Abstract
Nanotechnology involves the discovery and fabrication of nanoscale materials possessing unique physicochemical properties that are being employed in industry and medicine. Infectious Diseases clinicians and public health scientists utilize nanotechnology applications to diagnose, treat, and prevent infectious diseases. However, fundamental principles of nanotechnology are often presented in technical formats that presuppose an advanced knowledge of chemistry, physics, and engineering, thereby limiting the clinician’s grasp of the underlying science. While nanoscience is technically complex, it need not be out of reach of the clinical practitioner. The aim of this review is to introduce fundamental principles of nanotechnology in an accessible format, describe examples of current clinical infectious diseases and public health applications, and provide a foundation that will aid understanding of and appreciation for this burgeoning and important field of science.
Keywords: food and water safety, lateral flow chromatography, nanodiagnostics, nanomedicine, nanotechnology, nuclear magnetic resonance, surface plasmon resonance, vaccines
Nanotechnology has aptly been defined as “the design, characterization, production and application of structures and systems by controlled manipulation of size and shape at the nanometer scale (atomic, molecular and macromolecular scale) that produces structures, devices and systems with at least one novel/superior characteristic or property” [1]. Nanomedicine is the application of nanotechnology to the diagnosis, treatment, and prevention of disease [2]. Prevailing wisdom suggests that nanotechnology represents a very recent area of scientific endeavor and that its application to the field of clinical infectious diseases is a modern-day phenomenon. While this statement is correct in strictly scientific terms, it is interesting to note that the earliest application of nanomaterials to human health, dating back many centuries, involved the use of silver and gold colloids (ie, compounds composed of silver nanoparticles [AgNPs] and gold nanoparticles [AuNPs]) to keep food and water from spoiling and to prevent wounds from becoming infected [3, 4]. Aluminum salt nanoparticles (NPs) have been used as adjuvants in vaccines for nearly a century [5]. One of the first engineered NPs approved for clinical use in the treatment of human diseases was liposomal amphotericin B (LAmB) in the 1990s [6]. The red lines that develop on the lateral flow chromatography (LFC) assays, long used to detect the presence of pathogens, are functionalized NPs [7].
Today, clinical infectious diseases practitioners utilize a growing array of tools that harness the unique properties associated with nanotechnology [8]. These tools include rapid and point-of-care diagnostic tests (POCTs), antibiotics and antibiotic delivery vehicles, vaccines, and materials to purify food and water. The convergence of nanotechnology with clinical infectious diseases is robust and rapidly expanding. This review will present fundamental concepts of nanotechnology and will illustrate how this growing field of science is being applied to clinical infectious diseases.
Fundamental Nanotechnology Principles
Nanoscience is an amalgam of solid-state physics, materials science, surface chemistry, and quantum mechanics. Although the underlying science is complex and most often presented in highly technical formats, the fundamentals can be distilled into a few straightforward concepts that provide a strong working knowledge of nanoscience and an appreciation for its potential as a valuable clinical tool (Table 1). Nanotechnology involves the discovery, synthesis, and engineering of organic and inorganic materials having at least 1 dimension in the size range of approximately 1 to 1000 nanometers (nm); 1nm equals 1×10–9 meter. All materials within this size range possess unique physical properties relative to their bulk (macroscopic) counterparts. In addition, a variety of nanomaterials inherently display distinctive optical, electric, magnetic, and thermal behaviors that can be harnessed for medical purposes.
Table 1.
Fundamental Principles of Nanotechnology Relevant to Nanomedicine
| Nanoparticle sizes place them at a unique transition state between bulk material and individual atoms. |
| • They are comprised of several hundred to a few thousand atoms confined within the nanoparticle’s boundary |
| • They are typically smaller than the wavelengths of visible light |
| Nanoparticle properties are governed by both classical and quantum physics. |
| • Depending on the elemental constituents (eg, metals, silica, carbon), quantum physics can play a significant role in unique nanoparticle behavior |
| • The addition or subtraction of single atoms can profoundly affect nanoparticle size, shape, and energy levels |
| Nanoparticles have high surface area-to-volume ratios. |
| • High surface area imparts unique catalytic, adsorptive, and surface charge properties |
| Nanoparticle surface atoms tend to be reactive. |
| • Reactive surface atoms are amenable to surface coatings and ligand functionalization |
| Nanoparticle size is at the subcellular and biomolecule scale. |
| • The interactions at this scale underlie the nanotechnology–infectious diseases interface |
Nanomaterials represent a transition state between bulk solids and individual atoms resulting in unique behaviors governed by both classical and quantum physics [9]. Whereas bulk materials are composed of billions of atoms, NPs, by contrast, are composed of several hundred to a few thousand atoms. NP physical and chemical properties are a function of their constituent elements confined within small dimensional boundaries, and this differs significantly from properties at the macroscale. Depending on the materials involved, (eg, metals, semi-conductors, carbon, graphene), the addition or subtraction of only a few atoms can change the size and shape of the nanostructure, affect their discrete (quantized) electron energy levels, and profoundly alter NP properties. While the underlying nature of these quantum confinement effects is fundamentally complex and best left to quantum physicists to contemplate, the applications to clinical infectious diseases can be readily understood. As will be illustrated herein, these properties can be harnessed and tuned when engineered nanomaterials interact with biomolecules [8–10].
Nanomaterials have very large surface area-to-volume ratios. To illustrate this key concept, consider the following example: A cube having a 1-centimeter (cm) edge will have a volume of 1cm3 and a surface area of 6cm2. Were the same cube to be divided into 1-nm cubes, the volume remains the same, but the resulting total surface area now equals 60×106 cm2 [2]. This fundamental characteristic lends itself to biologically relevant catalytic and adsorptive behaviors [9, 11]. Moreover, this ratio dictates that as the size of a material decreases, the proportion of its surface atoms increases. Thus, at the nanoscale, surface atoms comprise a significant proportion of the NP structure. NP surface atoms, having fewer neighbors with whom to form chemical bonds, tend to be reactive. As a consequence of this reactivity, it is relatively easy to attach a variety of coatings and ligands to them. Surface coatings can modify NP solubility, biocompatibility, and pharmacology. A variety of biologically active ligands capable of being attached to NP surfaces include immunoglobulins, oligonucleotides, proteins, aptamers, enzymes, avidin, biotin, sugars, phospholipids, polymers, and drugs [12]. These NP bioconjugates are described as being functionalized; they have been engineered to perform a specific biological function.
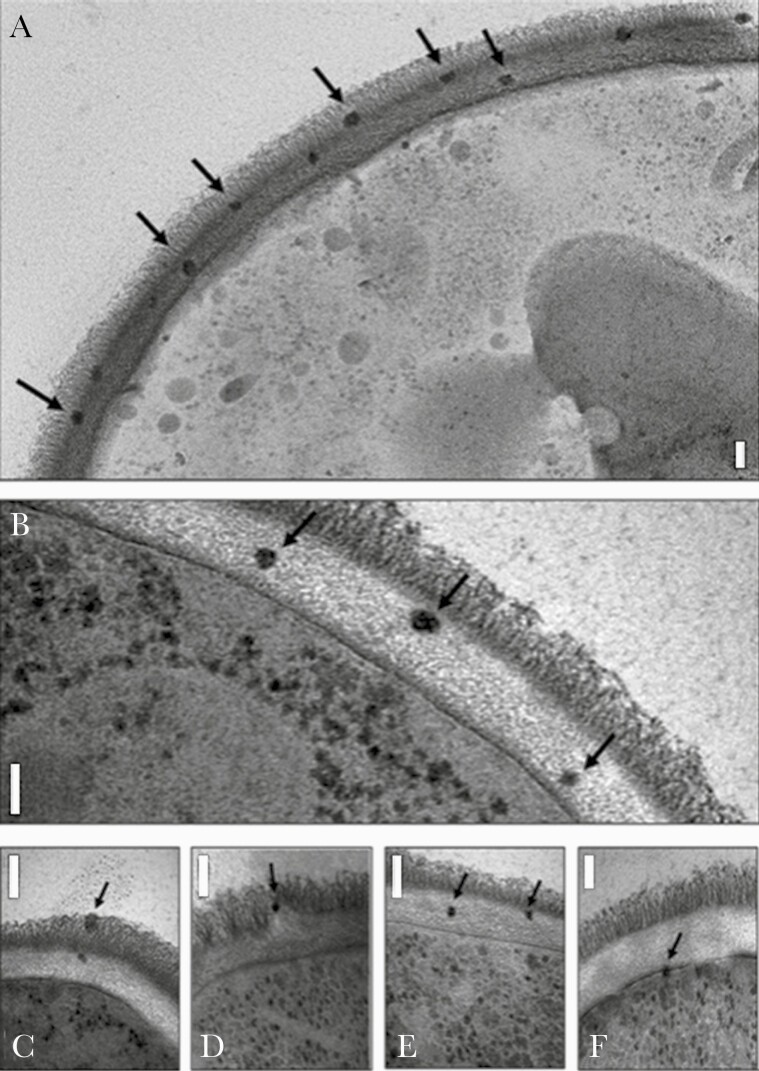
As in the case of LAmB, NPs may possess surface and core structures that are amenable to carrying biologically active payloads and in so doing alter pharmacokinetic, pharmacodynamic, and toxicological properties [13]. Similarly, a variety of NP-based vaccines incorporating antigens and adjuvants on their surface or within their matrix structures are capable of eliciting strong immunogenicity [14, 15]. Furthermore, reactivity of certain nanomaterials can be exploited to preserve food, decontaminate water, and disinfect wounds [16–18]. NPs are comparable in size to subcellular structures and biomolecules; interactions at this scale represent the key feature of the nanotechnology–clinical infectious diseases interface. This is beautifully illustrated in the photomicrograph of LAmB localized to the cell wall and cell membrane of Candida albicans (Figure 1) [19].
Figure 1.
Electron micrograph images of Candida albicans incubated with AmBisome, showing intact liposomes in the outer (A, C, and D) and inner (A, B, C, and E) cell wall and at the cell membrane (F), indicated by arrows. The granular particles in the cytoplasm are ribosomes, not liposomes. Bars represent 100nm. The photomicrograph originally appeared in Walker et al [19] (open access journal published by the American Society of Microbiology).
Applications to Clinical Infectious Diseases
There are currently numerous approved applications of nanotechnology that aid in the diagnosis, treatment, and prevention of infectious diseases. Many more nanotechnology applications are in various stages of development [8]. While a detailed discussion of each of these applications is beyond the scope of this review, a general description of their underlying nanotechnology features will provide a foundation upon which their clinical potential can be understood. Several examples of nanotechnology applications in current use and those in late stages of development as of this writing will be presented.
Nanotechnology and Infectious Diseases Diagnosis
The large surface area-to-volume ratio and reactive nature of their surface atoms create the opportunity to functionalize nanostructures with affinity ligands capable of binding to pathogen-associated analyte targets. These functionalized nanostructures transduce the binding events into optical, electric, mechanical, or magnetic output signals [8]. Rapid analysis, multiplexing capacity, ability to identify resistance determinants, and POCT capabilities are valuable features of the technology [20–22]. Currently, there are hundreds of nanobiosensing assays in various stages of development for which performance characteristics and economic impact remain to be defined [23, 24].
Two nanobiosensing platforms in current clinical use utilize optical and nuclear magnetic resonance (NMR) signals to detect a variety of infectious diseases agents [7, 25]. Moreover, they are rapidly expanding their capabilities by adding pathogen targets and antimicrobial resistance determinants to their existing diagnostic panels. When applied to the detection of fungal and bacterial bloodstream infections, these nanobiosensing modalities can shorten the time to initiate optimal antibiotics [26–28].
Optical Nanobiosensors
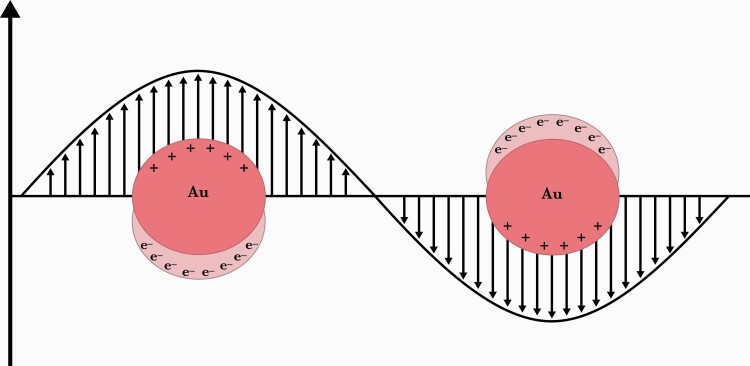
Assays based on optical nanobiosensors can harness the unique interaction of AuNPs and AgNPs with the electromagnetic photon energy of light [29–31]. These noble metal NPs, comprised of several hundred to a few thousand atoms, have dimensions much smaller than the wavelengths encompassed by the visible light spectrum (Figure 2). The incident light wave causes the ensemble of the metal’s free surface electrons to collectively oscillate in resonance with the light’s discreet (quantized) wavelength frequency. The collective, coherent, oscillating NP surface electrons are called plasmons, and their surface plasmon resonance (SPR) produces strong light absorption and emission phenomena [7, 11, 31].
Figure 2.
Schematic representation of metal nanoparticles in localized surface plasmon resonance. Interaction of the electromagnetic waves with the gold nanoparticle (Au) surface electrons (e–) induces a surface plasmon resonance that produces strong light emission. The figure originally appeared in Cordeiro et al [30] (open access journal published by MDPI).
SPR can be tuned by modifying NP composition, size, shape, proximity to other NPs, and the surrounding polarizable environment (dielectric medium) [32]. Because of their relative ease of fabrication, functionalization, stability, and unique SPR properties, AuNPs and AgNPs are widely employed as plasmonic nanobiosensors. SPR light emission by these NPs is several orders of magnitude greater than that of organic fluorophores. Moreover, unlike the rapidly fading fluorescence of fluorophore dyes, the light emission induced by SPR in metallic NPs does not fade. Interestingly, these unique SPR properties were harnessed centuries ago by artisans who incorporated colloids of gold and silver NPs into stained glass [31].
The application of SPR to infectious diseases diagnostics is best illustrated by the commonly used LFC platform, a familiar assay technique exemplified by the home pregnancy test. When functionalized AuNPs bind to their target analytes and aggregate on the LFC test strip, their optical signals appear as easily visible red lines; in this case the AuNPs absorb light in the green region and emit light in the red region of the visible light spectrum [23, 33]. In addition to the most commonly employed LFC formats that rely on the SPR properties of AuNPs, there are other less often used optical LFC methods that utilize functionalized NPs such as colored latex nanobeads, fluorescent dye containing liposomes, carbon, and selenium [23, 34, 35].
In general, LFC assays are qualitative, or semi-quantitative, and can be less sensitive compared to nucleic acid amplification tests. Given their relative low cost, ease of use, and rapid detection capability, LFC assays are often used as screening tests and are commonly employed in clinical laboratory, POCT, and at-home settings. In addition, LFC assays are frequently employed to detect contaminated food and water sources [33]. There are many variables including, but not limited to, pretest probability, presence or absence of symptoms, pathogen concentration and viability at the time of testing, proper sample collection, sensitivity, and specificity that require careful consideration when interpreting LFC test results (and, for that matter, all diagnostic test results) and influencing medical decision making. Discussion of these very important and complex considerations with respect to LFC assays are beyond the scope of this review, and the reader is directed to excellent references on the topic [23, 33, 35–42].
The World Health Organization (WHO), the US Food and Drug Administration (FDA), and other regulatory agencies have authorized and/or approved hundreds of LFC assays for detection of a variety of viruses, bacteria, fungi, and parasites [23, 33, 37, 43–47]. We provide a representative sample of commonly used diagnostic LFC tests (Table 2).
Table 2.
| Pathogen | Test Name | Specimen Type | Optical Signal | Test Performance | Reference |
|---|---|---|---|---|---|
| HIV | Oraquick Advance Rapid HIV-1/2 Antibody Test | Oral fluid, blood | AuNP | >99% agreement with confirmatory tests | Package insert |
| Influenza virus | Quidel Sofia Influenza A+B FIAc | Nasopharyngeal fluid | Fluorescent NP | A=97% sensitivity
B=90% sensitivity |
Package insert |
| HCV | Oraquick HCV Rapid Antibody Test | Oral fluid, serum | AuNP | Oral=98% sensitivity
Serum=100% sensitivity |
[44] |
| Ebola virus | CorGENIX ReEBOV Antigen Rapid Test | Blood | AuNP | 100% sensitivity | Package insert |
| SARS-CoV-2d | Celltrion DiaTrust COVID-19 Ag Rapid Test | NPS | AuNP | 93% sensitivitye | Package insert |
| Ellume COVID-19 Home Test | NPS | Fluorescent NP | 95% sensitivity | Package insert | |
| Cellex qSARS-CoV-2 IgG/IgM Rapid Test | Blood | AuNP | 95% sensitivity | Package insert | |
| Tuberculosis | Alere Determine TB-LAM Ag Test | Urine | AuNP | 54% sensitivityf | [45] |
| Group A Streptococcus | Wondfro StrepA Rapid Test | Throat swab | AuNP | 95% sensitivity
LOD 1.5×105 organisms/mL |
Package insert |
| Cryptococcus | Immy CrAg Cryptococcal IFA | Blood, CSF | AuNP | LOD 1ng/mL | Package insert |
| Plasmodium falciparum | Becton Dickinson ParaSight – F Test | Blood | Dye-loaded liposomes | <500 parasitemia/µL blood, 83% sensitivity
>500 parasitemia/µL blood, 98% sensitivity |
[46] |
| Plasmodium species | OptiMAL-IT | Blood | AuNP | <500 parasitemia/µL blood, 60% sensitivity for Pf 50% sensitivity for non-Pf >500 parasitemia/µL blood 100% sensitivity for Pf 89% sensitivity for non-Pf |
[47] |
Abbreviations: AuNP, gold nanoparticle; CSF, cerebrospinal fluid; HCV, hepatitis C virus; HIV, human immunodeficiency virus; LOD, limit of detection; NP, nanoparticle; NPS, nasopharyngeal swab; Pf, Plasmodium falciparum; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
This list represents a very small sample of authorized or approved lateral flow chromatography assays in common use. They were selected to illustrate a variety of pathogens and optical signals. Many other examples use NPs that the manufacturer deems proprietary information.
The mention of specific products is intended to serve as examples of nanotechnology applications in common clinical use and should not be considered an endorsement.
Requires use of an integrated instrument for analysis.
Test performance in real-world conditions and in different populations varies depending on a number of factors, including when in the course of the infection the test is performed.
Positive percentage agreement compared to reverse-transcription polymerase chain reaction.
Pooled sensitivity across 6 studies ranged from 23% to 84% in HIV-infected patients.
It is particularly noteworthy that, at this moment in time, there are >100 nanobiosensor-based LFC tests that have recently received WHO and FDA emergency use authorization (EUA) for detection of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [36, 38, 39, 48]. Of these, several have received EUA for at-home settings [36]. This highlights the critical public health importance of rapid, simple-to-perform, and available POCTs during outbreaks (eg, SARS-CoV-2, influenza, Ebola). Consequently, there is enormous interest in the role that nanobiosensor-based strategies play in this context.
Automated SPR-based nanobiosensing assays are capable of rapid, multiplexed detection of a variety of pathogens (Luminex VERIGENE System) [49, 50]. In this example, the presence of target oligonucleotides bound to functionalized AuNPs are optically identified at specific locations on a chip microarray [49, 51]. Sensitivity is enhanced by the strong SPR emission of AgNPs that are subsequently added to the assay sample where they chemically combine with the microarray’s bound AuNPs. The assay requires use of an integrated instrument that performs the test, reads, and records the microarray optical signals. Bacterial panels are culture based and performed on oligonucleotide targets extracted from the sample; where applicable, the target analytes can include specific resistance genes. Viral panels incorporate nucleic acid amplification steps in the automated procedure. In general, performance characteristics including sensitivity, specificity, and limit of detection (LOD) are excellent; their details can be obtained in the respective technical support package inserts (Luminexcorp.com).
The potential to integrate optical nanobiosensors with miniaturized, self-contained, and low-cost digital health tools and mobile communication technologies (eg, mobile phone applications, or “apps”) represents an important area of applied nanobioscience [52, 53]. In their proof-of-concept study, Chin and colleagues [52] developed a hand-held, automated human immunodeficiency virus (HIV) enzyme-linked immunosorbent assay that uses antibody functionalized AuNPs for ligand-receptor binding and signal transduction. Their system integrates assay performance with Web-based readout and reporting of results. Its low cost, ease of use, and self-contained capability have important implications for POCTs in both resource-poor and resource-rich regions of the world [53]. Other important benefits of digitally integrated diagnostic test workflows include (1) the ability for standardized data sets (including harmonized coding specifications) to be captured and transmitted directly from the testing device to clinicians and/or public health authorities in real time; (2) improvements in diagnostic data quality, completeness, interoperability, and utility; (3) data prepopulation in software or digital health tools that are used with tests, which dramatically reduces user burden associated with data capture and reporting; and (4) high quality real-world data captured, aggregated, and analyzed at scale, which supports a robust evaluation of postmarket device performance. This type of digital diagnostic test technology has recently been commercialized and applied to smartphone apps that accompany point-of-care and at-home SARS-CoV-2 LFC tests (eg, BD Veritor At-Home COVID-19 Test, Abbott BinaxNOW COVID-19 Antigen Self-Test, Ellume COVID-19 Home Test) and an at-home SARS-CoV-2 molecular test (eg, Cue COVID-19 Test Kit) capable of capturing standardized data sets and reporting results to public health authorities.
NMR Nanobiosensors
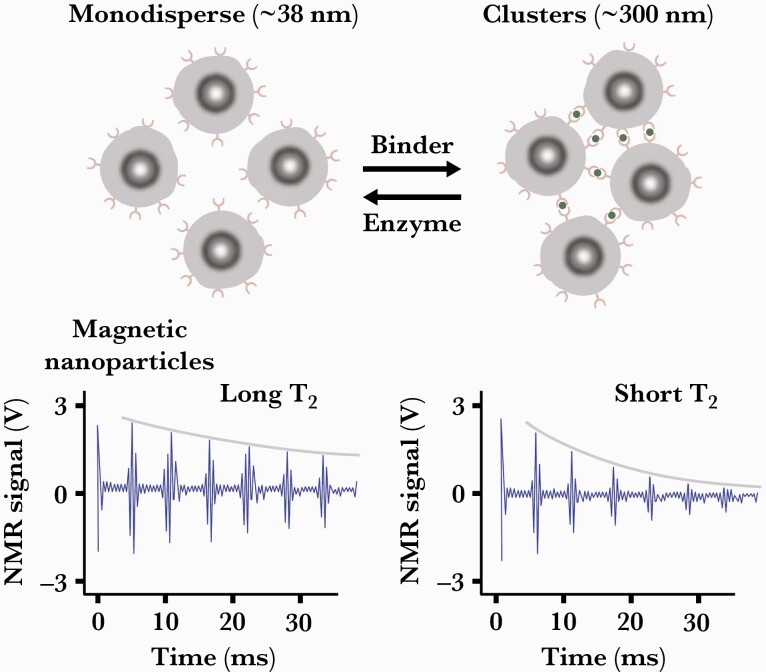
NMR nanobiosensing assays harness the signals produced by the nuclear spins of the billions of water molecules present in the assay sample. The technology (T2 Biosystems) is fundamentally similar to that used in magnetic resonance imaging, albeit on an in vitro and much smaller scale. When the sample being assayed is subjected to a strong external magnetic field, the magnetic moments of the water protons uniformly align parallel to the external magnetic field vector. A radiofrequency pulse aligned perpendicular to the external magnetic field induces the magnetic moments of the water protons to tip 90° in the transverse plane. The now-energized water protons release their energy over time as they return to their more thermodynamically relaxed state. This transverse relaxation is designated T2, and its time sequence is detected in the form of an NMR signal [10, 25]. When ligand functionalized magnetic nanoparticles (MNPs) are added to the assay sample they bind to and aggregate around their analyte targets. The aggregated MNPs create localized magnetic field nonuniformities that alter the T2 relaxation rate of the nearby water protons and hence the NMR signals produced (Figure 3).
Figure 3.
Magnetic relaxation switching assays involve assembly of magnetic nanoparticle.
clusters using target biomarker as cross-linking bridge, or disassembly of preformed clusters using an enzyme or competitive binding. Clustering magnetic nanoparticles causes them to more efficiently dephase the nuclear spins of neighboring water molecules, shortening the transverse relaxation time (T2). Likewise, disassembly of clusters increases T2 relaxation time. Reproduced with permission from Haun et al [25]. Abbreviation: NMR, nuclear magnetic resonance.
Since it relies on NMR as the output signal, this nanobiosensing platform can be performed on turbid samples such as whole blood. Moreover, the automated format incorporates nucleic acid amplification of the specific target analyte. The method is rapid and non–culture based. It is capable of detecting bloodstream infections due to a variety of commonly occurring Candida and bacterial species with LOD as low as 1 colony-forming unit/mL, sensitivity of >90%, and specificity of >98% [54–56]. The assay platform has recently received FDA EUA for detection of SARS-CoV-2 in respiratory specimens. The NMR nanobiosensing equipment is not currently portable; however, attempts at miniaturizing the components are being pursued [57].
Nanotechnology and Infectious Diseases Therapeutics
At a time when multidrug resistance is emerging at an alarming rate, the development of novel nanoantibiotics and the reformulation of existing antibiotics with nano-based drug delivery platforms represent an area of great importance. There are 2 major classes of NPs in infectious disease therapeutics. The first NP class consists of a variety of inorganic and organic nanomaterials that possess intrinsic antimicrobial activity [58–61]. The second NP class utilizes their small size, large surface, and unique surface chemistry to incorporate antimicrobials either on the surface of, or within the core of biodegradable and biocompatible NPs, thus allowing for effective drug delivery [62, 63]. Extensively studied and currently relevant examples of these nanoantimicrobials include AgNPs, liposomes, and nanosuspensions.
AgNPs
Metallic silver, silver salts, and AgNP colloids have a long history of preventing and treating infectious diseases [3, 18]. Silver-based compounds were the mainstay of antimicrobial therapy until the 1940s when they were supplanted by antibiotics. Due to the rise in antibiotic resistance, there has been a resurgence of interest in the antimicrobial activity of silver in the form of AgNPs. While their precise antimicrobial mechanisms of action have not been fully elucidated, studies suggest a direct toxic effect by the AgNPs on bacterial cell walls and their release of silver ions (Ag+) capable of disrupting the cell membrane and a variety of intracellular functions [18, 64]. These antimicrobial mechanisms confer both broad-spectrum activity and a high barrier to resistance.
AgNP antimicrobials are predominantly utilized as topical agents where they are incorporated into a variety of wound dressings, and have been found to promote wound healing [18, 65]. The small size and large surface area of the dressing’s integrated AgNPs enhance their solubility, promote wound coverage in high concentration, and release Ag+ in relatively low (nontoxic) and sustained concentrations. Attempts at incorporating AgNPs on the surface of catheters and implantable medical devices in an effort to prevent biofilm formation and infections has not yet achieved universal success, and remains an area of active investigation [66–68].
Silver possesses antimicrobial activity against viruses, bacteria, fungi, and parasites. However, several concerns exist regarding the use of silver nanoparticles. Pharmacokinetics of silver has not been standardized and Ag+ has exhibited toxicity to animal cells in vitro. In humans, gray skin discoloration (argyria) can occur from dermal deposition of silver [69]. Argyria is not associated with significant dermal toxicity, and is reversible upon cessation of the silver product. Reversible elevations of liver enzymes and serum silver levels due to absorption of AgNPs from burn wound dressings have been reported [70]. The unregulated use of AgNPs in household items, textiles, cosmetics, and paints, along with concerns for environmental contamination and potential for human toxicity, warrant careful study [69, 71].
Liposomes
Liposomes are NPs that are currently being used as vehicles for drug delivery [6, 13]. Their large surface area-to-volume ratio and unique surface structure allows engineering of pharmacologically active, biocompatible, and biodegradable nanostructures. LAmB incorporates amphotericin B (AmB) into the phospholipid and cholesterol bilayer of liposomes measuring 60–80nm in diameter [72]. When administered intravenously, the liposomes remain intact for prolonged periods within the bloodstream and produce high AmB peak and area under the concentration–time curve levels. Attenuation of renal and infusion-related toxicities is achieved through the incorporation of AmB in the lipid bilayer. The reasons for these attenuated toxicities have not been fully elucidated. Studies suggest that the encapsulation of AmB into liposomes significantly reduces proinflammatory cytokine release by mononuclear cells [73, 74]. Nephrotoxicity may also be attenuated due to liposome size that limits glomerular filtration, and the preferential deposition of LAmB into reticuloendothelial-rich organs [73–75]. Ultrastructure analysis suggests that the liposomes transit through the fungal cell wall (Figure 1) where they bind with ergosterol in the cell membrane [19]. In addition to the direct antifungal action of AmB, liposomes possess intrinsic immunomodulatory activity that can have additive antifungal effects [73]. LAmB is administered intravenously as well as via the inhalational route. In the latter case, both treatment and prophylaxis of pulmonary fungal infections are being explored. Details of LAmB pharmacology are described in excellent reviews [13, 73].
Liposomal amikacin administered by the inhalational route, has recently been FDA- approved for the treatment of refractory Mycobacterium avium complex [76]. Currently, this NP is also in late stages of clinical investigation for the treatment of Pseudomonas aeruginosa infection in cystic fibrosis patients [77]. This approach provides for a targeted, localized drug administration that reduces systemic exposure of potentially toxic antibiotics. Moreover, the liposome’s ability to penetrate biofilms and be taken up by alveolar macrophages may reflect additional pharmacologic benefits [73]. LAmB ability to penetrate biofilms is inferred on the basis of animal model, in vitro catheter, and ultrastructural studies that demonstrate its activity in Candida biofilms relative to AmB and other comparator antifungals [78–80].
Nanosuspensions
A variety of nanotechnology approaches to the treatment and prevention of HIV infection are being developed. Long-acting nanosuspensions of the antiretroviral drugs cabotegravir and rilpivirine have recently been FDA-approved [81–83]. Nanosuspension technology involves the preparation of poorly water-soluble drugs at nanoscale size where the large surface area and associated surface charge significantly enhance dissolution solubility and bioavailability [84]. The combination of small size, low aqueous solubility, long half-life, and high potency permits high drug loading in relatively small injection volumes, thus facilitating depot administration [85]. Nanosuspension technology is also being applied to a variety of antibiotic formulations that are in varying stages of development [86, 87].
Nanotechnology and Prevention of Infectious Diseases
Nanotechnology applications to clinical infectious diseases will have its greatest public health impact in the area of vaccine development, as well as food and water safety. NP size, surface characteristics, and the ability to engineer and manipulate their physicochemical properties are actively being applied to these infectious diseases prevention strategies.
Vaccines
Despite a wealth of accumulated knowledge regarding vaccine science, there remain gaps in our understanding of the detailed events that take place at the subcellular and biomolecular level. However, it is reasonable to assert that what does happen at that level occurs at the nanoscale, and its implications with respect to vaccine development cannot be overstated [5, 14, 15, 88]. To emphasize this point in the current era of the SARS-CoV-2 pandemic, it is worth considering a quote from Shin et al: “Nanoparticles and viruses operate at the same length scale—this is what makes nanotechnology approaches in vaccine development and immunoengineering so powerful” [88].
The presentation and processing of immunogens is dependent on particle size and epitope conformation. On that basis, nanotechnology applications have played a significant role in both traditional, modern, and next-generation vaccine development. For example, subunit vaccines employ a variety of nanoparticles capable of mimicking natural virus size and conformation. They often require the addition of nanoparticle adjuvants to elicit enhanced immunogenicity at targeted sites of action [5, 14, 15]. Virus-like particle (VLP) vaccines are comprised of self-assembling, nonreplicating protein and peptide subunits, and are in current use against hepatitis B and human papillomaviruses [15]. The vaccines also contain aluminum salt adjuvant nanoparticles whose large surface area adsorbs and/or clusters around the VLP antigens and initiates a localized inflammatory response.
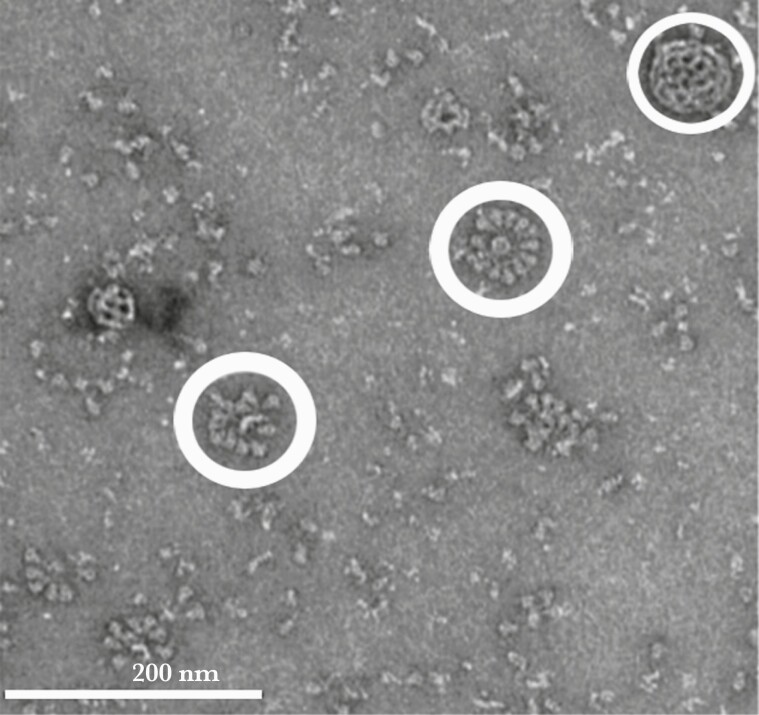
Nanotechnology is being applied to the development of next-generation vaccines, several of which are in late stages of development. Examples include vaccines against SARS-CoV-2 and respiratory syncytial virus, where protein antigen subunits combine to form rosette-like nanoparticle arrays that mimic the size and conformation of native virus (Figure 4) [89, 90]. These vaccines also require nanoparticle adjuvants for enhanced immunostimulation. Messenger RNA (mRNA) vaccines that utilize lipid nanocarriers have recently been authorized by the FDA and WHO for use against SARS-CoV-2 [91]. The lipid NPs carry the mRNAs within their core, protect them from enzyme degradation, promote cell entry, and possess adjuvant properties [92]. At the time of this writing, there are many vaccines in various stages of development to combat SARS-CoV-2 infection. They are utilizing traditional, modern, and next-generation vaccine strategies, and they are all employing nanotechnology.
Figure 4.
Electron micrograph image of severe acute respiratory syndrome coronavirus 2 nanoparticle subunit vaccine candidate demonstrating rosette conformation (thick circles) of full-length spike proteins attached to polysorbate 80 detergent. In addition, a saponin-cholesterol phospholipid adjuvant nanoparticle (Matrix-M) displays a cage-like structure (thin circle). Reproduced with permission from Bangaru et al [89].
Food and Water Safety
From a public health perspective, nanotechnology is being employed in water decontamination and disinfection, as well as in food preservation and safety [16, 17, 93–95]. Nanomaterial characteristics, including mechanical strength, large surface area, and reactive surfaces possessing catalytic, adsorptive, and antimicrobial properties, are being exploited to de-foul, disinfect, and preserve [96]. In addition, nanobiosensing strategies similar to those being used in infectious diseases diagnostic efforts are being utilized to identify contaminated water and food before they are consumed [33, 94].
Water and food safety sciences are complex. They require consideration of the delicate balance between the benefits of safety measures and the potentially harmful effects of forming toxic disinfection byproducts, often encountered with chemical disinfectants [95]. The potential for NP environmental contamination as a byproduct of food and water safety requires consideration and careful study [95]. With respect to water safety, additional considerations include the applicability and advantages of point-of-use vs centralized water treatment strategies. This is well illustrated by the development of an affordable, portable filtration device that utilizes nanocomposites to purify water and its widespread applicability to geographic regions with limited resources [17]. The ingenious filtration device utilizes green technology to produce self-assembled sheets containing chitosan fibers, aluminum hydroxide NPs, and AgNPs. The sheets remove both chemical toxins and microbial contaminants. The portable device was developed in India by Pradeep et al, and costs the equivalent of approximately US$16, thus making it widely affordable in resource-poor countries [17, 97]. Currently, scaled-up versions of the nano-based filtration device is providing centralized water purification to >10 million people in India [98].
With respect to food preservation, a variety of nanofilms, nanocoatings, and nanocomposites possessing unique gas permeability, thermal, moisture, and antimicrobial properties are being evaluated for packaging and storage [16, 94]. The availability of raw materials, ease and cost of fabrication, and environmental impact of nanostructures used for these purposes are just some of the many complex issues that impact these public health measures.
CONCLUSIONS
Nanotechnology has enhanced our ability to diagnose, treat, and prevent infectious diseases. It has significantly benefited healthcare in resource-rich as well as resource-poor parts of the world. There are concerns to be addressed and studied with respect to its limitations, the potential for nanoparticle toxicities, and their effect on the environment. Nanotechnology is based on innovative science that, for the foreseeable future, will continue to influence changes in the practice of clinical infectious diseases and public health. In their effort to understand and appreciate nanotechnology applications to medicine, clinicians and public health practitioners can benefit from resources that communicate and demystify the technical complexities of the science.
Acknowledgments
The authors thank Dr Ann Tobin, Dr Robin Hampton, and Dr Charles Hyman for helpful discussion and comments; James A. Kaplan for graphic arts; and Nicole Becher for technical assistance with preparation of tables.
Disclaimer. This publication reflects the views of the authors and should not be construed to represent FDA’s views or policies. The mention of specific products is intended to serve as examples of nanotechnology applications in common clinical use and should not be considered as an endorsement.
Potential conflicts of interest. Both authors: No reported conflicts of interest.
Both authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1. Bawa R. Patents and nanomedicine. Nanomedicine (Lond) 2007; 2:351–74. [DOI] [PubMed] [Google Scholar]
- 2. Kim BY, Rutka JT, Chan WC.. Nanomedicine. N Engl J Med 2010; 363:2434–43. [DOI] [PubMed] [Google Scholar]
- 3. Barillo DJ, Marx DE.. Silver in medicine: a brief history BC 335 to present. Burns 2014; 40(Suppl 1):S3–8. [DOI] [PubMed] [Google Scholar]
- 4. Dykman L, Khlebtsov N.. Gold nanoparticles in biomedical applications: recent advances and perspectives. Chem Soc Rev 2012; 41:2256–82. [DOI] [PubMed] [Google Scholar]
- 5. Shardlow E, Mold M, Exley C.. Unraveling the enigma: elucidating the relationship between the physicochemical properties of aluminium-based adjuvants and their immunological mechanisms of action. Allergy Asthma Clin Immunol 2018; 14:80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Lopez-Berestein G. Liposomes as carriers of antimicrobial agents. Antimicrob Agents Chemother 1987; 31:675–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Anker JN, Hall WP, Lyandres O, et al. . Biosensing with plasmonic nanosensors. Nat Mater 2008; 7:442–53. [DOI] [PubMed] [Google Scholar]
- 8. Tobin E. Nanomedicine applications for infectious diseases. In: Brenner S, ed. The Clinical Nanomedicine Handbook. Boca Raton, FL: CRC Press; 2013. [Google Scholar]
- 9. Roduner E. Size matters: why nanomaterials are different. Chem Soc Rev 2006; 35:583–92. [DOI] [PubMed] [Google Scholar]
- 10. Lowery T. Nanomaterials-Based Magnetic Relaxation Switch Biosensors. Nanotechnologies for the Life Sciences. Weinheim, Germany: Wiley-VCH Verlag GmbH & Co; 2007. [Google Scholar]
- 11. Eustis S, el-Sayed MA.. Why gold nanoparticles are more precious than pretty gold: noble metal surface plasmon resonance and its enhancement of the radiative and nonradiative properties of nanocrystals of different shapes. Chem Soc Rev 2006; 35:209–17. [DOI] [PubMed] [Google Scholar]
- 12. Sapsford KE, Algar WR, Berti L, et al. . Functionalizing nanoparticles with biological molecules: developing chemistries that facilitate nanotechnology. Chem Rev 2013; 113:1904–2074. [DOI] [PubMed] [Google Scholar]
- 13. Groll AH, Rijnders BJA, Walsh TJ, et al. . Clinical pharmacokinetics, pharmacodynamics, safety and efficacy of liposomal amphotericin B. Clin Infect Dis 2019; 68:260–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Peek LJ, Middaugh CR, Berkland C.. Nanotechnology in vaccine delivery. Adv Drug Deliv Rev 2008; 60:915–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Smith DM, Simon JK, Baker JR.. Applications of nanotechnology for immunology. Nat Rev Immunol 2013; 13:592–605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Bajpai VK, Kamle M, Shukla S, et al. . Prospects of using nanotechnology for food preservation, safety, and security. J Food Drug Anal 2018; 26:1201–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Sankar MU, Aigal S, Maliyekkal SM, et al. . Biopolymer-reinforced synthetic granular nanocomposites for affordable point-of-use water purification. Proc Natl Acad Sci U S A 2013; 110:8459–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Fong J, Wood F.. Nanocrystalline silver dressings in wound management: a review. Int J Nanomedicine 2006; 1:441–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Walker L, Sood P, Lenardon MD, et al. The viscoelastic properties of the fungal cell wall allow traffic of AmBisome as intact liposome vesicles. mBio 2018; 9:e02383-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Rosi NL, Mirkin CA.. Nanostructures in biodiagnostics. Chem Rev 2005; 105:1547–62. [DOI] [PubMed] [Google Scholar]
- 21. Kaittanis C, Santra S, Perez JM.. Emerging nanotechnology-based strategies for the identification of microbial pathogenesis. Adv Drug Deliv Rev 2010; 62:408–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Hauck TS, Giri S, Gao Y, Chan WC.. Nanotechnology diagnostics for infectious diseases prevalent in developing countries. Adv Drug Deliv Rev 2010; 62:438–48. [DOI] [PubMed] [Google Scholar]
- 23. Posthuma-Trumpie GA, Korf J, van Amerongen A.. Lateral flow (immuno)assay: its strengths, weaknesses, opportunities and threats. A literature survey. Anal Bioanal Chem 2009; 393:569–82. [DOI] [PubMed] [Google Scholar]
- 24. Caliendo AM, Gilbert DN, Ginocchio CC, et al. ; Infectious Diseases Society of America. . Better tests, better care: improved diagnostics for infectious diseases. Clin Infect Dis 2013; 57(Suppl 3):S139–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Haun JB, Yoon TJ, Lee H, Weissleder R.. Magnetic nanoparticle biosensors. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2010; 2:291–304. [DOI] [PubMed] [Google Scholar]
- 26. Bork JT, Leekha S, Heil EL, et al. . Rapid testing using the Verigene gram-negative blood culture nucleic acid test in combination with antimicrobial stewardship intervention against gram-negative bacteremia. Antimicrob Agents Chemother 2015; 59:1588–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Pogue JM, Heil EL, Lephart P, et al. An antibiotic stewardship program blueprint for optimizing Verigene BC-GN within an institution: a tale of two cities. Antimicrob Agents Chemother 2018; 62:e02538-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Clancy CJ, Nguyen MH.. T2 magnetic resonance for the diagnosis of bloodstream infections: charting a path forward. J Antimicrob Chemother 2018; 73:iv2–5. [DOI] [PubMed] [Google Scholar]
- 29. Tokel O, Inci F, Demirci U.. Advances in plasmonic technologies for point of care applications. Chem Rev 2014; 114:5728–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Cordeiro M, Ferreira Carlos F, Pedrosa P, et al. Gold nanoparticles for diagnostics: advances towards points of care. Diagnostics (Basel) 2016; 6:43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Link S, El-Sayed MA.. Optical properties and ultrafast dynamics of metallic nanocrystals. Annu Rev Phys Chem 2003; 54:331–66. [DOI] [PubMed] [Google Scholar]
- 32. Moores A, Goettmann F.. The plasmon band in noble metal nanoparticles: an introduction to theory and applications. N J Chemistry 2006; 30:1121–32. [Google Scholar]
- 33. Banerjee R, Jaiswal A.. Recent advances in nanoparticle-based lateral flow immunoassay as a point-of-care diagnostic tool for infectious agents and diseases. Analyst 2018; 143:1970–96. [DOI] [PubMed] [Google Scholar]
- 34. Gao X, Xu LP, Zhou SF, et al. Recent advances in nanoparticles-based lateral flow biosensors. Am J Biomed Sci 2014; 6:41–57. [Google Scholar]
- 35. Koczula KM, Gallotta A.. Lateral flow assays. Essays Biochem 2016; 60:111–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Rubin R. COVID-19 testing moves out of the clinic and into the home. JAMA 2021; 326:1362–4. [DOI] [PubMed] [Google Scholar]
- 37. Liu Y, Zhan L, Qin Z, et al. . Ultrasensitive and highly specific lateral flow assays for point-of-care diagnosis. ACS Nano 2021; 15:3593–611. [DOI] [PubMed] [Google Scholar]
- 38. Allan-Blitz LT, Klausner JD.. A real-world comparison of SARS-CoV-2 rapid antigen testing vs PCR testing in Florida. J Clin Microbiol 2021; 59:e0110721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Somborac Bačura A, Dorotić M, Grošić L, et al. . Current status of the lateral flow immunoassay for the detection of SARS-CoV-2 in nasopharyngeal swabs. Biochem Med (Zagreb) 2021; 31:020601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Guglielmi G. Fast coronavirus tests: what they can and can’t do. Nature 2020; 585:496–8. [DOI] [PubMed] [Google Scholar]
- 41. Coffey KC, Diekema DJ, Morgan DJ.. Interpreting SARS-CoV-2 test results. JAMA 2021; 326:1528–9. [DOI] [PubMed] [Google Scholar]
- 42. Hanson KE, Altayar O, Calliendo AM, et al. Infectious Diseases Society of America guidelines on the diagnosis of COVID-19: antigen testing. 2021. https://www.idsociety.org/practice-guideline/covid-19-guideline-antigen-testing. Accessed 18 October 2021.
- 43. Bouzid D, Zanella MC, Kerneis S, et al. . Rapid diagnostic tests for infectious diseases in the emergency department. Clin Microbiol Infect 2021; 27:182–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Cha YJ, Park Q, Kang ES, et al. . Performance evaluation of the OraQuick hepatitis C virus rapid antibody test. Ann Lab Med 2013; 33:184–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Global Laboratory Initiative. Practical implementation of lateral flow urine lipoarabinomannan assay (LF-LAM) for detection of active tuberculosis in people living with HIV. https://stoptb.org/wg/gli/assets/documents/practical-implementation-lf-lam.pdf. Accessed 20 November 2021.
- 46. Forney JR, Magill AJ, Wongsrichanalai C, et al. . Malaria rapid diagnostic devices: performance characteristics of the ParaSight F device determined in a multisite field study. J Clin Microbiol 2001; 39:2884–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Ratsimbasoa A, Randriamanantena A, Raherinjafy R, et al. . Which malaria rapid test for Madagascar? Field and laboratory evaluation of three tests and expert microscopy of samples from suspected malaria patients in Madagascar. Am J Trop Med Hyg 2007; 76:481–5. [PubMed] [Google Scholar]
- 48. Mahmoudinobar F, Britton D, Montclare JK.. Protein-based lateral flow assays for COVID-19 detection. Protein Eng Des Sel 2021; 34:1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Luminex. Verigene NanoGrid Technology. 2006. https://www.luminexcorp.com/verigene-nanogrid-technology/. Accessed 21 August 2020.
- 50. Buchan BW, Ginocchio CC, Manii R, et al. . Multiplex identification of gram-positive bacteria and resistance determinants directly from positive blood culture broths: evaluation of an automated microarray-based nucleic acid test. PLoS Med 2013; 10:e1001478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Taton TA, Mirkin CA, Letsinger RL.. Scanometric DNA array detection with nanoparticle probes. Science 2000; 289:1757–60. [DOI] [PubMed] [Google Scholar]
- 52. Chin CD, Cheung YK, Laksanasopin T, et al. . Mobile device for disease diagnosis and data tracking in resource-limited settings. Clin Chem 2013; 59:629–40. [DOI] [PubMed] [Google Scholar]
- 53. Whitesides GM. A glimpse into the future of diagnostics. Clin Chem 2013; 59:589–91. [DOI] [PubMed] [Google Scholar]
- 54. Mylonakis E, Clancy CJ, Ostrosky-Zeichner L, et al. . T2 magnetic resonance assay for the rapid diagnosis of candidemia in whole blood: a clinical trial. Clin Infect Dis 2015; 60:892–9. [DOI] [PubMed] [Google Scholar]
- 55. Clancy CJ, Pappas PG, Vazquez J, et al. . Detecting infections rapidly and easily for candidemia trial, part 2 (DIRECT2): a prospective, multicenter study of the T2Candida panel. Clin Infect Dis 2018; 66:1678–86. [DOI] [PubMed] [Google Scholar]
- 56. Nguyen MH, Clancy CJ, Pasculle AW, et al. . Performance of the T2Bacteria panel for diagnosing bloodstream infections: a diagnostic accuracy study. Ann Intern Med 2019; 170:845–52. [DOI] [PubMed] [Google Scholar]
- 57. Chun AL. Nanoparticles offer hope for TB detection. Nat Nanotechnol 2009; 4:698–9. [DOI] [PubMed] [Google Scholar]
- 58. Blecher K, Nasir A, Friedman A.. The growing role of nanotechnology in combating infectious disease. Virulence 2011; 2:395–401. [DOI] [PubMed] [Google Scholar]
- 59. Kang S, Pinault M, Pfefferle LD, Elimelech M.. Single-walled carbon nanotubes exhibit strong antimicrobial activity. Langmuir 2007; 23:8670–3. [DOI] [PubMed] [Google Scholar]
- 60. El-Sharif AA, Hussain MH.. Chitosan-EDTA new combination is a promising candidate for treatment of bacterial and fungal infections. Curr Microbiol 2011; 62:739–45. [DOI] [PubMed] [Google Scholar]
- 61. Nederberg F, Zhang Y, Tan JP, et al. . Biodegradable nanostructures with selective lysis of microbial membranes. Nat Chem 2011; 3:409–14. [DOI] [PubMed] [Google Scholar]
- 62. Zhang L, Pornpattananangku D, Hu CM, Huang CM.. Development of nanoparticles for antimicrobial drug delivery. Curr Med Chem 2010; 17:585–94. [DOI] [PubMed] [Google Scholar]
- 63. Huh AJ, Kwon YJ.. “Nanoantibiotics”: a new paradigm for treating infectious diseases using nanomaterials in the antibiotics resistant era. J Control Release 2011; 156:128–45. [DOI] [PubMed] [Google Scholar]
- 64. Durán N, Durán M, de Jesus MB, et al. . Silver nanoparticles: a new view on mechanistic aspects on antimicrobial activity. Nanomedicine 2016; 12:789–99. [DOI] [PubMed] [Google Scholar]
- 65. Paladini F, Pollini M.. Antimicrobial silver nanoparticles for wound healing application: progress and future trends. Materials (Basel) 2019; 12:2540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Roe D, Karandikar B, Bonn-Savage N, et al. . Antimicrobial surface functionalization of plastic catheters by silver nanoparticles. J Antimicrob Chemother 2008; 61:869–76. [DOI] [PubMed] [Google Scholar]
- 67. Kalfon P, de Vaumas C, Samba D, et al. . Comparison of silver-impregnated with standard multi-lumen central venous catheters in critically ill patients. Crit Care Med 2007; 35:1032–9. [DOI] [PubMed] [Google Scholar]
- 68. Seymour C. Audit of catheter-associated UTI using silver alloy-coated Foley catheters. Br J Nurs 2006; 15:598–603. [DOI] [PubMed] [Google Scholar]
- 69. Johnston HJ, Hutchison G, Christensen FM, et al. . A review of the in vivo and in vitro toxicity of silver and gold particulates: particle attributes and biological mechanisms responsible for the observed toxicity. Crit Rev Toxicol 2010; 40:328–46. [DOI] [PubMed] [Google Scholar]
- 70. Trop M, Novak M, Rodl S, et al. . Silver-coated dressing Acticoat caused raised liver enzymes and argyria-like symptoms in burn patient. J Trauma 2006; 60:648–52. [DOI] [PubMed] [Google Scholar]
- 71. Chopra I. The increasing use of silver-based products as antimicrobial agents: a useful development or a cause for concern? J Antimicrob Chemother 2007; 59:587–90. [DOI] [PubMed] [Google Scholar]
- 72. Adler-Moore J, Proffitt RT.. AmBisome: liposomal formulation, structure, mechanism of action and pre-clinical experience. J Antimicrob Chemother 2002; 49(Suppl 1):21–30. [DOI] [PubMed] [Google Scholar]
- 73. Adler-Moore J, Lewis RE, Brüggemann RJM, et al. . Preclinical safety, tolerability, pharmacokinetics, pharmacodynamics, and antifungal activity of liposomal amphotericin B. Clin Infect Dis 2019; 68:244–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Arning M, Kliche KO, Heer-Sonderhoff AH, Wehmeier A.. Infusion-related toxicity of three different amphotericin B formulations and its relation to cytokine plasma levels. Mycoses 1995; 38:459–65. [DOI] [PubMed] [Google Scholar]
- 75. Loo AS, Muhsin SA, Walsh TJ.. Toxicokinetic and mechanistic basis for the safety and tolerability of liposomal amphotericin B. Expert Opin Drug Saf 2013; 12:881–95. [DOI] [PubMed] [Google Scholar]
- 76. Shirley M. Amikacin liposome inhalation suspension: a review in Mycobacterium avium complex lung disease. Drugs 2019; 79:555–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Bilton D, Pressler T, Fajac I, et al. ; CLEAR-108 Study Group. . Amikacin liposome inhalation suspension for chronic Pseudomonas aeruginosa infection in cystic fibrosis. J Cyst Fibros 2020; 19:284–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Kuhn DM, Ghannoum MA.. Candida biofilms: antifungal resistance and emerging therapeutic options. Curr Opin Investig Drugs 2004; 5:186–97. [PubMed] [Google Scholar]
- 79. Seidler M, Salvenmoser S, Müller FM.. Liposomal amphotericin B eradicates Candida albicans biofilm in a continuous catheter flow model. FEMS Yeast Res 2010; 10:492–5. [DOI] [PubMed] [Google Scholar]
- 80. Schinabeck MK, Long LA, Hossain MA, et al. . Rabbit model of Candida albicans biofilm infection: liposomal amphotericin B antifungal lock therapy. Antimicrob Agents Chemother 2004; 48:1727–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Swindells S, Andrade-Villanueva JF, Richmond GJ, et al. . Long-acting cabotegravir and rilpivirine for maintenance of HIV-1 suppression. N Engl J Med 2020; 382:1112–23. [DOI] [PubMed] [Google Scholar]
- 82. Dizaj SM, Vazifehasl Zh, Salatin S, et al. . Nanosizing of drugs: effect on dissolution rate. Res Pharm Sci 2015; 10:95–108. [PMC free article] [PubMed] [Google Scholar]
- 83. US Food and Drug Administration. FDA Approves First Extended-Release, Injectable Drug Regimen for Adults Living with HIV. Silver Spring, MD: FDA; 2021. [Google Scholar]
- 84. Rabinow BE. Nanosuspensions in drug delivery. Nat Rev Drug Discov 2004; 3:785–96. [DOI] [PubMed] [Google Scholar]
- 85. Trezza C, Ford SL, Spreen W, et al. . Formulation and pharmacology of long-acting cabotegravir. Curr Opin HIV AIDS 2015; 10:239–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Van de Ven H, Paulussen C, Feijens PB, et al. PLGA nanoparticles and nanosuspensions with amphotericin B: potent in vitro and in vivo alternatives to Fungizone and AmBisome. J Control Release 2012; 161:795–803. [DOI] [PubMed] [Google Scholar]
- 87. Omolo CA, Kalhapure RS, Agrawal N, et al. . Formulation and molecular dynamics simulations of a fusidic acid nanosuspension for simultaneously enhancing solubility and antibacterial activity. Mol Pharm 2018; 15:3512–26. [DOI] [PubMed] [Google Scholar]
- 88. Shin MD, Shukla S, Chung YH, et al. . COVID-19 vaccine development and a potential nanomaterial path forward. Nat Nanotechnol 2020; 15:646–55. [DOI] [PubMed] [Google Scholar]
- 89. Bangaru S, Ozorowski G, Turner HL, et al. . Structural analysis of full-length SARS-CoV-2 spike protein from an advanced vaccine candidate. Science 2020; 370:1089–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Krueger S, Curtis JE, Scott DR, et al. . Structural characterization and modeling of a respiratory syncytial virus fusion glycoprotein nanoparticle vaccine in solution. Mol Pharm 2021; 18:359–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Nanomedicine and the COVID-19 vaccines. Nat Nanotech 2020; 15:963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Reichmuth AM, Oberli MA, Jaklenec A, et al. . mRNA vaccine delivery using lipid nanoparticles. Ther Deliv 2016; 7:319–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Nagar A, Pradeep T.. Clean water through nanotechnology: needs, gaps, and fulfillment. ACS Nano 2020; 14:6420–35. [DOI] [PubMed] [Google Scholar]
- 94. Duncan TV. Applications of nanotechnology in food packaging and food safety: barrier materials, antimicrobials and sensors. J Colloid Interface Sci 2011; 363:1–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Li Q, Mahendra S, Lyon DY, et al. . Antimicrobial nanomaterials for water disinfection and microbial control: potential applications and implications. Water Res 2008; 42:4591–602. [DOI] [PubMed] [Google Scholar]
- 96. Kokkinos PM, D, Venieri D.. Current trends in the application of nanomaterials for the removal of emerging micropollutants and pathogens from water. Molecules (Basel, Switzerland) 2020; 25:2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Gravotta L. Cheap nanotech filter clears hazardous microbes and chemicals from drinking water. Scientific American 2013. https://www.scientificamerican.com/article/cheap-nanotech-filter-water/. Accessed 10 November 2021. [Google Scholar]
- 98. Kumar R. From Lab to Field: IIT-M Start-up Takes Clean Water to People. New Delhi: India; 2021. https://www.indianarrative.com/india-news/from-lab-to-field-iit-m-start-up-takes-clean-water-to-people-77282.html. Accessed 10 November 2021 [Google Scholar]