Abstract
Purposes
To delineate operational changes in Kaiser Permanente Northern California breast care and evaluate the impact of these changes during the initial COVID-19 Shelter-in-Place period (SiP, 3/17/20–5/17/20).
Methods
By extracting data from institutional databases and reviewing electronic medical charts, we compared clinical and treatment characteristics of breast cancer patients diagnosed 3/17/20–5/17/20 to those diagnosed 3/17/19–5/17/2019. Outcomes included time from biopsy to consultation and treatment. Comparisons were made using Chi-square or Wilcoxon rank-sum tests.
Results
Fewer new breast cancers were diagnosed in 2020 during the SiP period than during a similar period in 2019 (n = 247 vs n = 703). A higher percentage presented with symptomatic disease in 2020 than 2019 (78% vs 37%, p < 0.001). Higher percentages of 2020 patients presented with grade 3 (37% vs 25%, p = 0.004) and triple-negative tumors (16% vs 10%, p = 0.04). A smaller percentage underwent surgery first in 2020 (71% vs 83%, p < 0.001) and a larger percentage had neoadjuvant chemotherapy (16% vs 11%, p < 0.001). Telehealth utilization increased from 0.8% in 2019 to 70.0% in 2020. Times to surgery and neoadjuvant chemotherapy were shorter in 2020 than 2019 (19 vs 26 days, p < 0.001, and 23 vs 28 days, p = 0.03, respectively).
Conclusions
During SiP, fewer breast cancers were diagnosed than during a similar period in 2019, and a higher proportion presented with symptomatic disease. Early-stage breast cancer diagnoses decreased, while metastatic cancer diagnoses remained similar. Telehealth increased significantly, and times to treatment were shorter in 2020 than 2019. Our system continued to provide timely breast cancer treatment despite significant pandemic-driven disruption.
Keywords: COVID-19, Presentation, Treatment times, Telehealth, Breast
Introduction
The COVID-19 pandemic has disrupted all aspects of healthcare, including the diagnosis and treatment of breast cancer. In March 2020, organizations issued guidelines for cancer care to help balance the urgency of cancer treatment against the need to conserve hospital resources and minimize exposure to COVID-19 [1–3]. Recommendations included delaying breast cancer surgery when possible by initiating neoadjuvant chemotherapy (NAC) for triple-negative and HER2 + cancers and neoadjuvant endocrine therapy (NET) for some early-stage ER + HER2- cancers. However, with uncertainty about the severity and duration of the pandemic, as well as variability in facility resources and patient factors, determining which guidelines to adopt and to what extent was unclear. Several groups have described guideline-driven changes to their institutional breast care workflows during the initial phases of the pandemic [4–8], but data are limited on the clinical impact of pandemic-driven changes [9–13]. Furthermore, these reports have revealed significant variability in guideline adherence between institutions and even between physicians within the same institution [14].
Kaiser Permanente Northern California (KPNC), a large integrated health care system, charged breast cancer subspecialty directors with reviewing published guidelines, evaluating their applicability to KPNC, and recommending operational changes for breast cancer care in response to California’s initial shelter-in-place (SiP) order on March 17, 2020. By May 17, 2020, the number of COVID infections had stabilized and KPNC resumed more normal workflows, including restarting elective surgeries which had been held since March 17. In this study, we describe the operational changes implemented during SiP (March 17—May 17, 2020), and the clinical impact of these changes on the diagnosis and treatment of breast cancer in KPNC.
Methods
KPNC is an integrated health system in Northern California serving 4.7 million patients across 21 medical centers with more than 250 outpatient facilities. This system provides both insurance coverage and healthcare to its members. KPNC membership represents the demographics of the Northern California population except at the extremes of income. This retrospective review was approved by the KPNC Institutional Review Board.
Operational changes during the initial SiP period (March 17 – May 17, 2020)
To minimize exposure to the COVID-19 virus during the initial pandemic period, KPNC leadership asked breast subspecialty leaders, most of whom are authors of this study, to develop and disseminate regional guidelines to reduce in-person visits and elective procedures for breast care (radiology, VA; surgical oncology, SBC and GK; medical oncology, TK and ET; radiation oncology, MP and SS). In the Results section, we summarize the systemwide operational change guidelines we developed and disseminated.
Study design, variables, and outcomes
Table 1 summarizes the study design. We identified patients with breast cancer diagnosed March 17—May 17, 2020 and those diagnosed March 17—May 17, 2019 using the KPNC Breast Cancer Tracking System (BCTS) database, a quality assurance program that identifies incident cancers within one month of diagnosis [15]. Patients were excluded if they had a previous history of breast cancer or had been diagnosed or treated outside of KPNC. We also excluded patients who had met with a breast surgeon prior to the breast cancer diagnosis for breast symptoms such as a breast mass, or those who were referred for excisional biopsy after an image-guided core needle biopsy revealed a high-risk lesion. Finally, we excluded those who had an established medical oncologist managing another malignancy prior to the breast cancer diagnosis.
Table 1.
Study Design
| Study design | Retrospective cohort |
|---|---|
| Participants | Patients newly diagnosed with breast cancer March 17-May 17, 2019 and March 17-May 17, 2020 |
| Data sources |
• Institutional breast cancer tracking database • Electronic health records |
| Data collection |
• Database queries • Chart review |
| Outcomes |
• Patient characteristics • Tumor characteristics • Consultation type, timing • Treatment type, timing |
Retrospective cohort study comparing patients newly diagnosed with breast cancer March 17, 2019 – May 17, 2019 to patients diagnosed March 17, 2020 – May 17, 2020
To determine patient characteristics, initial consultation type, and first treatment initiated within 5 months of diagnosis, we extracted data from the KPNC HealthConnect™ electronic health record (EHR) databases (Epic, Verona, WI, USA) and performed chart review. Data were tabulated in Excel spreadsheets. Patient characteristics included age at diagnosis, race, sex, body mass index (BMI), Charlson Comorbidity Index, presence or absence of breast-cancer-related genetic mutations, and whether patients were diagnosed with breast cancer after screening mammography or after diagnostic imaging for breast symptoms (i.e., palpable breast lumps, nipple discharge, or axillary adenopathy). Tumor characteristics included histology, stage, grade, and receptor status.
Initial breast cancer consultation visits were categorized as office, telephone, or video visits for each specialty. Initial treatment categories included surgery, neoadjuvant chemotherapy (NAC), neoadjuvant endocrine therapy (NET), palliative endocrine therapy, and no treatment. We classified breast surgery as partial mastectomy or mastectomy, and nodal surgery as sentinel lymph node biopsy or axillary lymph node dissection.
Time to surgical oncology consultation was defined as the time between the biopsy date and the date of the initial surgical visit. For NAC and NET patients, time to medical oncology consultation was defined as the interval between the biopsy date and the date of the initial medical oncology visit. The time to medical oncology consultation for patients with surgery as initial treatment was defined as the time between the surgery date and the first medical oncology visit.
Time to first treatment was defined as time between the biopsy date and the date of surgery for patients who had surgery first (time to surgery, TTS), and the time between the biopsy date and the date of the first chemotherapy infusion (time to neoadjuvant chemotherapy, TTN) for patients who underwent NAC. Time to adjuvant chemotherapy was defined as time from the surgery date to the first chemotherapy infusion.
Statistical analysis
Descriptive statistics were used to examine the distributions of the demographics, clinical characteristics, and outcome variables using the mean and standard deviation (SD) or median and interquartile range (IQR) for continuous variables and frequencies and percentages for categorical variables. Comparison of demographics, types of treatment, and outcomes parameters between the 2019 and 2020 cohorts were analyzed using the chi-square or Fisher’s exact test for categorical variables and Wilcoxon rank-sum tests for continuous variables. Hypothesis tests were two-sided and a p-value of < 0.05 was used for statistical significance. All statistical analyses were performed using IBM SPSS® Statistics (version 27.0) and SAS® SAS Institute (version 9.4).
Results
Operational changes during the initial SiP period (March 17 – May 17, 2020)
Breast Imaging: KPNC’s comprehensive cancer screening and tracking program included sending patients reminders for routine cancer screening via email and paper letters, checking screening status while rooming patients for doctor appointments, and tracking each patient’s completion of screening and results in a database. Before the pandemic, more than 10,000 reminders for screening mammograms were sent each week. With the SiP order on March 17, 2020, all active outreach for screening mammography was stopped, although patients wishing to have screening mammograms were accommodated. Patients referred for symptomatic breast disease and those called back from screening were evaluated with diagnostic imaging and biopsy as indicated.
Breast Cancer Diagnosis: Upon diagnosis of a breast cancer by image-guided breast biopsy, patients were referred to the facility’s breast care coordinator (BCC), who conducted a telephone intake visit to answer initial questions and determine which consultations were most appropriate for each patient. To minimize the risk of exposure to COVID-19 during SiP, BCCs were encouraged to offer telehealth for initial consultations rather than office visits whenever safe and feasible.
Prospective Multidisciplinary Breast Cancer Case Conference: Every case of newly diagnosed breast cancer in KPNC was reviewed in a multidisciplinary conference before initiation of treatment. Prior to the pandemic, most of these conferences were in person, with some attendees calling in. With the SiP order, all case conferences became virtual.
Surgical Oncology Consultation and Treatment: During SiP, initial surgery consultations occurred with a surgical oncologist only or with a multidisciplinary team consisting of a surgical oncologist, a medical oncologist, and at some facilities, a plastic surgeon and/or a radiation oncologist. Telehealth was encouraged to the extent possible for the surgeon-only appointments. Most multidisciplinary teams adopted hybrid office visit/telehealth appointments, with the patient meeting with the surgical oncologist in person, while family members and other clinical team members were present on video. Other multidisciplinary teams continued office visits with all parties present in person.
Although all KPNC elective surgeries were placed on hold due to the SiP order, cancer operations were not considered elective and were encouraged to continue. Surgical oncology directors developed guidelines during SiP about the timing and extent of surgical treatment based on patient factors, tumor characteristics, and the ability of the medical center to continue breast cancer surgeries safely (Table 2). Healthy patients with operable invasive tumors were encouraged to undergo surgery, especially if they had aggressive tumor subtypes and/or nodal disease. Patients with ductal carcinoma in situ (DCIS) were offered the option of waiting. However, final treatment decisions were left to the discretion of individual surgical oncologists and patients.
Table 2.
KPNC Surgical and Medical Oncology Treatment Guidelines March 17, 2020 – May 17, 2020
| Specialty | Category | Patient/Cancer Characteristics | Recommendation |
|---|---|---|---|
| Surgical Oncology | High Priority Case – Delay may be associated with significant impact on patient outcomes |
• Invasive Cancer – particularly if node-positive, triple-negative, HER2 + , locally advanced, recurrence, or lobular • Recent completion of neoadjuvant chemotherapy |
Proceed with surgery |
| Intermediate Priority Case – Delay may be associated with minimal-moderate impact on patient outcomes |
• Age ≥ 65 years, patients with comorbidities, low or intermediate-grade, T1, strongly ER + and Her2- invasive cancers • DCIS • Discordant breast biopsies or core biopsy showing ADH/atypical papilloma/radial scar/pleomorphic LCIS- weighing patient’s risk factors, imaging characteristics, and patient preference • Intraoperative radiotherapy cases |
Consider postponing • Prior to postponing any invasive cancer, consider axillary ultrasound to document normal axillary lymph nodes • Consider proceeding with DCIS surgery if palpable DCIS and/or extensive breast involvement on imaging, high grade, ER-, or any concerns for undiagnosed invasive component • Consider discussing with medical oncology if there is a benefit to starting endocrine therapy |
|
| Low Priority Case – Delay associated with low impact on patient outcome |
• Benign breast lumps (Fibroadenoma, phyllodes, papilloma) • Nipple discharge • Prophylactic mastectomy |
Cancel surgery | |
| Medical Oncology |
General suggestions: •To reduce immunosuppressing patients and hospital visits overall (labs, infusions. etc.): avoid neoadjuvant therapy for patients with operable cancers •For premenopausal patients on monthly Lupron (to facilitate aromatase inhibitor use), would continue monthly (rather than every 3 months) or consider switching to tamoxifen without Lupron •For patients whose coverage requires coming in for GCSF injections, switch to Fulphilia (biosimilar for Neulasta). For those who self-administer, continue with Zarxio at home •Port flushes can be done every 3 months, rather than monthly •Zometa (adjuvant or metastatic) can be delayed 2 + months •Multigated acquisition (MUGA) scans that have been stable while on HER2 antibodies can be delayed several months. Important to do first MUGA after starting HER2 antibodies to ensure ejection fraction has not decreased. MUGAs should be kept on schedule for those with marginal ejection fractions |
||
| Adjuvant | ER + /HER2- |
• Can start adjuvant endocrine therapy after surgery regardless of timing of radiation (i.e., can start prior to start of radiation treatment) • If surgery must be delayed, consider neoadjuvant endocrine therapy for selected patients • Carefully weigh pros/cons of chemotherapy for those for whom you would normally recommend it (e.g., node-positive, borderline or high Oncotype Dx scores) • Consider Oncotype Dx in all patients with 1–3 positive lymph nodes (carefully discussing lack of data in premenopausal patients) |
|
| HER2 + |
• Use HER2 antibodies/chemo after weighing risks/benefit • Consider adjuvant TDM-1 instead of weekly Herceptin/Taxol to reduce visits |
||
| Triple-negative |
• Use chemotherapy after weighing risks/benefits • Can consider Taxol every 2 weeks instead of weekly to reduce visits, but would require GCSF, associated with more neuropathy, and weekly associated with much less immunosuppression |
||
| Metastatic | If asymptomatic, please consider delaying initiation of chemo | ||
| ER + /HER2- | • Consider endocrine therapy alone (not adding palbociclib) to avoid labs, visits, and neutropenia | ||
| HER2 + | • Consider HER2 antibodies alone (+ endocrine therapy if ER +) | ||
| Triple-negative | • Weigh risks/benefit of chemo | ||
HER2: human epidermal growth factor receptor 2; ER: estrogen receptor; ADH: atypical ductal hyperplasia; DCIS: ductal carcinoma in situ; LCIS: lobular carcinoma in situ; MUGA: multigated acquisition; GCSF: granulocyte colony-stimulating factor
Medical Oncology Consultation and Treatment: Neoadjuvant endocrine therapy (NET) was offered to ER + patients for whom the risks of surgery during SIP were felt to outweigh the benefits due to the patients’ medical comorbidities and relatively indolent tumors (Table 2). Patients who met criteria for NAC but had operable tumors were encouraged to undergo surgery first. Adjuvant chemotherapy for HR + /HER2- was considered for those with node-positive, or borderline/high Oncotype Dx scores. The use of Oncotype Dx to guide chemotherapy recommendations for those with 1–3 positive nodes was also considered, even before RxPonder trial results were presented in December 2020 [16]. Finally, starting adjuvant endocrine therapy while awaiting radiation treatment was encouraged if a delay in starting radiation was anticipated.
Endocrine therapy alone was considered for those with metastatic ER + /HER2- disease. For HER2 + metastatic disease, HER2 antibody treatment was considered, and endocrine therapy was recommended for ER + /HER2 + patients. For triple-negative metastatic disease, chemotherapy was initiated for some patients after weighing the risks and benefits.
Intervals for routine port flushing were increased from 1 to 3 months to reduce unnecessary visits, and elective infusions were postponed. However, patients who wished to continue elective infusions were accommodated.
Presentation and diagnosis
A total of 1,681 screening mammograms were performed March 17-May 17, 2020, in contrast to the 180,724 mammograms performed March 17-May 17, 2019. During the 2 month period, only 54 breast cancers were detected by screening in the 2020 cohort, compared to 440 in the 2019 group.
Table 3 shows the demographic and clinical characteristics of the 703 patients in the 2019 cohort and the 247 patients in the 2020 cohort. The cohorts did not differ significantly by race, BMI, comorbidities, genetic mutation status, node positivity, or histologic subtypes. A higher percentage of patients were < 65 years in the 2020 cohort (66% vs 57% in 2019, p = 0.04). While the total number of breast cancer diagnoses was smaller in the 2020 cohort, a higher percentage of patients presented with symptomatic disease in 2020 than in 2019, (78% vs 37%, X2 (1, n = 950) = 121.5, p < 0.001). T-stages at presentation were higher in 2020: 78% of patients presented with T1c or greater tumors in 2020 vs 64% in 2019 (X2 (1, n = 950), = 15.1, p < 0.001). Absolute numbers of patients presenting with metastatic disease were similar in 2020 and 2019 (18 in 2020 and 17 in 2019), although with fewer diagnoses in 2020, these patients comprised a higher percentage of the cohort (7% in 2020 vs 2% in 2019, p = 0.001). Of patients with invasive disease, a higher percentage presented with grade 3 tumors in 2020 (37% in 2020 vs 25% in 2019, p = 0.004), and triple-negative tumors (16% vs 10%, p = 0.04).
Table 3.
Demographic and Clinical Characteristics of Patients Diagnosed with Breast Cancer March 17 – May 17, 2019 vs. March 17 – May 17, 2020
| Characteristic | 2019 (n = 703) | 2020 (n = 247) | |
|---|---|---|---|
| No (%) | No (%) | p-valuea | |
| Age, years | 0.04 | ||
| < 40 | 25 (4) | 14 (6) | |
| 40–64 | 373 (53) | 147 (60) | |
| ≥ 65 | 305 (43) | 86 (35) | |
| Race | 0.37 | ||
| White | 386 (55) | 143 (58) | |
| Asian | 143 (20) | 41 (17) | |
| Hispanic | 100 (14) | 29 (12) | |
| Black | 44 (6) | 19 (8) | |
| Other/Unknown | 30 (4) | 15 (6) | |
| BMI, N (%) | 0.63 | ||
| Obese (BMI ≥ 30) | 268 (38) | 90 (36) | |
| Charlson Comorbidity Index | 0.73 | ||
| Greater than 3 | 88 (12) | 33 (13) | |
| Genetic Mutation | 24 (3) | 15 (6) | 0.07 |
| Detection Method | < 0.001 | ||
| Screening Mammogram | 440 (63) | 54 (22) | |
| Symptomatic | 263 (37) | 193 (78) | |
| Histology | 0.16 | ||
| DCIS | 95 (14) | 21 (9) | |
| IDC | 489 (70) | 184 (75) | |
| ILC | 64 (9) | 26 (11) | |
| Other | 55 (8) | 16 (7) | |
| T Stage | 0.02 | ||
| Tis | 95 (14) | 21 (9) | |
| T1mi | 16 (2) | 6 (2) | |
| T1a | 46 (7) | 7 (3) | |
| T1b | 90 (13) | 19 (8) | |
| T1c | 177 (25) | 66 (27) | |
| T2 | 217 (31) | 98 (40) | |
| T3 | 37 (5) | 18 (7) | |
| T4 | 21 (3) | 10 (4) | |
| Tx/T0 | 4 (0) | 2 (1) | |
| ≥ T1c | 452 (64) | 192 (78) | < 0.001 |
| N Stageb | 0.88 | ||
| N1 | 125 (69) | 60 (68) | |
| N1m | 20 (11) | 8 (9) | |
| N2 | 14 (8) | 9 (10) | |
| N3 | 21 (12) | 11 (13) | |
| M Stage | 0.001 | ||
| M0 | 685 (97) | 227 (92) | |
| M1 | 17 (2) | 18 (7) | |
| Mx | 1 (0) | 2 (1) | |
| Invasive Cancer | 608 (87) | 229 (91) | 0.03 |
| Grade | 0.004 | ||
| 1 | 166 (27) | 44 (20) | |
| 2 | 277 (46) | 95 (42) | |
| 3 | 149 (25) | 83 (37) | |
| Unknown | 16 (3) | 4 (2) | |
| Receptor Status | 0.04 | ||
| HR + /HER2- | 460 (76) | 150 (67) | |
| HR + /HER2 + | 55 (9) | 29 (13) | |
| HR-/HER2 + | 28 (5) | 9 (4) | |
| HR-/HER2- | 59 (10) | 35 (16) | |
| Unknown | 4 (1) | 2 (1) |
BMI: Body Mass Index; DCIS: Ductal Carcinoma In Situ; IDC: Invasive Ductal Carcinoma; ILC: Invasive Lobular Carcinoma; HR: Hormone Receptor; HER2: Herceptin Epidermal Growth Factor Receptor 2
aChi-square or Fisher’s exact test
bTotal n = 268
Initial consultation timing and types
The total number of surgical oncology consultations decreased from 533 in 2019 to 169 in 2020 (Table 4). Medical oncology consultations for neoadjuvant therapy decreased from 94 in 2019 to 63 in 2020, and adjuvant therapy decreased from 182 to 78. Time to initial consultation was shorter in 2020 than 2019 for both surgical oncology consultations (median 7 vs 11 days, p < 0.001) and medical oncology consultations after surgery (median 13 vs 15 days, p = 0.01), but did not differ for neoadjuvant medical oncology consultations (median 12 vs 13.5 days, p = 0.19). Telehealth utilization for all initial consultations was higher in 2020 than 2019 (70.0% vs 0.8%, p < 0.001). For patients diagnosed in 2020, there were no significant differences in proportion of initial telehealth vs office consultation visits for patients BMI < 30 and BMI ≥ 30 (X2 (1, n = 238) = 1.86, 0.17 and X2 (1, n = 241) = 0.33, p = 0.56, respectively). Telehealth comprised 58% of initial surgical oncology consultations in 2020, compared to 0% in 2019. Similarly, a significantly higher percentage of initial medical oncology consultations in 2020 were telehealth than in 2019 (85.1% vs 2.5%, p < 0.001).
Table 4.
Initial New Physician Consultation Types and Timing by Specialty, for Patients Diagnosed with Breast Cancer March 17 – May 17, 2019 vs. March 17 – May 17, 2020
| Type of Visit | Total Visits, No. (%) | p-valuea | Median Days to Consultation (IQR) | p-valuea | ||
|---|---|---|---|---|---|---|
| 2019 | 2020 | 2019 | 2020 | |||
| All Patients | < 0.001 | N/A | ||||
| Office Visits | 822 (99.2%) | 92 (29.6%) | N/A | N/A | ||
| Telehealth Visits | 7 (0.8%) | 219 (70.4%) | N/A | N/A | ||
| Video Visits | 1 (0.1%) | 130 (41.8%) | N/A | N/A | ||
| Telephone Visits | 6 (0.7%) | 88 (28.3) | N/A | N/A | ||
| Surgical Oncology Firstb | < 0.001 | 11 (8–13) | 7(6–11) | < 0.001 | ||
| Office Visits | 553 (100%) | 71 (42.0%) | 11 (8–13) | 9 (7–12) | 0.12 | |
| Telehealth Visits | 0 (0%) | 98 (58.0%) | N/A | 7 (5–9.25) | N/A | |
| Video Visits | 0 (0%) | 57 (33.7%) | N/A | 7 (5.5–10) | N/A | |
| Telephone Visits | 0 (0%) | 41 (24.3%) | N/A | 7 (4–9) | N/A | |
| All Medical Oncologyc | < 0.001 | N/A | ||||
| Office Visits | 269 (97.5%) | 21 (14.9%) | N/A | N/A | ||
| Telehealth Visits | 7 (2.5%) | 120 (85.1%) | N/A | N/A | ||
| Video Visits | 1 (0.4%) | 73 (51.8%) | N/A | N/A | ||
| Telephone Visits | 6 (2.2%) | 47 (33.3%) | N/A | N/A | ||
| Neoadjuvant Medical Oncologyd | < 0.001 | 13.5 (10.5–21) | 12 (7–15) | 0.19 | ||
| Office Visits | 93 (98.9%) | 18 (28.6%) | 14 (11–21) | 9.5 (6.75–14.0) | 0.28 | |
| Telehealth Visits | 1 (1%) | 45 (71.4%) | N/A | 12 (8–17) | N/A | |
| Video Visits | 1 (1%) | 23 (35.9%) | N/A | 10 (7–13) | N/A | |
| Telephone Visits | 0 (0%) | 22 (34.4%) | N/A | 15 (10–20.5) | N/A | |
| Adjuvant Medical Oncologye | < 0.001 | 15 (10–21) | 13 (8–19) | 0.01 | ||
| Office Visits | 176 (96.7%) | 3 (3.8%) | 15 (10.25–21) | 13 (13–16) | 0.95 | |
| Telehealth Visits | 6 (3.3%) | 75 (96.2%) | 12.5 (7.75–21.5) | 13 (8–19) | 0.89 | |
| Video Visits | 0 (0%) | 50 (64.1%) | N/A | 14 (9–21) | N/A | |
| Telephone Visits | 6 (3.3%) | 25 (32.1%) | 12.5 (7.75–21.5) | 11 (7–14) | 0.71 | |
N/A: not applicable
aChi-square or Fisher’s exact test for categorical variables, Wilcoxon rank-sum test for continuous variables
bPatients who had surgery first including those who had adjuvant chemotherapy and excluding those who were previously followed by surgery prior to cancer diagnosis, Total n = 722
cTotal = 417
dPatients who had either chemotherapy or endocrine therapy first, Total n = 157
eTotal n = 261
Time to initial surgical consultation for telehealth appointments in 2020 was significantly shorter than for office visits (median 7 days vs 9 days, p = 0.03) (Table 4). Time to initial medical oncology consultation for patients with NAC/NET was not significantly different between visit types (median 12 days for telehealth vs 9.5 days for office visits, p = 0.60). Similarly, we found no difference in time to medical oncology consultation by visit type for patients with adjuvant chemotherapy (median 13 days for telehealth vs 13 days for office visits, p = 0.82).
Initial treatment types and intervals
Although most patients underwent surgery as their first treatment in 2020, the percentage was smaller (71% in 2020 vs 83% in 2019, p < 0.001), and a larger percentage started NAC than in 2019 (16% in 2020 vs 11% in 2019, p < 0.001) (Table 5). Six percent (n = 14) of the overall 2020 cohort received NET while awaiting surgery. However, this represented 46.7% of NET candidate patients (≥ 60 years with T1, HR + /HER2-, grade 1 or 2 breast cancer; n = 30). A higher proportion of patients underwent mastectomy in 2020 than in 2019 (33% vs 24%, p = 0.02).
Table 5.
First Treatment Type and Treatment Timing for Patients Diagnosed with Breast Cancer March 17 – May 17, 2019 vs. March 17 – May 17, 2020
| 2019 (n = 703) | 2020 (n = 247) | p-valuea | |
|---|---|---|---|
| No. (%) | No. (%) | ||
| First treatment type | < 0.001 | ||
| Surgery | 583 (83) | 176 (71) | |
| Neoadjuvant Chemotherapy | 75 (11) | 40 (16) | |
| Neoadjuvant HT | 3 (0) | 14 (6) | |
| Palliative Endocrine | 20 (3) | 10 (4) | |
| No treatment | 22 (3) | 7 (3) | |
| Breast operationsb | 0.02 | ||
| Partial Mastectomy | 441 (76) | 117 (67) | |
| Mastectomy | 139 (24) | 57 (33) | |
| Lymph node operationsc | 0.69 | ||
| ALND | 123 (23) | 39 (24) | |
| SLNB | 422 (77) | 123 (76) | |
| First treatment timing | |||
| Time to Surgeryd: median days (IQR) | 26 (20 – 36) | 19 (14 – 27) | < 0.001 |
| Time to Neoadjuvant Chemotherapye: median days (IQR) | 28 (21 – 32) | 23 (18 – 28) | 0.03 |
| Time to Adjuvant Chemotherapyf: median days (IQR) | 37 (30 – 48) | 34 (29 – 44) | 0.10 |
HT: Hormone therapy; ALND: Axillary Lymph Node Dissection; SLNB: Sentinel Lymph Node Biopsy; IQR: Interquartile range
aChi-square or Wilcoxon rank-sum tests
bTotal n = 754; cTotal n = 707
dTotal n = 759
eTotal n = 103
fTotal n = 261
Time to surgery (TTS) was significantly shorter for the 2020 group (median 19 days vs 26 days in 2019, p < 0.001) (Table 5). Time to neoadjuvant chemotherapy (TTN) also was shorter for the 2020 group (median 23 days vs 28 days in 2019, p = 0.03). Time to adjuvant chemotherapy did not differ between 2020 and 2019 (34 days vs 37 days, p = 0.09).
Discussion
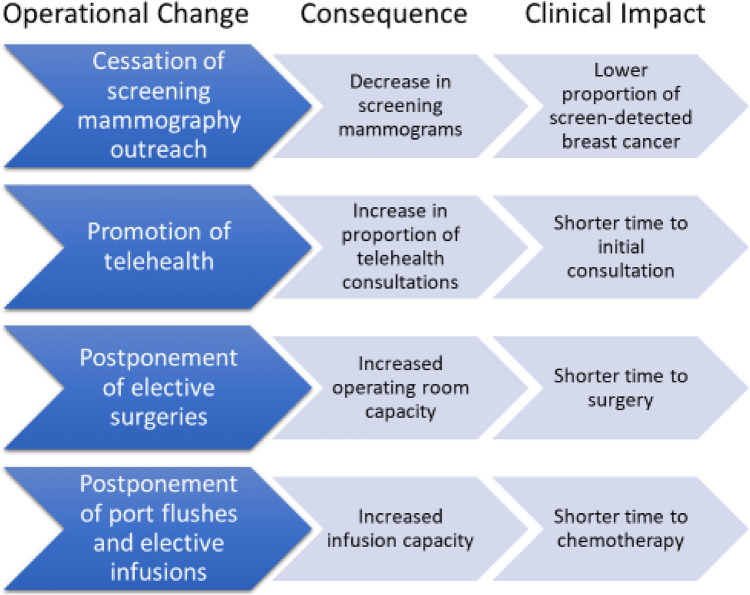
We describe operational changes in multidisciplinary breast cancer management and the diagnostic and treatment impact of these changes within a large, integrated health care system during SiP (Fig. 1). We found that screening mammography rates fell when screening mammography outreach was stopped. As a likely consequence, significantly fewer patients were diagnosed with breast cancer in 2020 than in 2019, and a larger proportion presented with symptomatic disease. The promotion of telehealth during SiP increased telehealth utilization significantly and probably contributed to decreased times to initial consultation. Finally, TTS and TTN were significantly shorter in 2020. We hypothesize this was due to increased operating room and infusion chair availability secondary to postponement of elective procedures and interventions. To our knowledge, our study is the first to describe both the systemwide operational changes and the likely sequelae of these changes on breast cancer management in an integrated health care system.
Fig. 1.
Summary of KPNC Breast Care Operational Changes During the Initial SiP Period (March 17, 2020 – May 17, 2020) and Clinical Impact. Abbreviations: KPNC: Kaiser Permanente Northern California; SiP: Shelter in Place
Others have described operational changes in their management of breast cancer [4–7, 11, 17]. One group promoted the use of neoadjuvant systemic therapies to delay definitive surgery until it had personal protective equipment and resources to resume breast cancer surgery [18]. KPNC breast cancer directors had considered using systemic therapy to temporize the need for surgery, but recommended against NAC in surgical candidates because of the high mortality risk in chemotherapy patients who contracted a COVID infection [19]. The “surgery first” approach was possible at KPNC because our facilities had the capacity to continue cancer operations during the initial SiP period, likely resulting in significantly shorter TTS in the 2020 cohort compared to the 2019 cohort. In contrast, others have reported delays in breast cancer treatment during the pandemic [11, 12, 20, 21]. A multicenter review of 432 patients revealed delays in breast cancer treatment during the initial period of the COVID-19 pandemic compared to pre-pandemic treatment times [12], and in a survey of patients with breast cancer, 44% reported delays in their cancer treatment [20].
The number of new breast cancer diagnoses decreased by 65%, similar to the decreases of 52–67% described by others in association with decreased outreach and screening mammography during SiP [9, 10, 13]. We hypothesize that the higher proportions of symptomatic disease and more aggressive tumors in 2020 than in 2019 reflect the changes in screening mammography outreach protocols rather than disease progression within the two-month initial SiP period. Despite the higher percentage of patients presenting with symptomatic disease, we noted a 27% decrease in the absolute number of symptomatic patients diagnosed, suggesting that many symptomatic patients may not have sought evaluation during SiP. An estimated 41% of U.S. adults reported delaying medical care during the pandemic due to concerns about COVID-19, and up to 12% avoided urgent or emergency care because of these concerns [22]. COVID-19 anxiety and fear in cancer patients has been associated with treatment delays and treatment refusal [12, 23]. Our results highlight the potential for disease progression as patients continue to delay care during the pandemic.
The significant decreases in TTS and TTN were unexpected. With national organizations recommending postponement of surgery for patients with breast cancer [2] and patients’ reluctance to come to the hospital for fear of the risk of COVID infection [24], we expected average TTS to be longer in the pandemic cohort. However, continuation of cancer operations in KPNC was encouraged as long as facilities could perform them safely. We presume that the increased operating room capacity due to postponement of elective operations, combined with the smaller volume of breast cancer patients, contributed to the shorter times to surgery. Telehealth also may have contributed to shorter TTS, allowing shorter times to initial consultation. Similarly, the shorter TTN likely was due to the increased availability of chemotherapy infusion chairs, since routine port flushes and elective infusions were postponed.
Telehealth utilization increased significantly across specialties for initial breast cancer consultations during SiP, with telehealth visits comprising 58% of initial surgical consultations in the 2020 cohort. This is consistent with the increase in telehealth medical oncology consultations reported for all cancer types in TPMG [25]. Others also have described a significant increase in cancer care video visits [26] and telehealth outpatient surgical consultations during SiP [27]. Cancer patients may prefer telehealth in order to minimize the number of medical facility visits and the risk of contracting COVID-19 [24]. Telehealth utilization may decrease as the pandemic is contained, but we anticipate it will become an established part of many practices now that both patients and providers have experienced its convenience and ease.
Our study has several limitations. First, we did not capture clinical outcomes. Due to the relative recentness of the pandemic, the impact of treatment algorithm changes on long-term outcomes such as local recurrence and breast cancer specific mortality may not become evident until years in the future. Comparisons of shorter-term outcomes such as emergency department (ED) visits and readmissions most likely were confounded significantly by pandemic-driven changes in ED workflows, hospital admission criteria, and patient reluctance to seek medical care. With these confounding factors and our historically low rate of complications [28, 29], we felt it was unlikely that we could draw meaningful conclusions regarding such shorter-term outcomes. A second limitation is that comparisons between cohorts from the two time periods were not adjusted for potential confounders. Finally, with the retrospective nature of our study, we did not capture patient-reported outcomes to evaluate patient satisfaction with telehealth, NET, and other care delivery changes driven by the pandemic.
In conclusion, our study describes operational changes and their clinical impact on breast cancer management in KPNC during the initial SiP period. Our study highlights the importance of screening mammography in the early detection of breast cancer, and we recommend that screening mammography be continued whenever safe and feasible to minimize the number of undetected cancers. We also demonstrate the efficacy of telehealth in cancer care and its value when in-person visits are not safe or feasible. Finally, we describe the capacity of an integrated health system to continue providing timely breast cancer treatment despite the constraints of the pandemic. Rapid implementation of coordinated workflow changes enabled continuation of breast cancer care even in the face of great uncertainty and resource limitations.
Looking forward, we are concerned about the prognosis of patients in our system with undiagnosed cancer. Although screening mammography outreach has resumed, as of December 8, 2020 screening mammography rates still were less than 50% of pre-pandemic levels because patients declined screening mammography. Given the 65% decrease in overall breast cancer diagnoses but the stable number of stage IV diagnoses, we suspect that the majority of undiagnosed patients have stage 1–3 disease, and we are concerned that stage progression could occur due to pandemic-related delays in diagnosis. Further studies on the long-term consequences of delay in breast cancer screening and diagnosis are warranted. We anticipate that with COVID vaccinations and better control of the pandemic, more patients will proceed with screening. In the meantime, breast care programs must prepare for a likely surge of new breast cancer patients in the near future.
Acknowledgements
The authors would like to thank Pamela Derish in the UCSF Department of Surgery for assistance in editing this manuscript and Jane Bethard-Tracy from the KPNC Breast Cancer Tracking Service for assistance with data collection.
Funding
Funding for this study was provided by Kaiser Permanente Northern California Graduate Medical Education, Kaiser Foundation Hospitals. Kaiser permanente
Data availability
The datasets generated during and/or analyzed during the current study are not publicly available due to patient privacy laws, but are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
Declarations
Conflict of interest
The authors report no conflicts of interest in any product mentioned or concept discussed in this article. This study has not been presented or published. An oral presentation with parts of this study was given at the 2020 San Antonio Breast Cancer Symposium on December 8, 2020. The authors have not published, posted, or submitted any related papers from the same study for publication.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Dietz JR, Moran MS, Isakoff SJ, et al. Recommendations for prioritization, treatment, and triage of breast cancer patients during the COVID-19 pandemic. The COVID-19 pandemic breast cancer consortium. Breast Cancer Res Treat. 2020;181:487–497. doi: 10.1007/s10549-020-05644-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bartlett DL, Howe JR, Chang G, et al. Management of cancer surgery cases during the COVID-19 pandemic: considerations. Ann Surg Oncol. 2020;27:1717–1720. doi: 10.1245/s10434-020-08461-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.COVIDSurg Collaborative, Global guidance for surgical care during the COVID-19 pandemic. Br J Surg. 2020;107:1097–1103. doi: 10.1002/bjs.11646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Al-Shamsi HO, Alhazzani W, Alhuraiji A, et al. A practical approach to the management of cancer patients during the novel coronavirus disease 2019 (COVID-19) pandemic: an international collaborative group. Oncologist. 2020;25:e936–e945. doi: 10.1634/theoncologist.2020-0213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Soran A, Gimbel M, Diego E. Breast cancer diagnosis, treatment and follow-up during COVID-19 pandemic. Eur J Breast Health. 2020;16:86–88. doi: 10.5152/ejbh.2020.240320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chan JJ, Sim Y, Ow SGW, et al. The impact of COVID-19 on and recommendations for breast cancer care: the Singapore experience. Endocr Relat Cancer. 2020;27:R307–R327. doi: 10.1530/ERC-20-0157. [DOI] [PubMed] [Google Scholar]
- 7.Ribeiro R, Wainstein AJA, de Castro Ribeiro HS, et al. Perioperative cancer care in the context of limited resources during the COVID-19 pandemic: Brazilian society of surgical oncology recommendations. Ann Surg Oncol. 2021;28:1289–1297. doi: 10.1245/s10434-020-09098-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zarzaur BL, Stahl CC, Greenberg JA, et al. Blueprint for restructuring a department of surgery in concert with the health care system during a pandemic: the university of Wisconsin experience. JAMA Surg. 2020;155:628. doi: 10.1001/jamasurg.2020.1386. [DOI] [PubMed] [Google Scholar]
- 9.Kaufman HW, Chen Z, Niles J, Fesko Y. Changes in the number of us patients with newly identified cancer before and during the coronavirus disease 2019 (COVID-19) pandemic. JAMA Netw Open. 2020;3:e2017267. doi: 10.1001/jamanetworkopen.2020.17267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nyante SJ, Benefield TS, Kuzmiak CM, Population-level impact of coronavirus disease,, et al. On breast cancer screening and diagnostic procedures. Cancer n/a: 2019 doi: 10.1002/cncr.33460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hawrot K, Shulman LN, Bleiweiss IJ, et al (2021) Time to Treatment Initiation for Breast Cancer During the 2020 COVID-19 Pandemic. JCO Oncology Practice OP.20.00807. 10.1200/OP.20.00807 [DOI] [PMC free article] [PubMed]
- 12.Vanni G, Tazzioli G, Pellicciaro M, et al (2020) Delay in Breast Cancer Treatments During the First COVID-19 Lockdown A Multicentric Analysis of 432 Patients. Anticancer Res 40:7119–7125 Doi: 10.21873/anticanres.14741 [DOI] [PubMed]
- 13.Li J, Wang H, Geng C, et al. Suboptimal declines and delays in early breast cancer treatment after COVID-19 quarantine restrictions in China: a national survey of 8397 patients in the first quarter of 2020. EClinicalMedicine. 2020;26:100503. doi: 10.1016/j.eclinm.2020.100503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mallick I, Chakraborty S, Baral S, et al. Prioritizing delivery of cancer treatment during a COVID-19 lockdown: the experience of a clinical oncology service in India. JCO Glob Oncol. 2021;7:99–107. doi: 10.1200/GO.20.00433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Callahan M, Sanderson J. A breast cancer tracking system. Perm J. 2000;4:36–39. [Google Scholar]
- 16.Kalinsky K, Barlow WE, Meric-Bernstam F, et al (2021) Abstract GS3–00: First results from a phase III randomized clinical trial of standard adjuvant endocrine therapy (ET) +/- chemotherapy (CT) in patients (pts) with 1–3 positive nodes, hormone receptor-positive (HR+) and HER2-negative (HER2-) breast cancer (BC) with recurrence score (RS) < 25: SWOG S1007 (RxPonder). Cancer Res 81:GS3-GS3–00. 10.1158/1538-7445.SABCS20-GS3-00
- 17.Satish T, Raghunathan R, Prigoff JG, et al (2021) Care Delivery Impact of the COVID-19 Pandemic on Breast Cancer Care. JCO Oncology Practice OP.20.01062. 10.1200/OP.20.01062 [DOI] [PubMed]
- 18.Sheng JY, Santa-Maria CA, Mangini N, et al. Management of breast cancer during the COVID-19 pandemic: a stage- and subtype-specific approach. JCO Oncol Pract. 2020;16:665–674. doi: 10.1200/OP.20.00364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Liang W, Guan W, Chen R, et al. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol. 2020;21:335–337. doi: 10.1016/S1470-2045(20)30096-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Papautsky EL, Hamlish T. Patient-reported treatment delays in breast cancer care during the COVID-19 pandemic. Breast Cancer Res Treat. 2020;184:249–254. doi: 10.1007/s10549-020-05828-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wu J, Bobo S, Henry S, et al (2021) Abstract PS6–32: Impact of COVID-19 on breast cancer care at a Bay Area academic center. Cancer Res 81:PS6-PS6–32. 10.1158/1538-7445.SABCS20-PS6-32
- 22.Czeisler MÉ, Marynak K, Clarke KEN, et al (2020) Delay or Avoidance of Medical Care Because of COVID-19-Related Concerns - United States, June 2020. MMWR Morb Mortal Wkly Rep 69:1250–1257. 10.15585/mmwr.mm6936a4 [DOI] [PMC free article] [PubMed]
- 23.Guven DC, Sahin TK, Aktepe OH, et al. Perspectives, Knowledge, and fears of cancer patients about COVID-19. Front Oncol. 2020;10:1553. doi: 10.3389/fonc.2020.01553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Vanni G, Materazzo M, Pellicciaro M, et al (2020) Breast Cancer and COVID-19: The Effect of Fear on Patients’ Decision-making Process. In Vivo 34:1651–1659. 10.21873/invivo.11957 [DOI] [PMC free article] [PubMed]
- 25.Liu R, Sundaresan T, Reed ME, et al. Telehealth in oncology during the COVID-19 outbreak: bringing the house call back virtually. JCO Oncology Practice. 2020;16:289–293. doi: 10.1200/OP.20.00199. [DOI] [PubMed] [Google Scholar]
- 26.Lonergan PE, Washington Iii SL, Branagan L, et al. Rapid Utilization of telehealth in a comprehensive cancer center as a response to COVID-19: cross-sectional analysis. J Med Internet Res. 2020;22:e19322. doi: 10.2196/19322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lazar AN, Nemeth SK, Kurlansky PA, et al. Adoption and usage of video telehealth in a large, academic department of surgery. Annals of Surgery Open. 2021;2:e040. doi: 10.1097/AS9.0000000000000040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Vuong B, Dusendang JR, Chang SB, et al. Outpatient mastectomy: factors influencing patient selection and predictors of return to care. J Am Coll Surg. 2021;232:35–44. doi: 10.1016/j.jamcollsurg.2020.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Patel AR, Vuong B, Kuehner GE, et al. Adoption of opioid-sparing and non-opioid regimens after breast surgery in a large, integrated health care delivery system. Ann Surg Oncol. 2020;27:4835–4843. doi: 10.1245/s10434-020-08897-6. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are not publicly available due to patient privacy laws, but are available from the corresponding author upon reasonable request.
Not applicable.