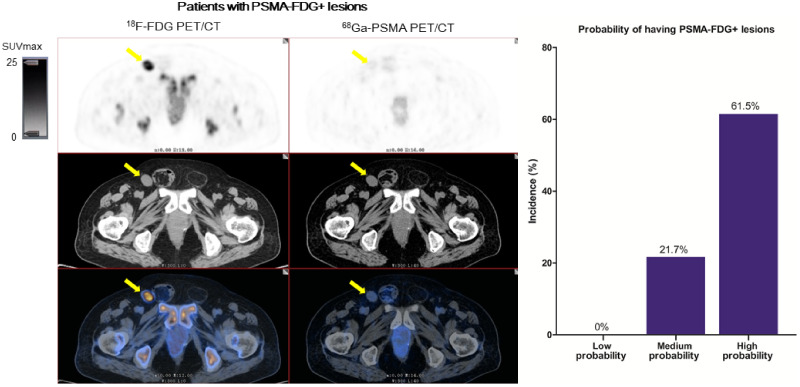
Visual Abstract
Keywords: 18F-FDG, 68Ga-PSMA, castration-resistant prostate cancer, negative PSMA
Abstract
68Ga-prostate-specific membrane antigen (68Ga-PSMA) PET/CT is a commonly used imaging modality in prostate cancers. However, few studies have compared the diagnostic efficiency between 68Ga-PSMA and 18F-FDG PET/CT and evaluated whether a heterogeneous metabolic phenotype (especially 68Ga-PSMA–negative [−], 18F-FDG–positive [+] lesions) exists in patients with castration-resistant prostate cancer (CRPC). We determined the added value of 18F-FDG PET/CT compared with 68Ga-PSMA PET/CT in CRPC patients and identified CRPC patients who may benefit from additional 18F-FDG PET/CT. Methods: The data of 56 patients with CRPC who underwent both 68Ga-PSMA and 18F-FDG PET/CT from May 2018 to February 2021 were retrospectively analyzed. The patients were classified into 2 groups: with or without 68Ga-PSMA−, 18F-FDG+ lesions. The differences in patient characteristics between the 2 groups and predictors of patients who have at least 1 68Ga-PSMA−, 18F-FDG+ lesion were analyzed. Results: Although both the detection rate (75.0% vs. 51.8%, P = 0.004) and the number of positive lesions (135 vs. 95) were higher for 68Ga-PSMA PET/CT than for 18F-FDG PET/CT, there were still 13 of 56 (23.2%) patients with at least 1 68Ga-PSMA−, 18F-FDG+ lesion. Prostate-specific antigen (PSA) and the Gleason score were both higher in patients with 68Ga-PSMA−, 18F-FDG+ lesions than in those without (P = 0.04 and P < 0.001, respectively). Multivariate regression analysis showed that the Gleason score (≥8) and PSA (≥7.9 ng/mL) were associated with the detection rate of patients who had 68Ga-PSMA−, 18F-FDG+ lesions (P = 0.01 and P = 0.04, respectively). The incidences of having 68Ga-PSMA−, 18F-FDG+ lesions in low-probability (Gleason score < 8 and PSA < 7.9 ng/mL), medium-probability (Gleason score ≥ 8 and PSA < 7.9 ng/mL or Gleason score < 8 and PSA ≥ 7.9 ng/mL), and high-probability (Gleason score ≥ 8 and PSA ≥ 7.9 ng/mL) groups were 0%, 21.7%, and 61.5%, respectively (P < 0.001). Conclusion: Gleason score and PSA are significant predictors of 68Ga-PSMA−, 18F-FDG+ lesions, and CRPC patients with a high Gleason score and PSA may benefit from additional 18F-FDG PET/CT.
Biochemical recurrence is a difficult problem after radical prostatectomy in prostate cancer (1). Androgen deprivation therapy (ADT) is the main treatment for biochemical recurrence. Despite inhibition of serum androgens, many patients develop castration-resistant prostate cancer (CRPC) and the tumor continues to grow, requiring multidrug therapy. Therefore, determining the location and degree of castration resistance is important to guide relevant treatment. However, the sensitivity of conventional imaging techniques such as MRI and CT is limited (2). The application of 68Ga-prostate-specific membrane antigen (PSMA) PET/CT can significantly improve imaging sensitivity in prostate cancer (3–6). Many studies have reported that the detection efficiency of 68Ga-PSMA PET is higher than that of conventional imaging methods (3,7).
At the same time, the effect of ADT on the detection efficiency of 68Ga-PSMA PET/CT has been controversial. Although some studies show that long-term use of ADT has no effect on 68Ga-PSMA PET/CT, Afshar-Oromieh et al. and Hoberuck et al. found that use of long-term ADT could reduce 68Ga-PSMA uptake and the visibility of tumor lesions in castration-sensitive prostate cancer (8,9). Weber et al. observed that 68Ga-PSMA PET/CT was positive in 75% of early-CRPC patients (10). These studies show that many CRPC patients might have negative 68Ga-PSMA PET/CT findings. Although 18F-FDG is not commonly used in prostate cancer because of its low diagnostic efficiency (11), Wang et al. (12) found that PSMA inhibition is associated with the upregulation of glucose transporter 1, which is positively associated with higher 18F-FDG uptake in CRPC. Several case reports have also shown that 18F-FDG PET/CT could be positive in those with negative 68Ga-PSMA PET/CT results (13,14). These findings have proven that 18F-FDG PET/CT could be complementary to 68Ga-PSMA PET/CT in partial-CRPC patients with the downregulation of PSMA expression. However, few studies have compared the diagnostic efficiency between 68Ga-PSMA PET/CT and 18F-FDG PET/CT in patients with CRPC.
Therefore, incorporating dual-tracer (68Ga-PSMA and 18F-FDG) PET/CT, we conducted this retrospective study to evaluate whether a heterogeneous metabolic phenotype (especially 68Ga-PSMA–negative [−], 18F-FDG–positive [+] disease) exists and whether incorporating 18F-FDG PET/CT with 68Ga-PSMA PET/CT has added value in the diagnosis of CRPC.
MATERIALS AND METHODS
Patients
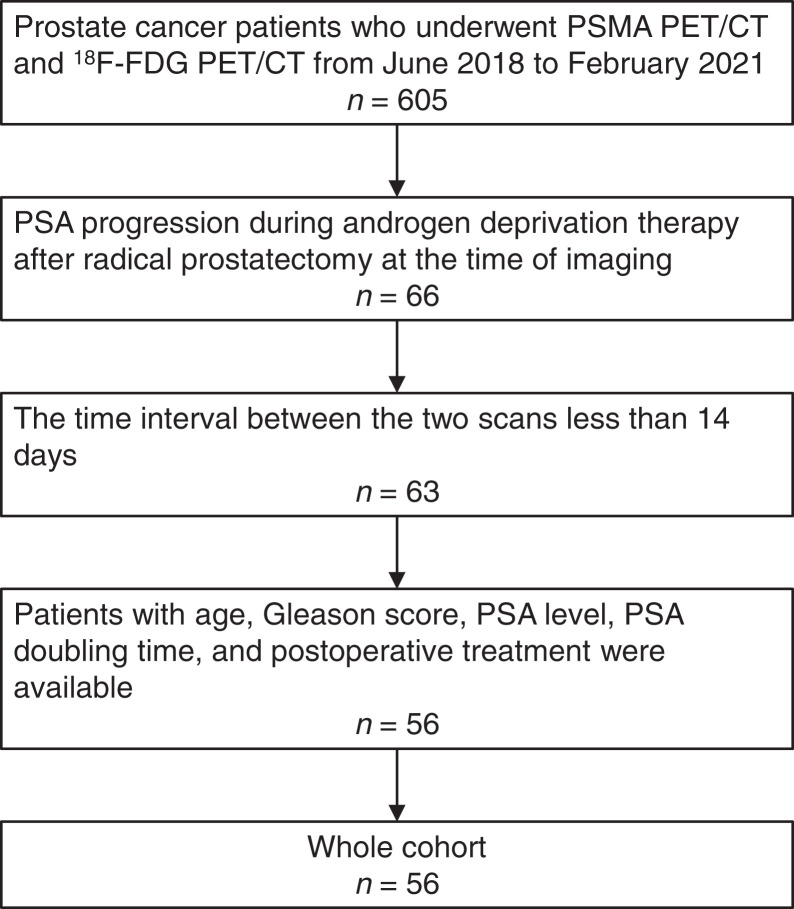
In Renji Hospital, 68Ga-PSMA and 18F-FDG PET/CT were routinely performed concomitantly in prostate cancer patients who were willing to undergo PET/CT. Fifty-six patients with CRPC were identified of 605 patients with prostate cancer who underwent both 68Ga-PSMA and 18F-FDG PET/CT from May 2018 to February 2021 (Fig. 1). Of these, 549 were excluded, and 56 patients were finally included in this study. Table 1 shows the patient characteristics. The inclusion criteria were patients who underwent radical prostatectomy, had histopathologically proven prostate cancer, showed prostate-specific antigen (PSA) progression during ADT therapy, underwent 68Ga-PSMA and 18F-FDG PET/CT at an interval of less than 14 d, and had available data on age, PSA, Gleason score, and ADT treatment. This retrospective study was approved by the Ethics Committee of Renji Hospital, and the requirement to obtain informed consent was waived.
FIGURE 1.
Patient recruitment flowchart.
TABLE 1.
Patient and Tumor Characteristics (n = 56)
| Characteristic | Data |
|---|---|
| Age (y) | |
| Mean ± SD | 69.6 ± 7.0 |
| Median | 70.0 (IQR, 63.0–75.0) |
| PSA doubling time (mo) | |
| Mean ± SD | 7.9 ± 6.4 |
| Median | 5.8 (IQR, 3.3–9.6) |
| PSA level (ng/mL) | |
| Mean ± SD | 12.5 ± 4.6 |
| Median | 5.0 (IQR, 1.5–14.5) |
| ADT duration time (mo) | |
| Mean ± SD | 25.5 ± 4.0 |
| Median | 16.5 (IQR, 8.0–33.0) |
| Two-scan interval (d) | |
| Mean ± SD | 7.3 ± 0.7 |
| Median | 7.0 (IQR, 1.0–12.0) |
| Gleason score | |
| 6 | 4 |
| 7 | 20 |
| 8 | 9 |
| 9 | 23 |
IQR = interquartile range.
68Ga-PSMA and 18F-FDG PET/CT
The patients fasted for 6 h before receiving the 18F-FDG injection, at a dose of 3.7 MBq/kg, and then remained quiet for 1 h before undergoing 18F-FDG PET/CT. The dose of 68Ga-PSMA was 1.85 MBq/kg, and the PSMA ligand was 68Ga-PSMA-11, which was synthesized as previously described (15). A PET/CT scan (Biograph mCT; Siemens) was obtained 50–60 min after injection of the 68Ga-PSMA. CT images (section thickness, 3 mm; automatic milliamp current, 120 kV) were obtained from the upper thigh to the skull. The PET scanning time for every bed position was 3 min. For better detection of local recurrence, forced diuresis with additional late imaging was performed to reduce retention of radioactive urine in the bladder. Additional late imaging was performed 1.5 h after early imaging. The patients received 20 mg of furosemide, drank at least 500 mL of water, and were asked to void frequently. Additional late imaging covered a range of 2 bed positions centered on the bladder.
Image Evaluation
Two nuclear medicine physicians with 8 and 12 y of PET/CT interpretation experience evaluated the images independently. When discrepancies occurred, they reached a consensus. In accord with interpretation guidelines (16–19), the experts evaluated the presence of positive lesions in local recurrence, lymph node metastasis, and distant metastasis. After excluding physiologic uptake and other important pitfalls, 68Ga-PSMA or 18F-FDG positivity was defined as focal avidity greater than the background mediastinal blood pool. Patients were considered to have positive PET/CT results if positive lesions were seen in cases of local recurrence, lymph node metastasis, or distant metastasis. PSA measurements, imaging examinations, and biopsies were used for follow-up. We used composite validation to verify these positive results.
Statistical Analysis
t testing or χ2 testing was used to evaluate the statistical significance of the correlation between clinicopathologic characteristics in patients with or without 68Ga-PSMA−, 18F-FDG+ lesions. Univariate and multivariate regression analyses were used to predict the detection rate of patients who had at least 1 68Ga-PSMA−, 18F-FDG+ lesion. All data were analyzed by SPSS (version 13.0; IBM).
RESULTS
Characteristics of Enrolled Patients with CRPC
The mean age was 70 y (interquartile range, 63–75 y), and the mean duration of ADT was 16.5 mo; 57.1% (32/56) of patients had a Gleason score of at least 8. The average PSA was 5.0 ng/mL (interquartile range, 1.5–14.5 ng/mL). The average interval between 68Ga-PSMA PET/CT and 18F-FDG PET/CT was 7.0 d (interquartile range, 1.0–12.0 d).
Detection Rate of 68Ga-PSMA PET/CT and 18F-FDG PET/CT
There was perfect agreement between the 2 nuclear medicine physicians for 68Ga-PSMA PET/CT and 18F-FDG PET/CT (both, κ = 0.97). 68Ga-PSMA PET/CT detected 135 lesions in 42 of the 56 patients, and 18F-FDG PET/CT detected 95 lesions in 29 of the 56 patients (Table 2). Overall, 169 lesions were detected in 48 of the 56 patients by 68Ga-PSMA PET/CT, 18F-FDG PET/CT, or both. The detection rate of CRPC per patient was significantly higher with 68Ga-PSMA PET/CT than with 18F-FDG PET/CT (75.0% vs. 51.8%, P = 0.004) (Table 2). Using a head-to-head comparison of PSMA uptake and 18F-FDG uptake, we were able to classify every lesion into the 68Ga-PSMA+, 18F-FDG+/− (68Ga-PSMA+, 18F-FDG+ or 68Ga-PSMA+, 18F-FDG−) group or the 68Ga-PSMA−, 18F-FDG+ group. Of the 169 lesions detected in 48 patients, 135 were 68Ga-PSMA+, 18F-FDG+/− and 34 lesions were 68Ga-PSMA−, 18F-FDG+. Among the 56 patients, 14.3% (8/56) patients had double-negative results, 62.5% (35/56) patients had only 68Ga-PSMA+, 18F-FDG+/− lesions, and 23.2% (13/56) patients had at least 1 68Ga-PSMA−, 18F-FDG+ lesion. Of the 8 patients with double-negative results, the mean PSA was 0.5 ± 0.3 ng/mL and the median Gleason score was 7 (range, 7–8). The representative images of 68Ga-PSMA−, 18F-FDG+ lesions and 68Ga-PSMA+, 18F-FDG− lesions are shown in Supplemental Figure 1 (supplemental materials are available at http://jnm.snmjournals.org).
TABLE 2.
Absolute Numbers and Percentages of Lesions Detected by 68Ga-PSMA or 18F-FDG PET/CT
| Tracer | |||
|---|---|---|---|
| Parameter | 68Ga-PSMA | 18F-FDG | P |
| Absolute numbers of positive lesions | 135 | 95 | 0.020 |
| Percentages of positive or negative findings | |||
| Positive findings | 42 (75%) | 29 (51.8%) | 0.004 |
| Negative findings | 14 (25%) | 27 (48.2%) | |
We verified the 68Ga-PSMA−, 18F-FDG+ lesions with composite validation (Table 3). Among the 13 CRPC patients with 68Ga-PSMA−, 18F-FDG+ lesions, 2 were verified by histopathology, 2 by decreasing PSA levels after radiotherapy, and 9 by imaging. All 68Ga-PSMA−, 18F-FDG+ lesions were verified as true-positive.
TABLE 3.
68Ga-PSMA−, 18F-FDG+ Lesion Validation
| Patients | Gleason score | PSA (ng/mL) | Lesions (n) | Lesions with 68Ga-PSMA−, 18F-FDG+ findings | Validated lesion | Validation method |
|---|---|---|---|---|---|---|
| 1 | 9 | 0.4 | 1 | Bone | Bone | Conventional imaging |
| 2 | 9 | 2.0 | 2 | Pelvic lymph nodes | Pelvic lymph nodes | Conventional imaging |
| 3 | 8 | 8.0 | 1 | Bone | Bone | Conventional imaging |
| 4 | 9 | 4.6 | 3 | Pelvic lymph nodes | Pelvic lymph nodes | Conventional imaging |
| 5 | 9 | 31.0 | 1 | Bone | Bone | Conventional imaging |
| 6 | 8 | 0.9 | 1 | Bone | Bone | Conventional imaging |
| 7 | 9 | 86.0 | 2 | Pelvic lymph nodes | Pelvic lymph nodes | Conventional imaging |
| 8 | 9 | 8.9 | 7 | Pelvic lymph nodes, bone | Pelvic lymph nodes | 18F-FDG PET/CT |
| 9 | 9 | 18.4 | 1 | Extrapelvic lymph nodes | Extrapelvic lymph nodes | 18F-FDG PET/CT |
| 10 | 8 | 2.1 | 1 | Pelvic lymph nodes | Pelvic lymph nodes | PSA response after SBRT |
| 11 | 9 | 84.7 | 2 | Pelvic lymph nodes | Pelvic lymph nodes | PSA response after SBRT |
| 12 | 8 | 8.0 | 10 | Local recurrence, pelvic lymph nodes, inguinal lymph nodes, bone | Inguinal lymph nodes | Pathologic confirmation |
| 13 | 9 | 15.9 | 2 | Pelvic lymph nodes and bone | Bone | Pathologic confirmation |
Conventional imaging = CT, MRI, or bone scintigraphy; SBRT = stereotactic body radiotherapy.
The Relationship Between Clinical Characteristics and Patients With or Without 68Ga-PSMA−, 18F-FDG+ Lesions
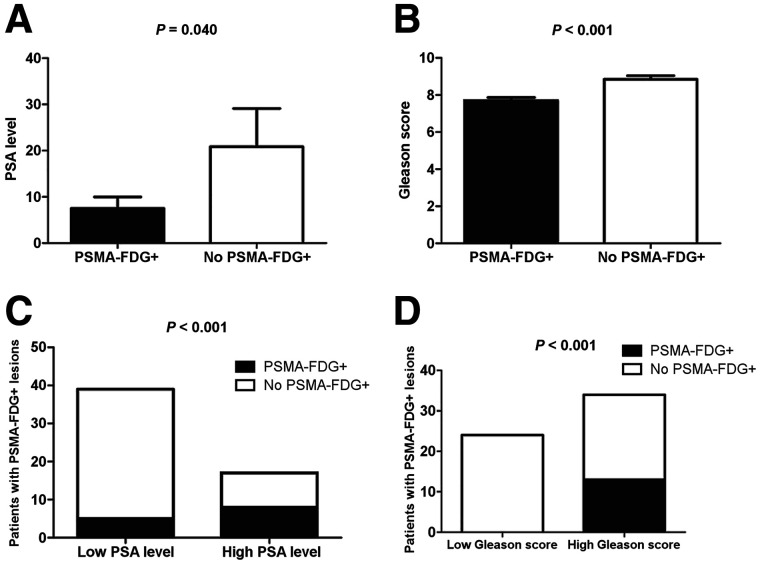
Although both the detection rate (75.0% vs. 51.8%, P = 0.004) and the number of positive lesions (135 vs. 95) for 68Ga-PSMA PET/CT were higher than those for 18F-FDG PET/CT, there were still 13 of 56 (23.2%) patients with at least 1 68Ga-PSMA−, 18F-FDG+ lesion. We then divided patients into 2 groups according to those with at least 1 68Ga-PSMA−, 18F-FDG+ lesions (n = 13) and those without (n = 43). Table 4 describes the association between clinical characteristics and patients with or without 68Ga-PSMA−, 18F-FDG+ lesions. There were no significant differences in age, scan interval, PSA doubling time, or ADT duration between the 2 groups. However, there were significant differences in PSA and Gleason score between the 2 groups. Namely, patients with 68Ga-PSMA−, 18F-FDG+ lesions had a higher PSA than those without (20.8 ± 8.3 vs. 7.5 ± 2.5 ng/mL; P = 0.04; Fig. 2A), and patients with 68Ga-PSMA−, 18F-FDG+ lesions had a higher Gleason score than those without (9 [range, 8–9] vs. 7 [range, 7–9]; P < 0.001; Fig. 2B).
TABLE 4.
Relationship Between Clinicopathologic Characteristics and Patients With or Without 68Ga-PSMA−, 18F-FDG+ Lesions
| 68Ga-PSMA−, 18F-FDG+ lesions | ||||
|---|---|---|---|---|
| Variable | Total (n) | No | Yes | P |
| Age (y) | ||||
| <70 | 31 | 23 | 8 | 0.609 |
| ≥70 | 25 | 20 | 5 | |
| PSA doubling time (mo) | ||||
| <6 | 29 | 22 | 7 | 0.203 |
| ≥6 | 27 | 24 | 3 | |
| PSA level (ng/mL) | 56 | 7.5 ± 2.5 | 20.8 ± 8.3 | 0.040 |
| ADT duration time (mo) | 56 | 16.8 ± 2.5 | 28.0 ± 5.1 | 0.238 |
| Two-scan interval (d) | 56 | 7.4 ± 0.8 | 6.9 ± 2.0 | 0.806 |
| Median Gleason score | 56 | 7 (IQR, 7–9) | 9 (IQR, 8–9) | <0.001 |
IQR = interquartile range.
FIGURE 2.
Analysis of 68Ga-PSMA−, 18F-FDG+ lesions according to PSA and Gleason score. (A) Patients with 68Ga-PSMA−, 18F-FDG+ lesions had higher PSA than patients without 68Ga-PSMA−, 18F-FDG+ lesions (20.8 ± 8.3 vs. 7.5 ± 2.5 ng/mL; P = 0.04). (B) Patients with 68Ga-PSMA−, 18F-FDG+ lesions had higher Gleason score than those without 68Ga-PSMA−, 18F-FDG+ lesions (8.8 ± 0.2 vs. 7.7 ± 0.2; P < 0.001). (C) Detection rate for patients with 68Ga-PSMA−, 18F-FDG+ lesions was higher in high-PSA than low-PSA group (12.8% vs. 47.1%, respectively; P < 0.001). (D) Detection rate for patients with 68Ga-PSMA−, 18F-FDG+ lesion was higher in high–Gleason score group than low–Gleason score group (38.2% vs. 0%, respectively; P < 0.001).
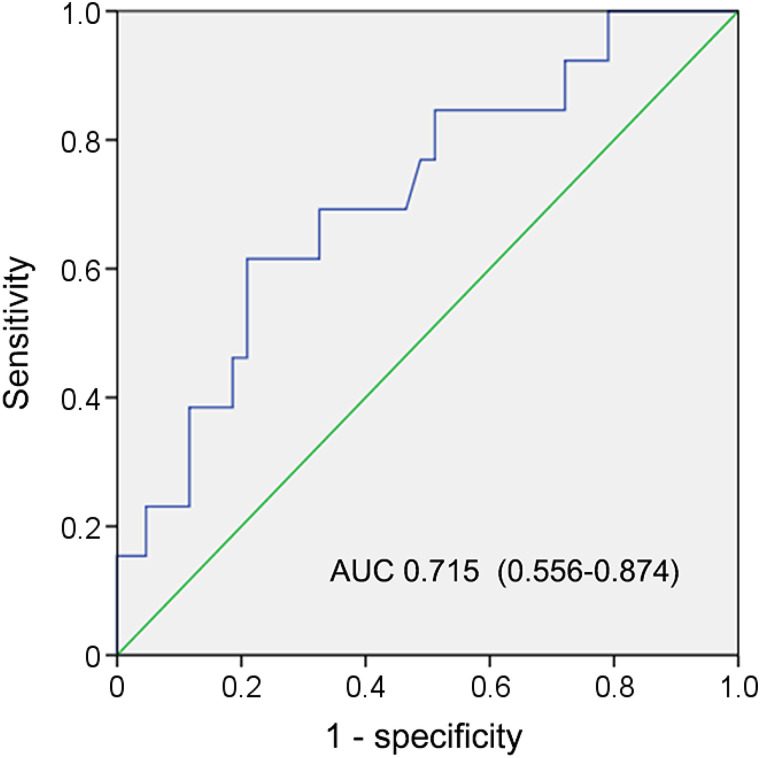
We then determined the optimal PSA threshold to predict at least 1 68Ga-PSMA−, 18F-FDG+ lesion in patients by using receiver-operating-characteristic curve analysis. The highest accuracy (75.0%) was obtained when PSA was 7.9 ng/mL (Fig. 3). We classified patients into low- or high-PSA groups (i.e., PSA < 7.9 vs. PSA ≥ 7.9 ng/mL). The detection rate of patients with 68Ga-PSMA−, 18F-FDG+ lesions was higher in the high-PSA group than in the low-PSA group (12.8% vs. 47.1%, respectively; P < 0.001; Fig. 2C).
FIGURE 3.
PSA for predicting 68Ga-PSMA−, 18F-FDG+ lesions in CRPC patients. Area under curve (AUC) was 0.715 (95% CI, 0.556–0.874; P = 0.02), and PSA of 7.9 ng/mL was determined for predicting patients with at least 1 68Ga-PSMA−, 18F-FDG+ lesion. Sensitivity and specificity for predicting at least 1 68Ga-PSMA−, 18F-FDG+ lesion were 61.5% (8/13) and 79.1% (34/43), respectively.
We also found that Gleason score was positively associated with patients with 68Ga-PSMA−, 18F-FDG+ lesions (P = 0.006). The detection rates of patients having 68Ga-PSMA−, 18F-FDG+ lesions with Gleason scores of 6, 7, 8, and 9 were 0%, 0%, 33.3%, and 43.5%, respectively. We classified patients into groups with a low or high Gleason score (Gleason score < 8 vs. ≥ 8). The detection rate of patients with 68Ga-PSMA−, 18F-FDG+ lesion was higher in the high group than in the low group (38.2% vs. 0%, respectively; P < 0.001; Fig. 2D).
Predictors of Patients with at Least One 68Ga-PSMA−, 18F-FDG+ Lesion
A multivariate regression analysis showed that PSA (odds ratio, 4.7; 95% CI, 1.1–20.8; P = 0.04) and Gleason score (odds ratio, 3.2; 95% CI, 1.3–7.7; P = 0.01) were significant predictors of patients with 68Ga-PSMA−, 18F-FDG+ lesions (Table 5). According to Gleason score and PSA, we classified patients for their probability of having 68Ga-PSMA−, 18F-FDG+ lesions: a low-probability group (low Gleason score and low PSA), a medium-probability group (low Gleason score and high PSA, or high Gleason score and low PSA), and a high-probability group (high Gleason score and high PSA). The incidences of patients with 68Ga-PSMA−, 18F-FDG+ lesions in the low-, medium-, and high-probability groups were 0%, 21.7%, and 61.5%, respectively (P < 0.001).
TABLE 5.
Univariate and Multivariate Regression to Predict 68Ga-PSMA−, 18F-FDG+ Lesions
| Univariate logistic regression | Multivariate logistic regression | |||
|---|---|---|---|---|
| Variable and intercept | OR | P | OR | P |
| Age (y) | 1.333 (0.396–4.484) | 0.642 | NA | NA |
| PSA doubling time (≥6 mo vs. <6 mo) | 0.721 (0.345–1.507) | 0.416 | NA | NA |
| PSA level (high vs. low) | 6.0 (1.6–23.0) | 0.008 | 4.7 (1.1–20.8) | 0.04 |
| ADT duration time | 0.985 (0.952–1.019) | 0.375 | NA | NA |
| Two-scan interval (d) | 0.986 (0.883–1.101) | 0.802 | NA | NA |
| Gleason score (high vs. low) | 3.3 (1.5–7.5) | 0.004 | 3.2 (1.3–7.7) | 0.01 |
NA = not available; these variables were eliminated in multivariate logistic regression model, so odds ratio and P values were not available.
Data in parentheses are 95% CIs.
Added Value of Staging by Incorporating 18F-FDG PET/CT with 68Ga-PSMA PET/CT
For staging (Table 6), the addition of 18F-FDG PET/CT could increase the detection rate of local recurrence, lymph node metastasis, distant metastasis, and any location from 14.3% to 19.6%, from 42.9% to 55.4%, from 35.7% to 39.3%, and from 75.0% to 85.7%, respectively, when compared with 68Ga-PSMA PET/CT alone. When patients were in a low-probability group, the addition of 18F-FDG PET/CT could not increase the detection rate regardless of local recurrence, lymph node metastasis, distant metastasis, or any location, compared with 68Ga-PSMA PET/CT alone. However, when patients were in a high-probability group, the addition of 18F-FDG PET/CT could increase the detection rate of local recurrence, lymph node metastasis, distant metastasis, and any location from 0% to 7.7%, from 30.8% to 61.5%, from 53.8% to 61.5%, and from 69.2% to 100%, respectively, when compared with 68Ga-PSMA PET/CT alone.
TABLE 6.
Detection Rate of Local Recurrence, Lymph Node Metastasis, and Distant Metastasis by 68Ga-PSMA PET/CT and 18F-FDG PET/CT
| 68Ga-PSMA PET/CT | 18F-FDG PET/CT | 68Ga-PSMA and 18F-FDG PET/CT | ||||||
|---|---|---|---|---|---|---|---|---|
| TNM stage | Patients (n) | Location | n | % | n | % | n | % |
| Total | 56 | Local recurrence | 8 | 14.3% | 5 | 8.9% | 11 | 19.6% |
| Lymph node metastasis | 24 | 42.9% | 14 | 25.0% | 31 | 55.4% | ||
| Distant metastasis | 20 | 35.7% | 17 | 30.4% | 22 | 39.3% | ||
| Any location | 42 | 75.0% | 29 | 51.8% | 48 | 85.7% | ||
| Low risk | 20 | Local recurrence | 3 | 15.0% | 1 | 5.0% | 3 | 15.0% |
| Lymph node metastasis | 11 | 55.0% | 2 | 10.0% | 11 | 55.0% | ||
| Distant metastasis | 6 | 30.0% | 1 | 5.0% | 6 | 30.0% | ||
| Any location | 16 | 80.0% | 4 | 20.0% | 16 | 80.0% | ||
| Moderate risk | 23 | Local recurrence | 5 | 21.7% | 3 | 13.0% | 7 | 30.4% |
| Lymph node metastasis | 9 | 39.1% | 6 | 26.1% | 12 | 52.2% | ||
| Distant metastasis | 7 | 30.4% | 7 | 30.4% | 8 | 34.8% | ||
| Any location | 17 | 73.9% | 14 | 60.9% | 19 | 82.6% | ||
| High risk | 13 | Local recurrence | 0 | 0.0% | 1 | 7.7% | 1 | 7.7% |
| Lymph node metastasis | 4 | 30.8% | 6 | 46.2% | 8 | 61.5% | ||
| Distant metastasis | 7 | 53.8% | 8 | 61.5% | 8 | 61.5% | ||
| Any location | 9 | 69.2% | 11 | 84.6% | 13 | 100% | ||
DISCUSSION
68Ga-PSMA PET/CT has been widely used in the diagnosis and biochemical recurrence of prostate cancer. However, partial CRPC might have negative 68Ga-PSMA PET/CT findings (10,20). For these patients, 18F-FDG PET/CT may be a compensatory diagnostic method. Combined 68Ga-PSMA and 18F-FDG scanning has been reported in the later phase of CRPC (21). 18F-FDG positivity has been used as an exclusion criterion before radioligand therapy or as an indicator of a poorer outcome after radioligand therapy (21). However, the patients scheduled for radioligand therapy are examined at a later stage of their carcinoma disease. In our study, using dual-tracer PET/CT (68Ga-PSMA and 18F-FDG), we retrospectively compared diagnostic efficiency between 68Ga-PSMA PET/CT and 18F-FDG PET/CT and evaluated the metabolic heterogeneity in patients who have just developed castration resistance. Although 68Ga-PSMA PET/CT showed both a higher detection rate and a higher number of positive lesions than 18F-FDG PET/CT, we also identified patients with 68Ga-PSMA−, 18F-FDG+ lesions, and incorporating 18F-FDG PET/CT with 68Ga-PSMA PET/CT had added value in the case of partial-CRPC patients, especially in those with a high PSA and a high Gleason score.
Although both the detection rate (75.0% vs. 51.8%, P = 0.004) and the number of positive lesions (135 vs. 95) were higher for 68Ga-PSMA PET/CT than for 18F-FDG PET/CT, there were still 13 of 56 (23.2%) patients with at least 1 68Ga-PSMA−, 18F-FDG+ lesions. We identified 34 68Ga-PSMA−, 18F-FDG+ lesions from 13 patients. The addition of 18F-FDG PET/CT could increase the detection rate of local recurrence, lymph node metastasis, distant metastasis, and any location from 14.3% to 19.6%, from 42.9% to 55.4%, from 35.7% to 39.3%, and from 75.0% to 85.7%, respectively, compared with 68Ga-PSMA PET/CT alone. These findings show that treatment could accordingly be administered for the 68Ga-PSMA−, 18F-FDG+ lesions. For locally recurrent or solitary bone metastases, salvage radiotherapy (22) may be feasible for the 68Ga-PSMA−, 18F-FDG+ lesions. For recurrent lymph node metastases, salvage radiotherapy or salvage node dissection (23) could be used for these 68Ga-PSMA−, 18F-FDG+ lesions. For patients with multiple lesions involved, systemic therapy may be the most appropriate treatment. However, only 23.2% of patients had at least 1 68Ga-PSMA−, 18F-FDG+ lesion. Therefore, identification of the most appropriate patients for 18F-FDG PET/CT is essential to optimize its use and avoid expensive and possibly unnecessary staging in low-risk patients.
We studied the correlation between clinicopathologic characteristics and the presence or absence of 68Ga-PSMA−, 18F-FDG+ lesions and found an excellent correlation between PSA and 68Ga-PSMA−, 18F-FDG+ lesions. Patients with 68Ga-PSMA−, 18F-FDG+ lesions had a higher PSA than did patients without. The receiver-operating-characteristic analysis suggested that PSA could be used to predict 68Ga-PSMA−, 18F-FDG+ lesions. Previous results have shown that the rate of 18F-FDG PET/CT positivity was associated with PSA level in the diagnosis of prostate cancer (11). Our study further demonstrated a positive correlation between PSA and detection rate in patients with 68Ga-PSMA−, 18F-FDG+ lesions. Aside from PSA, we also found that there was a good association between Gleason score and detection rate in patients with 68Ga-PSMA−, 18F-FDG+ lesions. The group with a high Gleason score had a significantly higher detection rate of 68Ga-PSMA−, 18F-FDG+ lesions than did the group with a low score in CRPC. Previous studies have shown that 18F-FDG PET/CT has diagnostic value in prostate cancer with a high Gleason score (11,24). Consistent with these previous results, our results further suggest that Gleason score is an important predictor of patients with 68Ga-PSMA−, 18F-FDG+ lesions. Our previous study (25) showed that 18F-FDG PET/CT has added value in patients with biochemical recurrence, who were different from the patients in this study, which included CRPC patients. In addition, the PSA level in 68Ga-PSMA−, 18F-FDG+ patients tended to be lower in CRPC patients than in patients with biochemically recurrent prostate cancer (25). This finding may be because the PSA level in patients with 68Ga-PSMA−, 18F-FDG+ lesions was affected in the initial stage of ADT treatment, though these patients later developed resistance.
Although 18F-FDG PET/CT has potential value in CRPC, its value-added mechanism is unclear. Bakht et al. (26) found that PSMA inhibition is associated with upregulation of glucose uptake–related genes and is positively associated with higher 18F-FDG uptake in PSMA-inhibited prostate cancer. Therefore, the added value of 18F-FDG could be attributed to the amplification of glucose uptake–related genes after long-time ADT.
We classified CRPC patients according to their incidence of having 68Ga-PSMA−, 18F-FDG+ lesions, based on Gleason score and PSA, into low-, medium-, and high-probability groups. The probability that patients would have 68Ga-PSMA−, 18F-FDG+ lesions was 0% in the low-probability group but 61.5% in the high-probability group. When patients were in a low-probability group, the addition of 18F-FDG PET/CT could not increase the detection rate at any location, when compared with 68Ga-PSMA PET/CT alone. However, when patients were in a high-probability group, the addition of 18F-FDG PET/CT could increase the detection rate of local recurrence, lymph node metastasis, distant metastasis, and any location from 0% to 7.7%, from 30.8% to 61.5%, from 53.8% to 61.5%, and from 69.2% to 100%, respectively, compared with 68Ga-PSMA PET/CT alone. These results suggest that 18F-FDG PET/CT is not appropriate for those with a low probability of having 68Ga-PSMA−, 18F-FDG+ lesions but is feasible for those with a high probability of having 68Ga-PSMA−, 18F-FDG+ lesions. For those with a low probability of having 68Ga-PSMA−, 18F-FDG+ lesions, other imaging tracers should be further explored.
The current study had some limitations, including its retrospective design and small sample size. The fact that the number of patients with 68Ga-PSMA−, 18F-FDG+ lesions was small may reduce the power of the multivariate regression analysis. In addition, though we found that when PSA 7.9 ng/mL was used as the threshold, PSA could predict 68Ga-PSMA−, 18F-FDG+ lesions, the area under the curve was not particular high. Therefore, the association between PSA level and patients with 68Ga-PSMA−, 18F-FDG+ lesion should be interpreted carefully, and prospective studies with a larger sample size are needed to further validate our findings. Furthermore, the lower detection rate of 68Ga-PSMA PET/CT in our study (75.0%) than in other studies (27) should also be interpreted carefully. The dose of PSMA-11 in our study was on the lower end of the recommended spectrum (18) and may have influenced the sensitivity.
CONCLUSION
Our study assessed the added value of 18F-FDG PET/CT compared with 68Ga-PSMA PET/CT in CRPC patients. We found that 18F-FDG PET/CT has additional value in 23.2% of CRPC patients, and Gleason score and PSA were significant predictors of 68Ga-PSMA−, 18F-FDG+ lesions. CRPC patients with a high Gleason score (≥8) and a high PSA (≥7.9 ng/mL) may benefit from 18F-FDG PET/CT.
DISCLOSURE
This work was supported by the National Natural Science Foundation of China (grant 81701724). No other potential conflict of interest relevant to this article was reported.
KEY POINTS.
QUESTION: Does a heterogeneous metabolic phenotype (especially 68Ga-PSMA−, 18F-FDG+ lesions) exist in patients with CRPC, and does 18F-FDG PET/CT have added value compared with 68Ga-PSMA PET/CT in CRPC patient?
PERTINENT FINDINGS: Although both the detection rate and the number of positive lesions were higher for 68Ga-PSMA PET/CT than for 18F-FDG PET/CT, there were still 13 of 56 patients with at least 1 68Ga-PSMA−, 18F-FDG+ lesion. The incidences of having 68Ga-PSMA−, 18F-FDG+ lesions in low-, medium-, and high-probability groups were 0%, 21.7%, and 61.5%, respectively (P < 0.001).
IMPLICATIONS FOR PATIENT CARE: Gleason score and PSA are significant predictors of 68Ga-PSMA−, 18F-FDG+ lesions, and CRPC patients with a high Gleason score and a high PSA may benefit from additional 18F-FDG PET/CT.
REFERENCES
- 1. Boorjian SA, Eastham JA, Graefen M, et al.A critical analysis of the long-term impact of radical prostatectomy on cancer control and function outcomes. Eur Urol. 2012;61:664–675. [DOI] [PubMed] [Google Scholar]
- 2. Briganti A, Abdollah F, Nini A, et al.Performance characteristics of computed tomography in detecting lymph node metastases in contemporary patients with prostate cancer treated with extended pelvic lymph node dissection. Eur Urol. 2012;61:1132–1138. [DOI] [PubMed] [Google Scholar]
- 3. Perera M, Papa N, Roberts M, et al.Gallium-68 prostate-specific membrane antigen positron emission tomography in advanced prostate cancer: updated diagnostic utility, sensitivity, specificity, and distribution of prostate-specific membrane antigen-avid lesions—a systematic review and meta-analysis. Eur Urol. 2020;77:403–417. [DOI] [PubMed] [Google Scholar]
- 4. Sprute K, Kramer V, Koerber S, et al.Diagnostic accuracy of 18F-PSMA-1007-PET/CT imaging for lymph node staging of prostate carcinoma in primary and biochemical recurrence. J Nucl Med. 2021;62:208–213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Tan N, Oyoyo U, Bavadian N, et al.PSMA-targeted radiotracers versus 18F fluciclovine for the detection of prostate cancer biochemical recurrence after definitive therapy: a systematic review and meta-analysis. Radiology. 2020;296:44–55. [DOI] [PubMed] [Google Scholar]
- 6. Fendler WP, Ferdinandus J, Czernin J, et al.Impact of 68Ga-PSMA-11 PET on the management of recurrent prostate cancer in a prospective single-arm clinical trial. J Nucl Med. 2020;61:1793–1799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Afshar-Oromieh A, Zechmann CM, Malcher A, et al.Comparison of PET imaging with a 68Ga-labelled PSMA ligand and 18F-choline-based PET/CT for the diagnosis of recurrent prostate cancer. Eur J Nucl Med Mol Imaging. 2014;41:11–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Afshar-Oromieh A, Debus N, Uhrig M, et al.Impact of long-term androgen deprivation therapy on PSMA ligand PET/CT in patients with castration-sensitive prostate cancer. Eur J Nucl Med Mol Imaging. 2018;45:2045–2054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Hoberuck S, Lock S, Winzer R, et al.[68Ga]Ga-PSMA-11 PET before and after initial long-term androgen deprivation in patients with newly diagnosed prostate cancer: a retrospective single-center study. EJNMMI Res. 2020;10:135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Weber M, Kurek C, Barbato F, et al.PSMA-ligand PET for early castration-resistant prostate cancer: a retrospective single-center study. J Nucl Med. 2021;62:88–91. [DOI] [PubMed] [Google Scholar]
- 11. Jadvar H. Imaging evaluation of prostate cancer with 18F-fluorodeoxyglucose PET/CT: utility and limitations. Eur J Nucl Med Mol Imaging. 2013;40(suppl 1):S5–S10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Wang J, Xu W, Wang B, et al.GLUT1 is an AR target contributing to tumor growth and glycolysis in castration-resistant and enzalutamide-resistant prostate cancers. Cancer Lett. 2020;485:45–55. [DOI] [PubMed] [Google Scholar]
- 13. Perez PM, Hope TA, Behr SC, van Zante A, Small EJ, Flavell RR. Intertumoral heterogeneity of 18F-FDG and 68Ga-PSMA uptake in prostate cancer pulmonary metastases. Clin Nucl Med. 2019;44:e28–e32. [DOI] [PubMed] [Google Scholar]
- 14. Parida GK, Tripathy S, Datta Gupta S, et al.Adenocarcinoma prostate with neuroendocrine differentiation: potential utility of 18F-FDG PET/CT and 68Ga-DOTANOC PET/CT over 68Ga-PSMA PET/CT. Clin Nucl Med. 2018;43:248–249. [DOI] [PubMed] [Google Scholar]
- 15. Demirci E, Sahin OE, Ocak M, Akovali B, Nematyazar J, Kabasakal L. Normal distribution pattern and physiological variants of 68Ga-PSMA-11 PET/CT imaging. Nucl Med Commun. 2016;37:1169–1179. [DOI] [PubMed] [Google Scholar]
- 16. Eiber M, Herrmann K, Calais J, et al.Prostate cancer molecular imaging standardized evaluation (PROMISE): proposed miTNM classification for the interpretation of PSMA-ligand PET/CT. J Nucl Med. 2018;59:469–478. [DOI] [PubMed] [Google Scholar]
- 17. Hofman MS, Hicks RJ, Maurer T, Eiber M. Prostate-specific membrane antigen PET: clinical utility in prostate cancer, normal patterns, pearls, and pitfalls. Radiographics. 2018;38:200–217. [DOI] [PubMed] [Google Scholar]
- 18. Fendler WP, Eiber M, Beheshti M, et al. 68Ga-PSMA PET/CT: joint EANM and SNMMI procedure guideline for prostate cancer imaging: version 1.0. Eur J Nucl Med Mol Imaging. 2017;44:1014–1024. [DOI] [PubMed] [Google Scholar]
- 19. Clark MS, Packard AT, Johnson DR, Johnson GB. Pitfalls of a mixed metabolic response at PET/CT. Radiographics. 2019;39:1461–1475. [DOI] [PubMed] [Google Scholar]
- 20. Weber M, Hadaschik B, Ferdinandus J, et al.Prostate-specific membrane antigen-based imaging of castration-resistant prostate cancer. Eur Urol Focus. 2021;7:279–287. [DOI] [PubMed] [Google Scholar]
- 21. Michalski K, Ruf J, Goetz C, et al.Prognostic implications of dual tracer PET/CT: PSMA ligand and [18F]FDG PET/CT in patients undergoing [177Lu] PSMA radioligand therapy. Eur J Nucl Med Mol Imaging. 2021;48: 2024–2030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Schmidt-Hegemann NS, Stief C, Kim TH, et al.Outcome after PSMA PET/CT based salvage radiotherapy in patients with biochemical recurrence after radical prostatectomy: a bi-institutional retrospective analysis. J Nucl Med. 2019;60:227–233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Jilg CA, Drendel V, Rischke HC, et al.Diagnostic accuracy of Ga-68-HBED-CC-PSMA-ligand-PET/CT before salvage lymph node dissection for recurrent prostate cancer. Theranostics. 2017;7:1770–1780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Oztürk H, Karapolat I. 18F-fluorodeoxyglucose PET/CT for detection of disease in patients with prostate-specific antigen relapse following radical treatment of a local-stage prostate cancer. Oncol Lett. 2016;11:316–322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Chen R, Wang Y, Shi Y, et al.Diagnostic value of 18F-FDG PET/CT in patients with biochemical recurrent prostate cancer and negative 68Ga-PSMA PET/CT. Eur J Nucl Med Mol Imaging. 2021;48:2970–2977. [DOI] [PubMed] [Google Scholar]
- 26. Bakht MK, Lovnicki JM, Tubman J, et al.Differential expression of glucose transporters and hexokinases in prostate cancer with a neuroendocrine gene signature: a mechanistic perspective for 18F-FDG imaging of PSMA-suppressed tumors. J Nucl Med. 2020;61:904–910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Fendler WP, Calais J, Eiber M, et al.Assessment of 68Ga-PSMA-11 PET accuracy in localizing recurrent prostate cancer: a prospective single-arm clinical trial. JAMA Oncol. 2019;5:856–863. [DOI] [PMC free article] [PubMed] [Google Scholar]