001 | NR | NR | - | I | 1.99 | 1.04–3.77 | S |
| Hong et al. (46) | 11.7 months | 10 months | 0.039 | 59.48% SR at 10 months | 49.69% SR at 10 months | - | NI | - | - | NS |
| Maestu et al. (47) | NR | NR | - | NR | NR | - | NI | - | - | NS |
| Kawahara et al. (48) | 11.4 months | 9.1 months | 0.0072 | NR | NR | - | NI | - | - | NS |
| Cerny et al. (49) | NR | NR | - | 59.48% SR at 10 months | 34% SR at 10 months | - | I | - | - | 0.0009 |
| Ma et al. (50) | 14.1 months | 7.6 months | <0.001 | 81.31% SR at 10 months | 32.83% SR at 10 months | - | NR | - | - | - |
| Umemura et al. (51) | 10.6 months | 10 months | 0.6653 | 50% SR at 10 months | 51.77% SR at 10 months | NS | NI | - | - | NS |
| Jacot et al. (52) | 7.5 months | 3.85 months | 0.0141 | NR | NR | - | I | 2.99 | 1.17–7.62 | 0.022 |
| Rechnitzer et al. (53) | 6.7 months | 3 months | <0.001 | NR | NR | - | I | - | - | <0.001 |
| Bose et al. (54) | NR | NR | <0.03 | NR | NR | - | NR | - | - | - |
| Rinaldi et al. (55) | 15.3 months | 10.3 months | 0.003 | NR | NR | - | I | 1.29 | 1.03–1.54 | 0.047 |
| Chan et al. (56) | NR | NR | - | NR | NR | - | NR | - | - | - |
| Huang et al. (57) | 14.5 months | 11 months | 0.008 | NR | NR | - | I | 1.49 | 1.04–2.13 | 0.03 |
OS, overall survival; NN, normonatremic; HN, hyponatremic; NR, Not reported; S-significant; NS, not significant; I, Independent factor; NI, Not independent factor; HR, hazard ratio; RR, risk ratio; SR, Survival rate; CI, confidence interval.
Meta-Analysis
The summary of the results of the statistical analysis is presented in Table 5.
Table 4.
Risk of bias assessment.
| Study participation | Study attrition | Prognostic factor measurement | Outcome measurement | Study confounding | Statistical analysis and reporting | Overall risk of bias | Included in quantitative synthesis | |
|---|---|---|---|---|---|---|---|---|
| Yang et al. (27) |

|
n.a. |

|

|

|

|

|
yes |
| Osterlind et al. (28) |

|
n.a. |

|

|

|

|

|
no |
| Zarzecka et al. (29) |

|
n.a. |

|

|

|

|

|
no |
| Hermes et al. (30) |

|
n.a. |

|

|

|

|

|
no |
| Wang et al. (31) |

|
n.a. |

|

|

|

|

|
no |
| Berardi et al. (32) |

|
n.a. |

|

|

|

|

|
yes |
| Sengupta et al. (33) |

|
n.a. |

|

|

|

|

|
no |
| Kobayashi et al. (34) |

|
n.a. |

|

|

|

|

|
no |
| Fucá et al. (35) |

|

|

|

|

|

|

|
yes |
| Alamoudi (36) |

|

|

|

|

|

|

|
no |
| Hansen et al. (37) |

|
n.a. |

|

|

|

|

|
yes |
| Svaton et al. (38) |

|
n.a. |

|

|

|

|

|
yes |
| Allan et al. (39) |

|
n.a. |

|

|

|

|

|
no |
| Li et al. (40) |

|
n.a. |

|

|

|

|

|
no |
| Osterlind et al. (41) |

|
n.a. |

|

|

|

|

|
no |
| Doshi et al. (42) |

|
n.a. |

|

|

|

|

|
no |
| Johnson et al. (43) |

|

|

|

|

|

|

|
no |
| Jacot et al. (45) |

|
n.a. |

|

|

|

|

|
no |
| Umemura et al. (51) |

|
n.a. |

|

|

|

|

|
no |
| Sagman et al. (44) |

|
n.a. |

|

|

|

|

|
no |
| Hong et al. (46) |

|
n.a. |

|

|

|

|

|
yes |
| Maestu et al. (47) |

|
n.a. |

|

|

|

|

|
no |
| Kawahara et al. (48) |

|
n.a. |

|

|

|

|

|
no |
| Cerny et al. (49) |

|
n.a. |

|

|

|

|

|
yes |
| Ma et al. (50) |

|
n.a. |

|

|

|

|

|
yes |
| Jacot et al. (52) |

|
n.a. |

|

|

|

|

|
no |
| Rechnitzer et al. (53) |

|
n.a. |

|

|

|

|

|
no |
| Bose et al. (54) |

|
n.a. |

|

|

|

|

|
no |
| Rinaldi et al. (55) |

|
n.a. |

|

|

|

|

|
no |
| Chan et al. (56) |

|
n.a. |

|

|

|

|

|
no |
| Huang et al. (57) |

|
n.a. |

|

|

|

|

|
no |
| ||||||||
| [CHART] | ||||||||
Table 5.
Results of the statistical analyses.
| Prevalence of hyponatremia | |||||
|---|---|---|---|---|---|
| Subgroups | RR | 95% CI | P value | Heterogeneity (I ∧ 2) |
P
value of heterogeneity |
| SCLC | 0.24 | [0.18; 0.30] | NS p = 0.425 |
96.56% | <0.001 <0.001 |
| NSCLC | 0.27 | [0.17; 0.39] | 98.57% | ||
| male vs. female | 1.15 | [0.86; 1.53] | NS p = 0.354 |
75.40% | <0.001 |
| <60 years vs. ≥60 years | 0.97 | [0.80; 1.19] | NS p = 0.773 |
0.00% | 0.657 |
| ECOG ≤ 1 vs. ECOG>1 | 0.80 | [0.59; 1.09] | NS p = 0.161 |
70.10% | 0.001 |
| LD vs. ED stage | 1.17 | [0.60; 2.28] | NS p = 0.655 |
87.8% | <0.001 |
| Overall survival rates | |||||
| Hyponatremic vs. Normonatremic patients: | |||||
| 10 months | 0.59 | [0.47; 0.74] | <0.001 | 81.1% | <0.001 |
| 20 months | 0.44 | [0.33; 0.59] | <0.001 | 40.5% | 0.109 |
| Hyponatremic SCLC vs. NSCLC patients at 10 months: | |||||
| SCLC | 0.42 | [0.27; 0.57] | <0.001 | 92.72% | <0.001 |
| NSCLC | 0.27 | [0.12; 0.44] | <0.001 | - | - |
| Corrected vs. Uncorrected hyponatremic patients: | |||||
| 10 months | 1.83 | [1.37; 2.44] | <0.001 | 0.0% | 0.775 |
| 20 months | 2.65 | [0.94; 7.50] | 0.067 | 23.3% | 0.271 |
ES, effect size; RR, risk ratio; CI, confidence interval; SCLC, small cell lung cancer; NSCLC, non-small cell lung cancer; ECOG, Eastern Cooperative Oncology Group performance status; LD, limited disease; ED, extensive disease.
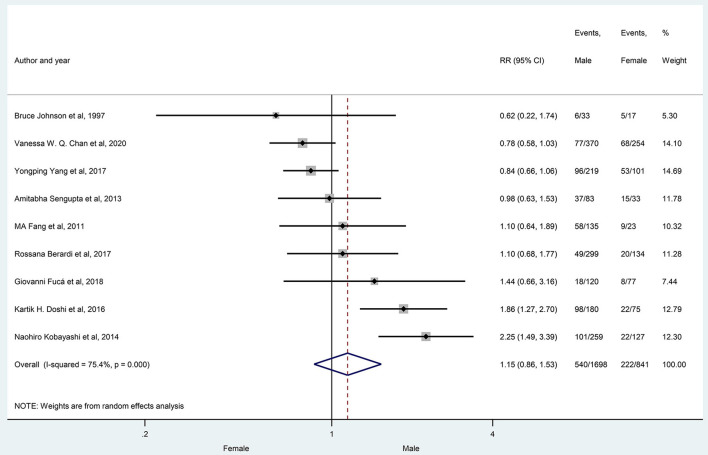
The prevalence of hyponatremia in patients with lung cancer varied between 3 and 94.8% (3–46.6% in SCLC and 5.3–94.8% in NSCLC) with an average of 25% (24% SCLC and 27% NSCLC) in the evaluated reports without significant differences between patients with SCLC vs. patients with NSCLC (29 articles, p = 0.425) (Figure 2), or in the subgroups created by the following criteria: gender (nine articles, p = 0.354), age (three articles, p = 0.773), ECOG state (nine articles, p = 0.161), the extent of disease (four articles, p = 0.655) (Figures 3–6).
Figure 2.
The prevalence of hyponatremia in patients with SCLC and NSCLC. Black diamonds represent the effect and vertical lines of the individual studies show the corresponding 95% CI. The size of the gray squares reflects the weight of a particular study. The blue diamond is the overall or summary effect. The outer edges of the diamonds represent the CIs. SCLC, small cell lung cancer; NSCLC, non-small cell lung cancer; ES, effect size.
Figure 3.
The prevalence of hyponatremia in male and female patients with lung cancer. Black diamonds represent the individual effects of studies, and vertical lines show the corresponding 95% CI. The size of the gray squares reflects the individual weight of a particular study. The blue diamond shows the overall or summary effect. The outer edges of the diamonds represent the CIs. RR, risk ratio.
Figure 6.
The prevalence of hyponatremia in patients with SCLC having limited and extensive disease stage. Black diamonds represent the individual effects of studies, and vertical lines show the corresponding 95% CI. The size of the gray squares reflects the individual weight of a particular study. The blue diamond shows the overall or summary effect. The outer edges of the diamonds represent the Cis. SCLC, small cell lung cancer, RR, risk ratio.
Figure 4.
The prevalence of hyponatremia in young (<60 years) and older patients (> 60 years) with lung cancer. Black diamonds represent the individual effects of studies, and vertical lines show the corresponding 95% CI. The size of the gray squares reflects the individual weight of a particular study. The blue diamond shows the overall or summary effect. The outer edges of the diamonds represent the CIs. RR, risk ratio.
Figure 5.
The prevalence of hyponatremia in patients with lung cancer having ECOG ≤ 1 and ECOG>1 performance state. Black diamonds represent the individual effects of studies, and vertical lines show the corresponding 95% CI. The size of the gray squares reflects the individual weight of a particular study. The blue diamond shows the overall or summary effect. The outer edges of the diamonds represent the CIs. ECOG, Eastern Cooperative Oncology Group performance status, RR, risk ratio.
The meta-analysis of the overall survival rates of lung cancer patients with hyponatremia was performed on eight selected studies (median follow-up time, 40 months), and a subgroup analysis was also carried out, comparing patients with SCLC and NSCLC.
The patients with hyponatremia, in comparison to patients with normonatremia, had significantly lower OS at 10 months (RR, 0.59 with 95% CI:0.47–0.74, p < 0.001) and 20 months (RR, 0.44 with 95% CI:0.33–0.59, p < 0.001), respectively, as shown in Figures 7, 8.
Figure 7.
The overall survival rates at 10 months, comparing patients with hyponatremic and normonatremic lung cancer. Black diamonds represent the individual effects of studies, and vertical lines show the corresponding 95% CI. The size of the gray squares reflects the individual weight of a particular study. The blue diamond shows the overall or summary effect. The outer edges of the diamonds represent the CIs. RR, risk ratio.
Figure 8.
The overall survival rates at 20 months, comparing patients with hyponatremic and normonatremic lung cancer. Black diamonds represent the individual effects of studies, and vertical lines show the corresponding 95% CI. The size of the gray squares reflects the individual weight of a particular study. The blue diamond shows the overall or summary effect. The outer edges of the diamonds represent the CIs. RR, risk ratio.
You can see in Figure 9 that there was a lower overall survival rate at 10 months in patients with hyponatremic NSCLC (RR, 0.27, with 95% CI:0.12–0.44, p < 0.001) compared to patients with hyponatremic SCLC (RR, 0.42, with 95% CI:0.27–0.57, p < 0.001). However, no direct statistical comparison was performed.
Figure 9.
The overall survival rates at 10 months in patients with hyponatremia, comparing patients with small cell and non-small cell lung cancer. Black diamonds represent the individual effects of studies, and vertical lines show the corresponding 95% CI. The size of the gray squares reflects the individual weight of a particular study. The blue diamond shows the overall or summary effect. The outer edges of the diamonds represent the CIs. SCLC, small cell lung cancer, NSCLC, non-small cell lung cancer, RR, risk ratio.
Information about the improvement of overall survival rates, following the correction of hyponatremia, was available in only four studies. In the selected articles, the changes in serum sodium levels were the result of oncologic, symptomatic, or supportive treatment, including at least two cycles of chemotherapy, sodium supplementation, or fluid restriction. After the correction of hyponatremia, the overall survival rates at 10 months were significantly higher when compared with the uncorrected hyponatremia group (RR, 1.83 with 95% CI: 1.37–2.44, p < 0.001) (Figure 10).
Figure 10.
The overall survival rates after correction of hyponatremia at 10 months (corrected and uncorrected hyponatremic patient groups). Black diamonds represent the individual effects of studies, and vertical lines show the corresponding 95% CI. The size of the gray squares reflects the individual weight of a particular study. The blue diamond shows the overall or summary effect. The outer edges of the diamonds represent the CIs. RR, risk ratio.
However, at 20 months, no significant differences were identified between these groups (RR, 2.65 with 95% CI:0.95–7.50, p = 0.067), as shown in Figure 11.
Figure 11.
The overall survival rates after correction of hyponatremia at 20 months (corrected and uncorrected hyponatremic patient groups). Black diamonds represent the individual effects of studies, and vertical lines show the corresponding 95% CI. The size of the gray squares reflects the individual weight of a particular study. The blue diamond shows the overall or summary effect. The outer edges of the diamonds represent the CIs. RR, risk ratio.
Discussion
Regarding the prevalence of hyponatremia in patients with lung cancer, discordant results have been published. In 1 review, the incidence of hyponatremia was suggested to be in the range of 15–75% among patients with lung cancer (3). At the same time, in another one, the prevalence varied in a lower range, from 5 to 36%, with an average of 15% in SCLC, and 4–2% in NSCLC (6). In other reviews, the prevalence of hyponatremia and/or SIADH was even less: Grohe et al. found hyponatremia in 14% of patients with SCLC and 2.7% of NSCLC (7), while Christoforos Efthymiou et al. showed that SIADH was present in 7–16% of patients with SCLC and was rare among patients with NSCLC (1%) (8).
To the best of our knowledge, ours is the first article which is not only a systematic review but also a meta-analysis evaluating the prevalence of hyponatremia in patients with lung cancer. According to our systematic search for the studies available in the literature, the prevalence of hyponatremia varies between 3. and 94.8%, with a pooled prevalence of 25% in patients with lung cancer. However, contrary to the general concept, our comprehensive evaluation and statistical analyses showed no significant difference in the prevalence of hyponatremia between SCLC and NSCLC (24 vs. 27% pooled mean frequency). The prevalence of hyponatremia has not differed significantly in the other subgroups either. So, in contrast to reviews (58, 59) and studies about the general hospitalized populations (60, 61), in patients with lung cancer, the presence of hyponatremia is not associated with age or gender. In respect of ECOG performance status or disease extent, ours is the first meta-analysis to describe that the prevalence of hyponatremia is independent of these parameters in lung cancer. Furthermore, ethnicity seems to be an important factor as well. In the article of K. Doshi and colleagues, ethnicity has been reported to be a risk factor in hyponatremia, but, unfortunately, we did not have enough data to analyze this aspect; therefore, more ethnicity-focused studies are needed (42).
The differences in the reported frequency values of low serum sodium levels in the cited reviews may be caused by the lack of appropriate studies; different eligibility criteria (hyponatremia at admission or during hospitalization); various patient groups (general hospitalized, general cancer, or only patients with SCLC/NSCLC and other subgroups); and different cut-off values (130–136 mmol/L).
Hyponatremia has an overall negative impact on mortality (5, 62–64). In a meta-analysis carried out in various hyponatremic patient groups, a significant association was revealed between hyponatremia and overall mortality (20). However, the 1,47,948 patients with hyponatremia included (from 81 studies) were not only subjects with different solid tumors but also patients with other medical conditions, such as myocardial infarction, heart failure, liver cirrhosis, and pulmonary infections. In a prospective study of patients with cancer, the mortality was higher among the patients with hyponatremic cancer compared to the whole cancer population (19.5 vs. 6.3%) (5). In a systematic review analyzing the prognostic role of hyponatremia in malignancies, including lung cancer, it was found to be a negative prognostic factor (19). In lung cancer, many authors also identified hyponatremia as an independent negative risk factor (30, 32, 34, 35). At the same time, Umemura and colleagues found that vasopressin (AVP) is a better prognostic factor than sodium levels (51). In another systematic review, the causes, diagnosis, management, and the role of hyponatremia were evaluated among patients with lung cancer. They concluded that hyponatremia is a negative prognostic and predictive factor. Furthermore, they highlighted the importance of assessing the serum sodium levels of patients with lung cancer to improve their quality of life (12). We could confirm that hyponatremia is significantly associated with worse overall survival rates in patients with lung cancer.
To the best of our knowledge, this is the first meta-analysis comparing the prognostic significance of pretreatment hyponatremia in patients with NSCLC and SCLC.
According to our analysis, hyponatremia has a more significant negative impact on the overall mortality of patients with NSCLC compared to patients with SCLC, since the overall survival rates at 10 months were significantly lower in patients with hyponatremic NSCLC than in hyponatremic SCLC ones (p < 0.001).
We found only one systematic review in the literature analyzing the independent prognostic role of hyponatremia in lung cancer (19). The authors identified hyponatremia as an independent risk factor in a poor outcome in 6 out of 13 and in 1 out of 3 studies in SCLC and NSCLC, respectively. However, this review has some limitations. In the included studies, hyponatremia was detected both before and during treatment, the overall survival times and rates were only partially available, and the multivariate analysis of hyponatremia was reported only in a few studies (19). In our systematic review, we came to similar conclusions regarding patients with SCLC: overall, out of the 12 studies, six identified hyponatremia as an independent prognostic factor. However, in NSCLC, we found low serum sodium to be a significant independent negative predictor more consistently (N = 10 vs. only one paper where it was not).
We also found another meta-analysis similar to ours after updating our systematic search. In contrast to our article, this meta-analysis evaluated the prognostic significance of hyponatremia in patients with NSCLC only, and it did not analyze the prevalence of hyponatremia and its effect on OS rates. In this systematic research, some low-quality studies with a too small number of data on prevalence and prognostic significance were also included; therefore, we chose not to include these articles in our evaluation. Regardless of the differences, their conclusion was similar to ours. They found that hyponatremia is a frequent electrolyte abnormality among patients with NSCLC, and it increases mortality, more pronounced if it remains uncorrected (65).
The improvement of hyponatremia correlated with mortality in retrospective studies in various medical conditions, including lung cancer (32, 66, 67). However, in some reviews, it was suggested that the impact of hyponatremia correction on mortality should be further investigated because it still seems to be unclear (68–70). In a meta-analysis, including 15 studies with 13,816 patients with hyponatremia, the correction of serum sodium level was achieved in 53.2% of cases, and it was associated with a significant reduction in overall mortality (23). Similarly, we found significantly higher OS rates at 10 months among patients with lung cancer if the hyponatremia was ameliorated compared to the uncorrected hyponatremia group. However, when assessed later, at 20 months, this benefit of the treatment was no longer statistically significant, which is in contrast with the observation of the previous meta-analysis carried out in various medical conditions, where reduced mortality could be detected even at 36 months. This discrepancy between the 10- and 20-month prognostic data may be related to the higher progression rate of lung cancer and, therefore, a shorter beneficial effect of the correction of the sodium level compared to other (malignant) diseases. However, statistical uncertainties (e.g., discrepant ratios of data available and/or loss of patients to follow-up during the time interval) may also influence this observation. In the meta-analysis of Cantini and colleagues, the risk of electrolyte disorders was evaluated among patients with lung cancer treated with immune checkpoint inhibitors (ICIs). Their conclusion was similar to ours; however, they investigated the incidence of treatment-related hyponatremia among patients with lung cancer. They stated that electrolyte disorders should be monitored frequently and corrected promptly because hyponatremia is associated with a worse prognosis (71).
These results show the importance of the correction of hyponatremia and raise awareness about further therapeutic options in treatment-resistant cases. Numerous papers highlighted the beneficial effect of V2R antagonists (mostly Tolvaptan), with close monitoring of sodium levels in the treatment of patients with hyponatremic lung cancer (72–78). These studies reported effective correction of hyponatremia and improvement of performance status, without any serious adverse events. However, according to our systematic search, in patients with lung cancer, the effect of V2R antagonist treatment on mortality has only been evaluated in one study, which also demonstrated the cost-effectiveness of tolvaptan treatment in Swedish patients with SIADH in pneumonia or SCLC (79). In a review of Fiordoliva et al., they found that SIADH is frequently associated with SCLC, but it should be expected in other lung cancer histotypes as well (12). We plan to investigate the diagnostic and therapeutic approaches of hyponatremia caused by SIADH in patients with lung cancer in the future.
Limitations
The most significant limitations of our meta-analysis are the following: the lack of randomized controlled trials, the low number of prospective studies, the limited number of studies available in respect of certain relevant questions, the considerable heterogeneity of the included studies, and the variable cut-off values for serum sodium levels, which all can lead to biased results. The heterogeneity among methods of measuring serum sodium is also a limiting factor. The severity of hyponatremia at the time of diagnosis could potentially have been influenced by some interfering factors, which were not fully evaluated, such as comorbidities and treatments. Also, the lack of randomization between uncorrected and corrected hyponatremia may lead to bias.
From the available data, no certain answer can be given to the question of whether the negative prognostic significance of resistant hyponatremia was due to oncological unresponsiveness or inappropriate sodium correction. Furthermore, incomplete data reporting of follow-up time and loss of the patients to follow-up, as well as the demographic characteristics of the patients with hyponatremia have also been important limitations, e.g., lack of information about demographic characteristics of patients with hyponatremia may distort the results. More data for a better understanding would be desirable.
Conclusions
In the literature, the prevalence of hyponatremia in patients with lung cancer varies between 3 and 94.8% with a pooled mean frequency of 25%. In our meta-analysis, no significant differences could be observed in the prevalence of hyponatremia among various subgroups, including SCLC vs. NSCLC.
Hyponatremia significantly and negatively influences the overall survival rates of these patients, especially in NSCLC where data are more consistent. Moreover, the improvement of serum sodium levels by specific or symptomatic treatment may improve the survival rates, at least in the short term.
Overall, the contradictions in the literature suggest that better-designed studies are necessary to assess the prevalence and prognostic significance of hyponatremia.
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
Author Contributions
MG, EB, BT, SK, MF, PH, ZS, EM, and LB: conception and design. EB and MS: administrative support. PH, EM, and LB: provision of study materials or patients. EB and MG: collection and assembly of data. EB, MG, and DN: data analysis and interpretation. MS: manuscript writing. MS: manuscript writing. All authors: funding acquisition. All authors read and approved the final manuscript.
Funding
This study was funded by GINOP-2.3.2-15-2016-00048 - STAY ALIVE co-financed by the European Union (European Regional Development Fund) within the framework of Programme Széchenyi 2020 and by the Human Resources Development Operational Programme Grant, Grant Number: EFOP 3.6.2-16-2017-00006 – LIVE LONGER, which is co-financed by the European Union (European Regional Development Fund) within the framework of Programme Széchenyi 2020.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmed.2021.671951/full#supplementary-material
A preferred reporting items for systematic reviews and meta-analyses (PRISMA) checklist.
A Quality in Prognostic Studies (QUIPS) tool for risk of bias assessment.
References
- 1.World Health Organization. Cancer; c2020. Available online at: https://www.who.int/news-room/fact-sheets/detail/cancer (accessed April 7, 2020).
- 2.World Health Organization . WHO GLOBOCAN; c2018. Cancer Tomorrow. Available online at: https://gco.iarc.fr/tomorrow/graphic-line?type=0&type_sex=0&mode=population&sex=0&populations=900&cancers=15&age_group=value&apc_male=0&apc_female=0&single_unit=500000&print=0 (accessed April 7, 2020).
- 3.Bowman BT. Electrolyte disorders associated with cancer. J Onco-Nephrol. (2017) 1:30–5. 10.5301/jo-n.5000004 [DOI] [Google Scholar]
- 4.Berardi R, Antonuzzo A, Blasi L, Buosi R, Lorusso V, Migliorino MR, et al. Practical issues for the management of hyponatremia in oncology. Endocrine. (2018) 61:158–64. 10.1007/s12020-018-1547-y [DOI] [PubMed] [Google Scholar]
- 5.Berghmans T, Paesmans M, Body JJ. A prospective study on hyponatraemia in medical cancer patients: epidemiology, aetiology and differential diagnosis. Support Care Cancer. (2000) 8:192–7. 10.1007/s005200050284 [DOI] [PubMed] [Google Scholar]
- 6.Sørensen JB, Andersen MK, Hansen HH. Syndrome of inappropriate secretion of antidiuretic hormone (SIADH) in malignant disease. J Intern Med. (1995) 238:97–110. 10.1111/j.1365-2796.1995.tb00907.x [DOI] [PubMed] [Google Scholar]
- 7.Grohé C, Berardi R, Burst V. Hyponatraemia–SIADH in lung cancer diagnostic and treatment algorithms. Crit Rev Oncol Hematol. (2015) 96:1–8. 10.1016/j.critrevonc.2015.04.005 [DOI] [PubMed] [Google Scholar]
- 8.Efthymiou C, Spyratos D, Kontakiotis T. Endocrine paraneoplastic syndromes in lung cancer. Hormones (Athens: ). (2018) 17:351–8. 10.1007/s42000-018-0046-0 [DOI] [PubMed] [Google Scholar]
- 9.Wang X, Liu M, Zhang L, Ma K. Syndrome of inappropriate antidiuretic hormone secretion: a poor prognosis in small-cell lung cancer. Arch Med Res. (2016) 47:19–24. 10.1016/j.arcmed.2015.12.006 [DOI] [PubMed] [Google Scholar]
- 10.List AF, Hainsworth JD, Davis BW, Hande KR, Greco FA, Johnson DH. The syndrome of inappropriate secretion of antidiuretic hormone (SIADH) in small-cell lung cancer. J Clin Oncol. (1986) 4:1191–8. 10.1200/JCO.1986.4.8.1191 [DOI] [PubMed] [Google Scholar]
- 11.Lokich JJ. The frequency and clinical biology of the ectopic hormone syndromes of small cell carcinoma. Cancer. (1982) 50:2111–4. [DOI] [PubMed] [Google Scholar]
- 12.Fiordoliva I, Meletani T, Baleani MG, Rinaldi S, Savini A, Di Pietro Paolo M, Berardi R. Managing hyponatremia in lung cancer: latest evidence and clinical implications. Ther Adv Med Oncol. (2017) 9:711–9. 10.1177/1758834017736210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dimitriadis GK, Angelousi A, Weickert MO, Randeva HS, Kaltsas G, Grossman A. Paraneoplastic endocrine syndromes. Endocr Relat Cancer. (2017) 24:R173–90. 10.1530/ERC-17-0036 [DOI] [PubMed] [Google Scholar]
- 14.Raftopoulos H. Diagnosis and management of hyponatremia in cancer patients. Support Care Cancer. (2007) 15:1341–7. 10.1007/s00520-007-0309-9 [DOI] [PubMed] [Google Scholar]
- 15.Liamis G, Megapanou E, Elisaf M, Milionis H. Hyponatremia-Inducing Drugs. Front Horm Res. (2019) 52:167–77. 10.1159/000493246 [DOI] [PubMed] [Google Scholar]
- 16.Oren RM. Hyponatremia in congestive heart failure. Am J Cardiol. (2005) 95(9A):2B–7B. 10.1016/j.amjcard.2005.03.002 [DOI] [PubMed] [Google Scholar]
- 17.Ginès P, Guevara M. Hyponatremia in cirrhosis: pathogenesis, clinical significance, and management. Hepatology. (2008) 48:1002–10. 10.1002/hep.22418 [DOI] [PubMed] [Google Scholar]
- 18.Peri A. Prognostic and predictive role of hyponatremia in cancer patients. J Cancer Metastasis Treat. (2019) 5:40. 10.20517/2394-4722.2019.1430406916 [DOI] [Google Scholar]
- 19.Castillo JJ, Vincent M, Justice E. Diagnosis and management of hyponatremia in cancer patients. Oncologist. (2012) 17:756–65. 10.1634/theoncologist.2011-0400 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Corona G, Giuliani C, Parenti G, Norello D, Verbalis JG, Forti G, et al. Moderate hyponatremia is associated with increased risk of mortality: evidence from a meta-analysis. PLoS ONE. (2013) 8:e80451. 10.1371/journal.pone.0080451 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Verbalis JG. Managing hyponatremia in patients with syndrome of inappropriate antidiuretic hormone secretion. J Hosp Med. (2010) 5 (Suppl 3):S18–26. 10.1002/jhm.783 [DOI] [PubMed] [Google Scholar]
- 22.Harper PG, Souhami RL, Spiro SG, Geddes DM, Guimaraes M, Fearon F, Smyth JF. Tumor size, response rate, and prognosis in small cell carcinoma of the bronchus treated by combination chemotherapy. Cancer Treat Rep. (1982) 66:463–70. [PubMed] [Google Scholar]
- 23.Corona G, Giuliani C, Verbalis JG, Forti G, Maggi M, Peri A. Hyponatremia improvement is associated with a reduced risk of mortality: evidence from a meta-analysis. PLoS ONE. (2015) 10:e0124105. 10.1371/journal.pone.0124105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Moher D, Liberati A, Tetzlaff J, Altman DG. The PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA Statement. PLoS Med. (2009) 6:e1000097. 10.1371/journal.pmed.1000097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hayden JA, Côté P, Bombardier C. Evaluation of the quality of prognosis studies in systematic reviews. Ann Intern Med. (2006) 144:427–37. 10.7326/0003-4819-144-6-200603210-00010 [DOI] [PubMed] [Google Scholar]
- 26.Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al. Cochrane Handbook for Systematic Reviews of Interventions. 2nd ed. Chichester: John Wiley & Sons. (2019). 10.1002/9781119536604 [DOI] [Google Scholar]
- 27.Yang Y, Sun N, Sun P, Zhang L. Clinical characteristics and prognosis of elderly small cell lung cancer patients complicated with hyponatremia: a retrospective analysis. Anticancer Res. (2017) 37:4681–6. 10.21873/anticanres.11872 [DOI] [PubMed] [Google Scholar]
- 28.Osterlind K, Hansen HH, Dombernowsky P, Hansen M, Andersen PK. Determinants of complete remission induction and maintenance in chemotherapy with or without irradiation of small cell lung cancer. Cancer Res. (1987) 47:2733–6. [PubMed] [Google Scholar]
- 29.Zarzecka M, Kubicki P, Kozielski J. Hyponatraemia - evaluation of prevalence in patients hospitalized in the Pulmonary Department and prognostic significance in lung cancer patients. Pneumonol Alergol Pol. (2014) 82:18–24. 10.5603/PiAP.2014.0004 [DOI] [PubMed] [Google Scholar]
- 30.Hermes A, Waschki B, Reck M. Hyponatremia as prognostic factor in small cell lung cancer–a retrospective single institution analysis. Respir Med. (2012) 106:900–4. 10.1016/j.rmed.2012.02.010 [DOI] [PubMed] [Google Scholar]
- 31.Wang W, Song Z, Zhang Y. Hyponatremia in small cell lung cancer is associated with a poorer prognosis. Transl Cancer Res. (2016) 5:36–43. 10.3978/j.issn.2218-676X.2016.01.04 [DOI] [Google Scholar]
- 32.Berardi R, Santoni M, Newsom-Davis T, Caramanti M, Rinaldi S, Tiberi M, et al. Hyponatremia normalization as an independent prognostic factor in patients with advanced non-small cell lung cancer treated with first-line therapy. Oncotarget. (2017) 8:23871–9. 10.18632/oncotarget.13372 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sengupta A, Banerjee SN, Biswas NM, Jash D, Saha K, Maji A, et al. The incidence of hyponatraemia and its effect on the ECOG performance status among lung cancer patients. J Clin Diagn Res. (2013) 7:1678–82. 10.7860/JCDR/2013/5900.3225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kobayashi N, Usui S, Yamaoka M, Suzuki H, Kikuchi S, Goto Y, et al. The influence of serum sodium concentration on prognosis in resected non-small cell lung cancer. Thorac Cardiovasc Surg. (2014) 62:338–43. 10.1055/s-0033-1359713 [DOI] [PubMed] [Google Scholar]
- 35.Fucà G, Galli G, Poggi M, Lo Russo G, Proto C, Imbimbo M, et al. Low baseline serum sodium concentration is associated with poor clinical outcomes in metastatic non-small cell lung cancer patients treated with immunotherapy. Target Oncol. (2018) 13:795–800. 10.1007/s11523-018-0599-5 [DOI] [PubMed] [Google Scholar]
- 36.Alamoudi OS. Lung cancer at a University Hospital in Saudi Arabia: a four-year prospective study of clinical, pathological, radiological, bronchoscopic, and biochemical parameters. Ann Thorac Med. (2010) 5:30–6. 10.4103/1817-1737.58957 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hansen O, Sørensen P, Hansen KH. The occurrence of hyponatremia in SCLC and the influence on prognosis: a retrospective study of 453 patients treated in a single institution in a 10-year period. Lung Cancer. (2010) 68:111–4. 10.1016/j.lungcan.2009.05.015 [DOI] [PubMed] [Google Scholar]
- 38.Svaton M, Fiala O, Pesek M, Bruha F, Mukensnabl P, Racek, et al. Predictive and prognostic significance of sodium levels in patients with NSCLC treated by erlotinib. Anticancer Res. (2014) 34:7461–5. [PubMed] [Google Scholar]
- 39.Allan SG, Stewart ME, Love S, Cornbleet MA, Smyth JF, Leonard RC. Prognosis at presentation of small cell carcinoma of the lung. Eur J Cancer. (1990) 26:703–5. 10.1016/0277-5379(90)90121-9 [DOI] [PubMed] [Google Scholar]
- 40.Li W, Chen X, Wang L, Wang Y, Huang C, Wang G, et al. The prognostic effects of hyponatremia and hyperchloremia on postoperative NSCLC patients. Curr Probl Cancer. (2019) 43:402–10. 10.1016/j.currproblcancer.2018.12.006 [DOI] [PubMed] [Google Scholar]
- 41.Osterlind K, Andersen PK. Prognostic factors in small cell lung cancer: multivariate model based on 778 patients treated with chemotherapy with or without irradiation. Cancer Res. (1986) 46:4189–94. [PubMed] [Google Scholar]
- 42.Doshi KH, Shriyan B, Nookala MK, Kannan S, Joshi A, Noronha V, et al. Prognostic significance of pretreatment sodium levels in patients of nonsmall cell lung cancer treated with pemetrexed-platinum doublet chemotherapy. J Cancer Res Ther. (2018) 14:1049–53. 10.4103/0973-1482.187296 [DOI] [PubMed] [Google Scholar]
- 43.Johnson BE, Chute JP, Rushin J, Williams J, Le PT, Venzon D, et al. prospective study of patients with lung cancer and hyponatremia of malignancy. Am J Respir Crit Care Med. (1997) 156:1669–78. 10.1164/ajrccm.156.5.96-10075 [DOI] [PubMed] [Google Scholar]
- 44.Sagman U, Maki E, Evans WK, Warr D, Shepherd FA, Sculier JP, et al. Small-cell carcinoma of the lung: derivation of a prognostic staging system. J Clin Oncol. (1991) 9:1639–49. 10.1200/JCO.1991.9.9.1639 [DOI] [PubMed] [Google Scholar]
- 45.Jacot W, Colinet B, Bertrand D, Lacombe S, Bozonnat MC, Daurès JP, et al. Quality of life and comorbidity score as prognostic determinants in non-small-cell lung cancer patients. Ann Oncol. (2008) 19:1458–64. 10.1093/annonc/mdn064 [DOI] [PubMed] [Google Scholar]
- 46.Hong X, Xu Q, Yang Z, Wang M, Yang F, Gao Y, et al. The value of prognostic factors in Chinese patients with small cell lung cancer: a retrospective study of 999 patients. Clin Respir J. (2018) 12:433–47. 10.1111/crj.12534 [DOI] [PubMed] [Google Scholar]
- 47.Maestu I, Pastor M, Gómez-Codina J, Aparicio J, Oltra A, Herranz C, et al. Pretreatment prognostic factors for survival in small-cell lung cancer: a new prognostic index and validation of three known prognostic indices on 341 patients. Ann Oncol. (1997) 8:547–53. 10.1023/A:1008212826956 [DOI] [PubMed] [Google Scholar]
- 48.Kawahara M, Fukuoka M, Saijo N, Nishiwaki Y, Ikegami H, Tamura T, et al. Prognostic factors and prognostic staging system for small cell lung cancer. Jpn J Clin Oncol. (1997) 27:158–65. 10.1093/jjco/27.3.158 [DOI] [PubMed] [Google Scholar]
- 49.Cerny T, Blair V, Anderson H, Bramwell V, Thatcher N. Pretreatment prognostic factors and scoring system in 407 small-cell lung cancer patients. Int J Cancer. (1987) 39:146–9. 10.1002/ijc.2910390204 [DOI] [PubMed] [Google Scholar]
- 50.Ma F, Liu X, Hu C, Huang M. [Clinical features and prognosis analysis of small-cell lung cancer complicated with hyponatremia]. Zhong Nan Da Xue Xue Bao Yi Xue Ban. (2011) 36:64–7. 10.3969/j.issn.1672-7347.2011.01.010 [DOI] [PubMed] [Google Scholar]
- 51.Umemura S, Segawa Y, Ueoka H, Hotta K, Kiura K, Takigawa N, et al. Serum level of arginine-vasopressin influences the prognosis of extensive-disease small-cell lung cancer. J Cancer Res Clin Oncol. (2007) 133:519–24. 10.1007/s00432-007-0196-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Jacot W, Quantin X, Boher JM, Andre F, Moreau L, Gainet M, et al. Association d'Enseignement et de Recherche des Internes en Oncologie. Brain metastases at the time of presentation of non-small cell lung cancer: a multi-centric AERIO analysis of prognostic factors. Br J Cancer. (2001) 84:903–9. bjoc.2000.1706 10.1054/bjoc.2000.1706 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Rechnitzer S, Bülzebruck H, Ebert W, Drings P. Pretreatment Prognostic Factors for Survival in Non-Small-Cell Lung Cancer: A Multivariate Analysis of 229 Patients Onkologie. Oncol Res Treat. (1998) 21:204–10. 10.1159/000026816 [DOI] [Google Scholar]
- 54.Bose CK, Dey S, Mukhopadhyay A. Hyponatremia of non-small cell lung cancer: Indian experience. Indian J Med Paediatr Oncol. (2011) 32:139–142. 10.4103/0971-5851.92810 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Rinaldi S, Santoni M, Leoni G, Fiordoliva I, Marcantognini G, Meletani T, et al. The prognostic and predictive role of hyponatremia in patients with advanced non-small cell lung cancer (NSCLC) with bone metastases. Support Care Cancer. (2019) 27:1255–61. 10.1007/s00520-018-4489-2 [DOI] [PubMed] [Google Scholar]
- 56.Chan VWQ, Henry MT, Kennedy MP. Hyponatremia and hypercalcemia: a study of a large cohort of patients with lung cancer. Transl Cancer Res. (2020) 9:222–30. 10.21037/tcr.2019.12.72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Huang LL, Hu XS, Wang Y, Li JL, Wang HY, Liu P, et al. Survival and pretreatment prognostic factors for extensive-stage small cell lung cancer: a comprehensive analysis of 358 patients. Thorac Cancer. (2021) 12:1943–51. 10.1111/1759-7714.13977 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Upadhyay A, Jaber BL, Madias NE. Incidence and prevalence of hyponatremia. Am J Med. (2006) 119 (7 Suppl 1):S30–5. 10.1016/j.amjmed.2006.05.005 [DOI] [PubMed] [Google Scholar]
- 59.Peri A. Management of hyponatremia: causes, clinical aspects, differential diagnosis and treatment. Expert Rev Endocrinol Metab. (2019) 14:13–21. 10.1080/17446651.2019.1556095 [DOI] [PubMed] [Google Scholar]
- 60.Hawkins RC. Age and gender as risk factors for hyponatremia and hypernatremia. Clin Chim Acta. (2003) 337 (1–2):169–72. 10.1016/j.cccn.2003.08.001 [DOI] [PubMed] [Google Scholar]
- 61.Mohan S, Gu S, Parikh A, Radhakrishnan J. Prevalence of hyponatremia and association with mortality: results from NHANES. Am J Med. (2013) 126:1127–37.e1. 10.1016/j.amjmed.2013.07.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Sterns RH, Silver SM. Complications and management of hyponatremia. Curr Opin Nephrol Hypertens. (2016) 25:114–119. 10.1097/MNH.0000000000000200 [DOI] [PubMed] [Google Scholar]
- 63.Verbalis JG, Goldsmith SR, Greenberg A, Korzelius C, Schrier RW, Sterns RH, et al. Diagnosis, evaluation, and treatment of hyponatremia: expert panel recommendations. Am J Med. (2013) 126 (10 Suppl 1):S1–42. 10.1016/j.amjmed.2013.07.006 [DOI] [PubMed] [Google Scholar]
- 64.Kraft MD, Btaiche IF, Sacks GS, Kudsk KA. Treatment of electrolyte disorders in adult patients in the intensive care unit. Am J Health Syst Pharm. (2005) 62:1663–82. 10.2146/ajhp040300 [DOI] [PubMed] [Google Scholar]
- 65.Sandfeld-Paulsen B, Aggerholm-Pedersen N, Winther-Larsen A. Hyponatremia as a prognostic factor in non-small cell lung cancer: a systematic review and meta-analysis. Transl Lung Cancer Res. (2021) 10:651–61. 10.21037/tlcr-20-877 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Petereit C, Zaba O, Teber I, Grohé C. Ist die Hyponatriämie (HN) ein Prognosefaktor für das Gesamtüberleben der Patienten mit Lungenkarzinom? [Is hyponatremia a prognostic marker of survival for lung cancer?]. Pneumologie. (2011) 65:565–71. 10.1055/s-0030-1256668 [DOI] [PubMed] [Google Scholar]
- 67.Balachandran K, Okines A, Gunapala R, Morganstein D, Popat S. Resolution of severe hyponatraemia is associated with improved survival in patients with cancer. BMC Cancer. (2015) 15:163. 10.1186/s12885-015-1156-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Sun L, Hou Y, Xiao Q, Du Y. Association of serum sodium and risk of all-cause mortality in patients with chronic kidney disease: a meta-analysis and sysematic review. Sci Rep. (2017) 7:15949. 10.1038/s41598-017-16242-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hoorn EJ, Zietse R. Diagnosis and treatment of hyponatremia: compilation of the guidelines. J Am Soc Nephrol. (2017) 28:1340–9. 10.1681/ASN.2016101139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Rusinaru D, Tribouilloy C, Berry C, Richards AM, Whalley GA, Earle N, et al. Relationship of serum sodium concentration to mortality in a wide spectrum of heart failure patients with preserved and with reduced ejection fraction: an individual patient data meta-analysis(†): Meta-Analysis Global Group in Chronic heart failure (MAGGIC). Eur J Heart Fail. (2012) 14:1139–46. 10.1093/eurjhf/hfs099 [DOI] [PubMed] [Google Scholar]
- 71.Cantini L, Merloni F, Rinaldi S, Lenci E, Marcantognini G, Meletani T, et al. Electrolyte disorders in advanced non-small cell lung cancer patients treated with immune check-point inhibitors: a systematic review and meta-analysis. Crit Rev Oncol Hematol. (2020) 151:102974. 10.1016/j.critrevonc.2020.102974 [DOI] [PubMed] [Google Scholar]
- 72.Dixon MB, Lien YH. Tolvaptan and its potential in the treatment of hyponatremia. Ther Clin Risk Manag. (2008) 4:1149–1155. 10.2147/TCRM.S3115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Thajudeen B, Salahudeen AK. Role of tolvaptan in the management of hyponatremia in patients with lung and other cancers: current data and future perspectives. Cancer Manag Res. (2016) 8:105–114. 10.2147/CMAR.S90169 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Fraser IM, Brooke AM, Dorey NE, Toy EW. Use of tolvaptan, a new V2 receptor antagonist, in the treatment of syndrome of inappropriate antidiuretic hormone secretion (SIADH) secondary to small cell lung cancer (SCLC) – a case series. Lung Cancer. (2014) 83:S69. 10.1016/S0169-5002(14)70187-4 [DOI] [Google Scholar]
- 75.Petereit C, Zaba O, Teber I, Lüders H, Grohé C. A rapid and efficient way to manage hyponatremia in patients with SIADH and small cell lung cancer: treatment with tolvaptan. BMC Pulm Med. (2013) 13:55. 10.1186/1471-2466-13-55 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Li-Ng M, Verbalis JG. Conivaptan: evidence supporting its therapeutic use in hyponatremia. Core Evid. (2010) 4:83–92. 10.2147/CE.S5997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Zeltser D, Rosansky S, van Rensburg H, Verbalis JG, Smith N, Conivaptan Study Group . Assessment of the efficacy and safety of intravenous conivaptan in euvolemic and hypervolemic hyponatremia. Am J Nephrol. (2007) 27:447–457. 10.1159/000106456 [DOI] [PubMed] [Google Scholar]
- 78.Annane D, Decaux G, Smith N, Conivaptan Study Group . Efficacy and safety of oral conivaptan, a vasopressin-receptor antagonist, evaluated in a randomized, controlled trial in patients with euvolemic or hypervolemic hyponatremia. Am J Med Sci. (2009) 337:28–36. 10.1097/MAJ.0b013e31817b8148 [DOI] [PubMed] [Google Scholar]
- 79.Jamookeeah C, Robinson P, O'Reilly K, Lundberg J, Gisby M, Ländin M, et al. Cost-effectiveness of tolvaptan for the treatment of hyponatraemia secondary to syndrome of inappropriate antidiuretic hormone secretion in Sweden. BMC Endocr Disord. (2016) 16:22. 10.1186/s12902-016-0104-z [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
A preferred reporting items for systematic reviews and meta-analyses (PRISMA) checklist.
A Quality in Prognostic Studies (QUIPS) tool for risk of bias assessment.
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.