Abstract
The time for battling cancer has never been more suitable than nowadays and fortunately against hepatocellular carcinoma (HCC) we do have a far-reaching arsenal. Moreover, because liver cancer comprises a plethora of stages-from very early to advanced disease and with many treatment options–from surgery to immunotherapy trials–it leaves the clinician a wide range of options. The scope of our review is to throw light on combination treatments that seem to be beyond guidelines and to highlight these using evidence-based analysis of the most frequently used combination therapies, discussing their advantages and flaws in comparison to the current standard of care. One particular combination therapy seems to be in the forefront: Transarterial chemoembolization plus ablation for medium-size non-resectable HCC (3-5 cm), which is currently at the frontier between Barcelona Clinic Liver Cancer classification A and B. Not only does it improve the outcome in contrast to each individual therapy, but it also seems to have similar results to surgery. Also, the abundance of immune checkpoint inhibitors that have appeared lately in clinical trials are bringing promising results against HCC. Although the path of combination therapies in HCC is still filled with uncertainty and caveats, in the following years the hepatology and oncology fields could witness an HCC guideline revolution.
Keywords: Hepatocellular carcinoma, Transarterial chemoembolization, Radiofrequency ablation, Microwave ablation, Systemic therapy, Immunotherapy combined treatments
Core Tip: The field of hepatocellular carcinoma has become highly interesting in recent years, given the emergence of a large amount of data on immunotherapy and combination treatment strategies. In this light, the current clinical practice guidelines may appear restrictive, especially in borderline cases, which have become a significant challenge in tumor boards across the world. The current review is designed to provide an exhaustive analysis of the most notable advances in the field, focusing on combination therapies and their role in the therapeutic algorithm, with the ultimate goal of aiding clinicians to navigate the Barcelona clinic liver cancer classification maze.
INTRODUCTION
Hepatocellular carcinoma (HCC) is the most common primary liver cancer, ranking sixth overall among malignancies in incidence and, disproportionately, fourth in mortality[1]. It is a multifaceted disease, atypical among cancers due to its intricate and non-linear prognostic indicators, as survival is closely intertwined with the tumor extension, the severity of the underlying liver disease, and overall fitness. To this point, there is a wide array of available techniques in the therapeutic arsenal against HCC. The options range from curative-intent solutions such as surgery or transplantation to local ablation, interventional radiology, and systemic therapies[2,3]. However, despite recent advances and potentially game-changing developments, HCC is still associated with a poor prognosis, with an incidence to mortality ratio dismally approaching number one[4].
Currently, the most frequently employed algorithm for standardizing care is the Barcelona Clinic Liver Cancer Classification (BCLC) and its subsequent updates[5]. Arguably the best algorithm to this point and backed by extensive validation, it still poses significant clinical dilemmas, especially for cases that do not fit perfectly in its boxes. In such scenarios, the comfort of evidence-based guideline recommendations tends to fade, leaving both the clinicians and the patients in uncharted waters, seeking the best path forward.
Consequently, multiple approaches have been attempted, combining available techniques in various shapes and forms with the ultimate goal of improving the overall outcome. These combinations try to negate the individual deficiencies of each method while augmenting their strengths, hoping to provide a perfect match for any specific clinical scenario. Ranging from an early tumor in an advanced, decompensated liver disease, to an advanced tumor in an otherwise relatively normal liver and anything in-between, the severity spectrum of HCC leaves room for epistemic, data-based improvisation. However, as the range of therapies is ever-increasing, there is a thin line between being too conservative and overtreating, as both extremes could lead to additional harm and cost. The current review aims to provide an evidence-based analysis of the most frequently used combination therapies, discussing their advantages and caveats in comparison to the current standard of care.
COMBINED TREATMENT TO FACILITATE CURE
Hepatic resection plus intraoperative ablation
Although surgical resection (SR) still represents the ideal and best option as treatment, having curative potential, unfortunately for patients diagnosed with liver malignancies, this treatment is feasible in only 10%-20% of cases[6]. The remaining 80%-90% of patients, which are not suitable for radical intervention, include patients with multiple tumors, located in both hepatic lobes with insufficient hepatic reserve[7]. In the case of HCC, according to the guidelines used in clinical practice (BCLC staging system), the patients with unresectable multicentric neoplasia and preserved liver function fall into stage B, being candidates only for transarterial chemoembolization (TACE)[8]. However, the results of various studies from the literature are beginning to support the use of combined techniques for this category of patients[9]. According to the literature, there are several radical options for bilobar localization, including two-stage resections, ALPPS technique (associating liver partition and portal vein ligation for staged hepatectomy), and combined techniques: Hepatic resections and ablative techniques[9].
Thereby, this complex treatment, which involves the combination of hepatectomies with simultaneous tumor ablations, has the role of increasing the proportion of patients who can become candidates for a radical, potentially curative treatment. However, although there are numerous reports in the literature underlining the clinical outcomes, a standard conduit and a therapeutic consensus in this field have not been yet established[6,10].
General indications for combined therapy are patients with multicentric, bilobar, unresectable neoplasms who are not candidates for curative resection but present with a compensated liver disease[11,12]. Other indications include inoperable tumors due to proximity to major vascular structures and/or the presence of liver cirrhosis with functional liver parenchyma, but not being able to tolerate a major resection[6,10]. Ablative techniques are indicated even if the tumors are in the proximity of a main portal branch, hepatic vein, or inferior vena cava[12].
The extrahepatic presence of neoplasia represents a contraindication for combined treatment, although there are authors who advocate for this treatment in the case of the associated resectable lung tumors or local invasion from liver tumors (in the diaphragm or adrenal gland)[9]. The association of ablation is not indicated when the tumor involves the right or left liver duct[12]. Incontestably, patients with decompensated liver disease, refractory ascites, coagulation disorders, and/or low-performance status (PS) cannot benefit from the combined resection-ablation treatment.
The intraoperative technique involves performing simultaneously, under general anesthesia and by laparotomy, both liver resections, and HCC ablation sessions. Most authors recommend performing resection first, followed by ultrasound-guided ablation, most commonly by tissue destruction by radiofrequency ablation (RFA)[6]. Microwave ablation (MWA) comes with some advantages, these being cited by some authors, but with a lower usage than RFA[9]. Although feasible, with comparable outcomes with “conventional” open surgery, the combined treatment performed by a laparoscopic approach is not a standardized technique, with only a few reports being found in the literature[13,14].
To exclude extrahepatic neoplasia it is mandatory to do a complete exploration of the entire abdominal cavity and intraoperative hepatic ultrasound (IOUS) to assess the topography and tumor relationships[15,16]. Anatomical resections are preferred whenever possible[17]. The indication for atypical, minor (1-2 segments), or major (> 3 segments) hepatectomy is determined by the preoperative assessment of the liver function, of the patient status, and by tumor extent. Also, to prevent blood loss, intermittent clamping of the afferent hepatic pedicle (Pringle maneuver) may be necessary. According to Qiu et al[6], it is required in less than 50% of cases[6]. Another advantage of this maneuver is the reduction of the cooling effect induced by the proximity of tumors to main vascular branches (“heat sink”), the rate of achieving a complete ablation being higher[18]. Evaluation of the necrotic area and the efficacy of RFA can be performed immediately by contrast-enhanced-IOUS (CE-IOUS), making possible repeated ablation sessions immediately[19].
Doing a literature survey guided by the words “hepatocellular carcinoma”, “combined”, “liver resection” and “ablative treatment”, using the PubMed database for titles in English published from 2010 to 2020, we found that the mortality reported for the surgery-ablative combined technique is around 1%-2% with a general incidence of complications in the range 22%-70%[6,20-25] (Table 1).
Table 1.
Morbidity, mortality, recurrence and survival after hepatic resection plus intraoperative ablation
|
Ref.
|
Patients, n (%)
|
MO, n (%)
|
Mo, n (%)
|
Recurrence
|
Survival rates (1 yr/3 yr/5 yr)
|
| Qiu et al[6], 2014 | 112 | 2 | 22.3 | 72.3% | 67.5%/32.5%/12.5% |
| Hou et al[23], 2016 | 51 | 0 | 70.6 | 54.9% | 88.2%/66.7%/52.9% |
| Zhang et al[24], 2014 | 114 | 0.9 | - | - | 34.4%/70.7%/40.7% |
| Huang et al[25], 2020 | 51 | - | - | - | 86.3%/66.6%/34.2% |
MO: Mortality; Mo: Morbidity.
In general, the literature supports the combined technique as feasible and safe, although the cited recurrence rate is high[6,20,23] (Table 1). The rate of major complications is reported to be around 15%, with a rate of acute liver failure of 1.8% and postoperative bleeding of 0.9%[6,21]. Other specific complications are biliary leaks (8.9%), postoperative ascites (11.6%), perihepatic abscesses (1.8%). 0.9% of patients with post-operative complications may require reinterventions[6,21]. No significant differences were reported between the rates of complications after combined techniques and conventional liver resections. Doing a literature survey guided by the words “hepatocellular carcinoma”, “combined”, “liver resection” and “ablative treatment”, using the PubMed database for titles in English published from 2010 to 2020, we found that the mortality reported for the surgery-ablative combined technique is around 1%-2% with a general incidence of complications in the range 22%-70%[6,20,21].
In regards to long-term survival, we found a range between 12% and 88%, depending on 1-, 3- and 5-year survival reports (Table 1). As can be seen, there is a wide range of results, most likely explained by the lack of standardization of the combined procedures and by the contribution of the case selection bias.
Most of the authors concluded that whenever resection can be performed, it must be chosen instead of ablation. However, RFA remains a feasible alternative in cases that are not suitable for resection, except for large tumors, over 5 cm[22]. Moreover, whenever possible, combined therapy should be indicated to the detriment of TACE, the latter being followed by a shorter 5-years survival: 52.9% (combined treatment) vs 9.8% (TACE)[23].
The results are more optimistic when comparing combined treatment with resections only, with overall survival (OS) at 1-, 3- and 5-years being comparable between the two groups: 86.3%, 66.6% and 34.2% vs 92.8%, 67% and 37%, respectively (P = 0.4)[6,21,24,26].
Supported by the data mentioned in the literature above, the combined treatment (resection plus ablation) is a feasible alternative to the therapeutic options already existing in current guidelines. The fact that more and more studies highlight the increase in the number of curative resections by using these techniques will certainly support their integration into the standard conducting algorithm for HCC.
Combination of locoregional therapies for early and intermediate stage including comparison with other therapies
Locoregional therapies (LRT) include TACE and local ablation techniques. Local ablation including percutaneous ethanol injections, RFA, MWA, laser- and cryoablation (CA) are considered alternative curative methods for early-stage HCC[27,28]. As 75% of HCCs nodules are inoperable at the time of the diagnosis, TACE plays an important role in the management of unresectable HCC and is considered the first-line therapy for BCLC stage B HCC based on Barcelona Clinic Liver Cancer guidelines[27,28].
Limits of LRT and rationale for combination therapy: For some patients undergoing TACE procedure the tumor necrosis rate is low with consequent frequent tumoral residue and high intrahepatic recurrence[27,29]. Along these lines there are several reasons for this limited efficacy: (1) The difficulty to embolize all the feeding arteries of the tumor; (2) Recanalization and angiogenesis which may occur after TACE with consequent tumor recurrence and metastasis; and (3) The re-establish of collateral circulation[27]. Ablations techniques like RFA present a high performance in tumors below 2-3 cm where complete necrosis may be achieved in up to 90% of cases[21]. However, RFA is less effective in medium-sized (3-5 cm) or large tumors (5-9 cm) where the efficacy dismally drops to 61% and 24% respectively[21]. When RFA was performed for lesions located near major vessels a heat sink effect was reported leading to an increased recurrence rate as well.
In this regard, the location of the lesion is a crucial factor when considering ablation on the grounds that some lesions cannot be successfully treated with thermal ablation without damaging adjacent structures (bile ducts, colon, diaphragm)[30]. Thereby, one intention-to-treat analysis found that 9% of small HCCs were not amenable to percutaneous ablation because of their location[30].
The rationale for combining TACE with ablation is to maximize the percentage of complete tumor response rate and thereby to reduce local recurrence rate due to incomplete or inadequate treatment of the adjacent hepatic parenchyma[9]. As follows, the synergy of the therapies leads to larger volumes of destructed tumoral tissue with consequent efficient treatment of presumed microsatellite nodules and microvascular invasion[9,27]. The sequencing of combined therapies is controversial, with most authors preferring TACE followed by RFA, although some prefer RFA followed by TACE or both techniques in the same session[9,27]. The theoretical advantages of performing TACE before ablation include (1) TACE reduces hepatic artery blood flow, thus diminishing heat sink effects and maximizing the size of the ablation zone; and (2) TACE can detect satellite lesions not seen on cross-sectional imaging[9,27].
TACE combined with RFA vs TACE: There are several papers published on this combination with different clinical scenarios. The most important ones are presented in Table 2.
Table 2.
Comparison of transarterial chemoembolization plus radiofrequency ablation to transarterial chemoembolization
|
Ref.
|
Treatment type, n (%)
|
Clinical scenario
|
Response rate mRECIST
|
Outcome
|
| Morimoto et al[111], 2013 | TACE + RFA (132) | HCC 1-5 cm, subcapsular | 98.5% CR | LTP (3 yr) 9.7%. OS (3, 5, 7 yr): 79.3%, 60.6%, 50.9% |
| Song et al[31], 2016 | TACE (71) vs TACE + RFA (87) vs RFA (43) | HCC within Milan | 81.6% vs 96.5% vs 97.6% (TACE vs TACE + RFA P = 0.019) | LTR (1, 3, 5 yr): 17%, 58%, 78% vs 6%, 33%, 54% vs 10%, 31%, 48% (TACE + RFA vs TACE P = 0.015; RFA vs TACE P = 0.005). OS (1, 3, 5 yr): 98%, 90%, 83% vs 98%, 95%, 90% vs 94%, 84%, 71% OS significantly higher (P = 0.019) for TACE + RFA vs TACE or RFA for lesions < 3 cm but not for lesions > 3 cm |
| Lee et al[32], 2018 | TACE (85) vs TACE + RFA (n = 82) | HCC BCLC 0 or A invisible for ultrasound | 97.6% vs 100% (CR) | LTP (1, 3, 5, 7 yr): 12.5%, 31%, 37% vs 7.3%, 16.5%, 16.5% (P = 0.013). Median TTP: 18 mo vs 24 mo (P = 0.037). OS (1, 3, 5 yr): 100%, 93.2%, 87.7% vs 100%, 96.6%, 87.4% (P = 0.686) |
| Liu et al[33], 2019 | TACE (195) vs TACE + RFA (209) | HCC B1 | N/A | Median PFS: 14 mo vs 20 mo. PFS (1, 3, 5 yr): 59.1%, 11.0%, 2.2% vs 71.8%, 26.6%, 13.0% (P < 0.001). OS (1, 3, 5 yr): 80.7%, 26.4%, 6.7% vs 83.7%, 45.8%, 24.8% (P = 0.003) |
| Hiraoka et al[73], 2017 | TACE (32) vs TACE + RFA (32) | HCC BCLC B1 + B2 | N/A | Median OS: 840 d vs 2466 d. OS (1, 3, 5 yr): 86.3%, 43.5%, 15.8% vs 100%, 78.6%, 62.3% (P < 0.001). Median TTP: 140 d vs 1148 d (P < 0.0001) |
| Ren et al[35], 2019 | TACE (271) vs TACE + RFA (128) | HCC BCLB A and B | 44.7% vs 85.9% (CR) | Median OS: 16 mo vs 59 mo (P < 0.001). Median PFS: 4 mo vs 45 mo. OS (1, 3, 5, 8 yr): 64.5%, 15.1%, 10.8%, 10.8% vs 90.6%, 76.6%, 68.0%, 68.0% |
| Chu et al[36], 2019 | TACE (314) vs TACE + RFA (109) vs RFA (115) | HCC 3.1-10 cm | 84.7% vs 95.4% vs 94.8% (CR) | RFS (5, 10, 15 yr): 59.1%, 11.0%, 2.2% vs 25.5%, 13.3%, 7.9% vs 9.2%, 2.9%, and 2.9% (P = 0.002). OS (5, 10, 15 yr): 16.2%, 10.9%, 7.7% vs 57.8%, 41.8%, 30.9% vs 35.2%, 11.9%, 11.9% (P = 0.022) |
| Liu et al[37], 2020 | TACE (124) vs TACE + RFA (77) | HCC 3-10 cm | N/A | Median PFS: 4 mo vs 9.13 mo (P < 0.001). PFS (1, 3, 5 yr): 11.9%, 0%, 0% vs 43%, 18%, 9.5%. Median OS: 12 mo vs 27.57 mo (P < 0.001). OS (1, 3, 5 yr): 48%, 6.5%, 0% vs 76.2%, 37.1%, 16.4% |
| Hyun et al[38], 2016 | TACE (54) vs TACE + RFA (37) | HCC not feasible for RFA | 57% vs 100% P < 0.01 (CR) | Median TTP: 29.7 mo vs 34.9 mo (P = 0.014). OS (1, 2, 3 yr): 91%, 79%, 71% vs 100%, 97%, 93% |
| Yang et al[39], 2020 | TACE + RFA special location (n = 37) vs TACE + RFA conventional location (n = 85) | HCC special locations | 91.9% vs 85.9% (CR) (NS) | Median PFS: 14 mo vs 17 mo (NS). Median OS: 32 mo vs 28 mo (NS). OS (1, 2 yr): 96.3%, 65% vs 89.9%, 63.3% (NS) |
| Hyun et al[112], 2016 | TACE + RFA (14) | HCC < 2 cm caudate lobe | 90.9% CR | LTP (1, 3, 5 yr): 0%, 12.5%, 12.5%. PFS (1, 3, 5 yr): 81.8%, 51.9%, 26%. OS (1, 3, 5 yr): 100%, 80.8%, 80.8% |
| Hyun et al[113], 2018 | TACE +RFA (69) | HCC < 3 cm not feasible for RFA | 100% CR | LTP (1, 3, 5, 7 yr): 4.4%, 6.8%, 8.2%, 9.5%, 9.5%. OS (1, 3, 5, 7 yr): 100%, 95%, 89%, 80%, 80% |
| Yan et al[114], 2018 | TACE + RFA single session (87) | HCC < 7 cm not resectable | 87.4% CR | LTP (1, 3, 5 yr): 0%, 29.9%, 55.2%. Median OS: 39 mo. OS (1, 3, 5 yr): 100%, 65.5%, 47.5% |
| Kim et al[115], 2019 | TACE + RFA (67) | BCLC A, non-surgical | N/A | PFS (1, 3, 5 yr): 86.8%, 55.9%, 29.7%. OS (1, 3, 5 yr): 100%, 93.4%, 83.5% |
| Duan et al[116], 2020 | TACE + RFA, one session (46) | HCC > 8 cm | N/A | PFS (2, 3 yr): 9.4 mo and 10.2 mo. OS (2, 3 yr): 18.4 mo and 26.4 mo |
| Zhang et al[117], 2020 | TACE + RFA (1) naive (40); (2) recurrent (36); and (3) hepatectomy | 1 tumor < 7 cm, up to 3 tumors < 3 cm, Child A or B | 62.5% vs 70% (CR + PR) | OS (1, 2, 3 yr): 97.5%, 84%, 66% (A) vs 90%, 82%, 66% (B) vs 90%, 79%, 63% (C) (A vs B vs C NS). DFS: 75%, 51%, 35% (A) vs 50%, 31%, 17% (B) vs 80%, 59%, 40% (C) (A vs B P = 0.013) |
| Wang et al[118], 2018 | TACE (13) vs TACE + RFA (13) | HCC with hepatic vein thrombus | 0% + 92.3% vs 46.2% + 53.7% (CR + PR) | Median OS: 6.5 mo vs 18 mo (P = 0.02) |
| Song et al[119], 2020 | TACE (63) vs TACE + RFA (96) | Recurrent HCC < 5 cm after HR | N/A | DFS (1, 3, 5 yr): 41.1%, 9.9%, 4.9% vs 55.1%, 22.5%, 9.7%. OS (1, 3, 5 yr): 75.9%, 30.7%, 11.3% vs 82.3%, 42.7%, 16.5% (NS) |
P value less than 0.05 (typically ≤ 0.05) is statistically significant. HCC: Hepatocellular carcinoma; RFA: Radiofrequency ablation; TACE: Transarterial chemoembolization; OS: Overall survival; PFS: Progression free survival; RFS: Recurrence free survival; TTP: Time to progression; LTP: Local tumor progression; LTR: Local tumor recurrence; DFS: Disease free survival; TR: Tumor recurrence; CR: Complete response.
By analyzing these studies, it was discovered that combination therapy might be beneficial compared to TACE alone, or RFA alone in several clinical scenarios.
BCLC-A patients: Song et al[31] have compared the results of 71 patients with HCC within Milan criteria treated by TACE to 87 and 43 patients treated by TACE + RFA and respectively RFA. The combination therapy yielded a significantly higher complete response rate according to mRECIST criteria in comparison to TACE (96.5% vs 81.6%, P = 0.019) and a lower rate of local tumor progression at 1, 3 and 5 years (6%, 33%, and 45% vs 17%, 58% and 78%). Nonetheless, what is surprising is the fact that OS was significantly higher for TACE + RFA vs TACE or RFA alone for lesions below 3 cm but not for lesions larger than 3 cm[31]. Similar results were reported as well by Lee et al[32] with lower recurrence rate at 1 and 3 years 7.3% and 16.5% vs 12.5% and 31% but no difference in OS[32].
BCLC-B patients: Liu et al[33] have compared a B1 HCC population treated by TACE (195 pts) vs TACE + RFA (209 pts) and found a significant difference in favor of combination therapy regarding progression-free survival (PFS) and OS[33]. Also, the same results [significantly higher median OS-840 vs 2466 d and median time-to-tumor progression (TTP)-140 vs 1148 d] were communicated by Hirooka et al[34] although on a small sample of patients[34]. Likewise, Ren et al[35] have investigated a large BCLC-A and B population treated either by TACE (271 pts) or TACE + RFA (128 pts) and concluded by all means that the combination therapy is significantly superior to TACE in terms of PFS and both median and cumulative OS[35]. The superiority of TACE + RFA over TACE in terms of longer OS and PFS was demonstrated also for medium and large nonresectable HCCs in 2 large recent trials[36,37].
The combination therapy was assessed also in patients with small lesions not feasible for RFA and lesions in unreachable locations (e.g., near the hepatic hilum, subdiaphragmatic, subcapsular). TACE procedures resolve this dilemma due to intratumoral accumulation of radio-opaque iodized oil used and give radiographic contrast to a small tumor either of poor conspicuity or even ultrasound (US) blind and also difficult to target spots such as hepatic dome[27]. An undoubtedly higher complete response rate (100% vs 54%, P < 0.01) and lower TTP rate were revealed by Hyun et al[38] in a series of patients with HCC not feasible for US-guided RFA treated by TACE (54 pts) or TACE + RFA (37 pts)[38]. However, Yang et al[39] compared the efficacy of combination therapy in 37 patients with HCC in special locations to 85 patients with HCC in convenient locations and found no differences in PFS and OS[39]. Three meta-analyses concerning the comparison of TACE + RFA vs TACE have been already published[40-42]. The last one issued in 2017 found that for intermediate-stage HCC, TACE plus RFA attained higher tumor response rates (OR = 6.08, 95%CI: 4.00-9.26, P < 0.00001), achieved longer recurrence-free survival (RFS) rates (ORRFS = 3.78, 95%CI: 2.38-6.02, P < 0.00001) and OS rates (OR1-year = 3.92, 95%CI: 2.41–6.39, P < 0.00001; OR3-year = 2.56; 95%CI: 1.81–3.60; P < 0.00001; OR5-year = 2.78; 95%CI: 1.77–4.38; P < 0.0001) when comparing to TACE alone. Unfortunately, as expected the number of complications were higher in the TACE plus RFA group than in the TACE alone group (OR = 2.74, 95%CI: 1.07-7.07, P = 0.04)[40].
The combination therapy TACE + RFA was also compared to RFA in several other meta-analyses[43-46]. The last one including 8 trials and 648 patients showed that RFA plus TACE is associated with a significant advantage in RFS [hazard ratio (HR) = 0.58; 95%CI: 0.42-0.80, P = 0.001], and OS (HR = 0.60; 95%CI: 0.47-0.76, P < 0.001). The authors concluded that TACE combined with RFA is more competent and appealing than RFA alone, especially for intermediate and large-size hepatic tumors or younger patients with HCC[47].
TACE combined with RFA vs hepatic resection: According to the BCLC guidelines published in 2018, the mainstay of curative therapy for early HCC is RFA or surgery-either HR or liver transplantation. As mentioned earlier, even though HR is a cornerstone treatment in non-metastatic disease, some patients are considered inappropriate candidates due to underlying liver disease[48]. We have emphasized in the previous lines that the combination therapy of TACE and RFA is superior and beneficial for the treatment of early and intermediate HCC vs the therapies used individually. Along these lines, the comparison of the outcome of TACE + RFA vs HR is of paramount importance as some of the patients are not ideal surgical candidates. Several comparative studies are presented in Table 3.
Table 3.
Comparison transarterial chemoembolization + radiofrequency ablation to other curative therapies
|
Ref.
|
Treatment type, n (%)
|
Clinical scenario
|
Response rate mRECIST
|
Outcome
|
| Saviano et al[49], 2017 | TACE + RFA (n = 25) vs HR (n = 29) | HCC 3.0-8.8 cm, solitary HCC 3-5 cm | N/A | OS (1, 3 yr): 89.4%, 48.2% vs 91.8%, 79.3% (P = 0.117). TR (1, 3 yr): 42.4%, 76.0% vs 29.5%, 45.0% (P = 0.034); LTP (3 yr): 58.1% vs 21.8% (P = 0.005). TR: 75.1% vs 35.4% (P = 0.016); LTP: 55.7% vs 16.0% P = 0.013) |
| Pan et al[50], 2017 | TACE + RFA (n = 154) vs HR (n = 176) | Within Up-To Seven criteria | N/A | Median OS: 56 mo vs 58 mo (NS). OS (1, 3, 5 yr): 96.1%, 76.7%, 41.3% vs 96.1%, 86.4%, 46.2% (P = 0.138). Median OS (beyond Milan): 52 mo vs 45 mo (P = 0.023) |
| Liu et al[51], 2016 | TACE + RFA (n = 100) vs HR (n = 100) | Within Milan | N/A | OS (1, 3, 5 yr): 96%, 67.2%, 45.7% vs 97%, 83.7%, 61.9% (P = 0.007). RFS (1, 3, 5 yr): 83%, 44.9%, 35.5% vs 94%, 68.2%, 48.4% (P = 0.026). Complications rate: 11% vs 23%, P = 0.024) |
| Lin et al[52], 2020 | TACE (n = 231) vs TACE + RFA (n = 57) vs HR (n = 140) | BCLC-B | N/A | OS (1, 3, 5 yr): 69.5%, 37.0%, 15.2% vs 86.0%, 57.9%, 38.2% vs 89.2%, 69.4%, 61.2%. OS higher HR vs TACE + RFA (P = 0.009), HR vs TACE (P < 0.001) and TACE + RFA vs TACE (P = 0.004) |
| Wei et al[63], 2020 | TACE + RFA (n = 107) vs HR (n = 79) | Recurrent HCC < 5 cm after HR | N/A | DFS (1, 3, 5 yr): 58.2%, 35.2%, 29.6% vs 64.8%, 41.6%, 38.3% (P = 0.258). OS (1, 3, 5 yr): 84.6%, 66.9%, 49.1% vs 84.8%, 60.2%, 51.9% (P = 0.871). Lower major complication rates (P = 0.009) and shorter hospital stay (P < 0.001) for TACE + RFA |
| Sheta et al[64], 2016 | TACE (n = 20) vs TACE + RFA (n = 20) vs TACE + MWA (n = 10) | Non resectable single lesion HCC > 4 cm | 50% vs 70% vs 80% (CR at 6 mo) | LTR (1, 3, 6 mo): 30% vs 5% vs 0% (P = 0.027); 14.3% vs 15.8% vs 10% (NS); 16.7% vs 12.5% vs 11.1% (NS). Complications rate: 40% vs 10% vs 10% |
| Yuan et al[65], 2019 | TACE + RFA (n = 41) vs TACE + MWA (n = 34) | HCC > 3 cm. HCC 3-5 cm. HCC > 5 cm | 68.3 vs 85.3% (NS). 73.5 vs 88.5% (NS). 42.9 vs 75% (P = 0.041) | DFS (1, 2, 3 yr): 53%, 29%, 12% vs 58%, 38%, 29% (P = 0.07). OS (1, 2, 3 yr): 68%, 36%, 14% vs 79%, 53%, 38% (P = 0.393) |
| Thornton et al[120], 2017 | TAE/TACE + RFA (n = 15) vs TAE/TACE + MWA (n = 20) | BCLC 0 and A | 80% vs 95% (NS) | LTR: 30% vs 0% |
| Vasnani et al[67], 2016 | TACE + RFA (n = 11) vs TACE + MWA (n = 31) | HCC within Milan | 91% vs 67% (CR) 45% vs 35% (rates of complete tumor coagulation on pathology) |
mRECIST: Modified RECIST; n: Number of patients; BCLC: Barcelona Clinic Liver Cancer; TACE: Transarterial chemoembolization; RFA: Radiofrequency ablation; MWA: Microwave ablation; HR: Hepatic resection; OS: Overall survival; PFS: Progression free survival; TTP: Time to progression; LTP: Local tumor progression; DFS: Disease free survival; TR: Tumor recurrence; CR: Complete response; NS: Not significative; N/A: Not applicable.
For small single HCC (2-3 cm) TACE + RFA have similar outcomes to HR in terms of disease free survival (DFS) and OS but with significantly lower complications rate and hospital stay[49]. However, as expected for medium size and larger lesions tumor recurrence (75% vs 35.4%, P = 0.005) and local tumor progression (LTP) (55.7% vs 16%, P = 0.013) are significantly lower for HR[49]. Moreover, it seems that for patients fitting the up-to seven criteria both treatment options provide similar DFS and OS. However for patients outside the Milan criteria but within up to seven criteria there was an increased median OS when HR was performed[50]. For patients within the Milan criteria, Takuma and colleague reported similar DFS and OS, whereas Liu et al[51] found a significantly increased DFS and OS for patients who had undergone HR[51]. In a recent study, Lin et al[52] demonstrated a significantly higher 5 years OS (61.2% vs 38.2%, P = 0.009) for HR in patients with HCC BCLC-B[52].
Beyond a shadow of a doubt, the association of TACE with RFA has proved its benefit in terms of controlling much more suitably early and intermediate primary liver cancer compared to the therapies used alone and we consider that it has gained its place in the treatment of HCC. Moreover, combination therapy is also an important alternative when surgery is not feasible.
TACE combined with MWA: MWA is a dielectric heating technique that generates an electromagnetic field surrounding the needle tip, consequently producing the coagulation necrosis of the target area. Along with RFA, MWA is an established thermal ablation method, best-suited for treating early-stage HCC with curative intent. Being non-inferior to RFA in the standard guideline setting, MWA boosts some theoretical advantages: Higher efficacy in larger nodules, quicker heating, lower procedural time, and higher heating temperatures[53]. Therefore, like RFA, MWA has often been combined with TACE in a multitude of clinical scenarios. These situations either fit outside the standardized BCLC boxes or aim to improve the relatively dim prognosis of the well-established path in intermediate or advanced HCC.
As addressed by Renzulli et al[54], intermediate-and advanced-stage HCC often leads the clinicians in the uncertain waters of an imperfect solution[54]. The typical case resembles the following: Curative intent solution off the board, unsatisfactory gold-standard (TACE), and technically treatable nodules within ablative reach. This has led to a recent surge of interest with regards to combining TACE and MWA, with numerous papers published on this topic in the past decade. The most important articles are comprised in Table 4, which will provide the cornerstone for the upcoming discussion.
Table 4.
Available studies on the transarterial chemoembolization plus microwave ablation
|
Ref.
|
Treatment type, n (%)
|
Clinical scenario
|
Response rate (CR + PR) mRECIST
|
Outcome
|
| Ni et al[59], 2020 | TACE + MWA (546) | BCLC B | N/A | Median PFS: 6.5 mo. Median OS: 35 mo |
| Ni et al[121], 2019 | TACE + MWA (349) | Up to 3 nodules, 5-8 cm diameter | 77.1% | Median PFS: 4.8 mo. Median OS: 28 mo |
| Chen et al[47], 2017 | TACE (96) vs TACE + MWA (48) | HCC ≤ 5 cm | 46.3% vs 92.1% | 2-yr PFS: 57.3% vs 10.4%; 2-yr OS: NS |
| Smolock et al[56], 2018 | TACE (16) vs TACE + MWA (22) | HCC 3-5 cm | 76% vs 95% (NS) | Median PFS: 4.2 mo vs 22.3 mo. Median OS: 14.8 mo vs 18.5 mo. 3-yr OS: 42.1% vs 79% |
| Zheng et al[57], 2018 | TACE (166) vs TACE + MWA (92) | Solitary HCC > 5 cm; 2-3 nodules > 3 cm; 4-10 nodules regardless of size | 55.4% vs 81.5% | Median PFS: 12.5 mo vs 26.6 mo. Median OS: 6.7 mo vs 17.1 mo. 3-yr OS: 11.4% vs 32.6% |
| Zhang et al[60], 2018 | TACE (100) vs TACE + MWA (50) | BCLC-B | 55% vs 74%. At 6-mo, including stable disease | Median PFS: 6.1 mo vs 10.1 mo. Median OS: 14.4 mo vs 18.5 mo. 3-yr OS: 42.1% vs 79%. 5-yr OS: 21% vs 67.7% |
| Wang et al[61], 2020 | TACE (111) vs TACE + MWA (72) | Recurrent (post-surgery) BCLC-B | N/A | Median PFS: N/A. Median OS: 14.4 mo vs 26.7 mo. 5-yr PFS: 13.0% vs 21.7%. 5-yr OS: 27.9% vs 43.3% |
| Li et al[29], 2020 | MWA (88) vs TACE + MWA (62) | BCLC-B | N/A | 3-yr PFS: 34.5% vs 32.5% (NS). 3-yr OS: 47.6% vs 49.2% (NS) |
| Biederman et al[55], 2017 | TACE + MWA (80) vs Radiation segmentectomy (41) | Unresectable, solitary, ≤ 3 cm | CR 82.5% vs 82.9% (NS) | Median PFS: 12.1 mo vs 11.1 mo (NS). 90-d mortality: 0% all groups. Median OS: N/A |
| Ni et al[59], 2020 | TACE + Sorafenib (n = 75) vs TACE + Sorafenib + MWA (77) | BCLC C | 12% vs 46.7% | Median PFS: 3 mo vs 6 mo. Median OS: 13 mo vs 19 mo |
| Sheta et al[64], 2016 | TACE (20) vs TACE + RFA (20) vs TACE + MWA (10) | Unrsesectable, solitary | 6-mo CR–50% vs 70% vs 80% | Median PFS: N/A. Median OS: N/A |
| Wei et al[63], 2020 | TACE + MWA (48) vs TACE + Cryoablation (60) | BCLC B | 73.3% vs 33.4% | Median PFS: 8.8 mo vs 9.3 mo (NS). Median OS: 20.9 vs 13 mo (NS) |
TACE: Transarterial chemoembolization; MWA: Microwave ablation; RFA: Radiofrequency ablation; HCC: Hepatocellular carcinoma; BCLC: Barcelona Clinic Liver Cancer classification system; CR: Complete response; PR: Partial response according to the mRECIST criteria; PFS: Progression-free survival; OS: Overall survival; NS: Non-significant; N/A: Not available.
As shown in Table 4, there is a wide array of clinical scenarios in which the TACE + MWA approach was tested. However, the large majority of the data comes from the most heterogeneous class: Intermediate-stage, BCLC-B. However, most of the HCC disease spectrum stood for trial, ranging from small, solitary unresectable nodules, up to 10 nodules and more advanced BCLC-C tumors[55-58]. The overall results were promising, if not always definitely positive. Therefore, both the empirical and the epistemological conclusions suggest that TACE + MWA can be safely employed whenever it is technically feasible with regards to tumor characteristics, vascularization, and percutaneous approach.
Three key aspects define therapeutic efficacy in HCC: Treatment response according to the mRECIST criteria, PFS, and OS. In this regard, the TACE + MWA combination appeared to exceed the standard of care in most of the clinical scenarios, in all three aspects (Table 4). Of course, the precise survival data varies widely, as the study designs were extremely heterogeneous, even within the same BCLC class. In intermediate-stage HCC, OS ranged from 17.1 mo[57] to 35 mo[59] as it reliably extended OS compared to TACE alone by 4 to 12 mo[56,57,60,61]. To objectivize the apparent benefit, a meta-analysis was recently performed by Liu et al[62], comparing TACE with TACE + MWA in single and up to three HCC nodules exceeding 5 cm[62]. The analysis included over 1700 patients, all from Chinese-conducted studies, and showed a significantly higher OS for the latter (1-year OS rate: RR = 1.36, 95%CI: 1.28–1.44; 2-year OS rate: RR = 1.56, 95%CI: 1.40–1.74, and 3-year OS rate: RR = 2.07, 95%CI: 1.67–2.57, P < 0.001). However, when compared to other non-standard therapies, such as radiation segmentectomy[55] or CA[63] the differences in outcome were not statistically significant, which might further suggest the rather suboptimal standard of care for intermediate HCC. Other clinical scenarios have shown a benefit of combination therapy, as TACE + MWA + Sorafenib outperformed TACE + Sorafenib in off-guideline advanced HCC[58]. However, it is unclear whether the available reports provide sufficient grounding for altering the current time-tested recommendations since no major randomized controlled trials are available. Furthermore, the overall quality of the data can be improved, as most studies are retrospective.
There are only a few small sample studies directly comparing MWA to RFA in combination with TACE, with no significant differences in major outcomes such as PFS and OS[64-66]. However, subgroup analysis suggests a higher response rate in larger tumors for MWA.
Per available data, TACE + MWA appears to laterally exceed the BCLC-B stage. On one hand, it might secure substantial survival benefits for nodules not amenable to curative intent solutions. One such scenario might be large, borderline BCLC-A nodules, unfit for resection due to portal hypertension, yet fit for transplantation, but living in a low-transplantation rate medical system. On the other hand, for BCLC-C patients, TACE + MWA appears to bring survival benefits, especially when adding sorafenib into the mix. The further one navigates beyond BCLC-B, the lesser the strength of the data. Within BCLC-B though, TACE + MWA shines the brightest, appearing to add a substantial survival benefit[67].
TACE combined with other local therapies: In a RCT comparing CA + drug-eluting bead (DEB)-TACE to CA monotherapy in large tumors (mean: 7.2 ± 4.5 cm and 6.5 ± 3.8 cm, respectively), the combination group demonstrated superior OS [16.8 mo vs 13.4 mo, respectively (P = 0.0493)] and significantly increased PFS [8.1 mo vs 6.0 mo, respectively (P = 0.0089)]. Interstitial laser therapy (ILT) uses optical fibers to create cytotoxic temperatures via the conversion of absorbed infrared light to heat with consequent coagulative necrosis[68]. TACE followed by ILT was used in patients with tumors up to 8 cm that has obtained a median OS of 36 mo (95%CI: 29.3–42.6)[69].
Stereotactic body radiation therapy (SBRT) can provide satisfactory local control with a low incidence of radiation-induced liver disease in patients with unresectable HCC that are not amenable to thermal ablation[68]. Via radiosensitization of tissue TACE/TAE may serve as targeting fiducials for SBRT. TACE + SBRT was compared to SBRT in a retrospective study including patients with tumors with a median size of 8.5 cm. An increased 5-year OS was reported in the combination arm vs SBRT arm (46.9 vs 32.9%, P = 0.047)[70]. In addition to cytotoxic properties, SBRT seems to be a potent activator of the adaptive immune system, and thus might stand as an interesting player in the field of immunotherapy[68].
COMBINED TREATMENT TO ENHANCE PALLIATION AND INCREASE SURVIVAL
TACE + systemic therapies–the downfall of an era
As mentioned previously, current guidelines recommend local ablative therapies (RFA/MWA) for patients with early-stage HCC (BCLC-0) and TACE for patients with intermediate-stage BCLC-B or BCLC-A patients not suitable for resection or ablation[2]. Unfortunately, many of these patients experience a progression of the disease and a worsening of the liver function after repeated sessions of percutaneous ablation or TACE[71]. The OPTIMIS study, which assessed the outcomes of HCC patients treated with TACE alone, or with TACE followed by sorafenib found that the proportion of patients with a progressive disease increases with each subsequent TACE, while the objective response rates decline as the number of TACE sessions increased (first TACE: 40%; second TACE: 26%; third TACE: 24%; and fourth TACE: 25%)[72]. Moreover, up to 30% of these patients experienced a deterioration of liver function. Similarly, Hiraoka et al[73] reported that the liver function deteriorated with repeated TACE[73]. These findings emphasize the importance of the appropriate timing of switching from local therapy to systemic therapies to obtain the maximal benefits of other therapies and consequently improve the OS.
As a result of an exclusively arterial vascularization of HCC tumors and comprising the fact that the normal surrounding liver parenchyma is vascularized from branches of the portal vein, TACE and other image-guided transcatheter treatments were born to destruct arterial tumoral vessels and hence inducing tumor necrosis[74]. TACE procedure is based on an intra-arterial infusion of a chemotherapy agent such as doxorubicin or cisplatin, frequently embedded in lipiodol as a vehicle to increase the availability of the drug. Furthermore, the tumoral blood vessels could be embolized with different agents such as gelatine sponge particles, metallic coils, polyvinyl alcohol, starch microspheres and autologous blood clots leading to an increased tumoricidal and ischemic effect[27]. According to the European Association for the Study of the Liver (EASL), the median survival for untreated patients at an intermediate-stage [BCLC-B–multinodular disease, good PS, without vascular invasion or extrahepatic spread] is around 16 mo and in rigorously selected candidates TACE can increase the survival up to 3 years[2].
TACE causes local hypoxia in the tumor, building up an expression of hypoxia response genes in tumor cells regulated by hypoxia-inducible factor-1 alpha (HIF-1α). The response triggers vascular endothelial growth factor (VEGF) expression and thus leading to the formation of neovascularization, and thereby forming a vicious cycle leading to tumor recurrence and metastasis[75]. In this regard, studies have conceded that dynamic changes in serum HIF-1α and VEGF levels occur after TACE in HCC patients[76-78]. Along these lines, Jia et al[76] investigated the expression levels of serum HIF-1α and VEGF before and after TACE and analyzed the correlations between prognosis factors and serum HIF-1α as well as VEGF levels[76]. The serum HIF-1α and VEGF levels of HCC patients pre-TACE, 1 d, 1-wk, 1-mo post-TACE were analyzed using ELISA and compared with that of 20 healthy volunteers[76]. The study revealed that the expression levels of serum HIF-1α and VEGF in HCC patients were significantly higher than those in the control group. One day after TACE, both serum HIF-1α and VEGF levels reached the peak values. One-week post-TACE, expression levels of them were decreased, but still significantly higher than those before TACE. The levels of both HIF-1α and VEGF incomplete response group 1-mo post-TACE were significantly lower than those in partial response, stable disease, or progressive disease groups. Thus, HIF-1α and VEGF might be important predictors of TACE efficacy.
Along these lines, hepatologists and oncologists hypothesized whether the combination of systemic therapy and TACE might be beneficial in terms of survival in HCC patients. In an effort to address this problem, several trials with TACE and anti-angiogenic therapies have emerged. Nevertheless, some challenges have arisen. Firstly, TACE is addressed to BCLC-B class patients which is a heterogeneous group due to the wide range of liver function (Child-Pugh A or B cirrhosis) and variable lesion number and dimension. Secondly, the use of chemotherapy, degree of selectivity and management of adverse effects have to be considered. The GIDEON trial, the first observational trial of more than 3000 patients with HCC BCLC A to C treated with sorafenib or in combination with TACE reported that sorafenib could be safely associated or used sequentially with TACE[79]. Thereby, taking this assumption into account, several randomized controlled trials have been reported, as seen in Table 5.
Table 5.
Chemoembolization plus systemic therapies
|
Trial
|
Experimental arms, n (%)
|
Outcomes
|
| TACE + sorafenib | ||
| SPACE trial (Lencioni et al[80], 2016) | DEB-TACE plus sorafenib (154) vs DEB-TACE plus placebo (153) | 5.6 mo vs 5.5 mo; HR: 0.797 (95%CI: 0.588–1.080); P = 0.072 |
| TACE 2 trial (Meyer et al[81], 2017) | DEB-TACE plus sorafenib (157) vs DEB-TACE plus placebo (156) | 7.8 mo vs 7.7 mo; HR: 1.03 (95%CI: 0.75–1.42); P = 0.85 |
| STAH trial (Park et al[82], 2019) | cTACE plus sorafenib (170) vs sorafenib (169) | 12.8 mo vs 10.8 mo; HR: 0.91 (95%CI: 0.69–1.21); P = 0.290 |
| TACTICS trial (Kudo et al[83], 2020) | cTACE plus sorafenib (80) vs cTACE (76) | 25.2 mo vs 13.5 mo; HR: 0.59 (95%CI: 0.41–0.87); P = 0.006 |
| TACE + other therapies | ||
| BRISK-TA trial (Kudo et al[85], 2014) | cTACE or DEB-TACE plus brivanib (249) vs cTACE plus placebo (253) | 26.4 mo vs 26.1 mo; HR: 0.90 (95%CI: 0.66-1.23); P = 0.53 |
| ORIENTAL trial (Kudo et al[86], 2018) | cTACE plus orantinib (445) vs cTACE plus placebo (444) | 31.1 mo vs 32.3 mo; HR: 1.090 (95%CI: 0.878–1.352); P = 0.435 |
| TACE combined with celecoxib and lanreotide (Tong et al[89], 2017) | TACE (n = 35) vs TACE + C + L (36) | 7.5 mo vs 15.0 mo; HR: 0.534 (95%CI: 0.321-0.888); P = 0.016 |
| TACE combined with thalidomide (Wu et al[87], 2014) | TACE + thalidomide (56) | 21 mo (95%CI: 16–28 mo) |
| TACE plus bevacizumab (Pinter et al[88], 2015) | TACE + bevacizumab (20) vs TACE + placebo (20) | 5.3 mo vs 13.7 mo; HR: 1.7 (95%CI: 0.8-3.6); P = 0.195 |
HR: Hepatic resection; TACE: Transarterial chemoembolization.
SPACE prospective randomized phase II trial included 307 patients with BCLC-B HCC randomly allocated to DEB-TACE with sorafenib 400 mg twice daily and DEB-TACE with placebo. Unfortunately, there was no difference in TTP between the 2 arms (169 d vs 166 d in the sorafenib and placebo arms, respectively, P = 0.072) and no impact on OS (P = 0.29) was observed[80].
A year later a phase III trial of TACE with sorafenib (TACE-2) from the United Kingdom emerged and included 313 patients randomized to sorafenib or placebo with DEB-TACE 2–5 wk later and additional TACE on demand[81]. The study aimed to reduce the adverse effects induced by combination treatment and to increase the prospect of continuing the drug at the time of the TACE procedure. Sadly, as in the SPACE trial, this RCT was also negative with a median PFS of 7.9 vs only 7.8 mo in the sorafenib and placebo arms, respectively (P = 0.94), and median OS of 21.1 and 19.7 mo in the sorafenib and placebo groups, respectively (P = 0.57). Moreover, discouraging results were also reported by the STAH trial in 2019 when comparing the combined treatment with sorafenib alone. The authors justify their results due to delays in starting sorafenib after TACE and/or low daily sorafenib doses[82].
TACTICS trial was the only phase II RCT that attested to the benefits of TACE-sorafenib synergy and met its primary endpoint for the treatment of intermediate stage HCC[83]. The authors reported a median PFS significantly longer in the TACE plus sorafenib group vs TACE alone group (25.2 vs 13.5 mo; P = 0.006). Moreover, the innovation of the trial stands in the modification of PFS, defined as time-to-unTACEable progression (TTUP), characterized as untreatable tumor progression, transient deterioration to Child-Pugh C, or appearance of vascular invasion/extrahepatic spread. Patients in the combination group received sorafenib 400 mg once daily for 2–3 wk before TACE, followed by 800 mg once daily during on-demand conventional TACE sessions until time to untreatable TTUP. Howbeit, a further analysis conceded that the TACTICS trial did not show an improved OS in the combination group as compared with TACE alone although significantly better PFS was consistently observed. However, the OS in TACE plus sorafenib arm showed the longest OS (36.2 mo) with the longest ΔOS (5.4 mo) as compared with the previous TACE combination trials[84]. The authors explain that the major reason for the negative OS result was due to many post-trial active treatments (other systemic treatments and immunotherapy agents) performed in the sorafenib group (76.3%), which implies that the OS endpoint in the TACE combination trial may not be feasible anymore in the current era of personalized medicine and immunotherapy.
It seems likely that other TKIs (brivanib, orantinib) and thalidomide derivatives as well combined with conventional TACE failed to meet the primary endpoint of OS[85-87]. Moreover, the addition of bevacizumab to TACE raised some safety concerns related to sepsis and vascular complications of the combination treatment[88]. Nevertheless, encouraging results were published by Tong et al[89] which have compared TACE alone with TACE combined with the selective COX-2 inhibitor, celecoxib and the somatostatin analog, lanreotide in advanced HCC[89]. The patients receiving the combination therapy had a median OS of 15 mo compared to 7.5 mo for those receiving TACE alone, and a subgroup analysis of advanced patients demonstrated an OS of 13 mo for the combination and 4.5 mo for TACE alone (P = 0.013). Likewise, encouraging results published in 2020 confirmed that the use of lenvatinib-TACE sequential treatment after progression during lenvatinib therapy was associated with better post-progression survival (HR = 0.08; 95%CI: 0.01–0.71; P = 0.023)[90].
Overall, up to this point, the literature has failed to support the use of multi-kinase inhibitors in combination with TACE. However, with the emergence of immunotherapy, combined strategies are encouraged. Moreover, because intermediate stage liver cancer is very heterogeneous a personalized approach is the key to a better outcome for patients.
TACE + immunotherapy–a new beginning?
It has been noted for several years that LRTs result in the release of tumor antigens, which are taken up by antigen-presenting cells (mainly dendritic cells) and which have been shown to activate a tumor-specific immune response a[91,92]. This evidence suggests that l LRTs may boost the response to immune-oncology drugs. Preliminary results of the phase I/II PETAL clinical trial (NCT03397654) showed good tolerability of pembrolizumab after TACE without cumulative side effects. Additionally, several clinical trials testing immune checkpoint inhibitors (ICI) as neoadjuvant or adjuvant therapy in patients treated with LRTs are currently running (Table 6).
Table 6.
Summary of ongoing clinical trials evaluating combination therapy of immune checkpoint inhibitors with locoregional therapies
| BCLC stage | Estimated/included patients | Clinical trial identifier | Phase | Arm |
| 0 | 530 | NCT03383458 | III | Arm 1: RFA/MWA/curative resection + nivolumab (neoadjuvant) vs Arm 2: RFA/MWA/curative resection |
| B | 26 | NCT03397654 (PETAL) | Ib | Single arm: TACE followed by pembrolizumab |
| B | 950 | NCT04246177 LEAP-012 | III | Arm 1: TACE + lenvatinib + pembrolizumab vs Arm 2: TACE |
| B | 49 | NCT03572582 (IMMUTACE) | II | Single arm: TACE + Nivolumab |
| B | 522 | NCT04268888 TACE-3 | II/III | Arm 1: DEB-TACE + Nivolumab vs Arm 2: DEB-TACE |
| B | 765 | NCT04340193, CheckMate 74W | III | Arm 1: TACE + nivolumab + ipilimumab vs Arm 2: TACE + nivolumab + placebo |
| A | 50 | NCT03939975 | II | Single arm: Pembrolizumab or nivolumab or toripalimab. For participants with stable disease or atypical progression to immunotherapy therapy, RFA or MWA is performed additionally |
| B | 130 | NCT03864211 | I/II | Single arm: RFA or MWA followed by Toripalimab |
| B | 61 | NCT01853618 | I/II | Single arm: Tremelimumab + RFA or TACE |
| B | 30 | NCT03638141 | II | Single arm: Initial DEB-TACE followed by Durvalumab + tremelimumab |
| B | 22 | NCT03937830 | II | Single arm: Durvalumab and bevacizumab + TACE |
| B/C | 600 | NCT03778957 EMERALD-1 | III | Arm 1: TACE + durvalumab vs Arm 2: TACE +bevacizumab + durvalumab |
| A/B | 662 | NCT04102098 IMbrave050 | III | Atezolizumab plus bevacizumab in HCC patients at high risk of recurrence after surgical resection or ablation vs Active surveillance in HCC patients at high risk of recurrence after surgical resection or ablation |
BCLC: Barcelona Clinic Liver Cancer Classification; RFA: Radiofrequency ablation; MWA: Microwave ablation; HCC: Hepatocellular carcinoma.
However, despite the strong antitumor response induced by ICI, not all patients experienced an objective response[93]. Currently there is no biomarker to predict response or resistance to immunotherapy in HCC. Emerging evidence revealed that VEGF is not only a proangiogenic factor but that VEGF also plays an important role in the development of the immunosuppressive tumor microenvironment (e.g. inhibition of dendritic cell maturation, accumulation of dendritic cell maturation, accumulation of myeloid-derived suppressor cells and induction of T reg cells). Voron et al[94] showed that targeting VEGF-A can decrease the VEGF-induced expression of inhibitory receptors mediating CD8+T cell exhaustion[94]. Given these results, the association of anti-angiogenic therapy [e.g. inhibitors of VEGF (bevacizumab) or TKIs (lenvatinib)] with ICI seems to overcome tumor-intrinsic resistance to immune checkpoint blockade.
LRTs are minimally invasive therapies; however, it has become clear that the treatment regimens adopted 15 years ago will change in the next few years. In the light of new evidence, three groups might benefit from the combined therapy (locoregional therapy and immunotherapy), including (1) Patients with a high risk of recurrence after a complete response by local ablation; (2) Patients who progressed under TACE; and (3) Patients with poor predictors of response to immunotherapy, when such predictors will be validated, possibly including NAFLD as underlying liver disease.
Currently, there are several trials underway evaluating different combinations (ICI ± anti-angiogenic therapy) as options for patients treated with LRTs (Table 6). It is important to remember that immunotherapies represent a two-edged sword, thus we must find the right timing, dose and combination of immunotherapy for a robust response and minimal side effects.
Ablation + other treatments
HCC is an attractive target for immunotherapy due to several reasons: (1) Usually, HCC develops on a background of chronic inflammation (cirrhosis or chronic hepatitis); (2) In the context of cirrhosis there is an immunosuppressive environment; and (3) Immune evasion was described in patients with liver cancer[95].
Combining thermal ablation with immunotherapy is a very appealing approach. Thermally-induced necrosis can act as a permanent source of tumor antigens, the sub-lethal zone around the necrotic zone can generate inflammatory cytokines, and the thermal stress is capable of making HCC cells more sensitive to immune therapies[48,96,97]. The field of immunotherapy in HCC (different from other cancer entities) was only recently unraveled. However, some preliminary studies with RFA and immunotherapy combinations (immune-ablation) have been already published. When used in a palliative setting, tremelimumab (anti-CTLA4) in combination with RFA or TACE in 32 HCC patients showed indeed interesting results. The patients received a total of six doses of tremelimumab at a 4-wk interval followed by an intentionally incomplete RFA or TACE to induce anti-tumor response at the ablation tumor junction. Patients with clinical response had an increase in CD8+ T cells in tumor biopsies obtained 6 wk after treatment. More interestingly, some patients experienced tumor responses in untreated lesions[98]. In another study, Ma et al[99] injected RetroNectin activated killer cells 14 d after RFA in patients with an HCC less than 4 cm[99]. They reported no severe adverse events, recurrences, or deaths during a seven-month follow-up. Using a similar approach, Cui et al[100] studied the combination of RFA and cellular therapy. Mononuclear cells from 30-HCC patients (early, intermediate and advanced stage) were harvested and induced into natural killer cells, γδT cells and cytokine-induced killer (CIK) cells which were subsequently infused back into RFA-treated patients for three or six courses. The combination improved PFS and reduced HCC recurrence compared to RFA alone[100].
Adjuvant treatment for HCC patients is an unsolved medical need. Using cellular-based immunotherapy Lee et al[101] studied the use of CIK cells injected after RFA (n = 69), ethanol injection (n = 13), or surgery (32) in patients with early-stage HCC[101,102]. They reported a better OS and cancer-specific survival in patients treated with a combined approach vs those treated with RFA, percutaneous ethanol injection (PEI), or surgery alone (P = 0.006 and P = 0.02). Similar findings were also reported in one multicentre randomized open-label phase 3 trial of adjuvant immunotherapy with CIK cells. The study included 230 patients with HCC treated by SR, RFA, or PEI. Patients were assigned randomly to receive immunotherapy or no adjuvant therapy. Adjuvant CIK cell therapy increased both recurrence-free survival and OS[103].
The use of monoclonal antibodies in combination with RFA was also studied in HCC. Either injected during RFA (131I-chTNT) or after RFA (131I metuximab) in an adjuvant setting both combinations showed improved PFS or OS[103,104]. More data about studies investigating the combination of RFA with different immunotherapy strategies are depicted in Table 7.
Table 7.
Radiofrequency ablation combined with immunotherapy
|
Ref.
|
BCLC, n (%)
|
Treatment, n (%)
|
Results
|
Level of evidence
|
| Cui et al[100], 2014 | A (10); B (10); C (10) | RFA and cellular immunotherapy 8-11 d after RFA vs RFA alone | Higher PFS (P < 0.001). Six courses had better survival prognosis than three courses | III |
| Ma et al[99], 2010 | A (7) | RFA and autologous RAK cells 14 d after RFA | No severe adverse events, recurrences or deaths during a seven month follow-up | IV |
| Duffy et al[98], 2017 | C (21) | Tremelimumab every 4 wk and subtotal RFA on day 36 | Median OS-12.3 mo. Median time to progression–7.4 mo. A significant increase of CD3+ and CD8+ immune cells infiltrates in lesions not treated by RFA | III |
| Lee et al[102], 2015 | A (114) | PEI (13); RFA (69); Surgery (32) and adjuvant CIK cells vs PEI, RFA or Surgery alone | OS was significantly longer in the immunotherapy group than in control group (P = 0.006). CSS was significantly longer in the immunotherapy group (P = 0.02) | II |
| Tu et al[103], 2014 | A and B | RFA and monoclonal antibody (131I-chTNT) injection during ablation vs RFA alone | Increased OS. Improved progression-free survival. Increased circulating white blood cells | IV |
| Bian et al[104], 2014 | 0 + A (94); B (33) | RFA and adjuvant 131I metuximab vs RFA alone | Prevention of tumor recurrence | II |
| Lee et al[101], 2019 | 0 and A (239) | RFA or PEI or Surgery plus CIK vs RFA or PEI or surgery alone | Increased recurrence-free survival and OS | I |
BCLC: Barcelona Clinic Liver Cancer Classification; RFA: Radiofrequency ablation; PEI: Percutaneous ethanol injection; CIK: Cytokine-induced Killer; OS: Overall survival; CSS: Cyberchondria severity scale; PFS: Progression-free survival.
Nevertheless, the treatment of HCC will change over the following years. Not much has changed in the last 20 years but the era of immunotherapy has started and we will probably witness groundbreaking changes in the years that will come. New treatments, new guidelines, from single option to multiple options and from RFA to “immune-ablation” the burden has moved from scientist to clinicians: It is an interesting world out there.
Assuming similar suppositions with the TACE procedure, RFA was also combined with VEGF inhibitors. One study showed that Bevacizumab is useful in preventing the rapid progression of residual HCC following RFA in a rat model[105,106]. EMERALD-2 is an ongoing A Phase III, multicenter study of Durvalumab monotherapy or in combination with Bevacizumab as adjuvant therapy in patients with HCC who are at high risk of recurrence after resection or RFA (NCT03847428). Last but not least, nanoparticle-mediated drug delivery systems have also gained ground in oncology. The lyso-thermosensitive liposomal doxorubicin (LTLD) treatment aims to deliver doxorubicin at the peripheral thermal ablation zone, where the thermal elevation is suboptimal. When heated to 40 °C, LTLD releases a 25-fold greater concentration of Doxorubicin. The HEAT study is a global randomized, double-blind, dummy-controlled trial comparing RFA plus LTLD vs RFA alone that enrolled 701 patients with ≤ 4 unresectable up to seven HCC lesions. No differences in PFS and OS were found. The subgroup post hoc analysis showed improved efficacy when the thermal ablation indwell time for a solitary lesion was ≥ 45 min and increased treatment time per tumor volume was associated with better OS in the RFA + LTLD group[107,108]. The subsequent phase III OPTIMA study (NCT02112656) was halted in the interim analysis for futility reasons.
An exhaustive report about the combination of different tyrosine kinase inhibitors, distinctive ICI, or even their combo in the advanced liver cancer setting is beyond the scope of our review. However, we consider it far-reaching to mention one of 2020’s revolutions–the association of bevacizumab (VEGF inhibitor) with atezolizumab (PD-L1 inhibitor) that brings encouraging data for unresectable HCC patients[109].
Although the combination of TACE and TKI’s seemed promising in terms of inhibiting hypoxia-activated tumoral growth factors, studies do not appear to benefit any amalgam therapy compared to TACE monotherapy. To such a degree, one might say that the association of TACE and TKI’s might have seen its downfall. Hence, the attention of the hepatology and oncology community was diverted to a new star-immunotherapy. In both the association with TACE and ablation, ICI have quietly demonstrated their benefit in trials. Although this path is still filled with uncertainty and caveats, in the following years we will witness an HCC guideline revolution.
THE PLACE OF COMBINED THERAPY IN THE BCLC/EASL-HCC GUIDELINES
In tumor boards across the world, the most debatable section of the BCLC classification appears to be ranging from non-resectable BCLC-A to the less severe spectrum of the BCLC-C class, which is, by excellence, the appanage of interventional therapies[110]. Conventionally, the available treatments are dichotomized in curative intent and, possibly mislabelled, palliative therapies. The first group comprises RFA, MWA, PEI, CA, irreversible electroporation, and the latter includes bland trans-arterial embolization, conventional TACE, DEB-TACE, and endovascular radiotherapy-selective internal radiation therapy[2,3].
However, the basis for the aforementioned dichotomy might be fading, as research published in the past decade has shown that combination therapy is at least technically feasible, with the most relevant results being discussed in the previous sections. This has prompted a discussion with regards to the place of combination therapy in the therapeutic algorithm, as some of the approaches might be suited for second-, or even first-line choices for a select group of patients. On the other hand, it might be important to recognize that over-complicating a relatively straightforward algorithm could lead to disputable therapeutic choices and widespread heterogeneous interpretation, rendering data collection difficult.
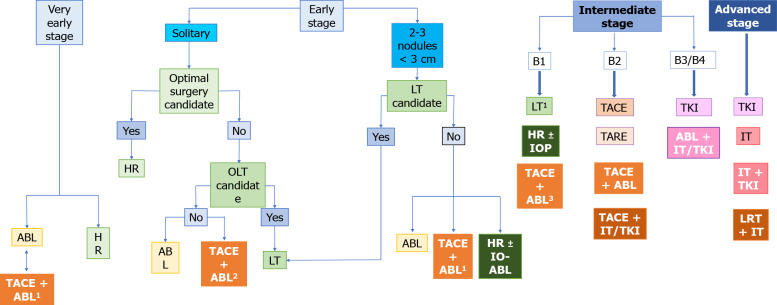
As discussed earlier, one particular combination therapy appears to stand-out among other approaches: TACE-ablation for small, non-resectable HCC (3-5 cm), which is currently at the threshold between BCLC-A and -B. Not only does it improve outcomes in comparison to each individual therapy alone, but, according to limited data, it also appears to generate outcomes similar to surgery, which otherwise would have not been available[30]. A proposed alteration of the BCLC classification, which speculates on the potential role of combination therapies based on the available data previously discussed, is shown in Figure 1.
Figure 1.
The place of combined therapy in the Barcelona clinic liver cancer classification algorithm. 1Lesion not seen at ultrasound or in inappropiate positions; 2Lesion > 3 cm; 3Within Up-to-seven criteria. ABL: Ablation; HR: Hepatic resection; IOP: Intraoperatiove ablation; IT: Immunotherapy; LT: Liver transplantation; LRT: Locoregional therapy; OLT: Orthotopic liver transplantation; TACE: Transarterial chemoembolization; TARE: Transarterial radioembolization; TKI: Tyrosine kinase inhibitors.
CONCLUSION
Of course, our proposal is based on the best available data and still needs further consensus validation, but might provide a foundation for future recommendations, as well as hinting towards potential areas of future development. There is a great need for well-designed, large-scale randomized controlled trials to adequately assess the benefits of combination therapy. Moreover, there are multiple nuances open for debate. Which is the best radiological method for combination therapy: Bland TAE, TACE, or DEB-TACE? Which ablative technique has the most benefits? Should treatments be applied in the same session or sequential? Which is the best sequence? The authors strongly believe that methodically addressing these questions could ultimately lead to a truly personalized approach, hoping to improve the quality of life and OS of HCC patients.
Footnotes
Conflict-of-interest statement: Jean-François Dufour: Advisory committees: Abbvie, Bayer, BMS, Falk, Genfit, Genkyotex, Gilead Science, HepaRegenix, Intercept, Lilly, Merck, Novartis. Speaking and teaching: Abbvie, Bayer, BMS, Genfit, Gilead Science, Novartis.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review started: March 3, 2021
First decision: April 19, 2021
Article in press: November 5, 2021
Specialty type: Oncology
Country/Territory of origin: Switzerland
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Aoki H S-Editor: Fan JR L-Editor: A P-Editor: Fan JR
Contributor Information
Zeno Sparchez, 3rd Medical Department, “Ïuliu Hatieganu” University of Medicine and Pharmacy, Institute for Gastroenterology and Hepatology, Cluj-Napoca 400162, Romania.
Pompilia Radu, Department of Visceral Surgery and Medicine, Hepatology, Inselspital, Bern University Hospital, University of Bern, Bern 3010, Switzerland. radupompilia@yahoo.com.
Adrian Bartos, Department of Surgery, “Ïuliu Hatieganu” University of Medicine and Pharmacy, Institute for Gastroenterology and Hepatology, Cluj-Napoca 400162, Romania.
Iuliana Nenu, 3rd Medical Department, “Ïuliu Hatieganu” University of Medicine and Pharmacy, Institute for Gastroenterology and Hepatology, Cluj-Napoca 400162, Romania.
Rares Craciun, 3rd Medical Department, “Ïuliu Hatieganu” University of Medicine and Pharmacy, Institute for Gastroenterology and Hepatology, Cluj-Napoca 400162, Romania.
Tudor Mocan, 3rd Medical Department, “Ïuliu Hatieganu” University of Medicine and Pharmacy, Institute for Gastroenterology and Hepatology, Cluj-Napoca 400162, Romania.
Adelina Horhat, 3rd Medical Department, “Ïuliu Hatieganu” University of Medicine and Pharmacy, Institute for Gastroenterology and Hepatology, Cluj-Napoca 400162, Romania.
Mihaela Spârchez, Department of Mother and Child, 2nd Paediatric Clinic, “Ïuliu Hatieganu” University of Medicine and Pharmacy, Cluj-Napoca 400177, Romania.
Jean-François Dufour, Department for BioMedical Research, Hepatology, University of Bern, Bern 3008, Switzerland.
References
- 1.GBD 2015 Mortality and Causes of Death Collaborators. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet. 2016;388:1459–1544. doi: 10.1016/S0140-6736(16)31012-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Forner A, Da Fonseca LG, Díaz-González Á, Sanduzzi-Zamparelli M, Reig M, Bruix J. Controversies in the management of hepatocellular carcinoma. JHEP Rep. 2019;1:17–29. doi: 10.1016/j.jhepr.2019.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Vogel A, Cervantes A, Chau I, Daniele B, Llovet JM, Meyer T, Nault JC, Neumann U, Ricke J, Sangro B, Schirmacher P, Verslype C, Zech CJ, Arnold D, Martinelli E ESMO Guidelines Committee. Hepatocellular carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018;29:iv238–iv255. doi: 10.1093/annonc/mdy308. [DOI] [PubMed] [Google Scholar]
- 4.Global Burden of Disease Liver Cancer Collaboration, Akinyemiju T, Abera S, Ahmed M, Alam N, Alemayohu MA, Allen C, Al-Raddadi R, Alvis-Guzman N, Amoako Y, Artaman A, Ayele TA, Barac A, Bensenor I, Berhane A, Bhutta Z, Castillo-Rivas J, Chitheer A, Choi JY, Cowie B, Dandona L, Dandona R, Dey S, Dicker D, Phuc H, Ekwueme DU, Zaki MS, Fischer F, Fürst T, Hancock J, Hay SI, Hotez P, Jee SH, Kasaeian A, Khader Y, Khang YH, Kumar A, Kutz M, Larson H, Lopez A, Lunevicius R, Malekzadeh R, McAlinden C, Meier T, Mendoza W, Mokdad A, Moradi-Lakeh M, Nagel G, Nguyen Q, Nguyen G, Ogbo F, Patton G, Pereira DM, Pourmalek F, Qorbani M, Radfar A, Roshandel G, Salomon JA, Sanabria J, Sartorius B, Satpathy M, Sawhney M, Sepanlou S, Shackelford K, Shore H, Sun J, Mengistu DT, Topór-Mądry R, Tran B, Ukwaja KN, Vlassov V, Vollset SE, Vos T, Wakayo T, Weiderpass E, Werdecker A, Yonemoto N, Younis M, Yu C, Zaidi Z, Zhu L, Murray CJL, Naghavi M, Fitzmaurice C. The Burden of Primary Liver Cancer and Underlying Etiologies From 1990 to 2015 at the Global, Regional, and National Level: Results From the Global Burden of Disease Study 2015. JAMA Oncol. 2017;3:1683–1691. doi: 10.1001/jamaoncol.2017.3055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Llovet JM, Brú C, Bruix J. Prognosis of hepatocellular carcinoma: the BCLC staging classification. Semin Liver Dis. 1999;19:329–338. doi: 10.1055/s-2007-1007122. [DOI] [PubMed] [Google Scholar]
- 6.Qiu J, Chen S, Wu H. Long-term outcomes after hepatic resection combined with radiofrequency ablation for initially unresectable multiple and bilobar liver malignancies. J Surg Res. 2014;188:14–20. doi: 10.1016/j.jss.2013.11.1120. [DOI] [PubMed] [Google Scholar]
- 7.Schindl MJ, Redhead DN, Fearon KC, Garden OJ, Wigmore SJ Edinburgh Liver Surgery and Transplantation Experimental Research Group (eLISTER) The value of residual liver volume as a predictor of hepatic dysfunction and infection after major liver resection. Gut. 2005;54:289–296. doi: 10.1136/gut.2004.046524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Forner A, Reig ME, de Lope CR, Bruix J. Current strategy for staging and treatment: the BCLC update and future prospects. Semin Liver Dis. 2010;30:61–74. doi: 10.1055/s-0030-1247133. [DOI] [PubMed] [Google Scholar]
- 9.Arellano RS. What's New in Percutaneous Ablative Strategies for Hepatocellular Carcinoma and Colorectal Hepatic Metastases? Curr Oncol Rep. 2020;22:105. doi: 10.1007/s11912-020-00967-y. [DOI] [PubMed] [Google Scholar]
- 10.de Jong KP, Wertenbroek MW. Liver resection combined with local ablation: where are the limits? Dig Surg. 2011;28:127–133. doi: 10.1159/000323823. [DOI] [PubMed] [Google Scholar]
- 11.Eisele RM, Zhukowa J, Chopra S, Schmidt SC, Neumann U, Pratschke J, Schumacher G. Results of liver resection in combination with radiofrequency ablation for hepatic malignancies. Eur J Surg Oncol. 2010;36:269–274. doi: 10.1016/j.ejso.2009.07.188. [DOI] [PubMed] [Google Scholar]
- 12.Pawlik TM, Izzo F, Cohen DS, Morris JS, Curley SA. Combined resection and radiofrequency ablation for advanced hepatic malignancies: results in 172 patients. Ann Surg Oncol. 2003;10:1059–1069. doi: 10.1245/ASO.2003.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ledoux G, Amroun K, Rhaiem R, Cagniet A, Aghaei A, Bouche O, Hoeffel C, Sommacale D, Piardi T, Kianmanesh R. Fully laparoscopic thermo-ablation of liver malignancies with or without liver resection: tumor location is an independent local recurrence risk factor. Surg Endosc. 2021;35:845–853. doi: 10.1007/s00464-020-07456-0. [DOI] [PubMed] [Google Scholar]
- 14.Belli G, D'Agostino A, Fantini C, Cioffi L, Belli A, Russolillo N, Langella S. Laparoscopic radiofrequency ablation combined with laparoscopic liver resection for more than one HCC on cirrhosis. Surg Laparosc Endosc Percutan Tech. 2007;17:331–334. doi: 10.1097/SLE.0b013e31806d9c65. [DOI] [PubMed] [Google Scholar]
- 15.Donadon M, Costa G, Torzilli G. State of the art of intraoperative ultrasound in liver surgery: current use for staging and resection guidance. Ultraschall Med. 2014;35:500–511; quiz 512-513. doi: 10.1055/s-0034-1385515. [DOI] [PubMed] [Google Scholar]
- 16.Mathuram Thiyagarajan U, Brown R, Dasari BVM. Liver Resection and Role of Extended Cytology and Histology: Response to: Viganò L, Costa G, Cimino MM, Procopio F, Donadon M, Del Fabbro D, Belghiti J, Kokudo N, Makuuchi M, Vauthey JN, Torzilli G. R1 Resection for Colorectal Liver Metastases: a Survey Questioning Surgeons about Its Incidence, Clinical Impact, and Management. J Gastrointest Surg. 2018 Oct; 22(10):1752-1763. J Gastrointest Surg. 2019;23:1283–1284. doi: 10.1007/s11605-019-04189-x. [DOI] [PubMed] [Google Scholar]
- 17.Ju M, Yopp AC. The Utility of Anatomical Liver Resection in Hepatocellular Carcinoma: Associated with Improved Outcomes or Lack of Supportive Evidence? Cancers (Basel) 2019;11 doi: 10.3390/cancers11101441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Poch FGM, Neizert CA, Gemeinhardt O, Geyer B, Eminger K, Rieder C, Niehues SM, Vahldiek J, Thieme SF, Lehmann KS. Intermittent Pringle maneuver may be beneficial for radiofrequency ablations in situations with tumor-vessel proximity. Innov Surg Sci. 2018;3:245–251. doi: 10.1515/iss-2018-0008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bartoș A, Bartos D, Spârchez Z, Iancu I, Ciobanu L, Iancu C, Breazu C. Laparoscopic Contrast-Enhanced Ultrasonography for Real Time Monitoring of Laparoscopic Radiofrequency Ablation for Hepatocellular Carcinoma: an Observational Pilot Study. J Gastrointestin Liver Dis. 2019;28:457–462. doi: 10.15403/jgld-263. [DOI] [PubMed] [Google Scholar]
- 20.Abdalla EK, Vauthey JN, Ellis LM, Ellis V, Pollock R, Broglio KR, Hess K, Curley SA. Recurrence and outcomes following hepatic resection, radiofrequency ablation, and combined resection/ablation for colorectal liver metastases. Ann Surg. 2004;239:818–825; discussion 825-827. doi: 10.1097/01.sla.0000128305.90650.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Xu LL, Zhang M, Yi PS, Zheng XB, Feng L, Lan C, Tang JW, Ren SS, Xu MQ. Hepatic resection combined with radiofrequency ablation vs hepatic resection alone for multifocal hepatocellular carcinomas: A meta-analysis. J Huazhong Univ Sci Technolog Med Sci. 2017;37:974–980. doi: 10.1007/s11596-017-1836-3. [DOI] [PubMed] [Google Scholar]
- 22.Chiappa A, Bertani E, Zbar AP, Foschi D, Fazio N, Zampino M, Belluco C, Orsi F, Della Vigna P, Bonomo G, Venturino M, Ferrari C, Biffi R. Optimizing treatment of hepatic metastases from colorectal cancer: Resection or resection plus ablation? Int J Oncol. 2016;48:1280–1289. doi: 10.3892/ijo.2016.3324. [DOI] [PubMed] [Google Scholar]
- 23.Hou YF, Wei YG, Yang JY, Wen TF, Xu MQ, Yan LN, Li B. Combined hepatectomy and radiofrequency ablation vs TACE in improving survival of patients with unresectable BCLC stage B HCC. Hepatobiliary Pancreat Dis Int. 2016;15:378–385. doi: 10.1016/s1499-3872(16)60089-9. [DOI] [PubMed] [Google Scholar]
- 24.Zhang T, Zeng Y, Huang J, Liao M, Wu H. Combined resection with radiofrequency ablation for bilobar hepatocellular carcinoma: a single-center experience. J Surg Res. 2014;191:370–378. doi: 10.1016/j.jss.2014.03.048. [DOI] [PubMed] [Google Scholar]
- 25.Huang Y, Song J, Zheng J, Jiang L, Yan L, Yang J, Zeng Y, Wu H. Comparison of Hepatic Resection Combined with Intraoperative Radiofrequency Ablation, or Hepatic Resection Alone, for Hepatocellular Carcinoma Patients with Multifocal Tumors Meeting the University of California San Francisco (UCSF) Criteria: A Propensity Score-Matched Analysis. Ann Surg Oncol. 2020;27:2334–2345. doi: 10.1245/s10434-020-08231-0. [DOI] [PubMed] [Google Scholar]
- 26.Philips P, Scoggins CR, Rostas JK, McMasters KM, Martin RC. Safety and advantages of combined resection and microwave ablation in patients with bilobar hepatic malignancies. Int J Hyperthermia. 2017;33:43–50. doi: 10.1080/02656736.2016.1211751. [DOI] [PubMed] [Google Scholar]
- 27.Li W, Ni CF. Current status of the combination therapy of transarterial chemoembolization and local ablation for hepatocellular carcinoma. Abdom Radiol (NY) 2019;44:2268–2275. doi: 10.1007/s00261-019-01943-2. [DOI] [PubMed] [Google Scholar]
- 28.Xu Z, Xie H, Zhou L, Chen X, Zheng S. The Combination Strategy of Transarterial Chemoembolization and Radiofrequency Ablation or Microwave Ablation against Hepatocellular Carcinoma. Anal Cell Pathol (Amst) 2019;2019:8619096. doi: 10.1155/2019/8619096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Li X, Chen B, An C, Cheng Z, Han Z, Liu F, Yu J, Liang P. Transarterial chemoembolization combined with microwave ablation vs microwave ablation only for Barcelona clinic liver cancer Stage B hepatocellular carcinoma: A propensity score matching study. J Cancer Res Ther. 2020;16:1027–1037. doi: 10.4103/jcrt.JCRT_380_19. [DOI] [PubMed] [Google Scholar]
- 30.Young S, Golzarian J. Locoregional Therapies in the Treatment of 3- to 5-cm Hepatocellular Carcinoma: Critical Review of the Literature. AJR Am J Roentgenol. 2020;215:223–234. doi: 10.2214/AJR.19.22098. [DOI] [PubMed] [Google Scholar]
- 31.Song MJ, Bae SH, Lee JS, Lee SW, Song DS, You CR, Choi JY, Yoon SK. Combination transarterial chemoembolization and radiofrequency ablation therapy for early hepatocellular carcinoma. Korean J Intern Med. 2016;31:242–252. doi: 10.3904/kjim.2015.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lee H, Yoon CJ, Seong NJ, Jeong SH, Kim JW. Comparison of Combined Therapy Using Conventional Chemoembolization and Radiofrequency Ablation Versus Conventional Chemoembolization for Ultrasound-Invisible Early-Stage Hepatocellular Carcinoma (Barcelona Clinic Liver Cancer Stage 0 or A) Korean J Radiol. 2018;19:1130–1139. doi: 10.3348/kjr.2018.19.6.1130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Liu F, Chen M, Mei J, Xu L, Guo R, Lin X, Zhang Y, Peng Z. Transarterial Chemoembolization Combined with Radiofrequency Ablation in the Treatment of Stage B1 Intermediate Hepatocellular Carcinoma. J Oncol. 2019;2019:6298502. doi: 10.1155/2019/6298502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hirooka M, Hiraoka A, Ochi H, Kisaka Y, Joko K, Michitaka K, Hiasa Y. Transcatheter Arterial Chemoembolization With or Without Radiofrequency Ablation: Outcomes in Patients With Barcelona Clinic Liver Cancer Stage B Hepatocellular Carcinoma. AJR Am J Roentgenol. 2018;210:891–898. doi: 10.2214/AJR.17.18177. [DOI] [PubMed] [Google Scholar]
- 35.Ren Y, Cao Y, Ma H, Kan X, Zhou C, Liu J, Shi Q, Feng G, Xiong B, Zheng C. Improved clinical outcome using transarterial chemoembolization combined with radiofrequency ablation for patients in Barcelona clinic liver cancer stage A or B hepatocellular carcinoma regardless of tumor size: results of a single-center retrospective case control study. BMC Cancer. 2019;19:983. doi: 10.1186/s12885-019-6237-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chu HH, Kim JH, Yoon HK, Ko HK, Gwon DI, Kim PN, Sung KB, Ko GY, Kim SY, Park SH. Chemoembolization Combined with Radiofrequency Ablation for Medium-Sized Hepatocellular Carcinoma: A Propensity-Score Analysis. J Vasc Interv Radiol. 2019;30:1533–1543. doi: 10.1016/j.jvir.2019.06.006. [DOI] [PubMed] [Google Scholar]
- 37.Liu W, Xu H, Ying X, Zhang D, Lai L, Wang L, Tu J, Ji J. Radiofrequency Ablation (RFA) Combined with Transcatheter Arterial Chemoembolization (TACE) for Patients with Medium-to-Large Hepatocellular Carcinoma: A Retrospective Analysis of Long-Term Outcome. Med Sci Monit. 2020;26:e923263. doi: 10.12659/MSM.923263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hyun D, Cho SK, Shin SW, Park KB, Park HS, Choo SW, Do YS, Choo IW, Lee MW, Rhim H, Lim HK. Early Stage Hepatocellular Carcinomas Not Feasible for Ultrasound-Guided Radiofrequency Ablation: Comparison of Transarterial Chemoembolization Alone and Combined Therapy with Transarterial Chemoembolization and Radiofrequency Ablation. Cardiovasc Intervent Radiol. 2016;39:417–425. doi: 10.1007/s00270-015-1194-0. [DOI] [PubMed] [Google Scholar]
- 39.Yang BS, Liu LX, Yuan M, Hou YB, Li QT, Zhou S, Shi YX, Gao BL. Multiple imaging modality-guided radiofrequency ablation combined with transarterial chemoembolization for hepatocellular carcinoma in special locations. Diagn Interv Radiol. 2020;26:131–139. doi: 10.5152/dir.2019.18540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yang DJ, Luo KL, Liu H, Cai B, Tao GQ, Su XF, Hou XJ, Ye F, Li XY, Tian ZQ. Meta-analysis of transcatheter arterial chemoembolization plus radiofrequency ablation vs transcatheter arterial chemoembolization alone for hepatocellular carcinoma. Oncotarget. 2017;8:2960–2970. doi: 10.18632/oncotarget.13813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cao JH, Zhou J, Zhang XL, Ding X, Long QY. Meta-analysis on radiofrequency ablation in combination with transarterial chemoembolization for the treatment of hepatocellular carcinoma. J Huazhong Univ Sci Technolog Med Sci. 2014;34:692–700. doi: 10.1007/s11596-014-1338-5. [DOI] [PubMed] [Google Scholar]
- 42.Wang Y, Deng T, Zeng L, Chen W. Efficacy and safety of radiofrequency ablation and transcatheter arterial chemoembolization for treatment of hepatocellular carcinoma: A meta-analysis. Hepatol Res. 2016;46:58–71. doi: 10.1111/hepr.12568. [DOI] [PubMed] [Google Scholar]
- 43.Lu Z, Wen F, Guo Q, Liang H, Mao X, Sun H. Radiofrequency ablation plus chemoembolization vs radiofrequency ablation alone for hepatocellular carcinoma: a meta-analysis of randomized-controlled trials. Eur J Gastroenterol Hepatol. 2013;25:187–194. doi: 10.1097/MEG.0b013e32835a0a07. [DOI] [PubMed] [Google Scholar]
- 44.Ni JY, Liu SS, Xu LF, Sun HL, Chen YT. Meta-analysis of radiofrequency ablation in combination with transarterial chemoembolization for hepatocellular carcinoma. World J Gastroenterol. 2013;19:3872–3882. doi: 10.3748/wjg.v19.i24.3872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Liu Z, Gao F, Yang G, Singh S, Lu M, Zhang T, Zhong Z, Zhang F, Tang R. Combination of radiofrequency ablation with transarterial chemoembolization for hepatocellular carcinoma: an up-to-date meta-analysis. Tumour Biol. 2014;35:7407–7413. doi: 10.1007/s13277-014-1976-z. [DOI] [PubMed] [Google Scholar]
- 46.Chen L, Sun J, Yang X. Radiofrequency ablation-combined multimodel therapies for hepatocellular carcinoma: Current status. Cancer Lett. 2016;370:78–84. doi: 10.1016/j.canlet.2015.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Chen QF, Jia ZY, Yang ZQ, Fan WL, Shi HB. Transarterial Chemoembolization Monotherapy Versus Combined Transarterial Chemoembolization-Microwave Ablation Therapy for Hepatocellular Carcinoma Tumors ≤5 cm: A Propensity Analysis at a Single Center. Cardiovasc Intervent Radiol. 2017;40:1748–1755. doi: 10.1007/s00270-017-1736-8. [DOI] [PubMed] [Google Scholar]
- 48.Gui CH, Baey S, D'cruz RT, Shelat VG. Trans-arterial chemoembolization + radiofrequency ablation vs surgical resection in hepatocellular carcinoma - A meta-analysis. Eur J Surg Oncol. 2020;46:763–771. doi: 10.1016/j.ejso.2020.01.004. [DOI] [PubMed] [Google Scholar]
- 49.Saviano A, Iezzi R, Giuliante F, Salvatore L, Mele C, Posa A, Ardito F, De Gaetano AM, Pompili M HepatoCATT Study Group. Liver Resection vs Radiofrequency Ablation plus Transcatheter Arterial Chemoembolization in Cirrhotic Patients with Solitary Large Hepatocellular Carcinoma. J Vasc Interv Radiol. 2017;28:1512–1519. doi: 10.1016/j.jvir.2017.06.016. [DOI] [PubMed] [Google Scholar]
- 50.Pan T, Mu LW, Wu C, Wu XQ, Xie QK, Li XS, Lyu N, Li SL, Deng HJ, Jiang ZB, Lin AH, Zhao M. Comparison of Combined Transcatheter Arterial Chemoembolization and CT-guided Radiofrequency Ablation with Surgical Resection in Patients with Hepatocellular Carcinoma within the Up-to-seven Criteria: A Multicenter Case-matched Study. J Cancer. 2017;8:3506–3513. doi: 10.7150/jca.19964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Liu H, Wang ZG, Fu SY, Li AJ, Pan ZY, Zhou WP, Lau WY, Wu MC. Randomized clinical trial of chemoembolization plus radiofrequency ablation vs partial hepatectomy for hepatocellular carcinoma within the Milan criteria. Br J Surg. 2016;103:348–356. doi: 10.1002/bjs.10061. [DOI] [PubMed] [Google Scholar]
- 52.Lin CW, Chen YS, Lo GH, Hsu YC, Hsu CC, Wu TC, Yeh JH, Hsiao P, Hsieh PM, Lin HY, Shu CW, Hung CM. Comparison of overall survival on surgical resection vs transarterial chemoembolization with or without radiofrequency ablation in intermediate stage hepatocellular carcinoma: a propensity score matching analysis. BMC Gastroenterol. 2020;20:99. doi: 10.1186/s12876-020-01235-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Facciorusso A, Di Maso M, Muscatiello N. Microwave ablation vs radiofrequency ablation for the treatment of hepatocellular carcinoma: A systematic review and meta-analysis. Int J Hyperthermia. 2016;32:339–344. doi: 10.3109/02656736.2015.1127434. [DOI] [PubMed] [Google Scholar]
- 54.Renzulli M, Tovoli F, Clemente A, Ierardi AM, Pettinari I, Peta G, Marasco G, Festi D, Piscaglia F, Cappabianca S, Carrafiello G, Golfieri R. Ablation for hepatocellular carcinoma: beyond the standard indications. Med Oncol. 2020;37:23. doi: 10.1007/s12032-020-01348-y. [DOI] [PubMed] [Google Scholar]
- 55.Biederman DM, Titano JJ, Bishay VL, Durrani RJ, Dayan E, Tabori N, Patel RS, Nowakowski FS, Fischman AM, Kim E. Radiation Segmentectomy vs TACE Combined with Microwave Ablation for Unresectable Solitary Hepatocellular Carcinoma Up to 3 cm: A Propensity Score Matching Study. Radiology. 2017;283:895–905. doi: 10.1148/radiol.2016160718. [DOI] [PubMed] [Google Scholar]
- 56.Smolock AR, Cristescu MM, Hinshaw A, Woo KM, Wells SA, Ziemlewicz TJ, Lubner MG, Dalvie PS, Louis Hinshaw J, Brace CL, Ozkan OS, Lee FT Jr, Laeseke P. Combination transarterial chemoembolization and microwave ablation improves local tumor control for 3- to 5-cm hepatocellular carcinoma when compared with transarterial chemoembolization alone. Abdom Radiol (NY) 2018;43:2497–2504. doi: 10.1007/s00261-018-1464-9. [DOI] [PubMed] [Google Scholar]
- 57.Zheng L, Li HL, Guo CY, Luo SX. Comparison of the Efficacy and Prognostic Factors of Transarterial Chemoembolization Plus Microwave Ablation vs Transarterial Chemoembolization Alone in Patients with a Large Solitary or Multinodular Hepatocellular Carcinomas. Korean J Radiol. 2018;19:237–246. doi: 10.3348/kjr.2018.19.2.237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ni JY, Sun HL, Luo JH, Jiang XY, Chen D, Wang WD, Chen YT, Huang JH, Xu LF. Transarterial Chemoembolization and Sorafenib Combined with Microwave Ablation for Advanced Primary Hepatocellular Carcinoma: A Preliminary Investigation of Safety and Efficacy. Cancer Manag Res. 2019;11:9939–9950. doi: 10.2147/CMAR.S224532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ni JY, Fang ZT, Sun HL, An C, Huang ZM, Zhang TQ, Jiang XY, Chen YT, Xu LF, Huang JH. A nomogram to predict survival of patients with intermediate-stage hepatocellular carcinoma after transarterial chemoembolization combined with microwave ablation. Eur Radiol. 2020;30:2377–2390. doi: 10.1007/s00330-019-06438-8. [DOI] [PubMed] [Google Scholar]
- 60.Zhang R, Shen L, Zhao L, Guan Z, Chen Q, Li W. Combined transarterial chemoembolization and microwave ablation vs transarterial chemoembolization in BCLC stage B hepatocellular carcinoma. Diagn Interv Radiol. 2018;24:219–224. doi: 10.5152/dir.2018.17528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wang C, Liao Y, Qiu J, Yuan Y, Zhang Y, Li K, Zou R, Wang Y, Zuo D, He W, Zheng Y, Li B. Transcatheter arterial chemoembolization alone or combined with ablation for recurrent intermediate-stage hepatocellular carcinoma: a propensity score matching study. J Cancer Res Clin Oncol. 2020;146:2669–2680. doi: 10.1007/s00432-020-03254-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Liu C, Li T, He JT, Shao H. TACE combined with microwave ablation therapy vs. TACE alone for treatment of early- and intermediate-stage hepatocellular carcinomas larger than 5 cm: a meta-analysis. Diagn Interv Radiol. 2020;26:575–583. doi: 10.5152/dir.2020.19615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wei J, Cui W, Fan W, Wang Y, Li J. Unresectable Hepatocellular Carcinoma: Transcatheter Arterial Chemoembolization Combined With Microwave Ablation vs. Combined With Cryoablation. Front Oncol. 2020;10:1285. doi: 10.3389/fonc.2020.01285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sheta E, El-Kalla F, El-Gharib M, Kobtan A, Elhendawy M, Abd-Elsalam S, Mansour L, Amer I. Comparison of single-session transarterial chemoembolization combined with microwave ablation or radiofrequency ablation in the treatment of hepatocellular carcinoma: a randomized-controlled study. Eur J Gastroenterol Hepatol. 2016;28:1198–1203. doi: 10.1097/MEG.0000000000000688. [DOI] [PubMed] [Google Scholar]
- 65.Yuan P, Zhang Z, Kuai J. Analysis on efficacy and safety of TACE in combination with RFA and MWA in the treatment of middle and large primary hepatic carcinoma. J BUON. 2019;24:163–170. [PubMed] [Google Scholar]
- 66.Abdelaziz AO, Abdelmaksoud AH, Nabeel MM, Shousha HI, Cordie AA, Mahmoud ShH, Medhat E, Omran D, Elbaz TM. Transarterial Chemoembolization Combined with Either Radiofrequency or Microwave Ablation in Management of Hepatocellular Carcinoma. Asian Pac J Cancer Prev. 2017;18:189–194. doi: 10.22034/APJCP.2017.18.1.189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Vasnani R, Ginsburg M, Ahmed O, Doshi T, Hart J, Te H, Van Ha TG. Radiofrequency and microwave ablation in combination with transarterial chemoembolization induce equivalent histopathologic coagulation necrosis in hepatocellular carcinoma patients bridged to liver transplantation. Hepatobiliary Surg Nutr. 2016;5:225–233. doi: 10.21037/hbsn.2016.01.05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Lewis AR, Padula CA, McKinney JM, Toskich BB. Ablation plus Transarterial Embolic Therapy for Hepatocellular Carcinoma Larger than 3 cm: Science, Evidence, and Future Directions. Semin Intervent Radiol. 2019;36:303–309. doi: 10.1055/s-0039-1697641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Zangos S, Eichler K, Balzer JO, Straub R, Hammerstingl R, Herzog C, Lehnert T, Heller M, Thalhammer A, Mack MG, Vogl TJ. Large-sized hepatocellular carcinoma (HCC): a neoadjuvant treatment protocol with repetitive transarterial chemoembolization (TACE) before percutaneous MR-guided laser-induced thermotherapy (LITT) Eur Radiol. 2007;17:553–563. doi: 10.1007/s00330-006-0343-x. [DOI] [PubMed] [Google Scholar]
- 70.Su TS, Lu HZ, Cheng T, Zhou Y, Huang Y, Gao YC, Tang MY, Jiang HY, Lian ZP, Hou EC, Liang P. Long-term survival analysis in combined transarterial embolization and stereotactic body radiation therapy vs stereotactic body radiation monotherapy for unresectable hepatocellular carcinoma >5 cm. BMC Cancer. 2016;16:834. doi: 10.1186/s12885-016-2894-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Wang B, Xu H, Gao ZQ, Ning HF, Sun YQ, Cao GW. Increased expression of vascular endothelial growth factor in hepatocellular carcinoma after transcatheter arterial chemoembolization. Acta Radiol. 2008;49:523–529. doi: 10.1080/02841850801958890. [DOI] [PubMed] [Google Scholar]
- 72.Peck-Radosavljevic M, Kudo M, Raoul J-L, Lee HC, Decaens T, Heo J, Lin S-M, Shan H, Yang Y, Bayh I, Nakajima K, Cheng A-L. Outcomes of patients with hepatocellular carcinoma (HCC) treated with transarterial chemoembolization (TACE): Global OPTIMIS final analysis. J Clin Oncol. 2018;36:4018. [Google Scholar]
- 73.Hiraoka A, Kumada T, Kudo M, Hirooka M, Koizumi Y, Hiasa Y, Tajiri K, Toyoda H, Tada T, Ochi H, Joko K, Shimada N, Deguchi A, Ishikawa T, Imai M, Tsuji K, Michitaka K Real-life Practice Experts for HCC (RELPEC) Study Group and HCC 48 Group (hepatocellular carcinoma experts from 48 clinics) Hepatic Function during Repeated TACE Procedures and Prognosis after Introducing Sorafenib in Patients with Unresectable Hepatocellular Carcinoma: Multicenter Analysis. Dig Dis. 2017;35:602–610. doi: 10.1159/000480256. [DOI] [PubMed] [Google Scholar]
- 74.Lencioni R. Loco-regional treatment of hepatocellular carcinoma. Hepatology. 2010;52:762–773. doi: 10.1002/hep.23725. [DOI] [PubMed] [Google Scholar]
- 75.Unruh A, Ressel A, Mohamed HG, Johnson RS, Nadrowitz R, Richter E, Katschinski DM, Wenger RH. The hypoxia-inducible factor-1α is a negative factor for tumor therapy. Oncogene. 2003;22:3213–20. doi: 10.1038/sj.onc.1206385. [DOI] [PubMed] [Google Scholar]
- 76.Jia ZZ, Jiang GM, Feng YL. Serum HIF-1alpha and VEGF levels pre- and post-TACE in patients with primary liver cancer. Chin Med Sci J. 2011;26:158–162. doi: 10.1016/s1001-9294(11)60041-2. [DOI] [PubMed] [Google Scholar]
- 77.Li X, Feng GS, Zheng CS, Zhuo CK, Liu X. Expression of plasma vascular endothelial growth factor in patients with hepatocellular carcinoma and effect of transcatheter arterial chemoembolization therapy on plasma vascular endothelial growth factor level. World J Gastroenterol. 2004;10:2878–2882. doi: 10.3748/wjg.v10.i19.2878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Jia ZZ, Huang YQ, Feng YL, Jiang GM. [Correlations between serum hypoxia inducible factor-1α, vascular endothelial growth factor and computed tomography perfusion imaging at pre-and post-TACE in patients with primary hepatic carcinoma] Zhonghua Yi Xue Za Zhi. 2013;93:1472–1475. [PubMed] [Google Scholar]
- 79.Geschwind JF, Kudo M, Marrero JA, Venook AP, Chen XP, Bronowicki JP, Dagher L, Furuse J, Ladrón de Guevara L, Papandreou C, Sanyal AJ, Takayama T, Ye SL, Yoon SK, Nakajima K, Lehr R, Heldner S, Lencioni R. TACE Treatment in Patients with Sorafenib-treated Unresectable Hepatocellular Carcinoma in Clinical Practice: Final Analysis of GIDEON. Radiology. 2016;279:630–640. doi: 10.1148/radiol.2015150667. [DOI] [PubMed] [Google Scholar]
- 80.Lencioni R, Llovet JM, Han G, Tak WY, Yang J, Guglielmi A, Paik SW, Reig M, Kim DY, Chau GY, Luca A, Del Arbol LR, Leberre MA, Niu W, Nicholson K, Meinhardt G, Bruix J. Sorafenib or placebo plus TACE with doxorubicin-eluting beads for intermediate stage HCC: The SPACE trial. J Hepatol. 2016;64:1090–1098. doi: 10.1016/j.jhep.2016.01.012. [DOI] [PubMed] [Google Scholar]
- 81.Meyer T, Fox R, Ma YT, Ross PJ, James MW, Sturgess R, Stubbs C, Stocken DD, Wall L, Watkinson A, Hacking N, Evans TRJ, Collins P, Hubner RA, Cunningham D, Primrose JN, Johnson PJ, Palmer DH. Sorafenib in combination with transarterial chemoembolisation in patients with unresectable hepatocellular carcinoma (TACE 2): a randomised placebo-controlled, double-blind, phase 3 trial. Lancet Gastroenterol Hepatol. 2017;2:565–575. doi: 10.1016/S2468-1253(17)30156-5. [DOI] [PubMed] [Google Scholar]
- 82.Park JW, Kim YJ, Kim DY, Bae SH, Paik SW, Lee YJ, Kim HY, Lee HC, Han SY, Cheong JY, Kwon OS, Yeon JE, Kim BH, Hwang J. Sorafenib with or without concurrent transarterial chemoembolization in patients with advanced hepatocellular carcinoma: The phase III STAH trial. J Hepatol. 2019;70:684–691. doi: 10.1016/j.jhep.2018.11.029. [DOI] [PubMed] [Google Scholar]
- 83.Kudo M, Ueshima K, Ikeda M, Torimura T, Tanabe N, Aikata H, Izumi N, Yamasaki T, Nojiri S, Hino K, Tsumura H, Kuzuya T, Isoda N, Yasui K, Aino H, Ido A, Kawabe N, Nakao K, Wada Y, Yokosuka O, Yoshimura K, Okusaka T, Furuse J, Kokudo N, Okita K, Johnson PJ, Arai Y TACTICS study group. Randomised, multicentre prospective trial of transarterial chemoembolisation (TACE) plus sorafenib as compared with TACE alone in patients with hepatocellular carcinoma: TACTICS trial. Gut. 2020;69:1492–1501. doi: 10.1136/gutjnl-2019-318934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Kudo M, Ueshima K, Ikeda M, Torimura T, Aikata H, Izumi N, Yamasaki T, Hino K, Kuzuya T, Isoda N, Yasui K, Aino H, Ido A, Kawabe N, Nakao K, Wada Y, Yoshimura K, Okusaka T, Furuse J, Arai Y. TACTICS: Final overall survival (OS) data from a randomized, open label, multicenter, phase II trial of transcatheter arterial chemoembolization (TACE) therapy in combination with sorafenib as compared with TACE alone in patients (pts) with hepatocellular. J Clin Oncol. 2021;39:270. [Google Scholar]
- 85.Kudo M, Han G, Finn RS, Poon RT, Blanc JF, Yan L, Yang J, Lu L, Tak WY, Yu X, Lee JH, Lin SM, Wu C, Tanwandee T, Shao G, Walters IB, Dela Cruz C, Poulart V, Wang JH. Brivanib as adjuvant therapy to transarterial chemoembolization in patients with hepatocellular carcinoma: A randomized phase III trial. Hepatology. 2014;60:1697–1707. doi: 10.1002/hep.27290. [DOI] [PubMed] [Google Scholar]
- 86.Kudo M, Cheng AL, Park JW, Park JH, Liang PC, Hidaka H, Izumi N, Heo J, Lee YJ, Sheen IS, Chiu CF, Arioka H, Morita S, Arai Y. Orantinib vs placebo combined with transcatheter arterial chemoembolisation in patients with unresectable hepatocellular carcinoma (ORIENTAL): a randomised, double-blind, placebo-controlled, multicentre, phase 3 study. Lancet Gastroenterol Hepatol. 2018;3:37–46. doi: 10.1016/S2468-1253(17)30290-X. [DOI] [PubMed] [Google Scholar]
- 87.Wu J, Ng J, Christos PJ, Goldenberg AS, Sparano J, Sung MW, Hochster HS, Muggia FM. Chronic thalidomide and chemoembolization for hepatocellular carcinoma. Oncologist. 2014;19:1229–1230. doi: 10.1634/theoncologist.2014-0283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Pinter M, Ulbrich G, Sieghart W, Kölblinger C, Reiberger T, Li S, Ferlitsch A, Müller C, Lammer J, Peck-Radosavljevic M. Hepatocellular Carcinoma: A Phase II Randomized Controlled Double-Blind Trial of Transarterial Chemoembolization in Combination with Biweekly Intravenous Administration of Bevacizumab or a Placebo. Radiology. 2015;277:903–912. doi: 10.1148/radiol.2015142140. [DOI] [PubMed] [Google Scholar]
- 89.Tong H, Wei B, Chen S, Xie YM, Zhang MG, Zhang LH, Huang ZY, Tang CW. Adjuvant celecoxib and lanreotide following transarterial chemoembolisation for unresectable hepatocellular carcinoma: a randomized pilot study. Oncotarget. 2017;8:48303–48312. doi: 10.18632/oncotarget.15684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Kawamura Y, Kobayashi M, Shindoh J, Kobayashi Y, Okubo S, Tominaga L, Kajiwara A, Kasuya K, Iritani S, Fujiyama S, Hosaka T, Saitoh S, Sezaki H, Akuta N, Suzuki F, Suzuki Y, Ikeda K, Arase Y, Hashimoto M, Kozuka T, Kumada H. Lenvatinib-Transarterial Chemoembolization Sequential Therapy as an Effective Treatment at Progression during Lenvatinib Therapy for Advanced Hepatocellular Carcinoma. Liver Cancer. 2020;9:756–770. doi: 10.1159/000510299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Hiroishi K, Eguchi J, Baba T, Shimazaki T, Ishii S, Hiraide A, Sakaki M, Doi H, Uozumi S, Omori R, Matsumura T, Yanagawa T, Ito T, Imawari M. Strong CD8(+) T-cell responses against tumor-associated antigens prolong the recurrence-free interval after tumor treatment in patients with hepatocellular carcinoma. J Gastroenterol. 2010;45:451–458. doi: 10.1007/s00535-009-0155-2. [DOI] [PubMed] [Google Scholar]
- 92.Greten TF, Duffy AG, Korangy F. Hepatocellular carcinoma from an immunologic perspective. Clin Cancer Res. 2013;19:6678–6685. doi: 10.1158/1078-0432.CCR-13-1721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Xu F, Jin T, Zhu Y, Dai C. Immune checkpoint therapy in liver cancer. J Exp Clin Cancer Res. 2018;37:110. doi: 10.1186/s13046-018-0777-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Voron T, Colussi O, Marcheteau E, Pernot S, Nizard M, Pointet AL, Latreche S, Bergaya S, Benhamouda N, Tanchot C, Stockmann C, Combe P, Berger A, Zinzindohoue F, Yagita H, Tartour E, Taieb J, Terme M. VEGF-A modulates expression of inhibitory checkpoints on CD8+ T cells in tumors. J Exp Med. 2015;212:139–148. doi: 10.1084/jem.20140559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Yoo SY, Badrinath N, Woo HY, Heo J. Oncolytic Virus-Based Immunotherapies for Hepatocellular Carcinoma. Mediators Inflamm. 2017;2017:5198798. doi: 10.1155/2017/5198798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Fagnoni FF, Zerbini A, Pelosi G, Missale G. Combination of radiofrequency ablation and immunotherapy. Front Biosci. 2008;13:369–381. doi: 10.2741/2686. [DOI] [PubMed] [Google Scholar]
- 97.Gameiro SR, Higgins JP, Dreher MR, Woods DL, Reddy G, Wood BJ, Guha C, Hodge JW. Combination therapy with local radiofrequency ablation and systemic vaccine enhances antitumor immunity and mediates local and distal tumor regression. PLoS One. 2013;8:e70417. doi: 10.1371/journal.pone.0070417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Duffy AG, Ulahannan SV, Makorova-Rusher O, Rahma O, Wedemeyer H, Pratt D, Davis JL, Hughes MS, Heller T, ElGindi M, Uppala A, Korangy F, Kleiner DE, Figg WD, Venzon D, Steinberg SM, Venkatesan AM, Krishnasamy V, Abi-Jaoudeh N, Levy E, Wood BJ, Greten TF. Tremelimumab in combination with ablation in patients with advanced hepatocellular carcinoma. J Hepatol. 2017;66:545–551. doi: 10.1016/j.jhep.2016.10.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Ma H, Zhang Y, Wang Q, Li Y, He J, Wang H, Sun J, Pan K, Chen M, Xia J. Therapeutic safety and effects of adjuvant autologous RetroNectin activated killer cell immunotherapy for patients with primary hepatocellular carcinoma after radiofrequency ablation. Cancer Biol Ther. 2010;9:903–907. doi: 10.4161/cbt.9.11.11697. [DOI] [PubMed] [Google Scholar]
- 100.Cui J, Wang N, Zhao H, Jin H, Wang G, Niu C, Terunuma H, He H, Li W. Combination of radiofrequency ablation and sequential cellular immunotherapy improves progression-free survival for patients with hepatocellular carcinoma. Int J Cancer. 2014;134:342–351. doi: 10.1002/ijc.28372. [DOI] [PubMed] [Google Scholar]
- 101.Lee JH, Lee JH, Lim YS, Yeon JE, Song TJ, Yu SJ, Gwak GY, Kim KM, Kim YJ, Lee JW, Yoon JH. Sustained efficacy of adjuvant immunotherapy with cytokine-induced killer cells for hepatocellular carcinoma: an extended 5-year follow-up. Cancer Immunol Immunother. 2019;68:23–32. doi: 10.1007/s00262-018-2247-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Lee JH, Lee JH, Lim YS, Yeon JE, Song TJ, Yu SJ, Gwak GY, Kim KM, Kim YJ, Lee JW, Yoon JH. Adjuvant immunotherapy with autologous cytokine-induced killer cells for hepatocellular carcinoma. Gastroenterology. 2015;148:1383–91.e6. doi: 10.1053/j.gastro.2015.02.055. [DOI] [PubMed] [Google Scholar]
- 103.Tu J, Ji J, Wu F, Zhang D, Ying X, Zhao Z. [Efficacies of ¹³¹I-chTNT plus radiofrequency ablation for the treatment of advanced hepatocellular carcinoma] Zhonghua Yi Xue Za Zhi. 2014;94:3586–3588. [PubMed] [Google Scholar]
- 104.Bian H, Zheng JS, Nan G, Li R, Chen C, Hu CX, Zhang Y, Sun B, Wang XL, Cui SC, Wu J, Xu J, Wei D, Zhang X, Liu H, Yang W, Ding Y, Li J, Chen ZN. Randomized trial of [131I] metuximab in treatment of hepatocellular carcinoma after percutaneous radiofrequency ablation. J Natl Cancer Inst. 2014;106 doi: 10.1093/jnci/dju239. [DOI] [PubMed] [Google Scholar]
- 105.Guan Q, Gu J, Zhang H, Ren W, Ji W, Fan Y. Correlation between vascular endothelial growth factor levels and prognosis of hepatocellular carcinoma patients receiving radiofrequency ablation. Biotechnol Biotechnol Equip. 2015;29:119–123. doi: 10.1080/13102818.2014.981776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Kong J, Kong J, Pan B, Ke S, Dong S, Li X, Zhou A, Zheng L, Sun WB. Insufficient radiofrequency ablation promotes angiogenesis of residual hepatocellular carcinoma via HIF-1α/VEGFA. PLoS One. 2012;7:e37266. doi: 10.1371/journal.pone.0037266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Tak WY, Lin SM, Wang Y, Zheng J, Vecchione A, Park SY, Chen MH, Wong S, Xu R, Peng CY, Chiou YY, Huang GT, Cai J, Abdullah BJJ, Lee JS, Lee JY, Choi JY, Gopez-Cervantes J, Sherman M, Finn RS, Omata M, O'Neal M, Makris L, Borys N, Poon R, Lencioni R. Phase III HEAT Study Adding Lyso-Thermosensitive Liposomal Doxorubicin to Radiofrequency Ablation in Patients with Unresectable Hepatocellular Carcinoma Lesions. Clin Cancer Res. 2018;24:73–83. doi: 10.1158/1078-0432.CCR-16-2433. [DOI] [PubMed] [Google Scholar]
- 108.Celik H, Wakim P, Pritchard WF, Castro M, Leonard S, Karanian JW, Dewhirst MW, Lencioni R, Wood BJ. Radiofrequency Ablation Duration per Tumor Volume May Correlate with Overall Survival in Solitary Hepatocellular Carcinoma Patients Treated with Radiofrequency Ablation Plus Lyso-Thermosensitive Liposomal Doxorubicin. J Vasc Interv Radiol. 2019;30:1908–1914. doi: 10.1016/j.jvir.2019.04.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Finn RS, Qin S, Ikeda M, Galle PR, Ducreux M, Kim TY, Kudo M, Breder V, Merle P, Kaseb AO, Li D, Verret W, Xu DZ, Hernandez S, Liu J, Huang C, Mulla S, Wang Y, Lim HY, Zhu AX, Cheng AL IMbrave150 Investigators. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N Engl J Med. 2020;382:1894–1905. doi: 10.1056/NEJMoa1915745. [DOI] [PubMed] [Google Scholar]
- 110.Kudo M, Chung H, Osaki Y. Prognostic staging system for hepatocellular carcinoma (CLIP score): its value and limitations, and a proposal for a new staging system, the Japan Integrated Staging Score (JIS score) J Gastroenterol. 2003;38:207–215. doi: 10.1007/s005350300038. [DOI] [PubMed] [Google Scholar]
- 111.Morimoto M, Numata K, Kondo M, Moriya S, Morita S, Maeda S, Tanaka K. Radiofrequency ablation combined with transarterial chemoembolization for subcapsular hepatocellular carcinoma: a prospective cohort study. Eur J Radiol. 2013;82:497–503. doi: 10.1016/j.ejrad.2012.09.014. [DOI] [PubMed] [Google Scholar]
- 112.Hyun D, Cho SK, Shin SW, Rhim H, Koh KC, Paik SW. Treatment of Small Hepatocellular Carcinoma (≤2 cm) in the Caudate Lobe with Sequential Transcatheter Arterial Chemoembolization and Radiofrequency Ablation. Cardiovasc Intervent Radiol. 2016;39:1015–1022. doi: 10.1007/s00270-016-1314-5. [DOI] [PubMed] [Google Scholar]
- 113.Hyun D, Cho SK, Shin SW, Park KB, Lee SY, Park HS, Do YS. Combined transarterial chemoembolization and radiofrequency ablation for small treatment-naïve hepatocellular carcinoma infeasible for ultrasound-guided radiofrequency ablation: long-term outcomes. Acta Radiol. 2018;59:773–781. doi: 10.1177/0284185117735349. [DOI] [PubMed] [Google Scholar]
- 114.Yan JY, Zhang JL, Wang MQ, Yuan K, Bai YH, Wang Y, Xin HN, Wang ZJ, Liu FY, Duan F, Fu JX. Combined transcatheter arterial chemoembolization and radiofrequency ablation in single-session for solitary hepatocellular carcinoma larger than 7 cm. Asia Pac J Clin Oncol. 2018;14:300–309. doi: 10.1111/ajco.12817. [DOI] [PubMed] [Google Scholar]
- 115.Kim AR, Park E, Kwon SY, Park SJ, Kim YJ, Yoo BC, Choe WH, Kim JH, Hwang JH, Park SW, Park HS, Yu MH, Jeon HJ. Efficacy and Safety of Combined Radiofrequency Ablation with Transarterial Chemoembolization in Patients with Barcelona Clinic Liver Cancer Stage A Hepatocellular Carcinoma Ineligible for Curative Treatment. Korean J Gastroenterol. 2019;73:167–176. doi: 10.4166/kjg.2019.73.3.167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Duan F, Bai YH, Cui L, Li XH, Yan JY, Wang MQ. Simultaneous transarterial chemoembolization and radiofrequency ablation for large hepatocellular carcinoma. World J Gastrointest Oncol. 2020;12:92–100. doi: 10.4251/wjgo.v12.i1.92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Zhang Y, Zhang MW, Fan XX, Mao DF, Ding QH, Zhuang LH, Lv SY. Drug-eluting beads transarterial chemoembolization sequentially combined with radiofrequency ablation in the treatment of untreated and recurrent hepatocellular carcinoma. World J Gastrointest Surg. 2020;12:355–368. doi: 10.4240/wjgs.v12.i8.355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Wang Y, Ma L, Yuan Z, Zheng J, Li W. Percutaneous thermal ablation combined with TACE vs TACE monotherapy in the treatment for liver cancer with hepatic vein tumor thrombus: A retrospective study. PLoS One. 2018;13:e0201525. doi: 10.1371/journal.pone.0201525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Song Q, Ren W, Fan L, Zhao M, Mao L, Jiang S, Zhao C, Cui Y. Long-Term Outcomes of Transarterial Chemoembolization Combined with Radiofrequency Ablation Versus Transarterial Chemoembolization Alone for Recurrent Hepatocellular Carcinoma After Surgical Resection. Dig Dis Sci. 2020;65:1266–1275. doi: 10.1007/s10620-019-05733-0. [DOI] [PubMed] [Google Scholar]
- 120.Thornton LM, Cabrera R, Kapp M, Lazarowicz M, Vogel JD, Toskich BB. Radiofrequency vs Microwave Ablation After Neoadjuvant Transarterial Bland and Drug-Eluting Microsphere Chembolization for the Treatment of Hepatocellular Carcinoma. Curr Probl Diagn Radiol. 2017;46:402–409. doi: 10.1067/j.cpradiol.2017.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Ni JY, Fang ZT, An C, Sun HL, Huang ZM, Zhang TQ, Jiang XY, Chen YT, Xu LF, Huang JH. Comparison of albumin-bilirubin grade, platelet-albumin-bilirubin grade and Child-Turcotte-Pugh class for prediction of survival in patients with large hepatocellular carcinoma after transarterial chemoembolization combined with microwave ablation. Int J Hyperthermia. 2019;36:841–853. doi: 10.1080/02656736.2019.1646927. [DOI] [PubMed] [Google Scholar]