Abstract
King Cobra (Ophiophagus hannah) has a significant place in many cultures, and is a medically important venomous snake in the world. Envenomation by this snake is highly lethal, manifested mainly by neurotoxicity and local tissue damage. King Cobra may be part of a larger species complex, and is widely distributed across Southeast Asia, southern China, northern and eastern regions as well as the Western Ghats of India, indicating potential geographical variation in venom composition. There is, however, only one species-specific King Cobra antivenom available worldwide that is produced in Thailand, using venom from the snake of Thai origin. Issues relating to the management of King Cobra envenomation (e.g., variation in the composition and toxicity of the venom, limited availability and efficacy of antivenom), and challenges faced in the research of venom (in particular proteomics), are rarely addressed. This article reviews the natural history and sociocultural importance of King Cobra, cases of snakebite envenomation caused by this species, current practice of management (preclinical and clinical), and major toxinological studies of the venom with a focus on venom proteomics, toxicity and neutralization. Unfortunately, epidemiological data of King Cobra bite is scarce, and venom proteomes reported in various studies revealed marked discrepancies in details. Challenges, such as inconsistency in snake venom sampling, varying methodology of proteomic analysis, lack of mechanistic and antivenomic studies, and controversy surrounding antivenom use in treating King Cobra envenomation are herein discussed. Future directions are proposed, including the effort to establish a standard, comprehensive Pan-Asian proteomic database of King Cobra venom, from which the venom variation can be determined. Research should be undertaken to characterize the toxin antigenicity, and to develop an antivenom with improved efficacy and wider geographical utility. The endeavors are aligned with the WHO´s roadmap that aims to reduce the disease burden of snakebite by 50% before 2030.
Keywords: Proteomics, Venomics, Antivenom, Neutralization, Neglected tropical disease
Introduction
Snakebite envenomation is a medical emergency characterized by the toxic manifestation of snake venom. It is a major public health problem, a common environmental and occupational hazard affecting many impoverished and geopolitically marginalized populations in the tropics and subtropics [1]. Close to 5 million snakebites occur yearly, resulting in 1.8-2.7 million cases of envenomation. Approximately 81,000-138,000 envenomed victims die each year, while about three times as many survivors continue to suffer various permanent physical disabilities and psychological distress [2,3]. The highest incidence and mortality rates are recorded in South Asia, Southeast Asia and Sub-Saharan Africa, reflecting a combination of factors associated with the developing status, high population density, and venomous snake abundance in the regions [4-6]. The exact extent of the disease burden of snakebite is largely underestimated due to scarce epidemiological data. Climate changes, increased trading of venomous snakes, and the adaptation of venomous species to anthropogenic-modified environments, have also indirectly increased the frequency of contact and conflict between humans and snakes [7,8].
In the past, snakebite envenomation received little attention globally despite its dramatic socio-economic impact on the poor [5,9,10]. It was until 2017 that the World Health Organization (WHO) formally reinstated snakebite envenomation as a priority neglected tropical Disease [11]. Several strategies have since been put forth, with the aim to halve the disease burden of snakebite envenomation by 2030. In this context, South Asia and Southeast Asia are homes to abundant venomous snakes, and are indeed hotspots of snakebite envenomation for solution. Each medically important species has its unique venom composition, and the diversity of venom modulates the pharmacological activities, resulting in varying toxic manifestations and effectiveness of antivenom treatment [12]. Moreover, intra-species variation in snake venom is widely recognized, adding complexity to the management of snakebite as antivenom products are usually manufactured using snake venom sourced from certain locale without considering the potential in-space (geographical) and in-time (ontogenic) variability of venom composition, as well as composition variation possibly caused by sexual dimorphism of the snake [13,14]. This remains true for the case of King Cobra (Ophiophagus hannah), a medically important elapid that has a wide biogeographical distribution in Asia. This article reviews the current status of King Cobra from the biological and medical perspectives, highlighting the impact of King Cobra bite on the community and the need for proper management. Recent advances, controversies and challenges in the proteomics, toxicity and neutralization studies of King Cobra venom are discussed.
Distribution, taxonomy and behavior
King Cobra is the world’s largest venomous snake, with adults capable of growing up to 6 meters in length. It widely distributes throughout Southeast Asia on the Malay Archipelago (to the exceptions of territories located eastward from the Indonesian islands of Java and Sulawesi), in southern China (including the insular Hainan Province and Hong Kong SAR), and in parts of India (in the northern and eastern parts of India, and disjunctly, in southwestern India along the Western Ghats of Tamil Nadu) [15] (Figure 1A). Depending on the geographical origin, habitat and stage of growth, King Cobras can exist in an array of different colors, including glossy black, pale olive, gray, brownish-green or yellowish golden, with or without white crossbars or chevrons on the body (Figure 1B-1E). King Cobras usually inhabit tropical forests although specimens were also sighted in a wide range of habitats, including mangrove forest, highlands, wetlands, scrublands and anthropogenically modified environments such as agricultural fields [15-17].
Figure 1. (A) Natural distribution of King Cobra in Southeast Asia, southern China, north and north-eastern India, and the Western Ghat of India. (B) Adult King Cobra, Malaysia, with pale olive coloration (photo by Chia How Lee, reprinted with permission). (C) Adult King Cobra, likely Western Ghats of India, in glossy black color with cross-bars (photo by Michael Allen Smith from Seattle, USA, CC BY-SA 2.0, via Wikimedia Commons). (D) Adult King Cobra, Odisha State, eastern India, with brownish bronze coloration and crossbars (photo by Mark O'Shea, https://www.markoshea.info/oba3-1_india01.php, reprinted with permission). (E) Juvenile King Cobra (Malaysia) in growth transition, with crossbars which will disappear into adulthood (photo by Chia How Lee, reprinted with permission). (F) Schematic representation of the phylogenetics of King Cobra in relation to extant elapids (tree simplified from Figueroa et al. [19]).
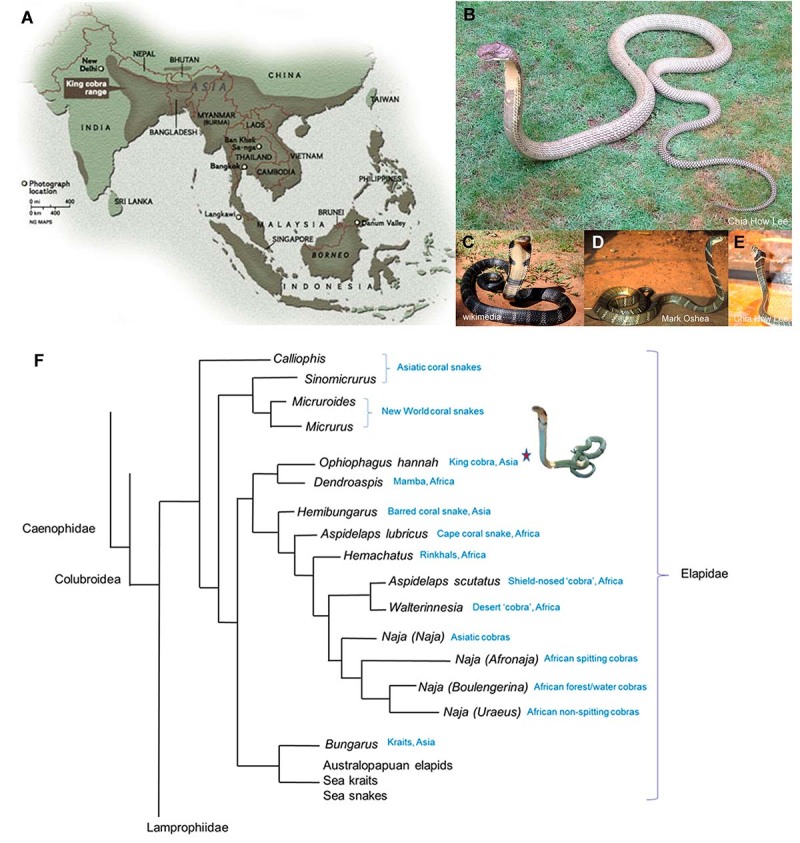
The taxonomy of King Cobra was first described as Hamadryas hannah (Cantor) in 1836. The genus Ophiophagus was proposed by Günther in 1864, aptly attributing the name to its propensity to eat snakes (Ophio-: snake, -phagous: eating, according to the Greek etymology). Ophiophagus hannah was formally accepted as the valid name for King Cobra in 1945 [18]. Although having a “cobra” in its common name and displaying hooding feature, the King Cobra is not a “true cobra” as it differs on several anatomical characters from those of the Naja genus; instead, it was more closely related to mambas (Dendroaspis spp.), based on more recent genetic analysis of its phylogeny [19] (Figure 1F). It is currently the monotypic species of the Ophiophagus genus, although King Cobras from various regions may be part of a larger species complex. For instance, mitochondrial DNA analysis on King Cobras in Thailand suggested a deep phylogenetic divergence between the northern and southern specimens [20]. In fact, besides variation in morphology and coloration within the species of King Cobra, the venom properties in terms of composition and biological activities tend to vary greatly across specimens from different geographic locations [21,22]. However, polymorphism and venom variation are also known to occur without phylogenetic implication, and serve no indication of speciation [23]. Hence, further phylogenomic study based on more specimens from different geographical populations of King Cobra is needed to shed light on its true taxonomy.
The King Cobra primarily feeds on other snakes, both venomous and non-venomous, but it probably also feeds on other reptiles and small mammals when food is scarce (Figure 2). A broad range of preys has been documented including rat snakes, pythons, cobras, kraits, vipers and other King Cobras - A cannibalistic trait is present in this species. Though a snake-eating species, King Cobra is not fully immune to snake venoms, as it can be envenomed and killed by another King Cobra or venomous snakes. Almost exclusively diurnal, the King Cobra is an active predator capable of traveling a long distance, tracking its prey over a prolonged duration [24]. This day-traveler also contributes to the low incidence of envenomation in humans as accidental encounter is less frequent compared with the Naja species (cobras) which are relatively more aggressive, and ambushing vipers/pit vipers which are resting motionless and often camouflaged. Additionally, despite its fearless and aggressive reputation, field scientists often described the King Cobra as a placid snake, only manifesting aggressive behavior when disturbed or cornered, while preferring visual and acoustic threats display over attack. Typically, as a sign of warning, it would raise its upper body above the ground, spreading its hood while emitting a loud growling hiss [24,25].
Figure 2. Feeding behavior of King Cobra. Ophiophagy is observed in (A, B) adult and (C) juvenile King Cobras, feeding on a corn snake, Gerald’s water snake, and bridle snake, respectively (A, photo by viperskin, CC BY-NC-SA 2.0, via Flickr; B, photo by Lip Kee, CC BY-SA 2.0, via Flickr; C, photo by Chia How Lee, reprinted with permission). These were smooth-scaled preys that King Cobras prefer over snakes with keeled scales. (D) Opportunistic feeding on rodents, in captive environments (photo by Chia How Lee, reprinted with permission).

Sociocultural impact and interaction with humans
The King Cobra holds cultural importance in many Asian populations. It is depicted in the Hindu mythology along with cobras (Naja genus), and are considered as the descendant of Nagas, serpent/snake deities. In the 1940 documentary film ‘Wheels across India’, Armand Denis recorded a religious ceremony in Burma (Myanmar), where a priestess was kissing a King Cobra on its head several times and making an offering to the snake as the God of Fertility (Figure 3A). Snake performances are still held in many parts of Asia to this day for both educational and touristic purposes, such as that conducted in the Snake Farm of the Queen Saovabha Memorial Institute, Bangkok (Figure 3B). In Ban Khok Sa-nga (Thailand), also known as the King Cobra Village, residents commonly own King Cobra and snake performances are held daily (Figure 3C). In parts of Asia, snakes including King Cobra are hunted, farmed and traded for food, leather and medicinal uses which are believed to offer health and lifestyle benefits, from fatigue relief to boosting virility (Figure 3D-3G).
Figure 3. Relationship between King Cobra and humans. (A) Footage from ‘Wheels across India’ (Armand Denis, Leila Roosevelt; 1940) of a religious ceremony held in Burma (Myanmar) involving a priestess attempting to kiss a King Cobra on its head three times to obtain the favor of the Snake God of Fertility. (B) Snake educational show at the snake farm of Queen Saovabha Memorial Institute, Bangkok (screenshot from a YouTube clip: www.youtube.com/watch?v=fWh-XOKJgxM). (C) Snake show as a touristic attraction: performer at the Ban Khok Sa-nga King Cobra Village (Thailand) attempting to hold the King Cobra’s head in his mouth (photo by Andrea Pistolesi, https://andreapistolesi.wordpress.com/2010/02/23/who-is-the-snake-who-is-the-victim/). (D) Bags of King Cobras in a wildlife trade market, China. (E) King Cobra medicinal liquor (photo by Viethavvh, CC BY-SA 3.0, via Wikimedia Commons). (F) Village cooking of King Cobra, a source of animal protein in rural areas (screenshot from a YouTube clip: www.youtube.com/watch?v=2Ut0KJv16hg). (G) Menu with “snake thick soup” placed against century-old wooden drawers marked with “venomous snake” (where live snakes were kept) in a restaurant, Hong Kong (©zolimacitymag.com, all rights reserved). Such exotic foods are considered a delicacy and traditional health tonic.

The King Cobra is currently listed in CITES Appendix II, and globally “Vulnerable” by the International Union for Conservation of Nature IUCN, notwithstanding considerable variation of local assessment in different countries. Further taxonomic revision will likely re-define King Cobra into a species complex, and inevitably aggravate the status of each newly erected species.
King Cobra is also a favorite subject of many snake enthusiasts, and “internet celebrities” for posting their interaction with the deadly snake on social media. This amateur practice is gaining popularity in recent years but it is undoubtedly dangerous, as the interaction typically involves bare-hand handling of the snake and exaggerated actions such as petting and kissing the snake (Figure 4). Unnecessary handling and accidental provocation of the snake would lead to its defensive bite and envenomation. Although these people could be well-trained snake handlers, the practice of unnecessary posing (barehand handling, petting, kissing) with the venomous snakes in social media is controversial, as non-trained persons might be tempted to imitate the action. Climate change (global warming, flash floods) and other anthropogenic factors that result in the clearing of wildlife habitat also contribute to the increasing incidence of snake intrusion into human settlement, resulting in human-snake conflicts. These will emerge as the leading causes of King Cobra envenomation in the future, considering that awareness practice and protective equipment may reduce the risk of snakebite among the rural and agricultural populations.
Figure 4. (A-D) Posting on social media showing close contact with the King Cobra. Sources: A-B, “whispering” (from www.facebook.com/wira.penyelamat.3); C, kissing (screenshot from a YouTube clip, www.youtube.com/watch?v=qN5x9raeaMI); D, petting and stroking (screenshot from a YouTube clip, www.youtube.com/watch?v=sX6DS98BhUk). (E) A snake catcher demonstrating the classical pose of “holding up the King Cobra” barehandedly (screenshot from a YouTube clip https://www.youtube.com/watch?v=0ypL-l2laPQ). (F) Snake intrusion alert on a hot day: a King Cobra was spotted slithering into a house in Malaysia (screenshot from a YouTube clip: www.youtube.com/watch?v=F6L_YaWxIA4).

Medical importance
King Cobra is classified as a venomous snake of secondary medical importance (WHO Category 2). By definition, venomous snakes listed in Category 2 are highly venomous species capable of causing morbidity, disability or death, for which exact epidemiological or clinical data may be lacking; and/or are less frequently implicated (due to their activity cycles, behavior, habitat preferences or occurrence in areas remote to large human populations) [1]. The majority of envenomation cases resulted from mishandling, and professions working in close contact with venomous snakes (e.g., venom extraction worker, reptile caretaker, snake rescuer) are thus exposed to a higher risk of envenomation [26,27]. Clinical cases of King Cobra envenomation reported in scientific literature consisted of bites occurring in snake handling environment [26-31]. The notorious popularity of this species and its consequent usage in recreational performances (e.g., snake charming, snake dance) also led to documented envenomation cases [31]. News media also commonly reported casualties arising from such close contacts [32-34].
Clinical cases of King Cobra envenomation
The global epidemiological data of snakebite is scarce. For envenomation caused by King Cobra, there were only nine clinical reports available in the literature published between 1971 and 2020. Of these, three cases were reported from Myanmar, one each from Thailand and Malaysia, and four occurred outside Asia: two in the USA, one in the UK and one in the Netherlands. King Cobra envenomation in humans typically manifests through local tissue damage and systemic neurotoxicity, with a constellation of signs and symptoms similar to that caused by cobras (Naja spp.) in Asia. Envenomed patients commonly develop intense local reactions characterized by pain and swelling resulting in tissue necrosis, and descending paralysis that leads to respiratory failure - the principal mode of fatality in envenomation by King Cobra [35].
Table 1 summarizes the reported cases of King Cobra envenomation in humans, with details of the clinical manifestation and the use of antivenom (if any) for treatment. All cases exhibited local envenomation effects at varying degrees of severity, most were presented with noticeable signs of aggravating inflammation (pain and intensive swelling around the bite site, which could be extending to the entire bitten limb and adjacent body structures) [31,36]. In severe envenomation, tissue necrosis developed requiring surgical interventions and amputation [37]. Non-specific systemic effects were also reported (e.g., dizziness, vertigo, nausea), accompanied with signs of neuromuscular paralysis such as ptosis, limb weakness, dysphagia, dysarthria and flaccid paralysis. Paralysis of respiratory muscle results in asphyxia and generalized hypoxia, with death ensues from multiorgan failure [26,27,31,36,38].
Table 1. Clinical cases reported on Ophiophagus hannah envenomation and treatment.
| Country | Cases | Local effect | Systemic effect | Intervention | Reference |
|---|---|---|---|---|---|
| Myanmar | 3 | Pain, extensive swelling, necrosis | Dizziness, ptosis, ophthalmoplegia, respiratory failure, flaccid paralysis | Artificial respiration; antivenom: OhMAV (20 vials), surgical debridement | Tin et al. [31] |
| UK | 1 | Swelling, ischemia | Dizziness, ptosis, dysphagia, hypertension, bradycardia | Artificial respiration; antivenom: OhMAV (20 vials) | Veto et al. [26] |
| USA | 1 | Pain, swelling | Visual hallucination, lethargy, headache, myalgias, periodic syncope | Antivenom: OhMAV (50 vials) | Wetzel and Christy [27] |
| Malaysia | 1 | Pain, extensive swelling | Dizziness, nausea, vomiting, blurred vision, dysphagia, dysarthria, flaccid paralysis, tachycardia | Artificial respiration; antivenom: OhMAV (33 vials) | Ismail et al. [36] |
| Thailand | 1 | Pain, extensive swelling, infection | Drowsiness, ptosis, dysphagia, dysarthria, syncope, hypotension, respiratory failure | Artificial respiration; antivenom: OhMAV (115 vials) | Ganthavorn [28] |
| USA | 1 | Pain, swelling | Confusion, ptosis, dysphagia, dysarthria, flaccid paralysis | Artificial respiration; antivenom: OhMAV (15 vials) | Gold and Plye [29] |
| The Netherlands | 1 | Blistering, cellulitis, digital ischemia and skin necrosis | None | Wound debridement, digital amputation | Imran et al. [37] |
OhMAV: Thai Ophiophagus hannah Monovalent Antivenom.
In all cases reported, the diagnosis was made clinically through history taking or identification of the biting snake. The treatment consisted of local effect management through specialized wound care and control of secondary infection, and treatment of systemic neurotoxicity with intubation and assisted ventilation [26,28,29,31,36]. The antivenom specific to King Cobra, i.e., Ophiophagus hannah Monovalent Antivenom (OhMAV) produced in Thailand was administered in eight out of the nine cases, indicated by the onset of neurological manifestation. The antivenom treatment was somewhat effective in reversing the systemic toxicity at high dosage ranging from 20 to 115 vials, indicating limited neutralization potency of the antivenom.
Management and potential challenges
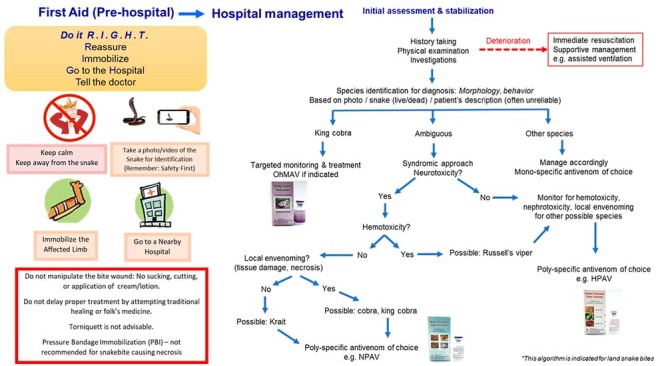
In general, when a bite occurs, the first thing the injured should do is to move beyond the striking distance of the snake (to prevent repeated bite). The management begins with first aid as part of pre-hospital care, while the patient is transferred to the nearest medical facility that manages snakebite as soon as possible. Figure 5 illustrates the basic first aid of snakebite the public should be aware of, and a simplified algorithm for clinical management of snakebite envenomation (with a focus on King Cobra bite). Principles and issues pertaining to the management are described below.
Figure 5. First aid of snakebite and a simplified algorithm for clinical management of snakebite (with a focus on King Cobra) envenomation. OhMAV: Ophiophagus hannah Monovalent Antivenom; NPAV: Neuro Polyvalent Antivenom; HPAV: Hemato Polyvalent Antivneom.
Pre-hospital first aid
The use of a tight (arterial) tourniquet, as widely practiced by many folks using a belt, shoelace, clothing or even fence wire, severs the blood and lymphatic circulation distally and thus potentially prevent or reduce the systemic spread of venom from the bitten site. This practice, however, is highly controversial and now discouraged especially in bites that can cause tissue damage, as tourniquets can concentrate the venom at the bitten site, promoting severe necrosis, gangrene, and resulting in the need to amputate, besides the dangers of “sudden release” (bolus of venom being released systemically leading to overwhelming toxic activity when the tourniquet is removed abruptly). Pressure bandage immobilization (PBI), much advocated by Australian practitioners, is an alternative that effectively reduces lymphatic drainage (the main route of venom spread) without compromising blood circulation. However, PBI too has a limited role in cytotoxic and hemotoxic bite, again, for the risk of worsened local tissue damage as seen in bites caused by most venomous Asiatic snakes, including the King Cobra. Thus, light bandaging along with immobilization of the bitten limb has been a more common practice here while the victim is being transferred at the soonest for medical care.
Species identification
The identification of species is crucial for the selection of an appropriate antivenom, which is usually species-specific. The identification of snake species is, however, clinically challenging. Diagnosis based on the victim’s description is usually less reliable, while captured specimen of the biting snake may not be always available - in fact, it is advisable not to capture the snake to avoid secondary bite. Increasingly, people have the possibility of getting a photo or video of the offending snake in a bite by using the cell phone. This should be recommended, followed by the consultation with an expert who is able to identify the snake.
Alternatively, the syndrome-based approach is useful for diagnosis when the description of snake is unclear, or the offending snake photo or specimen is unavailable. This however can be equally challenging in areas where several sympatric species that produce similar envenoming effects. In Asia, envenomation by the majority of cobra and krait species (land snakes) produces similar neuromuscular paralytic effect, as with the King Cobra. The kraits, nonetheless, do not cause a significant local envenoming effect, and thus it can be relatively easy to rule out krait envenoming based on clinical observation. Between cobras (Naja spp.) and King Cobra, the envenoming effects are rather similar, yet requiring totally different specific antivenom for effective treatment. From the practical standpoint, King Cobra envenomation typically occurs in captive environments and rarely in the wild, therefore species identification has not been much of a problem in King Cobra envenomation, and the mono-specific King Cobra antivenom can be opted as the first-line treatment accordingly. In a worse scenario where the neurotoxic species cannot be identified, the patient will have to rely on the availability of a polyvalent antivenom that is able to cover the different neurotoxic species in the region (Neuro Polyvalent Antivenom, product of Queen Saovabha Memorial Institute, Bangkok). A brief guide on the syndromic identification of King Cobra bite and antivenom recommendation is illustrated in Figure 5.
Antivenom production and efficacy
The Thai Red Cross Society, Queen Saovabha Memorial Institute (Bangkok, Thailand) is the only antivenom producer in the world that manufactures the specific antivenom, Ophiophagus hannah Monovalent Antivenom (OhMAV), raised against the King Cobra venom of Thai origin. A polyvalent antivenom, Neuro Polyvalent Antivenom (NPAV) that can be used to treat the envenomation caused by four medically important elapids in the region (Ophiophagus hannah, Naja kaouthia, Bungarus candidus, Bungarus fasciatus) is also available. These products are commonly used in the region, including Malaysia, as both countries shared similar medically important venomous herpetofauna [39]. In Myanmar, the Philippines, Indonesia and China, local production of antivenom is available to support domestic demand, but none of the antivenoms from these countries covers King Cobra. Anecdotal records showed that non-specific heterologous antivenoms such as antivenoms for cobra (Naja) envenomation were used as an alternative to treat King Cobra envenomation in China and Indonesia, but the treatment outcome has been poor. Although hetero-specific antivenoms may cross-neutralize the venoms of closely related species which share compositional and antigenic similarities [40], King Cobra and Naja cobras have variable venom antigenicity that limits immunorecognition of toxins and cross-neutralization by antivenom immunoglobulins [22]. Moreover, the use of high doses of less effective hetero-specific antivenom could pose a greater risk of hypersensitivity reaction with fatal outcome to the patients [41].
Snake venoms are integrated phenotypes used for predatory, digestive and defensive purposes, and are subject to evolution with adaptation to changes in ecological niches. Intraspecific variation of snake venom composition is a widely recognized phenomenon, and this has a huge impact on the effectiveness of antivenom used. Compositional variation of snake venom within a species from different locales is often accompanied with variable expression and antigenicity of the key toxins, rendering the antivenom from a single production source less potent or even ineffective [42-45]. Although in some instances, the reversal of toxicity can be achieved by increasing the dosage of antivenom administered, but this inevitably exposes the patient to a greater risk of hypersensitivity reaction, increases the treatment cost and rapidly exhausts the antivenom reserves. In King Cobra envenomation, the effectiveness of OhMAV in regions outside Thailand is questionable when considering the extensive biogeographical distribution of King Cobra. The dosage of antivenom for King Cobra envenomation tends to be exceptionally high (beyond 20 vials), probably because of the need to neutralize a massive amount of venom injected - In author’s experience, King Cobra is capable of delivering more than 1 gram (dry weight) of venom in one bite, and it has the propensity to hold its bite on the bitten subject for a while (several minutes), instead of the bite-and-release strike as seen in other snakes.
Treatment of local envenomation
King Cobra envenomation can result in extensive tissue necrosis, which requires serial wound debridement, followed by reconstructive surgery using skin grafting and/or a flap, or, amputation in severe cases [37]. To prevent later sequelae, local tissue damages caused by King Cobra bite are best attended soon with surgical debridement to remove as much venom as possible. However, there is no consensus on the time to conduct such intervention to achieve the most optimal outcome. Early antivenom administration (intravenous) may potentially reduce the spread of venom’s local activity, but the efficacy has not been well established. In cases presenting with severe and progressive swelling, inadequately informed clinicians might view it as compartment syndrome needing fasciotomies, and often the decision to send the patient to surgery for a fasciotomy is taken without the supporting tests to measure the intra-compartmental pressure. In King Cobra envenomation, such radical surgery is unjustified as the swelling is typically subdermal instead of deep tissue as produced by crush injuries.
King Cobra venom: composition and bioactivity studies
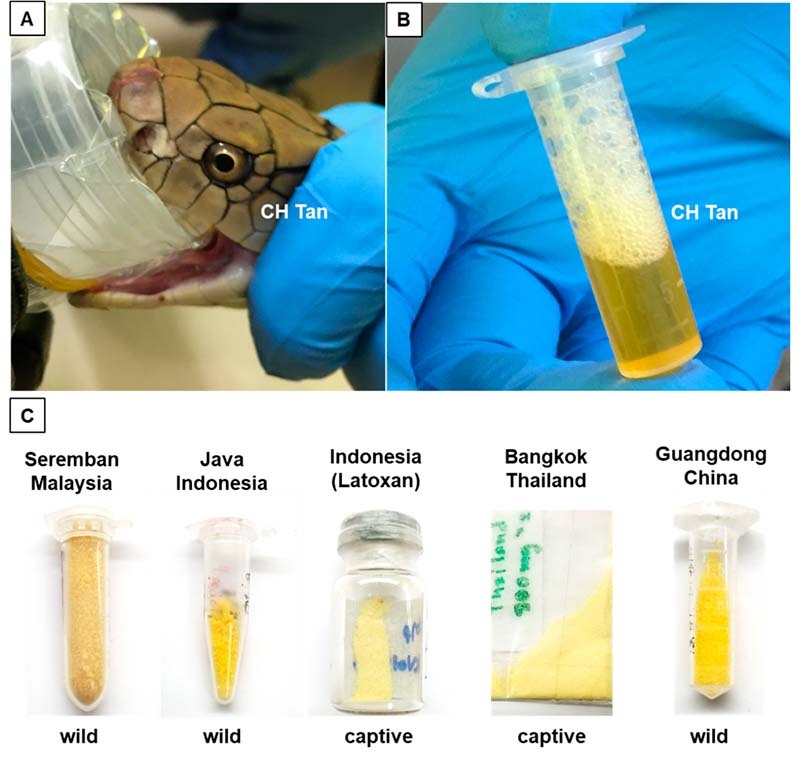
The studies are usually conducted on reconstituted venom powder, obtained through lyophilization (freeze-drying) or desiccation of crude venom milked from live snakes. King Cobra venom can be readily collected by inducing the snake to bite through a film covering a clean container, or, by directly placing its fangs over the edge of a collecting device. The venom of King Cobra is typically golden yellow in color, often viscous, and lyophilization yields powder with comparable coloration in different shades of yellow which is indicative of the presence of a flavin adenine dinucleotide (FAD)-containing flavoenzyme, L-amino acid oxidase (LAAO) (Figure 6).
Figure 6. Collection of King Cobra venom. (A) Milking of venom by inducing the snake to bite through a film-covered clean container. (B) Freshly milked venom with its bright golden color. (C) Venom powder in different shades of yellow obtained through lyophilization for long-term storage.
The investigation of King Cobra venom properties dated back to the 1960s when Tu et al. [46] studied the hemorrhagic and proteolytic activities of the venom. Various bioactive components had been isolated from the venom and characterized, including various proteins from the families of three-finger toxins (3FTx) [47,48], snake venom metalloproteinases (SVMP) [49], cysteine-rich secretory proteins (CRiSP) [50], phospholipases A2 (PLA2s) [51], L-amino acid oxidases (LAAOs) [52], Kunitz-type serine protease inhibitors (KSPIs) [53], cobra venom factors (CVFs) [54] and ohanin (vespryn) [55]. The global protein profile of King Cobra venom was only available with the advent of proteomics, which attempted to unveil the variety and abundances of all toxins (proteins) in the venom comprehensively. Thus far, a total of nine proteomic studies on King Cobra venom have been reported, with noticeable discrepancies in details. The proteomes were summarized in Table 2 and Table 3 for profiling based on quantitative and non-quantitative approaches, respectively.
Table 2. Quantitative proteomic profiling of King Cobra (Ophiophagus hannah) venom.
| Reference | Petras et al. [56] | Tan et al. [57] | Liu et al. [58] |
|---|---|---|---|
| Geographical origin | Indonesia, unspecified locale | Malaysia, Seremban (southwestern peninsula) | Indonesia, unspecified locale |
| Specimen habitat and source | Captivity, commercial source (Latoxan) | Wild | Captivity, commercial source (Latoxan) |
| Number of specimens | Unspecified | 1 | A few |
| Specimen growth state | Unspecified | Adult | Adult |
| Specimen sex | Unspecified | Unspecified | Unspecified |
| Methodology | C18 reverse-phase HPLC Reducing SDS-PAGE In-gel digestion LC-MS/MS | Reducing SDS-PAGE In-gel digestion LC-MS/MS | C18 reverse-phase HPLC Reducing SDS-PAGE In-gel digestion LC-MS/MS |
| Quantitation | Relative protein abundance of toxin (% of total venom proteins) and number of protein subtype (number in parentheses) | ||
| Toxin family | |||
| 3FTx | 64.2 (≥ 20) | 43.0 (13) | 60.65 (21) |
| Long neurotoxin | 37.1 | 26.7 | ~33.5 |
| Short neurotoxin | 10.2 | ~11.9 | |
| Weak toxin/neurotoxin | 13.7 | 3.9 | ~3.4 |
| Muscarinic toxin-like protein | 4.7 | - | - |
| Cytotoxin/cardiotoxin | 8.7 | 0.5 | 11.9 |
| SVMP | 11.9 (2-3) | 24.4 (12) | 15.63 (11) |
| PLA2 | 2.8 (5) | 4.0 (1) | 3.66 (3) |
| CRiSP | 6.5 (1) | 8.7 (1) | 7.46 (2) |
| KSPI | 3.3 (1) | 1.0 (1) | 2.93 (1) |
| LAAO | 0.5 (1) | 5.7 (2) | - |
| CVF / OVF | - | 2.8 (4) | - |
| Vespryn | 1.1 (1) | 5.7 (1) | 1.83 (2) |
| ILGF | 0.7 (1) | - | - |
| NP | 0.2 (3) | - | - |
| SVSP | - | 0.7 (1) | - |
| Cystatin | Trace (1) | 0.7 (1) | - |
| PDE | - | 1.1 (2) | - |
| NGF | - | 0.8 (1) | - |
| AChE | - | 0.1 (1) | - |
| Neprilysin | - | 0.7 (2) | - |
| PLB | - | 0.2 (1) | - |
| 5NT | - | 0.4 (1) | - |
| VEGF | - | - | - |
| CTL | - | - | - |
| Waprin | - | - | - |
| Others | - | - | 7.84 (9) Unknown protein |
3FTx: three-finger toxin; 5NT: 5' nucleotidase; AChE: acetylcholinesterase; CRiSP: cysteine-rich secretory protein; CTL: C-type lectin; CVF: cobra venom factor; HPLC: high performance liquid chromatography; ILGF: insulin-like growth factor; KSPI: Kunitz-type serine protease inhibitor; LAAO: L-amino acid oxidase; LC-MS/MS: liquid chromatography-tandem mass spectrometry; NGF: nerve growth factor; NP: natriuretic peptide; OVF: Ophiophagus venom factor; PAGE: polyacylamide gel electrophoresis; PDE: phosphodiesterase; PLA2: phospholipase A2; PLB: phospholipase-B; SVMP: snake venom metalloproteinase; SVSP: snake venom serine protease; VEGF: vascular endothelial growth factor.Note: 3FTx comprises of long neurotoxin, short neurotoxin, weak toxin/neurotoxin, muscarinic toxin-like protein and cytotoxin/cardiotoxin.
Table 3. Non-quantitative proteomic profiling of King Cobra (Ophiophagus hannah) venom.
| Reference | Vonk et al. [59] | Vejayan et al. [60] | Danpaiboon et al. [61] | Melani et al. [62] | Kunalan et al. [63] | Wongtay et al. [64] |
|---|---|---|---|---|---|---|
| Geographical origin | Indonesia, Bali Island | Malaysia, specific locale unknown | Thailand, specific locale unknown | Malaysia, specific locale unknown | Malaysia, Perlis (northwest Malaysia) | Thailand, Phetchabun, Songkhla, Suratthani |
| Specimen habitat and source | Unspecified | Unspecified, from local vendor | Captivity, from Queen Saovabha Memorial Institute | Captivity, from Kentucky Reptile Zoo in USA | Captivity, from a snake enthusiast | Unclear, captive-born was included |
| Number of specimens | 1 | Unspecified | Unspecified | 2 | Unspecified | One per locale, total of 3 |
| Specimen growth state | Adult | Unspecified | Unspecified | Unspecified | Unspecified | Unspecified |
| Specimen sex | Unspecified | Unspecified | Unspecified | Unspecified | Unspecified | Unspecified |
| Methodology | Whole venom in-solution digestion LC-MS/MS | 2D-PAGE In-gel digestion MALDI-TOF MS | 2D-PAGE In-gel digestion LC-MS/MS | GELFrEE Solution isoelectric focusing In-solution digestion LC-MS/MS | Gel-filtration chromatography In-solution digestion LC-MS/MS | 2D-PAGE In-gel digestion MALDI-TOF MS |
| Detection | Occurrence of toxin in percentage1 (number of proteins in parentheses) | Number of proteins | Number of proteins | Number of proteins (number of distinct proteoforms in parentheses) | Occurrence of toxin in percentage1 (number of proteins in parentheses) | +: Detected |
| Toxin family | ||||||
| 3FTx | 41.1% (30) | 7 | 16 | 57 (29) | 31.0% (55) | + |
| SVMP | 24.7% (18) | - | 4 | 14 | 25.0% (44) | - |
| PLA2 | 5.5% (4) | 4 | 5 | 10 (9) | 5.0% (9) | + |
| CRiSP | 6.8% (5) | - | 3 | 3 (3) | 9.0% (15) | + |
| KSPI | 4.1% (3) | - | 3 | 2 (8) | 3.0% (6) | - |
| LAAO | 1.4% (1) | - | 1 | 2 | 2.0% (3) | - |
| CVF / OVF | 5.5% (4) | 1 | 1 | 4 | 4.0% (7) | - |
| Vespryn | 1.4% (1) | 1 | 2 | 3 (4) | 1.0% (2) | + |
| ILGF | - | - | 1 | 3 | 3.0% (5) | - |
| NP | 1.4% (1) | - | - | - | - | + |
| SVSP | 4.1% (3) | 1 | - | 1 | 1.0% (2) | - |
| Cystatin | - | - | - | - | - | - |
| PDE | 1.4% (1) | - | 1 | - | 6.0% (11) | - |
| NGF | - | - | - | - | 1.0% (2) | - |
| AChE | - | - | - | - | 1.0% (2) | - |
| Neprilysin | - | - | - | - | 1.0% (1) | - |
| PLB | - | - | - | - | 1.0% (1) | - |
| 5NT | - | - | 1 | - | 2.0% (3) | - |
| VEGF | 2.7% (2) | - | - | 3 | 1.0% (1) | + |
| CTL | - | - | - | - | 1.0% (2) | + |
| Waprin | - | - | - | 1 | - | - |
| Others | - | 1 Thioredoxin | 4 Hypothetical 4 Other | 2 Disintegrin 25 non-toxin 1 SVGF | 2.0% (3) SVGF 1.0% (2) Endonuclease | + (unspecified) |
3FTx: three-finger toxin; 5NT: 5' nucleotidase; AChE: acetylcholinesterase; CRiSP: cysteine-rich secretory protein; CTL: C-type lectin; CVF: cobra venom factor; GELFrEE: Gel-eluted liquid fraction entrapment electrophoresis; ILGF: insulin-like growth factor; KSPI: Kunitz-type serine protease inhibitor; LAAO: L-amino acid oxidase; LC-MS/MS: liquid chromatography-tandem mass spectrometry; NGF: nerve growth factor; NP: natriuretic peptide; OVF: Ophiophagus venom factor; PAGE: polyacylamide gel electrophoresis; PDE: phosphodiesterase; PLA2: phospholipase A2; PLB: phospholipase-B; SVGF: snake venom growth factor; SVMP: snake venom metalloproteinase; SVSP: snake venom serine protease; VEGF: vascular endothelial growth factor.
+: indicates detection; -: indicates not reported.
A ratio obtained whereby the number of proteins in a family is divided by the total number of all proteins detected in proteome.
Three-finger toxins (3FTx)
Molecular cloning and recent -omic studies demonstrated the diversity of three-finger toxins that include alpha-neurotoxins, muscarinic toxin-like proteins (MTLPs), weak neurotoxins (WNTXs) and cardiotoxins/cytotoxins (CTXs) in King Cobra venom [47,56,57,59]. Of these, the alpha-neurotoxins (long neurotoxin, LNTX and short neurotoxin, SNTX) are the most diversely and abundantly expressed (Table 2 and Table 3). SNTX and LNTX are the main neurotoxins with high lethal activity (median lethal dose, LD50 ~0.05-0.20 µg/g, in mice), commonly found in the venoms of most Asian elapids (Naja cobras, kraits, sea snakes) [57,65-67]. MTLP and WNTX belong to the short-chain and non-conventional subfamilies of 3FTx, respectively. MTPLs interact with muscarinic acetylcholine receptors (mAChR) [68] while WNTXs bind to both muscular and neuronal nAChR but lack lethal activity (LD50 = 4-80 µg/g) [69,70]. CTXs, on the other hand, exhibit a more diverse array of pharmacological activities. CTX isolated from Naja cobra venoms typically show cytolytic and cytotoxic activities [71]. The CTX from King Cobra venom, i.e., beta-cardiotoxin (β-CTX), was shown to bind to β-1 and β-2 adrenergic receptors of cardiomyocytes, and exhibit negative chronotropic activity [48,72]. Cardiac complication has not been well established in clinical envenomation caused by King Cobra, presumably due to the low abundance of this toxin in the venom.
Quantitative proteomics showed that 3FTxs are the most abundant toxins in King Cobra venom from Malaysia (accounting for 43% of the total venom protein; 13 subtypes) [57] and Indonesia (64.2%, ≥ 20 subtypes [56], and 60.65%, 21 subtypes [58]). Among these, α-NTXs accounted for the majority of 3FTx (Table 2). Consistently, LNTX were present at a higher relative abundance than SNTX in the Malaysian (26.7% LNTX, 7.5% SNTX) and the Indonesian (33.5% LNTX, 11.9% SNTX) specimens. The abundance of CTX was rather variable, constituting 8.7-11.9% of total venom proteins in the Indonesian King Cobra venom [56,58] while only found at a negligible amount in the Malaysian King Cobra venom proteome (0.5%) [57]. Interestingly, β-CTX was recently isolated from the Thai King Cobra venom, also at a very low recovery rate (0.53% w/w). The existing data tend to indicate that the King Cobras from Malaysia and Thailand, and perhaps the Indochinese Peninsula, probably share a venom phenotype in which very low amount of CTX is expressed, in contrast to the insular populations from Indonesia. The abundances of non-conventional 3FTx, on the other hand, were consistent in the Malaysian and Indonesian King Cobra venom proteomes (~4%). In proteomic studies where relative abundances of proteins were not reported [59-64], 3FTxs overall showed the highest number of toxin subtypes detected in the venom (Table 3).
Snake venom metalloproteinases (SVMPs)
The SVMPs are multi-domain zinc-dependent enzymes of varying molecular weights. They are the principal toxins responsible for vasculature damage and may interfere with hemostasis, contributing to hemorrhage and coagulopathy in envenomation caused by vipers and pit vipers [73,74]. Compared to most cobra venoms in which SVMPs were little (< 1% of total venom proteins), SVMPs in King Cobra venom were found to be more diverse and abundantly expressed, accounting for 24.4% (12 subtypes) [57], 11.9% (2-3 subtypes) [56] and 15.63% (11 subtypes) [58] of total venom proteins in three separate Southeast Asian samples (Table 2). All SVMPs identified in King Cobra venom belong to the PIII class, which is made up of a metalloproteinase (M) domain, a disintegrin (D) domain and a cysteine-rich (C) domain. PIII-SVMPs are generally more potent hemorrhagin than PI-SVMPs (containing only M domain) and PII-SVMPs (containing M and D domain) [73]. The genomic and transcriptomic studies of King Cobra [57,59] revealed that the metalloproteinase of their PIII-SVMPs have only six cysteine residues, in contrast to most viperid SVMPs whose M domains contain an additional seventh cysteine residue at varying positions. Previous studies correlated the presence of the seventh cysteinyl residue in some SVMPs to vascular apoptosis-inducing activity, and indicated that in PIIIa subclass, the seventh cysteinyl residue at position 195 may be implicated in alternative disulfide bond pairing and proteolytic processing whereby proteolysis/autolysis produces a biologically active DC (disintegrin- and cysteine-rich) domain [73]. The evolutionary and medical implication of the sequence variation seen in King Cobra SVMP (PIII) has yet to be dissected out. Clinically, hemorrhagic effect is not observed in human envenomation caused by King Cobra, although its venom has been found to show hemorrhagic activity in rabbits and hares [75]. In addition, a 63−66 kDa hemorrhagin (with SVMP biochemical properties) was isolated from King Cobra venom previously and shown to exhibit species-sensitive hemorrhagic and lethal activities in rabbits, while its toxicity in mice was minimum [76]. Apparently, the pathophysiological role of King Cobra SVMP in human envenomation is under-explored, although its substantial abundance suggests involvement in local tissue damage and necrosis resulting from their inflammatory and proteolytic activities which are instrumental for foraging and digestive purposes.
Cysteine-rich secretory proteins (CRiSPs)
Cysteine-rich secretory proteins (CRiSPs) were the third most abundant protein family detected in King Cobra venom proteomes, constituting 6.5% to 8.7% of total venom proteins [56-58]. Ophanin, a CRiSP isolated from O. hannah venom [50], was commonly identified in the venom proteomes reported. The biological activities of CRiSP include the inhibition of voltage-dependant Ca2+ channels and blockade of smooth muscle contraction [50]. Despite its substantial abundance, the pathogenic role of Ophanin and other CRiSP proteoforms in King Cobra envenomation remains unknown.
Phospholipases A2 (PLA2s)
In King Cobra venom proteome, PLA2s were represented by a rather small number of subtypes and a low abundance (2.8-4% of total venom protein) [56-58]. The PLA2s in King Cobra venom consist of two distinct subgroups: group IA, including acidic PLA2s which exhibited mild proinflammatory activity but were non-lethal to mice at a high dose of 10 µg/g [77], and group IB with weak lethality and cardiotoxicity in mice [51]. While snake venom PLA2s are commonly present in most snake venoms implicated in the pathophysiology of envenomation, the function of PLA2 in King Cobra venom is likely ancillary without direct lethal activity.
L-amino acid oxidases (LAAOs)
King Cobra venom is a rich source of snake venom LAAO - the intense yellow coloration of the venom reflects the high content of FAD-containing LAAO in the venom (Figure 6). LAAO was reported to constitute close to 6% of total venom proteins in the Malaysian King Cobra venom proteome [57]. In the venom proteome of Indonesian specimen, the enzyme was either undetected [58] or present at only 0.5% of total venom proteins [56], although the Indonesian King Cobra venom too exhibits strong LAAO enzymatic activity [21], and shows yellow coloration (Figure 6). The substantial amount of LAAO in the Malaysian King Cobra venom [57] supported its strong enzymatic activity [21], and suggests the involvement of this enzyme in the development of local tissue damage (cytotoxic activity) and pain in King Cobra envenomation [78].
Minor toxin proteins
Several protein families of low abundances had been reported in proteomic studies of King Cobra venom. Among these were Kunitz-type serine protease inhibitors (KSPIs), which accounted for 1.0-3.3% of total venom proteins [56-58]. Snake venom KSPIs exhibit various biological functions including trypsin/chymotrypsin inhibition, anticoagulant and neurotoxic activity [79-81]. The role of KSPI in King Cobra venom, however, remains not well characterized to date. Vespryn, a novel snake venom protein family, was reported at a relative abundance between 1% and 5% of total venom proteins. Ohanin is a vespryn protein specific to King Cobra and capable of inducing hypolocomotion in mice. The absence of peripheral neurotoxicity of vespryn, nonetheless, implies a direct action on the central nervous system [55]. The hypolocomotion effect of ohanin probably contributes toward the predatory function of King Cobra venom.
Other minor protein families were reported rather variably in the King Cobra venom proteomes. Four subtypes of cobra venom factors (CVFs), including Ophiophagus venom factor (OVF) accounted for 2.8% of the Malaysian King Cobra venom proteome [57] while none was reported in the proteomes of Indonesian specimens [56,58]. CVF/OVF are non-lethal proteins structurally similar to complement C3 homolog (CC3H), and presumably involved in the activation of complement system. The release of C3a and increases vascular permeability and thus, facilitates systemic spread of the venom [82]. Other proteins of minor abundance are present at a relatively low or negligible amount in the venom, and these probably play an ancillary role in the predatory or digestive function of the venom. These minor components include proteins from the families of snake venom serine protease (SVSP) [57,59,60,62,63], phosphodiesterase (PDE) [57,59,61,63], acetylcholinesterase (AChE) [57,63], phospholipase-B (PLB) [57,63], C-type lectin (CTL) [63], 5’-nucleotidase (5NT) [57,60,63], natriuretic peptide (NP) [56,59,64], insulin-like growth factor (ILGF) [56,61-63], nerve growth factor (NGF) [57,63], vascular endothelial growth factor (VEGF) [59,62-64], cystatin [56,57], neprilysin [57,63] and waprin [63].
Reflections on venom proteomics
The proteomics of King Cobra venom sourced from different geographical locales has been studied to varying depths. Specimens studied thus far were from Malaysia (peninsula) [21,57,60,62,63], Indonesia (Java Island and Bali Island) [21,56,58,59], Thailand [21,61,64] and China [21], while distant and disjunct populations from India, Borneo and the Philippines have not been investigated. Comparison of the proteomes revealed remarkable variations in terms of the subtypes and abundances of toxin proteins identified (Table 2). These quantitative and qualitative differences probably reflected true intra-species venom variability of King Cobra from allopatric populations, and therefore were regarded as geographical variation in venom composition. However, the “proteomic differences” across the above studies were more likely due to sampling and technical reasons. Firstly, the origins and sources of the venom samples were diverse: the direct geographic provenance, from which the precise locales within each country is often not disclosed [56,58,60-62]. Information on the age, sex and number of snakes contributed to the venom sample was often lacking or incomplete. Additionally, some samples were either obtained from the wild specimen [57,59,64], or, presumably captive or farmed specimens through commercial serpentaria [56,58,62], or unreported [60,61,63]. On the other hand, the “functional proteomic approach” reported by Chang et al. [21] focused on characterizing enzymatic activities and identification of only 2 protein families (neurotoxins and phospholipases A2) without a comprehensive profile of all venom proteins. Possible interference from factors relating to the sex, developmental (ontogenic) stage and habitat of the snake cannot be excluded when addressing the variability of snake venom composition. Comparison of the findings and interpretation for intra-species variation is, therefore, challenging.
The second and perhaps more influential factor is the methodology of analysis. The different approaches employed in each study resulted in varying depths of proteomic profiling, which could be confounding when comparison is made, resulting in misinterpretation of geographical venom variation. Proteomics in snake venom research aims to comprehensively profile the diversity (how many?) and protein abundance (how much?) of all toxins present in a venom. The identification of high protein diversity is usually achieved using protein decomplexation strategies that deploy chromatographic or electrophoretic separation methods of venom proteins, prior to mass spectrometry analysis [83-86]. The sensitivity of the detection tool and the depth of database used in the bioinformatic analysis play crucial role in this regard. On the other hand, the quantitation methods of protein abundance in venom proteomics varied greatly. Of the nine proteomic studies of King Cobra venom to date, four listed and numbered the proteins identified [60-62,64], while five, in addition to protein name listing, also reported the “percentage” of individual protein or protein family found in the proteome [56-59,63] (Tables 2 and 3). Among the latter five studies that reported protein percentages, three truly quantified the relative protein abundance as percentage in terms of total venom proteins, estimated based on parameters such as peak area under the curve (chromatography), intensity of gel band on electrophoresis, and/or spectral input of peptide ions in mass spectrometry [56-58]. In two other studies including the proteome reported alongside the King Cobra’s genomics [59,63] (Table 3), the “protein percentages” reported indicated the ratio of a protein number, mathematically derived through dividing the number of proteins (those of the same family) by the total number of all proteins detected. Using this simple ratio calculating method, the more diverse a protein family is, the more “abundant” it will appear to be, but this association is not necessarily true and is not compatible with the analysis for “relative protein abundance”. Therefore, the inadequate information of the geographical sample of venom, and the different methods of analysis applied impede meaningful comparison of King Cobra venom proteomes reported in different studies.
Variation in King Cobra venom toxicity and neutralization
Earlier, Tan et al. [87] reported no remarkable variation in the toxicity of King Cobra venoms sampled from limited sources (two specimens from peninsular Malaysia, one specimen from southern Thailand, and one from a commercial source of unspecified locality). Chang et al. [21] subsequently compared the lethality and enzymatic activities of King Cobra venoms from five geographic locales, i.e., Thailand, Malaysia, Indonesia and two Chinese localities (Hainan and Guangxi) (the number of snakes in each geographical sample was not known). Their results demonstrated marked variations in the enzymatic activities and taxon-specific lethality between the Chinese and the Southeast Asian King Cobra venoms. The venom samples from Southeast Asia (Thailand, Malaysia, Indonesia) exhibited higher metalloproteinase, acetylcholinesterase and alkaline phosphatase activities than the Chinese samples. Taxon-specific lethality test was conducted in mice and lizards (Eutropis multifasciata) via intra-peritoneal route. It revealed that the Chinese samples were more lethal to mice with an i.p. LD50 of 0.5−0.6 µg/g, comparing to samples from Indonesia (1.1 µg/g), Malaysia (2.9 µg/g) and Thailand (3.5 µg/g), whereas the Southeast Asian samples were more lethal to lizards (Malaysia: 13.9 µg/g; Indonesia: 17.6 µg/g; Chinese/Guangxi: 30.2 µg/g; Thai and Hainanese samples not done) [21]. Another study compared the toxicity of three King Cobra venom samples, each derived from a single snake of Thai origin in different provinces (Phetchabun, Songkhla and Suratthani) [64], and found that the Phetchabun King Cobra venom was more cytotoxic and more lethal (however LD50 not reported) than the two other samples. Considering the very small sample number of this latter study, a conclusion from the geographic perspective could not be drawn as the variation observed is likely inter-individual difference.
More recently, the lethal activities of King Cobra venoms from four distant geographical locales (Thailand, Malaysia, Indonesia, and China) were shown to vary considerably [22]. The Chinese and Indonesian King Cobra venoms were the most lethal to mice (intravenous LD50 ~0.50 µg/g) in comparison to the Malaysian (LD50 = 0.90 µg/g) and Thai (LD50 = 1.04 µg/g) specimens. In vivo neutralization study revealed poor cross-neutralization of King Cobra venom lethalities by non-specific antivenoms, i.e., the Chinese Naja atra Monovalent Antivenom (NaMAV) and Indonesian Serum Antibisa Ular (SABU), which are commonly used in the respective countries to treat King Cobra envenomation [22]. The species-specific antivenom, Thai OhMAV effectively neutralized the lethality of the King Cobra venoms to different extents. The study expressed the efficacy of the antivenom in median effect dose (ED50), defined as the volume dose of antivenom needed to protect half of the tested mice from venom-induced lethality. OhMAV was found more efficacious in neutralizing the venom of Thai origin (median effective dose, ED50 = 39.37 µL) than it was against the Malaysian (ED50 = 129.09 µL), Indonesian (ED50 = 139.58 µL) and Chinese (ED50 = 170.16 µL) venom samples. The observation strongly supports the species-specificity of OhMAV toward its immunogen of Thai origin, and underscores the impact of King Cobra venom variation on the effectiveness of antivenom treatment (Figure 7).
Figure 7. Schematic representation of geographical variation in the lethality of King Cobra venom sourced from different locales: Bangkok (Thailand), Seremban (West Malaysia), Guangzhou (China) and Java Island (Indonesia). The differential potency of King Cobra-specific antivenom, OhMAV and locally used non-specific antivenoms, NaMAV (China) and SABU (Indonesia), in neutralizing the venom lethality was illustrated. OhMAV: Ophiophagus hannah Monovalent Antivenom (produced in Thailand); NaMAV: Naja atra Monovalent Antivenom (produced in China); SABU: Serum Anti Bisa Ullar (produced in Indonesia).
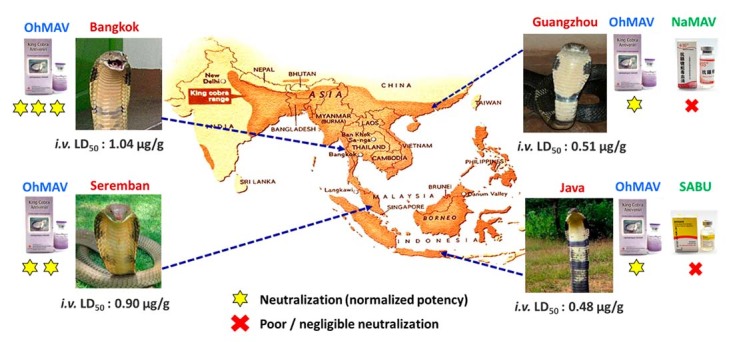
The existing data tend to support that the biochemistry and toxicity of King Cobra venom vary geographically, with a marked divergence between the Chinese and most Southeast Asian populations. From the ecological perspective, the variation in taxon-specific toxicity (in which the Chinese King Cobra venom was more lethal to mice while the Southeast Asian King Cobra venoms were more lethal to lizards) [21] suggests a possible dietary shift from mainly ophiophagy toward an enlarged scope of prey including mammals in the Chinese King Cobra, or inversely a dietary specialization from opportunistic feeding toward ophiophagy in the Southeast Asian King Cobra. The taxa-dependent toxicity of the venom is likely a result of arms races between King Cobra and available prey. Geographical differences in prey abundance, both ophidian and mammalian, between subtropical China (at a latitude above the tropic of Cancer, 23°26’) and tropical Southeast Asia, as well as anthropogenic factors such as habitat destruction or cultural practice (China being a well-known region historically suffering from pest rodents [88]), could have all contributed to the geographical variation in King Cobra venom, shaping divergent venom phenotype in the species.
Antivenomics to decipher the limitation of antivenom neutralization
Antivenomics is applied to examine the immunorecognition of an antivenom to protein components in a venom. Through the identification of toxins that are poorly bound immunologically, the weakness of an antivenom can be identified for improvement. The information on King Cobra antivenomics is however limited. Using a 2DE-immunoblotting technique, Danpaiboon et al. [61] earlier investigated the immunological binding of Thai King Cobra venom by human single-chain variable antibody fragments specific to N. kaouthia LNTX (NkLN-HuScFv, produced in laboratory). The experimental hetero-specific antibody bound to a short and a long neurotoxin in King Cobra venom, while weakly cross-neutralized the lethality of the venom. Another study by Liu et al. [58] reported the cross-reactivity (applying 1DE-immunoblot) and cross-neutralization of Indonesian O. hannah venom by the Taiwanese neurotoxic bivalent antivenom (TNBAV, CDC product), which is raised against Bungarus multicinctus and Naja atra venoms. Similarly, the finding showed that TNBAV cross-immunoreacted with most O. hannah HPLC-eluted venom fractions but failed to cross-neutralize the venom toxicity in mice [58]. The phenomenon further supports the need to use a species-specific antivenom, i.e., OhMAV, to achieve effective neutralization in King Cobra envenomation [22]. The differential efficacy of OhMAV against King Cobra venom of different locales, however, awaits elucidation with antivenomic approaches.
Conclusions
King Cobra (Ophiophagus hannah) has a wide geographical distribution in Asia, and is a medically important venomous snake capable of causing fatal envenomation. Epidemiological data of King Cobra envenomation, as with that of snakebite in the region, is relatively scarce. Worldwide, the only species-specific antivenom indicated for treating King Cobra envenomation (Ophiophagus hannah Monovalent Antivenom, OhMAV) is produced in Thailand by the Queen Saovabha Memorial Institute, Bangkok, raised against the venom of Thai King Cobra. Clinical reports showed that high doses of the mono-specific King Cobra antivenom, often beyond 20 vials, were needed in the treatment of King Cobra envenomation. Preclinical assessment of OhMAV neutralization activity revealed variable efficacy against the venoms of King Cobra from different locales, indicating vast geographical variation in the composition and antigenicity of the venom. Geographical variation of the venom, however, has not been comprehensively investigated despite a number of proteomic studies reported on King Cobra venom. Discrepancies in the venom proteomes reported across the different studies were observed, presumably due to differences in the sampling and method of analysis, which resulted in varying depths of venom profiling and incompatible quantitation of protein abundances. Comparison of King Cobra venom proteomes reported from different studies for the interpretation of geographical venom variability thus remains challenging. Furthermore, mechanistic and antivenomic studies are also lacking to elucidate the clinical pathophysiology of envenomation and the variable effectiveness of OhMAV in neutralizing King Cobra venom.
Future works should be tailored toward a comprehensive proteomic study of King Cobra venom from various locales, taking consideration into the potential variation arising from ontogenic factor and the environment of habitat (wild v.s. captivity). A standard method of proteomic analysis should be adopted, preferably one that incorporates the protein decomplexation strategy and high-resolution mass spectrometry coupled with deep database mining for better detection of protein diversity. The quantitation of protein should also be standardized, and in this context, the term “percentage” should be used cautiously to denote relative protein abundance, so that the composition of a protein is defined by its amount with respect to the total venom proteins. Furthermore, through high-performance liquid chromatography of the venom (protein decomplexation), it is possible to isolate the various venom proteins eluted for further functional investigation. This will help identify the key toxins and elucidate their toxicity, providing deeper insights into the pathophysiology and treatment of envenomation according to the variability of venom. Furthermore, the antivenomic approach based on either enzyme-link immunosorbent assay, immunoblotting or affinity chromatography, should follow to examine the degree of immunorecognition or immunocapturing capacity of OhMAV toward the different toxin components in the venom, for specimens originated from different geographical populations. Importantly, the immunologically-derived antivenomic findings must be interpreted against the in vivo efficacy of OhMAV, in which the venom and isolated toxin principles are subject to neutralization as outlined in functional antivenomics [65,89]. These are crucial to demonstrate the weakness or limitation of the antivenom in neutralizing the King Cobra venom of different geographical sources, so that the antivenom manufacturing can be improved to yield a product with higher potency and broader geographical utility. One promising approach is the potential use of fully human monoclonal antibodies as a recombinant antivenom targeting a particular animal toxin, for instance, dendrotoxins from the African Black Mamba [90]. Although recombinant antivenoms are possible alternatives to animal-derived antivenoms [91], the application probably remains far-reached in authors’ opinion. A simple, pragmatic and affordable approach can be taken to improve the existing antivenom production through a better process of antibody purification, and, by developing a pan-regional antivenom from immunogen mixture(s) containing the different target toxins in a formula optimized for hyperimmunization in host animals [92,93]. This shall contribute toward better management of snakebite envenomation in the region, in line with the WHO’s strategy to halve the disease burden of snakebite by 2030.
Acknowledgments
The authors are grateful to the photographers for the images used in the paper.
Footnotes
Availability of data and materials: Not applicable.
Funding: This work was supported by Special Research Grant (BKS003-2020) from the University of Malaya, Malaysia.
Ethics approval: Not applicable.
Consent for publication: Not applicable.
References
- World Health Organization . Guidelines for the management of snakebites WHO Regional Office for Southeast Asia. 2016. [Google Scholar]
- Gutierrez JM, Calvete JJ, Habib AG, Harrison RA, Williams DJ, Warrell DA. Snakebite envenoming. Nat Rev Dis Primers. 2017;3:17063. doi: 10.1038/nrdp.2017.63. [DOI] [PubMed] [Google Scholar]
- World Health Organization . Fact Sheets. 2021. [20 June 2021]. Snakebite envenoming. In Fact Sheets https://www.who.int/news-room/fact-sheets/detail/snakebite-envenoming . [Google Scholar]
- Chippaux JP. Snake-bites: appraisal of the global situation. Bull World Health Organ. 1998;76(5):515–524. [PMC free article] [PubMed] [Google Scholar]
- Harrison RA, Hargreaves A, Wagstaff SC, Faragher B, Lalloo DG. Snake envenoming: A disease of poverty. PLoS Negl Trop Dis. 2009;3(12):e569. doi: 10.1371/journal.pntd.0000569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasturiratne A, Wickremasinghe AR, de Silva N, Gunawardena NK, Pathmeswaran A, Premaratna R, Savioli L, Lalloo DG, de Silva HJ. The global burden of snakebite: A literature analysis and modelling based on regional estimates of envenoming and deaths. PLoS Med. 2008;5(11):e218. doi: 10.1371/journal.pmed.0050218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Needleman RK, Neylan IP, Erickson T. Potential environmental and ecological effects of global climate change on venomous terrestrial species in the wilderness. Wilderness Environ Med. 2018;29(2):226–238. doi: 10.1016/j.wem.2017.11.004. [DOI] [PubMed] [Google Scholar]
- Nori J, Carrasco PA, Leynaud GC. Venomous snakes and climate change: ophidism as a dynamic problem. Clim Change. 2014;122(1):67–80. [Google Scholar]
- Kasturiratne A, Pathmeswaran A, Wickremasinghe AR, Jayamanne SF, Dawson A, Isbister GK, de Silva HJ, Lalloo DG. The socio-economic burden of snakebite in Sri Lanka. PLoS Negl Trop Dis. 2017;11(7):e0005647. doi: 10.1371/journal.pntd.0005647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaiyapuri S, Vaiyapuri R, Ashokan R, Ramasamy K, Nattamaisundar K, Jeyaraj A, Chandran V, Gajjeraman P, Baksh MF, Gibbins JM, Hutchinson EG. Snakebite and its socio-economic impact on the rural population of Tamil Nadu, India. PLoS One. 2013;8(11):e80090. doi: 10.1371/journal.pone.0080090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chippaux JP. Snakebite envenomation turns again into a neglected tropical disease! J Venom Anim Toxins incl Trop Dis. 2017;23:38. doi: 10.1186/s40409-017-0127-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams D, Gutiérrez J, Calvete J, Wüster W, Ratanabanangkoon K, Paiva O, Brown NI, Casewell NR, Harrison RA, Rowley PD, O'Shea M, Jensen SD, Winkel KD, Warrell DA. Ending the drought: New strategies for improving the flow of affordable, effective antivenoms in Asia and Africa. J Proteomics. 2011;74(9):1735–1767. doi: 10.1016/j.jprot.2011.05.027. [DOI] [PubMed] [Google Scholar]
- Casewell NR, Jackson TNW, Laustsen AH, Sunagar K. Causes and consequences of snake venom Variation. Trends Pharmacol Sci. 2020;41(8):570–581. doi: 10.1016/j.tips.2020.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amorim FG, Costa TR, Baiwir D, De Pauw E, Quinton L, Sampaio SV. Proteopeptidomic, functional and immunoreactivity characterization of Bothrops moojeni snake venom: Influence of snake gender on venom composition. Toxins (Basel) 2018;10(5) doi: 10.3390/toxins10050177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stuart B, Wogan G, Grismer L, Auliya M, Inger RF, Lilley R, Chan-Ard T, Thy N, Nguyen TQ. Ophiophagus hannah. 2012. [2020 3rd June]. https://www.iucnredlist.org/species/177540/1491874 . [Google Scholar]
- Das A, Nair MTV, Ahmed MF, Sharma PK. Distribution of King Cobra (Ophiophagus hannah) in northeastern India with new altitudinal record and notes on its habitat. Tiger Paper. 2008;35:1–6. [Google Scholar]
- Marshall BM, Strine CT, Jones MD, Artchawakom T, Silva I, Suwanwaree P, Goode M. Space fit for a king: spatial ecology of King Cobras ( Ophiophagus hannah ) in Sakaerat Biosphere Reserve, Northeastern Thailand. Amphibia-Reptilia. 2019;40:163–178. [Google Scholar]
- Bogert CM. Hamadryas Preoccupied for the King Cobra. Copeia. 1945;1945(1):47 [Google Scholar]
- Figueroa A, McKelvy AD, Grismer LL, Bell CD, Lailvaux SP. A species-level phylogeny of extant snakes with description of a new colubrid subfamily and genus. PLoS One. 2016;11(9):e0161070. doi: 10.1371/journal.pone.0161070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suntrarachun S, Chanhome L, Sumontha M. Phylogenetic analysis of the King Cobra, Ophiophagus hannah in Thailand based on mitochondrial DNA sequences. Asian Biomed. 2014;8:269–274. [Google Scholar]
- Chang HC, Tsai TS, Tsai IH. Functional proteomic approach to discover geographic variations of King Cobra venoms from Southeast Asia and China. J Proteomics. 2013;89:141–153. doi: 10.1016/j.jprot.2013.06.012. [DOI] [PubMed] [Google Scholar]
- Tan KY, Ng TS, Bourges A, Ismail AK, Maharani T, Khomvilai S, Sitprija V, Tan NH, Tan CH. Geographical variations in King Cobra (Ophiophagus hannah) venom from Thailand, Malaysia, Indonesia and China: On venom lethality, antivenom immunoreactivity and in vivo neutralization. Acta Trop. 2020;203:105311. doi: 10.1016/j.actatropica.2019.105311. [DOI] [PubMed] [Google Scholar]
- Thorpe RS, Pook CE, Malhotra A. Phylogeography of the Russell's viper (Daboia russelii) complex in relation to variation in the colour pattern and symptoms of envenoming. Herpetol J. 2007;17(4):209–218. [Google Scholar]
- Bhaisare D, Ramanuj VP, Shankar P, Shankar M, Goode M, Whitaker R. Observations on a wild King Cobra (Ophiophagus hannah), with emphasis on foraging and diet. IRCF Reptiles Amphib. 2010;17(2):95–102. [Google Scholar]
- Leakey JHE. Observations made on King Cobras in Thailand during May 1966. J Nat Res Council Thailand. 1969;5:1–10. [Google Scholar]
- Veto T, Price R, Silsby JF, Carter JA. Treatment of the first known case of King Cobra envenomation in the United Kingdom, complicated by severe anaphylaxis. Anaesthesia. 2007;62(1):75–78. doi: 10.1111/j.1365-2044.2006.04866.x. [DOI] [PubMed] [Google Scholar]
- Wetzel WW, Christy NP. A King Cobra bite in New York City. Toxicon. 1989;27(3):393–395. doi: 10.1016/0041-0101(89)90186-4. [DOI] [PubMed] [Google Scholar]
- Ganthavorn S. A case of King Cobra bite. Toxicon. 1971;9(3):293–294. doi: 10.1016/0041-0101(71)90084-5. [DOI] [PubMed] [Google Scholar]
- Gold BS, Pyle P. Successful treatment of neurotoxic King Cobra envenomation in Myrtle Beach, South Carolina. Ann Emerg Med. 1998;32(6):736–738. doi: 10.1016/s0196-0644(98)70075-7. [DOI] [PubMed] [Google Scholar]
- Ismail AK. In: Toxinology: Clinical Toxinology in Asia Pacific and Africa. Gopalakrishnakone P, Faiz A, Fernando R, Gnanathasan C, Habib A, Yang CC, editors. Vol. 2. Dordrecht: Springer Netherlands; 2015. Snakebite and Envenomation Management in Malaysia; pp. 71–102. [Google Scholar]
- Tin M, Rai M, Maung C, Tun P, Warrell DA, editors. Bites by the King Cobra (Ophiophagus hannah) in Myanmar: successful treatment of severe neurotoxic envenoming. Q J Med. 1991;80(3):751–762. [PubMed] [Google Scholar]
- Post TJ. Snake charmer dies after being bitten by King Cobra. [2020 17th June];The Jakarta Post. 2020 The Jakarta Post https://www.thejakartapost.com/news/2020/01/28/snake-charmer-dies-after-being-bitten-by-king-cobra.html . [Google Scholar]
- Moyer J. Indonesian singer performing with King Cobra dies after being bitten onstage. [2020 June 17th];Washington Post. 2016 Washington Post https://www.washingtonpost.com/news/morning-mix/wp/2016/04/08/indonesian-singer-performing-with-king-cobra-dies-after-being-bitten-onstage . [Google Scholar]
- Alagesh T. Zookeeper in coma after cobra attack. 2012;2012 [Google Scholar]
- Tan CH, Tan NH. In: Snake Venoms. Gopalakrishnakone P, Inagaki H, Mukherjee AK, Rahmy TR, Vogel C-W, editors. Dordrecht: Springer Netherlands; 2015. Toxinology of Snake Venoms: The Malaysian Context; pp. 1–37. [Google Scholar]
- Ismail A, MohdFyzal B, Soo C, Yeap BL, Ah MF. Locked-in syndrome following a King Cobra (Ophiophagus hannah) envenomation. Med Health. 2017;12:357–362. [Google Scholar]
- Imran R, Vanat Q, Hausien O, Jose R. King Cobra bite - Can early decompression prevent digital amputation? JPRAS Open. 2020;27:12–16. doi: 10.1016/j.jpra.2020.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- White J, Weinstein S, Bates D. University of Adelaide Clinical Toxinology Resource. 2015. http://www.toxinology.com/fusebox.cfm?fuseaction=main.snakes.display&id=SN0048
- Ministry of Health Malaysia . Guideline: Management of Snakebite. 2017. [Google Scholar]
- Ledsgaard L, Jenkins TP, Davidsen K, Krause KE, Martos-Esteban A, Engmark M, Andersen MR, Lund O, Laustsen AH. Antibody cross-reactivity in antivenom research. Toxins (Basel) 2018;10(10):393. doi: 10.3390/toxins10100393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Silva HA, Ryan NM, de Silva HJ. Adverse reactions to snake antivenom, and their prevention and treatment. Br J Clin Pharmacol. 2016;81(3):446–452. doi: 10.1111/bcp.12739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lingam TMC, Tan KY, Tan CH. Proteomics and antivenom immunoprofiling of Russell's viper (Daboia siamensis) venoms from Thailand and Indonesia. J Venom Anim Toxins incl Trop Dis. 2020;26:e20190048-e. doi: 10.1590/1678-9199-JVATITD-2019-0048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan KY, Tan CH, Fung SY, Tan NH. Venomics, lethality and neutralization of Naja kaouthia (monocled cobra) venoms from three different geographical regions of Southeast Asia. J Proteomics. 2015;120:105–125. doi: 10.1016/j.jprot.2015.02.012. [DOI] [PubMed] [Google Scholar]
- Pla D, Sanz L, Quesada-Bernat S, Villalta M, Baal J, Chowdhury MAW, León G, Gutiérrez JM, Kuch U, Calvete JJ. Phylovenomics of Daboia russelii across the Indian subcontinent. Bioactivities and comparative in vivo neutralization and in vitro third-generation antivenomics of antivenoms against venoms from India, Bangladesh and Sri Lanka. J Proteomics. 2019;207:103443. doi: 10.1016/j.jprot.2019.103443. [DOI] [PubMed] [Google Scholar]
- Gutierrez JM, Sanz L, Flores-Diaz M, Figueroa L, Madrigal M, Herrera M, Villalta M, León G, Estrada R, Borges A, Alape-Girón A, Calvete JJ. Impact of regional variation in Bothrops asper snake venom on the design of antivenoms: Integrating antivenomics and neutralization approaches. J Proteome Res. 2010;9(1):564–577. doi: 10.1021/pr9009518. [DOI] [PubMed] [Google Scholar]
- Tu AT, Toom PM, Ganthavorn S. Hemorrhagic and proteolytic activities of Thailand snake venoms. Biochem Pharmacol. 1967;16(11):2125–2130. doi: 10.1016/0006-2952(67)90010-x. [DOI] [PubMed] [Google Scholar]
- Li J, Zhang H, Liu J, Xu K. Novel genes encoding six kinds of three-finger toxins in Ophiophagus hannah (King Cobra) and function characterization of two recombinant long-chain neurotoxins. Biochem J. 2006;398(2):233–242. doi: 10.1042/BJ20060004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajagopalan N, Pung YF, Zhu YZ, Wong PT, Kumar PP, Kini RM. Beta-cardiotoxin: a new three-finger toxin from Ophiophagus hannah (King Cobra) venom with beta-blocker activity. FASEB J. 2007;21(13):3685–3695. doi: 10.1096/fj.07-8658com. [DOI] [PubMed] [Google Scholar]
- Guo XX, Zeng L, Lee WH, Zhang Y, Jin Y. Isolation and cloning of a metalloproteinase from King Cobra snake venom. Toxicon. 2007;49(7):954–965. doi: 10.1016/j.toxicon.2007.01.003. [DOI] [PubMed] [Google Scholar]
- Yamazaki Y, Hyodo F, Morita T. Wide distribution of cysteine-rich secretory proteins in snake venoms: isolation and cloning of novel snake venom cysteine-rich secretory proteins. Arch Biochem Biophys. 2003;412(1):133–141. doi: 10.1016/s0003-9861(03)00028-6. [DOI] [PubMed] [Google Scholar]
- Zhang H-l, Xu S-j, Wang Q-y, Song S-y, Shu Y-y, Lin Z-j. Structure of a cardiotoxic phospholipase A2 from Ophiophagus hannah with the “pancreatic loop”. J Struct Biol. 2002;138(3):207–215. doi: 10.1016/s1047-8477(02)00022-9. [DOI] [PubMed] [Google Scholar]
- Tan NH, Saifuddin MN. Isolation and characterization of an unusual form of L-amino acid oxidase from King Cobra (Ophiophagus hannah) venom. Biochem Int. 1989;19(4):937–944. [PubMed] [Google Scholar]
- He YY, Liu SB, Lee WH, Qian JQ, Zhang Y. Isolation, expression and characterization of a novel dual serine protease inhibitor, OH-TCI, from King Cobra venom. Peptides. 2008;29(10):1692–1699. doi: 10.1016/j.peptides.2008.05.025. [DOI] [PubMed] [Google Scholar]
- Zeng L, Sun Q-Y, Jin Y, Zhang Y, Lee W-H, Zhang Y. Molecular cloning and characterization of a complement-depleting factor from King Cobra, Ophiophagus hannah. Toxicon. 2012;60(3):290–301. doi: 10.1016/j.toxicon.2012.04.344. [DOI] [PubMed] [Google Scholar]
- Pung YF, Wong PT, Kumar PP, Hodgson WC, Kini RM. Ohanin, a novel protein from King Cobra venom, induces hypolocomotion and hyperalgesia in mice. J Biol Chem. 2005;280(13):13137–13147. doi: 10.1074/jbc.M414137200. [DOI] [PubMed] [Google Scholar]
- Petras D, Heiss P, Süssmuth RD, Calvete JJ. Venom proteomics of Indonesian King Cobra, Ophiophagus hannah: integrating top-down and bottom-up approaches. J Proteome Res. 2015;14(6):2539–2556. doi: 10.1021/acs.jproteome.5b00305. [DOI] [PubMed] [Google Scholar]
- Tan CH, Tan KY, Fung SY, Tan NH. Venom-gland transcriptome and venom proteome of the Malaysian King Cobra (Ophiophagus hannah) BMC Genomics. 2015;16(1):687. doi: 10.1186/s12864-015-1828-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu CC, You CH, Wang P-J, Yu JS, Huang GJ, Liu CH, Hsieh WC, Lin CC. Analysis of the efficacy of Taiwanese freeze-dried neurotoxic antivenom against Naja kaouthia, Naja siamensis and Ophiophagus hannah through proteomics and animal model approaches. PLoS Negl Trop Dis. 2017;11(12):e0006138. doi: 10.1371/journal.pntd.0006138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vonk FJ, Casewell NR, Henkel CV, Heimberg AM, Jansen HJ, McCleary RJR, Kerkkamp HME, Vos RA, Guerreiro I, Calvete JJ, Wuster W, Woods AE, Logan JM, Harrison RA, Castoe TA, Jason de Koning AP, Pollock DD, Yandell M, Calderon D, Renjifo C, Currier RB, Salgado D. The King Cobra genome reveals dynamic gene evolution and adaptation in the snake venom system. Proc Natl Acad Sci U S A. 2013;110(51):20651–20656. doi: 10.1073/pnas.1314702110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vejayan J, Khoon TL, Ibrahim H. Comparative analysis of the venom proteome of four important Malaysian snake species. J Venom Anim Toxins incl Trop Dis. 2014;20(1):6. doi: 10.1186/1678-9199-20-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danpaiboon W, Reamtong O, Sookrung N, Seesuay W, Sakolvaree Y, Thanongsaksrikul J, Dong-din-on F, Srimanote P, Thueng-in K, Chaicumpa W. Ophiophagus hannah venom: proteome, components bound by Naja kaouthia antivenin and neutralization by N. kaouthia neurotoxin-specific human ScFv. Toxins (Basel) 2014;6(5):1526–1558. doi: 10.3390/toxins6051526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melani RD, Skinner OS, Fornelli L, Domont GB, Compton PD, Kelleher NL. Mapping proteoforms and protein complexes from King Cobra venom using both denaturing and native top-down proteomics. Mol Cell Proteomics. 2016;15(7):2423–2434. doi: 10.1074/mcp.M115.056523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kunalan S, Othman I, Syed Hassan S, Hodgson WC. Proteomic Characterization of two medically important malaysian snake venoms, Calloselasma rhodostoma (Malayan Pit Viper) and Ophiophagus hannah (King Cobra) Toxins (Basel) 2018;10(11):434. doi: 10.3390/toxins10110434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wongtay P, Sangtanoo P, Sangvanich P, Karnchanatat A. Variation in the protein composition and biological activity of King Cobra (Ophiophagus hannah) venoms. Protein J. 2019;38(5):565–575. doi: 10.1007/s10930-019-09852-5. [DOI] [PubMed] [Google Scholar]
- Tan NH, Wong KY, Tan CH. Venomics of Naja sputatrix, the Javan spitting cobra: A short neurotoxin-driven venom needing improved antivenom neutralization. J Proteomics. 2017;157:18–32. doi: 10.1016/j.jprot.2017.01.018. [DOI] [PubMed] [Google Scholar]
- Wong KY, Tan CH, Tan NH. Venom and purified toxins of the Spectacled Cobra (Naja naja) from Pakistan: Insights into toxicity and antivenom neutralization. Am J Trop Med Hyg. 2016;94(6):1392–1399. doi: 10.4269/ajtmh.15-0871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ranawaka UK, Lalloo DG, de Silva HJ. Neurotoxicity in snakebite--the limits of our knowledge. PLoS Negl Trop Dis. 2013;7(10):e2302. doi: 10.1371/journal.pntd.0002302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kini RM, Doley R. Structure, function and evolution of three-finger toxins: Mini proteins with multiple targets. Toxicon. 2010;56(6):855–867. doi: 10.1016/j.toxicon.2010.07.010. [DOI] [PubMed] [Google Scholar]
- Ogay AY, Rzhevsky DI, Murashev AN, Tsetlin VI, Utkin YN. Weak neurotoxin from Naja kaouthia cobra venom affects haemodynamic regulation by acting on acetylcholine receptors. Toxicon. 2005;45(1):93–99. doi: 10.1016/j.toxicon.2004.09.014. [DOI] [PubMed] [Google Scholar]
- Utkin YN, Kukhtina VV, Kryukova EV, Chiodini F, Bertrand D, Methfessel C, Tsetlin VI. "Weak toxin" from Naja kaouthia is a nontoxic antagonist of alpha 7 and muscle-type nicotinic acetylcholine receptors. J Biol Chem. 2001;276(19):15810–15815. doi: 10.1074/jbc.M100788200. [DOI] [PubMed] [Google Scholar]
- Chong HP, Tan KY, Tan CH. Cytotoxicity of snake venoms and cytotoxins from two Southeast Asian Cobras (Naja sumatrana, Naja kaouthia): Exploration of anticancer potential, selectivity, and cell death mechanism. Front Mol Biosci. 2020;7:583587. doi: 10.3389/fmolb.2020.583587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lertwanakarn T, Suntravat M, Sanchez EE, Boonhoh W, Solaro RJ, Wolska BM, Martin JL, de Tombe PP, Tachampa K. Suppression of cardiomyocyte functions by beta-CTX isolated from the Thai King Cobra (Ophiophagus hannah) venom via an alternative method. J Venom Anim Toxins incl Trop Dis. 2020;26:e20200005. doi: 10.1590/1678-9199-JVATITD-2020-0005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox JW, Serrano SM. Structural considerations of the snake venom metalloproteinases, key members of the M12 reprolysin family of metalloproteinases. Toxicon. 2005;45(8):969–985. doi: 10.1016/j.toxicon.2005.02.012. [DOI] [PubMed] [Google Scholar]
- FS Markland, Jr, Swenson S. Snake venom metalloproteinases. Toxicon. 2013;62:3–18. doi: 10.1016/j.toxicon.2012.09.004. [DOI] [PubMed] [Google Scholar]
- Weissenberg S, Ovadia M, Kochva E. Species specific sensitivity towards the hemorrhagin of Ophiophagus hannah (Elapidae) Toxicon. 1987;25(5):475–481. doi: 10.1016/0041-0101(87)90283-2. [DOI] [PubMed] [Google Scholar]
- Tan NH, Saifuddin MN. Isolation and characterization of a hemorrhagin from the venom of Ophiophagus hannah (King Cobra) Toxicon. 1990;28(4):385–392. doi: 10.1016/0041-0101(90)90076-j. [DOI] [PubMed] [Google Scholar]
- Tan NH, Saifuddin MN. Purification and characterization of two acidic phospholipase A2 enzymes from King Cobra (Ophiophagus hannah) snake venom. Int J Biochem. 1990;22(5):481–487. doi: 10.1016/0020-711x(90)90261-z. [DOI] [PubMed] [Google Scholar]
- Panagides N, Jackson TN, Ikonomopoulou MP, Arbuckle K, Pretzler R, Yang DC, Ali SA, Koludarov I, Dobson J, Sanker B, Asselin A, Santana RC, Hendrikx I, van der Ploeg H, Tai-A-Pin J, van den Bergh R, Kerkkamp HMI, Vonk FJ, Naude A, Strydom MA, Jacobsz L, Dunstan N, Jaeger M, Hodgson WC, Miles J, Fry BG. How the cobra got its flesh-eating venom: Cytotoxicity as a defensive innovation and its co-evolution with hooding, aposematic marking, and spitting. Toxins (Basel) 2017;9(3):103. doi: 10.3390/toxins9030103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cardle L, Dufton MJ. Foci of amino acid residue conservation in the 3D structures of the Kunitz BPTI proteinase inhibitors: How do variants from snake venom differ? Protein Eng. 1997;10(2):131–136. doi: 10.1093/protein/10.2.131. [DOI] [PubMed] [Google Scholar]
- Harvey AL. Twenty years of dendrotoxins. Toxicon. 2001;39(1):15–26. doi: 10.1016/s0041-0101(00)00162-8. [DOI] [PubMed] [Google Scholar]
- Mukherjee AK, Dutta S, Kalita B, Jha DK, Deb P, Mackessy SP. Structural and functional characterization of complex formation between two Kunitz-type serine protease inhibitors from Russell's Viper venom. Biochimie. 2016;(128-129):138–147. doi: 10.1016/j.biochi.2016.08.005. [DOI] [PubMed] [Google Scholar]
- Vogel CW, Fritzinger DC. Cobra venom factor: Structure, function, and humanization for therapeutic complement depletion. Toxicon. 2010;56(7):1198–1222. doi: 10.1016/j.toxicon.2010.04.007. [DOI] [PubMed] [Google Scholar]
- Tan CH, Tan KY, Tan NH. A protein decomplexation strategy in snake venom proteomics. Methods Mol Biol. 2019;1871:83–92. doi: 10.1007/978-1-4939-8814-3_5. [DOI] [PubMed] [Google Scholar]
- Lomonte B, Calvete JJ. Strategies in ‘snake venomics’ aiming at an integrative view of compositional, functional, and immunological characteristics of venoms. J Venom Anim Toxins incl Trop Dis. 2017;23:26. doi: 10.1186/s40409-017-0117-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calvete JJ. Venomics: Integrative venom proteomics and beyond. Biochem J. 2017;474(5):611–634. doi: 10.1042/BCJ20160577. [DOI] [PubMed] [Google Scholar]
- Dutta S, Chanda A, Kalita B, Islam T, Patra A, Mukherjee AK. Proteomic analysis to unravel the complex venom proteome of eastern India Naja naja: Correlation of venom composition with its biochemical and pharmacological properties. J Proteomics. 2017;156:29–39. doi: 10.1016/j.jprot.2016.12.018. [DOI] [PubMed] [Google Scholar]
- Tan NH, Saifuddin MN. Enzymatic and toxic properties of Ophiophagus hannah (King Cobra) venom and venom fractions. Toxicon. 1989;27(6):689–695. doi: 10.1016/0041-0101(89)90020-2. [DOI] [PubMed] [Google Scholar]
- Xh Liu. Rodent biology and management: Current status, opinion and challenges in China. J Integr Agric. 2019;18(4):830–839. [Google Scholar]
- Tan CH, Wong KY, Tan KY, Tan NH. Venom proteome of the yellow-lipped sea krait, Laticauda colubrina from Bali: Insights into subvenomic diversity, venom antigenicity and cross-neutralization by antivenom. J Proteomics. 2017;166:48–58. doi: 10.1016/j.jprot.2017.07.002. [DOI] [PubMed] [Google Scholar]
- Laustsen AH, Karatt-Vellatt A, Masters EW, Arias AS, Pus U, Knudsen C, Oscoz S, Slavny P, Griffiths DT, Luther AM, Leah RA, Lindholm M, Lomonte B, Gutiérrez JM, McCafferty J. In vivo neutralization of dendrotoxin-mediated neurotoxicity of black mamba venom by oligoclonal human IgG antibodies. Nat Commun. 2018;9(1):3928. doi: 10.1038/s41467-018-06086-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laustsen AH, Maria Gutierrez J, Knudsen C, Johansen KH, Bermudez-Mendez E, Cerni FA, Jurgensen JA, Ledsgaard L, Martos-Esteban A, Ohlenschlaeger M, Pus U, Andersen MR, Lomonte B, Engmark M, Pucca MB. Pros and cons of different therapeutic antibody formats for recombinant antivenom development. Toxicon. 2018;146:151–175. doi: 10.1016/j.toxicon.2018.03.004. [DOI] [PubMed] [Google Scholar]
- Ratanabanangkoon K, Tan KY, Pruksaphon K, Klinpayom C, Gutiérrez JM, Quraishi NH, Tan CH. A pan-specific antiserum produced by a novel immunization strategy shows a high spectrum of neutralization against neurotoxic snake venoms. Sci Rep. 2020;10(1):11261. doi: 10.1038/s41598-020-66657-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ratanabanangkoon K, Tan KY, Eursakun S, Tan CH, Simsiriwong P, Pamornsakda T, Wiriyarat W, Klinpayom C, Tan NH. A simple and novel strategy for the production of a pan-specific antiserum against elapid snakes of Asia. PLoS Negl Trop Dis. 2016;10(4):e0004565-e. doi: 10.1371/journal.pntd.0004565. [DOI] [PMC free article] [PubMed] [Google Scholar]


