Abstract
Congenital microcephaly causes smaller than average head circumference relative to age, sex and ethnicity and is most usually associated with a variety of neurodevelopmental disorders. The underlying etiology is highly heterogeneous and can be either environmental or genetic. Disruption of any one of multiple biological processes, such as those underlying neurogenesis, cell cycle and division, DNA repair or transcription regulation, can result in microcephaly. This etiological heterogeneity manifests in a clinical variability and presents a major diagnostic and therapeutic challenge, leaving an unacceptably large proportion of over half of microcephaly patients without molecular diagnosis. To elucidate the clinical and genetic landscapes of congenital microcephaly, we sequenced the exomes of 191 clinically diagnosed patients with microcephaly as one of the features. We established a molecular basis for microcephaly in 71 patients (37%), and detected novel variants in five high confidence candidate genes previously unassociated with this condition. We report a large number of patients with mutations in tubulin-related genes in our cohort as well as higher incidence of pathogenic mutations in MCPH genes. Our study expands the phenotypic and genetic landscape of microcephaly, facilitating differential clinical diagnoses for disorders associated with most commonly disrupted genes in our cohort.
Keywords: molecular genetics, neurology, high-throughput nucleotide sequencing, medical genetics, human genetics
1. Introduction
Microcephaly is caused by reduced brain volume and is defined as the deviation of an individual’s occipitofrontal head circumference (OFC) by more than two standard deviations (SD) below the mean for age, sex and ethnicity. The condition is also commonly associated with neurodevelopmental disorders and frequently associated with neurological features such as intellectual disability (ID) as well as developmental delay (DD) and epilepsy [1]. Microcephaly affects approximately 2% to 3% of the population worldwide [2] with both genetic and environmental etiology. In patients with established etiology of microcephaly, the rate of cases resulting from genetic and environmental causes is close to even at 52% and 48%, respectively [2]. Microcephaly can be present at birth (primary microcephaly—PM) or develop postnatally (secondary microcephaly—SM). This distinction is useful in diagnosis and elucidation of genetic etiology [3]. PM is often a result of defects in neurogenesis, while SM is associated with progressive neurodegenerative disease [4,5]. Microcephaly can present as an isolated clinical symptom but more frequently as a phenotypic feature of a variety of genetic syndromes. As of March 2021 the clinical feature “microcephaly” returns nearly 1600 entries in the Online Mendelian Inheritance in Man (OMIM) database [6]. The genetic and clinical heterogeneity of this condition therefore constitutes a major diagnostic and consequently therapeutic challenge. In order to properly assess the underlying cause of microcephaly, patients require thorough physical examination, analysis of medical records with attention to maternal disease and infections during pregnancy as well as adverse events during labor and diagnostic investigations including brain imaging, biochemical tests and chromosomal and genetic analyses. These steps help discern whether the basis for microcephaly in the patient is fundamentally environmental, genetic or multifactorial, which in turn can help determine further diagnostic and treatment options.
Exome sequencing (ES) has proved highly effective in identifying genetic causes for a range of Mendelian disorders, with a diagnostic rate of up to 50% [7]. The number of genes associated with microcephaly has increased over the last few years, primarily due to broad application of ES for research and diagnostic purposes. For example, the number of known genes causing autosomal recessive primary microcephaly (MCPH) has more than doubled from 12 to 28 in 5 years ([8,9], OMIM Phenotypic series: PS251200). Diagnosis using ES does not require prior knowledge of specific genes responsible for clinical phenotype. It is particularly advantageous for accurately and rapidly diagnosing genetically heterogeneous conditions with microcephaly as one of the features that can be caused by mutation in one of several hundred genes. In addition to better understanding the etiology of this specific disorder, identification of novel genes, the mutations of which cause microcephaly, can help elucidate biological mechanisms crucial to brain development and associated diseases.
Three previous studies have used ES to investigate the genetic basis of microcephaly in cohorts of patients presenting with this disorder [10,11,12]. Despite good progress in unraveling the genetic etiology of the condition, a large portion of over 50% of patients presenting with microcephaly remain without specific molecular diagnosis, making the most effective therapeutic measures for the patient difficult to establish. In this study, we carried out ES on 191 Polish patients clinically diagnosed with microcephaly. We established molecular diagnosis where possible and identified 12 previously unassociated potential disease genes. Our work further adds to mounting evidence that ES is the diagnostic method of choice in patients presenting with microcephaly.
2. Materials and Methods
We performed ES for a cohort of 191 Polish patients, including 3 sib-pairs, with clinically diagnosed microcephaly. Patients were referred between 2017 and 2019 from different medical centers in Poland and were given comprehensive clinical evaluation in specialized genetic and neurology clinics. Patients enrolled in our project had OFC <−2 SD below average for age and sex and negative history of environmental exposures. All patients underwent brain imaging studies to detect potential brain malformations. Out of 191 patients, 149 of them had either karyotyping or array comparative genomic hybridization analysis (aCGH) performed prior to ES. Informed consent was obtained from participating families, and the study protocol was approved by the Ethics Committee of the Institute of Mother and Child in Warsaw.
Whole-exome sequencing was performed by CeGaT GmbH, Germany. Sequencing libraries were prepared using Agilent SureSelect All Exon V6 sample preparation kits and carried out on Illumina NovaSeq 6000 sequencer, via 2 × 100 bp reads. Genomic data processing was based on an in-house developed pipeline, consisting of read alignment to a hg38 reference genome using Burrows-Wheeler Aligner software (BWA v0.7.16) [13], duplicated reads removal with Picard (v2.11.0) and variant calling with Genome Analysis Toolkit HaplotypeCaller (GATK v4.0b4) [14] through bcbio-nextgen toolkit (v.1.0.5) [15]. Variant annotation was carried out with Ensembl Variant Effect Predictor (v96) [16] using resources from the ClinVar clinical database [17], gnomAD v3.0 population database [18] and dbNSFP v4.1 functional predictions database [19]. Copy number variant (CNV) analysis was carried out with CoNIFER (v0.2.2) [20]. ES quality control was caried out with the Mosdepth (v0.2.4) [21], FastQC (v0.11.5), Bcftools (v1.5) [22], QualiMap (v2.2.2a) [23], Samtools (v1.5) [22] and MultiQC (v1.8) [24].
In this study we used a mixed approach. We first screened probands only, using ES and an in silico microcephaly gene panel to find patients with pathogenic or likely pathogenic variants in known genes associated with the disorder. Then, for selected negative cases both parents underwent ES to discover causative mutations in previously unassociated genes that might lead to microcephaly.
The in silico gene panel used to screen patients for variants in genes associated with microcephaly consisted of over 800 genes. These genes were manually selected from several gene panels customized for patients with microcephaly, and additional genes were selected from various databases such as OMIM or DECIPHER. The detailed gene list with details regarding coverage across all samples is included in Table S2. Selected variants from ES were confirmed by Sanger sequencing using an ABI AB3730 capillary sequencer. Detailed information regarding genes and exon coverage and coverage thresholds for each sample, based on RefSeq genes from NCBI, is available in Table S3. Sequencing quality control data from mentioned tools are available as a MultiQC report in File S4.
Molecular diagnosis for patients was established according to the following classification: (I) definitive diagnosis—the variant found is located in a gene with established association with microcephaly, zygosity and inheritance pattern in the family is in accordance with the underlying disease and the variant is reported in the ClinVar database as a pathogenic or likely pathogenic variant; (II) likely diagnosis—the variant found is located in a gene with established association with microcephaly, zygosity and inheritance pattern in the family is in accordance with the underlying disease and the variant is not reported in the ClinVar database but is predicted to be pathogenic/likely pathogenic by ACMG classification; (III) uncertain diagnosis—the variant is located in a gene with established association with microcephaly, unreported in the ClinVar database (or is reported as a variant of uncertain significance (VUS)) and is predicted to be VUS by the American College of Medical Genetics and Genomics (ACMG) classification; (IV) novel—the variant is located in a gene with no established association with microcephaly, but zygosity and inheritance pattern in the family indicate a possible pathogenic nature and published data suggest that it may interfere with processes relevant to microcephaly etiopathogenesis.
3. Results
3.1. Clinical Characteristics of the Cohort
We investigated 191 unrelated subjects with microcephaly of suspected genetic etiology, including 97 females and 94 males with average age of 5 years at last clinical evaluation (median: 3.2 years, range: 0 to 18 years). All investigated subjects were determined to have exclusively European descent, with no known consanguinity between parents. PM was suspected in 77 patients (40%) and SM in 110 patients (58%). For the remaining four patients (2%) the time of onset of microcephaly could not be determined. Three subjects (3/191) reported a family history of microcephaly with an affected sibling presenting with similar phenotype and clinically unaffected parents. Many patients presented with additional clinical findings including cognitive impairment (both ID and DD), abnormal muscle tone, epilepsy, dysmorphic facial features and short stature (Table 1). The most frequent findings on brain images were corpus callosum abnormalities, gyrification alterations, delayed myelination and cerebellum abnormalities.
Table 1.
Major clinical features and their frequency in patients enrolled in this study.
| Clinical Feature | HPO Number | Frequency |
|---|---|---|
| Microcephaly | HP:0000252 | 191 (100%) |
| Primary microcephaly | HP:0011451 | 77 (40.3%) |
| Secondary microcephaly | HP:0005484 | 110 (57.6%) |
| Unknown onset | HP:0000252 | 4 (2.1%) |
| Cognitive impairment (DD/ID) | HP:0100543 | 168 (87.9%) |
| Abnormal cerebral morphology | HP:0002060 | 133 (69.6%) |
| Abnormal corpus callosum morphology | HP:0001273 | 55 (28.8%) |
| Abnormal myelination | HP:0012447 | 35 (18.3%) |
| Abnormal cortical gyration | HP:0002536 | 28 (14.7%) |
| Abnormal cerebellum morphology | HP:0001317 | 28 (14.7%) |
| Ventriculomegaly | HP:0002119 | 22 (11.5%) |
| Abnormality of the nervous system | HP:0000707 | 166 (86.9%) |
| Abnormal muscle tone | HP:0003808 | 99 (51.8%) |
| Seizure | HP:0001250 | 76 (39.8%) |
| Refractory status epilepticus | HP:0032867 | 25 (13.1%) |
| Epileptic encephalopathy | HP:0200134 | 10 (5.2%) |
| Hemiplegia/hemiparesis or tetraplegia/tetraparesis | HP:0004374 HP:0030182 |
22 (11.5%) |
| Abnormality of movement | HP:0100022 | 17 (8.9%) |
| Stereotypy | HP:0000733 | 13 (6.8%) |
| Short stature | HP:0004322 | 61 (31.9%) |
| Abnormal facial shape | HP:0001999 | 75 (39.3%) |
| Strabismus | HP:0000486 | 27 (14.1%) |
| Abnormal heart morphology | HP:0001627 | 19 (9.9%) |
| Hearing impairment | HP:0000365 | 13 (6.8%) |
Of 191 total cases, definitive molecular diagnosis of microcephaly was made in 36/191 (19%) cases and a likely diagnosis in a further 35/191 (18%), while in another 11/191 (6%) cases we found variants of unknown significance in genes previously associated with microcephaly. Taken together they give a total of 82/191 cases, or a 43% diagnostic rate. We further found potential novel variants in previously unassociated candidate genes in 11/191 (6%) cases. For the remaining 98/191 (51%) cases we were not able to find causative or potential variants. In 4/191 (2.5%) cases we found more than one likely pathogenic variant in distinct genes linked to microcephaly. Altogether, we identified 132 (of which 115 were unique) variants affecting 65 genes in 93 patients (Table S1). Of the 132 variants, 88 (67%) were missense, 19 (14%) frameshift, 13 (10%) nonsense, 7 (5%) canonical splice site, 4 (3%) located within small nuclear RNA gene and 1 (1%) inframe duplication. Sixty-three variants were of de novo origin, and 72 were not previously reported in the ClinVar database. Following ACMG guidelines [25] 65 of the identified 132 variants (49%) were classified as pathogenic, 29 (22%) as likely pathogenic, another 29 (22%) as VUS and the remaining 9 (7%) variants as likely benign. In total we observed autosomal dominant inheritance in 42/78 (54%) cases, autosomal recessive in 26/78 (33%) cases, X-linked dominant in 7/78 (9%) cases and X-linked recessive in 3/78 (4%) cases. For the remaining 15 cases inheritance was either unknown, as in the case of variants in novel genes (11 cases), or the patient had more than one possible molecular diagnosis (four cases). Detailed information about patients’ phenotype as well as genetic findings are in Table S1. Taken together we found pathogenic/likely pathogenic variants or VUS in 54 different genes associated with microcephaly, as well as novel variants in 12 previously unassociated genes. Results of CNV analysis with Conifer software were examined for occurrence of heterozygous variants in dominant genes as well as homozygous variants in recessive genes linked with microcephaly. In patients with known pathogenic heterozygous variants in genes with recessive inheritance mode, we also looked at Conifer data in search for possible CNVs in the gene of interest; however, we did not find such cases in our cohort.
3.2. Definitive and Likely Diagnosis Cases
In our cohort, we found variants in a larger number of genes encoding tubulins previously linked with microcephaly. Two of these genes in particular, TUBA1A (five patients) and TUBB3 (five patients), showed the highest number of pathogenic mutations. Out of 71 patients with definitive or likely diagnosis, 15 (21%) overall had a causative variant in a tubulin gene. Aside from tubulins, other frequently mutated genes in our cohort are CTNNB1, FOXG1 and TSEN54, each identified in four out of 71 cases (6%), and ASPM, EFTUD2 and PDHA1, each in three out of 71 patients (4%). For conditions associated with the CTNNB1, FOXG1, EFTUD2 and PDHA1 genes, mutations are normally inherited in a dominant manner, and all our patients were found with de novo heterozygous variants. All patients with TSEN54 mutations, which cause pontocerebellar hypoplasia type 2A (PCH2A), an autosomal recessive disorder, shared the same variant NM_207346.3: c.919G > T: p.(Ala307Ser) in homozygous state inherited from carrier parents. This variant is over-represented in the European population (excluding Finnish) with a frequency of 1.7 × 10−3, in comparison to 2 × 10−4 in other populations, according to gnomAD v3 database. All PCH2A patients we report here have progressive microcephaly with average SD at birth nearing 0 and −4.9 at the time of last examination (1 year 9 months average) as well as cerebellum and/or brainstem hypoplasia, DD, hypertonia and epilepsy.
Overall we found pathogenic/likely pathogenic variants in 45 unique genes related to microcephaly, of which 27 have autosomal dominant inheritance, 12 autosomal recessive inheritance, 4 X-linked dominant inheritance and 1 each X-linked recessive and X-linked inheritance. In these genes we identified 54 missense, 16 frameshift, 13 nonsense, 7 canonical splice site and 4 variants located within the small nuclear RNA gene.
3.3. Dual Molecular Diagnosis
Among 71/191 cases with definitive or likely diagnosis, we found dual molecular diagnoses in four patients (Table 2).
Table 2.
Comparison of phenotype of patients with possible dual molecular diagnosis and most common clinical features of patients with mutations in those genes.
| Patient | Sex | Phenotype | Variant and Inheritance | Zygosity | OMIM Syndrome | OMIM ID | Syndrome Main Features | Pubmed |
|---|---|---|---|---|---|---|---|---|
| T50 | M | SM, ID, Dandy-Walker malformation, anterior commissure agenesis, dysmorphic facial features | TUBB2B-NM_178012.5 c.1171C>T p.(Arg391Cys) dn | het | Cortical dysplasia, complex, with other brain malformations 7 | 610031 | DD, polymicrogyria, corpus callosum agenesis, brainstem hypoplasia | [26] |
| HUWE1-NM_031407.7 c.11434G>A p.(Val3812Met) mat | hemi | Mental retardation, X-linked syndromic, Turner type | 309590 | DD, ID, hypotonia, speech delay, microcephaly, epilepsy, dysmorphic facial features | [27] | |||
| S78 | M | PM, DD, axial hypotonia, epileptic encephalopathy, limb hypertonia, EEG abnormalities | MCPH1-NM_024596.5 c.664T>C p.(Cys222Arg) | hom | Microcephaly 1, primary, autosomal recessive | 251200 | ID, microcephaly, short stature | [28] |
| SCN8A-NM_014191.4 c.5630A>G p.(Asn1877Ser) dn | het | Epileptic encephalopathy, early infantile, 13 | 614558 | DD, epilepsy, myoclonus, extrapyramidal signs | [29] | |||
| S177 | F | SM, DD, hypotonia, dysmorphic facial features | HUWE1-NM_031407.7 c.9208C>T p.(Arg3070Cys) dn | het | Mental retardation, X-linked syndromic, Turner type | 309590 | DD, ID, hypotonia, speech delay, microcephaly, epilepsy, dysmorphic facial features | [27] |
| SATB2-NM_015265.4 c.490G>A p.(Asp164Asn) dn | het | Glass syndrome | 612313 | DD, speech delay, dental anomalies, behavioural difficulties, feeding issues, abnormal brain neuroimaging, dysmorphic facial features | [30] | |||
| S188 | M | PM, DD, bilateral polymicrogyria, epilepsy, hypertonia, tetraplegia, nystagmus, strabismus convergent, cryptorchidism | KCNT1-NM_020822.3 c.1720G>A p.(Glu574Lys) dn | het | Epileptic encephalopathy, early infantile, 14 | 614959 | DD, epilepsy, absent speech, hypotonia, spasticity, microcephaly | [31] |
| GRIN1-NM_007327.4 c.1665G>T p.(Met555Ile) dn | het | Neurodevelopmental disorder with or without hyperkinetic movements and seizures, autosomal dominant | 614254 | DD, epilepsy, hypotonia, absent speech, movement disorders, spasticity, visual impairment, bilateral polymicrogyria | [32] |
PM—primary microcephaly, SM—secondary microcephaly, DD—developmental delay, ID—intellectual disability, dn—de novo, mat—maternally inherited, het—heterozygous, hemi—hemizygous, hom—homozygous.
In patient T50 with SM (−3.1 SD), DD, ID, radiological findings of Dandy–Walker malformation and anterior commissure agenesis as well as dysmorphic facial features, we found a heterozygous de novo variant NM_178012.5: c.1171C>T: p.(Arg391Cys) in TUBB2B gene and a hemizygous, maternally inherited variant NM_031407.7: c.11434G>A: p.(Val3812Met) in HUWE1. Neither are reported in the ClinVar database. The former mutation causes amino acid substitution and has high deleterious scores in 20/20 prediction algorithms, as well as pathogenic ACMG classification. In addition, a neighboring variant causing the amino acid change p.Arg391His is classified as likely pathogenic in the ClinVar database. The latter mutation was previously classified as VUS under ACMG guidelines. In patient S177 presenting with SM (−3.2 SD), DD, hypotonia and dysmorphic facial features, we identified a heterozygous de novo variant NM_031407.7: c.9208C>T: p.(Arg3070Cys) in HUWE1 and another de novo heterozygous variant NM_015265.4: c.490G>A: p.(Asp164Asn) in the SATB2 gene. While the HUWE1 variant is reported in the ClinVar database as pathogenic/likely pathogenic, the SATB2 variant was not previously reported though is likely pathogenic according to ACMG classification. In patient S188 with PM (−3.9 SD), bilateral polymicrogyria, DD, epilepsy, spastic tetraplegia, nystagmus, convergent strabismus and cryptorchidism we found two heterozygous de novo variants in two genes encoding ion channel subunits: KCNT1-NM_020822.3: c.1720G>A: p.(Glu574Lys) and GRIN1-NM_007327.4: c.1665G>T: p.(Met555Ile). Both variants have pathogenic classification according to ACMG and are not reported in the ClinVar database. Patient S78 presented clinically with PM (−3.7 SD), DD, epileptic encephalopathy, axial hypotonia, limb hypertonia and EEG abnormalities and has a homozygous mutation NM_024596.5: c.664T>C: p.(Cys222Arg) in the MCPH1 gene inherited from carrier parents and a heterozygous de novo variant NM_014191.4: c.5630A>G: p.(Asn1877Ser) in the SCN8A gene. Both variants are reported in the ClinVar database with conflicting interpretations of pathogenicity, where besides submissions classifying both variants as VUS, the MPCH1 variant has one additional likely benign submission, while SCN8A has four pathogenic and two likely pathogenic submissions. Further, under the ACMG guidelines the MPCH1 variant is classified as likely benign, while SCN8A is classified as pathogenic.
3.4. Novel Gene Associations
We identified variants in 12 genes not previously associated with human diseases. To assess possible status as novel candidate genes, mutations of which may cause microcephaly in our patients, we grouped the genes into two categories, high or low confidence, based on the level of evidence. Results of analysis of the high confidence candidates are presented below; Table S1 shows information on the low confidence candidates.
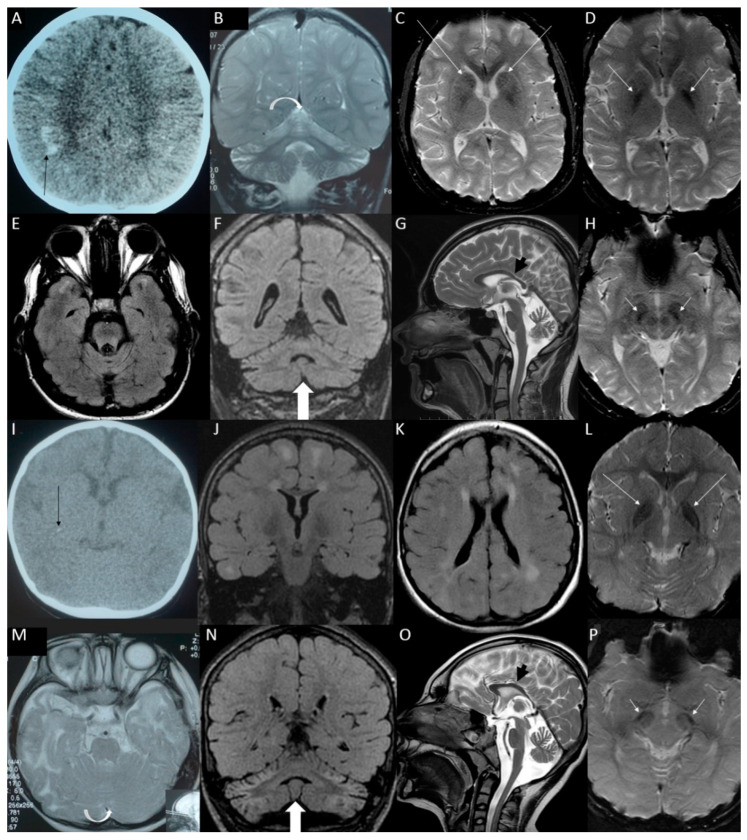
SUPV3L1: In a pair of siblings (S19a, S19b) with similar phenotype consisting of SM (−3.1 and −4.3 SD, respectively), delayed myelination, white matter abnormalities, profound DD, epilepsy and axial hypotonia, we identified a homozygous variant NM_003171.4: c.1093C>T: p.(Arg365Trp) inherited from healthy carrier parents. SUPV3L1 gene encodes an RNA/DNA helicase, and the identified variant is reported in the gnomAD v3 population database with very low frequency (1.7 × 10−5) and no homozygous occurrences; 11/21 predictive algorithms classify it as damaging. Figure 1 contains detailed descriptions of brain imaging findings for both siblings.
Figure 1.
Brain imaging of sibling with SUPV3L1 homozygous variant c.1093C>T:p.(Arg365Trp)—the older brother (patient S19a) in panels. (A) Computed tomography (CT) at the early childhood with marked subcortical calcifications in both frontal and parietal lobes (black arrow). (B) Subsequent magnetic resonance imaging (MRI) scans at the middle childhood with marked shrunken bright cerebellum (white arrow). (C–H) Brain MRI at the late adolescence. (E) FLAIR hyperintensities remained in the temporal poles only, (F) atrophic cerebellum but not so FLAIR-hyperintense as earlier and as in the younger sister (white arrow), (G) the corpus callosum is shorter and thinner than normal (black arrow), (D) iron or calcium deposits in the globi pallidi (white arrows), (H) in substantia nigra (white arrows) (C) and in the caudate nuclei (white arrows); brain imaging of sibling with SUPV3L1 homozygous variant c.1093C>T:p.(Arg365Trp)—the younger sister (patient S19b): (I) CT at the early childhood revealed two punctate subcortical calcifications in the right cerebral hemisphere (black arrow). (M) MRI taken as the toddler with normal cerebellum with slight widening of the cerebellar sulci (white arrow). (J–P) MRI at the early adolescence: (J,K) myelination has progressed, but there are white matter hyperintensities on FLAIR sequence in both cerebral hemispheres, (N) shrunken bright cerebellum (white arrow), (O) shortened and thinned corpus callosum (black arrow), (L) iron or calcium deposits in the globi pallidi (white arrows) (P) and in substantia nigra (white arrows).
DHX9: In patient T91 with SM (−2.8 SD), corpus callosum dysgenesis, DD, moderate ID, ataxia, hypotonia, convergent strabismus and dysmorphic facial features we identified a de novo variant NM_001357.5: c.3497G>C: p.(Arg1166Pro) in the DHX9 gene encoding the DNA and RNA helicase. It is classified by 15/20 in silico predictions as damaging and is not reported in the gnomAD v3 database.
MSI1: In patient T20 with PM (−4.9 SD), mild ID, hypotonia, dysmorphic facial features and syndactyly trio analysis revealed the presence of two de novo variants, in CBLC and MSI1 genes. One is a missense variant NM_012116.3: c.806G>T: p.(Arg269Leu) in the CBLC gene which encodes a member of Cbl family of E3 ubiquitin ligases. The variant found is not reported in the gnomAD v3 database and is classified by 16/21 predictive algorithms as damaging. The MSI1 gene encodes a protein containing two conserved tandem RNA recognition motifs in which we identified a frameshift variant NM_002442.3: c.594dup: p.(Arg199GlufsTer180). It is not reported in the gnomAD v3 database, and although there is no record of this variant in the dbNSFP functional prediction database, it is classified as pathogenic by ACMG guidelines.
ELFN1 and CCDC112: Using trio analysis we identified two de novo missense variants in the ELFN1 and CCDC112 genes in patient T62 with SM (−3.6), delayed myelination, asymmetric cerebral hemispheres, epileptic encephalopathy, hypotonia, sensorineural hearing loss, myopia and astigmatism. Variant NM_001128636.3: c.946C>T: p.(Arg316Cys) in ELFN1 gene encoding postsynaptic protein that regulates circuit dynamics in the central nervous system is classified as damaging by 3/14 prediction algorithms and is not reported in the gnomAD v3 database. We also identified the variant NM_152549.2: c.59A>G: p.(His20Arg) in CCDC112 gene encoding a coiled-coil domain containing protein. It is not reported in the gnomAD v3 population database and is classified as damaging by 1/20 in silico algorithms.
4. Discussion
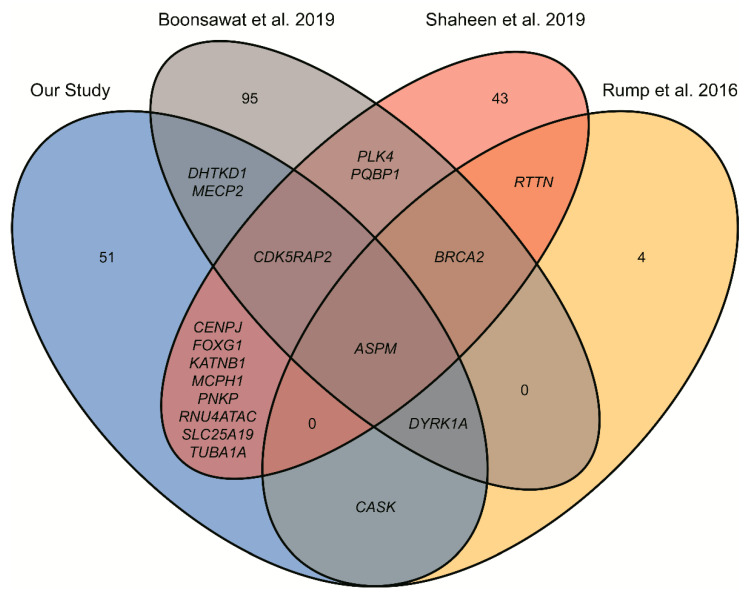
In our study we examined the full exomes of 191 Polish patients with clinically diagnosed microcephaly using routine exome sequencing technology. We identified 12 novel candidate disease genes and established a definitive molecular diagnosis in 19% of the patients, likely diagnosis for 18% of patients and found variants of unknown significance in genes associated with microcephaly in 6% of our patients. In aggregate, it gives a diagnostic yield of 43%, falling within the range reported for ES [7] and similar to results in previous studies regarding incidence of microcephaly [10,11] (29% and 48%, respectively). Comparison of these studies (Figure 2) reflects the high genetic heterogeneity of microcephaly, with the highest number of overlapping gene identifications at 8, between our study and that of Shaheen et al. (Figure 2). However, the only gene found in all four studies is ASPM, which could be explained by the fact that it is the most frequent gene mutated in patients with MCPH [8].
Figure 2.
Venn diagram showing number and symbols of unique and shared genes identified in four studies on different microcephaly patients.
Patients with mutations in tubulin genes, the largest group with 15 cases in our cohort, demonstrated malformations of cortical development, with a wide range of disorders, including microcephaly, abnormal gyral patterns of cerebral cortex such as lissencephaly or polymicrogyria as well as presence of heterotopic grey matter. The high incidence of pathogenic or likely pathogenic variants in tubulin genes in our cohort underscores the relevance of microtubules and microtubule-associated proteins in brain development and the likelihood of microcephaly in the case of their disruption. However, this is the first study of microcephalic patients in which a high percentage of over 20% of cases result from mutations in tubulin genes.
Our study allows for the further delineation of phenotype of microcephalic patients, especially in the case of several genes in which mutations were identified in more than one patient. Our findings seem to confirm the existence of sex bias in the neurodevelopmental disorder with spastic diplegia and visual defects caused by variants in CTNNB1, as all our patients are females [33]. With recent reports of eight additional patients, five of whom were male, and four patients from our study the sex ratio in this disorder is approximately 63% females to 37% males [34,35]. In one case (S87) we identified a frameshift variant NM_001904.4: c.1665del: p.(Thr556HisfsTer14) that had been reported only once in the literature, while the other three (S42, S63, S145) were harboring more common pathogenic variants already reported in the ClinVar database. While all our patients with mutations in the CTNNB1 gene had a similar phenotype with microcephaly (−2.3 to −4.3 SD), DD and abnormal muscle tone, patient S87 presented with uncommon finding of delayed myelination as evidenced on brain MRI studies. Three patients were found with strabismus, which supports the observation that ocular abnormalities are frequent findings in patients with CTNNB1-associated neurodevelopmental disorder [34].
Patients with congenital Rett syndrome due to pathogenic variants in FOXG1 presented with typical clinical features such as progressive microcephaly, DD, corpus callosum anomalies, intellectual disability, epilepsy and strabismus but also rare findings including nystagmus (case S8 and S106) and previously unreported hypoplastic cranial nerves I and II (S8) [36].
Mandibulofacial dysostosis with microcephaly caused by heterozygous variants in EFTUD2 gene is characterized by the presence of progressive microcephaly, dysmorphic features and hearing loss. Most of these symptoms were present in our patients (S39, S79, S80); however, patient S80 was found with a congenital heart defect that was not previously associated with EFTUD2 defect. [37]. PDHA1 mutations cause pyruvate dehydrogenase E1-α deficiency, a rare X-linked syndrome in which a high proportion of females with heterozygous variants manifest severe symptoms. Our patients (S37, S46, S89) with mutations in this gene were all females and displayed neurologic manifestations of this syndrome, with structural abnormalities in the central nervous system, mainly in the corpus callosum as well as DD [38]. Patients with pathogenic variants in the PDHA1 gene usually develop microcephaly postnatally; however, in the case of patient S89, lower OFC was observed at birth, and the abnormalities of the brain were more severe when compared to the other two patients. This could be the result of the frameshift variant causing a premature stop codon that usually results in absent protein, whereas the other two patients had missense mutations, which are linked with less severe phenotype [39]. In contrast to those disorders with dominant inheritance pattern, bi-allelic mutations in ASPM cause autosomal recessive primary microcephaly 5, a rare syndrome belonging to the MCPH group. Our patients presented with OFC reduction without other congenital abnormalities, except for patient S49 with left side hemiplegia. Cortical malformations are quite a rare clinical finding in this condition; however, brain MRI revealed right brain hemisphere polymicrogyria in the previously mentioned patient and pachygyria in patient S52 [40]. All patients had compound heterozygous loss-of-function (LoF) mutations, as was expected considering the non-consanguineous cohort, and two patients (S49, S52) had a pathogenic variant NM_018136.5: c.7782_7783del: p.(Lys2595SerfsTer6) common in the European population [41].
Patients with variants in genes responsible for MCPH accounted for seven out of 71 resolved cases (9.9%). This frequency is almost two times higher than the prevalence (5.4% and 5.7%) reported in other cohort studies of microcephalic patients [10,11]. We demonstrated higher incidence of cases with mutations in MCPH genes which is surprising considering lack of reported consanguinity in our cohort, especially having taken into account the number of cases caused by autosomal recessive conditions, which constitute about a third (32.9%) of total resolved cases with about 70% of them caused by compound heterozygous variants. A similar number was reported in a study of 62 unrelated patients with microcephaly, where 37.5% of patients had syndrome with autosomal recessive inheritance pattern [11]. In contrast, a study with 35 patients of whom six were from consanguineous families reported this pattern of inheritance in 70% of cases [10]. We argue that in the event of no reported consanguinity or similarly affected siblings, dominant inheritance pattern should be more common in patients with microcephaly.
In our study we found four cases with more than one possible molecular cause that could lead to microcephaly, a phenomenon described as a dual molecular diagnosis and reported in approximately 5% cases with molecular diagnoses [42]. Table 2 presents the patients’ phenotypes and molecular findings compared with the most common clinical features reported in the literature found in patients with pathogenic variants in these genes. In each case, due to similar clinical features caused by mutations in distinct microcephaly genes, we could not exclude the possibility of overlapping phenotype. We identified a heterozygous variant in TUBB2B and a hemizygous variant in HUWE1 in patient T50. Both genes cause syndromes with highly variable phenotype, especially in the case of HUWE1. Considering the X-linked inheritance pattern of this gene and the fact that this variant was inherited from a healthy mother, establishing a definitive molecular cause based solely on phenotype features is difficult. In a female patient S177 we identified a heterozygous de novo mutation in HUWE1 and another de novo heterozygous variant in SATB2 gene. In the case of previously mentioned HUWE1, the disruptions of which affect mainly males, recent studies report incidents of severely affected females, all of whom had de novo heterozygous variants [27]. On the other hand heterozygous variants in SATB2 can lead to Glass syndrome, which is characterized by ID, dysmorphic facial features, cleft palate and crowded teeth, with variable clinical expression. Considering the phenotype of this patient, the de novo character of both mutations, as well as the high pathogenicity scores for SATB2 variant, we cannot exclude the possibility of overlapping phenotype. In patient S188 we again identified two de novo heterozygous mutations in GRIN1 and KNCT1 genes. Disruption of these genes can lead to severe DD and seizures, both of which are present in this case. In the case of GRIN1 both dominant and recessive inheritance pattern is reported, with causative heterozygous missense substitutions of de novo origin usually found in functional domains of the protein and resulting in LoF [43]. The variant c.1665G>T found in our patient is located between the ligand binding and transmembrane domains and was previously reported in a large Chinese cohort of patients, classified as likely pathogenic although without any information about patients’ clinical picture [44]. Considering similar phenotypes resulting from mutations of those genes we cannot denote a causal variant or rule out a possible dual molecular cause of phenotype present in our patient. Finally patient S78 with a homozygous variant in MCPH1 inherited from both parents also had a pathogenic heterozygous variant in the SCN8A gene. Considering the phenotype of the patient, and likely benign reports regarding the variant identified in MCPH1, it is more probable that the mutation in SCN8A is the main cause of the disorder. However, without functional studies regarding the variant c.664T>C in MPCH1 gene we cannot exclude a possible additive effect of this variant on the clinical picture of this patient.
This study identified novel variants in 12 genes, of which 11 were previously unrelated to microcephaly. The five high confidence candidate genes identified in our cohort are SUPV3L1, DHX9, MSI1, ELFN1 and CCDC112. (Low confidence candidates and their possible link to microcephaly are included in Table S1). In siblings S19a and S19b we identified a homozygous variant in the SUPV3L1 gene that encodes NTP-dependent RNA/DNA helicase, localized mainly in the mitochondrial matrix and in a smaller fraction in the nucleus [45,46]. In yeast, Suv3 homolog interacts with various proteins implicated in DNA replication, recombination and repair, as well as chromatin repair and genome stability [47]. In human cell lines SUPV3L1 interacts with BLM and WRN proteins, involved in genome repair via homologous recombination [48]. Pathogenic variants in BLM cause Bloom syndrome, one of the clinical features of which is microcephaly [49]. Functional studies using knock-out of the SUPV3L1 orthologue in mouse and zebrafish mutants showed embryonic lethality [50,51]; knock-down in mammalian cell lines results in apoptosis [46]. The amino acid substitution p.Arg365Trp localizes in the helicase C-terminal domain, which may impair function.
For patient T91, trio-analyses identified a heterozygous de novo variant in the DHX9 gene. This encodes a helicase that unwinds both DNA and RNA and is important in fundamental processes such as DNA replication, transcriptional activation, post-transcriptional RNA regulation, mRNA translation and RNA-mediated gene silencing [52,53]. Loss of DHX9 leads to an increase in circular-RNA-producing genes [54]. It also promotes formation of R-loops in cells with impaired splicing factors that can block DNA replication [55]. DHX9 is also implicated in regulation of DNA transcription, translation, RNA processing and transport and maintenance of genome stability and is implicated in the development of many cancer types [56]. The initial data on SUPV3L1 and DHX9 may suggest implication of the aforementioned genes in normal brain development.
In two patients we found de novo variants in two distinct genes previously unassociated with microcephaly. In patient T20 we identified a heterozygous frameshift variant resulting in the introduction of a new stop codon downstream of the original stop in MSI1 and a heterozygous amino acid substitution variant in the CBLC gene. Lack of expression in the brain and no obvious metabolic function of the latter gene raises questions regarding the possibility of it being a possible cause of microcephaly. The MSI1 gene on the other hand encodes an RNA binding protein, which can inhibit translation initiation, stabilize RNA and influence alternative splicing [57,58,59]. Many mRNA targets of the mouse homolog Msi1 are involved in key cellular processes such as cell cycle, proliferation and survival [60]. The human protein MSI1 binds to and is a translational activator of MCPH1, a gene associated with MCPH which has various role in the cell cycle. In particular, it acts as G2/M checkpoint arrest via maintenance of inhibitory phosphorylation of cyclin-dependent kinase 1 [61]. Additionally MSI1 interacts with the Zika virus genome and enables viral replication [62]. Two siblings from a consanguineous family presenting with phenotype similar to MPCH had a homozygous missense variant in MSI1, as well as other homozygous variants in ACACB, DKK4 and DTX3L genes [62]. Although our patient presents with a heterozygous variant, the unknown pattern of inheritance of the MSI1 gene and resemblance of patient clinical features to MCPH were observed. It is therefore conceivable that heterozygous LoF variants in this gene cause microcephaly.
In patient T62 we found a de novo heterozygous variant in CCDC112, and another de novo heterozygous variant in ELFN1. CCDC112 is a centriolar satellite protein and co-localizes with PCM1 which is a component of centriolar satellites and is essential for anchoring microtubules to the centrosome [63]. It is also implicated in ciliogenesis in male germ cells [64]. Defects in genes encoding centriolar satellite proteins, which are an important part of the centrosome, are linked with microcephaly [65]. In the case of ELFN1 four heterozygous variants were reported in a cohort of Japanese patients with neurological disorders, such as epilepsy or autism spectrum disorder [66]. Functional studies show that ELFN1 mutants display seizures, motor abnormalities and hyperactivity [67]. Moreover, the ELFN1 protein is present in excitatory synapses, where it acts as a regulator of presynaptic release [68]. We speculate that the combined effect of variants in both of these genes may be responsible for the clinical picture of the patient (Supplementary Table S1). However, impact of disruptions of both the CCDC112 and ELFN1 genes is as yet largely unknown. Further information is therefore necessary.
Although the predictive algorithms and the current knowledge about the functions and interactions of proteins encoded by these genes indicate their potentially pathogenic nature, there is a need for functional studies or the identification of a larger number of patients with changes in these genes who display a similar phenotype to classify them as new genes, the mutations of which are responsible for microcephaly and other neurodevelopmental disorders.
5. Conclusions
Analysis of full exome data for 191 clinically diagnosed congenital microcephaly patients in our study provides a diagnostic rate of 37% and possibly pathogenic genetic variants in 12 genes previously unassociated with microcephaly. In addition, this is the first study of microcephalic patients in which such a large group of patients have mutations in tubulin-related genes. We also found that the number of patients with pathogenic mutations in MCPH genes is almost two times higher in comparison to similar previous studies. Our results indicate that in a group of patients with microcephaly and no reported consanguinity, the dominant inheritance pattern is more prevalent. Our observations expand the phenotypic and genetic landscape of microcephaly and support the routine use of exome sequencing when genetic etiology is suspected. Our findings further provide closer phenotypic delineation for variants in the most commonly disrupted genes in our own and other cohorts, facilitating differential clinical diagnoses for disorders associated with these genes.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/genes12122014/s1, Table S1: Patients details, Table S2: Gene list, Table S3: Gene coverage, File S4: MultiQC report.
Author Contributions
Conceptualization, W.W. and P.G.; methodology, W.W and P.G.; software, T.G.; validation, M.D., T.G. and M.M.; formal analysis, T.G. and M.B.-F.; investigation, M.D.; resources, E.B.-O., D.A.-M., M.B.-S., P.B., E.B., J.C., T.C., E.C., A.E.-M., J.F.-M., D.G.-B., E.G.-B., A.G.-C., M.G.-P., B.G., A.J.-T., E.J., M.J. (Magdalena Janeczko), D.J.-P., D.K., A.K., T.K., I.K., M.K.-W., K.K., A.K.-K., A.L., D.M.-G., A.M.-K., H.M., T.P.-G., A.P. (Aleksandra Pietrzyk), R.P., A.P. (Antoni Pyrkosz), M.R.-D., R.S., M.W., Z.Z.-M., E.S. and E.O.; data curation, T.G. and M.D.; writing—original draft preparation, M.D.; writing—review and editing, W.W., M.M. and M.J. (Marta Jurek); visualization, M.D.; supervision, W.W. and P.G.; funding acquisition, W.W. and P.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by National Science Centre, Poland, grant number 2015/19/B/NZ2/01824.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Ethics Committee of Institute of Mother and Child in Warsaw, Poland (Committee Opinion No. 29/2016 from 23 June 2016).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to them containing information that could compromise research participant privacy.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Woods C.G. Human microcephaly. Curr. Opin. Neurobiol. 2004;14:112–117. doi: 10.1016/j.conb.2004.01.003. [DOI] [PubMed] [Google Scholar]
- 2.Von Der Hagen M., Pivarcsi M., Liebe J., von Bernuth H., Di Donato N., Hennermann J.B., Bührer C., Wieczorek D., Kaindl A.M. Diagnostic approach to microcephaly in childhood: A two-center study and review of the literature. Dev. Med. Child Neurol. 2014;56:732–741. doi: 10.1111/dmcn.12425. [DOI] [PubMed] [Google Scholar]
- 3.Woods C.G., Parker A. Investigating microcephaly. Arch. Dis. Child. 2013;98:707–713. doi: 10.1136/archdischild-2012-302882. [DOI] [PubMed] [Google Scholar]
- 4.Faheem M., Naseer M.I., Rasool M., Chaudhary A.G., Kumosani T.A., Ilyas A.M., Pushparaj P.N., Ahmed F., Algahtani H.A., Al-Qahtani M.H., et al. Molecular genetics of human primary microcephaly: An overview. BMC Med. Genom. 2015;8((Suppl. S1)):S4. doi: 10.1186/1755-8794-8-S1-S4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Seltzer L.E., Paciorkowski A.R. Genetic disorders associated with postnatal microcephaly. Am. J. Med. Genet. Part C Semin. Med. Genet. 2014;166:140–155. doi: 10.1002/ajmg.c.31400. [DOI] [PubMed] [Google Scholar]
- 6.Alcantara D., O’Driscoll M. Congenital microcephaly. Am. J. Med. Genet. Part C Semin. Med. Genet. 2014;166:124–139. doi: 10.1002/ajmg.c.31397. [DOI] [PubMed] [Google Scholar]
- 7.Yang Y., Muzny D.M., Xia F., Niu Z., Person R., Ding Y., Ward P., Braxton A., Wang M., Buhay C., et al. Molecular Findings Among Patients Referred for Clinical Whole-Exome Sequencing. JAMA. 2014;312:1870–1879. doi: 10.1001/jama.2014.14601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Barbelanne M., Tsang W.Y. Molecular and Cellular Basis of Autosomal Recessive Primary Microcephaly. BioMed Res. Int. 2014;2014:547986. doi: 10.1155/2014/547986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Perez Y., Bar-Yaacov R., Kadir R., Wormser O., Shelef I., Birk O., Flusser H., Birnbaum R.Y. Mutations in the microtubule-associated protein MAP11 (C7orf43) cause microcephaly in humans and zebrafish. Brain. 2019;142:574–585. doi: 10.1093/brain/awz004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rump P., Jazayeri O., van Dijk-Bos K.K., Johansson L.F., van Essen A.J., Verheij J.B.G.M., Veenstra-Knol H.E., Redeker E.J.W., Mannens M.M.A.M., Swertz M.A., et al. Whole-exome sequencing is a powerful approach for establishing the etiological diagnosis in patients with intellectual disability and microcephaly. BMC Med. Genom. 2015;9:7. doi: 10.1186/s12920-016-0167-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Boonsawat P., Joset P., Steindl K., Oneda B., Gogoll L., Azzarello-Burri S., Sheth F., Datar C., Verma I.C., Puri R.D., et al. Elucidation of the phenotypic spectrum and genetic landscape in primary and secondary microcephaly. Genet. Med. 2019;21:2043–2058. doi: 10.1038/s41436-019-0464-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shaheen R., Maddirevula S., Ewida N., Alsahli S., Abdel-Salam G.M.H., Zaki M.S., Al Tala S., Alhashem A., Softah A., Al-Owain M., et al. Genomic and phenotypic delineation of congenital microcephaly. Genet. Med. 2018;21:545–552. doi: 10.1038/s41436-018-0140-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Li H., Durbin R. Fast and Accurate Short Read Alignment with Burrows-Wheeler Transform. Bioinformatics. 2009;25:1754–1760. doi: 10.1093/bioinformatics/btp324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Poplin R., Ruano-Rubio V., DePristo M.A., Fennell T.J., Carneiro M.O., van der Auwera G.A., Kling D.E., Gauthier L.D., Levy-Moonshine A., Roazen D., et al. Scaling accurate genetic variant discovery to tens of thousands of samples. BioRxiv. 2017:201178. doi: 10.1101/201178. [DOI] [Google Scholar]
- 15.Chapman B., Kirchner R., Pantano L., Khotiainsteva T., Smet M.D., Beltrame L., Saveliev V., Guimera R.V., Nau-menko S., Kern J., et al. Bcbio/Bcbio-Nextgen: V1.1.9. Zenodo; Geneva, Switzerland: 2019. [DOI] [Google Scholar]
- 16.McLaren W., Gil L., Hunt S.E., Riat H.S., Ritchie G.R.S., Thormann A., Flicek P., Cunningham F. The Ensembl Variant Effect Predictor. Genome Biol. 2016;17:122. doi: 10.1186/s13059-016-0974-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Landrum M.J., Lee J.M., Benson M., Brown G., Chao C., Chitipiralla S., Gu B., Hart J., Hoffman D., Hoover J., et al. ClinVar: Public archive of interpretations of clinically relevant variants. Nucleic Acids Res. 2016;44:D862–D868. doi: 10.1093/nar/gkv1222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang K., Li M., Hakonarson H. ANNOVAR: Functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 2010;38:e164. doi: 10.1093/nar/gkq603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Liu X., Li C., Mou C., Dong Y., Tu Y. dbNSFP v4: A comprehensive database of transcript-specific functional predictions and annotations for human nonsynonymous and splice-site SNVs. Genome Med. 2020;12:103. doi: 10.1186/s13073-020-00803-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Krumm N., Sudmant P.H., Ko A., O’Roak B.J., Malig M., Coe B.P., Quinlan A.R., Nickerson D.A., Eichler E.E., Project N.E.S. Copy number variation detection and genotyping from exome sequence data. Genome Res. 2012;22:1525–1532. doi: 10.1101/gr.138115.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pedersen B.S., Quinlan A.R. Mosdepth: Quick Coverage Calculation for Genomes and Exomes. Bioinformatics. 2018;34:867–868. doi: 10.1093/bioinformatics/btx699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Danecek P., Bonfield J.K., Liddle J., Marshall J., Ohan V., Pollard M.O., Whitwham A., Keane T., McCarthy S.A., Davies R.M., et al. Twelve years of SAMtools and BCFtools. GigaScience. 2021;10:giab008. doi: 10.1093/gigascience/giab008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.García-Alcalde F., Okonechnikov K., Carbonell J., Cruz L.M., Götz S., Tarazona S., Dopazo J., Meyer T.F., Conesa A. Qualimap: Evaluating next-generation sequencing alignment data. Bioinformatics. 2012;28:2678–2679. doi: 10.1093/bioinformatics/bts503. [DOI] [PubMed] [Google Scholar]
- 24.Ewels P., Magnusson M., Lundin S., Käller M. MultiQC: Summarize analysis results for multiple tools and samples in a single report. Bioinformatics. 2016;32:3047–3048. doi: 10.1093/bioinformatics/btw354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Richards S., Aziz N., Bale S., Bick D., Das S., Gastier-Foster J., Grody W.W., Hegde M., Lyon E., Spector E., et al. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 2015;17:405–423. doi: 10.1038/gim.2015.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bahi-Buisson N., Poirier K., Fourniol F., Saillour Y., Valence S., Lebrun N., Hully M., Bianco C.F., Boddaert N., Elie C., et al. The wide spectrum of tubulinopathies: What are the key features for the diagnosis? Brain. 2014;137:1676–1700. doi: 10.1093/brain/awu082. [DOI] [PubMed] [Google Scholar]
- 27.Moortgat S., Berland S., Aukrust I., Maystadt I., Baker L., Benoit V., Caro-Llopis A., Cooper N.S., Debray F.-G., Faivre L., et al. HUWE1 variants cause dominant X-linked intellectual disability: A clinical study of 21 patients. Eur. J. Hum. Genet. 2018;26:64–74. doi: 10.1038/s41431-017-0038-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Darvish H., Esmaeeli-Nieh S., Monajemi G.B., Mohseni M., Ghasemi-Firouzabadi S., Abedini S.S., Bahman I., Jamali P., Azimi S., Mojahedi F., et al. A clinical and molecular genetic study of 112 Iranian families with primary microcephaly. J. Med. Genet. 2010;47:823–828. doi: 10.1136/jmg.2009.076398. [DOI] [PubMed] [Google Scholar]
- 29.Gardella E., Møller R. Phenotypic and genetic spectrum of SCN 8A -related disorders, treatment options, and outcomes. Epilepsia. 2019;60:S77–S85. doi: 10.1111/epi.16319. [DOI] [PubMed] [Google Scholar]
- 30.Zarate Y.A., Bosanko K.A., Caffrey A.R., Bernstein J.A., Martin D.M., Williams M.S., Berry-Kravis E.M., Mark P., Manning M.A., Bhambhani V., et al. Mutation update for the SATB2 gene. Hum. Mutat. 2019;40:1013–1029. doi: 10.1002/humu.23771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Borlot F., Abushama A., Morrison-Levy N., Jain P., Vinayan K.P., Abukhalid M., Aldhalaan H.M., Almuzaini H.S., Gulati S., Hershkovitz T., et al. KCNT1-related epilepsy: An international multicenter cohort of 27 pediatric cases. Epilepsia. 2020;61:679–692. doi: 10.1111/epi.16480. [DOI] [PubMed] [Google Scholar]
- 32.Platzer K., Lemke J.R. GRIN1-Related Neurodevelopmental Disorder. In: Adam M.P., Ardinger H.H., Pagon R.A., Wallace S.E., Bean L.J., Mirzaa G., Amemiya A., editors. GeneReviews®. University of Washington; Seattle, WA, USA: 1993. [PubMed] [Google Scholar]
- 33.Ke Z., Chen Y. Case Report: A de novo CTNNB1 Nonsense Mutation Associated with Neurodevelopmental Disorder, Retinal Detachment, Polydactyly. Front. Pediatr. 2020;8:850. doi: 10.3389/fped.2020.575673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rossetti L.Z., Bekheirnia M.R., Lewis A.M., Mefford H.C., Golden-Grant K., Tarczy-Hornoch K., Briere L.C., Sweetser D.A., Walker M.A., Kravets E., et al. Missense variants in CTNNB1 can be associated with vitreoretinopathy—Seven new cases of CTNNB1 -associated neurodevelopmental disorder including a previously unreported retinal phenotype. Mol. Genet. Genom. Med. 2021;9:e1542. doi: 10.1002/mgg3.1542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Verhoeven W.M., Egger J.I., E Jongbloed R., van Putten M.M., Zandwijk M.D.B.-V., Zwemer A.-S., Pfundt R., Willemsen M.H. A de novo CTNNB1 novel splice variant in an adult female with severe intellectual disability. Int. Med. Case Rep. J. 2020;13:487–492. doi: 10.2147/IMCRJ.S270487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mitter D., Pringsheim M., Kaulisch M., Plümacher K.S., Schröder S., Warthemann R., Jamra R.A., Baethmann M., Bast T., Büttel H.-M., et al. FOXG1 syndrome: Genotype–phenotype association in 83 patients with FOXG1 variants. Genet. Med. 2018;20:98–108. doi: 10.1038/gim.2017.75. [DOI] [PubMed] [Google Scholar]
- 37.Huang L., Vanstone M.R., Hartley T., Osmond M., Barrowman N., Allanson J., Baker L., Dabir T.A., Dipple K.M., Dobyns W., et al. Mandibulofacial Dysostosis with Microcephaly: Mutation and Database Update. Hum. Mutat. 2016;37:148–154. doi: 10.1002/humu.22924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Brown G.K., Otero L.J., Legris M., Brown R.M. Syndrome of the Month Pyruvate Dehydrogenase Deficiency. Amino Acids. 1994;31:875–879. doi: 10.1136/jmg.31.11.875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Pavlu-Pereira H., Silva M.J., Florindo C., Sequeira S., Ferreira A.C., Duarte S., Rodrigues A.L., Janeiro P., Oliveira A., Gomes D., et al. Pyruvate dehydrogenase complex deficiency: Updating the clinical, metabolic and mutational landscapes in a cohort of Portuguese patients. Orphanet J. Rare Dis. 2020;15:298. doi: 10.1186/s13023-020-01586-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Verloes A., Drunat S., Passemard S. ASPM Primary Microcephaly. In: Adam M.P., Ardinger H.H., Pagon R.A., Wallace S.E., Bean L.J., Mirzaa G., Amemiya A., editors. GeneReviews®. University of Washington; Seattle, WA, USA: 1993. [PubMed] [Google Scholar]
- 41.Abdel-Hamid M.S., Ismail M.F., Darwish H.A., Effat L.K., Zaki M.S., Abdel-Salam G. Molecular and phenotypic spectrum of ASPM-related primary microcephaly: Identification of eight novel mutations. Am. J. Med. Genet. Part A. 2016;170:2133–2140. doi: 10.1002/ajmg.a.37724. [DOI] [PubMed] [Google Scholar]
- 42.Posey J.E., Harel T., Liu P., Rosenfeld J.A., James R.A., Akdemir Z.H.C., Walkiewicz M., Bi W., Xiao R., Ding Y., et al. Resolution of Disease Phenotypes Resulting from Multilocus Genomic Variation. N. Engl. J. Med. 2017;376:21–31. doi: 10.1056/NEJMoa1516767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lemke J.R., Geider K., Helbig K.L., Heyne H.O., Schütz H., Hentschel J., Courage C., Depienne C., Nava C., Heron D., et al. Delineating the GRIN1 phenotypic spectrum. Neurology. 2016;86:2171–2178. doi: 10.1212/WNL.0000000000002740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dong X., Liu B., Yang L., Wang H., Wu B., Liu R., Chen H., Chen X., Yu S., Chen B., et al. Clinical exome sequencing as the first-tier test for diagnosing developmental disorders covering both CNV and SNV: A Chinese cohort. J. Med. Genet. 2020;57:558–566. doi: 10.1136/jmedgenet-2019-106377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Minczuk M. Localisation of the human hSuv3p helicase in the mitochondrial matrix and its preferential unwinding of dsDNA. Nucleic Acids Res. 2002;30:5074–5086. doi: 10.1093/nar/gkf647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Szczesny R.J., Obriot H., Paczkowska A., Jedrzejczak R., Dmochowska A., Bartnik E., Formstecher P., Polakowska R., Stepien P.P. Down-regulation of human RNA/DNA helicase SUV3 induces apoptosis by a caspase- and AIF-dependent pathway. Biol. Cell. 2007;99:323–332. doi: 10.1042/BC20060108. [DOI] [PubMed] [Google Scholar]
- 47.Sharma S., Doherty K.M., Brosh R.M. Mechanisms of RecQ helicases in pathways of DNA metabolism and maintenance of genomic stability. Biochem. J. 2006;398:319–337. doi: 10.1042/BJ20060450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.De Renty C., Ellis N.A. Bloom’s syndrome: Why not premature aging? Ageing Res. Rev. 2017;33:36–51. doi: 10.1016/j.arr.2016.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Bouman A., van Koningsbruggen S., Karakullukcu M.B., Schreuder W.H., Lakeman P. Bloom syndrome does not always present with sun-sensitive facial erythema. Eur. J. Med. Genet. 2018;61:94–97. doi: 10.1016/j.ejmg.2017.10.010. [DOI] [PubMed] [Google Scholar]
- 50.Ni T.T., Lu J., Zhu M., Maddison L.A., Boyd K.L., Huskey L., Ju B., Hesselson D., Zhong T.P., Page-McCaw P., et al. Conditional control of gene function by an invertible gene trap in zebrafish. Proc. Natl. Acad. Sci. USA. 2012;109:15389–15394. doi: 10.1073/pnas.1206131109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Pereira M., Mason P., Szczesny R., Maddukuri L., Dziwura S., Jedrzejczak R., Paul E., Wojcik A., Dybczynska L., Tudek B. Interaction of human SUV3 RNA/DNA helicase with BLM helicase; loss of the SUV3 gene results in mouse embryonic lethality. Mech. Ageing Dev. 2007;128:609–617. doi: 10.1016/j.mad.2007.09.001. [DOI] [PubMed] [Google Scholar]
- 52.Lee T., Di Paola D., Malina A., Mills J.R., Kreps A., Grosse F., Tang H., Zannis-Hadjopoulos M., Larsson O., Pelletier J. Suppression of the DHX9 Helicase Induces Premature Senescence in Human Diploid Fibroblasts in a p53-dependent Manner. J. Biol. Chem. 2014;289:22798–22814. doi: 10.1074/jbc.M114.568535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Capitanio J.S., Montpetit B., Wozniak R.W. Human Nup98 regulates the localization and activity of DExH/D-box helicase DHX9. Elife. 2017;6:e18825. doi: 10.7554/elife.18825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Aktas T., Avsar Ilik I., Maticzka D., Bhardwaj V., Pessoa Rodrigues C., Mittler G., Manke T., Backofen R., Akhtar A. DHX9 suppresses RNA processing defects originating from the Alu invasion of the human genome. Nature. 2017;544:115–119. doi: 10.1038/nature21715. [DOI] [PubMed] [Google Scholar]
- 55.Chakraborty P., Huang J.T.J., Hiom K. DHX9 helicase promotes R-loop formation in cells with impaired RNA splicing. Nat. Commun. 2018;9:4346. doi: 10.1038/s41467-018-06677-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lee T., Pelletier J. The biology of DHX9 and its potential as a therapeutic target. Oncotarget. 2016;7:42716–42739. doi: 10.18632/oncotarget.8446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kawahara H., Imai T., Imataka H., Tsujimoto M., Matsumoto K., Okano H. Neural RNA-binding protein Musashi1 inhibits translation initiation by competing with eIF4G for PABP. J. Cell Biol. 2008;181:639–653. doi: 10.1083/jcb.200708004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cambuli F., Correa B., Rezza A., Burns S., Qiao M., Uren P., Kress E., Boussouar A., Galante P., Penalva L., et al. A Mouse Model of Targeted Musashi1 Expression in Whole Intestinal Epithelium Suggests Regulatory Roles in Cell Cycle and Stemness. St. Cells. 2015;33:3621–3634. doi: 10.1002/stem.2202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Murphy D., Cieply B., Carstens R., Ramamurthy V., Stoilov P. The Musashi 1 Controls the Splicing of Photoreceptor-Specific Exons in the Vertebrate Retina. PLoS Genet. 2016;12:e1006256. doi: 10.1371/journal.pgen.1006256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Abreu R.D.S., Sanchez-Diaz P.C., Vogel C., Burns S.C., Ko D., Burton T.L., Vo D.T., Chennasamudaram S., Le S.-Y., Shapiro B.A., et al. Genomic Analyses of Musashi1 Downstream Targets Show a Strong Association with Cancer-related Processes. J. Biol. Chem. 2009;284:12125–12135. doi: 10.1074/jbc.M809605200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Siskos N., Stylianopoulou E., Skavdis G., Grigoriou M. Molecular Genetics of Microcephaly Primary Hereditary: An Overview. Brain Sci. 2021;11:581. doi: 10.3390/brainsci11050581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Chavali P.L., Stojic L., Meredith L.W., Joseph N., Nahorski M.S., Sanford T.J., Sweeney T.R., Krishna B.A., Hosmillo M., Firth A.E., et al. Neurodevelopmental protein Musashi-1 interacts with the Zika genome and promotes viral replication. Science. 2017;357:83–88. doi: 10.1126/science.aam9243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Gupta G.D., Coyaud E., Gonçalves J., Mojarad B.A., Liu Y., Wu Q., Gheiratmand L., Comartin D., Tkach J.M., Cheung S.W., et al. A Dynamic Protein Interaction Landscape of the Human Centrosome-Cilium Interface. Cell. 2015;163:1484–1499. doi: 10.1016/j.cell.2015.10.065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Vandenbrouck Y., Pineau C., Lane L. The Functionally Unannotated Proteome of Human Male Tissues: A Shared Resource to Uncover New Protein Functions Associated with Reproductive Biology. J. Proteome Res. 2020;19:4782–4794. doi: 10.1021/acs.jproteome.0c00516. [DOI] [PubMed] [Google Scholar]
- 65.Kodani A., Yu T.W., Johnson J.R., Jayaraman D., Johnson T.L., Al-Gazali L., Sztriha L., Partlow J.N., Kim H., Krup A.L., et al. Centriolar satellites assemble centrosomal microcephaly proteins to recruit CDK2 and promote centriole duplication. Elife. 2015;4:e07519. doi: 10.7554/eLife.07519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Tomioka N.H., Yasuda H., Miyamoto H., Hatayama M., Morimura N., Matsumoto Y., Suzuki T., Odagawa M., Odaka Y.S., Iwayama Y., et al. Elfn1 recruits presynaptic mGluR7 in trans and its loss results in seizures. Nat. Commun. 2014;5:4501. doi: 10.1038/ncomms5501. [DOI] [PubMed] [Google Scholar]
- 67.Dolan J., Mitchell K.J. Mutation of Elfn1 in Mice Causes Seizures and Hyperactivity. PLoS ONE. 2013;8:e80491. doi: 10.1371/journal.pone.0080491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Cao Y., Sarria I., Fehlhaber K.E., Kamasawa N., Orlandi C., James K.N., Hazen J.L., Gardner M.R., Farzan M., Lee A., et al. Mechanism for Selective Synaptic Wiring of Rod Photoreceptors into the Retinal Circuitry and Its Role in Vision. Neuron. 2015;87:1248–1260. doi: 10.1016/j.neuron.2015.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to them containing information that could compromise research participant privacy.