Abstract
Purpose
Radiotherapy-induced sensorineural hearing loss (RISNHL) is a common adverse effect in patients with head and neck cancer. Given that there are few studies on the pathogenesis of RISNHL at present, we summarized the possible pathogenesis of RISNHL and possible protective measures found at present by referring to relevant literatures.
Methods
We performed a comprehensive literature search in the PubMed database, using keywords “sensorineural hearing loss,” “radiotherapy,” and “cancer,” among others. The literature was examined for the possible mechanism and preventive measures of sensorineural hearing loss induced by radiotherapy.
Results
We found that the incidence of RISNHL was closely related to the damage directly caused by ionizing radiation and the radiation-induced bystander effect. It also depends on the dose of radiation and the timing of chemotherapy. Studies confirmed that RISNHL is mainly involved in post-RT inflammatory response and changes in reactive oxygen species, mitogen-activated protein kinase, and p53 signaling pathways, leading to specific manners of cell death. We expect to reduce the incidence of hearing loss through advanced radiotherapy techniques, dose limitation of organs at risk, application of cell signaling inhibitors, use of antioxidants, induction of cochlear hair cell regeneration, and cochlear implantation.
Conclusion
RISNHL is associated with radiation damage to DNA, oxidative stress, and inflammation of cochlear cells, stria vascularis endothelial cells, vascular endothelial cells, spiral ganglion neurons, and other supporting cells. At present, the occurrence mechanism of RISNHL has not been clearly illustrated, and further studies are needed to better understand the underlying mechanism, which is crucial to promote the formulation of better strategies and prevent the occurrence of RISNHL.
1. Introduction
Radiotherapy (RT) is commonly used and sometimes the first choice in treating head and neck tumors, especially nasopharyngeal carcinoma (NPC). Unfortunately, the complex anatomy and location of the tumor often lead to the exposure of the auditory pathway to ionizing radiation, which may result in conductive hearing loss (CHL) and sensorineural hearing loss (SNHL). SNHL is a tardive and irreversible complication that can be observed in patients with inner ear exposure to radiation fields. The incidence of SNHL varies widely, because of various reasons such as radiation dose, age, and hearing sensitivity of patients, ranging from 0 to 85% for low-frequency (<4 kHz) and from 27% to 95% for high-frequency (>4 kHz) SNHL. The affected hearing range usually starts with high frequencies and progresses toward lower frequencies [1].
Despite the increased interest in RT-induced ototoxicity in tumors of the head and neck tumors, there is little research on the mechanism and protection of radiotherapy-induced SNHL (RISNHL). This article is aimed at describing the progress made to understand the mechanism of RISNHL and the approaches developed so far for its protection or to reduce the associated complications.
2. Cochlear Structure and Function
The impairment of cochlear structure has been shown to be associated with SNHL. A healthy neonatal baby's cochlea contains ~3500 inner hair cells (IHC) and 12,000 outer hair cells (OHC) (Figure 1) [2]. OHC comprises the basal and apical OHC that respond to higher and lower frequency sounds, respectively. Cochlear hair cells interlock with supporting cells to form an epithelial layer on top of the basilar membrane. Sound energy causes membrane motion and triggers K+ influx and hair cell (HC) receptor potential. Depolarization of OHCs forms the basis for cochlear sensitivity and frequency resolution, while depolarization of IHCs releases glutamate from ribbon synapses at their basal poles. Spiral ganglion neurons are bipolar neurons. When stimulated by IHCs, the bipolar spiral ganglion neurons innervating the IHCs transmit auditory information to the auditory center through extended synapses [3]. Cochlear hair cells (HCs) and spiral ganglion neurons constitute the basis of hearing function. These cells can hardly regenerate after death in mammals, making it increasingly important to protect these tissues during chemoradiotherapy.
Figure 1.
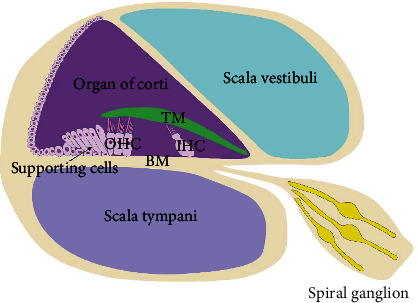
Cochlea structure diagram. BM: basement membrane; TM: tectorial membrane; IHC: inner hair cells; OHC: outer hair cells.
3. Mechanism of RISNHL in Head and Neck Cancer
According to precious studies, ionizing radiation- (IR-) induced direct cell damage and radiation-induced bystander effect (RIBE) are responsible for hearing loss. Radiation exerts multiple effects by disrupting chemical bonds in all the basic components of the cells. These damages classified as direct and indirect damage to the deoxyribonucleic acid (DNA) could lead to multiple manners of cell fate decision, including apoptosis, autophagy, necrosis, and mitotic catastrophe [4]. The cell fate decision mainly depends on the type of cell and the severity of cell damage [5]. Furthermore, we describe the possible mechanism of cell damage by IR at the cellular and molecular levels (Figure 2).
Figure 2.
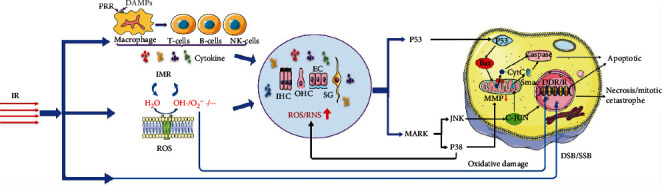
Mechanisms of cell damage induced by ionizing radiation (IR). Radiation can either damage DNA directly or cause oxidative damage to DNA through oxygen free radicals produced by ionizing water molecules, leading to cell death. Damage-associated molecular pattern molecules (DAMPs) will be released after cell damage or death, which activates macrophages and other antigen-presenting cells and enhances inflammatory and immune responses (IMR). Moreover, IMR and reactive oxygen species (ROS) can interact to change the cochlea microenvironment and induce cell death through p53 and mitogen-activated protein kinase (MAPK) signaling pathways. DDR/R: DNA damage response and repair reactions pathway; EC: endothelial cells; IHC: inner hair cells; OHC: outer hair cells; SG: spiral ganglion; MMP: mitochondrial membrane potential; DSB: double-strand breaks; SSB: single-strand breaks.
3.1. Direct Effects of Radiation Induction
3.1.1. Radiation-Induced DNA Damage
Damage is caused by IR primarily through local high-energy deposition in DNA [6] which causes single-strand breaks (SSB) and double-strand breaks (DSB) in DNA. After that, damages could be repaired [7]. DNA damage response and repair reaction pathways (DDR/R) mainly include single-strand break repair (SSBR), base excision repair (BER), and nucleotide excision repair (NER), which can initiate senescence or apoptosis when necessary. However, previous studies have confirmed that direct damage accounts for only about one-third of the biological effects of radiation. Most of the damage caused by radiation is mediated by free radicals generated during radiation exposure (which is classified as indirect damage), because most cells in the body are rich in water molecules [8, 9]. Increased ROS and NO after radiation have been proved to play a key role in the genotoxic effects of ionizing radiation [10].
3.1.2. Radiation-Induced Formation of Reactive Oxygen Species and Signaling Pathway Activation
IR-induced DNA damage is mainly mediated by free radicals produced by the interaction between radiation and water molecules, which is classified as indirect damage [11]. Moreover, a consensus has been reached that reactive oxygen species (ROS) and reactive nitrogen species (RNS) in the cochlea often trigger caspase-mediated cell death after exposure to loud sound and ototoxic drugs [12, 13]. The excessive production of ROS and RNS induced by IR destroys the balance in the body and leads to oxidative stress. Oxidative stress can aggravate the inflammatory response, which in turn induces the production of ROS/RNS [14]. Oxidative active products such as ROS/RNS/nitric oxide (NO) can cause oxidative damage to DNA, and typical DNA oxidative damage leads to SSB and DSB [15, 16]. Meanwhile, a high level of NO/ROS leads to mitochondrial membrane leakage and causes functional defects. There is increasing evidence that oxidative stress products produced postradiation lead to the release of cytochrome C, decrease of mitochondrial membrane potential(MMP), and activation of p53, C-Jun N-terminal kinase (JNK), and mitogen-activated protein kinase (MAPK) in auditory cells [17–19]. At present, many studies have found that ROS can also lead to membrane lipid peroxidation, and the products of lipid peroxidation induced cell apoptosis through a variety of signaling pathways [20]. Overall, it can be inferred that ROS/RNS can lead to cell damage or death through various pathways.
MAPKs are intracellular proteins present in all eukaryotic cells and respond to extracellular and intracellular stimuli after phosphorylation. MAPKs intercept plasma membrane-bound receptor signals to activate transcription factors in the nucleus, collaborating with gene expression and facilitating the regulation of cell proliferation, differentiation, motility, and survival [21]. The JNK, also known as a pressure-activated protein kinase, is an important member of the MAPK family [22]. Furthermore, it has been shown that JNK is a major contributor to oxidative stress damage in mammalian inner ear cells after trauma. Inactive JNK located in the cytoplasm is activated upon exposure to environmental pressure and transported to the nucleus, consequently activating several transcription factors [23]. These transcription factors can regulate the cell stress response according to different influencing factors and lead to different outcomes (cell survival or death). Another way of JNK regulation is via its transportation to the mitochondria, which results in the release of the second mitochondria-derived activator of caspase (SMAC). SMACs can promote cell apoptosis by activating caspases [24]. The other important key mediator in the MAPK signaling pathway is the p38 family. Apoptosis mediated by p38 in HEI-OC1 cells was observed in a previous study [25]. The pharmacological inhibition of p38 before radiation exposure protects auditory cells in vitro from radiation by moderating changes in mitochondrial membrane potential (MMP) and generation of NO. Furthermore, p38 inhibitors protect neuroblasts from radiation-induced damage in vivo. These findings suggest that inhibition of p38 may be a plausible strategy for protecting the mitochondria from radiation.
The activation of p53 is known to induce cell cycle arrest, facilitating the conception of the apoptotic pathway. The proapoptotic member Bax is transcriptionally upregulated after p53 phosphorylation [12], prompting pore formation in the mitochondrial membrane. Activated nuclear p53 transports to the mitochondria, where it interacts with and inactivates prosurvival Bcl-2 proteins, simultaneously damaging the HCs directly [26]. Studies showed that phosphorylation of p53 protein increased dramatically at 3 h postirradiation. Several p53-regulated genes associated with cell cycle regulation and arrest were also found to increase, corresponding to the phosphorylation of p53 [27]. These findings suggest that the inhibition of p38/p53 activation could be a potential therapeutic strategy.
3.1.3. Inflammatory Cell Recruitment
Radiation can induce inflammatory and immune response (IMR) and cause DNA damage [28]. Damaged cells release a variety of damage-associated molecular patterns (DAMPs), which activate antigen-presenting cells such as macrophages through pattern recognition receptor (PRR), causing IMR [29]. Several studies investigating the changes in immunomodulatory parameters after radiation have shown that these parameters maintain a chronic inflammatory state leading to radiation-related advanced pathology [28]. Furthermore, the continuous inflammatory process has been demonstrated as the principal cause of late IR effects [30]. After IR, the cells induce macrophages to produce various cytokines, such as IL-1, IL-6, IL-8, and TNF-α, causing cell damages [31]. Earlier studies have also reported an influx of inflammatory cells in the cochlea after acoustic trauma [32, 33]. Many studies have found that the stria vascularis plays a crucial role in the inflammatory process in the stria area by recruiting macrophages [34–36]. The role of this process in radiation-induced inflammatory response needs to be confirmed by future experiments. IR-induced IMR can alter the microenvironment of cochlear cells, leading to the occurrence of radiation-induced fibrosis [37, 38]. This could be one of the pathogeneses of RISNHL.
3.1.4. Radiation-Induced Bystander Effect (RIBE)
RIBE has been extensively studied in recent years [6, 39, 40]. It was first proposed by Mole in the 1950s [41] and has been demonstrated in many studies, such as on breast cancer cells [42], human umbilical vein endothelial cells (HUVECs) [43], and lung cancer cell lines [44]. RIBE mainly includes DNA damage, malignant transformation, and cell death in bystander cells. The bystander cells are not exposed to radiation directly, but the asymmetry of DNA damage repair between the observer cells and the irradiated cells activates DDR/R of the observer cells and even induces their death.
Oxidative stress also plays an important role in the RIBE. The main source of ROS causing bystander cell damage is the decomposition of irradiated cells [45]. At the same time, NADPH oxidase, bound to the plasma membrane of the observing cell, continuously produces ROS. Some studies have also shown that cytokines can stimulate COX-2 in bystander cells to release large amounts of ROS/NO during the production of prostaglandin E2, which promotes the transmission of RIBE [46]. RNS/NO is also produced by irradiated cells and are transmitted to bystander cells. The production of ROS/RNS/NO could lead to damage or death of bystander cells through a variety of pathways, similar to directly irradiated cells (Figure 3).
Figure 3.
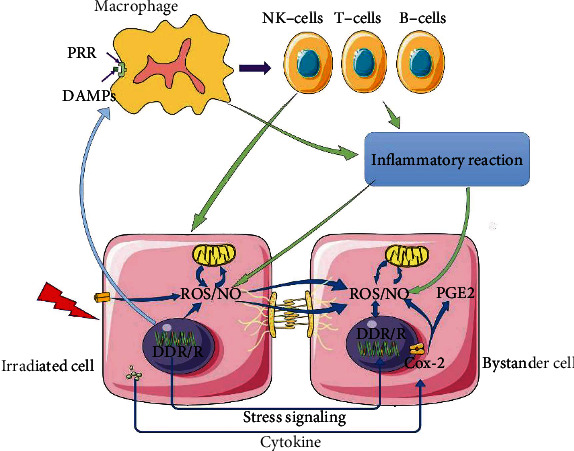
Radiation-induced bystander effect (RIBE). Ionizing radiation can directly cause DNA damage and activate the DNA damage response and repair reactions pathway (DDR/R), while the latter activates macrophages through the release of damage-associated molecular pattern molecules (DAMPs), thereby further enhancing the inflammatory and immune response. The reactive oxygen species (ROS)/nitric oxide (NO) and various cytokines produced by the damaged cells eventually crosstalk with the bystander cells through various paths, causing damage to the cells. PGE2: prostaglandin E2; PRR: pattern recognition receptor.
These results indicated that in the process of head and neck cancer radiotherapy, the cochlea cells could be damaged even without direct irradiation. Meanwhile, controlling the bystander effect with systemic/peripheral protection, rather than targeting the cochlea, may also reduce radiation damage. These all need to be explored.
3.2. Cell Death
Asenov et al. performed a histopathological examination of the temporal bone in patients with nasopharyngeal carcinoma (NPC) receiving chemotherapy and RT and revealed that denaturation of the stria vascularis, atrophy of the spiral ligament, reduction of spiral ganglion cells, and occasional loss of hair cells were the main changes in the cochlea [47].
3.2.1. Cochlear HC Death
The loss of cochlear hair cells is primarily responsible for RISNHL [48]. The cochlea is reported sensitive to radiation [49]. Many studies have proved that free radical accumulation is an early event in HCs injury process [50, 51]. ROS have been found to originate from a wide range of sources, including direct radiation and bystander effects. For instance, an in vitro study showed that the fluorescence intensity of an oxidation-sensitive probe increased in a dose-dependent manner 1 h after the HC line OC-K3 was exposed to different doses of radiation (2/20/100 Gy) [27]. Furthermore, Sha et al. have speculated that the basal OHCs (respond to higher frequency sounds) could be more susceptible to free radical damage than the apical (respond to lower frequency sounds) OHCs due to a significantly lower level of the antioxidant-glutathione in the basal OHCs than the apical OHCs [52]. These results partially illustrated the underlying mechanism that RISNHL affects the high frequencies of the hearing range and progresses toward lower frequencies, which are consistent with clinical observations. And another study found that the probability of low-frequency SNHL after intensity-modulated radiation therapy (IMRT) treatment of NPC was 6–22%, and the probability of high-frequency SNHL was 37–51.2% [53]. It also indicates that the order of cell death caused by different sensitivity to ROS affects the degree of hearing loss.
What role do classic cellular signaling pathways play in this process? Previous studies have observed abnormal activation of JNK and P53 signaling pathways during the apoptosis of OHCs [54, 55]. The activation of the inducible transcription factor c-Jun is the core event in JNK-mediated apoptosis of oxidative stress-damaged auditory HCs [56]. However, several contradictory studies have also been reported questioning the role of JNK in hair cell apoptosis. For instance, a recent study showed that apoptosis occurred in a dose-dependent manner, mainly at 72 h postirradiation, whereas the early activation of c-JUN began at 3 h and then decreased at 24 h postirradiation, suggesting that the c-JUN pathway may not lead to radiation-induced apoptosis [27]. Therefore, further research is needed to elucidate the relationship between JNK, P53, and HC death after radiation.
3.2.2. Stria Vascularis Endothelial Cell Death and Vascular Endothelial Cell Death
The stria vascularis is a specially stratified epithelial tissue of the outer wall of the cochlear duct with inner capillaries and is damaged after radiation exposure. These damages are not only caused by the direct effect of IR but also by various cytokines released by the irradiated cells. It has been proved that after irradiation, vascular endothelial cells in the stria vascularis was activated to produce aseptic inflammation [57, 58] which leads to thrombosis [59], endothelial cell contraction, and cell death. Additionally, Hellweg showed that radiation-induced genotoxicity is the main cause of aseptic inflammation, which activates the nuclear factor (NF)-κB pathway [60]. Studies have also shown that the damage of the stria vascularis endothelial cells after high-dose irradiation may be related to the sphingolipid ceramide pathway [61], which is usually activated in epithelial cells after radiation-induced DNA damage. Reportedly, stria vascularis endothelial cell damage mainly affects the short- and medium-term hearing of patients after RT, especially in patients receiving single-fraction high-dose RT [14], whereas as evident from the histological results, stria vascularis cells were rarely damaged in fractionated RT. [62]
3.2.3. Spiral Ganglion Neurons
It is well known that neurons are not capable of mitosis. However, owing to the damage of connective tissue cells (oligodendrocytes, astrocytes) involved in myelination and other supporting functions, nerve tissue is radiosensitive as a whole [63].
Though spiral ganglion cells were greatly affected by radiation during fractional RT [62], their function impairments after RT are controversial. In a study of NPC patients who underwent radiotherapy alone, no significant difference was found in evoked response audiometry and pure-tone threshold audiometry(PTA) before and after radiotherapy [64].
However, another study showed that after RT, patients' PTA increased significantly. The latency of auditory brainstem response (ABR) waves was prolonged or even disappeared at 1 year after RT, the amplitude of I-IV waves was reduced, and the latency between the peaks of I-V waves was significantly prolonged [65]. The experimental results of Lau et al. are also consistent with the above experiments [66]. Previous studies have revealed that the first wave (I) of ABR reflects the activity of the spiral ganglion cells [67]. The sign of neuroinflammation is the occurrence of radiation-induced fibrosis. Its pathological process includes axonal injury and demyelination, as well as the proliferation of fibrotic tissue around the nerve trunk and the decrease of vascular supply [37]. Krysta et al. found that the density of spiral ganglion cell bodies and surrounding protrusions exposed to 60 Gy IR was significantly decreased in mice, and the related extracellular matrix was also lost. In the 20 Gy IR group, only a slight threshold shift, with pathological examination revealing signs of vacuolation and separation of spiral ganglion cells, and early demyelination [68]. While demyelination is also found responsible for the hearing loss caused by excessive sound stimulation [69] and aging [70]. These findings indicate that inhibition of the demyelination of spiral ganglion neurons can help alleviate hearing loss.
4. Protection Measures
Radiation-related severe late complications, such as hearing loss, negatively affect the quality of life. Therefore, the prevention and treatment of SNHL have also attracted attention. Different strategies have been tested at the clinical and preclinical levels to reduce the incidence of RT induced hearing impairment. From hearing loss to cell death-induced deafness, we can reduce the occurrence of sensorineural hearing loss from many aspects. These include improved radiotherapy technology, limitation of normal tissue dose, application of signaling inhibitors, use of antioxidants, induction of cochlear hair cell regeneration, and cochlear implant.
4.1. Improvement of Radiotherapy and Chemotherapy Regiments
The degree of hearing loss increases with the dose of the inner ear [71, 72]. The most important measure to prevent RISNHL is to reduce the radiation dose in the cochlea [73]. A recent study found that the effect of fractionated stereotactic radiotherapy (fSRT) on the total dose of cochlea may be less than that of stereotactic radiosurgery (SRS) [74]. Studies have also shown that NPC patients have an increased risk of developing high-frequency SNHL when the radiation dose to internal auditory canal (IAC) was IAC − Dmax > 42.13 Gy or IAC − Dmean > 32.71 Gy [75]. Hence, reducing the cochlear dose seems to be critical to reducing the incidence of SNHL [73]. Furthermore, the dose distribution of organs at risk (OARs) is related to the T stage, especially tumor volume (GTV) [76, 77]. It has been demonstrated that the cochlear radiation dose can be reduced depending on different T stages, using the technique of dose-limiting stratification scheme, for example, by setting the limit dose threshold of 45 Gy or lower for T1 and T2 stages or by slightly adjusting the angle of the specific beam radiated to the target [78]. A comparative study of Smartarc-based volumetric-modulated arc therapy (VMAT-S) and stepped field intensity-modulated radiotherapy (IMRT) treatment of locally advanced NPC found that the average cochlear dose of the VMAT-S regimen was lower than that of the IMRT regimen [79].
Previous studies have found that the incidence of SNHL increases when chemoradiotherapy (CRT) is used. One of the studies found that the incidence of SNHL was 84% in CRT group, but only 26% in RT group [80, 81]. But for many advanced tumors, we usually need a combination of radiotherapy and chemotherapy. Many clinical studies have proved the superiority of CRT [82]. Platinum-based chemotherapy regimens have been shown to have definite ototoxic side effects. The molecular mechanisms of cisplatin-induced ototoxicity include the imbalance of endogenous oxidation system and antioxidant system, cochlear inflammatory response, and abnormal activation or inactivation of P53, HSP, CDK2, etc. [83]. A comparative study indicated a higher incidence of ototoxicity in patients receiving cisplatin after RT than in patients receiving chemotherapy before RT. [84] It may be associated with increased exposure to cisplatin in the Organ of Corti or auditory nerve after RT. It has been shown that radiation induces increased vesicle transport, phosphatase activity, and endothelial cell tight junction opening, which increase the permeability of the blood–brain barrier in a dose-dependent manner. The increased permeability increases the exposure of the Organ of Corti or auditory nerve to cisplatin. This explains the increased incidence of SNHL after concurrent cisplatin administration or RT. [85] In the application of platinum-based chemotherapy, drugs that reduce ototoxicity can also be used prophylactically, including drugs targeting endogenous antioxidant system (allopurinol, Ebselen, NOX3/RNS inhibitor, curcumin, ferulic acid), drugs targeting cochlear inflammation (TNF-α neutralizer, capsaicin, EGCG), and drugs targeting p53/HSP/CDK2. Drug-coated nanoparticles can also be used to increase the blood drug concentration in the cochlea, so as to reduce the ototoxicity of chemotherapeutic drugs [83, 86].
4.2. Signal Pathway Inhibitors
Cell damage and death are the result of cell signaling pathway transduction. It may be possible to save irradiated cells by inhibiting part of the signaling pathway. Previous studies have investigated the role of JNK inhibitors and P38 inhibitors in hearing protection. AM-111, a JNK inhibitor developed by Auris Medical, has been shown to protect hearing in animals and is currently undergoing clinical trials to study the treatment of sudden deafness [87]. Considering that RISNHL is also involved in JNK signaling pathway, it is expected to be used in RISNHL. P38 mitogen-activated protein kinase inhibitors such as SB203580 have been shown to prevent RISNHL in both cell and animal studies [25].
4.3. Antioxidant Drugs
The occurrence of RISNHL is closely related to oxidative stress. Many antioxidant drugs have been studied to treat various types of hearing loss. Studies have shown that mitochondrial targeted antioxidants are far superior to nontargeted cellular antioxidants in reducing mitochondrial oxidative damage [88]. Nontargeted antioxidants have been shown to protect ototoxicity in many cell experiments and animal experiments. This paper introduces the application of several antioxidants in hearing loss (Table 1), such as melatonin, amifostine, L-carnitine, methylprednisolone, piracetam, and epicatechin [89–94]. These drugs have not yet been tested in clinical trials to prove their feasibility in humans, and further clinical studies are expected. The concentration of traditional antioxidants in mitochondria is very low. Mitochondrial-targeted antioxidants discovered in recent years can accumulate in mitochondria and effectively inhibit oxidative stress response. MitoQ and SkQR1 are two kinds of mitochondrial targeted oxidants, which have been proved to have protective effects against ototoxicity in many studies [95, 96].
Table 1.
Studies on the protective effect of antioxidants on various SNHL.
| Drug | Experimental design | Outcome | Mechanism | Reference |
|---|---|---|---|---|
| MT | Animal experiment: rats | DPOAE: M+RT group and RT+M group>RT group | Inhibit production of ROS7; enhance the DNA repair process | [89] |
| AMF | Animal experiment: guinea pigs | Degree of cochlear hair cell damage: IRR + AMF group < RT group. No difference between 100 mg/kg and 200 mg/kg AMF groups |
Be hydrolyzed into active component in vivo, which has sulfhydryl group that scavenges free radicals | [90] |
| LC | Animal experiment: guinea pigs | Histopathological examination: LC can ameliorate radiation-induced cochlear damage in guinea pigs. | Improving mitochondrial oxygen utilization and scavenging free-radicals | [91] |
| MP | Clinical trials: NPC patients | Pure tone audiometry; DPOAE; ABR: the use of MP during RT can reduce the early RISNHL | Mechanisms to protect the OHC: 1. Affects transcription and inhibits caspase-mediated apoptosis 2. Inhibits inflammatory response 3. Promotes GSH synthesis |
[92] |
| PIR | Animal experiment: male albino guinea pigs | Histopathologic examination: PIR might reduce radiation-induced cochlear damage in guinea pigs | Increases oxygenation in the tumor cells with its rheological effects and decreases apoptosis in surrounding healthy cells | [93] |
| EC | Cell experiment: HEI-OC1 and UB-OC1 Animal experiment: embryos/rats |
EC can increase the survival rate of HEI-OC1 cells after radiotherapy. EC can inhibit the ABR threshold in the rats. |
Inhibits ROS production and MAPK activity | [94] |
MT: melatonin; AMF: amifostine; LC: L-carnitine; MP: methylprednisolone; PIR: piracetam; EC: epicatechin; GSH: glutathione.
4.4. Cochlea HC Regeneration
Cochlear HCs rarely regenerate. However, in recent years, it has been proposed that HCs can be regenerated by Atoh1 gene transfer. In this study, adenovirus containing Atoh1 gene was injected into the cochlea of deaf guinea pigs. Eight weeks after injection, different degrees of hair cell regeneration were observed, and hearing was significantly improved. “CGF166”—an adenovirus vector encoding the human unregulated transcription factor “Hath1”—is currently in clinical trials [87]. If proven safe and effective, this could be a major milestone in hearing loss treatment.
4.5. Cochlear Implants
Cochlear implants allow sound waves to bypass damaged HCs and travel to the brain. Hybrid cochlear implants are designed for adults with high-frequency SNHL [97]. Cochlear implantation is undoubtedly a good method for patients with SNHL whose HCs are damaged but the afferent nerve pathway is not damaged. A case-control comparative study indicated that RISNHL was mainly due to the damage of the cochlea structure, and the function of the auditory nerve was basically preserved [98]. However, there is a contrary suggestion that there is damage to the retrocochlear auditory pathway following radiotherapy, including demyelination and neuroinflammation as mentioned above [65]. More complete clinical studies are needed to verify whether the retrocochlear auditory pathway is damaged after radiotherapy.
5. Conclusions
Currently, the possible mechanisms of RISNHL can be simplified as cells exposed to radiation lead to cell death through direct or indirect DNA damage, inflammatory cell recruitment, ROS-mediated oxidative stress response, and activation of multiple signaling pathways (such as MAPK and P53). Cells that are not exposed to radiation die in various forms as a result of a series of oxidative stress and inflammatory responses released or triggered by damaged cells. Meanwhile, ROS generated by ionizing H2O do harm to DNA or mitochondrial membrane through oxidative stress reaction and inflammatory reaction. Sensorineural hearing loss is closely related to the damage of hair cells, stria vascularis, vascular endothelial cells, and spiral ganglion. Histopathological examination of patients after radiotherapy and chemotherapy showed loss of hair cells, striatal degeneration, atrophy of spiral ligaments, and reduction of spiral ganglion cells. The selection of radiation dose and timing of combined chemotherapy are key factors for the occurrence of RISNHL.
The most important way to prevent RISNHL is to reduce radiation damage to the hearing system through dose stratification with precise radiotherapy techniques. In view of various molecular mechanisms of cell death, signal pathway inhibitors, antioxidants, hair cell regeneration, and cochlear implant can be used to prevent or alleviate hearing loss after radiation. Various protective measures known to date are the result of in vitro evaluations or animal studies. Further clinical trials are needed to confirm their protective effect.
Acknowledgments
This work was supported by the Research Funding Program of Norman Bethune Biomedical Engineering Center (grant no. BQEGCZX2019025) and Science and Technology Department of Jilin Province Key Research and Development Projects (grant no. 20200404148YY).
Data Availability
All data, models, and code generated or used during the study appear in the submitted article.
Ethical Approval
The study was done with appropriate approval of the Institutional Ethical Committee and have been performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki.
Consent
Consent in not necessary.
Conflicts of Interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Authors' Contributions
All authors contributed to the study conception and design. Analysis was performed by all authors. The first draft of the manuscript was written by Wenxia Shi, Xue Hou, and Xueying Bao and revised by all co-authors. All authors read and approved the final manuscript. Wenxia Shi, Xue Hou, and Xueying Bao contributed equally to this work.
References
- 1.Mujica-Mota M. M., Waissbluth S., Daniel S. J. Characteristics of radiation-induced sensorineural hearing loss in head and neck cancer: a systematic review. HEAD and NECK. . 2013;35(11):1662–1668. doi: 10.1002/hed.23201. [DOI] [PubMed] [Google Scholar]
- 2.Wong A. C., Ryan A. F. Mechanisms of sensorineural cell damage, death and survival in the cochlea. Frontiers in Aging Neuroscience . 2015;7:p. 58. doi: 10.3389/fnagi.2015.00058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Schettino A. E., Lauer A. M. The efficiency of design-based stereology in estimating spiral ganglion populations in mice. Hearing Research . 2013;304:153–158. doi: 10.1016/j.heares.2013.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Selzer E., Hebar A. Basic principles of molecular effects of irradiation. Wiener Medizinische Wochenschrift . 2012;162(3-4):47–54. doi: 10.1007/s10354-012-0052-9. [DOI] [PubMed] [Google Scholar]
- 5.Offer H., Erez N., Zurer I., et al. The onset of p53-dependent DNA repair or apoptosis is determined by the level of accumulated damaged DNA. Carcinogenesis . 2002;23(6):1025–1032. doi: 10.1093/carcin/23.6.1025. [DOI] [PubMed] [Google Scholar]
- 6.Klammer H., Mladenov E., Li F., Iliakis G. Bystander effects as manifestation of intercellular communication of DNA damage and of the cellular oxidative status. Cancer Letters . 2015;356(1):58–71. doi: 10.1016/j.canlet.2013.12.017. [DOI] [PubMed] [Google Scholar]
- 7.Ciccia A., Elledge S. J. The DNA damage response: making it safe to play with knives. Molecular Cell . 2010;40(2):179–204. doi: 10.1016/j.molcel.2010.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yan S., Brown S. L., Kolozsvary A., Freytag S. O., Lu M., Kim J. H. Mitigation of radiation-induced skin injury by AAV2-mediated MnSOD gene therapy. The Journal of Gene Medicine . 2008;10(9):1012–1018. doi: 10.1002/jgm.1226. [DOI] [PubMed] [Google Scholar]
- 9.Farhood B., Ashrafizadeh M., khodamoradi E., et al. Targeting of cellular redox metabolism for mitigation of radiation injury. Life Sciences . 2020;250:p. 117570. doi: 10.1016/j.lfs.2020.117570. [DOI] [PubMed] [Google Scholar]
- 10.Farhood B., Mortezaee K., Motevaseli E., et al. Selenium as an adjuvant for modification of radiation response. Journal of Cellular Biochemistry . 2019;120(11):18559–18571. doi: 10.1002/jcb.29171. [DOI] [PubMed] [Google Scholar]
- 11.Mikkelsen R. B., Wardman P. Biological chemistry of reactive oxygen and nitrogen and radiation-induced signal transduction mechanisms. Oncogene . 2003;22(37):5734–5754. doi: 10.1038/sj.onc.1206663. [DOI] [PubMed] [Google Scholar]
- 12.Hu B. H., Henderson D., Nicotera T. M. Extremely rapid induction of outer hair cell apoptosis in the chinchilla cochlea following exposure to impulse noise. Hearing Research . 2006;211(1-2):16–25. doi: 10.1016/j.heares.2005.08.006. [DOI] [PubMed] [Google Scholar]
- 13.Henderson D., Bielefeld E. C., Harris K. C., Hu B. H. The role of oxidative stress in noise-induced hearing loss. Ear and Hearing . 2006;27(1):1–19. doi: 10.1097/01.aud.0000191942.36672.f3. [DOI] [PubMed] [Google Scholar]
- 14.Mujica-Mota M. A., Lehnert S., Devic S., Gasbarrino K., Daniel S. J. Mechanisms of radiation-induced sensorineural hearing loss and radioprotection. Hearing Research . 2014;312:60–68. doi: 10.1016/j.heares.2014.03.003. [DOI] [PubMed] [Google Scholar]
- 15.Havaki S., Kotsinas A., Chronopoulos E., Kletsas D., Georgakilas A., Gorgoulis V. G. The role of oxidative DNA damage in radiation induced bystander effect. Cancer Letters . 2015;356(1):43–51. doi: 10.1016/j.canlet.2014.01.023. [DOI] [PubMed] [Google Scholar]
- 16.Kryston T. B., Georgiev A. B., Pissis P., Georgakilas A. G. Role of oxidative stress and DNA damage in human carcinogenesis. Mutation Research . 2011;711(1-2):193–201. doi: 10.1016/j.mrfmmm.2010.12.016. [DOI] [PubMed] [Google Scholar]
- 17.Eriksson D., Stigbrand T. Radiation-induced cell death mechanisms. Tumour Biology . 2010;31(4):363–372. doi: 10.1007/s13277-010-0042-8. [DOI] [PubMed] [Google Scholar]
- 18.Tabuchi K., Nishimura B., Nakamagoe M., Hayashi K., Nakayama M., Hara A. Ototoxicity: mechanisms of cochlear impairment and its prevention. Current Medicinal Chemistry . 2011;18(31):4866–4871. doi: 10.2174/092986711797535254. [DOI] [PubMed] [Google Scholar]
- 19.Baker K., Staecker H. Low dose oxidative stress induces mitochondrial damage in hair cells. Anatomical Record . 2012;295(11):1868–1876. doi: 10.1002/ar.22594. [DOI] [PubMed] [Google Scholar]
- 20.Su L. J., Zhang J. H., Gomez H., et al. Reactive oxygen species-induced lipid peroxidation in apoptosis, autophagy, and ferroptosis. Oxidative Medicine and Cellular Longevity . 2019;2019:13. doi: 10.1155/2019/5080843.5080843 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Johnson G. L. Defining MAPK interactomes. ACS Chemical Biology . 2011;6(1):18–20. doi: 10.1021/cb100384z. [DOI] [PubMed] [Google Scholar]
- 22.Murai N., Kirkegaard M., Järlebark L., Risling M., Suneson A., Ulfendahl M. Activation of JNK in the inner ear following impulse noise exposure. Journal of Neurotrauma . 2008;25(1):72–77. doi: 10.1089/neu.2007.0346. [DOI] [PubMed] [Google Scholar]
- 23.Su B., Karin M. Mitogen-activated protein kinase cascades and regulation of gene expression. Current Opinion in Immunology . 1996;8(3):402–411. doi: 10.1016/S0952-7915(96)80131-2. [DOI] [PubMed] [Google Scholar]
- 24.Chauhan D., Li G., Hideshima T., et al. JNK-dependent Release of Mitochondrial Protein, Smac, during Apoptosis in Multiple Myeloma (MM) Cells. The Journal of Biological Chemistry . 2003;278(20):17593–17596. doi: 10.1074/jbc.C300076200. [DOI] [PubMed] [Google Scholar]
- 25.Shin Y. S., Hwang H. S., Kang S. U., Chang J. W., Oh Y. T., Kim C. H. Inhibition of p38 mitogen-activated protein kinase ameliorates radiation- induced ototoxicity in zebrafish and cochlea-derived cell lines. Neurotoxicology . 2014;40:111–122. doi: 10.1016/j.neuro.2013.12.006. [DOI] [PubMed] [Google Scholar]
- 26.Marchenko N. D., Zaika A., Moll U. M. Death Signal-induced Localization of p53 Protein to Mitochondria: The Journal of Biological Chemistry . 2000;275(21):16202–16212. doi: 10.1074/jbc.275.21.16202. [DOI] [PubMed] [Google Scholar]
- 27.Low W.-K., Tan M. G. K., Sun L., Chua A. W. C., Goh L. K., Wang D. Y. Dose-dependant radiation-induced apoptosis in a cochlear cell-line. Apoptosis . 2006;11(12):2127–2136. doi: 10.1007/s10495-006-0285-4. [DOI] [PubMed] [Google Scholar]
- 28.Lumniczky K., Impens N., Armengol G., et al. Low dose ionizing radiation effects on the immune system. Environment International . 2021;149:p. 106212. doi: 10.1016/j.envint.2020.106212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pateras I. S., Havaki S., Nikitopoulou X., et al. The DNA damage response and immune signaling alliance: is it good or bad? Nature decides when and where. Pharmacology & Therapeutics . 2015;154:36–56. doi: 10.1016/j.pharmthera.2015.06.011. [DOI] [PubMed] [Google Scholar]
- 30.Bentzen S. M. Preventing or reducing late side effects of radiation therapy: radiobiology meets molecular pathology. Nature Reviews Cancer . 2006;6(9):702–713. doi: 10.1038/nrc1950. [DOI] [PubMed] [Google Scholar]
- 31.Neher M. D., Weckbach S., Flierl M. A., Huber-Lang M. S., Stahel P. F. Molecular mechanisms of inflammation and tissue injury after major trauma--is complement the "bad guy"? Journal of Biomedical Science . 2011;18(1) doi: 10.1186/1423-0127-18-90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wakabayashi K., Fujioka M., Kanzaki S., et al. Blockade of interleukin-6 signaling suppressed cochlear inflammatory response and improved hearing impairment in noise-damaged mice cochlea. Neuroscience Research . 2010;66(4):345–352. doi: 10.1016/j.neures.2009.12.008. [DOI] [PubMed] [Google Scholar]
- 33.Hirose K., Discolo C. M., Keasler J. R., Ransohoff R. Mononuclear phagocytes migrate into the murine cochlea after acoustic trauma. The Journal of Comparative Neurology . 2005;489(2):180–194. doi: 10.1002/cne.20619. [DOI] [PubMed] [Google Scholar]
- 34.Zhang W., Dai M., Fridberger A., et al. Perivascular-resident macrophage-like melanocytes in the inner ear are essential for the integrity of the intrastrial fluid-blood barrier. Proceedings of the National Academy of Sciences of the United States of America . 2012;109(26):10388–10393. doi: 10.1073/pnas.1205210109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Shi X. Resident macrophages in the cochlear blood-labyrinth barrier and their renewal via migration of bone-marrow-derived cells. Cell and Tissue Research . 2010;342(1):21–30. doi: 10.1007/s00441-010-1040-2. [DOI] [PubMed] [Google Scholar]
- 36.Wright H. L., Moots R. J., Bucknall R. C., Edwards S. W. Neutrophil function in inflammation and inflammatory diseases. Rheumatology . 2010;49(9):1618–1631. doi: 10.1093/rheumatology/keq045. [DOI] [PubMed] [Google Scholar]
- 37.Azzam P., Mroueh M., Francis M., Daher A. A., Zeidan Y. H. Radiation-induced neuropathies in head and neck cancer: prevention and treatment modalities. Ecancermedicalscience . 2020;14:p. 1133. doi: 10.3332/ecancer.2020.1133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Straub J. M., New J., Hamilton C. D., Lominska C., Shnayder Y., Thomas S. M. Radiation-induced fibrosis: mechanisms and implications for therapy. Journal of Cancer Research and Clinical Oncology . 2015;141(11):1985–1994. doi: 10.1007/s00432-015-1974-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nikitaki Z., Mavragani I. V., Laskaratou D. A., et al. Systemic mechanisms and effects of ionizing radiation: A new ‘old’ paradigm of how the bystanders and distant can become the players. Seminars in Cancer Biology . 2016;37-38:77–95. doi: 10.1016/j.semcancer.2016.02.002. [DOI] [PubMed] [Google Scholar]
- 40.Yakovlev V. A. Role of nitric oxide in the radiation-induced bystander effect. Redox Biology . 2015;6:396–400. doi: 10.1016/j.redox.2015.08.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Mole R. H. Whole body irradiation; radiobiology or medicine? The British Journal of Radiology . 1953;26(305):234–241. doi: 10.1259/0007-1285-26-305-234. [DOI] [PubMed] [Google Scholar]
- 42.Luce A., Courtin A., Levalois C., et al. Death receptor pathways mediate targeted and non-targeted effects of ionizing radiations in breast cancer cells. Carcinogenesis . 2009;30(3):432–439. doi: 10.1093/carcin/bgp008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Xiao L., Liu W., Li J., et al. Irradiated U937 cells trigger inflammatory bystander responses in human umbilical vein endothelial cells through the p38 pathway. Radiation Research . 2014;182(1):111–121. doi: 10.1667/RR13736.1. [DOI] [PubMed] [Google Scholar]
- 44.Shareef M. M., Cui N., Burikhanov R., et al. Role of tumor necrosis factor-alpha and TRAIL in high-dose radiation-induced bystander signaling in lung adenocarcinoma. Cancer Research . 2007;67(24):11811–11820. doi: 10.1158/0008-5472.CAN-07-0722. [DOI] [PubMed] [Google Scholar]
- 45.Azzam E. I., de Toledo S. M., Little J. B. Oxidative metabolism, gap junctions and the ionizing radiation-induced bystander effect. Oncogene . 2003;22(45):7050–7057. doi: 10.1038/sj.onc.1206961. [DOI] [PubMed] [Google Scholar]
- 46.Cheki M., Yahyapour R., Farhood B., et al. COX-2 in radiotherapy: a potential target for radioprotection and radiosensitization. Current Molecular Pharmacology . 2018;11(3):173–183. doi: 10.2174/1874467211666180219102520. [DOI] [PubMed] [Google Scholar]
- 47.Asenov D. R., Kaga K., Tsuzuku T. Changes in the audiograms of a nasopharyngeal cancer patient during the course of treatment: a temporal bone histopathological study. Acta Oto-Laryngologica . 2007;127(10):1105–1110. doi: 10.1080/00016480601127026. [DOI] [PubMed] [Google Scholar]
- 48.Tan P. X., Du S. S., Ren C., Yao Q. W., Yuan Y. W. Radiation-induced cochlea hair cell death: mechanisms and protection. Asian Pacific Journal of Cancer Prevention : APJCP . 2013;14(10):5631–5635. doi: 10.7314/APJCP.2013.14.10.5631. [DOI] [PubMed] [Google Scholar]
- 49.Hasegawa T., Kida Y., Kato T., Iizuka H., Yamamoto T. Factors associated with hearing preservation after gamma knife surgery for vestibular schwannomas in patients who retain serviceable hearing. Journal of Neurosurgery . 2011;115(6):1078–1086. doi: 10.3171/2011.7.JNS11749. [DOI] [PubMed] [Google Scholar]
- 50.Kurabi A., Keithley E. M., Housley G. D., Ryan A. F., Wong A. C. Cellular mechanisms of noise-induced hearing loss. Hearing Research . 2017;349:129–137. doi: 10.1016/j.heares.2016.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Choung Y. H., Taura A., Pak K., Choi S. J., Masuda M., Ryan A. F. Generation of highly-reactive oxygen species is closely related to hair cell damage in rat organ of Corti treated with gentamicin. Neuroscience . 2009;161(1):214–226. doi: 10.1016/j.neuroscience.2009.02.085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sha S. H., Taylor R., Forge A., Schacht J. Differential vulnerability of basal and apical hair cells is based on intrinsic susceptibility to free radicals. Hearing Research . 2001;155(1-2):1–8. doi: 10.1016/S0378-5955(01)00224-6. [DOI] [PubMed] [Google Scholar]
- 53.Wang J., Chen Y. Y., Tai A., et al. Sensorineural hearing loss after combined intensity modulated radiation therapy and cisplatin-based chemotherapy for nasopharyngeal carcinoma. Translational Oncology . 2015;8(6):456–462. doi: 10.1016/j.tranon.2015.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wang J., Ruel J., Ladrech S., Bonny C., van de Water T. R., Puel J. L. Inhibition of the c-Jun N-terminal kinase-mediated mitochondrial cell death pathway restores auditory function in sound-exposed animals. Molecular Pharmacology . 2007;71(3):654–666. doi: 10.1124/mol.106.028936. [DOI] [PubMed] [Google Scholar]
- 55.Feghali J. G., Liu W., Van De Water T. R. L-n-acetyl-cysteine protection against cisplatin-induced auditory neuronal and hair cell toxicity. The Laryngoscope . 2001;111(7):1147–1155. doi: 10.1097/00005537-200107000-00005. [DOI] [PubMed] [Google Scholar]
- 56.Zine A., van de Water T. R. The MAPK/JNK signalling pathway offers potential therapeutic targets for the prevention of acquired deafness. CNS & Neurological Disorders Drug Targets . 2004;3(4):325–332. doi: 10.2174/1568007043337166. [DOI] [PubMed] [Google Scholar]
- 57.Vanhoutte P. M., Shimokawa H., Feletou M., Tang E. H. Endothelial dysfunction and vascular disease - a 30th anniversary update. Acta Physiologica Scandinavica . 2017;219(1):22–96. doi: 10.1111/apha.12646. [DOI] [PubMed] [Google Scholar]
- 58.Tousoulis D., Kampoli A. M., Tentolouris Nikolaos Papageorgiou C., Stefanadis C., Stefanadis C. The role of nitric oxide on endothelial function. Current Vascular Pharmacology . 2012;10(1):4–18. doi: 10.2174/157016112798829760. [DOI] [PubMed] [Google Scholar]
- 59.Kerr R., Stirling D., Ludlam C. A. Interleukin 6 and haemostasis. British Journal of Haematology . 2001;115(1):3–12. doi: 10.1046/j.1365-2141.2001.03061.x. [DOI] [PubMed] [Google Scholar]
- 60.Hellweg C. E. The Nuclear Factor κB pathway: A link to the immune system in the radiation response. Cancer Letters . 2015;368(2):275–289. doi: 10.1016/j.canlet.2015.02.019. [DOI] [PubMed] [Google Scholar]
- 61.Schwartz I., Kim C. S., Shin S. O. Ultrastructural changes in the cochlea of the guinea pig after fast neutron irradiation. Otolaryngology and Head and Neck Surgery . 1994;110(4):419–427. doi: 10.1177/019459989411000412. [DOI] [PubMed] [Google Scholar]
- 62.Bohne B. A., Marks J. E., Glasgow G. P. Delayed effects of ionizing radiation on the ear. The Laryngoscope . 1985;95, 7 Part 1:818–828. doi: 10.1288/00005537-198507000-00014. [DOI] [PubMed] [Google Scholar]
- 63.Rubin P. The Franz Buschke lecture: late effects of chemotherapy and radiation therapy: a new hypothesis. International Journal of Radiation Oncology • Biology • Physics . 1984;10(1):5–34. doi: 10.1016/0360-3016(84)90408-5. [DOI] [PubMed] [Google Scholar]
- 64.Low W. K., Burgess R., Fong K. W., Wang D. Y. Effect of radiotherapy on retro-cochlear auditory pathways. The Laryngoscope . 2005;115(10):1823–1826. doi: 10.1097/01.mlg.0000175061.59315.58. [DOI] [PubMed] [Google Scholar]
- 65.Li J. J., Guo Y. K., Tang Q. L., et al. Prospective study of sensorineural hearing loss following radiotherapy for nasopharyngeal carcinoma. The Journal of Laryngology and Otology . 2010;124(1):32–36. doi: 10.1017/S0022215109991435. [DOI] [PubMed] [Google Scholar]
- 66.Lau S. K., Wei W. I., Sham J. S., Choy D. T., Hui Y. Early changes of auditory brain stem evoked response after radiotherapy for nasopharyngeal carcinoma--a prospective study. The Journal of Laryngology and Otology . 1992;106(10):887–892. doi: 10.1017/S002221510012119X. [DOI] [PubMed] [Google Scholar]
- 67.Milloy V., Fournier P., Benoit D., Noreña A., Koravand A. Auditory brainstem responses in tinnitus: a review of who, how, and what? Frontiers in Aging Neuroscience . 2017;9:p. 237. doi: 10.3389/fnagi.2017.00237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Gasser Rutledge K. L., Prasad K. G., Emery K. R., Mikulec A. A., Varvares M., Gratton M. A. Short-term peripheral auditory effects of cranial Irradiation. The Annals of Otology, Rhinology, and Laryngology . 2015;124(11):903–910. doi: 10.1177/0003489415591205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Tagoe T., Barker M., Jones A., Allcock N., Hamann M. Auditory nerve perinodal dysmyelination in noise-induced hearing loss. The Journal of Neuroscience . 2014;34(7):2684–2688. doi: 10.1523/JNEUROSCI.3977-13.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Xing Y., Samuvel D. J., Stevens S. M., Dubno J. R., Schulte B. A., Lang H. Age-related changes of myelin basic protein in mouse and human auditory nerve. PLoS One . 2012;7(4, article e34500) doi: 10.1371/journal.pone.0034500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Jacob J. T., Carlson M. L., Schiefer T. K., Pollock B. E., Driscoll C. L., Link M. J. Significance of cochlear dose in the radiosurgical treatment of vestibular schwannoma: controversies and unanswered questions. Neurosurgery . 2014;74(5):466–474. doi: 10.1227/NEU.0000000000000299. [DOI] [PubMed] [Google Scholar]
- 72.Nader M. E., Gidley P. W. Challenges of hearing rehabilitation after radiation and chemotherapy. Journal of Neurological Surgery Part B. Skull Base . 2019;80(2):214–224. doi: 10.1055/s-0039-1677865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lamaj E., Vu E., van Timmeren J. E., et al. Cochlea sparing optimized radiotherapy for nasopharyngeal carcinoma. Radiation Oncology . 2021;16(1):p. 64. doi: 10.1186/s13014-021-01796-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Chung L. K., Ung N., Sheppard J. P., et al. Impact of cochlear dose on hearing preservation following stereotactic radiosurgery and fractionated stereotactic radiotherapy for the treatment of vestibular schwannoma. Journal of Neurological Surgery Part B. Skull Base . 2018;79(4):335–342. doi: 10.1055/s-0037-1607968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Zhu W., Chen F., Li J., et al. Dosimetric parameters associated with conductive or sensorineural hearing loss 5 years after intensity-modulated radiation therapy in nasopharyngeal carcinoma. Acta Oto-Laryngologica . 2019;139(3):263–268. doi: 10.1080/00016489.2019.1566778. [DOI] [PubMed] [Google Scholar]
- 76.Yao J. J., Chen F. P., Zhou G. Q., et al. A prospective study on radiation doses to organs at risk (OARs) during intensity-modulated radiotherapy for nasopharyngeal carcinoma patients. Oncotarget . 2016;7(16):21742–21752. doi: 10.18632/oncotarget.7826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Yao J. J., Zhou G. Q., Lin L., et al. Dose-volume factors associated with ear disorders following intensity modulated radiotherapy in nasopharyngeal carcinoma. Scientific Reports . 2015;5(1) doi: 10.1038/srep13525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Zhang C., Liu L. X., Li W. Z., et al. Cochlea sparing with a stratified scheme of dose limitation employed in intensity-modulated radiotherapy for nasopharyngeal carcinoma: a dosimetry study. Medical Dosimetry . 2019;44(3):226–232. doi: 10.1016/j.meddos.2018.08.003. [DOI] [PubMed] [Google Scholar]
- 79.Gao J., Qian T. L., Tao C. Z., et al. SmartArc-based volumetric modulated arc therapy can improve the middle ear, vestibule and cochlea sparing for locoregionally advanced nasopharyngeal carcinoma: a dosimetric comparison with step-and-shoot intensity-modulated radiotherapy. The British Journal of Radiology . 2015;88(1053):p. 20150052. doi: 10.1259/bjr.20150052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Mahdavi S. R., Rezaeyan A., Nikoofar A., Bakhshandeh M., Farahani S., Cheraghi S. Comparison of radiation and chemoradiation-induced sensorineural hearing loss in head and neck cancer patients. Journal of Cancer Research and Therapeutics . 2020;16(3):539–545. doi: 10.4103/jcrt.JCRT_891_16. [DOI] [PubMed] [Google Scholar]
- 81.Scobioala S., Parfitt R., Matulat P., et al. Impact of radiation technique, radiation fraction dose, and total cisplatin dose on hearing : retrospective analysis of 29 medulloblastoma patients. Strahlentherapie und Onkologie . 2017;193(11):910–920. doi: 10.1007/s00066-017-1205-y. [DOI] [PubMed] [Google Scholar]
- 82.Yang Q., Cao S. M., Guo L., et al. Induction chemotherapy followed by concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in locoregionally advanced nasopharyngeal carcinoma: long-term results of a phase III multicentre randomised controlled trial. European Journal of Cancer . 2019;119:87–96. doi: 10.1016/j.ejca.2019.07.007. [DOI] [PubMed] [Google Scholar]
- 83.Mukherjea D., Dhukhwa A., Sapra A., et al. Strategies to reduce the risk of platinum containing antineoplastic drug-induced ototoxicity. Expert Opinion on Drug Metabolism & Toxicology . 2020;16(10):965–982. doi: 10.1080/17425255.2020.1806235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Kortmann R. D., Kühl J., Timmermann B., et al. Postoperative neoadjuvant chemotherapy before radiotherapy as compared to immediate radiotherapy followed by maintenance chemotherapy in the treatment of medulloblastoma in childhood: results of the German prospective randomized trial HIT '91. International Journal of Radiation Oncology • Biology • Physics . 2000;46(2):269–279. doi: 10.1016/S0360-3016(99)00369-7. [DOI] [PubMed] [Google Scholar]
- 85.van Vulpen M., Kal H. B., Taphoorn M. J., El-Sharouni S. Y. Changes in blood-brain barrier permeability induced by radiotherapy: implications for timing of chemotherapy? (Review) Oncology Reports . 2002;9(4):683–688. doi: 10.3892/or.9.4.683. [DOI] [PubMed] [Google Scholar]
- 86.Bhatta P., Dhukhwa A., Sheehan K., et al. Capsaicin protects against cisplatin ototoxicity by changing the STAT3/STAT1 ratio and activating cannabinoid (CB2) receptors in the cochlea. Scientific Reports . 2019;9(1) doi: 10.1038/s41598-019-40425-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Crowson M. G., Hertzano R., Tucci D. L. Emerging therapies for sensorineural hearing loss. Otology & Neurotology . 2017;38(6):792–803. doi: 10.1097/MAO.0000000000001427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Fujimoto C., Yamasoba T. Mitochondria-targeted antioxidants for treatment of hearing loss: A Systematic Review. Antioxidants . 2019;8(4):p. 109. doi: 10.3390/antiox8040109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Karaer I., Simsek G., Gul M., et al. Melatonin protects inner ear against radiation damage in rats. The Laryngoscope . 2015;125(10):E345–E349. doi: 10.1002/lary.25376. [DOI] [PubMed] [Google Scholar]
- 90.Lessa R. M., Oliveira J. A., Rossato M., Ghilardi Netto T. Analysis of the cytoprotective effect of amifostine on the irradiated inner ear of guinea pigs: an experimental study. Brazilian Journal of Otorhinolaryngology . 2009;75(5):694–700. doi: 10.1590/S1808-86942009000500014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Altas E., Ertekin M. V., Gundogdu C., Demirci E. L-carnitine reduces cochlear damage induced by gamma irradiation in Guinea pigs. Annals of Clinical and Laboratory Science . 2006;36(3):312–318. [PubMed] [Google Scholar]
- 92.Chen J., Zhao Y., Zhou X., et al. Methylprednisolone use during radiotherapy extenuates hearing loss in patients with nasopharyngeal carcinoma. The Laryngoscope . 2016;126(1):100–103. doi: 10.1002/lary.25527. [DOI] [PubMed] [Google Scholar]
- 93.Altas E., Ertekin M. V., Kuduban O., Gundogdu C., Demirci E., Sutbeyaz Y. Effects of piracetam supplementation on cochlear damage Occuring in guinea pigs exposed to irradiation. Biological & Pharmaceutical Bulletin . 2006;29(7):1460–1465. doi: 10.1248/bpb.29.1460. [DOI] [PubMed] [Google Scholar]
- 94.Pyun J. H., Kang S. U., Hwang H. S., et al. Epicatechin inhibits radiation-induced auditory cell death by suppression of reactive oxygen species generation. Neuroscience . 2011;199:410–420. doi: 10.1016/j.neuroscience.2011.09.012. [DOI] [PubMed] [Google Scholar]
- 95.Kim Y. R., Baek J. I., Kim S. H., et al. Therapeutic potential of the mitochondria-targeted antioxidant MitoQ in mitochondrial-ROS induced sensorineural hearing loss caused by _Idh2_ deficiency. Redox Biology . 2019;20:544–555. doi: 10.1016/j.redox.2018.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Murphy M. P. Understanding and preventing mitochondrial oxidative damage. Biochemical Society Transactions . 2016;44(5):1219–1226. doi: 10.1042/BST20160108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Landier W. Ototoxicity and cancer therapy. Cancer . 2016;122(11):1647–1658. doi: 10.1002/cncr.29779. [DOI] [PubMed] [Google Scholar]
- 98.Chang W. W., Yeung K. N., Luk B. P., Leung K. K. Y., Sung J. K. K., Tong M. C. F. Cochlear implantation in postirradiated ears: a case-control comparative study. Laryngoscope Investigative Otolaryngology . 2020;5(6):1163–1167. doi: 10.1002/lio2.486. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data, models, and code generated or used during the study appear in the submitted article.


