Abstract
Background
Non-alcoholic fatty liver disease (NAFLD) is a well-established risk factor for cardiovascular disease, with ethnic and regional differences noted. With the recent surge of research within this field, we re-examine the evidence associating NAFLD with subclinical atherosclerosis, and investigate potential regional differences.
Methods
This is a systematic review and meta-analysis. PubMed and EMBASE were systematically searched for publications from January 1967 to July 2020 using standardised criteria. Original, observational studies investigating the association between NAFLD and either carotid intima-media thickness (CIMT) and/or coronary artery calcification (CAC) were included. Key outcomes included differences in mean CIMT, the presence of increased CIMT, the presence of CAC and the development/progression of CAC. Pooled ORs and pooled standard differences in means were calculated using random-effects models. Between-study heterogeneity was quantified using the Q statistic and I². Subgroup analyses stratified by region of study (Asian vs Western) were also conducted.
Results
64 studies involving a total of 172 385 participants (67 404 with NAFLD) were included. 44 studies assessed the effect of NAFLD on CIMT, with the presence of NAFLD associated with increased CIMT (OR 2.00, 95% CI 1.56 to 2.56). 22 studies assessed the effects of NAFLD on CAC score, with the presence of NAFLD associated with the presence of any coronary calcification (OR 1.21, 95% CI 1.12 to 1.32), and the development/progression of CAC (OR 1.26, 95% CI 1.04 to 1.52). When stratified by region, these associations remained consistent across both Asian and Western populations (p>0.05). The majority (n=39) of studies were classified as ‘high quality’, with the remaining 25 of ‘moderate quality’.
Conclusions
There is a significant positive association between various measures of subclinical atherosclerosis and NAFLD, seen across both Western and Asian populations. These results re-emphasise the importance of early risk evaluation and prophylactic intervention measures to preclude progression to clinical cardiovascular disease in patients with NAFLD.
Keywords: metabolic syndrome, meta-analysis, atherosclerosis
Key questions.
What is already known about this subject?
Non-alcoholic fatty liver disease (NAFLD) is a significant, independent risk factor for cardiovascular disease (CVD), with recent evidence positing this association to extend to the preclinical stages of CVD. Previous meta-analyses have quantified positive associations between NAFLD and subclinical atherosclerotic markers before, though the majority of included studies were published before 2016. The last 5 years, however, has experienced a large surge of research in this field, especially within large Asian populations that have not been included in previous meta-analyses. Ethnic and regional differences in the associations between NAFLD and subclinical atherosclerosis have been suggested within individual studies, but have yet to be synthesised across the available literature.
What does this study add?
This meta-analysis serves as a timely update of the existing literature, incorporating the results of over 21 new studies comprising over 100 000 participants (~50 000 with NAFLD) from both Western and Asian regions. The results reinforce the significant positive association between NAFLD and subclinical atherosclerosis (as defined by increased carotid intima-media thickness and coronary artery calcification scores), and further confirm these associations to be consistent across both Western and Asian populations. Lastly, this is the first meta-analysis to demonstrate that the associations between NAFLD and subclinical atherosclerosis are not just crosssectional but also longitudinal.
Key questions.
How might this impact on clinical practice?
This study highlights that NAFLD serves as an important atherogenic risk factor in both Western and Asian populations, and reemphasises the role of early risk evaluation and prophylactic intervention measures to preclude progression to clinical CVD in NAFLD. By confirming a longitudinal association between NAFLD and subclinical atherosclerotic markers, these results also provide potential insight into the causal relationship between NAFLD and subclinical atherosclerosis.
Introduction
Non-alcoholic fatty liver disease (NAFLD) encompasses a wide spectrum of pathological hepatic conditions ranging from simple steatosis to non-alcoholic steatohepatitis, and may ultimately progress to advanced fibrosis, cirrhosis, and end-stage liver disease.1–3 Over the last 20 years, NAFLD has become the leading cause of chronic liver disease, with an estimated 1 billion people affected worldwide.4 NAFLD has increasingly been recognised as the hepatic manifestation of the metabolic syndrome (MetS), and is one facet of a multisystem disease, with close relations to abdominal obesity, type 2 diabetes mellitus (T2DM), insulin resistance and hyperlipidaemia.5–7
Cardiovascular disease (CVD) is the leading cause of mortality in patients with NAFLD, with a large body of evidence demonstrating NAFLD to be a significant, independent risk factor for CVD.2 3 5 8 It is now widely hypothesised that NAFLD is not merely a marker of CVD, but may be actively involved in CVD pathogenesis.3 9–11 This association extends to preclinical CVD, with recent work identifying NAFLD as a risk factor for early subclinical atherosclerosis, and as a strong independent predictor of incident CVD.3 8 12 This holds highly important implications for the screening and early evaluation of CVD in patients with NAFLD. Carotid intima-media thickness (CIMT) and coronary artery calcification (CAC) are the two most established and widely studied surrogate measures of subclinical atherosclerosis, and a growing body of literature has investigated this relationship between NAFLD and CAC/CIMT over the last decade.3 12 13
NAFLD is no longer considered a disease only prevalent in affluent Western countries, with rapidly growing rates of NAFLD reported within Asia in particular.4 14 Ethnic and regional differences in NAFLD prevalence, severity and outcomes have been identified between Western, Hispanic and Asian populations,15 16 and have been attributed to factors including lifestyle, environment, insulin resistance, body composition (adipose distribution and muscle bulk) and genetics.14 15 17 18 Asian populations are especially susceptible with cardiometabolic complications such as NAFLD seen to develop within a much shorter period, within younger patient populations and in those with lower body mass index.18 19 These disparities may possibly extend to differential associations between NAFLD and subclinical atherosclerosis.20–22
Previous meta-analyses have quantified the associations between NAFLD and subclinical atherosclerotic markers before, with the majority of included studies published before 2016.13 23–25 However, the last 5 years has experienced a large surge in research within this area, especially within large Asian populations that have not been reported in previous meta-analyses.26–35 We aim to evaluate the relationship between NAFLD and subclinical atherosclerosis including these updated studies, and to further investigate potential regional differences in these associations.
Methodology
This meta-analysis was conducted and reported according to the Meta-analysis Of Observational Studies in Epidemiology statement36 and was registered in the International Prospective Register of Systematic Reviews (registration number: CRD42020204784).
Search strategy
A comprehensive literature search was performed via the MEDLINE and EMBASE databases to identify potentially relevant publications in the English language, with a date range from January 1967 to July 2020. The databases were systematically searched using a combination of the following keywords linked with appropriate Boolean logic: (Fatty Liver OR NAFLD OR Hepatic Steatosis OR Non-alcoholic fatty liver disease) AND ((subclinical atherosclerosis OR Preclinical atherosclerosis) OR (Coronary calcium OR Calcium Score OR Coronary Calcification) OR (“Carotid Intima-media thickness” OR CIMT OR IMT OR “‘intima media thickness”’)). Relevant references identified from the bibliographies of pertinent articles or review papers were also retrieved.
Eligibility (inclusion and exclusion) criteria
The eligibility criteria was based on the PICOS framework as recommended by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement.37
Participants: studies had to be conducted on adult participants. Studies conducted on ‘special populations’ including adolescent/paediatric populations, and those defined by additional pathologies such as HIV, severe CVD or liver transplants were rejected. Populations with existing metabolic conditions such as MetS and diabetes mellitus were accepted.
Exposures (intervention): studies had to have a defined exposure of ‘NAFLD’ or ‘fatty liver’ or ‘hepatic steatosis’, as diagnosed by either ultrasound (US), liver biopsy, CT, magnetic resonance spectroscopy (MRS) or Fatty Liver Index.
Outcomes: study outcomes had to report on either the (1) presence (cross-sectional) of CAC (CAC score >0), (2) progression (longitudinal) of CAC score, and/or (3) on CIMT. The presence of calcified coronary artery plaques was accepted as a measure of CAC score>0. Studies had to specify how CAC and CIMT were recorded and defined, and also had to quantitatively assess the association between NAFLD and CAC/CIMT, respectively, either via logistic regression for categorical outcomes or via comparison of means techniques (t-test/analysis of variance (ANOVA)) for continuous outcomes.
Comparison: studies had to include a ‘healthy’ control group of participants without NAFLD, preferably from the same population as the exposure group.
Study design: we included observational studies (cross-sectional, case–control, retrospective, prospective), which reported quantitative outcomes. Descriptive studies, reviews and studies on animals were excluded. Studies with sample sizes <50 were also excluded.
Using our search strategy, a total of 1007 titles were initially identified. Two authors (MYZW and JJLY) assessed the titles independently according to the predefined inclusion and exclusion criteria. Studies were first screened by title and abstract. The full-text articles deemed potentially relevant were then obtained and systematically included after detailed examination. The following data were extracted: (a) study: year, region, design; (b) patients: mean age, gender, sample size; (c) method of NAFLD evaluation: US, CT, MRS, liver biopsy or composite index; (d) outcomes: outcome type (CIMT or CAC) and method of outcome definition; (e) analysis: statistical techniques used, primary outcomes (mean±SD, ORs with 95% CIs), confounders adjustment.
For studies reporting multiple multivariable-adjusted models, we extracted those reflecting the greatest degree of control for potential confounders. Any discrepancies in data quantification were resolved by discussion among the investigators.
Study quality evaluation
The quality of observational studies was assessed using a modified version of the Newcastle–Ottawa Scale (NOS) for cohort and cross-sectional studies.38–40 The NOS awards a maximum of 9 stars to assess quality based on three main aspects: (a) the selection and representativeness of the participants (maximum 4 stars), (b) the comparability of groups (maximum 2 stars), and (c) the ascertainment of exposure (for case–control) or outcome (for prospective and cross-sectional) (maximum 3 stars). Following previous reviews, studies assigned 0–4, 5–7, and ≥8 stars were considered as low, medium and high quality, respectively.41–43
Data synthesis and statistical analysis
Outcomes were broadly grouped according to four main categories:
Differences in mean CIMT (continuous).
Presence of increased CIMT (categorical).
Presence of CAC (categorical).
Development/progression of CAC (categorical, longitudinal).
All outcomes were pooled using DerSimonian-Laird random-effects model. The continuous and categorical outcome was reported as pooled standard differences (Std Diff) in means and ORs with 95% CI. We further conducted subgroup analysis to look into regional differences between Asian versus Western populations. We defined ‘Western’ studies to comprise of studies conducted in North America, Europe and Australia, while ‘Asian’ studies comprised of those conducted in South Asian, East Asian and Southeast Asian countries. Lastly, additional subgroup analysis on the Std Diff in mean CIMT within the subset of participants with diabetes was conducted.
The heterogeneity of pooled estimates between studies was quantified using the Q statistic and I². A value of I² of 0%–25% indicates no heterogeneity, 26%–50% low heterogeneity, 51%–75% moderate heterogeneity and 76%–100% high heterogeneity. Funnel plots and Egger’s regression test were used to assess publication bias. P<0.05 was considered as statistical significance.
All statistical analyses were conducted using the Comprehensive Meta-Analysis Software V.3.3.
Results
Search strategy and description of studies
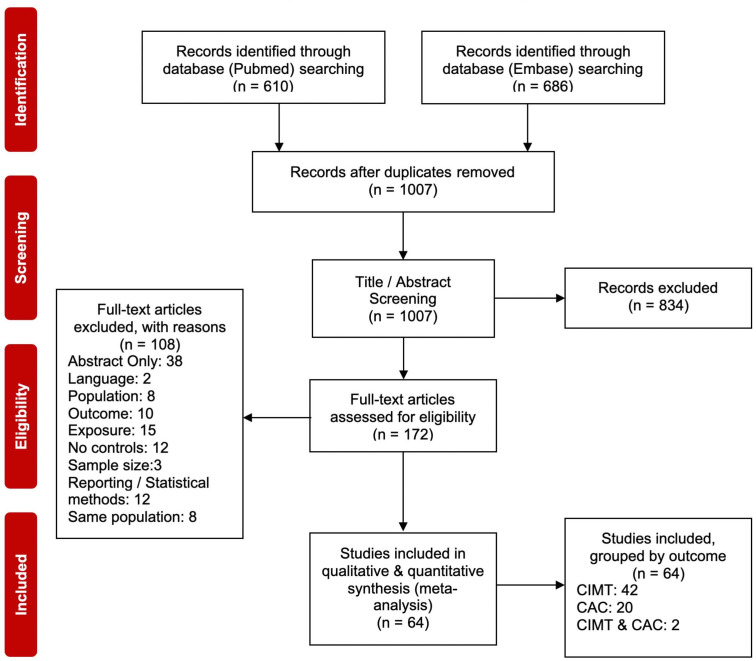
The initial search yielded 1007 potentially relevant titles, where 835 articles were excluded on the basis of title and abstract screen. A total of 172 titles underwent full-length review, of which 108 were further excluded (figure 1). A final total of 64 studies, involving 67 404 patients with NAFLD and 104 981 controls were included in the meta-analysis. Tables 1–3 describe the detailed characteristics of the included studies, grouped by study outcome. These included studies were carried out in Asia (n=32), Western Europe (n=15), the Middle East (n=10) and America (n=7; North America: 6, South America: 1). Sixty studies were cross-sectional and four were prospective cohort studies.
Figure 1.
Study selection PRISMA flow diagram. CAC, coronary artery calcification; CIMT, carotid intima-media thickness; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Table 1.
Characteristics of included studies which conducted a comparison of carotid-intima media thickness (CIMT) means between those with NAFLD and those without
| Name, year | Study region | Study population | Study size | n (%) NAFLD | Age (NAFLD+ vs NAFLD−) | % male (NAFLD+ vs NAFLD−) | NAFLD assessment | Outcome assessment | Confounder adjustment | NOS (max=9) |
| Oni et al22 2019 | North America | Population based | 4123 | 729 (17.7) | 61 vs 63 | 47.0 vs 44.0 | CT, LS ratio <1 | Ultrasound, mean IMT (L&R) | – | 7 |
| Mohammadzadeh et al44 2019 | Iran | Hospital based | 300 | 150 (50.0) | 49.9 vs 52.5 | 65.3 vs 57.3 | Ultrasound | Ultrasound, mean IMT (L&R) | – | 6 |
| Yi et al45 2018 | Asia | Outpatient clinic | 1981 | 1888 (95.3) | 45.9 vs 44.8 | 63.4 vs 40.1 | Ultrasound | Ultrasound, mean of max IMT (L&R) | – | 6 |
| Kim et al30 2018 | Asia | Population (health screen) | 819 | 330 (40.3) | 53.4 vs 53.1 | 64.2 vs 41.5 | Ultrasound | Ultrasound, mean IMT (L&R) | – | 6 |
| Vanjiappan et al46 2018 | Asia | Hospital based, patients with T2DM | 124 | 73 (58.9) | Overall=53.8 | Overall=54.0 | Ultrasound | Ultrasound, mean of max IMT (L&R) | – | 6 |
| Gummesson et al47 2018 | Europe | Population based | 1015 | 106 (10.4) | 58.3 vs 57.5 | 71.7 vs 52.5 | CT, liver HU <40 | Ultrasound, mean IMT | – | 7 |
| Cetindağlı et al48 2017 | Turkey | Outpatient clinic | 120 | 93 (77.5) | 34.5 vs 33.8 | 100 vs 100 | Ultrasound and biopsy | Ultrasound, mean IMT (6 measurements) | Age/sex-matched controls | 7 |
| Guo et al33 2017 | Asia | Hospital based, patients with T2DM | 8571 | 4340 (50.6) | 57.4 vs 61.9 | 54.6 vs 55.9 | Ultrasound | Ultrasound, mean IMT (6 measurements) | Age | 7 |
| Hong et al35 2016 | Asia | Population (health screen) | 955 | 342 (35.8) | 53 vs 51 (median) | 48.8 vs 42.1 | Ultrasound | Ultrasound, mean IMT (99 computer points) | – | 7 |
| Zhang et al49 2016 | Asia | Outpatient clinic, patients with T1DM | 722 | 123 (17.0) | 47.4 vs 46.0 | 52.8 vs 51.1 | Ultrasound | Ultrasound, mean IMT (6 measurements) | Age, sex, BMI, WC, SBP, DBP, total cholesterol, TAG, LDL, HDL, MetS, ALT, AST, GGT, hsCRP, medications | 8 |
| Ozturk et al50 2015 | Turkey | Outpatient clinic, MetS(−) | 82 | 41 (50.0) | 32.8 vs 31.8 | 100 vs 100 | Biopsy | Ultrasound, mean IMT (L&R) | – | 6 |
| Asakawa et al55 2014 | Asia | Population (health screen) | 76 | 24 (31.6) | 61.5 vs 61.0 (median) | 91.7 vs 75.0 | Ultrasound | Ultrasound, max IMT | – | 6 |
| Ayaz et al54 2014 | Turkey | Outpatient clinic | 90 | 60 (66.7) | 44.5 vs 39.5 (median) | 36.7 vs 26.7 | Ultrasound | Ultrasound, mean IMT (8 measurements) | – | 6 |
| Kim et al53 2014 | Asia | Population (health screen), MetS(−) | 1285 | 180 (14.0) | 55.7 vs 55.7 | 58.0 vs 36.0 | CT, liver minus spleen <5 | Ultrasound, mean IMT (4 measurements) | – | 7 |
| Kim et al52 2014 | Asia | Hospital based, patients with T2DM | 1211 | 747 (61.7) | 56.7 vs 55.6 | 51.0 vs 41.8 | Ultrasound | Ultrasound, mean IMT (6 measurements) | – | 6 |
| Nahandi et al51 2014 | Iran | Hospital based, patients without diabetes | 102 | 50 (49.0) | 43.3 vs 43.1 | 32.0 vs 40.4 | Ultrasound | Ultrasound, mean of max IMT (L&R) | HLP, sex, Smk, HT, obesity, walking, liver enzymes | 8 |
| Dogru et al58 2013 | Europe | Outpatient clinic | 189 | 115 (60.8) | 31 vs 28 (median) | 100 vs 100 | Liver biopsy | Ultrasound, mean IMT (6 measurements) | – | 6 |
| Kucukazman et al57 2013 | Europe | Outpatient clinic | 161 | 117 (72.7) | 45.8 vs 45.4 | 44 vs 32 | Ultrasound | Ultrasound, mean IMT (6 measurements) | – | 6 |
| Mishra et al56 2013 | Asia | Population based | 645 | 101 (15.7) | 31.6 vs 27.1 | 100 vs 100 | Ultrasound | Ultrasound, mean of max IMT (L&R) | – | 7 |
| Huang et al62 2012 | Asia | Population based | 8632 | 2590 (30.0) | 58.5 vs 58.5 | 31.4 vs 30.9 | Ultrasound | Ultrasound, max IMT (L&R) | – | 7 |
| Kang et al61 2012 | Asia | Outpatient (health screen), MetS(−) | 413 | 157 (38.0) | 52.0 vs 52.5 | 51.0 vs 41.8 | Ultrasound | Ultrasound, mean IMT (L&R) | – | 7 |
| Thakur et al59 2012 | Asia | Hospital based | 80 | 40 (50.0) | 42.1 vs 41.9 | 67.5 vs 67.5 | Ultrasound | Ultrasound, mean IMT (6 measurements) | – | 7 |
| Colak et al63 2012 | Turkey | Outpatient clinic | 87 | 57 (65.5) | 44.2 vs 42.7 | 45.6 vs 46.7 | Liver biopsy | Ultrasound, mean IMT (6 measurements) | – | 6 |
| Agarwal et al66 2011 | Asia | Hospital based, patients with T2DM | 124 | 71 (57.3) | 57 vs 61 | 52.5 vs 58.5 | Ultrasound | Ultrasound, mean IMT | – | 6 |
| Mohammadi et al65 2011 | Iran | Hospital based | 335 | 250 (74.6) | 46.6 vs 44.9 | 55.6 vs 54.1 | Ultrasound | Ultrasound, mean IMT (6 measurements) | HT, DM, HLP, hyperglycaemia | 8 |
| Poanta et al64 2011 | Europe | Outpatient clinic, patients with T2DM | 56 | 38 (67.9) | 59.4 vs 61.5 | 50.0 vs 83.3 | Ultrasound | Ultrasound | – | 5 |
| Kilciler et al69 2010 | Europe | Outpatient clinic | 114 | 60 (52.6) | 31.7 vs 30.3 | 100 vs 100 | Biopsy | Ultrasound, mean IMT (L&R) | Age-matched controls | 6 |
| Salvi et al68 2010 | Europe | Population based | 220 | 92 (41.8) | 50.7 vs 49.3 | 54.3 vs 36.7 | Ultrasound | Ultrasound, mean IMT (6 measurements) | – | 7 |
| Vlachopoulos et al67 2010 | Europe | Outpatient clinic | 51 | 28 (54.9) | 55.4 vs 51.5 | 52.3 vs 64.3 | Biopsy | Ultrasound, mean IMT (L&R) | Age/sex-matched controls | 6 |
| Gastaldelli et al73 2009 | Europe | Population based | 842 | 234 (27.8) | 42 vs 45 | 69.7 vs 24.0 | Fatty liver index >60 | Ultrasound, mean IMT (10 measurements) | – | 6 |
| Karakurt et al72 2009 | Turkey | Not mentioned | 66 | 40 (60.6) | 53 vs 53 | 30.0 vs 42.3 | Ultrasound | Ultrasound, mean IMT (6 measurements) | – | 5 |
| Petit et al71 2009 | Europe | Hospital based, patients with T2DM | 101 | 61 (60.4) | 60.3 vs 60.1 | 44.2 vs 50.0 | MR spectroscopy, liver fat content >5.5% | Ultrasound, mean IMT (6 measurements) | – | 6 |
| Ramilli et al70 2009 | Europe | Outpatient clinic | 154 | 90 (58.4) | 59.3 vs 60.1 | 51.1 vs 45.3 | Ultrasound | Ultrasound, mean of max IMT (L&R) | Age, sex, BMI, Smk, HT, dyslipidaemia, DM | 8 |
| Fracanzani et al74 2008 | Europe | Hospital based | 375 | 125 (33.3) | 50.5 vs 52 | 87.2 vs 87.2 | Ultrasound+biopsy | Ultrasound, mean IMT (6 measurements) | – | 7 |
| Aygun et al75 2008 | Turkey | Hospital based | 80 | 40 (50.0) | 43.2 vs 38.8 | 47.5 vs 50.0 | Biopsy | Ultrasound | Age/sex-matched controls | 7 |
| Targher et al77 2006 1 | Europe | Outpatient clinic, patients with T2DM | 200 | 100 (50.0) | 55 vs 56 | 64.0 vs 67.0 | Ultrasound | Ultrasound, mean IMT (6 measurements) | Age/sex-matched controls | 7 |
| Targher et al76 2006 2 | Europe | Outpatient clinic | 245 | 85 (24.7) | 45 vs 45 | 58.8 vs 59.4 | Biopsy | Ultrasound, mean IMT (6 measurements) | Age, sex, BMI, Smk, LDL, HOMA-IR, MetS | 8 |
| Brea et al79 2005 | Europe | Hospital based | 80 | 30 (50.0) | 53.2 vs 51.6 | 50.0 vs 50.0 | Ultrasound | Ultrasound, mean IMT | – | 7 |
| Targher et al78 2005 | Europe | Outpatient clinic | 90 | 50 (55.5) | 46 vs 46 | 60.0 vs 65 | Biopsy | Ultrasound, mean IMT (6 measurements) | Age, Sex, HOMA-IR, MetS | 8 |
BMI, body mass index; L&R, left and right; MetS, metabolic syndrome; MR, magnetic resonance; NAFLD, non-alcoholic fatty liver disease; NOS, Newcastle–Ottawa Scale; T1DM, type 1 diabetes mellitus; T2DM, type 2 diabetes mellitus.
Table 2.
Characteristics of included studies which investigated the association between NAFLD and Increased CIMT
| Name, year | Study region | Study population | Study size | n (%) NAFLD | Age (NAFLD+ vs NAFLD−) | % male (NAFLD+ vs NAFLD−) | NAFLD assessment | Outcome assessment | Outcome definition | Confounder adjustment | NOS (max=9) |
| Mohammadzadeh et al44 2019 | Other: Iran | Hospital based | 300 | 150 (50.0) | 49.9 vs 52.5 | 65.3 vs 57.3 | Ultrasound | Ultrasound, mean IMT (L&R) | CIMT >0.8 | Age, BMI, HLP, HTN, DM | 8 |
| Tan et al80 2019 | Asia | Government officials (health screen) | 131 | 84 (64.1) | Overall=47.1 | 84.0 vs 60.7 | Ultrasound (Fibroscan, Controlled attenuation parameter (CAP) ≥263 dB/min) | Ultrasound, mean IMT (6 measurements) | CIMT >0.8 | Age, sex, WC, ALT, DM, HT | 8 |
| Oni et al22 2019 | North America | Population based | 4123 | 729 (17.7) | 61 vs 63 | 47.0 vs 44.0 | CT, LS ratio <1 | Ultrasound, mean internal carotid IMT (L&R) | CIMT >1.0 | Age, sex, ethnicity, SBP, fasting glucose, lipid-lowering meds, HT meds, LDL, Smk, BMI, logCRP | 9 |
| Yi et al45 2018 | Asia | Outpatient clinic | 1981 | 1888 (95.3) | 45.9 vs 44.8 | 63.4 vs 40.1 | Ultrasound | Ultrasound, mean of max IMT (L&R) | – | Sex, SBP, FPG, TG, TC, LDL, ALT, AST, GGT, Cr | 6 |
| Zheng et al28 2018 | Asia | Population based | 4112 | 1571 (38.2) | 56.2 vs 55.6 | 64.4 vs 35.6 | Ultrasound | Ultrasound, max IMT (L&R) | CIMT >0.8 | Age, sex, BMI, exercise, Smk, WC, TG, LDL, DM, HT | 9 |
| Martínez-Alvarado et al81 2014 | Mexican | Population based | 429 | 122 (28.4) | 52.1 vs 54.1 | 0.0 vs 0.0 | CT, LS ratio <1 | Ultrasound, mean IMT (10 measurements) | >75th sex/age-specific percentile | Age, HT, hypercholesterolaemia, hyperTAG, HDL, WC, HOMA-IR | 9 |
| Lankarani et al82 2013 | Other: Iran | Population based | 580 | 290 (50.0) | 46.4 vs 45.4 | 44.8 vs 40.0 | Ultrasound | Ultrasound, mean IMT (6 measurements) | CIMT >0.8 | Age, sex, WC, DM, HT, TAG, HDL | 9 |
| Huang et al62 2012 | Asia | Population based | 8632 | 2590 (30.0) | 58.5 vs 58.5 | 31.4 vs 30.9 | Ultrasound | Ultrasound, max IMT (L&R) | CIMT >0.8 | Age, sex, alcohol, Smk, exercise, BMI, LDL, central obesity, FBG, TG, BP, HDL, HOMA-IR | 9 |
| Kang et al61 2012 | Asia | Outpatient (health screen), MetS(−) participants | 413 | 157 (38.0) | 52.0 vs 52.5 | 51.0 vs 41.8 | Ultrasound | Ultrasound, mean IMT (L&R) | CIMT >1.0 | Age, BP, BMI, WC, lipid profile, liver enzymes | 8 |
| Thakur et al59 2012 | Asia | Hospital based | 80 | 40 (50.0) | 42.1 vs 41.9 | 67.5 vs 67.5 | Ultrasound | Ultrasound, mean IMT (6 measurements) | CIMT >0.556 | Generalised and abdominal obesity, MetS, fasting insulin, dyslipidaemia, SBP, DBP, hsCRP | 8 |
| Kim et al83 2009 | Asia | Population (health screen) | 1021 | 507 (49.7) | – | 62.5 vs 46.5 | Ultrasound | Ultrasound, mean of max IMT (L&R) | CIMT >0.8 | Age, sex, WC, SBP, fasting glucose, total/HDL cholesterol ratio, Smk, alcohol | 9 |
| Fracanzani et al74 2008 | Europe | Hospital based | 375 | 125 (33.3) | 50.5 vs 52.0 | 87.2 vs 87.2 | Ultrasound+biopsy | Ultrasound, mean IMT (6 measurements) | CIMT >0.64 | Sex, Smk, HDL, LDL, TAG, fasting glucose, MetS, DM, BMI, AAT | 8 |
| Brea et al79 2005 | Europe | Hospital based | 80 | 40 (50.0) | 53.2 vs 51.6 | 50.0 vs 50.0 | Ultrasound | Ultrasound, mean IMT | CIMT top quartile | Sex, age, BMI, SBP, DBP, DM, lab serum values | 8 |
BMI, body mass index; CIMT, carotid intima-media thickness; Cr, creatinine; DM, diabetes mellitus; L&R, left and right; MetS, metabolic syndrome; NAFLD, non-alcoholic fatty liver disease; NOS, Newcastle–Ottawa Scale.
Table 3.
Characteristics of included studies which investigated the association between NAFLD and CAC presence, development or progression
| Name, year | Study region | Study population | Study size | n (%) NAFLD | Age (NAFLD+ vs NAFLD−) | % male (NAFLD+ vs NAFLD−) | NAFLD assessment | Outcome assessment | Outcome definition | Confounder adjustment | NOS (max=9) |
| CAC presence (CAC >0 and CAC >100) | |||||||||||
| Jacobs et al85 2016 | North America | Population based | 250 | 71 (28.4) | 66.8 vs 67.8 | 43.7 vs 43.0 | CT, Liver Spleen ratio ≤1.1 | MDCT, Agatston method | CAC >100 & CAC >0 | Age, sex, HR, Smk, creatinine, BMI, alcohol, total cholesterol, HDL, TAG, VAT/SAT/WC | 9 |
| Chhabra et al92 2013 | North America | Population (health screen) | 377 | 43 (11.4) | Overall=57.1 | Overall=52.0 | CT, spleen minus liver >10 | MDCT, Agatston method | CAC >100 | Age, sex, Smk, LDL, HT, DM, MetS | 9 |
| Kim et al60 2012 | Asia | Population (health screen) | 4023 | 1617 (40.2) | 57.5 vs 56.4 | 73.0 vs 52.5 | Ultrasound | 16 & 64 slice MDCT, Agatston method | CAC >100 & CAC >0 | Age, sex, BMI, WC, alcohol, Smk, physical activity, DM, HT, total cholesterol, TAG, HDL, CRP | 9 |
| Chen et al94 2010 | Asia | Population (health screen) | 295 | 121 (41.0) | Overall=52.6 | Overall=65.8 | Ultrasound and CT | 64 slice MDCT, Agatston method | CAC >100 | Age, sex, BMI, Smk, HT, DM, fasting glucose, total cholesterol, TAG, HDL, LDL, ALT, AST, serum uric acid, gallbladder stones | 9 |
| Jung et al93 2010 | Asia | Population (health screen) | 928 | 219 (34.4) | 54.0 vs 51.7 | 72.8 vs 49.5 | Ultrasound | 64 slice MDCT, Agatston method | CAC >100 | Age, Sex, BMI, WHR, uric acid, SBP, DBP, GGT, TAG, HDL, fasting glucose, Smk, DM, HT, statins | 9 |
| Kim et al26 2020 | Asia | Population (health screen) | 7259 | 3328 (45.8) | Overall=54 | Overall=59.5 | Ultrasound | 64 slice MDCT, Agatston method | CAC >0 | Age, sex, HT, DM, obesity, abdominal obesity, eGFR, CRP, Smk, alcohol, AST, ALT, GGT | 9 |
| Oni et al22 2019 | North America | Population based | 4123 | 729 (17.7) | 61 vs 63 | 47 vs 44 | CT, LS ratio <1 | EBCT or MDCT, Agatston method | CAC >0 | Age, gender, ethnicity, SBP, fasting glucose, lipid-lowering meds, HT meds, LDL, Smk, BMI, logCRP | 9 |
| Chang et al27 2019 | Asia | Population (health screen) | 86 911 | 34 382 (39.6) | 41.1 vs 40.3 | 89.1 vs 64.7 | Ultrasound | 64 slice MDCT, Agatston method | CAC >0 | Age, sex, BMI, Smk, physical activity, education, total caloric intake, family history of CVD, DM, HT, LDL, meds, dyslipidaemia, hsCRP, HOMA-IR | 9 |
| Gummesson et al47 2018 | Europe | Population based | 1015 | 106 (10.4) | 58.3 vs 57.5 | 71.7 vs 52.5 | CT, liver HU <40 | MDCT, Agatston method | CAC >0 | Sex, age, education, BMI, alcohol, Smk, sedentary time, waist, VAT, physical activity, DM, HT, LDL, HDL, TG, CRP, insulin, hsCRP | 9 |
| Cho et al31 2018 | Asia | Population (health screen) | 798 | 272 (34.1) | 53.4 vs 54.1 | 91.2 vs 72.2 | Ultrasound | 64 slice MDCT, Agatston method | CAC >0 | Age, sex, BMI, Smk, alcohol, exercise, LDL-cholesterol, hsCRP | 9 |
| Lee et al29 2018 | Asia | Population (health screen) | 5121 | 1979 (38.6) | 54.0 vs 53.7 | 77.6 vs 62.1 | Ultrasound | 64 slice MDCT, Agatston method | CAC >10 | Age, sex, obesity, DM, HT, HLP, Smk, family history of CAD, hsCRP | 9 |
| Wu et al32 2017 | Asia | Population based | 2345 | 1272 (54.2) | Overall=55.7 | Overall=44.1 | Ultrasound | 64 slice MDCT, Agatston method | CAC >0 | Age, sex, Smk, HT, DM, HC, LDL, physical activity, education, income | 9 |
| Kim et al84 2016 | Asia | Population (health screen) | 1473 | 677 (46.0) | – | 68.4 vs 47.1 | Ultrasound | 64 slice MDCT, Agatston method | CAC >0 | Age, sex, Smk, alcohol, exercise, BMI, WC, SBP, total cholesterol, TAG, HDL, LDL, blood urea nitrogen, creatinine, glucose, hsCRP | 9 |
| Kang et al88 2015 | Asia | Population (health screen) | 772 | 346 (44.8) | 50.0 vs 48.6 | 83.5 vs 55.4 | Ultrasound | Presence of calcified coronary plaques | Presence of calcified plaques | Age, Smk, HT, DM, LDL, HDL, MetS | 8 |
| Mellinger et al86 2015 | North America | Population (health screen) | 3014 | 512 (17.0) | Overall=51.1 | Overall=49.5 | CT, liver phantom ratio <0.33 | MDCT, Agatston method | CAC >0 | Age, sex, alcohol, Smk, menopause, HRT, BMI | 9 |
| Kim et al87 2015 | Asia | Population (health screen), postmenopausal women | 754 | 129 (17.1) | 59.5 vs 57.1 | 0.0 vs 0.0 | Ultrasound | 64 slice MDCT, Agatston method | CAC >0 | Age, BMI, SBP, DBP, fasting glucose, total cholesterol, LDL, TAG, HDL, CRP, HOMA-IR | 8 |
| VanWagner et al89 2014 | North America | Population based | 2424 | 232 (9.57) | 50.5 vs 49.9 | 58.2 vs 41.1 | CT, liver HU ≤40 | ECG-gated CT, Agatston method | CAC >0 | Age, race, sex, study centre, income, education, alcohol, Smk, physical activity, BMI | 9 |
| Sung et al90 2012 | Asia | Population (health screen) | 10 153 | 3784 (37.3) | Overall=49.1 | Overall=76.3 | Ultrasound | 64 slice MDCT, Agatston method | CAC >0 | Age, sex, TAG, HDL, LDL, WC, SBP, alcohol, Smk, activity, Hx CHD, Hx HTN, Hx DM, HOMA-IR | 9 |
| Santos et al91 2007 | South America | Population (health screen) | 505 | 204 (40.4) | 48 vs 46 | 100 vs 100 | Ultrasound | EBCT, Agatston method | CAC >0 | Age, pulse pressure, BMI, Smk, alcohol, MetS, LDL, TG/HDL ratio, fasting glucose, BP medication, lipid medication, ALT/AST ratio, GGT | 9 |
| CAC development/progression | |||||||||||
| Cho et al31 2018 | Asia | Population (health screen), MetS(−) participants | 798 | 272 (34.1) | 53.4 vs 54.1 | 91.2 vs 72.2 | Ultrasound | 64 slice MDCT, Agatston method | Incident CAC or increase by >2.5 units between baseline & final square root of CAC score | Age, sex, BMI, Smk, alcohol, exercise, LDL-C, hsCRP, follow-up interval, baseline CAC score | 9 |
| Kang et al96 2017 | Asia | Population (health screen), non-obese participants | 447 | 105 (23.5) | Overall=54.1 | Overall=70.9 | Ultrasound | 64 slice MDCT, Agatston method | Incident CAC or increase by >2.5 units between baseline & final square root of CAC score | Age, sex, WC, alcohol, Smk, exercise, baseline CAC, LDL, hsCRP, follow-up interval | 8 |
| Kim et al95 2017 | Asia | Population (health screen) | 1575 | 734 (46.6) | 40.0 vs 398 | 94.8 vs 85.0 | Ultrasound | 64 slice MDCT, Agatston method | Any development (incidence) | Age, sex, ALT, Smk, FBS, LDL, BMI | 9 |
| Park et al34 2016 | Asia | Population (health screen) | 1732 | 846 (48.8) | 57.1 vs 57.4 | 81.3 vs 67.7 | Ultrasound | 256 slice MDCT, Agatston method | Development & progression (>10 CAC from baseline) | Age, sex, BMI, HT, DM, hypercholesterolaemia, TAG, HDL, GFR, Smk, WC, incident DM, lipid- lowering agents | 9 |
BMI, body mass index; CAC, coronary artery calcification; CAD, coronary artery disease; CRP, C reactive protein; CVD, cardiovascular disease; DM, diabetes mellitus; MetS, metabolic syndrome; NAFLD, non-alcoholic fatty liver disease; NOS, Newcastle–Ottawa Scale.
Measurement of exposures and outcomes
The presence of NAFLD was largely determined by US (n=46), with other studies using CT (n=8), biopsy (N=8), Fatty Liver Index (n=1) and MRS (n=1). Twenty-two studies investigated the effects of NAFLD on CAC score, with one study using the presence of calcified coronary artery plaques as a proxy for CAC >0. Forty-four studies investigated the effects of NAFLD on CIMT score. CIMT was assessed via B-mode US of bilateral carotid arteries, with majority of studies (n=18) commonly averaging the mean CIMT over six measurements (three on each carotid artery).
Methodological quality
Tables 1–3 and online supplemental table 1 detail the NOS risk of bias evaluation for the various studies. Of the 60 cross-sectional studies, the majority (n=35) were classified as ‘high quality’ (≥8 stars) with the remaining 25 classified as ‘moderate quality’ (5–7 stars). All four prospective studies were classified as ‘high quality’.
openhrt-2021-001850supp001.pdf (3.3MB, pdf)
Effect of NAFLD on CIMT
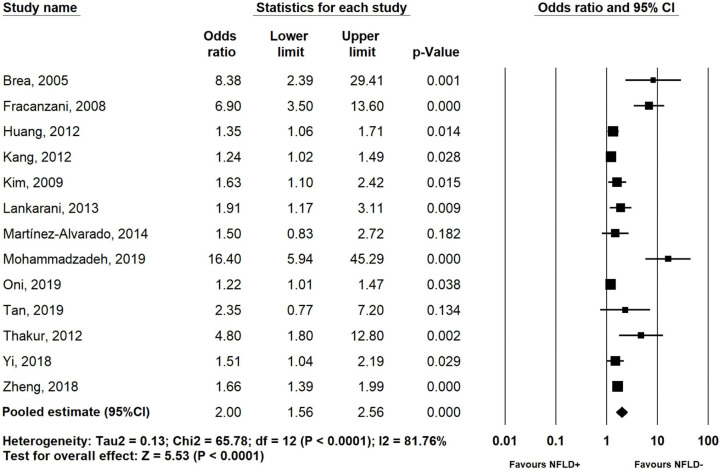
Figures 2 and 3 summarise the studies which investigated the effects of NAFLD on CIMT. Forty-four studies, with a total of 41 189 individuals, assessed the effect of NAFLD on CIMT. Thirty-nine studies investigated the mean differences in CIMT between NAFLD and controls,22 30 33 35 44–79 while 13 studies used logistic regression to quantify the associations between NAFLD and an ‘increased CIMT’.22 28 44 45 59 61 62 74 79–83 Increased CIMT was defined as >0.8 mm in six studies, >1.0 mm in two studies and via other stratification methods in the remaining five studies.
Figure 2.
Forest plots showing relationship between NAFLD and presence of increased CIMT. CIMT, carotid intima-media thickness; NFLD, non-alcoholic fatty liver disease.
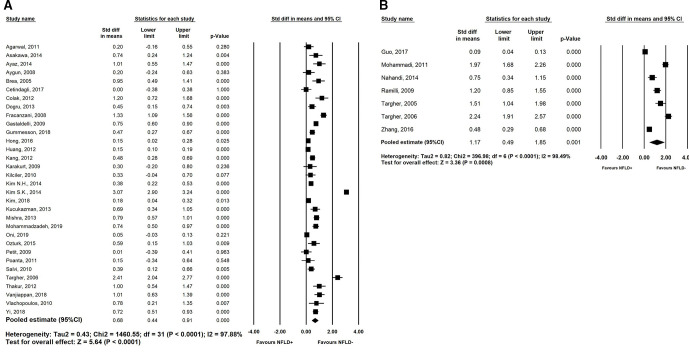
Figure 3.
(A) Forest plots showing pooled standard differences in unadjusted CIMT means between NAFLD(+) and NAFLD(−) groups. (B) Forest plots showing pooled standard differences in adjusted CIMT means between NAFLD(+) and NAFLD(−) groups. CIMT, carotid intima-media thickness; NFLD, non-alcoholic fatty liver disease.
Compared with participants without NAFLD, the presence of NAFLD was significantly associated with an increased CIMT, with a pooled OR of 2.00 (95% CI 1.56 to 2.56, Pheterogeneity<0.001, I2=81.8%, figure 2). Likewise, subjects with NAFLD had a higher mean CIMT than subjects without, both across studies which adjusted for confounders (pooled Std Diff in means: 1.17, 95% CI: 0.49 to 1.85, figure 3B), and in studies which compared unadjusted means (pooled Std Diff in means: 0.68, 95% CI: 0.44 to 0.91, figure 3A). For all CIMT outcomes, a sensitivity analysis including only studies of ‘high quality’ was performed, with similar results obtained.
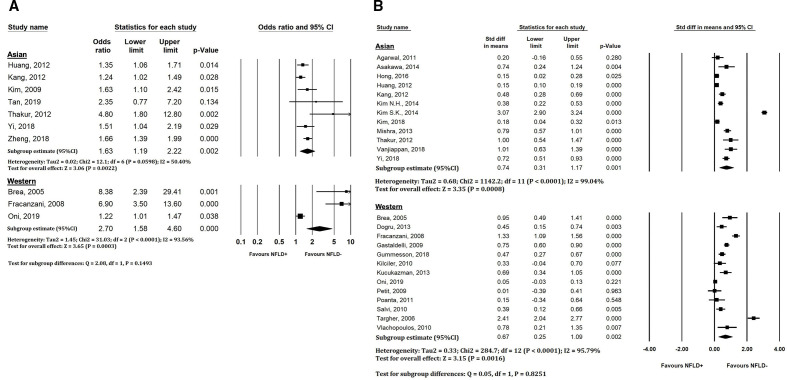
Subgroup analyses
We further stratified the associations between NAFLD and an increased risk of increased CIMT by study region (figure 4A). The pooled ORs for increased CIMT were (OR: 1.63, 95% CI: 1.19 to 2.22, Pheterogeneity=0.06, I2=50%, n=7 studies) in Asian populations vs (OR: 2.70, 95% CI: 1.58 to 4.60, Pheterogeneity<0.001, I2=93.6%, n=3 studies) in Western populations (Pdifference=0.15). Likewise, the pooled Std Diff in mean CIMT were 0.75 (95% CI: 0.31 to 1.17) in Asian populations (n=12 studies) vs 0.67 (95% CI: 0.25 to 1.09) in Western populations (Pdifference=0.83) (figure 4B). Lastly, when analysing the subset of studies conducted on participants with T2DM, no Std Diff in CIMT means were found between those with and without NAFLD (Std Diff in means: 0.99, 95% CI:−0.21 to 2.20, n=7 studies) (online supplemental figure 1).
Figure 4.
(A) Forest plots showing relationship between NAFLD and presence of increased CIMT, stratified by region of study. (B) Forest plots showing pooled standard differences in CIMT means between NAFLD(+) and NAFLD(−) groups, stratified by region of study. CIMT, carotid intima-media thickness; NFLD, non-alcoholic fatty liver disease.
Effect of NAFLD on CAC score
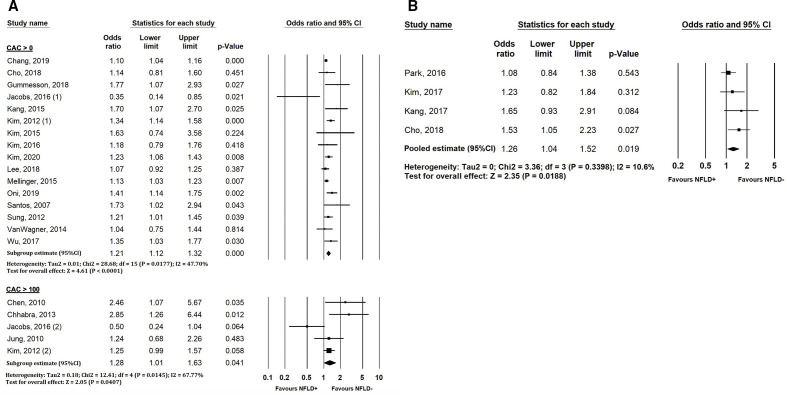
Figure 5 summarises the studies investigating the associations between NAFLD and CAC score. Twenty-two studies, with a total of 136 294 individuals, assessed the effect of NAFLD on CAC score. Sixteen studies investigated the cross-sectional associations between NAFLD and the presence of CAC score >0,22 26 27 29 31 32 47 60 84–91 five studies investigated the cross-sectional associations between NAFLD and the presence of CAC score >100,60 80 85 92–94 and four studies investigated the longitudinal influence of NAFLD on CAC score progression/development.31 34 95 96
Figure 5.
(A) Forest plots showing relationship between NAFLD and CAC scores >0 and >100. (B) Forest plots showing relationship between NAFLD and the development/progression of CAC. CAC, coronary artery calcification; NFLD, non-alcoholic fatty liver disease.
Compared with participants without NAFLD, the presence of NAFLD was significantly associated with the presence of both CAC score >0 (pooled OR: 1.21, 95% CI 1.12 to 1.32, Pheterogeneity=0.018, I2=47.7%), and CAC score >100 (pooled OR: 1.28, 95% CI 1.01 to 1.63, Pheterogeneity=0.015, I2=67.8%), (figure 5A). Likewise, NAFLD was significantly associated with the development/progression of CAC with a pooled OR of 1.26 (95% CI 1.04 to 1.52, Pheterogeneity=0.34, I2=10.6%) (figure 5B).
Subgroup analyses
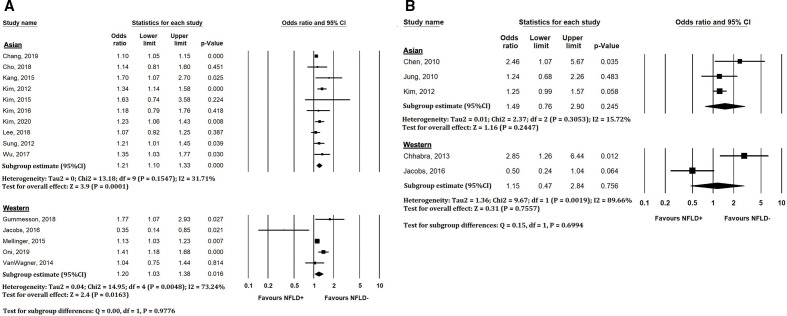
As with the CIMT analysis, we further stratified the associations of NAFLD with CAC score >0 based on ethnicity (figure 6). The pooled associations between NAFLD and CAC score >0 were (OR: 1.21 95% CI 1.10 to 1.33, Pheterogeneity=0.15, I2=31.7%, n=10 studies) in Asian populations vs (OR: 1.20 95% CI 1.03 to 1.38, Pheterogeneity=0.004, I2=73%, n=5 studies) in Western populations (Pdifference=0.98). There were too few studies to conclusively compare ethnic differences for the associations with CAC score >100, or for the progression/development of CAC.
Figure 6.
(A) Forest plots showing relationship between NAFLD and CAC score >0, stratified by region of study. (B) Forest plots showing relationship between NAFLD and CAC score >100, stratified by region of study. CAC, coronary artery calcification; NFLD, non-alcoholic fatty liver disease.
Evaluation of publication bias
When assessing the studies that investigated the relationships between NAFLD and CIMT, the funnel plot showed asymmetry (online supplemental figures 2 and 3), with studies favouring increased Std Diff in means CIMT (Egger’s, p<0.05) and positive ORs for increased CIMT (Egger’s, p=0.002). For studies investigating the relationships between NAFLD and CAC outcomes (online supplemental figures 4 and 5), the funnel plots excluded bias with symmetrical distribution of studies on both sides of the mean, while the Egger’s test was non-significant (p=0.07 for CAC presence, and p=0.15 for CAC progression/development).
Discussion
In this meta-analysis, we evaluated the associations of NAFLD with two established markers of subclinical atherosclerosis, synthesising the results of 64 published studies with a total of 172 385 patients. In line with existing literature, we have demonstrated that subjects with NAFLD have an increased risk of prevalent subclinical atherosclerosis than those without, even after adjustment for common cardiometabolic risk factors. Our subgroup analyses also revealed these associations to be consistent across both Western and Asian populations. This is also the first meta-analysis to demonstrate that subjects with NAFLD are at increased risk of development and progression of subclinical atherosclerosis. This may provide additional insights into screening and surveillance strategies for patients with NAFLD,2 potentially identifying higher-risk NAFLD populations, and may also provide further insight into the role of NAFLD in the development of CVD.
Our meta-analysis serves as a timely update to build on the previous work of Zhou et al, Kapuria et al and Jaruvongvanich et al,23–25 incorporating the results of over 21 new studies published from 2016 and 2020, comprising over 100 000 participants (~50 000 of which have NAFLD). The inclusion of these new studies enables us to conduct a more robust analysis of the differences between ethnic populations, with a larger number of studies conducted in both Western and Asian populations. Our overall findings of the associations between NAFLD and an increased risk of subclinical atherosclerosis (as measured by CIMT and/or CAC score) are in agreement with existing literature, further reinforcing the findings of previous studies and meta-analyses.11 12 23–25 97 In addition to these associations with subclinical atherosclerosis, other meta-analyses have also found NAFLD to be significantly associated with increased cardiovascular mortality, coronary artery disease (CAD), incident CVD events, and other subclinical manifestations of CVD including abnormalities in myocardial metabolism, ventricular structure and function.98–100 Our findings reiterate how the increased risk of CVD in patients with NAFLD can be attributed to an increased underlying subclinical atherosclerotic burden, and suggest that patients with NAFLD should be considered at high risk of atherosclerotic CVD.
Interestingly, we did not observe differential associations between NAFLD and both CAC or CIMT across Asian and Western populations. Our subgroup analyses found similar associations between NAFLD and CAC in both Asian (OR: 1.21 (1.10 to 1.33)) and Western regions (OR: 1.20 (1.03 to 1.38)), with a Pdifference=0.98. Likewise, similar associations between NAFLD and increased CIMT were found across both regions. Despite literature suggesting ethnic differences in the pathogenesis, severity and outcomes of NAFLD,14 17 101 remarkably few studies have specifically investigated these ethnic differences in the context of associations with subclinical atherosclerosis. The Multi-Ethnic Study of Atherosclerosis found a positive association between NAFLD and both CAC and increased CIMT in white and Hispanic individuals, but not in Chinese individuals.21 22 While we did not specifically look at ethnic differences, our results show that NAFLD serves as an important atherogenic risk factor in both Western and Asian populations.
The associations between NAFLD and atherosclerotic CVD were originally considered epiphenomena due to a shared confluence of metabolic risk factors.102 However, increasingly, evidence has now recognised that NAFLD is an independent risk factor for CVD, with NAFLD thought to play an active role in the systemic release of proatherogenic and proinflammatory mediators, with additional contributions to insulin resistance and abnormal atherogenic lipid profiles, all of which increase the risk of atherogenesis.3 5 8 These potential pathways and mechanisms are covered in detail in other reviews.2 3 103 Nonetheless, the interplay between NAFLD, MetS, diabetes and CVD remains complex. Evidence on the effect of NAFLD on subclinical atherosclerosis within subjects with T2DM, for example, remains equivocal.33 52 71 77 In our subgroup analysis of studies conducted within populations with T2DM, our forest plots did not show significant Std Diff in mean CIMT between those with NAFLD and those without (p=0.107). Diabetes is a potent risk factor for both CAD and CVD, and may have thus masked subtler associations between NAFLD and subclinical atherosclerosis. Alternatively, this may also highlight the role of insulin resistance in mediating the relationship between NAFLD and atherosclerosis.104
Only recently have studies begun to investigate the longitudinal associations between NAFLD and CAC progression/development, with this paper being the first meta-analysis to synthesise the results of four studies published from 2016 onwards.31 34 95 96 We demonstrated that patients with NAFLD are at greater risk of development/progression of CAC, even after adjustment for known confounders. While our results do not elucidate the exact pathophysiological mechanisms by which NAFLD may affect CAC development/progression, they do provide insight into the causal relationship between NAFLD and subclinical atherosclerosis. It should be noted that Park et al found differential associations between NAFLD and CAC development and progression, reporting that NAFLD might play a role in the early development of atherosclerosis, but not in the progression to more severe degrees of atherosclerosis.34 Future studies may be warranted to confirm such observations.
Strengths of our study include the large participant numbers, the assessment of various modalities of subclinical atherosclerosis including CIMT and CAC, the large number of studies from both Western and Asian populations enabling robust analysis of regional differences, and our analysis of not just cross-sectional, but longitudinal outcomes (CAC development and progression). Nonetheless, our results should be interpreted with caution, taking into consideration certain limitations. Heterogeneity was consistently present across the different subclinical atherosclerotic outcomes. This can be attributed to differences in study design, population characteristics, the use of different cut-off definitions for both CAC and increased CIMT, the adjustment for different cardiometabolic confounders and the different modalities of NAFLD diagnosis. In addition, even though liver biopsy remains the gold standard for NAFLD evaluation, US was the most common modality used in the NAFLD assessment in the included studies, and is cited to have diminished accuracy when it comes to the diagnosis of milder hepatic steatosis.105 106 While we did not find regional differences in the results, we could not perform actual ethnic comparisons as these data were not available. Whether these regional data accurately reflect ethnic data is uncertain and also the influence of cultural and socioeconomic factors cannot be quantified. Nevertheless, this provides one of the first combined regional comparison of such results. Finally, potential publication bias exists with regard to the studies investigating CIMT-related outcomes.
Conclusion
In conclusion, this meta-analysis reports a significant positive association between NAFLD and subclinical atherosclerosis, as defined by increased CIMT and CAC scores. These observed associations are not just cross-sectional, but also longitudinal, and are seen across both Western and Asian populations. These results re-emphasise the importance of early risk evaluation and prophylactic intervention measures to preclude progression to clinical CVD in NAFLD.
Footnotes
MYZW and JJLY contributed equally.
Contributors: MYZW—conception of idea, crafting of research question, design of inclusion/exclusion criteria, collection of data (literature search), statistical analysis, quality (risk of bias) evaluation, figure creation, writing of the manuscript, and writing and drafting of the manuscript, guarantor. JJLY—conception of idea, crafting of research question, design of inclusion/exclusion criteria, collection of data (literature search), drafting of the manuscript and editing of the manuscript. RS—design of inclusion/exclusion criteria, collection of data (literature search), statistical analysis, quality (risk of bias) evaluation, figure creation and editing of the manuscript, guarantor. MC—conception of idea, crafting of research question, design of inclusion/exclusion criteria and editing of the manuscript. GBBG—conception of idea, crafting of research question, design of inclusion/exclusion criteria and editing of the manuscript. KKY—conception of idea, crafting of research question, design of inclusion/exclusion criteria, editing of the manuscript and guarantor.
Funding: KKY has received research funding from Medtronic, Boston Scientific, Amgen, AstraZeneca, Shockwave Medical (all significant, via institution); consulting or honoraria fees (all modest) from Medtronic, Boston Scientific, Abbott Vascular, Amgen, Bayer, Novartis; speaker or proctor fees from Abbott Vascular, Boston Scientific, Medtronic, Philips, Shockwave Medical, Alvimedica, Menarini, AstraZeneca, Amgen, Bayer.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement
Data are available upon reasonable request.
Ethics statements
Patient consent for publication
Not required.
Ethics approval
Ethical approval is not applicable for this systematic review/meta-analysis.
References
- 1.Angulo P. Nonalcoholic fatty liver disease. N Engl J Med 2002;346:1221–31. 10.1056/NEJMra011775 [DOI] [PubMed] [Google Scholar]
- 2.Anstee QM, Targher G, Day CP. Progression of NAFLD to diabetes mellitus, cardiovascular disease or cirrhosis. Nat Rev Gastroenterol Hepatol 2013;10:330–44. 10.1038/nrgastro.2013.41 [DOI] [PubMed] [Google Scholar]
- 3.Stahl EP, Dhindsa DS, Lee SK, et al. Nonalcoholic fatty liver disease and the Heart: JACC state-of-the-art review. J Am Coll Cardiol 2019;73:948–63. 10.1016/j.jacc.2018.11.050 [DOI] [PubMed] [Google Scholar]
- 4.Loomba R, Sanyal AJ. The global NAFLD epidemic. Nat Rev Gastroenterol Hepatol 2013;10:686–90. 10.1038/nrgastro.2013.171 [DOI] [PubMed] [Google Scholar]
- 5.Adams LA, Anstee QM, Tilg H, et al. Non-alcoholic fatty liver disease and its relationship with cardiovascular disease and other extrahepatic diseases. Gut 2017;66:1138–53. 10.1136/gutjnl-2017-313884 [DOI] [PubMed] [Google Scholar]
- 6.Marchesini G, Brizi M, Bianchi G, et al. Nonalcoholic fatty liver disease: a feature of the metabolic syndrome. Diabetes 2001;50:1844–50. 10.2337/diabetes.50.8.1844 [DOI] [PubMed] [Google Scholar]
- 7.Musso G, Gambino R, Bo S, et al. Should nonalcoholic fatty liver disease be included in the definition of metabolic syndrome?: a cross-sectional comparison with adult treatment panel III criteria in nonobese nondiabetic subjects. Diabetes Care 2008;31:562–8. 10.2337/dc07-1526 [DOI] [PubMed] [Google Scholar]
- 8.Targher G, Day CP, Bonora E. Risk of cardiovascular disease in patients with nonalcoholic fatty liver disease. N Engl J Med 2010;363:1341–50. 10.1056/NEJMra0912063 [DOI] [PubMed] [Google Scholar]
- 9.Fuchs M, Sanyal AJ. Lipotoxicity in NASH. J Hepatol 2012;56:291–3. 10.1016/j.jhep.2011.05.019 [DOI] [PubMed] [Google Scholar]
- 10.Targher G, Bertolini L, Rodella S, et al. Nonalcoholic fatty liver disease is independently associated with an increased incidence of cardiovascular events in type 2 diabetic patients. Diabetes Care 2007;30:2119–21. 10.2337/dc07-0349 [DOI] [PubMed] [Google Scholar]
- 11.Oni ET, Agatston AS, Blaha MJ, et al. A systematic review: burden and severity of subclinical cardiovascular disease among those with nonalcoholic fatty liver; should we care? Atherosclerosis 2013;230:258–67. 10.1016/j.atherosclerosis.2013.07.052 [DOI] [PubMed] [Google Scholar]
- 12.Sookoian S, Pirola CJ. Non-Alcoholic fatty liver disease is strongly associated with carotid atherosclerosis: a systematic review. J Hepatol 2008;49:600–7. 10.1016/j.jhep.2008.06.012 [DOI] [PubMed] [Google Scholar]
- 13.Cai J, Zhang S, Huang W. Association between nonalcoholic fatty liver disease and carotid atherosclerosis: a meta-analysis. Int J Clin Exp Med 2015;8:7673–8. [PMC free article] [PubMed] [Google Scholar]
- 14.Farrell GC, Wong VW-S, Chitturi S. Nafld in Asia—as common and important as in the West. Nat Rev Gastroenterol Hepatol 2013;10:307–18. 10.1038/nrgastro.2013.34 [DOI] [PubMed] [Google Scholar]
- 15.Mohanty SR, Troy TN, Huo D, et al. Influence of ethnicity on histological differences in non-alcoholic fatty liver disease. J Hepatol 2009;50:797–804. 10.1016/j.jhep.2008.11.017 [DOI] [PubMed] [Google Scholar]
- 16.Rich NE, Oji S, Mufti AR, et al. Racial and ethnic disparities in nonalcoholic fatty liver disease prevalence, severity, and outcomes in the United States: a systematic review and meta-analysis. Clin Gastroenterol Hepatol 2018;16:198–210. 10.1016/j.cgh.2017.09.041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pan J-J, Fallon MB. Gender and racial differences in nonalcoholic fatty liver disease. World J Hepatol 2014;6:274–83. 10.4254/wjh.v6.i5.274 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wong RJ, Ahmed A. Obesity and non-alcoholic fatty liver disease: disparate associations among Asian populations. World J Hepatol 2014;6:263–73. 10.4254/wjh.v6.i5.263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yoon K-H, Lee J-H, Kim J-W, et al. Epidemic obesity and type 2 diabetes in Asia. The Lancet 2006;368:1681–8. 10.1016/S0140-6736(06)69703-1 [DOI] [PubMed] [Google Scholar]
- 20.Remigio-Baker RA, Allison MA, Forbang NI, et al. Race/ethnic and sex disparities in the non-alcoholic fatty liver disease-abdominal aortic calcification association: the multi-ethnic study of atherosclerosis. Atherosclerosis 2017;258:89–96. 10.1016/j.atherosclerosis.2016.11.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Al Rifai M, Silverman MG, Nasir K, et al. The association of nonalcoholic fatty liver disease, obesity, and metabolic syndrome, with systemic inflammation and subclinical atherosclerosis: the multi-ethnic study of atherosclerosis (MESA). Atherosclerosis 2015;239:629–33. 10.1016/j.atherosclerosis.2015.02.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Oni E, Budoff MJ, Zeb I, et al. Nonalcoholic fatty liver disease is associated with arterial distensibility and carotid intima-media thickness: (from the multi-ethnic study of atherosclerosis). Am J Cardiol 2019;124:534–8. 10.1016/j.amjcard.2019.05.028 [DOI] [PubMed] [Google Scholar]
- 23.Jaruvongvanich V, Wirunsawanya K, Sanguankeo A, et al. Nonalcoholic fatty liver disease is associated with coronary artery calcification: a systematic review and meta-analysis. Digestive and Liver Disease 2016;48:1410–7. 10.1016/j.dld.2016.09.002 [DOI] [PubMed] [Google Scholar]
- 24.Kapuria D, Takyar VK, Etzion O, et al. Association of hepatic steatosis with subclinical atherosclerosis: systematic review and Meta‐Analysis. Hepatology Communications 2018;2:877–87. 10.1002/hep4.1199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhou Y-Y, Zhou X-D, Wu S-J, et al. Nonalcoholic fatty liver disease contributes to subclinical atherosclerosis: a systematic review and meta-analysis. Hepatol Commun 2018;2:376–92. 10.1002/hep4.1155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kim S-H, Park HY, Lee HS, et al. Association between non-alcoholic fatty liver disease and coronary calcification depending on sex and obesity. Sci Rep 2020;10:1025. 10.1038/s41598-020-57894-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chang Y, Ryu S, Sung K-C, et al. Alcoholic and non-alcoholic fatty liver disease and associations with coronary artery calcification: evidence from the Kangbuk Samsung health study. Gut 2019;68:1667–75. 10.1136/gutjnl-2018-317666 [DOI] [PubMed] [Google Scholar]
- 28.Zheng J, Zhou Y, Zhang K, et al. Association between nonalcoholic fatty liver disease and subclinical atherosclerosis: a cross-sectional study on population over 40 years old. BMC Cardiovasc Disord 2018;18:147. 10.1186/s12872-018-0877-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lee SB, Park G-M, Lee J-Y, et al. Association between non-alcoholic fatty liver disease and subclinical coronary atherosclerosis: an observational cohort study. J Hepatol 2018;68:1018–24. 10.1016/j.jhep.2017.12.012 [DOI] [PubMed] [Google Scholar]
- 30.Kim H-J, Park H-B, Suh Y, et al. Comparison of carotid intima-media thickness and coronary artery calcium score for estimating subclinical atherosclerosis in patients with fatty liver disease. Cardiovasc J Afr 2018;29:93–8. 10.5830/CVJA-2017-052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cho YK, Kang YM, Yoo JH, et al. The impact of non-alcoholic fatty liver disease and metabolic syndrome on the progression of coronary artery calcification. Sci Rep 2018;8:12004. 10.1038/s41598-018-30465-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wu R, Hou F, Wang X, et al. Nonalcoholic fatty liver disease and coronary artery calcification in a northern Chinese population: a cross sectional study. Sci Rep 2017;7:9933. 10.1038/s41598-017-09851-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Guo K, Zhang L, Lu J, et al. Non-Alcoholic fatty liver disease is associated with late but not early atherosclerotic lesions in Chinese inpatients with type 2 diabetes. J Diabetes Complications 2017;31:80–5. 10.1016/j.jdiacomp.2016.09.008 [DOI] [PubMed] [Google Scholar]
- 34.Park HE, Kwak M-S, Kim D, et al. Nonalcoholic fatty liver disease is associated with coronary artery calcification development: a longitudinal study. J Clin Endocrinol Metab 2016;101:3134–43. 10.1210/jc.2016-1525 [DOI] [PubMed] [Google Scholar]
- 35.Hong HC, Hwang SY, Ryu JY, et al. The synergistic impact of nonalcoholic fatty liver disease and metabolic syndrome on subclinical atherosclerosis. Clin Endocrinol 2016;84:203–9. 10.1111/cen.12940 [DOI] [PubMed] [Google Scholar]
- 36.Stroup DF, Berlin JA, Morton SC, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. meta-analysis of observational studies in epidemiology (moose) group. JAMA 2000;283:2008–12. 10.1001/jama.283.15.2008 [DOI] [PubMed] [Google Scholar]
- 37.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med 2009;6:e1000100. 10.1371/journal.pmed.1000100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Deeks JJ, Dinnes J, D'Amico R. Evaluating non-randomised intervention studies. In: Health technology assessment. 7. Winchester, England, 2003: 1–173. [DOI] [PubMed] [Google Scholar]
- 39.Herzog R, Álvarez-Pasquin MJ, Díaz C, et al. Are healthcare workers' intentions to vaccinate related to their knowledge, beliefs and attitudes? A systematic review. BMC Public Health 2013;13:154. 10.1186/1471-2458-13-154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wells GSB, O'Connell J, Robertson J. The Newcastle-Ottawa scale (NOS) for assessing the quality of nonrandomised studies in meta-analysis, 2011. Available: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp
- 41.McPheeters ML, Kripalani S, Peterson NB, et al. Closing the quality gap: revisiting the state of the science (Vol. 3: quality improvement interventions to address health disparities). Evid Rep Technol Assess 2012:1–475. [PMC free article] [PubMed] [Google Scholar]
- 42.Chmielowska M, Fuhr DC. Intimate partner violence and mental ill health among global populations of Indigenous women: a systematic review. Soc Psychiatry Psychiatr Epidemiol 2017;52:689–704. 10.1007/s00127-017-1375-z [DOI] [PubMed] [Google Scholar]
- 43.Lo CK-L, Mertz D, Loeb M. Newcastle-Ottawa scale: comparing reviewers' to authors' assessments. BMC Med Res Methodol 2014;14:45. 10.1186/1471-2288-14-45 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Mohammadzadeh A, Shahkarami V, Shakiba M, et al. Association of non-alcoholic fatty liver disease with increased carotid intima-media thickness considering other cardiovascular risk factors. Iranian Journal of Radiology 2019;16. 10.5812/iranjradiol.14260 [DOI] [Google Scholar]
- 45.Yi X, Liu Y-H, Zhou X-F, et al. The influence of abdominal and ectopic fat accumulation on carotid intima-media thickness: a Chongqing study. J Stroke Cerebrovasc Dis 2018;27:1992–7. 10.1016/j.jstrokecerebrovasdis.2018.02.052 [DOI] [PubMed] [Google Scholar]
- 46.Vanjiappan S, Hamide A, Ananthakrishnan R, et al. Nonalcoholic fatty liver disease in patients with type 2 diabetes mellitus and its association with cardiovascular disease. Diabetes Metab Syndr 2018;12:479–82. 10.1016/j.dsx.2018.01.001 [DOI] [PubMed] [Google Scholar]
- 47.Gummesson A, Strömberg U, Schmidt C, et al. Non-alcoholic fatty liver disease is a strong predictor of coronary artery calcification in metabolically healthy subjects: a cross-sectional, population-based study in middle-aged subjects. PLoS One 2018;13:e0202666. 10.1371/journal.pone.0202666 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Cetindağlı I, Kara M, Tanoglu A, et al. Evaluation of endothelial dysfunction in patients with nonalcoholic fatty liver disease: association of selenoprotein P with carotid intima-media thickness and endothelium-dependent vasodilation. Clin Res Hepatol Gastroenterol 2017;41:516–24. 10.1016/j.clinre.2017.01.005 [DOI] [PubMed] [Google Scholar]
- 49.Zhang L, Guo K, Lu J, et al. Nonalcoholic fatty liver disease is associated with increased carotid intima-media thickness in type 1 diabetic patients. Sci Rep 2016;6:26805. 10.1038/srep26805 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ozturk K, Uygun A, Guler AK, et al. Nonalcoholic fatty liver disease is an independent risk factor for atherosclerosis in young adult men. Atherosclerosis 2015;240:380–6. 10.1016/j.atherosclerosis.2015.04.009 [DOI] [PubMed] [Google Scholar]
- 51.Nahandi MZ, Khoshbaten M, Ramazanzadeh E, et al. Effect of non-alcoholic fatty liver disease on carotid artery intima-media thickness as a risk factor for atherosclerosis. Gastroenterol Hepatol Bed Bench 2014;7:55–62. [PMC free article] [PubMed] [Google Scholar]
- 52.Kim S-K, Choi YJ, Huh BW, et al. Nonalcoholic fatty liver disease is associated with increased carotid intima-media thickness only in type 2 diabetic subjects with insulin resistance. J Clin Endocrinol Metab 2014;99:1879–84. 10.1210/jc.2013-4133 [DOI] [PubMed] [Google Scholar]
- 53.Kim NH, Park J, Kim SH, et al. Non-alcoholic fatty liver disease, metabolic syndrome and subclinical cardiovascular changes in the general population. Heart 2014;100:938–43. 10.1136/heartjnl-2013-305099 [DOI] [PubMed] [Google Scholar]
- 54.Ayaz T, Kirbas A, Durakoglugil T, et al. The relation between carotid intima media thickness and serum osteoprotegerin levels in nonalcoholic fatty liver disease. Metab Syndr Relat Disord 2014;12:283–9. 10.1089/met.2013.0151 [DOI] [PubMed] [Google Scholar]
- 55.Asakawa Y, Takekawa H, Suzuki K, et al. Relationship between fatty liver disease and intracranial main artery stenosis. Intern Med 2014;53:1259–64. 10.2169/internalmedicine.53.2067 [DOI] [PubMed] [Google Scholar]
- 56.Mishra S, Yadav D, Gupta M, et al. A study of carotid atherosclerosis in patients with non-alcoholic fatty liver disease. Ind J Clin Biochem 2013;28:79–83. 10.1007/s12291-012-0286-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kucukazman M, Ata N, Yavuz B, et al. Evaluation of early atherosclerosis markers in patients with nonalcoholic fatty liver disease. Eur J Gastroenterol Hepatol 2013;25:147–51. 10.1097/MEG.0b013e32835a58b1 [DOI] [PubMed] [Google Scholar]
- 58.Dogru T, Genc H, Tapan S, et al. Plasma fetuin-A is associated with endothelial dysfunction and subclinical atherosclerosis in subjects with nonalcoholic fatty liver disease. Clin Endocrinol 2013;78:712–7. 10.1111/j.1365-2265.2012.04460.x [DOI] [PubMed] [Google Scholar]
- 59.Thakur ML, Sharma S, Kumar A, et al. Nonalcoholic fatty liver disease is associated with subclinical atherosclerosis independent of obesity and metabolic syndrome in Asian Indians. Atherosclerosis 2012;223:507–11. 10.1016/j.atherosclerosis.2012.06.005 [DOI] [PubMed] [Google Scholar]
- 60.Kim D, Choi S-Y, Park EH, et al. Nonalcoholic fatty liver disease is associated with coronary artery calcification. Hepatology 2012;56:605–13. 10.1002/hep.25593 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kang JH, Cho KI, Kim SM, et al. Relationship between nonalcoholic fatty liver disease and carotid artery atherosclerosis beyond metabolic disorders in non-diabetic patients. J Cardiovasc Ultrasound 2012;20:126–33. 10.4250/jcu.2012.20.3.126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Huang Y, Bi Y, Xu M, et al. Nonalcoholic fatty liver disease is associated with atherosclerosis in middle-aged and elderly Chinese. Arterioscler Thromb Vasc Biol 2012;32:2321–6. 10.1161/ATVBAHA.112.252957 [DOI] [PubMed] [Google Scholar]
- 63.Colak Y, Karabay CY, Tuncer I, et al. Relation of epicardial adipose tissue and carotid intima-media thickness in patients with nonalcoholic fatty liver disease. Eur J Gastroenterol Hepatol 2012;24:613–8. 10.1097/MEG.0b013e3283513f19 [DOI] [PubMed] [Google Scholar]
- 64.Poanta LI, Albu A, Fodor D. Association between fatty liver disease and carotid atherosclerosis in patients with uncomplicated type 2 diabetes mellitus. Med Ultrason 2011;13:215–9. [PubMed] [Google Scholar]
- 65.Mohammadi A, Bazazi A, Ghasemi-Rad M. Evaluation of atherosclerotic findings in patients with nonalcoholic fatty liver disease. Int J Gen Med 2011;4:717–22. 10.2147/IJGM.S25753 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Agarwal AK, Jain V, Singla S, et al. Prevalence of non-alcoholic fatty liver disease and its correlation with coronary risk factors in patients with type 2 diabetes. J Assoc Physicians India 2011;59:351–4. [PubMed] [Google Scholar]
- 67.Vlachopoulos C, Manesis E, Baou K, et al. Increased arterial stiffness and impaired endothelial function in nonalcoholic fatty liver disease: a pilot study. Am J Hypertens 2010;23:1183–9. 10.1038/ajh.2010.144 [DOI] [PubMed] [Google Scholar]
- 68.Salvi P, Ruffini R, Agnoletti D, et al. Increased arterial stiffness in nonalcoholic fatty liver disease: the Cardio-GOOSE study. J Hypertens 2010;28:1699–707. 10.1097/HJH.0b013e32833a7de6 [DOI] [PubMed] [Google Scholar]
- 69.Kilciler G, Genc H, Tapan S, et al. Mean platelet volume and its relationship with carotid atherosclerosis in subjects with non-alcoholic fatty liver disease. Ups J Med Sci 2010;115:253–9. 10.3109/03009734.2010.500062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ramilli S, Pretolani S, Muscari A, et al. Carotid lesions in outpatients with nonalcoholic fatty liver disease. World J Gastroenterol 2009;15:4770–4. 10.3748/wjg.15.4770 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Petit JM, Guiu B, Terriat B, et al. Nonalcoholic fatty liver is not associated with carotid intima-media thickness in type 2 diabetic patients. J Clin Endocrinol Metab 2009;94:4103–6. 10.1210/jc.2009-0541 [DOI] [PubMed] [Google Scholar]
- 72.Karakurt F, Carlioglu A, Koktener A, et al. Relationship between cerebral arterial Pulsatility and carotid intima media thickness in diabetic and non-diabetic patients with non-alcoholic fatty liver disease. J Endocrinol Invest 2009;32:63–8. 10.1007/BF03345681 [DOI] [PubMed] [Google Scholar]
- 73.Gastaldelli A, Kozakova M, Højlund K, et al. Fatty liver is associated with insulin resistance, risk of coronary heart disease, and early atherosclerosis in a large European population. Hepatology 2009;49:1537–44. 10.1002/hep.22845 [DOI] [PubMed] [Google Scholar]
- 74.Fracanzani AL, Burdick L, Raselli S, et al. Carotid artery intima-media thickness in nonalcoholic fatty liver disease. Am J Med 2008;121:72–8. 10.1016/j.amjmed.2007.08.041 [DOI] [PubMed] [Google Scholar]
- 75.Aygun C, Kocaman O, Sahin T, et al. Evaluation of metabolic syndrome frequency and carotid artery intima-media thickness as risk factors for atherosclerosis in patients with nonalcoholic fatty liver disease. Dig Dis Sci 2008;53:1352–7. 10.1007/s10620-007-9998-7 [DOI] [PubMed] [Google Scholar]
- 76.Targher G, Bertolini L, Padovani R, et al. Relations between carotid artery wall thickness and liver histology in subjects with nonalcoholic fatty liver disease. Diabetes Care 2006;29:1325–30. 10.2337/dc06-0135 [DOI] [PubMed] [Google Scholar]
- 77.Targher G, Bertolini L, Padovani R, et al. Non-Alcoholic fatty liver disease is associated with carotid artery wall thickness in diet-controlled type 2 diabetic patients. J Endocrinol Invest 2006;29:55–60. 10.1007/BF03349177 [DOI] [PubMed] [Google Scholar]
- 78.Targher G, Bertolini L, Padovani R, et al. Associations between liver histology and carotid intima-media thickness in patients with nonalcoholic fatty liver disease. Arterioscler Thromb Vasc Biol 2005;25:2687–8. 10.1161/01.ATV.0000189299.61568.79 [DOI] [PubMed] [Google Scholar]
- 79.Brea A, Mosquera D, Martín E, et al. Nonalcoholic fatty liver disease is associated with carotid atherosclerosis: a case-control study. Arterioscler Thromb Vasc Biol 2005;25:1045–50. 10.1161/01.ATV.0000160613.57985.18 [DOI] [PubMed] [Google Scholar]
- 80.Tan EC-L, Tai M-LS, Chan W-K, et al. Association between non-alcoholic fatty liver disease evaluated by transient elastography with extracranial carotid atherosclerosis in a multiethnic Asian community. JGH Open 2019;3:117–25. 10.1002/jgh3.12114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Martínez-Alvarado MDR, Juárez-Rojas JG, Medina-Urrutia AX, et al. Association of fatty liver with cardiovascular risk factors and subclinical atherosclerosis in a Mexican population. Rev Invest Clin 2014;66:407–14. [PubMed] [Google Scholar]
- 82.Lankarani KB, Mahmoodi M, Lotfi M, et al. Common carotid intima-media thickness in patients with non-alcoholic fatty liver disease: a population-based case-control study. Korean J Gastroenterol 2013;62:344–51. 10.4166/kjg.2013.62.6.344 [DOI] [PubMed] [Google Scholar]
- 83.Kim HC, Kim DJ, Huh KB. Association between nonalcoholic fatty liver disease and carotid intima-media thickness according to the presence of metabolic syndrome. Atherosclerosis 2009;204:521–5. 10.1016/j.atherosclerosis.2008.09.012 [DOI] [PubMed] [Google Scholar]
- 84.Kim BJ, Cheong ES, Kang JG, et al. Relationship of epicardial fat thickness and nonalcoholic fatty liver disease to coronary artery calcification: from the Caesar study. J Clin Lipidol 2016;10:619–26. 10.1016/j.jacl.2016.01.008 [DOI] [PubMed] [Google Scholar]
- 85.Jacobs K, Brouha S, Bettencourt R, et al. Association of nonalcoholic fatty liver disease with visceral adiposity but not coronary artery calcification in the elderly. Clinical Gastroenterology and Hepatology 2016;14:1337–44. 10.1016/j.cgh.2016.01.010 [DOI] [PubMed] [Google Scholar]
- 86.Mellinger JL, Pencina KM, Massaro JM, et al. Hepatic steatosis and cardiovascular disease outcomes: an analysis of the Framingham heart study. J Hepatol 2015;63:470–6. 10.1016/j.jhep.2015.02.045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Kim MK, Ahn CW, Nam JS, et al. Association between nonalcoholic fatty liver disease and coronary artery calcification in postmenopausal women. Menopause 2015;22:1323–7. 10.1097/GME.0000000000000503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kang MK, Kang BH, Kim JH. Nonalcoholic fatty liver disease is associated with the presence and morphology of subclinical coronary atherosclerosis. Yonsei Med J 2015;56:1288–95. 10.3349/ymj.2015.56.5.1288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.VanWagner LB, Ning H, Lewis CE, et al. Associations between nonalcoholic fatty liver disease and subclinical atherosclerosis in middle-aged adults: the coronary artery risk development in young adults study. Atherosclerosis 2014;235:599–605. 10.1016/j.atherosclerosis.2014.05.962 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Sung K-C, Wild SH, Kwag HJ, et al. Fatty liver, insulin resistance, and features of metabolic syndrome: relationships with coronary artery calcium in 10,153 people. Diabetes Care 2012;35:2359–64. 10.2337/dc12-0515 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Santos RD, Nasir K, Conceição RD, et al. Hepatic steatosis is associated with a greater prevalence of coronary artery calcification in asymptomatic men. Atherosclerosis 2007;194:517–9. 10.1016/j.atherosclerosis.2007.01.026 [DOI] [PubMed] [Google Scholar]
- 92.Chhabra R, O'Keefe JH, Patil H, et al. Association of coronary artery calcification with hepatic steatosis in asymptomatic individuals. Mayo Clin Proc 2013;88:1259–65. 10.1016/j.mayocp.2013.06.025 [DOI] [PubMed] [Google Scholar]
- 93.Jung D-H, Lee Y-J, Ahn H-Y, et al. Relationship of hepatic steatosis and alanine aminotransferase with coronary calcification. Clin Chem Lab Med 2010;48:1829–34. 10.1515/CCLM.2010.349 [DOI] [PubMed] [Google Scholar]
- 94.Chen C-H, Nien C-K, Yang C-C, et al. Association between nonalcoholic fatty liver disease and coronary artery calcification. Dig Dis Sci 2010;55:1752–60. 10.1007/s10620-009-0935-9 [DOI] [PubMed] [Google Scholar]
- 95.Kim J, Lee DY, Park SE, et al. Increased risk for development of coronary artery calcification in subjects with non-alcoholic fatty liver disease and systemic inflammation. PLoS One 2017;12:e0180118. 10.1371/journal.pone.0180118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Kang YM, Jung CH, Cho YK, et al. Fatty liver disease determines the progression of coronary artery calcification in a metabolically healthy obese population. PLoS One 2017;12:e0175762. 10.1371/journal.pone.0175762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Ampuero J, Gallego-Durán R, Romero-Gómez M. Association of NAFLD with subclinical atherosclerosis and coronary-artery disease: meta-analysis. Rev Esp Enferm Dig 2015;107:10–16. [PubMed] [Google Scholar]
- 98.Bonci E, Chiesa C, Versacci P, et al. Association of nonalcoholic fatty liver disease with subclinical cardiovascular changes: a systematic review and meta-analysis. Biomed Res Int 2015;2015:1–11. 10.1155/2015/213737 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Musso G, Gambino R, Cassader M, et al. Meta-Analysis: natural history of non-alcoholic fatty liver disease (NAFLD) and diagnostic accuracy of non-invasive tests for liver disease severity. Ann Med 2011;43:617–49. 10.3109/07853890.2010.518623 [DOI] [PubMed] [Google Scholar]
- 100.Targher G, Byrne CD, Lonardo A, et al. Non-Alcoholic fatty liver disease and risk of incident cardiovascular disease: a meta-analysis. J Hepatol 2016;65:589–600. 10.1016/j.jhep.2016.05.013 [DOI] [PubMed] [Google Scholar]
- 101.Szanto KB, Li J, Cordero P, et al. Ethnic differences and heterogeneity in genetic and metabolic makeup contributing to nonalcoholic fatty liver disease. Diabetes Metab Syndr Obes 2019;12:357–67. 10.2147/DMSO.S182331 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.McKimmie RL, Daniel KR, Carr JJ, et al. Hepatic steatosis and subclinical cardiovascular disease in a cohort enriched for type 2 diabetes: the diabetes heart study. Am J Gastroenterol 2008;103:3029–35. 10.1111/j.1572-0241.2008.02188.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Francque SM, van der Graaff D, Kwanten WJ. Non-alcoholic fatty liver disease and cardiovascular risk: pathophysiological mechanisms and implications. J Hepatol 2016;65:425–43. 10.1016/j.jhep.2016.04.005 [DOI] [PubMed] [Google Scholar]
- 104.Gaggini M, Morelli M, Buzzigoli E, et al. Non-alcoholic fatty liver disease (NAFLD) and its connection with insulin resistance, dyslipidemia, atherosclerosis and coronary heart disease. Nutrients 2013;5:1544–60. 10.3390/nu5051544 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Hernaez R, Lazo M, Bonekamp S, et al. Diagnostic accuracy and reliability of ultrasonography for the detection of fatty liver: a meta-analysis. Hepatology 2011;54:1082–90. 10.1002/hep.24452 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Lee SS, Park SH. Radiologic evaluation of nonalcoholic fatty liver disease. WJG 2014;20:7392–402. 10.3748/wjg.v20.i23.7392 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
openhrt-2021-001850supp001.pdf (3.3MB, pdf)
Data Availability Statement
Data are available upon reasonable request.