Abstract
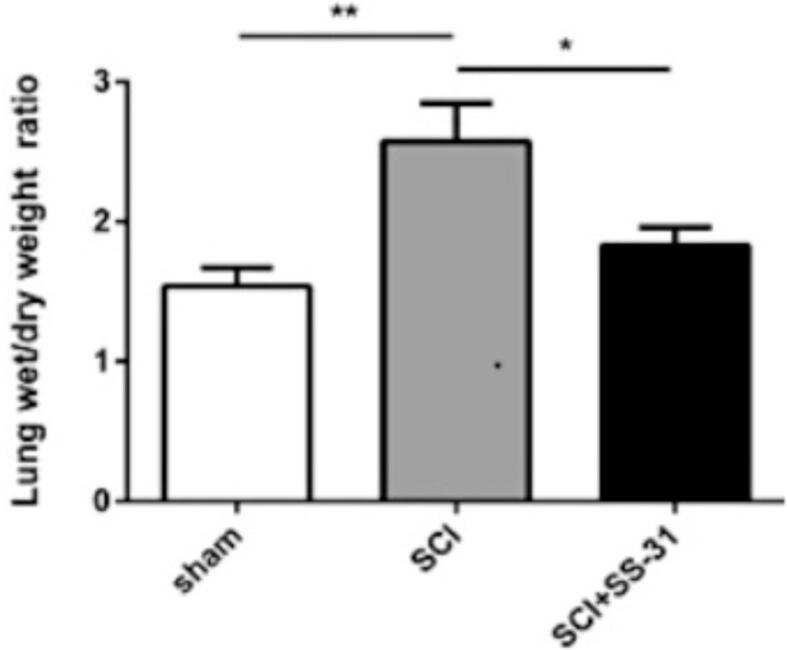
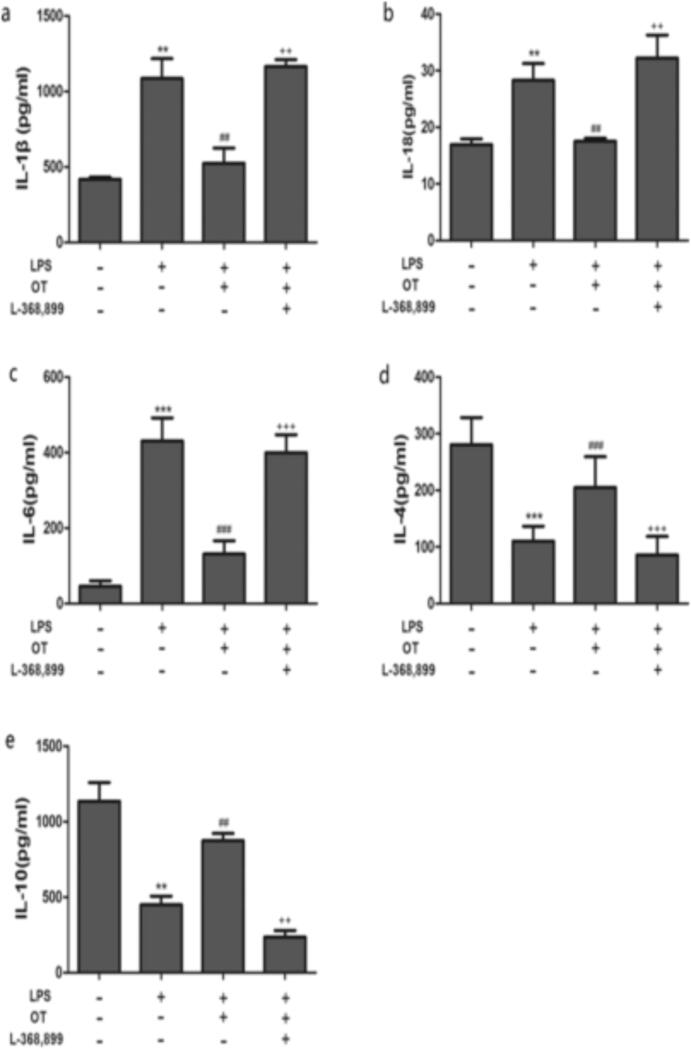
Lung fluid accumulation was determined using wet/dry lung mass ratio. Rats subjected to LPS-induced acute lung injury (2.8 ± 0.33, P < 0.05) presented with a significantly higher wet to dry lung weight ration ratio than sham rats (1.6 ± 0.23, P < 0.05). These results demonstrate that acutely inured rats' lungs were oedematous. On the other hand, treatment with scutellarin alone and in combination with a JNK inhibitor, SP600125, both significantly attenuated pulmonary edema as shown via reduced wet/dry lung mass ratios (1.7 ± 0.09 and 1.8 ± 0.23; P < 0.05, respectively). These results showed that the interventions were effective against LPS-induced edema of the lungs. However, the difference between treatment groups' weight ratios was not statistically significant (P > 0.05). In the sham control rats, the levels of ROS and SOD production were maintained at a low and at a high concentration, respectively (P < 0.05). However, following LPS infusion, the ROS levels skyrocketed while that of SOD decreased significantly relative to the control rats (P < 0.05). Furthermore, we noted that pre-treatment with scutellarin reduced the ROS levels in LPS-injured rats while the SOD was increased to near control levels (P < 0.05). Moreover, the combined effect of scutellarin and JNK inhibitor SP600125 on the levels of ROS and the SOD activity followed a similar trend to that of scutellarin alone albeit with a lower magnitude of change. Our results also showed that the combinatorial treatment was not significantly different from scutellarin alone in terms of influence on the levels of ROS production and SOD activity (P > 0.05). The effect of Scutellarin on broncho-alveolar lavage fluid (BALF) cytokine secretion The expression of interleukins-1β, −18 and −6 in the broncho-alveolar lavage fluid were significantly upregulated by LPS infusion (P < 0.05). The rise was, however, attenuated via pre-treatment with scutellarin only or in conjunction with SP600125, a JNK inhibitor (all P < 0.05). On the contrary, we observed that LPS injection caused a reduction of interlekins −4 and −10 secreted in the BALF. Pre-treatment with scutellarin alone (P < 0.05) and not in combination with SP600125 or SP600125 was able to significantly reverse this noted down-regulation (all P > 0.05).
Keywords: Scutellarin, Lung fluid accumulation, Mitochondrial dysfunction, Oxidative stress, Down-regulation
1. Introduction
Acute lung injury is a constellation of vascular endothelium and alveolar epithelium barrier injuries that are associated with PaO2/FiO2 values between 200 and 300 mmHg as well as significant morbidity and mortality (Ferguson et al., 2002, Rubenfeld et al., 2005). The disorder, primarily inflammatory, is associated with excessive immune cyto-activation, pro-inflammatory mediator secretion and disruption of membrane functions (Matthay and Zimmerman, 2005). Its clinical management involves mechano-ventilatory as well as pharmacological strategies. To date, the most successful interventions have been mechanical ventilation of acute lung injury patients (The Acute Respiratory Distress Syndrome Network, 2000). Numerous clinical trials revealed that pharmacotherapeutic modalities hold promises but have largely failed to mitigate against morbidity and mortality in these patients. As the advances in the understanding of pathophysiological processes underlying lung injury come ashore, efficacy testing of several drugs has fallen by the wayside (Steinberg et al., 2006, Morris et al., 2008, Liu et al., 2008) thereby warranting continued search for pharmacological agents that would mitigate cellular damage, reverse acute lung injury, and increase survival among patients.
The region of interest in acute lung injury is the site of gaseous exchange comprising the surfactant layer sitting on the alveolar type 1 cells which are separated from the vascular endothelial cells by respective basement membranes and an interstitial space. Several stess, including viral infections (coronavirus, influenza, and adenovirus), idiopathic, severe sepsis, burns, smoke inhalation and drug poisoning will invariably cause cellular and fluid liberation within this region of interest. The infiltrating neutrophils and alveolar macrophages will invade the interstitium. The alveolar macrophages exudate includes cytokines such as interleukins-1,-6, −8, −10, and TNF-α, that are chemoattractant and stimulatory to neutrophils and fibroblasts (Matthay et al., 2002). In turn, neutrophils liberate copious amounts of oxidizing agents, peptidases, leukotrienes, and other inflammation promoting molecules. The mitochondria have featured as leading site of subcellular pathological processes that have a strong bearing towards cytoprotection or cytotoxicity (Yu et al., 2016, Lee et al., 2017). During acute lung injury, the mitochondria may lose the ability maintain normal calcium ion and reactive oxygen species (ROS) concentration leading to excessive mitochondrial free radical leakage, mitochondrial DNA damage and out-of-control ROS production (de Prost et al., 2011, Du et al., 2014). The overall outcome of these pathological processes would be unmitigated mitochondrial dysfunction, oxidative stress and apoptosis of the cellular components of the inflamed lung tissue.
Scutellarin is a natural bioactive flavonoid extracted from Erigeron breviscapus. It has been shown to have antiinflammatory and antioxidative activity, antiapoptotic, antihyperglycermic, neuroprotective, and anti-tumorigenic effects (Hong and Liu, 2004, Luo et al., 2008, Ma et al., 2008, Wang et al., 2011, Yang et al., 2017, Hu et al., 2018) in several efficacy studies. Therefore, scutellarin suggests appear to be a versatile molecule that has immense potential for clinical applications in treating disorders such as acute lung injury whole hallmarks include oxidative stress, inflammation, and mitochondrial dysfunction. Thus, we examined the effect of scutellarin on acute lung injury in an LPS-induced pulmonary injury model.
2. Materials and methods
2.1. Animal husbandry and experimental modeling
After getting approval to conduct the experiments from the, 42 two-month-old male Sprague-Dawley rats weighing between 185 and 220 g were bought from the. The rats were accorded unbridled access to standard rat chow and water. They were allowed to get accustomed to the new environment with a 12-hour light and darkness cycle for 7 days. Before the experimentation, these rats were randomly subdivided into six groups (n = 6 each) namely: (i) control (saline vehicle), (ii) LPS-group (6 mg/kg, dissolved in normal saline), (iii) LPS plus Scutellarin, (iv) LPS plus SP600125 (a specific inhibitor of JNK), (v) LPS plus Scutellarin plus SP600125, (vi) LPS plus normal saline. To induce acute lung injury, the rats were anesthetized by isoflurane inhalation and then a 24-gauge cannula inserted into the jugular vein for the administration of LPS. Scutellarin (dose) or SP600125 (15 mg/kg) was administrated per intraperitoneal an hour before LPS infusion. An equal volume of sterile normal saline acted as a control vehicle. The rats were allowed to survive and sacrificed 3 days after LPS application.
2.2. Extracting lung tissue
All lungs (left side) tissue was carefully removed following trans-cardiac perfusion with precooled normal saline, and then kept at −80 °C awaiting quantitative real-time PCR (RT-qPCR), Western Blotting, measurement of myeloperoxidase, mitochondrial bioactivity, SOD and ROS generation.
2.3. Lung edema evaluation
Whole lungs (right) were rapidly dissected out and cleaned of extra-pulmonary connective tissue. The lungs’ wet mass was obtained and then they were placed in an over to dry at 65 °C over 4 nights. They were weighed again to obtain dry mass. The wet to dry weight ratio was computed as an indicator of fluid accumulation inside lungs.
2.4. RT-qPCR
Extraction of RNA and DNA was carried out using the reagent Trizol (Life technologies, USA) according to accompanying instructions. The primer set used in this assay were constituted using Primer3 suite and are listed in Table 1. The expression of targeted genes was compared to 18S rRNA or GAPDH, as applicable, and Δ Δ Ct log-fold-change method used for quantitation (n = 6). The general RT-qPCR procedures followed were in line with previously published protocols (Zhu et al., 2017).
Table 1.
RT-qPCR primers.
| Gene | Forward primers(5′-3′) | Reverse primers(5′-3′) |
|---|---|---|
| IL − 1β | ATGAAAGACGGCACACCCAC | AAGGCAGAGTCTTCGGTGAG |
| IL − 6 | CAACCAAGAGGTGAGTGCTTC | GGTGTCCTCTTTCCCACACTG |
| IL − 18 | GGAAGACCAGAGACATCCACTG | ACACTAGACCAAAGGGCTTG |
| IL − 4 | CTGTAGGGCTTCCAAGGTGC | CTCTCATTGTGCCAGGTCACT |
| IL − 10 | GCCAGTTAGAAAGCCACCAC | GGTTCAGCCTGTTTCCCAAC |
| Mfn1 | TGGGGAGGTGCTGTCTCGGA | ACCAATCCCGCTGGGGAGGA |
| ACTIN | GGCTGTATTCCCCTCCATCG | CCAGTTGGTAACAATGCCATGT |
2.5. Reactive oxygen species (ROS) and SOD evaluation
The frozen lungs were broken to tiny pieces and homogenized in lytic enzymes for 1 h at 37 °C. The homogenate was then filtered, centrifuged and its supernatant eluted. A 2, 7-Dichloro-dihydrofluorescein-diacetate solution was mixed with the supernatant and put in incubator at 37 °C for half an hour. A resulting solution then rinsed thrice with serum free medium, and then suspended in phosphate buffer saline. Commercial kits purchased from (Nanjing Jincheng Bioengineering Institute, China) were used to assess ROS production and SOD activity following recommended protocols (n = 6 mice/group).
2.6. Western blot
Lung samples were homogenized and protein obtained using Abcam's 10X RIPA lysis buffer (Abcam, UK). The concentration of protein in samples was determined using a BCATM protein assay kit (Pierce, USA) according protocol given by the manufacturer. 30 µg per lane total protein was electroseparated and electrotransferred onto polyvinylidene-difluoride membranes (BioRad, USA). 5% skimmed milk was used as a blocking buffer for 1 h at room temperature. The following primary antibodies were used: mitofusin-1 protein (Mfn 1), c-jun, phosphor-c-jun, cytochrome c (Cyt C) (All 1: 1000; Cell Signaling Tech, USA), cleaved caspase-3 (1: 2000; Santa Cruz Biotech, USA), IL-1β, IL-6 (all 1: 1500; Cell Signaling Technology, USA), and GAPDH (1: 1500; Santa Cruz Biotech, USA) overnight at 4 °C. The next day, the strips of membrane were incubated with an HRP conjugated secondary antibody (). For optical density quantitation, each immunoband intensity was standardised to GAPDH (n = 6 mice/group).
2.7. Myeloperoxidase (MPO) activity: Bronchoalveolar lavage fluid (BALF)
MPO activity was used as a sign of pulmonary invasion by macrophages as well as neutrophils (Loria et al., 2008). This was measured at 450 nm according to instructions contained in commercial kits bought at Wuhan Huamei Biotechnology CO., PR China.
2.8. BALF cytokines levels
Rat BALF concentration of cytokines, interleukins-1β, −18, −6, −4, and −10 we determined using commercial ELISA kits as dictated in the manufacturer’s manual. The reading were obtained using amount of light absorbed at 450 nm on an ELx800 Absorbance Microplate Reader (Life Sciences, USA).
2.9. Statistical analysis
The data are represented by Mean ± Standard Error of the Mean. The tatistical significance of differences between various experimental and control groupings were computed using one-way analysis of variance supplemented with Tukey’s post-hoc testing where appropriate. For scenarios whereby data does not follow a normal distribution, the statistical significance of the difference between groups were checked with one-way ANOVA followed by Dunn’s post-hoc method. All p-values<0.05 were taken as statistically significant differences.
3. Results
3.1. Scutellarin attenuates fluid accumulating in lungs
Lung fluid accumulation was determined using wet/dry lung mass ratio. Rats subjected to LPS-induced acute lung injury (2.8 ± 0.33, P < 0.05) presented with a significantly higher wet to dry lung weight ration ratio than sham rats (1.6 ± 0.23, P < 0.05, Fig. 1). These results demonstrate that acutely inured rats' lungs were edematous. On the other hand, treatment with scutellarin alone and in combination with a JNK inhibitor, SP600125, both significantly attenuated pulmonary edema as shown via reduced wet/dry lung mass ratios (1.7 ± 0.09 and 1.8 ± 0.23; P < 0.05, respectively). These results showed that the interventions were effective against LPS-induced edema of the lungs. However, the difference between treatment groups' weight ratios was not statistically significant (P > 0.05).
Fig. 1.
Scutellarin attenuates fluid accumulating in lungs.
3.2. Scutellarin protects against LPS-induced pulmonary edema.
Pulmonary edema was assessed using wet/dry mass ratios at 3 days after LPS induction. *denotes significance at P < 0.05. Data presented is Mean ± SEM; n = 6.
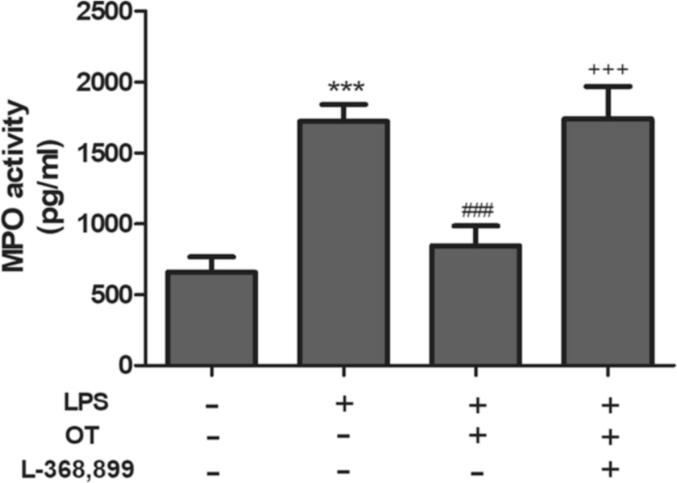
3.3. The effect of scutellarin on MPO activity
The MPO activity is a known biomarker of neutrophil and macrophage pulmonary infiltration (Loria et al., 2008). As Fig. 2 shows, LPS infusion resulted in a robust rise in MPO activity when compared with that seen in saline infused rats (P < 0.05). In addition, the rise was significantly inhibited by scutellarin pre-treatment (P < 0.05). Furthermore, JNK inhibitor SP600125 given an hour prior to LPS insult restored MPO activity to near normal levels (P < 0.05). There was no evidence of synergism between scutellarin and SP600125 treatments as the difference between the scutellarin only and the scutellarin plus SP600125 was not statistically significant.
Fig. 2.
Effect of Scutellarin on MPO activity.
3.4. The effects of scutellarin on pulmonary MPO activity
The data is represented by Mean ± standard error of the mean. The * denotes P < 0.05 for significant differences. LPS-induced acute lung injury vs normal saline sham control. LPS + Scutellarin vs LPS and LPS + scutellarin + SP600125. n = 6
3.5. Effect of scutellarin on ROS content and SOD activity
In the sham control rats, the levels of ROS and SOD production were maintained at a low and at a high concentration, respectively (P < 0.05, Table 2). However, following LPS infusion, the ROS levels skyrocketed while that of SOD decreased significantly relative to the control rats (P < 0.05; Table 2). Furthermore, we noted that pre-treatment with scutellarin reduced the ROS levels in LPS-injured rats while the SOD was increased to near control levels (P < 0.05, Table 1). Moreover, the combined effect of scutellarin and JNK inhibitor SP600125 on the levels of ROS and the SOD activity followed a similar trend to that of scutellarin alone albeit with a lower magnitude of change. Our results also showed that the combinatorial treatment was not significantly different from scutellarin alone in terms of influence on the levels of ROS production and SOD activity (P > 0.05, Table 1).
Table 2.
ROS production and SOD activity following LPS-induced acute lung injury Oxidative stress measurements.
| Parameters | Control | LPS | LPS + Scutellarin | LPS + Scutellrin + SP600125 |
|---|---|---|---|---|
| ROS | 109.11 ± 1.11 | 152.3 ± 4.11* | 129.12 ± 2.95* | 149.99 ± 4.21 |
| SOD | 60.77 ± 1.88 | 51.25 ± 1.44* | 66.23 ± 2.01* | 52.78 ± 1.85 |
ROS production and SOD activity following experimental acute lung injury caused by LPS plus treatment with SP600125 or scutellarin. LPS treatment robustly augmented the levels of ROS generation while decreasing SOD activity. However, after scutellarin significantly downregulate the levels of ROS and augmented SOD activity (*P < 0.05, 2)
The effect of Scutellarin on broncho-alveolar lavage fluid (BALF) cytokine secretion
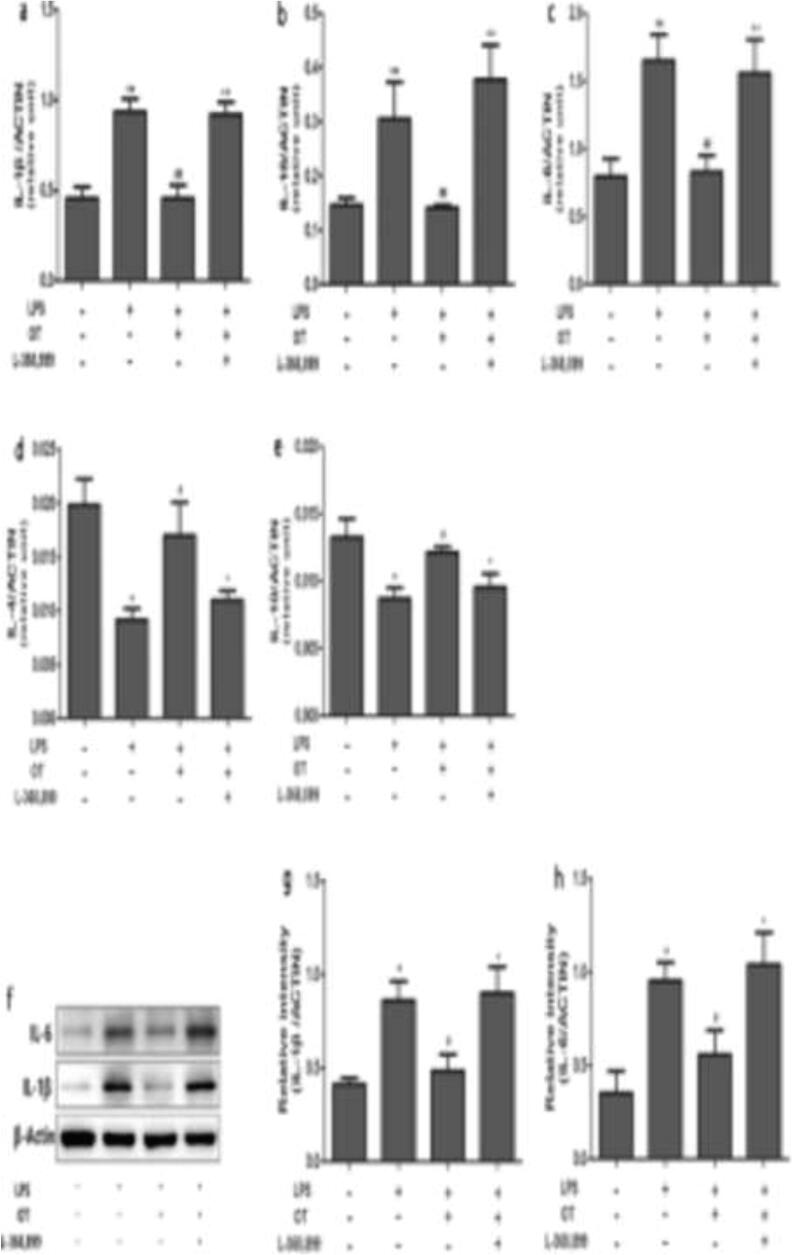
The expression of interleukins-1β, −18 and −6 in the broncho-alveolar lavage fluid were significantly upregulated by LPS infusion (P < 0.05). The rise was, however, attenuated via pre-treatment with scutellarin only or in conjunction with SP600125, a JNK inhibitor (all P < 0.05). On the contrary, we observed that LPS injection caused a reduction of interlekins −4 and −10 secreted in the BALF. Pre-treatment with scutellarin alone (P < 0.05) and not in combination with SP600125 or SP600125 was able to significantly reverse this noted down-regulation (all P > 0.05). In addition, the trend of the levels of expression of these cytokines was also concordant with RT qPCR and Western blot assays shown in Fig. 3. Fig. 4.
Fig. 3.
The effects of scutellarin treatment on inflammatory cytokine expression in BALF of rats suffering from LPS-induced acute lung injury. The BALF expression of interleukins IL-1β (i), IL-18 (ii), IL-6 (iii), IL-4 (iv), and IL-10 (v) determined by ELISA. Data is expressed as mean ± standard error of the mean. The * denotes P < 0.05 as significant; n = 6.
Fig. 4.
The effect of scutellarin on the cytokine expression in the lungs of LPS-induced rats (RT-qPCR and Western blot).
The gene assay: IL-1β (i), IL-18 (ii) IL-6 (iii), IL-4 (iv), and IL-10 (v). The confirmatory protein expression immunoblotting of selected IL-1β; IL-6 normalised to β-Actin levels (vi). The data represented by mean ± standard error of the mean. The * denotes when P < 0.05, statistically significant; n = 6.
As depicted in Fig. 5, LPS insult significantly increased c-jun, c-jun phosphrylation and cleaved caspase 3 expression than observed in the control groups (P < 0.05). However, scutellarin pre-treatment significantly lowered the expression of these proteins relative to that observed in the LPS induction-only rats (P < 0.05). Furthermore, SP600125 pre-treatment alone or in combination with scutellarin also significantly reduced c-jun cleaved caspase 3 and phospho-c-jun protein overexpression relative to the controls. The results were corroborated by densitometry evaluation on the western blots.
Fig. 5.
The mechanism of Scutellarin recovery in LPS-induced acute lung injured rats.
3.6. The mechanism of scutellarin recovery in LPS-induced acute lung injured rats
The Western blot analysis revealed overexpression of pro apoptotic and JNK signaling pathways. Pre-treatment with scutellarin slowed JNK signaling and also reduced pro-apoptotic cleaved caspase 3 expression * denotes P < 0.05. The data depicts Mean ± standard error of the mean for 6 rats per group.
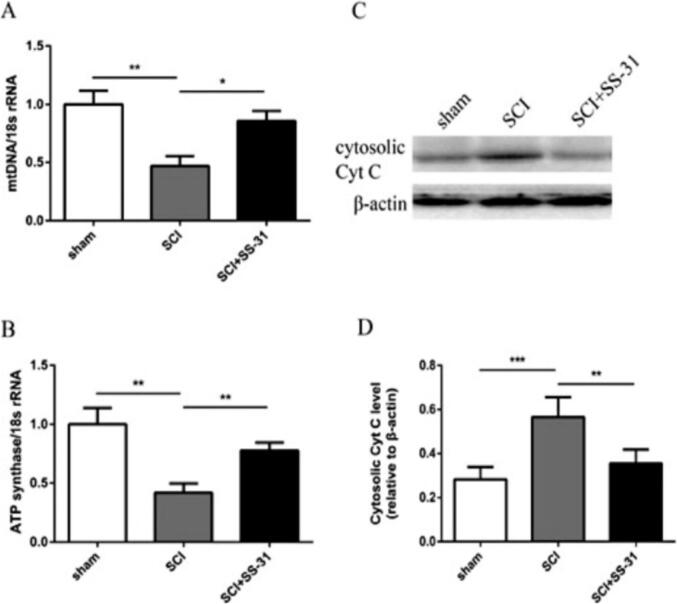
3.7. Scutellarin restrains mitochondrial dysfunction
Our results show that LPS induced acute lung injury presents with significantly reduced mitochondrial DNA amount (P < 0.05), ATP synthases activity (P < 0.05), as well as heightened cytosolic liberation of Cyt C (P < 0.05) relative to controls. We also observed that pre-treatment with scutellarin restores mitochondrial DNA amount, ATP synthases function, as well as inhibits cytoplasmic Cyt C release at day three after LPS-induced lung injury. LPS infusion also reduced mitofusin-1 protein (Mfn1) expression which as then significantly revered by scutellarin treatment.
3.8. Scutellarin attenuates mitochondrial dysfunction.
Fig. 6 A shows RT-qPCR assays of ATP-synthases and mt DNA genes at day 3 after LPS induced lung-injury. Fig. 6B represents cytochrome c and mitofusin-1 protein (Mfn1) expression western blots at day 3 after LPS-induced lung injury.* depicts P < 0.05. The data represents mean ± standard error of the mean; n = 6.
Fig. 6.
Scutellarin attenuates mitochondrial dysfunction.
4. Discussion
Clinically, acute lung injury is a serious condition accounting for the majority of acute respiratory failure among seriously ill patients worldwide. Its management remains largely supportive (The Acute Respiratory Distress Syndrome Network, 2000), creating an ongoing need for continued research. In the present study, we used an LPS injection lung injury model to demonstrate scutellarin’s role in mitigating against oxidative stress, inflammation and apoptosis which collectively underpin severe adult respiratory distress seen in patients with acute lung injuries. In particular, our results indicated that the role of scutellarin is through interfering with the downstream JNK signalling, reduction of oxidative stress as well as improving antioxidant SOD activity and being antiapoptotic. Scutellarin also worked against LPS-induced decline in the number of the mitochondria. Furthermore, a JNK kinase inhibitor, also abrogated LPS-induced oxidative stress, inflammation and apoptosis in the injured lungs. Therefore, we posited that prevention of JNK dependent c-jun phospho-activation partially mediates the attenuation of acute injury to lungs in vivo.
Insult to the lungs kicks starts a series of pathological reactions, chief among them being the alveolar macrophage chemotaxis of neutrophils to the alveolar interstitium (Barnes, 2009, Grommes and Soehnlein, 2011). Once at the site of injury, neutrophils along with alveolar macrophages and fibroblasts secrete cell damaging oxidizing agents, peptidases, leukotrienes and cytokines that creates a positive feedback leading to setting in of local edema should inflammation fail to resolve quickly (Abraham, 2003). As observed in the present study, the biochemical markers of pulmonary infiltration by neutrophils and macrophages were all elevated in injured lung tissue (Loria et al., 2008). The injured lung wet to dry weight ratio was also skewed in favor of heightened pulmonary edema in the LPS-injured rats. These results created replicated a standard previously reported in human patients (Matthay et al., 1984) and animal model studies (Matsuyama et al., 2008). Interestingly, our present study showed that pretreatment with scutellarin reversed the severity of these biomarkers setting it as a good candidate for managing acute lung injuries as well as their clinical sequelae acute respiratory distress syndrome. Scutellarin appears to reduce neutrophil infiltration as shown by dwarfed myeloperoxidase activity, a feat that has been touted as a plausible therapeutic target in mice (Abraham et al., 2000) and being pursued in clinical trials (Egners et al., 2016). Intriguingly, JNK inhibitor SP600125 also significantly attenuated these parameters, suggesting that the beneficial effects maybe mediated partially by blockage of c-jun phosphorylation and activation. JNK substrate, c-jun, is an immediate-early gene that is responsive to lung injury and its phosphoactivation has been associated with pulmonary cytotoxicity, among other functions (Bennett, 2006).
Following acute lung injury, the recruited monocytes and macrophages along with neutrophils secrete copious amounts of cytokines such as interleukins-1β, −6, and −18 which intercede the ensuing inflammatory response (Matthay et al., 2002). The expression of these pro-inflammatory interleukins in the broncho-alveolar lavage fluid were significantly upregulated by LPS infusion while anti-inflammatory IL-4 and IL-10 were down regulated in the current study. These trends are concordant with the previous reports by An et al., (2019) and (Sawada et al., 2013) in mice and rats, respectively. IL-18, a known pro-inflammatory cytokine, recruits other inflammation mediating factors (Jordan et al., 2001) and was dysregulated in this study. We investigated the effects of scutellarin on and found that it reverses LPS-induced interleukin dysregulation. In addition, JNK inhibitor SP600125, showed a similar but weaker trend. These results suggest that scutellarin acts by promoting anti-inflammatory cytokine expression while suppressing pro-inflammatory ones. These dichotomous roles of scutellarin have been observed before in stroke (Ma et al., 2008) and neurodegeneration (Hong and Liu, 2004), diabetes (Luo et al., 2008), nervous injury (Wang et al., 2011), and cancer (Yang et al., 2017).
The ultimate result of alveolar cellular infiltration and subsequent secretion of pro-inflammatory molecules in the alveolar interstitium offsets the endogenous scavenging capacity leading to rapid generation of reactive oxygen and nitrogen species. These oxidizing agents can destroy lipid, proteins and DNA leading to apoptosis. Manganese superoxide dismutase (Mn SOD is antitoxidant and retrieves superoxide anions that would drive cytotoxic processes in the injured lungs (Hart et al., 2015). As observed currently, LPS induced high ROS production as well as apoptosis while dwindling the endogenous SOD scavenging mechanism. These results are in line with prior observations in mice, rats and ARDS patients. Pretreatment with scutellarin reversed these LPS induced changes suggesting that its antioxidant functions are at play in injured lungs. The mitochondria is a metabolic center as well as integrator of cell death or survival via the mitochondrial apoptotic pathway. In this study, LPS injection dwindled mitochondrion numbers while pretreatment with scutellarin rescued mitochondrial loss. In addition, inhibition of cytochrome c liberation and mitofusin-1 protein overexpression where mediated by scutellarin pretreatment. In response to injury, the mitochondria shifts the dynamic equilibrium between fission and fusion in order to preserve mitochondrial functions (Ni et al., 2015).
5. Conclusions
In conclusion, the current study has revealed that scutellarin alone and in combination with JNK inhibitor SP600125 is protective against acute lung injury induced by LPS by inhibiting inflammation, apoptotic as well as oxidative stress in a rat model. The mechanism appears to encompass inhibition of the JNK/c-jun/Phospho-c-jun/cleaved caspase 3 signaling pathway. These results sets scutellarin as a novel therapeutic strategy in the management of acute lung injuries.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Footnotes
Peer review under responsibility of King Saud University.
References
- Abraham E. Neutrophils and acute lung injury. Crit. Care Med. 2003;31(Supplement):S195–S199. doi: 10.1097/01.CCM.0000057843.47705.E8. [DOI] [PubMed] [Google Scholar]
- Abraham E., Carmody A., Shenkar R., Arcaroli J. Neutrophils as early immunologic effectors in hemorrhage- or endotoxemia-induced acute lung injury. Am. J. Physiol. Lung Cell. Mol. Physiol. 2000;279(6):L1137–L1145. doi: 10.1152/ajplung.2000.279.6.L1137. [DOI] [PubMed] [Google Scholar]
- An X., Sun X., Hou Y., Yang X., Chen H., Zhang P., Wu J. Protective effect of oxytocin on LPS-induced acute lung injury in mice. Sci. Rep. 2019;9(1) doi: 10.1038/s41598-019-39349-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnes P.J. The cytokine network in chronic obstructive pulmonary disease. Am. J. Res. Cell Mol. Biol. 2009;41(6):631–638. doi: 10.1165/rcmb.2009-0220tr. [DOI] [PubMed] [Google Scholar]
- Bennett B.L. c-Jun N-terminal kinase-dependent mechanisms in respiratory disease. Eur. Respir. J. 2006;28(3):651–661. doi: 10.1183/09031936.06.00012106. [DOI] [PubMed] [Google Scholar]
- de Prost N., Ricard J.D., Saumon G., et al. Ventilator-induced lung injury: historical perspectives and clinical implications. Ann. Int. Care. 2011;1:1–15. doi: 10.1186/2110-5820-1-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du Q., Wang C., Zhang N., Li G., Zhang M., Li L., Zhang Q., Zhang J. In vivo study of the effects of exogenous hydrogen sulfide on lung mitochondria in acute lung injury in rats. BMC Anesthesiol. 2014;14:117. doi: 10.1186/1471-2253-14-117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Egners A., Erdem M., Cramer T. The Response of Macrophages and Neutrophils to Hypoxia in the Context of Cancer and Other Inflammatory Diseases. Mediators Inflamm. 2016;2016:1–10. doi: 10.1155/2016/2053646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferguson N.D., Meade M.O., Hallett D.C., Stewart T.E. High values of the pulmonary artery wedge pressure in patients with acute lung injury and acute respiratory distress syndrome. Intensive Care Med. 2002;28(8):1073–1077. doi: 10.1007/s00134-002-1354-y. [DOI] [PubMed] [Google Scholar]
- Grommes J., Soehnlein O. Contribution of neutrophils to acute lung injury. Mol. Med. 2011;17(3-4):293–307. doi: 10.2119/molmed.2010.00138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hart P.C., Mao M., de Abreu A.L., Ansenberger-Fricano K., Ekoue D.N., Ganini D., Kajdacsy-Balla A., Diamond A.M., Minshall R.D., Consolaro M.E., Santos J.H., Bonini M.G. MnSOD upregulation sustains the Warburg effect via mitochondrial ROS and AMPK-dependent signalling in cancer. Nat. Commun. 2015;5(6):6053. doi: 10.1038/ncomms7053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hong H., Liu G.Q. Protection against hydrogen peroxide-induced cytotoxicity in PC12 cells by scutellarin. Life Sci. 2004;74(24):2959–2973. doi: 10.1016/j.lfs.2003.09.074. [DOI] [PubMed] [Google Scholar]
- Hu X., Teng S., He J., Sun X., Du M., Kou L., Wang X. Pharmacological basis for application of scutellarin in Alzheimer's disease: Antioxidation and antiapoptosis. Mol. Med. Rep. 2018;18(5):4289–4296. doi: 10.3892/mmr.2018.9482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jordan, J.A., Guo, R.F., Yun, E.C., Sarma, V., Warner, R.L., Crouch, L.D., Senaldi, G., Ulich, T.R., Ward, P.A., 2001. Role of IL-18 in acute lung inflammation. The Journal of Immunology, 167(12), pp.7060-7068. J. Immunol. 167(12), 7060-7068, https://doi.org/10.4049/jimmunol.167.12.7060. [DOI] [PubMed]
- Lee Y.L., Obiako B., Gorodnya O.M., Ruchko M.V., Kuck J.L., Pastukh V.M., Wilson G.L., Simmons J.D., Gillespie M.N. Mitochondrial DNA Damage Initiates Acute Lung Injury and Multi-Organ System Failure Evoked in Rats by Intra-Tracheal Pseudomonas Aeruginosa. Shock. 2017;48(1):54–60. doi: 10.1097/SHK.0000000000000838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu K.D., Levitt J., Zhuo H., Kallet R.H., Brady S., Steingrub J., Tidswell M., Siegel M.D., Soto G., Peterson M.W., Chesnutt M.S., Phillips C., Weinacker A., Thompson B.T., Eisner M.D., Matthay M.A. Randomized clinical trial of activated protein C for the treatment of acute lung injury. Am. J. Respir. Crit. Care Med. 2008;178(6):618–623. doi: 10.1164/rccm.200803-419OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loria V., Dato I., Graziani F., Biasucci L.M. Myeloperoxidase: a new biomarker of inflammation in ischemic heart disease and acute coronary syndromes. Mediators Inflamm. 2008;2008:1–4. doi: 10.1155/2008/135625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo P., Tan Z.H., Zhang Z.F., Zhang H., Liu X.F., Mo Z.J. Scutellarin isolated from Erigeron multiradiatus inhibits high glucose-mediated vascular inflammation. Yakugaku Zasshi. 2008;128(9):1293–1299. doi: 10.1248/yakushi.128.1293. [DOI] [PubMed] [Google Scholar]
- Ma J.Y., Jiang W.W., Zhou Z.T., Li J.M., Wang H.Y. The promoting angiogenesis and anti-inflammation effect of scutellarin on polyglycolic acid scaffold of balb/c mice model. J. Asian Nat. Prod. Res. 2008;10(12):1147–1153. doi: 10.1080/10286020802361255. [DOI] [PubMed] [Google Scholar]
- Matsuyama H., Amaya F., Hashimoto S., Ueno H., Beppu S., Mizuta M., Shime N., Ishizaka A., Hashimoto S. Acute lung inflammation and ventilator-induced lung injury caused by ATP via the P2Y receptors: an experimental study. Respir. Res. 2008;9(1) doi: 10.1186/1465-9921-9-79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matthay M.A., Bhattacharya S., Gaver D., Ware L.B., Lim L.H.K., Syrkina O., Eyal F., Hubmayr R. Ventilator-induced lung injury: in vivo and in vitro mechanisms. Am. J. Physiol. 2002;283(4):L678–L682. doi: 10.1152/ajplung.00154.2002. [DOI] [PubMed] [Google Scholar]
- Matthay, M.A., Eschenbacher, W.L., Goetzl, E.J., 1984. Elevated concentrations of leukotriene D4 in pulmonary edema fluid of patients with the adult respiratory distress syndrome. J. Clin. Immunol. 4, 479–83, https://doi.org/ 10.1007/BF00916578. [DOI] [PubMed]
- Matthay M.A., Zimmerman G.A. Acute lung injury and the acute respiratory distress syndrome: four decades of inquiry into pathogenesis and rational management. Am. J. Respir. Cell. Mol. Biol. 2005;33(4):319–327. doi: 10.1165/rcmb.F305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris P.E., Papadakos P., Russell J.A., Wunderink R., Schuster D.P., Truwit J.D., Vincent J.-L., Bernard G.R. A double-blind placebo-controlled study to evaluate the safety and efficacy of L-2-oxothiazolidine-4-carboxylic acid in the treatment of patients with acute respiratory distress syndrome. Crit. Care Med. 2008;36(3):782–788. doi: 10.1097/CCM.0B013E318164E7E4. [DOI] [PubMed] [Google Scholar]
- Ni H.M., Williams J.A., Ding W.X. Mitochondrial dynamics and mitochondrial quality control. Redox. Biol. 2015;4:6–13. doi: 10.1016/j.redox.2014.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rubenfeld G.D., Caldwell E., Peabody E., Weaver J., Martin D.P., Neff M., Stern E.J., Hudson L.D. Incidence and outcomes of acute lung injury. N. Engl. J. Med. 2005;353(16):1685–1693. doi: 10.1056/NEJMoa050333. [DOI] [PubMed] [Google Scholar]
- Sawada M., Kawayama T., Imaoka H., Sakazaki Y., Oda H., Takenaka S.-I., Kaku Y., Azuma K., Tajiri M., Edakuni N., Okamoto M., Kato S., Hoshino T., Idzko M. IL-18 induces airway hyperresponsiveness and pulmonary inflammation via CD4+ T cell and IL-13. PLoS One. 2013;8(1):e54623. doi: 10.1371/journal.pone.005462310.1371/journal.pone.0054623.g00110.1371/journal.pone.0054623.g00210.1371/journal.pone.0054623.g00310.1371/journal.pone.0054623.g00410.1371/journal.pone.0054623.g005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinberg K.P., Hudson L.D., Goodman R.B., Hough C.L., Lanken P.N., Hyzy R., Thompson B.T., Ancukiewicz M. National Heart, Lung, and Blood Institute, and Acute Respiratory Distress Syndrome Clinical Trials Network: Efficacy and safety of corticosteroids for persistent acute respiratory distress syndrome. N. Engl. J. Med. 2006;354:1671–1684. doi: 10.1056/NEJMoa051693. [DOI] [PubMed] [Google Scholar]
- Network T.A.R.D.S. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N. Engl. J. Med. 2000;342(18):1301–1308. doi: 10.1056/NEJM200005043421801. [DOI] [PubMed] [Google Scholar]
- Wang S., Wang H., Guo H., Kang L., Gao X., Hu L. Neuroprotection of Scutellarin is mediated by inhibition of microglial inflammatory activation. Neuroscience. 2011;185:150–160. doi: 10.1016/j.neuroscience.2011.04.005. [DOI] [PubMed] [Google Scholar]
- Yang N., Zhao Y., Wang Z., Liu Y., Zhang Y. Scutellarin suppresses growth and causes apoptosis of human colorectal cancer cells by regulating the p53 pathway. Mol. Med. Rep. 2017;15(2):929–935. doi: 10.3892/mmr.2016.6081. [DOI] [PubMed] [Google Scholar]
- Yu J.B., Shi J., Wang D., et al. Heme oxygenase-1/carbon monoxide regulated mitochondrial dynamic equilibrium contributes to the attenuation of endotoxin-induced acute lung injury in rats and in lipopolysaccharide-activated RAW264.7 cells. Anesthesiology. 2016;125:1190–1201. doi: 10.1097/ALN.0000000000001333. [DOI] [PubMed] [Google Scholar]
- Zhu L.L., Li M.Q., He F., Zhou S.B., Jiang W. Mitochondria targeted peptide attenuates mitochondrial dysfunction, controls inflammation and protects against spinal cord injury-induced lung injury. Cell. Physiol. Biochem. 2017;44(1):388–400. doi: 10.1159/000484919. [DOI] [PubMed] [Google Scholar]