Abstract
Inflammation plays a prominent role in the development of atherosclerosis and other cardiovascular diseases, and anti-inflammatory agents may improve cardiovascular outcomes. For years, colchicine has been used as a safe and well-tolerated agent in diseases such as gout and familial Mediterranean fever. The widely available therapeutic has several anti-inflammatory effects however that have proven effective in a broad spectrum of cardiovascular diseases as well. It is considered standard-of-care therapy for pericarditis, and several clinical trials have evaluated its role in pericarditis, post-operative- and post-ablation atrial fibrillation, post-pericardiotomy syndrome, coronary artery disease, percutaneous coronary interventions, and cerebrovascular disease. We aim to summarize colchicine’s pharmacodynamics and the mechanism behind its anti-inflammatory effect, outline thus far accumulated evidence on treatment with colchicine in cardiovascular disease, and present ongoing randomized clinical trials. We also emphasize real-world clinical implications that should be considered on the basis of the merits and limitations of completed trials. Altogether, colchicine’s simplicity, low cost, and effectiveness may provide an important addition to other standard cardiovascular therapies. Ongoing studies will address complementary questions pertaining to the use of low-dose colchicine for the treatment of cardiovascular disease.
Keywords: Colchicine, Inflammation, Coronary Artery Disease, Pericarditis, Atrial Fibrillation, Post-Pericardiotomy Syndrome, Percutaneous Coronary Intervention, Cerebrovascular Disease
Introduction
Colchicine has carried many names and been used for centuries.1, 2 The botanical alkaloid compound is derived from the flower Colchicum autumnale, a medicinal plant described as early as 1550 B.C. in the Egyptian Ebers papyrus to treat pain and swelling. The plant and therapeutic lend their names to the ancient Colchis (home to mythological sorceresses Medea and Circe), roughly corresponding to the current republic of Georgia. Through the years, colchicine has been frequently described both for its medicinal use in gout (as early as 129 to 200 C.E.) as well as its poisonous effects at high doses. Having survived years of scrutiny, colchicine use is rapidly expanding as it is currently considered for a variety of inflammatory conditions. It currently carries Food & Drug Administration approved indications for prevention and treatment of gout flares, as well as treatment of familial Mediterranean fever (FMF). It is also utilized off-label for other inflammatory conditions including pericarditis, calcium pyrophosphate disease, and Adamantiades-Behcet’s syndrome.3–6 Over the years, many studies have shown that inflammation may significantly contribute to a wider variety of pathologies than traditionally thought, including cardiovascular diseases such as atherosclerosis and atrial fibrillation (AF). Therefore, anti-inflammatory strategies are now being carefully examined for potential broader application across a range of cardiovascular diseases. Given wide availability, low cost, and a rather favorable side effect (tolerability) profile, colchicine has emerged as a potentially useful oral cardiovascular treatment targeting the inflammatory axis. We will, hereafter, review the role inflammation plays in cardiovascular disease, including various anti-inflammatory strategies, and then examine past and current evidence regarding colchicine in cardiovascular disease.
Mechanism of Action
Colchicine is an oral therapeutic agent that binds tubulin and inhibits tubulin polymerization, with subsequent disruption of the cellular cytoskeleton, mitosis, and intracellular transport activities. Colchicine preferentially accumulates in neutrophils due to the lack of the P-glycoprotein membrane efflux pump and thereby largely impacts neutrophil activity (Figure 1).7 Specifically, colchicine has been shown to inhibit the directed migration of neutrophils to an inflamed focus (chemotaxis)8 and decrease adhesion of neutrophils to inflamed endothelium via diminished quantitative surface-expression of L-selectin adhesion molecules.9 Colchicine further inhibits the adhesion of leukocytes to inflamed endothelium via decreased qualitative expression of E-selectin adhesion molecules on endothelial cells,9 downregulation of tumor necrosis factor (TNF) receptors on macrophages and endothelial cells,10 and reduced monocyte/macrophage secretion of TNF-α.11 Finally, colchicine has been shown to suppress protein tyrosine phosphorylation in neutrophils with subsequent inhibition of both intracellular mobilization and extracellular release of granular enzymes, such as matrix metalloproteinases, neutrophil elastase, and α-defensins.12, 13
Figure 1. Inflammation in cardiovascular disease and anti-inflammatory targets of colchicine and canakinumab.
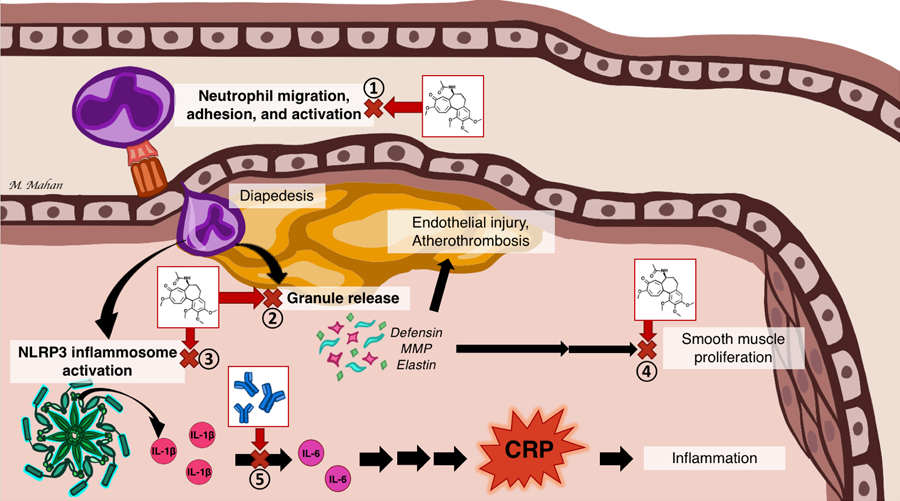
Colchicine has broad anti-inflammatory effects, here illustrated here in atherosclerosis. Colchicine inhibits (1) neutrophil migration, adhesion, activation, (2), neutrophil release of defensins, MMP, and elastin, (3) NLRP3 activation of the inflammatory pathway with release of IL-1β, in turn activating IL-6, and eventually stimulation CRP, and (4) smooth muscle proliferation and vascular stenosis. (5) Canakinumab is a monoclonal antibody that specifically targets the upstream inflammatory mediator IL-1β.
CRP = C-reactive protein; IL = Interleukin; MMP = Matrix Metalloproteinases, NLRP3 = NLR Family Pyrin Domain Containing 3, TGF = transforming growth factor
Though the exact mechanism of action remains incompletely elucidated, data show that colchicine also suppresses the assembly and activation of the NLR Family Pyrin Domain Containing 3 (NLRP3) inflammasome, with a resultant decrease in inflammasome-mediated production of interleukin (IL)-1β and IL-18.14, 15 Both IL-1β and IL-18 are also activated extracellularly by neutrophil enzymes (e.g. proteinase 3), the release of which are also inhibited by colchicine.16–18 Together, these lead to an overall decrease in IL-6 production and C-Reactive Protein (CRP) concentration. Finally, colchicine may suppress the proliferation of myofibroblasts, smooth muscle cell proliferation, and fibrosis.19
Dosing, Safety, and Tolerability
Colchicine acts in a dose-dependent manner, and most side effects reverse with lower doses or cessation of treatment.20 Although it is approved for higher doses in acute gout (4.8 mg over 6 hours load in patients without advanced kidney disease), colchicine generates similar peak plasma concentrations and is associated with improved tolerability with a lower dose regimen (1.0–1.2 mg followed by 0.5–0.6 mg one hour later).21 Maintenance dosage is 0.5–0.6 mg once or twice daily for acute disease; and 0.3–0.6 mg daily for chronic prevention. Dosage adjustments are recommended for acutely ill patients with impaired renal function or body weight under 70 kg. Furthermore, dosage formulations vary based on geographic location, as low-dose formulation tablets are available in 0.6 mg tablets in the United States versus 0.5 mg tablets in Australia. This disparity is evident in the differing dosages used in randomized clinical trials (RCTs) discussed below. Given the absence of data comparing efficacy and toxicity characteristics between the two formulations, preparation strength differences are most likely due to commercial manufacturing purposes. Proposed dosing of colchicine per indication can be found in Table 1.
Table 1.
Dosing and duration for colchicine stratified by indication
| Clinical Indication | Dose* | Duration |
|---|---|---|
| Pericardial disease | ||
| Acute and Recurrent Pericarditis | ▪ ≥70 kg: 0.5 mg twice daily‡ ▪ <70 kg: 0.5 mg once daily‡ |
3 months |
| Dressler’s Syndrome | ||
| Post-Pericardiotomy Syndrome | 1 month post cardiac surgery | |
| Atrial fibrillation | ||
| Post-Operative Atrial Fibrillation | 0.5 mg once or twice daily | 1–3 months |
| Post-Ablation Atrial Fibrillation | ||
| Coronary artery disease | ||
| Stable Coronary Artery Disease | 0.5 mg daily | Possibly indefinite |
| Acute Coronary Syndrome | ||
In the United States 0.6 mg tablets are available, while 0.5 mg tablets are used in other countries.
First day loading dose of 1–2 mg optional but not required.
kg = kilograms; mg = milligrams; TBD = To be determined.
Gastro-intestinal intolerance including diarrhea, nausea, vomiting and abdominal pain is the most common adverse effect, occurring in approximately 10–20% of patients, followed by myalgias; however lower daily doses at 0.5 mg daily or longer term treatment durations over 12 weeks may attenuate significant gastrointestinal intolerance.22 At very high dosage over prolonged periods of time, colchicine may lead to myelosuppression, neuromuscular toxicity, liver damage, and dermatological issues.23 A comprehensive safety analysis of colchicine in 35 double-blind randomized trials including 8659 participants with various inflammatory conditions confirmed known gastrointestinal adverse events (RR 1.74; 95% CI 1.32–2.30) yet did not demonstrate an increase in mortality or rate of adverse events related to the liver, neuropathy, muscle, infection, or hematological disturbances.24 Such findings, however, do not incorporate less-controlled, real-world environments where patients often carry several co-morbidities and take medications which may alter clearance of the therapeutic.
Colchicine is metabolized by the CYP3A4 enzyme and cleared by the P-glycoprotein efflux pump in the bile and kidneys.20 Colchicine may therefore accumulate to toxic levels in patients with chronic kidney disease (CKD) if the dose remains unadjusted, especially if chronically used with acute on chronic kidney insults or worsening kidney function over time. Significant dosage reductions or even avoidance are recommended for hemodialysis patients.25 Furthermore, strong inhibitors of the CYP3A4 and P-glycoprotein efflux pathways such as clarithromycin, certain immunosuppressives, and azole antifungal agents also increase serum or cellular concentrations of colchicine and should be avoided (Figure 2).20, 26 A few cardiac medications including certain calcium channel blockers (verapamil) and antiarrhythmics (amiodarone) may also alter clearance; lower colchicine dose is then advised (typically once daily dosage). Statins are generally well tolerated with colchicine despite their own risk of musculoskeletal effects, although rhabdomyolysis has been reported with combination therapy.27 Fatalities related to colchicine are very rare, and remain limited to suicide attempts or in patients with advanced organ dysfunction in conjunction with very strong P-glycoprotein inhibitors such as clarithromycin.28 A contraindication exists for patients using a potent CYP3A4 or P-glycoprotein inhibitor with either renal or hepatic impairment.
Figure 2. Various CYP-3A4- and P-glycoprotein inhibitors that effect colchicine metabolism.
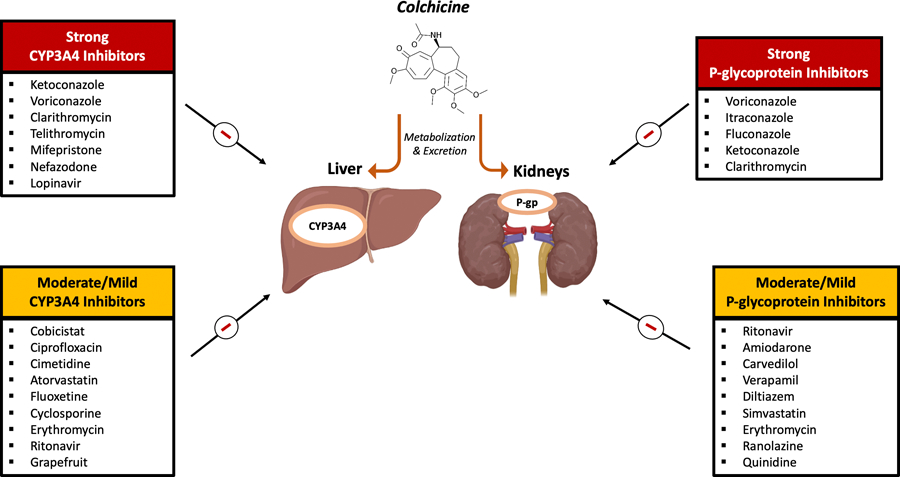
CYP-3A4- and P-glycoprotein inhibitors may alter the liver and kidney’s ability to clear colchicine. Concomitant administration of strong inhibitors with colchicine should generally be avoided, while moderate/mild inhibitors may cautiously be used with colchicine through dose reductions and shared decision making.
P-gp = P-glycoprotein. Figure designed with BioRender.
Colchicine in Pericardial Disease
Acute and Recurrent Pericarditis
Pericarditis arises from inflammation of the pericardium. Given the rich innervation of the pericardium, pericarditis may be associated with disabling or recurrent chest pain. CRP concentrations are often elevated in cases of acute pericarditis and indicate higher chance of recurrence.29 Several clinical trials have been conducted for both acute 30–32 and recurrent pericarditis 33–35 (Table 2) utilizing a colchicine loading dose ranging 1–2 mg followed by 0.5–1 mg daily for subsequent maintenance therapy of variable duration (longer for chronic/recurrent disease). Although all studies were modest in size without more than a few hundred participants, findings have been mostly favorable, apart from a contradicting small study.32 Most studies, however, focused on isolated pericarditis; hence, data regarding pericarditis with concurrent myocardial injury are scarce. Colchicine is currently recommended by the European Society of Cardiology guidelines (ESC, class I recommendation – level of evidence A) as a first-line treatment for both acute and recurrent pericarditis and may be administered with conventional anti-inflammatory regimens (aspirin or non-steroidal anti-inflammatory drugs).6 The American College of Cardiology/American Heart Association (ACC/AHA) guidelines thus far only recommend colchicine for pericarditis after myocardial infarction (MI) (class IIb recommendation – level of evidence C).5 Colchicine treatment is not guided by inflammatory markers nor by symptoms but is typically given for 1–3 months in acute pericarditis and at least 6 months in recurrent pericarditis.
Table 2.
Summary of key studies investigating colchicine as adjunct in inflammatory pericardial disease
| STUDY | DESIGN | POPULATION | INTERVENTION | CONTROL | PRIMARY OUTCOME | COLCHICINE EFFECT | NOTABLE ADVERSE EVENTS* |
|---|---|---|---|---|---|---|---|
| ACUTE PERICARDITIS | |||||||
|
| |||||||
|
Colchicine in addition to conventional therapy for acute pericarditis: results of the COlchicine for acute PEricarditis (COPE)
Imazio et al, 2005 30 |
Prospective, randomized, open-label | Acute Pericarditis (n = 120) |
Colchicine 1.0 to 2.0 mg for the first day and then 0.5 to 1.0 mg/d for 3 months | Conventional treatment | Recurrence Rate at 18 months | 10.7% vs. 32.3%; p=0.004 | Gastrointestinal intolerance (8.3%) |
|
A randomized trial of colchicine for acute pericarditis (ICAP) Imazio et al, 2013 31 |
Prospective, randomized, double-blind, placebo controlled | Acute Pericarditis (n = 240) |
Colchicine 0.5 mg BID for 3 months for patients weighing >70 kg; 0.5 mg QD for patients weighing ≤70 kg |
Placebo | Incessant or recurrent pericarditis at 18 months | 16.7% vs. 37.5%; p<0.001; RRR 0.56 [95% CI 0.30–0.72] |
None |
|
Colchicine Administered in the First Episode of Acute Idiopathic Pericarditis: A Randomized Multicenter Open-label Study
Sambola et al, 2019 32 |
Prospective, randomized, open-label | Acute Pericarditis (n = 110) |
Colchicine 1 mg BID for 3 months for patients weighing >70 kg; 0.5 mg BID for patients weighing ≤70 kg |
Conventional treatment | Recurrence Rate at 24 months | 10.9% vs. 13.5%; p=0.34; HR 1.53 [95% CI 0.7–3.4] |
Gastrointestinal intolerance (13.5%) |
|
| |||||||
| RECURRENT PERICARDITIS | |||||||
|
| |||||||
|
Colchicine as first-choice therapy for recurrent pericarditis: results of the CORE (COlchicine for REcurrent pericarditis) trial
Imazio et al, 2005 33 |
Prospective, randomized, open-label | Recurrent Pericarditis (n = 83) |
Colchicine 1.0–2.0 mg the first day and then 0.5–1.0 mg QD for 6 months | Conventional treatment | Recurrence Rate at 20 months | 24.0% vs 50.6%; p=0 .02; | Gastrointestinal intolerance (7%) |
|
Colchicine for recurrent pericarditis (CORP): a randomized trial.
Imazio et al, 2011 34 |
Prospective, randomized, double blind, placebo controlled | Recurrent Pericarditis (n = 120) |
Colchicine 1.0–2.0 mg on the first day followed by a maintenance dose of 0.5 to 1.0 mg QD, for 6 months. | Conventional treatment | Recurrence Rate at 18 months | 24% vs. 55%; p<0.001; ARR 0.31 [95% CI 0.13–0.46]; RRR 0.56 [95% CI 0.27–0.73] |
Gastrointestinal intolerance (7%) |
|
Efficacy and safety of colchicine for treatment of multiple recurrences of pericarditis (CORP-2): a multicentre, double-blind, placebo-controlled, randomised trial. Imazio et al, 2014 35 |
Prospective, randomized, double-blind, placebo controlled | Recurrent Pericarditis (n = 240) |
Colchicine 0.5 mg BID for 6 months for patients weighing more than 70 kg or 0.5 mg QD for patients weighing 70 kg or less | Conventional treatment | Recurrence rate at 18 months | 21.6% vs. 42.5%; p=0.0009; RR 0.49 [95% CI 0.24–0.65]; |
Gastrointestinal intolerance (7.5%) Hepatotoxicity (2.5%) |
|
| |||||||
| POST-PERICARDIOTOMY SYNDROME | |||||||
|
| |||||||
|
Colchicine for the prevention of postpericardiotomy syndrome Finkelstein et al, 2002 38 |
Prospective randomized, double-blind, placebo controlled | Cardiac Surgery (n = 163) |
POD 3: Colchicine 1.5 mg QD for 1 month | Placebo | PPS at 3 months | 10.6% vs. 21.9%; p<0.135 | Not reported |
|
COlchicine for the Prevention of the Post-pericardiotomy Syndrome (COPPS): a multicentre, randomized, double-blind, placebo-controlled trial Imazio et al, 2010 39 |
Prospective, randomized, double-blind, placebo controlled | Cardiac Surgery (n = 336) |
POD 3: Colchicine 1 mg BID for one day, followed by 0.5mg BID until 1 month (dose reduced for </=70kg) | Placebo | PPS at 12 months | 8.9% vs. 21.1%; p=0.002; RRR 0.579 [95% CI 0.273–0.756] | Gastrointestinal intolerance (8.9%) |
|
Colchicine for prevention of post-pericardiotomy syndrome and postoperative atrial fibrillation: the COPPS-2 randomized clinical trial Imazio et al, 2014 40 |
Prospective, randomized, double blind, placebo controlled | Cardiac surgery (n = 360) |
Colchicine 0.5 mg BID (0.5mg daily if <70kg), 48–72hrs pre-operatively until 1 month post-operatively | Placebo | PPS at 3 months | 19.4% vs.29.4%; Absolute Difference 10.0% [95% CI 1.1%−8.7%] | Gastrointestinal intolerance (14.4%) |
Notable adverse events include events significantly increased compared to the control group. If no comparison was made, events were included that were deemed of interest.
Non-significant PPS reduction was observed in the colchicine arm
ARR = Absolute risk reduction; BID = twice daily; HR = Hazard ratio; NNT = Number needed to treat; POD = Post-operative day; PPS= Post-Pericardiotomy Syndrome; QD = daily; RR = Relative risk; RRR = Relative risk reduction.
Dressler’s Syndrome
Late post-MI pericarditis (or Dressler’s) syndrome is typically diagnosed two to eight weeks after acute MI and is believed to be immune-mediated. In the contemporary era of primary revascularization, it occurs infrequently (0.1–0.5%).36 Expectedly for a rather rare condition, data from RCTs are lacking. Extrapolating from studies in pericarditis, both ESC (class IIa recommendation – level of evidence B) and ACC/AHA guidelines (class IIb recommendation – level of evidence C) include colchicine in the recommended therapeutic regimen for Dressler’s syndrome (in combination with aspirin), dosed in a similar manner as for pericarditis.5, 6
Post-Pericardiotomy Syndrome
Post-pericardiotomy syndrome (PPS) is an immune-mediated pericardial inflammatory syndrome encountered after surgery involving the pericardium, similar to Dressler’s. PPS should be differentiated from isolated pericardial- or pleural effusion seen after cardiac surgery, and specific diagnostic criteria exist.37 Colchicine should be considered for PPS treatment according to ESC guidelines (class IIa – level of evidence B) using the pericarditis treatment regimens.6 The role of colchicine in the prevention of PPS has been evaluated in three double-blinded placebo-controlled RCTs (Table 2), involving 859 patients in total using colchicine 0.5–1.5 mg daily for one month post-operatively. One small study (n = 163) observed a numerically, but not significantly, lower rate of PPS with colchicine versus placebo (11% vs. 17%).38 Two somewhat larger trials including 336 and 360 patients showed a significant effect of colchicine on PPS prevention (9% vs. 21%; and 19% vs. 29%, respectively).39, 40 Hence, ESC guidelines indicate that colchicine should be considered for PPS prevention (class IIa – level of evidence A) for one month post cardiac surgery.6 Of note, a recent randomized trial (n = 197) focused specifically on patients with a non-inflammatory, moderate to large-sized pericardial effusion post cardiac surgery demonstrated that delayed administration of colchicine 7–30 days after surgery did not reduce effusion size or prevent cardiac tamponade, indicating that early administration of the therapeutic may be of particular importance.41
Colchicine in Atrial Fibrillation
Though inflammatory cytokine signaling is known to be elevated in AF, one study suggests it is the activation of the NLRP3 inflammasome that plays a key role in the secretion of the inflammatory cytokines associated with the occurrence of AF.42 Increases in inflammatory cytokines not only promote ectopic firing of the atria, but also stimulates myocyte remodeling and fibrosis that leads to more permanent states of arrhythmia.43
Post-Operative Atrial Fibrillation
Post-operative atrial fibrillation (POAF) occurs in around 0.8% of patients undergoing non-cardiac surgery and 16% following cardiac surgery, and is associated with significant morbidity, including risk of embolic stroke, increased length of stay and higher health care costs.44 Higher post-operative levels of inflammatory markers such as IL-6 and CRP are associated with POAF, and colchicine may suppress POAF through a variety of pathways including the NLRP3 inflammasome cascade which can promote AF.43
A meta-analysis of the three largest double-blind RCTs investigating the role of colchicine in the prevention of POAF in 912 patients found that peri-operative colchicine reduced the incidence of AF by 35% (RR 0.65; 95% CI 0.46–0.91) over a follow-up period of one to six months (Table 3). 40, 45–47 Two subsequent studies did not show a reduced incidence of POAF after surgery with colchicine treatment. These two studies, however, had a smaller, open-label design (n = 140 and n = 152, respectively) with only short-term follow-up period (up to hospital discharge).48, 49 Furthermore, all above studies used varying dosing regimens and durations (Table 3), thereby complicating between-trial comparisons. ACC/AHA guidelines suggest colchicine can be considered for post-operative AF prevention (class IIb - level of evidence B) while ESC guidelines do not include colchicine treatment post-operatively.50, 51 The ongoing Colchicine For The Prevention Of Perioperative Atrial Fibrillation In Patients Undergoing Thoracic Surgery (COP-AF; NCT03310125) investigates the efficacy of short-term post-operative colchicine administration for the prevention of POAF: target enrollment of 2,800 patients with a primary outcome of POAF at 14 days.
Table 3.
Summary of key studies investigating colchicine as adjunct in post-procedural atrial fibrillation
| STUDY | DESIGN | POPULATION | INTERVENTION | CONTROL | PRIMARY OUTCOME | COLCHICINE EFFECT | NOTABLE ADVERSE EVENTS* |
|---|---|---|---|---|---|---|---|
| POST-OPERATIVE ATRIAL FIBRILLATION | |||||||
|
| |||||||
|
Colchicine reduces postoperative atrial fibrillation: results of the Colchicine for the Prevention of the Postpericardiotomy Syndrome (COPPS) atrial fibrillation substudy Imazio et al, 2011 45 |
Prospective, randomized, double blind, placebo controlled | Any cardiac surgery (n = 336) |
Colchicine 1 mg BID on day 3 post-operatively, followed by 0.5mg BID for 1 month (dose reduced for </=70kg) | Placebo | Post-operative AF at 1 months | 12.0% vs. 22.0%, p=0.021; RRR 0.45 [95% CI 0.34–0.94]; |
Gastrointestinal intolerance (9.5%) |
|
Colchicine for prevention of post-pericardiotomy syndrome and postoperative atrial fibrillation: the COPPS-2 randomized clinical trial Imazio et al, 2014 40 |
Prospective, randomized, double blind, placebo controlled | Any cardiac surgery (n = 360) |
Colchicine 0.5 mg BID (0.5mg daily if <70kg) from 48–72 hours prior to surgery then 1 month post-operatively | Placebo | PPS (Secondary outcome- Post-operative AF) at 3 months |
33.9% vs. 41.7%; Absolute Difference 7.8% [95% CI −2.2%−17.6%] | Gastrointestinal intolerance (14.4%) |
|
Low dose Colchicine in prevention of atrial fibrillation after coronary artery bypass graft: a double blind clinical trial Sarzaeem et al, 2014 46 |
Prospective, randomized, blinded, placebo controlled | Coronary artery bypass surgery (n = 216) |
Colchicine 1 mg BID 2 doses prior to surgery then 0.5mg BID for 5 days |
Placebo | Post-operative AF at 6 months | 14.8% vs. 30.6%; p=0.006 | Not reported |
|
Colchicine to Reduce atrial fibrillation in the postoperative period of myocardial revascularization Zarpelon et al, 2016 48 |
Prospective, randomized, open-label | Elective coronary artery bypass surgery (n = 140) |
Colchicine 1 mg BID pre-operatively, then 0.5 mg BID until hospital discharge | No colchicine therapy | Post-operative AF at hospital discharge | 7.04% vs. 13.04%; p=0.271; RRR 0.46 [95% CI −0.53–0.81) |
Not reported |
|
Effect of colchicine on the incidence of atrial fibrillation in open heart surgery patients: END-AF Trial Tabbalat et al, 2020 49 |
Prospective randomized, open-label, placebo controlled | Elective cardiac surgery (n = 152) |
Colchicine 1 mg once 12–24 hours pre-operatively, followed by 0.5 mg QD until hospital discharge | Placebo 12–24 hours pre-operatively, followed by placebo until hospital discharge | Post-operative AF at hospital discharge | 16% vs. 18.3%; p=0.88; OR 0.85 [95% CI 0.37–1.99] |
Gastrointestinal intolerance (2.4%) |
|
| |||||||
| POST-PULMONARY VEIN ISOLATION ATRIAL FIBRILLATION | |||||||
|
| |||||||
|
Colchicine for prevention of early atrial fibrillation recurrence after pulmonary vein isolation: a randomized controlled study Deftereos et al, 2012 52 |
Prospective, randomized, double blind, placebo controlled |
Pulmonary vein isolation (n = 161) |
Colchicine 0.5 mg BID from day 1 post-procedure for 3 months | Placebo | Post-procedure AF at 3 months | 16% vs. 33.5%; p=0.01; OR 0.38 [95% CI 0.18–0.80] |
Gastrointestinal intolerance (8.6%) |
|
Colchicine for prevention of atrial fibrillation recurrence after pulmonary vein isolation: Mid-term efficacy and effect on quality of life Deftereos et al, 2014 53 |
Prospective, randomized, double blind, placebo controlled |
Pulmonary vein isolation (n = 223) |
Colchicine 0.5 mg BID from day 1 post-procedure for 3 months | Placebo | Post-procedure AF at 15 months | 31.1% vs. 49.5%; p=0.01; RRR 0.37; OR 0.46 [95% CI 0.26–0.81] |
Gastrointestinal intolerance (9.7%) |
Notable adverse events include events significantly increased compared to the control group. If no comparison was made, events were included that were deemed of interest AF = Atrial Fibrillation; BID = twice a day; NNT = Number needed to treat; OR = Odds ratio; PPS= Post-Pericardiotomy Syndrome; RRR = Relative risk reduction; QD = daily
Post-Ablation Atrial Fibrillation
AF recurrence following catheter-based radiofrequency ablation pulmonary vein isolation (RFA PVI) procedures has been associated, at least in part, with an ongoing inflammatory process.43 The effect of colchicine in preventing AF recurrence after RFA PVI for paroxysmal AF has only been evaluated in one small double-blinded placebo-controlled trial at 3 months (n = 161) and in an extension study at 12 months follow up (n = 223) (Table 3).52, 53 A 3-month regimen of colchicine 0.5 mg twice daily was associated with decreased post-procedural AF recurrence at both 3 months and 12 months. These findings were accompanied by a reduction in IL-6 and CRP levels and an improvement in self-perceived quality of life indices in the midterm. Evidence thus far is scarce and there are currently no recommendations from scientific society guidelines for post RFA PVI AF, although a consensus document from American and European Scientific Societies recommends the use of colchicine for pericardial symptoms after ablation.54 The ongoing Impact of Short-course Colchicine Versus Placebo After Pulmonary Vein Isolation (IMPROVE-PVI Pilot; NCT04160117) is planning to investigate AF recurrence with 10 days of 0.6mg colchicine per day after catheter ablation for long-term recurrence of AF up to 24 months post RFA-PVI.
Colchicine in Coronary Artery Disease
While the normal coronary endothelium is relatively resistant to adhesion by circulating leukocytes, inflamed endothelium in the setting of atherosclerosis attracts the migration, adhesion, and activation of leukocytes, of which neutrophils make up a large component.55 Subsequent release of neutrophil granular enzymes include matrix metalloproteinases, which play a role in the increased vulnerability of atherosclerotic plaque;56 neutrophil elastase, which cleaves tissue factor pathway inhibitor and restores factor Xa activity, contributing to the generation of thrombin;57 and α-defensins, which are associated with altered fibrin formation, larger lipid- and macrophage-rich plaques, and larger thrombus size.58, 59 Activated neutrophils additionally release neutrophil extracellular traps, externalized nucleosomes with neutrophil enzymes adherent to chromatin that accumulate in rupture-prone plaque and further attract leukocytes and platelets.60 Platelets, in turn, adhere to exposed collagen in ruptured or eroded atherosclerotic plaque and aggregate with other circulating leukocytes.61 Colchicine not only reduces the migration, adhesion, and activation of neutrophils, but may also have a potential downstream impact on the inflammatory/thrombotic interface via a reduction in the release of neutrophil granular enzymes related to thrombosis (e.g., neutrophil elastase and α-defensin), and inhibition of neutrophil-platelet aggregates.62 Secondly, both IL-1β and IL-18 play a role in the development and instability of atherosclerotic plaque and subsequent IL-6/CRP production.16, 17 Colchicine not only reduces the NLRP3 inflammasome-mediated production of IL-1β and IL-18, but also reduces the concentration of neutrophil enzymes (e.g. proteinase 3) that activate these cytokines extracellularly.18 In patients with established coronary artery disease (CAD), colchicine has been shown to incrementally reduce CRP concentrations on a background of aspirin and statin therapy,63 as well as reduce low-attenuation plaque volume on coronary computed tomography angiography over time after acute coronary syndrome (ACS).64 In patients with gout, colchicine has resulted in improved arterial endothelial function with treatment associated reductions in CRP concentrations.20, 65
Direct support of an independent effect of inflammation on atherosclerosis, i.e., the inflammatory hypothesis, came from the large randomized Canakinumab Anti-Inflammatory Thrombosis Outcomes Study (CANTOS).66 Canakinumab, a parenteral anti-IL-1β monoclonal antibody approved for juvenile idiopathic arthritis and Still’s disease, led to a reduction in major adverse cardiovascular events while low density lipoprotein-cholesterol concentrations remained unaffected in patients with prior MI and elevated high-sensitivity CRP. Patients who received canakinumab and achieved on-treatment reductions in IL-6 and high-sensitivity CRP had improved outcomes compared to placebo and to those who did not achieve on-treatment reductions in inflammatory markers.67 However, canakinumab is costly, appeared associated with an increased incidence of fatal infections, yet was also associated with a large reduction in lung cancer mortality; hence, an oncological indication was further pursued by the manufacturer instead. Conversely, another large randomized trial demonstrated no reduction in major adverse events with low-dose methotrexate, an antimetabolite that blocks the alternate adenosine-mediated inflammatory pathway; however, concentrations of IL-1β, IL-6, and CRP also did not change with treatment.68 Taken together, the data support the IL-1β/IL-6/CRP pathway as an important focus of therapeutic investigations in targeting cardiovascular inflammation.
Stable CAD
Colchicine’s effect in CAD was initially examined retrospectively in the gout and FMF populations. An early case-control series of patients with FMF suggested that taking long-term prophylactic colchicine decreased the incidence of CAD compared to their untreated counterparts.69 Colchicine use was also associated with a lower rate of CAD when compared to non-users in a retrospective study of patients with gout; this risk was notably lower in patients without concomitant kidney disease.70 A retrospective analysis of 1,288 patients with gout first hinted that prophylactic long-term colchicine use was associated with a lower rate of MI.71 However, no adjustment for potential confounders was performed, and the optimal timing of colchicine treatment remained unclear. A subsequent retrospective study of 1,002 gout patients also indicated that colchicine was associated with a 49% lower risk of the composite of MI, stroke, or transient ischemic attack and a 73% relative risk reduction in all-cause mortality compared to untreated patients.72 Acknowledging the important limitations that retrospective studies carry, these studies provided a foundation for RCTs investigating colchicine in coronary artery disease (Table 4).
Table 4.
Summary of key studies investigating colchicine as adjunct in atherosclerosis including acute coronary syndrome and stable coronary artery disease
| STUDY | DESIGN | POPULATION | INTERVENTION | CONTROL | PRIMARY OUTCOME | COLCHICINE EFFECT | NOTABLE ADVERSE EVENTS* |
|---|---|---|---|---|---|---|---|
| STABLE CORONARY ARTERY DISEASE | |||||||
|
| |||||||
|
Low-dose colchicine for secondary prevention of cardiovascular disease (LoDoCo trial) Nidorf et al, 2013 73 |
Prospective, randomized, observer-blinded, open-label | Stable CAD (n = 532) |
Colchicine 0.5 mg QD until study completion | Conventional treatment | Composite of ACS, out-of-hospital cardiac arrest, or non-cardioembolic ischemic stroke at median follow up of 3 years | 5.3% vs. 16%; p<0.001; HR 0.33 [95% CI 0.18–0.59] |
Gastrointestinal intolerance (2.5%) Myalgia (0.9%) Myositis (1 case) |
|
Colchicine in Patients with Chronic Coronary Disease (LoDoCo2 trial) Nidorf et al, 2020 74 |
Prospective, randomized, double blind, placebo controlled |
Stable CAD (n = 5,522) |
Colchicine 0.5 mg QD until study completion | Placebo | Composite of CV death, spontaneous (nonprocedural) MI, ischemic stroke, or ischemia-driven coronary revascularization at median follow up of 28.6 months | 6.8% vs. 9.6%; p<0.001; HR 0.69 [95% CI 0.57–0.83] |
Noncardiovascular death (1.9%; HR 1.51 [95% CI 0.99–2.31]) Myalgia‡ (21.2%; cumulative incidence ratio 1.15 [95% CI 1.01–1.31]) Gastrointestinal intolerance (15.4% during run-in period) |
|
| |||||||
| ACUTE CORONARY SYNDROME | |||||||
|
| |||||||
|
Colchicine in Patients With Acute Coronary Syndrome: The Australian COPS Randomized Clinical Trial Tong et al, 2020 79 |
Prospective, randomized, double blind, placebo controlled | Patients hospitalized with ACS (n = 795) |
Colchicine 0.5 mg BID for the first month, then 0.5 mg QD for 11 months | Placebo | Composite of all-cause mortality, ACS, ischemia-driven (unplanned) urgent revascularization, and non-cardioembolic ischemic stroke at 12 months | 6.1% vs. 9.5%; p=0.09; HR 0.65 [95% CI 0.38–1.09] |
Total Death (8 vs. 1; p=0.017) Noncardiovascular death (5 versus 0; p=0.024) |
|
Colchicine for Left Ventricular Remodeling Treatment in Acute Myocardial Infarction (COVERT-MI) Mewton et al, 202180 |
Phase 2, prospective, randomized, double-blind, placebo controlled |
ACS (n = 194) |
Colchicine 2mg bolus followed by Colchicine 0.5mg BID for 5 days |
Placebo | Infarct size by CMR at 5 days | 26 vs. 28.4 grams; p=0.84 | LV Thrombus (22.2% vs. 7.4%; p=0.01)\ Gastrointestinal Intolerance (34.4% vs. 10.1%; p=0.0001) |
|
| |||||||
| POST MYOCARDIAL INFARCTION | |||||||
|
| |||||||
|
Efficacy and Safety of Low-Dose Colchicine after Myocardial Infarction (COLCOT trial) Tardif et al, 2019 82 |
Prospective, randomized, double blind, placebo controlled |
Colchicine 0.5mg QD until study completion | Placebo | Composite of death from CV causes, resuscitated cardiac arrest, MI, stroke, or urgent hospitalization for angina leading to coronary revascularization at median follow up of 22.6 months | 5.5% vs. 7.1%; p=0.02; HR 0.77 [95% CI 0.61–0.96] | Pneumonia (0.9% vs. 0.4%; p=0.03) | |
Notable adverse events include events significantly increased compared to the control group. If no comparison was made, events were included that were deemed of interest
Higher rate of non-cardiovascular death in the colchicine group, longer follow-up results still awaited.
Only reported among the cohort from the Netherlands
ACS = acute coronary syndrome; BID = twice daily; CAD = coronary artery disease; CV = cardiovascular; g = grams; HR = Hazard ratio; MI = myocardial infarction; QD = once daily
The Low-Dose Colchicine (LoDoCo) study was an open-label pilot trial (n = 532) studying low-dose colchicine in patients with angiographically proven CAD who were clinically stable for at least 6 months on optimal medical therapy.73 At a median 3-year follow up, the colchicine group had a significantly lower composite rate of ACS, out-of-hospital cardiac arrest or non-cardioembolic ischemic stroke (5.3% vs 16%; p<0.001). Due to the small size and lack of placebo control in this study, larger studies were warranted to confirm such positive findings in this pilot study. Subsequently, the larger LoDoCo2 trial was designed as a multi-center, double-blind placebo-controlled randomized trial clinically stable, optimally treated patients from Australia and the Netherlands with documented CAD.74 The investigators implemented a 30 day open-label run-in period of colchicine treatment for 6,582 patients initially enrolled, during which ~10% withdrew due to perceived side effects (mostly gastrointestinal). A total of 5,522 patients eventually underwent randomization. Although focused on stable CAD patients, most patients (85%) randomized had a history of ACS, and almost all were treated with statins (94%) and other optimal guideline directed therapy. At a median follow-up of 29 months, colchicine reduced the risk of the primary composite cardiovascular endpoint of cardiovascular death, MI, ischemic stroke or ischemia-driven coronary revascularization by 31% compared to placebo (6.8% vs. 9.6%; p< 0.001) driven mostly by the occurrence of spontaneous MI and ischemia-driven revascularization. There were no significant mortality differences between groups, although there was a non-significant increase in non-cardiovascular death noted in the colchicine arm compared to placebo (53 vs. 35 events, respectively; HR 1.51; CI 95% 0.99–2.31). No potential etiology of such non-cardiovascular deaths, including causes as infection or cancer, were able to explain illustrated differences. Furthermore, there were no observed differences in hospitalizations due to infection, pneumonia or gastrointestinal reasons between the colchicine and placebo groups. There was a higher rate of myalgia with the colchicine group compared to placebo (21.2% vs. 18.5%; HR 1.15; 95% CI 1.01–1.31; data on myalgias were only collected in the Netherlands cohort). Within pre-specified sub-group analyses the effects of colchicine on the primary endpoint were generally consistent, with important exceptions including females or patients with moderate CKD stage 3A (although such sub-groups had lower power, representing 15.3% and 5.5% of the total population, respectively). There was also a region dependent variation of treatment effect, larger in Australia (HR 0.51; 95% CI 0.39–0.67) compared to the Netherlands (HR 0.92; 95% CI 0.72–1.20). As geographical baseline characteristics were similar, this finding may have been rooted in chance but should call for further investigation. Finally, a subgroup analysis of LoDoCo2 confirmed the benefit of colchicine treatment in stable CAD patients regardless of the history and timing of a prior ACS within the patient population.75 Altogether, the LoDoCo2 trial served as a strong validation for the benefit colchicine may carry in stable atherosclerotic coronary disease.
ACS
Colchicine also has been investigated immediately after ACS. Preliminary in vivo data illustrated how pharmacological inhibition of the NLRP3 inflammasome with colchicine could attenuate MI reperfusion injury and limit infarct size in mouse models.76 A pilot study (n = 151, n = 60 in the MRI sub-study) illustrated how colchicine may also reduce the levels of cardiac biomarkers and late-gadolinium-enhancement-defined infarct size when administered within 12 hours of symptom onset in patients with ST-elevation MI.77 The following double-blind pilot-study of Low Dose Colchicine after Myocardial Infarction (LoDoCo-MI) however did not observe a significant difference in high-sensitivity CRP levels among 237 patients admitted with an acute MI after 30 days of treatment with daily low-dose colchicine (0.5 mg daily) compared to placebo.78
Larger randomized studies were necessary to adequately investigate colchicine’s effect after ACS. In the Colchicine in Patients with Acute Coronary Syndromes (COPS) study (n = 795), colchicine was started during index hospitalization of ACS, with a higher dose of colchicine (0.5 mg twice daily) compared to LoDoCo2 during the first month.79 The composite primary endpoint of all-cause mortality, ACS, ischemia-driven (unplanned) urgent revascularization, and noncardioembolic ischemic stroke did not differ significantly between colchicine and control arms after 12 months of low-dose colchicine following ACS (p=0.09). However, in a post hoc analysis using a primary outcome similar to the LoDoCo and LoDoCo2 trials (a composite of cardiovascular death, ACS, stroke, and urgent revascularization; excluding non-cardiovascular death), there was a significant benefit found within the colchicine group (p=0.019). Notably, COPS did illustrate a statistically significant increase in non-cardiovascular deaths in the colchicine arm, with 5 and 0 in the colchicine and control arms, respectively (p=0.024). The cause of death was related to sepsis in 4 out of the 5 events, although of note is that most of these patients (3 out of 5) had discontinued colchicine within the first 30 days of the trial and were not taking colchicine at the time of death. Longer-term follow up of the COPS trial is currently being collected, and results should further clarify the effect of colchicine in CAD and whether this statistical difference in non-cardiovascular death upholds.
The Colchicine for Left Ventricular Infarct Size Reduction in Acute Myocardial Infarction (COVERT-MI) multicenter study (n = 192) investigated colchicine’s effect targeting the initial inflammatory response that occurs with reperfusion injury in ACS.80 Patients admitted for PCI after a first episode of ST segment-elevation myocardial infarction were randomized to five days of high dose colchicine (2mg loading dose followed by 0.5 mg twice daily) or placebo. In contrast to the earlier pilot study77, no differences were demonstrated between groups in the primary outcome of gadolinium-enhanced infarct size on cardiac magnetic resonance imaging (p = 0.87), in biomarkers of inflammation (including CRP), or myocardial injury (at 6, 24, and 48 hours follow up). There were furthermore no differences in longer term secondary outcomes of infarct size and left ventricle (LV) remodeling (defined as change in LV end-diastolic volume) at 3 months follow up. Notably, the study demonstrated a higher number of patients with LV thrombus in the colchicine group (18) than in the placebo group (6) at 5 days, a difference that was no longer observed at 3 months. There was no significant difference in the number of embolic events. There have not been any pro-thrombotic reports of colchicine previously, and given the small sample size, the increased incidence of LV thrombus may again be due to chance.
While the LoDoCo trials illustrated a significant benefit of low dose colchicine in patients with stable CAD, the therapeutic has yet to illustrate benefit during index hospitalization for ACS. This may be due to differences in ACS pathophysiology, proposed to feature a series of phenotypes not all of them being inflammation dependent.81 Variations in trial design including the dosing and timing of colchicine administration may also account for disparities. Ongoing studies should further explore such differences.
Post Myocardial Infarction
The large double-blind, placebo-controlled COLchicine Cardiovascular Outcomes Trial (COLCOT) tested low-dose colchicine in 4,745 patients randomized within 30 days after MI.82 Treatment was started with a median of 14 days after stabilization from initial MI. The majority of patients were on established optimal treatment including a statin (98–99%). Median follow-up was23 months. Colchicine significantly reduced the primary composite endpoint of cardiovascular death, resuscitated cardiac arrest, MI, stroke, or urgent hospitalization for angina requiring coronary revascularization by 23% (HR 0.77; 95% CI 0.61–0.96; p=0.02). The benefit of colchicine was most pronounced in the components of urgent hospitalizations for angina requiring revascularization (HR 0.50; 95% CI 0.31–0.81) and stroke (HR 0.26; 95% CI 0.10–0.70). Colchicine was also associated with a 34% reduction in the total number of both first and recurrent primary endpoint events during follow-up (rate ratio 0.66; 95% CI 0.51–0.86). The therapeutic was well tolerated with minimal adverse events, although there was a small increase in hospitalization for non-fatal pneumonia in the treatment group versus control (0.9% vs. 0.4%; p=0.03). COLCOT has been the only trial powered enough to investigate improved outcomes from colchicine within a month of an MI.
Pooled Efficacy, Guidelines, and Adverse Effects
Pooled data from these large RCTs further emphasize the value that colchicine carries in CAD. A 2020 meta-analysis pooling data from the above COLCOT, COPS, LoDoCo and LoDoCo2 trials included a total of 11,594 patients and showed that, compared to placebo, colchicine was associated with significant reductions of 32% in the incidence of the composite of cardiovascular mortality, MI, ischemic stroke, and urgent coronary revascularization, 38% for MI, 62% for stroke, and 44% for urgent coronary revascularization.83 There were no significant differences in mortality endpoints or other adverse events as hospitalization for gastrointestinal events, infection, or pneumonia; although there was a numerical non-significant increase in non-cardiovascular death in colchicine treated patients (RR 1.38; 95% CI 0.99–1.93). Another meta-analysis from 2021 illustrated similar results with addition of a randomized trial investigating colchicine after bare metal stent implantation in diabetic patients (discussed below).84, 85 Importantly, neither meta-analysis established significant drug-drug interactions with colchicine and aspirin, statins, or other guideline-directed cardiovascular therapeutics. Based on notable RCTs as COLCOT and LoDoCo2, 2021 ESC guidelines on the prevention of cardiovascular disease have recommended colchicine be considered for the secondary prevention of cardiovascular disease, in particular for those with uncontrolled risk factors and recurrent cardiac events despite optimal medical therapy (class IIb recommendation – level of evidence A).86 Health Canada has since also approved low-dose colchicine for the reduction of atherothrombotic events in patients with existing coronary disease.87
Signals of a non-cardiovascular mortality effect nonetheless caution the benefit demonstrated in colchicine treatment for atherosclerosis.74, 79 Such concerns are new and have not been illustrated during treatment for approved indications such as gout and FMF, and thus far, no clear biological root-cause for the non-cardiovascular deaths observed has been determined. Although the increased incidence of sepsis illustrated in the non-cardiovascular deaths of COPS may raise concern considering the potential immunosuppressive effect of colchicine, the majority of treated patients had discontinued the medication long before their fatal event. Higher doses of colchicine in the initial month could have also affected outcomes, and if so, it would be pertinent to further investigate this concern. Additionally, non-cardiovascular deaths in LoDoCo2 were unable to be attributed to a specific cause such as infection or cancer. Given the low number of events and wide confidence intervals, observed differences in non-cardiovascular mortality may be attributed to limited power and play of chance. Nonetheless, continued pharmacovigilance is advisable. Upcoming randomized trials should increase power with longer follow up and improve quality of evidence with on-treatment analyses in order to further understand the nuances of such mortality signals.
Colchicine in Percutaneous Coronary Intervention
Percutaneous coronary intervention (PCI) involves an induced endovascular injury and an increased local inflammatory response.88 Initial studies focused on the prevention of restenosis since colchicine may target inflammatory factors and halt local smooth muscle cell proliferation (Table 5). However, two early studies using daily colchicine after balloon angioplasty, in 130 and 50 patients respectively, failed to show any impact on restenosis.89, 90 Post-angioplasty restenosis is largely driven by arterial elastic recoil and remodeling, while in-stent restenosis is mainly driven by neointimal hyperplasia and local inflammation. Such in-stent restenosis processes may be more effectively targeted by colchicine. A subsequent randomized trial examined the effect of 6 months of treatment with colchicine on neointima formation and restenosis in 196 diabetic patients after bare-metal stent implantation.85 The intravascular-ultrasound-defined restenosis rate and in-stent lumen area loss were significantly lower in the colchicine arm. Given these early positive results, the ongoing Oral Colchicine in Argentina to Prevent Restenosis (ORCA; NCT04382443) trial is comparing bare-metal stent plus colchicine versus drug-eluting stent alone with a planned enrollment of 450 patients. If comparable, adjunctive colchicine with bare metal stent implantation may offer a safe alternative for those with a contraindication to drug-eluting stents.
Table 5.
Summary of key studies investigating colchicine as adjunct in percutaneous coronary interventions
| STUDY | DESIGN | POPULATION | INTERVENTION | CONTROL | PRIMARY OUTCOME | COLCHICINE EFFECT | NOTABLE ADVERSE EVENTS* |
|---|---|---|---|---|---|---|---|
| PERCUTANEOUS CORONARY INTERVENTION | |||||||
|
| |||||||
|
Ineffectiveness of colchicine for the prevention of restenosis after coronary angioplasty
O’Keefe et al, 1992 89 |
Prospective, randomized, double blind, placebo controlled | PTCA (n = 130) |
Colchicine 0.6 mg BID until study completion | Placebo | Restenosis at 6 months | 46% vs. 45%; p=non-significant | Gastrointestinal intolerance (28%) |
|
Combination of lovastatin, enalapril, and colchicine does not prevent restenosis after percutaneous transluminal coronary angioplasty Freed et al, 1995 90 |
Prospective, open label |
PTCA (n = 50) |
Colchicine 0.6 mg BID + Lovastatin 20 mg QD + Enalapil 2.5–10 mg BID (titrated to SBP >100mmHg) + Aspirin 81 mg QD until study completion | N/A | Late loss in lumen diameter at 16 weeks | 0.5 mm ± 0.8mm† | Gastrointestinal intolerance (18%) |
|
Colchicine treatment for the prevention of bare-metal stent restenosis in diabetic patients Deftereos et al, 2013 85 |
Prospective, randomized, double blind, placebo controlled | BMS PCI in Diabetics (n = 196) |
Colchicine 0.5 mg BID until study completion | Placebo | Angio- and IVUS-ISR at 6 months | Angio-ISR: 16% vs. 33%; p=0.007; OR 0.38 (95% CI 0.18–0.79); IVUS-ISR: 24% vs. 43%; p=0.006; OR 0.42 [95% CI 0.22–0.81]; |
Gastrointestinal intolerance (16% vs. 7%; p=0.058) |
|
Effects of Acute Colchicine Administration Prior to Percutaneous Coronary Intervention: COLCHICINE-PCI Randomized Trial
Shah et al, 2020 94 |
Prospective, randomized, double blind, placebo controlled | ACS or Stable CAD PCI (n = 400) |
Colchicine 1.2 mg once prior to PCI, and 0.6 mg once after PCI | Placebo at matching timepoints | PCI-related myocardial injury at 6 to 8 hours and 22 to 24 hours post-PCI | 57.3% vs. 64.2%; p=0.19 | Gastrointestinal intolerance (9.3% vs. 3.2%; p=0.001) |
|
Colchicine to Prevent Periprocedural Myocardial Injury in Percutaneous Coronary Intervention: The COPE-PCI Pilot Trial Cole et al, 2021 95 |
Prospective, randomized, double-blind, placebo controlled | NSTEMI or Stable Angina (n = 75) |
Colchicine 1mg followed by 0.5mg 1 hour later, 6–24 hours pre-procedure |
Placebo at matching timepoints | Major and minor periprocedural myocardial injury at 24 hours post-PCI | Minor: 58% vs. 85%; p=0.01 Major: 31% vs. 54%; p=0.04 |
None |
Notable adverse events include events significantly increased compared to the control group. If no comparison was made, events were included that were deemed of interest
Indicating no prevention of restenosis
BID = twice daily; BMS = Bare metal stent; ISR = In stent restenosis; IVUS = Intravascular ultrasound; PCI = Percutaneous Coronary Intervention; PTCA = Percutaneous Coronary Angioplasty; MI = Myocardial infarction; OR = Odds ratio; QD = daily; TVR = Target Vessel Revascularization
Interestingly, elevations in inflammatory markers may be detectable as early as one hour after PCI and peri-procedural inflammation has been associated with short- and long-term major adverse cardiovascular events even in the contemporary era.91, 92 Given that a loading dose of high-intensity statin reduces PCI-related MI, colchicine’s effect was also examined recently as a “prophylactic” regimen initiated in conjunction with PCI.93 The double-blind Colchicine-PCI randomized trial included 400 patients with either ACS or stable CAD treated with either colchicine (1.2 mg prior and 0.6 mg after PCI) versus placebo one to two hours pre-procedure.94 There were no significant differences in the primary outcome of type 4 MI/injury (57.3% vs. 64.2%; p=0.19) nor any other clinical outcomes at 30-day follow up. However, the rise in inflammatory markers (IL-6 and high-sensitivity CRP) was significantly dampened in the colchicine compared to placebo group at approximately 24 hours post-PCI. The subsequent double-blind COlchicine to Prevent PeriprocEdural myocardial Injury in PCI (COPE-PCI) trial demonstrated a significant reduction in periprocedural MI when administering a loading dose of colchicine earlier (1 mg followed by 0.5 mg one hour later, 6–24 hours pre-PCI) in 75 patients with non-ST elevation myocardial infarction and stable angina undergoing PCI.95 The biomarker analyses of Colchicine-PCI and COPE-PCI are forthcoming. Considering the limited sample sizes thus far in PCI trials and their mixed results, these results should only be considered as hypothesis generating.
Colchicine in Cerebrovascular Disease
Atherosclerotic cerebrovascular disease accounts for half of all ischemic strokes, whether due to large artery disease (e.g. carotid, proximal middle cerebral artery) or small vessel disease (deep perforating arteries). Despite current optimal medical therapy that includes antihypertensive agents, antithrombotic agents, and lipid-lowering therapy, the annualized risk of recurrent stroke or transient ischemic attack after initial event is up to 5%.96 In those with known large artery atherosclerosis, the risk is doubled. Therefore, new therapeutic approaches to prevent atherosclerotic stroke are warranted, particularly targeting the inflammatory component, which has been relatively neglected thus far despite data showing the link between plaque inflammation and higher risk of stroke recurrence.97
Initial data suggest colchicine reduces risk of stroke in high-risk populations. After recent ACS, COLCOT showed a markedly lower risk of stroke in the colchicine group compared to the placebo group (HR 0.26; 95% CI 0.10–0.70).82 Multiple meta-analyses have thus far reported impressive stroke reductions associated with colchicine use compared to placebo, which has led to many ongoing prospective investigations.83, 84 There are currently two larger randomized trials examining the effects of colchicine in stroke survivors. The ongoing open-label Colchicine for prevention of vascular inflammation in non-cardio embolic stroke trial (CONVINCE; NCT02898610) will assess its effects in patients who have recently suffered a non-cardioembolic transient ischemic attack or ischemic stroke, with a planned recruitment of 2,623. The second study is the planned Australian-based multi-center Colchicine After Stroke to Prevent Event Recurrence (CASPER) trial, which will test the effects of adding colchicine to optimal medical therapy in order to reduce major adverse cardiovascular events in stroke survivors with a persistently elevated high-sensitivity CRP of >2 mg/L. Thus far evidence of colchicine in stroke is only preliminary, however if confirmed by above larger randomized trials, stroke may be only one of a number of clinical manifestations of inflammatory cerebrovascular atherosclerotic processes that may be targeted with anti-inflammatory therapies such as colchicine.
Ongoing and Future Trials of Colchicine in Atherosclerosis
Although the above CAD studies included patients with peripheral artery disease (PAD), no studies have specifically examined the effects of colchicine on chronic PAD, another partly inflammatory process. Given promising results with colchicine in CAD, further investigation is anticipated in patients with PAD and peripheral percutaneous arterial interventions. In addition, there are other large studies ongoing that will further add to our understanding of colchicine in atherosclerosis. The ongoing, placebo-controlled, randomized trial Colchicine and Spironolactone in Patients With MI / SYNERGY Stent Registry (CLEAR SYNERGY; NCT03048825) has a 2×2 factorial design and plans to enroll 7,000 participants with large MI undergoing primary PCI. It will include investigation of low-dose colchicine initiated within 3 days of PCI on the composite primary endpoint of cardiovascular death, MI, or stroke compared with placebo up to 5 years of follow-up. The COLchicine effects on Cardiovascular Outcomes in Acute Coronary Syndrome Study (COLCARDIO-ACS, ACTRN12616000400460) plans to recruit 3,000 ACS survivors with a persistently elevated high sensitivity CRP (>2 mg/L 4–6 weeks post-event) in a prospective, randomized, placebo-controlled trial.
Clinical Implications
Already standard-of-care for pericarditis, colchicine’s possible role is expanding to other areas of cardiovascular inflammation in a stepwise fashion. Examining the expanding body of evidence of colchicine in cardiovascular disease, we can summarize the following: 1. Although not FDA approved per se, the use of colchicine for pericarditis has strong evidence and is currently recommended by the practice guidelines; 2. colchicine can effectively decrease AF and PPS after cardiac surgery, however additional RCTs are warranted to determine colchicine’s effect after RFA-PVI; 3. recent large, well designed RCTs and meta-analyses have illustrated substantial benefit of colchicine in CAD; 4. larger RCTs should further examine the use and timing of colchicine in relation to PCI after mixed preliminary evidence; and 5. retrospective analyses indicate a possible indication for colchicine in atherosclerotic cerebrovascular disease, and ongoing prospective studies are eagerly awaited.
Colchicine’s dosing, timing, and duration of administration should continue to be clarified for each condition treated (Table 1). Although higher doses are used in gout and FMF, randomized cardiovascular trials used low-dose daily colchicine with success in secondary prevention.74, 82 Given its dose-dependent side effect profile, higher doses should only be used short-term in acute cardiovascular conditions (i.e. acute pericarditis, acute POAF, etc.), while a lower dose regimen appears preferable for longer term treatment exceeding ~1 week. Particularly for indications as chronic CAD or post MI, colchicine can be taken in conjunction with other optimal secondary prevention medical therapy for years given periodic evaluations of tolerability and shared physician-patient decision making. This may especially serve those with uncontrolled cardiovascular risk factors and recurrent cardiovascular events, as indicated by the 2021 ESC guidelines.86 Additionally, given the suggested benefits of long-term colchicine administration in CAD, longer-term colchicine administration may be further examined for many other parallel cardiovascular indications, as after PCI or coronary artery bypass graft surgery. Accumulating evidence in the various cardiovascular indications will likely further help elucidate optimal timing and use.
Target populations that may benefit from colchicine need yet to be further delineated. Neither LoDoCo trials or COLCOT selected patients based on elevated inflammatory risk, currently defined as high-sensitivity CRP >2 mg/L.73, 74, 82 Indeed, after excluding patients with highly elevated high-sensitivity CRP concentration (>10 mg/L), a substudy of LoDoCo2 (n=174) demonstrated a median baseline high-sensitivity CRP concentration of 1.52 mg/L, which was significantly reduced 34% with colchicine treatment.98 Although a subgroup of COLCOT (n=207) demonstrated an elevated median baseline high-sensitivity CRP concentration of 4.28 mg/L early after MI, reductions in this inflammatory marker did not differ over time by treatment group.82 Colchicine, therefore, may improve clinical outcomes irrespective of CRP concentrations and could be considered for all patients with CAD . However, a biomarker-specific strategy may provide a targeted patient subset that may enable precision medicine and amplify the benefit/risk ratio. Notably, the 2020 proteomic study of 174 colchicine-treated patients nested within the 30 day run-in phase of LoDoCo2 not only demonstrated a significant reduction in high-sensitivity CRP with colchicine treatment, but reductions in various proteins associated with NLRP3 inflammasome (i.e IL-1 β, IL-6, and IL-18) and others related to neutrophil function as well.98 Many of these protein reductions, however, did not necessarily correlate with change in high-sensitivity CRP, and we should therefore continue to scrutinize our current definition of high inflammatory risk. The ongoing biomarker substudy of CLEAR SYNERGY (NCT03874338) plans to examine detailed neutrophil profiles and determine clinical, biomarker, and genetic predictors of heterogeneity to colchicine treatment response in survivors of a large MI. Certain pharmacogenomic determinants of colchicine safety have already been identified in COLCOT.99 Further studies investigating the role of inflammatory markers and clinical outcomes in parallel may clarify if the overall population benefits are rather uniform or mostly driven by a major response among patients with exaggerated inflammatory profile.
One must recognize that colchicine is not tolerated by everyone. Early gastrointestinal intolerance has been common in many cardiovascular trials and should be monitored in patients when starting colchicine therapy. Furthermore, the majority of RCTs to date excluded patients with severe renal dysfunction, and findings from above trials cannot be extrapolated to this population. Preliminary data from LoDoCo2 among patients with CKD stage 3A or worse may even indicate that colchicine loses its beneficial cardiovascular effect with worsening renal disease. Given that patients with CKD represent a substantial proportion of the CAD population, it will be imperative to investigate whether renal dosing of colchicine in such patients could safely lead to similar positive clinical outcomes. Until then, colchicine treatment in patients with severe renal dysfunction should remain relatively contra-indicated given its partial renal clearance.
All-in-all, while anti-inflammatory therapy with canakinumab would have likely been too costly to implement on a large scale, colchicine has been a widely available oral agent for centuries and is reasonably inexpensive in most areas of the world. A cost effectiveness study of COLCOT reported cost savings with the addition of colchicine to standard therapy after MI.100 However, colchicine may cost more in certain countries, including areas as the United States which were not included in the study. The ever-growing evidence of cardiovascular benefit that colchicine carries may forecast relevant updates in upcoming practice guidelines for indications such as CAD or cardiac surgery. If colchicine use does expand over the coming years, caution must be taken to avoid cost increases, due to supply limitations or elsewise.
Conclusion
There is broad evidence regarding colchicine’s benefits in acute and recurrent pericarditis, and emerging indications for its use in post-procedural AF, CAD and stroke based on its targeting of cardiovascular inflammation. Indeed, colchicine may potentially become an important addition to other standard cardiovascular therapies. Guidelines already recommend colchicine for cardiovascular diseases as pericarditis and ESC has recently included coronary artery disease as a potential indication as well. Questions however remain regarding its use in patients with severe chronic kidney disease and its potential effect on non-cardiovascular mortality. Large ongoing trials (Table 6) should further clarify how best to utilize this old therapeutic agent in cardiac disease in the most safe and effective manner. Given colchicine’s promise, they are eagerly awaited.
Table 6.
Ongoing major registered randomized clinical trials with colchicine in cardiovascular disease
| STUDY | DESIGN | TARGET POPULATION | INTERVENTION | PRIMARY OUTCOME | PLANNED FOLLOW UP | COUNTRY |
|---|---|---|---|---|---|---|
| ATRIAL FIBRILLATION | ||||||
|
| ||||||
|
Colchicine For The Prevention Of Perioperative Atrial Fibrillation In Patients Undergoing Thoracic Surgery (COP-AF)
NCT03310125 |
Phase 3 prospective, randomized, double blind |
Major Thoracic Surgery Patients (n = 2,800) |
Colchicine 0.5mg BID for 10 days | POAF | 14 days | Canada |
|
COlchicine in Cardiac Surgery (COCS)
NCT04224545 |
Phase 4, prospective, randomized, double-blind |
CABG and/or AVR Patients (n = 1,000) |
Colchicine 1mg QD a day before surgery, and 2, 3, 4, 5 days after surgery | POAF | 7 days | Russia |
|
Impact of Short-course Colchicine Versus Placebo After Pulmonary Vein Isolation (IMPROVE-PVI Pilot)
NCT04160117 |
Phase 3, prospective, randomized, double-blind |
RFA-PVI (n = 200) |
Colchicine 0.6mg BID for 10 days | AF recurrence | 2 years | Canada |
|
| ||||||
| CORONARY ARTERY DISEASE | ||||||
|
| ||||||
|
Colchicine and Spironolactone in Patients With MI / SYNERGY Stent Registry (CLEAR SYNERGY)
NCT03048825 |
Phase 3, prospective, randomized, blinded, double-dummy 2×2 factorial design |
STEMI or NSTEMI status-post PCI (n = 7,000) |
Drug 1: Colchicine 0.5mg BID Drug 2: Spironolactone 25mg QD Device: SYNERGY Bioabsorbable Polymer Drug-Eluting Stent |
MACE | 1 year | NY, USA |
|
COLchicine effects on Cardiovascular Outcomes in Acute Coronary Syndrome Study (COLCARDIO-ACS) ACTRN12616000400460 |
Prospective, randomized, double blind, placebo controlled | ACS + hs-CRP >2mg/L 4–6 weeks post event (n = 3,000) |
Colchicine 0.5mg QD for 3 years | Cardiac Events | 3 years | Australia |
|
Effect of Colchicine in Patients With Myocardial Infarction
NCT04218786 |
Phase 2, prospective, randomized, double-blind | ACS (n = 800) |
Colchicine 0.5mg QD for 3 monts | MACE | 3 months | Pakistan |
|
| ||||||
| PCI | ||||||
|
| ||||||
|
Oral Colchicine in Argentina to Prevent Restenosis (ORCA)
NCT04382443 |
Phase 4, prospective, randomized, open-label |
PCI (n = 450) |
Arm 1: Colchicine 0.5mg BID for 3 months+ BMS Arm 2: DES |
MACE | 1 year | Argentina |
|
| ||||||
|
CEREBROVASCULAR
DISEASE | ||||||
|
| ||||||
|
Colchicine for Prevention of Vascular Inflammation in Non-cardio Embolic Stroke (CONVINCE)
NCT02898610 |
Phase 3, prospective, randomized, open-label |
Stroke/TIA (n = 2,623) |
Conventional treatment + Colchicine 0.5mg QD for 60 months | MACE | 60 months | Ireland |
|
Colchicine after stroke to prevent event recurrence (CASPER)
Registration ID to be confirmed |
Prospective, randomized, double-blind, placebo controlled | Stroke/TIA + hs-CRP >2 mg/L at 4–6 weeks post-event (n = unknown) |
Conventional treatment + Colchicine 0.5mg QD for 60 months | MACE | To be confirmed | Australia |
ACS = Acute Coronary Syndrome; AF = Atrial Fibrillation; AVR = Aortic Valve Replacement; BID = twice daily; BMS = Bare Metal Stent; CABG = Coronary Artery Bypass Graft; CMR = Cardiac MRI; DES = Drug-Eluting Stent; hs-CRP = high-sensitivity C-reactive protein; MACE = Major Adverse Cardiac Events; PCI = Percutaneous Coronary Intervention; POAF = Post-operative Atrial Fibrillation; RFA-PVI = radiofrequency ablation pulmonary vein isolation; QD = daily; TIA = Transient Ischemic Attack;
Acknowledgement
The authors thank Marielle Mahan for her expert assistance with the medical illustrations.
Funding
Clare Arnott holds National Health and Medical Research Council/Medical Research Future Fund Priority Investigator Grant and an New South Wales Health Early-Mid Career Research Grant (both of which are not for colchicine studies). Sanjay Patel holds the following Australian National Health and Medical Research Council/Medical Research Future Fund funded colchicine grants: COLCARDIO-ACS GA65779, CASPER GA82107, IMPACT-ICO GA85492. These studies are supported by Aspen Pharmacare Australia who are providing drug and placebo. Sanjay Patel also holds a New South Wales Cardiovascular Health Fellowship to support colchicine research. Mark Parsons receives funding from the National Health and Medical Research Council Program Grant in Stroke (ID 1113352). Jason Kovacic received funding from the NIH (R01HL130423, R01HL135093, R01HL148167–01A1) and New South Wales health grant (RG194194).
Abbreviations
- ACC/AHA
American College of Cardiology/American Heart Association
- ACS
Acute Coronary Syndrome
- AF
Atrial Fibrillation
- CAD
Coronary Artery Disease
- CRP
C-Reactive Protein
- IL
Interleukin
- ESC
European Society of Cardiology
- FMF
Familial Mediterranean Fever
- LV
Left Ventricle
- MI
Myocardial Infarction
- NLRP3
NLR Family Pyrin Domain Containing 3
- PAD
Peripheral Artery Disease
- PCI
Percutaneous Coronary Intervention
- POAF
Post-Operative Atrial Fibrillation
- PPS
Post-Pericardiotomy Syndrome
- RCT
Randomized Clinical Trial
- RFA PVI
Radiofrequency Ablation Pulmonary Vein Isolation
Footnotes
Disclosures
Binita Shah reports funding from the Veterans Affairs Office of Research and Development (iK2CX001074) and the National Heart, Lung, Blood Institute of the National Institutes of Health (R01HL146206) for studies of colchicine in coronary artery disease; is on the advisory board for Philips Volcano and serves as a consultant for Terumo Medical. Sanjay Patel reports involvement with Aspen Pharmacare through colchicine-related research studies as above. Jean-Claude Tardif has received research grants from Amarin, AstraZeneca, Ceapro, DalCor Pharmaceuticals, Esperion, Ionis, Novartis, Pfizer, RegenXBio and Sanofi; honoraria from AstraZeneca, DalCor Pharmaceuticals, HLS Pharmaceuticals and Pendopharm; minor equity interest from DalCor Pharmaceuticals; and is an author of patents on pharmacogenomics-guided CETP-inhibition, use of colchicine after MI, and use of colchicine in coronavirus infection (Dr. Tardif has waived his rights in patents on colchicine and does not stand to gain financially). The other authors have no conflicts to disclose and no competing interests.
References
- 1.Nerlekar N, Beale A and Harper RW. Colchicine — a short history of an ancient drug. Medical Journal of Australia 2014;201:687–688. [DOI] [PubMed] [Google Scholar]
- 2.Imazio M and Nidorf M. Colchicine and the heart. European Heart Journal 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Beck C, Morbach H, Richl P, Stenzel M and Girschick HJ. How can calcium pyrophosphate crystals induce inflammation in hypophosphatasia or chronic inflammatory joint diseases? Rheumatol Int 2009;29:229–238. [DOI] [PubMed] [Google Scholar]
- 4.Esatoglu SN and Hatemi G. Update on the treatment of Behçet’s syndrome. Intern Emerg Med 2019;14:661–675. [DOI] [PubMed] [Google Scholar]
- 5.O’Gara PT, Kushner FG, Ascheim DD, Casey DE Jr., Chung MK, de Lemos JA, Ettinger SM, Fang JC, Fesmire FM, Franklin BA, et al. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation 2013;127:e362–425. [DOI] [PubMed] [Google Scholar]
- 6.Adler Y, Charron P, Imazio M, Badano L, Barón-Esquivias G, Bogaert J, Brucato A, Gueret P, Klingel K, Lionis C, et al. 2015 ESC Guidelines for the diagnosis and management of pericardial diseases: The Task Force for the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology (ESC)Endorsed by: The European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J 2015;36:2921–2964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Klimecki WT, Futscher BW, Grogan TM and Dalton WS. P-glycoprotein expression and function in circulating blood cells from normal volunteers. Blood 1994;83:2451–2458. [PubMed] [Google Scholar]
- 8.Caner JE. Colchicine inhibition of chemotaxis. Arthritis Rheum 1965;8:757–764. [DOI] [PubMed] [Google Scholar]
- 9.Cronstein BN, Molad Y, Reibman J, Balakhane E, Levin RI and Weissmann G. Colchicine alters the quantitative and qualitative display of selectins on endothelial cells and neutrophils. J Clin Invest 1995;96:994–1002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ding AH, Porteu F, Sanchez E and Nathan CF. Downregulation of tumor necrosis factor receptors on macrophages and endothelial cells by microtubule depolymerizing agents. J Exp Med 1990;171:715–727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Li Z, Davis GS, Mohr C, Nain M and Gemsa D. Inhibition of LPS-induced tumor necrosis factor-alpha production by colchicine and other microtubule disrupting drugs. Immunobiology 1996;195:624–639. [DOI] [PubMed] [Google Scholar]
- 12.Roberge CJ, Gaudry M, de Médicis R, Lussier A, Poubelle PE and Naccache PH. Crystal-induced neutrophil activation. IV. Specific inhibition of tyrosine phosphorylation by colchicine. J Clin Invest 1993;92:1722–1729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wright DG and Malawista SE. Mobilization and extracellular release of granular enzymes from human leukocytes during phagocytosis: inhibition by colchicine and cortisol but not by salicylate. Arthritis Rheum 1973;16:749–758. [DOI] [PubMed] [Google Scholar]
- 14.Martinon F, Pétrilli V, Mayor A, Tardivel A and Tschopp J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 2006;440:237–241. [DOI] [PubMed] [Google Scholar]
- 15.Park YH, Wood G, Kastner DL and Chae JJ. Pyrin inflammasome activation and RhoA signaling in the autoinflammatory diseases FMF and HIDS. Nat Immunol 2016;17:914–921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kirii H, Niwa T, Yamada Y, Wada H, Saito K, Iwakura Y, Asano M, Moriwaki H and Seishima M. Lack of interleukin-1beta decreases the severity of atherosclerosis in ApoE-deficient mice. Arterioscler Thromb Vasc Biol 2003;23:656–660. [DOI] [PubMed] [Google Scholar]
- 17.Mallat Z, Corbaz A, Scoazec A, Besnard S, Lesèche G, Chvatchko Y and Tedgui A. Expression of interleukin-18 in human atherosclerotic plaques and relation to plaque instability. Circulation 2001;104:1598–1603. [DOI] [PubMed] [Google Scholar]
- 18.Garlanda C, Dinarello CA and Mantovani A. The interleukin-1 family: back to the future. Immunity 2013;39:1003–1018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bauriedel G, Heimerl J, Beinert T, Welsch U and Höfling B. Colchicine antagonizes the activity of human smooth muscle cells cultivated from arteriosclerotic lesions after atherectomy. Coron Artery Dis 1994;5:531–539. [PubMed] [Google Scholar]
- 20.Slobodnick A, Shah B, Krasnokutsky S and Pillinger MH. Update on colchicine, 2017. Rheumatology (Oxford) 2018;57:i4–i11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Terkeltaub RA, Furst DE, Bennett K, Kook KA, Crockett RS and Davis MW. High versus low dosing of oral colchicine for early acute gout flare: Twenty-four-hour outcome of the first multicenter, randomized, double-blind, placebo-controlled, parallel-group, dose-comparison colchicine study. Arthritis Rheum 2010;62:1060–1068. [DOI] [PubMed] [Google Scholar]
- 22.Andreis A, Imazio M, Avondo S, Casula M, Paneva E, Piroli F and De Ferrari GM. Adverse events of colchicine for cardiovascular diseases: a comprehensive meta-analysis of 14 188 patients from 21 randomized controlled trials. J Cardiovasc Med (Hagerstown) 2021;22:637–644. [DOI] [PubMed] [Google Scholar]
- 23.Nidorf SM, Eikelboom JW and Thompson PL. Colchicine for secondary prevention of cardiovascular disease. Curr Atheroscler Rep 2014;16:391. [DOI] [PubMed] [Google Scholar]
- 24.Stewart S, Yang KCK, Atkins K, Dalbeth N and Robinson PC. Adverse events during oral colchicine use: a systematic review and meta-analysis of randomised controlled trials. Arthritis Res Ther 2020;22:28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Medani S and Wall C. Colchicine toxicity in renal patients - Are we paying attention? Clin Nephrol 2016;86:100–105. [DOI] [PubMed] [Google Scholar]
- 26.U. S. Food and Drug Administration. FDA Center for Drug Evaluation and Research, Guidance for Industry; Cytochrome P450 Enzyme- and Transporter-Mediated Drug Interactions. Clinical Drug Interaction Studies January 2020, accessed 19 October 2021. https://www.fda.gov/media/134581/download. [Google Scholar]
- 27.Tufan A, Dede DS, Cavus S, Altintas ND, Iskit AB and Topeli A. Rhabdomyolysis in a patient treated with colchicine and atorvastatin. Ann Pharmacother 2006;40:1466–1469. [DOI] [PubMed] [Google Scholar]
- 28.Hung IF, Wu AK, Cheng VC, Tang BS, To KW, Yeung CK, Woo PC, Lau SK, Cheung BM and Yuen KY. Fatal interaction between clarithromycin and colchicine in patients with renal insufficiency: a retrospective study. Clin Infect Dis 2005;41:291–300. [DOI] [PubMed] [Google Scholar]
- 29.Imazio M, Brucato A, Maestroni S, Cumetti D, Dominelli A, Natale G and Trinchero R. Prevalence of C-reactive protein elevation and time course of normalization in acute pericarditis: implications for the diagnosis, therapy, and prognosis of pericarditis. Circulation 2011;123:1092–1097. [DOI] [PubMed] [Google Scholar]
- 30.Imazio M, Bobbio M, Cecchi E, Demarie D, Demichelis B, Pomari F, Moratti M, Gaschino G, Giammaria M, Ghisio A, et al. Colchicine in addition to conventional therapy for acute pericarditis: results of the COlchicine for acute PEricarditis (COPE) trial. Circulation 2005;112:2012–2016. [DOI] [PubMed] [Google Scholar]
- 31.Imazio M, Brucato A, Cemin R, Ferrua S, Maggiolini S, Beqaraj F, Demarie D, Forno D, Ferro S, Maestroni S, et al. A randomized trial of colchicine for acute pericarditis. The New England journal of medicine 2013;369:1522–1528. [DOI] [PubMed] [Google Scholar]
- 32.Sambola A, Roca Luque I, Mercé J, Alguersuari J, Francisco-Pascual J, García-Dorado D and Sagristà-Sauleda J. Colchicine Administered in the First Episode of Acute Idiopathic Pericarditis: A Randomized Multicenter Open-label Study. Rev Esp Cardiol (Engl Ed) 2019;72:709–716. [DOI] [PubMed] [Google Scholar]
- 33.Imazio M, Bobbio M, Cecchi E, Demarie D, Pomari F, Moratti M, Ghisio A, Belli R and Trinchero R. Colchicine as first-choice therapy for recurrent pericarditis: results of the CORE (COlchicine for REcurrent pericarditis) trial. Arch Intern Med 2005;165:1987–1991. [DOI] [PubMed] [Google Scholar]
- 34.Imazio M, Brucato A, Cemin R, Ferrua S, Belli R, Maestroni S, Trinchero R, Spodick DH and Adler Y. Colchicine for recurrent pericarditis (CORP): a randomized trial. Ann Intern Med 2011;155:409–414. [DOI] [PubMed] [Google Scholar]
- 35.Imazio M, Belli R, Brucato A, Cemin R, Ferrua S, Beqaraj F, Demarie D, Ferro S, Forno D, Maestroni S, et al. Efficacy and safety of colchicine for treatment of multiple recurrences of pericarditis (CORP-2): a multicentre, double-blind, placebo-controlled, randomised trial. Lancet 2014;383:2232–2237. [DOI] [PubMed] [Google Scholar]
- 36.Imazio M, Negro A, Belli R, Beqaraj F, Forno D, Giammaria M, Trinchero R, Adler Y and Spodick D. Frequency and prognostic significance of pericarditis following acute myocardial infarction treated by primary percutaneous coronary intervention. Am J Cardiol 2009;103:1525–1529. [DOI] [PubMed] [Google Scholar]
- 37.Imazio M, Brucato A, Ferrazzi P, Spodick DH and Adler Y. Postpericardiotomy syndrome: a proposal for diagnostic criteria. J Cardiovasc Med (Hagerstown) 2013;14:351–3. [DOI] [PubMed] [Google Scholar]
- 38.Finkelstein Y, Shemesh J, Mahlab K, Abramov D, Bar-El Y, Sagie A, Sharoni E, Sahar G, Smolinsky AK, Schechter T, et al. Colchicine for the prevention of postpericardiotomy syndrome. Herz 2002;27:791–794. [DOI] [PubMed] [Google Scholar]
- 39.Imazio M, Trinchero R, Brucato A, Rovere ME, Gandino A, Cemin R, Ferrua S, Maestroni S, Zingarelli E, Barosi A, et al. COlchicine for the Prevention of the Post-pericardiotomy Syndrome (COPPS): a multicentre, randomized, double-blind, placebo-controlled trial. Eur Heart J 2010;31:2749–2754. [DOI] [PubMed] [Google Scholar]
- 40.Imazio M, Brucato A, Ferrazzi P, Pullara A, Adler Y, Barosi A, Caforio AL, Cemin R, Chirillo F, Comoglio C, et al. Colchicine for Prevention of Postpericardiotomy Syndrome and Postoperative Atrial Fibrillation: The COPPS-2 Randomized Clinical Trial. JAMA 2014;312:1016–1023. [DOI] [PubMed] [Google Scholar]
- 41.Meurin P, Lelay-Kubas S, Pierre B, Pereira H, Pavy B, Iliou MC, Bussière JL, Weber H, Beugin JP, Farrokhi T, et al. Colchicine for postoperative pericardial effusion: a multicentre, double-blind, randomised controlled trial. Heart 2015;101:1711–1716. [DOI] [PubMed] [Google Scholar]
- 42.Yao C, Veleva T, Scott L Jr., Cao S, Li L, Chen G, Jeyabal P, Pan X, Alsina KM, Abu-Taha ID, et al. Enhanced Cardiomyocyte NLRP3 Inflammasome Signaling Promotes Atrial Fibrillation. Circulation 2018;138:2227–2242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Varghese B, Feldman DI, Chew C, Valilis E, Blumenthal RS, Sharma G and Calkins H. Inflammation, atrial fibrillation, and the potential role for colchicine therapy. Heart Rhythm O2 2021;2:298–303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gialdini G, Nearing K, Bhave PD, Bonuccelli U, Iadecola C, Healey JS and Kamel H. Perioperative atrial fibrillation and the long-term risk of ischemic stroke. Jama 2014;312:616–622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Imazio M, Brucato A, Ferrazzi P, Rovere Maria E, Gandino A, Cemin R, Ferrua S, Belli R, Maestroni S, Simon C, et al. Colchicine Reduces Postoperative Atrial Fibrillation. Circulation 2011;124:2290–2295. [DOI] [PubMed] [Google Scholar]
- 46.Sarzaeem M, Shayan N, Bagheri J, Jebelli M and Mandegar M. Low dose Colchicine in prevention of atrial fibrillation after coronary artery bypass graft: a double blind clinical trial. Tehran-Univ-Med-J 2014;72:147–154. [Google Scholar]
- 47.Lee JZ, Singh N, Howe CL, Low SW, Huang JJ, Ortega G, Lee KS and Pandit A. Colchicine for Prevention of Post-Operative Atrial Fibrillation: A Meta-Analysis. JACC Clin Electrophysiol 2016;2:78–85. [DOI] [PubMed] [Google Scholar]
- 48.Zarpelon CS, Netto MC, Jorge JCM, Fabris CC, Desengrini D, Jardim MdS and Silva DGd. Colchicine to Reduce Atrial Fibrillation in the Postoperative Period of Myocardial Revascularization. Arq Bras Cardiol 2016;107:4–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Tabbalat RA, Alhaddad I, Hammoudeh A, Khader YS, Khalaf HA, Obaidat M and Barakat J. Effect of Low-dose ColchiciNe on the InciDence of Atrial Fibrillation in Open Heart Surgery Patients: END-AF Low Dose Trial. J Int Med Res 2020;48:300060520939832–300060520939832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.January CT, Wann LS, Alpert JS, Calkins H, Cigarroa JE, Cleveland JC, Conti JB, Ellinor PT, Ezekowitz MD, Field ME, et al. 2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation. Circulation 2014;130:e199–e267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kirchhof P, Benussi S, Kotecha D, Ahlsson A, Atar D, Casadei B, Castella M, Diener H-C, Heidbuchel H, Hendriks J, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. European Heart Journal 2016;37:2893–2962. [DOI] [PubMed] [Google Scholar]
- 52.Deftereos S, Giannopoulos G, Kossyvakis C, Efremidis M, Panagopoulou V, Kaoukis A, Raisakis K, Bouras G, Angelidis C, Theodorakis A, et al. Colchicine for prevention of early atrial fibrillation recurrence after pulmonary vein isolation: a randomized controlled study. J Am Coll Cardiol 2012;60:1790–1796. [DOI] [PubMed] [Google Scholar]
- 53.Deftereos S, Giannopoulos G, Efremidis M, Kossyvakis C, Katsivas A, Panagopoulou V, Papadimitriou C, Karageorgiou S, Doudoumis K, Raisakis K, et al. Colchicine for prevention of atrial fibrillation recurrence after pulmonary vein isolation: mid-term efficacy and effect on quality of life. Heart Rhythm 2014;11:620–628. [DOI] [PubMed] [Google Scholar]
- 54.Calkins H, Hindricks G, Cappato R, Kim YH, Saad EB, Aguinaga L, Akar JG, Badhwar V, Brugada J, Camm J, et al. 2017 HRS/EHRA/ECAS/APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation: Executive summary. Europace 2018;20:157–208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Naruko T, Ueda M, Haze K, van der Wal AC, van der Loos CM, Itoh A, Komatsu R, Ikura Y, Ogami M, Shimada Y, et al. Neutrophil infiltration of culprit lesions in acute coronary syndromes. Circulation 2002;106:2894–2900. [DOI] [PubMed] [Google Scholar]
- 56.Shu J, Ren N, Du JB, Zhang M, Cong HL and Huang TG. Increased levels of interleukin-6 and matrix metalloproteinase-9 are of cardiac origin in acute coronary syndrome. Scand Cardiovasc J 2007;41:149–154. [DOI] [PubMed] [Google Scholar]
- 57.Petersen LC, Bjørn SE and Nordfang O. Effect of leukocyte proteinases on tissue factor pathway inhibitor. Thromb Haemost 1992;67:537–541. [PubMed] [Google Scholar]
- 58.Higazi M, Abdeen S, Abu-Fanne R, Heyman SN, Masarwy A, Bdeir K, Maraga E, Cines DB and Higazi AA. Opposing effects of HNP1 (α-defensin-1) on plasma cholesterol and atherogenesis. PLoS One 2020;15:e0231582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Abu-Fanne R, Stepanova V, Litvinov RI, Abdeen S, Bdeir K, Higazi M, Maraga E, Nagaswami C, Mukhitov AR, Weisel JW, et al. Neutrophil α-defensins promote thrombosis in vivo by altering fibrin formation, structure, and stability. Blood 2019;133:481–493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Quillard T, Araújo HA, Franck G, Shvartz E, Sukhova G and Libby P. TLR2 and neutrophils potentiate endothelial stress, apoptosis and detachment: implications for superficial erosion. Eur Heart J 2015;36:1394–1404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Furman MI, Barnard MR, Krueger LA, Fox ML, Shilale EA, Lessard DM, Marchese P, Frelinger AL 3rd, Goldberg RJ and Michelson AD. Circulating monocyte-platelet aggregates are an early marker of acute myocardial infarction. J Am Coll Cardiol 2001;38:1002–1006. [DOI] [PubMed] [Google Scholar]
- 62.Shah B, Allen N, Harchandani B, Pillinger M, Katz S, Sedlis SP, Echagarruga C, Samuels SK, Morina P, Singh P, et al. Effect of Colchicine on Platelet-Platelet and Platelet-Leukocyte Interactions: a Pilot Study in Healthy Subjects. Inflammation 2016;39:182–189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Nidorf M and Thompson PL. Effect of colchicine (0.5 mg twice daily) on high-sensitivity C-reactive protein independent of aspirin and atorvastatin in patients with stable coronary artery disease. Am J Cardiol 2007;99:805–807. [DOI] [PubMed] [Google Scholar]
- 64.Vaidya K, Arnott C, Martínez GJ, Ng B, McCormack S, Sullivan DR, Celermajer DS and Patel S. Colchicine Therapy and Plaque Stabilization in Patients With Acute Coronary Syndrome: A CT Coronary Angiography Study. JACC Cardiovasc Imaging 2018;11:305–316. [DOI] [PubMed] [Google Scholar]
- 65.Toprover M, Shah B, Oh C, Igel TF, Romero AG, Pike VC, Curovic F, Bang D, Lazaro D, Krasnokutsky S, et al. Initiating guideline-concordant gout treatment improves arterial endothelial function and reduces intercritical inflammation: a prospective observational study. Arthritis research & therapy 2020;22:169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C, Fonseca F, Nicolau J, Koenig W, Anker SD, et al. Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. The New England journal of medicine 2017;377:1119–1131. [DOI] [PubMed] [Google Scholar]
- 67.Ridker PM, MacFadyen JG, Everett BM, Libby P, Thuren T and Glynn RJ. Relationship of C-reactive protein reduction to cardiovascular event reduction following treatment with canakinumab: a secondary analysis from the CANTOS randomised controlled trial. Lancet 2018;391:319–328. [DOI] [PubMed] [Google Scholar]
- 68.Ridker PM, Everett BM, Pradhan A, MacFadyen JG, Solomon DH, Zaharris E, Mam V, Hasan A, Rosenberg Y, Iturriaga E, et al. Low-Dose Methotrexate for the Prevention of Atherosclerotic Events. The New England journal of medicine 2019;380:752–762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Langevitz P, Livneh A, Neumann L, Buskila D, Shemer J, Amolsky D and Pras M. Prevalence of ischemic heart disease in patients with familial Mediterranean fever. Isr Med Assoc J 2001;3:9–12. [PubMed] [Google Scholar]
- 70.Shah B, Toprover M, Crittenden DB, Jeurling S, Pike VC, Krasnokutsky S, Xia Y, Fisher MC, Slobodnick A, Tenner CT, et al. Colchicine Use and Incident Coronary Artery Disease in Male Patients With Gout. Can J Cardiol 2020;36:1722–1728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Crittenden DB, Lehmann RA, Schneck L, Keenan RT, Shah B, Greenberg JD, Cronstein BN, Sedlis SP and Pillinger MH. Colchicine use is associated with decreased prevalence of myocardial infarction in patients with gout. J Rheumatol 2012;39:1458–1464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Solomon DH, Liu C-C, Kuo IH, Zak A and Kim SC. Effects of colchicine on risk of cardiovascular events and mortality among patients with gout: a cohort study using electronic medical records linked with Medicare claims. Ann Rheum Dis 2016;75:1674–1679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Nidorf SM, Eikelboom JW, Budgeon CA and Thompson PL. Low-dose colchicine for secondary prevention of cardiovascular disease. J Am Coll Cardiol 2013;61:404–410. [DOI] [PubMed] [Google Scholar]
- 74.Nidorf SM, Fiolet ATL, Mosterd A, Eikelboom JW, Schut A, Opstal TSJ, The SHK, Xu XF, Ireland MA, Lenderink T, et al. Colchicine in Patients with Chronic Coronary Disease. The New England journal of medicine 2020;383:1838–1847. [DOI] [PubMed] [Google Scholar]
- 75.Opstal TSJ, Fiolet ATL, van Broekhoven A, Mosterd A, Eikelboom JW, Nidorf SM, Thompson PL, Duyvendak M, van Eck JWM, van Beek EA, et al. Colchicine in Patients With Chronic Coronary Disease in Relation to Prior Acute Coronary Syndrome. Journal of the American College of Cardiology 2021;78:859–866. [DOI] [PubMed] [Google Scholar]
- 76.Mastrocola R, Penna C, Tullio F, Femminò S, Nigro D, Chiazza F, Serpe L, Collotta D, Alloatti G, Cocco M, et al. Pharmacological Inhibition of NLRP3 Inflammasome Attenuates Myocardial Ischemia/Reperfusion Injury by Activation of RISK and Mitochondrial Pathways. Oxid Med Cell Longev 2016;2016:5271251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Deftereos S, Giannopoulos G, Angelidis C, Alexopoulos N, Filippatos G, Papoutsidakis N, Sianos G, Goudevenos J, Alexopoulos D, Pyrgakis V, et al. Anti-Inflammatory Treatment With Colchicine in Acute Myocardial Infarction: A Pilot Study. Circulation 2015;132:1395–1403. [DOI] [PubMed] [Google Scholar]
- 78.Hennessy T, Soh L, Bowman M, Kurup R, Schultz C, Patel S and Hillis GS. The Low Dose Colchicine after Myocardial Infarction (LoDoCo-MI) study: A pilot randomized placebo controlled trial of colchicine following acute myocardial infarction. Am Heart J 2019;215:62–69. [DOI] [PubMed] [Google Scholar]
- 79.Tong David C, Quinn S, Nasis A, Hiew C, Roberts-Thomson P, Adams H, Sriamareswaran R, Htun Nay M, Wilson W, Stub D, et al. Colchicine in Patients With Acute Coronary Syndrome. Circulation 2020;142:1890–1900. [DOI] [PubMed] [Google Scholar]
- 80.Mewton N, Roubille F, Bresson D, Prieur C, Bouleti C, Bochaton T, Ivanes F, Dubreuil O, Biere L, Hayek A, et al. Effect of Colchicine on Myocardial Injury in Acute Myocardial Infarction. Circulation 2021;144:859–869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Lawler PR, Bhatt DL, Godoy LC, Lüscher TF, Bonow RO, Verma S and Ridker PM. Targeting cardiovascular inflammation: next steps in clinical translation. European Heart Journal 2021;42:113–131. [DOI] [PubMed] [Google Scholar]
- 82.Tardif JC, Kouz S, Waters DD, Bertrand OF, Diaz R, Maggioni AP, Pinto FJ, Ibrahim R, Gamra H, Kiwan GS, et al. Efficacy and Safety of Low-Dose Colchicine after Myocardial Infarction. The New England journal of medicine 2019;381:2497–2505. [DOI] [PubMed] [Google Scholar]
- 83.Samuel M, Tardif JC, Bouabdallaoui N, Khairy P, Dubé MP, Blondeau L and Guertin MC. Colchicine for Secondary Prevention of Cardiovascular Disease: A Systematic Review and Meta-analysis of Randomized Controlled Trials. Can J Cardiol 2021;37:776–785. [DOI] [PubMed] [Google Scholar]
- 84.Fiolet ATL, Opstal TSJ, Mosterd A, Eikelboom JW, Jolly SS, Keech AC, Kelly P, Tong DC, Layland J, Nidorf SM, et al. Efficacy and safety of low-dose colchicine in patients with coronary disease: a systematic review and meta-analysis of randomized trials. Eur Heart J 2021;42:2765–2775. [DOI] [PubMed] [Google Scholar]
- 85.Deftereos S, Giannopoulos G, Raisakis K, Kossyvakis C, Kaoukis A, Panagopoulou V, Driva M, Hahalis G, Pyrgakis V, Alexopoulos D, et al. Colchicine treatment for the prevention of bare-metal stent restenosis in diabetic patients. J Am Coll Cardiol 2013;61:1679–1685. [DOI] [PubMed] [Google Scholar]
- 86.Visseren FLJ, Mach F, Smulders YM, Carballo D, Koskinas KC, Bäck M, Benetos A, Biffi A, Boavida J-M, Capodanno D, et al. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice: Developed by the Task Force for cardiovascular disease prevention in clinical practice with representatives of the European Society of Cardiology and 12 medical societies With the special contribution of the European Association of Preventive Cardiology (EAPC). European Heart Journal 2021;42:3227–3337.34458905 [Google Scholar]
- 87.Health Canada approval of low-dose colchicine for cardiovascular disease based on the COLCOT study. Institute de Cardiologie de Montreal 27 August 2021, accessed 19 October 2021. https://www.icm-mhi.org/en/pressroom/news/health-canada-approval-low-dose-colchicine-cardiovascular-disease-based-colcot-study. [Google Scholar]
- 88.Almagor M, Keren A and Banai S. Increased C-reactive protein level after coronary stent implantation in patients with stable coronary artery disease. Am Heart J 2003;145:248–253. [DOI] [PubMed] [Google Scholar]
- 89.O’Keefe JH, McCallister BD, Bateman TM, Kuhnlein DL, Ligon RW and Hartzler GO. Ineffectiveness of colchicine for the prevention of restenosis after coronary angioplasty. Journal of the American College of Cardiology 1992;19:1597–1600. [DOI] [PubMed] [Google Scholar]
- 90.Freed M, Safian RD, O’Neill WW, Safian M, Jones D and Grines CL. Combination of lovastatin, enalapril, and colchicine does not prevent restenosis after percutaneous transluminal coronary angioplasty. The American Journal of Cardiology 1995;76:1185–1188. [DOI] [PubMed] [Google Scholar]
- 91.Herrmann J, Lennon Ryan J, Barsness Gregory W, Sandhu Gurpreet S, Gulati R, Best Patricia JM, Sorajja P, Bresnahan John F, Mathew V, Bell Malcolm R, et al. High Sensitivity C-Reactive Protein and Outcomes Following Percutaneous Coronary Intervention in Contemporary Practice. Circulation: Cardiovascular Interventions 2012;5:783–790. [DOI] [PubMed] [Google Scholar]
- 92.Shah B, Baber U, Pocock SJ, Krucoff MW, Ariti C, Gibson CM, Steg PG, Weisz G, Witzenbichler B, Henry TD, et al. White Blood Cell Count and Major Adverse Cardiovascular Events After Percutaneous Coronary Intervention in the Contemporary Era: Insights From the PARIS Study (Patterns of Non-Adherence to Anti-Platelet Regimens in Stented Patients Registry). Circ Cardiovasc Interv 2017;10(9):e004981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Di Sciascio G, Patti G, Pasceri V, Gaspardone A, Colonna G and Montinaro A. Efficacy of atorvastatin reload in patients on chronic statin therapy undergoing percutaneous coronary intervention: results of the ARMYDA-RECAPTURE (Atorvastatin for Reduction of Myocardial Damage During Angioplasty) Randomized Trial. J Am Coll Cardiol 2009;54:558–565. [DOI] [PubMed] [Google Scholar]
- 94.Shah B, Pillinger M, Zhong H, Cronstein B, Xia Y, Lorin JD, Smilowitz NR, Feit F, Ratnapala N, Keller NM, et al. Effects of Acute Colchicine Administration Prior to Percutaneous Coronary Intervention: COLCHICINE-PCI Randomized Trial. Circ Cardiovasc Interv 2020;13:e008717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Cole J, Htun N, Lew R, Freilich M, Quinn S and Layland J. Colchicine to Prevent Periprocedural Myocardial Injury in Percutaneous Coronary Intervention: The COPE-PCI Pilot Trial. Circulation: Cardiovascular Interventions 2021;14:e009992. [DOI] [PubMed] [Google Scholar]
- 96.Amarenco P, Lavallée PC, Labreuche J, Albers GW, Bornstein NM, Canhão P, Caplan LR, Donnan GA, Ferro JM, Hennerici MG, et al. One-Year Risk of Stroke after Transient Ischemic Attack or Minor Stroke. The New England journal of medicine 2016;374:1533–1542. [DOI] [PubMed] [Google Scholar]
- 97.Tarkin JM, Joshi FR and Rudd JH. PET imaging of inflammation in atherosclerosis. Nat Rev Cardiol 2014;11:443–457. [DOI] [PubMed] [Google Scholar]
- 98.Opstal Tjerk SJ, Hoogeveen Renate M, Fiolet Aernoud TL, Silvis Max JM, The Salem HK, Bax Willem A, de Kleijn Dominique PV, Mosterd A, Stroes Erik SG and Cornel Jan H. Colchicine Attenuates Inflammation Beyond the Inflammasome in Chronic Coronary Artery Disease. Circulation 2020;142:1996–1998. [DOI] [PubMed] [Google Scholar]
- 99.Dubé MP, Legault MA, Lemaçon A, Lemieux Perreault LP, Fouodjio R, Waters DD, Kouz S, Pinto FJ, Maggioni AP, Diaz R, et al. Pharmacogenomics of the Efficacy and Safety of Colchicine in COLCOT. Circ Genom Precis Med 2021;14:e003183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Samuel M, Tardif JC, Khairy P, Roubille F, Waters DD, Grégoire JC, Pinto FJ, Maggioni AP, Diaz R, Berry C, et al. Cost-effectiveness of low-dose colchicine after myocardial infarction in the Colchicine Cardiovascular Outcomes Trial (COLCOT). Eur Heart J Qual Care Clin Outcomes 2021;7:486–495. [DOI] [PMC free article] [PubMed] [Google Scholar]


