Abstract
According to the World Health Organization (WHO), 47 million people display mental health disorders Worldwide. In addition, epidemiological studies have shown that the extension of life expectancy and the increase in aged population will significantly impact the prevalence of several mental impairments. Although there are strategies for preventing and alleviating mental illnesses, such as pharmacological and psychological approaches, limited results have been observed. Thus, the search for new therapeutics for managing psychiatric disorders has explored multiple roads. In recent years, it has been demonstrated that physical activity and exercise promote health benefits. On the other hand, among the neurobiological systems that participate in the genesis and development of mental disruptions, the endocannabinoid system has been suggested as an active player. Supporting this hypothesis, data suggest that the elements comprising the endocannabinoid system, such as the CB1/CB2 cannabinoid receptors, endogenous ligands (N-arachidonoylethanolamine [anandamide, AEA] and 2-arachidonoylglycerol [2-AG]), transporters and the enzymes involved in the biosynthesis and degradation of the AEA and 2-AG, modulate mental diseases. In this review, we discuss that the endocannabinoid system might be considered as a modulator for the positive outcomes of exercise in the management of mental disorders. Clinically, this promising field might be exploited by targeting the elements of the endocannabinoid system aimed to increase the exercise benefits applied to patients with mental illnesses.
Keywords: Anandamide, endocannabinoids, physical activity, mental health, exercise, depression
1. INTRODUCTION
The burden of mental disorders continues to grow up world-wide with significant impacts on multiple areas, including health. According to the literature, two key factors seem to participate in the increase of the prevalence of mental diseases: (i) The decline of mortality rates and, (ii) The expansion of life expectancy [1-4]. With the enhancement of aged population, an increase in the number of mental disorders, including depression, bipolar disorders, schizophrenia, and many others is expected in parallel [1, 5, 6].
Although a diversity of treatments aimed for managing mental diseases are currently used, such as pharmacological means and psychosocial support, limited results have been observed [7-10]. Additional efforts have been achieved by using non-pharmacological approaches for treating mental illnesses, including phototherapy, nutritional supplements, and physical exercise [11-14]. In this regard, multiple findings have demonstrated that physical activity and exercise promote health benefits in patients with psychiatric disorders [15-18]. Exercise influences multiple neurobiological networks, including the endocannabinoid system [19-22], therefore, it is likely that this endogenous signaling arrangement might influence the effectiveness of exercise as a treatment for managing mental disorders.
2. THE ENDOCANNABINOID SYSTEM
Cannabis sativa contains over 100 compounds called cannabinoids, among the most abundant are delta-9-tetrahydrocannabinol (Δ9-THC) and cannabidiol (CBD) [23-25]. Whereas Δ9-THC binds to transmembranal receptors to induce multiple neurophysiological effects, the mechanism of action of CBD remains to be fully described [23, 26, 27]. With the characterization of the receptors that recognized Δ9-THC, named CB1 and CB2 cannabinoid receptors, the search for their natural endogenous ligands was driven during the 1980s-1990s until the discovery of lipids that were produced by the mammalian body that naturally bind to the cannabinoid receptors. These endogenous compounds, anandamide (arachidonoylethanolamine [anandamide, AEA]) and 2-arachidonoylglycerol (2-AG) were eventually named endocannabinoids [28, 29]. Further elements were added to the endocannabinoid system family, including enzymes responsible for the synthesis and degradation of AEA and 2-AG, transporters, and additional endogenous ligands [30, 31].
At this date, the endocannabinoid system is a complex signaling network comprised of at least two G-protein coupled receptors, the CB1 and CB2 cannabinoid receptors, their endogenous ligands (AEA and 2-AG, as the most studied so far), and the enzymes involved in the synthesis and degradation of AEA and 2-AG, as well as a membrane transporter [28-31]. Although the description of the functioning of the endocannabinoid system is complex and goes beyond the scope of this review, briefly (Fig. 1) AEA is generated from its membrane precursor, N-arachidonoyl phosphatidylethanolamine (NAPE), through cleavage by a phospholipase D (NAPE–PLD) whereas AEA´s degradation includes the engagement of ethanolamine and arachidonic acid via the activity of the fatty acid amide hydrolase (FAAH) enzyme. It has been suggested that once synthesized, AEA crosses the cellular membrane via the action of the anandamide membrane transporter (AMT).
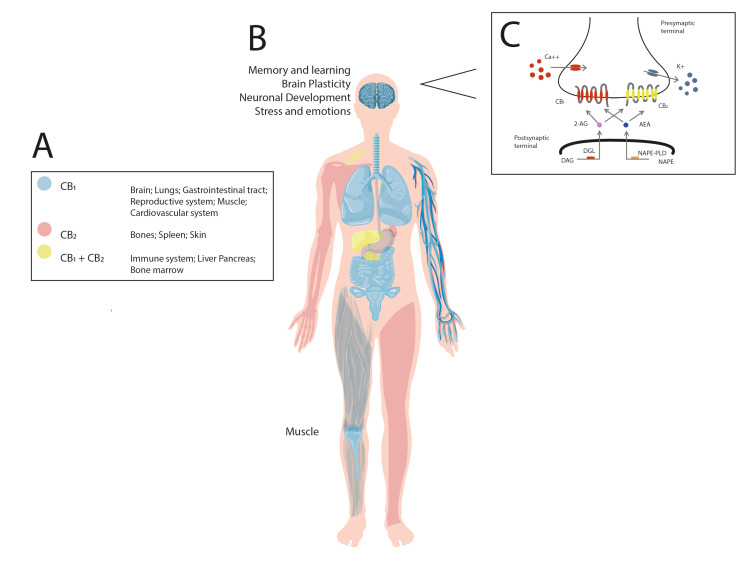
Fig. (1).
The endocannabinoid system. The drawing represents the distribution in human body of the endocannabinoid components, including the CB1 and CB2 cannabinoid receptors, as well as the biosynthesis and degradation enzymatic routes for arachidonoylethanolamine (anandamide, AEA) or 2-arachidonoylglycerol (2-AG) governed by the fatty acid amide hydrolase (FAAH) and monoacylglycerol lipase (MAGL), respectively. Once synthetized, AEA and 2-AG bind to the cannabinoid receptors via the involvement of membrane transporter (for AEA, anandamide membrane transporter [AMT]). As shown, the presence of the cannabinoid receptors in multiple physiological systems such as brain, lung, gastrointestinal tract, etc. (Panel A) suggest the modulatory role of both receptors in the control of a diversity of complex functions, including memory and learning, brain plasticity, neuronal development, stress and emotions, among many others (Panel B). In addition, the image illustrates that arachidonic acid-containing diacylglycerol (DAG), diacylglycerol lipase (DAGL) synthesizes 2-AG whereas NAPE (N-arachidonoyl phosphatidylethanolamine) and N-acyl phosphatidylethanolamine phospholipase D (NAPE-PLD) participates in the formation of AEA. Once released, both lipids (2-AG and AEA) bind and activate the cannabinoid receptors, which in turn, promotes calcium (Ca2+) influx and potassium (K+) efflux for modulating neurotransmission (Panel C). (A higher resolution/colour version of this figure is available in the electronic copy of the article).
On the other hand, 2-AG is formed by the activity of phospholipase C (PLC) and diacylglycerol lipase (DAGL), whereas its degradation is mediated by monoacylglycerol lipase (MAGL) [32, 33]. Once synthetized, AEA and 2-AG bind to the cannabinoid receptors for triggering a diversity of intracellular responses, such as increasing Ca2+ influx and decreasing K+ outflux, leading to a short- or long-term suppression of the release of neurotransmitters.
The distribution of the endocannabinoid system elements (i.e., the CB1 and CB2 cannabinoid receptors) in the whole body, including the central nervous system, has been a point of interest in the function of memory and learning, brain plasticity, neuronal development, modulation of stress and emotions, among many others [34-37] (Fig. 1). The activation of the endocannabinoid system has been recently recognized as an important modulatory network for controlling the function of the brain by regulating the secretion of a diversity of neurochemicals.
3. A COMPREHENSIVE OVERVIEW OF MENTAL HEALTH DISORDERS
Mental disorders are a wide term considering health complex problems characterized by patterns of behaviors with dysfunctional processes of thoughts, emotions, or perceptions [38, 39]. In addition, the mental illnesses include specific symptoms and signs inducing distress or limiting personal functioning in multiple areas of life [40]. The compendium of mental disruptions comprises depression, anxiety disturbances, post-traumatic stress disorder (PTSD), and schizophrenia, among many others. The major categories of mental disturbances described in the latest edition of the Diagnostic and Statistical Manual of Mental Disorders Fifth Edition provide standardized diagnostic criteria are also present in the eleventh edition of the International Classification of Diseases [41, 42]. Further studies have revealed interesting interactions between mental health disorders and neurobiological signaling, including the endocannabinoid system [43-48].
4. THE MODULATORY ROLE OF THE ENDOCANNABINOID SYSTEM IN MENTAL DISORDERS
Basic and clinical studies have explored the causal relationship and likely involvement of the endocannabinoid system in the genesis and development of mental disturbances [49-54]. In the following sections, we provide a broad revision of the current knowledge in the field of neurobiological role of the CB1/CB2 cannabinoid receptors, AEA and 2-AG, and the enzymes NAPE, NAPE–PLD, FAAH, AMT, MGL in mental health issues. However, given the expansion of the knowledge in the area, it is indeed ambitious to describe all the experimental findings related to the modulatory properties of the endocannabinoid system on psychiatric disturbances. Thus, we highlight the current comprehension of the functional role of the endocannabinoid system in common mental health disorders, including depression, anxiety, PTSD, and schizophrenia [1, 55-57].
4.1. Cannabinoid Receptors and Depression, Anxiety, PTSD, and Schizophrenia
4.1.1. Cannabinoid Receptors and Depression
Several studies suggest the putative role of the cannabinoid receptors in the modulation of mental health disorders such as depression (Table 1). For instance, administrations of the inverse agonist CB1 cannabinoid receptor, rimonabant, induces symptoms of anxiety and depression and even suicidal ideation [58-60]. Conversely, antidepressive effects have been found by using CB1 cannabinoid receptor antagonists [61-63]. In line with the current pharmacological findings, mice lacking the CB1 cannabinoid receptor display abnormal patterns at different behavioral paradigms associated with mood disorders. These alterations include a wider spectrum of neurobiological variables, from disturbed molecular functions to dysregulation of neurotransmitter systems related to depression [64-66].
Table 1.
Effects of the elements of the endocannabinoid system on the modulation on depression, anxiety, post-traumatic stress disorder (PTSD), and schizophrenia. Further studies have described the physiological role of the cannabinoid receptors, AEA, 2-AG, the enzymes engaged in the biosynthesis and degradation of the endocannabinoids (NAPE, NAPE-PLD, FAAH, AMT, DAGL, and MAGL) on control depression, anxiety, PTSD, and schizophrenia.
| - | Depression | Anxiety | PTSD | Schizophrenia |
|---|---|---|---|---|
| Cannabinoid receptors | The activation of the CB1 cannabinoid receptor induces depression-like behavior The role of the CB2 cannabinoid receptor in depression is unknown |
The absence of the CB1 cannabinoid receptor increases depression-like behavior The role of the CB2 cannabinoid receptor in anxiety is unknown |
Th activation of the CB1 cannabinoid receptor reduces PTSD symptoms The engagement of the CB2 cannabinoid receptor role in PTSD is unknown |
The levels of the CB1 cannabinoid receptor mRNA in schizophrenia is decreased The role of the CB2 cannabinoid receptor role in schizophrenia is unknown |
| AEA and 2-AG | The levels of the endocannabinoids in depression are decreased | Stress reduces levels of the endocannabinoids | The contents of the endocannabinoids in PTSD are decreased | The levels of the endocannabinoids in schizophrenia are increased |
| NAPE, NAPE-PLD, FAAH, AMT, DAGL, and MAGL | The NAPE-PLD contents are decreased in depression The FAAH gene has been related to depression The FAAH levels are enhanced in depression The DAGL contents are decreased in depression The MAGL levels are increased in depression The AMT activity in depression is unknown |
The blockade of FAAH and MAGL activity decreases anxiety-like behavior The role of NAPE, NAPE-PLD, AMT, DAGL, and MAGL in depression is unknown |
Inconclusive results of role of FAAH in PTSD The role of NAPE, NAPE-PLD, AMT, DAGL, and MAGL in PTSD is unknown |
The FAAH and MAGL levels are enhanced in schizophrenia The NAPE and DAGL contents are decreased in schizophrenia The NAPE-PLD and AMT levels in schizophrenia are unknown |
In conclusion, the current evidence supports the hypothesis that the CB1 cannabinoid receptor plays an important role in depression and suggests that modulation of the activity of this receptor might be a pharmacological target for novel
antidepressant therapeutical approaches for treating mood disorders. Moreover, the comprehension of the mechanism of action of the CB1 cannabinoid receptors on depression has fostered the development of synthetic compounds that regulate the function of these receptors; howbeit, targeting the CB1 cannabinoid receptors only in those target brain areas where there is a presumably perturbed mood-related function will be the new challenge in future studies [67, 68]. While a possible therapeutic use of the CB1 cannabinoid receptor in depression has been discussed in this review, the findings in regards the engagement of the CB2 cannabinoid receptor in mood disturbances should be determined.
4.1.2. Cannabinoid Receptors and Anxiety
Since the CB1 cannabinoid receptor has been localized in several brain areas, including the amygdala, the activation of this receptor has been related to a modulatory function of aversive memories [69-71]. For example, the CB1 cannabinoid receptor knockout mice show an increase in anxiety-like behavior [72]. In addition, pharmacological experiments have demonstrated that the activation of the CB1 cannabinoid receptor precipitates the episodes of anxiety, whereas its blockade exerts anxiety-related behaviors [73-75] (Table 1).
Thus, since the CB1 cannabinoid receptor is widely localized in the central nervous system, then it displays a complex signaling network that responds to different modes of synaptic neurotransmission modulation. For instance, the high levels of the CB1 cannabinoid receptors on inhibitory (GABAergic interneurons) and at a lesser extent on excitatory (glutamatergic) terminals [76], as well as on dopamine D1-expressing neurons, play a modulatory role on different emotional behaviors such as social and cognitive activity, which are affected in mental health illness [77-79]. This is why such psychiatry disorders are modulated by this complex circuitry expressed at some synapses in all brain regions related to the processes of stress. Finally, and because the localization of the CB2 cannabinoid receptor has been widely described outside of the central nervous system [37], no current data in regards to the role of this receptor in anxiety is available.
4.1.3. Cannabinoid Receptors and PTSD
The management of PTSD includes pharmacological intervention by using diverse compounds, such as antidepressive drugs [80]. However, in recent years, it has been studied the likely neurobiological role of the CB1 cannabinoid receptors as a therapeutic element for the management of PTSD (Table 1). In this regard, the very first evidence showing the specific involvement of the CB1 cannabinoid receptors in neurons expressed dopamine D1 receptors in extinction of aversive memories was reported from Carsten T. Wotjak´s laboratory [79, 81]. The same group reported for the first time that the enhancement in contents of AEA controlled acute fear relief, while the increase in 2-AG levels promoted the expression of conditioned fear primarily via the involvement of the CB1 cannabinoid receptor on GABAergic neurons [82]. Further advances have been achieved in the field. For example, the administration of nabilone, a synthetic CB1 cannabinoid receptor agonist, induces positive outcomes for the treatment of PTSD since 72% of participants reported total cessations of severity of the symptoms of this disease [83]. Complementary studies have demonstrated that using in vivo imaging studies in subjects with PTSD, there was an increase in the CB1 cannabinoid receptor availability [84]. Despite the psychiatric side effects of the administrations of rimonabant such as anxiety, depression and suicidal tendencies, there is still interest in the development of CB1 cannabinoid receptor antagonists, including TM38837, as a pharmacological approach for the management of fear. In this regard, Micale and coworkers (2019) reported that TM38837, given either per os (100mg/Kg) or icv (10 or 30μg), increased fear response in mice subjected to tone fear conditioning paradigm. Importantly, authors reported that pharmacological effects induced by TM38837 were only at a dose 10 times higher than rimonabant [85]. Due to the distribution of the CB1 cannabinoid receptors in the central nervous system, it became a target over the CB2 cannabinoid receptors, which have been mapped at peripheral sites [34-37]; however, their localization may not imply direct neural connections between the external tissues -indeed several body regions might not directly innervate to the brain centers involved in fear control. Instead, these conditions might be proposed as hypothesis-generating, addressing future likely functional connectivity studies.
Finally, PTSD is a complex clinical outcome of experiencing a severe emotional trauma, and importantly, not all subjects exposed to the same traumatic experience develop PTSD; the existence of individual susceptibility conditions has been suggested [86, 87]. Thus, the research of individual susceptibility on animal models that mimic the symptoms of PTSD has a limited coherent framework. Indeed, more studies are needed to explore the functional role of the CB1 or CB2 cannabinoid receptors on PTSD, considering the individual susceptibility and resilience factors.
4.1.4. Cannabinoid Receptors and Schizophrenia
Since the wider distribution of the CB1 cannabinoid receptors is present in the central nervous system [36, 38], then it has been suggested to display critical functional properties in the regulation of mental disorders, including schizophrenia. From imaging studies to pharmacological experiments and genetic analyses, the current data suggest that the CB1 cannabinoid receptor is engaged in the genesis, development, and control of schizophrenia [88-92] (Table 1). A closer look at the studies shows a significant decrease in the CB1 cannabinoid receptor mRNA levels in postmortem brain of subjects that displayed schizophrenia [93], whereas autoradiographic studies of the same receptor have shown higher binding in the prefrontal cortex in schizophrenic subjects [94, 95]. In addition, recent pieces of evidence have demonstrated that there was an epigenetic alteration of the CB1 cannabinoid receptors in the peripheral blood mononuclear cells collected from schizophrenic patients. Moreover, data suggested that due to the transcriptional regulation of the CB1 cannabinoid receptor found in schizophrenic subjects through the DNA methylation might be considered as a biomarker for the disease [96]. Similar findings were reported by Stark, et al. (2019) using an animal model of schizophrenia, induced by prenatal methylazoxymethanol acetate (MAM) exposure. According to the report, animals under the MAM experimental condition showed a reduction in DNA methylation at the CNR1 promoter, which was associated with an enhancement in the CB1 cannabinoid receptor gene and protein expression. Moreover, the changes observed in the CB1 cannabinoid receptor matched with the negative symptoms displayed by the MAM rats [97].
Lastly, in a review, Kucerova et al. (2014) discussed the preclinical and clinical findings linked to the dysfunction of the CB1 cannabinoid receptors in schizophrenia. In addition, the authors summarized the promising therapeutic approaches for treating schizophrenia by using CB1 cannabinoid receptor antagonists/inverse agonists, such as AVE1625, which seems to ameliorate the positive-like symptoms in psychotomimetic-induced hyperactivity and latent inhibition deficit models. Notwithstanding, it is recognized that the involvement of the CB2 cannabinoid receptors in schizophrenia still lacks sufficient experimental evidence [92].
4.2. Endocannabinoid Levels and Depression, Anxiety, PTSD, and Schizophrenia
4.2.1. Endocannabinoid Levels and Depression
Experimental and clinical studies aimed to characterize the levels AEA and 2-AG in depression have found lower profiles of these lipids in such mental conditions [98-102] (Table 1). These associative data have been consistent with pharmacological studies, which have demonstrated that targeting the endocannabinoid endogenous tone by enhancers or inhibitors of AEA or 2-AG seem to exert positive therapeutic outcomes for mental disorders [103, 104]. Given the recent discovery of new lipids with cannabimimetic properties, such as N-arachidonoyl dopamine (NADA) and virodhamine [105], then, limited current data are available in regards to their involvement in depression.
4.2.2. Endocannabinoid Levels and Anxiety
The endocannabinoids exert effects on emotional functions since their contents have been described in recent years [106-110]. For instance, exposition to stressful conditions causes anxiety-like behavior and reduces AEA brain contents by enhancing the activity of the FAAH in the amygdala [106]. Although the knowledge about new elements belonging to the endocannabinoid system has brought advanced insights, including the characterization of NADA and virodhamine [105], no evidence is available regarding their neurobiological role on the modulation of anxiety (Table 1).
4.2.3. Endocannabinoid Levels and PTSD
Fascinating data have been reported in PTSD since the levels of the endocannabinoids are decreased if the traumatic experience occurred during childhood. Further complexity to the phenomena is added due to other variables, such as the age of the subject, which play a critical contribution in the likely role of the endocannabinoids and PTSD [110, 111]. Recent discoveries have reported the likely link among the contents of endocannabinoids and the severity of PTSD [111-113]. However, further studies are needed for describing the mechanism of action of AEA or 2-AG into the regulation of PTSD (Table 1).
4.2.4. Endocannabinoid Levels and Schizophrenia
Most of the findings point out that there is an increase in the levels of endocannabinoids in schizophrenic patients (Table 1). Data from several biological samples have reached out similar conclusions [113-116]. Due to that the descriptive studies do not allow to discriminate whether the disrupted levels of the endocannabinoids are cause or consequence of schizophrenia; further studies still are needed to describe the mechanism of action of AEA and 2-AG in the establishment of schizophrenia. Moreover, the participation of additional components of the endocannabinoid signaling, including NADA and virodhamine, in schizophrenia, remains to be addressed as well.
4.3. The Engagement of NAPE, NAPE-PLD, FAAH, AMT, MGL, and DAGL in Depression, Anxiety, PTSD, and Schizophrenia
4.3.1. Enzymes Involved in the Functioning of the Endocannabinoid System Participating in Depression
As described previously, the route for AEA synthesis engages the activity of NAPE-PLD, whereas 2-AG is synthesized from membrane phospholipids via sequential activation of the PLC as well as DAGL. Following their release, AEA and 2-AG´s neurochemical functions are terminated through their subsequent hydrolysis by the activity of either the FAAH or MAGL, respectively [116-118]. In recent years, data suggest that the enzymatic activity of FAAH, DAGL, or MAGL is associated with mental alterations [119-121] (Table 1). For example, a polymorphism of the FAAH gene has been associated with depression [122]. In line with this finding, Vinod et al., (2012) reported that Wistar Kyoto rat strain, a genetic model of depression, displayed higher levels of FAAH in frontal cortex and hippocampus [123]. Similar results were published by Smaga et al., (2017) since the authors found that using an experimental model of depression, the removal of olfactory bulb, caused a decrease in the levels of NAPE-PLD, MAGL whereas contents of FAAH and DAGL were enhanced [124]. Although some achievements have been made in the field of the engagement of the enzymes involved in the functioning of the endocannabinoid system in depression, it remains to be described whether the pharmacological manipulation of the activity of NAPE, NAPE–PLD, FAAH, AMT, MGL, and DAGL might cause positive effects for managing depression.
4.3.2. Enzymes Involved in the Functioning of the Endocannabinoid System Participating in Anxiety
Anxiety is also a mental disorder that has been associated with the dysregulation of the endocannabinoid system (Table 1). For example, the knockout mice lacking DAGL show significant behavioral alterations such as reduction in the exploration of the open field as well as anxiety-related behaviors [125]. Similar findings have been reported when FAAH or MAGL activity is blocked by pharmacological means. In this regard, the inhibition of the enzymatic activity of either FAAH or MAGL has been associated with reducing anxiety-like behaviors [126, 127]. Despite these interesting results, it is worthy to mention that the blockade of FAAH or MAGL promotes an endogenous increase of AEA and 2-AG, respectively. Thus, the link among the functioning of these enzymes and anxiety modulation might be an indirect effect since it is likely that outcomes might be the result of the influence of the enhanced levels of AEA and 2-AG. Whereas whether NAPE, NAPE–PLD, and AMT participate in anxiety, remains to be studied.
4.3.3. Enzymes Involved in the Functioning of the Endocannabinoid System Participating in PTSD
Few findings are available in regards to the role of NAPE, NAPE–PLD, FAAH, AMT, MAGL, and DAGL on PTSD [128]. Among the current data, Vimalanathan et al., (2020) used an experimental model or PTSD finding that the blockade FAAH activity by URB597 did not reduce the anxiety-like behavior in experimental models [129]. However, this result is not in concordance to previous reports since other groups have shown that chronic administrations of URB597 (0.2, 0.3, 0.4 mg/kg, i.p.) controlled PTSD symptoms [130]. The differences in results among comparative studies, including experimental design, dosage, animal strain, limit to drawing solid conclusions in regards to the role of the enzymes engaged in the biosynthesis and degradation of the endocannabinoids in the modulation of PTSD. As one can assume, future studies are requested to address the functional role of NAPE, NAPE–PLD, FAAH, AMT, MAGL, or DAGL in PTSD (Table 1).
4.3.4. Enzymes Involved in the Functioning of the Endocannabinoid System Participating in Schizophrenia
From imaging studies to pharmacological experimental reports, current findings suggest that the enzymes involved in the synthesis and degradation of endocannabinoids play a modulatory role in schizophrenia [131, 132]. For example, patients with the first episode of psychosis showed an increase in the expression of FAAH and MAGL, whereas a decrease in the levels of NAPE and DAGL was found as compared to healthy controls [133]. Due to the relative recent focus of studying the importance of the function of NAPE, NAPE-PLD, FAAH, AMT, MAGL or DAGL on schizophrenia, limited data are available. We hope that in the future, data from basic and clinical trials describing the importance of NAPE, NAPE–PLD, FAAH, AMT, MAGL or DAGL on schizophrenia might consider these enzymes as biological markers to allow earlier diagnosis and treatments of patients with schizophrenia (Table 1).
5. BENEFITS OF PHYSICAL EXERCISE IN MENTAL DISORDERS
Mental disorders represent a significant economic cost for society and health care system [134]. Globally, an estimated 264 million people suffer from depression, 284 million subjects complain about anxiety disorders, whereas 20 million patients present schizophrenia [1]. Remarkably, the burden of mental disorders continues to grow causing serious impacts on additional areas such as social relationships and economics around the world [135]. Further complexity in mental disorders includes the impact of comorbidity of suffering from various diseases simultaneously [136]. In addition, mental disabilities are commonly linked to non-communicable chronic illnesses, such as cardiovascular diseases, type 2 diabetes, cancer, and chronic respiratory, among many other health issues [137]. The causes of mental disruptions comprise genetic and external influences [138-144], where the lifestyle seems to impact the onset and development of psychiatric disturbances. For example, low rates of exercising along with higher rates of smoking induce mental disorders [145, 146]. Thus, as we can assume, external factors also exert an influence on the genesis and development of mental diseases.
Mental disorders have been tackled using several treatments, such as cognitive behavior therapy and pharmacological interventions [147-149]. Interestingly, physical activity and exercise have shown to produce benefits in the prevention and the delaying of the onset of several psychiatric disorders [150]. For the purpose of the current review, we would like to provide the following definitions of physical activity and exercise. While physical activity is defined as any bodily movement produced by the contraction of skeletal muscles, being an umbrella term that includes subcategories like sports, leisure activities, dance, exercise is understood as every planned, structured, repetitive, and purposeful intervention [151, 152]. It is worthy to mention that exercise is always physical activity, but physical activity is not necessarily an exercise [152, 153].
There are data supporting that insufficient physical activity is one of the leading risk factors for death worldwide [154]. Between 60 to 85% of people in the world show sedentary lifestyles leading to health complications. In this era of exponential growth of sedentarism, the implementation of lifestyle modifications might be a cost-effective way to improve the health status of the population. Moreover, the benefits of regular physical activity or exercise on mental disorders have been previously demonstrated [1]. Since exercise has been linked to neuronal processes such as neurogenesis and synaptogenesis, then the positive effects of exercise have been associated with such neurobiological phenomena leading to promote additional outcomes in cognitive, emotional, and behavioral measurements [155]. For example, exercise interventions seem to facilitate certain brain areas functioning, inducing well-being and controlling mental disorders such as depression and anxiety [156]. The positive impact of exercise on mental health has been recently suggested as a therapeutic mean for the management of mental impairments during pandemic periods [157, 158]. In sum, the available literature suggests that exercise produces benefits in health and these strategies might be considered as therapeutic elements for managing mental disorders.
5.1. Exercise and Depression
The association between exercise and depression has been addressed since the early 1900s. Several studies addressing depression and exercise include case studies, cross-sectional designs, meta-analyses, and systematic reviews of meta-analyses, among many other approaches. Currently, the consensus accepts the positive outcomes of the treatment of depressive symptoms with exercise interventions [159, 160]. However, some findings support that subjects suffering from depression tend to be less physically active than healthy peers [161, 162]. This phenomenological issue might limit the exerting positive effects of exercise in patients with depression. In line with this perspective, Rethorst et al., (2009) reported that participants displaying depression showed significantly lower depression-related scores than the control group after acute exercise [163]. Similarly, significant benefits in the relief from depressive disorders were found after chronic exercise interventions in children, adolescent and adult subjects [164-166]. Moreover, there is cross-sectional data of the inverse association among physical inactivity and mental health. The Aerobics Center Longitudinal Study (ACLS) [167] evaluated the associations between physical activity and mental health, in which a lower depressive symptomatology and greater emotional well-being was found in physically active subjects. It is worthy to mention that the type and mode of exercise interventions varies between studies, leading to suggest that the impact of exercise might depend on the kind and method of exercise applied. However, aerobic exercise has been the most widely implemented intervention in depression [164-168]. Nevertheless, the efficacy of other types of exercise such as resistance/strength has been demonstrated as well [169]. In this regard, endurance and resistance exercise interventions displayed equally effective outcomes in the amelioration of depressive symptoms [163-170] and the combination of both types of exercise in the same intervention resulted in larger effects than practicing endurance exercise or resistance exercise separately [163].
On the other hand, the length of the interventions in exercise is a variable that plays a critical role as part of the therapeutic effects of exercise in depression. For example, acute bouts of exercise as well as chronic exercise interventions have showed positive outcomes in the remission of depressive symptoms and mood related disorders. In line with this observation, Bartholomew et al., (2005), focusing on patients with major depressive disorder, demonstrated that a single bout of moderate-intensity aerobic exercise (30 min of moderate-intensity treadmill training) exerted greater effects on depression than a 30 min period of quiet rest [171]. Furthermore, single studies with chronic exercise interventions also demonstrated validity to ameliorate depressive symptoms. The SMILE study [172] describes a randomized controlled trial that included 202 adults with major depressive disorder and randomized them to one of four conditions during 16 weeks: Condition 1= Supervised exercise in a group setting (45 min sessions, 3 times per week including 10 min of warm-up, 30 min of walking or jogging on a treadmill at an intensity range between 70-88% of their maximum heart rate [HRmax], and 5 min of cool-down); Condition 2= Home-based exercise (with participants receiving the same exercise prescription but exercised at home on their own with minimal contact from the study staff); Condition 3= Antidepressant medication and Condition 4= Placebo pill. The remission rates of depressive symptoms were found as follows: Condition 1: Supervised exercise group= 45%; Condition 2: Home-based exercise group= 40%; Condition 3: Medication group= 47%; Condition 4: Placebo pill group= 31%. Similar findings have been reported in the Depressed Adolescents Treated with Exercise (DATE) study [173] since vigorous exercise intervention (>12 Kcal/kg/week [KKW]) or a control stretching group (< 4 KKW) for 12 weeks showed a decrease in depressive symptoms. In addition, Brand et al., (2018) studied 129 inpatients from a psychiatric hospital suffering different mental disorders and showed that a single session at a moderate intensity of Nordic walking, workout/ gymnastics or ball sports produced benefits on mood [174].
Despite the promising findings of exercise in depression, further complexity has been added to the interventions since additional variables play an active role in the modulation of depression. For example, the age of the subjects as well as the intensity of the exercise exert influence in the remission of depression [175]. In this regard, there is still no agreement on the precise exercise intensity to achieve the best dose-response, with intensities ranging from high to moderate or from moderate to mild for endurance exercise interventions [176, 177]. Besides, limited evidence is available regarding the influence that exercise intensity exerts on depressed mood [178]. Although most of the literature reports the positive effects on the mental health of moderate intensity in the exercise interventions, there is a need for developing research defining the dose-response relationship and prescription of exercise for the improvement in depression [179]. Taking together, we conclude that results have demonstrated the efficacy of exercise interventions in patients with major depressive disorders and outcomes from these non-invasive treatments seem to be comparable to the use of antidepressant medications.
5.2. Exercise and Anxiety
Anxiety disorders have been considered the most frequent mental disorders (14.0%) in European countries across all age groups [180], as well as the most common mental disorder in the USA, affecting more than 25 million subjects [181]. Anxiety is managed by using psychological programs as well as pharmacological treatments, including the prescription of antidepressant [182-184]. However, these therapeutic approaches show limited results. The search for novel medical options for managing anxiety has focused on exercise. In this regard, a reduction of anxiety symptoms when comparing exercise programs with no-treatment has been demonstrated [184-189]. However, contradictory data have been published since non-significant benefits of exercise on anxiety symptoms have been observed as well. For example, a bout of 15 min running at a moderate intensity (65-75% of their HRmax) did not produce significant improvements in anxiety symptoms as well as in cortisol levels [165]. Thus, the research of the influence of exercise on anxiety disorders remains to be explored in detail.
5.3. Exercise and PTSD
Only a few randomized controlled trials have been reported studying the impact of exercise programs on PTSD. Despite the limitation of available data in this field, the results are encouraging. For example, a 12-week exercise program showed positive outcomes in adults suffering with PTSD [190, 191]. The exercise intervention group displayed a reduction of PTSD and depressive symptoms as well as an improved sleep quality in comparison with the control group only receiving the usual PTSD care. Complementary studies using endurance exercise interventions showed positive outcomes on PTSD symptoms [192].
As mentioned previously, the lack of data regarding the influence that chronic exercise interventions have on PTSD, as well as on the response of biochemical markers, including cortisol, highlights the need to explore the involvement of physical activity and exercise on the modulation of PTSD via the engagement of biochemical markers. Researchers suggest that after 8-week exercise, intervention promoted changes in the cortisol and endocannabinoid contents in juvenile subjects [193]. Whether the physical activity or exercise interventions might exert log-term effects on the symptoms in PTSD remains to be elucidated.
5.4. Exercise and Schizophrenia
The benefits that different exercise interventions induce on patients with schizophrenia have been reported [184]. For instance, Oertel-Knöchel et al., (2014) showed that a combination of 30 min of cognitive training followed by 45 min of endurance exercise that included 10 min warm-up, 25 min of circuit training at an intensity of 60-70% of their HRmax, and 10 min of cool-down, performed 3 times per week for 4 weeks, induced a positive effect on cognitive performance and individual psychopathology on both, depressive and schizophrenic patients [194]. Additional data have shown that 12 weeks of endurance training (cycling) decreased the severity of schizophrenia symptoms in comparison with the control group that played table football. The exercise group attended 30 min sessions, 3 times per week at an intensity corresponding to a blood lactate concentration of about 1-5 to 2mmol/L [195]. Similar results were found by Wang et al. (2018) on the symptomatology of adult schizophrenic patients receiving antipsychotic treatment. The participants were randomly assigned to a high-intensity endurance intervention (exercise training that included 40 min sessions with 5 min of warm-up (walking), 30 min of exercise, and 5 min of cool-down) or a control group attending a low-intensity stretching program that included, among others, flexibility and balance exercises. The authors found that severities of schizophrenic symptoms were diminished in patients that attended the endurance exercise intervention [196]. Regarding the exercise intensity in both groups, the authors stated that the intensity of the endurance intervention was based on each individual´s age adjusted HRmax. However, the exact intensity was not specified for any of the two groups. Moreover, Gorczynski and Faulkner (2010) examined the health benefits of exercise in schizophrenic patients and found that in spite of the small number and size of the randomized controlled trials and the implementation of various measures of physical and mental health, regular exercise programs shows beneficial effects on schizophrenia [197]. However, it remains to tackle some limitations of considering exercise as part of medical programs for managing schizophrenia since patients suffering from this mental disease tend to have less-active lifestyles compared to healthy controls [198].
Finally, Duraiswamy et al., (2007) compared the effects of a 4-month exercise or yoga intervention in schizophrenic patients. The exercise intervention included brisk walking, jogging and several exercises in standing posture [199]. The yoga group performed an integrated yoga treatment. Both interventions included 15 days of directed training (60 min sessions, 5 days per week for 3 weeks) and 3 months of auto-training following the same sequence. Results showed a reduction in the symptoms of schizophrenia in both groups after the intervention.
Additional research is needed to elaborate a comprehensive hallmark of the positive therapeutic effects of exercise on schizophrenia. Moreover, the integration of current knowledge of the diversity of methodological approaches, including sample sizes, proper control groups, evaluation tools, physical exercise and training variables such as type, mode, volume, frequency, and different approaches prescribing physical interventions regarding dose-response relationship is demanded [179, 200].
6. MODULATION OF THE ENDOCANNABINOID SYSTEM AS MEDIATOR FOR ENHANCING THE BENEFITS OF EXERCISE ON MENTAL HEALTH
Although the exercise modulates mental disturbances, the neurobiological mechanism involved in this phenomenon remains to be described in detail [201]. Multiple explanations have been offered for the understanding of the positive effects of exercise by modulating brain systems and in turn, promoting mental health [202]. Amongst the neurobiological networks that respond to exercise, the endocannabinoid system is included [203]. Remarkably, the research on the field has situated the endocannabinoid system as one of the possible moderators through which exercise may cause benefits on mental disturbances [19, 20, 204]. Following this idea, current findings have demonstrated an increase in the levels of the endocannabinoids after acute sessions of exercise. Elevated AEA contents were found in runners and cyclists but not in sedentary controls after 1 hour of moderate intensity exercise (70-80% of their HRmax), whereas 2-AG displayed no significant changes [204]. Similar results were reported by posterior studies where the levels of AEA were increased after 30 min of treadmill running (70-80% of their HRmax) in a posterior study [205]. Another study showed that AEA levels also increased in a group of cyclists during intense exercise (60min at 55% of their maximal power output [Wmax] followed by 30 min at 75% of their Wmax) and in the 15 min of recovery, whereas 2-AG concentrations remained stable [206]. However, the exercise-induced psychological modifications modulated, among others, by endocannabinoid signaling changes might be dependent on the intensity of the intervention [207]. In this regard, high-intensity running for 30 min promoted a significant increase in exercise-induced contents of AEA, while low-intensity walking for 30 min caused no significant variations of AEA levels [205]. Furthermore, the contents of the endocannabinoids were described after four different treadmill running intensities (walk= <50%, light jog= 70%, moderate-intensity run= 80%, and high-intensity run= 90% of the HRmax) in a posterior study [207]. The results showed that the two exercise interventions with a moderate intensity (70-80% of the HRmax) promoted variations in the levels of AEA. In a similar line, a sample of female subjects suffering from depression that exercised in separate sessions for 20 min on a cycle ergometer at both moderate-intensity or preferred-intensity, only found an enhancement in the AEA levels after the moderate-intensity session [21]. The intensity was established to be a “13” in the rating of perceived exertion (RPE), according to Borg et al. (1998) rating scale from 6 to 20 for the moderate-intensity session. It is worthy to mention that intensity was freely manipulated by the participants in the preferred-intensity session [208]. The reported heart rates of both groups (M = 126.7; SD = 20.8 for the moderate-intensity and M = 131.1; SD = 28.7 for the preferred-intensity group) as well as the RPE (M = 13.1; SD = 0.18 for the moderate-intensity and M = 12.7; SD = 1.9 for the preferred-intensity group) did not show significant differences between groups. According to the authors, the intrasubject variability in the way participants exercised during the preferred-intensity session (both lower or harder intensity compared to the compulsory moderate intensity) might hide the potential effect of exercise if the intensity is a necessary threshold to elicit exercise-induced changes in endocannabinoids [21]. Comparable studies have shown significant increases in circulating endocannabinoids (AEA and 2-AG) in healthy adults subjected to low, moderate or high levels of moderate to vigorous physical activity [MVPA] per week after their participation in prescribed or preferred exercise [201]. The prescribed exercise included a 60 min session with 10-min warm-up at 40-60% of their estimated maximal oxygen consumption [VO2max], 45 min at 70-75% of their VO2max, and 5 min of cool down, in the preferred exercise session, participants self-selected the intensity and the length of the session. Overall, participants performed significantly more work (% VO2max × duration in minutes) in the prescribed versus the preferred condition. It should be noted that increases in AEA were larger in the prescribed condition in comparison with the preferred one, but both activated the endocannabinoid system. However, there were non-significant differences in the endocannabinoid’s concentration between samples with varying physical activity levels.
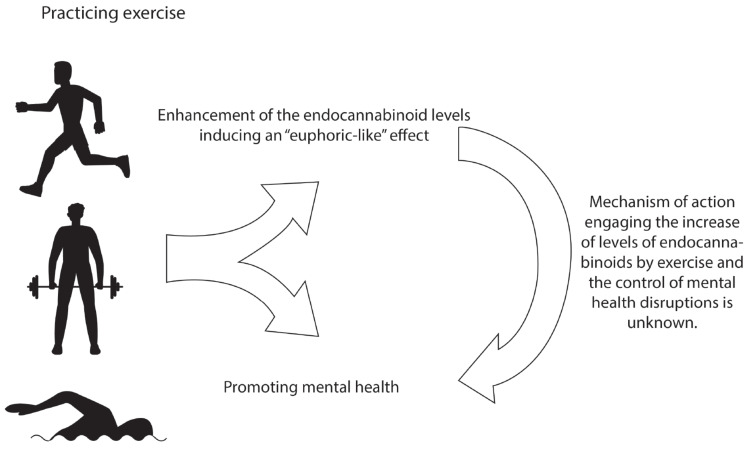
As reviewed, exercise influences several neurobiological elements, including the endocannabinoid system, suggesting a putative modulatory role of the endocannabinoid system on the effectiveness of physical activity and exercise as treatments for managing mental disorders [19-22]. Despite the lack of a mechanism of action that might provide further comprehension of the process in which the endocannabinoid system moderates the positive effects of exercise in depression, anxiety, PTSD and schizophrenia, it has been suggested that one critical element engaged in this putative mechanism might involve a euphoric-like feeling (Fig. 2). This assumption is based on previous reports that suggest that a euphoric feeling is present in subjects after exercise, leading to the release of several neurochemicals, including the endocannabinoids [19-22, 208, 209]. Thus, the data suggest that physical activity and exercise could increase the levels of the endocannabinoids, which in turn might produce changes in mood [21, 210-212]. Whether the increase in AEA and 2-AG or the involvement of the enzymes that modulates the functioning of the endocannabinoid system may underlie the rewarding and pleasurable effects of exercise promoting the positive outcomes described in mental health issues, it is a gray area that remains to be studied as a potential mechanism of action. Moreover, future studies should consider the patience’s preference to a specific exercise routine that influences the endocannabinoid system.
Fig. (2).
The benefits of physical exercise benefits in endocannabinoid system and mental health. The positive effects of physical exercise on boosting the endocannabinoid system activity for modulating the mental health functioning. Current evidence has shown that exercise increases the activity of the endocannabinoids by presumably inducing an “euphoric-like” effect. In addition, practicing exercise controls mental health symptomatology. However, further studies are needed to describe the mechanism of action that engages the increase in the contents of the endocannabinoids and the control of mental health disruptions. (A higher resolution/colour version of this figure is available in the electronic copy of the article).
CONCLUSION
The demographic projections suggest a growth in longevity worldwide in the next years [1-4]. The increase in the number of aged populations will be also associated with the coexistence of medical conditions, including mental health disorders [1, 5, 6]. Despite the further advances for managing mental disturbances, such as the pharmacological means, limited results have been observed, leading to the search for novelty approaches. In this regard, promising strategies include the endocannabinoid system as an effective tool for the enhancement of the positive effects of exercise in the treatment of mental issues [19-22]. In this review, we summarize results collected in studies aimed at evaluating the role of the endocannabinoid system as a modulator for the positive outcomes of exercise in the control of mental disorders, including depression, anxiety, PTSD, and schizophrenia.
The endocannabinoid system comprises a multiple biological elements such as the CB1/CB2 cannabinoid receptors, endogenous ligands -AEA and 2-AG-, AMT and enzymes involved in the biosynthesis and degradation of the AEA and 2-AG, including NAPE, NAPE–PLD, FAAH, DAGL, and MAGL [28-33]. Because the distribution of the endocannabinoid system components has been mapped in multiple organs, including the central nervous system, it has been suggested that this signaling system might be engaged by controlling several complex phenomena such as pain perception, emotion modulation and, mental health disparities [34-37]. Through the selective pharmacological or genetic manipulations of the CB1 cannabinoid receptors [1, 55-68], AEA, 2-AG [98-106, 110-116], enzymes related to the functioning of the endocannabinoid signaling sch as FAAH, DAGL, and MGL [119-133], their relationship with a wider variety of mental disfunctions has been demonstrated [1, 55-68]. However, the role of the CB2 cannabinoid receptors, NAPE, NAPE–PLD, FAAH, AMT, MGL, and DAGL on depression, anxiety, PTSD, schizophrenia is not fully known. Perhaps due to the limited distribution of the CB2 cannabinoid receptors, expression in brain areas implicated in the development of psychiatric disorders or the recent discovery of the enzymes linked to the endocannabinoids biosynthesis and degradation, scarce evidence is available in regards the relationship of these endocannabinoid system components in mental disturbances. Indeed, future experiments should be aimed at unlocking the putative role of the CB2 cannabinoid receptors, NAPE, NAPE–PLD, FAAH, AMT, MGL, and DAGL on depression, anxiety, PTSD, schizophrenia and describe the mechanisms by which elements might exert their modulatory properties in psychiatric disorders.
While the work outlined above does clearly delineate the relevance of the endocannabinoid system on mental health issues, additional elements may be related to the control of psychiatric disturbances. Such variable, exercise, seems to exert positive effects on the management of mental illnesses [159, 160]. For example, it has been reported that different exercise interventions modulate depressive symptoms [161-179]. In addition, clinical evidence has demonstrated that practicing exercise induces positive results for the management of anxiety, [161-189], PTSD [190-192], and schizophrenia [194-199].
It has been proposed that exercise modulates mental health issues by controlling multiple neurobiological networks [201, 202]. Remarkably, the endocannabinoid system is also related to exercise outcomes [203]. In this regard, several studies have shown that exercise enhances the endogenous tone of the endocannabinoids, including practicing high intensity running for 30 min [19, 20, 204-212].
Due to the relationship between endocannabinoid signaling and exercise, it is crucial to clearly distinguish the possible result of modulation of the positive effects of exercise in mental illness by the engagement of the endocannabinoid system. Given the functional link between the elements that comprise the endocannabinoid network and exercise, it bears speculation that these two variables work in concert to control psychiatric disturbances. However, the set of data certainly suggest that the endocannabinoid signaling modulates exercise outcomes. Thus, it remains to be determined how the functioning of the endocannabinoid system regulates the positive effects derived from exercise in the control of psychiatric disturbances.
Indeed, there are gray areas that require further attention to explore the likely enhancement of the endocannabinoid tone for increasing the exercise benefits for managing depression, anxiety, PTSD, and schizophrenia. In addition, the implications of variables such as exercise intensity, volume, type and mode of exercise, or length of the intervention, or type of exercise, require further attention in regards to the dose-response relationship and the likely impact on the endocannabinoid system functioning in mental health treatment. Especially, a standardization of the exercise intensities [152, 213] might facilitate the accurate assessment of the real benefits that training at different intensities might have in the treatment of specific disorders as well as for the change of the endocannabinoids concentration. Given that the endocannabinoid system shows deteriorating profile in mental disorders, thus, creative strategies targeting the enhancement of this neurobiological signaling network, beyond the pharmacological perspectives, will be needed in the future.
FUTURE PERSPECTIVES
An outbreak caused by a novel coronavirus (SARS-CoV-2, previously known as 2019-nCoV), the coronavirus disease 2019 (COVID-19), which was originated in the Chinese city of Wuhan, has rapidly spread around the world causing severe acute respiratory syndrome [214, 215]. COVID-19 symptoms include cough, fatigue, fever, and gastrointestinal infection. Importantly, elderly subjects are susceptible to infection and prone to critical outcomes [216-219]. Recent articles suggest that COVID-19 could be associated with psychiatric disturbances [158, 159, 220-224]. For instance, current data have shown that patients that suffered and recovered from COVID-19 displayed significant rates of anxiety, depression, and PTSD [225-227]. We believe that the present review would provide further stimulation for assessing novel therapeutic means such as the manipulation of the endocannabinoid system to enhance the benefits of physical exercise in mental disorders, highlighting the recovered subjects from COVID-19 who could likely develop psychiatric diseases.
ACKNOWLEDGEMENTS
Declared none.
CONSENT FOR PUBLICATION
Not applicable.
FUNDING
This work was supported by Escuela de Medicina, Uni-versidad Anáhuac Mayab (Grant: PresInvEMR2019) given to E M-R.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
REFERENCES
- 1.GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990-2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392(10159):1789–1858. doi: 10.1016/S0140-6736(18)32279-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tucci V., Moukaddam N. We are the hollow men: The worldwide epidemic of mental illness, psychiatric and behavioral emergencies, and its impact on patients and providers. J. Emerg. Trauma Shock. 2017;10(1):4–6. doi: 10.4103/0974-2700.199517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Prince M., Ali G.C., Guerchet M., Prina A.M., Albanese E., Wu Y.T. Recent global trends in the prevalence and incidence of dementia, and survival with dementia. Alzheimers Res. Ther. 2016;8(1):23. doi: 10.1186/s13195-016-0188-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Richter D., Wall A., Bruen A., Whittington R. Is the global prevalence rate of adult mental illness increasing? Systematic review and meta-analysis. Acta Psychiatr. Scand. 2019;140(5):393–407. doi: 10.1111/acps.13083. [DOI] [PubMed] [Google Scholar]
- 5.Steel Z., Marnane C., Iranpour C., Chey T., Jackson J.W., Patel V., Silove D. The global prevalence of common mental disorders: a systematic review and meta-analysis 1980-2013. Int. J. Epidemiol. 2014;43(2):476–493. doi: 10.1093/ije/dyu038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Schürmann J., Margraf J. Age of anxiety and depression revisited: A meta-analysis of two European community samples (1964-2015). Int. J. Clin. Health Psychol. 2018;18(2):102–112. doi: 10.1016/j.ijchp.2018.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fusar-Poli P., Davies C., Solmi M., Brondino N., De Micheli A., Kotlicka-Antczak M., Shin J.I., Radua J. preventive treatments for psychosis: umbrella review (just the evidence). Front. Psychiatry. 2019;10:764. doi: 10.3389/fpsyt.2019.00764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Brown J.V.E., Walton N., Meader N., Todd A., Webster L.A., Steele R., Sampson S.J., Churchill R., McMillan D., Gilbody S., Ekers D. Pharmacy-based management for depression in adults. Cochrane Database Syst. Rev. 2019;12(12):CD013299. doi: 10.1002/14651858.CD013299.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ansari F., Pourjafar H., Tabrizi A., Homayouni A. The effects of probiotics and prebiotics on mental disorders: a review on depression, anxiety, alzheimer, and autism spectrum disorders. Curr. Pharm. Biotechnol. 2020;21(7):555–565. doi: 10.2174/1389201021666200107113812. [DOI] [PubMed] [Google Scholar]
- 10.Liu J., Chua J.J., Chong S.A., Subramaniam M., Mahendran R. The impact of emotion dysregulation on positive and negative symptoms in schizophrenia spectrum disorders: A systematic review. J. Clin. Psychol. 2020;76(4):612–624. doi: 10.1002/jclp.22915. [DOI] [PubMed] [Google Scholar]
- 11.Chen P. Optimized Treatment Strategy for Depressive Disorder. Adv. Exp. Med. Biol. 2019;1180:201–217. doi: 10.1007/978-981-32-9271-0_11. [DOI] [PubMed] [Google Scholar]
- 12.Arvanitakis Z., Shah R.C., Bennett D.A. diagnosis and management of dementia. JAMA. 2019;322(16):1589–1599. doi: 10.1001/jama.2019.4782. [Review]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Missotten P., Farag L., Delye S., Muller A., Grotz C., Adam S. Role of “light therapy” among older adults with dementia: an overview and future perspectives. Gériatr. Psychol. Neuropsychiatr. Vieil. 2019;17(1):83–91. doi: 10.1684/pnv.2019.0786. [DOI] [PubMed] [Google Scholar]
- 14.Głąbska D., Guzek D., Groele B., Gutkowska K. Fruit and vegetable intake and mental health in adults: a systematic review. Nutrients. 2020;12(1):E115. doi: 10.3390/nu12010115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Korman N., Armour M., Chapman J., Rosenbaum S., Kisely S., Suetani S., Firth J., Siskind D., Siskind D. High Intensity Interval training (HIIT) for people with severe mental illness: A systematic review & meta-analysis of intervention studies- considering diverse approaches for mental and physical recovery. Psychiatry Res. 2020;284:112601. doi: 10.1016/j.psychres.2019.112601. [DOI] [PubMed] [Google Scholar]
- 16.de Almeida S.I.L., Gomes da Silva M., Marques A.S.P.D. Home-based physical activity programs for people with dementia: systematic review and meta-analysis. Gerontologist. 2020;60(8):600–608. doi: 10.1093/geront/gnz176. [DOI] [PubMed] [Google Scholar]
- 17.Sampaio A., Marques-Aleixo I., Seabra A., Mota J., Marques E., Carvalho J. Physical fitness in institutionalized older adults with dementia: association with cognition, functional capacity and quality of life. Aging Clin. Exp. Res. 2020;32(11):2329–2338. doi: 10.1007/s40520-019-01445-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Enette L., Vogel T., Merle S., Valard-Guiguet A.G., Ozier-Lafontaine N., Neviere R., Leuly-Joncart C., Fanon J.L., Lang P.O. Effect of 9 weeks continuous vs. interval aerobic training on plasma BDNF levels, aerobic fitness, cognitive capacity and quality of life among seniors with mild to moderate Alzheimer’s disease: a randomized controlled trial. Eur. Rev. Aging Phys. Act. 2020;17:2. doi: 10.1186/s11556-019-0234-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Watkins B.A. Endocannabinoids, exercise, pain, and a path to health with aging. Mol. Aspects Med. 2018;64:68–78. doi: 10.1016/j.mam.2018.10.001. [DOI] [PubMed] [Google Scholar]
- 20.Loprinzi P.D., Zou L., Li H. the endocannabinoid system as a potential mechanism through which exercise influences episodic memory function. Brain Sci. 2019;9(5):112. doi: 10.3390/brainsci9050112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Meyer J.D., Crombie K.M., Cook D.B., Hillard C.J., Koltyn K.F. Serum endocannabinoid and mood changes after exercise in major depressive disorder. Med. Sci. Sports Exerc. 2019;51(9):1909–1917. doi: 10.1249/MSS.0000000000002006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Moosavi Sohroforouzani A., Shakerian S., Ghanbarzadeh M., Alaei H. Treadmill exercise improves LPS-induced memory impairments via endocannabinoid receptors and cyclooxygenase enzymes. Behav. Brain Res. 2020;380:112440. doi: 10.1016/j.bbr.2019.112440. [DOI] [PubMed] [Google Scholar]
- 23.Maccarrone M., Bab I., Bíró T., Cabral G.A., Dey S.K., Di Marzo V., Konje J.C., Kunos G., Mechoulam R., Pacher P., Sharkey K.A., Zimmer A. Endocannabinoid signaling at the periphery: 50 years after THC. Trends Pharmacol. Sci. 2015;36(5):277–296. doi: 10.1016/j.tips.2015.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Russo E.B., Marcu J. Cannabis pharmacology: the usual suspects and a few promising leads. Adv. Pharmacol. 2017;80:67–134. doi: 10.1016/bs.apha.2017.03.004. [DOI] [PubMed] [Google Scholar]
- 25.Pacher P., Kogan N.M., Mechoulam R. Beyond THC and Endocannabinoids. Annu. Rev. Pharmacol. Toxicol. 2020;60:637–659. doi: 10.1146/annurev-pharmtox-010818-021441. [DOI] [PubMed] [Google Scholar]
- 26.McPartland J.M., Duncan M., Di Marzo V., Pertwee R.G. Are cannabidiol and Δ(9) -tetrahydrocannabivarin negative modulators of the endocannabinoid system? A systematic review. Br. J. Pharmacol. 2015;172(3):737–753. doi: 10.1111/bph.12944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Freeman A.M., Petrilli K., Lees R., Hindocha C., Mokrysz C., Curran H.V., Saunders R., Freeman T.P. How does cannabidiol (CBD) influence the acute effects of delta-9-tetrahydrocannabinol (THC) in humans? A systematic review. Neurosci. Biobehav. Rev. 2019;107:696–712. doi: 10.1016/j.neubiorev.2019.09.036. [DOI] [PubMed] [Google Scholar]
- 28.Di Marzo V., Piscitelli F. The endocannabinoid system and its modulation by phytocannabinoids. Neurotherapeutics. 2015;12(4):692–698. doi: 10.1007/s13311-015-0374-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Biernacki M., Skrzydlewska E. Metabolism of endocannabinoids. Postepy Hig. Med. Dosw. 2016;70(0):830–843. doi: 10.5604/17322693.1213898. [DOI] [PubMed] [Google Scholar]
- 30.Tsuboi K., Uyama T., Okamoto Y., Ueda N. Endocannabinoids and related N-acylethanolamines: biological activities and metabolism. Inflamm. Regen. 2018;38:28. doi: 10.1186/s41232-018-0086-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Freitas H.R., Isaac A.R., Malcher-Lopes R., Diaz B.L., Trevenzoli I.H., De Melo Reis R.A. Polyunsaturated fatty acids and endocannabinoids in health and disease. Nutr. Neurosci. 2018;21(10):695–714. doi: 10.1080/1028415X.2017.1347373. [DOI] [PubMed] [Google Scholar]
- 32.Gil-Ordóñez A., Martín-Fontecha M., Ortega-Gutiérrez S., López-Rodríguez M.L. Monoacylglycerol lipase (MAGL) as a promising therapeutic target. Biochem. Pharmacol. 2018;157:18–32. doi: 10.1016/j.bcp.2018.07.036. [DOI] [PubMed] [Google Scholar]
- 33.Toczek M., Malinowska B. Enhanced endocannabinoid tone as a potential target of pharmacotherapy. Life Sci. 2018;204:20–45. doi: 10.1016/j.lfs.2018.04.054. [DOI] [PubMed] [Google Scholar]
- 34.Bisogno T., Berrendero F., Ambrosino G., Cebeira M., Ramos J.A., Fernandez-Ruiz J.J., Di Marzo V. Brain regional distribution of endocannabinoids: implications for their biosynthesis and biological function. Biochem. Biophys. Res. Commun. 1999;256(2):377–380. doi: 10.1006/bbrc.1999.0254. [DOI] [PubMed] [Google Scholar]
- 35.Kendall D.A., Yudowski G.A. Cannabinoid receptors in the central nervous system: their signaling and roles in disease. Front. Cell. Neurosci. 2017;10:294. doi: 10.3389/fncel.2016.00294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Busquets-Garcia A., Bains J., Marsicano G. CB1 Receptor Signaling in the Brain: Extracting Specificity from Ubiquity. Neuropsychopharmacology. 2018;43(1):4–20. doi: 10.1038/npp.2017.206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zou S., Kumar U. Cannabinoid receptors and the endocannabinoid system: signaling and function in the central nervous system. Int. J. Mol. Sci. 2018;19(3):833. doi: 10.3390/ijms19030833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Jain A., Mitra P. Bipolar Affective Disorder. 1st ed. Florida: StatPearls; 2020. [Google Scholar]
- 39.Habtewold T.D., Rodijk L.H., Liemburg E.J., Sidorenkov G., Boezen H.M., Bruggeman R., Alizadeh B.Z. A systematic review and narrative synthesis of data-driven studies in schizophrenia symptoms and cognitive deficits. Transl. Psychiatry. 2020;10(1):244. doi: 10.1038/s41398-020-00919-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Maguire J., McCormack C., Mitchell A., Monk C. Neurobiology of maternal mental illness. Handb. Clin. Neurol. 2020;171:97–116. doi: 10.1016/B978-0-444-64239-4.00005-9. [DOI] [PubMed] [Google Scholar]
- 41.Regier D.A., Kuhl E.A., Kupfer D.J. The DSM-5: Classification and criteria changes. World Psychiatry. 2013;12(2):92–98. doi: 10.1002/wps.20050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cooper R. Understanding the DSM-5: stasis and change. Hist. Psychiatry. 2018;29(1):49–65. doi: 10.1177/0957154X17741783. [DOI] [PubMed] [Google Scholar]
- 43.Fusar-Poli P., Salazar de Pablo G., Correll C.U., Meyer-Lindenberg A., Millan M.J., Borgwardt S., Galderisi S., Bechdolf A., Pfennig A., Kessing L.V., van Amelsvoort T., Nieman D.H., Domschke K., Krebs M.O., Koutsouleris N., McGuire P., Do K.Q., Arango C. Prevention of psychosis: advances in detection, prognosis, and intervention. JAMA Psychiatry. 2020;77(7):755–765. doi: 10.1001/jamapsychiatry.2019.4779. [DOI] [PubMed] [Google Scholar]
- 44.Witusik A., Pietras T. Music therapy as a complementary form of therapy for mental disorders. Pol. Merkuriusz Lek. 2019;47(282):240–243. [PubMed] [Google Scholar]
- 45.Sarris J., Sinclair J., Karamacoska D., Davidson M., Firth J. Medicinal cannabis for psychiatric disorders: a clinically-focused systematic review. BMC Psychiatry. 2020;20(1):24. doi: 10.1186/s12888-019-2409-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zagórska A., Marcinkowska M., Jamrozik M., Wiśniowska B., Paśko P. From probiotics to psychobiotics - the gut-brain axis in psychiatric disorders. Benef. Microbes. 2020;11(8):717–732. doi: 10.3920/BM2020.0063. [DOI] [PubMed] [Google Scholar]
- 47.Gordovez F.J.A., McMahon F.J. The genetics of bipolar disorder. Mol. Psychiatry. 2020;25(3):544–559. doi: 10.1038/s41380-019-0634-7. [DOI] [PubMed] [Google Scholar]
- 48.Smigielski L., Jagannath V., Rössler W., Walitza S., Grünblatt E. Epigenetic mechanisms in schizophrenia and other psychotic disorders: a systematic review of empirical human findings. Mol. Psychiatry. 2020;25(8):1718–1748. doi: 10.1038/s41380-019-0601-3. [DOI] [PubMed] [Google Scholar]
- 49.Rubino T., Zamberletti E., Parolaro D. Endocannabinoids and mental disorders. Handb. Exp. Pharmacol. 2015;231:261–283. doi: 10.1007/978-3-319-20825-1_9. [DOI] [PubMed] [Google Scholar]
- 50.Aran A., Eylon M., Harel M., Polianski L., Nemirovski A., Tepper S., Schnapp A., Cassuto H., Wattad N., Tam J. Lower circulating endocannabinoid levels in children with autism spectrum disorder. Mol. Autism. 2019;10:2. doi: 10.1186/s13229-019-0256-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Romero-Sanchiz P., Nogueira-Arjona R., Pastor A., Araos P., Serrano A., Boronat A., Garcia-Marchena N., Mayoral F., Bordallo A., Alen F., Suárez J., de la Torre R., Pavón F.J., Rodríguez de Fonseca F. Plasma concentrations of oleoylethanolamide in a primary care sample of depressed patients are increased in those treated with selective serotonin reuptake inhibitor-type antidepressants. Neuropharmacology. 2019;149:212–220. doi: 10.1016/j.neuropharm.2019.02.026. [DOI] [PubMed] [Google Scholar]
- 52.Papagianni E.P., Stevenson C.W. Cannabinoid regulation of fear and anxiety: an update. Curr. Psychiatry Rep. 2019;21(6):38. doi: 10.1007/s11920-019-1026-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Arjmand S., Behzadi M., Kohlmeier K.A., Mazhari S., Sabahi A., Shabani M. Bipolar disorder and the endocannabinoid system. Acta Neuropsychiatr. 2019;31(4):193–201. doi: 10.1017/neu.2019.21. [DOI] [PubMed] [Google Scholar]
- 54.Chadwick V.L., Rohleder C., Koethe D., Leweke F.M. Cannabinoids and the endocannabinoid system in anxiety, depression, and dysregulation of emotion in humans. Curr. Opin. Psychiatry. 2020;33(1):20–42. doi: 10.1097/YCO.0000000000000562. [DOI] [PubMed] [Google Scholar]
- 55.Kendrick T., Pilling S. Common mental health disorders--identification and pathways to care: NICE clinical guideline. Br. J. Gen. Pract. 2012;62(594):47–49. doi: 10.3399/bjgp12X616481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Identification of risk loci with shared effects on five major psychiatric disorders: a genome-wide analysis. Lancet. 2013;381(9875):1371–1379. doi: 10.1016/S0140-6736(12)62129-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Black N., Stockings E., Campbell G., Tran L.T., Zagic D., Hall W.D., Farrell M., Degenhardt L. Cannabinoids for the treatment of mental disorders and symptoms of mental disorders: a systematic review and meta-analysis. Lancet Psychiatry. 2019;6(12):995–1010. doi: 10.1016/S2215-0366(19)30401-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Moreira F.A., Crippa J.A. The psychiatric side-effects of rimonabant. Br. J. Psychiatry. 2009;31(2):145–153. doi: 10.1590/S1516-44462009000200012. [DOI] [PubMed] [Google Scholar]
- 59.Botsford S.L., Yang S., George T.P. Cannabis and cannabinoids in mood and anxiety disorders: impact on illness onset and course, and assessment of therapeutic potential. Am. J. Addict. 2020;29(1):9–26. doi: 10.1111/ajad.12963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Nguyen T., Thomas B.F., Zhang Y. Overcoming the psychiatric side effects of the cannabinoid CB1 receptor antagonists: current approaches for therapeutics development. Curr. Top. Med. Chem. 2019;19(16):1418–1435. doi: 10.2174/1568026619666190708164841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ostadhadi S., Haj-Mirzaian A., Nikoui V., Kordjazy N., Dehpour A.R. Involvement of opioid system in antidepressant-like effect of the cannabinoid CB1 receptor inverse agonist AM-251 after physical stress in mice. Clin. Exp. Pharmacol. Physiol. 2016;43(2):203–212. doi: 10.1111/1440-1681.12518. [DOI] [PubMed] [Google Scholar]
- 62.Fang G., Wang Y. Effects of rTMS on hippocampal endocannabinoids and depressive-like behaviors in adolescent rats. Neurochem. Res. 2018;43(9):1756–1765. doi: 10.1007/s11064-018-2591-y. [DOI] [PubMed] [Google Scholar]
- 63.Wośko S., Serefko A., Szopa A., Wlaź P., Wróbel A., Wlaź A., Górska J., Poleszak E., Wlaź A., Serefko A. CB1 cannabinoid receptor ligands augment the antidepressant-like activity of biometals (magnesium and zinc) in the behavioural tests. J. Pharm. Pharmacol. 2018;70(4):566–575. doi: 10.1111/jphp.12880. [DOI] [PubMed] [Google Scholar]
- 64.Aso E., Ozaita A., Serra M.À., Maldonado R. Genes differentially expressed in CB1 knockout mice: involvement in the depressive-like phenotype. Eur. Neuropsychopharmacol. 2011;21(1):11–22. doi: 10.1016/j.euroneuro.2010.06.007. [DOI] [PubMed] [Google Scholar]
- 65.Walsh K.B., Andersen H.K. Molecular pharmacology of synthetic cannabinoids: delineating cb1 receptor-mediated cell signaling. Int. J. Mol. Sci. 2020;21(17):6115. doi: 10.3390/ijms21176115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Fraguas-Sánchez A.I., Torres-Suárez A.I. Medical use of cannabinoids. Drugs. 2018;78(16):1665–1703. doi: 10.1007/s40265-018-0996-1. [DOI] [PubMed] [Google Scholar]
- 67.Micale V., Di Marzo V., Sulcova A., Wotjak C.T., Drago F. Endocannabinoid system and mood disorders: priming a target for new therapies. Pharmacol. Ther. 2013;138(1):18–37. doi: 10.1016/j.pharmthera.2012.12.002. [DOI] [PubMed] [Google Scholar]
- 68.Micale V., Tabiova K., Kucerova J., Drago F. In: Role of the endocannabinoid system in depression: from preclinical to clinical evidence. Cannabinoid Modulation of Emotion, Memory, and Motivation; Campolongo, P. Fattore L., editor. Vol. 1. New York: Springer; 2015. pp. 97–129. [Google Scholar]
- 69.Ganon-Elazar E., Akirav I. Cannabinoid receptor activation in the basolateral amygdala blocks the effects of stress on the conditioning and extinction of inhibitory avoidance. J. Neurosci. 2009;29(36):11078–11088. doi: 10.1523/JNEUROSCI.1223-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Shoshan N., Segev A., Abush H., Mizrachi Zer-Aviv T., Akirav I. Cannabinoids prevent the differential long-term effects of exposure to severe stress on hippocampal- and amygdala-dependent memory and plasticity. Hippocampus. 2017;27(10):1093–1109. doi: 10.1002/hipo.22755. [DOI] [PubMed] [Google Scholar]
- 71.Lisboa S.F., Vila-Verde C., Rosa J., Uliana D.L., Stern C.A.J., Bertoglio L.J., Resstel L.B., Guimaraes F.S. Tempering aversive/traumatic memories with cannabinoids: a review of evidence from animal and human studies. Psychopharmacology (Berl.) 2019;236(1):201–226. doi: 10.1007/s00213-018-5127-x. [DOI] [PubMed] [Google Scholar]
- 72.Valverde O., Torrens M. CB1 receptor-deficient mice as a model for depression. Neuroscience. 2012;204:193–206. doi: 10.1016/j.neuroscience.2011.09.031. [DOI] [PubMed] [Google Scholar]
- 73.Moreira F.A., Grieb M., Lutz B. Central side-effects of therapies based on CB1 cannabinoid receptor agonists and antagonists: focus on anxiety and depression. Best Pract. Res. Clin. Endocrinol. Metab. 2009;23(1):133–144. doi: 10.1016/j.beem.2008.09.003. [DOI] [PubMed] [Google Scholar]
- 74.Beyer C.E., Dwyer J.M., Piesla M.J., Platt B.J., Shen R., Rahman Z., Chan K., Manners M.T., Samad T.A., Kennedy J.D., Bingham B., Whiteside G.T. Depression-like phenotype following chronic CB1 receptor antagonism. Neurobiol. Dis. 2010;39(2):148–155. doi: 10.1016/j.nbd.2010.03.020. [DOI] [PubMed] [Google Scholar]
- 75.Ivy D., Palese F., Vozella V., Fotio Y., Yalcin A., Ramirez G., Mears D., Wynn G., Piomelli D. Cannabinoid CB2 receptors mediate the anxiolytic-like effects of monoacylglycerol lipase inhibition in a rat model of predator-induced fear. Neuropsychopharmacology. 2020;45(8):1330–1338. doi: 10.1038/s41386-020-0696-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Marsicano G., Lutz B. Expression of the cannabinoid receptor CB1 in distinct neuronal subpopulations in the adult mouse forebrain. Eur. J. Neurosci. 1999;11(12):4213–4225. doi: 10.1046/j.1460-9568.1999.00847.x. [DOI] [PubMed] [Google Scholar]
- 77.Hudson R., Rushlow W., Laviolette S.R. Phytocannabinoids modulate emotional memory processing through interactions with the ventral hippocampus and mesolimbic dopamine system: implications for neuropsychiatric pathology. . Psychopharmacol. . 2018;235(2):447–458. doi: 10.1007/s00213-017-4766-7. [DOI] [PubMed] [Google Scholar]
- 78.Terzian A.L., Micale V., Wotjak C.T. Cannabinoid receptor type 1 receptors on GABAergic vs. glutamatergic neurons differentially gate sex-dependent social interest in mice. Eur. J. Neurosci. 2014;40(1):2293–2298. doi: 10.1111/ejn.12561. [DOI] [PubMed] [Google Scholar]
- 79.Micale V., Stepan J., Jurik A., Pamplona F.A., Marsch R., Drago F., Eder M., Wotjak C.T. Extinction of avoidance behavior by safety learning depends on endocannabinoid signaling in the hippocampus. J. Psychiatr. Res. 2017;90:46–59. doi: 10.1016/j.jpsychires.2017.02.002. [DOI] [PubMed] [Google Scholar]
- 80.Astill Wright L., Sijbrandij M., Sinnerton R., Lewis C., Roberts N.P., Bisson J.I. Pharmacological prevention and early treatment of post-traumatic stress disorder and acute stress disorder: a systematic review and meta-analysis. Transl. Psychiatry. 2019;9(1):334. doi: 10.1038/s41398-019-0673-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Terzian A.L., Drago F., Wotjak C.T., Micale V. The dopamine and cannabinoid interaction in the modulation of emotions and cognition: assessing the role of cannabinoid cb1 receptor in neurons expressing dopamine D1 receptors. Front. Behav. Neurosci. 2011;5:49. doi: 10.3389/fnbeh.2011.00049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Llorente-Berzal A., Terzian A.L., di Marzo V., Micale V., Viveros M.P., Wotjak C.T. 2-AG promotes the expression of conditioned fear via cannabinoid receptor type 1 on GABAergic neurons. Psychopharmacology (Berl.) 2015;232(15):2811–2825. doi: 10.1007/s00213-015-3917-y. [DOI] [PubMed] [Google Scholar]
- 83.Cowling T., MacDougall D. Nabilone for the Treatment of Post-Traumatic Stress Disorder: A Review of Clinical Effectiveness and Guidelines. 1st ed. Ottawa: Canadian Agency for Drugs and Technologies in Health; 2019. [PubMed] [Google Scholar]
- 84.Neumeister A., Normandin M.D., Pietrzak R.H., Piomelli D., Zheng M.Q., Gujarro-Anton A., Potenza M.N., Bailey C.R., Lin S.F., Najafzadeh S., Ropchan J., Henry S., Corsi-Travali S., Carson R.E., Huang Y., Corsi-Travali S., Carson R.E., Huang Y. Elevated brain cannabinoid CB1 receptor availability in post-traumatic stress disorder: a positron emission tomography study. Mol. Psychiatry. 2013;18(9):1034–1040. doi: 10.1038/mp.2013.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Micale V., Drago F., Noerregaard P.K., Elling C.E., Wotjak C.T. The cannabinoid CB1 antagonist TM38837 with limited penetrance to the brain shows reduced fear-promoting effects in Mice. Front. Pharmacol. 2019;10:207. doi: 10.3389/fphar.2019.00207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Bryant R.A. Post-traumatic stress disorder: a state-of-the-art review of evidence and challenges. World Psychiatry. 2019;18(3):259–269. doi: 10.1002/wps.20656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Colucci P., Marchetta E., Mancini G.F., Alva P., Chiarotti F., Hasan M.T., Campolongo P. Predicting susceptibility and resilience in an animal model of post-traumatic stress disorder (PTSD). Transl. Psychiatry. 2020;10(1):243. doi: 10.1038/s41398-020-00929-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Leweke F.M., Mueller J.K., Lange B., Fritze S., Topor C.E., Koethe D., Rohleder C. Role of the endocannabinoid system in the pathophysiology of schizophrenia: implications for pharmacological intervention. CNS Drugs. 2018;32(7):605–619. doi: 10.1007/s40263-018-0539-z. [DOI] [PubMed] [Google Scholar]
- 89.Varlow C., Boileau I., Wey H.Y., Liang S.H., Vasdev N. Classics in neuroimaging: imaging the endocannabinoid pathway with PET. ACS Chem. Neurosci. 2020;11(13):1855–1862. doi: 10.1021/acschemneuro.0c00305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Tao R., Li C., Jaffe A.E., Shin J.H., Deep-Soboslay A., Yamin R., Weinberger D.R., Hyde T.M., Kleinman J.E. Cannabinoid receptor CNR1 expression and DNA methylation in human prefrontal cortex, hippocampus and caudate in brain development and schizophrenia. Transl. Psychiatry. 2020;10(1):158. doi: 10.1038/s41398-020-0832-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Sloan M.E., Grant C.W., Gowin J.L., Ramchandani V.A., Le Foll B. Endocannabinoid signaling in psychiatric disorders: a review of positron emission tomography studies. Acta Pharmacol. Sin. 2019;40(3):342–350. doi: 10.1038/s41401-018-0081-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Kucerova J., Tabiova K., Drago F., Micale V. Therapeutic potential of cannabinoids in schizophrenia. Recent Patents CNS Drug Discov. 2014;9(1):13–25. doi: 10.2174/1574889809666140307115532. [DOI] [PubMed] [Google Scholar]
- 93.Muguruza C., Morentin B., Meana J.J., Alexander S.P., Callado L.F. Endocannabinoid system imbalance in the postmortem prefrontal cortex of subjects with schizophrenia. J. Psychopharmacol. 2019;33(9):1132–1140. doi: 10.1177/0269881119857205. [DOI] [PubMed] [Google Scholar]
- 94.Dalton V.S., Long L.E., Weickert C.S., Zavitsanou K. Paranoid schizophrenia is characterized by increased CB1 receptor binding in the dorsolateral prefrontal cortex. Neuropsychopharmacology. 2011;36(8):1620–1630. doi: 10.1038/npp.2011.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Volk D.W., Eggan S.M., Horti A.G., Wong D.F., Lewis D.A. Reciprocal alterations in cortical cannabinoid receptor 1 binding relative to protein immunoreactivity and transcript levels in schizophrenia. Schizophr. Res. 2014;159(1):124–129. doi: 10.1016/j.schres.2014.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.D’Addario C., Micale V., Di Bartolomeo M., Stark T., Pucci M., Sulcova A., Palazzo M., Babinska Z., Cremaschi L., Drago F., Carlo Altamura A., Maccarrone M., Dell’Osso B. A preliminary study of endocannabinoid system regulation in psychosis: Distinct alterations of CNR1 promoter DNA methylation in patients with schizophrenia. Schizophr. Res. 2017;188:132–140. doi: 10.1016/j.schres.2017.01.022. [DOI] [PubMed] [Google Scholar]
- 97.Stark T., Ruda-Kucerova J., Iannotti F.A., D’Addario C., Di Marco R., Pekarik V., Drazanova E., Piscitelli F., Bari M., Babinska Z., Giurdanella G., Di Bartolomeo M., Salomone S., Sulcova A., Maccarrone M., Wotjak C.T., Starcuk Z., Jr, Drago F., Mechoulam R., Di Marzo V., Micale V. Peripubertal cannabidiol treatment rescues behavioral and neurochemical abnormalities in the MAM model of schizophrenia. Neuropharmacology. 2019;146:212–221. doi: 10.1016/j.neuropharm.2018.11.035. [DOI] [PubMed] [Google Scholar]
- 98.Hill M.N., Miller G.E., Ho W.S., Gorzalka B.B., Hillard C.J. Serum endocannabinoid content is altered in females with depressive disorders: a preliminary report. Pharmacopsychiatry. 2008;41(2):48–53. doi: 10.1055/s-2007-993211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Hill M.N., Miller G.E., Carrier E.J., Gorzalka B.B., Hillard C.J. Circulating endocannabinoids and N-acyl ethanolamines are differentially regulated in major depression and following exposure to social stress. Psychoneuroendocrinology. 2009;34(8):1257–1262. doi: 10.1016/j.psyneuen.2009.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Wang H.N., Wang L., Zhang R.G., Chen Y.C., Liu L., Gao F., Nie H., Hou W.G., Peng Z.W., Tan Q. Anti-depressive mechanism of repetitive transcranial magnetic stimulation in rat: the role of the endocannabinoid system. J. Psychiatr. Res. 2014;51:79–87. doi: 10.1016/j.jpsychires.2014.01.004. [DOI] [PubMed] [Google Scholar]
- 101.Wingenfeld K., Dettenborn L., Kirschbaum C., Gao W., Otte C., Roepke S. Reduced levels of the endocannabinoid arachidonylethanolamide (AEA) in hair in patients with borderline personality disorder - a pilot study. Stress. 2018;21(4):366–369. doi: 10.1080/10253890.2018.1451837. [DOI] [PubMed] [Google Scholar]
- 102.Smaga I., Gawlinski D., Brodowicz J., Filip M. Brain region-dependent changes in the expression of endocannabinoid-metabolizing enzymes in rats following antidepressant drugs. J. Physiol. Pharmacol. 2019;70(5):10. doi: 10.26402/jpp.2019.5.06. [DOI] [PubMed] [Google Scholar]
- 103.Cristino L., Bisogno T., Di Marzo V. Cannabinoids and the expanded endocannabinoid system in neurological disorders. Nat. Rev. Neurol. 2020;16(1):9–29. doi: 10.1038/s41582-019-0284-z. [DOI] [PubMed] [Google Scholar]
- 104.Pertwee R.G. Targeting the endocannabinoid system with cannabinoid receptor agonists: pharmacological strategies and therapeutic possibilities. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2012;367(1607):3353–3363. doi: 10.1098/rstb.2011.0381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Reggio P.H. Endocannabinoid binding to the cannabinoid receptors: what is known and what remains unknown. Curr. Med. Chem. 2010;17(14):1468–1486. doi: 10.2174/092986710790980005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Coccaro E.F., Hill M.N., Robinson L., Lee R.J. Circulating endocannabinoids and affect regulation in human subjects. Psychoneuroendocrinology. 2018;92:66–71. doi: 10.1016/j.psyneuen.2018.03.009. [DOI] [PubMed] [Google Scholar]
- 107.Navarrete F., García-Gutiérrez M.S., Jurado-Barba R., Rubio G., Gasparyan A., Austrich-Olivares A., Manzanares J. Endocannabinoid system components as potential biomarkers in psychiatry. Front. Psychiatry. 2020;11:315. doi: 10.3389/fpsyt.2020.00315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Gao W., Walther A., Wekenborg M., Penz M., Kirschbaum C. Determination of endocannabinoids and N-acylethanolamines in human hair with LC-MS/MS and their relation to symptoms of depression, burnout, and anxiety. Talanta. 2020;217:121006. doi: 10.1016/j.talanta.2020.121006. [DOI] [PubMed] [Google Scholar]
- 109.Bedse G., Hill M.N., Patel S. 2-Arachidonoylglycerol modulation of anxiety and stress adaptation: from grass roots to novel therapeutics. Biol. Psychiatry. 2020;88(7):520–530. doi: 10.1016/j.biopsych.2020.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Dow-Edwards D. Sex differences in the interactive effects of early life stress and the endocannabinoid system. Neurotoxicol. Teratol. 2020;80:106893. doi: 10.1016/j.ntt.2020.106893. [DOI] [PubMed] [Google Scholar]
- 111.Neumeister A. The endocannabinoid system provides an avenue for evidence-based treatment development for PTSD. Depress. Anxiety. 2013;30(2):93–96. doi: 10.1002/da.22031. [DOI] [PubMed] [Google Scholar]
- 112.Wilker S., Pfeiffer A., Elbert T., Ovuga E., Karabatsiakis A., Krumbholz A., Thieme D., Schelling G., Kolassa I.T. Endocannabinoid concentrations in hair are associated with PTSD symptom severity. Psychoneuroendocrinology. 2016;67:198–206. doi: 10.1016/j.psyneuen.2016.02.010. [DOI] [PubMed] [Google Scholar]
- 113.Pinna G. Biomarkers for PTSD at the Interface of the Endocannabinoid and Neurosteroid Axis. Front. Neurosci. 2018;12:482. doi: 10.3389/fnins.2018.00482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Koethe D., Pahlisch F., Hellmich M., Rohleder C., Mueller J.K., Meyer-Lindenberg A., Torrey E.F., Piomelli D., Leweke F.M. Familial abnormalities of endocannabinoid signaling in schizophrenia. World J. Biol. Psychiatry. 2019;20(2):117–125. doi: 10.1080/15622975.2018.1449966. [DOI] [PubMed] [Google Scholar]
- 115.Potvin S., Mahrouche L., Assaf R., Chicoine M., Giguère C.É., Furtos A., Godbout R. Peripheral endogenous cannabinoid levels are increased in schizophrenia patients evaluated in a psychiatric emergency setting. Front. Psychiatry. 2020;11:628. doi: 10.3389/fpsyt.2020.00628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Stasiulewicz A., Znajdek K., Grudzień M., Pawiński T., Sulkowska A.J.I. A guide to targeting the endocannabinoid system in drug design. Int. J. Mol. Sci. 2020;21(8):2778. doi: 10.3390/ijms21082778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Maccarrone M. Metabolism of the Endocannabinoid Anandamide: Open Questions after 25 Years. Front. Mol. Neurosci. 2017;10:166. doi: 10.3389/fnmol.2017.00166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Chanda D., Neumann D., Glatz J.F.C. The endocannabinoid system: Overview of an emerging multi-faceted therapeutic target. Prostaglandins Leukot. Essent. Fatty Acids. 2019;140:51–56. doi: 10.1016/j.plefa.2018.11.016. [DOI] [PubMed] [Google Scholar]
- 119.Rodríguez-Muñoz M., Sánchez-Blázquez P., Callado L.F., Meana J.J., Garzón-Niño J. Schizophrenia and depression, two poles of endocannabinoid system deregulation. Transl. Psychiatry. 2017;7(12):1291. doi: 10.1038/s41398-017-0029-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Ibarra-Lecue I., Pilar-Cuéllar F., Muguruza C., Florensa-Zanuy E., Díaz Á., Urigüen L., Castro E., Pazos A., Callado L.F. The endocannabinoid system in mental disorders: Evidence from human brain studies. Biochem. Pharmacol. 2018;157:97–107. doi: 10.1016/j.bcp.2018.07.009. [DOI] [PubMed] [Google Scholar]
- 121.Gärtner A., Dörfel D., Diers K., Witt S.H., Strobel A., Brocke B. Impact of FAAH genetic variation on fronto-amygdala function during emotional processing. Eur. Arch. Psychiatry Clin. Neurosci. 2019;269(2):209–221. doi: 10.1007/s00406-018-0944-9. [DOI] [PubMed] [Google Scholar]
- 122.Monteleone P., Bifulco M., Maina G., Tortorella A., Gazzerro P., Proto M.C., Di Filippo C., Monteleone F., Canestrelli B., Buonerba G., Bogetto F., Maj M. Investigation of CNR1 and FAAH endocannabinoid gene polymorphisms in bipolar disorder and major depression. Pharmacol. Res. 2010;61(5):400–404. doi: 10.1016/j.phrs.2010.01.002. [DOI] [PubMed] [Google Scholar]
- 123.Vinod K.Y., Xie S., Psychoyos D., Hungund B.L., Cooper T.B., Tejani-Butt S.M. Dysfunction in fatty acid amide hydrolase is associated with depressive-like behavior in Wistar Kyoto rats. PLoS One. 2012;7(5):e36743. doi: 10.1371/journal.pone.0036743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Smaga I., Jastrzębska J., Zaniewska M., Bystrowska B., Gawliński D., Faron-Górecka A., Broniowska Ż., Miszkiel J., Filip M. Changes in the brain endocannabinoid system in rat models of depression. Neurotox. Res. 2017;31(3):421–435. doi: 10.1007/s12640-017-9708-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Jenniches I., Ternes S., Albayram O., Otte D.M., Bach K., Bindila L., Michel K., Lutz B., Bilkei-Gorzo A., Zimmer A. Anxiety, stress, and fear response in mice with reduced endocannabinoid levels. Biol. Psychiatry. 2016;79(10):858–868. doi: 10.1016/j.biopsych.2015.03.033. [DOI] [PubMed] [Google Scholar]
- 126.Patel S., Hill M.N., Cheer J.F., Wotjak C.T., Holmes A. The endocannabinoid system as a target for novel anxiolytic drugs. . Neurosci. Biobehav. Rev. 2017;76(Pt A):56–66. doi: 10.1016/j.neubiorev.2016.12.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Bedse G., Bluett R.J., Patrick T.A., Romness N.K., Gaulden A.D., Kingsley P.J., Plath N., Marnett L.J., Patel S. Therapeutic endocannabinoid augmentation for mood and anxiety disorders: comparative profiling of FAAH, MAGL and dual inhibitors. Transl. Psychiatry. 2018;8(1):92. doi: 10.1038/s41398-018-0141-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Berardi A., Schelling G., Campolongo P. The endocannabinoid system and Post Traumatic Stress Disorder (PTSD): From preclinical findings to innovative therapeutic approaches in clinical settings. Pharmacol. Res. 2016;111:668–678. doi: 10.1016/j.phrs.2016.07.024. [DOI] [PubMed] [Google Scholar]
- 129.Vimalanathan A., Gidyk D.C., Diwan M., Gouveia F.V., Lipsman N., Giacobbe P., Nobrega J.N., Hamani C. Endocannabinoid modulating drugs improve anxiety but not the expression of conditioned fear in a rodent model of post-traumatic stress disorder. Neuropharmacology. 2020;166:107965. doi: 10.1016/j.neuropharm.2020.107965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Fidelman S., Mizrachi Zer-Aviv T., Lange R., Hillard C.J., Akirav I. Chronic treatment with URB597 ameliorates post-stress symptoms in a rat model of PTSD. Eur. Neuropsychopharmacol. 2018;28(5):630–642. doi: 10.1016/j.euroneuro.2018.02.004. [DOI] [PubMed] [Google Scholar]
- 131.Kruk-Slomka M., Banaszkiewicz I., Slomka T., Biala G. Effects of fatty acid amide hydrolase inhibitors acute administration on the positive and cognitive symptoms of schizophrenia in mice. Mol. Neurobiol. 2019;56(11):7251–7266. doi: 10.1007/s12035-019-1596-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Watts J.J., Jacobson M.R., Lalang N., Boileau I., Tyndale R.F., Kiang M., Ross R.A., Houle S., Wilson A.A., Rusjan P., Mizrahi R. Imaging brain fatty acid amide hydrolase in untreated patients with psychosis. Biol. Psychiatry. 2020;88(9):727–735. doi: 10.1016/j.biopsych.2020.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Bioque M., García-Bueno B., Macdowell K.S., Meseguer A., Saiz P.A., Parellada M., Gonzalez-Pinto A., Rodriguez-Jimenez R., Lobo A., Leza J.C., Bernardo M. FLAMM-PEPs study—Centro de Investigación Biomédica en Red de Salud Mental. Peripheral endocannabinoid system dysregulation in first-episode psychosis. Neuropsychopharmacol. 2013;38(13):2568–2577. doi: 10.1038/npp.2013.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Razzouk D. Mental Health Economics: The Costs and Benefits of Psychiatric Care. 1st ed. Switzerland: Springer; 2017. [Google Scholar]
- 135.World Health Organization Mental disorders. who.int/en/news-room/fact-sheets/detail/mental-disorders
- 136.Plana-Ripoll O., Pedersen C.B., Holtz Y., Benros M.E., Dalsgaard S., de Jonge P., Fan C.C., Degenhardt L., Ganna A., Greve A.N., Gunn J., Iburg K.M., Kessing L.V., Lee B.K., Lim C.C.W., Mors O., Nordentoft M., Prior A., Roest A.M., Saha S., Schork A., Scott J.G., Scott K.M., Stedman T., Sørensen H.J., Werge T., Whiteford H.A., Laursen T.M., Agerbo E., Kessler R.C., Mortensen P.B., McGrath J.J. Exploring comorbidity within mental disorders among a danish national population. JAMA Psychiatry. 2019;76(3):259–270. doi: 10.1001/jamapsychiatry.2018.3658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Ngo V.K., Rubinstein A., Ganju V., Kanellis P., Loza N., Rabadan-Diehl C., Daar A.S. Grand challenges: Integrating mental health care into the non-communicable disease agenda. PLoS Med. 2013;10(5):e1001443–e1001443. doi: 10.1371/journal.pmed.1001443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Saini S.M., Mancuso S.G., Mostaid M.S., Liu C., Pantelis C., Everall I.P., Bousman C.A. Meta-analysis supports GWAS-implicated link between GRM3 and schizophrenia risk. Transl. Psychiatry. 2017;7(8):e1196. doi: 10.1038/tp.2017.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Paul K.C., Haan M., Mayeda E.R., Ritz B.R. Ambient air pollution, noise, and late-life cognitive decline and dementia risk. Annu. Rev. Public Health. 2019;40:203–220. doi: 10.1146/annurev-publhealth-040218-044058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Barbon A., Magri C. RNA Editing and modifications in mood disorders. Genes (Basel) 2020;11(8):E872. doi: 10.3390/genes11080872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Trifu S.C., Vlăduţi A., Trifu A.I. Genetic aspects in schizophrenia. Receptoral theories. Metabolic theories. Rom. J. Morphol. Embryol. 2020;61(1):25–32. doi: 10.47162/RJME.61.1.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Baselmans B.M.L., Yengo L., van Rheenen W., Wray N.R. Risk in relatives, heritability, SNP-based heritability, and genetic correlations in psychiatric disorders: a review. . Biol. Psychiat. . 2020;S0006-3223(20):31669–31673. doi: 10.1016/j.biopsych.2020.05.034. [DOI] [PubMed] [Google Scholar]
- 143.Godos J., Currenti W., Angelino D., Mena P., Castellano S., Caraci F., Galvano F., Del Rio D., Ferri R., Grosso G. diet and mental health: review of the recent updates on molecular mechanisms. Antioxidants. 2020;9(4):346. doi: 10.3390/antiox9040346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Garcia-Rizo C., Bitanihirwe B.K.Y. Implications of early life stress on fetal metabolic programming of schizophrenia: A focus on epiphenomena underlying morbidity and early mortality. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2020;101:109910. doi: 10.1016/j.pnpbp.2020.109910. [DOI] [PubMed] [Google Scholar]
- 145.Thornicroft G. Physical health disparities and mental illness: the scandal of premature mortality. Br. J. Psychiatry. 2011;199(6):441–442. doi: 10.1192/bjp.bp.111.092718. [DOI] [PubMed] [Google Scholar]
- 146.Ashworth M., Schofield P., Das-Munshi J. Physical health in severe mental illness. Br. J. Gen. Pract. 2017;67(663):436–437. doi: 10.3399/bjgp17X692621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Stern S., Linker S., Vadodaria K.C., Marchetto M.C., Gage F.H. Prediction of response to drug therapy in psychiatric disorders. Focus Am. Psychiatr. Publ. 2019;17(3):294–307. doi: 10.1176/appi.focus.17304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Catanzano M., Bennett S.D., Sanderson C., Patel M., Manzotti G., Kerry E., Coughtrey A.E., Liang H., Heyman I., Shafran R. Brief psychological interventions for psychiatric disorders in young people with long term physical health conditions: A systematic review and meta-analysis. J. Psychosom. Res. 2020;136:110187. doi: 10.1016/j.jpsychores.2020.110187. [DOI] [PubMed] [Google Scholar]
- 149.Christ C., Schouten M.J.E., Blankers M., van Schaik D.J., Beekman A.T., Wisman M.A., Stikkelbroek Y.A., Dekker J.J. Internet and computer-based cognitive behavioral therapy for anxiety and depression in adolescents and young adults: systematic review and meta-analysis. J. Med. Internet Res. 2020;22(9):e17831. doi: 10.2196/17831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Zschucke E., Gaudlitz K., Ströhle A. Exercise and physical activity in mental disorders: Clinical and experimental evidence. J. Prev. Med. Public Health. 2013;46(Suppl. 1):S12–S21. doi: 10.3961/jpmph.2013.46.S.S12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.American College of Sports Medicine . ACSM’s Guidelines for Exercise Testing and Prescription. 10th ed. Philadelphia: Wolters Kluwer; 2017. [Google Scholar]
- 152.Budde H., Schwarz R., Velasques B., Ribeiro P., Holzweg M., Machado S., Brazaitis M., Staack F., Wegner M. The need for differentiating between exercise, physical activity, and training. Autoimmun. Rev. 2016;15(1):110–111. doi: 10.1016/j.autrev.2015.09.004. [DOI] [PubMed] [Google Scholar]
- 153.Wegner M., Amatriain-Fernández S., Kaulitzky A., Murillo-Rodriguez E., Machado S., Budde H. Systematic review of meta-analyses: exercise effects on depression in children and adolescents. Front. Psychiatry. 2020;11:81. doi: 10.3389/fpsyt.2020.00081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.World Health Organization Physical activity. who.int/news-room/fact-sheets/detail/physical-activity
- 155.Budde H., Wegner M., Soya H., Voelcker-Rehage C., McMorris T. Neuroscience of exercise: neuroplasticity and its behavioral consequences. Neural Plast. 2016;2016:3643879. doi: 10.1155/2016/3643879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Budde H., Velasques B., Ribeiro P., Soya H. Editorial: Neuromodulation of exercise: impact on different kinds of behavior. Front. Neurosci. 2020;14:455. doi: 10.3389/fnins.2020.00455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Amatriain-Fernández S., Murillo-Rodríguez E.S., Gronwald T., Machado S., Budde H. Benefits of physical activity and physical exercise in the time of pandemic. Psychol. Trauma. 2020;12(S1):S264–S266. doi: 10.1037/tra0000643. [DOI] [PubMed] [Google Scholar]
- 158.Amatriain-Fernández S., Gronwald T., Murillo-Rodríguez E., Imperatori C., Solano A.F., Latini A., Budde H. Physical Exercise potentials against viral diseases like COVID-19 in the elderly. Front. Med. (Lausanne) 2020;7:379. doi: 10.3389/fmed.2020.00379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Craft L.L., Perna F.M. The benefits of exercise for the clinically depressed. Prim. Care Companion J. Clin. Psychiatry. 2004;6(3):104–111. doi: 10.4088/PCC.v06n0301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Stern S., Linker S., Vadodaria K.C., Marchetto M.C., Gage F.H. Prediction of response to drug therapy in psychiatric disorders. Open Biol. 2018;8(5):180031. doi: 10.1098/rsob.180031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Martinsen E.W. Benefits of exercise for the treatment of depression. Sports Med. 1990;9(6):380–389. doi: 10.2165/00007256-199009060-00006. [DOI] [PubMed] [Google Scholar]
- 162.Achttien R., van Lieshout J., Wensing M., van der Sanden M.N., Staal J.B. Symptoms of depression are associated with physical inactivity but not modified by gender or the presence of a cardiovascular disease; a cross-sectional study. BMC Cardiovasc. Disord. 2019;19(1):95. doi: 10.1186/s12872-019-1065-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Rethorst C.D., Wipfli B.M., Landers D.M. The antidepressive effects of exercise: a meta-analysis of randomized trials. Sports Med. 2009;39(6):491–511. doi: 10.2165/00007256-200939060-00004. [DOI] [PubMed] [Google Scholar]
- 164.Wegner M., Helmich I., Machado S., Nardi A.E., Arias-Carrion O., Budde H. Effects of exercise on anxiety and depression disorders: review of meta- analyses and neurobiological mechanisms. CNS Neurol. Disord. Drug Targets. 2014;13(6):1002–1014. doi: 10.2174/1871527313666140612102841. [DOI] [PubMed] [Google Scholar]
- 165.Wegner M., Müller-Alcazar A., Jäger A., Machado S., Arias-Carrión O., Budde H. Psychosocial stress but not exercise increases cortisol and reduces state anxiety levels in school classes - results from a stressor applicable in large group settings. CNS Neurol. Disord. Drug Targets. 2014;13(6):1015–1020. doi: 10.2174/1871527313666140612103425. [DOI] [PubMed] [Google Scholar]
- 166.Hu M.X., Turner D., Generaal E., Bos D., Ikram M.K., Ikram M.A., Cuijpers P., Penninx B.W.J.H. Exercise interventions for the prevention of depression: a systematic review of meta-analyses. BMC Public Health. 2020;20(1):1255. doi: 10.1186/s12889-020-09323-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Galper D.I., Trivedi M.H., Barlow C.E., Dunn A.L., Kampert J.B. Inverse association between physical inactivity and mental health in men and women. Med. Sci. Sports Exerc. 2006;38(1):173–178. doi: 10.1249/01.mss.0000180883.32116.28. [DOI] [PubMed] [Google Scholar]
- 168.de Souza Moura A.M., Lamego M.K., Paes F., Ferreira Rocha N.B., Simoes-Silva V., Rocha S.A., de Sá Filho A.S., Rimes R., Manochio J., Budde H., Wegner M., Mura G., Arias-Carrión O., Yuan T.F., Nardi A.E., Machado S. Comparison among aerobic exercise and other types of interventions to treat depression: a systematic review. CNS Neurol. Disord. Drug Targets. 2015;14(9):1171–1183. doi: 10.2174/1871527315666151111120714. [DOI] [PubMed] [Google Scholar]
- 169.North T.C., McCullagh P., Tran Z.V. Effect of exercise on depression. Exerc. Sport Sci. Rev. 1990;18:379–415. doi: 10.1249/00003677-199001000-00016. [DOI] [PubMed] [Google Scholar]
- 170.Craft L.L., Landers D.M. The effect of exercise on clinical depression and depression resulting from mental illness: a meta-analysis. J. Sport Exerc. Psychol. 1998;20(4):339–357. doi: 10.1123/jsep.20.4.339. [DOI] [Google Scholar]
- 171.Bartholomew J.B., Morrison D., Ciccolo J.T. Effects of acute exercise on mood and well-being in patients with major depressive disorder. Med. Sci. Sports Exerc. 2005;37(12):2032–2037. doi: 10.1249/01.mss.0000178101.78322.dd. [DOI] [PubMed] [Google Scholar]
- 172.Blumenthal J.A., Babyak M.A., Doraiswamy P.M., Watkins L., Hoffman B.M., Barbour K.A., Herman S., Craighead W.E., Brosse A.L., Waugh R., Hinderliter A., Sherwood A. Exercise and pharmacotherapy in the treatment of major depressive disorder. Psychosom. Med. 2007;69(7):587–596. doi: 10.1097/PSY.0b013e318148c19a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Hughes C.W., Barnes S., Barnes C., Defina L.F., Nakonezny P., Emslie G.J. Depressed adolescents treated with exercise (DATE): A pilot randomized controlled trial to test feasibility and establish preliminary effect sizes. Ment. Health Phys. Act. 2013;6(2):119–131. doi: 10.1016/j.mhpa.2013.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Brand S., Colledge F., Ludyga S., Emmenegger R., Kalak N., Sadeghi Bahmani D., Holsboer-Trachsler E., Pühse U., Gerber M. Acute bouts of exercising improved mood, rumination and social interaction in inpatients with mental disorders. Front. Psychol. 2018;9:249. doi: 10.3389/fpsyg.2018.00249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Budde H., Velasques B., Ribeiro P., Machado S., Emeljanovas A., Kamandulis S., Skurvydas A., Wegner M. Does intensity or youth affect the neurobiological effect of exercise on major depressive disorder? Neurosci. Biobehav. Rev. 2018;84:492–494. doi: 10.1016/j.neubiorev.2016.09.026. [DOI] [PubMed] [Google Scholar]
- 176.Bueno-Antequera J., Munguía-Izquierdo D. Exercise and Depressive Disorder. Adv. Exp. Med. Biol. 2020;1228:271–287. doi: 10.1007/978-981-15-1792-1_18. [DOI] [PubMed] [Google Scholar]
- 177.Gronwald T., Velasques B., Ribeiro P., Machado S., Murillo-Rodríguez E., Ludyga S., Yamamoto T., Budde H. Increasing exercise’s effect on mental health: Exercise intensity does matter. Proc. Natl. Acad. Sci. USA. 2018;115(51):E11890–E11891. doi: 10.1073/pnas.1818161115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Paolucci E.M., Loukov D., Bowdish D.M.E., Heisz J.J. Exercise reduces depression and inflammation but intensity matters. Biol. Psychol. 2018;133:79–84. doi: 10.1016/j.biopsycho.2018.01.015. [DOI] [PubMed] [Google Scholar]
- 179.Herold F., Törpel A., Hamacher D., Budde H., Gronwald T. A Discussion on different approaches for prescribing physical interventions - four roads lead to rome, but which one should we choose? J. Pers. Med. 2020;10(3):55. doi: 10.3390/jpm10030055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Wittchen H.U., Jacobi F., Rehm J., Gustavsson A., Svensson M., Jönsson B., Olesen J., Allgulander C., Alonso J., Faravelli C., Fratiglioni L., Jennum P., Lieb R., Maercker A., van Os J., Preisig M., Salvador-Carulla L., Simon R., Steinhausen H.C. The size and burden of mental disorders and other disorders of the brain in Europe 2010. Eur. Neuropsychopharmacol. 2011;21(9):655–679. doi: 10.1016/j.euroneuro.2011.07.018. [DOI] [PubMed] [Google Scholar]
- 181.American Psychiatric Association Help With Anxiety Disorders. psychiatry.org/patients-families/anxiety-disorders/what-are-anxiety-disorders
- 182.Creswell C., Waite P., Cooper P.J. Assessment and management of anxiety disorders in children and adolescents. Arch. Dis. Child. 2014;99(7):674–678. doi: 10.1136/archdischild-2013-303768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Bandelow B., Michaelis S., Wedekind D. Treatment of anxiety disorders. Dialogues Clin. Neurosci. 2017;19(2):93–107. doi: 10.31887/DCNS.2017.19.2/bbandelow. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Taylor A.H. In: Physical activity, anxiety, and stress. Physical Activity and Psychological Well-Being; Biddle, J.H.; Fox, K.R. Boutcher S.H., editor. Vol. 1. London: Taylor & Francis Group; 2000. pp. 10–45. [Google Scholar]
- 185.Vancampfort D., Rosenbaum S., Probst M., Stubbs B. In: Aerobic exercise in people with schizophrenia: from efficacy to effectiveness. The exercise effect on mental health: neurobiological mechanisms; Budde, H. Wegner M., editor. Vol. 1. New York: Taylor & Francis Group; 2018. pp. 312–333. [Google Scholar]
- 186.Wipfli B.M., Rethorst C.D., Landers D.M. The anxiolytic effects of exercise: a meta-analysis of randomized trials and dose-response analysis. J. Sport Exerc. Psychol. 2008;30(4):392–410. doi: 10.1123/jsep.30.4.392. [DOI] [PubMed] [Google Scholar]
- 187.Anderson E., Shivakumar G. Effects of exercise and physical activity on anxiety. Front. Psychiatry. 2013;4:27. doi: 10.3389/fpsyt.2013.00027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.de Souza Moura A.M., Lamego M.K., Paes F., Ferreira Rocha N.B., Simoes-Silva V., Rocha S.A., de Sá Filho A.S., Rimes R., Manochio J., Budde H., Wegner M., Mura G., Arias-Carrión O., Yuan T.F., Nardi A.E., Machado S. Effects of aerobic exercise on anxiety disorders: a systematic review. CNS Neurol. Disord. Drug Targets. 2015;14(9):1184–1193. doi: 10.2174/1871527315666151111121259. [DOI] [PubMed] [Google Scholar]
- 189.Mumm J., Bischoff S. In: StrÖhle, A. Exercise and anxiety disorders. The exercise effect on mental health: neurobiological mechanisms; Budde, H. Wegner M., editor. Vol. 1. New York: Taylor & Francis Group; 2018. pp. 334–357. [Google Scholar]
- 190.Rosenbaum S., Sherrington C., Tiedemann A. Exercise augmentation compared with usual care for post-traumatic stress disorder: a randomized controlled trial. Acta Psychiatr. Scand. 2015;131(5):350–359. doi: 10.1111/acps.12371. [DOI] [PubMed] [Google Scholar]
- 191.Rosenbaum S., Vancampfort D., Steel Z., Newby J., Ward P.B., Stubbs B. Physical activity in the treatment of Post-traumatic stress disorder: A systematic review and meta-analysis. Psychiatry Res. 2015;230(2):130–136. doi: 10.1016/j.psychres.2015.10.017. [DOI] [PubMed] [Google Scholar]
- 192.Hegberg N.J., Hayes J.P., Hayes S.M. Exercise intervention in PTSD: A narrative review and rationale for implementation. Front. Psychiatry. 2019;10:133–133. doi: 10.3389/fpsyt.2019.00133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Budde H., Akko D.P., Ainamani H.E., Murillo-Rodríguez E., Weierstall R. The impact of an exercise training intervention on cortisol levels and post-traumatic stress disorder in juveniles from an Ugandan refugee settlement: study protocol for a randomized control trial. Trials. 2018;19(1):364–364. doi: 10.1186/s13063-018-2753-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Oertel-Knöchel V., Mehler P., Thiel C., Steinbrecher K., Malchow B., Tesky V., Ademmer K., Prvulovic D., Banzer W., Zopf Y., Schmitt A., Hänsel F. Effects of aerobic exercise on cognitive performance and individual psychopathology in depressive and schizophrenia patients. Eur. Arch. Psychiatry Clin. Neurosci. 2014;264(7):589–604. doi: 10.1007/s00406-014-0485-9. [DOI] [PubMed] [Google Scholar]
- 195.Pajonk F.G., Wobrock T., Gruber O., Scherk H., Berner D., Kaizl I., Kierer A., Müller S., Oest M., Meyer T., Backens M., Schneider-Axmann T., Thornton A.E., Honer W.G., Falkai P. Hippocampal plasticity in response to exercise in schizophrenia. Arch. Gen. Psychiatry. 2010;67(2):133–143. doi: 10.1001/archgenpsychiatry.2009.193. [DOI] [PubMed] [Google Scholar]
- 196.Wang P.W., Lin H.C., Su C.Y., Chen M.D., Lin K.C., Ko C.H., Yen C.F. Effect of aerobic exercise on improving symptoms of individuals with schizophrenia: a single blinded randomized control study. Front. Psychiatry. 2018;9:167. doi: 10.3389/fpsyt.2018.00167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Gorczynski P., Faulkner G. Exercise therapy for schizophrenia. Cochrane Database Syst. Rev. 2010;5(5):CD004412. doi: 10.1002/14651858.CD004412.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Vancampfort D., Firth J., Schuch F.B., Rosenbaum S., Mugisha J., Hallgren M., Probst M., Ward P.B., Gaughran F., De Hert M., Carvalho A.F., Stubbs B. Sedentary behavior and physical activity levels in people with schizophrenia, bipolar disorder and major depressive disorder: a global systematic review and meta-analysis. World Psychiatry. 2017;16(3):308–315. doi: 10.1002/wps.20458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Duraiswamy G., Thirthalli J., Nagendra H.R., Gangadhar B.N. Yoga therapy as an add-on treatment in the management of patients with schizophrenia--a randomized controlled trial. Acta Psychiatr. Scand. 2007;116(3):226–232. doi: 10.1111/j.1600-0447.2007.01032.x. [DOI] [PubMed] [Google Scholar]
- 200.Gronwald T., Budde H. Commentary: physical exercise as personalized medicine for dementia prevention? Front. Physiol. 2019;10:1358. doi: 10.3389/fphys.2019.01358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Brellenthin A.G., Crombie K.M., Hillard C.J., Koltyn K.F. Endocannabinoid and mood responses to exercise in adults with varying activity levels. Med. Sci. Sports Exerc. 2017;49(8):1688–1696. doi: 10.1249/MSS.0000000000001276. [DOI] [PubMed] [Google Scholar]
- 202.Mikkelsen K., Stojanovska L., Polenakovic M., Bosevski M., Apostolopoulos V. Exercise and mental health. Maturitas. 2017;106:48–56. doi: 10.1016/j.maturitas.2017.09.003. [DOI] [PubMed] [Google Scholar]
- 203.Dietrich A., McDaniel W.F. Endocannabinoids and exercise. Br. J. Sports Med. 2004;38(5):536–541. doi: 10.1136/bjsm.2004.011718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Sparling P.B., Giuffrida A., Piomelli D., Rosskopf L., Dietrich A. Exercise activates the endocannabinoid system. Neuroreport. 2003;14(17):2209–2211. doi: 10.1097/00001756-200312020-00015. [DOI] [PubMed] [Google Scholar]
- 205.Raichlen D.A., Foster A.D., Gerdeman G.L., Seillier A., Giuffrida A. Wired to run: exercise-induced endocannabinoid signaling in humans and cursorial mammals with implications for the ‘runner’s high’. J. Exp. Biol. 2012;215(Pt 8):1331–1336. doi: 10.1242/jeb.063677. [DOI] [PubMed] [Google Scholar]
- 206.Heyman E., Gamelin F.X., Goekint M., Piscitelli F., Roelands B., Leclair E., Di Marzo V., Meeusen R. Intense exercise increases circulating endocannabinoid and BDNF levels in humans--possible implications for reward and depression. Psychoneuroendocrinology. 2012;37(6):844–851. doi: 10.1016/j.psyneuen.2011.09.017. [DOI] [PubMed] [Google Scholar]
- 207.Raichlen D.A., Foster A.D., Seillier A., Giuffrida A., Gerdeman G.L. Exercise-induced endocannabinoid signaling is modulated by intensity. Eur. J. Appl. Physiol. 2013;113(4):869–875. doi: 10.1007/s00421-012-2495-5. [DOI] [PubMed] [Google Scholar]
- 208.Borg G. Borg’s Perceived Exertion and Pain Scales. 1st ed. Champaign: Human Kinetics; 1998. [Google Scholar]
- 209.Lin T.W., Kuo Y.M. Exercise benefits brain function: the monoamine connection. Brain Sci. 2013;3(1):39–53. doi: 10.3390/brainsci3010039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Schönke M., Martinez-Tellez B., Rensen P.C. Role of the endocannabinoid system in the regulation of the skeletal muscle response to exercise. Curr. Opin. Pharmacol. 2020;52:52–60. doi: 10.1016/j.coph.2020.05.003. [DOI] [PubMed] [Google Scholar]
- 211.Fuss J., Gass P. Endocannabinoids and voluntary activity in mice: runner’s high and long-term consequences in emotional behaviors. Exp. Neurol. 2010;224(1):103–105. doi: 10.1016/j.expneurol.2010.03.016. [DOI] [PubMed] [Google Scholar]
- 212.Stone N.L., Millar S.A., Herrod P.J.J., Barrett D.A., Ortori C.A., Mellon V.A., O’Sullivan S.E. An Analysis of endocannabinoid concentrations and mood following singing and exercise in healthy volunteers. Front. Behav. Neurosci. 2018;12:269. doi: 10.3389/fnbeh.2018.00269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Garber C.E., Blissmer B., Deschenes M.R., Franklin B.A., Lamonte M.J., Lee I.M., Nieman D.C., Swain D.P. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med. Sci. Sports Exerc. 2011;43(7):1334–1359. doi: 10.1249/MSS.0b013e318213fefb. [DOI] [PubMed] [Google Scholar]
- 214.2019. covid19.who.int/
- 215.Kinross P., Suetens C., Gomes Dias J., Alexakis L., Wijermans A., Colzani E., Monnet D.L. Rapidly increasing cumulative incidence of coronavirus disease (COVID-19) in the European Union/European Economic Area and the United Kingdom, 1 January to 15 March 2020. Euro Surveill. 2020;25(11):10. doi: 10.2807/1560-7917.ES.2020.25.11.2000285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Chen J., Qi T., Liu L., Ling Y., Qian Z., Li T., Li F., Xu Q., Zhang Y., Xu S., Song Z., Zeng Y., Shen Y., Shi Y., Zhu T., Lu H. Clinical progression of patients with COVID-19 in Shanghai, China. J. Infect. 2020;80(5):e1–e6. doi: 10.1016/j.jinf.2020.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Ahsan W., Alhazmi H.A., Patel K.S., Mangla B., Al Bratty M., Javed S., Najmi A., Sultan M.H., Makeen H.A., Khalid A., Mohan S., Taha M.M.E., Sultana S. Recent advancements in the diagnosis, prevention, and prospective drug therapy of COVID-19. Front. Public Health. 2020;8:384. doi: 10.3389/fpubh.2020.00384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Jotz G.P., Stein A., Sirena S., Barros E., Baldisserotto J., Figueiredo J.A.P., Lavinsky J., Steier L., Dora C. The COVID-19 pandemic and planetary health. a critical review of epidemiology, prevention, clinical characteristics and treatments for oral, head and neck health professionals. do we have a roadmap? Int. Arch. Otorhinolaryngol. 2020;24(3):e351–e358. doi: 10.1055/s-0040-1714143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Pergolizzi J.V., Jr, Magnusson P., LeQuang J.A., Breve F., Paladini A., Rekatsina M., Yeam C.T., Imani F., Saltelli G., Taylor R., Jr, Wollmuth C., Varrassi G. The current clinically relevant findings on covid-19 pandemic. Anesth. Pain Med. 2020;10(2):e103819. doi: 10.5812/aapm.103819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Salari N., Hosseinian-Far A., Jalali R., Vaisi-Raygani A., Rasoulpoor S., Mohammadi M., Rasoulpoor S., Khaledi-Paveh B. Prevalence of stress, anxiety, depression among the general population during the COVID-19 pandemic: a systematic review and meta-analysis. Global. Health. 2020;16(1):57. doi: 10.1186/s12992-020-00589-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Steardo L., Jr, Steardo L., Verkhratsky A. Psychiatric face of COVID-19. Transl. Psychiatry. 2020;10(1):261. doi: 10.1038/s41398-020-00949-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Sher L. The impact of the COVID-19 pandemic on suicide rates. QJM. 2020;113(10):707–712. doi: 10.1093/qjmed/hcaa202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Imperatori C., Dakanalis A., Farina B., Pallavicini F., Colmegna F., Mantovani F., Clerici M. Global storm of stress-related psychopathological symptoms: a brief overview on the usefulness of virtual reality in facing the mental health impact of COVID-19. Cyberpsychol. Behav. Soc. Netw. 2020;23(11):782–788. doi: 10.1089/cyber.2020.0339. [DOI] [PubMed] [Google Scholar]
- 224.Yamamoto T., Uchiumi Ch., Suzuki N., Yoshimoto J., Murillo-Rodríguez E. The psychological impact of ‘mild lockdown’ in Japan during the COVID-19 pandemic: a nationwide survey under a declared state of emergency. medRxiv. 2020 doi: 10.3390/ijerph17249382. [DOI] [PMC free article] [PubMed]
- 225.Troyer E.A., Kohn J.N., Hong S. Are we facing a crashing wave of neuropsychiatric sequelae of COVID-19? Neuropsychiatric symptoms and potential immunologic mechanisms. Brain Behav. Immun. 2020;87:34–39. doi: 10.1016/j.bbi.2020.04.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Rogers J.P., Chesney E., Oliver D., Pollak T.A., McGuire P., Fusar-Poli P., Zandi M.S., Lewis G., David A.S. Psychiatric and neuropsychiatric presentations associated with severe coronavirus infections: a systematic review and meta-analysis with comparison to the COVID-19 pandemic. Lancet Psychiatry. 2020;7(7):611–627. doi: 10.1016/S2215-0366(20)30203-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Gennaro Mazza M., De Lorenzo R., Conte C., Poletti S., Vai B., Bollettini I., Melloni E.M.T., Furlan R., Ciceri F., Rovere-Querini P. COVID-19 BioB Outpatient Clinic Study group; Benedetti, F. Anxiety and depression in COVID-19 survivors: role of inflammatory and clinical predictors. Brain Behav. Immun. 2020;89:594–600. doi: 10.1016/j.bbi.2020.07.037. [DOI] [PMC free article] [PubMed] [Google Scholar]