Abstract
Trigeminal neuralgia associated with brainstem lesions is currently considered as a rare condition and only few patients have been reported so far in literature. Tohyama and colleagues recently proposed the nosological entity of trigeminal neuralgia associated with solitary pontine lesion, trying to categorize it as a new clinical syndrome on its own. Based on this description, trigeminal neuralgia associated with solitary pontine lesion patients have an identical clinical presentation compared to other patients with trigeminal neuralgia but have a solitary pontine lesion. The nature of the pontine lesion has been attributed to several etiologies, including ischemia, demyelination or previous pontine viral neuritis. In those patients with a putative demyelinating lesion, a definite diagnosis of multiple sclerosis cannot be made due to the lack of dissemination in space. Very little is known in relation to the cerebrospinal fluid characteristics of this population of patients.
We present a case of a 42-year-old man suffering of trigeminal neuralgia associated with solitary pontine lesion with a possible demyelinating etiology. The patient herein described had an atypical trigeminal neuralgia associated with a single pontine lesion. The MRI characteristics of the lesion, along with the presence of oligoclonal bands in the cerebrospinal fluid, suggested a demyelinating etiology. Trigeminal neuralgia associated with a solitary pontine lesion may be categorized as a possible manifestation of solitary sclerosis. Future research need to reveal which features can predict the risk of conversion to clinically defined multiple sclerosis and which treatments modify this risk.
Keywords: solitary sclerosis, multiple sclerosis, demyelination, trigeminal neuralgia
Introduction
Trigeminal neuralgia (TN) associated with brainstem lesions is currently considered as a rare condition. 1 In fact, only a total of 13 cases of TN patients with brainstem lesions were reported2-7 until the recent paper by Tohyama and colleagues who analyzed 24 patients affected by TN associated with solitary pontine lesion (SPL-TN), trying to categorize it as a new clinical syndrome on its own. 1 Based on this description, SPL-TN patients have an identical clinical presentation compared to other patients with TN but they have a solitary pontine lesion. The lesions are primarily found along the trigeminal pontine pathway, especially within the area of the trigeminal brainstem sensory nuclear complex. The etiological diagnosis of these patients can be difficult in some cases and very little is known in relation to the cerebrospinal fluid (CSF) characteristics of this population of patients.
We present a case of a 42-year-old man presenting with trigeminal neuralgia associated with a solitary pontine lesion with a possible demyelinating etiology.
Case Description
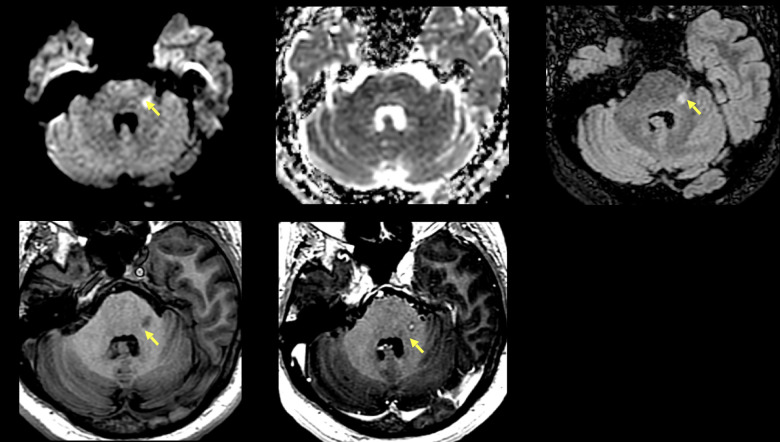
A 42-year-old Caucasian man presented to the emergency department with continuous left facial aching and burning pain. The symptoms started 7 days before the admission and gradually worsened. His past medical history was unremarkable and he was on no regular medication. He was admitted to the Neurology Unit and neurological examination documented paresthesia in the territory of the second and third branch of left trigeminal nerve. Brain and spinal cord MRI revealed only a single, distinct left enhancing pontine lesion in close proximity to the emergence of the trigeminal nerve (Figure 1). Axial T2 FLAIR showed a well-demarcated, hyperintense lesions in the left ventrolateral pons located between the trigeminal root entry zone and the trigeminal nuclei, showing Gadolinium enhancement in T1 sequences. No further supratentorial or spinal cord lesion was evident. Blood examination, including autoimmune screening, research for neurotropic viruses, cell based assays testing anti-aquaporin-4 and anti-myelin oligodendrocyte glycoprotein antibodies were all negative. CSF analysis demonstrated 7 unmatched oligoclonal bands with no cells and normal protein concentration. Given the putative demyelinating etiology of the lesion and absence of any evidence of dissemination in space, the patient was diagnosed with clinically isolated syndrome (CIS) and started on high dose intravenous steroids (IV methylprednisolone 1 g/day for 5 days) with almost complete and sustained benefit. A new brain and spinal cord MRI repeated after one month showed a significant reduction of contrast enhancement (Figure 2). After 6 months his neurological examination did not show any progression and he did not report any further clinical event.
Figure 1.
MRI images at symptom onset (Figure 1a-e) and 1 month later (figure 1d-f).##The figure shows the distinct left demyelinating pontine lesion in axial DWI (1a), ADC maps (1b), 3D - CUBE T2 FLAIR (1c), BRAVO (1d) and post-contrast T1-weighted (1e) sequences.
Figure 2.
MRI images one month later (figure 1d-f). the figure shows the evolution of the distinct left demyelinating pontine lesion in axial DWI (1a), ADC maps (1b), 3D - CUBE T2 FLAIR (1c), BRAVO (1d) and post-contrast T1-weighted (1e) sequences.
Discussion
The new clinical syndrome termed SPL-TN was recently described by Tohyama and colleagues. 1 These authors reported a case series of 24 patients experiencing TN with classical clinical features and a solitary pontine lesion not fulfilling the 2017 revised McDonald criteria for the diagnosis of MS. 8 The patients reported by Tohyama and colleagues showed brainstem lesions along the affected trigeminal pontine pathway, being the trigeminal brainstem sensory nuclear complex the area showing the highest frequency of involvement. 1 The trigeminal brainstem sensory nuclear complex is made up by the main trigeminal sensory nucleus and the spinal trigeminal nucleus. The impairment of inhibitory mechanisms within the spinal trigeminal nucleus, which contains the vast majority of synapses of nociceptive fibers (Adelta and C fibers) which mediates cranial pain sensation, is thought to be responsible for the paroxysmal pain in TN.9,10
SPL-TN represents a peculiar diagnostic entity, also due to the lack of response to surgical treatment. 1 Tohyama et al. compared the characteristics of demyelinating lesions due to MS and SPL-TN lesions. They found that, on conventional MRI, MS plaques show a more diffuse and less demarcated shape as compared to SPL-TN lesions. Using tractography, and analyzing only the voxels where the trigeminal brainstem tracts were present within the SPL-TN lesion or MS plaque, all diffusivity metrics showed significant differences. By using diffusion tensor imaging (DTI) analyses of the SPL-TN lesions an abnormal brainstem trigeminal fiber microstructure within SPL-TN lesions was demonstrated, but not involving the whole lesion itself, suggesting these peculiar DTI characteristics are typical of SPL-TN and possibly different from classic demyelinating MS lesions. 1 SPL-TN can have multiple different underlying disease mechanisms, in fact the etiology of these lesions has been attributed to several conditions, including ischemia,2,4,5 demyelination 7 or previous pontine viral neuritis. 6
The patient herein described had an atypical TN associated with a single pontine lesion with putative demyelinating etiology. 1 In fact, the MRI characteristics of the lesion and their evolution, along with the presence of CSF oligoclonal bands, suggested a demyelinating etiology, albeit the MRI did not show any evidence of dissemination in space. In this patient a diagnosis of solitary sclerosis (SS) could be made. When we compare the patients with SPL-TN described by Tohyama and colleagues and the patient herein described, it is important to note that many of the patients described by Tohyama and colleagues have been followed with serial MR images and there was no change in the shape and size of the lesion, whereas the case herein described showed some change in the size of the lesion with time, along with a reduction in Gadolinium enhancement. Differences and common features of TN secondary to multiple sclerosis and classic and idiopathic TN 11 are summarized in Table 1.
Table 1.
Differences and Common Features of Trigeminal Neuralgia Secondary to Multiple Sclerosis and Classic and Idiopathic Trigeminal Neuralgia.
| TN secondary to MS | classical and idiopathic TN | |
|---|---|---|
| Pain characteristics | sudden, usually unilateral, brief, stabbing or electrical shock-like, recurrent pain | |
| distribution | consistent with one or more divisions of the fifth cranial nerve | |
| Duration of paroxysmal attacks | from a fraction of a second to 2 min | |
| Triggers | typically evoked by stimulating cutaneous or mucous trigeminal territories, but spontaneous attacks are possible too | |
| frequency of the pain attacks | from 1 to over 50 a day | |
| Remission | lack of general consensus about the occurrence of remission periods | pain-free intervals of often complete remission lasting from weeks to years |
| Symptoms between the paroxysms | Some patients may experience also concomitant continuous, dull, burning, or tingling pain | |
| Gender differences | more common in women than in men | |
| Side differences | right side is more frequently involved than the left side | |
| Age at onset | Earlier (40 to 50 years) | After 50 years |
| Bilateral pain | 18% | Uncommon |
| Sensory deficits | About 37% | Uncommon |
TN, trigeminal neuralgia; MS, multiple sclerosis.
SS is also considered a rare entity, clinically characterized by a progressive course and radiologically by a single focal demyelinating lesion within the central nervous system, involving the cerebral white matter, the cervicomedullary junction or the spinal cord.12,13 Nevertheless, patients with a solitary white matter inflammatory lesion without a progressive course exist and have been described. 14 The term SS has been recently used to describe patients with CIS, if at the time of the clinical event, the patient has a lesion consistent with MS that cannot be explained by any other pathology, but does not fulfill the MS imaging criteria. 15 This definition takes into account that SS patients can definitely evolve to MS, or may also evolve into a progressive disease (with no clinical relapses), named “progressive SS”. The pathophysiology of SS is poorly understood but it certainly falls within the so-called “clinical-radiologic paradox” of MS, pointing out how in these patients there is a lack of correspondence between clinical progression and radiological extent of involvement.13,16 Moreover, it is not known which factors favor the development of new MRI lesions in these patients, which can ultimately lead to the fulfillment of the current MS diagnostic criteria. Interestingly, this can happen also several years after the onset of the SS lesion. 12
On the basis of this definition of SS, SPL-TN may be categorized as a possible manifestation of SS. Future research need to reveal which features can predict the risk of conversion to clinically defined MS in patients with SPL-TN with a putative demyelinating pathology and which treatments can modify this risk.
Footnotes
Authors’ Note: Informed consent was obtained from the patient.
Declaration of Conflicting Interests: The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iD
Domenico Plantone  https://orcid.org/0000-0001-6666-7244
https://orcid.org/0000-0001-6666-7244
References
- 1.Tohyama S, Hung PS, Cheng JC, Zhang JY, Halawani A. Trigeminal neuralgia associated with a solitary pontine lesion: clinical and neuroimaging definition of a new syndrome. Pain. 2020;161(5):916–925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Peker S, Akansel G, Sun I, Pamir NM. Trigeminal neuralgia due to pontine infarction. Headache. 2004;44(10):1043–1045. doi:10.1111/j.1526-4610.2004.4200_1.x [DOI] [PubMed] [Google Scholar]
- 3.Neetu S, Sunil K, Ashish A, Jayantee K, Kant MU. Microstructural abnormalities of the trigeminal nerve by diffusion-tensor imaging in trigeminal neuralgia without neurovascular compression. Neuroradiol J. 2016;29(1):13–18. doi:10.1177/1971400915620439 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kim JS, Kang JH, Lee MC. Trigeminal neuralgia after pontine infarction. Neurology. 1998;51(5):1511–1512. doi:10.1212/WNL.51.5.1511 [DOI] [PubMed] [Google Scholar]
- 5.Katsuno M, Teramoto A. Secondary trigeminal neuropathy and neuralgia resulting from pontine infarction. J Stroke Cerebrovasc Dis. 2010;19(3):251–252. doi:10.1016/j.jstrokecerebrovasdis.2009.04.005 [DOI] [PubMed] [Google Scholar]
- 6.Chang JW, Choi JY, Yoon Y, Park YG, Chung SS. Unusual causes of trigeminal neuralgia treated by gamma knife radiosurgery. report of two cases. J Neurosurg. 2002;97(5 suppl):533–535. doi 10.3171/jns.2002.97.supplement [DOI] [PubMed] [Google Scholar]
- 7.Arrese I, Lagares A, Alday R, Ramos A, Rivas JJ, Lobato RD. Typical trigeminal neuralgia associated with brainstem white matter lesions on MRI in patients without criteria of multiple sclerosis. Acta Neurochir (Wien). 2008;150(11):1157–1161. doi:10.1007/s00701-008-0024-4 [DOI] [PubMed] [Google Scholar]
- 8.Thompson AJ, Banwell BL, Barkhof F, et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018;17(2):162–173. doi:10.1016/S1474-4422(17)30470-2 [DOI] [PubMed] [Google Scholar]
- 9.Harries AM, Mitchell RD. Percutaneous glycerol rhizotomy for trigeminal neuralgia: safety and efficacy of repeat procedures. Br J Neurosurg. 2011;25(2):268–272. doi:10.3109/02688697.2011.558946 [DOI] [PubMed] [Google Scholar]
- 10.Kugelberg E, Lindblom U. The mechanism of the pain in trigeminal neuralgia. J Neurol Neurosurg Psychiatry. 1959;22(1):36–43. doi:10.1136/jnnp.22.1.36 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Di Stefano G, Maarbjerg S, Truini A. Trigeminal neuralgia secondary to multiple sclerosis: from the clinical picture to the treatment options. J Headache Pain. 2019;20(1):20. doi:10.1186/s10194-019-0969-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lebrun C, Cohen M, Mondot L, Ayrignac X, Labauge P. A. Case report of solitary sclerosis: This is really multiple sclerosis. Neurol Ther. 2017;6(2):259–263. doi:10.1007/s40120-017-0082-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Keegan BM, Kaufmann TJ, Weinshenker BG, et al. Progressive solitary sclerosis: Gradual motor impairment from a singleCNSdemyelinating lesion. Neurology. 2016;87(16):1713–1719. doi:10.1212/WNL.0000000000003235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Xia Y, Shoemaker T, Gorelick N, McArthur JC. Solitary sclerosis presenting as isolated spontaneous paroxysmal dysarthria. eNeurologicalSci. 2019;14(January):98–100. doi:10.1016/j.ensci.2019.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zeydan B, Flanagan EP, Keegan BM, Weinshenker BG, Kantarci OH. Solitary Sclerosis in MS Disease Course Continuum. May 2019. Accessed June 18, 2021. https://cmsc.confex.com/cmsc/2019/meetingapp.cgi/Paper/6361.
- 16.Barkhof F. The clinico-radiological paradox in multiple sclerosis revisited. Curr Opin Neurol. 2002;15(3):239–245. doi:10.1097/00019052-200206000-00003 [DOI] [PubMed] [Google Scholar]