I. Yasuda, K. Hasegawa, Y. Sakamaki, H. Muraoka, T. Kawaguchi, E. Kusahana, T. Ono, T. Kanda, H. Tokuyama, S. Wakino, H. Itoh: Pre-emptive Short-term Nicotinamide Mononucleotide Treatment in a Mouse Model of Diabetic Nephropathy. J Am Soc Nephrol 32: 1355–1370, 2021.
The Figure 3 in the paper shows that the data were composed of five experimental groups although the experiment was originally done with four experimental groups, that is, db/m mice, db/db mice, db/db mice treated with 100 mg NMN, and db/db mice treated with 300 mg NMN. The data on mice treated with 500 mg NMN in Figure 3 were already presented in Figures 1E and 2E. This duplication was discovered after publication. The correct Figure 3, correct corresponding results for Figure 3, and correct Figure 3 legends and corresponding results part are as follows:
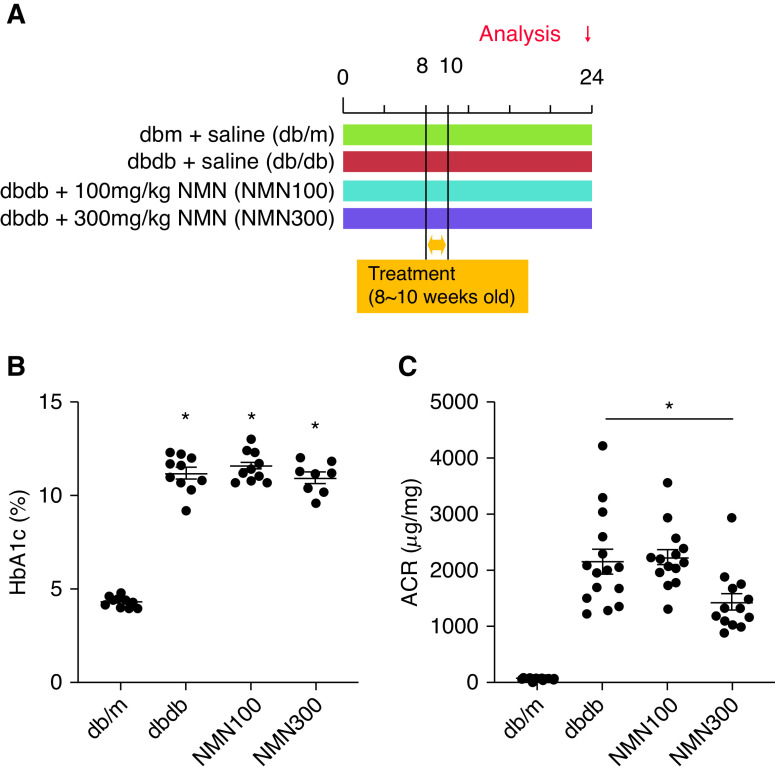
Figure 3.
Dose-response study of NMN treatment. (A) Schematic diagram illustrating the dose-response study. (B) HbA1c at 24 weeks of age in the four groups (db/m, db/db, NMN100, and NMN300; n=15). Statistical significance is represented by an asterisk. *P<0.05 versus db/m. (C) Urine ACR at 24 weeks of age in the four groups (db/m, db/db, NMN100, and NMN300; n=15). All data are shown as mean±SEM. Statistical significance between each group is represented by a horizontal bar. *P<0.05 by ANOVA with Tukey’s post hoc test.
Results
Dose-response Study of NMN Treatment
We further examined the dose-dependent effects of short-term NMN treatment (Figure 3A). The effects of short-term treatment with two additional doses of NMN, 100 and 300 mg/kg, on HbA1c levels and urine ACR levels were evaluated at 24 weeks of age. HbA1c levels of db/db mice treated with 100 mg/kg NMN (NMN100) and db/db mice treated with 300 mg/kg NMN (NMN300) were not different from those in db/db mice (Figure 3B). In comparison with db/db mice, the NMN300 group exhibited reduced ACR, whereas the NMN100 group did not show a significant reduction in ACR (Figure 3C). We have therefore observed dose-dependent effects of short-term NMN treatment on diabetic albuminuria.
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
See related original article, “Pre-emptive Short-term Nicotinamide Mononucleotide Treatment in a Mouse Model of Diabetic Nephropathy,” in Vol. 32, Iss. 6, on pages 1355–1370.