Abstract
T-cell immunity associated with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection or vaccination in solid organ transplant recipients (SOTRs) is poorly understood. To address this, we measured T-cell responses in 50 SOTRs with prior SARS-CoV-2 infection. The majority of patients mounted SARS-CoV-2–specific CD4+ T-cell responses against spike (S), nucleocapsid, and membrane proteins; CD8+ T-cell responses were generated to a lesser extent. CD4+ T-cell responses correlated with antibody levels. Severity of disease and mycophenolate dose were moderately associated with lower proportions of antigen-specific T cells. Relative to nontransplant controls, SOTRs had perturbations in both total and antigen-specific T cells, including higher frequencies of total PD-1+ CD4+ T cells. Vaccinated SOTRs (n = 55) mounted significantly lower proportions of S-specific polyfunctional CD4+ T cells after 2 doses, relative to unvaccinated SOTRs with prior coronavirus disease 2019. Together, these results suggest that SOTRs generate robust T-cell responses following natural infection that correlate with disease severity but generate comparatively lower T-cell responses following mRNA vaccination.
Keywords: transplantation, COVID-19, SARS-CoV-2, T cells, vaccination
Solid organ transplant recipients mount antigen-specific T-cell responses after SARS-CoV-2 infection that correlate with antibodies and disease severity. Compared to natural infection, vaccine responses to 2 doses of mRNA vaccine result in comparably lower frequencies of antigen-specific CD4+ T cells.
Solid organ transplant recipients (SOTRs) are at increased risk for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, with mortality rates ranging from 10% to 30% [1–4]. Profound immune disturbances have been identified in immunocompetent individuals with acute coronavirus disease 2019 (COVID-19), including lymphopenia and decreased T-cell counts [5, 6]. Most immunocompetent individuals mount SARS-CoV-2–specific T cells. While CD4+ T-cell responses may outnumber CD8+ T-cell responses in some studies [7, 8], both branches of T-cell immunity are induced following infection. A phenotype of T-cell exhaustion, associated with expression of specific cell surface receptors, such as programmed cell death protein 1 (PD-1) or T-cell immunoglobulin and mucin-domain containing-3 (TIM-3), has also been observed in severe cases [9]. The magnitude of SARS-CoV-2–specific T cells ranges from 0.01% to 1% of circulating T cells [7, 10–15] and may be related to disease severity [16]. These cells primarily target spike (S) [10] and other SARS-CoV-2 antigens, including the nucleocapsid (NP) and membrane (Mb) proteins [7, 13–15]. Antigen-specific CD4+ T cells appear to be Th1-polarized, evidenced by production of interferon gamma (IFN-γ) and interleukin 2 (IL-2) as effectors [7, 10, 13, 17].
Although we have some understanding of antibody response following SARS-CoV-2 infection in the immunocompromised setting, we know far less about T-cell responses in SOTRs [18]. Most studies describing T-cell responses in transplant patients are limited by small sample sizes, obviating the capacity to draw links with outcomes or clinical parameters, such as severity of disease. Most T-cell studies also suffer from severity bias with few studies evaluating the T-cell response in milder COVID disease. Few studies have also directly compared immune responses to SARS-CoV-2 between transplant recipients and the general population or examined how the magnitude of T-cell response in SOTRs varies between natural infection and vaccination. In the general population, SARS-CoV-2 vaccination induces potent antibody and T-cell responses [19, 20]. Although there is evidence of decreased antibody responses in vaccinated SOTRs [21–23], the impact on T-cell responses is less well understood. Here we provide a detailed look at the T-cell response in 50 SOTRs with prior SARS-CoV-2 infection. We provide comparisons to previously infected nontransplant controls and to vaccinated transplant recipients, and describe how T-cell responses during natural infection correlate with antibody responses and severity of disease.
MATERIALS AND METHODS
Study Design and Ethics
This single-center study was performed at the University Health Network (UHN) Transplant Centre. The primary cohort comprised 50 SOTRs diagnosed with COVID-19 from March 2020–March 2021. Inpatients and outpatients were included if they had a positive nucleic acid test for SARS-CoV-2 on a respiratory specimen. Peripheral blood mononuclear cells (PBMCs) and serum were collected at approximately 4–6 weeks after symptom onset. A second cohort of nontransplanted controls with prior COVID-19 was included for comparison. PBMCs were obtained during convalescence (>14 days post–symptom onset) from COVID-19 clinic outpatients at UHN (n = 13), or via UHN’s PRESERVE-Pandemic Response Biobank for coronavirus samples (n = 7). All infected patients were followed for outcomes up to 90 days. The third cohort consisted of vaccinated SOTRs who had no previous history of COVID-19 (n = 55). PBMCs were collected 4–6 weeks after the second dose of mRNA vaccine; design and ethical considerations for the vaccinated cohort are described elsewhere [24]. All vaccinated SOTRs were negative for anti–receptor binding domain (RBD) antibody before vaccination. The study was approved by the UHN research ethics board. All patients or their delegates provided informed consent.
T-Cell Assessment
A total of 106 cryopreserved PBMCs were thawed and rested for 2 hours prior to incubation with overlapping peptides (15-mers with 11 amino acid overlaps; PepTivator, Miltenyi Biotec) corresponding to SARS-CoV-2 S, NP, or Mb proteins (final concentration 5 µg/mL per peptide, based on preliminary optimization experiments). Cells were incubated overnight with peptides, a co-stimulatory antibody cocktail (BD Biosciences), and a protein transport inhibitor (ThermoFisher Scientific). Intracellular cytokine staining was used to measure the frequency of SARS-CoV-2–specific T cells, as has been done by others [20, 25, 26]. Phorbol 12-myristate 13-acetate/ionomycin was used as a positive control and cells treated with media alone were used as a negative (media) control. Following incubation at 37°C, cells were stained with a viability dye (Zombie Aqua, Biolegend), Fc blocked (BD Biosciences), and incubated with a surface marker antibody cocktail (CD3, CD4, CD8, PD-1, and TIM-3). Cells were then fixed, permeabilized, and incubated with an antibody cocktail to detect intracellular cytokines (IFN-γ and IL-2). Supplementary Table 1 lists the antibodies used in this study. Flow cytometry was performed on an LSR II BGRV (BD Biosciences) at the SickKids-UHN Flow Cytometry Facility. Representative gating is shown in Supplementary Figure 1. Frequencies of CD4+ and CD8+ T cells were measured in terms of cells expressing IFN-γ and IL-2 alone, or both cytokines simultaneously (polyfunctional T cells). The frequency of antigen-specific T cells was determined by subtracting the frequency of cytokine-positive T cells in untreated comparators from the frequency in peptide-stimulated samples. A positive T-cell response was defined as a frequency exceeding 0.01%, the limit of quantitation for this study. Results below this threshold were set to 0.005%, or 50 cells per 106 CD4+/CD8+ T cells. A minimum number of 100000 live, CD3+ T cells were required for samples to be included in the flow analysis. Vaccine-specific T-cell responses were assessed by stimulating isolated PBMCs with S peptides using the same protocol described above. Total CD4+ and CD8+ T cells were assessed using non-peptide-stimulated PBMCs (media controls). Total T cells were used to characterize cell surface markers associated with T-cell exhaustion (PD-1, TIM-3).
Antibody Testing
Serologic testing for anti–SARS-CoV-2 antibody was performed using an anti-NP chemiluminescent microparticle immunoassay (Abbott Laboratories) [27] and an anti-S RBD electrochemiluminescent immunoassay (Roche) [28]. Index measurements of ≥1.4 and ≥0.8 U/mL were considered positive for anti-NP and anti-S antibodies, respectively.
Statistical Analysis
Demographics were analyzed using descriptive statistics. Categorical variables were compared using a 2-tailed Fisher exact test. Continuous variables were compared using Mann–Whitney U test, the Kruskal–Wallis test, or Spearman correlation. Dunn correction for multiple comparisons was used when performing the Kruskal–Wallis test. Statistical significance was defined at the level of P < .05. All statistical analyses were performed with Prism version 9 (GraphPad Software). Data are available upon reasonable request.
RESULTS
Patient Demographics
Fifty SOTRs diagnosed with COVID-19 had PBMCs collected and tested at a median of 38.5 days (interquartile range [IQR], 36.0–51.3 days) from symptom onset. Demographic information for the 50 SOTRs and 20 nontransplant controls are described in Table 1. SOTRs were primarily male (72.0%) with a median age of 55.5 years. Kidney transplant recipients comprised 48.0% of the cohort. The median time from transplant to COVID-19 diagnosis was 5.9 years (IQR, 1.8–9.4 years). At diagnosis, most SOTRs (98%) were treated with calcineurin inhibitors (CNIs), primarily tacrolimus (70%), along with anti-metabolites (78.0%) and prednisone (76.0%). Hospitalization for COVID-19 occurred in 46.0% (n = 23) of cases, with oxygen supplementation, intensive care unit admission, and mechanical ventilation occurring in 24%, 6.0%, and 2.0%, respectively. No deaths were recorded in this SOTR cohort.
Table 1.
Patient and Control Demographics
| Characteristic | SOTRs | Controls | P Value |
|---|---|---|---|
| No. | 50 | 20 | |
| Age, y, median (IQR) | 55.5 (47.0–61.5) | 52.5 (36.3–56.8) | .18 |
| Sex, No. (%) | |||
| Male | 36 (72.0) | 14 (70.0) | |
| Female | 14 (28.0) | 6 (30.0) | >.99 |
| Time from symptom onset to sample, d, median (IQR) | 38.5 (36.0–51.3) | 41.5 (20.5–53.8) | .49 |
| Time from transplant to COVID-19, y, median (IQR) | 5.9 (1.8–9.4) | … | |
| Disease severity, No. (%) | |||
| WHO score 1–4 | 38 (76.0) | 6 (30.0) | .76 |
| WHO score 5–9 | 12 (24.0) | 14 (70.0) | |
| Type of transplant, No. (%) | |||
| Kidney | 24 (48.0) | … | |
| Kidney-pancreas | 3 (6.0) | … | |
| Heart | 3 (6.0) | … | |
| Liver | 13 (26.0) | … | |
| Lung | 7 (14.0) | … | |
| Immunosuppression at time of COVID-19, No. (%) | |||
| Calcineurin inhibitor | 49 (98.0) | … | |
| Cyclosporin | 14 (28.0) | … | |
| Tacrolimus | 35 (70.0) | … | |
| Anti-metabolite | 39 (78.0) | … | |
| Azathioprine | 6 (12.0) | … | |
| Mycophenolate | 33 (66.0) | … | |
| Steroid | … | ||
| Prednisone | 38 (76.0) | … | |
Abbreviations: COVID-19, coronavirus disease 2019; IQR, interquartile range; SOTR, solid organ transplant recipient; WHO, World Health Organization.
SARS-CoV-2–Specific T Cells in SOTRs
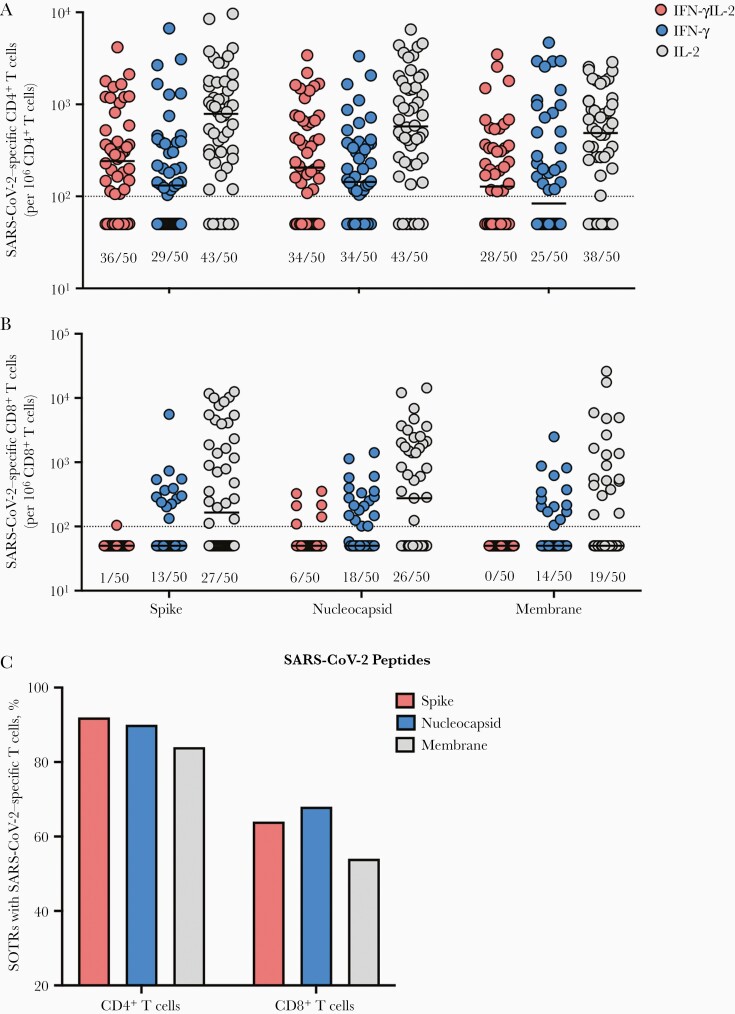
The frequency of SARS-CoV-2–specific T cells in SOTRs was measured after stimulation with SARS-CoV-2 S, NP, or Mb protein peptides. The proportions of SOTRs who mounted S-reactive CD4+ T cells was 58.0% (IFN-γ monofunctional), 86.0% (IL-2 monofunctional), and 72.0% (polyfunctional) (Figure 1A). The proportion that had detectable NP-specific CD4+ T cells was 68.0% (IFN-γ monofunctional), 86.0% (IL-2 monofunctional), and 68.0% (polyfunctional). Last, the proportion of individuals with detectable Mb-specific CD4+ T cells was 50.0% (IFN-γ monofunctional), 76.0% (IL-2 monofunctional), and 56% (polyfunctional). The percentage of SOTRs with at least 1 positive SARS-CoV-2–reactive CD4+ T-cell population was 92% for S, 90% for NP, and 84% for Mb (Figure 1C), representing the overall proportion of individuals with detectable CD4+ T-cell responses against each antigen. The percentage positive for all 3 cytokine populations following S, NP, or Mb stimulation was 50%, 58%, and 38%, respectively.
Figure 1.
Antigen-specific T cells in peptide-stimulated CD4+ and CD8+ T cells. Proportions of spike-, nucleoprotein- and membrane-specific CD4+ (A) and CD8+ (B) T cells are shown. Individual patients are shown by colored dots. The fraction underneath each bar corresponds to the proportion of solid organ transplant recipients (SOTRs) positive for each corresponding cytokine population. Bars show median. Horizontal dotted line indicates limit of quantification, 0.01%. C, Proportion of SOTRs with severe acute respiratory syndrome coronavirus 2–reactive T cells. All data shown were collected from 50 SOTRs. Abbreviations: IFN-γ, interferon gamma; IL-2, interleukin 2; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; SOTR, solid organ transplant recipients.
The SARS-CoV-2–directed CD8+ T-cell response was less pronounced (Figure 1B). The proportion of SOTRs who mounted S-reactive CD8+ T cells was 26.0% (IFN-γ monofunctional), 54.0% (IL-2 monofunctional), and 2.0% (polyfunctional). The proportion that had detectable NP-specific CD8+ T cells was 36.0% (IFN-γ monofunctional), 52.0% (IL-2 monofunctional), and 12.0% (polyfunctional). Last, the proportion of individuals with detectable Mb-specific CD8+ T cells was 28.0% (IFN-γ monofunctional) and 38.0% (IL-2 monofunctional). No polyfunctional Mb-directed CD8+ T cells were detected in the SOTR group. The percentage of SOTRs with at least 1 positive SARS-CoV-2–reactive CD8+ T-cell population was 64% for S, 68% for NP, and 54% for Mb (Figure 1C). The percentage of patients who were positive for all 3 cytokine populations following S, NP, or Mb stimulation were 2%, 10%, and 0%, respectively. Together, these data suggest that the majority of SOTRs generate SARS-CoV-2–specific T-cell responses following natural infection.
Relationship Between Antigen-Specific T-Cell and Antibody Responses in SOTRs
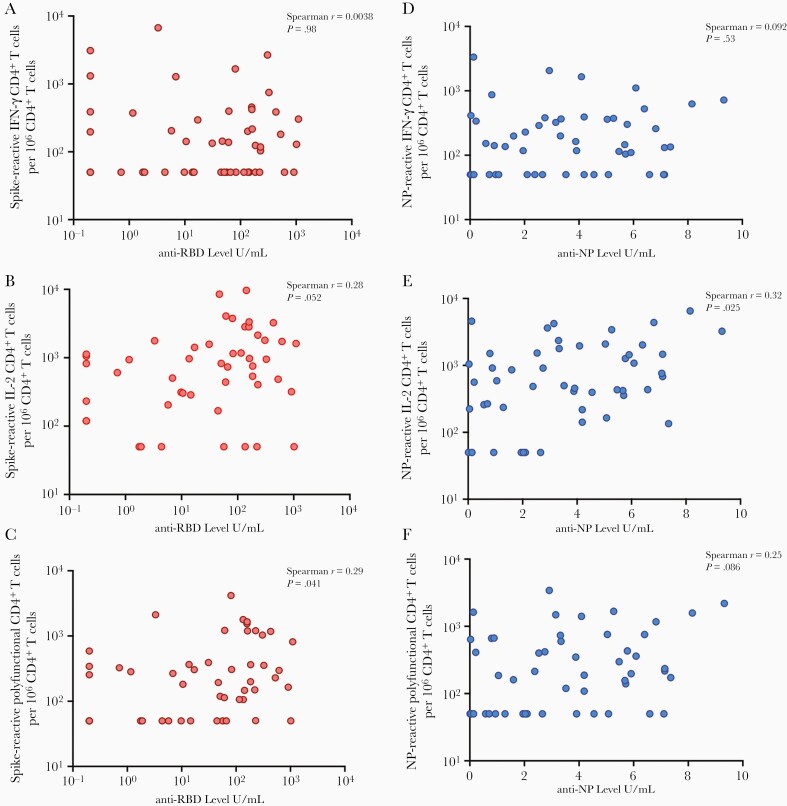
Previously, we measured SARS-CoV-2–specific antibody responses in the 50 SOTRs in our cohort. Those results, including analysis of factors associated with antibody response, are published elsewhere [2]. Anti-NP and anti-S RBD antibody levels in sera were measured at the same time as T-cell responses were assessed. Antibody levels were compared with proportions of S- or NP-directed CD4+ T-cell responses. This was done to identify antigen-specific CD4+ T cells that may be important for driving antibody responses. Six of the 50 naturally infected patients (12%) did not develop anti-S antibodies at sampling time. All 6 of these patients mounted anti-S T-cell responses. A larger proportion were anti-NP negative at sampling time (13/50 [26%]). T-cell responses were found in all but 3 (10/13 [76.9%]) of these patients.
Proportions of S-specific CD4+ T cells correlated only moderately with levels of anti-S RBD antibodies, particularly among IL-2 monofunctional (P = .052) and polyfunctional (P = .041) cells (Figure 2A–C). We found a similar relationship with respect to NP, where the magnitude of NP-specific IL-2 monofunctional (P = .025) or polyfunctional CD4+ T cells (P = .086) had a trend toward correlating with anti-NP antibody levels in blood (Figure 2D–F). Interestingly, IFN-γ monofunctional CD4+ T cells poorly correlated with antibody responses.
Figure 2.
Correlation of antibody levels and magnitude of antigen-specific T-cell response in solid organ transplant recipients (SOTRs). Proportions of spike-specific interferon gamma (IFN-γ) monofunctional (A), interleukin 2 (IL-2) monofunctional (B), or IFN-γ and IL-2 polyfunctional (C) CD4+ T cells relative to levels of anti-spike receptor-binding domain (RBD) antibodies (U/mL). Similar plots are shown for nucleoprotein (NP)–specific IFN-γ monofunctional (D), IL-2 monofunctional (E), or IFN-γ and IL-2 polyfunctional (F) CD4+ T cells relative to levels of anti-NP antibody level (U/mL). Each dot corresponds to 1 participating SOTR. Spearman r for each comparison is shown in the top right of each plot, along with the corresponding P value.
T-Cell Responses and Severity of SARS-CoV-2 Infection in SOTRs
SOTRs were categorized according to severity of clinical COVID-19 disease and compared with respect to total and antigen-specific T-cell responses. Those not receiving oxygen supplementation were considered to have milder COVID-19, consistent with World Health Organization (WHO) severity scores of 1–4, and those requiring oxygen supplementation, or any other higher level of hospital care (n = 12 [24%]), comprised the moderate-to-severe SOTR group, consistent with WHO severity scores of 5–9 [2].
No differences in total CD4+ and CD8+ T cells were found with respect to disease severity (Supplementary Figure 2A–C).We also found no differences in frequencies of CD4+ or CD8+ T cells expressing markers associated with T-cell exhaustion, namely PD-1 and TIM-3 (Supplementary Figure 2D–G).
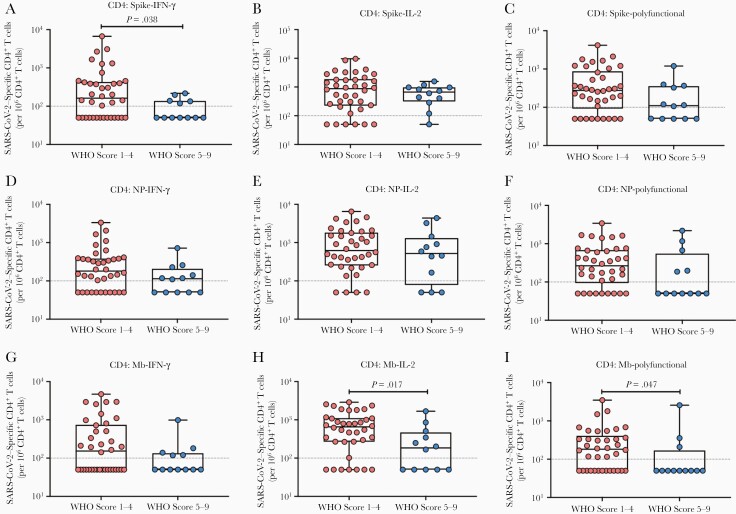
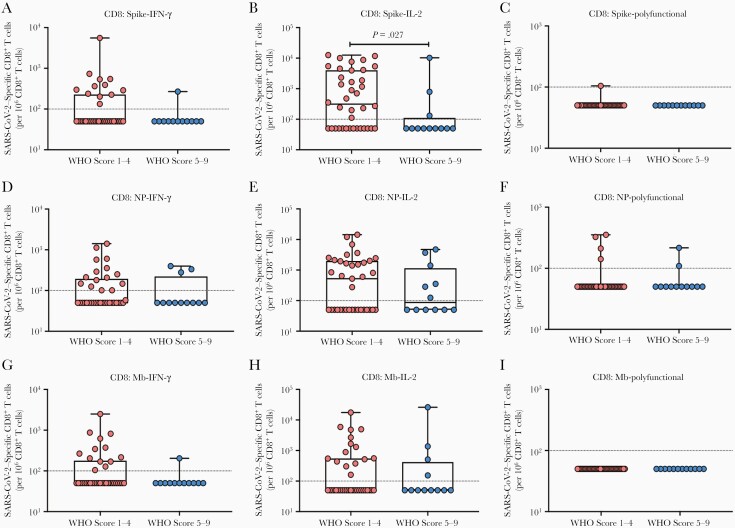
We next compared frequencies of SARS-CoV-2–specific CD4+ (Figure 3) and CD8+ (Figure 4) T cells according to disease severity. In general, those with higher WHO disease scores had lower proportions of antigen-specific CD4+ and CD8+ T cells, with significant differences observed among S-specific IFN-γ–expressing CD4+ T cells (Figure 3A; P = .038), and Mb-specific CD4+ T cells expressing IL-2 alone (Figure 3H; P = .017) or Mb-specific polyfunctional CD4+ T cells (Figure 3I; P = .047). Among antigen-specific CD8+ T cells, we noted a similar pattern; in particular, the proportions of S-specific IL-2–expressing CD8+ T cells (Figure 4B; P = .027) were significantly lower in SOTRs with higher disease scores.
Figure 3.
Impact of disease severity on severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) antigen–specific CD4+ T cells according to cytokine subpopulations. Proportions of spike-specific (A–C), nucleoprotein (NP)–specific (D–F), and membrane (Mb)–specific (G–I) CD4+ T cells were compared with respect to disease severity: World Health Organization (WHO) scores 1–4 (milder coronavirus disease 2019 [COVID-19]) vs 5–9 (moderate-to-severe COVID-19). The proportion of interferon gamma (IFN-γ) monofunctional (A, D, G), interleukin 2 (IL-2) monofunctional (B, E, H), or IFN-γ and IL-2 polyfunctional (C, F, I) T cells are shown with each solid organ transplant recipient represented by a dot. Bars show median ± interquartile range. Dotted line indicates the limit of quantification, 0.01%.
Figure 4.
Impact of disease severity on severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) antigen-specific CD8+ T cells according to cytokine subpopulations. Proportions of spike-specific (A–C), nucleoprotein (NP)–specific (D–F), and membrane (Mb)–specific (G–I) CD8+ T cells were compared with respect to disease severity: World Health Organization (WHO) scores 1–4 (milder coronavirus disease 2019 [COVID-19]) vs 5–9 (moderate-to-severe COVID-19). The proportion of interferon gamma (IFN-γ) monofunctional (A, D, G), interleukin 2 (IL-2) monofunctional (B, E, H), or IFN-γ and IL-2 polyfunctional (C, F, I) T cells are shown with each solid organ transplant recipient represented by a dot. Bars show median ± interquartile range. Dotted line indicates the limit of quantification, 0.01%.
Impact of Immunosuppression on SARS-CoV-2–Specific T Cells in SOTRs
Next, we investigated the impact of immunosuppression at diagnosis on SARS-CoV-2–specific T cells. To minimize the number of comparisons, we only analyzed the impact of immunosuppression on S-specific T cells. These T cells were assessed in composite: proportions of monofunctional and polyfunctional T cells were pooled together and expressed as total S-reactive CD4+ or CD8+ T cells.
The majority of SOTRs received a CNI (98%). We compared the magnitude of total S-specific T-cell responses according to type of CNI and found no significant differences for either CD4+ or CD8+ T cells (Supplementary Figures 3A and 4B). We also found no correlation between the blood level of tacrolimus, the most commonly used CNI, and the magnitude of total S-specific CD4+ or CD8+ T cells (Supplementary Figure 3C). Use of anti-metabolites, namely mycophenolate or azathioprine, did not significantly impact proportions of total S-specific CD4+ or CD8+ T cells (Supplementary Figure 3D and 3E); however, total daily dose (TDD) of mycophenolate had a weak inverse correlation with the magnitude of total S-specific CD4+ T cells (Spearman r = –0.35, P = .048; Supplementary Figure 3F). Regarding steroids, no significant differences in total S-specific T cells were measured according to use, or TDD of prednisone (Supplementary Figure 3G–I).
Bulk and SARS-CoV-2–Specific T-Cell Responses in SOTRs Compared to Nontransplant Controls
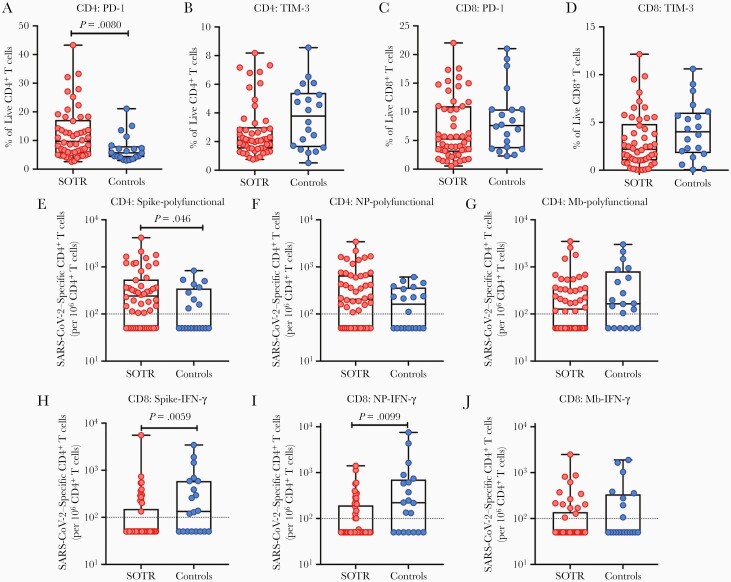
We next compared T-cell responses in 50 SOTRs and 20 nontransplant controls who were similar in age, sex, and time from symptom onset to blood collection (Table 1). Although SOTRs and controls had similar proportions of CD3+ T cells in the peripheral blood, we noted a significantly lower frequency of total CD4+ T cells and a significantly higher proportion of total CD8+ T cells in SOTRs (Supplementary Figure 4). We also compared the proportions of total PD-1+ or TIM-3+ T cells between groups: SOTRs were characterized by significantly higher frequencies of PD-1–expressing total CD4+ T cells relative to controls (P = .008; Figure 5A). No differences were found with respect to TIM-3+ CD4+ T cells, or CD8+ T cells expressing markers of exhaustion (Figure 5B–D).
Figure 5.
Programmed cell death protein 1 (PD-1)– or T-cell immunoglobulin and mucin-domain containing-3 (TIM-3)–expressing total T cells, and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)–specific T cells between solid organ transplant recipients (SOTRs) and controls. PD-1– or TIM-3–expressing total CD4+ T cells (A and B, respectively), and PD-1– or TIM-3–expressing total CD8+ T cells (C and D, respectively) between 50 SOTRs and 20 nontransplant controls. Proportion of spike-specific (E), nucleoprotein (NP)–specific (F), or membrane (Mb)–specific (G) polyfunctional CD4+ T cells, and total spike-specific (H), NP-specific (I), or Mb-specific (J) interferon gamma (IFN-γ) monofunctional CD8+ T cells between SOTRs and nontransplant controls. Each patient is presented by a dot. Bars show median ± interquartile range. Dotted line indicates the limit of quantification, 0.01%.
Last, we examined whether the magnitude of the SARS-CoV-2–specific T-cell response varied between SOTRs and controls. To minimize number of comparisons, we analyzed only antigen-specific polyfunctional CD4+ T cells, and CD8+ IFN-γ monofunctional T cells as these are common subsets used to assess quality of T-cell response during natural infection and in vaccine studies [29]. S-specific polyfunctional CD4+ T cells were more proportionally abundant in SOTRs compared to controls (P = .046; Figure 5E), but no differences were seen with respect to NP-specific or Mb-specific polyfunctional CD4+ T cells (Figure 5F and 5G). With respect to CD8+ T cells, antigen-specific T cells were consistently more abundant in controls than in SOTRs, particularly among S-directed (P = .0059) or NP-directed (P = .0099) T-cell responses (Figure 5H–J). All together, these data suggest that immune responses to SARS-CoV-2 vary between SOTRs and nontransplant controls at the global and antigen-specific T-cell level.
Magnitude of T-Cell Response Between SARS-CoV-2 Natural Infection and Vaccination in SOTRs
Last, we compared the magnitude of the T-cell response between naturally infected SOTRs and SOTRs receiving mRNA-based SARS-CoV-2 vaccine. The demographics of the vaccine cohort are found in Supplementary Table 2. Both groups were similar with respect to sex and time from transplant. The vaccinated group was significantly older (55.5 vs 65.5 years, P < .0001). Seven SOTRs received 2 doses of BNT162b2 (Pfizer), and 48 received 2 doses of mRNA-1273 (Moderna). Factors determining vaccine response to mRNA-1273 have been described elsewhere [24]. For the purpose of this study, we specifically compared S-specific polyfunctional CD4+ T cells between groups as this type of cellular response is commonly used to assess immunogenicity [29–31].
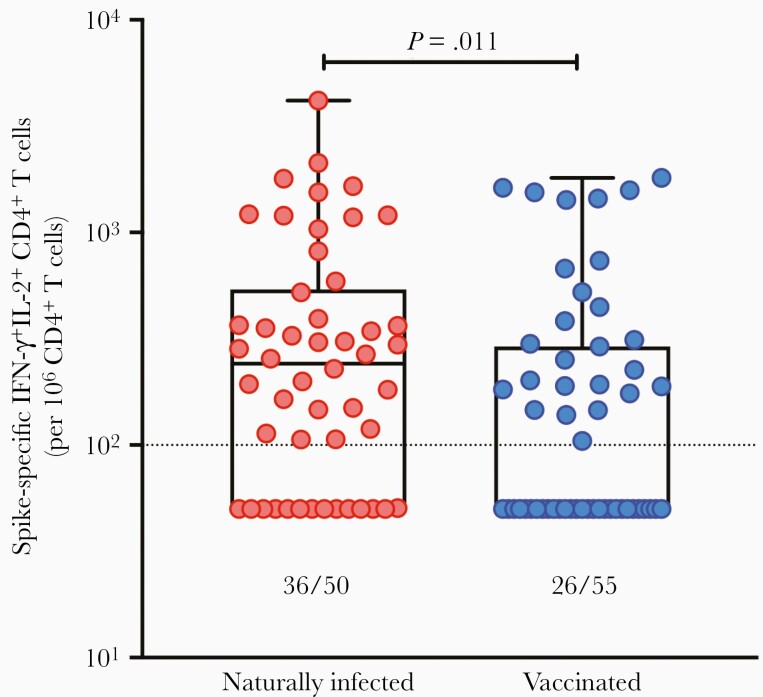
Relative to naturally infected SOTRs, those receiving mRNA vaccination mounted proportionally less abundant polyfunctional CD4+ T-cell responses (P = .011; Figure 6). A total of 47.3% of SOTRs had detectable antigen-specific T-cell responses at 4–6 weeks after second dose, compared to 72.0% of SOTRs who had detectable polyfunctional S-specific responses after recovery from natural infection. Neither the use nor the dose or level of immunosuppression (CNIs, anti-metabolites, prednisone) was statistically associated with spike-specific CD4+ T cells within the vaccinated cohort (Supplementary Figure 5). Furthermore, no differences in immunosuppression were measured between vaccinated and naturally infected SOTRs (P > .05 for all comparisons, data not shown). These results suggest that the T-cell responses are comparatively lower in SOTRs vaccinated with 2 doses of mRNA vaccine, with a greater proportional response in naturally infected SOTRs.
Figure 6.
Spike-specific polyfunctional CD4+ T-cell response in recovered solid organ transplant recipients (SOTRs) with natural infection vs SOTRs vaccinated against severe acute respiratory syndrome coronavirus 2. Polyfunctional CD4+ T-cell responses were measured in 50 SOTRs with natural infection and 55 SOTRs vaccinated with 2 doses of mRNA vaccine. Each patient is represented by a dot. Bars show median ± interquartile range. The fraction beneath each box and whisker plot indicates the number of SOTRs with positive T-cell responses in each group. Dotted line indicates the limit of quantification, 0.01%. Abbreviations: IFN-γ, interferon gamma, IL-2, interleukin 2.
DISCUSSION
Our study provides a number of novel and key findings. SARS-CoV-2–specific CD4+ T-cell responses were generated in most SOTRs with natural infection (84%–92%). Anti-S and anti-NP responses were most prominent, but Mb-directed CD4+ T-cell responses were regularly detected. As has been found for the general population [7, 10], the CD8+ T-cell response in SOTRs was lower. The overall magnitude of antigen-specific CD4+ and CD8+ T-cell response measured in our study is similar to the proportion observed for the general population [7, 10–15], along with the directionality of T-cell responses against S and NP antigens. Other studies have reported vigorous SARS-CoV-2–specific T-cell responses against S, NP, and Mb in SOTRs in convalescence [32, 33]; in one of the larger studies, the proportion of liver transplant recipients who developed SARS-CoV-2–specific CD4+ and CD8+ T-cell responses was 90.3% and 83.9%, respectively, by 103 days after COVID-19 diagnosis [32].
In our study, the functionality of the CD4+ T-cell response was primarily driven by IL-2–producing CD4+ T cells and IFN-γ+IL-2+ polyfunctional responses, but IFN-γ monofunctional responses were also commonly identified. Unlike CD4+ T cells, CD8+ polyfunctional T-cell responses were uncommonly detected. In the literature there is often a preponderance of using IFN-γ–related readouts to assess T-cell responses following infection or vaccination. Our results suggest that a significant portion of antigen-specific T-cell responses in SOTRs may be missed if IL-2 is not taken into consideration. IL-2–producing CD4+ T cells—represented in both IL-2 monofunctional and polyfunctional T cells—correlated with anti-S RBD and anti-NP antibody levels, while monofunctional IFN-γ–producing CD4+ T cells did not. Furthermore, of the T-cell responses that were significantly less abundant in SOTRs who developed severe disease, many were IL-2 expressing, further underscoring the need to consider IL-2, and potentially other effectors, in assays that assess T-cell responses.
In addition to lower proportions of T-cell populations expressing IL-2, higher severity scores in SOTRs were also associated with lower frequencies of S-specific IFN-γ monofunctional CD4+ T cells. These results suggest that a balance of antigen-specific T-cell responses may be required for optimal control of infection in SOTR. Many drugs in the SOTR setting target IL-2 and IFN-γ, such as mycophenolate, a potent T- and B-cell inhibitor. In line with our results, others have reported on the potentially negative impact of mycophenolate on anti–SARS-CoV-2 responses, both in terms of infection and vaccination [2, 21, 34–37]. Our results suggest that reducing the dosage of immunosuppression, specifically for mycophenolate, may be an advantageous step toward maximizing the induction of SARS-CoV-2–specific T cells during natural infection, but this needs to be weighed carefully in light of risk for graft rejection.
In our study, SOTRs experienced several disturbances in total T cells, notably increased frequencies of PD-1–expressing CD4+ T cells relative to nontransplant controls. In acutely infected immunocompetent patients, severity of COVID-19, including death, was associated with PD-1 expression on T cells [9, 38]. Although PD-1 is implicated as a marker of T-cell exhaustion, its exact role here is unknown, and could also be associated with an activated cell state, or immunosuppression relating to transplantation. Recently Rha et al [39] showed that PD-1–expressing SARS-CoV-2–specific T cells were not exhausted, but functional in both acutely infected and convalescent immunocompetent persons. While it is possible that the increased frequency of PD-1–expressing cells may in turn negatively regulate SARS-CoV-2–specific, or other antigen-specific T-cell responses, future studies will need to directly evaluate the role of PD-1 and other exhaustion markers in the pathogenesis of COVID-19 in SOTRs.
Importantly, our study directly compares T-cell responses postvaccination to postinfection in the immunocompromised setting. We identify that the proportion of SOTRs who generate T-cell responses after 2-dose SARS-CoV-2 mRNA vaccination (47.3%) was significantly lower than the proportion of SOTRs who generate comparable T-cell responses after natural infection (72.0%). This is in contrast to the general population where mRNA vaccine generates a greater antibody and T-cell response relative to natural infection. Several studies have now shown that humoral and cellular vaccine responses are diminished in immunocompromised populations [21, 24, 34, 40–45]. Specific immunosuppressives such as mycophenolate may contribute to this, although our data suggest this may not be the case. Further interventions to expand antibody and T-cell immunogenicity, such as additional vaccine doses, may be required to optimize vaccine immunity in this cohort. One limitation of these data is that the natural infection cohort was significantly younger than the vaccine cohort; therefore, it is possible that the lower responses seen in the vaccinated cohort may be partly due to older age. Also, while our data show a quantifiable difference in T-cell response, these data do not suggest that those with natural infection are more likely than vaccinated patients to be protected against subsequent viral challenge. Furthermore, the authors discourage deliberate exposure to SARS-CoV-2 in immunologically naive SOTRs because of the high risk for serious COVID-19 complications and continue to strongly encourage all SOTRs to be vaccinated.
Our study is limited by a lack of longitudinal follow-up data, owing to the cross-sectional nature of the study. Uneven numbers of organ transplant types limited our ability to look at role of type of transplant on T-cell responses. Furthermore, we only examined effect of baseline immunosuppression, and it is possible that changes to immunosuppression during the course of illness may have also impacted T-cell responses. We also did not assess the maturation subtypes of antigen-specific T cells. Since we did not always correct for multiple comparisons, we recognize the preliminary nature of our data. Future studies with larger cohorts of SOTRs will be required to confirm these observations. However, these limitations are countered by several strengths. We believe this study provides important information to the scientific community and fills many gaps in knowledge with respect to our understanding of T-cell responses in immunocompromised persons with COVID-19.
Supplementary Data
Supplementary materials are available at The Journal of Infectious Diseases online. Supplementary materials consist of data provided by the author that are published to benefit the reader. The posted materials are not copyedited. The contents of all supplementary data are the sole responsibility of the authors. Questions or messages regarding errors should be addressed to the author.
Notes
Disclaimer. The contents of this work are solely the responsibility of the authors and do not necessarily represent the official views of the University Health Network (UHN).
Financial support. This publication was supported in part by funding from the Public Health Agency of Canada, through the COVID-19 Immunity Task Force and Vaccine Surveillance Reference Group (D. K., A. H., V. H. F.). This publication was also supported by UHN’s PRESERVE-Pandemic Response Biobank for coronavirus samples, University Health Network Biospecimen Services (REB number 20–5364).
Potential conflicts of interest. A. H. has received clinical trial grants from Roche and Merck, and advisory fees from Merck. D. K. has received clinical trial grants from Roche and Merck, and advisory fees from Roche and Merck. V. K. is a consultant at Abbott Diagnostic Laboratories. All other authors report no potential conflicts of interest.
All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
Presented in part: 2021 American Transplant Congress, Virtual, 4–9 June 2021.
REFERENCES
- 1. Elias M, Pievani D, Randoux C, et al. . COVID-19 infection in kidney transplant recipients: disease incidence and clinical outcomes. J Am Soc Nephrol 2020; 31:2413–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Marinelli T, Ferreira VH, Ierullo M, et al. . Prospective clinical, virologic, and immunologic assessment of COVID-19 in transplant recipients. Transplantation 2021; 105:2175–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Pereira MR, Mohan S, Cohen DJ, et al. . COVID-19 in solid organ transplant recipients: initial report from the US epicenter. Am J Transplant 2020; 20:1800–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Kates OS, Haydel BM, Florman SS, et al. . COVID-19 in solid organ transplant: a multi-center cohort study [manuscript published online ahead of print 7 August 2020]. Clin Infect Dis 2020. doi:10.1093/cid/ciaa1097. [Google Scholar]
- 5. Huang C, Wang Y, Li X, et al. . Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020; 395:497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Chen G, Wu D, Guo W, et al. . Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest 2020; 130:2620–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Grifoni A, Weiskopf D, Ramirez SI, et al. . Targets of T cell responses to SARS-CoV-2 coronavirus in humans with COVID-19 disease and unexposed individuals. Cell 2020; 181:1489–501.e15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Law JC, Koh WH, Budylowski P, et al. . Systematic examination of antigen-specific recall T cell responses to SARS-CoV-2 versus influenza virus reveals a distinct inflammatory profile. J Immunol 2021; 206:37–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Diao B, Wang C, Tan Y, et al. . Reduction and functional exhaustion of T cells in patients with coronavirus disease 2019 (COVID-19). Front Immunol 2020; 11:827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Weiskopf D, Schmitz KS, Raadsen MP, et al. . Phenotype and kinetics of SARS-CoV-2-specific T cells in COVID-19 patients with acute respiratory distress syndrome. Sci Immunol 2020; 5:eabd2071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Braun J, Loyal L, Frentsch M, et al. . SARS-CoV-2-reactive T cells in healthy donors and patients with COVID-19. Nature 2020; 587:270–4. [DOI] [PubMed] [Google Scholar]
- 12. Rydyznski Moderbacher C, Ramirez SI, Dan JM, et al. . Antigen-specific adaptive immunity to SARS-CoV-2 in acute COVID-19 and associations with age and disease severity. Cell 2020; 183:996–1012.e19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Sattler A, Angermair S, Stockmann H, et al. . SARS-CoV-2-specific T cell responses and correlations with COVID-19 patient predisposition. J Clin Invest 2020; 130:6477–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Peng Y, Mentzer AJ, Liu G, et al. ; Oxford Immunology Network Covid-19 Response T cell Consortium; ISARIC4C Investigators. Broad and strong memory CD4+ and CD8+ T cells induced by SARS-CoV-2 in UK convalescent individuals following COVID-19. Nat Immunol 2020; 21:1336–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Jung JH, Rha MS, Sa M, et al. . SARS-CoV-2-specific T cell memory is sustained in COVID-19 convalescent patients for 10 months with successful development of stem cell-like memory T cells. Nat Commun 2021; 12:4043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Chen Z, John Wherry E.. T cell responses in patients with COVID-19. Nat Rev Immunol 2020; 20:529–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Ni L, Ye F, Cheng ML, et al. . Detection of SARS-CoV-2-specific humoral and cellular immunity in COVID-19 convalescent individuals. Immunity 2020; 52:971–7.e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Phadke VK, Scanlon N, Jordan SC, Rouphael NG.. Immune responses to SARS-CoV-2 in solid organ transplant recipients [manuscript published online ahead of print 4 March 2021]. Curr Transplant Rep 2021. doi:10.1007/s40472-021-00322-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Sahin U, Muik A, Derhovanessian E, et al. . COVID-19 vaccine BNT162b1 elicits human antibody and Th1 T cell responses. Nature 2020; 586:594–9. [DOI] [PubMed] [Google Scholar]
- 20. Jackson LA, Anderson EJ, Rouphael NG, et al. ; mRNA-1273 Study Group. An mRNA vaccine against SARS-CoV-2—preliminary report. N Engl J Med 2020; 383:1920–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Boyarsky BJ, Werbel WA, Avery RK, et al. . Immunogenicity of a single dose of SARS-CoV-2 messenger RNA vaccine in solid organ transplant recipients. JAMA 2021; 325:1784–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Boyarsky BJ, Werbel WA, Avery RK, et al. . Antibody response to 2-dose SARS-CoV-2 mRNA vaccine series in solid organ transplant recipients. JAMA 2021; 325:2204–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Marion O, Del Bello A, Abravanel F, et al. . Safety and immunogenicity of anti-SARS-CoV-2 messenger RNA vaccines in recipients of solid organ transplants. Ann Intern Med 2021; 174:1336–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Hall VG, Ferreira VH, Ierullo M, et al. . Humoral and cellular immune response and safety of two-dose SARS-CoV-2 mRNA-1273 vaccine in solid organ transplant recipients [manuscript published online ahead of print 4 August 2021]. Am J Transplant 2021. doi:10.1111/ajt.16766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Le Bert N, Tan AT, Kunasegaran K, et al. . SARS-CoV-2-specific T cell immunity in cases of COVID-19 and SARS, and uninfected controls. Nature 2020; 584:457–62. [DOI] [PubMed] [Google Scholar]
- 26. Anderson EJ, Rouphael NG, Widge AT, et al. ; mRNA-1273 Study Group. Safety and immunogenicity of SARS-CoV-2 mRNA-1273 vaccine in older adults. N Engl J Med 2020; 383:2427–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Bryan A, Pepper G, Wener MH, et al. . Performance characteristics of the Abbott Architect SARS-CoV-2 IgG assay and seroprevalence in Boise, Idaho. J Clin Microbiol 2020; 58:e00941-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. National SARS-CoV-2 Serology Assay Evalution Group. Performance characteristics of five immunoassays for SARS-CoV-2: a head-to-head benchmark comparison. Lancet Infect Dis 2020; 20:1390–400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Panagioti E, Klenerman P, Lee LN, van der Burg SH, Arens R.. Features of effective T cell-inducing vaccines against chronic viral infections. Front Immunol 2018; 9:276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. L’Huillier AG, Hirzel C, Ferreira VH, et al. . Evaluation of recombinant herpes zoster vaccine for primary immunization of varicella-seronegative transplant recipients. Transplantation 2021; 105:2316–23. [DOI] [PubMed] [Google Scholar]
- 31. Hirzel C, L’Huillier AG, Ferreira VH, et al. . Safety and immunogenicity of adjuvanted recombinant subunit herpes zoster vaccine in lung transplant recipients. Am J Transplant 2021; 21:2246–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Fernández-Ruiz M, Olea B, Almendro-Vázquez P, et al. . T cell-mediated response to SARS-CoV-2 in liver transplant recipients with prior COVID-19. Am J Transplant 2021; 21:2785–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Candon S, Guerrot D, Drouot L, et al. . T cell and antibody responses to SARS-CoV-2: experience from a French transplantation and hemodialysis center during the COVID-19 pandemic. Am J Transplant 2021; 21:854–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Boyarsky BJ, Werbel WA, Avery RK, et al. . Antibody response to 2-dose SARS-CoV-2 mRNA vaccine series in solid organ transplant recipients. JAMA 2021; 325:2204–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Benotmane I, Gautier-Vargas G, Cognard N, et al. . Weak anti-SARS-CoV-2 antibody response after the first injection of an mRNA COVID-19 vaccine in kidney transplant recipients. Kidney Int 2021; 99:1487–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Colmenero J, Rodríguez-Perálvarez M, Salcedo M, et al. . Epidemiological pattern, incidence, and outcomes of COVID-19 in liver transplant patients. J Hepatol 2021; 74:148–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Rozen-Zvi B, Yahav D, Agur T, et al. . Antibody response to SARS-CoV-2 mRNA vaccine among kidney transplant recipients: a prospective cohort study. Clin Microbiol Infect 2021; 27:1173.e1–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Zheng M, Gao Y, Wang G, et al. . Functional exhaustion of antiviral lymphocytes in COVID-19 patients. Cell Mol Immunol 2020; 17:533–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Rha MS, Jeong HW, Ko JH, et al. . PD-1-expressing SARS-CoV-2-specific CD8(+) T cells are not exhausted, but functional in patients with COVID-19. Immunity 2021; 54:44–52.e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Monin L, Laing AG, Muñoz-Ruiz M, et al. . Safety and immunogenicity of one versus two doses of the COVID-19 vaccine BNT162b2 for patients with cancer: interim analysis of a prospective observational study. Lancet Oncol 2021; 22:765–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Boyarsky BJ, Ruddy JA, Connolly CM, et al. . Antibody response to a single dose of SARS-CoV-2 mRNA vaccine in patients with rheumatic and musculoskeletal diseases [manuscript published online ahead of print 23 March 2021]. Ann Rheum Dis 2021. doi: 10.1136/annrheumdis-2021-220289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Schramm R, Costard-Jäckle A, Rivinius R, et al. . Poor humoral and T-cell response to two-dose SARS-CoV-2 messenger RNA vaccine BNT162b2 in cardiothoracic transplant recipients. Clin Res Cardiol 2021; 110:1142–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Sattler A, Schrezenmeier E, Weber UA, et al. . Impaired humoral and cellular immunity after SARS-CoV2 BNT162b2 (Tozinameran) prime-boost vaccination in kidney transplant recipients. J Clin Invest 2021; 131:e150175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Cucchiari D, Egri N, Bodro M, et al. . Cellular and humoral response after MRNA-1273 SARS-CoV-2 vaccine in kidney transplant recipients. Am J Transplant 2021; 21:2727–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Herrera S, Colmenero J, Pascal M, et al. . Cellular and humoral immune response after mRNA-1273 SARS-CoV-2 vaccine in liver and heart transplant recipients [manuscript published online ahead of print 22 July 2021]. Am J Transplant 2021. doi: 10.1111/ajt.16768. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.