Abstract
Arbuscular mycorrhizal fungi (AMF), which form symbioses with most land plants, could benefit their hosts and potentially play important roles in revegetation of degraded lands. However, their application in revegetation of desert grasslands still faces challenges and uncertainties due to the unclear specificity of AMF-plant interactions. Here, Caragana korshinskii and Caragana microphylla were inoculated with either conspecific (home) or heterospecific (away) AM fungal communities from the rhizosphere of three common plant species (C. korshinskii, C. microphylla and Hedysarum laeve) in Kubuqi Desert, China. AMF communities of the inocula and their home and away effects on growth and nutrition status of two Caragana species were examined. Results showed that AMF communities of the three inocula from C. korshinskii, H. laeve and C. microphylla were significantly different, and were characterized by high abundance of Diversispora, Archaeospora, and Glomus, respectively. The shoot biomass, photosynthetic rate, foliar N and P contents of C. korshinskii only significantly increased under home AMF inoculation by 167.10%, 73.55%, 9.24%, and 23.87%, respectively. However, no significant effects of AMF on C. microphylla growth were found, regardless of home or away AMF. Positive correlations between C. korshinskii biomass and the abundance of AMF genus Diversispora were found. Our study showed strong home advantage of using native AMF community to enhance C. korshinskii growth in the desert and presented a potentially efficient way to use native AMF in restoration practices.
Keywords: arbuscular mycorrhizal fungi, inoculum source, home advantage, Caragana, desert grassland
1. Introduction
Arbuscular mycorrhizal fungi (AMF) provide benefits to their host plants [1,2] and show functional diversity assumed to have implications for revegetation of degraded lands [3,4,5]. By forming biotrophic symbiosis with plants, AMF could enhance plant uptake of relatively immobile nutrients, particularly phosphorus, and several micronutrients [6,7,8]. Besides, AMF could also provide a large number of other benefits to host plants such as maintaining and ameliorating soil structure, pathogen and herbivory protection, alleviation of drought and salinity stress [9,10,11,12,13].
However, the benefit of AMF in terms of increased plant growth is not always evident [14,15]. Neutral or negative effects of AMF on plant growth in both greenhouse [16,17] and field experiments [18] were reported. Furthermore, the effects of AMF on plant growth seemed to be host specific [19]. The AMF-plant symbiosis is not formed stochastically, but depends on their functional traits [20,21]. Both plant hosts and AMF have been shown to preferentially allocate resources to higher quality partners [22]. Consequently, due to the simultaneous processes of coevolution and bi-direction selection, the plant-AMF usually achieved good match for each other. In addition, the behavior of AMF was also dramatically affected by local soil properties, such as soil texture and structure, organic matter, and pH [23]. Thus, theoretically, the native AMF should benefit the local plant better than that from foreign habitats, which has been reported previously [4]. For example, native AMF enhanced the growth of Sorghastrum nutans more than fungi from an alternate system in the greenhouse [24]. However, the knowledge on the host specific benefits provided by AMF to plants obtained from greenhouses should be further tested in field studies.
Desertification is one of the most serious global environment problems, especially in China [25]. Specifically, 2.62 million km2 of the drylands, accounting for 27.33% of the country’s territorial area, is desertified and caused serious ecological problems [26]. In such desert land, characterized by low plant cover and soil fertility, the below-ground microorganisms were severely disturbed resulting in a relatively low level of beneficial soil microbes, such as AMF [27,28]. Usually, in northern China, most shrubs in arid regions are prevalently colonized by AMF and the symbiosis is critical to their survival and regrowth [29,30,31]. With the loss of AMF after desertification, the growth of the shrubs might be dampened [32,33]. Caragana korshinskii and Caragana microphylla are common brushes of the desert in northern China and are frequently used for revegetation due to its well adaptation to water and nutrient deficit [34,35]. Furthermore, they are extensively colonized by AMF and host specific interactions between C. microphylla and AMF was detected in desertified grasslands in northern China [36], indicating the potential importance of AMF in benefiting their growth. However, the effects of AMF on revegetation of C. korshinskii and C. microphylla are still not clear in practice. Thus, field experimental tests of the effectiveness of AMF on plant growth in desertified area and their proper applications are an urgent demand [3,4,5].
Here we investigate the inoculation effect of home (i.e., conspecific AMF inoculation) and away (i.e., heterospecific AMF inoculation) AMF on the growth of C. korshinskii and C. microphylla in Kubuqi Desert continuously for three years. The Kubuqi Desert is the seventh largest desert and is a major dust source in China. The home and away AMF inocula were obtained from the roots and soils of three host species (C. korshinskii, C. microphylla and Hedysarum laeve) in the desert. We hypothesized that: (1) AMF inoculation could enhance the plant performance in the desert; (2) AMF inocula from the rhizosphere of different host species differed in their ability to promote plant growth, and specifically, plants would grow better under home AMF relative to away AMF.
2. Materials and Methods
2.1. Study Site
The study site was located in the eastern part of the Kubuqi Desert in Zhungeer Banner in the north of the Erdos Plateau in Inner Mongolia, China (40°04′47.13″ N, 110°46′34.83″ E). The mean annual temperature here is 6.2–8.7 °C and annual precipitation is 420 mm with more than 60% of the precipitation occurring from July to September. The soil texture is arenosol and contained 1.0 g kg−1 soil organic carbon. The total N and available P were 0.09 g kg−1 and 4.36 mg kg−1 respectively. The site was previously dominated by natural occurring Artemisia Ordosicaonce but with a very low plant coverage (<10%).
2.2. Trap Culture of AMF
In June 2014, 10 individuals of C. korshinskii, H. laeve and C. microphylla with similar growth conditions were selected at the study site. Samples containing root and rhizosphere soils of each individual were collected from the top 0–15 cm using soil auger with 5-cm diameter and were mixed for each plant species. AMF spore density in these samples were measured by wet-sieving (paired sieves: 750 and 38 μm) and sucrose-gradient centrifugation [27]. The spore density was 133 ± 26, 123 ± 38, and 98 ± 47 spores per 100 g soil for C. korshinskii, H. laeve and C. microphylla, respectively (Figure S1). Desert soil samples nearby were also collected at the same time and steam-sterilized (121 °C, 1 h, twice) to serve as growth substrate for following trap culture of AMF. The collected root and rhizosphere soil samples of the three shrubs containing AMF community were mixed with sterilized growth substrate in a 1:2 ratio (v/v) in 20 sterilized plastic pots (20 cm diameter ×15 cm depth), respectively. Then, 20 seeds of maize (sterilized with 75% alcohol) were sown on the surface of each plastic pots. To equalize microbial communities of the trap culture soils except AMF, soil sievates containing microbes except AMF from the three shrubs were added to the pots. The field soil sievates were obtained by blending the collected root and soil samples and water in a 1:2 ratio and passing the slurry through a 38 μm sieve. The relatively large AM fungal spores and hyphae were trapped on the sieve, while smaller organisms passed through [37]. All trap culture pots were amended with 150 mL non-sterilized soil sievates, with 50 mL from each of the three species. Supplied with tap water when needed, maize was grown for 14 weeks at a temperature of 25 °C in a greenhouse to obtain enough AMF spores. Then, the above ground parts of the maize were removed, and all contents underground were harvested separately as inocula for the following experiment. Maize roots were removed from the soil, cut into 1 cm lengths, and then mixed back with the soil. So, the inocula from C. korshinskii (A), H. leave (B) and C. microphylla (C) consisted of AM-colonized root pieces, spores, and hyphae originated. The number of spores in the inocula A, B and C were 382 ± 57, 325 ± 33, and 367 ± 76 AMF spores per 100 g inoculum respectively (Figure S1). No significant differences in the spore density were detected among them. Though AM fungal communities could change during the trap culture, the influence was minor considering the relative short-term conditioning time [38].
2.3. Seedling Preparation
Seeds of C. korshinskii and C. microphylla were collected at the study site at the same time as soil sampling. In 2016, 1000 plump seeds were disinfected with 75% alcohol, washed with sterilized tap water and then pre-germinated in 100 pots (i.e., 10 seeds per pot) for each shrub using the same sterilized growth substrate as in the AMF trap culture. After germination, the number of seedlings in each pot was thinned to 3 and were cultivated in a greenhouse for 2 months from May to June to make sure that seedlings were strong enough to survive after transplanting to the field.
2.4. Field Experiment Design
In the July of 2016, an area of 8 m × 15 m was fenced in the study site to setup the field experiment. Plants grown in the fenced area were removed manually, and the field was tilled to potentially disrupt the hyphae of AMF and create a uniform environment. Then the fenced area was separated to 112 subplots (0.5 m length × 0.5 m width × 0.1 m depth) with 0.5 m distance from each other.
The seedlings with a good status and nearly equal height (about 15 cm) obtained from seedling preparation were transplanted to field site. Each subplot received 3 C. korshinskii or C. microphylla seedlings randomly. AMF inocula from the trap culture were applied at the time of seedling transplantation. Briefly, a small hole was dug by sterilized spade, and 100 g of inoculum (average 358 AMF spores, Figure S1) were put on the bottom and then seedlings were placed on the top of the inoculum. Weeds in the field were controlled manually twice a year in June and September. Thus, the field experiment consisted of 2 factors including 2 host species and 4 AMF inocula with a replicate of 14. There were 112 microcosms in total in this study (i.e., 2 host species × 4 AMF inocula × 14 replicates). The 4 AMF treatments included inocula originated from C. korshinskii (A), H. leave (B) and C. microphylla (C), adding sterilized-inocula (mixture of A, B, and C) as control (CK).
2.5. Field Measurements
In July 2017 and 2018, the height and canopy diameter of plants were measured by steel tape. Gas-exchange characteristics, including photosynthetic rate, transpiration rate, and stomatal conductance (GH2O) were measured on leaves in situ with a portable leaf gas exchange system (GFS-3000, Walz GmbH, Effeltrich, Bavaria, Germany). All gas-exchange characteristics were measured between 9:00 and 11:30 (24 h) on sunny days and data were measured at a temperature between 25–30 °C, relative humidity about 70%, and CO2 concentration inside the chamber of ambient level approximate 400 ppm.
In July 2017, soil cores (5 cm diameter) were collected to a depth of 20 cm in the root area of the plants. For each treatment, 10 of 14 replicates with nearly equal plant height were sampled adding up to a total of 80 samples. For each sample, the plant roots were collected and transferred to the lab with ice. After gentle washing of the roots with tap water, a subset of the roots was stained with trypan blue and checked for AMF colonization rate using the magnified gridline intersect method [39]. An additional subset of roots was stored at −20 °C for subsequent molecular analysis.
Plants were harvested in August 2019. Plant height was measured before harvest and dry (65 °C, 72 h) weights of shoots were recorded to determine the average shoot biomass per individual. Dried leaf and shoot tissues were ground to powder before analyzing for total N and P. Foliar P was determined colorimetrically after digestion with sulfuric acid using the molybdenum blue method [40] and foliar N was determined by the Kjeldahl method [41].
2.6. DNA Extraction and Sequencing
Total DNA of the root samples was extracted by the CTAB (cetyltrimethylammonium bromide) protocol [42]. Briefly, roots were cut into 1 cm pieces and 20 fragments of them were randomly picked up for DNA extraction. We also extracted DNA from 0.25 g fresh soil samples of three AMF inocula after trap culture. 5 replicates of each inoculum were extracted using DNA extraction kits (DNeasy PowerSoil, Qiagen, Valencia, CA, USA) according to the manufacturer’s protocol.
Glomeromycotina sequences were amplified by nested PCR with the SSU rRNA gene primers NS31-AML2 and AMV4.5NF–AMDGR as previous study [43]. We used the Quantitative Insights into Microbial Ecology (QIIME v1.7.0) [44] and the UPARSE pipeline [45] to treat raw sequences. In brief, sequences below the quality score of 20 and fewer than 200 bp in length were excluded. Primer-free sequences were de-replicated and chimeric sequences were removed. The sequences were binned into operational taxonomic units (OTUs) with 97% similarity in USEARCH version 11 [46], and the most abundant sequence from each OTU was selected as a representative sequence for that OTU. Then, a Basic Local Alignment Search Tool (BLAST) search against the GenBank database at the National Center for Biotechnology Information (NCBI) to detect non Glomeromycotina sequences. Rare OTUs with relative abundance less than 0.01% (total abundance less than 510) were also removed, resulting 100 abundant AMF OTUs for downstream analysis. Representative sequences of each OTU in this study were deposited in GenBank (MK318664-MK318763). Further, the BLAST function was used to retrieve closely related reference Glomeromycotina sequences for the representative sequences of OTUs. Neighbor-joining (NJ) phylogenetic analysis [47] was computed in MEGA version 7 with 1000 bootstrap replicates to evaluate the tree and with Henningsomyces candidus as outgroup to root the tree.
2.7. Statistic Analysis
Statistical analysis was conducted using R (version 3.5.0). Analysis of variance (ANOVA) was performed to test for differences in plant responses to each inoculation treatment of the two Caragana plants, followed by Tukey’s HSD post-hoc analysis. Within the one-way ANOVA, multiple comparisons were done with orthogonal, a priori contrasts (inoculated vs. un-inoculated and home vs. away) to test if there was a net inoculation effect overall and if there was a home advantage under home AMF and plant combinations.
AM fungal community’s Shannon–Wiener diversity and species richness were calculated using the vegan package [48]. Indicator species analysis of AMF was performed using indicspecies [49]. Permutational multivariate analysis (PERMANOVA) was used to test whether host species (C. korshinskii and C. microphylla) and inoculum sources (A, B, C, and CK) explained differences in AM fungal community (sequence number dataset, wisconsin-square root transformed), and we visualized these differences at a community level using nonmetric multidimensional scaling (NMDS) using Bray–Curtis distances. Linear regression was performed to explore relationships between plant variables and AMF abundance. Additionally, to estimate the causal effects of AMF inoculation on host plant performance, structural equation models (SEM) were calculated using SPSS Amos v. 23.0.
3. Result
3.1. AM Fungal Community Properties
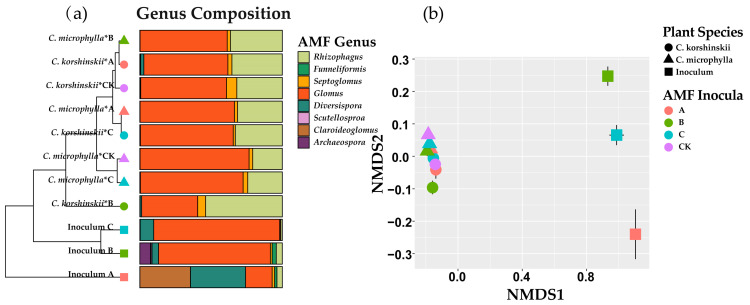
All samples of inocula yielded positive PCR products of the expected size (250 bp) and the rarefaction curve suggested that our sampling captured most of the community (Figure S2). Differences in Shannon diversity (F = 3.848, p = 0.058) and composition of AMF community (R2 = 0.400, p = 0.075) were detected among the three inocula. Inoculum B showed the highest AMF diversity relative to inoculum A and C (Figure S3). 100 OTUs could be assigned to putative AMF and all the identified OTUs were affiliated with genus levels within the families Archaeosporaceae, Claroideoglomeraceae, Diversisporaceae, Gigasporaceae, and Glomeraceae. Diversispora was significantly enriched in inoculum A (Figure S4), but Archaeospora was significantly enriched in inoculum B. Glomus was significantly more abundant in inoculum C (Table S1). The NMDS analysis also showed the dramatically different in AM fungal community among the three inocula (Figure 1).
Figure 1.
Relative abundance of AM fungal genus in original inocula (A: C. korshinskii, B: H. leave, C: C. microphylla, and CK: sterilized inocula) and plant roots after one year of growth (a), and non-metric multidimensional scaling (NMDS) ordination plot illustrating differences in AMF OTU composition among the original inocula and plant roots (b).
In addition, 92 of 100 AMF OTUs were found associated with Caragana roots. Though AMF richness was neither influenced by host plant species nor inoculum sources, the Shannon diversity was significantly influenced by inoculum sources (F = 5.027, p = 0.003) and their interaction (F = 3.901, p = 0.012) (Figure S5). The community structure of AMF associated with C. korshinskii and C. microphylla roots was significantly influenced by both inoculum sources (F = 1.558, p = 0.033) and plant species (F = 8.528, p = 0.001) according to PERMANOVA result. AM fungal communities inside C. korshinskii and C. microphylla roots were significantly different.
3.2. Plant Response to AMF Inoculation
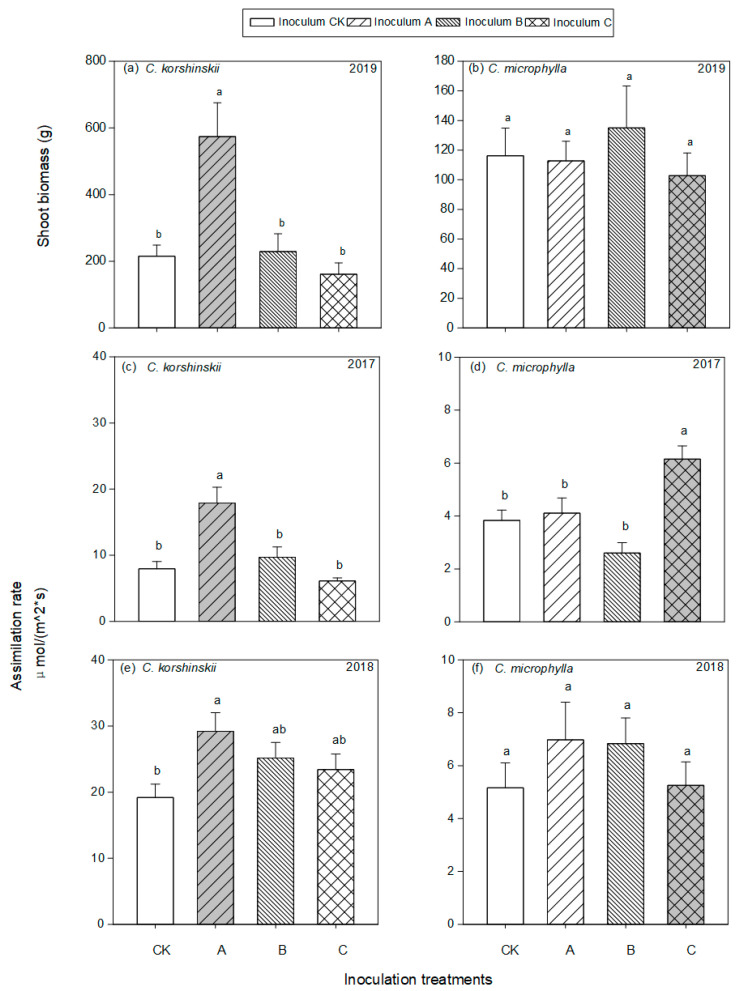
Host species, AMF inoculation, and experimental year showed significant effects on most of the plant measurements (Table 1). C. korshinskii was significantly higher in shoot biomass (F = 25.276, p < 0.001) relative to C. microphylla, while foliar N (F = 53.833, p < 0.001) and P (F = 9.538, p = 0.003) showed reverse pattern (Table S2). AMF inoculation significantly increased the shoot biomass and photosynthetic rate of C. korshinskii by 49.53%, and 26.96% relative to CK, respectively. However, no significant effects of AMF inoculation were found for C. microphylla, except for increased photosynthetic rate by 11.74% in 2017 (Figure 2).
Table 1.
Results of three-way analysis of variances showing the effects of host plant species, AMF inoculation, year and their interactions on plant performance.
| Height | Canopy | Photosynthetic Rate |
Transpiration Rate |
Vapor Pressure Deficit |
Stomatal Conductance |
Colonization Rate |
Shoot Biomass |
Foliar N | Foliar P | Foliar N:P | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Host | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | 0.2394 | <0.001 | <0.001 | 0.003 | 0.011 |
| Inoculation | <0.001 | 0.001 | 0.001 | 0.189 | 0.000 | 0.002 | 0.0241 | <0.001 | 0.102 | 0.163 | 0.076 |
| Year | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |||||
| Host:Inoculation | <0.001 | 0.001 | 0.029 | 0.026 | 0.501 | 0.015 | 0.0646 | 0.0119 | 0.0406 | 0.0130 | 0.5085 |
| Host:Year | <0.001 | 0.012 | <0.001 | <0.001 | <0.001 | 0.002 | |||||
| Inoculation:Year | 0.026 | 0.001 | 0.584 | 0.435 | 0.307 | 0.213 | |||||
| Host:Inoculation:Year | 0.769 | 0.341 | 0.035 | 0.151 | 0.033 | 0.109 |
Figure 2.
Shoot biomass (a,b) and photosynthetic rate (c–f) of C. korshinskii and C. microphylla plants in 3 years. Inoculation treatments including inocula originating from rhizosphere of C. korshinskii (A), H. leave (B), C. microphylla (C) and sterilized control (CK). Bars indicate means with standard error (n = 7 for shoot biomass and n = 14 for photosynthetic rate). Different letters above bars indicated significant differences at p < 0.05 level according to Tukey’s test. Grey bars indicated home AMF and plant combinations.
Furthermore, the different responses of C. korshinskii to home and away AMF inoculation was found (Table 2). Home AMF significantly increased the shoot biomass, foliar N, and foliar P of C. korshinskii by 194.35%, 15.20%, and 25.69% relative to away AMF inoculation, respectively (p < 0.001, p = 0.002, p = 0.009). However, no significant differences in C. microphylla responses to home and away AMF inoculation, except for increased photosynthetic rate in 2017 (p < 0.001) and Foliar N:P (p = 0.003).
Table 2.
Results of orthogonal contrasts for photosynthetic rate, biomass, and nutrient content of Caragana species among inoculated or un-inoculated and inoculated with home AMF (home) or other inocula (away). ‘+/−’ means a positive/negative effect of AMF inoculation or home advantage of conspecific inocula. Statistically significant effects (P) were shown behind.
| C. korshinskii | C. microphylla | |||||||
|---|---|---|---|---|---|---|---|---|
| Inoculated vs. Un-Inoculated | Home vs. Away | Inoculated vs. Un-Inoculated | Home vs. Away | |||||
| Shoot biomass | + | 0.144 | + | <0.001 | + | 0.977 | − | 0.38 |
| Photosynthetic rate (2017) | + | 0.132 | + | <0.001 | + | 0.376 | + | <0.001 |
| Photosynthetic rate (2018) | + | 0.017 | + | 0.102 | − | 0.426 | − | 0.144 |
| Foliar N | + | 0.854 | + | 0.002 | − | 0.002 | + | 0.089 |
| Foliar P | + | 0.244 | + | 0.009 | − | 0.008 | − | 0.444 |
| Foliar N:P | − | 0.397 | − | 0.365 | + | 0.383 | + | 0.003 |
3.3. The Association between AM Fungal Community and Plant Performance
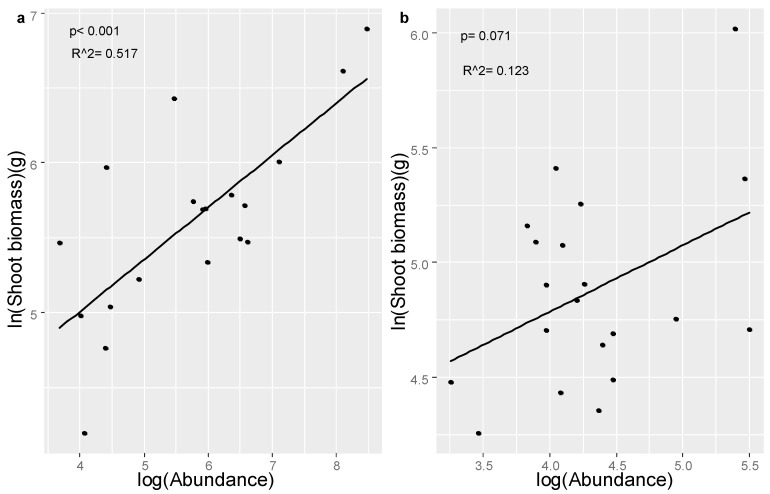
Linear regression revealed a significant positive relationship between C. korshinskii shoot biomass and the Shannon diversity of the AMF (R2 = 0.282, p = 0.019, Figure S6) and between C. microphylla shoot biomass and the richness of associated AM fungal communities (R2 = 0.219, p = 0.037, Figure S7). Furthermore, significant positively relationship between C. korshinskii shoot biomass and the abundance of AMF genus Diversispora was detected (R2 = 0.517, p < 0.001, Figure 3). The abundance of Diversispora was also marginally correlated with the shoot biomass of C. microphylla plants (R2 = 0.123, p = 0.071).
Figure 3.
Correlations between shoot biomass of C. korshinskii ((a), n = 19) and C. microphylla ((b), n = 20) and the corresponding relative abundance of AMF Diversispora. Both plant biomass and AMF abundance were log transformed. Statistically significant effects (p) and the coefficient of determination (R2) were presented.
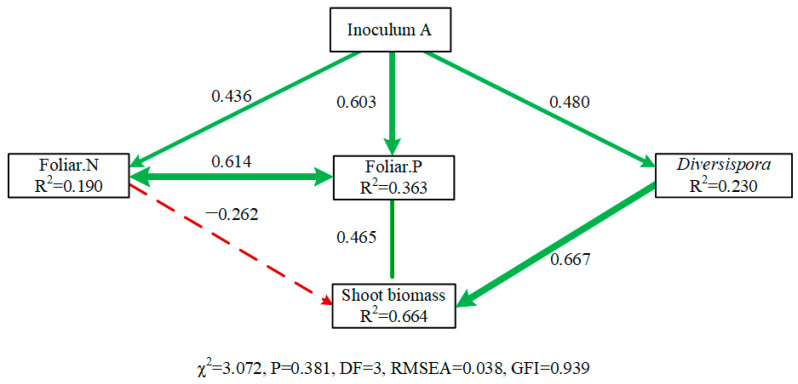
The SEM results with a good fit showed the effects of home AMF inoculation on plant performance for C. korshinskii plants (Figure 4). The models explained 66.4% of the variance in plant shoot biomass and the path coefficients (λ) for direct effects were displayed (Table S3). According to the standardized total effects, the abundance of Diversispora AMF (λ = 0.667) was the strongest predictor for plant shoot biomass. The effect of home AMF inoculation (inoculum A) on plant shoot biomass was mainly through the indirect path mediated by the influence of P uptake, and the abundance of AMF genus Diversispora.
Figure 4.
Structural equation model (SEM) showed home AMF inoculation effects on C. korshinskii plant performance. Solid and dashed lines indicated significant (p < 0.05), and non-significant pathways (p > 0.05). The double headed arrow indicated correlations. Numbers above the arrows showed the standardized path coefficients. Arrow width is proportional to the strength of different pathways. Green and red lines indicated positive and negative pathways. R2 below each dependent variable represented the proportion of variance explained.
4. Discussion
Beneficial effects of conspecific (home) AMF communities on shrub growth were detected here. These results suggested that native AMF might aid revegetation efforts in the desertified grasslands in north China. However, the different host origin effects of native AM fungal communities on plant growth and nutrition revealed that plant performance was depending on both host plant identity and fungal partners, and home advantage for conspecific AM fungal community was proved only for C. korshinskii plants.
Our first hypothesis was that native AMF inoculation would enhance plant performance. In this study, C. korshinskii plants showed increased shoot biomass when inoculated with native AM fungal communities, which was in line with previous report [50]. However, the effect of AMF inoculation also depended heavily on the host plant identity as has been reported [51]. The inoculation only increased the biomass of C. korshinskii plants, but not for C. microphylla. The fact that un-inoculated C. microphylla plants had higher nutrient also showed that the feedback between plant and AMF was host specific.
The different effects of home AMF on C. korshinskii and C. microphylla might be due to the differences in their physiology and interaction specificity to AMF [36]. Naturally in the Mongolia plateau, C. korshinskii and C. microphylla form a geographical cline with C. korshinskii distributed in extremely dry regions where soil water content is low and temperature is high. Whereas C. microphylla plants are distributed in semi-arid regions with relatively higher humidity [52]. Further studies on the physiological characteristics of these species revealed that C. korshinskii were able to maintain high photosynthetic rate and water use efficiency in more stressed environment compared to C. microphylla plants [53,54,55]. Thus, the C. korshinskii-AMF symbioses might be important for adaption of C. korshinskii to desert condition. And in this study, the higher photosynthetic rate of C. korshinskii species implied its higher ability to allocate more carbon to its fungi partner than that of C. microphylla plants. This may explain the higher colonization rate and more sensitive response of C. korshinskii species to AMF inoculation relative to C. microphylla. Regardless of why the two legume species showed varied response to AMF inocula, the inoculation effect holds implications for revegetation that the introduction of beneficial microorganism such as native AMF should be considered.
For the second hypothesis, the three AMF inocula had different effects on plant performance and the functional differences of native AM fungal communities from the three host rhizospheres were proved. Inoculum A produced the highest photosynthetic rate and the highest shoot biomass for C. korshinskii when compared with other inocula. Plants are known to have varied response to either different species of AMF, or different AM fungal communities [56,57,58]. For instance, AMF collected from abandoned field or grassland had varied effects on the plants [59]. Likewise, another study found that AMF collected from the field with different levels of long-term manure addition differently affect plant shoot biomass and P concentration [60]. Specifically, the situation when native AMF enhanced the plant growth more than the foreign one was described as “local adaptation” [16], and lots of studies reported that AMF locally adapted to the environment would be more beneficial [61,62,63,64]. Our results showed that the home AMF treatment (i.e., inoculum A for C. korshinskii) increased the biomass, N and P content of their plant host, indicating home advantage.
To further explore the home advantage of C. korshinskii and the corresponding AMF, molecular analysis was performed. Overall, we detected 8 genera among inocula and Caragana plant roots. Indicator species analysis showed that the genus Diversispora was the indicator of inoculum A and its abundance was significantly higher than the other two inocula. Interestingly, the abundance of Diversispora retrieved from the colonized roots was positively correlated with the plant biomass at harvest. Taken together, Diversispora that came from the inoculum may contribute to the biomass promotion, in line with previous findings [65]. It has been known that AMF differed in allocating biomass to intraradical hyphae, extraradical hyphae and spores [66,67]. Recent study found that edaphophilic AMF such as Diversispora with high investment to extraradical hyphae were more abundant in shrub roots [68]. Though we could not assure that Diversispora directly from the inoculum colonized Caragana roots well, according to the SEM, the home AMF treatment (inoculum A) had a positive effect on the abundance of Diversispora and the Diversispora also had a positive effect on plant biomass. Taken together, Diversispora might be the key AMF group in C. korshinskii growth in desertified areas. Previous study inoculating plants with AMF from Diversispora showed the highest biomass compared with other two species from Paraglomus and Claroideoglomus [69], but knowledge regarding the systemic functional difference among AMF genera affiliated with different guilds were still insufficient. Further study should further explore the different roles of edaphophilic and rhizophilic AMF to optimize the inoculation benefits.
5. Conclusions
In conclusion, this study explored the relationship between Caragana plant performance and the home advantage of conspecific AM fungal community in the Kubuqi Desert. C. korshinskii tend to show better performance when inoculated with home AMF, which may partly relate to Diversispora. We demonstrated that the growth of leguminous C. korshinskii can be enhanced in the desert with the help of conspecific native AMF community. Considering the vital roles AMF played, the reconstitution of the soil fungi may be a key step in restoration programs for such desert environments. For further studies, large scale field experiments containing different combinations of AMF community and host plants are needed and the evaluation on long-term effects of the introduced AMF on plants and soil should also be considered.
Acknowledgments
We appreciate the support of the Inner Mongolia Academy of Forestry. Many staffs involved in setting the experiment, the measurement of plant variables, and collecting samples but not listed are deeply appreciated.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/jof7121077/s1, Figure S1. Spore density of AMF in soil samples taken in 2014 (a) and AMF inoculum A, B and C prepared in 2016 (b). Spores were extracted from 20 g soil samples and then counted. Figure S2. Rarefaction curves of AMF species richness along the number of sequences obtained from the 3 inocula (a) and rhizospheres of 2 Caragana plant roots (b). The rarefaction curve suggested that our sampling captured most of the AMF community. Figure S3. S Shannon diversity (a) and species richness (b) of AMF community associated with inoculum A, B, and C. No significant difference was found among the inoculum A, B and C. Figure S4. The relative abundance of AMF belonging to Diversispora associated with 3 inocula. Apparently, Diversispora was significantly enriched in inoculum A. Figure S5. Shannon diversity of AMF associated with C. korshinskii, and C. microphylla plants with different AMF inoculation treatments. Inoculation treatments including inocula originating from rhizosphere of C. korshinskii (A), H. leave (B), C. microphylla (C), and sterilized control (CK). Figure S6. Linear regression indicating a significant positive relationship between C. korshinskii plant shoot biomass and the Shannon diversity of rhizosphere AMF community. Figure S7. Linear regression indicating a significant positive relationship between C. microphylla plant shoot biomass and the richness of rhizosphere AMF community. Table S1. Results of indicator species analysis of AMF genus associated with 3 inocula. Table S2. Foliar and shoot nutrient of Caragana species under different AMF treatments. Table S3. Standardized total effects of the SEM.
Author Contributions
Conceptualization, B.J. and Y.L.; investigation, X.G. and P.W.; writing—original draft preparation, X.G.; writing—review and editing, B.J., Z.W., J.Z. and Y.L.; visualization, X.G. and Y.L.; project administration, B.J.; funding acquisition, B.J. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Natural Science Foundation of China (31761123001-1); State Forestry and Grassland Administration (201404204-05A); the National Natural Science Foundation of China (31770542, 41871067).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Available upon reasonable request.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Brundrett M.C., Tedersoo L. Evolutionary history of mycorrhizal symbioses and global host plant diversity. New Phytol. 2018;220:1108–1115. doi: 10.1111/nph.14976. [DOI] [PubMed] [Google Scholar]
- 2.Smith S.E., Read D.J. Mycorrhizal Symbiosis. 3rd ed. Academic Press; Oxford, UK: 2008. [Google Scholar]
- 3.Koziol L., Schultz P.A., House G.L., Bauer J.T., Middleton E.L., Bever J.D. The Plant Microbiome and Native Plant Restoration: The Example of Native Mycorrhizal Fungi. Bioscience. 2018;68:996–1006. doi: 10.1093/biosci/biy125. [DOI] [Google Scholar]
- 4.Vahter T., Bueno C.G., Davison J., Herodes K., Hiiesalu I., Kasari-Toussaint L., Oja J., Olsson P.A., Sepp S.K., Zobel M., et al. Co-introduction of native mycorrhizal fungi and plant seeds accelerates restoration of post-mining landscapes. J. Appl. Ecol. 2020;57:1741–1751. doi: 10.1111/1365-2664.13663. [DOI] [Google Scholar]
- 5.Moreno-Mateos D., Alberdi A., Morrien E., van der Putten W.H., Rodriguez-Una A., Montoya D. The long-term restoration of ecosystem complexity. Nat. Ecol. Evol. 2020;4:676–685. doi: 10.1038/s41559-020-1154-1. [DOI] [PubMed] [Google Scholar]
- 6.Reynolds H.L., Vogelsang K.M., Hartley A.E., Bever J.D., Schultz P.A. Variable responses of old-field perennials to arbuscular mycorrhizal fungi and phosphorus source. Oecologia. 2006;147:348–358. doi: 10.1007/s00442-005-0270-6. [DOI] [PubMed] [Google Scholar]
- 7.Calonne-Salmon M., Plouznikoff K., Declerck S. The arbuscular mycorrhizal fungus Rhizophagus irregularis MUCL 41833 increases the phosphorus uptake and biomass of Medicago truncatula, a benzo[a]pyrene-tolerant plant species. Mycorrhiza. 2018;28:761–771. doi: 10.1007/s00572-018-0861-9. [DOI] [PubMed] [Google Scholar]
- 8.Frey B., Schuepp H. Acquisition of nitrogen by external hyphae of arbuscular mycorrhizal fungi associated with Zea mays L. New Phytol. 1993;124:221–230. doi: 10.1111/j.1469-8137.1993.tb03811.x. [DOI] [PubMed] [Google Scholar]
- 9.Volpe V., Chitarra W., Cascone P., Volpe M.G., Bartolini P., Moneti G., Pieraccini G., Di Serio C., Maserti B., Guerrieri E., et al. The Association with Two Different Arbuscular Mycorrhizal Fungi Differently Affects Water Stress Tolerance in Tomato. Front. Plant Sci. 2018;9:1480. doi: 10.3389/fpls.2018.01480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Paymaneh Z., Sarcheshmehpour M., Bukovská P., Jansa J. Could indigenous arbuscular mycorrhizal communities be used to improve tolerance of pistachio to salinity and/or drought? Symbiosis. 2019;79:269–283. doi: 10.1007/s13199-019-00645-z. [DOI] [Google Scholar]
- 11.Pereyra M., Grilli G., Galetto L. Root-associated fungi increase male fitness, while high simulated herbivory decreases indirect defenses in Croton lachnostachyus plants. Plant Ecol. 2018;220:29–39. doi: 10.1007/s11258-018-0900-9. [DOI] [Google Scholar]
- 12.Kaur J., Chavana J., Soti P., Racelis A., Kariyat R. Arbuscular mycorrhizal fungi (AMF) influences growth and insect community dynamics in Sorghum-sudangrass (Sorghum × drummondii) Arthropod-Plant Interact. 2020;14:301–315. doi: 10.1007/s11829-020-09747-8. [DOI] [Google Scholar]
- 13.Cruz-Silva A., Figueiredo A., Sebastiana M. First Insights into the Effect of Mycorrhizae on the Expression of Pathogen Effectors during the Infection of Grapevine with Plasmopara viticola. Sustainability. 2021;13:1226. doi: 10.3390/su13031226. [DOI] [Google Scholar]
- 14.Ryan M.H., Graham J.H. Little evidence that farmers should consider abundance or diversity of arbuscular mycorrhizal fungi when managing crops. New Phytol. 2018;220:1092–1107. doi: 10.1111/nph.15308. [DOI] [PubMed] [Google Scholar]
- 15.Albornoz F.E., Dixon K.W., Lambers H. Revisiting mycorrhizal dogmas: Are mycorrhizas really functioning as they are widely believed to do? Soil Ecol. Lett. 2021;3:73–82. doi: 10.1007/s42832-020-0070-2. [DOI] [Google Scholar]
- 16.Klironomos J.N. Variation in plant response to native and exotic arbuscular mycorrhizal fungi. Ecology. 2003;84:2292–2301. doi: 10.1890/02-0413. [DOI] [Google Scholar]
- 17.Pizano C., Kitajima K., Graham J.H., Mangan S.A. Negative plant-soil feedbacks are stronger in agricultural habitats than in forest fragments in the tropical Andes. Ecology. 2019;100:e02850. doi: 10.1002/ecy.2850. [DOI] [PubMed] [Google Scholar]
- 18.Hijri M. Analysis of a large dataset of mycorrhiza inoculation field trials on potato shows highly significant increases in yield. Mycorrhiza. 2016;26:209–214. doi: 10.1007/s00572-015-0661-4. [DOI] [PubMed] [Google Scholar]
- 19.Pizano C., Mangan S.A., Graham J.H., Kitajima K. Host-specific effects of soil microbial filtrates prevail over those of arbuscular mycorrhizae in a fragmented landscape. Ecol. Appl. 2017;27:1946–1957. doi: 10.1002/eap.1579. [DOI] [PubMed] [Google Scholar]
- 20.Bever J.D., Richardson S.C., Lawrence B.M., Holmes J., Watson M. Preferential allocation to beneficial symbiont with spatial structure maintains mycorrhizal mutualism. Ecol. Lett. 2009;12:13–21. doi: 10.1111/j.1461-0248.2008.01254.x. [DOI] [PubMed] [Google Scholar]
- 21.Kiers E.T., Duhamel M., Beesetty Y., Mensah J.A., Franken O., Verbruggen E., Fellbaum C.R., Kowalchuk G.A., Hart M.M., Bago A., et al. Reciprocal rewards stabilize cooperation in the mycorrhizal symbiosis. Science. 2011;333:880–882. doi: 10.1126/science.1208473. [DOI] [PubMed] [Google Scholar]
- 22.Ji B.M., Bever J.D. Plant preferential allocation and fungal reward decline with soil phosphorus: Implications for mycorrhizal mutualism. Ecosphere. 2016;7:e01256. doi: 10.1002/ecs2.1256. [DOI] [Google Scholar]
- 23.Garbeva P., Veen J.A.V., Elsas J.D.v. MICROBIAL DIVERSITY IN SOIL: Selection of Microbial Populations by Plant and Soil Type and Implications for Disease Suppressiveness. Annu. Rev. Phytopathol. 2004;42:243–270. doi: 10.1146/annurev.phyto.42.012604.135455. [DOI] [PubMed] [Google Scholar]
- 24.Ji B., Bentivenga S.P., Casper B.B. Evidence for ecological matching of whole AM fungal communities to the local plant-soil environment. Ecology. 2010;91:3037–3046. doi: 10.1890/09-1451.1. [DOI] [PubMed] [Google Scholar]
- 25.Feng Q., Ma H., Jiang X., Wang X., Cao S. What Has Caused Desertification in China? Sci. Rep. 2015;5:15998. doi: 10.1038/srep15998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Guoqian W., Xuequan W., Bo W., Qi L. Desertification and Its Mitigation Strategy in China. J. Resour. Ecol. 2012;3:97–104. doi: 10.5814/j.issn.1674-764x.2012.02.001. [DOI] [Google Scholar]
- 27.Carrillo-Garcia Á., De La Luz J.L.L., Bashan Y., Bethlenfalvay G.J. Nurse Plants, Mycorrhizae, and Plant Establishment in a Disturbed Area of the Sonoran Desert. Restor. Ecol. 2002;7:321–335. doi: 10.1046/j.1526-100X.1999.72027.x. [DOI] [Google Scholar]
- 28.Yu J., Xue Z., He X., Liu C., Steinberger Y. Shifts in composition and diversity of arbuscular mycorrhizal fungi and glomalin contents during revegetation of desertified semiarid grassland. Appl. Soil Ecol. 2017;115:60–67. doi: 10.1016/j.apsoil.2017.03.015. [DOI] [Google Scholar]
- 29.Hu D., Baskin J.M., Baskin C.C., Wang Z., Zhang S., Yang X., Huang Z. Arbuscular mycorrhizal symbiosis and achene mucilage have independent functions in seedling growth of a desert shrub. J. Plant Physiol. 2019;232:1–11. doi: 10.1016/j.jplph.2018.11.010. [DOI] [PubMed] [Google Scholar]
- 30.Chen Z., He X., Guo H., Yao X., Chen C. Diversity of arbuscular mycorrhizal fungi in the rhizosphere of three host plants in the farming–pastoral zone, north China. Symbiosis. 2012;57:149–160. doi: 10.1007/s13199-012-0186-y. [DOI] [Google Scholar]
- 31.Wu Y.Q., Wang Y.Y., Lang Y.F., Bai J.H., Liu W.J. Dynamics of Arbuscular Mycorrhizal Fungi and Dark Septate Endophytic Fungi under the Canopy of Caragana korshinskii Kom. Bangladesh J. Bot. 2018;47:633–641. [Google Scholar]
- 32.Koske R.E., Gemma J.N. Mycorrhizae and succession in plantings of beachgrass in sand dunes. Am. J. Bot. 1997;84:118–130. doi: 10.2307/2445889. [DOI] [Google Scholar]
- 33.Lal R. Restoring Soil Quality to Mitigate Soil Degradation. Sustainability. 2015;7:5875–5895. doi: 10.3390/su7055875. [DOI] [Google Scholar]
- 34.Liu Y., He L., An L., Helgason T., Feng H. Arbuscular mycorrhizal dynamics in a chronosequence of Caragana korshinskii plantations. FEMS Microbiol. Ecol. 2009;67:81–92. doi: 10.1111/j.1574-6941.2008.00597.x. [DOI] [PubMed] [Google Scholar]
- 35.Li X.R., Xiao H.L., Zhang J.G., Wang X.P. Long-term ecosystem effects of sand-binding vegetation in the Tengger Desert, northern China. Restor. Ecol. 2004;12:376–390. doi: 10.1111/j.1061-2971.2004.00313.x. [DOI] [Google Scholar]
- 36.Ma F., Na X., Xu T. Drought responses of three closely related Caragana species: Implication for their vicarious distribution. Ecol. Evol. 2016;6:2763–2773. doi: 10.1002/ece3.2044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Koide R.T., Li M. Appropriate controls for vesicular-arbuscular mycorrhiza research. New Phytol. 1989;111:35–44. doi: 10.1111/j.1469-8137.1989.tb04215.x. [DOI] [Google Scholar]
- 38.Ke P.J., Zee P.C., Fukami T. Dynamic plant-soil microbe interactions: The neglected effect of soil conditioning time. New Phytol. 2021;231:1546–1558. doi: 10.1111/nph.17420. [DOI] [PubMed] [Google Scholar]
- 39.Mc G.T., Miller M.H., Evans D.G., Fairchild G.L., Swan J.A. A new method which gives an objective measure of colonization of roots by vesicular-arbuscular mycorrhizal fungi. New Phytol. 1990;115:495–501. doi: 10.1111/j.1469-8137.1990.tb00476.x. [DOI] [PubMed] [Google Scholar]
- 40.Murphy J., Riley J.P. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta. 1962;27:31–36. doi: 10.1016/S0003-2670(00)88444-5. [DOI] [Google Scholar]
- 41.Bremner J.M., Tabatabai M.A. Use of an ammonia electrode for determination of ammonium in Kjeldahl analysis of soils. Commun. Soil Sci. Plant Anal. 2008;3:159–165. doi: 10.1080/00103627209366361. [DOI] [Google Scholar]
- 42.Allen G.C., Flores-Vergara M.A., Krasynanski S., Kumar S., Thompson W.F. A modified protocol for rapid DNA isolation from plant tissues using cetyltrimethylammonium bromide. Nat Protoc. 2006;1:2320–2325. doi: 10.1038/nprot.2006.384. [DOI] [PubMed] [Google Scholar]
- 43.Muneer M.A., Tarin M.W.K., Chen X., Afridi M.S., Iqbal A., Munir M.Z., Zheng C., Zhang J., Ji B. Differential response of mycorrhizal fungi linked with two dominant plant species of temperate grassland under varying levels of N-addition. Appl. Soil Ecol. 2022;170:104272. doi: 10.1016/j.apsoil.2021.104272. [DOI] [Google Scholar]
- 44.Caporaso J.G., Kuczynski J., Stombaugh J., Bittinger K., Bushman F.D., Costello E.K., Fierer N., Pena A.G., Goodrich J.K., Gordon J.I., et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods. 2010;7:335–336. doi: 10.1038/nmeth.f.303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Edgar R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods. 2013;10:996–998. doi: 10.1038/nmeth.2604. [DOI] [PubMed] [Google Scholar]
- 46.Edgar R.C., Haas B.J., Clemente J.C., Quince C., Knight R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics. 2011;27:2194–2200. doi: 10.1093/bioinformatics/btr381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Saitou N., Nei M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987;4:406–425. doi: 10.1093/oxfordjournals.molbev.a040454. [DOI] [PubMed] [Google Scholar]
- 48.Oksanen J., Blanchet F.G., Kindt R., Legendre P., Minchin P.R., O’hara R., Simpson G.L., Solymos P., Stevens M.H.H., Wagner H. Package ‘vegan’. Community Ecol. Package Version. 2013;2:1–295. [Google Scholar]
- 49.De Caceres M., Legendre P. Associations between species and groups of sites: Indices and statistical inference. Ecology. 2009;90:3566–3574. doi: 10.1890/08-1823.1. [DOI] [PubMed] [Google Scholar]
- 50.Shi Z.Y., Wang Y.M., Xu S.X., Lan Z.J., Mickan B.S., Zhang X.L., Wang F.Y. Arbuscular Mycorrhizal Fungi Enhance Plant Diversity, Density and Productivity of Spring Ephemeral Community in Desert Ecosystem. Not. Bot. Horti Agrobot. Cluj-Napoca. 2017;45:301–307. doi: 10.15835/nbha45110766. [DOI] [Google Scholar]
- 51.Hoeksema J.D., Chaudhary V.B., Gehring C.A., Johnson N.C., Karst J., Koide R.T., Pringle A., Zabinski C., Bever J.D., Moore J.C., et al. A meta-analysis of context-dependency in plant response to inoculation with mycorrhizal fungi. Ecol. Lett. 2010;13:394–407. doi: 10.1111/j.1461-0248.2009.01430.x. [DOI] [PubMed] [Google Scholar]
- 52.Ma C.C., Gao Y.B., Liu H.F., Wang J.L., Guo H.Y. Interspecific transition among Caragana microphylla, C. davazamcii and C. korshinskii along geographic gradient. I. Ecological and RAPD evidence. Acta Bot. Sin. 2003;45:1218–1227. [Google Scholar]
- 53.Xie L., Ma C., Guo H., Li Q., Gao Y. Distribution pattern of Caragana species under the influence of climate gradient in the Inner Mongolia region, China. J. Arid. Land. 2013;6:311–323. doi: 10.1007/s40333-013-0227-2. [DOI] [Google Scholar]
- 54.Ma C.C., Gao Y.B., Guo H.Y., Wang J.L. Photosynthesis, transpiration, and water use efficiency of Caragana microphylla, C. intermedia, and C. korshinskii. Photosynthetica. 2004;42:65–70. doi: 10.1023/B:PHOT.0000040571.63254.c2. [DOI] [Google Scholar]
- 55.Ma C.-C., Guo H.-Y., Wu J.-B., Wang J.-L., Qi S.-X., Wei Y.-R., Gao Y.-B. Acclimation of Photosynthetic Traits of Caragana Species to Desert Environment in Inner Mongolian Plateau. Arid. Land Res. Manag. 2013;28:87–101. doi: 10.1080/15324982.2013.808717. [DOI] [Google Scholar]
- 56.van der Heijden M.G.A., Boller T., Wiemken A., Sanders I.R. Different Arbuscular Mycorrhizal Fungal Species Are Potential Determinants of Plant Community Structure. Ecology. 1998;79:2082–2091. doi: 10.1890/0012-9658(1998)079[2082:DAMFSA]2.0.CO;2. [DOI] [Google Scholar]
- 57.Frew A., Wilson B.A.L. Different mycorrhizal fungal communities differentially affect plant phenolic-based resistance to insect herbivory. Rhizosphere. 2021;19:100365. doi: 10.1016/j.rhisph.2021.100365. [DOI] [Google Scholar]
- 58.Bi Y.L., Wang K., Wang J. Effect of different inoculation treatments on AM fungal communities and the sustainability of soil remediation in Daliuta coal mining subsidence area in northwest China. Appl. Soil Ecol. 2018;132:107–113. doi: 10.1016/j.apsoil.2018.08.009. [DOI] [Google Scholar]
- 59.Pankova H., Lepinay C., Rydlova J., Voriskova A., Janouskova M., Dostalek T., Munzbergova Z. Arbuscular mycorrhizal fungi and associated microbial communities from dry grassland do not improve plant growth on abandoned field soil. Oecologia. 2018;186:677–689. doi: 10.1007/s00442-017-4054-6. [DOI] [PubMed] [Google Scholar]
- 60.Qin Z.F., Zhang H.Y., Feng G., Christie P., Zhang J.L., Li X.L., Gai J.P. Soil phosphorus availability modifies the relationship between AM fungal diversity and mycorrhizal benefits to maize in an agricultural soil. Soil Biol. Biochem. 2020;144:107790. doi: 10.1016/j.soilbio.2020.107790. [DOI] [Google Scholar]
- 61.Ruiz-Lozano J.M., Azcón R. Symbiotic efficiency and infectivity of an autochthonous arbuscular mycorrhizal Glomus sp. from saline soils and Glomus deserticola under salinity. Mycorrhiza. 2000;10:137–143. doi: 10.1007/s005720000075. [DOI] [Google Scholar]
- 62.Middleton E.L., Richardson S., Koziol L., Palmer C.E., Yermakov Z., Henning J.A., Schultz P.A., Bever J.D. Locally adapted arbuscular mycorrhizal fungi improve vigor and resistance to herbivory of native prairie plant species. Ecosphere. 2015;6:276. doi: 10.1890/ES15-00152.1. [DOI] [Google Scholar]
- 63.Harris-Valle C., Esqueda M., Gutierrez A., Castellanos A.E., Gardea A.A., Berbara R. Physiological response of Cucurbita pepo var. pepo mycorrhized by Sonoran desert native arbuscular fungi to drought and salinity stresses. Braz. J. Microbiol. 2018;49:45–53. doi: 10.1016/j.bjm.2017.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Bauer J.T., Koziol L., Bever J.D. Local adaptation of mycorrhizae communities changes plant community composition and increases aboveground productivity. Oecologia. 2020;192:735–744. doi: 10.1007/s00442-020-04598-9. [DOI] [PubMed] [Google Scholar]
- 65.Wu Q.-S., Zou Y.-N., Huang Y.-M. The arbuscular mycorrhizal fungus Diversispora spurca ameliorates effects of waterlogging on growth, root system architecture and antioxidant enzyme activities of citrus seedlings. Fungal Ecol. 2013;6:37–43. doi: 10.1016/j.funeco.2012.09.002. [DOI] [Google Scholar]
- 66.Maherali H., Klironomos J.N. Influence of phylogeny on fungal community assembly and ecosystem functioning. Science. 2007;316:1746–1748. doi: 10.1126/science.1143082. [DOI] [PubMed] [Google Scholar]
- 67.Han Y., Feng J., Han M., Zhu B. Responses of arbuscular mycorrhizal fungi to nitrogen addition: A meta-analysis. Glob. Chang. Biol. 2020;26:7229–7241. doi: 10.1111/gcb.15369. [DOI] [PubMed] [Google Scholar]
- 68.Weber S.E., Diez J.M., Andrews L.V., Goulden M.L., Aronson E.L., Allen M.F. Responses of arbuscular mycorrhizal fungi to multiple coinciding global change drivers. Fungal Ecol. 2019;40:62–71. doi: 10.1016/j.funeco.2018.11.008. [DOI] [Google Scholar]
- 69.Yang W., Guo X.J., Wang Q. Contribution of different arbuscular mycorrhizal fungal inoculum to Elymus nutans under nitrogen addition. Ann. Microbiol. 2018;68:689–696. doi: 10.1007/s13213-018-1375-6. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Available upon reasonable request.