Abstract
For practical use of pluripotent stem cells (PSCs) for disease modelling, drug screening, and regenerative medicine, the cell differentiation process needs to be properly refined to generate end products with consistent and high quality. To construct and optimize a robust cell-induction process, a myriad of cell culture conditions should be considered. In contrast to inefficient brute-force screening, statistical design of experiments (DOE) approaches, such as factorial design, orthogonal array design, response surface methodology (RSM), definitive screening design (DSD), and mixture design, enable efficient and strategic screening of conditions in smaller experimental runs through multifactorial screening and/or quantitative modeling. Although DOE has become routinely utilized in the bioengineering and pharmaceutical fields, the imminent need of more detailed cell-lineage specification, complex organoid construction, and a stable supply of qualified cell-derived material requires expedition of DOE utilization in stem cell bioprocessing. This review summarizes DOE-based cell culture optimizations of PSCs, mesenchymal stem cells (MSCs), hematopoietic stem cells (HSCs), and Chinese hamster ovary (CHO) cells, which guide effective research and development of PSC-derived materials for academic and industrial applications.
Keywords: design of experiments (DOE), induced pluripotent stem cell (iPSC), embryonic stem cell (ESC), mesenchymal stem cell (MSC), hematopoietic stem cell (HSC), Chinese hamster ovary (CHO) cell, cell differentiation
1. Introduction
Growing cell-based therapeutics utilizing pluripotent stem cells (PSCs), mesenchymal stem cells (MSCs), hematopoietic stem cells (HSCs), or chimeric antigen receptor T (CAR-T) cells offer novel disease treatment approaches. The use of PSCs, represented by induced pluripotent stem cells (iPSCs) and embryonic stem cells (ESCs), opens the door for more precise disease modelling, drug screening, and regenerative medicine [1]. The use of iPSCs, which overcomes the ethical issues associated with ESCs, especially broadens prospects for disease- or, even, patient-specific models and autologous cell treatments. Currently, the efficacies of PSC-derived cell therapy products are being assessed in over 30 clinical trials worldwide [2].
However, several issues surrounding the use and application of PSCs must be resolved to take advantage of their potential benefits. Among the hundreds of types of human cells [3], most have never been properly differentiated from PSCs or maintained in vitro, restricting their research and industrial use. Even if differentiation protocols are available, comparisons of PSC-derived cells and reference cells, such as primary cultures, often exhibit different cell states and functions. Although these differences are partly resolved by advanced cultivation technologies (e.g., three-dimensional (3D) culture, co-culture of different types of cells, or organoid construction), further improvements are anticipated. Moreover, for the broader use of PSC-based therapy and regenerative medicine, both stable mass production of PSC-derived materials and cost reduction should be achieved [4]. In addition, to overcome safety issues of heterogeneity, genetic instability, and tumorigenicity that sometimes hamper smooth PSC therapeutic applications [5,6,7], the whole process from PSC establishment to differentiation requires further screening and optimization.
To appropriately assess a cell-differentiation process, there are considerable requirements to evaluate in addition to the general process of cell expansion. During cell expansion, cells grow and increase through cell division; thus, it is critical to maintain cell quality and condition so as not to disturb homologous self-renewal [8]. Therefore, for the expansion of stem cells or other cell types (e.g., to produce a recombinant protein or antibody), cells are cultivated in specific optimized media and conditions. In contrast, during cell differentiation, cells often divide heterogeneously [9], exhibit altered cell characteristics, and mature in a temporal manner. Moreover, during prolonged culture processes, cells mature but no longer increase in number; instead, unnecessary cells are excluded in some cases.
Therefore, in general, stem cell differentiation processes are divided into phases based on cell maturation stages, with different media and culture conditions used for each phase. Compositions of media and extracellular matrixes (ECM) have primarily been determined through optimization based on findings from research of in vivo embryonic development [10]. Recently, cytokines, chemical compounds, small molecules, and micro-RNAs have been utilized to control cell-lineage specification [11,12]. Additionally, novel culture methods and devices have further broadened PSC applications while offering a myriad of possible conditions to optimize [13].
As the necessities for screening and optimization of PSC differentiation multiply, so do time and costs for detailed or large-scale experiments, which can hamper productivity. At present, PSC culture is relatively expensive and requires diligent medium changes. In addition, recent advancements in culture technologies can prolong the cultivation period to months, this inevitably makes it difficult to confirm experimental reproducibility.
To circumvent such difficulties in cell culture optimizations, a number of design of experiments (DOE) approaches have been implemented for cell expansion and differentiation processes—including PSC technology [14,15]. DOE enables effective condition screening and optimization while reducing experimental runs by virtue of statistics. Since the first application of analysis of variance (ANOVA) toward improved crop yield by Fisher in the 1920s, various approaches have supported academic and industry researchers. Recently, the perceived utility and spread of user-friendly statistical software have supported DOE applications in biological experiments [16,17]. Among such studies, numerous reports from the field of fermentation that achieved efficient optimizations of bacterial growth and fungi growth, and improve final product yield, suggest great applicability of DOE for mammalian cell culture optimization [18,19,20,21].
The use of DOE is not only recommended for experimental efficiency, but also for the reliability of cell-derived material production processes required by governments and regulatory authorities. DOE methodologies are now standardized by the International Organization for Standardization (ISO), while authorities including the United States Food and Drug Administration (FDA) and European Medicines Agency (EMA) recommend using DOE for pharmaceutical product development [14,22]. Notably, the success of Quality by Design (QbD) concepts in medical fields has urged DOE-based process evaluations in cell production facilities [23,24,25].
In this review, we collect and summarize DOE investigations of mammalian cell cultivation processes to support readers incorporating DOE approaches for their own experiments. First, we briefly introduce major experimental designs and their characteristics. Then, we discuss previous articles assessing stem cells, Chinese hamster ovary (CHO) cells, other cell types, and miscellaneous cell culture-associated processes. Finally, we describe the limitations and issues to be resolved for more effective PSC expansion and differentiation.
2. DOE Approaches
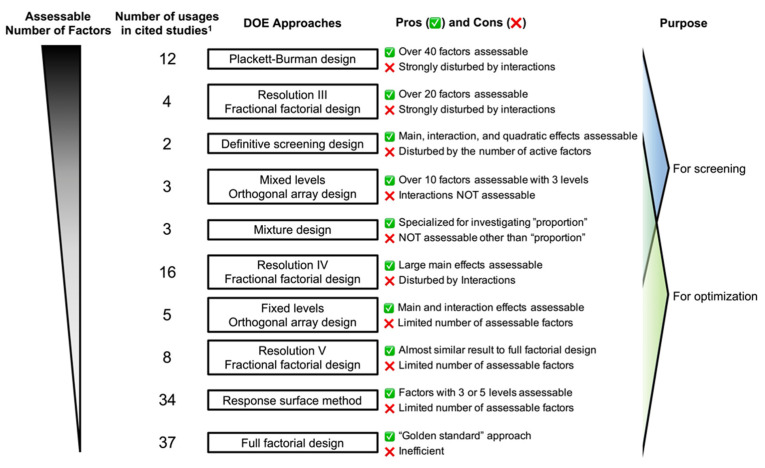
In DOE applications, a trade-off between model accuracy and experimental efficiency is inevitable when estimating “main effects”, “interaction effects”, and “quadratic effects” of investigated factors for “response variables” (Figure 1). Response variables are the indicators measured and improved, e.g., marker gene/protein expression, cell yield, or cell purity. Main effects reflect the contribution of the investigated factor itself; interaction effects reflect synergistic or counteracting effects elicited by a combination of two or more investigated factors; quadratic effects reflect the curvature contributed by each investigated factor. Generally, specifying significant main effects should be prioritized over evaluating higher-order effects because main effects tend to be more robust and reproducible. In addition, three or more higher-order interactions are often neglected in DOE approaches. Compared with other scientific or industrial experiments, chemical or biological interactions largely hinder effective investigations because they are often unforeseeable and uncontrollable.
Figure 1.
Pros and cons of each DOE approach. DOE, design of experiments. 1 Number of usages in cited studies were determined based on, in total, 124 DOE-based experiments.
2.1. One Factor at a Time Approach
The most classical and simple experimental method is the “one factor at a time” (OFAT) approach. OFAT clarifies the main effect of a single factor at multiple levels using a moderate experimental scale. However, the accuracy and reproducibility of results are often inferior to those of other DOE approaches, and screenings easily end up with inefficient quasi-optimizations [26].
2.2. Full Factorial Design
Compared with OFAT, DOE enhances screening efficiency and result reproducibility by selecting optimal conditions through investigations of multiple factors and levels. Full factorial designs provide a clear estimation of the investigated response variables in an experimental space [27]. However, its practical use is limited to small experimental scales because investigation of more factors and levels requires a tremendous number of experimental runs.
2.3. Fractional Factorial Design
Fractional factorial designs are used to estimate main effects and interactions with a reduced number of experimental runs. By compromising the resolution of an assay [28], its experimental scale can be reduced to 1/2, 1/4, 1/8, 1/16, and so on of the original full factorial design. All main effects and two-order interactions remain estimable by “resolution V or more” designs to almost the same extent as full factorial designs, but the risk of missing optimal conditions increases as resolution decreases to “IV” or “III”. The Plackett–Burman design, another fractional factorial design, has been widely used for screening of media compositions owing to its relatively higher screening efficiency focusing on only large main effects. Because fractional factorial designs generally handle two-level factors, usages are limited to screening and further optimizations would be recommended for numerical factors possessing significant contributions.
2.4. Orthogonal Array Designs
The use of orthogonal array designs enables screening of multiple factors with multiple levels [29]. Fixed two-level (e.g., L4, L8, L16, and so on) or three-level (e.g., L9, L27, and so on) arrays are used to estimate all main effects and interactions of interest. Mixed-level arrays (e.g., L12, L18, L36, and so on) enable efficient identification of large main effects by ignoring interaction effects, based on the belief that controlling main effects is much easier and robust than controlling interactions and quadric effects [30]. Larger arrays offer extremely high experimental efficiency and ready-made analytical formats (e.g., various ways of signal-to-noise ratio calculation and two-step optimizations, and so on) enable efficient screening and even optimized fine-tuning. In addition, robust parameter design (RPD), which intentionally includes a “noise factor” in experimental arrays, essentially focuses on enhancing quality stability. Especially, L18 array-based RPDs have been widely utilized in industrial fields for the product designs and manufacturing process designs, which should be robust to uncontrollable factors, such as differences in lot size and lot-to-lot differences [31].
2.5. Response Surface Method (RSM)
Key factors selected through DOE screenings can be further optimized through DOE optimizations, represented by the RSM. With RSM, main effects, interactions, and quadratic effects can be efficiently deduced, and the modeled response surface gives clear hints for further optimization. More logically, statistical approaches like steepest ascent analysis offer optimal conditions. Despite of its powerful modelling availability, because RSM requires three or five levels for each factor, the number of assessable factors would be limited practically. Another concern would be the effects of outlier data, missing values, and extremely high or low values, on model constructions, which restrict the experimental space to conservatively small.
2.6. Definitive Screening Design (DSD)
DSD is an emerging DOE that possesses fascinating merits [32]. With small experimental runs ((2 × number of factors) + 1), main effects can be defined, large interactions can be detected, and quadratic effects can be estimated. These features readily offer a response surface for factor selection and further optimization from the initial screening results. However, the use of DSD is especially recommended for the screening of novel factors with unknown effects because more than three active factors in the design fail to construct good models. In addition, other screening designs specialized for main effect detection, such as fractional factorial designs and orthogonal array designs, would more precisely identify factors possessing main effects.
2.7. Mixture Design
Another potent approach available for media improvement is mixture design, which is especially suitable for blending-ratio optimization. This method has been vigorously utilized in the food, beverage, and drug fields [33], and its use for cell culture improvements should facilitate optimization of any proportional investigation, e.g., ECM composition, co-culture cell ratio, or nutrient composition. Although the usages are limited to the ratio or proportional assessments, mixture designs offer response surface models and experimental design spaces could be set more flexibly than general RSM avoiding unnecessary experimental conditions.
2.8. Selection of DOE Approach
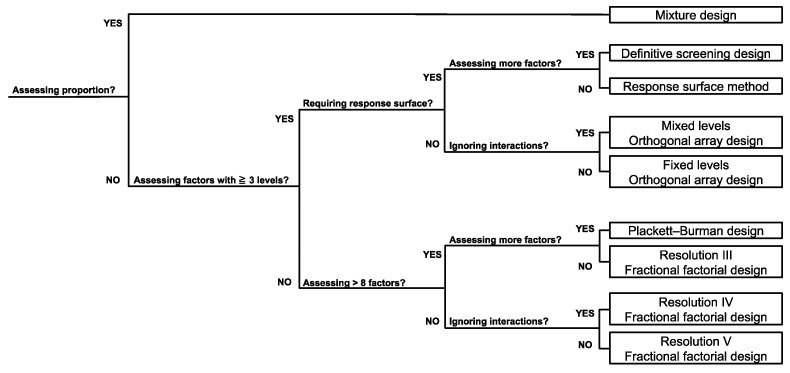
From the major strategies described above, experimenters can choose and construct appropriate experimental designs to achieve specific goals (Figure 2). For efficient multifactorial screening, the use of low-resolution fractional factorial screenings, DSD, and mixed-level orthogonal array designs is recommended. Based on the screening results, optimal conditions can be chosen through fixed-level orthogonal array designs, high-resolution fractional factorial designs, full factorial designs, and RSM assessing a selected small number of factors. Above all, because it is crucial in DOE to reasonably select the response variables, factors, and levels of factors to be investigated, those settings should be fully discussed after a vigorous literature research for marked process improvement.
Figure 2.
Decision tree for DOE selection.
3. Stem Cell Expansion and Differentiation
In this chapter, we select DOE reports investigating PSC, MSC, or HSC cultivations. These representative stem cells, in common, require efficient cell expansion and differentiation for advanced research and application. Although the required media components are totally different, DOE facilitates efficient screening and optimization as a universal framework whose methodology can be applied for future investigations of any cell culture paradigm. In each section, commonly used DOE and distinguished works are briefly introduced.
3.1. PSC Expansion and Differentiation
For investigations of PSC expansion (Table 1), full factorial screenings have been used to select cytokines and ECM that maintain ESC pluripotency [34] and optimize bioreactor parameters for aggregate ESC cultures [35]. RSM was used to investigate the basic cell signaling of ESCs under hypoxic conditions [36], develop novel human iPSC (hiPSC) maintenance media [37], and increase the yield of automated ESC [38] and microcarrier PSC culture systems [39]. In these RSM studies, models were developed for optimizations of cell growth rate, colony-forming efficiency, population doubling, and cell yield of PSCs.
Table 1.
DOE studies investigating PSC expansion.
| Cells | Experimental Design | Number of Factors | Factors | Year | Ref. |
|---|---|---|---|---|---|
| Murine ESC | Full factorial 24 | 4 | LIF, FGF4, Fibronectin, Laminin | 2004 | [34] |
| Full factorial 23 | 3 | FGF4, Fibronectin, Laminin | |||
| Human ESC | Full factorial 32 | 2 | Seeding density, Agitation speed | 2014 | [35] |
| Murine ESC | RSM | 3 | CHIR99021, LIF, PD0325901 | 2012 | [36] |
| Human ESC | RSM | 4 | Seeding density, Media volume, Media exchange time, Duration between passages | 2013 | [38] |
| Human iPSC | RSM | 2 | bFGF, NRG1β1 | 2015 | [37] |
| Human iPSC | RSM | 2 | Seeding density, Agitation speed | 2016 | [39] |
Abbreviations: LIF, leukemia inhibitory factor; FGF4, fibroblast growth factor 4; bFGF, basic fibroblast growth factor 2; NRG1β1, neuregulin 1 β1.
For investigations of PSC differentiation (Table 2), full factorial designs were used to screen additives during endoderm induction from embryoid bodies [40]; optimize ECM and cytokines during hepatic, cardiac, or mesodermal progenitor lineage induction [41,42,43]; optimize additive doses for retinal organoid production [44]. Fractional factorial screenings have also been used for definitive endoderm differentiation [45]. Mixed-level orthogonal arrays were used to screen cytokines for choroidal endothelium differentiation [46] and optimize additive doses for four distinct types of endodermal cell induction [47]. RSM was used to optimize hydrogel peptide concentrations for neural progenitor maturation [48], optimize the ECM composition for cardiomyocyte differentiation [49], and screen nutrient compositions driving trilineage specification from hiPSCs based on the response surface models for endodermal, mesodermal, and ectodermal protein expressions [50].
Table 2.
DOE studies investigating PSC differentiation.
| Cells | Purpose | Experimental Design | Number of Factors | Factors | Year | Ref. |
|---|---|---|---|---|---|---|
| Murine ESC | Endodermal differentiation | Full factorial 25 | 5 | Glucose, Insulin, bFGF, Retinoic acid, EGF | 2004 | [40] |
| Full factorial 32 | 2 | Retinoic acid, EGF | ||||
| Murine ESC | Hepatocyte differentiation | Full factorial 25 | 5 | Collagen I, Collagen III, Collagen IV, Laminin, Fibronectin | 2005 | [41] |
| Murine ESC | Cardiac cell differentiation | Full factorial 25 | 5 | Collagen I, Collagen III, Collagen IV, Laminin, Fibronectin | 2008 | [42] |
| Full factorial 24 | 4 | Wnt3a, Activin A, BMP4, FGF4 | ||||
| Human iPSC | Mesodermal progenitor differentiation | Full factorial 27 | 7 | Collagen I, Collagen III, Collagen IV, Collagen V, Laminin, Fibronectin, Vitronectin | 2015 | [43] |
| Human iPSC | Retinal organoid differentiation | Full factorial 25 | 5 | Initial cell density, 1-Thioglycerol, BMP4, KSR, Lipids | 2018 | [44] |
| Full factorial 24 | 4 | Initial cell density, CHIR99201, BMP4, SU5402 | ||||
| Human iPSC | Definitive endoderm differentiation | 24−1 Resolution IV | 4 | Activin A, GDF8, Wortmannin, CHIR99201 | 2020 | [45] |
| Human iPSC | Choroidal endothelium cell differentiation | L12 | 5 | CTGF, CTNNB1, SHC1, TWEAKR, VEGFB | 2017 | [46] |
| Human iPSC | Four endodermal cell differentiation | L18 | 8 | Retinoic acid, CHIR99201(early phase), bFGF(later phase), Sodium butyrate, bFGF(early phase), CHIR99201(later phase), (LDN193189, BMP4), A-83-01 | 2021 | [47] |
| Human iPSC-derived | Mature neuron differentiation | RSM | 3 | RGD, YIGSR, IKVAV 1 | 2015 | [48] |
| neural progenitor cell | RSM | 2 | RGD, IKVAV 1 | |||
| RSM | 2 | RGD, IKVAV 1 | ||||
| Murine iPSC | Cardiomyocyte differentiation | Full factorial 23 | 3 | Collagen I, Laminin, Fibronectin | 2015 | [49] |
| RSM | 3 | Collagen I, Laminin, Fibronectin | ||||
| Full factorial 23 | 3 | Collagen I, Fibronectin, TSP1 | ||||
| Human iPSC | Trilineage bifurcation | RSM | 3 | O2 tension, Glucose, Pyruvate | 2021 | [50] |
Abbreviations: bFGF, basic fibroblast growth factor 2; EGF, epidermal growth factor; BMP4, bone morphogenetic protein 4; FGF4, fibroblast growth factor 4; KSR, KnockOut™ Serum Replacement; GDF8, myostatin or growth differentiation factor 8; CTGF, connective tissue growth factor; CTNNB1, β catenin; SHC1, steroid receptor coactivator homology 2 domain-containing transforming protein 1; TWEAKR, TNF-related weak inducer of apoptosis receptor; VEGFB, vascular endothelial growth factor B; TSP1, thrombospondin.1 RGD, YIGSR, and IKVAV are peptides containing the indicated amino acid residues.
During early in vivo development, human embryos are exposed to dramatic environmental changes upon maternal blood perfusion [51]. From this perspective, Esteban et al. constructed a full factorial 33 RSM design space composed of O2, glucose, and pyruvate to assess how the absence and presence of these key nutrients affects trilineage specification upon spontaneous differentiation of hiPSC. By assessing key marker gene sets, they revealed that O2 deprivation promoted ectodermal differentiation; O2 deprivation and low glucose synergistically promoted mesodermal differentiation; high O2 and low glucose synergistically promoted endodermal differentiation. The temporal transition of the generated response surfaces clearly showed the different contributions of the three factors.
Although 3D cultures strongly facilitate PSC-derived cell maturation compared with two-dimensional cultures, the culture processes involved are relatively complicated and need longer culture periods, which require detailed condition screenings. Jung et al. showed that interactions between three ECM components (collagen I, fibronectin, and laminin 111) largely contributed to cardiac troponin T protein expression and finely tuned the matrix composition via an RSM with just 15 runs [49]. Importantly, the optimal cell matrix reproducibly enhanced cardiomyocyte differentiation.
To investigate definitive endoderm differentiation and patterning, we constructed an L18 array design including whole anterior-posterior endoderm by inputting eight cell signaling modifiers, thereby increasing the screening efficiency 243-fold (18 runs vs. 4374 runs) [47]. RNA expression of 18 end products seemed like “melting pots”, in which some achieved specific differentiation and others produced mixtures of cell lineages. These varied results enabled the selection of optimal conditions for upregulating desirable genes, while downregulating undesirable ones for specific anterior foregut, hepatic, pancreatic, and mid-hindgut cell inductions. Following initial screening of two hiPSC lines, the constructed protocols were successfully applied to an additional five hiPSC lines.
Another mixed-level orthogonal array design approach reported by Songstad et al. screened five cell signaling proteins that facilitate differentiation of hiPSC-derived embryoid bodies into choroidal endothelial cells [46]. An L12 array facilitated the five-factor screening 2.7-fold (12 runs vs. 32 runs) and identified positive contributions. Notably, the screening results revealed a significant contribution of connective tissue growth factor (CTGF) through cell signal inhibitor analysis.
Survival and differentiation efficiency of neural cells largely depend on the ECM and its peptide compositions. Lam et al. developed a novel hydrogel matrix and optimized the gel formulation by investigating the ratio of three adhesion peptide components via repeated RSM optimizations [48]; the results revealed positive or negative interactions between the components. Along with component optimization, both cell survival and cell spreading efficiency were improved, and immunostaining and cell sorting analysis verified significant neuronal maturation.
As shown in Table 1 and Table 2, most DOE studies investigating PSCs focused on full factorial or RSM optimization. This suggests a need and opportunities for wider screening of uninvestigated factors to overcome technical limitations of PSC expansion and differentiation. Although investigations of ECM are likely more difficult than those of soluble factors, the use of ECM array platforms has enabled high-throughput full factorial screening of up to 128 combinations with replicates [41,42,43]. Incremental iPSC differentiation studies from the late 2010s presage further DOE exploitations in this field.
3.2. MSC and HSC Expansion, and Differentiation
For investigations of MSCs (Table 3), full factorial screenings were used to identify key parameters for automated expansion [52] and screen additives for chondrogenic differentiation [53]. Fractional factorial screening was used to develop a serum-free expansion medium [54]. Orthogonal arrays were used to optimize primary MSC culture conditions from umbilical cord blood, readily identified the significant contributions of initial cell density and cytokine doses for MSC growth [55]. RSM was used to optimize osteoblast and tenocyte differentiations, which offered models for marker RNA expression levels [56,57].
Table 3.
DOE studies investigating MSC expansion and differentiation.
| Purpose | Experimental Design | Number of Factors | Factors | Year | Ref. |
|---|---|---|---|---|---|
| MSC expansion | Full factorial 24 | 4 | Seeding density, Fetal calf serum, Media volume, Culture time | 2008 | [52] |
| Chondrocyte differentiation | Full factorial 25 | 5 | TGFβ1, BMP2, DEX, FGF2, IGF1 | 2014 | [53] |
| MSC expansion | 24−1 Resolution IV | 4 | Hydrocortisone, bFGF, Human albumin, SITE supplement 1 | 2007 | [54] |
| MSC expansion | L8 | 4 | Seeding density, Cytokines 2, Serum, Stromal cells | 2009 | [55] |
| L8 | 6 | SCF, TPO, FL, IL-3, GM-CSF, G-CSF | |||
| Osteoblast differentiation | RSM | 4 | Culture duration, O2 tension, Seeding density, Two media 3 | 2011 | [56] |
| Tenocyte differentiation | RSM | 2 | TGFβ3, Culture days | 2020 | [57] |
Abbreviations: TGFβ1, transforming growth factor β1; BMP2, bone morphogenetic protein 2; DEX, dexamethasone; FGF2, fibroblast growth factor 2; IGF1, insulin-like growth factor 1; bFGF, basic fibroblast growth factor 2; SCF, stem cell factor; TPO, thrombopoietin; FL, FMS-like tyrosine kinase 3 ligand; IL-3, interleukin-3; GM-CSF, granulocyte macrophage-colony stimulating factor; G-CSF, granulocyte-colony stimulating factor; TGFβ3, transforming growth factor β3.1 SITE supplement contains sodium selenite, bovine insulin, human transferrin, and ethanolamine.2 Cytokines contain stem cell factor, FL, TPO, IL-3, G-CSF, and GM-CSF.3 Two media are α-MEM supplemented with ascorbate-2-phosphate or α-MEM supplemented with ascorbate-2-phosphate, DEX, and β-glycerophosphate.
For clinical use of cell-derived materials, one of the big challenges is the development of chemically defined, animal origin-free cell culturing processes. After pre-screening of over 15 compounds, Liu et al. chose and screened four additives using a resolution IV 24−1 fractional factorial design, and examined the viability of human cord blood MSCs [54]. A mere eight runs of cultivation defined a serum-free medium capable of yielding MSCs possessing the same growth ability and adipogenic, chondrogenic, and osteogenic differentiation potentials as those maintained in serum-containing media.
For efficient differentiation of human MSCs into chondrocytes, Jakobsen et al. screened five cytokines with 25 full factorial design [53]. For each of the 32 cultivation runs, the expression of 364 chondrogenic genes was analyzed. The gene set contained both desirable articular cartilage markers and undesirable bone or adipose markers, thus enabling detection of marked contributions of transforming growth factor β1 (TGFβ1) and dexamethasone (DEX) to chondrogenesis.
For investigations of HSC (Table 4), full factorial designs were used to screen cytokines for long-term culture-initiating cells and colony-forming cell bifurcation [58,59,60], as well as platelet production [61]. Fractional factorial designs were used to screen the cytokines relative to HSC expansion [62,63] and its lineage commitments toward erythroid, granulocyte, megakaryocyte, and dendric cell [64,65,66,67]. RSM was used for the detailed optimization of HSC expansion [68] and to identify long-term culture-initiating cells or colony-forming cell bifurcation based on cell yields [69] and megakaryocyte induction was optimized through the modeling of cell yield, cell expansion ratio, and platelet production [70]. The resulting cytokine usages, lineage commitments, and DOE approaches for HSC investigations were diligently reviewed by Lim et al. [71].
Table 4.
DOE studies investigating HSC expansion and differentiation.
| Purpose | Experimental Design | Number of Factors | Factors | Year | Ref. |
|---|---|---|---|---|---|
| LTC-IC and CFC bifurcation | Full factorial 25 | 5 | FL, SF, IL-3, IL-6, (G-CSF, NGFβ) | 1996 | [58] |
| LTC-IC and CFC bifurcation | Full factorial 23 | 3 | FL, SF, IL-3 | 1997 | [59] |
| LTC-IC and CFC bifurcation | Full factorial 26 | 6 | FL, SF, IL-3, (IL-6, sIL-6R), TPO, IL-1 | 1998 | [60] |
| Megakaryocyte and platelet differentiation | Full factorial 24 | 4 | SCF, IL-3, IL-6, IL-9 | 2013 | [61] |
| HSC expansion | 29−5 Resolution III | 9 | TPO, IL-3, SCF, FL, G-CSF, GM-CSF, IL-6, sIL-6R, EPO | 2003 | [62] |
| 24−1 Resolution IV | 4 | TPO, IL-3, SCF, FL | |||
| 28−4 Resolution IV | 8 | Albumax, BSA, TF, Glutamine, Hydrocortisone, Peptone, 2-ME, Insulin | |||
| 24 | 4 | BSA, Insulin, TF, 2-ME | |||
| 27−3 Resolution IV | 7 | TPO, IL-3, SCF, FL, G-CSF, GM-CSF, IL-6 | |||
| HSC expansion | Full factorial 24 | 4 | BSA, Insulin, TF, 2-ME | 2004 | [63] |
| 210−6 Resolution III | 10 | TPO, IL-3, SCF, FL, IL-11, IL-6, GM-CSF, G-CSF, SCGF, HGF | |||
| Erythroid cell, granulocyte, and megakaryocyte differentiation | 27−3 Resolution IV | 7 | FL, SCF, IL-3, (MGDF, G-CSF), IL-11, IL-6, EPO | 2001 | [64] |
| Full factorial 24 | 4 | IL-3, IL-11, IL-6, EPO | |||
| Megakaryocyte differentiation | 28−3 Resolution IV | 8 | TPO, IL-3, SCF, FL, IL-11, IL-6, GM-CSF, IL-9 | 2009 | [65] |
| Dendritic cell differentiation | 28−4 Resolution IV | 8 | SCF, FL, IL-1β, GM-CSF, TNFα, IL-4, IL-6, TGFβ1 | 2019 | [66] |
| 25−1 Resolution V | 5 | SCF, FL, IL-1β, GM-CSF, TNFα | |||
| HSC differentiation ability | 25−1 Resolution V | 5 | SCF, FL, TPO, SDF-1, Fucoidan | 2011 | [67] |
| Full factorial 23 | 3 | SCF, FL, TPO | |||
| HSC expansion | RSM | 4 | SCF, FL, TPO, LIF | 2010 | [68] |
| LTC-IC and CFC bifurcation | Full factorial 25 | 5 | IL-11, SF, FL, TPO, Temperature | 2002 | [69] |
| RSM | 3 | IL-11, SF, FL | |||
| Megakaryocyte and platelet differentiation | Plackett–Burman | 11 | SCF, FL, IL-11, MIP-1α, IL-1α, IL-1β, IL-8, IFN-γ, VEGF, MCP-1, β-thromboglobuline | 2005 | [70] |
| Plackett–Burman | 9 | IL-9, IL-8, IL-6, IL-1α, IL-1β, SCF, FL, MIP-1α, IFN-γ | |||
| 25−1 Resolution V | 5 | SCF, FL, IL-6, IL-9, EPO | |||
| Full factorial 24 | 4 | SCF, FL, IL-6, IL-9 | |||
| RSM | 4 | TPO, SCF, IL-6, IL-9 |
Abbreviations: FL, FMS-like tyrosine kinase 3 ligand; SF, steel factor; IL-3, interleukin-3; IL-6, interleukin-6; G-CSF, granulocyte-colony stimulating factor; NGFβ, nerve growth factor β; sIL-6R, soluble IL-6 receptor; TPO, thrombopoietin; IL-1, interleukin-1; SCF, stem cell factor; MGDF, megakaryocyte growth and development factor; IL-11, interleukin-11; EPO, erythropoietin; GM-CSF, granulocyte macrophage-colony stimulating factor; TF, transferrin; 2-ME, 2-mercaptoethanol; SCGF, stem cell growth factor α; HGF, hepatocyte growth factor; IL-1β, interleukin-1β; TNFα, tumor necrosis factor α; IL-4, interleukin-4; TGFβ1, transforming growth factor β1; SDF-1, stromal cell-derived factor-1; MIP-1α, macrophage inhibitory protein-1α; IL-1α, interleukin-1α; IL-8, interleukin-8; IFN-γ, interferon γ; VEGF, vascular endothelial growth factor; MCP-1, monocyte chemoattractant protein-1.
To optimize media components for HSCs and their derivatives, Yao et al. reported numerous DOE results and employed systematic approaches by combining fractional factorial screening and steepest ascent. In their 2003 report, Yao and colleagues isolated HSCs from cord blood and screened numerous cytokines and serum components by low-resolution fractional factorial screenings [63]. The established medium expanded HSCs, colony-forming cells, and white blood cells more efficiently than other published results. Similar strategies were applied for direct HSC expansion from umbilical cord blood mononuclear cells to omit the HSC-isolation phase, which increased the experimental efficiency up to 64-fold (16 runs vs. 1024 runs) [64]. They also achieved megakaryocyte or platelet production [65] and potent antigen-presenting dendric cell differentiation from their HSCs [66] by applying fractional factorial screenings.
.
Differentiation of HSCs has been thoroughly investigated by DOE. The significant factors chosen from low-resolution fractional screening, including Plackett–Burman designs, seem to reproducibly contribute to the desired cell expansions and differentiations, probably due to their larger main effects compared with the interaction effects brought about by other factors. Notably, the wide varieties of target cell lineages and cytokines investigated suggest the applicability of multifactorial screening to enhance differentiation.
4. CHO Cell Expansion
CHO cell lines are exemplary mammalian cells whose culture processes have been thoroughly investigated using various DOE (Table 5). Indeed, detailed media optimizations utilizing DOE have been reported for effective recombinant protein and antibody production processes. We collected DOE investigations evaluating cell yield and/or viability as response variables [72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95], or selecting desirable clones [96,97].
Table 5.
DOE studies investigating CHO cell expansion.
| Experimental Design | Number of Factors | Factors | Year | Ref. |
|---|---|---|---|---|
| Full factorial 23 | 3 | Glucose, Glutamine, Inorganic salts | 2004 | [72] |
| Full factorial 25 | 5 | Feed volume at days 3, 5, 7, 10, and 12 | 2019 | [73] |
| 25−1 Resolution V | 5 | Sodium hypoxanthine-thymidine, Antioxidant, ITS 1, Fatty acids supplement, Polyamines supplement | 2006 | [74] |
| 24−1 Resolution IV | 5 | Amino acid feed, Glucose Feed, Temperature, pH | 2011 | [96] |
| Full factorial 31 × 22 | 3 | Glucose feed, Temperature shift, pH control frequency | ||
| Plackett–Burman | 20 | BSA, Transferrin, Insulin, Sodium pyruvate, Putrescine, Glucose, Ala, Arg, Asn, Asp, Cys, Gln, Glu, Gly, Ser, Met, (Pro, His, Hydroxyproline), (Thr, Val, Ile), (Leu, Trp, Lys), (Phe, Tyr) | 1992 | [75] |
| Plackett–Burman | 4 | Oleic acid, Linoleic acid, Cholesterol, (Choline, Ethanolamine) | 1995 | [76] |
| Plackett–Burman | 21 | Ala, Arg, (Asn, Asp), Cys, Gln, Glu, Gly, Ser, Met, (Phe, Tyr), (Thr, Val, Ile), (Leu, Trp, Lys), (Pro, His), Insulin, Transferrin, Ethanolamine, Pluronic F68, Phosphatidylcholine, Putrescine, Linoleic acid, Hydrocortisone | 1998 | [77] |
| Plackett–Burman | 21 | Ala, Arg, (Asn, Asp), Cys, Gln, Glu, Gly, Ser, Met, (Phe, Tyr), (Thr, Val, Ile), (Leu, Trp, Lys), (Pro, His), Insulin, Transferrin, Ethanolamine, Pluronic F68, Phosphatidylcholine, Hydrocortisone, Sodium selenite, Glutathione | 1999 | [78] |
| Plackett–Burman | 21 | Ala, Arg, (Asn, Asp), Cys, Gln, Glu, Gly, Ser, Met, (Phe, Tyr), (Thr, Val, Ile), (Leu, Trp, Lys), (Pro, His), Sodium selenite, Insulin, Transferrin, Hydrocortisone, Ethanolamine, Phosphatidylcholine, Glutathione, Pluronic F68 | 1999 | [79] |
| RSM | 2 | Glucose, Gln | 2005 | [80] |
| RSM | 2 | Glucose, NaCl | ||
| 27−3 Resolution IV | 7 | Insulin, Meat peptone, Yeast extract, SerEx, BSA, Linoleic acid–BSA, Dextran sulfate | 2006 | [81] |
| RSM | 2 | Insulin, SerEx | ||
| RSM | 5 | Gln, Essential amino acids supplement, Non-essential amino acids supplement, ITS 1, Lipids | 2007 | [82] |
| RSM | 3 | Yeastolate, Soy, Wheat | 2009 | [83] |
| Plackett–Burman | 17 | Ethanolamine, Sodium selenite, Putrescine, Hydrocortisone, Lipids, Sodium pyruvate, Ascorbic acid, Glutathione, Choline chloride, D-calcium pantothenate, Folic acid, Niacinamide, Pyridoxine-hydrochloride, Riboflavin, Thiamine hydrochloride, Cyanocobalamin, I-inositol | 2013 | [84] |
| RSM | 3 | Lipids, Putrescine, Ammonium ferric citrate | ||
| RSM | 3 | Temperature, pH, Seeding density, Culture duration | 2013 | [85] |
| RSM | 3 | Glucose, Asn, Gln | 2015 | [86] |
| Plackett–Burman | 19 | 19 amino acids (Gln excluded) | 2015 | [87] |
| RSM | 4 | Asp, Glu, Arg, Gly | ||
| RSM | 3 | pH, O2 tension, CO2 tension | 2017 | [88] |
| 28−4 Resolution IV | 8 | 8 kinds of commercial supplements | 2020 | [89] |
| RSM | 4 | 4 kinds of commercial supplements | ||
| Full factorial 23 | 3 | 3 kinds of commercial supplements | ||
| Plackett–Burman | 8 | Sodium selenite, Transferrin, Albumin, Insulin, Tocopherol, Tween 80, Fatty acids, Synthetic cholesterol | 2019 | [90] |
| Box–Behnken RSM | 3 | Transferrin, Insulin, Tween 80 | ||
| Plackett–Burman | 15 | Gln, Asp, Lys, Trp, Thr, Val, His, Vitamin B1, Thymidine, Deoxy-cytidine, 3-methyl-oxobutyrate, Deoxy-guanosine, Vitamin B6, Vitamin A, Arachidonate | 2020 | [91] |
| RSM | 2 | Thr, Arachidonate | ||
| Mixture Design | 6 | Hexoses, Energy provider compounds | 2007 | [92] |
| Mixture Design | 20 | 20 amino acids | 2013 | [97] |
| Mixture Design | 43 | 19 amino acids (Gln excluded), Disodium phosphate, Magnesium sulfate, Calcium chloride, Myo-inositol, Sodium pyruvate, D-biotin, Choline Chloride, Folic acid, Niacinamide, D-pantothenic acid, Potassium chloride, Pyridoxine, Riboflavin, Thiamine, Ferric ammonium citrate, Vitamin B12, Hypoxanthine, Thymidine, Putrescine, Ethanolamine, Zinc sulfate, Cupric sulfate, Pluronic, Sodium selenite | 2013 | [93] |
| DSD | 5 | pH, Shifted temperature, Seeding density, Viable cell density at first feeding, Viable cell density at temperature shift | 2019 | [94] |
| DSD | 6 | DMEM fraction, Cellgro trace element A, Cellgro trace element B, Insulin, Ca2+, Mg2+ | 2021 | [95] |
1 ITS supplement contains sodium selenite, bovine insulin, and human transferrin.
Although the base media used for PSC differentiation mostly depend on commercial media, such as RPMI, Eagle’s, and Ham’s media with some supplements, nutrient levels in CHO culture media have been optimized through detailed investigations, sometimes for each clone. Torkashvand et al., achieved a 1.7-fold increase of monoclonal antibody (mAb) titer by screening 19 amino acids in 20 runs with a Plackett–Burman design, followed by RSM dose optimization of the four key amino acids [76]. Obvious inhibitory effects arising from some unnecessary amino acid additions highlight the importance of dedicated evaluations of base media for cell quality improvements.
Selecting the right cell strain or clone can resolve difficulties in later research and developmental stages, in some cases because some human PSC characteristics originate from the process used to establish the clone [98,99]. In a CHO cell line-development process, Mora et al. employed factorial DOE to feed 10 CHO clones with 24 different feed plans [73]. This screening revealed that the clones could be divided into early and late responders, which differed in their peak timing for molecule production. On the basis of the screening results, they modified the feeding strategy and improved the molecule titers of seven investigated clones (up to 34% increase), while reducing about 40% of hands-on time for culture maintenance by skipping unnecessary media changes.
CHO cells originally maintained adherently in the presence of serum are often adapted to serum-free cell suspension culture to improve productivity and quality, especially in industry. This adaptation process was optimized by Wu et al., who screened five additives employing DSD in just 15 experimental runs [95]. Consequently, with the established medium condition, the adaptation process that conventionally took 66 days was successfully shortened to just 27 days.
Screening of wider design spaces with more data points can be investigated through mixture design approaches. Jordan et al. prepared 10 different nutrient cocktails, each containing different combinations of 20 amino acids, and further systematically blended the 10 cocktails at various combinations and ratios. From the resulting 192 media, the medium generating the highest mAb titer was chosen [97]. More detailed statistical analysis revealed the limiting dose of each amino acid, suggesting that further optimization of medium components is possible on the basis of the accumulated data. Likewise, Rouiller et al. included 43 nutrients in 16 cocktails, and mixed them to obtain 376 media [93]. Some media increased the mAb titer up to 1.4-fold compared with the control condition, and further improvement was predicted through the identification of key components.
In addition to screening cytokines, the base medium components for CHO cell culture have been minutely investigated. Such approaches should be applied not only for PSC expansion processes, but also to optimize base media for differentiation of cells to increase yield and/or purity of the end material. Screening efficiency was further increased by regarding groups of nutrients as a factor, based on their roles in metabolic pathways. Notably, the use of mixed-level orthogonal array designs, mixture designs, and DSDs for cell culture have great potential to further accelerate media development because of their high screening efficiency.
5. Other Cell Expansion, Cell Differentiation, and Cell-Material Development Processes
Cell isolation and/or selective expansion of target cells (Table 6) also requires intricate screening, and this process needs to be robust enough for reproducible utilization for different cell origins and donors. To improve human cell yields, culture conditions for pancreatic duct cells were assessed by full factorial design [100], or co-culture of intestinal Caco-2 cells and goblet HT29-MTX cells [101], umbilical vein endothelial cells [102], cytotoxic T lymphocytes [103], prostate cancer cells [104], and immortalized erythroblasts [105] were investigated by RSM modeling of cell numbers. In addition to these human cell types, Vero cells [106,107] and murine cells [108,109,110] have been assessed by fractional factorial and orthogonal array designs.
Table 6.
DOE studies investigating other cell expansion.
| Cells | Experimental Design | Number of Factors | Factors | Year | Ref. |
|---|---|---|---|---|---|
| Human pancreatic duct cell | Full factorial 25 | 5 | bFGF, EGF, HGF, KGF, VEGF | 2012 | [100] |
| Vero | 210−6 Resolution III | 10 | (20 amino acids, Vitamin B1, Magnesium sulfate, Sodium phosphate), (Vitamins H, B2, and B9, Thymidine, Uracil, Xanthine, Hypoxanthine), (Vitamins B12, B3, and B7, Choline chloride, Pyridoxal), (Vitamins B3, B6, and BX, Putrescin), (Vitamins A, D2, and K3, Linoleic acids, Lipoic acids), (Deoxyribose, Adenine, Adenosine, Ethanolamine), (Plant and yeast extracts, EGF, Insulin), (Sodium citrate, Ferric chloride), (Glucose, Pyruvate), (Other) | 2010 | [106] |
| Murine hybridoma | 29−4 Resolution IV | 9 | Serum, Dissolved oxygen, Temperature, pH, Glucose, Glutamine, Lactate, Ammonium, Base medium concentration | 1993 | [108] |
| Murine myeloma | 25−1 Resolution V | 5 | pH, Temperature, Dissolved oxygen, Early/late feed regime, Seeding density | 2000 | [109] |
| Murine hybridoma | L8 | 4 | Stirring speed, Fetal bovine or calf serum, Serum concentration, Glucose and glutamine supplement | 2002 | [110] |
| Vero | L8 | 4 | Cytodex 1, Regulation of glucose, Initial glucose, Gln | 2006 | [107] |
| Caco-2 and HT29-MTX cells | L18 | 4 | MEM or DMEM medium, Seeding time, Seeding density, and Caco-2/HT29-MTX ratio | 2010 | [101] |
| Human umbilical vein endothelial cell (HUVEC) | Full factorial 24 | 4 | RGDS, IKVAV, YIGSR, Q11 1 | 2011 | [102] |
| RSM | 3 | RGDS, IKVAV, YIGSR 1 | |||
| Human peripheral blood mononuclear cell | 24−1 Resolution IV | 4 | Phosphatidyl choline, Polyamine supplement, Antioxidant supplement, Cholesterol | 2010 | [103] |
| RSM | 2 | Polyamine supplement, Cholesterol | |||
| Human prostate cancer cells | Plackett–Burman | 16 | Transferrin, Sodium selenite, Sodium L-ascorbate, Ferric citrate, L-glutathione, BSA, EGF, bFGF, Ethanolamine, Linoleic acid, Arachidonate, Thioglycerol, Hydrocortisone, Yeast hydrolysate, Penicillin-Streptomycin Solution, Succinic Acid | 2017 | [104] |
| RSM | 3 | EGF, FGF, Linoleic acid | |||
| Immortalized human erythroblast | 29−4 Resolution IV | 9 | BSA, EPO, Holo-transferrin, Hydrocortisone, Insulin, Fatty acid supplement, Lipid mixture solution, Non-essential amino acids supplement, SCF | 2018 | [105] |
| RSM | 3 | BSA, EPO, Fatty acid supplement |
Abbreviations: bFGF, basic fibroblast growth factor 2; EGF, epidermal growth factor; HGF, hepatocyte growth factor; KGF, keratinocyte growth factor; VEGF, vascular endothelial growth factor; MEM, Minimum Essential Medium with Earle’s Salts; DMEM, Dulbecco’s Modified Eagle’s Medium; EPO, erythropoietin; SCF, stem cell factor.1 RGDS, IKVAV, and YIGSR are peptides containing the indicated amino acid residues. Q11 is the structural peptide used as control matrix.
To improve cell differentiation (Table 7), human chondrocytes were differentiated into cartilage [111,112], human periosteum-derived cells and osteosarcoma cells were investigated for skeletal tissue development [113], human adipose-derived stromal cells and murine embryonic fibroblasts were differentiated into osteoblasts [114,115], human hepatoma cells were differentiated into hepatocytes [116], and mouse pluripotent embryonic carcinoma were differentiated into neuronal cells [117]. In these RSM optimizations, response surfaces of cell spreading, osteogenic markers [115], and metabolites [116] were obtained.
Table 7.
DOE studies investigating other cell differentiation.
| Cells | Purpose | Experimental Design | Number of Factors | Factors | Year | Ref. |
|---|---|---|---|---|---|---|
| Mouse pluripotent embryonic carcinoma | Neuronal cell differentiation | Full factorial 23 | 3 | 2D- or 3D-culture, IKVAV 1, ECM stiffness | 2012 | [117] |
| Human chondrocytes | Cartilage differentiation | 212−4 Resolution VI | 12 | BMP2, Insulin, IGF1, Testosterone, Parathyroid hormone, IL-1RA, Growth hormone, 17β-estradiol, Triiodothyronine, 1α-25-dihydroxy vitamin D3, FGF2, DEX | 2007 | [111] |
| Human chondrocytes | Articular chondrocyte differentiation | 25−1 Resolution V | 5 | TGFβ1, ASC, ITS, DEX, Linoleic acid | 2012 | [112] |
| Full factorial 23 | 3 | TGFβ1, DEX, Glucose | ||||
| Full factorial 22 | 2 | DEX, Glucose | ||||
| Human bone progenitor cells | Skeletal tissue development | 25−1 Resolution V | 5 | Medium volume, Seeding density, Human periosteum-derived cell or osteosarcoma cell, Seeding timing, Foamed titanium or 3D fiber-deposited titanium | 2011 | [113] |
| Human adipose-derived stromal cells | Osteoblast differentiation | 12 × 12 Hadamard matrix 2 | 8 | Two human adipose-derived stromal cells suppliers, Seeding density, DMEM/F12 or DMEM, Human platelet lysate or Fetal bovine serum, L-ascorbate-2-phosphate, β-glycerophosphate, DEX, BMP9 | 2019 | [114] |
| Human hepatoma cell | Hepatocyte differentiation | 27−4 Resolution III | 7 | Human serum albumin, HGF, Oncostatin M, DEX, FGF4, EGF, Nicotinamide | 2008 | [116] |
| RSM | 3 | Oncostatin M, HGF, FGF4 | ||||
| Murine embryonic fibroblast cell | Osteoblast differentiation | RSM | 2 | Matrix stiffness, Collagen I | 2010 | [115] |
Abbreviations: BMP2, bone morphogenetic protein 2; IGF1, insulin-like growth factor 1; IL-1RA, interleukin-1 receptor antagonist; FGF2, fibroblast growth factor 2; DEX, dexamethasone; BMP9, bone morphogenetic protein 9; HGF, hepatocyte growth factor; FGF4, fibroblast growth factor 4; EGF, epidermal growth factor. 1 IKVAV is a peptide containing the indicated amino acid residues.2 Hadamard matrix is a fixed level orthogonal array.
The use of DOE is not limited to cell cultivation and can also be applied for other cell-related purposes (Table 8), such as cell storage, cell transportation, and related materials and devices, which are especially important issues for industrial cell usage. The refrigerated storage conditions for retinal pigment epithelial cells [118] and epithelial cell sheets [119] were optimized through full and fractional factorial screening. In addition, the virus inactivation process of Vero cells [120] was investigated using an orthogonal array. Optimal freezing conditions for CHO and human embryonic kidney (HEK) cells were identified by RSM through the modelling of the first doubling time after the cell thawing [121].
Table 8.
DOE studies investigating other cell-related processes.
| Cells | Purpose | Experimental Design | Number of Factors | Factors | Year | Ref. |
|---|---|---|---|---|---|---|
| Human retinal pigment epithelial cells | Cell storage condition | Full factorial 25 | 5 | Adenosine, Allopurinol, β-Glycerophosphate, L-Ascorbic acid, Taurine | 2018 | [118] |
| Human epithelial cell sheets | Cell storage condition | 210−4 Resolution IV | 10 | 1% Glycerol, L-Ascorbic acid, Allopurinol, Sodium pyruvate, Adenosine, Taurine, L-Glutathione, Hydrocortizone, LiCl, Antimycin-A | 2018 | [119] |
| 210−4 Resolution IV | 10 | 0.75% Glycerol, 3% Glycerol, Icilin, Menthol, Dimethyl (S)-(−)-malate, Methyl pyruvate, N-Acetyl-L-Cys, Insulin, Acetovanillone, N-(2-Mercaptopropionyl)glycine | ||||
| Full factorial 55 | 5 | L-Carnosine, Dimethyl sulfoxide, Fenoldopam mesylate, Glycerol, LIF | ||||
| Full factorial 55 | 5 | Glycerol, Aspirin, Melatonin, Lactic acid, ATP | ||||
| Vero | Virus inactivation | L9 | 4 | Temperature, Treatment time, pH, Ethanol | 2019 | [120] |
| CHO cell and HEK293 | Cell freezing and refreezing condition | RSM | 3 | Freezing density, Dimethyloxide, Seeding density | 2008 | [121] |
Abbreviations: LIF, leukemia inhibitory factor.
For prostate cancer cell expansion toward vaccine development, Zhao et al. screened 16 compounds in 20 runs using the Plackett–Burman approach [117]. This screening indicated that epidermal growth factor (EGF), fibroblast growth factor (FGF), and linoleic acid had the highest positive effects on cell yield. The best doses of these additives were determined by RSM in another 20 runs of experiments. The established medium was confirmed to support prostate cancer cell growth equal to or greater than serum-containing medium.
During in vitro chondrocyte expansion, some mature chondrocytes undesirably dedifferentiate into immature fibrous chondrocytes. This obstacle to chondrocyte amplification is overcome through redifferentiation culture, in which expanded fibrous chondrocytes are again matured while avoiding hypertrophic differentiation. This cell bifurcation was inspected by two different DOE approaches. First, Enochson et al. optimized the redifferentiation medium by assessing the gene expression of mature and immature chondrocyte markers [112]. Five-factor fractional factorial screening (25−1) extracted contributors, while subsequent RSM optimizations established the medium, to induce histologically better micromass formation than conventional medium. Throughout the investigation, gene expression of collagens was not ideally controlled; thus indicating that collagen expression might be regulated by other cell signaling pathways and that further screening of collagen regulators could lead to quality improvement. Liu et al. screened 12 candidate regulators for auricular chondrocyte redifferentiation by monitoring glycosaminoglycan accumulation as a response variable by 212−4 fractional factorial design [111], which increased screening efficiency 16-fold (256 runs vs. 4096 runs). A combination of three cytokines was found to promote glycosaminoglycan accumulation while preventing hypertrophic differentiation. Efficacy of the optimized medium was validated by redifferentiation of chondrocytes from different sources (articular and rib), mechanical property analysis of 3D-cultured cartilage pellets, and pellet implantation into nude mice.
In this chapter, we confirmed that DOE facilitated optimization of cell culture processes, regardless of the cell lineages involved. Although an enormous number of factors were incorporated in DOE for different purposes, DOE successfully identified positive factors and optimal conditions. The versatility of DOE promises to accelerate PSC-derived material research and development in any situation.
6. Concluding Remarks
The presented efforts adopting DOE approaches in mammalian cell culture confirm that DOE-based screening and optimization of stem cell culture facilitates novel differentiation protocol developments and strategic evidence accumulation. Moreover, technical hurdles arising from different cell characteristics of donors or clones might be overcome by the construction of robust universal media components or donor-specific customized media through high-throughput optimization. In particular, multifactorial screening has good opportunities to reveal novel cell signaling regulation, which is not only important for its biological meaning, but also desirable for stable and high-quality cell-differentiated materials.
There is still room for more effective utilization of DOEs, especially for PSC investigations. Indeed, because most previous studies were based on closed settings with full factorial or RSM designs, rather than open for novel factor searching [122], the high experimental efficiency of DOE is not fully utilized. In addition, because most PSC studies were designed to assess cell-signaling modifiers, nutrient composition could also be improved, as evaluated in CHO and other cell investigations [123]. Emerging metabolome analysis and online monitoring of medium compositions along the differentiation process should offer hints for further multifactorial screening. The significant contributions of basal media composition on PSC lineage commitment [50] suggest that further addition or removal of energy sources can improve the quality and/or yield of PSC and PSC-derived materials.
The timing and duration of additive treatments seem to be poorly investigated, although differentiating cells mature during the cultivation process. For instance, in the course of endoderm differentiation, the timing and duration of retinoic acid exposure significantly affects lineage commitment both in vivo and in vitro [124]. To assess time-dependent regulation of cell signaling, three or more multilevel screenings using orthogonal arrays or DSDs might be more suitable. However, because incorporation of time-related factors in DOEs tends to enlarge interaction effects, experimental plans should be carefully reviewed.
Another underexplored time-related issue is simultaneous investigation of media components from different cultivation phases. For example, if a differentiation process consists of three phases, the first, second, and third phases are often sequentially developed, and then a problematic phase might be refined. This “one phase at a time” approach leads to quasi-optimal or frail conditions in the same manner as OFAT [15]. Particularly for cell differentiation processes, it is strongly supposed that the cell status (e.g., epigenetic regulation) in earlier phases largely influences the cell-lineage commitment of later phases; thus, interactions between factors used in different phases would have significant effects. DOE investigations of whole-process development methodologies, such as the studies summarized in Section 6 or reviewed elsewhere [125,126], offer hints for better experimental planning.
To screen numerous conditions appropriately, novel devices and platforms (such as those used for ECM optimizations) can decrease experimental cost, human error, and statistical experimental error. Utilization of robotics should especially be considered when incorporating many factors or implementing a large-scale mixture design, an intricate media preparation or media-change scheme [97,127]. The use of robots can further promote sequential optimizations through DOE planning, subsequent cell culture, cell characterization by omics analysis and/or cell sorting, and validation of candidate optimal protocols without human intervention. Those acquired data should be particularly suitable for further in silico modeling of cell culture processes [128,129,130].
The use of DOE has no drawbacks if the experimenters have enough knowledge and skills in the research field. Because DOE prompts research activities whose concrete utilities have been ubiquitously elucidated, its use and understanding in academia, industry, and administrative authorities are more essential than ever. We are certain that wider DOE utilization will reveal new biological insights and novel solutions to establish more efficient cell differentiation processes for further cell material applications.
Author Contributions
R.Y. wrote the draft of the manuscript; R.Y. and K.S. wrote the final version of the manuscript. H.T. gave administrative support, and final approval of manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This work was partly supported by the Japan Agency for Medical Research and Development (AMED) Research Center Network for Realization of Regenerative Medicine (H.T.), along with Grants-in-Aid 18K19589 from the Ministry of Education, Culture, Sports, Science and Technology of Japan (K.S.).
Conflicts of Interest
R.Y. is employee of Eiken Chemical Co., Ltd. Authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Yamanaka S. Pluripotent Stem Cell-Based Cell Therapy—Promise and Challenges. Cell Stem Cell. 2020;27:523–531. doi: 10.1016/j.stem.2020.09.014. [DOI] [PubMed] [Google Scholar]
- 2.Umezawa A., Sato Y., Kusakawa S., Amagase R., Akutsu H., Nakamura K. Research and Development Strategy for Future Embryonic Stem Cell-Based Therapy in Japan. JMA J. 2020;3:287–294. doi: 10.31662/jmaj.2018-0029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Vickaryous M.K., Hall B.K. Human cell type diversity, evolution, development, and classification with special reference to cells derived from the neural crest. Biol. Rev. Camb. Philos. Soc. 2006;81:425–455. doi: 10.1017/S1464793106007068. [DOI] [PubMed] [Google Scholar]
- 4.Kim M.H., Kino-Oka M. Designing a blueprint for next-generation stem cell bioprocessing development. Biotechnol. Bioeng. 2020;117:832–843. doi: 10.1002/bit.27228. [DOI] [PubMed] [Google Scholar]
- 5.Rohani L., Johnson A.A., Naghsh P., Rancourt D.E., Ulrich H., Holland H. Concise Review: Molecular Cytogenetics and Quality Control: Clinical Guardians for Pluripotent Stem Cells. Stem Cells Transl. Med. 2018;7:867–875. doi: 10.1002/sctm.18-0087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sullivan S., Stacey G.N., Akazawa C., Aoyama N., Baptista R., Bedford P., Bennaceur Griscelli A., Chandra A., Elwood N., Girard M., et al. Quality control guidelines for clinical-grade human induced pluripotent stem cell lines. Regen. Med. 2018;13:859–866. doi: 10.2217/rme-2018-0095. [DOI] [PubMed] [Google Scholar]
- 7.Ortuño-Costela M.D.C., Cerrada V., García-López M., Gallardo M.E. The Challenge of Bringing iPSCs to the Patient. Int. J. Mol. Sci. 2019;20:6305. doi: 10.3390/ijms20246305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Li W., Fan Z., Lin Y., Wang T.Y. Serum-Free Medium for Recombinant Protein Expression in Chinese Hamster Ovary Cells. Front. Bioeng. Biotechnol. 2021;9:172. doi: 10.3389/fbioe.2021.646363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Brown K., Loh K.M., Nusse R. Live Imaging Reveals that the First Division of Differentiating Human Embryonic Stem Cells Often Yields Asymmetric Fates. Cell Rep. 2017;21:301–307. doi: 10.1016/j.celrep.2017.09.044. [DOI] [PubMed] [Google Scholar]
- 10.Fowler J.L., Ang L.T., Loh K.M. A critical look: Challenges in differentiating human pluripotent stem cells into desired cell types and organoids. Wiley Interdiscip. Rev. Dev. Biol. 2019;9:e368. doi: 10.1002/wdev.368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yao T., Asayama Y. Animal-cell culture media: History, characteristics, and current issues. Reprod. Med. Biol. 2017;16:99–117. doi: 10.1002/rmb2.12024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lin Z., Xiao Z., Lan L., Ya T., Liu M., Qu K., Wang Z., Zeng Z.L., Lin X.L., Tan L.L., et al. MicroRNAs: Important Regulators of Induced Pluripotent Stem Cell Generation and Differentiation. Stem Cell Rev. Rep. 2018;14:71–81. doi: 10.1007/s12015-017-9785-6. [DOI] [PubMed] [Google Scholar]
- 13.Matsumoto R., Yamamoto T., Takahashi Y. Complex organ construction from human pluripotent stem cells for biological research and disease modeling with new emerging techniques. Int. J. Mol. Sci. 2021;22:10184. doi: 10.3390/ijms221910184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kirouac D.C., Zandstra P.W. The Systematic Production of Cells for Cell Therapies. Cell Stem Cell. 2008;3:369–381. doi: 10.1016/j.stem.2008.09.001. [DOI] [PubMed] [Google Scholar]
- 15.Toms D., Deardon R., Ungrin M. Climbing the mountain: Experimental design for the efficient optimization of stem cell bioprocessing. J. Biol. Eng. 2017;11:35. doi: 10.1186/s13036-017-0078-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hanrahan G., Lu K. Application of factorial and response surface methodology in modern experimental design and optimization. Crit. Rev. Anal. Chem. 2006;36:141–151. doi: 10.1080/10408340600969478. [DOI] [Google Scholar]
- 17.Shaw R., Festing M.F.W., Peers I., Furlong L. Use of factorial designs to optimize animal experiments and reduce animal use. ILAR J. 2002;43:223–232. doi: 10.1093/ilar.43.4.223. [DOI] [PubMed] [Google Scholar]
- 18.Mandenius C.-F., Brundin A. Bioprocess optimization using design-of-experiments methodology. Biotechnol. Prog. 2008;24:1191–1203. doi: 10.1002/btpr.67. [DOI] [PubMed] [Google Scholar]
- 19.Gündoʇdu T.K., Deniz I., Çalişkan G., Şahin E.S., Azbar N. Experimental design methods for bioengineering applications. Crit. Rev. Biotechnol. 2016;36:368–388. doi: 10.3109/07388551.2014.973014. [DOI] [PubMed] [Google Scholar]
- 20.Singh V., Haque S., Niwas R., Srivastava A., Pasupuleti M., Tripathi C.K.M. Strategies for fermentation medium optimization: An in-depth review. Front. Microbiol. 2017;7:2087. doi: 10.3389/fmicb.2016.02087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Uhoraningoga A., Kinsella G.K., Henehan G.T., Ryan B.J. The goldilocks approach: A review of employing design of experiments in prokaryotic recombinant protein production. Bioengineering. 2018;5:89. doi: 10.3390/bioengineering5040089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Politis S.N., Colombo P., Colombo G., Rekkas D.M. Design of experiments (DoE) in pharmaceutical development. Drug Dev. Ind. Pharm. 2017;43:889–901. doi: 10.1080/03639045.2017.1291672. [DOI] [PubMed] [Google Scholar]
- 23.Sommeregger W., Sissolak B., Kandra K., von Stosch M., Mayer M., Striedner G. Quality by control: Towards model predictive control of mammalian cell culture bioprocesses. Biotechnol. J. 2017;12:1600546. doi: 10.1002/biot.201600546. [DOI] [PubMed] [Google Scholar]
- 24.Puskeiler R., Kreuzmann J., Schuster C., Didzus K., Bartsch N., Hakemeyer C., Schmidt H., Jacobs M., Wolf S. The way to a design space for an animal cell culture process according to Quality by Design (QbD) BMC Proc. 2011;5:P12. doi: 10.1186/1753-6561-5-S8-P12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Marasco D.M., Gao J., Griffiths K., Froggatt C., Wang T., Wei G. Development and characterization of a cell culture manufacturing process using quality by design (QbD) principles. Adv. Biochem. Eng. Biotechnol. 2014;139:93–121. doi: 10.1007/10_2013_217. [DOI] [PubMed] [Google Scholar]
- 26.Czitrom V. One-factor-at-a-time versus designed experiments. Am. Stat. 1999;53:126–131. doi: 10.1080/00031305.1999.10474445. [DOI] [Google Scholar]
- 27.Li X., Sudarsanam N., Frey D.D. Regularities in data from factorial experiments. Complexity. 2006;11:32–45. doi: 10.1002/cplx.20123. [DOI] [Google Scholar]
- 28.Collins L.M., Dziak J.J., Li R. Design of Experiments with Multiple Independent Variables: A Resource Management Perspective on Complete and Reduced Factorial Designs. Psychol. Methods. 2009;14:202–224. doi: 10.1037/a0015826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rao R.S., Kumar C.G., Prakasham R.S., Hobbs P.J. The Taguchi methodology as a statistical tool for biotechnological applications: A critical appraisal. Biotechnol. J. 2008;3:510–523. doi: 10.1002/biot.200700201. [DOI] [PubMed] [Google Scholar]
- 30.Taguchi G. System of Experimental Design: Engineering Methods to Optimize Quality and Minimize Costs. UNIPUB/Kraus International Publications; Millwood, NY, USA: 1987. [Google Scholar]
- 31.Taguchi G., Chowdhury S., Wu Y. Taguchi’s Quality Engineering Handbook. John Wiley & Sons, Inc.; Hoboken, NJ, USA: 2004. [Google Scholar]
- 32.Jones B. 21st century screening experiments: What, why, and how. Qual. Eng. 2016;28:98–106. doi: 10.1080/08982112.2015.1100462. [DOI] [Google Scholar]
- 33.Galvan D., Effting L., Cremasco H., Conte-Junior C.A. Recent Applications of Mixture Designs in Beverages, Foods, and Pharmaceutical Health: A Systematic Review and Meta-Analysis. Foods. 2021;10:1941. doi: 10.3390/foods10081941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Prudhomme W., Daley G.Q., Zandstra P., Lauffenburger D.A. Multivariate proteomic analysis of murine embryonic stem cell self-renewal versus differentation signaling. Proc. Natl. Acad. Sci. USA. 2004;101:2900–2905. doi: 10.1073/pnas.0308768101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hunt M.M., Meng G., Rancourt D.E., Gates I.D., Kallos M.S. Factorial experimental design for the culture of human embryonic stem cells as aggregates in stirred suspension bioreactors reveals the potential for interaction effects between bioprocess parameters. Tissue Eng. Part C Methods. 2014;20:76–89. doi: 10.1089/ten.tec.2013.0040. [DOI] [PubMed] [Google Scholar]
- 36.Barbosa H.S.C., Fernandes T.G., Dias T.P., Diogo M.M., Cabral J.M.S. New insights into the mechanisms of embryonic stem cell self-renewal under Hypoxia: A multifactorial analysis approach. PLoS ONE. 2012;7:e38963. doi: 10.1371/journal.pone.0038963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Marinho P.A., Chailangkarn T., Muotri A.R. Systematic optimization of human pluripotent stem cells media using Design of Experiments. Sci. Rep. 2015;5:9834. doi: 10.1038/srep09834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ratcliffe E., Hourd P., Guijarro-Leach J., Rayment E., Williams D.J., Thomas R.J. Application of response surface methodology to maximize the productivity of scalable automated human embryonic stem cell manufacture. Regen. Med. 2013;8:39–48. doi: 10.2217/rme.12.109. [DOI] [PubMed] [Google Scholar]
- 39.Badenes S.M., Fernandes T.G., Cordeiro C.S.M., Boucher S., Kuninger D., Vemuri M.C., Diogo M.M., Cabral J.M.S. Defined essential 8′ medium and vitronectin efficiently support scalable xeno-free expansion of human induced pluripotent stem cells in stirred microcarrier culture systems. PLoS ONE. 2016;11:e0151264. doi: 10.1371/journal.pone.0151264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Chang K.H., Zandstra P.W. Quantitative screening of embryonic stem cell differentiation: Endoderm formation as a model. Biotechnol. Bioeng. 2004;88:287–298. doi: 10.1002/bit.20242. [DOI] [PubMed] [Google Scholar]
- 41.Flaim C.J., Chien S., Bhatia S.N. An extracellular matrix microarray for probing cellular differentiation. Nat. Methods. 2005;2:119–125. doi: 10.1038/nmeth736. [DOI] [PubMed] [Google Scholar]
- 42.Flaim C.J., Teng D., Chien S., Bhatia S.N. Combinatorial signaling microenvironments for studying stem cell fate. Stem Cells Dev. 2008;17:29–39. doi: 10.1089/scd.2007.0085. [DOI] [PubMed] [Google Scholar]
- 43.Kumar N., Richter J., Cutts J., Bush K.T., Trujillo C., Nigam S.K., Gaasterland T., Brafman D., Willert K. Generation of an expandable intermediate mesoderm restricted progenitor cell line from human pluripotent stem cells. eLife. 2015;4:e08413. doi: 10.7554/eLife.08413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hallam D., Hilgen G., Dorgau B., Zhu L., Yu M., Bojic S., Hewitt P., Schmitt M., Uteng M., Kustermann S., et al. Human-Induced Pluripotent Stem Cells Generate Light Responsive Retinal Organoids with Variable and Nutrient-Dependent Efficiency. Stem Cells. 2018;36:1535–1551. doi: 10.1002/stem.2883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Li L., Tan D., Liu S., Jiao R., Yang X., Li F., Wu H., Huang W. Optimization of Factor Combinations for Stem Cell Differentiations on a Design-of-Experiment Microfluidic Chip. Anal. Chem. 2020;92:14228–14235. doi: 10.1021/acs.analchem.0c03488. [DOI] [PubMed] [Google Scholar]
- 46.Songstad A.E., Worthington K.S., Chirco K.R., Giacalone J.C., Whitmore S.S., Anfinson K.R., Ochoa D., Cranston C.M., Riker M.J., Neiman M., et al. Connective Tissue Growth Factor Promotes Efficient Generation of Human Induced Pluripotent Stem Cell-Derived Choroidal Endothelium. Stem Cells Transl. Med. 2017;6:1533–1546. doi: 10.1002/sctm.16-0399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yasui R., Sekine K., Yamaguchi K., Furukawa Y., Taniguchi H. Robust parameter design of human induced pluripotent stem cell differentiation protocols defines lineage-specific induction of anterior-posterior gut tube endodermal cells. Stem Cells. 2021;39:429–442. doi: 10.1002/stem.3326. [DOI] [PubMed] [Google Scholar]
- 48.Lam J., Carmichael S.T., Lowry W.E., Segura T. Hydrogel design of experiments methodology to optimize hydrogel for iPSC-NPC culture. Adv. Healthc. Mater. 2015;4:534–539. doi: 10.1002/adhm.201400410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Jung J.P., Hu D., Domian I.J., Ogle B.M. An integrated statistical model for enhanced murine cardiomyocyte differentiation via optimized engagement of 3D extracellular matrices. Sci. Rep. 2015;5:18705. doi: 10.1038/srep18705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Esteban P.P., Patel H., Veraitch F., Khalife R. Optimization of the nutritional environment for differentiation of human-induced pluripotent stem cells using design of experiments—A proof of concept. Biotechnol. Prog. 2021;37:e3143. doi: 10.1002/btpr.3143. [DOI] [PubMed] [Google Scholar]
- 51.Hu K., Yu Y. Metabolite availability as a window to view the early embryo microenvironment in vivo. Mol. Reprod. Dev. 2017;84:1027–1038. doi: 10.1002/mrd.22868. [DOI] [PubMed] [Google Scholar]
- 52.Thomas R.J., Hourd P.C., Williams D.J. Application of process quality engineering techniques to improve the understanding of the in vitro processing of stem cells for therapeutic use. J. Biotechnol. 2008;136:148–155. doi: 10.1016/j.jbiotec.2008.06.009. [DOI] [PubMed] [Google Scholar]
- 53.Jakobsen R.B., Østrup E., Zhang X., Mikkelsen T.S., Brinchmann J.E. Analysis of the effects of five factors relevant to in vitro chondrogenesis of human mesenchymal stem cells using factorial design and high throughput mRNA-profiling. PLoS ONE. 2014;9:e96615. doi: 10.1371/journal.pone.0096615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Liu C.H., Wu M.L., Hwang S.M. Optimization of serum free medium for cord blood mesenchymal stem cells. Biochem. Eng. J. 2007;33:1–9. doi: 10.1016/j.bej.2006.08.005. [DOI] [Google Scholar]
- 55.Fan X., Liu T., Liu Y., Ma X., Cui Z. Optimization of primary culture condition for mesenchymal stem cells derived from umbilical cord blood with factorial design. Biotechnol. Prog. 2009;25:499–507. doi: 10.1002/btpr.68. [DOI] [PubMed] [Google Scholar]
- 56.Decaris M.L., Leach J.K. Design of experiments approach to engineer cell-secreted matrices for directing osteogenic differentiation. Ann. Biomed. Eng. 2011;39:1174–1185. doi: 10.1007/s10439-010-0217-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kwon S.S., Kim H., Shin S.J., Lee S.Y. Optimization of tenocyte lineage-related factors from tonsil-derived mesenchymal stem cells using response surface methodology. J. Orthop. Surg. Res. 2020;15:109. doi: 10.1186/s13018-020-01623-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Petzer A.L., Zandstra P.W., Piret J.M., Eaves C.J. Differential cytokine effects on primitive (CD34+CD38-) human hematopoietic cells: Novel responses to Flt3-ligand and thrombopoietin. J. Exp. Med. 1996;183:2551–2558. doi: 10.1084/jem.183.6.2551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zandstra P.W., Conneally E., Petzer A.L., Piret J.M., Eaves C.J. Cytokine manipulation of primitive human hematopoietic cell self-renewal. Proc. Natl. Acad. Sci. USA. 1997;94:4698–4703. doi: 10.1073/pnas.94.9.4698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zandstra P.W., Conneally E., Piret J.M., Eaves C.J. Ontogeny-associated changes in the cytokine responses of primitive human haemopoietic cells. Br. J. Haematol. 1998;101:770–778. doi: 10.1046/j.1365-2141.1998.00777.x. [DOI] [PubMed] [Google Scholar]
- 61.Panuganti S., Schlinker A.C., Lindholm P.F., Papoutsakis E.T., Miller W.M. Three-stage ex vivo expansion of high-ploidy megakaryocytic cells: Toward large-scale platelet production. Tissue Eng. Part A. 2013;19:998–1014. doi: 10.1089/ten.tea.2011.0111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Yao C.L., Liu C.H., Chu I.M., Hsieh T.B., Hwang S.M. Factorial designs combined with the steepest ascent method to optimize serum-free media for ex vivo expansion of human hematopoietic progenitor cells. Enzyme Microb. Technol. 2003;33:343–352. doi: 10.1016/S0141-0229(03)00144-3. [DOI] [Google Scholar]
- 63.Yao C.L., Chu I.M., Hsieh T.B., Hwang S.M. A systematic strategy to optimize ex vivo expansion medium for human hematopoietic stem cells derived from umbilical cord blood mononuclear cells. Exp. Hematol. 2004;32:720–727. doi: 10.1016/j.exphem.2004.05.021. [DOI] [PubMed] [Google Scholar]
- 64.Case J., Rice A., Wood J., Gaudry L., Vowels M., Nordon R.E. Characterization of cytokine interactions by flow cytometry and factorial analysis. Cytometry. 2001;43:69–81. doi: 10.1002/1097-0320(20010101)43:1<69::AID-CYTO1020>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- 65.Chen T.W., Yao C.L., Chu I.M., Chuang T.L., Hsieh T.B., Hwang S.M. Large generation of megakaryocytes from serum-free expanded human CD34+ cells. Biochem. Biophys. Res. Commun. 2009;378:112–117. doi: 10.1016/j.bbrc.2008.11.019. [DOI] [PubMed] [Google Scholar]
- 66.Hsu S.C., Lu L.C., Chan K.Y., Huang C.H., Cheng S.L., Chan Y.S., Yang Y.S., Lai Y.T., Yao C.L. Large-scale production and directed induction of functional dendritic cells ex vivo from serum-free expanded human hematopoietic stem cells. Cytotherapy. 2019;21:755–768. doi: 10.1016/j.jcyt.2019.04.059. [DOI] [PubMed] [Google Scholar]
- 67.Irhimeh M.R., Fitton J.H., Ko K.H., Lowenthal R.M., Nordon R.E. Formation of an adherent hematopoietic expansion culture using fucoidan. Ann. Hematol. 2011;90:1005–1015. doi: 10.1007/s00277-011-1185-4. [DOI] [PubMed] [Google Scholar]
- 68.Andrade P.Z., Dos Santos F., Almeida-Porada G., Lobato Da Silva C., Joaquim J.M. Systematic delineation of optimal cytokine concentrations to expand hematopoietic stem/progenitor cells in co-culture with mesenchymal stem cells. Mol. Biosyst. 2010;6:1207–1215. doi: 10.1039/b922637k. [DOI] [PubMed] [Google Scholar]
- 69.Audet J., Miller C.L., Eaves C.J., Piret J.M. Common and distinct features of cytokine effects on hematopoietic stem and progenitor cells revealed by dose-response surface analysis. Biotechnol. Bioeng. 2002;80:393–404. doi: 10.1002/bit.10399. [DOI] [PubMed] [Google Scholar]
- 70.Cortin V., Garnier A., Pineault N., Lemieux R., Boyer L., Proulx C. Efficient in vitro megakaryocyte maturation using cytokine cocktails optimized by statistical experimental design. Exp. Hematol. 2005;33:1182–1191. doi: 10.1016/j.exphem.2005.06.020. [DOI] [PubMed] [Google Scholar]
- 71.Lim M., Ye H., Panoskaltsis N., Drakakis E.M., Yue X., Cass A.E.G., Radomska A., Mantalaris A. Intelligent bioprocessing for haemotopoietic cell cultures using monitoring and design of experiments. Biotechnol. Adv. 2007;25:353–368. doi: 10.1016/j.biotechadv.2007.02.002. [DOI] [PubMed] [Google Scholar]
- 72.Deshpande R.R., Wittmann C., Heinzle E. Microplates with integrated oxygen sensing for medium optimization in animal cell culture. Cytotechnology. 2004;46:1–8. doi: 10.1007/s10616-004-6401-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Mora A., Nabiswa B., Duan Y., Zhang S., Carson G., Yoon S. Early integration of Design of Experiment (DOE) and multivariate statistics identifies feeding regimens suitable for CHO cell line development and screening. Cytotechnology. 2019;71:1137–1153. doi: 10.1007/s10616-019-00350-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Sandadi S., Ensari S., Kearns B. Application of fractional factorial designs to screen active factors for antibody production by Chinese hamster ovary cells. Biotechnol. Prog. 2006;22:595–600. doi: 10.1021/bp050300q. [DOI] [PubMed] [Google Scholar]
- 75.Castro P.M.L., Hayter P.M., Ison A.P., Bull A.T. Application of a statistical design to the optimization of culture medium for recombinant interferon-gamma production by Chinese hamster ovary cells. Appl. Microbiol. Biotechnol. 1992;38:84–90. doi: 10.1007/BF00169424. [DOI] [PubMed] [Google Scholar]
- 76.Castro P.M., Ison A.P., Hayter P.M., Bull A.T. CHO cell growth and recombinant interferon-γ production: Effects of BSA, Pluronic and lipids. Cytotechnology. 1995;19:27–36. doi: 10.1007/BF00749752. [DOI] [PubMed] [Google Scholar]
- 77.Kim E.J., Kim N.S., Lee G.M. Development of a serum-free medium for the production of humanized antibody from Chinese hamster ovary cells using a statistical design. Vitr. Cell. Dev. Biol. Anim. 1998;34:757–761. doi: 10.1007/s11626-998-0029-6. [DOI] [PubMed] [Google Scholar]
- 78.Kim E.J., Kim N.S., Lee G.M. Development of a serum-free medium for dihydrofolate reductase-deficient Chinese hamster ovary cells (DG44) using a statistical design: Beneficial effect of weaning of cells. In Vitro Cell. Dev. Biol. Anim. 1999;35:178–182. doi: 10.1007/s11626-999-0024-6. [DOI] [PubMed] [Google Scholar]
- 79.Lee G.M., Kim E.J., Kim N.S., Yoon S.K., Ahn Y.H., Song J.Y. Development of a serum-free medium for the production of erythropoietin by suspension culture of recombinant Chinese hamster ovary cells using a statistical design. J. Biotechnol. 1999;69:85–93. doi: 10.1016/S0168-1656(99)00004-8. [DOI] [PubMed] [Google Scholar]
- 80.Sandadi S., Ensari S., Kearns B. Heuristic optimization of antibody production by Chinese hamster ovary cells. Biotechnol. Prog. 2005;21:1537–1542. doi: 10.1021/bp0501266. [DOI] [PubMed] [Google Scholar]
- 81.Liu C.H., Chang T.Y. Rational development of serum-free medium for Chinese hamster ovary cells. Process Biochem. 2006;41:2314–2319. doi: 10.1016/j.procbio.2006.06.008. [DOI] [Google Scholar]
- 82.Parampalli A., Eskridge K., Smith L., Meagher M.M., Mowry M.C., Subramanian A. Developement of serum-free media in CHO-DG44 cells using a central composite statistical design. Cytotechnology. 2007;54:57–68. doi: 10.1007/s10616-007-9074-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Kim S.H., Lee G.M. Development of serum-free medium supplemented with hydrolysates for the production of therapeutic antibodies in CHO cell cultures using design of experiments. Appl. Microbiol. Biotechnol. 2009;83:639–648. doi: 10.1007/s00253-009-1903-1. [DOI] [PubMed] [Google Scholar]
- 84.Zhang H., Wang H., Liu M., Zhang T., Zhang J., Wang X., Xiang W. Rational development of a serum-free medium and fed-batch process for a GS-CHO cell line expressing recombinant antibody. Cytotechnology. 2013;65:363–378. doi: 10.1007/s10616-012-9488-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Nagashima H., Watari A., Shinoda Y., Okamoto H., Takuma S. Application of a Quality by Design approach to the cell culture process of monoclonal antibody production, resulting in the establishment of a Design space. J. Pharm. Sci. 2013;102:4274–4283. doi: 10.1002/jps.23744. [DOI] [PubMed] [Google Scholar]
- 86.Liu Y., Zhang W., Deng X., Poon H.F., Liu X., Tan W.S., Zhou Y., Fan L. Chinese hamster ovary cell performance enhanced by a rational divide-and-conquer strategy for chemically defined medium development. J. Biosci. Bioeng. 2015;120:690–696. doi: 10.1016/j.jbiosc.2015.04.016. [DOI] [PubMed] [Google Scholar]
- 87.Torkashvand F., Vaziri B., Maleknia S., Heydari A., Vossoughi M., Davami F., Mahboudi F. Designed amino acid feed in improvement of production and quality targets of a therapeutic monoclonal antibody. PLoS ONE. 2015;10:e0140597. doi: 10.1371/journal.pone.0140597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Brunner M., Fricke J., Kroll P., Herwig C. Investigation of the interactions of critical scale-up parameters (pH, pO2 and pCO2) on CHO batch performance and critical quality attributes. Bioprocess Biosyst. Eng. 2017;40:251–263. doi: 10.1007/s00449-016-1693-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Mayrhofer P., Reinhart D., Castan A., Kunert R. Rapid development of clone-specific, high-performing perfusion media from established feed supplements. Biotechnol. Prog. 2020;36:e2933. doi: 10.1002/btpr.2933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Puente-Massaguer E., Badiella L., Gutiérrez-Granados S., Cervera L., Gòdia F. A statistical approach to improve compound screening in cell culture media. Eng. Life Sci. 2019;19:315–327. doi: 10.1002/elsc.201800168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Fouladiha H., Marashi S.A., Torkashvand F., Mahboudi F., Lewis N.E., Vaziri B. A metabolic network-based approach for developing feeding strategies for CHO cells to increase monoclonal antibody production. Bioprocess Biosyst. Eng. 2020;43:1381–1389. doi: 10.1007/s00449-020-02332-6. [DOI] [PubMed] [Google Scholar]
- 92.Didier C., Etcheverrigaray M., Kratje R., Goicoechea H.C. Crossed mixture design and multiple response analysis for developing complex culture media used in recombinant protein production. Chemom. Intell. Lab. Syst. 2007;86:1–9. doi: 10.1016/j.chemolab.2006.07.007. [DOI] [Google Scholar]
- 93.Rouiller Y., Périlleux A., Collet N., Jordan M., Stettler M., Broly H. A high-throughput media design approach for high performance mammalian fed-batch cultures. MAbs. 2013;5:501–511. doi: 10.4161/mabs.23942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Nie L., Gao D., Jiang H., Gou J., Li L., Hu F., Guo T., Wang H., Qu H. Development and Qualification of a Scale-Down Mammalian Cell Culture Model and Application in Design Space Development by Definitive Screening Design. AAPS PharmSciTech. 2019;20:246. doi: 10.1208/s12249-019-1451-7. [DOI] [PubMed] [Google Scholar]
- 95.Wu S., Rish A.J., Skomo A., Zhao Y., Drennen J.K., Anderson C.A. Rapid serum-free/suspension adaptation: Medium development using a definitive screening design for Chinese hamster ovary cells. Biotechnol. Prog. 2021;37:e3154. doi: 10.1002/btpr.3154. [DOI] [PubMed] [Google Scholar]
- 96.Legmann R., Benoit B., Fedechko R.W., Deppeler C.L., Srinivasan S., Robins R.H., Mccormick E.L., Ferrick D.A., Rodgers S.T., Russo A.P. A Strategy for clone selection under different production conditions. Biotechnol. Prog. 2011;27:757–765. doi: 10.1002/btpr.577. [DOI] [PubMed] [Google Scholar]
- 97.Jordan M., Voisard D., Berthoud A., Tercier L., Kleuser B., Baer G., Broly H. Cell culture medium improvement by rigorous shuffling of components using media blending. Cytotechnology. 2013;65:31–40. doi: 10.1007/s10616-012-9462-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Kuroda T., Yasuda S., Tachi S., Matsuyama S., Kusakawa S., Tano K., Miura T., Matsuyama A., Sato Y. SALL3 expression balance underlies lineage biases in human induced pluripotent stem cell differentiation. Nat. Commun. 2019;10:2175. doi: 10.1038/s41467-019-09511-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Loh K.M., Lim B. A precarious balance: Pluripotency factors as lineage specifiers. Cell Stem Cell. 2011;8:363–369. doi: 10.1016/j.stem.2011.03.013. [DOI] [PubMed] [Google Scholar]
- 100.Hoesli C.A., Johnson J.D., Piret J.M. Purified human pancreatic duct cell culture conditions defined by serum-free high-content growth factor screening. PLoS ONE. 2012;7:e33999. doi: 10.1371/journal.pone.0033999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Chen X.M., Elisia I., Kitts D.D. Defining conditions for the co-culture of Caco-2 and HT29-MTX cells using Taguchi design. J. Pharmacol. Toxicol. Methods. 2010;61:334–342. doi: 10.1016/j.vascn.2010.02.004. [DOI] [PubMed] [Google Scholar]
- 102.Jung J.P., Moyano J.V., Collier J.H. Multifactorial optimization of endothelial cell growth using modular synthetic extracellular matrices. Integr. Biol. 2011;3:185–196. doi: 10.1039/c0ib00112k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Jeon M.K., Lim J.B., Lee G.M. Development of a serum-free medium for in vitro expansion of human cytotoxic T lymphocytes using a statistical design. BMC Biotechnol. 2010;10:70. doi: 10.1186/1472-6750-10-70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Zhao A., Chen F., Ning C., Wu H., Song H., Wu Y., Chen R., Zhou K., Xu X., Lu Y., et al. Use of real-time cellular analysis and Plackett-Burman design to develop the serum-free media for PC-3 prostate cancer cells. PLoS ONE. 2017;12:e0185470. doi: 10.1371/journal.pone.0185470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Lee E., Lim Z.R., Chen H.Y., Yang B.X., Lam A.T.L., Chen A.K.L., Sivalingam J., Reuveny S., Loh Y.H., Oh S.K.W. Defined Serum-Free Medium for Bioreactor Culture of an Immortalized Human Erythroblast Cell Line. Biotechnol. J. 2018;13:e1700567. doi: 10.1002/biot.201700567. [DOI] [PubMed] [Google Scholar]
- 106.Petiot E., Fournier F., Gény C., Pinton H., Marc A. Rapid screening of serum-free media for the growth of adherent vero cells by using a small-scale and non-invasive tool. Appl. Biochem. Biotechnol. 2010;160:1600–1615. doi: 10.1007/s12010-009-8674-0. [DOI] [PubMed] [Google Scholar]
- 107.Trabelsi K., Rourou S., Loukil H., Majoul S., Kallel H. Optimization of virus yield as a strategy to improve rabies vaccine production by Vero cells in a bioreactor. J. Biotechnol. 2006;121:261–271. doi: 10.1016/j.jbiotec.2005.07.018. [DOI] [PubMed] [Google Scholar]
- 108.Gaertner J.G., Dhurjati P. Fractional Factorial Study of Hybridoma Behavior. 2. Kinetics of Nutrient Uptake and Waste Production. Biotechnol. Prog. 1993;9:309–316. doi: 10.1021/bp00021a010. [DOI] [PubMed] [Google Scholar]
- 109.Moran E.B., McGowan S.T., McGuire J.M., Frankland J.E., Oyebade I.A., Waller W., Archer L.C., Morris L.O., Pandya J., Nathan S.R., et al. A systematic approach to the validation of process control parameters for monoclonal antibody production in fed-batch culture of a murine myeloma. Biotechnol. Bioeng. 2000;69:242–255. doi: 10.1002/1097-0290(20000805)69:3<242::AID-BIT2>3.0.CO;2-I. [DOI] [PubMed] [Google Scholar]
- 110.Kallel H., Zaïri H., Rourou S., Essafi M., Barbouche R., Dellagi K., Fathallah D.M. Use of Taguchi’s methods as a basis to optimize hybridoma cell line growth and antibody production in a spinner flask. Cytotechnology. 2002;39:9–14. doi: 10.1023/A:1022437514334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Liu G., Kawaguchi H., Ogasawara T., Asawa Y., Kishimoto J., Takahashi T., Chung U.I., Yamaoka H., Asato H., Nakamura K., et al. Optimal combination of soluble factors for tissue engineering of permanent cartilage from cultured human chondrocytes. J. Biol. Chem. 2007;282:20407–20415. doi: 10.1074/jbc.M608383200. [DOI] [PubMed] [Google Scholar]
- 112.Enochson L., Brittberg M., Lindahl A. Optimization of a chondrogenic medium through the use of factorial design of experiments. Biores. Open Access. 2012;1:306–313. doi: 10.1089/biores.2012.0277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Chen Y., Bloemen V., Impens S., Moesen M., Luyten F.P., Schrooten J. Characterization and optimization of cell seeding in scaffolds by factorial design: Quality by design approach for Skeletal tissue engineering. Tissue Eng. Part C Methods. 2011;17:1211–1221. doi: 10.1089/ten.tec.2011.0092. [DOI] [PubMed] [Google Scholar]
- 114.Kuterbekov M., Machillot P., Baillet F., Jonas A.M., Glinel K., Picart C. Design of experiments to assess the effect of culture parameters on the osteogenic differentiation of human adipose stromal cells. Stem Cell Res. Ther. 2019;10:256. doi: 10.1186/s13287-019-1333-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Chen W.L.K., Likhitpanichkul M., Ho A., Simmons C.A. Integration of statistical modeling and high-content microscopy to systematically investigate cell-substrate interactions. Biomaterials. 2010;31:2489–2497. doi: 10.1016/j.biomaterials.2009.12.002. [DOI] [PubMed] [Google Scholar]
- 116.Dong J., Mandenius C.F., Lübberstedt M., Urbaniak T., Nüssler A.K.N., Knobeloch D., Gerlach J.C., Zeilinger K. Evaluation and optimization of hepatocyte culture media factors by design of experiments (DoE) methodology. Cytotechnology. 2008;57:251–261. doi: 10.1007/s10616-008-9168-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Li Q., Cheung W.H., Chow K.L., Ellis-Behnke R.G., Chau Y. Factorial analysis of adaptable properties of self-assembling peptide matrix on cellular proliferation and neuronal differentiation of pluripotent embryonic carcinoma. Nanomed. Nanotechnol. Biol. Med. 2012;8:748–756. doi: 10.1016/j.nano.2011.09.001. [DOI] [PubMed] [Google Scholar]
- 118.Pasovic L., Utheim T.P., Reppe S., Khan A.Z., Jackson C.J., Thiede B., Berg J.P., Messelt E.B., Eidet J.R. Improvement of Storage Medium for Cultured Human Retinal Pigment Epithelial Cells Using Factorial Design. Sci. Rep. 2018;8:5688. doi: 10.1038/s41598-018-24121-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Reppe S., Jackson C.J., Ringstad H., Tønseth K.A., Bakke H., Eidet J.R., Utheim T.P. High throughput screening of additives using factorial design to promote survival of stored cultured epithelial sheets. Stem Cells Int. 2018;2018:6545876. doi: 10.1155/2018/6545876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Khosravi R., Hosseini S.N., Javidanbardan A., Khatami M., Kaghazian H., Mousavi Nasab S.D. Optimization of non-detergent treatment for enveloped virus inactivation using the Taguchi design of experimental methodology (DOE) Prep. Biochem. Biotechnol. 2019;49:686–694. doi: 10.1080/10826068.2019.1599398. [DOI] [PubMed] [Google Scholar]
- 121.Kleman M.I., Oellers K., Lullau E. Optimal conditions for freezing cho-s and HEK293-EBNA cell lines: Influence of ME2SO, freeze density, and pei-mediated transfection on devitalization and growth of cells, and expression of recombinant protein. Biotechnol. Bioeng. 2008;100:911–922. doi: 10.1002/bit.21832. [DOI] [PubMed] [Google Scholar]
- 122.Kennedy M., Krouse D. Strategies for improving fermentation medium performance: A review. J. Ind. Microbiol. Biotechnol. 1999;23:456–475. doi: 10.1038/sj.jim.2900755. [DOI] [Google Scholar]
- 123.Hook A.L., Anderson D.G., Langer R., Williams P., Davies M.C., Alexander M.R. High throughput methods applied in biomaterial development and discovery. Biomaterials. 2010;31:187–198. doi: 10.1016/j.biomaterials.2009.09.037. [DOI] [PubMed] [Google Scholar]
- 124.Rankin S.A., Mccracken K.W., Luedeke D.M., Han L., Wells J.M., Shannon J.M., Zorn A.M. Timing is everything: Reiterative Wnt, BMP and RA signaling regulate developmental competence during endoderm organogenesis. Dev. Biol. 2017;434:121–132. doi: 10.1016/j.ydbio.2017.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Debevec V., Srčič S., Horvat M. Scientific, statistical, practical, and regulatory considerations in design space development. Drug Dev. Ind. Pharm. 2018;44:349–364. doi: 10.1080/03639045.2017.1409755. [DOI] [PubMed] [Google Scholar]
- 126.Papaneophytou C. Design of Experiments as a Tool for Optimization in Recombinant Protein Biotechnology: From Constructs to Crystals. Mol. Biotechnol. 2019;61:873–891. doi: 10.1007/s12033-019-00218-x. [DOI] [PubMed] [Google Scholar]
- 127.Sasamata M., Shimojo D., Fuse H., Nishi Y., Sakurai H., Nakahata T., Yamagishi Y., Sasaki-Iwaoka H. Establishment of a Robust Platform for Induced Pluripotent Stem Cell Research Using Maholo LabDroid. SLAS Technol. 2021;26:441–453. doi: 10.1177/24726303211000690. [DOI] [PubMed] [Google Scholar]
- 128.Rodriguez-Granrose D., Jones A., Loftus H., Tandeski T., Heaton W., Foley K.T., Silverman L. Design of experiment (DOE) applied to artificial neural network architecture enables rapid bioprocess improvement. Bioprocess Biosyst. Eng. 2021;44:1301–1308. doi: 10.1007/s00449-021-02529-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Zou M., Zhou Z.W., Fan L., Zhang W.J., Zhao L., Liu X.P., Wang H.B., Tan W.S. A novel method based on nonparametric regression with a Gaussian kernel algorithm identifies the critical components in CHO media and feed optimization. J. Ind. Microbiol. Biotechnol. 2020;47:63–72. doi: 10.1007/s10295-019-02248-5. [DOI] [PubMed] [Google Scholar]
- 130.Grzesik P., Warth S.C. One-Time Optimization of Advanced T Cell Culture Media Using a Machine Learning Pipeline. Front. Bioeng. Biotechnol. 2021;9:614324. doi: 10.3389/fbioe.2021.614324. [DOI] [PMC free article] [PubMed] [Google Scholar]