Abstract
Irritable bowel syndrome (IBS) and non-alcoholic fatty liver disease (NAFLD) are amongst the most common gastrointestinal and liver conditions encountered in primary and secondary care. Recently, there has been interest in the apparent co-incidence of NAFLD in patients with IBS mainly driven by improved understanding of their shared risk factors and pathophysiology. In this paper we summarize the shared risk factors which include; overlapping nutritional and dietary factors as well as shared putative mechanisms of pathophysiology. These include changes in the gut microbiome, gut permeability, immunity, small bowel bacterial overgrowth and bile acid metabolism. This paper describes how these shared risk factors and etiological factors may have practical clinical implications for these highly prevalent conditions. It also highlights some of the limitations of current epidemiological data relating to estimates of the overlapping prevalence of the two conditions which have resulted in inconsistent results and, therefore the need for further research. Early recognition and management of the overlap could potentially have impacts on treatment outcomes, compliance and morbidity of both conditions. Patients with known IBS who have abnormal liver function tests or significant risk factors for NAFLD should be investigated appropriately for this possibility. Similarly, IBS should be considered in patients with NAFLD and symptoms of abdominal pain associated with defecation, an altered bowel habit and bloating.
Keywords: Irritable bowel syndrome, Non-alcoholic fatty liver disease, Metabolic syndrome, Obesity, Prevalence, Pathophysiology
Core Tip: Irritable bowel syndrome (IBS) and non-alcoholic fatty liver disease (NAFLD) are amongst the most common gastrointestinal and liver conditions encountered in primary and secondary care. There has been interest in the apparent co-incidence of NAFLD in patients with IBS mainly driven by improved understanding of their shared risk factors and pathophysiology. In this paper we summarize the shared risk factors which include; overlapping nutritional and dietary factors as well as shared putative mechanisms of pathophysiology. Physicians should be aware of the possibility of co-existence of IBS and NAFLD and consider investigating patients with IBS or NAFLD with clinical features of the other condition.
INTRODUCTION
Irritable Bowel syndrome (IBS) is a disorder of gut-brain interaction (DGBI) resulting in recurrent abdominal pain associated with defecation and an altered bowel habit. Patients are considered to have IBS when they fulfill the Rome IV diagnostic criteria which include an altered bowel habit (constipation, diarrhea or a mix of both), associated with frequent abdominal pain and abdominal bloating or distension for at least 6 mo prior to diagnosis[1]. A recent systematic review and meta-analysis has shown a worldwide prevalence of IBS of 9.2% with significant regional variability[2]. In the United Kingdom, DGBIs such as IBS are very common, and account for around a third of gastroenterology outpatient referrals[3]. IBS can be debilitating often resulting in an increasing risk of anxiety or depression[4] with symptoms such as fecal incontinence that can be difficult to manage leading to poor quality of life and distress[5]. There is often significant clinician prejudice and frustration towards patients with IBS[6] resulting in unfair public perceptions and significant stigmatization[7].
Non-alcoholic fatty liver disease (NAFLD) is characterized by the accumulation of more than 5% of fat in the liver in the absence of a secondary cause. It is one of the major causes of liver disease worldwide and its pathogenesis is linked to metabolic syndrome, obesity and Type 2 diabetes. The population based prevalence of NAFLD is between 25%-44% but rises to 70% in patients with Type 2 diabetes[8,9]. NAFLD is recognized as a heterogeneous condition with variable rates of progression. In certain patients isolated steatosis leads to steatohepatitis and fibrosis, progressing ultimately to cirrhosis, decompensated liver disease and sometimes hepatocellular carcinoma. Population based screening studies have shown a prevalence of advanced fibrosis in 8% of patients rising to 27% in those with risk factors[10,11]. Unfortunately, the majority of patients are only diagnosed with liver disease when they present with advanced disease and many are of working age. Consequently, liver disease is responsible for the loss of 38000 and 22000 working life years, in men and women, respectively. NAFLD has been increasing in incidence in the western world with a predictable commensurate increase in liver transplant in both the United States and Europe[12-14].
There is increasing recognition that both IBS and NAFLD share a number of overlapping risk and aetiological factors leading to growing interest in the possibility of an association between the two conditions. However, there is limited high quality data on the concomitance of IBS and NAFLD. As a result, IBS symptoms may not be routinely screened for in hepatology clinics and vice versa. Therefore, the aims of this article are to summarize the current understanding of relevant overlapping pathophysiological and aetiological factors, and to highlight areas for future research and their clinical implications.
THE PREVALENCE OF CO-EXISTING IBS AND NAFLD
Table 1 summarizes the literature on the co-existing prevalence of IBS and NAFLD to date. Most studies have examined the incidence of NAFLD in previously diagnosed IBS. Unfortunately, a review of the literature of concomitant IBS and NAFLD revealed a very high variability in estimates of the prevalence from 12.9% to 74%, with significant differences in methodology in the diagnostic approaches for both conditions and the populations studied[15-17]. Amongst the reasons for this heterogeneity and variability include the change in the Rome criteria for IBS from Rome III, to the current Rome IV iteration, which is known to be more restrictive[18]. From a hepatology perspective, it is notable that all the studies to date have used raised liver transaminases, with a negative viral hepatitis screen, in the absence of excessive alcohol consumption, and abdominal ultrasound to diagnose NAFLD, which in the absence of objective liver fibrosis assessment could be considered sub-optimal.
Table 1.
Summarizes the literature on the co-existing prevalence of irritable bowel syndrome and non-alcoholic fatty liver disease to date
|
Author
|
Population studied
|
Study design
|
No. patients
|
Criteria for IBS diagnosis
|
IBS subtypes
|
Criteria for NAFLD diagnosis
|
Prevalence of NAFLD in IBS/ IBS in NAFLD
|
Outcomes
|
| Hasanain et al[15] | IBS | Cross sectional study | 100 patients with IBS | Rome III | IBS-C: 45%; IBS-D: 23%; IBS-M: 32%, | Ultrasound; No history of alcohol exposure; No exposure to steatogenic medications; Negative viral screen | 74% of those with IBS had co-existing NAFLD | Moderate/severe NAFLD significantly associated with moderate/severe IBS (OR: 2.4, 95%CI: 1.3-62.7, P = 0.026) |
| Shin et al[16] | Healthy individuals via NHANES | Cross sectional study | 2345 patients with IBS | Rome IV | IBS-C: 1023; IBS-D: 1322 | Raised ALT or AST; Absence of excessive alcohol; Negative viral hepatitis screen | Prevalence of NAFLD in IBS-D: 12.9% (95%CI: 9.8-15.9); IBS-C: 9.0% (95%CI: 7.0-11.0) | NAFLD associated with diarrhoea vs normal bowel pattern (OR: 1.340, 95%CI: 1.007-1.784) and constipation (OR: 1.445, 95%CI: 1.028-2.031) |
| Arasteh et al[17] | IBS | Cohort study | 1067 patients with IBS | Rome IV | IBS-D: 57 (5.3%); IBS-C: 380 (35.6%); IBS-U: 630 (59%) | Not documented | 3.7% | Liver disease not associated with IBS (Coefficient: 0.26, OR: 1.30, 95%CI: 0.92-1.82) |
| Lee et al[19] | IBS vs control | Retrospective, cross sectional, case control study | 83 IBS patients; 260 age and sex matched control | Rome III | IBS-C: 14.8%; IBS-D: 49.4%; IBS-M: 31.3%; IBS-U: 4.5% | Investigated raised ALT, GGT, AST and features of metabolic syndrome | 16.9% of IBS patients had raised ALT; 24.1% had raised GGT | Significantly higher ALT in patients with IBS (16.9% vs 7.7%; P = 0.015); Significantly higher GGT in patients with IBS (24.1% vs 11.5%; P = 0.037); Significantly higher prevalence of metabolic syndrome in patients with IBS (32.5% vs 12.7%; P < 0.001) |
| Sarmini et al[73] | IBS vs control | Observational study | 637942 | Clinical diagnosis | Not documented | Not documented | Not available | Patients with IBS significantly more likely to develop NAFLD compared to non-IBS group (OR: 3.204, 95%CI: 3.130-3.279, P < 0.001) |
| Singh et al[24] | NAFLD | Retrospective analysis | 632 | Clinical diagnosis | Not documented | Ultrasound; Alcohol consumption < 20 g/d; Normal aetiological liver screen | 186 (29.4%) patients with NAFLD had clinical diagnosis of IBS | IBS symptoms are highly prevalent in those with NAFLD |
| Jones-Pauley et al[22] | NAFLD | Cross-sectional study | 130 | Rome IV | Not documented | Not documented | 38 (29.2%) patients with NAFLD met Rome IV IBS criteria | High prevalence of IBS in patients with NAFLD; Significant increase in prevalence of depression (18.4% vs 5.4%, P = 0.01) and anxiety (31.6% vs 9.8%, P = 0.002) in those with co-existing IBS compared to those with NAFLD without IBS |
IBS: Irritable bowel syndrome; NAFLD: Non-alcoholic fatty liver disease; IBS-C: Constipation predominant IBS; IBS-D: Diarrhoea predominant IBS; IBS-M: Mixed IBS; IBS-U: Unsubtyped IBS; OR: Odds ratio; CI: Cumulative incidence; ALT: Alanine aminotransferase; AST: Aspartate aminotransferase; GGT: Gamma-glutamyl transferase.
Shin et al[16] found that the prevalence of presumed NAFLD was 12.9% in patients with diarrhoea predominant IBS (IBS-D) compared to 9.0% in patients with constipation predominant IBS (IBS-C), although the reasons for this apparent difference are unclear and merit further investigation. In an interesting study by Lee et al[19], rather than evaluating patients with a formal diagnosis of NAFLD, the authors assessed the incidence of elevated liver transaminases and the metabolic syndrome in patients with IBS, compared to an age and sex matched control group. Those with IBS were found to have a significantly higher alanine aminotransferase (ALT) (16.9% vs 7.7%; P = 0.015) and Gamma-glutamyl transferase (GGT) (24.1% vs 11.5%; P = 0.037) compared to the control group, and there was a significantly higher prevalence of metabolic syndrome in the IBS group (32.5% vs 12.7%; P < 0.001).
To our knowledge, there have only been three previous reports on the incidence of functional bowel symptoms in patients with NAFLD. Appleby et al[20] found that in 127 patients with NAFLD, 25% had chronic diarrhea, and 12% had features of bile acid diarrhoea with both being associated with a raised NAFLD fibrosis score. Furthermore, Singh et al[21] studied 632 patients in India diagnosed with fatty liver disease and found that 29.4% had co-existing clinical features of IBS. Similar findings were reported by Jones-Pauley et al[22] in a cross sectional study looking at IBS diagnosed by Rome IV criteria in 130 NAFLD patients and as many as 38 (29.2%) patients had IBS based on Rome IV criteria. Interestingly, depression and anxiety were found to be more prevalent in the IBS cohort, compared to the non-IBS cohort, indicating the detrimental effect of co-existing bowel symptoms may have on quality of life, and the resulting need for a multi-systems approach in NAFLD patients with IBS symptoms.
In summary, regardless of the iteration of the Rome IBS diagnostic criteria used and the highlighted limitations of the previous studies, the data summarized in Table 1 on the co-existing prevalence of IBS in patients with NAFLD consistently report a much higher prevalence of IBS than that reported in global prevalence studies using either Rome III or Rome IV diagnostic criteria[2].
OVERLAPPING ETIOLOGICAL FACTORS IBS AND NAFLD
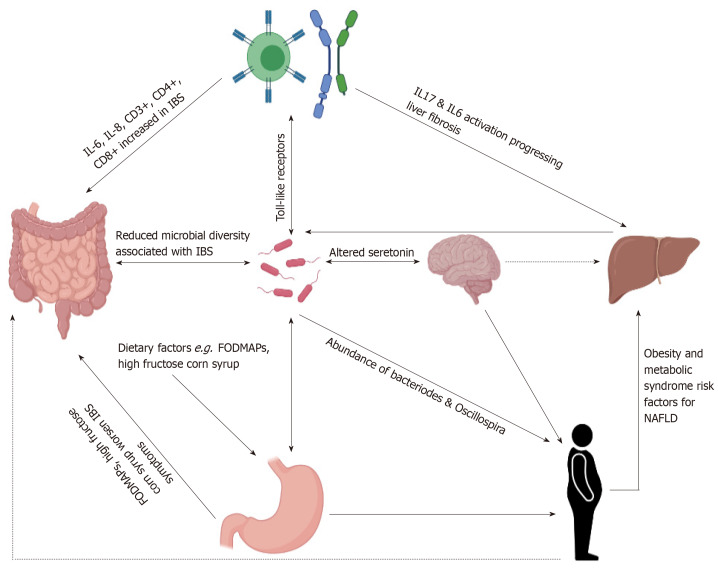
Multiple etiological factors overlap between IBS and NAFLD leading to interest in possible associations including obesity, gut microbiome, dietary factors and immune mediated causes as illustrated in Figure 1.
Figure 1.
Schematic illustration summarizing associations and co-existing etiologies of irritable bowel syndrome and non-alcoholic fatty liver disease. IBS: Irritable bowel syndrome; NAFLD: Non-alcoholic fatty liver disease; IL: Interleukin.
OBESITY
NAFLD is intrinsically linked with obesity, diabetes and the metabolic syndrome. In obese populations, NAFLD has a prevalence of up to 95%[23]. Excess adipose tissue exhausting peripheral storage capacity resulting in deposition in the liver and increased insulin resistance is thought to be the main culprit for NAFLD pathogenesis[24]. Weight loss through diet and exercise reduces hepatic steatosis and fibrosis, and in 109 obese patients[25]. Lassailly et al[26] showed that bariatric surgery resolved non-alcoholic steatohepatitis (NASH) within a year.
The association between IBS and obesity is more unclear[27]. Aro et al[28] found a significant association between the obesity and IBS symptoms such as abdominal pain and diarrhoea using the Abdominal Symptom Questionnaire as well as a positive association between obesity and a formal diagnosis of IBS. However, these have not been confirmed in several other studies[29-31]. Interestingly, Lee et al[30] found visceral abdominal adiposity was associated with increased risk of IBS-D. There is evidence that IBS is more prevalent in patients who are obese[32]. Schneck et al[33] described a cohort of patients with obesity undergoing bariatric surgery of which 30% fulfilled Rome III criteria for IBS. Further evidence for role of obesity in IBS is supported by the observation that increased visceral adiposity enhances perception of luminal stimuli, dysmotility and abdominal pain[34]. Higher body mass indexes have been associated with accelerated colonic and rectosigmoid transit and increased stool frequency[35]. Furthermore, weight loss through diet or bariatric surgery has been shown to improve symptoms[32,36]. Aasbrenn et al[37] prospectively analyzed the effect of a weight loss program on bowel symptoms using the IBS severity scoring system (IBS-SSS) and Gastrointestinal Symptom Rating Scale and found that there were significant improvements in the IBS-SSS in patients with IBS compared to those without.
MICROBIOME
The gut microbiota plays a vital role in the intestinal barrier function, metabolism of nutrients and development of immune tolerance and response. Dysregulation of the microbiome has been shown to be a component for the development of both NAFLD and IBS[38].
Long-term perturbation of the gut microbiota has been shown to contribute to metabolic syndrome and fatty liver disease[39]. Several mechanisms have been proposed on how the gut microbiota results in NAFLD development. This includes increased intestinal permeability leading greater lipopolysaccharide exposure to the host. This, in turn, results in toll like receptor (predominantly TLR4) activation of the innate immune system, causing liver inflammation as they are transported from the gut to the liver. Additionally, microbially produced metabolites, such as lactate and ethanol, can directly activate inflammatory cascades within the liver. Enterohepatic bile acid homeostasis is important for multiple processes, including fat absorption, inflammation, immunity and microbial diversity. Significant differences have been noted in bile acid composition in metabolic diseases associating with progression of NAFLD[38,40].
Patients with hepatic steatosis and NASH have been shown to have increased Proteobacteria, Enterobacteriaceae, Escherichia and Citrobacter with reductions in abundance of Rikenellaceae, Ruminococcaceae, Anaerosporobacter and Coprococcus[39,40] Reductions in Bifidobacteria have also been observed and Bifidobacteria possibly reduce gut wall permeability to lipopolysaccherides, suggesting a relationship with the development of disease[39]. Interestingly, Frost et al[39] followed up patients who had incidental findings of fatty liver or diabetes and found changes in Clostridium XIVa as a result of dysbiosis with a strong association for increasing fatty acid biosynthesis. Type 2 diabetes is also noted to result in increased gut permeability. Aron-Wisnewsky et al[40] found significant overlap in microbial signatures between patients with NAFLD and NASH with obesity and diabetes, finding changes in abundance of Oscillospira and Bacteriodes. Further evidence on the importance of the gut microbiome in metabolic syndrome, is shown by fecal microbiota transplant being associated with a temporary improvement in peripheral insulin resistance[41].
Changes in intestinal microbial diversity is also thought to contribute to the development of IBS as the microbiota impacts on intestinal motility and sensitivity. Some patients with IBS have been shown to have changes in the Fermicutes-to-Bacteriodes ratio, reduced lactobacilli and bifidobacterial as well as reduced microbial diversity[38,42].
The gut-brain-microbiome axis is known to have an important role in glucose regulation. Gut microbiota modulation produces changes in the immune, neurotransmitter and monoaminergic activity of this axis. Serotonin secretion affects motility, pain perception but also plays a role in mood control[43]. NAFLD and the gut-brain axis may also be inter-related. There is evidence that depression is associated with NAFLD. However, disentangling the multiple contributors to depression in multi-factorial disease states (as often seen in patients with metabolic syndrome) can be exceptionally difficult[44,45].
Dysregulation of the microbiome itself can lead to poor glycaemic control, acting through nitric oxide formation which affects the neuronal response to gut hormone Glucagon-like peptide-1 (GLP-1)[46]. The GLP-1 receptor antagonist, Semaglutide, has been shown to reduce liver fat and NASH resolution in patients with NAFLD[47]. It has also been used to treat weight loss and type-2 diabetes mellitus[48]. Given the known functions of GLP-1 on the gut microbiota, the effect seen in these studies may well be related to beneficial alterations in microbiome composition[49].
DIETARY FACTORS
Dietary factors have been shown to be integral to the management of both IBS and NAFLD. Weight loss through diet and exercise is the mainstay of NAFLD management. Adherence to a Mediterranean diet reduces hepatic steatosis and achieves a greater weight loss in patients with NAFLD[50]. By contrast, patients with IBS have been shown to have a poorer adherence to a Mediterranean diet than healthy controls[50], a dietary factor which may therefore be relevant in the development of NAFLD in those with IBS. There is also some evidence that conservative weight loss can help IBS symptoms. Aasbrenn et al[37] found that a weight loss program resulted in a significant improvement in IBS symptoms as assessed by IBS-SSS questionnaires and Gastrointestinal Symptom Rating Scale[37].
Certain food groups appear to worsen IBS symptoms and contribute to NAFLD development. High fructose corn syrup (HFCS) is a disaccharide which is frequently used in artificial sweeteners, processed, canned and baked goods worldwide. HFCS has been shown to induce IBS symptoms through increased osmotic pressure and bacterial fermentation resulting in gas production, abdominal bloating and pain[51]. HFCS has also been shown to downregulate the insulin signaling pathway which would contribute to the pathogenesis of NAFLD[52]. Fructose consumption has also been shown to increase intestinal permeability potentially leading to the development of both NAFLD and IBS through the processes already outlined[53].
Certainly more research into the dietary implications on NAFLD and IBS is needed. Many patients with IBS notice that ‘healthy’ foods such as fruit and vegetables can make their symptoms worse and this results in some of them adopting a more ‘unhealthy’ diet which may lead to weight gain. There is evidence that a low FODMAP diet with excludes some fruits and vegeatables improves IBS symptoms however to the authors’ knowledge, there is a paucity of data on the effects of a low FODMAP diet on the progression of NAFLD.
IMMUNE MEDIATED FACTORS
Chronic inflammation is a critical driver of progressive disease in NAFLD and significant advances have been made to understand the role of inflammation[54,55]. The role of toll-like receptors (TLRs) and macrophage activation has already been discussed. Additionally, Natural killer cells and natural killer T cells contribute to inflammation by releasing cytokines and reactive oxygen species[56]. Tumor necrosis factor (TNF)-α, alongside other cytokines and growth factors, have also been shown to possible have a role in the development of NAFLD and NASH, in both animals and humans[38]. TNF-α in combination with interleukin (IL)-6 stimulates the production of leptin activating neutrophils and the innate immune system[38]. In addition, adaptive immune responses drive NASH as hepatic infiltration of B cells and CD4 and CD8 T cells exacerbate parenchymal injury and inflammation[56]. B cells play a profibrogenic role involving the stimulation of hepatic stellate cells and liver macrophages[57]. CD4+ T cells differentiate to type-17 T helper cells, producing IL-17 which has been implicated in the progression of NAFLD[58]. The balance of the adaptive immune cellular compartment within the liver can transition from a pro-resolution composition to pro-inflammatory subset, driving disease and fibrosis.
In IBS, a similar chronic low-grade inflammatory picture has also been described. The innate immune system is implicated with an increased number of mast cells throughout the intestines in some patients[59]. The adaptive immune response is also important with CD3+, CD4+ and CD8+ T cells increased in intestines and blood of patients with IBS[38]. Interestingly, an increase in IL-6 and IL-8 with reduced anti-inflammatory cytokines has been seen in serum of IBS patients[59]. The role of TLRs is also felt to be important with IL-6 and other cytokines acting through this mechanism[38]. TNF-α can act on the nervous system to cause hypersensitivity, gastric hypomotility and nausea[59].
SMALL INTESTINAL BACTERIAL OVERGROWTH
Small intestinal bacterial overgrowth (SIBO) can cause abdominal pain, bloating and chronic diarrhea. Although an area of controversy due to conflicting evidence, a number of previous studies have suggested that some patients with IBS have a relatively high prevalence of SIBO[60,61]. A recent metanalysis has shown that patients with IBS were more likely to test positive for SIBO than healthy controls[61]. Further circumstantial evidence for the gut-brain-microbiome-liver axis can be drawn from the effects of the non-absorbable antibiotic Rifaximin in both IBS and in liver disease. Whilst the mechanism is unclear, improvement in IBS symptoms have been demonstrated in patients in randomized controlled trials of Rifaximin[62,63]. Rifaximin is also often used to treat SIBO[64], a condition which has been shown to affect cognitive function in a subset of patients who present with brain fog[65]. Interestingly, treatment with Rifaximin has recently been shown in brain imaging studies to alter neuronal connectivity and increase cognitive flexibility through its effect on the gut microbiome particularly in beta and theta frequencies with a particular focus on the insular cortex, a region known to be affected in patients with IBS[66]. Furthermore, Rifaximin has an immunomodulatory action counteracting the pro-inflammatory response seen in gut microbiota dysbiosis[67]. In liver disease, Rifaximin is an established treatment for hepatic encephalopathy, with its effects attributed to alterations in the gut microbiome and resultant positive effects on cognitive function. Specifically in patients with biopsy proven NASH, Rifaximin has also been shown to reduce insulin resistance, inflammation and NAFLD fat scores[68]. Therefore, the effects of Rifaximin are multifactorial including reduced endotoxemia, modulation of inflammatory cytokines, and intestinal permeability as well as changing functional brain connectivity[62,66].
Further overlapping evidence for SIBO in this context comes from the obesity literature. There is evidence that obesity reduces gut motility, which may predispose to SIBO due to stasis, and plausibly this is thought to damage barrier function, which can result in bacterial translocation and altered gut-liver axis[53]. Furthermore, changes in the gut-liver axis may well a result of increased intestinal permeability. A high prevalence of SIBO has been observed in obese subjects however the association between NAFLD and SIBO is less clear[53]. Studies have found the prevalence of SIBO in NAFLD to range from 39%-60% albeit in small numbers of patients. However, more recently, some research found 8% of NAFLD patients in their cohort had SIBO and there was no evidence that SIBO was associated with a higher risk of fibrosis[69-71].
BILE ACID DIARRHOEA
Bile acid malabsorption is a cause of chronic diarrhea and has been shown to be associated with an increased NAFLD fibrosis score. Hepatic bile acid production is regulated by Fibroblast growth factor 19 (FGF19) and Farnesoid-X-receptor (FXR) and obeticholic acid (a FXR agonist) has shown therapeutic potential in both bile acid related diarrhea and NAFLD[20]. Appleby et al[20] found that increased hepatic bile acid production and diarrhea were associated with an increased NAFLD score. Of further relevance to the link with NAFLD, bile acid diarrhoea has also been shown to be associated with raised body mass index[72]. This is therefore an important point to be considered in clinical practice when evaluating patients with suspected overlapping IBS and NAFLD, as up to a third of patients meeting the criteria for IBS-D have been shown to have bile salt malabsorption when investigated[72], and this condition should therefore be excluded in the context of watery diarrhea.
APPLICABILITY TO CLINICAL PRACTICE
Pulling this together, there is consistent evidence to show that IBS and NAFLD have a similar pathogenesis and therefore applying this to clinical practice, physicians should be aware that NAFLD may co-exist silently in patients with IBS and vice versa. Patients with IBS and incidental findings of elevated liver enzymes or with risk factors for NAFLD should be considered for non-invasive liver screening through ultrasound and appropriately available non-invasive fibrosis assessment using FIB-4 scoring, enhanced liver fibrosis testing or mechanical liver stiffness measurement.
Conversely, patients with NAFLD may not admit to the debilitating symptoms of IBS due to stigma or feeling that their symptoms are not relevant to their liver consultation. Screening for positive clinical features of IBS and targeted treatment for both conditions in unison may aid compliance with treatment, improve quality of life and ultimately improve morbidity.
However, as highlighted in this review, there is a lack of large, high quality cross-sectional data on the incidence of IBS in NAFLD patients and vice versa. To date, studies have been limited to the use of ultrasound and blood tests to diagnose NAFLD, however there is a lack of data that quantifies a fibrosis score which may be useful to correlate with IBS severity. From the currently available data (summarized in Table 1), whilst there is a suggestion that the IBS-D sub-type may be more common than IBS-C in patients with NAFLD, whether this is a genuine finding merits further evaluation in studies which have excluded bile salt malabsorption with appropriate investigations given its apparent independent association with NAFLD.
CONCLUSION
IBS and NAFLD are common conditions that can have significant effects on both physical and mental health[73], as well as significant healthcare and socioeconomic implications. There is some evidence that patients with IBS are more likely to develop NAFLD, and there are multiple different pathophysiological mechanisms that could contribute to both conditions, however more data is needed. Until such data clarifies this picture, the possibility of these conditions existing concomitantly should be considered proactively and investigated appropriately.
Footnotes
Conflict-of-interest statement: None of the authors have any conflicts of interest to disclose.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Corresponding Author's Membership in Professional Societies: British Society of Gastroenterology; American Gastroenterological Association.
Peer-review started: April 17, 2021
First decision: July 27, 2021
Article in press: October 27, 2021
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: United Kingdom
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Du Y S-Editor: Wang JJ L-Editor: A P-Editor: Wang JJ
Contributor Information
Huw Purssell, Hepatology, Manchester University NHS Foundation Trust, Manchester M23 9LT, United Kingdom; Division of Diabetes, Endocrinology and Gastroenterology, University of Manchester, Manchester M23 9LT, United Kingdom.
Peter J Whorwell, Division of Diabetes, Endocrinology and Gastroenterology, University of Manchester, Manchester M23 9LT, United Kingdom; Neurogastroenterology Unit, Wythenshawe Hospital, Manchester University NHS Foundation Trust, Manchester M23 9LT, United Kingdom.
Varinder S Athwal, Hepatology, Manchester University NHS Foundation Trust, Manchester M23 9LT, United Kingdom; Division of Diabetes, Endocrinology and Gastroenterology, University of Manchester, Manchester M23 9LT, United Kingdom.
Dipesh H Vasant, Division of Diabetes, Endocrinology and Gastroenterology, University of Manchester, Manchester M23 9LT, United Kingdom; Neurogastroenterology Unit, Department of Gastroenterology, Manchester University NHS Foundation Trust, Wythenshawe Hospital, Manchester M23 9LT, United Kingdom. dipesh.vasant@manchester.ac.uk.
References
- 1.Mearin F, Lacy BE, Chang L, Chey WD, Lembo AJ, Simren M, Spiller R. Bowel Disorders. Gastroenterology. 2016 doi: 10.1053/j.gastro.2016.02.031. [DOI] [PubMed] [Google Scholar]
- 2.Oka P, Parr H, Barberio B, Black CJ, Savarino EV, Ford AC. Global prevalence of irritable bowel syndrome according to Rome III or IV criteria: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2020;5:908–917. doi: 10.1016/S2468-1253(20)30217-X. [DOI] [PubMed] [Google Scholar]
- 3.Shivaji UN, Ford AC. Prevalence of functional gastrointestinal disorders among consecutive new patient referrals to a gastroenterology clinic. Frontline Gastroenterol. 2014;5:266–271. doi: 10.1136/flgastro-2013-100426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zamani M, Alizadeh-Tabari S, Zamani V. Systematic review with meta-analysis: the prevalence of anxiety and depression in patients with irritable bowel syndrome. Aliment Pharmacol Ther. 2019;50:132–143. doi: 10.1111/apt.15325. [DOI] [PubMed] [Google Scholar]
- 5.Atarodi S, Rafieian S, Whorwell PJ. Faecal incontinence-the hidden scourge of irritable bowel syndrome: a cross-sectional study. BMJ Open Gastroenterol. 2014;1:e000002. doi: 10.1136/bmjgast-2014-000002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dixon-Woods M, Critchley S. Medical and lay views of irritable bowel syndrome. Fam Pract. 2000;17:108–113. doi: 10.1093/fampra/17.2.108. [DOI] [PubMed] [Google Scholar]
- 7.Hearn M, Whorwell PJ, Vasant DH. Stigma and irritable bowel syndrome: a taboo subject? Lancet Gastroenterol Hepatol. 2020;5:607–615. doi: 10.1016/S2468-1253(19)30348-6. [DOI] [PubMed] [Google Scholar]
- 8.Blachier M, Leleu H, Peck-Radosavljevic M, Valla DC, Roudot-Thoraval F. The burden of liver disease in Europe: a review of available epidemiological data. J Hepatol. 2013;58:593–608. doi: 10.1016/j.jhep.2012.12.005. [DOI] [PubMed] [Google Scholar]
- 9.Targher G, Byrne CD, Tilg H. NAFLD and increased risk of cardiovascular disease: clinical associations, pathophysiological mechanisms and pharmacological implications. Gut. 2020;69:1691–1705. doi: 10.1136/gutjnl-2020-320622. [DOI] [PubMed] [Google Scholar]
- 10.Roulot D. Screening for Liver Fibrosis in General or At-Risk populations using Transient Elastrography. Liver Elastrography. Springer Nature, 2020: 545-550. [Google Scholar]
- 11.Harman DJ, Ryder SD, James MW, Jelpke M, Ottey DS, Wilkes EA, Card TR, Aithal GP, Guha IN. Direct targeting of risk factors significantly increases the detection of liver cirrhosis in primary care: a cross-sectional diagnostic study utilising transient elastography. BMJ Open. 2015;5:e007516. doi: 10.1136/bmjopen-2014-007516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Public Health England. Liver disease: applying All Our Health. [cited 23 March 2021]. Available from: https://www.gov.uk/government/publications/liver-disease-applying-all-our-health/liverdisease-applying-all-our-health .
- 13.Cotter TG, Charlton M. Nonalcoholic Steatohepatitis After Liver Transplantation. Liver Transpl. 2020;26:141–159. doi: 10.1002/lt.25657. [DOI] [PubMed] [Google Scholar]
- 14.Haldar D, Kern B, Hodson J, Armstrong MJ, Adam R, Berlakovich G, Fritz J, Feurstein B, Popp W, Karam V, Muiesan P, O'Grady J, Jamieson N, Wigmore SJ, Pirenne J, Malek-Hosseini SA, Hidalgo E, Tokat Y, Paul A, Pratschke J, Bartels M, Trunecka P, Settmacher U, Pinzani M, Duvoux C, Newsome PN, Schneeberger S European Liver and Intestine Transplant Association (ELITA) Outcomes of liver transplantation for non-alcoholic steatohepatitis: A European Liver Transplant Registry study. J Hepatol. 2019;71:313–322. doi: 10.1016/j.jhep.2019.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hasanain AF, Abdel-Rahman ME, Ali AM, Abdel-Aal SM. Nonalcoholic fatty liver disease among patients with irritable bowel syndrome: prevalence and contribution to disease severity. Gastroenterol Hepatol Endoscopy. 2018;3 (3):1–4. [Google Scholar]
- 16.Shin A, Xu H, Imperiale TF. Associations of chronic diarrhoea with non-alcoholic fatty liver disease and obesity-related disorders among US adults. BMJ Open Gastroenterol. 2019;6:e000322. doi: 10.1136/bmjgast-2019-000322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Arasteh P, Maharlouei N, Eghbali SS, Amini M, Lankarani KB, Malekzadeh R. A Comprehensive Look at Irritable Bowel Syndrome and its Associated Factors Considering the Rome IV Criteria: A Penalized Smoothly Clipped Absolute Deviation Regression Approach in the Pars Cohort Study. Middle East J Dig Dis. 2018;10:149–159. doi: 10.15171/mejdd.2018.104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Vasant DH. Global prevalence of irritable bowel syndrome: time to consider factors beyond diagnostic criteria? Lancet Gastroenterol Hepatol. 2020;5:879–880. doi: 10.1016/S2468-1253(20)30211-9. [DOI] [PubMed] [Google Scholar]
- 19.Lee SH, Kim KN, Kim KM, Joo NS. Irritable Bowel Syndrome May Be Associated with Elevated Alanine Aminotransferase and Metabolic Syndrome. Yonsei Med J. 2016;57:146–152. doi: 10.3349/ymj.2016.57.1.146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Appleby RN, Moghul I, Khan S, Yee M, Manousou P, Neal TD, Walters JRF. Non-alcoholic fatty liver disease is associated with dysregulated bile acid synthesis and diarrhea: A prospective observational study. PLoS One. 2019;14:e0211348. doi: 10.1371/journal.pone.0211348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Singh SP, Kar SK, Panigrahi MK, Misra B, Pattnaik K, Bhuyan P, Meher C, Agrawal O, Rout N, Swain M. Profile of patients with incidentally detected nonalcoholic fatty liver disease (IDNAFLD) in coastal eastern India. Trop Gastroenterol. 2013;34:144–152. doi: 10.7869/tg.118. [DOI] [PubMed] [Google Scholar]
- 22.Jones-Pauley M, Franco L, Tamimi O, Nashatian L, Victor D, Quigley EM. Mo1503 Irritable bowel syndrome symptoms in non-alcoholic fatty liver disease patients are an indicator of depression and anxiety. Gastroenterology. 2020;158 (6):S–1429. doi: 10.1097/MCG.0000000000001770. [DOI] [PubMed] [Google Scholar]
- 23.Pallayova M, Taheri S. Non-alcoholic fatty liver disease in obese adults: clinical aspects and current management strategies. Clin Obes. 2014;4:243–253. doi: 10.1111/cob.12068. [DOI] [PubMed] [Google Scholar]
- 24.Godoy-Matos AF, Silva Júnior WS, Valerio CM. NAFLD as a continuum: from obesity to metabolic syndrome and diabetes. Diabetol Metab Syndr. 2020;12:60. doi: 10.1186/s13098-020-00570-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Marchesini G, Petta S, Dalle Grave R. Diet, weight loss, and liver health in nonalcoholic fatty liver disease: Pathophysiology, evidence, and practice. Hepatology. 2016;63:2032–2043. doi: 10.1002/hep.28392. [DOI] [PubMed] [Google Scholar]
- 26.Lassailly G, Caiazzo R, Buob D, Pigeyre M, Verkindt H, Labreuche J, Raverdy V, Leteurtre E, Dharancy S, Louvet A, Romon M, Duhamel A, Pattou F, Mathurin P. Bariatric Surgery Reduces Features of Nonalcoholic Steatohepatitis in Morbidly Obese Patients. Gastroenterology. 2015;149:379–88; quiz e15. doi: 10.1053/j.gastro.2015.04.014. [DOI] [PubMed] [Google Scholar]
- 27.Pickett-Blakely O. Obesity and irritable bowel syndrome: a comprehensive review. Gastroenterol Hepatol (N Y) 2014;10:411–416. [PMC free article] [PubMed] [Google Scholar]
- 28.Aro P, Ronkainen J, Talley NJ, Storskrubb T, Bolling-Sternevald E, Agréus L. Body mass index and chronic unexplained gastrointestinal symptoms: an adult endoscopic population based study. Gut. 2005;54:1377–1383. doi: 10.1136/gut.2004.057497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Akhondi N, Memar Montazerin S, Soltani S, Saneei P, Hassanzadeh Keshteli A, Esmaillzadeh A, Adibi P. General and abdominal obesity in relation to the prevalence of irritable bowel syndrome. Neurogastroenterol Motil. 2019;31:e13549. doi: 10.1111/nmo.13549. [DOI] [PubMed] [Google Scholar]
- 30.Lee CG, Lee JK, Kang YS, Shin S, Kim JH, Lim YJ, Koh MS, Lee JH, Kang HW. Visceral abdominal obesity is associated with an increased risk of irritable bowel syndrome. Am J Gastroenterol. 2015;110:310–319. doi: 10.1038/ajg.2014.422. [DOI] [PubMed] [Google Scholar]
- 31.Talley NJ, Howell S, Poulton R. Obesity and chronic gastrointestinal tract symptoms in young adults: a birth cohort study. Am J Gastroenterol. 2004;99:1807–1814. doi: 10.1111/j.1572-0241.2004.30388.x. [DOI] [PubMed] [Google Scholar]
- 32.Pugliese G, Muscogiuri G, Barrea L, Laudisio D, Savastano S, Colao A. Irritable bowel syndrome: a new therapeutic target when treating obesity? Hormones (Athens) 2019;18:395–399. doi: 10.1007/s42000-019-00113-9. [DOI] [PubMed] [Google Scholar]
- 33.Schneck AS, Anty R, Tran A, Hastier A, Amor IB, Gugenheim J, Iannelli A, Piche T. Increased Prevalence of Irritable Bowel Syndrome in a Cohort of French Morbidly Obese Patients Candidate for Bariatric Surgery. Obes Surg. 2016;26:1525–1530. doi: 10.1007/s11695-015-1907-0. [DOI] [PubMed] [Google Scholar]
- 34.Emerenziani S, Guarino MPL, Trillo Asensio LM, Altomare A, Ribolsi M, Balestrieri P, Cicala M. Role of Overweight and Obesity in Gastrointestinal Disease. Nutrients. 2019;12 doi: 10.3390/nu12010111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sadik R, Björnsson E, Simrén M. The relationship between symptoms, body mass index, gastrointestinal transit and stool frequency in patients with irritable bowel syndrome. Eur J Gastroenterol Hepatol. 2010;22:102–108. doi: 10.1097/MEG.0b013e32832ffd9b. [DOI] [PubMed] [Google Scholar]
- 36.Clements RH, Gonzalez QH, Foster A, Richards WO, McDowell J, Bondora A, Laws HL. Gastrointestinal symptoms are more intense in morbidly obese patients and are improved with laparoscopic Roux-en-Y gastric bypass. Obes Surg. 2003;13:610–614. doi: 10.1381/096089203322190835. [DOI] [PubMed] [Google Scholar]
- 37.Aasbrenn M, Lydersen S, Farup PG. A Conservative Weight Loss Intervention Relieves Bowel Symptoms in Morbidly Obese Subjects with Irritable Bowel Syndrome: A Prospective Cohort Study. J Obes. 2018;2018:3732753. doi: 10.1155/2018/3732753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Scalera A, Di Minno MN, Tarantino G. What does irritable bowel syndrome share with non-alcoholic fatty liver disease? World J Gastroenterol. 2013;19:5402–5420. doi: 10.3748/wjg.v19.i33.5402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Frost F, Kacprowski T, Rühlemann M, Pietzner M, Bang C, Franke A, Nauck M, Völker U, Völzke H, Dörr M, Baumbach J, Sendler M, Schulz C, Mayerle J, Weiss FU, Homuth G, Lerch MM. Long-term instability of the intestinal microbiome is associated with metabolic liver disease, low microbiota diversity, diabetes mellitus and impaired exocrine pancreatic function. Gut. 2021;70:522–530. doi: 10.1136/gutjnl-2020-322753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Aron-Wisnewsky J, Vigliotti C, Witjes J, Le P, Holleboom AG, Verheij J, Nieuwdorp M, Clément K. Gut microbiota and human NAFLD: disentangling microbial signatures from metabolic disorders. Nat Rev Gastroenterol Hepatol. 2020;17:279–297. doi: 10.1038/s41575-020-0269-9. [DOI] [PubMed] [Google Scholar]
- 41.Zhang Z, Mocanu V, Cai C, Dang J, Slater L, Deehan EC, Walter J, Madsen KL. Impact of Fecal Microbiota Transplantation on Obesity and Metabolic Syndrome-A Systematic Review. Nutrients. 2019;11 doi: 10.3390/nu11102291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bhattarai Y, Muniz Pedrogo DA, Kashyap PC. Irritable bowel syndrome: a gut microbiota-related disorder? Am J Physiol Gastrointest Liver Physiol. 2017;312:G52–G62. doi: 10.1152/ajpgi.00338.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Agustí A, García-Pardo MP, López-Almela I, Campillo I, Maes M, Romaní-Pérez M, Sanz Y. Interplay Between the Gut-Brain Axis, Obesity and Cognitive Function. Front Neurosci. 2018;12:155. doi: 10.3389/fnins.2018.00155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tomeno W, Kawashima K, Yoneda M, Saito S, Ogawa Y, Honda Y, Kessoku T, Imajo K, Mawatari H, Fujita K, Hirayasu Y, Nakajima A. Non-alcoholic fatty liver disease comorbid with major depressive disorder: The pathological features and poor therapeutic efficacy. J Gastroenterol Hepatol. 2015;30:1009–1014. doi: 10.1111/jgh.12897. [DOI] [PubMed] [Google Scholar]
- 45.Kim D, Yoo ER, Li AA, Tighe SP, Cholankeril G, Harrison SA, Ahmed A. Depression is associated with non-alcoholic fatty liver disease among adults in the United States. Aliment Pharmacol Ther. 2019;50:590–598. doi: 10.1111/apt.15395. [DOI] [PubMed] [Google Scholar]
- 46.Grasset E, Burcelin R. The gut microbiota to the brain axis in the metabolic control. Rev Endocr Metab Disord. 2019;20:427–438. doi: 10.1007/s11154-019-09511-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Newsome PN, Buchholtz K, Cusi K, Linder M, Okanoue T, Ratziu V, Sanyal AJ, Sejling AS, Harrison SA NN9931-4296 Investigators. A Placebo-Controlled Trial of Subcutaneous Semaglutide in Nonalcoholic Steatohepatitis. N Engl J Med. 2021;384:1113–1124. doi: 10.1056/NEJMoa2028395. [DOI] [PubMed] [Google Scholar]
- 48.Nauck MA, Meier JJ. MANAGEMENT OF ENDOCRINE DISEASE: Are all GLP-1 agonists equal in the treatment of type 2 diabetes? Eur J Endocrinol. 2019;181:R211–R234. doi: 10.1530/EJE-19-0566. [DOI] [PubMed] [Google Scholar]
- 49.Everard A, Cani PD. Gut microbiota and GLP-1. Rev Endocr Metab Disord. 2014;15:189–196. doi: 10.1007/s11154-014-9288-6. [DOI] [PubMed] [Google Scholar]
- 50.Kataria R, Pickett-Blakely O. The Mediterranean Diet in Gastrointestinal and liver diseases. Curr treatm opt gastroenterol. 2020;18:718–728. [Google Scholar]
- 51.Ikechi R, Fischer BD, DeSipio J, Phadtare S. Irritable Bowel Syndrome: Clinical Manifestations, Dietary Influences, and Management. Healthcare (Basel) 2017;5 doi: 10.3390/healthcare5020021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Collison KS, Saleh SM, Bakheet RH, Al-Rabiah RK, Inglis AL, Makhoul NJ, Maqbool ZM, Zaidi MZ, Al-Johi MA, Al-Mohanna FA. Diabetes of the liver: the link between nonalcoholic fatty liver disease and HFCS-55. Obesity (Silver Spring) 2009;17:2003–2013. doi: 10.1038/oby.2009.58. [DOI] [PubMed] [Google Scholar]
- 53.Paolella G, Mandato C, Pierri L, Poeta M, Di Stasi M, Vajro P. Gut-liver axis and probiotics: their role in non-alcoholic fatty liver disease. World J Gastroenterol. 2014;20:15518–15531. doi: 10.3748/wjg.v20.i42.15518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Gao B, Tsukamoto H. Inflammation in Alcoholic and Nonalcoholic Fatty Liver Disease: Friend or Foe? Gastroenterology. 2016;150:1704–1709. doi: 10.1053/j.gastro.2016.01.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Pellicoro A, Ramachandran P, Iredale JP, Fallowfield JA. Liver fibrosis and repair: immune regulation of wound healing in a solid organ. Nat Rev Immunol. 2014;14:181–194. doi: 10.1038/nri3623. [DOI] [PubMed] [Google Scholar]
- 56.Sutti S, Albano E. Adaptive immunity: an emerging player in the progression of NAFLD. Nat Rev Gastroenterol Hepatol. 2020;17:81–92. doi: 10.1038/s41575-019-0210-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Thapa M, Chinnadurai R, Velazquez VM, Tedesco D, Elrod E, Han JH, Sharma P, Ibegbu C, Gewirtz A, Anania F, Pulendran B, Suthar MS, Grakoui A. Liver fibrosis occurs through dysregulation of MyD88-dependent innate B-cell activity. Hepatology. 2015;61:2067–2079. doi: 10.1002/hep.27761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Tang Y, Bian Z, Zhao L, Liu Y, Liang S, Wang Q, Han X, Peng Y, Chen X, Shen L, Qiu D, Li Z, Ma X. Interleukin-17 exacerbates hepatic steatosis and inflammation in non-alcoholic fatty liver disease. Clin Exp Immunol. 2011;166:281–290. doi: 10.1111/j.1365-2249.2011.04471.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ghoshal UC. Marshall and Warren Lecture 2019: A paradigm shift in pathophysiological basis of irritable bowel syndrome and its implication on treatment. J Gastroenterol Hepatol. 2020;35:712–721. doi: 10.1111/jgh.15032. [DOI] [PubMed] [Google Scholar]
- 60.Ford AC, Spiegel BM, Talley NJ, Moayyedi P. Small intestinal bacterial overgrowth in irritable bowel syndrome: systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2009;7:1279–1286. doi: 10.1016/j.cgh.2009.06.031. [DOI] [PubMed] [Google Scholar]
- 61.Ghoshal UC, Nehra A, Mathur A, Rai S. A meta-analysis on small intestinal bacterial overgrowth in patients with different subtypes of irritable bowel syndrome. J Gastroenterol Hepatol. 2020;35:922–931. doi: 10.1111/jgh.14938. [DOI] [PubMed] [Google Scholar]
- 62.Pimentel M, Lembo A, Chey WD, Zakko S, Ringel Y, Yu J, Mareya SM, Shaw AL, Bortey E, Forbes WP TARGET Study Group. Rifaximin therapy for patients with irritable bowel syndrome without constipation. N Engl J Med. 2011;364:22–32. doi: 10.1056/NEJMoa1004409. [DOI] [PubMed] [Google Scholar]
- 63.Meyrat P, Safroneeva E, Schoepfer AM. Rifaximin treatment for the irritable bowel syndrome with a positive lactulose hydrogen breath test improves symptoms for at least 3 months. Aliment Pharmacol Ther. 2012;36:1084–1093. doi: 10.1111/apt.12087. [DOI] [PubMed] [Google Scholar]
- 64.Pimentel M, Saad RJ, Long MD, Rao SSC. ACG Clinical Guideline: Small Intestinal Bacterial Overgrowth. Am J Gastroenterol. 2020;115:165–178. doi: 10.14309/ajg.0000000000000501. [DOI] [PubMed] [Google Scholar]
- 65.Rao SSC, Rehman A, Yu S, Andino NM. Brain fogginess, gas and bloating: a link between SIBO, probiotics and metabolic acidosis. Clin Transl Gastroenterol. 2018;9:162. doi: 10.1038/s41424-018-0030-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Sometti D, Ballan C, Wang H, Braun C, Enck P. Effects of the antibiotic rifaximin on cortical functional connectivity are mediated through insular cortex. Sci Rep. 2021;11:4479. doi: 10.1038/s41598-021-83994-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ponziani FR, Gerardi V, Pecere S, D'Aversa F, Lopetuso L, Zocco MA, Pompili M, Gasbarrini A. Effect of rifaximin on gut microbiota composition in advanced liver disease and its complications. World J Gastroenterol. 2015;21:12322–12333. doi: 10.3748/wjg.v21.i43.12322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Abdel-Razik A, Mousa N, Shabana W, Refaey M, Elzehery R, Elhelaly R, Zalata K, Abdelsalam M, Eldeeb AA, Awad M, Elgamal A, Attia A, El-Wakeel N, Eldars W. Rifaximin in nonalcoholic fatty liver disease: hit multiple targets with a single shot. Eur J Gastroenterol Hepatol. 2018;30:1237–1246. doi: 10.1097/MEG.0000000000001232. [DOI] [PubMed] [Google Scholar]
- 69.Miele L, Valenza V, La Torre G, Montalto M, Cammarota G, Ricci R, Mascianà R, Forgione A, Gabrieli ML, Perotti G, Vecchio FM, Rapaccini G, Gasbarrini G, Day CP, Grieco A. Increased intestinal permeability and tight junction alterations in nonalcoholic fatty liver disease. Hepatology. 2009;49:1877–1887. doi: 10.1002/hep.22848. [DOI] [PubMed] [Google Scholar]
- 70.Rafiei R, Bemanian M, Rafiei F, Bahrami M, Fooladi L, Ebrahimi G, Hemmat A, Torabi Z. Liver disease symptoms in non-alcoholic fatty liver disease and small intestinal bacterial overgrowth. Rom J Intern Med. 2018;56:85–89. doi: 10.1515/rjim-2017-0042. [DOI] [PubMed] [Google Scholar]
- 71.Lira MMP, de Medeiros Filho JEM, Baccin Martins VJ, da Silva G, de Oliveira Junior FA, de Almeida Filho ÉJB, Silva AS, Henrique da Costa-Silva J, de Brito Alves JL. Association of worsening of nonalcoholic fatty liver disease with cardiometabolic function and intestinal bacterial overgrowth: A cross-sectional study. PLoS One. 2020;15:e0237360. doi: 10.1371/journal.pone.0237360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Aziz I, Mumtaz S, Bholah H, Chowdhury FU, Sanders DS, Ford AC. High Prevalence of Idiopathic Bile Acid Diarrhea Among Patients With Diarrhea-Predominant Irritable Bowel Syndrome Based on Rome III Criteria. Clin Gastroenterol Hepatol. 2015;13:1650–5.e2. doi: 10.1016/j.cgh.2015.03.002. [DOI] [PubMed] [Google Scholar]
- 73.Sarmini MT, Asfari MM, Alomari M, Al-Khadra Y, Khoudari G, Dasarathy S, McCullough AJ. Assessing the relation between Irritable bowel syndrome and nonalcoholic liver disease. Am J Gastroenterol. 2019;114:S277–S278. [Google Scholar]