Key Points
Question
What are the hypothetical annual out-of-pocket costs of guideline-recommended medications for the treatment of older adults with multiple chronic conditions in 2009 and 2019?
Findings
In this cross-sectional study of hypothetical older adults with 8 common chronic conditions enrolled in 3599 Medicare prescription drug plans in 2009 and 3618 Medicare prescription drug plans in 2019, the median inflation-adjusted out-of-pocket costs for guideline-recommended medications decreased between 2009 and 2019, with the exception of costs for conditions for which brand-name medications without generic alternatives became guideline recommended. For a cluster of 5 commonly comorbid conditions (chronic obstructive pulmonary disease, hypertension, osteoarthritis, osteoporosis, and type 2 diabetes), the median annual cost was $1999 in 2019, a 12% decrease from 2009.
Meaning
In this study, although inflation-adjusted out-of-pocket costs of guideline-recommended medications for older adults with multiple chronic conditions generally decreased between 2009 and 2019, costs remained high and may have presented a substantial financial burden for Medicare beneficiaries.
Abstract
Importance
Most adults 65 years or older have multiple chronic conditions. Managing these conditions with prescription drugs can be costly, particularly for older adults with limited incomes.
Objective
To estimate hypothetical out-of-pocket costs associated with guideline-recommended outpatient medications for the initial treatment of 8 common chronic diseases among older adults with Medicare prescription drug plans (PDPs).
Design, Setting, and Participants
This retrospective cross-sectional study used 2009 and 2019 Medicare prescription drug plan formulary files to estimate annual out-of-pocket costs among hypothetical patients enrolled in Medicare Advantage or stand-alone Medicare Part D plans. A total of 3599 PDPs in 2009 and 3618 PDPs in 2019 were included after inclusion and exclusion criteria were applied. Costs associated with guideline-recommended medications for 8 of the most common chronic diseases (atrial fibrillation, chronic obstructive pulmonary disease [COPD], heart failure with reduced ejection fraction, hypercholesterolemia, hypertension, osteoarthritis, osteoporosis, and type 2 diabetes), alone and in 2 clusters of commonly comorbid conditions, were examined.
Main Outcomes and Measures
Annual out-of-pocket costs for each chronic condition, inflation adjusted to 2019 dollars.
Results
Among 3599 Medicare PDPs in 2009, 1998 were Medicare Advantage plans and 1601 were stand-alone plans; among 3618 Medicare PDPs in 2019, 2719 were Medicare Advantage plans and 899 were stand-alone plans. For an older adult enrolled in any Medicare PDP in 2019, the median annual out-of-pocket costs for individual conditions varied, from a minimum of $32 (IQR, $6-$48) for guideline-recommended management of osteoporosis (a decrease from $128 [IQR, $102-$183] in 2009) to a maximum of $1579 (IQR, $1524-$2229) for guideline-recommended management of atrial fibrillation (an increase from $91 [IQR, $73-$124] in 2009). For an older adult with a cluster of 5 commonly comorbid conditions (COPD, hypertension, osteoarthritis, osteoporosis, and type 2 diabetes) enrolled in any PDP, the median out-of-pocket cost in 2019 was $1999 (IQR, $1630-$2564), a 12% decrease from $2284 (IQR, $1920-$3107) in 2009. For an older adult with all 8 chronic conditions (atrial fibrillation, COPD, diabetes, hypercholesterolemia, heart failure, hypertension, osteoarthritis, and osteoporosis) enrolled in any PDP, the median out-of-pocket cost in 2019 was $3630 (IQR, $3234-$5197), a 41% increase from $2571 (IQR, $2185-$3719) in 2009.
Conclusions and Relevance
In this cross-sectional study, out-of-pocket costs for guideline-recommended outpatient medications for the initial treatment of 8 common chronic diseases varied by condition. Although costs generally decreased between 2009 and 2019, particularly with regard to conditions for which generic drugs were available, out-of-pocket costs remained high and may have presented a substantial financial burden for Medicare beneficiaries, especially older adults with conditions for which brand-name drugs were guideline recommended.
This cross-sectional study assesses the out-of-pocket costs associated with guideline-recommended outpatient medications for the initial treatment of 8 common chronic diseases among hypothetical older adults with Medicare prescription drug plans.
Introduction
The typical older adult in the US manages multiple chronic conditions, such as diabetes, cardiovascular disease, and pulmonary disease. In the Medicare population, 66% of adults older than 65 years have been diagnosed with 2 or more chronic conditions, and 16% of adults have 6 or more conditions.1 Optimal management of multiple chronic conditions often requires pharmacological treatment, which can be a burden on patients for 2 reasons. First, medication regimens are complex, and the potential exists for drug interactions and medication error.2,3,4,5 Second, the out-of-pocket costs incurred by older adults, a population with limited incomes, are substantial.6 High out-of-pocket costs may also reduce medication adherence and limit the benefit of guideline-recommended treatment while increasing the burden of treatment among patients with multiple chronic conditions.7
In 2003, the US Congress passed the Medicare Prescription Drug, Improvement, and Modernization Act,8 offering prescription drug coverage for Medicare beneficiaries through stand-alone Medicare Part D prescription drug plans (PDPs), an alternative to prescription coverage available through the Medicare Part C program, also called Medicare Advantage PDPs. In addition, PDPs require out-of-pocket spending on the part of Medicare beneficiaries, both through insurance premiums and prescription cost sharing (eg, flat-fee copayments, coinsurance, or deductibles).8 Given increasing drug prices and coinsurance requirements, many Medicare beneficiaries may experience increasing cost burdens.9,10,11,12,13
Before Medicare launched PDPs in 2006, a seminal study by Boyd et al14 estimated the cost of a prescription drug regimen for a hypothetical patient with 5 specific chronic conditions: chronic obstructive pulmonary disease (COPD), hypertension, osteoporosis, osteoarthritis, and type 2 diabetes. Based on 2004 drug prices and assuming no prescription drug coverage, the patient’s medications would have cost $6544 annually (inflation adjusted to June 2019), more than 20% of typical patients’ median annual income.14,15,16
Prescription drug pricing remains a major bipartisan issue, and further research on the financial burden of treating multiple chronic conditions among older adults can inform policy proposals.17 Therefore, we emulated the study originally conducted by Boyd et al,14 estimating the 2009 and 2019 out-of-pocket expenses associated with guideline-recommended outpatient medications for the initial treatment of multiple chronic conditions among older adults enrolled in stand-alone or Medicare Advantage PDPs. Our cross-sectional study aimed to provide a detailed assessment of variation over time in the financial burden of out-of-pocket expenses for prescription drugs among older patients with multiple chronic conditions.18,19
Methods
This cross-sectional study estimated the annual out-of-pocket costs in 2009 and 2019 (inflation adjusted to 2019 dollars) for the hypothetical older adult adhering to guideline-recommended initial pharmacological treatment for common chronic diseases. The study was deemed exempt from review by the Yale Human Research Protection Program because of its use of publicly available data that did not involve human participants. This study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline for cross-sectional studies.
Guideline Selection
We selected clinical practice guidelines for the initial outpatient pharmacological treatment of 9 common chronic diseases identified by Boyd et al14 that were commonly managed in primary care settings: atrial fibrillation, chronic heart failure (ejection fraction unspecified), COPD, hypercholesterolemia, hypertension, osteoarthritis, osteoporosis, stable angina, and type 2 diabetes. We obtained 2 groups of guidelines published by professional organizations in the US: (1) guidelines that were the most up-to-date editions as of 2009 and (2) guidelines that were the most up-to-date editions as of 2019 (eTable 2 in the Supplement, with abbreviations for terms listed in eTable 1 in the Supplement).
Guideline Recommendations
Recommendations for outpatient medical therapy for diseases of moderate severity were abstracted from each clinical practice guideline (eTable 3 in the Supplement). For example, within the recommendations for the management of heart failure, we assumed patients were diagnosed with stage C heart failure with reduced ejection fraction (HFrEF) and were categorized as New York Heart Association class II or class III. Within the recommendations for the management of cholesterol, we assumed patients had clinical atherosclerotic cardiovascular disease.
We assumed the hypothetical older adult had no intensification of treatment (eg, from first-line to second-line pharmacological therapy) for 1 year. We only included strong recommendations or those of moderate strength (equivalent to class 1 and class 2a in the American College of Cardiology/American Heart Association guideline20) based on high-quality or moderate-quality evidence (equivalent to level A and level B in the American College of Cardiology/American Heart Association guideline20). For each recommendation, we identified the classes of prescription drugs recommended. Furthermore, for cases in which a single dosage of a drug was recommended and multiple dosages were available on the market, we included only formulations of the recommended dosage.
After abstracting recommendations, we excluded stable angina from further analysis because no specific prescription drugs were recommended for treatment of the condition based on the most recent guideline, which was published in 2012.21 The remaining 8 conditions (atrial fibrillation, COPD, HFrEF, hypercholesterolemia, hypertension, osteoarthritis, osteoporosis, and type 2 diabetes) were included in the analysis.
Data Sources
We used PDP formulary files from the Centers for Medicare & Medicaid Services from the second quarters of 2009 and 2019. These files provided information submitted by insurance companies to the Centers for Medicare & Medicaid Services and included the associated formulary, the tier assignment for all covered medications, the copayment type (eg, dollar copayment vs percentage coinsurance), and the copayment amount. We noted whether each plan was a Medicare Advantage PDP, which covered Part A and Part B benefits in addition to prescription drugs, or a stand-alone PDP, which covered only prescription drugs.
We included plans that had no deductibles or reduced deductibles. We excluded special needs plans, which restricted enrollment to institutionalized individuals, individuals with dual eligibility, and individuals with severe or disabling chronic conditions. We also excluded plans outside of the 50 states and Puerto Rico (eg, plans offered in other US territories). All plans not meeting these exclusion criteria that were active in either 2009 or 2019 were included. We assumed patients filled their prescriptions at preferred retail pharmacies.22
We limited our analysis to PDPs that submitted full details about cost sharing, coverage phase, and tier structures for the drugs in our sample. If a plan’s formulary did not include one of the drugs recommended by guidelines or equivalent drugs within the drug class, we excluded the plan for that specific condition. In 2009, no plans were excluded for atrial fibrillation, diabetes, heart failure, hypertension, hyperlipidemia, and osteoarthritis; of 3599 plans, only 3 (0.8%) associated with osteoporosis and 88 (2.4%) associated with the management of COPD were excluded. In 2019, each plan's formulary included at least 1 of the drugs recommended by guidelines or an equivalent drug within the class; therefore, in 2019 no plans were excluded based on their drug availability (eTable 7 in the Supplement).
Out-of-Pocket Costs
The primary outcome was median annual out-of-pocket costs across all PDPs for the pharmacological treatment of each chronic disease using guideline-recommended medications. Our estimates were deliberately conservative; if multiple guideline-recommended drugs were available within the same drug class for the same indications, we assumed patients and physicians would choose the least expensive drug covered by the patient’s insurance.
We estimated annual out-of-pocket costs according to the 4 benefit phases of the standard PDP. In 2009, the 4 benefit phases included a deductible, an initial coverage phase until total drug costs reached $2700, a coverage gap until out-of-pocket spending reached $4350, and a catastrophic coverage phase.23 In 2019, the 4 benefit phases included a deductible, an initial coverage phase until total drug costs reached $3820, a coverage gap until out-of-pocket spending reached $5100, and a catastrophic coverage phase.23 The Part D coverage gap closed between 2009 and 2019, meaning that out-of-pocket spending for prescription refills decreased from 100% to 25% of the drug’s retail price.23 We did not include insurance plan premiums in the estimates. When calculating the median out-of-pocket expenditures, we adjusted for inflation using June 2019 as the index month and year.15
In addition to examining the median annual out-of-pocket costs to treat single conditions, we assessed the median annual out-of-pocket costs to treat 2 clusters of comorbidities. Cluster A included 5 commonly comorbid chronic conditions (identical to the hypothetical patient with multiple chronic conditions described by Boyd et al14): COPD, hypertension, osteoarthritis, osteoporosis, and type 2 diabetes. Cluster B included all 8 commonly comorbid chronic conditions: atrial fibrillation, COPD, HFrEF, hypercholesterolemia, hypertension, osteoarthritis, osteoporosis, and type 2 diabetes.
Statistical Analysis
First, we estimated median annual out-of-pocket costs based on the guideline-recommended treatment regimens for all 8 single conditions and the 2 clusters of commonly comorbid conditions. As an additional analysis, we included HFrEF for 2019 based on 2 different approaches, both of which were guideline recommended. Approach 1 assumed the patient was prescribed an angiotensin-converting enzyme inhibitor or an angiotensin receptor blocker, an evidence-based β blocker, and an aldosterone antagonist. Approach 2 assumed the patient was prescribed an angiotensin receptor neprilysin inhibitor, an evidence-based β blocker, and an aldosterone antagonist. For each condition or cluster of conditions, we calculated the percent change in median annual out-of-pocket costs between 2009 and 2019. Analyses were stratified by insurance type (any PDP, Medicare Advantage PDP, or stand-alone PDP) and state. The Wilcoxon rank sum test was used to compare median annual out-of-pocket costs in 2009 and 2019 within the same insurance type.
Second, for each condition or cluster of conditions, we compared the median annual out-of-pocket costs between Medicare Advantage PDPs and stand-alone PDPs using the Wilcoxon rank sum test for both 2009 and 2019. All data were analyzed using RStudio, version 1.3.959 (R Foundation for Statistical Computing). The significance threshold was 2-sided P < .05.
Results
A total of 3599 Medicare PDPs in 2009 and 3618 Medicare PDPs in 2019 were included. In the study sample, over this period, the composition of PDP types increasingly shifted toward Medicare Advantage PDPs, with 1998 Medicare Advantage PDPs and 1601 stand-alone PDPs in 2009 compared with 2719 Medicare Advantage PDPs and 899 stand-alone PDPs in 2019.
Guideline-Recommended Treatment
A detailed list of the medication-associated recommendations abstracted from clinical practice guidelines are available in eTable 4 and eTable 5 in the Supplement. In total, for the 8 chronic conditions of interest, 15 distinct drug classes were recommended at least once in clinical practice guidelines: anticoagulants, angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, angiotensin receptor neprilysin inhibitors, β blockers, biguanides, bisphosphonates, bronchodilators, calcium channel blockers, nonsteroidal anti-inflammatory drugs, aldosterone antagonists, sodium-glucose transporter protein 2 inhibitors, statins, sulfonylureas, and thiazide diuretics. Across these 15 drug classes, the total number of medications included was 47 in 2009 and 58 in 2019 (eTable 6 in the Supplement).
Annual Out-of-Pocket Costs for Single Conditions
Between 2009 and 2019, the median inflation-adjusted annual out-of-pocket costs for older adults enrolled in any PDP decreased for 6 of the 8 chronic conditions: COPD (from $1218 [IQR, $1031-$1915] in 2009 to $845 [IQR, $701-$1143] in 2019), HFrEF (assuming approach 1: from $268 [IQR, $220-$445] in 2009 to $103 [IQR, $36-$180] in 2019), hypercholesterolemia (from $128 [IQR, $81-$181] to $36 [IQR, $11-$72] in 2019), hypertension (from $141 [IQR, $106-$194] in 2009 to $48 [IQR, $8-$75] in 2019), osteoarthritis (from $96 [IQR, $73-$120] in 2009 to $48 [IQR, $12-$84] in 2019), and osteoporosis (from $128 [IQR, $102-$183] in 2009 to $32 [IQR, $6-$48] in 2019).
For older adults enrolled in any PDP in 2019, the median annual out-of-pocket costs varied from a minimum of $32 (IQR, $6-$48) for osteoporosis to a maximum of $1579 (IQR, $1524-$2229) for atrial fibrillation (an increase from $91 [IQR, $73-$124] in 2009) (Table 1). From 2009 to 2019, the percent change in median out-of-pocket costs ranged from −75% for osteoporosis to 1634% for atrial fibrillation. Patterns of median annual out-of-pocket costs by condition were similar when stratified by stand-alone PDPs (eg, percent change from 2009-2019 ranging from −81% for osteoporosis to 2195% for atrial fibrillation) and Medicare Advantage PDPs (eg, percent change from 2009-2019 ranging from −71% for osteoporosis to 1938% for atrial fibrillation).
Table 1. Percent Change in Estimated Annual Out-of-Pocket Costs in 2009 and 2019 Among Hypothetical Patients With Specific Chronic Conditions Enrolled in Medicare Prescription Drug Plans.
| Condition | Cost, median (IQR), $a | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| All PDPs | Medicare Advantage PDPs | Stand-alone PDPs | ||||||||||
| 2009 | 2019 | Percent change | P value | 2009 | 2019 | Percent change | P value | 2009 | 2019 | Percent change | P value | |
| Single conditions | ||||||||||||
| Atrial fibrillation | 91 (73-124) | 1579 (1524-2229) | 1634 | <.001 | 76 (59-101) | 1552 (1501-1604) | 1938 | <.001 | 103 (91-143) | 2372 (2156-2655) | 2195 | <.001 |
| COPD | 1218 (1031-1915) | 845 (701-1143) | −31 | <.001 | 1162 (944-1509) | 858 (740-1252) | −26 | <.001 | 1579 (1161-1929) | 744 (656-836) | −53 | <.001 |
| Type 2 diabetes | 172 (139-212) | 992 (594-1288) | 477 | <.001 | 146 (118-199) | 1017 (592-1251) | 595 | <.001 | 186 (158-341) | 893 (669-1445) | 381 | <.001 |
| HFrEF approach 1b | 268 (220-445) | 103 (36-180) | −62 | <.001 | 230 (181-345) | 107 (10-181) | −54 | <.001 | 314 (267-571) | 82 (36-141) | −74 | <.001 |
| HFrEF approach 2c | 268 (220-445) | 1018 (663-1214) | 280 | <.001 | 230 (181-345) | 1041 (641-1203) | 352 | <.001 | 314 (267-571) | 929 (743-1343) | 196 | <.001 |
| Hypercholesterolemia | 128 (81-181) | 36 (11-72) | −72 | <.001 | 93 (73-142) | 36 (9-71) | −61 | <.001 | 155 (128-845) | 36 (12-90) | −77 | <.001 |
| Hypertension | 141 (106-194) | 48 (8-75) | −66 | <.001 | 140 (85-174) | 48 (7-78) | −66 | <.001 | 168 (130-195) | 36 (24-60) | −78 | <.001 |
| Osteoarthritis | 96 (73-120) | 48 (12-84) | −50 | <.001 | 75 (48-101) | 48 (12-96) | −36 | <.001 | 107 (95-322) | 37 (12-61) | −65 | <.001 |
| Osteoporosis | 128 (102-183) | 32 (6-48) | −75 | <.001 | 115 (81-141) | 34 (6-49) | −71 | <.001 | 141 (116-591) | 27 (12-48) | −81 | <.001 |
| Multiple chronic conditions | ||||||||||||
| Cluster A conditionsd | 2284 (1920-3107) | 1999 (1630-2564) | −12 | <.001 | 2199 (1771-2459) | 2148 (1699-2789) | −2 | .46 | 2880 (2209-3406) | 1704 (1539-2182) | −41 | <.001 |
| Cluster B conditionse | 2571 (2185-3719) | 3630 (3234-5197) | 41 | <.001 | 2436 (2009-2853) | 3553 (3210-5172) | 46 | <.001 | 3196 (2511-4847) | 3750 (3472-5279) | 17 | <.001 |
Abbreviations: COPD, chronic obstructive pulmonary disease; HFrEF, heart failure with reduced ejection fraction; PDP, prescription drug plan.
Out-of-pocket costs were inflation adjusted to 2019 dollars.
Approach 1 assumed the patient was prescribed an angiotensin-converting enzyme inhibitor or angiotensin receptor blocker, an evidence-based β blocker, and an aldosterone antagonist in 2019.
Approach 2 assumed the patient was prescribed an angiotensin receptor neprilysin inhibitor (sacubitril-valsartan), an evidence-based β blocker, and an aldosterone antagonist in 2019.
Cluster A conditions include COPD, type 2 diabetes, hypertension, osteoarthritis, and osteoporosis.
Cluster B conditions include atrial fibrillation, COPD, type 2 diabetes, HFrEF, hypercholesterolemia, hypertension, osteoarthritis, and osteoporosis. For HFrEF, it was assumed the patient was prescribed an angiotensin-converting enzyme inhibitor or angiotensin receptor blocker, an evidence-based β blocker, and an aldosterone antagonist in 2019.
In 2009, patients with any single condition had higher median annual out-of-pocket costs when enrolled in stand-alone vs Medicare Advantage PDPs (eg, COPD: $1579 [IQR, $1161-$1929] vs $1162 [IQR, $944-$1509]; P < .001) (Table 2). In 2019, only patients with atrial fibrillation had higher median annual out-of-pocket costs when enrolled in stand-alone vs Medicare Advantage PDPs ($2372 [IQR, $2156-$2655] vs $1522 [IQR, $1501-$1604]; P < .001). Median annual out-of-pocket costs in 2019 were similar among patients enrolled in stand-alone vs Medicare Advantage PDPs for guideline-recommended management of type 2 diabetes ($893 [IQR, $669-$1445] vs $1017 [IQR, $592-$1251]; P = .72), HFrEF (assuming approach 2: $929 [IQR, $743-$1343] vs $1041 [IQR, $641-$1203]; P = .60), hypercholesterolemia ($36 [IQR, $12-$90] vs $36 [IQR, $9-$71]; P = .48), or osteoporosis ($27 [IQR, $12-$48] vs $34 [IQR, $6-$49]; P = .46). Higher median annual out-of-pocket costs in 2019 were observed among patients enrolled in Medicare Advantage vs stand-alone PDPs for guideline-recommended management of COPD ($858 [IQR, $740-$1252] vs $744 [IQR, $656-$836]; P < .001), HFrEF (assuming approach 1: $107 [IQR, $10-$181] vs $82 [IQR, $36-$141]; P < .001), hypertension ($48 [IQR, $7-$78] vs $36 [IQR, $24-$60]; P < .001), or osteoarthritis ($48 [IQR, $12-$96] vs $37 [IQR, $12-$61]; P < .001).
Table 2. Estimated Annual Out-of-Pocket Costs in 2009 and 2019 Among Hypothetical Patients With Specific Chronic Conditions Enrolled in Medicare Advantage and Medicare Stand-Alone Prescription Drug Plans.
| Condition | Cost, median (IQR), $a | |||||
|---|---|---|---|---|---|---|
| 2009 | 2019 | |||||
| Medicare Advantage PDP | Stand-alone PDP | P value | Medicare Advantage PDP | Stand-alone PDP | P value | |
| Single conditions | ||||||
| Atrial fibrillation | 76 (59-101) | 103 (91-143) | <.001 | 1552 (1501-1604) | 2372 (2156-2655) | <.001 |
| COPD | 1162 (944-1509) | 1579 (1161-1929) | <.001 | 858 (740-1252) | 744 (656-836) | <.001 |
| Type 2 diabetes | 146 (118-199) | 186 (158-341) | <.001 | 1017 (592-1251) | 893 (669-1445) | .72 |
| HFrEF approach 1b | 230 (181-345) | 314 (267-571) | <.001 | 107 (10-181) | 82 (36-141) | <.001 |
| HFrEF approach 2c | NA | NA | NA | 1041 (641-1203) | 929 (743-1343) | .60 |
| Hypercholesterolemia | 93 (73-142) | 155 (128-845) | <.001 | 36 (9-71) | 36 (12-90) | .48 |
| Hypertension | 140 (85-174) | 168 (130-195) | <.001 | 48 (7-78) | 36 (24-60) | <.001 |
| Osteoarthritis | 75 (48-101) | 107 (95-322) | <.001 | 48 (12-96) | 37 (12-61) | <.001 |
| Osteoporosis | 115 (81-141) | 141 (116-591) | <.001 | 34 (6-49) | 27 (12-48) | .46 |
| Multiple chronic conditions | ||||||
| Cluster A conditionsd | 2199 (1771-2459) | 2880 (2209-3406) | <.001 | 2148 (1699-2789) | 1704 (1539-2182) | <.001 |
| Cluster B conditionse | 2436 (2009-2853) | 3196 (2511-4847) | <.001 | 3553 (3210-5172) | 3750 (3472-5279) | <.001 |
Abbreviations: COPD, chronic obstructive pulmonary disease; HFrEF, heart failure with reduced ejection fraction; NA, not applicable; PDP, prescription drug plan.
Out-of-pocket costs were inflation adjusted to 2019 dollars.
Approach 1 assumed the patient was prescribed an angiotensin-converting enzyme inhibitor or angiotensin receptor blocker, an evidence-based β blocker, and an aldosterone antagonist in 2019.
Approach 2 assumed the patient was prescribed an angiotensin receptor neprilysin inhibitor (sacubitril-valsartan), an evidence-based β blocker, and an aldosterone antagonist in 2019.
Cluster A conditions include COPD, type 2 diabetes, hypertension, osteoarthritis, and osteoporosis.
Cluster B conditions include atrial fibrillation, COPD, type 2 diabetes, HFrEF, hypercholesterolemia, hypertension, osteoarthritis, and osteoporosis. For HFrEF, it was assumed the patient was prescribed an angiotensin-converting enzyme inhibitor or angiotensin receptor blocker, an evidence-based β blocker, and an aldosterone antagonist in 2019.
Annual Out-of-Pocket Costs for Multiple Chronic Conditions
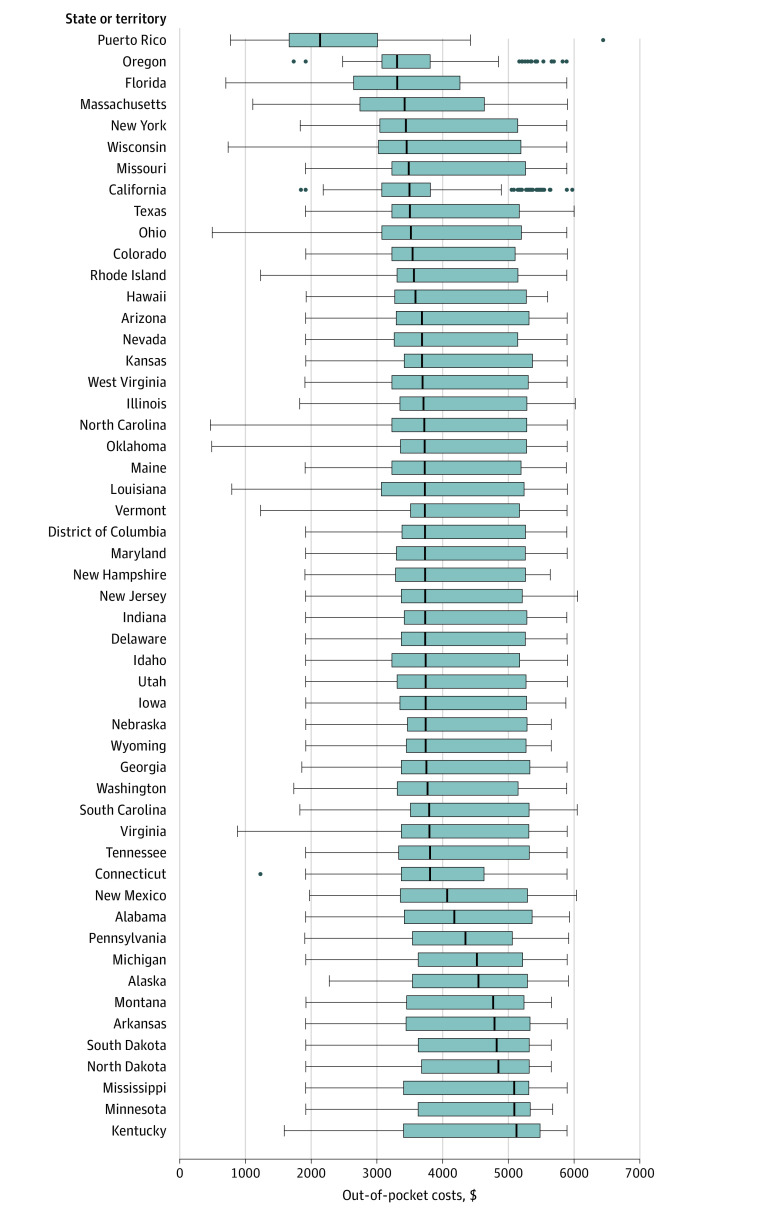
For an older adult enrolled in any PDP receiving treatment for the 5 commonly comorbid cluster A conditions, the median annual out-of-pocket costs were $1999 (IQR, $1630-$2564) in 2019, a 12% decrease from $2284 (IQR, $1920-$3107) in 2009 (Table 1). Among those with Medicare Advantage PDPs, the median 2019 out-of-pocket costs were $2148 (IQR, $1699-$2789), a 2% decrease from $2199 (IQR, $1771-$2459) in 2009. Among those with stand-alone PDPs, the median 2019 out-of-pocket costs were $1704 (IQR, $1539-$2182), a 41% decrease from $2880 (IQR, $2209-$3406) in 2009. State-level variations in annual out-of-pocket costs for cluster A conditions are shown in Figure 1.
Figure 1. Out-of-Pocket Costs for Cluster A Conditions in 2019.
Includes costs for all prescription drug plans. Cluster A conditions include chronic obstructive pulmonary disease, type 2 diabetes, hypertension, osteoarthritis, and osteoporosis. The box plot lines, from left end of each box to the right end, represent the 25th percentile, median percentile, and 75th percentile. The whiskers extend to the minimum and maximum values but no further than 1.5 times the IQR from the 25th and 75th percentiles. Dots beyond the end of the whiskers represent outliers.
For an older adult enrolled in any PDP receiving treatment for all 8 commonly comorbid cluster B conditions, the median out-of-pocket costs were $3630 (IQR, $3234-$5197) in 2019, a 41% increase from $2571 (IQR, $2185-$3719) in 2009 (Table 1). Among those with Medicare Advantage PDPs, the median 2019 out-of-pocket costs were $3553 (IQR, $3210-$5172), a 46% increase from $2436 (IQR, $2009-$2853) in 2009. Among those with stand-alone PDPs, the median 2019 out-of-pocket costs were $3750 (IQR, $3472-$5279), a 17% increase from $3196 (IQR, $2511-$4847) in 2009. State-level variations in annual out-of-pocket costs for cluster B conditions are shown in Figure 2.
Figure 2. Out-of-Pocket Costs for Cluster B Conditions in 2019.
Includes costs for all prescription drug plans. Cluster B conditions include atrial fibrillation, chronic obstructive pulmonary disease, type 2 diabetes, hypercholesterolemia, heart failure with reduced ejection fraction (assuming the patient was prescribed an angiotensin-converting enzyme inhibitor or angiotensin receptor blocker), hypertension, osteoarthritis, and osteoporosis. The box plot lines, from the left end of each box to the right end, represent the 25th percentile, median percentile, and 75th percentile. The whiskers extend to the minimum and maximum values but no further than 1.5 times the IQR from the 25th and 75th percentiles. Dots beyond the end of the whiskers represent outliers.
In 2019, patients receiving treatment for the 5 commonly comorbid cluster A conditions had higher median annual out-of-pocket costs when enrolled in Medicare Advantage vs stand-alone PDPs ($2148 [IQR, $1699-$2789] vs $1704 [IQR, $1539-$2182]; P < .001) (Table 2). In contrast, patients in 2019 receiving treatment for all 8 commonly comorbid cluster B conditions had higher median annual out-of-pocket costs when enrolled in stand-alone vs Medicare Advantage PDPs ($3750 [IQR, $3472-$5279] vs $3553 [IQR, $3210-$5172]; P < .001).
Discussion
In response to increasing prescription drug costs and to better understand the impact of the Medicare Prescription Drug, Improvement, and Modernization Act for out-of-pocket medication costs among Medicare beneficiaries, this cross-sectional study estimated the median inflation-adjusted annual out-of-pocket costs associated with guideline-recommended outpatient medical therapy for Medicare beneficiaries with 8 common chronic conditions in 2009 and 2019. We found wide variation in out-of-pocket costs across chronic conditions. During this period, both generic and brand-name medications entered the market. For many common conditions, particularly conditions for which generic drugs were guideline recommended, annual out-of-pocket costs decreased. However, among those conditions for which brand-name drugs without generic versions became guideline recommended, out-of-pocket costs increased substantially between 2009 and 2019. These findings suggest that not all older adults experience equal burdens associated with out-of-pocket medication costs. Furthermore, our results suggest the need for both upstream solutions, such as drug pricing negotiations, as well as downstream solutions, such as capping out-of-pocket spending for prescription drugs among Medicare beneficiaries. In addition, physicians should be encouraged to engage in both deprescribing efforts and shared decision-making regarding the cost burden for individual patients to mitigate the overall financial burden of managing multiple chronic conditions.
For specific single chronic conditions, we found wide variations in out-of-pocket costs for Medicare beneficiaries. The greatest increases were for treatment of conditions such as atrial fibrillation, HFrEF, and type 2 diabetes and were associated with professional clinical guidelines recommending newer and more expensive therapies, such as sodium-glucose cotransporter 2 inhibitors, direct oral anticoagulants, and angiotensin receptor neprilysin inhibitors. Moreover, as new evidence has been generated suggesting improvements in clinical outcomes associated with these drugs, including evidence to support their use for other indications, they have been increasingly prescribed.24 For example, direct oral anticoagulants are also recommended for the treatment of symptomatic deep vein thrombosis and pulmonary embolism.25 After our study period, sodium-glucose cotransporter 2 inhibitors were recommended for the treatment of patients with HFrEF, which means a larger patient population is now likely to receive these medications.26 In addition, the newer drugs included in this study may continue to be potentially underused among patients who might benefit clinically27 but for whom costs may be prohibitive.28 Although newer medications may be associated with improvements in clinical outcomes, an increasing number of older adults will likely be exposed to the financial burden of treatment in coming years.
Comparing our estimated costs in 2019 with those reported by Boyd et al14 in 2004, it appears that the Medicare Prescription Drug, Improvement, and Modernization Act has substantially mitigated the impact of out-of-pocket medication costs for Medicare beneficiaries. Median annual out-of-pocket costs for the treatment of cluster A conditions decreased 69% (from $6544 in 200414 [inflation-adjusted to June 2019] to $1999 in 2019); these out-of-pocket costs represented 6.7% of the annual income of an average Medicare beneficiary in 2019 compared with more than 20% in 2004.6,14,16 However, drug pricing remains a problem for many patients, even those with Medicare PDP coverage. We also observed that stand-alone PDPs cost more than Medicare Advantage PDPs for the medication management of all 8 single conditions in 2009, whereas stand-alone PDPs cost more than Medicare Advantage PDPs only for the management of atrial fibrillation in 2019. Further research may clarify the reasons for these variations. Other studies have observed differences in medication spending and access with regard to racial disparities and state-level variations.29,30,31
Excessive out-of-pocket costs have been associated with worse medication adherence and, as a result, worse patient outcomes.32,33 One in 4 Medicare beneficiaries reported difficulties affording medications in 2019.34 Based on 2020 patterns in drug pricing and cost sharing, it has been estimated that 112 000 Medicare beneficiaries will die annually because of cost-associated medication nonadherence.35 Policy makers have considered upstream solutions to mitigate the financial burden of managing multiple chronic conditions, including legislation removing the noninterference clause, thereby enabling the Department of Health and Human Services to negotiate maximum drug prices and introduce measures to increase the availability of generic drugs.17 Previous studies reported that competition among brand-name drugs might not help reduce costs,19 but the availability of generic drugs may have an impact for prices.36 Notably, to be successful at mitigating costs for beneficiaries, Medicare PDPs might incentivize the use of bioequivalent and therapeutically interchangeable generic drugs.37
In addition, a number of downstream solutions have been proposed to mitigate the financial burden of managing multiple chronic conditions, including capping Medicare beneficiaries’ annual out-of-pocket spending for all prescription drugs.38 Clinical practice guidelines are also increasingly providing recommendations based on the cost and value of medical therapies39; however, greater adoption and transparency of medication cost considerations in clinical practice guidelines may further benefit patients.40 At the federal and state levels, proposals have been considered to anchor prices using value assessments.41,42,43 Clinicians may also further alleviate patients’ out-of-pocket cost burden through deprescribing efforts, which are acceptable to most Medicare beneficiaries.44,45
Limitations
This study has several limitations. First, we modeled treatment of hypothetical patients using clinical practice guidelines for pharmacological therapy recommendations. We did not integrate recommendations from the American Geriatrics Society46 or Choosing Wisely,47 and we did not account for nonpharmacological interventions, deprescribing efforts, or lack of adherence to guideline recommendations among physicians. Furthermore, our cost estimates assume adherence to guideline-recommended medical therapies over a 1-year period, although there may have been periods when beneficiaries did not purchase medications because of costs. Among high-income countries, the US has the highest prevalence of cost-associated nonadherence, including nonadherence among 16.8% of adults older than 55 years.48 Research has found that price increases are associated with fewer patients filling their drug prescriptions, regardless of health risks or the number of drugs prescribed.49
Second, our estimates do not account for patient preferences for specific drugs, including potential preferences for brand-name versions of drugs when generic versions are available.50 Third, our estimates are based on preferred in-network retail pharmacies, which do not reflect the experience of approximately 17% of Medicare Part D beneficiaries who use mail-order pharmacies,22 beneficiaries who choose to obtain prescription drugs through generic drug discount programs without using their insurance benefits,51,52 and beneficiaries who have other copayment offsets.53
Fourth, our estimates of annual out-of-pocket costs were deliberately conservative because we preferentially assumed that beneficiaries would be prescribed the lowest-cost medication option and because we did not account for insurance premiums, a major component of prescription drug coverage costs. Fifth, our estimates do not include other associated costs, such as travel for medical treatment, home modifications, caregiver expenses, and loss of employment income; we also do not account for the low-income subsidy, for which 30% of Medicare Part D beneficiaries were eligible in 2018.54
Conclusions
In this cross-sectional study, out-of-pocket costs for guideline-recommended outpatient medications for the initial treatment of 8 common chronic diseases varied by condition. Although costs generally decreased between 2009 and 2019, particularly for the treatment of conditions for which generic drugs were available, out-of-pocket costs remained high and may have presented a substantial financial burden for Medicare beneficiaries, especially those with conditions for which brand-name drugs were guideline recommended.
eTable 1. Abbreviations Used in Supplementary Online Content
eTable 2. Clinical Practice Guidelines
eTable 3. Characteristics of Hypothetical Patients
eTable 4. Recommendations From Clinical Practice Guidelines as of 2009
eTable 5. Recommendations From Clinical Practice Guidelines as of 2019
eTable 6. Drugs Included in Study Sample
eTable 7. Study Sample (No. of Plans) Included by Disease, PDP Type, and Year
References
- 1.Centers for Medicare & Medicaid Services . Chronic conditions among Medicare beneficiaries: chartbook: 2017 edition. Accessed August 20, 2020. https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/Chronic-Conditions/Downloads/cc_charts.zip
- 2.Field TS, Gurwitz JH, Harrold LR, et al. Risk factors for adverse drug events among older adults in the ambulatory setting. J Am Geriatr Soc. 2004;52(8):1349-1354. doi: 10.1111/j.1532-5415.2004.52367.x [DOI] [PubMed] [Google Scholar]
- 3.Gurwitz JH, Field TS, Harrold LR, et al. Incidence and preventability of adverse drug events among older persons in the ambulatory setting. JAMA. 2003;289(9):1107-1116. doi: 10.1001/jama.289.9.1107 [DOI] [PubMed] [Google Scholar]
- 4.Juurlink DN, Mamdani M, Kopp A, Laupacis A, Redelmeier DA. Drug-drug interactions among elderly patients hospitalized for drug toxicity. JAMA. 2003;289(13):1652-1658. doi: 10.1001/jama.289.13.1652 [DOI] [PubMed] [Google Scholar]
- 5.Flaherty JH, Perry HM III, Lynchard GS, Morley JE. Polypharmacy and hospitalization among older home care patients. J Gerontol A Biol Sci Med Sci. 2000;55(10):M554-M559. doi: 10.1093/gerona/55.10.M554 [DOI] [PubMed] [Google Scholar]
- 6.Koma W, Neuman T, Jacobson G, Smith K. Medicare beneficiaries’ financial security before the coronavirus pandemic. Kaiser Family Foundation. April 24, 2020. Accessed June 19, 2021. https://www.kff.org/medicare/issue-brief/medicare-beneficiaries-financial-security-before-the-coronavirus-pandemic/
- 7.Shippee ND, Shah ND, May CR, Mair FS, Montori VM. Cumulative complexity: a functional, patient-centered model of patient complexity can improve research and practice. J Clin Epidemiol. 2012;65(10):1041-1051. doi: 10.1016/j.jclinepi.2012.05.005 [DOI] [PubMed] [Google Scholar]
- 8.Social Security Administration . Compilation of the Social Security laws: title XVIII—health insurance for the aged and disabled. Social Security Administration; 2021. Accessed July 19, 2021. https://www.ssa.gov/OP_Home/ssact/title18/1800.htm
- 9.Briesacher BA, Zhao Y, Madden JM, et al. Medicare Part D and changes in prescription drug use and cost burden: national estimates for the Medicare population, 2000 to 2007. Med Care. 2011;49(9):834-841. doi: 10.1097/MLR.0b013e3182162afb [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Briesacher BA, Ross-Degnan D, Wagner AK, et al. Out-of-pocket burden of health care spending and the adequacy of the Medicare Part D low-income subsidy. Med Care. 2010;48(6):503-509. doi: 10.1097/MLR.0b013e3181dbd8d3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dusetzina SB, Keating NL, Huskamp HA. Proposals to redesign Medicare Part D—easing the burden of rising drug prices. N Engl J Med. 2019;381(15):1401-1404. doi: 10.1056/NEJMp1908688 [DOI] [PubMed] [Google Scholar]
- 12.Dusetzina SB, Conti RM, Yu NL, Bach PB. Association of prescription drug price rebates in Medicare Part D with patient out-of-pocket and federal spending. JAMA Intern Med. 2017;177(8):1185-1188. doi: 10.1001/jamainternmed.2017.1885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pearson CF, Brantley K, Frieder M. Majority of drugs now subject to coinsurance in Medicare Part D plans. Avalere Health. March 10, 2016. Accessed March 14, 2021. https://avalere.com/press-releases/majority-of-drugs-now-subject-to-coinsurance-in-medicare-part-d-plans
- 14.Boyd CM, Darer J, Boult C, Fried LP, Boult L, Wu AW. Clinical practice guidelines and quality of care for older patients with multiple comorbid diseases: implications for pay for performance. JAMA. 2005;294(6):716-724. doi: 10.1001/jama.294.6.716 [DOI] [PubMed] [Google Scholar]
- 15.US Bureau of Labor Statistics . CPI inflation calculator. US Bureau of Labor Statistics. Accessed June 19, 2020. https://www.bls.gov/data/inflation_calculator.htm
- 16.Foster R, Clemens MK. Additional information regarding comparisons of beneficiary income and out-of-pocket costs for Medicare supplementary medical insurance. Memorandum. Office of the Actuary, Centers for Medicare & Medicaid Services, Department of Health & Human Services. March 25, 2008. Accessed July 19, 2021. https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/ReportsTrustFunds/downloads/Beneficiaryoop.pdf
- 17.Gavulic KA, Dusetzina SB. Prescription drug priorities under the Biden administration. J Health Polit Policy Law. 2021;46(4):599-609. doi: 10.1215/03616878-8970810 [DOI] [PubMed] [Google Scholar]
- 18.Hernandez I, San-Juan-Rodriguez A, Good CB, Gellad WF. Changes in list prices, net prices, and discounts for branded drugs in the US, 2007-2018. JAMA. 2020;323(9):854-862. doi: 10.1001/jama.2020.1012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Liu P, Dhruva SS, Shah ND, Ross JS. Trends in within-class changes in US average wholesale prices for brand-name medications for common conditions from 2015 to 2020. JAMA Netw Open. 2021;4(1):e2035064. doi: 10.1001/jamanetworkopen.2020.35064 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Halperin JL, Levine GN, Al-Khatib SM, et al. Further evolution of the ACC/AHA clinical practice guideline recommendation classification system: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2016;133(14):1426-1428. [DOI] [PubMed] [Google Scholar]
- 21.Fihn SD, Gardin JM, Abrams J, et al. ; American College of Cardiology Foundation . 2012 ACCF/AHA/ACP/AATS/PCNA/SCAI/STS guideline for the diagnosis and management of patients with stable ischemic heart disease: executive summary: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, and the American College of Physicians, American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. Circulation. 2012;126(25):3097-3137. doi: 10.1161/CIR.0b013e3182776f83 [DOI] [PubMed] [Google Scholar]
- 22.Cubanski BJ, Biniek JF, Rae M, Damico A, Frederiksen B, Salganicoff A. Mail delays could affect mail-order prescriptions for millions of Medicare Part D and large employer plan enrollees. Kaiser Family Foundation. August 20, 2020. Accessed July 19, 2021. https://www.kff.org/coronavirus-covid-19/issue-brief/mail-delays-could-affect-mail-order-prescriptions-for-millions-of-medicare-part-d-and-large-employer-plan-enrollees/
- 23.Cubanski J, Neuman T, Damico A. Closing the Medicare Part D coverage gap: trends, recent changes, and what’s ahead. Kaiser Family Foundation. August 21, 2018. Accessed June 19, 2021. https://www.kff.org/medicare/issue-brief/closing-the-medicare-part-d-coverage-gap-trends-recent-changes-and-whats-ahead
- 24.Ziakas PD, Kourbeti IS, Poulou LS, Vlachogeorgos GS, Mylonakis E. Medicare Part D prescribing for direct oral anticoagulants in the United States: cost, use and the “rubber effect”. PLoS One. 2018;13(6):e0198674. doi: 10.1371/journal.pone.0198674 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ortel TL, Neumann I, Ageno W, et al. American Society of Hematology 2020 guidelines for management of venous thromboembolism: treatment of deep vein thrombosis and pulmonary embolism. Blood Adv. 2020;4(19):4693-4738. doi: 10.1182/bloodadvances.2020001830 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Maddox TM, Januzzi JL Jr, Allen LA, et al. ; Writing Committee . 2021 Update to the 2017 ACC expert consensus decision pathway for optimization of heart failure treatment: answers to 10 pivotal issues about heart failure with reduced ejection fraction: a report of the American College of Cardiology Solution Set Oversight Committee. J Am Coll Cardiol. 2021;77(6):772-810. doi: 10.1016/j.jacc.2020.11.022 [DOI] [PubMed] [Google Scholar]
- 27.McCoy RG, Dykhoff HJ, Sangaralingham L, et al. Adoption of new glucose-lowering medications in the U.S.—the case of SGLT2 inhibitors: nationwide cohort study. Diabetes Technol Ther. 2019;21(12):702-712. doi: 10.1089/dia.2019.0213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.McCoy RG, Van Houten HK, Deng Y, et al. Comparison of diabetes medications used by adults with commercial insurance vs Medicare Advantage, 2016 to 2019. JAMA Netw Open. 2021;4(2):e2035792. doi: 10.1001/jamanetworkopen.2020.35792 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lochner KA, Goodman RA, Posner S, Parekh A. Multiple chronic conditions among Medicare beneficiaries: state-level variations in prevalence, utilization, and cost, 2011. Medicare Medicaid Res Rev. 2013;3(3):mmrr.003.03.b02. doi: 10.5600/mmrr.003.03.b02 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chakravarty S. Did the Medicare prescription drug program lead to new racial and ethnic disparities? examining long-term changes in prescription drug access among minority populations. Soc Work Public Health. 2020;35(5):248-260. doi: 10.1080/19371918.2020.1785981 [DOI] [PubMed] [Google Scholar]
- 31.Clements JM, Ragina NP, Killingsworth J, et al. Differences in spending for diabetes and multiple chronic conditions in Michigan Medicare beneficiaries. Am J Manag Care. 2020;26(11):e362-e368. doi: 10.37765/ajmc.2020.88531 [DOI] [PubMed] [Google Scholar]
- 32.Khera R, Valero-Elizondo J, Das SR, et al. Cost-related medication nonadherence in adults with atherosclerotic cardiovascular disease in the United States, 2013 to 2017. Circulation. 2019;140(25):2067-2075. doi: 10.1161/CIRCULATIONAHA.119.041974 [DOI] [PubMed] [Google Scholar]
- 33.Bansilal S, Castellano JM, Garrido E, et al. Assessing the impact of medication adherence on long-term cardiovascular outcomes. J Am Coll Cardiol. 2016;68(8):789-801. doi: 10.1016/j.jacc.2016.06.005 [DOI] [PubMed] [Google Scholar]
- 34.Kaiser Family Foundation . Poll: nearly 1 in 4 Americans taking prescription drugs say it’s difficult to afford their medicines, including larger shares among those with health issues, with low incomes and nearing Medicare age. Kaiser Family Foundation. March 1, 2019. Accessed June 19, 2021. https://www.kff.org/health-costs/press-release/poll-nearly-1-in-4-americans-taking-prescription-drugs-say-its-difficult-to-afford-medicines-including-larger-shares-with-low-incomes/
- 35.Council for Informed Drug Spending Analysis . High drug prices and patient costs: millions of lives and billions of dollars lost. November 18, 2020. Accessed July 19, 2021. https://www.cidsa.org/publications/xcenda-summary
- 36.Warraich HJ, Salami JA, Khera R, Valero-Elizondo J, Okunrintemi V, Nasir K. Trends in use and expenditures of brand-name atorvastatin after introduction of generic atorvastatin. JAMA Intern Med. 2018;178(5):719-721. doi: 10.1001/jamainternmed.2018.0990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Vijay A, Gupta R, Liu P, Dhruva SS, Shah ND, Ross JS. Medicare formulary coverage of brand-name drugs and therapeutically interchangeable generics. J Gen Intern Med. 2020;35(6):1928-1930. doi: 10.1007/s11606-019-05432-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cubanski J, Neuman T, Orgera K, Damico A. No limit: Medicare Part D enrollees exposed to high out-of-pocket drug costs without a hard cap on spending. Issue brief. The Henry J. Kaiser Family Foundation. November 2017. Accessed July 19, 2021. https://files.kff.org/attachment/Issue-Brief-No-Limit-Medicare-Part-D-Enrollees-Exposed-to-High-Out-of-Pocket-Drug-Costs-Without-a-Hard-Cap-on-Spending
- 39.Buse JB, Wexler DJ, Tsapas A, et al. 2019 Update to: management of hyperglycemia in type 2 diabetes, 2018. a consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2020;43(2):487-493. doi: 10.2337/dci19-0066 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Schwartz JAT, Pearson SD. Cost consideration in the clinical guidance documents of physician specialty societies in the United States. JAMA Intern Med. 2013;173(12):1091-1097. doi: 10.1001/jamainternmed.2013.817 [DOI] [PubMed] [Google Scholar]
- 41.Dusetzina SB, Mello MM. Drug pricing reform in 2021—going big or going bipartisan? JAMA Health Forum. 2021;2(7):e212372. doi: 10.1001/jamahealthforum.2021.2372 [DOI] [PubMed] [Google Scholar]
- 42.Anderson JL, Heidenreich PA, Barnett PG, et al. ; ACC/AHA Task Force on Performance Measures; ACC/AHA Task Force on Practice Guidelines . ACC/AHA statement on cost/value methodology in clinical practice guidelines and performance measures: a report of the American College of Cardiology/American Heart Association Task Force on Performance Measures and Task Force on Practice Guidelines. Circulation. 2014;129(22):2329-2345. doi: 10.1161/CIR.0000000000000042 [DOI] [PubMed] [Google Scholar]
- 43.National Academy for State Health Policy . Comparison of provisions of states’ current drug affordability review board proposals. National Academy for State Health Policy. Updated March 19, 2019. Accessed July 19, 2021. https://www.nashp.org/comparison-of-bills-creating-state-prescription-drug-affordability-review-boards/
- 44.Halli-Tierney AD, Scarbrough C, Carroll D. Polypharmacy: evaluating risks and deprescribing. Am Fam Physician. 2019;100(1):32-38. [PubMed] [Google Scholar]
- 45.Reeve E, Wolff JL, Skehan M, Bayliss EA, Hilmer SN, Boyd CM. Assessment of attitudes toward deprescribing in older Medicare beneficiaries in the United States. JAMA Intern Med. 2018;178(12):1673-1680. doi: 10.1001/jamainternmed.2018.4720 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.American Geriatrics Society. Publications and tools: AGS guidelines, recommendations, and position statements. 2021. Accessed November 23, 2021. https://www.americangeriatrics.org/publications-tools
- 47.American Academy of Family Physicians. Clinical practice guidelines: choosing Wisely. 2021. Accessed November 23, 2021. https://www.aafp.org/family-physician/patient-care/clinical-recommendations/clinical-practice-guidelines/choosing-wisely.html
- 48.Morgan SG, Lee A. Cost-related non-adherence to prescribed medicines among older adults: a cross-sectional analysis of a survey in 11 developed countries. BMJ Open. 2017;7(1):e014287. doi: 10.1136/bmjopen-2016-014287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Chandra A, Flack E, Obermeyer Z. The health costs of cost-sharing. National Bureau of Economic Research; February 2021. Working paper 28439. Accessed October 9, 2021. doi: 10.3386/w28439 [DOI]
- 50.Kesselheim AS, Gagne JJ, Franklin JM, et al. Variations in patients’ perceptions and use of generic drugs: results of a national survey. J Gen Intern Med. 2016;31(6):609-614. doi: 10.1007/s11606-016-3612-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Liu P, Dhruva SS, Shah ND, Ross JS. Medicare beneficiary out-of-pocket costs for generic cardiovascular medications available through $4 generic drug discount programs. Ann Intern Med. 2018;169(11):817-819. doi: 10.7326/M18-0965 [DOI] [PubMed] [Google Scholar]
- 52.Trish E, Gascue L, Ribero R, Van Nuys K, Joyce G. Comparison of spending on common generic drugs by Medicare vs Costco members. JAMA Intern Med. 2021;181(10):1414-1416. doi: 10.1001/jamainternmed.2021.3366 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sen AP, Kang SY, Rashidi E, Ganguli D, Anderson G, Alexander GC. Characteristics of copayment offsets for prescription drugs in the United States. JAMA Intern Med. 2021;181(6):758-764. doi: 10.1001/jamainternmed.2021.0733 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kaiser Family Foundation. An overview of the Medicare Part D prescription drug benefit. Kaiser Family Foundation. October 13, 2021. Accessed June 19, 2021. https://www.kff.org/medicare/fact-sheet/an-overview-of-the-medicare-part-d-prescription-drug-benefit/
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
eTable 1. Abbreviations Used in Supplementary Online Content
eTable 2. Clinical Practice Guidelines
eTable 3. Characteristics of Hypothetical Patients
eTable 4. Recommendations From Clinical Practice Guidelines as of 2009
eTable 5. Recommendations From Clinical Practice Guidelines as of 2019
eTable 6. Drugs Included in Study Sample
eTable 7. Study Sample (No. of Plans) Included by Disease, PDP Type, and Year