Abstract
Objective
To assess medium-term organ impairment in symptomatic individuals following recovery from acute SARS-CoV-2 infection.
Design
Baseline findings from a prospective, observational cohort study.
Setting
Community-based individuals from two UK centres between 1 April and 14 September 2020.
Participants
Individuals ≥18 years with persistent symptoms following recovery from acute SARS-CoV-2 infection and age-matched healthy controls.
Intervention
Assessment of symptoms by standardised questionnaires (EQ-5D-5L, Dyspnoea-12) and organ-specific metrics by biochemical assessment and quantitative MRI.
Main outcome measures
Severe post-COVID-19 syndrome defined as ongoing respiratory symptoms and/or moderate functional impairment in activities of daily living; single-organ and multiorgan impairment (heart, lungs, kidneys, liver, pancreas, spleen) by consensus definitions at baseline investigation.
Results
201 individuals (mean age 45, range 21–71 years, 71% female, 88% white, 32% healthcare workers) completed the baseline assessment (median of 141 days following SARS-CoV-2 infection, IQR 110–162). The study population was at low risk of COVID-19 mortality (obesity 20%, hypertension 7%, type 2 diabetes 2%, heart disease 5%), with only 19% hospitalised with COVID-19. 42% of individuals had 10 or more symptoms and 60% had severe post-COVID-19 syndrome. Fatigue (98%), muscle aches (87%), breathlessness (88%) and headaches (83%) were most frequently reported. Mild organ impairment was present in the heart (26%), lungs (11%), kidneys (4%), liver (28%), pancreas (40%) and spleen (4%), with single-organ and multiorgan impairment in 70% and 29%, respectively. Hospitalisation was associated with older age (p=0.001), non-white ethnicity (p=0.016), increased liver volume (p<0.0001), pancreatic inflammation (p<0.01), and fat accumulation in the liver (p<0.05) and pancreas (p<0.01). Severe post-COVID-19 syndrome was associated with radiological evidence of cardiac damage (myocarditis) (p<0.05).
Conclusions
In individuals at low risk of COVID-19 mortality with ongoing symptoms, 70% have impairment in one or more organs 4 months after initial COVID-19 symptoms, with implications for healthcare and public health, which have assumed low risk in young people with no comorbidities.
Trial registration number
Keywords: COVID-19, epidemiology, health policy, public health
Strengths and limitations of this study.
This is an ongoing, prospective, longitudinal COVID-19 recovery study with biochemical and imaging characterisation of organ function, starting in April 2020 before recognition of ‘long-COVID’, proper testing availability and prospective COVID-19-related research.
By recruiting ambulatory patients with broad inclusion criteria, we focused on a real-world population at lower risk of COVID-19 severity and mortality.
Healthy controls were included for comparison, not individuals with postinfluenza symptoms, COVID-19 without symptoms or from general clinics, which further studies may explore.
The study population was not ethnically diverse despite disproportionate COVID-19 impact in non-white individuals.
To limit interaction and exposure between the trial team and the patients, pulse oximetry, spirometry, MRI assessment of the brain and muscle function were not included from the outset.
Introduction
Early in the COVID-19 pandemic, research and clinical practice focused on pulmonary manifestations.1 There is increasing evidence for direct multiorgan effects,2–7 as well as indirect effects on other organ systems and disease processes, such as cardiovascular diseases and cancers, through changes in healthcare delivery and patient behaviours.8–10 The clear long-term impact on individuals and health systems underlines the urgent need for a whole body approach with assessment of all major organ systems following SARS-CoV-2 infection. Quantitative MRI has recently been used to show multiorgan impairment in individuals post-COVID-19 hospitalisation,11 but has not been used in non-hospitalised individuals.
COVID-19 is the convergence of an infectious disease, undertreated non-communicable diseases and social determinants of health, described as a ‘syndemic’.12 Pre-existing non-communicable diseases and risk factors predict poor COVID-19 outcomes, whether intensive care admission or mortality.10 Research has emphasised acute SARS-CoV-2 infection, hospitalised individuals and COVID-19 mortality,13–15 which is likely to underestimate the true burden of COVID-19-related disease. Among those surviving acute infection, 10% report persistent symptoms for 12 weeks or longer after initial infection (‘long-COVID’, or ‘post COVID-19 syndrome’, PCS).16 However, PCS is yet to be fully defined.17–20 Neither severity of symptoms, nor medium-term and long-term pathophysiology across organ systems, nor the appropriate control populations are understood.
UK government policies have emphasised excess mortality risk in moderate-risk and high-risk conditions, including ‘shielding’10 and commissioning of a risk calculator to identify those at highest risk of COVID-19 severity and mortality.21 These policies assume that younger individuals without apparent underlying conditions are at low risk. However, unlike symptoms following critical illness22 or acute phase of other coronavirus infections,23 symptoms in PCS are commonly reported in individuals with low COVID-19 mortality risk, for example, female, young and no chronic comorbidities.14 The potential scale of PCS in ‘lower-risk’ individuals, representing up to 80% of the population,3 necessitates urgent policies across countries to monitor,24 treat19 and pay25 for long-term implications of COVID-19 and to mitigate impact on healthcare utilisation and economies.
Therefore, in a pragmatic, prospective cohort study of individuals with persistent symptoms at least 4 weeks following recovery from acute SARS-CoV-2 infection and at low risk of COVID-19 mortality, we investigated (1) the prevalence of multiorgan impairment, compared with healthy, age-matched controls; (2) the associations between typical COVID-19 symptoms and multiorgan impairment; and (3) the associations between hospitalisation, severity of symptoms and multiorgan impairment.
Methods
Patient population and study design
In an ongoing, prospective study, participants were recruited to the study following expression of interest on the study registration website. Participants learnt about the study through advertisement on social media or via recommendations from clinicians from four participant identification centres, the latter usually applied to patients who had been hospitalised. Assessment took place at two UK research imaging sites (Perspectum, Oxford; and Mayo Clinic Healthcare, London) between 1 April 2020 and 14 September 2020, completing baseline assessment by 14 September 2020 (figure 1). Participants with laboratory-confirmed SARS-CoV-2 infection (tested SARS-CoV-2-positive by oropharyngeal/nasopharyngeal swab by reverse-transcriptase PCR (n=62), a positive antibody test (n=63), or with strong clinical suspicion of infection with typical symptoms/signs and assessed as highly likely to have COVID-19 by two independent clinicians (n=73)) were eligible for enrolment. Exclusion criteria were symptoms of active respiratory viral infection (temperature >37.8°C or three or more episodes of coughing in 24 hours), hospital discharge in the last 7 days, and contraindications to MRI, including implanted pacemakers, defibrillators, other metallic implanted devices and claustrophobia. All participants gave written informed consent.
Figure 1.
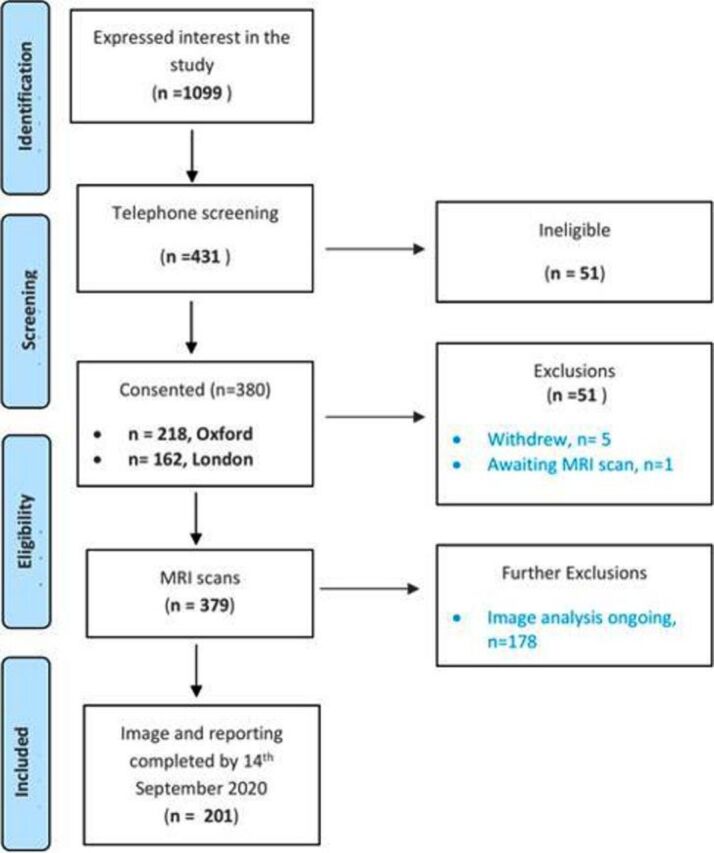
Flow from recruitment to enrolment of 201 patients with post-COVID-19 syndrome.
Assessment of PCS
Assessment included patient-reported validated questionnaires (quality of life, EQ-5D-5L,26 and Dyspnoea-1227) and fasting biochemical investigations (listed in online supplemental methods). PCS was classified as ‘severe’ (defined as persistent breathlessness, score of ≥10 on Dyspnoea-12, or reported moderate or greater problems with usual activities on EQ-5D-5L) or ‘moderate’. These thresholds were selected as the Dyspnoea-12 has been correlated with the Medical Research Council (MRC) dyspnoea grade, where level 3 warrants referral to rehabilitation services,27 and with EQ-5D-5L, less than 8% of the general population report moderate or greater problems with usual activities.28
bmjopen-2020-048391supp003.pdf (559.2KB, pdf)
Multiorgan impairment in PCS compared with healthy controls
We selected MRI as the imaging modality (as in UK Biobank) due to (1) safety (no radiation exposure, no need for intravenous contrast and minimal contact with the radiographer); (2) quantitative reproducibility (>95% acquisition and image processing success rate); (3) capacity for information sharing (digital data repository for independent analysis and research); and (4) rapid scalability (35 min scan to phenotype lung, heart, kidney, liver, pancreas and spleen). Multiorgan MRI data were collected at both study sites (Oxford: MAGNETOM Aera 1.5T; Mayo Healthcare London: MAGNETOM Vida 3T; both from Siemens Healthcare, Erlangen, Germany). The COVERSCAN multiparametric MRI assessment typically required 35 min per patient, including the lungs, heart, liver, pancreas, kidneys and spleen, by standardised methodology (online supplemental file 1). In brief, we assessed inflammation of the heart, kidneys, liver and pancreas with quantitative T1 relaxation mapping; lung function was characterised with a dynamic structural T2-weighted lung scan estimating lung capacity; ectopic fat accumulation in the liver and pancreas from proton density fat fraction; and volume of the liver and spleen measured from T1-weighted structural scan.
To determine impairment in each organ, we compared MRI-derived measurements from the heart, lungs, kidneys, liver, pancreas and spleen with reference ranges (online supplemental table 1), which were established as mean±2 SD from the healthy, age-matched control subjects (n=36) and validated by scoping literature review.11 We defined organ impairment if quantitative T1 mapping was outside the reference ranges for the heart, kidney, liver and pancreas, reduced estimated lung capacity from dynamic measurements in the lungs, or there was evidence of hepatomegaly, splenomegaly or ectopic fat accumulation.
bmjopen-2020-048391supp002.pdf (559.2KB, pdf)
Symptoms and multiorgan impairment
Associations between organ impairment and symptoms were visually assessed using a heat map, dividing those with impairments to an organ into columns and colouring the rows by percentage of reported symptoms.
Hospitalisation, severity and multiorgan impairment
We compared mean differences in quantitative organ metrics for hospitalised versus not hospitalised and moderate versus severe PCS using Kruskal-Wallis test (Fisher’s exact test for differences in binary outcomes). We defined multiorgan impairment as ≥2 organs with metrics outside the reference range. We investigated the associations between multiorgan impairment and (1) being hospitalised and (2) severe PCS with multivariate logistic regression models, adjusting for age, sex and body mass index (BMI).
Patient and public involvement and engagement
Patients and the public have directly and indirectly informed our research, from design to dissemination, with regular updates and webinars, including question and answer sessions with patients. Several clinician coauthors were indirectly informed by their patients in the COVERSCAN study (RB, AB) or PCS clinics (DW, MH, MC), who are members of organisations such as Long Covid SOS (eg, LH) and UKDoctors#Longcovid (eg, EA). LH and EA have been involved in the research, interpretation of results, understanding implications of our results and providing critical feedback to the manuscript.
Statistical analysis
We performed all analyses using R V.3.6.1, using descriptive statistics to summarise baseline characteristics and considering a p value less than 0.05 as statistically significant. Mean and SD were used for normally distributed continuous variables, median with IQR for non-normally distributed variables, and frequency and percentage for categorical variables. For group-wise comparison for absolute values between cases and healthy controls, we used Kruskal-Wallis test.
Results
Overall study population
Baseline characteristics
The study included 201 individuals (full details regarding hospitalisation: n=199; full questionnaire data to assign PCS severity: n=193). The mean age was 44.0 (range 21–71) years and the median BMI was 25.7 (IQR 23–28). Of the individuals, 71% were female, 88% were white, 32% were healthcare workers and 19% had been hospitalised with COVID-19. Assessments (symptoms, blood and MRI) had a median of 141 (IQR 110–162) days after initial symptoms. Medical history included smoking (3%), asthma (19%), obesity (20%), hypertension (7%), diabetes (2%) and prior heart disease (5%). The healthy control group had a mean age of 39 years (range 20–70), 40% were female, with a median BMI of 23 (IQR: 21–25) (table 1).
Table 1.
Baseline demographics and symptoms of 201 low-risk individuals with post-COVID-19 syndrome
| All patients (N=201) | Healthy controls (n=36) | P value | Not hospitalised (n=163) | Hospitalised (n=37) | P value | Moderate PCS (n=77) | Severe PCS (n=116) | P value | |
| Age (years), mean (SD) | 44 (11.0) | 39 (12.4) | 0.013 | 43 (10.9) | 50 (10.0) | 0.001 | 45 (12.2) | 44 (10.0) | 0.419 |
| Female, n (%) | 142 (70.6) | 14 (38.9) | 0.032 | 118 (72.4) | 23 (62.2) | 0.302 | 51 (66.2) | 85 (73.3) | 0.374 |
| BMI (kg/m2), median (IQR) | 25.7 (22.7–28.1) | 23.2 (21.4–23.1) | <0.001 | 25.3 (22.7–27.7) | 27.2 (23.1–31.0) | 0.063 | 25.8 (22.7–27.9) | 25.4 (22.5–28.2) | 0.639 |
| Ethnicity | |||||||||
| White | 176 (87.6) | 33 (91.7) | 148 (90.8) | 28 (75.7) | 67 (87.0) | 106 (91.4) | 0.178 | ||
| Mixed | 3 (1.5) | 0 (0) | 0.904 | 3 (1.8) | 0 (0) | 0.016 | 1 (1.3) | 2 (1.7) | |
| South Asian | 7 (3.5) | 3 (8.3) | 4 (2.5) | 3 (8.1) | 5 (6.5) | 0 (0) | |||
| Black | 4 (2.0) | 0 (0) | 1 (0.6) | 2 (5.4) | 2 (2.6) | 2 (1.7) | |||
| Comorbidities and risks | |||||||||
| Smoking | 0.244 | ||||||||
| Never | 133 (66.2) | 20 (60.6) | 108 (66.3) | 24 (64.9) | 55 (71.4) | 72 (61.7) | |||
| Current | 6 (3.0) | 8 (24.2) | <0.001 | 6 (3.7) | 0 (0) | 0.641 | 3 (3.9) | 3 (2.6) | |
| Ex-smoker | 62 (30.8) | 5 (15.2) | 49 (30.1) | 13 (35.1) | 19 (24.7) | 41 (35.3) | |||
| Healthcare worker | 64 (31.8) | 4 (12.1) | 0.009 | 50 (30.7) | 13 (35.1) | 0.695 | 33 (42.9) | 28 (24.1) | 0.007 |
| Asthma | 37 (18.4) | 0 (0) | 0.002 | 34 (20.9) | 3 (8.1) | 0.099 | 13 (16.9) | 22 (19.0) | 0.849 |
| BMI | |||||||||
| ≥25 kg/m2 | 113 (56.5) | 7 (20) | 87 (53.7) | 25 (67.6) | 0.144 | 46 (60.5) | 62 (53.4) | 0.374 | |
| ≥30 kg/m2 | 40 (20.0) | 0 (0) | 28 (17.3) | 12 (32.4) | 0.066 | 16 (21.1) | 24 (20.7) | 1.000 | |
| Hypertension | 13 (6.5) | 0 (0) | 0.001 | 11 (6.7) | 2 (5.4) | 1.000 | 6 (7.8) | 7 (6.0) | 0.771 |
| Diabetes | 4 (2.0) | 0 (0) | 0.104 | 4 (2.5) | 0 (0.0) | 1.000 | 4 (5.2) | 0 (0.0) | 0.024 |
| Previous heart disease | 9 (4.5) | 0 (0) | 0.001 | 8 (4.9) | 1 (2.7) | 1.000 | 3 (3.9) | 5 (4.3) | 1.000 |
| Symptoms | |||||||||
| Fatigue | 196 (98.0) | 159 (97.5) | 37 (100.0) | 1.000 | 73 (96.1) | 115 (99.1) | 0.302 | ||
| Shortness of breath | 176 (88.0) | 141 (86.5) | 35 (94.6) | 0.262 | 58 (76.3) | 112 (96.6) | <0.0001 | ||
| Muscle ache | 173 (86.5) | 142 (87.1) | 31 (83.8) | 0.597 | 66 (86.8) | 101 (87.1) | 1.000 | ||
| Headache | 165 (82.5) | 138 (84.7) | 27 (73.0) | 0.098 | 56 (73.7) | 102 (87.9) | 0.019 | ||
| Joint pain | 156 (78.0) | 127 (77.9) | 29 (78.4) | 1.000 | 56 (73.7) | 94 (81.0) | 0.284 | ||
| Chest pain | 152 (76.0) | 128 (78.5) | 24 (64.9) | 0.090 | 47 (61.8) | 98 (84.5) | 0.001 | ||
| Cough | 146 (73.0) | 117 (71.8) | 29 (78.4) | 0.539 | 55 (72.4) | 84 (72.4) | 1.000 | ||
| Fever | 144 (72.0) | 113 (69.3) | 31 (83.8) | 0.104 | 51 (67.1) | 86 (74.1) | 0.329 | ||
| Sore throat | 143 (71.5) | 120 (73.6) | 23 (62.2) | 0.165 | 50 (65.8) | 86 (74.1) | 0.256 | ||
| Diarrhoea | 118 (59.0) | 91 (55.8) | 27 (73.0) | 0.065 | 40 (52.6) | 76 (65.5) | 0.097 | ||
| Abnormal pain | 108 (54.0) | 91 (55.8) | 17 (45.9) | 0.361 | 30 (39.5) | 75 (64.7) | 0.001 | ||
| Wheezing | 98 (49.0) | 75 (46.0) | 23 (62.2) | 0.101 | 30 (39.5) | 64 (55.2) | 0.039 | ||
| Inability to walk | 80 (40.0) | 58 (35.6) | 22 (59.5) | 0.009 | 24 (31.6) | 50 (43.1) | 0.130 | ||
| Runny nose | 68 (34.0) | 55 (33.7) | 13 (35.1) | 0.85 | 24 (31.6) | 41 (35.3) | 0.642 | ||
| Time interval | |||||||||
| Initial symptoms to assessment (days), median (IQR) | 141 (110–162) | 141 (112–163) | 138 (97–150) | 0.106 | 121 (89–158) | 145 (121–163) | 0.001 | ||
| COVID-19-positive to assessment (days), median (IQR) | 71 (41–114) | 68 (35–112) | 105 (59–126) | 0.012 | 60 (43–98) | 78 (34–119) | 0.305 | ||
Data are presented as count (%).
Comparisons between managed at home versus hospitalised and between moderate versus PCS were conducted using Fisher’s exact test
BMI, body mass index; PCS, post-COVID-19 syndrome.
Regardless of hospitalisation, the most frequently reported symptoms were fatigue (98%), shortness of breath (88%), muscle ache (87%) and headache (83%) (table 1). Of the individuals, 99% had four or more and 42% had ten or more symptoms. Of individuals 70% reported ≥13 weeks off paid employment. Of the incidental structural findings observed on MRI (n=56), three were cardiac (atrial septal defect, bicuspid aortic valve and right atrial mass), one renal (hydronephrosis) and the rest were benign cysts.
Haematological investigations, including mean corpuscular haemoglobin concentration (24%), and renal, liver and lipid biochemistry, including potassium (38%), alanine transferase (14%), lactate dehydrogenase (17%), triglycerides (11%) and cholesterol (42%), were abnormally high in ≥10% of individuals. Bicarbonate (10%), phosphate (11%), uric acid (11%) and transferrin saturation (19%) were abnormally low in ≥10% of individuals (online supplemental table 1).
Single-organ and multiorgan impairment in PCS compared with healthy controls
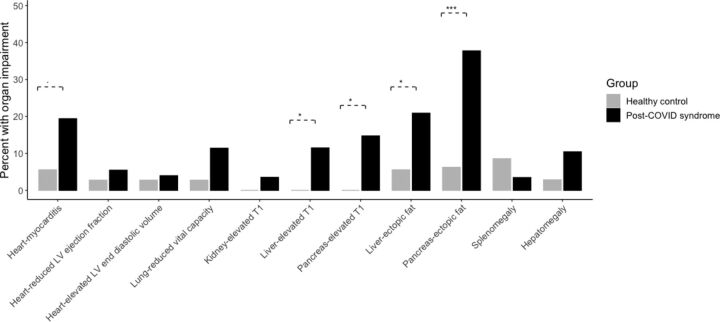
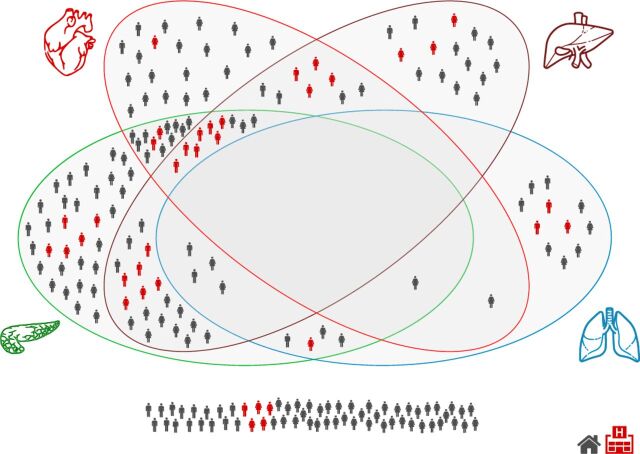
Organ impairment was more common in PCS than healthy controls (figure 2 and online supplemental figure 1). Impairment was present in the heart in 26% (myocarditis 19%, systolic dysfunction 9%), lung in 11% (reduced vital capacity), kidney in 4% (inflammation), liver in 28% (12% inflammation, 21% ectopic fat, 10% hepatomegaly), pancreas in 40% (15% inflammation, 38% ectopic fat) and spleen in 4% (splenomegaly) (figure 2 and table 2). Of the individuals, 70% had impairment in at least one organ and 29% had multiorgan impairment, with overlap across multiple organs (figure 3). Impairment in the liver, heart or lungs was associated with further organ impairment in 63%, 62% and 48% of individuals, respectively (figure 3).
Figure 2.
Percentage of patients (black) and controls (grey) with individual organ measures outside of the predefined normal range. Lines represent significant difference in the proportions between the two groups, with *p<0.05, **p<0.01, ***p<0.001. LV, left ventricular.
Table 2.
Evidence of organ impairment in 201 low-risk individuals with post-COVID-19 syndrome
| Measurement | All patients (N=201) | Healthy controls (n=36) | P value | Not hospitalised (n=163) | Hospitalised (n=37) | P value | Moderate PCS (n=77) | Severe PCS (n=116) | P value |
| Heart | |||||||||
| Left ventricular ejection fraction (%) | |||||||||
| Normal (>51%) | 190 (95.0) | 35 (97.2) | 0.699 | 155 (95.7) | 33 (89.1) | 0.124 | 72 (93.5) | 111 (95.7) | 0.353 |
| Impaired (≤51%) | 11 (5.0) | 1 (2.8) | 7 (4.3) | 4 (10.1) | 5 (6.4) | 5 (4.3) | |||
| Left ventricular end diastolic volume (mL) | |||||||||
| >264 mL in Men; >206 mL in Women | 8 (4.0) | 1 (2.8) | 1.00 | 4 (2.5) | 4 (10.8) | 0.040 | 4 (5.2) | 4 (3.4) | 0.715 |
| Evidence of myocarditis | |||||||||
| ≥3 segments with high T1 (≥1229 ms at 3T; ≥1015 ms at 1.5T) | 39 (19.4) | 2 (5.6) | 0.053 | 30 (18.4) | 8 (21.6) | 0.647 | 9 (11.7) | 29 (25.0) | 0.027 |
| Lungs | |||||||||
| Deep breathing fractional area change | (n=17 missing) | (n=13 missing) | (n=3 missing) | (n=8 missing) | (n=7 missing) | ||||
| <31% | 21 (11.4) | 1 (2.8) | 0.138 | 17 (11.3) | 4 (11.8) | 1 | 7 (10.1) | 13 (11.9) | 0.811 |
| Kidneys | |||||||||
| Kidney cortex T1 | (n=3 missing) | (n=3 missing) | (n=2 missing) | ||||||
| Normal (<1652 ms at 3T; <1227 ms at 1.5T) | 191 (96.5) | 36 (100.0) | 0.599 | 155 (96.9) | 35 (94.6) | 0.618 | 74 (98.7) | 112 (96.6) | 0.65 |
| Impaired (≥1652 ms at 3T; ≥1227 ms at 1.5T) | 7 (3.5) | 0 (0.0) | 5 (3.1) | 2 (5.4) | 1 (1.3) | 4 (3.4) | |||
| Pancreas | |||||||||
| Pancreatic inflammation (T1 in ms) | (n=11 missing) | (n=13 missing) | (n=7 missing) | (n=4 missing) | (n=4 missing) | (n=6 missing) | |||
| Normal <803 ms | 162 (85.3) | 23 (100.0) | 0.049 | 139 (89.1) | 22 (66.7) | 0.002 | 60 (82.2) | 95 (86.4) | 0.530 |
| Impaired ≥803 ms | 28 (14.7) | 0 (0) | 17 (10.9) | 11 (33.3) | 13 (17.8) | 15 (13.6) | |||
| Pancreatic fat | (n=4 missing) | ||||||||
| Normal <4.6% | 122 (62.2) | 30 (93.8) | <0.001 | 107 (66.9) | 14 (40.0) | 0.004 | 44 (57.9) | 72 (63.7) | 0.449 |
| Impaired ≥4.6% | 74 (37.8) | 2 (6.2) | 53 (33.1) | 21 (60.0) | 32 (42.1) | 41 (36.3) | |||
| Liver | |||||||||
| Liver inflammation (cT1 in ms) | (n=1 missing) | (n=1 missing) | (n=1 missing) | ||||||
| Normal <784 ms | 177 (88.5) | 36 (100) | 0.030 | 148 (91.4) | 28 (75.7) | 0.018 | 69 (90.8) | 101 (87.1) | 0.494 |
| Impaired ≥784 ms | 23 (11.5) | 0 (0) | 14 (8.6) | 9 (24.3) | 7 (9.2) | 15 (12.9) | |||
| Liver fat | |||||||||
| Normal <4.8% | 159 (79.1) | 34 (94.4) | 0.034 | 134 (82.2) | 24 (64.9) | 0.026 | 61 (79.2) | 91 (78.4) | 1 |
| Impaired ≥4.8% | 42 (20.9) | 2 (5.4) | 29 (17.8) | 13 (35.1) | 16 (20.8) | 25 (21.6) | |||
| Liver volume | (n=1 missing) | ||||||||
| Normal <1935 mL | 180 (89.6) | 34 (97.1) | 0.214 | 154 (94.5) | 25 (67.6) | <0.0001 | 68 (88.3) | 104 (89.7) | 0.816 |
| Impaired ≥1935 mL | 21 (10.4) | 1 (2.9) | 9 (5.5) | 12 (32.4) | 9 (11.7) | 12 (10.3) | |||
| Spleen | |||||||||
| Splenic volume (mL) | (n=1 missing) | ||||||||
| Normal <350 mL | 194 (96.5) | 32 (91.4) | 0.172 | 160 (98.2) | 33 (89.2) | 0.023 | 74 (96.1) | 112 (96.6) | 1 |
| Impaired ≥350 mL | 7 (3.5) | 3 (8.6) | 3 (1.8) | 4 (10.8) | 3 (3.9) | 4 (3.4) | |||
Data are presented as count (%).
Comparisons between managed at home versus hospitalised and between moderate versus PCS were conducted using Fisher’s exact test.
PCS, post-COVID-19 syndrome.
Figure 3.
Multiorgan impairment in low-risk individuals with post-COVID-19 syndrome by gender and hospitalisation.
bmjopen-2020-048391supp001.pdf (559.2KB, pdf)
Symptoms and multiorgan impairment
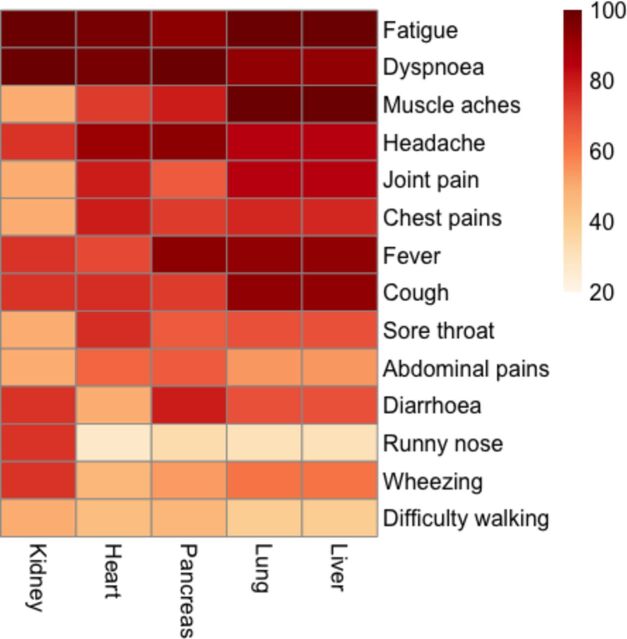
Hepatic and pulmonary impairment frequently clustered together, with fatigue, muscle aches, fever and cough commonly reported. Impairment in particular organs was associated with particular symptoms—pancreas: diarrhoea, fever, headache and dyspnoea; heart: headache, dyspnoea and fatigue; and kidney: wheezing, runny nose, diarrhoea, cough, fever, headache, dyspnoea and fatigue (figure 4).
Figure 4.
Percentage of reported symptoms during the acute phases of the illness within those with evidence of organ impairment for each organ separately. Darker red indicates higher percentage of reported symptoms per impaired organ. There are no distinct patterns of symptoms relating to each impaired organ, but a high burden of symptoms in individuals is highlighted.
Hospitalisation, severity and multiorgan impairment
The hospitalised group were older (p=0.001), had higher BMI (p=0.063), and were more likely to be non-white (p=0.016) and to report ‘inability to walk’ (p=0.009) than non-hospitalised individuals. There were no other statistically significant differences between risk factors or symptoms between the groups. Impairment of the liver, pancreas (eg, ectopic fat in the pancreas and liver, hepatomegaly) and ≥2 organs was higher in hospitalised individuals (all p<0.05) (figure 3 and table 2). In multivariate analyses, adjusting for age, sex and BMI, liver volume remained significantly associated with hospitalisation (p=0.001). Hospitalised individuals had high triglycerides (30% vs 7.2%, p=0.002), cholesterol (60% vs 38%, p=0.04) and low-density lipoprotein-cholesterol (57% vs 31%, p=0.01), and low transferrin saturation (38% vs 15%, p=0.01), compared with non-hospitalised individuals. erythrocyte sedimentation rate (ESR) (13%), bicarbonate (12%), uric acid (16%), platelet count (13%) and high-sensitivity C-reactive protein (CRP) (15%) were high in ≥10% of hospitalised individuals.
Of the individuals, 60% (n=120) had severe PCS, with 52% reporting persistent, moderate problems undertaking usual activities (level 3 or greater in the relevant EQ-5D-5L question; 34% reported Dyspnoea-12 score ≥10). Of those with severe PCS, 84% were not hospitalised and 73% were female. There were no differences in age, BMI or ethnicity between the groups. Individuals with severe PCS were more likely to report shortness of breath (p<0.001), headache (p=0.019), chest pain (p=0.001), abdominal pain (p=0.001) and wheezing (p=0.039). Of those with ‘severe’ PCS, 25% had myocarditis compared with 12% with moderate PCS (unadjusted: 0.023; adjustment for age, sex and BMI: p=0.04; online supplemental figure 2). Severe PCS was associated with higher mean cell haemoglobin concentration (28% vs 17%), cholesterol (46.2% vs 32.8%), CRP (10% vs 3.8%) and ESR (10% vs 6%) than moderate PCS, but these differences were not statistically significant (online supplemental table 3). Muscle aches, fever and coughing were common in severe PCS, and headache was common in individuals with inflammation of the pancreas (figure 4).
Discussion
We report three findings in the first COVID-19 recovery study to evaluate medium-term, multiorgan impairment. First, in low-risk individuals, there were chronic symptoms and mild impairment in the heart, lung, liver, kidney and pancreas 4 months post-COVID-19, compared with healthy controls. Second, cardiac impairment was more common in severe PCS. Third, we demonstrate feasibility and potential utility of community-based multiorgan assessment for PCS.
Comparison with other studies
Common symptoms were fatigue, dyspnoea, myalgia, headache and arthralgia, despite low risk of COVID-19 mortality or hospitalisation. COVID-19 impact models have included age, underlying conditions and mortality, but not morbidity, multiorgan impairment and chronic diseases.29 30 Even in non-hospitalised individuals, up to 10% of those infected have PCS,15 31 but studies of extrapulmonary manifestations emphasise acute illness.32 We describe mild rather than severe organ impairment, but the pandemic’s scale and high infection rates in lower risk individuals signal medium-term and longer-term COVID-19 impact, which cannot be ignored in healthcare or policy spheres.
Acute myocarditis and cardiogenic shock33 are documented in hospitalised patients with COVID-19.6 In American athletes, recent COVID-19 was associated with myocarditis.34 Although causality cannot be attributed and postviral syndromes have included similar findings,21 we show that a quarter of low-risk individuals with PCS have mild systolic dysfunction or myocarditis. The significance of these findings and the associations with contemporaneous abnormal echocardiography findings and long-term myocardial fibrosis and impairment are unknown. Cardiac impairment, a risk factor for severe COVID-19, may have a role in PCS. Two further findings that deserve investigation are pancreatic abnormalities, given the excess diabetes risk reported in PCS,15 and the preponderance of healthcare workers at increased PCS risk (as observed for COVID-19 mortality), possibly due to higher viral burden.
PCS is likely to be a syndrome rather than a single condition. Despite an immunological basis for individual variations in COVID-19 progression and severity,35 prediction models have high rates of bias, perform poorly,36 and focus on respiratory dysfunction and decisions for ventilation in acutely unwell patients, rather than multiorgan function. Ongoing long-term studies37 exclude non-hospitalised, low-risk individuals. During a pandemic, we studied subclinical organ impairment in PCS, showing low rates of incidental findings. As specialist PCS services are rolled out,38 39 multiorgan assessment, monitoring and community pathways have potential roles during and beyond COVID-19, but need to be evaluated.
Implications for research, clinical practice and public health
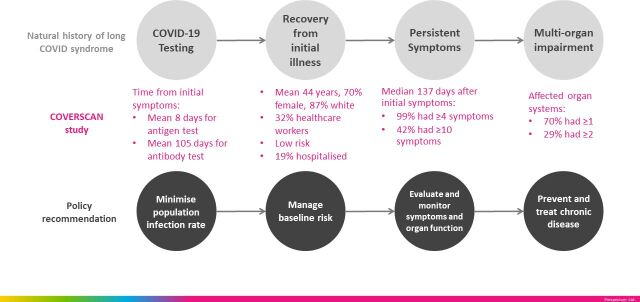
Our findings have three research implications. First, as countries face second waves, COVID-19 impact models should include PCS, whether quality of life, healthcare utilisation or economic effects. Second, there is urgent need for multiorgan assessment, including blood and imaging, as well as primary and secondary care data linkage, to define PCS. Third, longitudinal studies of clustering of symptoms and organ impairment will inform health services research to plan multidisciplinary care pathways. There are three management implications. First, we signal the need for multiorgan monitoring in at least the medium term, especially extrapulmonary sequelae. Care pathways involving MRI (with limited access in many clinical settings) need evaluation versus other modalities to detect organ impairment (eg, spirometry, N-terminal pro B-type natriuretic peptide (NT-pro-BNP), ECG, echocardiography, ultrasound and blood investigations). Second, until effective vaccines and treatments are widely available, ‘infection suppression’ (eg, social distancing, masks, physical isolation) is the prevention strategy. Third, whether understanding baseline risk or multiorgan complications, PCS requires management across specialties (eg, cardiology, gastroenterology) and disciplines (eg, epidemiology, diagnostics, laboratory science) (figure 5).
Figure 5.
Natural history of post-COVID-19 syndrome, the COVERSCAN study in low-risk individuals (N=201) and policy recommendations.
Limitations
There are some limitations. First, our cardiac MRI protocol excluded gadolinium contrast due to concerns regarding COVID-19-related renal complications, relying on native T1 mapping to characterise myocardial inflammation non-invasively (previously validated for acute myocarditis).40 Second, for organ impairment, we show association, not causation, and incidental findings are possible in asymptomatic individuals41; however, our findings are strengthened by comparison with healthy, age-matched controls, although not matched for sex or baseline comorbidities. Third, for pragmatic reasons, our controls were scanned using 1.5T, but we used 3T ranges as described in an analogous study with similar acquisition protocols. Therefore, we may be under-representing the true proportion of impairment in those individuals with PCS scanned at 3T. Fourth, further studies may explore different controls, for example, individuals with postinfluenza symptoms, COVID-19 without symptoms or from general clinics. We will investigate duration, trajectory, complications and recovery for specific symptoms and organ impairment in the follow-up phase. Fifth, our study population was not ethnically diverse, despite disproportionate COVID-19 impact in non-white individuals. Sixth, to limit interaction and exposure between the trial team and the patients, pulse oximetry, spirometry, MRI assessment of the brain and muscle function were not included from the outset.
Conclusions
Our study suggests PCS has a physiological basis, with measurable patient-reported outcomes and organ impairment. Future research should address longer-term follow-up of organ function beyond symptoms and blood investigations, even in lower risk individuals; prioritisation for imaging, investigation and referral; and optimal care pathways. Health system responses should emphasise infection suppression and management of pre-COVID-19 and post-COVID-19 risk factors and chronic diseases.
Supplementary Material
Footnotes
Twitter: @amibanerjee1
Contributors: Study design: AD, RB, JA, COVERSCAN team. Patient recruitment: RB, COVERSCAN team. Data collection: MW, COVERSCAN team. Data analysis: AD, AB, COVERSCAN team. Data interpretation: AB, AD, MW, RB. Initial manuscript drafting: AB, AD, RB. Critical review of early and final versions of the manuscript: all authors including JO and DJC. Specialist input: MW, AB (cardiology); RB, MH, DW, MC, DJC (general medicine); MH, MC, DW (long COVID-19); MB, RB (imaging); AD (statistics); AB (epidemiology/public health); MG (primary care); JA (healthcare management); LH, EA (patient and public involvement).
Funding: This work was supported by the UK’s National Consortium of Intelligent Medical Imaging (Industry Strategy Challenge Fund), Innovate UK (Grant 104688) and the European Union’s Horizon 2020 research and innovation programme (agreement no 719445). The research was designed, conducted, analysed and interpreted by the authors independently of the funding sources.
Competing interests: AD, RB and MB are employees of Perspectum.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available upon reasonable request from the corresponding author.
Ethics statements
Patient consent for publication
Not required.
Ethics approval
The study has received ethical approval (20/SC/0185).
References
- 1. World Health Organization . Clinical management of severe acute respiratory infection (SARI) when COVID-19 disease is suspected. interim guidance 13 March 2020, 2020. Available: https://apps.who.int/iris/handle/10665/331446
- 2. Pavon AG, Meier D, Samim D, et al. First documentation of persistent SARS-Cov-2 infection presenting with late acute severe myocarditis. Can J Cardiol 2020;36:1326.e5–1326.e7. 10.1016/j.cjca.2020.06.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Puntmann VO, Carerj ML, Wieters I. Outcomes of cardiovascular magnetic resonance imaging in patients recently recovered from coronavirus disease 2019 (COVID-19). JAMA Cardiol 2019;2020:1265–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Tabary M, Khanmohammadi S, Araghi F, et al. Pathologic features of COVID-19: a Concise review. Pathol Res Pract 2020;216:153097. 10.1016/j.prp.2020.153097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Alqahtani SA, Schattenberg JM. Liver injury in COVID-19: the current evidence. United European Gastroenterol J 2020;8:509–19. 10.1177/2050640620924157 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Somasundaram NP, Ranathunga I, Ratnasamy V, et al. The impact of SARS-Cov-2 virus infection on the endocrine system. J Endocr Soc 2020;4:1–22. 10.1210/jendso/bvaa082 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Farouk SS, Fiaccadori E, Cravedi P, et al. COVID-19 and the kidney: what we think we know so far and what we don't. J Nephrol 2020;33:1213–8. 10.1007/s40620-020-00789-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Lai A, Pasea L, Banerjee A. Estimating excess mortality in people with cancer and multimorbidity in the COVID-19 emergency. BMJ Open 2020;10:e043828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Banerjee A, Pasea L, Harris S, et al. Estimating excess 1-year mortality associated with the COVID-19 pandemic according to underlying conditions and age: a population-based cohort study. The Lancet 2020;395:1715–25. 10.1016/S0140-6736(20)30854-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Banerjee A, Chen S, Pasea L. Excess deaths in people with cardiovascular diseases during the COVID-19 pandemic. Eur J Prev Cardiol 2020. 10.1093/eurjpc/zwaa155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Raman B, Cassar MP, Tunnicliffe EM, et al. Medium-Term effects of SARS-CoV-2 infection on multiple vital organs, exercise capacity, cognition, quality of life and mental health, post-hospital discharge. EClinicalMedicine 2021;31:100683. 10.1016/j.eclinm.2020.100683 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Horton R. Offline: COVID-19 is not a pandemic. The Lancet 2020;396:874. 10.1016/S0140-6736(20)32000-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Shovlin CL, Vizcaychipi MP. Implications for COVID-19 triage from the ICNARC report of 2204 COVID-19 cases managed in UK adult intensive care units. Emerg Med J 2020;37:332–3. 10.1136/emermed-2020-209791 [DOI] [PubMed] [Google Scholar]
- 14. Docherty AB, Harrison EM, Green CA, et al. Features of 20 133 UK patients in hospital with covid-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ 2020;369:m1985–12. 10.1136/bmj.m1985 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Williamson EJ, Walker AJ, Bhaskaran K, et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020;584:430–6. 10.1038/s41586-020-2521-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Office for National Statistics . The prevalence of long COVID symptoms and COVID-19 complications, 2020. Available: https://www.ons.gov.uk/news/statementsandletters/theprevalenceoflongcovidsymptomsandcovid19complications
- 17. del Rio C, Collins LF, Malani P. Long-Term health consequences of COVID-19. JAMA 2020;324:1723. 10.1001/jama.2020.19719 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Carfì A, Bernabei R, Landi F, et al. Persistent symptoms in patients after acute COVID-19. JAMA 2020;324:603–5. 10.1001/jama.2020.12603 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Nabavi N. Long covid: how to define it and how to manage it. BMJ 2020;370:m3489. 10.1136/bmj.m3489 [DOI] [PubMed] [Google Scholar]
- 20. Greenhalgh T, Knight M, A’Court C, et al. Management of post-acute covid-19 in primary care. BMJ 2020;13:m3026. 10.1136/bmj.m3026 [DOI] [PubMed] [Google Scholar]
- 21. National Institute for Health Research . New risk prediction model could help improve guidance for people shielding from COVID-19, 2020. Available: https://www.nihr.ac.uk/news/new-risk-prediction-model-could-help-improve-guidance-for-people-shielding-from-covid-19/25096
- 22. Hill AD, Fowler RA, Pinto R, et al. Long-term outcomes and healthcare utilization following critical illness – a population-based study. Crit Care 2016;20:1–10. 10.1186/s13054-016-1248-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Perrin R, Riste L, Hann M, et al. Into the looking glass: post-viral syndrome post COVID-19. Med Hypotheses 2020;144:110055. 10.1016/j.mehy.2020.110055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. George PM, Barratt SL, Condliffe R, et al. Respiratory follow-up of patients with COVID-19 pneumonia. Thorax 2020;75:1009–16. 10.1136/thoraxjnl-2020-215314 [DOI] [PubMed] [Google Scholar]
- 25. Jiang DH, McCoy RG. Planning for the Post-COVID syndrome: how payers can mitigate long-term complications of the pandemic. J Gen Intern Med 2020;35:3036–9. 10.1007/s11606-020-06042-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Janssen MF, Pickard AS, Golicki D, et al. Measurement properties of the EQ-5D-5L compared to the EQ-5D-3L across eight patient groups: a multi-country study. Qual Life Res 2013;22:1717–27. 10.1007/s11136-012-0322-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Yorke J, Moosavi SH, Shuldham C, et al. Quantification of dyspnoea using descriptors: development and initial testing of the Dyspnoea-12. Thorax 2010;65:21–6. 10.1136/thx.2009.118521 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Hobbins A, Barry L, Kelleher D, et al. The health of the residents of ireland: population norms for Ireland based on the EQ-5D-5L descriptive system – a cross sectional study. HRB Open Res 2018;1:22. 10.12688/hrbopenres.12848.1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Gupta A, Madhavan MV, Sehgal K, et al. Extrapulmonary manifestations of COVID-19. Nat Med 2020;26:1017–32. 10.1038/s41591-020-0968-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Palmer K, Monaco A, Kivipelto M, et al. The potential long-term impact of the COVID-19 outbreak on patients with non-communicable diseases in Europe: consequences for healthy ageing. Aging Clin Exp Res 2020;32:1189–94. 10.1007/s40520-020-01601-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Menni C, Valdes AM, Freidin MB, et al. Real-Time tracking of self-reported symptoms to predict potential COVID-19. Nat Med 2020;26:1037–40. 10.1038/s41591-020-0916-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Mandal S, Barnett J, Brill S, et al. “Long-COVID”: a cross-sectional study of persisting symptoms, biomarker and imaging abnormalities following hospitalisation for COVID-19. Thorax 2020:thoraxjnl-2020-215818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Chau VQ, Giustino G, Mahmood K, et al. Cardiogenic shock and hyperinflammatory syndrome in young males with COVID-19. Circ Hear Fail 2020:556–9. [DOI] [PubMed] [Google Scholar]
- 34. Rajpal S, Tong MS, Borchers J, et al. Cardiovascular magnetic resonance findings in competitive athletes recovering from COVID-19 infection. JAMA Cardiol 2020:5–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Mathew D, Giles JR, Baxter AE, et al. Deep immune profiling of COVID-19 patients reveals distinct immunotypes with therapeutic implications. Science 2020;369:eabc8511. 10.1126/science.abc8511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Wynants L, Van Calster B, Collins GS, et al. Prediction models for diagnosis and prognosis of covid-19: systematic review and critical appraisal. BMJ 2020;369:m1328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. PHOSP-COVID . Post-HOSPitalisation COVID-19 study, 2020. Available: https://www.phosp.org/
- 38. NHS . NHS to offer ‘long covid’ sufferers help at specialist centres, 2020. Available: https://www.england.nhs.uk/2020/10/nhs-to-offer-long-covid-help/
- 39. National Institute for Healh and Care Excellence . COVID-19 rapid guideline: managing the long-term effects of COVID-19, 2020. Available: https://www.nice.org.uk/guidance/ng188 [PubMed]
- 40. Ferreira VM, Piechnik SK, Dall’Armellina E, et al. Native T1-mapping detects the location, extent and patterns of acute myocarditis without the need for gadolinium contrast agents. J Cardiovasc Magn Reson 2014;16:1–11. 10.1186/1532-429X-16-36 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Gibson LM, Paul L, Chappell FM, et al. Potentially serious incidental findings on brain and body magnetic resonance imaging of apparently asymptomatic adults: systematic review and meta-analysis. BMJ 2018;14:k4577. 10.1136/bmj.k4577 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjopen-2020-048391supp003.pdf (559.2KB, pdf)
bmjopen-2020-048391supp002.pdf (559.2KB, pdf)
bmjopen-2020-048391supp001.pdf (559.2KB, pdf)
Data Availability Statement
Data are available upon reasonable request from the corresponding author.