Abstract
Human corneal endothelium (HCE) is a single layer of hexagonal cells that lines the posterior surface of the cornea. It forms the barrier that separates the aqueous humor from the rest of the corneal layers (stroma and epithelium layer). This layer plays a fundamental role in maintaining the hydration and transparency of the cornea, which in turn ensures a clear vision. In vivo, human corneal endothelial cells (HCECs) are generally believed to be nonproliferating. In many cases, due to their nonproliferative nature, any damage to these cells can lead to further issues with Descemet’s membrane (DM), stroma and epithelium which may ultimately lead to hazy vision and blindness. Endothelial keratoplasties such as Descemet’s stripping automated endothelial keratoplasty (DSAEK) and Descemet’s membrane endothelial keratoplasty (DEK) are the standard surgeries routinely used to restore vision following endothelial failure. Basically, these two similar surgical techniques involve the replacement of the diseased endothelial layer in the center of the cornea by a healthy layer taken from a donor cornea. Globally, eye banks are facing an increased demand to provide corneas that have suitable features for transplantation. Consequently, it can be stated that there is a significant shortage of corneal grafting tissue; for every 70 corneas required, only 1 is available. Nowadays, eye banks face long waiting lists due to shortage of donors, seriously aggravated when compared with previous years, due to the global COVID-19 pandemic. Thus, there is an urgent need to find alternative and more sustainable sources for treating endothelial diseases, such as utilizing bioengineering to use of biomaterials as a remedy. The current review focuses on the use of biomaterials to repair the corneal endothelium. A range of biomaterials have been considered based on their promising results and outstanding features, including previous studies and their key findings in the context of each biomaterial.
Keywords: biomaterials, cornea, corneal transplantation, Descemet’s membrane (DM), human corneal endothelial cells (HCECs), scaffold, tissue engineering
Introduction
Human cornea and corneal endothelium
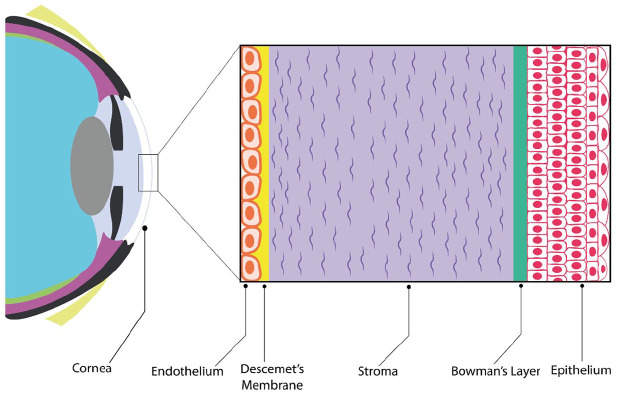
The transparent and multilayered structure at the front of the eye is the cornea. From the anterior to the posterior, the cornea comprises five layers shown in Figure 1: Epithelium, Bowman’s layer, stroma, Descemet’s membrane (DM), and endothelium. 1 In homeostasis, all these layers work together to achieve the cornea’s main function, which is vision, refracting the light into the crystalline lens through the pupil, which in turn focuses the light onto the retina. Corneal tissue’s transparency and hydration status are required as fundamental properties for obtaining a clear and normal vision.1–3
Figure 1.
Diagram of the human cornea location and structure.
Source: Author’s illustration.
This review specifically focuses on materials that can be used to support the corneal endothelial cell layer, which is derived from embryonic neural crest cells. 4 It acts as a barrier that separates the aqueous humor and other corneal layers. This layer is coating the cornea’s posterior surface as a thin monolayer of polygonal, mostly hexagonal cells. DM is the basement membrane of this layer; the endothelial cells are lying upon it3,5 with a firm connection to it, resulting in a mosaic pattern of dispersion. 6 The endothelial layer plays several essential roles in corneal homeostasis via controlling the corneal hydration using ionic pumps and leaky cell barriers. 7 The endothelium regulates nutrition and the metabolic pathways from and to the stroma; therefore, it controls the stromal hydration and consequently maintains the stromal thickness3,8,9 as well as the health of the stromal cells.
As the endothelial cells in human are arrested in the G1 phase of the cell cycle, they are characterized by limited mitotic ability in vivo.6,10–12 The mean corneal endothelial cell density decreases from 4000 cells/mm2 in newborns to 2300 cells/mm2 in the elderly. 10 An inverse relationship has been demonstrated between age and corneal endothelial cell density with density found to be reduced by approximately 0.6% annually in adults.14,15 The vision gradually decreases at a critical density of 500 cell/mm2, due to swelling which can ultimately lead to blindness. 13
Fuchs’ endothelial corneal dystrophy (FECD) is the commonest disorder of the corneal endothelium. Several hallmarks characterized and reported including an accelerated decline of endothelial function due to the low density of corneal endothelial cells (CECs). Those changes are accompanied by changes in DM, commonly known as guttae.16,17 It is worth mentioning that FECD is one of the most common reasons for endothelial keratoplasty (EK), as it accounts for 39% of all corneal transplants. 18 EKs such as Descemet’s stripping automated endothelial keratoplasty (DSAEK) and Descemet’s membrane endothelial keratoplasty (DMEK) are the preferred transplantation techniques used around the world for restoring vision when Fuchs’ endothelial dystrophy has advanced resulting in chronic corneal edema and consequent poor sight. Basically, in both techniques (i.e. DSAEK and DMEK), the diseased endothelium and its DM are removed and replaced by healthy donor endothelial cells from cadaveric donors that are in high demand globally.19–21
Worldwide, there is a long list of patients waiting for EK, and at the same time, there is a scarcity of donor corneas. Consequently, the continuing demand for native human corneas has driven research to find alternative solutions for transplantation. 18 It cannot be denied that a thorough understanding of corneal endothelial cell biology could greatly facilitate and contribute to innovation and further developments of cell culture. Due to the poor ability of CECs to regenerate in humans, many challenges have been faced to find the best cell culture conditions for CECs in vitro.8,22 Despite all this, researchers have overcome a substantial portion of the challenges23–25 which has led to rethinking additional therapy options. Thus, the pursuit for discovering viable alternatives to donor corneas is invaluable in order to increase supply of corneas for transplantation. A novel direction in tissue engineering is the development and harnessing of biomaterials and adapting them for use as cell scaffolds that would enhance the regenerative processes of diseased tissue; also, these biomaterials can be used to mimic native DM in tissue culture of endothelial cells as an alternative for overcoming the shortage of corneas for transplantation. 26
CEC culture
To date, a wide range of protocols and supplemented culture media has been developed to support HCEC survival and proliferation. Different percentages of serum and a variety group of supplements and mitogenic factors have been used such as basic fibroblast growth factor (bFGF), epidermal growth factor (EGF), and nerve growth factor (NGF).24,27–29 This point has been raised in the current review because it is a critical step in the tissue engineering field. It is known that culturing primary endothelial cells (in vitro) was the most common difficulty in terms of establishing a healthy and maintaining the culture long term. This is due to human corneal endothelial cells’ (HCECs) nature as they are nonregenerative in vivo. In addition, during the cell culture system, those cells could lose cellular morphology (hexagonal phenotype) to a mesenchymal-like transformation (fibroblastic phenotype) during long period of cell culture and hence their functionality deteriorates.
Nonetheless, gradually, primary endothelial cell culture obstacles have been overcome to a certain extent. For instance, healthy cultures of the extracted HCECs have been obtained using the dual media culture approach. 23 Basically, the dual media approach is based on switching between two different media (i.e. maintenance/stabilization M5 medium and the proliferative M4 medium). At the beginning, the isolated corneal endothelium was incubated in a serum-supplemented medium overnight to stabilize the cells and grant cells time to attach to the FNC-coated plates before cells start to expand in the culture. Preventing the mesenchymal-like transformation was one of the advantages of using this approach, in addition to the distinctive CECs’ morphology of the cultivated cells being preserved. Also, cells maintaining the expression of their functional genes and markers during their propagation in dual media system was another positive feature. 23
Several studies revealed the effective roles of Rho-associated kinase (ROCK) inhibitors on CECs in terms of proliferation, adhesion, and helping wound healing.24,25,30–32 Okumura and colleagues 25 reported that a selective ROCK inhibitor Y-27632 signaling has enhanced the adhesion of CECs to a substrate, improved cell proliferation, and suppressed apoptosis. Moreover, a study by a Japanese team was conducted to demonstrate the role of ROCK inhibitor Y-27632 for corneal endothelial wound healing. This study included in vitro and in vivo models (Japanese white rabbits), finding that ROCK inhibitor Y-27632 promotes corneal endothelial wound healing, both in vitro and in vivo. 30
Furthermore, in 2012, research carried out on rabbit and primate corneal endothelial dysfunction models has shown that using a cell-based therapy, combined with a ROCK inhibitor, may provide a promising approach in regenerative medicine for the treatment of corneal endothelial dysfunctions. The aforementioned study reached this result by transplantation of CECs in incorporation with ROCK inhibitor Y-27632, which successfully achieved the recovery of long-term corneal transparency. It must be noted that the normal hexagonal cells’ phenotype at a high cell density has been noticed after transplantation. Furthermore, corneal endothelium monolayer has expressed the basic functional markers (ZO-1 and Na+/K+-ATPase) of CECs in the presence of ROCK inhibitor Y-27632. 24
A pertinent investigation revealed that during the dual media approach, adding a selective ROCK inhibitor (Y-27632) into primary corneal endothelial culture had significantly increased the overall cell yield from 1.96- to 3.36-fold. 31 In a similar context, research performed by Okumura and colleagues investigated and explained the molecular mechanism by which ROCK inhibitors stimulated the proliferation of both monkey CECs and HCECs. The resulted data from this study demonstrated that ROCK inhibitors employ cyclin D (positive G1 regulator) and p27Kip1 (p27) (negative G1 regulator) via phosphatidylinositol 3-kinase (PI 3-kinase) signaling (cyclin D and p27 activities are necessary for G1/S progress) to regulate growth-synthesis (G1/S) phases of the cell cycle, ultimately promoting the proliferation of CECs. 32
A further study showed that the culture of HCECs can be established from older donor corneas over 65 years old when left to attach in the presence of a viscoelastic solution. Notably, the viscoelastic solution (Viscoat) in this study contained 3% sodium hyaluronate and 4% chondroitin sulfate. It is known that younger donor corneas have a better ability to proliferate and produce corneal endothelial culture in vitro than older donor corneas. However, obtaining younger corneas for research purposes is not easy because they are in high demand for cornea transplantation. Parekh and colleagues sought to find the best way to successfully exploit the older donor corneas. This research found that the viscous culture has forced the cells to attach speedily onto the coated base. In addition, the cell culture had reached 100% confluence after 9 days. Furthermore, passage 1 showed a significant increase of confluence (67%), compared with culture without Viscoat (19%). The most important point found in this study was that passaging HCECs with Viscoat maintained cellular morphology and the expression of vital proteins, such as cell-surface markers CD166, Na+/K+-ATPase, and ZO-1, as well as the proliferative marker Ki-67. 7
Tissue engineering of the corneal endothelium
It is estimated that around 39% of corneal transplants are a result of damage or dysfunction of endothelial cells. 18 As such, eye banks around the world are facing an increased demand to provide corneas that have suitable healthy endothelium for transplantation. At the same time, the eye banks suffer from an increasing shortage of donors. The statistics in 2016 worryingly indicated that there is a significant shortage of corneas for transplantation worldwide; that is, for every 70 corneas required, only 1 is available. 18 This serious shortage of corneas is currently even worse, as the COVID-19 pandemic has worsened the existing shortage of donor corneas and negatively impacted transplantations, especially after a recent report explained that SARS COV-2 has been found in the tears of positive patients. 33 Consequently, the United States and global eye bank associations have called to avoid using tissues from donors infected with or exposed to COVID-19. 34 In such circumstances, there is an urgent need to find alternative and more sustainable sources for treating endothelial diseases, such as applying bioengineering to use of biomaterials as a remedy. Bioengineering intensifies its efforts in this direction to achieve the main goal, which is alleviating the high demand for native corneas. So far, it can be said that research in this field has taken a great leap and an optimistic start, with encouraging results coming from using biomaterials as an alternative source for natural corneas. Yet, the search is still underway for finding the ideal and qualified biomaterial, by all standard criteria, which could be used for transplantation.
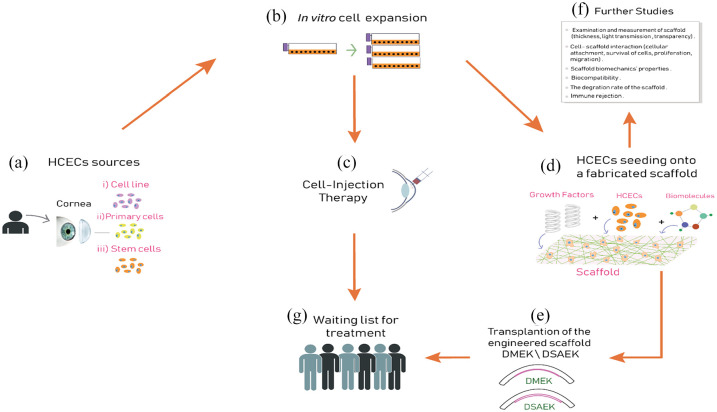
As pointed out, bioengineering research is aimed at alleviating the global shortage of donor cornea via using biomaterials. Based on the published literature, this review aims to summarize the current knowledge, the general manufacturing steps (Figure 2), current development direction, and planned goals on using fabricated substrates or cell injection therapy with CECs to treat corneal endothelial failure in humans. At the beginning, researchers obtained the CECs from three sources (Figure 2(a)): primary cells isolated from a donor cornea, 23 immortalized cell lines, 35 or stem cells. 36 Intriguingly, a study has found that genetically engineered (GE) pigs could provide a source of CECs for clinical transplantation due to the excellent proliferative capacity of cultured pig CECs, compared with HCEC cultures. This study mentioned that specific pathogen-free pigs would provide corneas to meet the current shortfalls of human cornea donors, and with the lowest risk of transferring disease to humans. 37
Figure 2.
Overview of the concept of corneal bioengineering and regeneration therapy of human corneal endothelial cells (HCECs), which ultimately is aimed at alleviating the global shortage in donor cornea tissues. General steps have been summarized. (a) The corneal endothelial cell sources: primary cells isolated from a donor cornea, immortalized cell lines, or stem cells. (b) In vitro cell expansion is the next step with the necessity to maintain healthy phenotype and morphology. The delivery system of the expanded corneal endothelial cells to the posterior corneal surface is achieved using (c) cell injection therapy or (d) using various types of biomaterials to fabricate corneal endothelial cell scaffold. (e) The implantation stage of the cultured scaffold into the anterior chamber through a similar DMEK/DSAEK transplantation. (f) Parallel studies on the scaffold are carried out to investigate whether the scaffold is working effectively. (g) Corneal bioengineering and regeneration therapy can alleviate the shortage of native corneas and help improve the quality of life for many patients who are waiting their turn for cornea tissue transplantation.
Source: Author’s illustration.
In vitro cell expansion is the next step (Figure 2(b)) with the necessity to maintain healthy phenotype and morphology. So far, numerous research studies have succeeded in tackling most of the challenges associated with endothelial cells’ culture by refining the types of media and supplements used in their in vitro culture. 38 Finally, the delivery system of the expanded CECs to the posterior corneal surface is achieved using one of three approaches proposed for this purpose; 8 these are as follows:
Fabrication of transplantable HCEC sheets grown/expanded on temperature-responsive dishes;39,40
Cell injection therapy where HCECs supplemented with a Rho-associated protein kinase inhibitor (ROCK inhibitor) are injected into the anterior chamber 41 (Figure 2(c));
Using corneal endothelial cell scaffolds, where endothelial cells are seeded onto fabricated substrates using various types of biomaterials (Figure 2(d)); this step is the main focus of this review.
Once the delivery system has been chosen, the implantation stage of the cultured scaffold into the anterior chamber is next. This is likely to be achieved through a delivery system similar to the currently used in DMEK/DSAEK transplantation, but replacing the native DM with a fabricated substrate instead. Importantly, parallel studies (before/during/after seeding) on the scaffold are carried out to investigate whether the scaffold is working effectively. These studies include, but are not limited to, examining cell–scaffold interaction, mechanical properties of the scaffold, immune rejection, biocompatibility, and biodegradability. Ultimately, it is expected that one cadaveric donor – who is the original source of endothelial cells (cells are multiplying in vitro for several passages) – can help and improve the quality of life of many patients (who will receive those cells and are waiting their turn for cornea tissue transplantation) which is the goal sought of corneal bioengineering and regeneration therapy.
Biomaterials
Currently, biomaterials occupy a top position in the bioengineering tissue field, due to their useful properties such as the comprehensive inclusion of necessary physical, chemical, and mechanical features which can guide cells and promote their functionality. The massive role that biomaterials have played in medicine is remarkable for many purposes, including diagnostic and therapeutic. From a healthcare perspective, the term biomaterial refers to materials which have unique properties making them suitable for direct contact with tissues of living organisms without causing negative immune reaction 42 and other associated properties. Basically, researchers are focusing on several important aspects of biomaterials for use in corneal repair: biocompatibility, transparency, suitable mechanical properties, appropriate biodegradability, toxicity, and being clinically compliant. 43 Pursuing sophisticated suitable scaffolds for HCECs is still underway, and some of these are discussed below.
Many studies have produced/developed scaffolds for HCECs using various sources of biomaterials, such as naturally derived biomaterials or synthetic origin ones. Reports have indicated that using biomaterials of natural origin as cell substrates can provide an extracellular matrix (ECM) that mimics the native environment for the cells; in addition, due to their biocompatibility and biodegradability, natural polymers are suitable in the biomedical field. 44 However, a group of synthetic polymers has also been successfully used. It is worth mentioning that synthetic polymers have excellent characteristics that can also qualify them to be used in the bioengineering field. They can be controlled to create scaffolds with customization of the desired properties. Furthermore, synthetic materials have excellent mechanical properties.43,45 Moreover, two or more synthetic polymers or natural polymers, or a mixture of both, can be blended. Such composite materials can be used to enhance and customize desirable properties such as mechanical strength or biocompatibility which qualify them to be suitable substrates for HCECs’ growth. 43 In the following sections, numerous biomaterials are described/highlighted according to their properties; encouraging outcomes and remarkable characteristics making them potentially suitable for clinical application as a scaffold for HCECs are also discussed.
Amniotic membrane
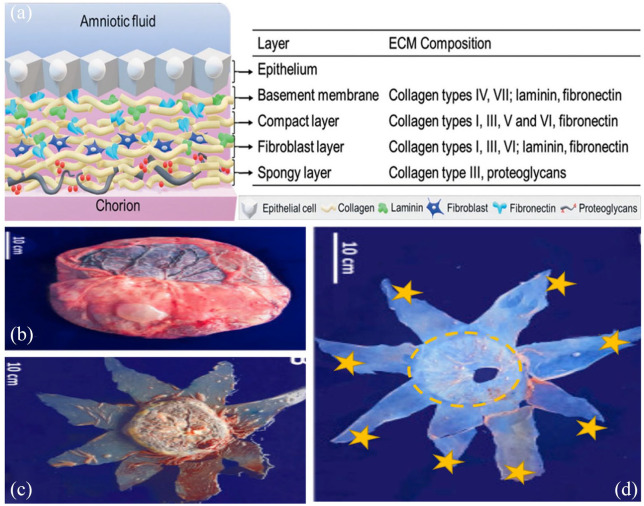
Amniotic membrane (AM) is the innermost layer of the placenta, along with a thick prominent basement membrane, a vascularized stroma, and a single epithelial layer46-48 (Figure 3(a)). Human amniotic membranes (HAMs) are routinely collected during cesarean section 49 due to its abundance and ease access. AM is well known for its wound healing, anti-angiogenic, and anti-inflammatory properties. 50 A study has shown that when obtaining a transparent AM for the purpose of ocular surface regeneration, having up to 85% level of transparency can be managed by considering two factors: the site of AM sample collection (distal or proximal to the placenta) and storage technique of AM. High transparency was found in the amniotic sac, distal to the placenta, and the best method for preserving it was by the freeze–dry approach as it has shown a higher level of transparency when compared with the freeze–thaw method of preservation. 51
Figure 3.
(a) The structure of human AM, and the composition of extracellular matrix for each layer (Source: Leal-Marin and colleagues 54 ). (b) The illustration displays placenta and fetal membranes. (c) The fetal bag (membranes) can be viewed on the placenta with a surgical incision noticeable. (d) An isolate and dispersed human AM is seen. The circle shows the placental human AM (proximal) area and the stars show the peripheral human AM (distal) areas (Source: Grémare and colleagues 52 ).
In addition, Tseng and colleagues 47 studied the effect of AM on tissue scarring in cornea transplantation, which may impact its transparency, and therefore the vision. The research showed that using AM has significantly reduced cornea scarring. Remarkably, Ishino and colleagues 49 suggested, in their research, that AM can be used as a carrier for CECs. Their results showed that when using AM to transplant cultivated HCECs, the cornea retained its transparency and thickness in a rabbit model. When examining the HCECs, they were similar in morphology and function in comparison with a normal cornea. The researchers found a density of more than 3000 cells/mm2 which is considered acceptable.
It can be stated that the result of using AM, as a scaffold of cells, has been encouraging. It is a naturally biocompatible and noncytotoxic material. 48 In addition, AM has anti-inflammatory and antimicrobial properties.46,50,52 Due to a lack of human leukocyte antigen (HLA) class II antigens, AM has a low immunogenicity as well. 52 Furthermore, a variety of soluble growth factors and cytokines are present within AM. 53 It also contains collagen and glycoproteins, which can enhance wound healing. 52 All these advantages could place AM at the top of the list of candidates to become a good corneal endothelial cell substrate/scaffold. However, several challenges of using AM in tissue engineering applications have been faced. AM has a low biomechanical consistency and its biodegradation rate is rapid. 54 Importantly, AM being a natural material, inter-donor variation (associated with attributes of the donor such as race, age, maternal health, and diet) and intra-donor variation remain a major concern. 55 In addition, there is an essential need to perform large amounts of expensive testing on AM donors to prevent the transmission of diseases such as hepatitis C, hepatitis B, human immunodeficiency virus (HIV), and syphilis. 54
Decellularized tissues
Numerous studies have reported that decellularized tissues are considered promising platforms and successful candidates in a variety of tissue engineering/regenerative medical applications (e.g. corneal transplantation), as well as in vitro modeling of diseased tissues. In the decellularization process, the tissue or organ undergoes serial steps of treatment.
There are several commonly used protocols such as utilizing enzymatic, chemical, and physical methods for decellularization. Most importantly, whatever the protocol, after decellularization, the ideal final product [decellularized extracellular matrix (dECM)] should not contain any cellular and antigenic molecules or nuclear materials belonging to the donor. This significantly reduces the risk of rejection. At the same time, this product must not adversely influence the tridimensional (3D) structure, organization, and mechanical integrity of the native ECM tissue.56,57 As a matter of fact, sodium dodecyl sulfate (SDS)58,59 and Triton X-10027 are widely utilized in the decellularization process of the cornea.
In that regard, there are reports that detail an optimized protocol to prepare an ultra-thin and decellularized Descemet stripping (DS) scaffold from porcine cornea. One such study used 0.1% SDS for decellularization, which was found to be most favorable for decellularizing the Descemet stripping endothelial (DSE) graft without structural deterioration. The authors also found that SDS at 1% deformed the stroma and damaged the DM. This study showed that incision of corneal endothelium, DM, and posterior stroma (the Descemet’s stripping endothelial keratoplasty (DSEK) graft) was more comfortable, in terms of handling, than the DMEK graft, which consists of the DM and endothelium without stroma. Moreover, they found that longer decellularization time led to lower transparency of grafts. 58 This finding was consistent with another study which found that using 0.5% or 1% SDS for 24 h led to an opaque and swollen decellularized porcine cornea matrix. 59
Another study found that using 2% Triton X-100 for decellularization of human corneal stroma produced a scaffold with acceptable mechanical properties and integral ECM proteins, which supported the biological functions of cells. 27 A further relevant study has shown that for decellularization of small intestine (SI) tissues, using Triton X-100 proved to efficiently remove all cells from the tissues and maintain collagen intensity and ECM components. Due to the resulting high transparency and optimum light transmittance appearing in decellularized tissue, when using Triton X-100, this study suggested that Triton X-100 could be safely used for efficient decellularization of SI tissues for corneal tissue engineering applications. 60
The use of detergents in the preparation of acellular corneal stroma can be harmful and any residue of these detergents must be washed out, as a study has claimed. 61 That is why, due to health and safety reasons, the researchers did not use detergent at all in their experiments, contrary to most other studies. Indeed, the aforementioned study has used a novel methodology to decellularize porcine cornea – without detergents – using high-hydrostatic-pressure (HHP) technology. The HHP process is performed in two steps: using high hydrostatic pressurization to disrupt cells, bacteria, and viruses in the tissue, as first step. Subsequently, a rinsing process is done to remove the residues of cellular components including lipids, proteins, and nucleic acids using a cultured medium, which is the second step. This study reported that no immune reaction occurred during the implantation time of acellular porcine corneas into rabbit cornea. In terms of transparency, the acellular porcine cornea maintained its transparency after implantation for at least 12 months. Acceptable results have been found as well, regarding biomechanical properties of the decellularized cornea, compared with the native cornea. Moreover, this study pointed to the possibility of HHP process for successfully reproducing decellularized corneas from rejected infected corneas (for transplantation), as HHP destroys and inactivates bacteria and viruses, not just the components of cells. 61 It can be said that this HHP procedure could be considered superior to other methods in terms of promising results as its ability for preventing the transmission of infectious diseases by eliminating bacteria and viruses is high. This is currently highly relevant as it could be beneficial for eliminating SARS-CoV-2 virus. Specifically, COVID-19 virus, as mentioned (in the introduction), can be found in the tears of infected patients, which hinders using their tissues for transplantation. As a consequence, the effectiveness of using HHP in eradicating pathogens from donor tissues must be investigated, because it can open a bright future for ensuring the sterilization of decellularized tissues.
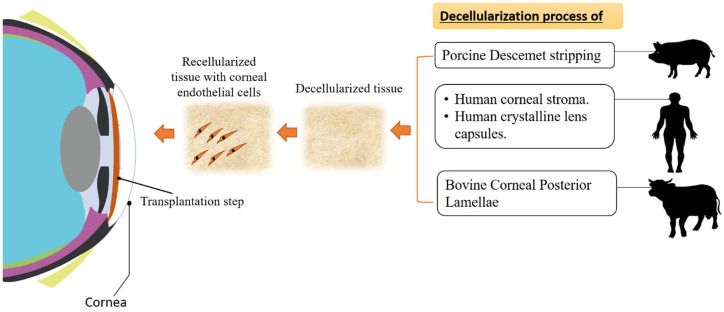
Usage of decellularized tissues provides benefits such as standardization in processing, in addition to consistency of the organ/tissue (critical), together offering an identical biological/physical microenvironment of the native tissue intended for cultured cells. Notably, there is a group of decellularized tissues which have been designed/investigated as an alternative substrate for CECs; for example, human corneal stroma, bovine corneal posterior lamellae, human crystalline lens capsule (LC), and porcine Descemet’s stripping (DS) scaffold are known (Figure 4).
Figure 4.
Diagram showing the general approach for using decellularized tissues for corneal endothelial cells’ bioengineering.
Source: Author’s illustration.
Decellularized human corneal stroma has been subjected to numerous in vitro experiments as a potential scaffold for CECs. 27 In one experiment, a thin layer of the decellularized corneal stroma at 120–200 µm thickness was engineered with 130 cells/mm2 of primary HCECs cultured upon its scaffold for 14 days. The results of this study have shown that decellularized corneal stroma may create a new source of high-quality corneal tissue for transplantation. The tight junction marker ZO-1, connexin-43, and the pump protein Na+/K+-ATPase were found, indicating that seeded cells can express markers of normal corneal endothelial cell functionality when grown on the human decellularized corneal stroma. This work demonstrates the fabricated scaffold has preserved its biomechanical and biochemical properties, similar to those of the normal DM, as cells were expanded and behaved similar to being in native tissue. 27
Decellularized bovine corneal posterior lamellae have been used as a potential scaffold for HCECs. 62 Regarding transparency, this decellularized tissue has recovered almost complete transparency after using 100% glycerol. Furthermore, the results after recellularization with human endothelial cells showed a relatively high cell density of HCECs. Cells also displayed acceptable growth and formed a viable monolayer of polygonal cells. Immunohistochemistry analysis was successfully used to demonstrate the differentiation and functionality of these endothelial cells by showing positive staining for AE5, collagen type VIII (differentiation markers), as well as positive result for ZO-1, CX-43, Na+/HCO3−, and Na+/K+-ATPase (functional markers) of CECs. 62
In another study, decellularized human crystalline LC was investigated as a potential scaffold for HCECs. 63 Due to LC being composed of collagen IV and laminin, it is granted features (i.e. elasticity, flexibility) that encouraged researchers to study it for the same purpose. 63 Culturing of primary HCECs on the decellularized human LC has shown good results in terms of cell adhesion, functional marker expression, and hexagonal morphology. The research has focused on two experimental aspects: First, comparing the physical properties of the LC scaffold with two other scaffolds (i.e. DM and HAM); second, the decellularized LC endothelial culture in terms of phenotype and morphology. The findings of this research showed that LC scaffold was thicker than DM; however, the LC scaffold has outperformed in the transparency experiments when compared with DM and HAM. Regarding the cell morphology, the cultured cells have shown hexagonal structure, such as those grown on plastic. Notably, the typical markers of healthy CECs, ZO-1 and Na+/K+-ATPase, only appeared when fibronectin was coated on LC. 63
In a recent study, an ultra-thin layer of decellularized DS scaffold from porcine cornea has been designed as a scaffold for hCEC-B4G12 cell line and induction of pluripotent stem cells (iPSCs). This ultra-thin layer has approximately 99 μm thickness compared with the ultra-thin DSEK and standard DSEK which are usually less than 100 and 200 μm, respectively. Findings of this study have shown that the dry DS graft at 40°C was able to maintain its transparency and ultra-thinness before cell seeding. The scaffold was shown to be a homogeneously reseeded graft after recellularization of cells on it, and the functional markers expression of CECs were confirmed by immunohistochemistry. 58
It can be said that most studies have shown that the results of using decellularized scaffolds for CECs are making good progress and moving toward an acceptable standard. Studies have mentioned that the success of these scaffolds and their distinct results are believed to be due to their ability to provide an appropriate microenvironment which mimics the native tissues. Ultimately, such a material can promote the functionality of the cells such as survival, migration, and differentiation, therefore improving ocular therapeutic effects after grafting.64–66
Silk fibroin
Silk is a natural fibrous protein. It is normally produced by certain mulberry silkworm (Bombyx mori) larvae to form cocoons at a specific stage of their life cycle. The protein fiber of silk is composed mainly of fibroin and minorly sericin. The ultrastructural arrangements of these proteins have been highlighted by Cao and Wang 67 where the central part of silk is fibroin (core). Fibroin is covered by sericin which in turn forms an outer glue-like coating. Practically, silk fibroin has been employed as a promising material for biomedical applications such as tissue engineering and regenerative medicine due to its unique properties. It has biocompatibility, mechanical robustness, controllable biodegradability, and hemostatic properties. In addition, it has been used for a long time as biomedical sutures during surgeries due to its noncytotoxicity and noninflammatory characteristics.67,68 Due to the presence of the glue-like family of sericins (incorporated in the silkworm cocoons), silk has low immunogenicity. 69
Silks have been reported to support proliferation and adhesion of a variety of cell types; specifically for corneal use, silk from mulberry Bombyx mori has been utilized with success. In addition, it has been found to be a suitable biomaterial to culture corneal limbal epithelial cells. 70 The silk membrane has been shown to be easily managed and also is a remarkably transparent film. The human limbal epithelial stem cells on the silk substrates have given acceptable results in terms of cell attachment and proliferation, compared with the plastic surface. 70 Another study has shown that HCECs do not show outstanding growth and attachment to the prepared silk fibroin (Bombyx mori) substrates without coating. 71 For this reason, the study used different coating materials for enhancing endothelial cell attachment and proliferation, including collagen IV, a chondroitin sulfate-laminin mixture, and a commercial FNC Coating Mix® (containing mix of fibronectin, collagen, and albumin). In addition, they compared the coated silk fibroin with uncoated silk membrane and the plastic surface. The results of this study found that collagen IV–coated fibroin yielded the highest mean cell counts when compared with the uncoated fibroin which yielded very low cell counts. Moreover, cells in collagen IV–coated fibroin have shown satisfactory attachment and the best proliferation (they became confluent within 2 weeks). 71
Non-mulberry silk was examined in a recent study 72 with corneal endothelial cell culture and compared with Bombyx mori silk. Aqueous silk fibroin was derived from three different sources: Philosamia ricini (PR), Antheraea assamensis, and Bombyx mori. Films with 15 μm thickness were manufactured to be used with CECs. The properties of all films were characterized and they have been compared with each other. All silk films derived from Philosamia ricini, Antheraea assamensis, and Bombyx mori had >90% transmittance of light with good mechanical properties and great tensile strength. As the reported transparency and refractive index values of silk films are close to that of human cornea, it makes a good candidate for cornea usage. CECs were found in vitro to attach and form a homogeneous and coherent monolayer on Philosamia ricini and Antheraea assamensis, in contrast with Bombyx mori. It is highly plausible that the reason behind this result is the existence of an arginine-glycine-aspartic acid (RGD)-peptide in the structure of the derived proteins of Philosamia ricini and Antheraea assamensis. It was also found that Philosamia ricini and Antheraea assamensis supported the growth of CECs and their functions without their phenotype changing, when compared with Bombyx mori. 72 It can be said that the results have clearly indicated that silk scaffolds are the best scaffold for endothelial cell culture. However, it is known that silk is used in many industrial and commercial fields such as clothes, parachutes, bicycle tires, furniture, and surgical sutures. Consequently, the question arises, whether the environment can cover all human needs of silk or whether this will massively increase the load on this natural resource.
Gelatin
Gelatin is one of the most common natural biomaterials. It is a heterogeneous water-soluble protein. Gelatin is obtained via the hydrolysis process of native collagen by breaking down the triple helix of collagen to such an extent that soluble collagen in warm water is produced (i.e. gelatin). 73 Around 95% of gelatin has mammalian origins such as porcine and bovine which raised some concerns about using it in the human medical field. Transmission of pathogenic vectors such as prions and the development of bovine spongiform encephalopathy is a potential risk. 73 To overcome this matter, there is an alternative direction for the production of gelatin from other sources such as fish bone, skin, and fin. Gelatin obtained from fish offers a solution in terms of reducing the risk of transmitting diseases, in addition to having good properties such as high viscosity, low melting temperature, and thermal stability. 74
Gelatin properties make it suitable for usage in tissue engineering. It is commercially available with low cost. Moreover, it has an excellent biocompatibility, controllable biodegradability, and low immunogenicity, nonharmful even upon enzymatic degradation. Notably, gelatin structure contains motifs such as arginine-glycine-aspartic acid (RGD) sequences 75 which is a biomimetic peptide, playing a vital role in encouraging cell adhesion to the matrix, preventing cell apoptosis, and hence enhancing the proliferation rate of cells and ultimately accelerating new tissue regeneration. 76 Gelatin hydrogel sheets have shown superior transparency, high elastic modulus, and a good permeability of albumin compared with those of atelocollagen sheets. The CECs have been seeded on these gelatin hydrogel sheets which have shown healthy functional activity where expression levels of ZO-1, Na+/K+-ATPase, and N-cadherin were noticed to be normal. In terms of the morphology, cells displayed a polygonal shape and achieved a continuous endothelial monolayer which is the normal structure of CECs. 77 However, gelatin lacking of sufficient mechanical strength needed for enduring surgical procedures remains an obstacle 78 which drives the researchers to create a bioactive hydrogel that is considered semi-synthetic, denoted as ‘gelatin methacrylate (GelMA)’. GelMA has been used in tissue bioengineering due to it retaining an excellent biocompatibility and bioactivity (e.g. enhance cell adhesion and proliferation), owing to the existence of cell-adhesive RGD motifs. 79 Furthermore, Rizwan and colleagues 78 created an improved scaffold for HCECs, called GelMA+ using sequential hybrid cross-linking using UV rays, with over eightfold increase in mechanical strength and it is more homogeneous as compared with regular GelMA. GelMA+ hydrogel has been investigated as a scaffold for CECs. 78 A simple nano-molding method was used to print patterns on the gelatin’s surface to help HCECs’ functionality and to work as a medium for transferring nutrients using polyterephthalate (PET) films as master stamps. It was also suggested that using 30% GelMA+ films could increase the ZO-1 and Na+/K+ ATPase expressions and make human corneal endothelial growth homogeneous. After the implant of GelMA+ in a rabbit’s cornea and examining it for over 4 months, it was found that it maintained its transparency, did not cause inflammation, and was slow in degrading, compared with GelMA. Thus, it was inferred that GelMA is a safe material and easy to customize its properties to potentially provide an excellent substrate for HCECs. 78
Collagen
Collagen is a primary structural protein for life and the most abundant in all connective tissues. Approximately, 25% of the total dry weight of mammals is collagen. Due to its essential role, it is considered as one of the most studied biomolecules of the ECM. Collagen lattice provides a substrate for cells and supports their functionality (e.g. proliferation, differentiation, cell attachment, migration). 80 A triple helix is a typical structure of most collagen proteins.81,82 Regarding the cornea, it derives its strength and elasticity from the presence of collagen protein (approximately, 71% of dry weight of stroma is collagen).83,84 Human corneal stroma contains types I, III, IV, 84 and V collagen. 85 Collagen type I is the predominant type in the stroma.84,85 As for the DM, studies have found that its elastic nature is due to the existence of various different types of collagen fibers. It is composed of collagen IV, V, VIII, XII, XVIII. 86
Nowadays, collagen is widely used in tissue engineering studies, due to its ability to create a 3D culture system which mimics real body tissues, unlike plastic dishes which lack the third dimension.87,88 In terms of preparation technique, it is both cheap and relatively simple as it does not require special skills. 89 Collagen lattice facilitates the cells to grow and behave in a manner somewhat similar to the native ECM in their morphology, differentiation, migration, adhesion, and proliferation. 90 However, owing to the natural cross-links’ dissociation during extraction procedures of native collagen, collagen can lack its mechanical stiffness in vivo as a result of exposure to the pressure from surrounding tissue or during handling in vitro as well. 91 In addition, there is concern for using collagen in biomedical applications, due to the probability of disease transmission and allergic reactions, as collagen is sourced from animal tissues.90,92 Despite lacking substantial evidence, 93 there are several studies that have expressed their concern in this regard and they aim at generating recombinant collagen from other sources such as transgenic silkworms 94 and yeast Pichia pastoris, 95 to avoid this drawback.
Research has been undertaken to understand the behavior of corneal cells using type I collagen as a physical model substrate.96,97 Regarding HCECs, several studies using type I collagen as a substrate have confirmed its suitability for endothelial cells. One of these studies has implemented an evaluation using type I collagen as a carrier for HCECs. This study revealed that the transplanted collagen sheet with cultured HCECs, grafted in the rabbit model, can maintain the proper function of the cells, as a pump for controlling corneal dehydration. 96 The researchers also noticed that after transplantation, fibroblast-like cells appeared in the stroma, attached to the collagen disk; collagen and fibronectin acted as a stimulant for these cells, but not endothelial cells, because fibroblast-like cells also appeared in the control sample (collagen disk without endothelial cells). Focusing on immunologic rejection, this study 96 found no apparent inflammatory reaction.
Controlling and improving the mechanical properties of collagen gel have been the focus of multiple studies. In this regard, using the plastic compression method 98 has improved the nanostructure surface and topography of collagenous matrix. In the plastic compression method, fluid from collagen gel (high water-containing) is rapidly removed by applying a compressive mechanical load or capillary suction. The produced matrix after compression has excellent functional mechanical properties (i.e. strength and compliance) with high cell viability. In addition, compressed collagen gel is denser and more controllable, compared with the uncompressed 98 (Figure 5).
Figure 5.
Analysis by scanning electron microscopy of collagen gel shows the difference between (a) uncompressed gel and (b) compressed gel. Collagen fibers on the surface of the uncompressed gel are displayed in a disorganized and very loosely arranged manner, whereas the collagen fibers in the compressed gel are more densely packed and homogeneous.
Source: Mi and colleagues. 128
HCECs have shown acceptable culture results with the compressed collagen gel. 97 A study by Levi and colleagues 97 has shown that HCECs cultured on compressed collagen retain their endothelial cell characteristics in terms of morphology and expression of tight junction protein ZO-1 and pump protein Na+/K+ ATPase which are functional markers. This study has mentioned that the mechanical strength of these compressed gels using a process termed Real Architecture For 3D Tissues (RAFT) is improved and sufficient to withstand the manipulation without rupture. Interestingly, a study by Jones and colleagues 99 revealed that similarly compressed collagen gels played a crucial role in driving limbal epithelial stem cell growth and phenotype. In this case, the corneal epithelial cells were shown to have an increased capacity for growth on compressed collagen gels (stiff) compared with uncompressed collagen gels (less stiff). This indicates that there is a positive relationship between ‘mechanical properties’ and ‘levels of cell differentiation’. This may also have relevance for CECs.
Chitosan
Chitosan (CHM) is an amino polysaccharide, produced from the deacetylation of chitin obtained from crustaceans and insects. 100 CHM weight ranges between 300 and over 1000 kDa. 128 It has diverse biological activities and therapeutic applications due to its properties, especially being biodegradable and nontoxic. CHM is considered as having antibacterial activity, fungicidal effects, antioxidant activity, and nutritional supplements. It has been established as a safe polymer for use in many experiments conducted on animals and limited clinical studies on humans. 100 It breaks down slowly by certain human enzymes, such as lysozyme, to amino sugars which are nontoxic, totally absorbed in the human body.102,103
CHM is a naturally derived bioactive polymer. The chemical and mechanical properties of CHM are affected by its manufacturing conditions, which affect the amount of deacetylation. 128 Cell adhesion and proliferation might be affected by degree of deacetylation as cells prefer low degree of deacetylation which support cell adhesion and growth. 102
CHM’s positive polarity allows for easier electrostatic reactions with compounds that have negative polarity (e.g. growth factors, cytokines, and nucleic acids), thus forming new compounds with these molecules and therefore changing the cell behavior during regeneration. 128
Recently, it has shown to be used in tissue engineering as a scaffold. Jorge and colleagues 104 found that using chitosan as a scaffold and then inducing endothelial cell damage caused inflammation in the cornea after 1 week. This affected the optical media transparency, coming to the conclusion that it is not biocompatible and not suitable for clinical application. When examining the cells under a microscope, it was found that the wound did not heal and there was a rupture with purulence on the cornea, iris, and the anterior chamber of the eye. In addition, the inflammation also caused the tissue to lose its normal structure, as well as thickening the cornea. This study showed that the CHM strengthens some immune cells’ functionality related to inflammatory responses such as macrophages, polymorphonuclear (PMN) leukocytes, and fibroblasts. In addition, the result of this study was consistent with another study which has found that chitosan activates immunocytes. 105 However, another study assessed the biocompatibility of chitosan implantation in a mammalian model in terms of histocompatibility and immune reactivity. This research demonstrated that chitosan has a high degree of biocompatibility in this animal model. The researchers attributed this inconsistency to the degree of CHM acetylation, where it was found that decreasing the acetylation in CHM lessens the possibility of causing inflammation. 106
Markedly, CHM lacks the mechanical capability to endure surgical procedures. Therefore, new studies and research were conducted to investigate CHM blend with different substances. Improving its chemical and mechanical properties has been proposed and conducted through combining CHM with other substances. Polycaprolactone (PCL), genipin, collagen, and hydroxyethyl sulfate–gelatin were blended with CHM for this reason.104,107–111
Continuing on the necessity to blend materials, research by Liang and colleagues 111 demonstrated that a membrane made of hydroxyethyl chitosan, gelatin, and chondroitin sulfate (HECTS) was suitable for rabbit CECs to attach and grow in culture. 111 This research also found that the characteristics of the membrane blend were relatively similar to the natural human cornea in terms of glucose permeability, transparency, and equilibrium water content and other small molecules. During the experiment, a monolayer of cultured rabbit CECs was formed on the membrane and the cells maintained normal morphology. The growth rate of these cells on the membrane was faster than the control sample (on plastic). In addition, degradability and biocompatibility have been assessed in a rat model and have shown acceptable results. Thus, the results from this study have pointed to the possibility that a HECTS-blended membrane can potentially be used as a scaffold for corneal endothelial cell growth and subsequent transplantation with the necessity of further studies in the future to improve this scaffold. 111
Having shown acceptable results when blending CHM, a subsequent investigation by Wang and colleagues 107 analyzed the blending of chitosan and PCL to form a scaffold in bovine corneal endothelial cell (BCEC) culture. The transparency of this scaffold was shown to be excellent at PCL25, PCL50, and PCL75, but not at PCL100 (Figure 6). The results of this study demonstrated that the content of PCL increased in the blends and BCECs showed greater degrees of adhesion and proliferation. Concerning the phenotype and expression of the differentiation markers (N-cadherin and tight junction marker ZO-1), the cells showed good results and they were confluent, maintaining their hexagonal morphology at high concentration of PCL50 and PCL75. 107 Wang and colleagues 112 found that chitosan/PCL blends have excellent topography (roughness) compared with pure chitosan membrane. BCECs have shown hexagonal morphology, an acceptable proliferation, and well-localized ZO-1 and Na+/K+ ATPase expression, indicating good functionality of cells. In addition, Wang and colleagues’ study has demonstrated that high amount of collagen type IV and reduced TGF-β2 expression in ECM on PCL25 membrane was found compared with tissue culture polystyrene (TCPS) substrate. This result was explained as the presence of collagen IV mimics the natural structure of DM and collagen IV as an essential component for maintaining CECs morphology. 112 However, no data were given for human cells which are significantly more challenging to grow in vitro somewhat weakening the impact of the study.
Figure 6.
Photos show the high transparency of ‘pure chitosan’ and largely high transparent ‘blended chitosan with polycaprolactone’ (PCL25, PLC50, PCL75, but not PCL100), comparing with tissue culture polystyrene (TCPS) plates.
Source: Wang and colleagues. 107
Agarose
Agarose is a nature-derived biopolymer. It is a polysaccharide extracted from algae. Agarose has been utilized in numerous biomedical applications due to its significant advantages over other biomaterials. First, it has a porous structure with an open porous network to facilitate the nutrients, oxygen permeation, and waste exchange. Second, it has strong mechanical properties and ability to endure surgical manipulation due to its elasticity (Figure 7(a)). Third, it displays the required biodegradability and biocompatibility. Fourth, agarose has a high ability to mimic human tissue where it provides an appropriate microenvironment for cellular activity such as the cell migration and proliferation. 113 However, the native agarose has poor cell attachment capacity due to lacking the appropriate chemical groups for cell adhesion. 114 Interestingly, agarose has a self-gelling behavior relating to oxygen and hydrogen existing in its structure. 115
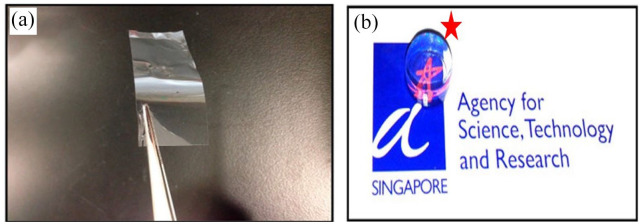
Figure 7.
Figure (a) shows agarose membrane is transparent and robust enough to be handled with a pair of forceps and (b) Despite the thickness was being ~8 mm, the conjugated agarose gel with fish-derived gelatin (AG) showed excellent transparency (the red star indicates the agarose gel).
Source: Seow and colleagues. 114
Investigating agarose, Seow and colleagues 114 studied the effects of modifying its structure by adding lysine (AK), poly lysine (AP), fish-derived gelatin (AG), and GRGD (AR) to improve its properties such as cell adhesion for usage as a carrier for rabbit CECs. The results demonstrated that cells successfully attached to agarose combined with AG. In addition, CECs on this material survived for more than 4 weeks in culture. Researchers observed that cells expressed CD166, Na+/K+ ATPase, and ZO-1, indicating a degree of functional activity. Furthermore, the transparent (AG) membranes allowed passage for more than 96% of visible light 114 (Figure 7(b)).
Poly-ε-lysine
Poly-ε-lysine (pεK) is a natural homo-poly-amino acid. It is safe and biocompatible for humans and has been used as a preservative in the food industry for many years. 116 The pεK hydrogel was one of the biopolymers which was studied due to its ability to mimic a natural ECM. In this context, research was conducted using HeLa cells (the first continuous cancer cell line) 117 to evaluate the feasibility of hydrogels containing pεK. A cell-binding RGD peptide was incorporated into the pεK hydrogel network to enhance cell adhesion and biomechanical material properties, a common approach used to improve cell spreading within relatively inert polymers. This study highlighted HG-PεK with molar ratio of 1:2 (mixing stock solutions of four-arm PEG-aldehydes with pεK) and formulated with low RGD concentrations provided enough stability and structural support, to afford cellular adhesion and proliferation. 116
In the corneal context, in vitro research has shown that pεK hydrogel has excellent cytocompatibility that qualifies it for use as antimicrobial bandage contact lenses. 118 As a hydrogel pεK is characterized as naturally antimicrobial. In addition, pεK hydrogel has useful features such as transparency, a porous matrix, sufficient mechanical properties for handling (Figure 8), being hydrophilic, and containing high water content. Interestingly, this hydrogel did not impede a functioning human corneal epithelial cell monolayer’s reformation while being tested as a lens. These features collectively can support the conclusion that using this hydrogel is possibly the best conceivable substitution for corneal ulcer treatment instead of eye drops. 118
Figure 8.
(a) Poly-ε-lysine hydrogel forms a thin transparent film. (b) Microporous structure of the poly-ε-lysine hydrogel under atomic force microscope (AFM). (c) Poly-ε-lysine hydrogels can be manipulated easily using forceps.
Source: Kennedy and colleagues. 119
In a study conducted by Kennedy and colleagues, 120 pεK hydrogels were used with 100–130 μm thickness and a diameter of 8 mm as substrates for CECs which were cultured at 1800 cells/mm², approximately 100,000 cells/(pεK) hydrogel. This study has shown that it is possible to control the mechanical and functional properties of pεK, which is a synthetic hydrogel, to make it suitable for creating a stable endothelial cell monolayer. The mechanical properties and transparency can also be modified depending on the formulation, percentage of cross-linking, and type of cross-linker. In the work done by Kennedy and colleagues, 120 the formulation has been optimized to provide high transparency, mechanical properties to allow loading and implantation from a clinical graft delivery device, high adhesion, and expansion of primary cells on the surface. This synthetic peptide shows great potential to be a suitable scaffold for creating a tissue-engineered corneal endothelial graft.
Discussion
Tissue-engineered corneal endothelial grafts are being developed in order to alleviate pressure on the increasing demand for transplant tissue due to a worldwide donor cornea shortage. It can be said that attempts are still underway and sophisticated materials and methods are being used toward finding the ideal scaffold in this area. However, many obstacles have been faced in this field; the most common difficulty was culturing primary endothelial cells (in vitro). As mentioned, previous studies have found two main obstacles which hinder them to establish a healthy long-term culture. First obstacle is proliferation ability of the CECs is very low. A mesenchymal-like transformation is the second obstacle, which leads to loss of the healthy cell morphology and hence their functionality will deteriorate. Therefore, a major focus has been placed to address these issues in order to obtain successful culture. Recently, the dual media culture approach, 23 adding ROCK inhibitors to the cell culture,24,31 and using Viscoat to establish HCECs culture from older donor corneas 7 have achieved good results in this regard.
Another difficulty, less common than obtaining primary endothelial cells (in vitro), is preventing corneal allograft rejection in patients who underwent penetrating keratoplasty (PKP). Interestingly, HLA matching (HLA-A, HLA-B, and HLA-DR) was studied to determine the effectiveness of HLA antigens in that regard. This study has confirmed that there is a significant correlation between the number of HLA (HLA-A, HLA-B, and HLA-DR) mismatches and the rate of allograft rejections. When two or more alleles of HLA-A, HLA-B, or HLA-DR are matched, the risk of immunological rejection is markedly reduced. Hence, clinical usage of cultured human endothelial cells causes HLA matching to become feasible in order to assist in reducing the risk pertaining to allograft rejection. 119
After this promising success, contemplating the delivery system of endothelial cells into the anterior chamber was the next step. Remarkably, in recent years, studies in corneal bioengineering and regeneration therapy have mainly aimed to alleviate the global deficit in eye banks, reducing the long waiting list of patients who are waiting for corneal transplantation. In that regard, exploiting biomaterials as an alternative to native tissues has emerged as a promising trend. Interestingly, it is expected that cells extracted from a single corneal donor can be a source for several endothelial cell cultures in the lab. Consequently, this can provide a considerable amount of eligible cells for seeding on many ideal scaffolds. These can be readily transplanted in many patients without the need to wait long. Ultimately, it will help improve the quality of human life.
The current review has discussed a group of biomaterials that have been most frequently used in this regard. A brief description of the studied biomaterials which are used for corneal endothelial cell bioengineering, including their general advantages, some limitations, mechanical and transparency properties, types of CECs, and in vivo studies of CECs with those scaffolds, is shown in Table 1. To date, a number of biomaterial scaffolds have been trialed for use as potential future alternatives to EK; however, it can be said that finding the optimal and medically approved scaffold for endothelial cells is still in progress, as of now.
Table 1.
Summary of the advantages and limitations of each biomaterial which has been used as scaffold-based tissue engineering for the corneal endothelium.
| Biomaterial | Advantages | Limitations | Mechanical properties | Transparency | Used corneal endothelial cell types | In vivo study of corneal endothelial cells in animal models |
|---|---|---|---|---|---|---|
| AM | Naturally biocompatible material
48
Inexpensive 48 Inert, noncytotoxic Antifibrosis activity46,50 Low immunogenicity due to a lack of HLA class II antigens 52 Owing to production of anti-inflammatory proteins (e.g. Interleukin-1 receptor antagonist (IL-1 RA) and IL-10, β-defensins), it has anti-inflammatory and antimicrobial activity46,50,52 Anti-angiogenic 49 Containing a variety of soluble growth factors and cytokines 53 It contains collagen and glycoproteins, which can enhance wound healing 52 |
Low biomechanical consistency
54
Rapid biodegradation 54 Requires large amount of expensive testing from donors to prevent the transmission of diseases 54 Inter-donor and intra-donor variations in the membrane 55 The potential for epidemic infections (e.g. human immunodeficiency virus, hepatitis B and C) 55 |
The fluctuation of mechanical stability such as elasticity, stiffness, and tensile strength is due to the composition of the placenta,
54
the preparatory method of AM,
53
and preservation procedures46,54
There are two mechanically distinct areas which are the placental area and peripheral area. The placental human amniotic membrane provides isotropic mechanical properties 52 (Figure 3) |
AM was found to be up to 85% as transparent as the human cornea (according to its original location from within the fetal sac and its method of preservation, as either can influence corneal transparency) 51 | HCECs
49
Cat corneal endothelial cells 122 |
Rabbit model
49
Cat model122,123 |
| Decellularized tissues: (a) decellularized corneal stroma | Generally, natural material, the best model of a given tissue’s native microenvironment (3D) in terms of providing growth/behavioral cues 123 | Limitation of mechanical properties Batch-to-batch variability (the differences between donor gender, age, genetics, and exposure to medicine) Difficulty in decellularization assessment, in terms of comprehensively assessing the protein content in each matrix |
||||
| Decellularized corneal stroma provides a unique ECM organization that supports cell functions, including cell adhesion, proliferation, and cell–cell interactions through offering appropriate mechanical properties and essential biological properties 27 | The decellularized human corneal stroma has provided acceptable mechanical properties in terms of tensile strength and Young’s modulus which were similar to the mechanical properties of the native corneal stroma 27 | Decellularized stroma has acceptable transparency and it maintained a 90% transparency after decellularization 124 | HCECs27,124 | Rabbits 124 | ||
| (b) Decellularized human crystalline lens capsules 63 | Biodegradation results of the LC was acceptable (after 13 hrs) compared with DM (after 17 h) 63 | Limitation in permeability test with corneal endothelial cultures in vivo model 63 | None | LC has excellent transparency even after the decellularization process 63 | Primary HCEnCs 63 | None |
| (c) Decellularized DS from porcine cornea 58 | Obtaining ultra-thin DS graft (approximately 99 μm in the dry graft) compared with ultra-thin and standard DSEK (which are less than 100 and 200 μm, respectively). In vivo biocompatibility 58 |
Limitation in permeability test with corneal endothelial cultures in vivo model | None | Compared with the control, the dry DS graft displayed high transparency (~92%) after cell seeding 58 | hCEC-B4G12 cell line
58
Induced pluripotent stem cell–derived corneal endothelial cells 58 |
Porcine eye 58 |
| (d) Decellularized bovine corneal posterior lamellae 62 | Inexpensive
62
Biocompatibility 62 |
Xenogeneic substrate need in vivo studies to test permeability with corneal endothelial 62 | None | Semitransparent to cloudy after decellularization process. Using 100% glycerol has changed the transparency into almost complete transparency 62 | Human corneal endothelial cells 62 | None |
| Silk fibroin | Natural fibrous protein
67
Biocompatibility 125 Controllable biodegradability69,125 Noncytotoxicity Noninflammatory characteristics 67 Hemostatic properties 67 Optimal water and oxygen permeability Thermal stability 126 Transparency 126 |
Inadequate elasticity Fragile and difficult during manipulation 126 High cost 125 |
Has excellent mechanical robustness 72 ,125 | Excellent transparency71,72,126 | B4G12 endothelial cell line
71
Primary human corneal endothelial cells71,72,127 HCEnC-21 T-cell line 72 Rabbit corneal endothelial cells 127 |
Rabbit model 127 |
| Gelatin | Natural material Biocompatible and noncytotoxic material and low Immunogenicity compared with the native collagen Approved by the Food and Drug Administration 74 Controllable biodegradability Containing RGD motifs 75 which enhance cell adhesion and proliferation |
Lack of mechanical strength
78
Risk of transmission of pathogenic vectors such as prions and the development of bovine spongiform encephalopathy 74 Production of gelatin methacrylate (GelMA+) is a relatively long process 78 Need to prove the results with corneal endothelial cells by in vivo study 78 |
GelMA+ has over eightfold increase in mechanical strength compared with regular GelMA 78 | Excellent transparency | Human corneal endothelial cells 78 | None |
| Collagen | Multisources protein Low immunogenicity Porous structure Permeability Biocompatibility Controllable biodegradability 90 Relatively cheap 89 |
Risk of disease transmission and allergic reactions due to its source from animal tissues 90 | Lack of mechanical stiffness 91 | Human corneal endothelial cells 96 | Rabbit 96 | |
| (a) Plastic compressed type I collagen gel | The compressed gels have similarity in structure and transparency with the normal corneal stroma
128
Customizable dimensions Ease of manipulation 128 Can rapidly produce dense, mechanically strong collagen scaffolds 128 |
Improved mechanical prosperities 128 | Transparency 128 | |||
| (b) Compressed collagen using RAFT method 97 | There is reduced variability among batches during production Quick production Easy production method 97 Due to material having tunable characteristics, it facilitates the user to generate constructs with different concentration of collagen or thickness (reliant on necessity) 97 Because of having adequate mechanical strength, it has capability for manipulation in a clinically applicable way with no breakage 97 |
Biodegradability time The effect of a functional endothelial layer on RAFT transparency 97 |
The mechanical strength of these compressed gels is improved and sufficient to withstand the manipulation without rupture 97 | None | Human corneal endothelial cells
97
Human corneal endothelial cell line (hCECL) B4G1297 |
None |
| Chitosan blends | Low costs
107
Biodegradable 100 Nontoxic 100 Acting as antibacterial, fungicidal compound100,102 Ability to accelerate wound healing100,102 Excellent biocompatibility100,102 Promotes ECM production 108 |
Poor solubility
102
Cell–scaffold interaction needs further studies 107 Lack study with human corneal endothelial cells and in vivo |
Blending with other materials can improve its mechanical properties104,107–111 | Good transparency 107 | Bovine corneal endothelial cells107,112
Rabbit corneal endothelial cells 111 |
None |
| Agarose | Natural material Biodegradability Biocompatibility Nontoxicity Porous structure allows to exchange nutrients, oxygen, and waste High ability to mimic human tissue and enhance cellular activities High cell–matrix interaction Great hydrophilicity and elasticity 113 |
Poor cell adhesive ability due to lacking appropriate chemical groups for enhancing cells’ attachment 114 | Strong mechanical properties 113 | Excellent optical transparency 114 | Rabbit corneal endothelial cells 114 | None |
| Poly-ε-lysine (pεK) | Being a synthetic material, it can be tightly controlled to produce customized characteristics of scaffold
119
Biocompatibility Porous matrix structure 118 Transparent and easy to manipulate118,119 |
Further in vivo studies with human corneal endothelial cells are needed | Sufficient mechanical properties for handling during surgery 118 | Excellent transparency118,120 | Human corneal endothelial cell line (HCEC-12)
119
Primary porcine endothelial cells 119 |
None |
AM, amniotic membrane; DM, Descemet’s membrane; DS, Descemet stripping; ECM, extracellular matrix; Descemet’s stripping endothelial keratoplasty DSEK; HCECs, human corneal endothelial cells; HCEnCs, human corneal endothelial cells; HLA, human leukocyte antigen; LC, lens capsule; RAFT, Real Architecture For 3D Tissues; RGD, arginine-glycine-aspartic acid.
For successful tissue engineering, scaffolds of CECs must meet specific requirements and characteristics of the ideal scaffold. These required properties include, but are not limited to, transparency, biocompatibility, biodegradability, permeability, and providing a proper microenvironment for cell activities in terms of biomechanical and biochemical properties.
Transparency
Successful bioengineered CECs should bear deep resemblance to natural cornea in terms of transparency. This ultimately will support obtaining a clear vision after treatment. Fortunately, transparency results of scaffolds were in fact outstanding in some biomaterials such as silk fibroin,71,72 pεK,118,120 chitosan,107,112 agarose, 114 decellularized human crystalline LCs, 63 and decellularized DS from porcine cornea. 58
Biocompatibility
Biocompatibility is a very recognized property within biomaterial studies in bioengineering field. Basically, it is defined as an appropriate host response against implanted material, while the material being required to perform the proper function. 121 It could indicate several aspects resulting from cell–biomaterial interactions. Thus, being biologically compatible materials means noncytotoxicity, low immunogenicity, not inducing deleterious reactions, and being noncarcinogenic. Furthermore, biocompatible scaffold allows cells to attach and grow with excellent viability. Most studies within the field of CECs have focused on biocompatibility of used scaffolds as key property for success in animal model studies in vivo. This critical step provides a result regarding the eye’s acceptance of the scaffold. All the biomaterials (synthetic or natural source) used have displayed acceptable biocompatibility, such as silk,71,72 collagen, decellularized tissues, pεK,118,120 and agarose. 114 However, some studies did not perform the biocompatibility test using HCECs, such as chitosan 107 and agarose 115 scaffolds, which were directly experimented on animal cells.
Biodegradation ability
Remarkable consideration has been paid to study the biodegradability of biomaterials. A major prerequisite for choosing a scaffold is that the material’s degradation time must meet the regeneration/curing process, and any change of mechanical qualities with degradation must match regeneration/healing process. Furthermore, the degradation products should be harmless and easily absorbed or removed from the body such as silk fibroins. 67 Decellularized human crystalline LCs have shown an acceptable degradation time (after 13 h) comparing with DM (after 17 h). 63
Permeability
Corneal endothelium acts as a leaky barrier between the corneal stroma (anterior) and anterior chamber (posterior). Protein and other nutrients (such as glucose) are actively transported, whereas water passively moves from the anterior chamber into the stroma through CECs. 129 Thus, the good permeability of scaffold can support and provide this function to continue. By in vitro test, collagen, 90 gelatin, 77 and chitosan 111 have displayed appropriate permeability.
Mechanical properties
It is known that the mechanical properties of scaffold play a vital role in cell adhesion and proliferation. In addition, high elasticity allows handling the scaffold under low risk of rupture during surgical procedures. Thus, many studies have focused on understanding and investigating the mechanical characteristics of the scaffold. AM has been found to have low mechanical stability; 54 however, this inconsistency could be related to the composition of the placenta 54 or resulting from the preparation method of AM. 53 Decellularized tissues have shown limited mechanical properties as well. 46 This could be a result of donor variation. However, silk fibroin,72,125 compressed collagen gel, 128 and agarose 113 have shown excellent mechanical robustness. Notably, chitosan scaffold was low in mechanical strength. 107 To overcome this drawback and improve chitosan’s mechanical strength, a blending approach with other substances such as PCL, genipin, collagen, and hydroxyethyl sulfate–gelatin104,107–111 has been conducted, which provided acceptable mechanical property for chitosan.
Finally, it can be stated that it is difficult to reach a definitive conclusion for which available biomaterial scaffold can be the best candidate for substituting DM. So far, attempts are still underway in finding the outstanding scaffold for CECs, with sophisticated methods being used. Most of the suggested biomaterials have shown promising results; however, some of them lack the preclinical trials on animal models (in vivo studies) (mentioned in Table 1) as it is crucial to analyze and understand the results of this type of study before moving from bench to bedside. This will be a vital step forward in the decision of choosing the most suited candidate for CECs.
According to the findings of several research works, collagen is considered to be the most extensively used material in corneal bioengineering applications 43 for many reasons. Structurally, it is the most abundant protein in human DM and cornea (as a whole), due to representing about 71% of corneal dry weight (primarily consisting of collagen type I). Furthermore, it is able to mimic the natural environment of cells. Also, there are various additional advantages for collagen as a biomaterial (mentioned in section ‘Collagen’) such as commercial availability, low immunogenicity, superior biodegradability, and excellent biocompatibility. In this context, plastic compression is a successful way to increase the strength of collagen and create a mimic environment of corneal tissue (details in section ‘Collagen’).
While collagen is the most widely employed material for corneal bioengineering applications, decellularized scaffold appears to be next. It is apparent from reviewing the literature that the decellularization research field is demonstrating significant growth. The dECM is becoming a promising tool to conduct regenerative medicine research, with numerous benefits (covered in the ‘Decellularized tissues’ section). In that regard, the HHP method could be considered as a powerful tool in reproducing scaffolds that are disease-free.
Future perspective and conclusions
Fuchs’ endothelial cell dystrophy has been characterized and reported by Hribek and colleagues 17 as a bilateral disease of the corneal endothelium in humans, clearly related to an accelerated decline of the CECs, which starts at the central region and thereafter spreads toward the peripheral region of the cornea. Due to the fact that HCECs do not have mitotic capability, the rest of the endothelial cells try to cover the empty spaces of the posterior surface by spreading and migration. However, gradually, those cells lose their healthy morphology (hexagonal) and size. Those changes are accompanied by changes in DM, which include an accumulation of ECM, commonly known as guttae (formation of posterior focal excrescences).16,17 Consequently, Fuchs’ dystrophy could induce changes in mechanical properties of DM.
From these indications, a study has aimed at understanding the mechanical properties of DM in Fuchs’ endothelial dystrophy patients. This study has found that the diseased DMs are much thicker than the control DM. In addition, the control sample had a porous homogeneous structure, while the diseased DM displayed different sizes of wide-spaced collagen structures. Moreover, the mechanical properties of the corneal posterior surface changed, not just in the guttae sites, but in the entire DM. Within the diseased DM, the wide-spaced collagen was found to be softer than the rest of the tissue, which in turn had a similar stiffness value to the control DM. 129 It is known that any change in the biomechanical properties of the surrounding microenvironment of cells could affect them by changing their behavior through mechanotransduction. 130 This suggests that such changes in Fuchs’ endothelial dystrophy could affect corneal endothelial cell behavior.
It is worth mentioning that a few research groups, currently working to improve properties of biomaterials by focusing on mechanical qualities of scaffolds, can take note of the aforementioned results to understand how to guide cells for migration toward the damage or proliferation for compensating damaged cells through understanding the mechanotransduction by which CECs respond to biophysical signals from substrates. This could play an important role in finding the most appropriate scaffold for corneal endothelial cell growth in vitro. Mechanotransduction alludes to every process wherein cells transform mechanical cues sent by the microenvironment to biochemical signals for adjusting their behavior to the environment. 130 It is commonly known that cells repeatedly sense the microenvironment and detect any modulating effect in their ECM, which is reflected on their behavior and hence their functionality. Recent numerous reviews conducted on different types of cells such as vascular endothelial cells, 131 skin cells, 132 immune cells, 133 and corneal epithelial stem cells 134 have revealed that mechanical cues play important roles in regulating fundamental cellular functions.
In conclusion, extensive work has been clearly done over the past few years to establish the ideal scaffold for HCECs which will be the alternative substitute for native corneas. Many characteristics have been assessed by examining various groups of biomaterials including biocompatibility, biodegradability, and cell–scaffold interaction. According to the encouraging results so far attained, it can be said that success is possible in the near future. However, there is a need to focus on biomaterials not only in general, but their individual properties with particular interest on their biomechanical qualities as a potentially important factor that influences the growth process and cell behavior to be as close as possible to the natural CECs in humans. These improvements could be made sooner if the role of mechanotransduction is elucidated for corneal endothelial cell homeostasis as it has been for the corneal epithelial cells.
Acknowledgments
Noor Ahmed Hussain is thankful for the scholarship by University of Jeddah that was provided through the Saudi Arabian Cultural Mission in the United Kingdom.
Footnotes
Author Note: Francisco C. Figueiredo is now affiliated to Department of Ophthalmology, Royal Victoria Infirmary, Newcastle upon Tyne Hospitals NHS Foundation Trust, Newcastle upon Tyne, UK.
Conflict of interest statement: The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: University of Jeddah, Kingdom of Saudi Arabia.
ORCID iD: Che J. Connon  https://orcid.org/0000-0001-8264-6531
https://orcid.org/0000-0001-8264-6531
Contributor Information
Noor Ahmed Hussain, University of Jeddah, Jeddah, Saudi ArabiaBiosciences Institute, Faculty of Medical Sciences, Newcastle University, Newcastle upon Tyne, UK.
Francisco C. Figueiredo, Biosciences Institute, Faculty of Medical Sciences, Newcastle University, Newcastle upon Tyne, UKDepartment of Ophthalmology, Royal Victoria Infirmary, Newcastle upon Tyne Hospitals NHS Foundation Trust, Newcastle upon Tyne, UK
Che J. Connon, Biosciences Institute, Faculty of Medical Sciences, Newcastle University, Newcastle upon Tyne NE1 7RU, UK.
References
- 1. Jirsova K. The cornea, anatomy and function no title. In: Jirsova K. (ed.) Light and specular microscopy of the cornea. Cham: Springer, 2017, pp. 1–21. [Google Scholar]
- 2. Sridhar MS. Anatomy of cornea and ocular surface. Indian J Ophthalmol 2018; 66: 190–194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Nishida T, Saika S, Morishige N. Cornea and sclera: anatomy and physiology. In: Mannis MJ, Holland EJ. (eds) Cornea: fundamentals, diagnosis and management. New York: Elsevier, 2010, pp. 1–22. [Google Scholar]
- 4. Bahn CF, Falls HF, Varley GA, et al. Classification of corneal endothelial disorders based on neural crest origin. Ophthalmology 1984; 91: 558–563. [DOI] [PubMed] [Google Scholar]
- 5. Soh YQ, Peh G, George BL, et al. Predicative factors for corneal endothelial cell migration. Invest Ophthalmol Vis Sci 2016; 57: 338–348. [DOI] [PubMed] [Google Scholar]
- 6. Yam GH, Seah X, Yusoff NZBM, et al. Characterization of human transition zone reveals a putative progenitor-enriched niche of corneal endothelium. Cells 2019; 8: 1244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Parekh M, Peh G, Mehta JS, et al. Passaging capability of human corneal endothelial cells derived from old donors with and without accelerating cell attachment. Exp Eye Res 2019; 189: 107814. [DOI] [PubMed] [Google Scholar]
- 8. Soh YQ, Peh GSL, Mehta JS. Translational issues for human corneal endothelial tissue engineering. J Tissue Eng Regen Med 2017; 11: 2425–2442. [DOI] [PubMed] [Google Scholar]
- 9. Chen S, Zhu Q, Sun H, et al. Advances in culture, expansion and mechanistic studies of corneal endothelial cells: a systematic review. J Biomed Sci 2019; 26: 1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Bartakova A, Alvarez-Delfin K, Weisman AD, et al. Novel identity and functional markers for human corneal endothelial cells. Investig Ophthalmol Vis Sci 2016; 57: 2749–2762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Hsueh YJ, Chen HC, Wu SE, et al. Lysophosphatidic acid induces YAP-promoted proliferation of human corneal endothelial cells via PI3K and ROCK pathways. Mol Ther Methods Clin Dev 2015; 2: 15014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Joyce NC. Cell cycle status in human corneal endothelium. Exp Eye Res 2005; 81: 629–638. [DOI] [PubMed] [Google Scholar]
- 13. Navaratnam J, Utheim T, Rajasekhar V, et al. Substrates for expansion of corneal endothelial cells towards bioengineering of human corneal endothelium. J Funct Biomater 2015; 6: 917–945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Wahlig S, Lovatt M, Peh GSL, et al. Corneal endothelial cells: methods for ex vivo expansion. In: Alió J, Alió del Barrio J, Arnalich-Montiel F. (eds) Corneal regeneration: essentials in ophthalmology. Cham: Springer, 2019, pp. 109–122. [Google Scholar]
- 15. Murphy C, Alvarado J, Juster R, et al. Prenatal and postnatal cellularity of the human corneal endothelium. A quantitative histologic study. Invest Ophthalmol Vis Sci 1984; 25: 312–322. [PubMed] [Google Scholar]
- 16. Biswas S, Munier FL, Yardley J, et al. Missense mutations in COL8A2, the gene encoding the α2 chain of type VIII collagen, cause two forms of corneal endothelial dystrophy. Hum Mol Genet 2001; 10: 2415–2423. [DOI] [PubMed] [Google Scholar]
- 17. Hribek A, Clahsen T, Horstmann J, et al. Fibrillar layer as a marker for areas of pronounced corneal endothelial cell loss in advanced Fuchs endothelial corneal dystrophy. Am J Ophthalmol 2021; 222: 292–301. [DOI] [PubMed] [Google Scholar]
- 18. Gain P, Jullienne R, He Z, et al. Global survey of corneal transplantation and eye banking. JAMA Ophthalmol 2016; 134: 167–173. [DOI] [PubMed] [Google Scholar]
- 19. Cardascia N, Pastore V, Bini V, et al. Graft detachment after Descemet’s stripping automated endothelial keratoplasty in bullous keratopathy and Fuchs dystrophy. Med Hypothesis Discov Innov Ophthalmol 2020; 9: 15–22. [PMC free article] [PubMed] [Google Scholar]
- 20. Thomas PB, Mukherjee AN, O’Donovan D, et al. Preconditioned donor corneal thickness for microthin endothelial keratoplasty. Cornea 2013; 32: e173–e178. [DOI] [PubMed] [Google Scholar]
- 21. Chen ES, Terry MA, Shamie N, et al. Descemet-stripping automated endothelial keratoplasty. Cornea 2008; 27: 514–520. [DOI] [PubMed] [Google Scholar]
- 22. Li W, Sabater AL, Chen YT, et al. A novel method of isolation, preservation, and expansion of human corneal endothelial cells. Invest Ophthalmol Vis Sci 2007; 48: 614–620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Peh GSL, Chng Z, Ang HP, et al. Propagation of human corneal endothelial cells: a novel dual media approach. Cell Transplant 2015; 24: 287–304. [DOI] [PubMed] [Google Scholar]
- 24. Okumura N, Koizumi N, Ueno M, et al. ROCK inhibitor converts corneal endothelial cells into a phenotype capable of regenerating in vivo endothelial tissue. Am J Pathol 2012; 181: 268–277. [DOI] [PubMed] [Google Scholar]
- 25. Okumura N, Ueno M, Koizumi N, et al. Enhancement on primate corneal endothelial cell survival in vitro by a rock inhibitor. Invest Ophthalmol Vis Sci 2009; 50: 3680–3687. [DOI] [PubMed] [Google Scholar]
- 26. Mahdavi SS, Abdekhodaie MJ, Mashayekhan S, et al. Bioengineering approaches for corneal regenerative medicine. Tissue Eng Regen Med 2020; 17: 567–593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Choi JS, Williams JK, Greven M, et al. Bioengineering endothelialized neo-corneas using donor-derived corneal endothelial cells and decellularized corneal stroma. Biomaterials 2010; 31: 6738–6745. [DOI] [PubMed] [Google Scholar]
- 28. Rao BM, Zandstra PW. Culture development for human embryonic stem cell propagation: molecular aspects and challenges. Curr Opin Biotechnol 2005; 16: 568–576. [DOI] [PubMed] [Google Scholar]
- 29. Peh GSL, Beuerman RW, Colman A, et al. Human corneal endothelial cell expansion for corneal endothelium transplantation: an overview. Transplantation 2011; 91: 811–819. [DOI] [PubMed] [Google Scholar]
- 30. Okumura N, Koizumi N, Ueno M, et al. Enhancement of corneal endothelium wound healing by Rho-associated kinase (ROCK) inhibitor eye drops. Br J Ophthalmol 2011; 95: 1006–1009. [DOI] [PubMed] [Google Scholar]
- 31. Peh GSL, Adnan K, George BL, et al. The effects of rho-associated kinase inhibitor Y-27632 on primary human corneal endothelial cells propagated using a dual media approach. Sci Rep 2015; 5: 9167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Okumura N, Nakano S, Kay EDP, et al. Involvement of cyclin D and p27 in cell proliferation mediated by ROCK inhibitors Y-27632 and Y-39983 during corneal endothelium wound healing. Investig Ophthalmol Vis Sci 2014; 55: 318–329. [DOI] [PubMed] [Google Scholar]
- 33. Thuret G, Courrier E, Poinard S, et al. One threat, different answers: the impact of COVID-19 pandemic on cornea donation and donor selection across Europe. Br J Ophthalmol. Epub ahead of print November 2020. DOI: 10.1136/bjophthalmol-2020-317938. [DOI] [PubMed] [Google Scholar]
- 34. Ang M, Moriyama A, Colby K, et al. Corneal transplantation in the aftermath of the COVID-19 pandemic: an international perspective. Br J Ophthalmol 2020; 104: 1477–1481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Schmedt T, Chen Y, Nguyen TT, et al. Telomerase immortalization of human corneal endothelial cells yields functional hexagonal monolayers. PLoS ONE 2012; 7: e51427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Zhang J, Ahmad AM, Ng H, et al. Successful culture of human transition zone cells. Clin Exp Ophthalmol 2020; 48: 689–700. [DOI] [PubMed] [Google Scholar]
- 37. Fujita M, Mehra R, Lee SE, et al. Comparison of proliferative capacity of genetically-engineered pig and human corneal endothelial cells. Ophthalmic Res 2013; 49: 127–138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. David MB, Balgos MJTD, Arnalich-Montiel F. Corneal endothelium: isolation and cultivation methods. In: Alió J, Alió del Barrio J, Arnalich-Montiel F. (eds) Corneal regeneration: essentials in ophthalmology. Cham: Springer, 2019, pp. 425–436. [Google Scholar]
- 39. Ide T, Nishida K, Yamato M, et al. Structural characterization of bioengineered human corneal endothelial cell sheets fabricated on temperature-responsive culture dishes. Biomaterials 2006; 27: 607–614. [DOI] [PubMed] [Google Scholar]
- 40. Sumide T, Nishida K, Yamato M, et al. Functional human corneal endothelial cell sheets harvested from temperature-responsive culture surfaces. FASEB J 2006; 20: 392–394. [DOI] [PubMed] [Google Scholar]
- 41. Kinoshita S, Koizumi N, Ueno M, et al. Injection of cultured cells with a ROCK inhibitor for bullous keratopathy. N Engl J Med 2018; 378: 995–1003. [DOI] [PubMed] [Google Scholar]
- 42. Bhat S, Kumar A. Biomaterials and bioengineering tomorrow’s healthcare. Biomatter 2013; 3: e24717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Chen Z, You J, Liu X, et al. Biomaterials for corneal bioengineering. Biomed Mater 2018; 13: 032002. [DOI] [PubMed] [Google Scholar]
- 44. Sionkowska A. Current research on the blends of natural and synthetic polymers as new biomaterials: review. Prog Polym Sci 2011; 36: 1254–1276. [Google Scholar]
- 45. Parekh M, Romano V, Hassanin K, et al. Biomaterials for corneal endothelial cell culture and tissue engineering. J Tissue Eng. Epub ahead of print February 2021. DOI: 10.1177/2041731421990536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Niknejad H, Deihim T, Solati-Hashjin M, et al. The effects of preservation procedures on amniotic membrane’s ability to serve as a substrate for cultivation of endothelial cells. Cryobiology 2011; 63: 145–151. [DOI] [PubMed] [Google Scholar]
- 47. Tseng SCG, Li DQ, Ma X. Suppression of transforming growth factor-beta isoforms, TGF-β receptor type II, and myofibroblast differentiation in cultured human corneal and limbal fibroblasts by amniotic membrane matrix. J Cell Physiol 1999; 179: 325–335. [DOI] [PubMed] [Google Scholar]
- 48. Arrizabalaga JH, Nollert MU. Human amniotic membrane: a versatile scaffold for tissue engineering. ACS Biomater Sci Eng 2018; 4: 2226–2236. [DOI] [PubMed] [Google Scholar]
- 49. Ishino Y, Sano Y, Nakamura T, et al. Amniotic membrane as a carrier for cultivated human corneal endothelial cell transplantation. Investig Ophthalmol Vis Sci 2004; 45: 800–806. [DOI] [PubMed] [Google Scholar]
- 50. Niknejad H, Peirovi H, Jorjani M, et al. Properties of the amniotic membrane for potential use in tissue engineering. Eur Cells Mater 2008; 15: 88–99. [DOI] [PubMed] [Google Scholar]
- 51. Connon CJ, Doutch J, Chen B, et al. The variation in transparency of amniotic membrane used in ocular surface regeneration. Br J Ophthalmol 2010; 94: 1057–1061. [DOI] [PubMed] [Google Scholar]
- 52. Grémare A, Jean-Gilles S, Musqui P, et al. Cartography of the mechanical properties of the human amniotic membrane. J Mech Behav Biomed Mater 2019; 99: 18–26. [DOI] [PubMed] [Google Scholar]
- 53. Chen B, Jones RR, Mi S, et al. The mechanical properties of amniotic membrane influence its effect as a biomaterial for ocular surface repair. Soft Matter 2012; 8: 8379–8387. [Google Scholar]
- 54. Leal-Marin S, Kern T, Hofmann N, et al. Human Amniotic Membrane: a review on tissue engineering, application, and storage. J Biomed Mater Res Part B Appl Biomater 2021; 109: 1198–1215. [DOI] [PubMed] [Google Scholar]
- 55. Dua HS, Rahman I, Miri A, et al. Variations in amniotic membrane: relevance for clinical applications. Br J Ophthalmol 2010; 94: 963–964. [DOI] [PubMed] [Google Scholar]
- 56. Crapo PM, Gilbert TW, Badylak SF. An overview of tissue and whole organ decellularization processes. Biomaterials 2011; 32: 3233–3243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Gilpin A, Yang Y. Decellularization strategies for regenerative medicine: from processing techniques to applications. Biomed Res Int 2017; 2017: 9831534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. An JH, Park SY, Kim GH, et al. Tissue engineered ultra-thin Descemet stripping corneal endothelial layers using porcine cornea and stem cells. Exp Eye Res 2020; 199: 108192. [DOI] [PubMed] [Google Scholar]
- 59. Du L, Wu X. Development and characterization of a full-thickness acellular porcine cornea matrix for tissue engineering. Artif Organs 2011; 35: 691–705. [DOI] [PubMed] [Google Scholar]
- 60. Oliveira AC, Garzón I, Ionescu AM, et al. Evaluation of small intestine grafts decellularization methods for corneal tissue engineering. PLoS ONE 2013; 8: e66538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Hashimoto Y, Funamoto S, Sasaki S, et al. Preparation and characterization of decellularized cornea using high-hydrostatic pressurization for corneal tissue engineering. Biomaterials 2010; 31: 3941–3948. [DOI] [PubMed] [Google Scholar]
- 62. Bayyoud T, Thaler S, Hofmann J, et al. Decellularized bovine corneal posterior lamellae as carrier matrix for cultivated human corneal endothelial cells. Curr Eye Res 2012; 37: 179–186. [DOI] [PubMed] [Google Scholar]
- 63. Van den Bogerd B, Ní Dhubhghaill S, Zakaria N. Characterizing human decellularized crystalline lens capsules as a scaffold for corneal endothelial tissue engineering. J Tissue Eng Regen Med 2018; 12: e2020–e2028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Jang J, Kim TG, Kim BS, et al. Tailoring mechanical properties of decellularized extracellular matrix bioink by vitamin B2-induced photo-crosslinking. Acta Biomater 2016; 33: 88–95. [DOI] [PubMed] [Google Scholar]
- 65. Jang J, Park HJ, Kim SW, et al. 3D printed complex tissue construct using stem cell-laden decellularized extracellular matrix bioinks for cardiac repair. Biomaterials 2017; 112: 264–274. [DOI] [PubMed] [Google Scholar]
- 66. Pati F, Jang J, Ha DH, et al. Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink. Nat Commun 2014; 5: 3935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Cao Y, Wang B. Biodegradation of silk biomaterials. Int J Mol Sci 2009; 10: 1514–1524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Gil ES, Panilaitis B, Bellas E, et al. Functionalized silk biomaterials for wound healing. Adv Healthc Mater 2013; 2: 206–217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Meinel L, Hofmann S, Karageorgiou V, et al. The inflammatory responses to silk films in vitro and in vivo. Biomaterials 2005; 26: 147–155. [DOI] [PubMed] [Google Scholar]
- 70. Chirila T, Barnard Zainuddin Z, et al. Bombyx mori silk fibroin membranes as potential substrata for epithelial constructs used in the management of ocular surface disorders. Tissue Eng Part A 2008; 14: 1203–1211. [DOI] [PubMed] [Google Scholar]
- 71. Madden PW, Lai JNX, George KA, et al. Human corneal endothelial cell growth on a silk fibroin membrane. Biomaterials 2011; 32: 4076–4084. [DOI] [PubMed] [Google Scholar]
- 72. Ramachandran C, Gupta P, Hazra S, et al. In vitro culture of human corneal endothelium on non-mulberry silk fibroin films for tissue regeneration. Transl Vis Sci Technol 2020; 9: 12–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Echave MC, Sánchez P, Pedraz JL, et al. Progress of gelatin-based 3D approaches for bone regeneration. J Drug Deliv Sci Technol 2017; 42: 63–74. [Google Scholar]
- 74. Karim AA, Bhat R. Fish gelatin: properties, challenges, and prospects as an alternative to mammalian gelatins. Food Hydrocoll 2009; 23: 563–576. [Google Scholar]
- 75. Wang H, Boerman OC, Sariibrahimoglu K, et al. Comparison of micro- vs. nanostructured colloidal gelatin gels for sustained delivery of osteogenic proteins: bone morphogenetic protein-2 and alkaline phosphatase. Biomaterials 2012; 33: 8695–8703. [DOI] [PubMed] [Google Scholar]
- 76. Wang F, Li Y, Shen Y, et al. The functions and applications of RGD in tumor therapy and tissue engineering. Int J Mol Sci 2013; 14: 13447–13462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Watanabe R, Hayashi R, Kimura Y, et al. A novel gelatin hydrogel carrier sheet for corneal endothelial transplantation. Tissue Eng Part A 2011; 17: 2213–2219. [DOI] [PubMed] [Google Scholar]
- 78. Rizwan M, Peh GSL, Ang HP, et al. Sequentially-crosslinked bioactive hydrogels as nano-patterned substrates with customizable stiffness and degradation for corneal tissue engineering applications. Biomaterials 2017; 120: 139–154. [DOI] [PubMed] [Google Scholar]
- 79. Yue K, Trujillo-de Santiago G, Alvarez MM, et al. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials 2015; 73: 254–271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Yang C, Hillas PJ, Báez JA, et al. The application of recombinant human collagen in tissue engineering. Biodrugs 2004; 18: 103–119. [DOI] [PubMed] [Google Scholar]
- 81. Berisio R, Vitagliano L, Mazzarella L, et al. Recent progress on collagen triple helix structure, stability and assembly. Protein Pept Lett 2002; 9: 107–116. [DOI] [PubMed] [Google Scholar]
- 82. Brodsky B, Ramshaw JAM. The collagen triple-helix structure. Matrix Biol 1997; 15: 545–554. [DOI] [PubMed] [Google Scholar]
- 83. Newsome DA, Foidart JM, Hassell JR, et al. Detection of specific collagen types in normal and keratoconus corneas. Invest Ophthalmol Vis Sci 1981; 20: 738–750. [PubMed] [Google Scholar]
- 84. Newsome DA, Gross J, Hassell JR. Human corneal stroma contains three distinct collagens. Invest Ophthalmol Vis Sci 1982; 22: 376–381. [PubMed] [Google Scholar]
- 85. Hassell JR, Birk DE. The molecular basis of corneal transparency. Exp Eye Res 2010; 91: 326–335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Gonzalez-Andrades M, Argüeso P, Gipson I. Part I: corneal regeneration: the concept, the facts, the potential: corneal anatomy. In: Alió JL, del Barrio JLA, Arnalich-Montiel F. (eds) Corneal regeneration: essentials in ophthalmology. Cham: Springer, 2019, pp. 3–12. [Google Scholar]
- 87. Kanta J. Collagen matrix as a tool in studying fibroblastic cell behavior. Cell Adh Migr 2015; 9: 308–316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Rajan N, Habermehl J, Coté MF, et al. Preparation of ready-to-use, storable and reconstituted type I collagen from rat tail tendon for tissue engineering applications. Nat Protoc 2007; 1: 2753–2758. [DOI] [PubMed] [Google Scholar]
- 89. Elsdale T, Bard J. Collagen substrata for studies on cell behavior. J Cell Biol 1972; 54: 626–637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Dong C, Lv Y. Application of collagen scaffold in tissue engineering: recent advances and new perspectives. Polymers (Basel) 2016; 8: 42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Friess W. Collagen: biomaterial for drug delivery. Eur J Pharm Biopharm 1998; 45: 113–136. [DOI] [PubMed] [Google Scholar]
- 92. Browne S, Zeugolis DI, Pandit A. Collagen: finding a solution for the source. Tissue Eng Part A 2013; 19: 1491–1494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Lynn AK, Yannas IV, Bonfield W. Antigenicity and immunogenicity of collagen. J Biomed Mater Res B Appl Biomater 2004; 71: 343–354. [DOI] [PubMed] [Google Scholar]
- 94. Adachi T, Tomita M, Shimizu K, et al. Generation of hybrid transgenic silkworms that express Bombyx mori prolyl-hydroxylase α-subunits and human collagens in posterior silk glands: production of cocoons that contained collagens with hydroxylated proline residues. J Biotechnol 2006; 126: 205–219. [DOI] [PubMed] [Google Scholar]
- 95. Nokelainen M, Tu H, Vuorela A, et al. High-level production of human type I collagen in the yeast Pichia pastoris. Yeast 2001; 18: 797–806. [DOI] [PubMed] [Google Scholar]
- 96. Mimura T, Yamagami S, Yokoo S, et al. Cultured human corneal endothelial cell transplantation with a collagen sheet in a rabbit model. Invest Ophthalmol Vis Sci 2004; 45: 2992–2997. [DOI] [PubMed] [Google Scholar]
- 97. Levis HJ, Peh GSL, Toh KP, et al. Plastic compressed collagen as a novel carrier for expanded human corneal endothelial cells for transplantation. PLoS ONE 2012; 7: e50993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Brown RA, Wiseman M, Chuo CB, et al. Ultrarapid engineering of biomimetic materials and tissues: fabrication of nano- and microstructures by plastic compression. Adv Funct Mater 2005; 15: 1762–1770. [Google Scholar]
- 99. Jones RR, Hamley IW, Connon CJ. Ex vivo expansion of limbal stem cells is affected by substrate properties. Stem Cell Res 2012; 8: 403–409. [DOI] [PubMed] [Google Scholar]
- 100. Sánchez-Machado D, López-Cervantes J, Correa-Murrieta MA, et al. Chitosan. In: Nabavi SM, Silva AS. (eds) Nonvitamin and nonmineral nutritional supplements. Cambridge, MA: Academic Press, 2019, pp. 485–493. [Google Scholar]
- 101. Rodríguez-Vázquez M, Vega-Ruiz B, Ramos-Zúñiga R, et al. Chitosan and its potential use as a scaffold for tissue engineering in regenerative medicine. Biomed Res Int 2015; 2015: 821279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Bakshi PS, Selvakumar D, Kadirvelu K, et al. Chitosan as an environment friendly biomaterial – a review on recent modifications and applications. Int J Biol Macromol 2020; 150: 1072–1083. [DOI] [PubMed] [Google Scholar]
- 103. Anal AK, Tobiassen A, Flanagan J, et al. Preparation and characterization of nanoparticles formed by chitosan-caseinate interactions. Colloids Surfaces B Biointerfaces 2008; 64: 104–110. [DOI] [PubMed] [Google Scholar]
- 104. Jorge EVG, Guillermo M, Judith Z, et al. In vivo biocompatibility of chitosan and collagen-vitrigel membranes for corneal scaffolding: a comparative analysis. Curr Tissue Eng 2016; 5: 123–129. [Google Scholar]
- 105. Ueno H. Topical formulations and wound healing applications of chitosan 2. Topical findings of healing with chitosan at early phase of experimental open skin wound. Adv Drug Deliv Rev 2001; 52: 105–115. [DOI] [PubMed] [Google Scholar]
- 106. VandeVord PJ, Matthew HWT, DeSilva SP, et al. Evaluation of the biocompatibility of a chitosan scaffold in mice. J Biomed Mater Res 2002; 59: 585–590. [DOI] [PubMed] [Google Scholar]
- 107. Wang TJ, Wang IJ, Chen S, et al. The phenotypic response of bovine corneal endothelial cells on chitosan/polycaprolactone blends. Colloids Surfaces B Biointerfaces 2012; 90: 236–243. [DOI] [PubMed] [Google Scholar]
- 108. Young TH, Wang IJ, Hu FR, et al. Fabrication of a bioengineered corneal endothelial cell sheet using chitosan/polycaprolactone blend membranes. Colloids Surfaces B Biointerfaces 2014; 116: 403–410. [DOI] [PubMed] [Google Scholar]
- 109. Mi FL, Tan YC, Liang HF, et al. In vivo biocompatibility and degradability of a novel injectable-chitosan-based implant. Biomaterials 2002; 23: 181–191. [DOI] [PubMed] [Google Scholar]
- 110. Huang C, Chen R, Ke Q, et al. Electrospun collagen-chitosan-TPU nanofibrous scaffolds for tissue engineered tubular grafts. Colloids Surfaces B Biointerfaces 2011; 82: 307–315. [DOI] [PubMed] [Google Scholar]
- 111. Liang Y, Liu W, Han B, et al. Fabrication and characters of a corneal endothelial cells scaffold based on chitosan. J Mater Sci Mater Med 2011; 22: 175–183. [DOI] [PubMed] [Google Scholar]
- 112. Wang YH, Young TH, Wang TJ. Investigating the effect of chitosan/ polycaprolactone blends in differentiation of corneal endothelial cells and extracellular matrix compositions. Exp Eye Res 2019; 185: 107679. [DOI] [PubMed] [Google Scholar]
- 113. Salati MA, Khazai J, Tahmuri AM, et al. Agarose-based biomaterials: opportunities and challenges in cartilage tissue engineering. Polymers (Basel) 2020; 12: 1–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Seow WY, Kandasamy K, Peh GSL, et al. Ultrathin, strong, and cell-adhesive agarose-based membranes engineered as substrates for corneal endothelial cells. ACS Biomater Sci Eng 2019; 5: 4067–4076. [DOI] [PubMed] [Google Scholar]
- 115. Khodadadi Yazdi M, Taghizadeh A, Taghizadeh M, et al. Agarose-based biomaterials for advanced drug delivery. J Control Release 2020; 326: 523–543. [DOI] [PubMed] [Google Scholar]
- 116. Lopez Mora N, Owens M, Schmidt S, et al. Poly-epsilon-lysine hydrogels with dynamic crosslinking facilitates cell proliferation. Materials (Basel) 2020; 13: 3851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117. Masters JR. HeLa cells 50 years on: the good, the bad and the ugly. Nat Rev Cancer 2002; 2: 315–319. [DOI] [PubMed] [Google Scholar]
- 118. Gallagher AG, Alorabi JA, Wellings DA, et al. A novel peptide hydrogel for an antimicrobial bandage contact lens. Adv Healthc Mater 2016; 5: 2013–2018. [DOI] [PubMed] [Google Scholar]
- 119. Khaireddin R, Wachtlin J, Hopfenmüller W, et al. HLA-A, HLA-B and HLA-DR matching reduces the rate of corneal allograft rejection. Graefes Arch Clin Exp Ophthalmol 2003; 241: 1020–1028. [DOI] [PubMed] [Google Scholar]
- 120. Kennedy S, Lace R, Carserides C, et al. Poly-ε-lysine based hydrogels as synthetic substrates for the expansion of corneal endothelial cells for transplantation. J Mater Sci Mater Med 2019; 30: 1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Black J. Biological performance of materials : fundamentals of biocompatibility. 4th ed. Boca Raton: : CRC/Taylor & Francis, 2006. [Google Scholar]
- 122. Wencan W, Mao Y, Wentao Y, et al. Using basement membrane of human amniotic membrane as a cell carrier for cultivated cat corneal endothelial cell transplantation. Curr Eye Res 2007; 32: 199–215. [DOI] [PubMed] [Google Scholar]
- 123. McCrary MW, Bousalis D, Mobini S, et al. Decellularized tissues as platforms for in vitro modeling of healthy and diseased tissues. Acta Biomater. Epub ahead of print 2020. DOI: 10.1016/j.actbio.2020.05.031. [DOI] [PubMed] [Google Scholar]
- 124. Arnalich-Montiel F, Moratilla A, Fuentes-Julián S, et al. Treatment of corneal endothelial damage in a rabbit model with a bioengineered graft using human decellularized corneal lamina and cultured human corneal endothelium. PLoS One 2019; 14: 1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Yukseloglu SM, Sokmen N, Canoglu S. Biomaterial applications of silk fibroin electrospun nanofibres. Microelectron Eng 2015; 146: 43–47. [Google Scholar]
- 126. Song JE, Sim BR, Jeon YS, et al. Characterization of surface modified glycerol/silk fibroin film for application to corneal endothelial cell regeneration. J Biomater Sci Polym Ed 2019; 30: 263–275. [DOI] [PubMed] [Google Scholar]
- 127. Vázquez N, Rodríguez-Barrientos CA, Aznar-Cervantes SD, et al. Silk fibroin films for corneal endothelial regeneration: Transplant in a rabbit descemet membrane endothelial keratoplasty. Investig Ophthalmol Vis Sci 2017; 58: 3357–3365. [DOI] [PubMed] [Google Scholar]
- 128. Mi S, Chen B, Wright B, et al. Plastic compression of a collagen gel forms a much improved scaffold for ocular surface tissue engineering over conventional collagen gels. J Biomed Mater Res A 2010; 95: 447–453. [DOI] [PubMed] [Google Scholar]
- 129. Xia D, Zhang S, Nielsen E, et al. The ultrastructures and mechanical properties of the Descemet’s membrane in Fuchs endothelial corneal dystrophy. Sci Rep 2016; 6: 4–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130. Panciera T, Azzolin L, Cordenonsi M, et al. Mechanobiology of YAP and TAZ in physiology and disease. Nat Rev Mol Cell Biol 2017; 18: 758–770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131. Gordon E, Schimmel L, Frye M. The importance of mechanical forces for in vitro endothelial cell biology. Front Physiol 2020; 11: 684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132. Biggs LC, Kim CS, Miroshnikova YA, et al. Mechanical forces in the skin: roles in tissue architecture, stability, and function. J Invest Dermatol 2020; 140: 284–290. [DOI] [PubMed] [Google Scholar]
- 133. Huse M. Mechanical forces in the immune system. Nat Rev Immunol 2017; 17: 679–690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134. Gouveia RM, Lepert G, Gupta S, et al. Assessment of corneal substrate biomechanics and its effect on epithelial stem cell maintenance and differentiation. Nat Commun 2019; 10: 1496. [DOI] [PMC free article] [PubMed] [Google Scholar]