Abstract
Diabetes is a driver of non-alcoholic fatty liver disease (NAFLD) and fibrosis. We determine current practices in examining liver fibrosis in people with diabetes and record prevalence levels in primary and secondary care. We extracted HbA1c results ≥48 mmol/mol to identify people with diabetes, then examined the proportion who had AST, ALT, and platelets results, facilitating calculation of non-invasive fibrosis tests (NIT), or an enhanced liver fibrosis score. Fibrosis markers were requested in only 1.49% (390/26,090), of which 29.7% (n = 106) had evidence of significant fibrosis via NIT. All patients at risk of fibrosis had undergone transient elastography (TE), biopsy or imaging. TE and biopsy data showed that 80.6% of people with raised fibrosis markers had confirmed significant fibrosis. We also show that fibrosis levels as detected by NIT are marginally lower in patients treated with newer glucose lowering agents (sodium-glucose transporter protein 2 inhibitors, dipeptidyl peptidase-4 inhibitors and glucagon-like peptide-1 receptor agonists). In conclusion by utilising a large consecutively recruited dataset we demonstrate that liver fibrosis is infrequently screened for in patients with diabetes despite high prevalence rates of advanced fibrosis. This highlights the need for cost-effectiveness analyses to support the incorporation of widespread screening into national guidelines and the requirement for healthcare practitioners to incorporate NAFLD screening into routine diabetes care.
Keywords: fibrosis, NAFLD, diabetes, screening, primary care, secondary care
1. Introduction
Non-alcoholic fatty liver disease (NAFLD) is the most common cause of liver disease in the UK and Europe [1], soon to become the most common indication for liver transplantation in the next decade [2], as a result of the obesity and associated type 2 diabetes (T2D) epidemics. Expert consensus has suggested NAFLD be re-named metabolic-associated fatty liver disease (MAFLD) to reflect its strong association with insulin resistance and the metabolic syndrome [3]. Type 2 diabetes is a condition characterised by peripheral insulin resistance with inadequate compensatory pancreatic beta-cell insulin secretion. Insulin resistance and systemic inflammation lead to accumulation of free fatty acids and consequentially hepatocyte triglyceride accumulation characterising NAFLD [4,5]. NAFLD is generally benign in the majority of individuals, however in up to 40% of people it can progress to liver fibrosis [6,7]. Liver fibrosis describes the development of fibrous tissue due to the replacement of healthy tissue by extracellular matrix proteins, in NAFLD this is the result of hepatotoxic injury and initially leads to non-alcoholic steatohepatitis (NASH) and chronically to liver fibrosis [8]. Liver fibrosis, rather than simple steatosis or NASH, is associated with an increased risk of liver-related morbidity and mortality [6,9], overall mortality [10], and cardiovascular disease [11,12].
One of the most significant predictors of fibrosis progression and the development of advanced fibrosis is diabetes, particularly T2D [13,14,15,16,17,18]. NAFLD is reported to be present in 40–70% of individuals with T2D [19,20,21]. Furthermore, UK diabetes prevalence according to Quality Outcome Framework data is now 7.1% (2020/21), with an additional large number of undiagnosed cases [22]. While the European Association for the Study of the Liver (EASL) guidelines [23] and American Diabetes Association guidelines [24] suggest surveillance for NAFLD in people with T2D, the American [25], Asian [26], and UK [27,28] guidelines acknowledge that individuals with T2D are at greater risk of NAFLD, yet do not advocate widespread screening.
We aimed to perform a cross-sectional analysis of the burden of significant liver fibrosis in individuals with diabetes from both primary and secondary care to understand the prevalence of potentially clinically significant liver disease in these settings; and to provide a snapshot into current practice of examining fibrosis markers and ongoing risk stratification in people with diabetes.
2. Materials and Methods
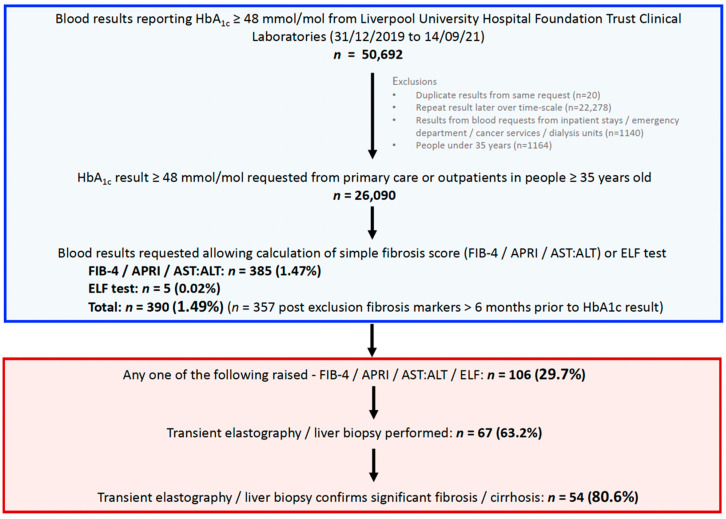
Screening with HbA1c We extracted glycated haemoglobin (HbA1c) results over a 21-month period (31 December 2019 to 14 September 21) from the Liverpool (University Hospital Foundation Trust) Clinical Laboratories and identified a cohort of individuals with an HbA1c ≥ 48 mmol/mol indicative of a diagnosis of diabetes (Figure 1). Individuals under 35 years old were excluded as fibrosis scores are inaccurate in this age group. Results from blood requests from inpatient stays, the emergency department, cancer services, and dialysis units were excluded, leaving those taken from primary care and other outpatient departments.
Figure 1.
Study flow chart and summary of results. HbA1c, glycated haemoglobin; FIB-4, fibrosis-4; APRI, aspartate transaminase to platelet ratio index; AST aspartate transaminase; ALT, alanine transaminase; ELF, enhanced liver fibrosis score.
2.1. Determination of Liver Biochemistry Results and Fibrosis Scores
We examined what proportion of these people had an aspartate transaminase (AST), alanine transaminase (ALT), and platelet levels taken within this time frame. From these results, we calculated three validated non-invasive scores of liver fibrosis, the fibrosis-4 (FIB-4) score [29], the AST to platelet ration index (APRI) [30], and the AST:ALT ratio (Supplementary Table S1) [31]. Significant fibrosis was defined as either a FIB-4 score > 2.67, APRI score ≥ 1.0, or AST:ALT ratio ≥ 1.0, where either the AST or ALT level was also >40 IU/L. We also included patients with an enhanced liver fibrosis score (ELF), based on tissue inhibitor metalloproteinases 1, amino-terminal pro-peptide of type III procollagen and hyaluronic acid [32]. Significant fibrosis was defined as an ELF score of >9.8. We then excluded results taken over 6 months prior to the HbA1c to ensure that individuals were likely to have diabetes at the time the fibrosis tests were taken. We additionally compared prevalence rates of liver fibrosis detected by primary and secondary care.
2.2. Confirmation of Fibrosis Identified with Non-Invasive Testing Using Transient Elastography (TE) and/or Liver Biopsy
We further examined what proportion of individuals identified as being at risk of significant liver fibrosis according to non-invasive tests (NITs), had gone on to have confirmatory testing with either TE or liver biopsy. TE suggestive of fibrosis was defined according to a liver stiffness measurement > 8 kPa (Fibroscan, Echosens, Paris, France). Histological evidence of significant fibrosis or cirrhosis was confirmed by percutaneous liver biopsy and verified by an experienced liver histopathologist.
2.3. Association between Advanced Fibrosis According to FIB-4 Score and Glucose Lowering Agents
We examined prescription data for glucose lowering agents for patients who had data available to calculate a FIB-4 score. We additionally examined the proportion of people with a raised FIB-4 score > 2.67 according the number and classes of glucose lowering medications prescribed.
2.4. Statistical Analysis
Results are presented as the median and interquartile range. Data validity was ensured by examining ten random NHS numbers of both included and excluded patients and cross-checking them across databases. Data was analysed using R version 4.1.1 (R Foundation for Statistical Computing, Vienna, Austria) and Excel Kutools.
2.5. Ethics
As all patient data was anonymised this project did not require national ethical approval; clinical audit approval was obtained locally (number 10864).
3. Results
3.1. Description of Study Cohort
We identified 26,090 individuals who had an HbA1c result ≥48 mmol/mol requested from primary care or secondary care (outpatients department). Data was available to calculate the APRI score, AST:ALT ratio and FIB-4 score in 385 (1.47%) of these individuals and a further 5 (0.02%) had an ELF score requested, meaning that overall 390 (1.49%) people with diabetes had undergone a non-invasive test for fibrosis. Following the exclusion of results taken >6 months prior to the HbA1c result, the final study cohort consisted of 357 individuals with diabetes (Figure 1). In total 134 (37.5%) results were ordered from primary care and 223 from outpatients (62.5%). Baseline demographic data and laboratory results from this cohort are presented in Table 1.
Table 1.
Baseline data from the cohort (n = 357).
| Variable | Demographic Factor/Laboratory Finding |
|---|---|
| Sex (n (%)) | 204 (57.1) M, 153 (42.9) F |
| Age (years) (Median (IQR)) | 60 (53–67) |
| HbA1c (mmol/mol) (Median (IQR)) | 62 (53–76) |
| AST (IU/L) (Median (IQR)) | 30 (21–48) |
| ALT (IU/L) (Median (IQR)) | 35 (23–53) |
| Platelets (×109/L) (Median (IQR)) | 223 (170–284) |
| ELF score | 10.1 (10–10.7) |
M, male; F, Female; IQR, interquartile range; HbA1c, glycated haemoglobin; AST aspartate transaminase; ALT, alanine transaminase; ELF, enhanced liver fibrosis.
3.2. Prevalence of Significant Fibrosis in Individuals with Diabetes According to Serum Fibrosis Scores
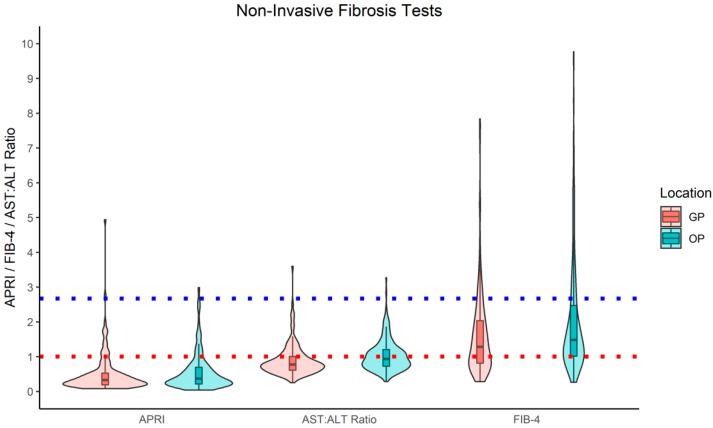
Between 13.7–19% individuals with diabetes were identified as having evidence of significant fibrosis using simple NITs (Table 2, Figure 2) and 80% (4/5) of people who had an ELF score requested had evidence of significant fibrosis. Using the previously described definitions of significant fibrosis (one or more of FIB-4 score > 2.67, APRI ≥ 1.0, AST:ALT ≥ 1.0, or ELF > 9.8), 106 (29.7%) people with diabetes were identified as being at risk. Of the 106 people at risk of significant fibrosis, 30 (28.3%) had fibrosis markers requested from primary care. Of the 76 outpatient blood requests, 66 (86.8%) came from the liver clinic. Overall fibrosis scores derived from blood requests sent from secondary care (34.1%) showed higher levels of significant fibrosis than primary care (22.4%) (Table 1, Figure 2). There was no positive correlation between HbA1c and fibrosis scores when examined on a continuous scale (Supplementary Figure S1).
Table 2.
Percentage of people with diabetes found to have evidence of significant fibrosis determined by non-invasive markers.
| Non-Invasive Serum Fibrosis Scores | Total, % (n) | Primary Care, % (n) | Secondary Care, % (n) |
|---|---|---|---|
| n = 357 | 37.5 (134) | 62.5 (223) | |
| FIB-4 > 2.67 | 19.0 (68) | 13.4 (18) | 22.4 (50) |
| APRI ≥ 1.0 | 13.7 (49) | 12.7 (17) | 14.3 (32) |
| AST:ALT ratio ≥ 1.0 and AST or ALT > 40 IU/L | 17.4 (62) | 11.2 (15) | 21.1 (47) |
| Any one of the above, or ELF > 9.8 | 29.7 (106) | 22.4 (30) | 34.1 (76) |
FIB-4, fibrosis-4; APRI, aspartate transaminase to platelet ratio index; AST aspartate transaminase; ALT, alanine transaminase; ELF, enhanced liver fibrosis test; kPa kilopascal.
Figure 2.
Summary Graphs of FIB-4, APRI, AST:ALT Ratio by Test Location. Red dotted line = cut-off for high-risk fibrosis via APRI and AST:ALT Ration. Blue dotted line = cut-off for high risk liver fibrosis via FIB-4, FIB-4 = Fibrosis 4, APRI = AST to Platelet Ratio Index, n = Number, GP = General Practice, OP = Outpatient.
3.3. Prevalence of People with Diabetes and At-Risk Serum Fibrosis Scores with Confirmed Significant Fibrosis/Cirrhosis
Of the 106 individuals with diabetes identified to be at risk of significant fibrosis using non-invasive serum markers, 67/106 (63.2%) went on to have transient elastography (TE/Fibroscan) (n = 50, 47.2%), liver biopsy (n = 24, 22.6%), or both (n = 7, 6.6%). In total 54/67 (80.6%) of these individuals had a liver stiffness measurement >8 kPa or evidence of significant fibrosis or cirrhosis at biopsy. All 39 people with raised fibrosis markers who did not receive a fibroscan or liver biopsy, had prior liver imaging via ultrasound (n = 30, 76.9%) or CT (n = 9, 23.1%), and 21/39 (53.8%) had evidence of cirrhosis
3.4. Prevalence of a Raised FIB-4 Score According to the Number and Class of Glucose Lowering Agent
Medication data was available for 91.6% (327/357) patients. A further 4 patients were excluded who did not have data to calculate a FIB-4 score (final sample n = 323). A breakdown of the number of drugs and subclasses of glucose lowering agents prescribed are shown in Table 3. Patients who were not prescribed any glucose lowering therapies had lower levels of fibrosis according to the FIB-4 score (12.5%), compared to those on treatment (19.5%), however glycaemic control was also improved (Table 3, Supplementary Figure S2). Patients treated with SGLT2 inhibitors (16.4%), GLP-1 receptors agonists (16.0%) and DDP-4 inhibitors (15.1%) trended towards having non-significantly lower levels of NIT fibrosis (Table 3, Supplementary Figure S3), whilst having no noticeable differences in glycaemic control. Patients treated with metformin (18.6%) and sulphonylureas (18.4%) had similar levels of fibrosis to the overall cohort. Patients treated with insulin trended towards having non-significantly higher levels of fibrosis (23.8%) and higher HbA1c levels (median 73 mmol/mol) (Table 3, Supplementary Figure S3).
Table 3.
Results of Non-Invasive Serum Fibrosis Tests for People with Diabetes according to Number and Sub-class of Glucose-Lowering Agents Prescribed.
| People with Diabetes Who Had an NIT% (n) | Median HbA1c [IQR] (mmol/mol) | FIB-4 > 2.67% (n) | |
|---|---|---|---|
| Number of Glucose Lowering Agents Prescribed | |||
| None | 12.4 (40) | 51 (49–55) | 12.5 (5) |
| 1 | 40.6 (131) | 58 (52–70) | 22.1 (29) |
| 2 | 29.1 (94) | 67 (56–80) | 17.0 (16) |
| ≥3 | 19.2 (62) | 73 (62–86) | 17.7 (11) |
| Subclasses of Glucose Lowering Agents Prescribed | |||
| SGLT2 inhibitors | 18.9 (61) | 67 (59–79) | 16.4 (10) |
| GLP-1 receptors agonists | 7.7 (25) | 69 (55–77) | 16.0 (4) |
| DDP-4 inhibitors | 26.6 (86) | 67 (57–80) | 15.1 (13) |
| Metformin | 65.0 (210) | 63 (53–77) | 18.6 (39) |
| Insulin | 24.8 (80) | 73 (62–87) | 23.8 (19) |
| Sulphonylurea | 15.2 (49) | 76 (63–86) | 18.4 (9) |
| Thiazolidinediones | 0.6 (2) | n/A | 0.0 (0) |
FIB-4, Fibrosis 4; SGLT-2 inhibitor, sodium-glucose cotransporter 2 inhibitor; GLP-1 receptor agonist, Glucagon-Like Peptide 1 Receptor Agonist; DDP-4 inhibitor, Dipeptidyl peptidase-4 inhibitor.
4. Discussion
4.1. Summary of Findings
In this brief report, we utilise real world UK regional data from local populations of people with diabetes and highlight two alarming findings. First, we demonstrate that <2% of people with diabetes are being screened for liver fibrosis, and that use of patented serum fibrosis biomarkers is minimal despite been advocated by the National Institute of Health and Clinical Excellence (NICE) as first line assessment for people with NAFLD [27]. Secondly, up to 29.7% of people with diabetes, in whom serum fibrosis markers were requested, were at risk of having significant liver fibrosis; subsequent confirmation of fibrosis was provided by second line tests, TE or liver biopsy, in a high proportion (80.6%) of cases. Thirdly, we report limited data showing a non-significant trend towards lower fibrosis scores in patients treated with DDP-4 inhibitors, SGLT-2 inhibitors, and GLP-1 receptor agonists. These findings reinforce the need for large prospective studies in this clinical population to develop cost-effective and easily implementable approaches to widespread screening for liver fibrosis in individuals with diabetes.
4.2. Comparison to the Existing Literature
While our estimates of fibrosis prevalence in people with diabetes are higher than comparable studies, there is consensus in the literature that clinically relevant liver fibrosis is highly prevalent in this group. Global meta-analysis data in 439 biopsied patient with NAFLD and T2D identified that 17% had advanced fibrosis [21]. Data from over 120,000 people with T2D from the Cleveland clinic suggests that 8.4% have a FIB-4 score >2.67; however, prevalence estimates varied widely depending on the non-invasive score used [33]. Among individuals with T2D and a reliable TE result in the NHANES study (n = 825), 15.4% had a liver stiffness measurement ≥9.7 kPa. In a recent cross-sectional study from the US, 561 individuals with T2D attending primary care or endocrinology clinics underwent non-invasive screening using serum markers and TE; liver biopsy was performed where there was a suggestion of fibrosis [34]. In total 9% of people with diabetes had advanced fibrosis (F3/F4) according to TE. Fibrosis prevalence levels with TE were similar to that estimated using the FIB-4 and APRI panels, and both modalities correlated well with biopsy findings. A similar analysis from the UK identified that 18.5% of people with T2D attending primary care clinics (n = 467) had a FIB-4 >1.3 for ≤65 years and >2.0 for >65 years, of which nearly two thirds had a TE >8 kPa [35].
4.3. Molecular Mechanisms Linking T2D and NAFLD
Pathogenic mechanisms linking T2D to NAFLD are complex; however, insulin resistance and inflammation are central [36]. High levels of circulating glucose and insulin increase rates of hepatic de novo lipogenesis leading to high levels of free fatty acids (FFA) in the liver; excess FFAs are stored as intrahepatic triglycerides [37]. Adiposity and the presence of insulin resistant adipose tissue leads to lipolysis; FFAs released from adipose tissue are taken up by peripheral tissues including the liver and muscle. NAFLD itself in turn leads to impairments in insulin signalling [38] and increased secretion of hepatokines. Adipokines are lipotoxic agents arising from chronically inflamed adipose tissue characterising T2D. These travel to the liver contributing to inflammation and NAFLD development [39]. Lipotoxicity, along with oxidative stress and a pro-inflammatory environment, result in steatohepatitis and eventually activation of hepatic stellate cells and extracellular matrix deposition. Clinical studies support this mechanism: stable isotope analyses show patients with increased hepatic adiposity have higher plasma FFA levels and ~3x greater de novo FFA synthesis [40].
4.4. Implications for Practice
We therefore propose that there is an urgent need for greater adoption of national and international guidelines to implement widespread screening for fibrosis in individuals with diabetes and undertake comprehensive cost-effectiveness analyses. Despite updated recommendations from the EASL advocating the use of NITs and that ALT, AST, and platelets should be part of the routine investigations in primary care in patients with suspected liver disease [41], a huge shift in practice towards more widespread screening is unlikely to be implemented in the UK without guidance from the NICE. Detection of liver cirrhosis, which develops insidiously and without abnormalities in liver biochemistry, allows entry of individuals into variceal and hepatocellular carcinoma surveillance programmes, the latter being particularly relevant for people with diabetes [42,43]. Liver fibrosis is a partially reversible state, achieved with weight loss (~7%) [44], while fibrosis progression may be retarded with optimisation of glycaemic control, so multi-component metabolic intervention programmes are likely to be highly effective. Detection of NAFLD, and associated fibrosis, will facilitate enrolment in relevant clinical trials, and may encourage prescription of glucose-lowering therapies that target steatosis, steatohepatitis, or even fibrosis (including DDP-4 inhibitors, GLP receptor agonists and SGLT2 inhibitors) [45]. The burden of NAFLD and liver fibrosis expands beyond the liver, with well-established associations with cardiovascular morbidity and mortality [11,12] and extrahepatic cancer [46], so the wider benefits of detection are considerable.
We additionally show that fewer patients treated with either GLP-1 receptor agonists, SGLT 2 inhibitors and DDP-4 inhibitors have elevated FIB-4 scores. Glucose lowering therapies are a potential therapy in NAFLD given the fact they reduce insulin resistance and thus potentially reduce liver fat. DDP-4 inhibitors have not shown therapeutic effect in NAFLD; however, data is limited so larger trials are required [45,47,48]. GLP-1 receptor agonists have shown more promising findings. One study reported GLP-1 agonists significantly reduce liver fat (relative reduction 42%) [49]. Similarly, in a larger randomised controlled trial (RCT) (n = 320), semaglutide therapy led to higher rate of NASH resolution than control. However, no clear dose–response relationship was reported between dosing regimens (0.1 mg vs 0.2 mg vs 0.4 mg) [50]. A meta-analysis (n = 4442) of patients treated with liraglutide demonstrated ALT reduction [51]. For SGLT-2 inhibitors, a large RCT, EMPA-REG OUTCOME, reported that empagliflozin reduced ALT with these findings independent of glycaemic control (HbA1c) [52]. Similarly, in a moderately sized Swedish trial dapagliflozin reduced liver fat and ALT but did not improve glycaemic control. The conflicting findings between these two trials may or may delineate that SGLT-2 inhibitors have beneficial effects on NAFLD independent of glycaemic control [53]. Altogether, these trials show that GLP-1 agonists and SGLT 2 inhibitors have beneficial effects on liver biochemistry and liver fat levels in NAFLD. However, future trials need to assess the effects of these glucose lowering therapies on liver fibrosis. This could be via measuring non-invasive fibrosis scores (i.e., FIB-4, APRI, AST:ALT ratio), conducting fibroscans, liver multi-scan MRI testing, and liver biopsies.
4.5. Strengths and Limitations
This dataset benefits from a systematic approach to screening individuals with diabetes in both primary and secondary care. However, there are several limitations. The dataset is biased by the fact that we were only able to examine fibrosis markers in people in whom clinicians requested an AST level, i.e., influenced by clinical suspicion of liver disease. Most outpatient requests were made from hepatology clinics, with an inevitable bias towards higher rates of fibrosis or cirrhosis. These factors would lead to an over-estimation of fibrosis prevalence compared to the overall population with diabetes. The positive predictive values of NITs are only moderate, so the true prevalence of fibrosis confirmed by biopsy would also have been lower. Furthermore the performance of NITs is less well validated and less reliable in the diabetes population [54,55]. Individuals with exemplary glycaemic control, with HbA1c < 48 mmol/mol would also have been overlooked, leading to a selection bias towards a sub-population of lesser metabolic health at higher risk of diabetes-related end-organ damage. This study was reliant on electronic medical records and therefore we were unable to reliably determine the aetiology of diabetes (type 1 or type 2 diabetes), or liver disease (including alcohol excess or viral hepatitis). Approximately 95% of people with diabetes in the UK are estimated to have T2D; however, and all individuals that we have assessed would have had either MAFLD or dual aetiology liver disease, given the fact that they had diabetes. We examined the current practice of examining fibrosis markers in individuals with diabetes over a 1 year window of an HbA1c. Current guidelines advise screening every 1–3 years in people with confirmed NAFLD [41], so some individuals may have had bloods taken which could have been used to calculate a fibrosis score outside this time period. This study was limited by a significant proportion of the data being extracted over the COVID-19 pandemic. This may have negatively affected screening rates for fibrosis markers in both primary and secondary care and therefore may have affected the results. In addition, this study was limited by omission of the body mass index (BMI) data, which was not widely available from patient records. While we were able to access prescription records, unfortunately data on duration of diabetes, duration a glucose lowering agent had been prescribed and historic prescription data was no available to allow a comprehensive assessment of the role of newer glucose lowering therapies on fibrosis levels.
5. Conclusions
In summary, we found very limited evidence of systematic screening for liver fibrosis: only 1.5% of individuals with diabetes had a NIT for assessment of fibrosis, despite evidence of a high prevalence of significant fibrosis (29.8%) in those assessed. We also show that fibrosis levels as detected by NIT is lower in patients treated with SGLT2 inhibitors, DDP-4 inhibitors, and GLP-1 receptor agonists. There is an urgent and unmet need to assess, develop, and implement cost-effective methods to provide widespread screening of individuals with diabetes for liver fibrosis and for healthcare practitioners to incorporate NAFLD screening into routine diabetes care. This will undoubtedly reap longer-term clinical benefits in reducing the hepatic and extra-hepatic burden of NAFLD in patients with diabetes.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/jcm10245755/s1, Table S1: Algorithms used to calculate Hepatic Steatosis Index and Fibrosis scores, Figure S1: Correlation between HbA1c and Fibrosis Marker scores; Figure S2: Percentage of people with a raised FIB-4 score according to Number of glucose lowering agents prescribed; Figure S3: Percentage of people with a raised FIB-4 score according to subclasses of glucose lowering agents prescribed.
Author Contributions
Conceptualization, all authors; methodology, all authors; formal analysis, L.J.D., M.K. and T.J.H.; data curation, A.S.D. and P.G.; writing—original draft preparation, T.J.H.; writing—review and editing, all authors; visualization, L.J.D.; supervision, D.J.C.; All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
This project did not require ethical approval however was approved by Aintree University Hospital Local Audit Committee (number 10864).
Informed Consent Statement
Patient consent was not required for this study, however we obtained local approval from Aintree University Hospital Audit Committee (Number: 10864).
Data Availability Statement
Data is available upon reasonable request to the authors.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Younossi Z.M., Koenig A.B., Abdelatif D., Fazel Y., Henry L., Wymer M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology. 2016;64:73–84. doi: 10.1002/hep.28431. [DOI] [PubMed] [Google Scholar]
- 2.Estes C., Razavi H., Loomba R., Younossi Z., Sanyal A.J. Modeling the epidemic of nonalcoholic fatty liver disease demonstrates an exponential increase in burden of disease. Hepatology. 2018;67:123–133. doi: 10.1002/hep.29466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Eslam M., Sanyal A.J., George J., Sanyal A., Neuschwander-Tetri B., Tiribelli C., Kleiner D.E., Brunt E., Bugianesi E., Yki-Järvinen H., et al. MAFLD: A Consensus-Driven Proposed Nomenclature for Metabolic Associated Fatty Liver Disease. Gastroenterology. 2020;158:1999–2014.e1. doi: 10.1053/j.gastro.2019.11.312. [DOI] [PubMed] [Google Scholar]
- 4.DeFronzo R.A., Ferrannini E., Groop L., Henry R.R., Herman W.H., Holst J.J., Hu F.B., Kahn C.R., Raz I., Shulman G.I., et al. Type 2 diabetes mellitus. Nat. Rev. Dis. Primers. 2015;1:15019. doi: 10.1038/nrdp.2015.19. [DOI] [PubMed] [Google Scholar]
- 5.Tomah S., Alkhouri N., Hamdy O. Non-alcoholic Fatty Liver Disease and Type 2 Diabetes: Where do Diabetologists stand? Clin. Diabetes Endocrinol. 2020;6:9. doi: 10.1186/s40842-020-00097-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.De A., Duseja A. Natural History of Simple Steatosis or Nonalcoholic Fatty Liver. J. Clin. Exp. Hepatol. 2020;10:255–262. doi: 10.1016/j.jceh.2019.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Singh S., Allen A.M., Wang Z., Prokop L.J., Murad M.H., Loomba R. Fibrosis Progression in Nonalcoholic Fatty Liver vs Nonalcoholic Steatohepatitis: A Systematic Review and Meta-analysis of Paired-Biopsy Studies. Clin. Gastroenterol. Hepatol. 2015;13:643–654.e9. doi: 10.1016/j.cgh.2014.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kisseleva T., Brenner D. Molecular and cellular mechanisms of liver fibrosis and its regression. Nat. Rev. Gastroenterol. Hepatol. 2021;18:151–166. doi: 10.1038/s41575-020-00372-7. [DOI] [PubMed] [Google Scholar]
- 9.Dulai P.S., Singh S., Patel J., Soni M., Prokop L.J., Younossi Z., Sebastiani G., Ekstedt M., Hagstrom H., Nasr P., et al. Increased risk of mortality by fibrosis stage in nonalcoholic fatty liver disease: Systematic review and meta-analysis. Hepatology. 2017;65:1557–1565. doi: 10.1002/hep.29085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kim D., Kim W.R., Kim H.J., Therneau T.M. Association between noninvasive fibrosis markers and mortality among adults with nonalcoholic fatty liver disease in the United States. Hepatology. 2013;57:1357–1365. doi: 10.1002/hep.26156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Simon T., Corey K., Cannon C., Blazing M., Park J., O’Donoghue M., Chung R., Giugliano R. The nonalcoholic fatty liver disease (NAFLD) fibrosis score, cardiovascular risk stratification and a strategy for secondary prevention with ezetimibe. Int. J. Cardiol. 2018;270:245–252. doi: 10.1016/j.ijcard.2018.05.087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Baratta F., Pastori D., Angelico F., Balla A., Paganini A.M., Cocomello N., Ferro D., Violi F., Sanyal A.J., del Ben M. Nonalcoholic Fatty Liver Disease and Fibrosis Associated With Increased Risk of Cardiovascular Events in a Prospective Study. Clin. Gastroenterol. Hepatol. 2020;18:2324–2331.e4. doi: 10.1016/j.cgh.2019.12.026. [DOI] [PubMed] [Google Scholar]
- 13.Adams L.A., Sanderson S., Lindor K.D., Angulo P. The histological course of nonalcoholic fatty liver disease: A longitudinal study of 103 patients with sequential liver biopsies. J. Hepatol. 2005;42:132–138. doi: 10.1016/j.jhep.2004.09.012. [DOI] [PubMed] [Google Scholar]
- 14.Koehler E.M., Plompen E.P.C., Schouten J.N.L., Hansen B.E., Darwish Murad S., Taimr P., Leebeek F.W.G., Hofman A., Stricker B.H., Castera L., et al. Presence of diabetes mellitus and steatosis is associated with liver stiffness in a general population: The Rotterdam study. Hepatology. 2016;63:138–147. doi: 10.1002/hep.27981. [DOI] [PubMed] [Google Scholar]
- 15.Bril F., Cusi K. Management of Nonalcoholic Fatty Liver Disease in Patients with Type 2 Diabetes: A Call to Action. Diabetes Care. 2017;40:419–430. doi: 10.2337/dc16-1787. [DOI] [PubMed] [Google Scholar]
- 16.Doycheva I., Cui J., Nguyen P., Costa E.A., Hooker J., Hofflich H., Bettencourt R., Brouha S., Sirlin C.B., Loomba R. Non-invasive screening of diabetics in primary care for NAFLD and advanced fibrosis by MRI and MRE. Aliment. Pharmacol. Ther. 2016;43:83–95. doi: 10.1111/apt.13405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.McPherson S., Hardy T., Henderson E., Burt A.D., Day C.P., Anstee Q.M. Evidence of NAFLD progression from steatosis to fibrosing-steatohepatitis using paired biopsies: Implications for prognosis and clinical management. J. Hepatol. 2015;62:1148–1155. doi: 10.1016/j.jhep.2014.11.034. [DOI] [PubMed] [Google Scholar]
- 18.Ekstedt M., Franzén L.E., Mathiesen U.L., Thorelius L., Holmqvist M., Bodemar G., Kechagias S. Long-term follow-up of patients with NAFLD and elevated liver enzymes. Hepatology. 2006;44:865–873. doi: 10.1002/hep.21327. [DOI] [PubMed] [Google Scholar]
- 19.Liu J., Ayada I., Zhang X., Wang L., Li Y., Wen T., Ma Z., Bruno M.J., de Knegt R.J., Cao W., et al. Estimating global prevalence of metabolic dysfunction-associated fatty liver diseasein overweight or obese adults. Clin. Gastroenterol. Hepatol. 2021 doi: 10.1016/j.cgh.2021.02.030. in press. [DOI] [PubMed] [Google Scholar]
- 20.Anstee Q.M., Targher G., Day C.P. Progression of NAFLD to diabetes mellitus, cardiovascular disease or cirrhosis. Nat. Rev. Gastroenterol. Hepatol. 2013;10:330–344. doi: 10.1038/nrgastro.2013.41. [DOI] [PubMed] [Google Scholar]
- 21.Younossi Z.M., Golabi P., de Avila L., Paik J.M., Srishord M., Fukui N., Qiu Y., Burns L., Afendy A., Nader F. The global epidemiology of NAFLD and NASH in patients with type 2 diabetes: A systematic review and meta-analysis. J. Hepatol. 2019;71:793–801. doi: 10.1016/j.jhep.2019.06.021. [DOI] [PubMed] [Google Scholar]
- 22.Public Health England Public Health England Diabetes Statistics. [(accessed on 1 November 2021)]. Available online: https://fingertips.phe.org.uk/profile/diabetes-ft/data#page/0.
- 23.European Association for the Study of the Liver (EASL) European Association for the Study of Diabetes (EASD) European Association for the Study of Obesity (EASO) EASL–EASD–EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J. Hepatol. 2016;64:1388–1402. doi: 10.1016/j.jhep.2015.11.004. [DOI] [PubMed] [Google Scholar]
- 24.Association A.D. Comprehensive medical evaluation and assessment of comorbidities: Standards of Medical Care in Diabetes-2020. Diabetes Care. 2020;43:S37–S47. doi: 10.2337/dc20-S004. [DOI] [PubMed] [Google Scholar]
- 25.Chalasani N., Younossi Z., Lavine J.E., Charlton M., Cusi K., Rinella M., Harrison S.A., Brunt E.M., Sanyal A.J. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018;67:328–357. doi: 10.1002/hep.29367. [DOI] [PubMed] [Google Scholar]
- 26.Wong V.W.S., Chan W.K., Chitturi S., Chawla Y., Dan Y.Y., Duseja A., Fan J., Goh K.L., Hamaguchi M., Hashimoto E., et al. Asia–Pacific Working Party on Non-alcoholic Fatty Liver Disease guidelines 2017—Part 1: Definition, risk factors and assessment. J. Gastroenterol. Hepatol. 2018;33:70–85. doi: 10.1111/jgh.13857. [DOI] [PubMed] [Google Scholar]
- 27.National Guideline Centre (UK) Non-Alcoholic Fatty Liver Disease (NAFLD): Assessment and Management. National Institute for Health and Care Excellence; London, UK: 2016. [PubMed] [Google Scholar]
- 28.National Guideline Centre (UK) Cirrhosis in over 16s: Assessment and Management. National Institute for Health and Care Excellence; London, UK: 2016. [PubMed] [Google Scholar]
- 29.Sterling R.K., Lissen E., Clumeck N., Sola R., Correa M.C., Montaner J., Sulkowski M.S., Torriani F.J., Dieterich D.T., Thomas D.L., et al. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology. 2006;43:1317–1325. doi: 10.1002/hep.21178. [DOI] [PubMed] [Google Scholar]
- 30.Wai C., Greenson J.K., Fontana R.J., Kalbfleisch J.D., Marrero J.A., Conjeevaram H.S., Lok A.S.-F. A simple noninvasive index can predict both significant fibrosis and cirrhosis in patients with chronic hepatitis C. Hepatology. 2003;38:518–526. doi: 10.1053/jhep.2003.50346. [DOI] [PubMed] [Google Scholar]
- 31.Williams A.L., Hoofnagle J.H. Ratio of serum aspartate to alanine aminotransferase in chronic hepatitis. Relationship to cirrhosis. Gastroenterology. 1988;95:734–739. doi: 10.1016/S0016-5085(88)80022-2. [DOI] [PubMed] [Google Scholar]
- 32.Lichtinghagen R., Pietsch D., Bantel H., Manns M.P., Brand K., Bahr M.J. The Enhanced Liver Fibrosis (ELF) score: Normal values, influence factors and proposed cut-off values. J. Hepatol. 2013;59:236–242. doi: 10.1016/j.jhep.2013.03.016. [DOI] [PubMed] [Google Scholar]
- 33.Singh A., Le P., Peerzada M.M., Lopez R., Alkhouri N. The Utility of Noninvasive Scores in Assessing the Prevalence of Nonalcoholic Fatty Liver Disease and Advanced Fibrosis in Type 2 Diabetic Patients. J. Clin. Gastroenterol. 2018;52:268–272. doi: 10.1097/MCG.0000000000000905. [DOI] [PubMed] [Google Scholar]
- 34.Lomonaco R., Leiva E.G., Bril F., Shrestha S., Mansour L., Budd J., Romero J.P., Schmidt S., Chang K.-L., Samraj G., et al. Advanced Liver Fibrosis Is Common in Patients With Type 2 Diabetes Followed in the Outpatient Setting: The Need for Systematic Screening. Diabetes Care. 2021;44:399–406. doi: 10.2337/dc20-1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mansour D., Grapes A., Herscovitz M., Cassidy P., Vernazza J., Broad A., Anstee Q.M., McPherson S. Embedding assessment of liver fibrosis into routine diabetic review in primary care. JHEP Rep. 2021;3:100293. doi: 10.1016/j.jhepr.2021.100293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Robertson R.P., Harmon J., Tran P.O.T., Poitout V. β-cell glucose toxicity, lipotoxicity, and chronic oxidative stress in type 2 diabetes. Diabetes. 2004;53((Suppl. 1)):S119–S124. doi: 10.2337/diabetes.53.2007.S119. [DOI] [PubMed] [Google Scholar]
- 37.Smith G.I., Shankaran M., Yoshino M., Schweitzer G.G., Chondronikola M., Beals J.W., Okunade A.L., Patterson B.W., Nyangau E., Field T., et al. Insulin resistance drives hepatic de novo lipogenesis in nonalcoholic fatty liver disease. J. Clin. Investig. 2020;130:1453–1460. doi: 10.1172/JCI134165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Samuel V.T., Liu Z.X., Qu X., Elder B.D., Bilz S., Befroy D., Romanelli A.J., Shulman G.I. Mechanism of hepatic insulin resistance in non-alcoholic fatty liver disease. J. Biol. Chem. 2004;279:32345–32353. doi: 10.1074/jbc.M313478200. [DOI] [PubMed] [Google Scholar]
- 39.Kantartzis K., MacHann J., Schick F., Fritsche A., Häring H.U., Stefan N. The impact of liver fat vs visceral fat in determining categories of prediabetes. Diabetologia. 2010;53:882–889. doi: 10.1007/s00125-010-1663-6. [DOI] [PubMed] [Google Scholar]
- 40.Lambert J.E., Ramos-Roman M.A., Browning J.D., Parks E.J. Increased de novo lipogenesis is a distinct characteristic of individuals with nonalcoholic fatty liver disease. Gastroenterology. 2014;146:726–735. doi: 10.1053/j.gastro.2013.11.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Berzigotti A., Tsochatzis E., Boursier J., Castera L., Cazzagon N., Friedrich-Rust M., Petta S., Thiele M. EASL Clinical Practice Guidelines on non-invasive tests for evaluation of liver disease severity and prognosis–2021 update. J. Hepatol. 2021;75:659–689. doi: 10.1016/j.jhep.2021.05.025. [DOI] [PubMed] [Google Scholar]
- 42.Rousseau M.C., Parent M.É., Pollak M.N., Siemiatycki J. Diabetes mellitus and cancer risk in a population-based case-control study among men from Montreal, Canada. Int. J. Cancer. 2006;118:2105–2109. doi: 10.1002/ijc.21600. [DOI] [PubMed] [Google Scholar]
- 43.Wang P., Kang D., Cao W., Wang Y., Liu Z. Diabetes mellitus and risk of hepatocellular carcinoma: A systematic review and meta-analysis. Diabetes. Metab. Res. Rev. 2012;28:109–122. doi: 10.1002/dmrr.1291. [DOI] [PubMed] [Google Scholar]
- 44.Vilar-Gomez E., Martinez-Perez Y., Calzadilla-Bertot L., Torres-Gonzalez A., Gra-Oramas B., Gonzalez-Fabian L., Friedman S.L., Diago M., Romero-Gomez M. Weight Loss Through Lifestyle Modification Significantly Reduces Features of Nonalcoholic Steatohepatitis. Gastroenterology. 2015;149:367–378.e5. doi: 10.1053/j.gastro.2015.04.005. [DOI] [PubMed] [Google Scholar]
- 45.Brown E., Hydes T., Hamid A., Cuthbertson D. Emerging and Established Therapeutic Approaches for Nonalcoholic Fatty Liver Disease. Clin. Ther. 2021;43:1476–1504. doi: 10.1016/j.clinthera.2021.07.013. [DOI] [PubMed] [Google Scholar]
- 46.Allen A.M., Hicks S.B., Mara K.C., Larson J.J., Therneau T.M. The risk of incident extrahepatic cancers is higher in non-alcoholic fatty liver disease than obesity—A longitudinal cohort study. J. Hepatol. 2019;71:1229–1236. doi: 10.1016/j.jhep.2019.08.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Cui J., Philo L., Nguyen P., Hofflich H., Hernandez C., Bettencourt R., Richards L., Salotti J., Bhatt A., Hooker J., et al. Sitagliptin vs. placebo for non-alcoholic fatty liver disease: A randomized controlled trial. J. Hepatol. 2016;65:369–376. doi: 10.1016/j.jhep.2016.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Macauley M., Hollingsworth K.G., Smith F.E., Thelwall P.E., Al-Mrabeh A., Schweizer A., Foley J.E., Taylor R. Effect of Vildagliptin on Hepatic Steatosis. J. Clin. Endocrinol. Metab. 2015;100:1578–1585. doi: 10.1210/jc.2014-3794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cuthbertson D.J., Irwin A., Gardner C.J., Daousi C., Purewal T., Furlong N., Goenka N., Thomas E.L., Adams V.L., Pushpakom S.P., et al. Improved Glycaemia Correlates with Liver Fat Reduction in Obese, Type 2 Diabetes, Patients Given Glucagon-Like Peptide-1 (GLP-1) Receptor Agonists. PLoS ONE. 2012;7:e50117. doi: 10.1371/journal.pone.0050117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Newsome P.N., Buchholtz K., Cusi K., Linder M., Okanoue T., Ratziu V., Sanyal A.J., Sejling A.-S., Harrison S.A. A Placebo-Controlled Trial of Subcutaneous Semaglutide in Nonalcoholic Steatohepatitis. N. Engl. J. Med. 2021;384:1113–1124. doi: 10.1056/NEJMoa2028395. [DOI] [PubMed] [Google Scholar]
- 51.Armstrong M.J., Houlihan D.D., Rowe I.A., Clausen W.H.O., Elbrønd B., Gough S.C.L., Tomlinson J.W., Newsome P.N. Safety and efficacy of liraglutide in patients with type 2 diabetes and elevated liver enzymes: Individual patient data meta-analysis of the LEAD program. Aliment. Pharmacol. Ther. 2013;37:234–242. doi: 10.1111/apt.12149. [DOI] [PubMed] [Google Scholar]
- 52.Sattar N., Fitchett D., Hantel S., George J.T., Zinman B. Empagliflozin is associated with improvements in liver enzymes potentially consistent with reductions in liver fat: Results from randomised trials including the EMPA-REG OUTCOME® trial. Diabetologia. 2018;61:2155–2163. doi: 10.1007/s00125-018-4702-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Eriksson J.W., Lundkvist P., Jansson P.-A., Johansson L., Kvarnström M., Moris L., Miliotis T., Forsberg G.-B., Risérus U., Lind L., et al. Effects of dapagliflozin and n-3 carboxylic acids on non-alcoholic fatty liver disease in people with type 2 diabetes: A double-blind randomised placebo-controlled study. Diabetologia. 2018;61:1923–1934. doi: 10.1007/s00125-018-4675-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Grecian S.M., McLachlan S., Fallowfield J.A., Kearns P.K.A., Hayes P.C., Guha N.I., Morling J.R., Glancy S., Williamson R.M., Reynolds R.M., et al. Non-invasive risk scores do not reliably identify future cirrhosis or hepatocellular carcinoma in Type 2 diabetes: The Edinburgh type 2 diabetes study. Liver Int. 2020;40:2252–2262. doi: 10.1111/liv.14590. [DOI] [PubMed] [Google Scholar]
- 55.Morling J.R., Fallowfield J.A., Guha I.N., Nee L.D., Glancy S., Williamson R.M., Robertson C.M., Strachan M.W.J., Price J.F. Using non-invasive biomarkers to identify hepatic fibrosis in people with type 2 diabetes mellitus: The Edinburgh type 2 diabetes study. J. Hepatol. 2014;60:384–391. doi: 10.1016/j.jhep.2013.10.017. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data is available upon reasonable request to the authors.