BACKGROUND:
Previous studies have focused on the development and evaluation of care bundles to reduce the risk of surgical site infection (SSI) throughout the perioperative period. A focused examination of the technical/surgical aspects of SSI reduction during CRS has not been conducted. This study aimed to develop an expert consensus on intraoperative technical/surgical aspects of SSI prevention by the surgical team during colorectal surgery (CRS).
STUDY DESIGN:
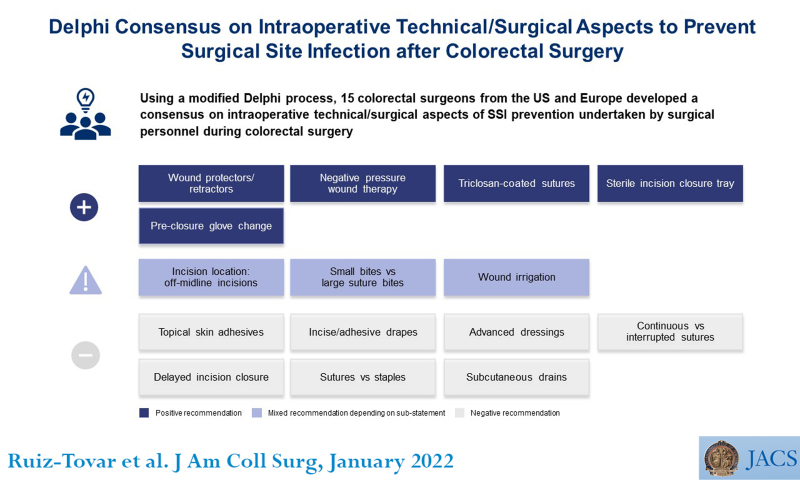
In a modified Delphi process, a panel of 15 colorectal surgeons developed a consensus on intraoperative technical/surgical aspects of SSI prevention undertaken by surgical personnel during CRS using information from a targeted literature review and expert opinion. Consensus was developed with up to three rounds per topic, with a prespecified threshold of ≥70% agreement.
RESULTS:
In 3 Delphi rounds, the 15 panelists achieved consensus on 16 evidence-based statements. The consensus panel supported the use of wound protectors/retractors, sterile incision closure tray, preclosure glove change, and antimicrobial sutures in reducing SSI along with wound irrigation with aqueous iodine and closed-incision negative pressure wound therapy in high-risk, contaminated wounds.
CONCLUSIONS:
Using a modified Delphi method, consensus has been achieved on a tailored set of recommendations on technical/surgical aspects that should be considered by surgical personnel during CRS to reduce the risk of SSI, particularly in areas where the evidence base is controversial or lacking. This document forms the basis for ongoing evidence for the topics discussed in this article or new topics based on newly emerging technologies in CRS.
In a modified Delphi process, a panel of 15 colorectal surgeons developed a consensus on intraoperative technical/surgical aspects of surgical site infection (SSI) prevention undertaken by surgical personnel during colorectal surgery to reduce the risk of SSI, particularly in areas where the evidence base is controversial or lacking.
Supplemental Digital Content is available in the text.
Colorectal surgery (CRS) is associated with one of the highest surgical site infection (SSI) rates after surgical procedures of all types, with reported incidences ranging up to 30%.1,2 CRS is associated with a particularly high risk of SSI resulting from contamination of the surgical site by the high bacterial load in the colon and rectum, as well as patient comorbidities which further increase risk.3,4 The increased risk of SSI contributes to a substantial clinical and economic burden, including higher costs, additional community care, longer hospitalization, reoperations, readmissions, reduced quality of life, and mortality.2,5,6
For these reasons, evidence-based surgical care bundles have been implemented as part of SSI prevention policies to reduce the incidence of SSI.1,7-9 Meta-analyses have shown that the implementation of such surgical care bundles, particularly those which include recommendations which have a level 1A evidence base, are associated with reduced SSI rates. One such meta-analysis, of 13 studies including 8,515 patients undergoing CRS, found that surgical care bundles significantly reduced the risk of SSI by half compared with standard care (relative risk [RR] = 0.55; 95% CI 0.39-0.77).9 Another meta-analysis, of 23 studies including 17,557 patients, similarly reported that bundles reduced superficial SSI by 40% (p < 0.001).1 However, it should be noted that the care bundles in the studies examined different interventions.
Components of these care bundles included preoperative, intraoperative, and postoperative components such as preoperative bathing, mechanical bowel preparation together with oral antibiotics, glycemic control, maintenance of perioperative normothermia, skin preparation, SSI education, and a variety of other technical and nontechnical items.1,7,9 Previous studies analyzing the value of surgical care bundles, as well as guidelines focused on prevention of SSI, have been directed mostly towards the use of bundles spanning the entire perioperative period. A more focused study centered strictly on the technical/surgical aspects that could be beneficially undertaken by surgical personnel during colorectal procedures to prevent SSI is lacking.
Therefore, the objective of the current study, using the Delphi process, was to develop an expert consensus on intraoperative technical/surgical aspects of SSI prevention undertaken by surgical personnel during CRS. The study was designed to provide a set of intraoperative recommendations, particularly where the evidence base is controversial or lacking, which can be used to add to and improve the technical/surgical components of the SSI care bundle.
METHODS
In October and November 2020, two virtual Delphi panel meetings were held. The Delphi panel included 15 colorectal surgeons from the US and Europe: two steering committee (SC) members and a working group of 13 members. The panel was selected based on research involvement in SSI bundles or enhanced recovery after surgery and/or the implementation of such measures, strong reputation of their affiliated hospital, leadership involvement in their affiliated hospital and/or a society, and SC recommendation. The SC agreed on 15 topics (Table 1) to be included in a Delphi consensus panel on intraoperative technical/surgical aspects performed by surgical personnel during CRS, with a focus on superficial/deep SSI prevention. Organ space SSIs were not considered in this study given the inherent differences in risk factors and preventive strategies between superficial/deep and organ space SSIs. The selection of topics was informed by literature review and expert opinion from the SC, particularly where the evidence base was lacking or controversial.
Table 1.
Summary of Statements on Technical/Surgical Aspects to Prevent Surgical Site Infection
| Topic (statement number) | Consensus statement |
|---|---|
| Incision location: off midline incision (1) | There is insufficient evidence that off-midline incision reduces the risk of SSI compared with midline incision. However, off-midline incision (when possible/appropriate) is associated with a reduced incisional hernia risk after (laparoscopic) colorectal surgery. |
| Wound protector/retractor (2) | Wound protectors/retractors are associated with reduced SSI risk when compared with no wound protectors/retractors. |
| Incise/adhesive drape (3) | There is insufficient evidence to support the role of incise/adhesive drape to reduce the risk of SSI. |
| Wound irrigation (4.1, 4.2) | Antibiotic incisional wound irrigation should not be used for reducing SSI risk. |
| Wound irrigation with aqueous iodine is associated with reduced SSI risk when compared with no irrigation in high-risk, contaminated wounds. | |
| Sterile incision closure tray (5) | Use of a separate, dedicated sterile incision closure tray may be useful in reducing SSI risk when compared with no sterile incision closure trays. |
| Preclosure glove change (6) | Preclosure glove changes may be useful in reducing SSI risk when compared with no glove change. |
| Small bites vs large bites (7) | There is insufficient evidence that a small bite suture technique reduces SSI risk compared with a large bite suture technique. However, the small bite suture technique is associated with reduced incisional hernia risk. |
| Triclosan coated suture (8) | Triclosan-coated or impregnated antimicrobial sutures are associated with a reduced risk of SSI compared with non-antimicrobial sutures. |
| Continuous vs interrupted sutures (9) | There is insufficient evidence to make a recommendation on the use of continuous or interrupted sutures on the impact of postoperative wound complication (SSI, incisional hernia, or wound dehiscence). |
| Sutures vs staples (10) | There is insufficient evidence to recommend the use of staples or subcuticular sutures for skin closure to reduce SSI. |
| Topical skin adhesive (11) | There is insufficient evidence to support the use of topical skin adhesives for superficial closure in reducing SSI. |
| Negative pressure wound therapy (12) | Closed-incision negative pressure wound therapy may be useful in reducing SSI risk when compared with no NPWT in open, high-risk, contaminated surgery. |
| Advanced dressing (13) | There is insufficient evidence to support the use of advanced dressings (for example silver dressings) as opposed to conventional dressings (such as standard gauze) to reduce the risk of SSI. |
| Delayed incision closure (14) | There is insufficient evidence to make a recommendation on the use of delayed incisional closure after open CRS. |
| Subcutaneous drains (15) | There is insufficient evidence to support the role of subcutaneous drains in reducing SSI risk. |
CRS, colorectal surgery; NPWT, negative pressure wound therapy; SSI, surgical site infection.
A targeted literature review was undertaken to identify and summarize published information from the last 10 years which aligned with the research question, specifically in colorectal or abdominal surgery. Electronic searches were performed in MEDLINE and PubMed databases, using keywords specific to the disease area and key topic (Supplemental Digital Content 1, available at http://links.lww.com/XCS/A9). Supplemental cascading searches were performed on the reference lists of recent systematic reviews, previously conducted Delphi consensus studies and guidelines in colorectal or abdominal surgery, as well as national or global guidelines which focused on SSI prevention. Meta-analyses, randomized controlled trials (RCTs), and observational (prospective or retrospective) studies were included. After review of 1,047 abstracts and supplementary searches, 56 studies were included for review and consideration by the Delphi panel.
All 15 members of the Delphi panel participated in the voting process. A modified version of the standard Delphi process10 was used, which involved anonymous voting based on reviewed literature and expert opinion discussing the available evidence for each topic. Consensus was developed with up to three rounds per topic and a prespecified required threshold of ≥70% agreement. The first round included an electronic survey, where a focused list of draft statements and information-seeking questions, to understand expert opinion and treatment practices, was addressed by panel participants. The second and third rounds were interactive and included clinical discussions around the summarized literature combined with iterative anonymous voting on revised consensus statements. Clinical discussion was led by the SC and focused on developing recommendations based on the published literature in colorectal or abdominal surgery. If no such evidence was available or a component of the care bundle was controversial, expert opinion was used to develop recommendations.
RESULTS
Incision location
Statement 1: There is insufficient evidence that off-midline incisions reduce the risk of SSI compared with midline incisions. However, off-midline incisions (when possible/appropriate) are associated with a reduced incisional hernia risk after (laparoscopic) colorectal surgery.
The most common approaches for specimen retrieval after laparoscopic colorectal resection are midline, transverse, and Pfannenstiel incisions.11 There is a paucity of evidence regarding the impact of incision location on SSI. However, evidence from a meta-analysis of mostly observational studies has shown that the use of midline incisions was associated with significantly higher risk of incisional hernia (odds ratio [OR] = 4.1; 95% CI 2.0-8.3) compared with off-midline (transverse or Pfannenstiel incisions).12 It should be noted that incision location depends on the laparoscopic CRS procedure, and off-midline incisions are not appropriate in all patients, such as those who are expected to require a stoma.
Wound protectors/retractors
Statement 2: Wound protectors/retractors are associated with reduced SSI risk when compared with no wound protectors/retractors.
Wound protectors (commonly referred to as plastic ring wound protectors, wound retractors, or wound-edge protectors) theoretically act as an impervious barrier to protect the incision from contamination with the surgical field after enterotomy/resection. Wound protectors currently feature a single or double ringed configuration to secure the device within the wound.
The value of wound protectors to prevent SSI after CRS has been reported in a recent meta-analysis of RCTs which found that wound protectors significantly reduced superficial SSI compared with no wound protectors after abdominal surgery (OR = 0.42; 95% CI 0.18-0.95).13 Similar findings were reported in another meta-analysis which performed a CRS subgroup analysis.14 The use of wound protectors has been shown to significantly reduce SSI after open and laparoscopic CRS, pancreaticoduodenectomy, and open elective digestive surgery.15-19
Several meta-analyses have addressed the potential benefit of dual-ring vs single-ring wound protectors in reducing SSI risk through subgroup analyses, but there is no clear direct evidence showing superiority for either device. Comparisons from current meta-analyses have compared relative risks for each device compared with no wound protector.14,20-23 A formal network meta-analysis has not been conducted but is warranted to better understand whether there are differences between device types.
Incise/adhesive drapes
Statement 3: There is insufficient evidence to support the role of incise/adhesive drapes to reduce the risk of SSI.
Incise drapes (plain or antimicrobial-impregnated) are plastic surgical drapes with a sterile adhesive transparent film that adheres to the skin with the intention of confining skin organisms during surgical procedures. Although used for more than 30 years,24 few studies have evaluated the impact of these drapes on SSI after colorectal or abdominal surgery. One observational study (n = 296) showed that use of plastic adhesive drapes impregnated with iodophor was associated with significantly lower wound infection compared with surgery without iodophor drapes in liver surgery (3.1% vs. 12.1%; p = 0.0096).25 Another observational study (n = 91) demonstrated similar rates of SSI between incise drapes and standard skin preparation with 10% povidone iodine soap in patients undergoing appendectomy.26 National guidelines from the Centers for Disease Control and Prevention (CDC), the World Health Organization (WHO), and the National Institute for Health and Care Excellence (NICE) do not recommend the use of incise drapes.27-29
During the Delphi process, panel members described other pragmatic uses of incise drapes including isolation of a stoma during surgery or prevention of drape slippage during minimally invasive cases, to maintain integrity of the operative field, where the patient is being repositioned frequently.
Wound irrigation
Statement 4.1: Antibiotic incisional wound irrigation should not be used for reducing SSI risk.
Statement 4.2: Wound irrigation with aqueous iodine is associated with reduced SSI risk when compared with no irrigation in high-risk, contaminated wounds.
Incisional wound irrigation involves the use of a solution in an open wound to remove cellular debris and reduce microbial contamination/bacterial load at the end of a surgical procedure. A variety of solutions may be used, including saline, an aqueous iodophor such as povidone-iodine (PVP-I), or antibiotics.30 Use of each irrigating solution during surgery is dependent on a number of factors and the patient population.30
A large meta-analysis of various types of surgery analyzed a subgroup of RCTs in clean and clean-contaminated surgery and reported that PVP-I significantly reduced SSI compared with saline irrigation (OR = 0.31; 95% CI 0.13-0.73).31 Conversely, antibiotic wound irrigation did not impact SSI risk when compared with saline (OR = 1.16; 95% CI 0.64-2.12).31 Another meta-analysis of RCTs in abdominal surgery reported that PVP-I irrigation resulted in significantly lower SSI compared with no irrigation (OR = 0.70; 95% CI 0.51-0.97), whereas no significant difference was observed between saline irrigation compared with no irrigation (OR = 0.64; 95% CI 0.28-1.46).32 Another observational cohort of 122 patients reported that irrigation with 0.05% chlorohexidine reduces SSI compared with saline irrigation (18% compared with 31.6%; p < 0.001). Guidelines from WHO and CDC have suggested that intraoperative subcutaneous/incisional irrigation with PVP-I should be used.27,28 However, CDC, NICE, and WHO have suggested uncertain tradeoffs or recommended against the use of antibiotic solutions for incisional wound irrigation, in particular the risk of promoting antimicrobial resistance.27-29
Despite the wealth of evidence from meta-analyses on wound irrigation, the information is heterogenous and may have a risk of bias, thereby creating challenges in drawing meaningful conclusions. The most compelling evidence appears to favor wound irrigation with PVP-I to reduce SSI whereas evidence for saline irrigation is weak and antibiotic irrigation unfavorable given risk/benefit tradeoffs.
Sterile incision closure tray
Statement 5: Use of a separate, dedicated sterile incision closure tray may be useful in reducing SSI risk when compared with no sterile incision closure trays.
Switching to an unused set of sterile instruments, specifically for incision closure, has become a common component of SSI prevention bundles.9 The use of a sterile incision closure tray can serve as part of a large, team-building effort for surgical personnel and signals that the procedure is ending. Use of sterile incision closure trays may have additional utility in specific situations: the avoidance of contamination when inserting an abdominal mesh for example.
The use of sterile incision closure trays to reduce SSI risk has not been evaluated independently but rather as a component of larger bundles. However, efforts have been made to compare surgical care bundles which include or exclude the use of sterile incision closure trays. For example, one meta-analysis concluded that bundles with sterile closure trays were associated with a significant SSI reduction compared with bundles without sterile closure trays in CRS procedures (58.6% compared with 33.1%; p = 0.019).1 It is difficult to draw evidence-based conclusions because other bundle components were not controlled for. In terms of guidelines, the American College of Surgeons (ACS) and Surgical Infection Society of North America (SIS-NA) recommend the use of new instruments for closure specifically for colorectal cases,33 whereas the WHO guideline do not.28
Although there is a lack of clear data on the efficacy of dedicated, sterile incision closure trays to reduce the risk of SSI, their use is recommended by the panel for several reasons. Intuitively, the use of sterile instruments for closure after CRS may reduce the risk of contamination. Additionally, these trays are commonly used, easy to implement, and inexpensive, making their use practical, as part of a care bundle to reduce SSIs.
Preclosure glove change
Statement 6: Preclosure glove changes may be useful in reducing SSI risk when compared with no glove changes.
Glove changes are an important consideration because micro-perforations can occur with increasing duration of wear during surgical procedures.34,35 Moreover, gloves may become contaminated once the bowel is opened. Evidence has shown that changing gloves reduces the degree of bacterial contamination.36
The direct impact of surgical glove changes on SSI has not been independently studied. The best evidence in CRS comes from the same meta-analysis which concluded that care bundles, which included preclosure glove changes, were associated with an SSI reduction compared with bundles without preclosure glove changes (56.9% vs. 28.5%; p = 0.002). However, other bundle components were not controlled for.1 Guidelines by ACS and SIS and the World Society of Emergency Surgery (WSES) recommend preclosure glove changes to reduce SSI risk.33,37
Small bites compared with large bites in musculo-fascial closure
Statement 7: There is insufficient evidence that a small bite suture technique reduces SSI risk compared with a large bite suture technique. However, the small bite suture technique is associated with reduced incisional hernia risk.
Small bite suturing involves approximately 5-mm tissue bites being placed 5 mm apart, whereas large bite suturing incorporates 10-mm tissue bites 10 mm apart.38 Evidence from a small meta-analysis of RCTs in elective laparotomies found no difference in SSI between the two techniques (OR = 0.67; 95% CI 0.39–1.15) but a significantly lower risk of incisional hernia was found following the use of small bites (OR = 0.41; 95% CI 0.19–0.86).39 Recent evidence from an observational study has reported fewer SSIs after a small bite technique for abdominal wound closure of midline laparotomy (17% vs. 28%; p = 0.02).38
Results from this evidence are difficult to interpret as wound outcome definitions may be heterogeneous across studies. However, the benefits of small bites to reduce overall complications, such as incisional hernia, may impact the rate of SSI. Future studies on suture bite technique are warranted.
Antimicrobial sutures
Statement 8: Triclosan-coated or impregnated antimicrobial sutures (TCS) are associated with a reduced risk of SSI compared with non-antimicrobial sutures.
TCS work to inhibit bacterial colonization, prevent formation of suture-prosthetic biofilm, and kill bacteria associated with SSI through the antimicrobial properties of triclosan.40 In one meta-analysis of RCTs, TCS were shown to significantly reduce SSI compared with non-antimicrobial sutures after CRS (RR = 0.67; 95% CI 0.48–0.94).41 This was consistent with the largest overall meta-analysis of 21 RCTs assessing colorectal, abdominal, and other surgery types (n = 6,462) showing that TCS significantly reduced SSIs (RR = 0.72; 95% CI 0.60–0.86).42 Similar results were found in a propensity score–matched analysis in colorectal cancer surgery (4.3% vs. 16.1%; p = 0.007).43 Another RCT in emergency laparotomy, through a midline approach, showed that TCS lowered incisional SSI compared with conventional sutures (7.6% compared with 23.4%; p = 0.009).44 Guidelines from NICE, WHO, CDC, ACS/SIS, and WSES either recommended their use or consideration of their use with evidence derived from several systematic reviews and meta-analyses.27-29,33,37 An economic analysis demonstrated that TCS used for incision closure after colorectal surgery were projected to significantly reduce median payer costs by at least $809 per patient compared with non-antimicrobial sutures.2
It is important that antimicrobial sutures are used for closure of all incisional wound layers to ensure a zone of microbial inhibition throughout the whole incision, mitigating the risk of wound contamination and SSI, particularly in obese patients. Antimicrobial polydioxanone sutures used for closure of the abdominal musculo-fascial layer are triclosan impregnated, not coated, and provide a prolonged antimicrobial effect which matches the slow resorption of polydioxanone. Triclosan-coated polyglactin sutures should be used for the subcutaneous layer and triclosan impregnated poliglecaprone for the subcuticular layer. Triclosan-coated sutures were shown in vivo and in vitro to be nontoxic, nonirritating, noncarcinogenic, and nonteratogenic.45 A 10-year clinical survey also showed no relationship between triclosan usage and antibiotic resistance.46
Continuous compared with interrupted sutures
Statement 9: There is insufficient evidence to make a recommendation on the use of continuous or interrupted sutures on the impact of postoperative wound complications (SSI, incisional hernia, or wound dehiscence).
Continuous and interrupted suturing techniques, for musculo-fascial closure after abdominal incisions, are each associated with benefits and drawbacks. Interrupted sutures are individually placed and, if one breaks, the wound theoretically remains intact. However, continuous suturing is considerably less time consuming and brings less foreign material into the wound, and there is little evidence to show it is inferior. A meta-analysis of four RCTs showed no differences in SSI (OR = 0.83; 95% CI 0.41–1.71) or incisional hernia (OR = 1.20; 95% CI 0.84–1.71) when comparing the continuous and interrupted suture techniques.39
Sutures compared with staples
Statement 10: There is insufficient evidence to recommend the use of staples or subcuticular sutures for skin closure to reduce SSIs.
It is uncertain whether the use of sutured or stapled skin closure impacts SSI risk after CRS. Three RCTs found that intracutaneous/subcuticular sutures were associated with similar SSI rates after elective gastrointestinal (GI) surgery, open laparotomy, and elective CRS.47-49 One RCT has reported that subcuticular sutures were associated with significantly lower SSIs compared with staples in a subgroup of patients undergoing lower GI surgery, but this difference was not observed in the overall SSI rate after elective open GI surgery.50 A propensity score analysis in elective colorectal resection also noted that subcuticular sutures were associated with significantly lower incisional SSI rates.51 However, the use of subcuticular sutures may be a proxy for surgical procedures in which there is less contamination, shorter incisions, and fewer unexpected findings, which are associated with lower SSI rates. Based on the available evidence, it is difficult to recommend one approach over another for CRS.
There are various other considerations, beyond an SSI risk, when choosing between sutures or staples. Staples and interrupted sutures are easy to remove when an SSI has developed to allow for free drainage and so may be advantageous in high-risk patients, although this was contested by some panel members. Alternatively, subcuticular absorbable sutures do not hinder local opening of an infected incision for drainage of localized pus. Furthermore, metal staples typically require removal at a later outpatient encounter, which may increase postoperative healthcare use and hospital visits. This may be particularly important in the era of COVID-19, where limiting hospital exposure is paramount. If cosmetic aspects are also considered, patients may prefer subcuticular sutures.
Topical skin adhesives
Statement 11: There is insufficient evidence to support the use of topical skin adhesives for superficial closure in reducing SSI.
Topical skin adhesives, also known as skin glues, are used in superficial skin closure to approximate wound edges of a surgical incision and have been shown to limit microbial penetration in a preclinical study.52 The potential benefits of topical skin adhesives in reduction of SSI has been noted in plastic and orthopedic surgery; however, further studies are required to understand the potential benefit in CRS.53,54
Negative pressure wound therapy
Statement 12: Closed-incision negative pressure wound therapy (NPWT) may be useful in reducing SSI risk when compared with no NPWT in open, high-risk, contaminated surgery.
Negative pressure wound therapy of a primarily closed incision involves use of a vacuum-sealed dressing applied to the wound surface to create a negative pressure. A large meta-analysis of RCTs found that NPWT was associated with significantly lower SSI compared with standard dressings on closed incisions after various types of surgery (RR = 0.61; 95% CI 0.49–0.76).55 The observed reduction remained significant among a subgroup of contaminated or clean/contaminated procedures (RR = 0.67; 95% CI 0.49–0.92). When analyzing a subgroup of studies in abdominal surgery, the summary result of 21 comparative observational studies also showed a significantly lower SSI risk (n = 2498; RR = 0.40; 95% CI 0.31–0.53), but the result was no longer significant for five randomized studies only (n = 792; RR = 0.56; 95% CI 0.30-1.03).55 Another meta-analysis of RCTs and observational studies similarly found NPWT to significantly reduce SSI compared with standard dressings for closed laparotomy incisions (OR = 0.25; 95% CI 0.12–0.52).56 The results remained significant in a subgroup study in CRS (OR = 0.16; 95% CI 0.07–0.36).56 Although indirectly applicable to CRS, recent studies may provide contradictory evidence regarding empiric NPWT use. A recent high-profile RCT of obese women undergoing caesarian section showed no difference in superficial SSI rates for patients receiving NPWT compared to traditional wound dressings.57
In general, guidelines from WHO, ACS/SIS, and WSES recommend the use of NPWT. The WHO notes its use may be beneficial to reduce the risk of SSI on closed incisions in high-risk wounds.28 The ACS/SIS and WSES have also noted potential benefits of SSI reduction, specifically over stapled skin in colorectal cases or in high-risk patients.33,37 The participants of the Delphi panel meeting also critically discussed the higher costs of the application. Further studies are warranted to better understand the cost-effectiveness of NPWT in CRS given the current cost of commercial NPWT products.
Advanced dressings
Statement 13: There is insufficient evidence to support the use of advanced dressings (for example silver dressings) as opposed to conventional dressings (such as standard gauze) to reduce the risk of SSI.
Dressings are commonly applied to closed wounds. For the sake of this Delphi, the panel considered dressings to be a class of flexible, removable, solid materials commonly used to cover the wounds immediately after closure. Dressings can contain traditional materials (e.g., gauze) or a variety of antimicrobial materials (e.g., silver) or other impregnations intended to promote healing. The panel defined dressings to be separate from Topical skin adhesives. Although dressings are commonly used either for pragmatic purposes (to manage wound exudate for example) or for a possible clinical benefit (such as reducing SSI), there is limited evidence that dressings give any benefit, with most available data being unrelated to CRS.58
One meta-analysis of RCTs found that ionized silver dressings, applied to a closed wound, were associated with fewer SSIs compared with placebo after CRS (RR = 0.55; 95% CI 0.35–0.85).59 A more recent RCT found that vitamin E and silicone dressings were associated with fewer SSIs when compared with conventional dressings applied to wounds closed with staples (3.4% vs. 17.2%; p = 0.013).60 An RCT of mupirocin dressings found no difference in SSI compared with standard gauze when applied to wounds closed with staples or sutures (2% vs. 3%; p = 0.56).61 Guidelines and recommendations are equally unclear. WHO guidelines suggest that advanced dressings should not be used, and the CDC noted uncertain tradeoffs of antimicrobial dressings.27,28 It was stated by the ACS that mupirocin topical antibiotic application can decrease SSI compared with a standard dressing.33 Use of antibiotics has to be traded off with the risk of promoting antibiotic resistance (see Statement 4.1).
There may be a potential benefit from the use of advanced dressings in reducing the risk of SSI, but current evidence is limited and heterogeneous. Future studies may be warranted to better understand the advantages of their use compared with standard dressings or not using any dressings.
Delayed incision closure
Statement 14: There is insufficient evidence to make a recommendation on the use of delayed incisional closure after open CRS.
Delayed incisional closure involves leaving the incisional skin layer open after a surgical procedure to be closed at a later date (delayed primary closure typically 3–5 days postoperatively; secondary closure is delayed longer than this after a dehisced or infected wound is clean after successful chronic wound management, often involving NPWT). This is electively chosen for incisions which are contaminated/dirty or at high risk for infection, allowing the open incision to be cleaned and observed to ensure infection is in control before closing the open skin layer of the incision. A systematic review and meta-analysis has reported mixed findings in management of contaminated abdominal incisions: delayed primary wound skin closure (undertaken approximately 3–5 days postoperatively) was associated with a lower SSI (RR = 0.64; 95% CI 0.51–0.79) when a fixed effect model was used, but a similar SSI was found (RR = 0.65; 95% CI 0.38–0.1.12) when a random effect model was used.62 The authors concluded that delayed primary skin closure may be the preferable choice after contaminated/dirty open abdominal procedures in patients with a high risk of infection and particularly in resource constrained environments.
Subcutaneous drains
Statement 15: There is insufficient evidence to support the role of subcutaneous drains in reducing SSI risk.
The purpose of placing a subcutaneous drain after CRS is to theoretically reduce the subcutaneous cavity of the incision and the risk of seroma.63 Evidence of their efficacy is mixed and limited to a small number of studies. An RCT involving reversal of elective ileostomy reported that subcutaneous drains were associated with similar SSI (14.0% vs. 17.0%; p = 0.68) compared with no drains.63 Two further RCTs, and a prospective observational study, all reported that subcutaneous drainage was associated with significantly lower superficial/incisional SSI when compared with no drains after CRS.64-66 Guidance from the WSES advocates that there is insufficient evidence on the use of subcutaneous drains for the prevention of SSI in high-risk patients.37
DISCUSSION
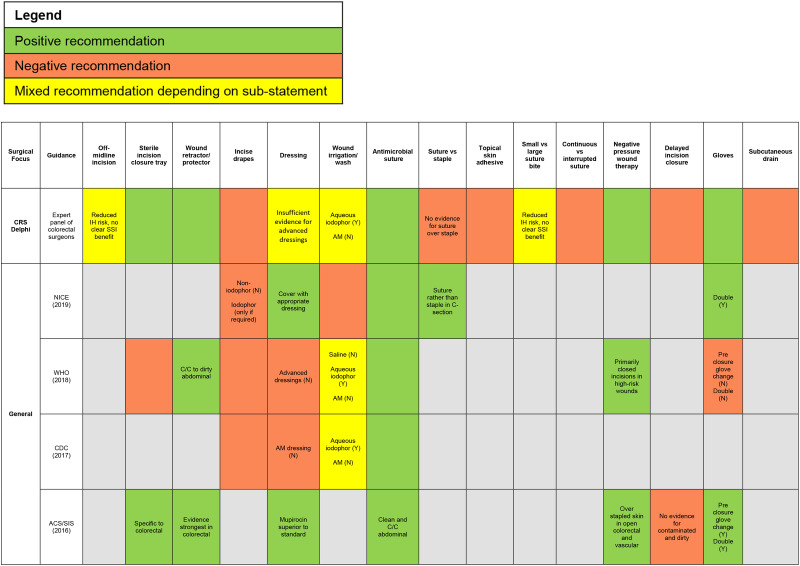
In conclusion, the high incidence of SSIs after CRS contributes to a large clinical and economic burden. The current study achieved consensus, using a modified Delphi method, to provide a more refined set of evidence-based SSI prevention recommendations involving intraoperative technical/surgical aspects that should be undertaken by surgical personnel during CRS (in addition to accepted SSI care bundles which are based on level 1A evidence from published systematic reviews and meta-analyses; Fig. 1).
Figure 1.
Comparison of recommendations between the colorectal Delphi and national/global guidelines. ACS, American College of Surgeons; AM, antimicrobial; C/C, clean-contaminated; CRS, colorectal surgery; IH, incisional hernia; N, No; NICE, National Institute of Health and Care Excellence; NPWT, negative pressure wound therapy; SIS, Surgical Infection Society; TSA, topical skin adhesive; Y, yes.
There are important limitations and considerations for this Delphi consensus. First, the Delphi panel only included representation from the US and Europe, and clinical practice, opinion, and economic feasibility may differ outside these regions. Secondly, organ space SSIs were not considered in this study given the inherent differences in risk factors and preventive strategies between superficial/deep (i.e., traditional “wound” infections) and organ space SSIs. Importantly, the recommendations included in this document are not exhaustive and thus need to be incorporated into a larger bundle of care (i.e., spanning the entire perioperative continuum) involving other equally important procedures such as adequate timing and redosing of prophylactic antibiotics, normothermia, and normoglycemia (advocated by local and national guidelines usually with a level 1A evidence base). Although identification of effective SSI reduction measures is important, implementation of properly chosen bundles is equally important and requires strategies that engage all perioperative stakeholders to produce effective results. Because some studies have shown that the effectiveness of SSI care bundles parallels compliance with elements of the bundle, audit of process measures and feedback schemes, as well as culture and communication, are paramount to reduction of SSI.
Finally, the recommendations contained in this Delphi consensus article were developed using the best available evidence and expert opinion, and not all recommendations may be suitable or applicable. When feasible, institutions should aim to incorporate the technical bundle components recommended in this study which are not already in place. Similarly, supplier contracting and rigid institutional formularies may create local logistical challenges to obtaining any recommended SSI product which are beyond the control of surgical personnel.
CONCLUSION
Using a modified Delphi method, consensus has been achieved on a tailored set of recommendations on technical/surgical aspects that should be considered by surgical personnel during CRS to reduce the risk of SSI, particularly in areas where the evidence base is controversial or lacking. Further research, through RCTs, systematic reviews, and meta-analysis, is warranted to add to the evidence advocated for the topics discussed in this article, or to assess new topics, based on emerging technologies, all of which may help prevent SSIs after CRS.
APPENDIX
Members of the Colorectal Delphi Facilitating Group: Joshua Dubé, MBIOTECH, EVERSANA Life Sciences Services, LLC, Burlington, ON, Canada; Nicole Ferko, MSC, Department of Surgery, University of Virginia Health System, Charlottesville, VA; Andrew Hogan, MSC, EVERSANA Life Sciences Services, LLC, Burlington, ON, Canada; Brian Po-Han Chen, SCM, and Walter Danker III, PhD, Ethicon, Inc, a Johnson & Johnson Company, Somerville, NJ.
Author Contributions
Study conception and design: Chen, Danker III, Dubé, Ferko, Hedrick, Hogan, Ruiz-Tovar
Acquisition of data: Boermeester, Bordeianou, Chang, Chen, Danker III, Dubé, Ferko, Gorgun, Hedrick, Hogan, Justinger, Lawson, Leaper, Mahmoud, Mantyh, McGee, Nfonsam, Rubio-Perez, Ruiz-Tovar, Wick
Analysis and interpretation of data: Boermeester, Bordeianou, Chang, Chen, Danker III, Dubé, Ferko, Gorgun, Hedrick, Hogan, Justinger, Lawson, Leaper, Mahmoud, Mantyh, McGee, Nfonsam, Rubio-Perez, Ruiz-Tovar, Wick
Drafting of manuscript: Chen, Danker III, Dubé, Ferko, Hedrick, Hogan, Ruiz-Tovar
Critical revision: Boermeester, Bordeianou, Chang, Gorgun, Hedrick, Justinger, Lawson, Leaper, Mahmoud, Mantyh, McGee, Nfonsam, Rubio-Perez, Ruiz-Tovar, Wick
Supplementary Material
Abbreviations and Acronyms
- ACS
- American College of Surgeons
- CRS
- colorectal surgery
- GI
- gastrointestinal
- NICE
- National Institute for Health and Care Excellence
- NPWT
- negative pressure wound therapy
- OR
- odds ratio
- PVP-I
- povidone-iodine
- RCT
- randomized controlled trial
- RR
- relative risk
- SC
- steering committee
- SIS-NA
- Surgical Infection Society of North America
- SSI
- surgical site infection
- TCS
- triclosan-coated/impregnated suture
- WSES
- World Society of Emergency Surgery
Disclosure Information: This study was supported by funding from Johnson & Johnson and Ethicon, Inc.
Disclosures outside the scope of this work: Dr Chang receives grant funding from Medicaroid and 11Health.
Disclaimer: The funding source had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Presented virtually at the American College of Surgeons 107th Annual Clinical Congress, Scientific Forum, October 2021.
Members of the Colorectal Delphi Facilitating Group who co-authored this article can be found in the Appendix.
Supplemental digital content for this article is available at http://links.lww.com/XCS/A9.
Contributor Information
Joshua Dubé, EVERSANA Life Sciences Services, LLC, Burlington, ON, Canada.
Nicole Ferko, Department of Surgery, University of Virginia Health System, Charlottesville, VA.
Andrew Hogan, EVERSANA Life Sciences Services, LLC, Burlington, ON, Canada.
Brian Po-Han Chen, Ethicon, Inc, a Johnson & Johnson Company, Somerville, NJ.
Walter Danker, III, Ethicon, Inc, a Johnson & Johnson Company, Somerville, NJ.
Collaborators: Joshua Dubé, Nicole Ferko, Andrew Hogan, Brian Po-Han Chen, and Walter Danker, III
REFERENCES
- 1.Zywot A, Lau CSM, Stephen Fletcher H, et al. Bundles prevent surgical site infections after colorectal surgery: Meta-analysis and systematic review. J Gastrointest Surg. 2017;21:1915–1930. [DOI] [PubMed] [Google Scholar]
- 2.Leaper DJ, Holy CE, Spencer M, et al. Assessment of the risk and economic burden of surgical site infection following colorectal surgery using a US Longitudinal Database: Is there a role for innovative antimicrobial wound closure technology to reduce the risk of infection? Dis Colon Rectum. 2020;63:1628–1638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Murray BW, Huerta S, Dineen S, et al. Surgical site infection in colorectal surgery: A review of the nonpharmacologic tools of prevention. J Am Coll Surg. 2010;211:812–822. [DOI] [PubMed] [Google Scholar]
- 4.Lawson EH, Hall BL, Ko CY. Risk factors for superficial vs deep/organ-space surgical site infections: Implications for quality improvement initiatives. JAMA Surg. 2013;148:849–858. [DOI] [PubMed] [Google Scholar]
- 5.Wang ZX, Jiang CP, Cao Y, et al. Systematic review and meta-analysis of triclosan-coated sutures for the prevention of surgical-site infection. Br J Surg. 2013;100:465–473. [DOI] [PubMed] [Google Scholar]
- 6.Badia JM, Casey AL, Petrosillo N, et al. Impact of surgical site infection on healthcare costs and patient outcomes: A systematic review in six European countries. J Hosp Infect. 2017;96:1–15. [DOI] [PubMed] [Google Scholar]
- 7.Liu Z, Dumville JC, Norman G, et al. Intraoperative interventions for preventing surgical site infection: An overview of Cochrane Reviews. Cochrane Database Syst Rev. 2018;2:CD012653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gorgun E, Rencuzogullari A, Ozben V, et al. An effective bundled approach reduces surgical site infections in a high-outlier colorectal unit. Dis Colon Rectum. 2018;61:89–98. [DOI] [PubMed] [Google Scholar]
- 9.Tanner J, Padley W, Assadian O, et al. Do surgical care bundles reduce the risk of surgical site infections in patients undergoing colorectal surgery? A systematic review and cohort meta-analysis of 8,515 patients. Surgery. 2015;158:66–77. [DOI] [PubMed] [Google Scholar]
- 10.Dalkey NC. The Delphi Method: An experimental study of group opinion. Rand Corp Public RM-58888-PR. Santa Monica: Rand Corp. 1969. [Google Scholar]
- 11.Sajid MS, Bhatti MI, Sains P, et al. Specimen retrieval approaches in patients undergoing laparoscopic colorectal resections: A literature-based review of published studies. Gastroenterol Rep (Oxf). 2014;2:251–261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lee L, Abou-Khalil M, Liberman S, et al. Incidence of incisional hernia in the specimen extraction site for laparoscopic colorectal surgery: Systematic review and meta-analysis. Surg Endosc. 2017;31:5083–5093. [DOI] [PubMed] [Google Scholar]
- 13.Sajid MS, Rathore MA, Sains P, et al. A systematic review of clinical effectiveness of wound edge protector devices in reducing surgical site infections in patients undergoing abdominal surgery. Updates Surg. 2017;69:21–28. [DOI] [PubMed] [Google Scholar]
- 14.Mihaljevic AL, Müller TC, Kehl V, et al. Wound edge protectors in open abdominal surgery to reduce surgical site infections: A systematic review and meta-analysis. PLoS One. 2015;10:e0121187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kobayashi H, Uetake H, Yasuno M, et al. Effectiveness of wound-edge protectors for preventing surgical site infections after open surgery for colorectal disease: A prospective cohort study with two parallel study groups. Dig Surg. 2019;36:83–88. [DOI] [PubMed] [Google Scholar]
- 16.Bressan AK, Aubin JM, Martel G, et al. Efficacy of a dual-ring wound protector for prevention of surgical site infections after pancreaticoduodenectomy in patients with intrabiliary stents: A randomized clinical trial. Ann Surg. 2018;268:35–40. [DOI] [PubMed] [Google Scholar]
- 17.Capolupo GT, Lauricella S, Mascianà G, et al. O-Ring protector in prevention of SSIs in laparoscopic colorectal surgery. JSLS. 2019;23:e2019.00048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chen CF, Tsai HL, Huang CW, et al. Impact of a dual-ring wound protector on outcome after elective surgery for colorectal cancer. J Surg Res. 2019;244:136–145. [DOI] [PubMed] [Google Scholar]
- 19.Itatsu K, Yokoyama Y, Sugawara G, et al. The benefits of a wound protector in preventing incisional surgical site infection in elective open digestive surgery: A large-scale cohort study. World J Surg. 2017;41:2715–2722. [DOI] [PubMed] [Google Scholar]
- 20.Edwards JP, Ho AL, Tee MC, et al. Wound protectors reduce surgical site infection: A meta-analysis of randomized controlled trials. Ann Surg. 2012;256:53–59. [DOI] [PubMed] [Google Scholar]
- 21.Kang SI, Oh HK, Kim MH, et al. Systematic review and meta-analysis of randomized controlled trials of the clinical effectiveness of impervious plastic wound protectors in reducing surgical site infections in patients undergoing abdominal surgery. Surgery. 2018;164:939–945. [DOI] [PubMed] [Google Scholar]
- 22.Zhang L, Elsolh B, Patel SV. Wound protectors in reducing surgical site infections in lower gastrointestinal surgery: An updated meta-analysis. Surg Endosc. 2018;32:1111–1122. [DOI] [PubMed] [Google Scholar]
- 23.Zhang MX, Sun YH, Xu Z, et al. Wound edge protector for prevention of surgical site infection in laparotomy: An updated systematic review and meta-analysis. ANZ J Surg. 2015;85:308–314. [DOI] [PubMed] [Google Scholar]
- 24.Alexander JW, Aerni S, Plettner JP. Development of a safe and effective one-minute preoperative skin preparation. Arch Surg. 1985;120:1357–1361. [DOI] [PubMed] [Google Scholar]
- 25.Yoshimura Y, Kubo S, Hirohashi K, et al. Plastic iodophor drape during liver surgery operative use of the iodophor-impregnated adhesive drape to prevent wound infection during high risk surgery. World J Surg. 2003;27:685–688. [DOI] [PubMed] [Google Scholar]
- 26.Al-Qahtani SM, Al-Amoudi HM, Al-Jehani S, et al. Post-appendectomy surgical site infection rate after using an antimicrobial film incise drape: A prospective study. Surg Infect (Larchmt). 2015;16:155–158. [DOI] [PubMed] [Google Scholar]
- 27.Berríos-Torres SI, Umscheid CA, Bratzler DW, et al.; Healthcare Infection Control Practices Advisory Committee. Centers for Disease Control and Prevention Guideline for the Prevention of Surgical Site Infection, 2017. JAMA Surg. 2017;152:784–791. [DOI] [PubMed] [Google Scholar]
- 28.WHO. Global guidelines for the prevention of surgical site infection. 2018. [PubMed]
- 29.NICE. Surgical site infections: Prevention and treatment. 2019.
- 30.Norman G, Atkinson RA, Smith TA, et al. Intracavity lavage and wound irrigation for prevention of surgical site infection. Cochrane Database Syst Rev. 2017;10:CD012234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.de Jonge SW, Boldingh QJJ, Solomkin JS, et al. Systematic review and meta-analysis of randomized controlled trials evaluating prophylactic intra-operative wound irrigation for the prevention of surgical site infections. Surg Infect (Larchmt). 2017;18:508–519. [DOI] [PubMed] [Google Scholar]
- 32.Mueller TC, Loos M, Haller B, et al. Intra-operative wound irrigation to reduce surgical site infections after abdominal surgery: A systematic review and meta-analysis. Langenbecks Arch Surg. 2015;400:167–181. [DOI] [PubMed] [Google Scholar]
- 33.Ban KA, Minei JP, Laronga C, et al. American College of Surgeons and Surgical Infection Society: Surgical site infection guidelines, 2016 update. J Am Coll Surg. 2017;224:59–74. [DOI] [PubMed] [Google Scholar]
- 34.Harnoss JC, Partecke LI, Heidecke CD, et al. Concentration of bacteria passing through puncture holes in surgical gloves. Am J Infect Control. 2010;38:154–158. [DOI] [PubMed] [Google Scholar]
- 35.Partecke LI, Goerdt AM, Langner I, et al. Incidence of microperforation for surgical gloves depends on duration of wear. Infect Control Hosp Epidemiol. 2009;30:409–414. [DOI] [PubMed] [Google Scholar]
- 36.Assadian O, Kramer A, Ouriel K, et al. Suppression of surgeons’ bacterial hand flora during surgical procedures with a new antimicrobial surgical glove. Surg Infect (Larchmt). 2014;15:43–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.De Simone B, Sartelli M, Coccolini F, et al. Intraoperative surgical site infection control and prevention: A position paper and future addendum to WSES intra-abdominal infections guidelines. World J Emerg Surg. 2020;15:10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.de Vries HS, Verhaak T, van Boxtel TH, et al. Implementation of the small bites closure of abdominal midline incisions in clinical practice is correlated with a reduction in surgical site infections. Hernia. 2020;24:839–843. [DOI] [PubMed] [Google Scholar]
- 39.Henriksen NA, Deerenberg EB, Venclauskas L, et al. Meta-analysis on materials and techniques for laparotomy closure: the MATCH Review. World J Surg. 2018;42:1666–1678. [DOI] [PubMed] [Google Scholar]
- 40.Russell AD. Similarities and differences in the responses of microorganisms to biocides. J Antimicrob Chemother. 2003;52:750–763. [DOI] [PubMed] [Google Scholar]
- 41.Uchino M, Mizuguchi T, Ohge H, et al.; SSI Prevention Guideline Committee of the Japan Society for Surgical Infection. The efficacy of antimicrobial-coated sutures for preventing incisional surgical site infections in digestive surgery: A systematic review and meta-analysis. J Gastrointest Surg. 2018;22:1832–1841. [DOI] [PubMed] [Google Scholar]
- 42.de Jonge SW, Atema JJ, Solomkin JS, et al. Meta-analysis and trial sequential analysis of triclosan-coated sutures for the prevention of surgical-site infection. Br J Surg. 2017;104:e118–e133. [DOI] [PubMed] [Google Scholar]
- 43.Yamashita K, Takeno S, Hoshino S, et al. Triclosan sutures for surgical site infection in colorectal cancer. J Surg Res. 2016;206:16–21. [DOI] [PubMed] [Google Scholar]
- 44.Ruiz-Tovar J, Llavero C, Jimenez-Fuertes M, et al. Incisional surgical site infection after abdominal fascial closure with triclosan-coated barbed suture vs triclosan-coated polydioxanone loop suture vs polydioxanone loop suture in emergent abdominal surgery: A randomized clinical trial. J Am Coll Surg. 2020;230:766–774. [DOI] [PubMed] [Google Scholar]
- 45.Barbolt TA. Chemistry and safety of triclosan, and its use as an antimicrobial coating on Coated VICRYL* Plus Antibacterial Suture (coated polyglactin 910 suture with triclosan). Surg Infect (Larchmt). 2002;3 Suppl 1:S45–S53. [DOI] [PubMed] [Google Scholar]
- 46.Russell AD. Whither triclosan? J Antimicrob Chemother. 2004;53:693–695. [DOI] [PubMed] [Google Scholar]
- 47.Imamura K, Adachi K, Sasaki R, et al. Randomized comparison of subcuticular sutures versus staples for skin closure after open abdominal surgery: A multicenter open-label randomized controlled trial. J Gastrointest Surg. 2016;20:2083–2092. [DOI] [PubMed] [Google Scholar]
- 48.Kobayashi S, Ito M, Yamamoto S, et al. Randomized clinical trial of skin closure by subcuticular suture or skin stapling after elective colorectal cancer surgery. Br J Surg. 2015;102:495–500. [DOI] [PubMed] [Google Scholar]
- 49.Maurer E, Reuss A, Maschuw K, et al. Superficial surgical site infection following the use of intracutaneous sutures versus staples. Dtsch Arztebl Int. 2019;116:365–371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Tsujinaka T, Yamamoto K, Fujita J, et al.; Clinical Study Group of Osaka University on Section of Risk Management. Subcuticular sutures versus staples for skin closure after open gastrointestinal surgery: A phase 3, multicentre, open-label, randomised controlled trial. Lancet. 2013;382:1105–1112. [DOI] [PubMed] [Google Scholar]
- 51.Yamaoka Y, Ikeda M, Ikenaga M, et al. Efficacy of skin closure with subcuticular sutures for preventing wound infection after resection of colorectal cancer: A propensity score-matched analysis. Langenbecks Arch Surg. 2015;400:961–966. [DOI] [PubMed] [Google Scholar]
- 52.Bhende S, Rothenburger S, Spangler DJ, et al. In vitro assessment of microbial barrier properties of Dermabond topical skin adhesive. Surg Infect (Larchmt). 2002;3:251–257. [DOI] [PubMed] [Google Scholar]
- 53.Ando M, Tamaki T, Yoshida M, et al. Surgical site infection in spinal surgery: A comparative study between 2-octyl-cyanoacrylate and staples for wound closure. Eur Spine J. 2014;23:854–862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Grimaldi L, Cuomo R, Brandi C, et al. Octyl-2-cyanoacrylate adhesive for skin closure: eight years experience. In Vivo. 2015;29:145–148. [PubMed] [Google Scholar]
- 55.Zwanenburg PR, Tol BT, Obdeijn MC, et al. Meta-analysis, meta-regression, and GRADE assessment of randomized and nonrandomized studies of incisional negative pressure wound therapy versus control dressings for the prevention of postoperative wound complications. Ann Surg. 2020;272:81–91. [DOI] [PubMed] [Google Scholar]
- 56.Sahebally SM, McKevitt K, Stephens I, et al. Negative pressure wound therapy for closed laparotomy incisions in general and colorectal surgery: A systematic review and meta-analysis. JAMA Surg. 2018;153:e183467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Tuuli MG, Liu J, Tita ATN, et al. Effect of prophylactic negative pressure wound therapy vs standard wound dressing on surgical-site infection in obese women after cesarean delivery: A randomized clinical trial. JAMA. 2020;324:1180–1189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Dumville JC, Gray TA, Walter CJ, et al. Dressings for the prevention of surgical site infection. Cochrane Database Syst Rev. 2016;12:CD003091. [DOI] [PubMed] [Google Scholar]
- 59.Nelson RL, Iqbal NM, Kravets A, et al. Topical antimicrobial prophylaxis in colorectal surgery for the prevention of surgical wound infection: A systematic review and meta-analysis. Tech Coloproctol. 2018;22:573–587. [DOI] [PubMed] [Google Scholar]
- 60.Ruiz-Tovar J, Llavero C, Perez-Lopez M, et al. Effects of the application of vitamin E and silicone dressings vs conventional dressings on incisional surgical site infection in elective laparoscopic colorectal surgery: A prospective randomized clinical trial. J Hosp Infect. 2019;102:262–266. [DOI] [PubMed] [Google Scholar]
- 61.Ahmad HF, Kallies KJ, Shapiro SB. The effect of mupirocin dressings on postoperative surgical site infections in elective colorectal surgery: A prospective, randomized controlled trial. Am J Surg. 2019;217:1083–1088. [DOI] [PubMed] [Google Scholar]
- 62.Tang S, Hu W, Hu L, et al. Primary versus delayed primary incision closure in contaminated abdominal surgery: A meta-analysis. J Surg Res. 2019;239:22–30. [DOI] [PubMed] [Google Scholar]
- 63.Lauscher JC, Schneider V, Lee LD, et al. Necessity of subcutaneous suction drains in ileostomy reversal (DRASTAR): A randomized, controlled bi-centered trial. Langenbecks Arch Surg. 2016;401:409–418. [DOI] [PubMed] [Google Scholar]
- 64.Fujii T, Tabe Y, Yajima R, et al. Effects of subcutaneous drain for the prevention of incisional SSI in high-risk patients undergoing colorectal surgery. Int J Colorectal Dis. 2011;26:1151–1155. [DOI] [PubMed] [Google Scholar]
- 65.Numata M, Godai T, Shirai J, et al. A prospective randomized controlled trial of subcutaneous passive drainage for the prevention of superficial surgical site infections in open and laparoscopic colorectal surgery. Int J Colorectal Dis. 2014;29:353–358. [DOI] [PubMed] [Google Scholar]
- 66.Watanabe J, Ota M, Kawamoto M, et al. A randomized controlled trial of subcutaneous closed-suction Blake drains for the prevention of incisional surgical site infection after colorectal surgery. Int J Colorectal Dis. 2017;32:391–398. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.