Abstract
Multivalent glycosylated materials (polymers, surfaces, and particles) often show high affinity toward carbohydrate binding proteins (e.g., lectins) due to the nonlinear enhancement from the cluster glycoside effect. This affinity gain has potential in applications from diagnostics, biosensors, and targeted delivery to anti-infectives and in an understanding of basic glycobiology. This perspective highlights the question of selectivity, which is less often addressed due to the reductionist nature of glycomaterials and the promiscuity of many lectins. The use of macromolecular features, including architecture, heterogeneous ligand display, and the installation of non-natural glycans, to address this challenge is discussed, and examples of selectivity gains are given.
Keywords: glycopolymers, multivalency, nanoparticles, carbohydrates, chemical glycobiology
Carbohydrates are diverse (macro)molecules that coat cell surfaces and lipids (and even RNA1) and are present on >50% of human proteins, fulfilling functions including recognition, signal transduction, and fertilization and as sites for pathogen invasion.2,3 The huge structural diversity of glycans arises from the assembly of monosaccharides via different glycoside linkages, at different ring positions and with specific stereochemistry, resulting in the inherent complexity of the glycome.4 Proteins that interact or “read”5 carbohydrates include enzymes, anticarbohydrate antibodies, adhesins, and lectins.6 Hence, the development of probes, binders and inhibitors of carbohydrate-binding proteins has broad biotechnological and biomedical value. For material scientists, the motivation to incorporate glycans is to mimic their multivalent presentation found on cell surfaces. The actual binding affinity of a carbohydrate to its target lectin is typically weak (Kd = 10–3–10–6 M) in comparison to antibody–antigen interactions, which can be <10–9 M. The presentation of multiple copies of the target carbohydrate on the cell surface gives rise to an increase in affinity greater than that of the linear sum of the individual sugars; this is known as the “cluster glycoside effect”.7−9 In short, polymers and particles bearing glycans can show affinity higher than that of a single “small molecule” of equal concentration, a concept that has been established now for around 40 years. In 1983, Lee et al.7 synthesized a series of oligosaccharides, based on N-acetyllactosamine-type glycans, and demonstrated their ability to inhibit the mammalian hepatic lectin binding to rabbit hepatocytes. This revealed inhibitory potency in the order tetraantennary > trianntenary ≫ biantennary ≫ monoantenneary, increasing from 1 mM to 1 nM, while only increasing the glycan concentration 3-fold. In 1996 Whitesides and co-workers showed that sialic acid–functional polyacrylamides could prevent influenza from agglutinating (i.e., stopping binding) erythrocytes, demonstrating the anti-infective potential of polymeric glycan mimetics.10 Kiessling showed nonlinear increases in affinity of well-defined ring opening metathesis polymerization (ROMP) derived mannosylated polymers toward Con A as a function of chain length.11 Of course, there are examples of medicinal chemistry approaches for small-molecule affinity, selectivity, and PK/PD profiles such as those developed for FimH inhibition.12,13 However, these are beyond the scope of this Perspective, which will focus on multivalent systems.
These (selected) early examples show the clear benefit of multivalent assemblies, which provide advantages over monovalent assemblies.14 Multivalency enables spanning of multiple binding sites (on the same or different lectins), chelation, subsite binding, clustering, and statistical rebinding among others, and the mechanisms of these have been reviewed extensively.3,15,16 A vast range of multivalent architectures are known, which will not be reviewed in this Perspective but include dendrimers,17−25 peptides,26,27 polymers,28−33 particles,34−37 viruses,38 and presentations designed to specifically interact with the binding sites.39,40 Multivalent inhibitors for antiadhesion have also been covered previously,16,24,41−48 and this Perspective does not aim to re-review these.
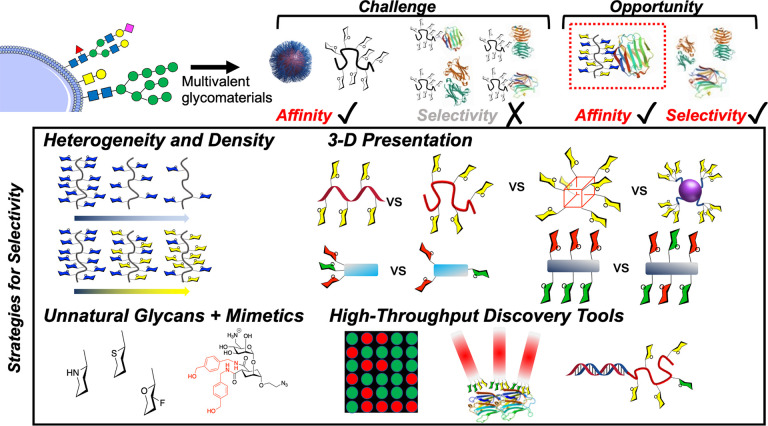
This Perspective aims to highlight potential macromolecular solutions to engineer selectivity into glycomaterials. Multivalent presentation almost always leads to an increase in affinity, but there is an exciting opportunity to develop macromolecular tools to increase selectivity. Figure 1 summarizes this challenge, and the approaches which are covered in this perspective will include glycan heterogeneity, control of 3D presentation, and the use of unnatural glycans. We also cover some emerging discovery approaches for the identification of selective binders.
Figure 1.
Scope of the Perspective on moving from high affinity to high affinity and high selectivity glycomaterials. The lower panel schematic shows strategies that are discussed here.
Diversity of Interactions and Promiscuity
In any applications spanning delivery, sensing, or inhibition, ensuring selectivity is essential: not against all lectins (or e.g. antibodies), but against those likely to be in competition in the same environment. For example, respiratory viruses, including SARS-CoV-2, have affinities toward sialic acids,49 as does influenza,50 and hence any glycan-based sensor would require a strategy for selectivity. The selective targeting of DC-SIGN over other C-type lectins present on dendritic cells presents a challenge in glycomaterial design for therapy or immune modulation.51 Blocking DC-SIGN can reduce HIV viral infection, but another lectin, Langerin, is implicated in clearing viral particles.52 Both DC-SIGN and Langerin bind mannosylated glycans (with subtle differences in their profile53), and hence achieving selectivity between these two C-type lectins would be essential if a glycomaterial were to be used. Furthermore, mannosylated polymers can also activate the complement pathway, which may limit their translation.54 To highlight the diversity of glycan interactions, Figure 2 shows data from the Consortium for Functional Glycomics (CFG) glycan array55 versus DC-SIGN. Any one lectin can bind multiple different glycans, in this case including not just high mannose but also fucoyslated glycans. The branching pattern of the glycans also affects the observed extent of binding.
Figure 2.
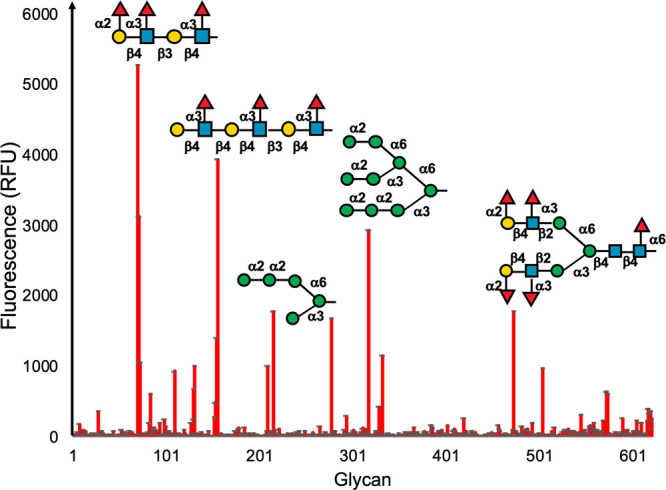
Extract of glycan-array data for DC-SIGN from the Consortium for Functional Glycomics (primscreen_5273, Human DC-SIGN-AF488 200 μg mL–1). Increased fluorescence shows more protein binding to the immobilized glycans, highlighting how the same lectin can bind structurally diverse glycans. Selected high-binding glycans are indicated.
The galectin family of lectins plays a crucial role in human physiology, but all have affinity toward β-galactosides56 with subtle differences in their glycan binding profiles,57 which is also controlled by the architecture (chimeric, tandem repeat, or prototype).58 Therefore, if the aim is to selectively identify a galectin as a biomarker, for example, the cross-reactivity question is crucial. Finally, cross reactivity is context dependent—cross reactivity from blood biopsies will be distinct from a wastewater containing the galactose-binding cholera toxin.59 Unlike small molecules, materials chemistry solutions offer a huge opportunity to control the 3D presentation, density, heterogeneity, and nature of the glycans, and this Perspective introduces some approaches being taken to address this problem, hoping to show that this is an area that is ripe for innovation.
Glycan Density
Perhaps the simplest tool to tune the glycan/lectin interface is tuning the side chain density of glycans, which can be achieved by copolymerization or postpolymerization modification.60 While outside of the scope of this Perspective, the glycan array literature already makes extensive use of variable density surface display, where the differences in density can promote/inhibit inter-/intralectin binding interactions and have been reviewed.61 It is also crucial to note that density changes achieved by addition of another glycan (which introduces potential secondary binders) is covered later in this Perspective, as the effects from these similar concepts can be very distinct.
Godula et al. employed a microarray platform with immobilized synthetic glycopolymers to investigate how glycan valency and spatial separation affect the binding mode of a panel of four GalNAc specific lectins (Figure 3A).62 SBA (soybean agglutinin) showed the highest binding to the most dense arrays, whereas HPA (Helix pomatia agglutinin) showed the highest binding to the lowest density, even though they are both GalNAc binding lectins, with the key difference being the lectins’ ability to form interchain cross-links. Hence, simple density tuning, with careful consideration of the lectin architecture, introduces selectivity. This raises the question of how binding affinities/selectivities for isolated glycans scales with multivalent systems and that they are not always linear relationships.63
Figure 3.
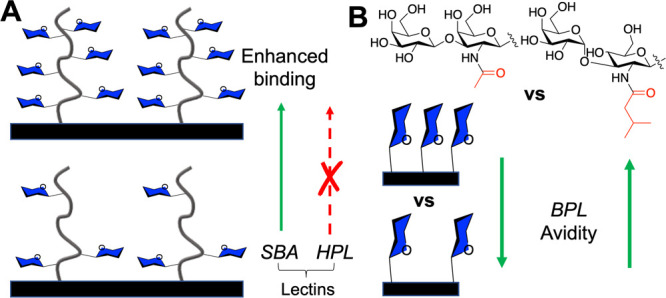
Effect of glycan density on lectin binding. (A) Glycopolymer surfaces show differential responses to SBA and HPL as a function of GalNAc side density.62 (B) Monolayers containing two distinct glycans show differential responses to BPL binding.64
Whitesides and co-workers prepared self-assembled monolayers bearing galactose ligands (Figure 3B) and evaluated binding toward BPL (Bauhinia purpurea lectin).64 An unnatural glycan (with an N-valeryl group and α replacing a β linkage) showed increased avidity with higher density but the opposite effect for the natural glycan, showing that selectivity was possible. It should be noted that characterization of glycopolymer and other multivalent glycostructures is challenging, as spacing/clustering of glycans on these scaffolds is mostly unknown and will almost certainly influence both the affinity and selectivity. Kwon et al. synthesized 6′-sialyllactose presenting PAMAM-based dendrimers with well-defined ligand densities and spacing. The G4 dendrimer outperformed larger/smaller dendrimers in an influenza inhibitory assay, with an estimated spacing of 3 nm between ligands estimated to be optimum.65 Smaller di- and trivalent ligands were also shown to be potent hemagglutinin inhibitors, enhancing >400-fold in comparison to monovalent ligands.66 The ligand density has been reported to be crucial for cholera toxin binding, with many studies reporting that low density (fewer galactose units) leads to maximum inhibition.67−69 On consideration of the inherent simplicity in changing density, this is a valuable tool for identifying selectivity (or preferentiality), whereby the lectin architecture, in addition to binding-site preference, can be exploited.
3D Presentation
The reductionist nature of the glycan presentation in many materials does not (yet) recapitulate the precise 3D presentation and valency control which is found in oligosaccharides. Figure 4A shows selected data from a glycan microarray against human influenza hemagglutinins.70 Biantennary glycans led to significant binding, in comparison to monoantennary glycans, and all binding (or rather binding signal, in the particular assay) was removed when just a trisaccharide (with the same final three monosaccharide units) was displayed. Asymmetric linkages also prevented binding. While the interactions are complex, involving multiple contacts, this example shows how glycan selectivity in Nature is driven by the presentation as much as the chemical nature. Modeling has shown how the chemical nature of how glycans are presented on arrays can lead to false negatives.71 A glycan array strategy was again used to discover bivalent ligands capable of spanning binding sites in LecA (from Pseudomonas aeruginosa), with only ligands with a precise match leading to enhanced affinity and selectivity in comparison to a Shiga toxin.72 A synthetic biology solution to controlling the presentation was shown by Branson et al., who precisely displayed just five copies of the GM1-oligosaccharide onto cholera toxin proximal to its binding sites (Figure 4B), ensuring that the glycans were spatially located for optimal engagement with cholera toxin, leading to nM affinity,73 which has also been modeled showing that the size of the multivalent core must match the receptor unit display and valency.74
Figure 4.
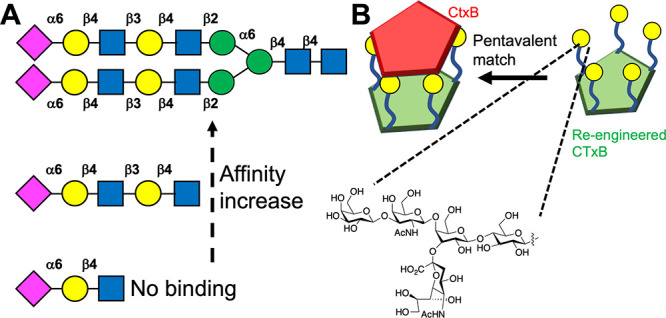
3D presentation of glycans affects binding. (A) Neu5Ac terminated glycans versus hemagglutinins, showing branching and sequence-length dependent binding.70 (B) A pentameric glycosylated cholera toxin B subunit (CTxB) has a 3D match to CTxB for nM inhibition.73
To tune the presentation of glycans, Kiessling and co-workers exploited ROMP to install cis or trans backbones, on otherwise identical polymers. The cis backbones led to an extended conformation, leading to stronger binding in comparison to trans backbones, mimicking native mucin presentation.69 Changing the side chain linker from an amide to an ester in glycopolymers, which in turn affects the flexibility of the glycan, was reported to dramatically alter the overall affinity of mannosylated glycopolymers.75 A proline macrocycle was used to control the presentation of mannose to discriminate between Langerin and DC-SIGN, increasing selectivity many thousand-fold. Both lectins bind to the mannose, but selectivity was achieved due to the spacing differences in the Langerin homotrimer, in comparison to the DC-SIGN homotetramer (Figure 5A).76 Bachem et al. used DNA-PNA scaffolds to precisely space and cluster glycans to selectively engage Langerin with 1150-fold increased affinity in comparison to the free ligand.77 In addition to precisely targeting 3D presentation to gain affinity, the presentation of glycans can affect a sensing outcome (which is not necessarily proportional to affinity). For example, SARS-CoV-2 spike protein binding in a flow-through assay was dependent on the length of polymeric linkers, connecting Neu5NAc to gold nanoparticles,49 and polymer chain length and chemistry tuned the outputs in gold nanoparticle aggregation assays (Figure 5B).34 The polymer architecture also gives rise to very different binding, with a linear sialic acid presenting polymer showing higher in vitro and in vivo activity for protection against influenza infection, attributed to the increased steric shielding by the linear polymer, in comparison to the compact dendrimer.78 Star branched polymannosides varying in only the number and length of arms showed differential responses to immobilized human lectins using SPR.79 These examples highlight how both precision and, more generally, macromolecular engineering could be easily exploited in the search for selectivity.
Figure 5.
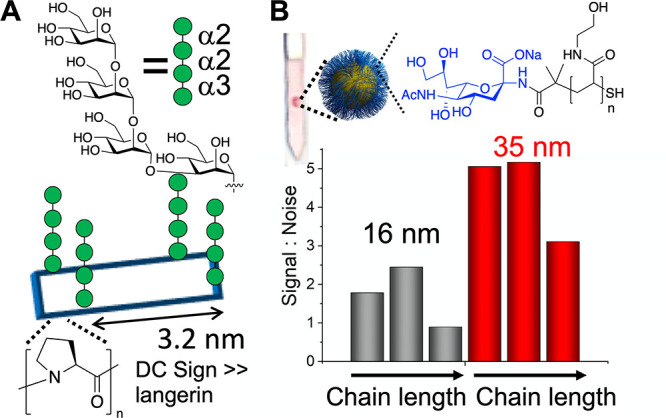
Glycan presentation affects overall binding. (A) Cyclic proline scaffolds bearing Man4, with selective DC-SIGN binding, over Langerin, even though both bind the glycan individually.76 (B) Flow-through detection of SARS-CoV-2 spike protein using polymer-tethered glyconanoparticles, with the signal controlled by diameter and chain length, with the same glycan. Image adapted from ref (49).
Glycan Heterogeneity and Targeting of Secondary Binding Sites
A classic description of lectins is also as “pattern recognition molecules”.80 Pathogens and the host share many glycans, and hence differentiating between these must be driven by a selection other than just their chemical identity. The primary binding site of a lectin is the subject of most focus, but allosteric (secondary) interactions can also be exploited, by incorporating multiple (smaller) glycans proximal to each other rather than as a single oligosaccharide, mimicking glycan branching. Hence, presenting multiple glycans can lead to selectivity gains (and is related to the previous section in that the 3D control of these can also matter).
Turnbull et al. probed the GM1 CTx interaction by ITC, showing that the sialic acid unit contributed 44% of the intrinsic binding energy, although the sialic acid ligand when used alone had no appreciable affinity (Figure 6A,B).81 This demonstrated that an approach to target secondary binding pockets, in addition to the primary β-galactose site, is a valid tool for gaining selectivity. Tran et al. used polymeric scaffolds bearing β-galactose as the primary ligand for CTx but also “clicked” (azide/alkyne) additional functional groups proximal to the galactose, to mimic the branched GM1 ganglioside.82 This approach led to a shift in IC50 from 584 μM (for sialic acid) to 0.014 μM for a fluorobenzyl derivative (Figure 6C). Gibson and co-workers showed that addition of aromatic secondary units (in addition to galactose) in a two-step postpolymerization strategy enabled the relative affinity (selectivity) of the glycopolymers toward CTx and PNA (peanut agglutinin) to be tuned by 20-fold.83,84 In contrast, using thiolactone chemistry (Figure 6D), a benzyl side chain reduced CTx inhibition but retained RCA120 inhibition, showing that the precise location and density of side chains has a significant effect.84 The density of side chains in CTx inhibition has also been reported in several studies, with lower galactose density often leading to increased inhibitory activity, showing that the “more is better” design principle is overly simplistic.27,68,69,85
Figure 6.
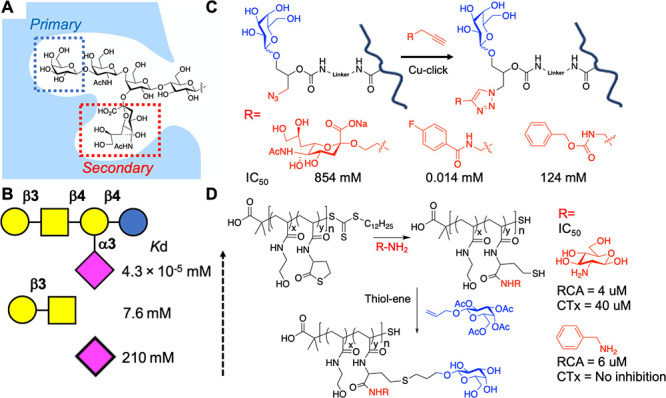
Targeting secondary binding sites in CTx. (A) Schematic of the GM1 glycan in the CTx binding site. (B) Affinity of glycans toward CTx from ITC.81 (C) Secondary binding site targeting via a click reaction proximal to the primary galactose unit and CTx inhibition.82 (D) Thiolactone ring opening to install secondary binding units and CTx/RCA120 inhibition.84
The importance of heterogeneity in biomimetics is in line with the complexity of the glycans in the glycocalyx,86 and the strongest binder may not be the only component essential for biological function. Worstell et al. used a nanocube-based sensing system to demonstrate that the addition of fucosyl GM1 into a mixture with GM2 led to enhanced binding affinity toward CTx, even though the fucosyl GM1 itself had minimal affinity.87 Similarly, galactose and fucose copolymers were more effective inhibitors of CTx binding to human enteroids than galacto- or fuco-polymers alone, due to the additional lower-affinity fucose-binding site.88,89 What is clear is that most materials strategies currently rely on trial and error to judge benefits (or not) from heterogeneity. The challenge of the polymer sequence for example, where extended sequences of one glycan may emerge, rather than a pure statistical distribution in a copolymer, makes quantification of the exact role of each component a major challenge.
The above examples support mechanisms where the heterogeneity increases affinity/selectivity by targeting secondary sites, but that is not the sole mode of action. An alternative mechanism of action for heterogeneity gains (and hence potentially selectivity) is due to steric shielding, from a nonbinding partner. Hartmann and co-workers have shown that nonbinding galactose units on a sequence-defined oligomer enhance inhibitory activity but do not change the overall affinity (Kd) due to a steric shielding effect, as shown by STD NMR (Figure 7A).90 Garcia-Fernandez and co-workers undertook an extremely detailed study using cyclodextrins as the scaffold for the heterogeneous display of glycan (Figure 7B).91 Selectivity between Con A and PNA was achieved not only by heterogeneity but also in their inhibitory action against glycosidases, tuning in selectivity toward maltase, isomalatase, and α-mannosidase. The data supported that sliding or steric shielding was again the mechanism for enhancement, rather than a secondary binding site (unlike the previous examples for CTx). Dendrimers bearing variable densities of mannose and galactose were immobilized on surfaces, and screening revealed “hot spots” where a specific presentation/ratio led to selectivity. The underpinning mechanism for this was not clear but showed the principle that heterogeneity could be deployed in a biosensor format.92 Otten et al. used mixtures of GalNAc and ManNAc in a gold nanoparticle biosensor format (for lectin aggregation). Nonlinear responses to glycan mixtures were seen, such that affinity for SBA could be retained but was significantly reduced toward RCA120 due to the addition of mannose.36
Figure 7.
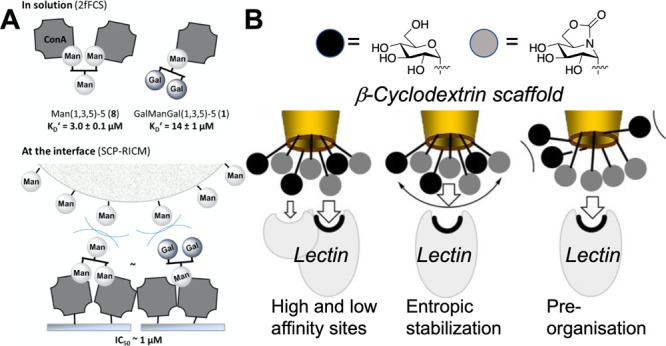
Heterogeneous presentation of high- and low-affinity glycans can enhance binding. (A) Sequence-defined polymers binding Con A in solution and at the interface (competition experiment).90 Reprinted with permission from ref (90). Copyright 2014 American Chemical Society. (B) Proposed modes of affinity enhancement for heterogeneous glycoclusters based on cyclodextrin scaffolds.91 Reprinted with permission from ref (91). Copyright 2017 John Wiley and Sons.
Variable-density glycopolymers have been immobilized onto glass slides.93 Using this strategy, individual glycan components from degraded heparin sulfate were screened for their ability to bind FGF2 (fibroblast growth factor 2).94 However, it is important to note that heterogeneity does not always lead to these increased gains and is material- and lectin-dependent. For example, dilution of an α-mannosylated glycopolymer with β-galactose side chains led to a decrease in DC-SIGN binding (measured by SPR; Figure 8A).33 Alternatively, the heterogeneity may play only a slight role, as seen for mannosylated gold nanoparticles for inhibiting FimH-driven adhesion (Figure 8B).95
Figure 8.
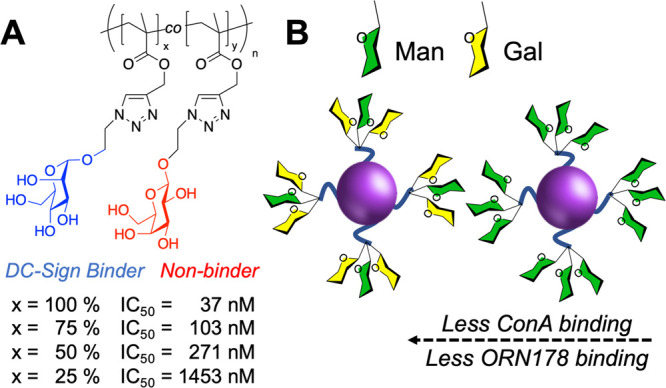
Glycan heterogeneity can reduce affinity. (A) Mannosylated glycopolymers show reduced inhibitory activity vs DC-SIGN on dilution with galactose.33 (B) Mannosylated glycoparticles with reduced affinity toward Con A and ORN178 (E. coli) as galactose is introduced.95
This subsection highlighted the inherent complexity (and huge potential) of heterogeneous and secondary-site targeting materials. A key observation is not all lectins respond to the dual effect of glycan dilution, showing that selectivity tuning is possible. The mechanism for selectivity is often subtle, covering steric shielding effects, targeting secondary sites, or allowing/preventing inter-/intralectin binding site spanning. However, subtle differences between systems can have large effects and high-throughput screening-based approaches, based on sequentially modified scaffolds, could play a role in dissecting these interactions.
Non-natural Glycans and Glycan Mimetics
To drive selectivity and affinity, a medicinal chemistry (e.g. not materials) approach would be to use non-natural glycans that have favorable binding and pharmacokinetic properties. Thiosugars (where the internal ring oxygen is substituted by a sulfur, rather than those with an anomeric thiol), iminosugars (NH replacement), and carbasugars (CH2) are established medicinal chemistry tools, especially as glycosidase inhibitors,96 and glycomimetic drugs have reached the clinic.97 The application of these approaches in multivalent systems is less common, as the synthetic burden may outweigh the intrinsic simplicity of many polymeric systems, but has already shown significant promise. Kiessling and co-workers developed a mannose mimetic that was displayed on BSA carrier proteins and functioned as a DC-SIGN agonist.98 The same group demonstrated that C-linked mannose glycopolymers were more potent binders of Con A than O-linked species30 and that selective positioning of sulfate groups on galactosylated polymers can tune the affinity between L- and P-selectins.99
Fieschi and co-workers have explored the use of glycomimietics to tune selectivity. They observed that C-6 sulfation in GlcNAc derivatives led to selectivity toward Langerin. They developed this further to identify inhibitors (Figure 9A) which only bound DC-SIGN and not Langerin100 and were incorporated into dendritic structures, which selectively inhibited HIV infection in a model study.101
Figure 9.
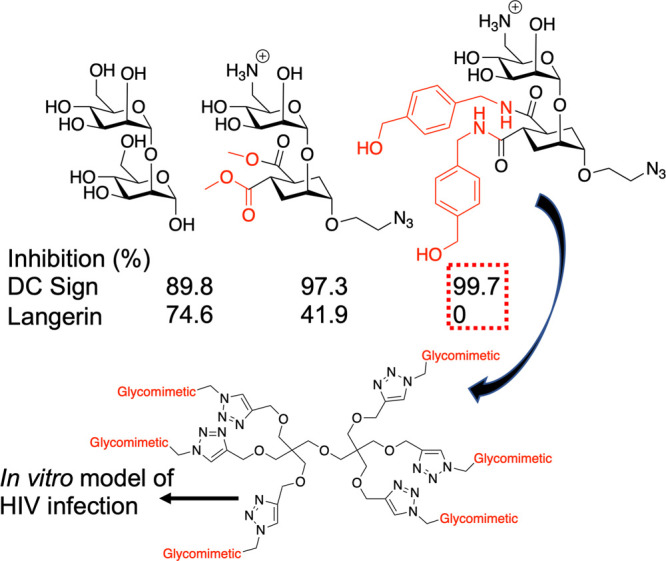
Glycomimetic strategy to identify selective DC-SIGN binders, with no inhibition of Langerin, and subsequent multivalent display.100,101
Fluorine is an appealing modification to glycans as a tool to modulate their pharmacokinetics, due to its small size and minimal effect on glycan conformation.102,103 Fluorine is not a hydrogen bond donor but is a weak acceptor, and hence the replacement of hydroxyls with fluorines can lead to significant changes in binding.104 Fluorinated phenyltriazolyl-thiogalactosides engaged with additional interactions with Galectin-3, in comparison to nonfluorinated species.105 Fluorination of the glycan portion of MUC-1 peptides resulted in differential antiserum responses.106 Site-specific fluorination at the terminal mannose C-6 in Man3GlcNAc was found to be crucial to Con A binding, but the branched mannose C-6 could tolerate fluorination, when it was displayed on a glycoprotein.107 Encouraged by this, Richards et al. employed a chemoenzymatic synthesis to obtain a library of fluorinated Lacto-N-biose derivatives, exploiting the promiscuity of the BiGalK and BiGalHexNAcP enzymes from Bifidobacterium infantis which tolerate fluorinated donors (Figure 10A). Incorporation of these onto multivalent gold nanoparticle platforms allowed the identification of specific fluorination sites to tune discrimination between Galectin-3 and Galectin-7 with the glycomaterials (Figure 10B,C).108
Figure 10.
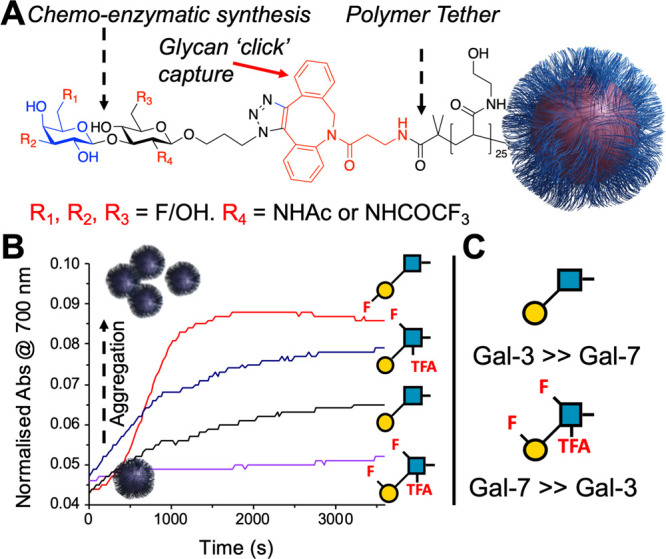
Fluorinated Lacto-N-biose-functional gold nanoparticles to bind galectins.108 (A) Schematic of glyconanoparticle structure (B) Aggregation kinetics of selected glyconanoparticles with Galectin-3. (C) Glycans identified (in multivalent format only) with switched affinity. Reproduced from ref (108) with permission from the Royal Society of Chemistry.
Directed Evolution, High-Throughput, and Biochemical Panning Approaches
High-throughput discovery approaches offer an alternative (or complementary) tool for the discovery of high-affinity and selective ligands. For example, robotics and parallel synthesis have been used for polymeric109,110 and inorganic111 materials. Due to selection and amplification tools, protein and nucleic acid based materials can be screened by phage112 or apatamer/SELEX113 technologies. However, for glycans, which are not template-directed and cannot be amplified, the discovery tools are fewer. Automated glycan synthesis is rapidly progressing but is still not a routine laboratory tool.114,115 It should be noted that glycan arrays are high-throughput, once the glycans are in hand,70 but have already been reviewed and are not covered here.116
Krauss and co-workers made libraries of peptides containing non-natural amino acids (bearing an alkyne) connected to mRNA. This mRNA-encoded library could be glycosylated (using glycosyl azides), followed by selection and PCR amplification of the “winning” binders. Using this approach, a library of 1013 glycopeptides were screened and a picomolar binder to the HIV neutralizing antibody 2G12 was identified,117 with a chemical glycosylation step being essential during selection rounds. Ng et al. used a related strategy whereby phase display was employed, followed by oxidation of terminal serine residues to aldehydes to capture amino-oxy mannose (Figure 11A).118 Selection against Con A (positive) and BSA (negative) (Figure 11B) led to ligands with increased selectivity via modulation of the peptide linkage, with an example hit being shown in Figure 10C. Interestingly, the same ligand for Con A binding showed high affinity to DC-SIGN which also binds high-mannose, highlighting again the selectivity challenge. A method where the peptide is not varied but the glycan immobilized onto an M13 bacteriophage is, termed a “liquid glycan array”, has also been reported.119 Related approaches to identify selective binders using DNA-encoded glycans as microarray alternatives can be used to pan hundreds of glycans.120 These methods all show huge potential for true high-throughput screening and are especially suitable for positive/negative selection to introduce selectivity.
Figure 11.
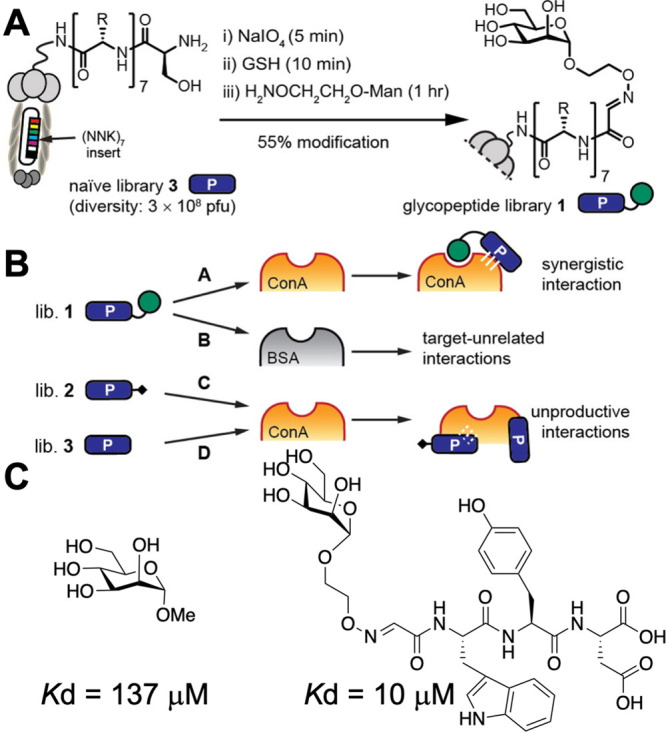
Phage-based screening for glycosylated peptides to enhance mannose binding to Con A.118 (A) Schematic of glycosylation at the serine N-terminus of peptides on phage. (B) Selection and enrichment processes. (C) Example of a discovered peptide sequence with enhanced Con A affinity in comparison to methyl-mannoside. Reprinted (adapted) with permission from ref (118). Copyright 2015 American Chemical Society.
Outlook and Opportunities
The aim of this perspective is to highlight that macromolecular and materials science has a huge potential to have an effect on glycoscience and that not only is the selectivity challenge tractable but also there exists a diverse range of strategies to achieve it. This perspective is not intended to be comprehensive but to introduce the reader to some current strategies that show promise in this challenging area.
By drawing from detailed “small molecule” studies, significant gains in selectivity are possible by exploiting the benefits of multivalency, including the ability to present multiple different glycans on the same scaffold, use steric shielding effects, and tune the linker chemistry. However, moving from simple monosaccharides to oligo or non-natural glycans is essential to ensure that this large step is taken. Advances in high-throughput materials discovery is well placed to support this, as well as exciting macromolecular tools based upon, for example, sequence-controlled121 and folded polymers,122 which show early promise.90 Recent advances in structural biology, including cryo-electron microscopy123 and the new computation tools to predict protein structure,124 will inevitably feed into this as well. It is also crucial, if glycomaterials are to be used in biological environments, to understand how the media affect the performance. The protein corona, where proteins absorb to nanoparticle surfaces,125 has been shown to introduce additional glycoproteins126 and hence there is the potential for a highly selective binder in “pure” solutions to lose function in an application. We anticipate that the next generation of glycomaterials will move beyond using simple monosaccharides against plant lectins (which have obvious value still) to real targets under biomedically relevant conditions.
Acknowledgments
M.I.G. thanks the ERC for a Consolidator Grant (866056). BBSRC/Innovate are thanked for funding the Specialty Glycans project BB/M02878X/1. UoW, the EPSRC (EP/R511808/1), and the BBSRC (BB/S506783/1) impact acceleration accounts are acknowledged. This project has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement no. 814236, supporting the NanoCarb ITN network, which is thanked for useful discussions. The glycan array resources (DC-SIGN screening primscreen_5273) were provided by the Consortium for Functional Glycomics grant numbers GM62116 and GM098791.
The authors declare no competing financial interest.
References
- Flynn R. A.; Pedram K.; Malaker S. A.; Batista P. J.; Smith B. A. H.; Johnson A. G.; George B. M.; Majzoub K.; Villalta P. W.; Carette J. E.; Bertozzi C. R. Small RNAs Are Modified with N-Glycans and Displayed on the Surface of Living Cells. Cell 2021, 184 (12), 3109–3124. 10.1016/j.cell.2021.04.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pilobello K. T.; Mahal L. K. Deciphering the Glycocode: The Complexity and Analytical Challenge of Glycomics. Curr. Opin. Chem. Biol. 2007, 11 (3), 300–305. 10.1016/j.cbpa.2007.05.002. [DOI] [PubMed] [Google Scholar]
- Bertozzi C. R.; Kiessling L. L. Chemical Glycobiology. Science 2001, 291 (5512), 2357–2364. 10.1126/science.1059820. [DOI] [PubMed] [Google Scholar]
- Adibekian A.; Stallforth P.; Hecht M.-L.; Werz D. B.; Gagneux P.; Seeberger P. H. Comparative Bioinformatics Analysis of the Mammalian and Bacterial Glycomes. Chem. Sci. 2011, 2 (2), 337–344. 10.1039/C0SC00322K. [DOI] [Google Scholar]
- Dedola S.; Rugen M. D.; Young R. J.; Field R. A. Revisiting the Language of Glycoscience: Readers, Writers and Erasers in Carbohydrate Biochemistry. ChemBioChem 2020, 21 (3), 423–427. 10.1002/cbic.201900377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lis H.; Sharon N. Lectins: Carbohydrate-Specific Proteins That Mediate Cellular Recognition. Chem. Rev. 1998, 98 (2), 637–674. 10.1021/cr940413g. [DOI] [PubMed] [Google Scholar]
- Lee Y. C.; Townsend R. R.; Hardy M. R.; Lönngren J.; Arnarp J.; Haraldsson M.; Lönn H. Binding of Synthetic Oligosaccharides to the Hepatic Gal/GalNAc Lectin. Dependence on Fine Structural Features. J. Biol. Chem. 1983, 258 (1), 199–202. 10.1016/S0021-9258(18)33240-X. [DOI] [PubMed] [Google Scholar]
- Lee Y. C.; Lee R. T. Carbohydrate-Protein Interactions: Basis of Glycobiology. Acc. Chem. Res. 1995, 28 (8), 321–327. 10.1021/ar00056a001. [DOI] [Google Scholar]
- Lundquist J. J.; Toone E. J. The Cluster Glycoside Effect. Chem. Rev. 2002, 102 (2), 555–578. 10.1021/cr000418f. [DOI] [PubMed] [Google Scholar]
- Sigal G. B.; Mammen M.; Dahmann G.; Whitesides G. M. Polyacrylamides Bearing Pendant α-Sialoside Groups Strongly Inhibit Agglutination of Erythrocytes by Influenza Virus: The Strong Inhibition Reflects Enhanced Binding through Cooperative Polyvalent Interactions. J. Am. Chem. Soc. 1996, 118 (16), 3789–3800. 10.1021/ja953729u. [DOI] [Google Scholar]
- Kanai M.; Mortell K. H.; Kiessling L. L. Varying the Size of Multivalent Ligands: The Dependence of Concanavalin A Binding on Neoglycopolymer Length. J. Am. Chem. Soc. 1997, 119 (41), 9931–9932. 10.1021/ja972089n. [DOI] [Google Scholar]
- Ernst B.; Magnani J. L. From Carbohydrate Leads to Glycomimetic Drugs. Nat. Rev. Drug Discovery 2009, 8 (8), 661–677. 10.1038/nrd2852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleeb S.; Pang L.; Mayer K.; Eris D.; Sigl A.; Preston R. C.; Zihlmann P.; Sharpe T.; Jakob R. P.; Abgottspon D.; Hutter A. S.; Scharenberg M.; Jiang X.; Navarra G.; Rabbani S.; Smiesko M.; Ledin N.; Bezenon J.; Schwardt O.; Maier T.; Ernst B. FimH Antagonists: Bioisosteres to Improve the in Vitro and in Vivo PK/PD Profile. J. Med. Chem. 2015, 58 (5), 2221–2239. 10.1021/jm501524q. [DOI] [PubMed] [Google Scholar]
- Liang R.; Yan L.; Loebach J.; Ge M.; Uozumi Y.; Sekanina K.; Horan N.; Gildersleeve J.; Thompson C.; Smith A.; Biswas K.; Still W. C.; Kahne D. Parallel Synthesis and Screening of a Solid Phase Carbohydrate Library. Science 1996, 274 (5292), 1520–1522. 10.1126/science.274.5292.1520. [DOI] [PubMed] [Google Scholar]
- Levine P. M.; Carberry T. P.; Holub J. M.; Kirshenbaum K. Crafting Precise Multivalent Architectures. MedChemComm 2013, 4 (3), 493–509. 10.1039/C2MD20338C. [DOI] [Google Scholar]
- Wittmann V.; Pieters R. J. Bridging Lectin Binding Sites by Multivalent Carbohydrates. Chem. Soc. Rev. 2013, 42 (10), 4492. 10.1039/c3cs60089k. [DOI] [PubMed] [Google Scholar]
- Thompson J. P.; Schengrund C.-L. Oligosaccharide-Derivatized Dendrimers: Defined Multivalent Inhibitors of the Adherence of the Cholera Toxin B Subunit and the Heat Labile Enterotoxin of E. Coli to GM1. Glycoconjugate J. 1997, 14 (7), 837–845. 10.1023/A:1018590021762. [DOI] [PubMed] [Google Scholar]
- Turnbull W. B.; Stoddart J. F. Design and Synthesis of Glycodendrimers. Rev. Mol. Biotechnol. 2002, 90 (3–4), 231–255. 10.1016/S1389-0352(01)00062-9. [DOI] [PubMed] [Google Scholar]
- Vrasidas I.; De Mol N. J.; Liskamp R. M. J.; Pieters R. J. Synthesis of Lactose Dendrimers and Multivalency Effects in Binding to the Cholera Toxin B Subunit. Eur. J. Org. Chem. 2001, 2001 (24), 4685–4692. . [DOI] [Google Scholar]
- Arosio D.; Vrasidas I.; Valentini P.; Liskamp R. M. J.; Pieters R. J.; Bernardi A. Synthesis and Cholera Toxin Binding Properties of Multivalent GM1Mimics. Org. Biomol. Chem. 2004, 2 (14), 2113. 10.1039/b405344c. [DOI] [PubMed] [Google Scholar]
- Branderhorst H. M.; Kooij R.; Salminen A.; Jongeneel L. H.; Arnusch C. J.; Liskamp R. M. J.; Finne J.; Pieters R. J. Synthesis of Multivalent Streptococcus Suis Adhesion Inhibitors by Enzymatic Cleavage of Polygalacturonic Acid and “click” Conjugation. Org. Biomol. Chem. 2008, 6 (8), 1425–1434. 10.1039/b800283e. [DOI] [PubMed] [Google Scholar]
- Johansson E. M. V.; Crusz S. A.; Kolomiets E.; Buts L.; Kadam R. U.; Cacciarini M.; Bartels K.-M.; Diggle S. P.; Cámara M.; Williams P.; Loris R.; Nativi C.; Rosenau F.; Jaeger K.-E.; Darbre T.; Reymond J.-L. Inhibition and Dispersion of Pseudomonas Aeruginosa Biofilms by Glycopeptide Dendrimers Targeting the Fucose-Specific Lectin LecB. Chem. Biol. 2008, 15 (12), 1249–1257. 10.1016/j.chembiol.2008.10.009. [DOI] [PubMed] [Google Scholar]
- Kadam R. U.; Bergmann M.; Hurley M.; Garg D.; Cacciarini M.; Swiderska M. A.; Nativi C.; Sattler M.; Smyth A. R.; Williams P.; Cámara M.; Stocker A.; Darbre T.; Reymond J.-L. A Glycopeptide Dendrimer Inhibitor of the Galactose-Specific Lectin LecA and of Pseudomonas Aeruginosa Biofilms. Angew. Chem., Int. Ed. 2011, 50 (45), 10631–10635. 10.1002/anie.201104342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernardi A.; Jiménez-Barbero J.; Casnati A.; De Castro C.; Darbre T.; Fieschi F.; Finne J.; Funken H.; Jaeger K.-E.; Lahmann M.; Lindhorst T. K.; Marradi M.; Messner P.; Molinaro A.; Murphy P. V.; Nativi C.; Oscarson S.; Penadés S.; Peri F.; Pieters R. J.; Renaudet O.; Reymond J.-L.; Richichi B.; Rojo J.; Sansone F.; Schäffer C.; Turnbull W. B.; Velasco-Torrijos T.; Vidal S.; Vincent S.; Wennekes T.; Zuilhof H.; Imberty A. Multivalent Glycoconjugates as Anti-Pathogenic Agents. Chem. Soc. Rev. 2013, 42 (11), 4709–4727. 10.1039/C2CS35408J. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reymond J. L.; Bergmann M.; Darbrea T. Glycopeptide Dendrimers as Pseudomonas Aeruginosa Biofilm Inhibitors. Chem. Soc. Rev. 2013, 42 (11), 4814–4822. 10.1039/c3cs35504g. [DOI] [PubMed] [Google Scholar]
- Wittmann V.; Seeberger S. Spatial Screening of Cyclic Neoglycopeptides: Identification of Polyvalent Wheat-Germ Agglutinin Ligands. Angew. Chem., Int. Ed. 2004, 43 (7), 900–903. 10.1002/anie.200352055. [DOI] [PubMed] [Google Scholar]
- Polizzotti B. D.; Kiick K. L. Effects of Polymer Structure on the Inhibition of Cholera Toxin by Linear Polypeptide-Based Glycopolymers. Biomacromolecules 2006, 7 (2), 483–490. 10.1021/bm050672n. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miura Y.; Koketsu D.; Kobayashi K. Synthesis and Properties of a Well-defined Glycopolymer via Living Radical Polymerization. Polym. Adv. Technol. 2007, 18 (8), 647–651. 10.1002/pat.873. [DOI] [Google Scholar]
- Baek M.-G.; Roy R. Relative Lectin Binding Properties of T-Antigen-Containing Glycopolymers: Copolymerization OfN-Acryloylated T-Antigen Monomer vs. Graft Conjugation of Aminated T-Antigen Ligands onto Poly(N-Acryloxysuccinimide). Macromol. Biosci. 2001, 1 (7), 305–311. . [DOI] [Google Scholar]
- Mortell K. H.; Weatherman R. V.; Kiessling L. L. Recognition Specificity of Neoglycopolymers Prepared by Ring-Opening Metathesis Polymerization. J. Am. Chem. Soc. 1996, 118 (9), 2297–2298. 10.1021/ja953574q. [DOI] [Google Scholar]
- Mammen M.; Dahmann G.; Whitesides G. M. Effective Inhibitors of Hemagglutination by Influenza Virus Synthesized from Polymers Having Active Ester Groups. Insight into Mechanism of Inhibition. J. Med. Chem. 1995, 38 (21), 4179–4190. 10.1021/jm00021a007. [DOI] [PubMed] [Google Scholar]
- Ladmiral V.; Mantovani G.; Clarkson G. J.; Cauet S.; Irwin J. L.; Haddleton D. M. Synthesis of Neoglycopolymers by a Combination of “Click Chemistry” and Living Radical Polymerization. J. Am. Chem. Soc. 2006, 128 (14), 4823–4830. 10.1021/ja058364k. [DOI] [PubMed] [Google Scholar]
- Becer C. R. R.; Gibson M. I. M. I.; Geng J.; Ilyas R.; Wallis R.; Mitchell D. A. D. A.; Haddleton D. M. D. M. High-Affinity Glycopolymer Binding to Human DC-SIGN and Disruption of DC-SIGN Interactions with HIV Envelope Glycoprotein. J. Am. Chem. Soc. 2010, 132 (43), 15130–15132. 10.1021/ja1056714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Georgiou P. G.; Baker A. N.; Richards S.-J.; Laezza A.; Walker M.; Gibson M. I. Tuning Aggregative versus Non-Aggregative Lectin Binding with Glycosylated Nanoparticles by the Nature of the Polymer Ligand. J. Mater. Chem. B 2020, 8 (1), 136–145. 10.1039/C9TB02004G. [DOI] [PubMed] [Google Scholar]
- Richards S.-J.; Baker A. N.; Walker M.; Gibson M. I. Polymer-Stabilized Sialylated Nanoparticles: Synthesis, Optimization, and Differential Binding to Influenza Hemagglutinins. Biomacromolecules 2020, 21 (4), 1604–1612. 10.1021/acs.biomac.0c00179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Otten L.; Vlachou D.; Richards S.-J.; Gibson M. I. Glycan Heterogeneity On Gold Nanoparticles Increases Lectin Discrimination Capacity in Label-Free Multiplexed Bioassays. Analyst 2016, 141 (14), 4305–4312. 10.1039/C6AN00549G. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X.; Ramström O.; Yan M. A Photochemically Initiated Chemistry for Coupling Underivatized Carbohydrates to Gold Nanoparticles. J. Mater. Chem. 2009, 19 (47), 8944–8949. 10.1039/b917900c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Astronomo R. D.; Kaltgrad E.; Udit A. K.; Wang S. K.; Doores K. J.; Huang C. Y.; Pantophlet R.; Paulson J. C.; Wong C. H.; Finn M. G.; Burton D. R. Defining Criteria for Oligomannose Immunogens for HIV Using Icosahedral Virus Capsid Scaffolds. Chem. Biol. 2010, 17 (4), 357–370. 10.1016/j.chembiol.2010.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bundle D. R.; Kitov P. I.; Sadowska J. M.; Mulvey G.; Armstrong G. D.; Ling H.; Pannu N. S.; Read R. J. Shiga-like Toxins Are Neutralized by Tailored Multivalent Carbohydrate. Nature 2000, 403 (6770), 669–672. 10.1038/35001095. [DOI] [PubMed] [Google Scholar]
- Varner C. T.; Rosen T.; Martin J. T.; Kane R. S. Recent Advances in Engineering Polyvalent Biological Interactions. Biomacromolecules 2015, 16 (1), 43–55. 10.1021/bm5014469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galan M. C.; Dumy P.; Renaudet O. Multivalent Glyco(Cyclo)Peptides. Chem. Soc. Rev. 2013, 42 (11), 4599–4612. 10.1039/C2CS35413F. [DOI] [PubMed] [Google Scholar]
- Jiménez Blanco J. L.; Ortiz Mellet C.; García Fernández J. M. Multivalency in Heterogeneous Glycoenvironments: Hetero-Glycoclusters, -Glycopolymers and -Glycoassemblies. Chem. Soc. Rev. 2013, 42 (11), 4518–4531. 10.1039/C2CS35219B. [DOI] [PubMed] [Google Scholar]
- Kiessling L. L.; Gestwicki J. E.; Strong L. E. Synthetic Multivalent Ligands as Probes of Signal Transduction. Angew. Chem., Int. Ed. 2006, 45 (15), 2348–2368. 10.1002/anie.200502794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharon N. Carbohydrates as Future Anti-Adhesion Drugs for Infectious Diseases. Biochim. Biophys. Acta, Gen. Subj. 2006, 1760 (4), 527–537. 10.1016/j.bbagen.2005.12.008. [DOI] [PubMed] [Google Scholar]
- Spain S. G.; Cameron N. R. A Spoonful of Sugar: The Application of Glycopolymers in Therapeutics. Polym. Chem. 2011, 2 (1), 60–68. 10.1039/C0PY00149J. [DOI] [Google Scholar]
- Branson T. R.; Turnbull W. B. Bacterial Toxin Inhibitors Based on Multivalent Scaffolds. Chem. Soc. Rev. 2013, 42 (11), 4613–4622. 10.1039/C2CS35430F. [DOI] [PubMed] [Google Scholar]
- Pieters R. J. Maximising Multivalency Effects in Protein–Carbohydrate Interactions. Org. Biomol. Chem. 2009, 7 (10), 2013–2025. 10.1039/b901828j. [DOI] [PubMed] [Google Scholar]
- Ambrosi M.; Cameron N. R.; Davis B. G. Lectins: Tools for the Molecular Understanding of the Glycocode. Org. Biomol. Chem. 2005, 3 (9), 1593–1608. 10.1039/b414350g. [DOI] [PubMed] [Google Scholar]
- Baker A. N.; Richards S. J.; Guy C. S.; Congdon T. R.; Hasan M.; Zwetsloot A. J.; Gallo A.; Lewandowski J. R.; Stansfeld P. J.; Straube A.; Walker M.; Chessa S.; Pergolizzi G.; Dedola S.; Field R. A.; Gibson M. I. The SARS-COV-2 Spike Protein Binds Sialic Acids and Enables Rapid Detection in a Lateral Flow Point of Care Diagnostic Device. ACS Cent. Sci. 2020, 6 (11), 2046–2052. 10.1021/acscentsci.0c00855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Childs R. A.; Palma A. S.; Wharton S.; Matrosovich T.; Liu Y.; Chai W.; Campanero-Rhodes M. A.; Zhang Y.; Eickmann M.; Kiso M.; Hay A.; Matrosovich M.; Feizi T. Receptor-Binding Specificity of Pandemic Influenza A (H1N1) 2009 Virus Determined by Carbohydrate Microarray. Nat. Biotechnol. 2009, 27 (9), 797–799. 10.1038/nbt0909-797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Figdor C. G.; Van Kooyk Y.; Adema G. J. C-Type Lectin Receptors on Dendritic Cells and Langerhans Cells. Nat. Rev. Immunol. 2002, 2 (2), 77–84. 10.1038/nri723. [DOI] [PubMed] [Google Scholar]
- de Witte L.; Nabatov A.; Geijtenbeek T. B. H. Distinct Roles for DC-SIGN+-Dendritic Cells and Langerhans Cells in HIV-1 Transmission. Trends Mol. Med. 2008, 14 (1), 12–19. 10.1016/j.molmed.2007.11.001. [DOI] [PubMed] [Google Scholar]
- Holla A.; Skerra A. Comparative Analysis Reveals Selective Recognition of Glycans by the Dendritic Cell Receptors DC-SIGN and Langerin. Protein Eng., Des. Sel. 2011, 24 (9), 659–669. 10.1093/protein/gzr016. [DOI] [PubMed] [Google Scholar]
- Lin K.; Kasko A. M. Carbohydrate-Based Polymers for Immune Modulation. ACS Macro Lett. 2014, 3 (7), 652–657. 10.1021/mz5002417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Consortium for Functional Glycomics.
- Rabinovich G. A.; Toscano M. A. Turning “sweet” on Immunity: Galectin-Glycan Interactions in Immune Tolerance and Inflammation. Nat. Rev. Immunol. 2009, 9 (5), 338–352. 10.1038/nri2536. [DOI] [PubMed] [Google Scholar]
- Hirabayashi J.; Hashidate T.; Arata Y.; Nishi N.; Nakamura T.; Hirashima M.; Urashima T.; Oka T.; Futai M.; Muller W. E.; Yagi F.; Kasai K. I. Oligosaccharide Specificity of Galectins: A Search by Frontal Affinity Chromatography. Biochim. Biophys. Acta, Gen. Subj. 2002, 1572 (2–3), 232–254. 10.1016/S0304-4165(02)00311-2. [DOI] [PubMed] [Google Scholar]
- de Jong C. G. H. M.; Gabius H.-J.; Baron W. The Emerging Role of Galectins in (Re)Myelination and Its Potential for Developing New Approaches to Treat Multiple Sclerosis. Cell. Mol. Life Sci. 2020, 77 (7), 1289–1317. 10.1007/s00018-019-03327-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schofield C. L.; Field R. A.; Russell D. A. Glyconanoparticles for the Colorimetric Detection of Cholera Toxin. Anal. Chem. 2007, 79 (4), 1356–1361. 10.1021/ac061462j. [DOI] [PubMed] [Google Scholar]
- Spain S. G.; Gibson M. I.; Cameron N. R. Recent Advances in the Synthesis of Well-Defined Glycopolymers. J. Polym. Sci., Part A: Polym. Chem. 2007, 45 (11), 2059–2072. 10.1002/pola.22106. [DOI] [Google Scholar]
- Rillahan C. D.; Paulson J. C. Glycan Microarrays for Decoding the Glycome. Annu. Rev. Biochem. 2011, 80, 797–823. 10.1146/annurev-biochem-061809-152236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Godula K.; Bertozzi C. R. Density Variant Glycan Microarray for Evaluating Cross-Linking of Mucin-like Glycoconjugates by Lectins. J. Am. Chem. Soc. 2012, 134 (38), 15732–15742. 10.1021/ja302193u. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang R.; Loebach J.; Horan N.; Ge M.; Thompson C.; Yan L.; Kahne D. Polyvalent Binding to Carbohydrates Immobilized on an Insoluble Resin. Proc. Natl. Acad. Sci. U. S. A. 1997, 94 (20), 10554–10559. 10.1073/pnas.94.20.10554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horan N.; Yan L.; Isobe H.; Whitesides G. M.; Kahne D. Nonstatistical Binding of a Protein to Clustered Carbohydrates. Proc. Natl. Acad. Sci. U. S. A. 1999, 96 (21), 11782–11786. 10.1073/pnas.96.21.11782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwon S. J.; Na D. H.; Kwak J. H.; Douaisi M.; Zhang F.; Park E. J.; Park J. H.; Youn H.; Song C. S.; Kane R. S.; Dordick J. S.; Lee K. B.; Linhardt R. J. Nanostructured Glycan Architecture Is Important in the Inhibition of Influenza A Virus Infection. Nat. Nanotechnol. 2017, 12 (1), 48–54. 10.1038/nnano.2016.181. [DOI] [PubMed] [Google Scholar]
- Lu W.; Du W.; Somovilla V. J.; Yu G.; Haksar D.; de Vries E.; Boons G.-J.; de Vries R. P.; de Haan C. A. M.; Pieters R. J. Enhanced Inhibition of Influenza A Virus Adhesion by Di- and Trivalent Hemagglutinin Inhibitors. J. Med. Chem. 2019, 62 (13), 6398–6404. 10.1021/acs.jmedchem.9b00303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S.; Kiick K. L. Architecture Effects on the Binding of Cholera Toxin by Helical Glycopolypeptides. Macromolecules 2008, 41 (3), 764–772. 10.1021/ma702128a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richards S.-J.; Jones M. W.; Hunaban M.; Haddleton D. M.; Gibson M. I. Probing Bacterial-Toxin Inhibition with Synthetic Glycopolymers Prepared by Tandem Post-Polymerization Modification: Role of Linker Length and Carbohydrate Density. Angew. Chem., Int. Ed. 2012, 51 (31), 7812–7816. 10.1002/anie.201202945. [DOI] [PubMed] [Google Scholar]
- Kruger A. G.; Brucks S. D.; Yan T.; Cárcarmo-Oyarce G.; Wei Y.; Wen D. H.; Carvalho D. R.; Hore M. J. A.; Ribbeck K.; Schrock R. R.; Kiessling L. L. Stereochemical Control Yields Mucin Mimetic Polymers. ACS Cent. Sci. 2021, 7 (4), 624–630. 10.1021/acscentsci.0c01569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Z.; Chinoy Z. S.; Ambre S. G.; Peng W.; McBride R.; de Vries R. P.; Glushka J.; Paulson J. C.; Boons G.-J. A General Strategy for the Chemoenzymatic Synthesis of Asymmetrically Branched N-Glycans. Science 2013, 341 (6144), 379–383. 10.1126/science.1236231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grant O. C.; Smith H. M. K.; Firsova D.; Fadda E.; Woods R. J. Presentation, Presentation, Presentation! Molecular-Level Insight into Linker Effects on Glycan Array Screening Data. Glycobiology 2014, 24 (1), 17–25. 10.1093/glycob/cwt083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Novoa A.; Eierhoff T.; Topin J.; Varrot A.; Barluenga S.; Imberty A.; Römer W.; Winssinger N. A LecA Ligand Identified from a Galactoside-Conjugate Array Inhibits Host Cell Invasion by Pseudomonas Aeruginosa. Angew. Chem., Int. Ed. 2014, 53 (34), 8885–8889. 10.1002/anie.201402831. [DOI] [PubMed] [Google Scholar]
- Branson T. R.; McAllister T. E.; Garcia-Hartjes J.; Fascione M. A.; Ross J. F.; Warriner S. L.; Wennekes T.; Zuilhof H.; Turnbull W. B. A Protein-Based Pentavalent Inhibitor of the Cholera Toxin B-Subunit. Angew. Chem., Int. Ed. 2014, 53 (32), 8323–8327. 10.1002/anie.201404397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liese S.; Netz R. R. Quantitative Prediction of Multivalent Ligand-Receptor Binding Affinities for Influenza, Cholera, and Anthrax Inhibition. ACS Nano 2018, 12 (5), 4140–4147. 10.1021/acsnano.7b08479. [DOI] [PubMed] [Google Scholar]
- Nagao M.; Kichize M.; Hoshino Y.; Miura Y. Influence of Monomer Structures for Polymeric Multivalent Ligands: Consideration of the Molecular Mobility of Glycopolymers. Biomacromolecules 2021, 22 (7), 3119–3127. 10.1021/acs.biomac.1c00553. [DOI] [PubMed] [Google Scholar]
- Wen H. C.; Lin C. H.; Huang J. S.; Tsai C. L.; Chen T. F.; Wang S. K. Selective Targeting of DC-SIGN by Controlling the Oligomannose Pattern on a Polyproline Tetra-Helix Macrocycle Scaffold. Chem. Commun. 2019, 55 (62), 9124–9127. 10.1039/C9CC03124C. [DOI] [PubMed] [Google Scholar]
- Bachem G.; Wamhoff E.-C.; Silberreis K.; Kim D.; Baukmann H.; Fuchsberger F.; Dernedde J.; Rademacher C.; Seitz O. Rational Design of a DNA-Scaffolded High-Affinity Binder for Langerin. Angew. Chem., Int. Ed. 2020, 59 (47), 21016–21022. 10.1002/anie.202006880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhatia S.; Lauster D.; Bardua M.; Ludwig K.; Angioletti-Uberti S.; Popp N.; Hoffmann U.; Paulus F.; Budt M.; Stadtmüller M.; Wolff T.; Hamann A.; Böttcher C.; Herrmann A.; Haag R. Linear Polysialoside Outperforms Dendritic Analogs for Inhibition of Influenza Virus Infection in Vitro and in Vivo. Biomaterials 2017, 138, 22–34. 10.1016/j.biomaterials.2017.05.028. [DOI] [PubMed] [Google Scholar]
- Abdouni Y.; Yilmaz G.; Monaco A.; Aksakal R.; Becer C. R. Effect of Arm Number and Length of Star-Shaped Glycopolymers on Binding to Dendritic and Langerhans Cell Lectins. Biomacromolecules 2020, 21 (9), 3756–3764. 10.1021/acs.biomac.0c00856. [DOI] [PubMed] [Google Scholar]
- Dam T. K.; Gerken T. A.; Brewer C. F. Thermodynamics of Multivalent Carbohydrate-Lectin Cross-Linking Interactions: Importance of Entropy in the Bind and Jump Mechanism. Biochemistry 2009, 48 (18), 3822–3827. 10.1021/bi9002919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turnbull W. B.; Precious B. L.; Homans S. W. Dissecting the Cholera Toxin-Ganglioside GM1 Interaction by Isothermal Titration Calorimetry. J. Am. Chem. Soc. 2004, 126 (4), 1047–1054. 10.1021/ja0378207. [DOI] [PubMed] [Google Scholar]
- Tran H.-A.; Kitov P. I.; Paszkiewicz E.; Sadowska J. M.; Bundle D. R. Multifunctional Multivalency: A Focused Library of Polymeric Cholera Toxin Antagonists. Org. Biomol. Chem. 2011, 9 (10), 3658–3671. 10.1039/c0ob01089h. [DOI] [PubMed] [Google Scholar]
- Jones M. W.; Otten L.; Richards S.-J.; Lowery R.; Phillips D. J.; Haddleton D. M.; Gibson M. I. Glycopolymers with Secondary Binding Motifs Mimic Glycan Branching and Display Bacterial Lectin Selectivity in Addition to Affinity. Chem. Sci. 2014, 5 (4), 1611–1616. 10.1039/C3SC52982G. [DOI] [Google Scholar]
- Wilkins L. E.; Badi N.; Du Prez F.; Gibson M. I. Double-Modified Glycopolymers from Thiolactones to Modulate Lectin Selectivity and Affinity. ACS Macro Lett. 2018, 7 (12), 1498–1502. 10.1021/acsmacrolett.8b00825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martyn B.; Biggs C. I.; Gibson M. I. Comparison of Systematically Functionalized Heterogeneous and Homogenous Glycopolymers as Toxin Inhibitors. J. Polym. Sci., Part A: Polym. Chem. 2019, 57 (1), 40–47. 10.1002/pola.29279. [DOI] [Google Scholar]
- Moremen K. W.; Tiemeyer M.; Nairn A. V. Vertebrate Protein Glycosylation: Diversity, Synthesis and Function. Nat. Rev. Mol. Cell Biol. 2012, 13 (7), 448–462. 10.1038/nrm3383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Worstell N. C.; Krishnan P.; Weatherston J. D.; Wu H. J. Binding Cooperativity Matters: A Gm1-like Ganglioside-Cholera Toxin b Subunit Binding Study Using a Nanocube-Based Lipid Bilayer Array. PLoS One 2016, 11 (4), e0153265 10.1371/journal.pone.0153265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wands A. M.; Cervin J.; Huang H.; Zhang Y.; Youn G.; Brautigam C. A.; Matson Dzebo M.; Björklund P.; Wallenius V.; Bright D. K.; Bennett C. S.; Wittung-Stafshede P.; Sampson N. S.; Yrlid U.; Kohler J. J. Fucosylated Molecules Competitively Interfere with Cholera Toxin Binding to Host Cells. ACS Infect. Dis. 2018, 4 (5), 758–770. 10.1021/acsinfecdis.7b00085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mandal P. K.; Branson T. R.; Hayes E. D.; Ross J. F.; Gavín J. A.; Daranas A. H.; Turnbull W. B. Towards a Structural Basis for the Relationship between Blood Group and the Severity of El Tor Cholera. Angew. Chem., Int. Ed. 2012, 51 (21), 5143–5146. 10.1002/anie.201109068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ponader D.; Maffre P.; Aretz J.; Pussak D.; Ninnemann N. M.; Schmidt S.; Seeberger P. H.; Rademacher C.; Nienhaus G. U.; Hartmann L. Carbohydrate-Lectin Recognition of Sequence-Defined Heteromultivalent Glycooligomers. J. Am. Chem. Soc. 2014, 136 (5), 2008–2016. 10.1021/ja411582t. [DOI] [PubMed] [Google Scholar]
- García-Moreno M. I.; Ortega-Caballero F.; Rísquez-Cuadro R.; Ortiz Mellet C.; García Fernández J. M. The Impact of Heteromultivalency in Lectin Recognition and Glycosidase Inhibition: An Integrated Mechanistic Study. Chem. - Eur. J. 2017, 23 (26), 6295–6304. 10.1002/chem.201700470. [DOI] [PubMed] [Google Scholar]
- Gade M.; Alex C.; Ben-Arye S. L.; Monteiro J. T.; Yehuda S.; Lepenies B.; Padler-Karavani V.; Kikkeri R. Microarray Analysis of Oligosaccharide-Mediated Multivalent Carbohydrate–Protein Interactions and Their Heterogeneity. ChemBioChem 2018, 19 (11), 1170–1177. 10.1002/cbic.201800037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Godula K.; Bertozzi C. R. Synthesis of Glycopolymers for Microarray Applications via Ligation of Reducing Sugars to a Poly(Acryloyl Hydrazide) Scaffold. J. Am. Chem. Soc. 2010, 132 (29), 9963–9965. 10.1021/ja103009d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang M. L.; Smith R. A. A.; Trieger G. W.; Godula K. Glycocalyx Remodeling with Proteoglycan Mimetics Promotes Neural Specification in Embryonic Stem Cells. J. Am. Chem. Soc. 2014, 136 (30), 10565–10568. 10.1021/ja505012a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toraskar S.; Gade M.; Sangabathuni S.; Thulasiram H. V.; Kikkeri R. Exploring the Influence of Shapes and Heterogeneity of Glyco-Gold Nanoparticles on Bacterial Binding for Preventing Infections. ChemMedChem 2017, 12 (14), 1116–1124. 10.1002/cmdc.201700218. [DOI] [PubMed] [Google Scholar]
- Gloster T. M.; Davies G. J. Glycosidase Inhibition: Assessing Mimicry of the Transition State. Org. Biomol. Chem. 2010, 8 (2), 305–320. 10.1039/B915870G. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith B. A. H.; Bertozzi C. R. The Clinical Impact of Glycobiology: Targeting Selectins, Siglecs and Mammalian Glycans. Nat. Rev. Drug Discovery 2021, 20 (3), 217–243. 10.1038/s41573-020-00093-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prost L. R.; Grim J. C.; Tonelli M.; Kiessling L. L. Noncarbohydrate Glycomimetics and Glycoprotein Surrogates as DC-SIGN Antagonists and Agonists. ACS Chem. Biol. 2012, 7 (9), 1603–1608. 10.1021/cb300260p. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manning D. D.; Hu X.; Beck P.; Kiessling L. L. Synthesis of Sulfated Neoglycopolymers: Selective P-Selectin Inhibitors. J. Am. Chem. Soc. 1997, 119 (13), 3161–3162. 10.1021/ja964046x. [DOI] [Google Scholar]
- Porkolab V.; Chabrol E.; Varga N.; Ordanini S.; Sutkevičiute I.; Thépaut M.; García-Jiménez M. J.; Girard E.; Nieto P. M.; Bernardi A.; Fieschi F. Rational-Differential Design of Highly Specific Glycomimetic Ligands: Targeting DC-SIGN and Excluding Langerin Recognition. ACS Chem. Biol. 2018, 13 (3), 600–608. 10.1021/acschembio.7b00958. [DOI] [PubMed] [Google Scholar]
- Ordanini S.; Varga N.; Porkolab V.; Thépaut M.; Belvisi L.; Bertaglia A.; Palmioli A.; Berzi A.; Trabattoni D.; Clerici M.; Fieschi F.; Bernardi A. Designing Nanomolar Antagonists of DC-SIGN-Mediated HIV Infection: Ligand Presentation Using Molecular Rods. Chem. Commun. 2015, 51 (18), 3816–3819. 10.1039/C4CC09709B. [DOI] [PubMed] [Google Scholar]
- Linclau B.; Ardá A.; Reichardt N.-C.; Sollogoub M.; Unione L.; Vincent S. P.; Jiménez-Barbero J. Fluorinated Carbohydrates as Chemical Probes for Molecular Recognition Studies. Current Status and Perspectives. Chem. Soc. Rev. 2020, 49 (12), 3863–3888. 10.1039/C9CS00099B. [DOI] [PubMed] [Google Scholar]
- Linclau B.; Golten S.; Light M.; Sebban M.; Oulyadi H. The Conformation of Tetrafluorinated Methyl Galactoside Anomers: Crystallographic and NMR Studies. Carbohydr. Res. 2011, 346 (9), 1129–1139. 10.1016/j.carres.2011.04.007. [DOI] [PubMed] [Google Scholar]
- Murray-Rust P.; Stallings W. C.; Monti C. T.; Preston R. K.; Glusker J. P. Intermolecular Interactions of the C-F Bond: The Crystallographic Environment of Fluorinated Carboxylic Acids and Related Structures. J. Am. Chem. Soc. 1983, 105 (10), 3206–3214. 10.1021/ja00348a041. [DOI] [Google Scholar]
- Kumar R.; Ignjatović M. M.; Peterson K.; Olsson M.; Leffler H.; Ryde U.; Nilsson U. J.; Logan D. T. Structure and Energetics of Ligand–Fluorine Interactions with Galectin-3 Backbone and Side-Chain Amides: Insight into Solvation Effects and Multipolar Interactions. ChemMedChem 2019, 14 (16), 1528–1536. 10.1002/cmdc.201900293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oberbillig T.; Mersch C.; Wagner S.; Hoffmann-Röder A. Antibody Recognition of Fluorinated MUC1 Glycopeptide Antigens. Chem. Commun. 2012, 48 (10), 1487–1489. 10.1039/C1CC15139H. [DOI] [PubMed] [Google Scholar]
- Orwenyo J.; Huang W.; Wang L. X. Chemoenzymatic Synthesis and Lectin Recognition of a Selectively Fluorinated Glycoprotein. Bioorg. Med. Chem. 2013, 21 (16), 4768–4777. 10.1016/j.bmc.2013.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richards S. J.; Keenan T.; Vendeville J. B.; Wheatley D. E.; Chidwick H.; Budhadev D.; Council C. E.; Webster C. S.; Ledru H.; Baker A. N.; Walker M.; Galan M. C.; Linclau B.; Fascione M. A.; Gibson M. I. Introducing Affinity and Selectivity into Galectin-Targeting Nanoparticles with Fluorinated Glycan Ligands. Chem. Sci. 2021, 12 (3), 905–910. 10.1039/D0SC05360K. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hook A. L.; Chang C. Y.; Yang J.; Luckett J.; Cockayne A.; Atkinson S.; Mei Y.; Bayston R.; Irvine D. J.; Langer R.; Anderson D. G.; Williams P.; Davies M. C.; Alexander M. R. Combinatorial Discovery of Polymers Resistant to Bacterial Attachment. Nat. Biotechnol. 2012, 30 (9), 868–875. 10.1038/nbt.2316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lynn D. M.; Anderson D. G.; Putnam D.; Langer R. Accelerated Discovery of Synthetic Transfection Vectors: Parallel Synthesis and Screening of a Degradable Polymer Library [20]. J. Am. Chem. Soc. 2001, 123 (33), 8155–8156. 10.1021/ja016288p. [DOI] [PubMed] [Google Scholar]
- Collins C.; Dyer M. S.; Pitcher M. J.; Whitehead G. F. S.; Zanella M.; Mandal P.; Claridge J. B.; Darling G. R.; Rosseinsky M. J. Accelerated Discovery of Two Crystal Structure Types in a Complex Inorganic Phase Field. Nature 2017, 546 (7657), 280–284. 10.1038/nature22374. [DOI] [PubMed] [Google Scholar]
- Pande J.; Szewczyk M. M.; Grover A. K. Phage Display: Concept, Innovations, Applications and Future. Biotechnol. Adv. 2010, 28 (6), 849–858. 10.1016/j.biotechadv.2010.07.004. [DOI] [PubMed] [Google Scholar]
- Stoltenburg R.; Reinemann C.; Strehlitz B. SELEX-A (r)Evolutionary Method to Generate High-Affinity Nucleic Acid Ligands. Biomol. Eng. 2007, 24 (4), 381–403. 10.1016/j.bioeng.2007.06.001. [DOI] [PubMed] [Google Scholar]
- Joseph A. A.; Pardo-Vargas A.; Seeberger P. H. Total Synthesis of Polysaccharides by Automated Glycan Assembly. J. Am. Chem. Soc. 2020, 142 (19), 8561–8564. 10.1021/jacs.0c00751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li T.; Liu L.; Wei N.; Yang J. Y.; Chapla D. G.; Moremen K. W.; Boons G. J. An Automated Platform for the Enzyme-Mediated Assembly of Complex Oligosaccharides. Nat. Chem. 2019, 11 (3), 229–236. 10.1038/s41557-019-0219-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oyelaran O.; Gildersleeve J. C. Glycan Arrays: Recent Advances and Future Challenges. Curr. Opin. Chem. Biol. 2009, 13 (4), 406–413. 10.1016/j.cbpa.2009.06.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horiya S.; Bailey J. K.; Temme J. S.; Guillen Schlippe Y. V.; Krauss I. J. Directed Evolution of Multivalent Glycopeptides Tightly Recognized by HIV Antibody 2G12. J. Am. Chem. Soc. 2014, 136 (14), 5407–5415. 10.1021/ja500678v. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng S.; Lin E.; Kitov P. I.; Tjhung K. F.; Gerlits O. O.; Deng L.; Kasper B.; Sood A.; Paschal B. M.; Zhang P.; Ling C. C.; Klassen J. S.; Noren C. J.; Mahal L. K.; Woods R. J.; Coates L.; Derda R. Genetically Encoded Fragment-Based Discovery of Glycopeptide Ligands for Carbohydrate-Binding Proteins. J. Am. Chem. Soc. 2015, 137 (16), 5248–5251. 10.1021/ja511237n. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sojitra M.; Sarkar S.; Maghera J.; Rodrigues E.; Carpenter E. J.; Seth S.; Ferrer Vinals D.; Bennett N. J.; Reddy R.; Khalil A.; Xue X.; Bell M. R.; Zheng R. B.; Zhang P.; Nycholat C.; Bailey J. J.; Ling C. C.; Lowary T. L.; Paulson J. C.; Macauley M. S.; Derda R. Genetically Encoded Multivalent Liquid Glycan Array Displayed on M13 Bacteriophage. Nat. Chem. Biol. 2021, 17 (7), 806–816. 10.1038/s41589-021-00788-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan M.; Zhu Y.; Liu X.; Lasanajak Y.; Xiong J.; Lu J.; Lin X.; Ashline D.; Reinhold V.; Smith D. F.; Song X. Next-Generation Glycan Microarray Enabled by DNA-Coded Glycan Library and Next-Generation Sequencing Technology. Anal. Chem. 2019, 91 (14), 9221–9228. 10.1021/acs.analchem.9b01988. [DOI] [PubMed] [Google Scholar]
- Lutz J.-F.; Ouchi M.; Liu D. R.; Sawamoto M. Sequence-Controlled Polymers. Science 2013, 341 (6146), 1238149. 10.1126/science.1238149. [DOI] [PubMed] [Google Scholar]
- Schmidt B. V. K. J.; Fechler N.; Falkenhagen J.; Lutz J.-F. Controlled Folding of Synthetic Polymer Chains through the Formation of Positionable Covalent Bridges. Nat. Chem. 2011, 3 (3), 234–238. 10.1038/nchem.964. [DOI] [PubMed] [Google Scholar]
- Kühlbrandt W. The Resolution Revolution. Science 2014, 343 (6178), 1443–1444. 10.1126/science.1251652. [DOI] [PubMed] [Google Scholar]
- Jumper J.; Evans R.; Pritzel A.; Green T.; Figurnov M.; Ronneberger O.; Tunyasuvunakool K.; Bates R.; Žídek A.; Potapenko A.; Bridgland A.; Meyer C.; Kohl S. A. A.; Ballard A. J.; Cowie A.; Romera-Paredes B.; Nikolov S.; Jain R.; Adler J.; Back T.; Petersen S.; Reiman D.; Clancy E.; Zielinski M.; Steinegger M.; Pacholska M.; Berghammer T.; Bodenstein S.; Silver D.; Vinyals O.; Senior A. W.; Kavukcuoglu K.; Kohli P.; Hassabis D. Highly Accurate Protein Structure Prediction with AlphaFold. Nature 2021, 596, 583–589. 10.1038/s41586-021-03819-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walczyk D.; Bombelli F. B.; Monopoli M. P.; Lynch I.; Dawson K. A. What the Cell “Sees” in Bionanoscience. J. Am. Chem. Soc. 2010, 132 (16), 5761–5768. 10.1021/ja910675v. [DOI] [PubMed] [Google Scholar]
- Wan S.; Kelly P. M.; Mahon E.; Stöckmann H.; Rudd P. M.; Caruso F.; Dawson K. A.; Yan Y.; Monopoli M. P. The “Sweet” Side of the Protein Corona: Effects of Glycosylation on Nanoparticle-Cell Interactions. ACS Nano 2015, 9 (2), 2157–2166. 10.1021/nn506060q. [DOI] [PubMed] [Google Scholar]