Abstract
Low‐dose methotrexate (MTX) is an immunosuppressant used to treat inflammatory bowel disease (IBD). SLCO1B1 genetic variation has been associated with delayed MTX clearance and increased toxicity. The purpose of this study was to evaluate the association between SLCO1B1 genetic variation and MTX‐induced nausea in children with IBD. We performed a single center retrospective chart analysis of 278 patients who were prescribed MTX for IBD. Two hundred two patients had banked DNA and were genotyped for three SLCO1B1 single nucleotide polymorphisms (SNPs; rs4149056, rs2306283, and rs11045819). Diplotypes were determined by combining the SNPs into *1, *4, *5, *14, *15, and *37 alleles. Incidence of nausea was abstracted from clinician notes. Prescriptions and demographics were extracted from the medical record. The cohort was 69.8% boys, 89.1% White, and 87.6% had a diagnosis of Crohn’s disease with a mean age of 16.0 (± 3.8) years. MTX‐induced nausea was noted in 34% of the cohort. MTX‐induced nausea was associated with the number of reduced‐function *15 alleles (p = 0.034) and occurred 2.26 times more often in patients with at least one *15 allele who did not initiate MTX treatment with concomitant ondansetron (p = 0.034). MTX‐induced nausea was significantly independently associated with SLCO1B1 diplotype (p = 0.006) after controlling for MTX dose group and concomitant ondansetron. Our data demonstrate that the SLCO1B1 *15 allele is associated with MTX‐induced nausea in pediatric patients with IBD. Additionally, *15 allele carriers could benefit from a dose reduction of MTX to reduce exposure and treatment initiation with concomitant ondansetron to reduce nausea.
Study Highlights.
WHAT IS THE CURRENT KNOWLEDGE ON THE TOPIC?
Low‐dose methotrexate (MTX) is commonly used for treatment of inflammatory bowel disease (IBD); however, it is well known by patients for having a potent side effect profile, characterized as MTX intolerance, which most notably includes nausea.
WHAT QUESTION DID THIS STUDY ADDRESS?
This study investigated the biological basis of MTX intolerance by testing the association between alleles of the MTX transporter SLCO1B1 and MTX intolerance.
WHAT DOES THIS STUDY ADD TO OUR KNOWLEDGE?
These results significantly add to pharmacogenetics literature on immune‐mediated disorders, because there are no existing studies investigating the relationship between dosing and response of MTX in pediatric IBD populations. Specifically, our paper provides evidence that pediatric patients with IBD that carry a variant allele of the MTX transporter SLCO1B1 have higher rates of MTX‐induced nausea.
HOW MIGHT THIS CHANGE CLINICAL PHARMACOLOGY OR TRANSLATIONAL SCIENCE?
Patients with this common allele may benefit from lower doses of MTX and concomitant prescription of an antiemetic, such as ondansetron. This study provides a rationale for the use genotype‐guided dosing for MTX in pediatric IBD populations.
INTRODUCTION
The inflammatory bowel diseases (IBDs), including ulcerative colitis (UC) and Crohn’s disease (CD), are disorders of chronic intestinal inflammation caused by a dysregulated immune response against bacterial flora. 1 Low‐dose methotrexate (MTX) is commonly used as an immunomodulator in treating autoimmune diseases, including rheumatoid arthritis and IBD. In the pediatric IBD population, MTX is often prescribed concomitantly with antitumor necrosis factor‐alpha (anti‐TNFα) treatment 2 because it suppresses anti‐drug antibody formation, preserving exposure to anti‐TNFα biologics. 3
There is substantial interindividual variation in the clearance of MTX in pediatric patients with leukemia. A genomewide association study revealed that inherited single nucleotide polymorphisms (SNPs) in SLCO1B1 are the most important genetic variations associated with clearance of high‐dose MTX. 4 SLCO1B1 encodes OATP1B1, an organic anion transporter responsible for the hepatic uptake of MTX. 5 Over 10% of the pharmacokinetic variability in high‐dose MTX clearance can be attributed to SLCO1B1 variants. 6 One or more SNPs make up a haplotype (named as a * allele), and two haplotypes combine to give a diplotype. The most widely studied variant in SLCO1B1 is rs4149056 (c. 521T>C, V174A), an SNP common to two reduced function alleles, *15 and *5. The rs4149056 variant decreases membrane localization and activity of the transporter, 7 resulting in slower MTX clearance. 8 The *5 and *15 alleles resulted in higher systemic exposure in patients with leukemia, 6 and these alleles are also associated with severe toxicities with irinotecan chemotherapy 9 and statin‐induced myopathy. 10 This variant seems to have more impact on simvastatin pharmacokinetics in children than in adults, 11 indicating there may be higher expression or activity in children. It has also been associated with an increased frequency of gastrointestinal toxicity and reduced frequency of hepatotoxicity in patients with juvenile systemic arthritis (JIA). 12 Meanwhile, the *14 allele, made up of two other coding SNPs (rs2306283 and rs11045819), is associated with increased expression, 13 increased function, and faster clearance of MTX 6 ; because clearance of MTX is negatively correlated to drug exposure, this allele is associated with poor response to MTX in children with arthritis. 14 Wildtype alleles (*1 and *37) are associated with normal clearance.
In addition to the MTX pharmacokinetic variability described above, there is variability in side effects to low‐dose MTX that are dose‐dependent. MTX intolerance is defined as gastrointestinal and behavioral symptoms occurring before or after MTX administration that may lead to treatment discontinuation and can be evaluated with a standardized questionnaire. 15 Among pediatric patients with IBD, 30–50% of patients reported symptoms of MTX intolerance, including adverse effects before MTX intake (anticipatory nausea) and when thinking of MTX (associative symptoms). 16 Associative symptoms of MTX arise through a classical conditioned response to nausea or vomiting after MTX administration. Prophylactic prescription of anti‐emetics did not prevent symptoms of MTX intolerance. 16 MTX intolerance was reported as early as the first published study to treat patients with IBD with MTX in 1998, where two of 14 patients reported nausea, leading to withdrawal of therapy, and is still being reported today, more than 25 years later. 17
Although MTX intolerance in pediatric IBD has been well‐described, the biologic causes or predictors of MTX intolerance in pediatric IBD are not known. The purpose of this study is to evaluate whether SLCO1B1 genetic variation is associated with MTX adverse effects, including nausea. The results of this study could contribute to guidelines for personalized dosing of MTX. Identifying genetic predictors of response will help clinicians optimize treatment and ultimately reduce the burden of disease. We hypothesize that variants in SLCO1B1 that are associated with increased systemic exposure to MTX will be associated with adverse effects.
METHODS
Study design and participants
We conducted a retrospective cohort study of MTX intolerance in pediatric patients with IBD. This study was approved by the Institutional Review Board at Cincinnati Children’s Hospital Medical Center (CCHMC). DNA samples were provided by the CCHMC Discover Together Biobank, a College of American Pathology accredited biorepository in which patients voluntarily consented to their DNA being used for CCHMC research studies. An initial electronic medical record query identified 316 pediatric patients with IBD treated with MTX at CCHMC from January 2010 to November 2018. The inclusion criteria were: indication for MTX was IBD (CD, UC, and IBD‐unclassified), oral or subcutaneous routes of MTX administration, and a dosage of less than or equal to 25 mg/week. 18 After exclusions, 202 patients with banked DNA and genotypes were included in the study cohort (Figure S1). The study was designed to have 80% power to detect an effect size of 2.0 at an alpha = 0.05 for MTX‐induced nausea.
MTX intolerance
Symptoms of MTX intolerance, including abdominal pain, nausea, vomiting, sore mouth, and behavioral symptoms, before, during, or after taking MTX were manually abstracted from the medical records, and were only included as positive when the clinician directly attributed the symptom to MTX. 8 Medical records were also reviewed for prescriptions (MTX, ondansetron, and metronidazole), demographics, diagnosis, indication, and reason for MTX discontinuation. Due to the small number of patients discontinuing MTX due to liver function enzyme increases (n = 7), diarrhea (n = 2), vomiting (n = 2), and abdominal pain (n = 0), we were not able to analyze any associations with SLCO1B1. CCHMC institutional guidelines recommend a weekly dose of 15 mg/m2 capped at 25 mg for patients taking MTX as a monotherapy and less than or equal to 10 mg/m2 capped at 15 mg for patients taking MTX as a combination therapy with a biologic medication. Therefore, we created two dose groups based on these recommendations. Patients that did not fit either of these criteria were included in the “other” dose group. Because subcutaneous administration provides higher MTX exposure than the same dose given orally, oral doses were converted to equivalent subcutaneous doses based on established dose conversions. 19 Metronidazole was included as it is an antibiotic used in IBD treatment for which nausea is a common side effect; 125 patients received metronidazole (Table 1). Ondansetron was included because it is an anti‐emetic commonly prescribed to patients to prevent nausea (n = 40) or after development of MTX‐induced nausea (n = 37). 20 Data on concomitant inhibitors/substrates of OATP1B1 were not collected (e.g., rifampin and statins). We were not reliably able to estimate medication adherence. The primary end point was nausea while on MTX and the secondary end point was discontinuation of MTX.
TABLE 1.
Patient demographics, methotrexate dosing, and SLCO1B1 alleles
| Parameters |
Values (n = 202) n (%) or median (range) |
|---|---|
| Age (years) | 16.0 (2.6–24.0) |
| Sex (male) | 141 (69.8%) |
| Race | |
| White | 180 (89.1%) |
| Black | 16 (7.9%) |
| Other | 6 (3.0%) |
| Diagnosis | |
| Crohn’s disease | 178 (87.6%) |
| Ulcerative colitis | 21 (10.4%) |
| Indeterminate colitis | 4 (2.0%) |
| Routes of MTX administration | |
| Oral | 122 (60.1%) |
| Subcutaneous | 81 (39.9%) |
| MTX Dose (mg) | 25 (4.5–25) |
| Capped at 10 mg | 7 (3.5%) |
| 10 mg/m2 | 20 (9.9%) |
| Capped at 15 mg | 31 (15.3%) |
| 15 mg/m2 | 56 (27.7%) |
| Capped at 25 mg | 80 (39.6%) |
| Other dose | 8 (4.0%) |
| MTX intolerance | 105 (52.0%) |
| MTX‐induced nausea | 69 (34.2%) |
| MTX Discontinued | 108 (53.5%) |
| Metronidazole prescribed | 125 (61.6%) |
| SLCO1B1 alleles | n = 404, n (%) |
|---|---|
| *1 (A‐C‐T) | 209 (51.7%) |
| *4 (A‐A‐T) | 0 (0%) |
| *5 (A‐C‐C) | 8 (4.0%) |
| *14 (G‐A‐T) | 59 (14.6%) |
| *15 (G‐C‐C) | 50 (12.4%) |
| *37 (G‐C‐T) | 69 (17.1%) |
Abbreviation: MTX, methotrexate.
Genotyping
Genotyping of three SNPs in SLCO1B1 (rs4149056, rs2306283, and rs11045819) was performed using TaqMan assays (Applied Biosystems, Beverly Hills, CA). 14 All genotypes were determined simultaneously and verified in replicate with a failure rate of 15.48% (n = 37/239), where at least one SNP failed genotyping in 37 samples. The observed genotype frequencies were consistent with Hardy‐Weinberg equilibrium (p > 0.1 for each SNP).
A patient’s SLCO1B1 alleles were determined by these three SNPs into *1 (wildtype), *4 (rs11045819), *5 (rs4149056), *14 (rs2306283 + rs11045819), *15 (rs2306283 + rs4149056), and *37 (rs2306283; Table S1), and each patient then has a combination of two haplotypes/alleles to make a diplotype. PharmVar is currently reviewing allele nomenclature for SLCO1B1 and has changed the names of alleles that were formerly known as *1a and *1b to *1 and *37, respectively.
Statistical methods
A χ2 test for trend was used to analyze the dose‐dependent effect of decreased function SLCO1B1 *15 alleles and incidence of nausea. A Fisher’s exact test was used for the association between concomitant ondansetron at MTX initiation and the frequency of nausea. A Mann‐Whitney U test was applied to compare the subcutaneous‐equivalent MTX dose between those with and without nausea. Diplotypes were ordered based on their relative association with MTX clearance 6 for ordinal analysis using a multivariable general linear model testing the relationship between the frequency of nausea and SLCO1B1 diplotypes, including dose group and ondansetron use as covariates. A type 1 error rate of less than 0.05 was considered statistically significant. Statistical analyses were completed using GraphPad Prism 8.0.1 and R 4.0.0.
RESULTS
Baseline characteristics and outcomes
The cohort was 69.8% boys, 89.1% White, and 87.6% had a diagnosis of CD with a mean age of 16.0 (± 3.8) years (Table 1). MTX was taken orally by 60.4% of patients and was prescribed at 15 mg/m2/week (dose capped at 25 mg) in 67.3% of patients and at 10 mg/m2/week (dose capped at 15 mg) in 25.2% of patients. Approximately half of the patients (n = 105/202) experienced MTX intolerance, 34.2% experienced MTX‐induced nausea, and 53.5% of patients discontinued MTX. All discontinuations were due to nausea and/or elevation of liver function enzymes. The subcutaneous equivalent dose of MTX was significantly higher in patients with nausea (n = 69) compared with patients without nausea (20.0 mg vs. 15.1 mg, p < 0.0001, Mann‐Whitney U test).
Association of SLCO1B1 genetic variation with MTX‐induced nausea and discontinuation
The number of decreased‐function SLCO1B1*15 alleles was tested for association with MTX intolerance and MTX‐induced nausea. Patients with at least one *15 allele reported a higher frequency of nausea (p = 0.034; Figure 1a) and four of five patients with two *15 alleles reported nausea.
FIGURE 1.
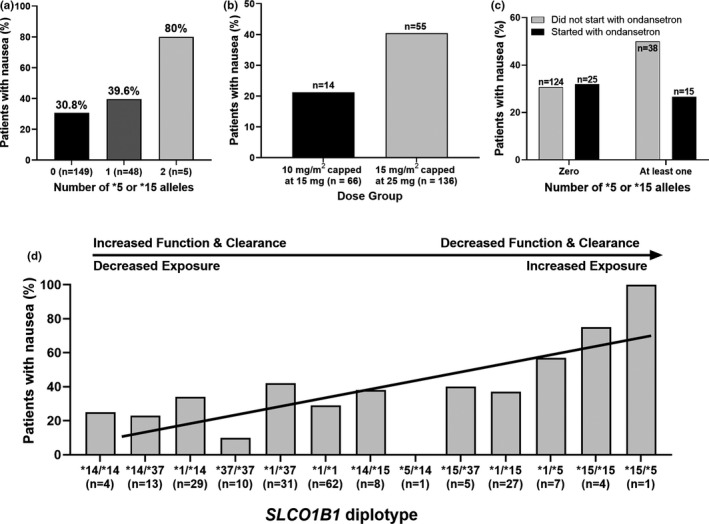
(a) MTX‐induced nausea is dependent upon the number of reduced‐function SLCO1B1*15 alleles (χ2 for trend, p = 0.034). (b) The frequency of nausea was greater in the 15 mg/m2 capped at 25 mg dose group than the less than or equal to 10 mg/m2 capped at 15 mg dose group (χ2 test with Yates’ correction, p = 0.011). (c) Concurrent use of ondansetron reduced nausea in patients with at least one SLCO1B1*15 allele (Fisher’s exact test, p = 0.14). (d) SLCO1B1 diplotype is associated with increased frequency of nausea (multivariable general linear model, p = 0.006). The diplotypes are ordered according to the predicted activity from a cohort treated with high‐dose MTX. 6 Data are adjusted for MTX dose group (p = 0.0002) and whether treatment was initiated with concurrent ondansetron (p = 0.441). Data fitted by multivariable general linear model. MTX, methotrexate
Patients taking 15 mg/m2 capped at 25 mg had a 2.5 odds ratio of developing nausea compared with patients taking less than or equal to 10 mg/m2 capped at 15 mg (p = 0.011, χ2 test with Yates’ correction, Figure 1b). There was no difference in the dose group representation between the zero *15 alleles and at least one *15 allele.
Frequency of nausea was independent of whether treatment was initiated with ondansetron (p = 0.66, χ2 test with Yates’ correction). In our cohort, 35% (n = 57/162) of patients that did not initiate with ondansetron experienced nausea and 30% (n = 12/40) of patients that did initiate with ondansetron experienced nausea. Ondansetron reduced the frequency of reported nausea from 50% to 26.6% in *15 allele carriers, which was comparable to the frequency of reported nausea in noncarriers, although this difference was not significant (p = 0.14; Figure 1c). The route of administration (oral vs. subcutaneous) and concomitant prescription of metronidazole were not significantly associated with intolerance or nausea (all p > 0.3, Fisher’s exact test).
SLCO1B1 diplotype was associated with increased frequency of nausea (p = 0.006, multivariable general linear model; Figure 1d) when adjusted for MTX dose group (p = 0.0002, multivariable general linear model) and treatment initiation with concurrent ondansetron (p = 0.44, multivariable general linear model) to prevent nausea. There was no association between SLCO1B1 diplotype and discontinuation of MTX or MTX intolerance (both p > 0.5, univariable general linear model).
DISCUSSION
This is the largest study that investigates the association between the SLCO1B1*15 allele and increased MTX‐induced nausea in patients with IBD. In addition to the observation that MTX‐induced nausea was more common in pediatric patients carrying SLCO1B1*15 alleles, patients taking higher doses of MTX were more likely to have nausea than patients taking lower doses of MTX. Initiation with ondansetron did not prevent nausea; however, it may help reduce the frequency of nausea in *15 allele carriers.
The 34.2% rate of MTX‐induced nausea observed in this study was slightly less than previous studies demonstrating 52.0% in pediatric patients with IBD 16 and 64% in pediatric patients with JIA. 21 One study of low‐dose MTX in patients with JIA found that the mean MTX polyglutamate levels were 1.4 times higher in patients who reported nausea compared with those who did not have nausea, indicating a possible exposure‐response relationship, 22 and we found patients receiving higher doses were more likely to report nausea. We likely found a reduced rate of nausea compared with the other prospective studies because of the retrospective design of our study. There are several other genes encoding transporters and enzymes in the folate pathway that may contribute to MTX‐induced nausea. Kyvsgaard et al recently found that the rs4149056 SNP in the SLCO1B1*5 and *15 alleles was not associated with MTX‐induced nausea in a sample of 121 pediatric patients with JIA, but did find an association with the rs1801133 SNP in MTHFR that we did not investigate in this study. 23
Our sample size of 202 patients is one of the largest for a retrospective cohort study of MTX intolerance. However, there are some limitations to this study. Reported side effects were abstracted from physician documented notes, which may have resulted in under‐reporting of these frequencies in our cohort. Adherence to MTX, ondansetron, and folic acid supplements have been associated with reducing the incidence of nausea, hepatic dysfunction, and MTX discontinutation, 24 but given the lack of a prospective questionnaire, we were not able to systematically evaluate MTX intolerance or adherence to these medications in our cohort. MTX was prescribed as a monotherapy or in combination therapy with a biologic medication; heterogeneity in the indication for MTX and lack of standardized follow‐up did not allow for assessment of disease response.
In conclusion, this study establishes an association between MTX‐induced nausea and a patient’s SLCO1B1 genotype. Moreover, we found that patients with at least one *15 allele could benefit from initiating MTX treatment with concurrent ondansetron to reduce nausea. Further investigation of pharmacodynamic measurements, such as MTX polyglutamates, in relation to SLCO1B1*15 allele status, may lend additional support to the hypothesis of slower clearance, higher systemic exposure, and more formation of polyglutamates being associated with nausea. A clinical trial of allele‐guided dosing is warranted; patients carrying the *15 allele may benefit from a lower dose than noncarriers to avoid MTX‐induced nausea, without compromising efficacy. There are opportunities to personalize dosing and supportive care based on SLCO1B1 genotype—there are currently no dosing recommendations for this gene‐drug pair.
CONFLICT OF INTEREST
L.B.R. has received research funding from the National Institutes of Health (NICHD) and BTG International. All other authors declared no competing interests for this work.
AUTHOR CONTRIBUTIONS
R.S.M., Z.L.T., L.J.M., M.J.R., and L.B.R. wrote the manuscript. R.S.M., M.J.R., and L.B.R. designed the research. R.S.M. performed the research. Z.L.T., L.J.M., and L.B.R. analyzed the data.
Supporting information
Supplementary Material
ACKNOWLEDGEMENTS
This study used samples, data, and services from the Discover Together Biobank at Cincinnati Children’s Research Foundation. We thank the Discover Together Biobank for support of this study, as well as participants and their families, whose help and participation made this work possible. We appreciate the data warehouse extraction performed by Parth Divekar and genotyping by Dr. Marc Sudman.
Mehta RS, Taylor ZL, Martin LJ, Rosen MJ, Ramsey LB. SLCO1B1 *15 allele is associated with methotrexate‐induced nausea in pediatric patients with inflammatory bowel disease. Clin Transl Sci. 2022;15:63–69. 10.1111/cts.13130
Funding information
This project was financially supported by institutional funding from Cincinnati Children’s Hospital Medical Center.
REFERENCES
- 1. Chang JT. Pathophysiology of inflammatory bowel diseases. N Engl J Med. 2020;383:2652‐2664. [DOI] [PubMed] [Google Scholar]
- 2. Herfarth HH, Kappelman MD, Long MD, Isaacs KL. Use of methotrexate in the treatment of inflammatory bowel diseases. Inflamm Bowel Dis. 2016;22:224‐233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Vermeire S, Noman M, Van Assche G, Baert F, D’Haens G, Rutgeerts P. Effectiveness of concomitant immunosuppressive therapy in suppressing the formation of antibodies to infliximab in Crohn’s disease. Gut. 2007;56:1226‐1231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Ramsey LB, Panetta JC, Smith C, et al. Genome‐wide study of methotrexate clearance replicates SLCO1B1. Blood. 2013;121:898‐904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Niemi M, Pasanen MK, Neuvonen PJ. Organic anion transporting polypeptide 1B1: A genetically polymorphic transporter of major importance for hepatic drug uptake. Pharmacol Rev. 2011;63:157‐181. [DOI] [PubMed] [Google Scholar]
- 6. Ramsey LB, Bruun GH, Yang W, et al. Rare versus common variants in pharmacogenetics: SLCO1B1 variation and methotrexate disposition. Genome Res. 2012;22:1‐8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Grant DM. Pharmacogenetics. In Fetal Neonatal Physiol. Elsevier; 2017: 222‐229. [Google Scholar]
- 8. Treviño LR, Shimasaki N, Yang W, et al. Germline genetic variation in an organic anion transporter polypeptide associated with methotrexate pharmacokinetics and clinical effects. J Clin Oncol. 2009;27:5972‐5978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Takane H, Miyata M, Burioka N, et al. Severe toxicities after irinotecan‐based chemotherapy in a patient with lung cancer: A homozygote for the SLCO1B1*15 allele. Ther Drug Monit. 2007;29:666‐668. [DOI] [PubMed] [Google Scholar]
- 10. Romaine SPR, Bailey KM, Hall AS, Balmforth AJ. The influence of SLCO1B1 (OATP1B1) gene polymorphisms on response to statin therapy. Pharmacogenomics J. 2010;10:1‐11. [DOI] [PubMed] [Google Scholar]
- 11. Wagner JB, Abdel‐Rahman S, Van Haandel L, et al. Impact of SLCO1B1 genotype on pediatric simvastatin acid pharmacokinetics. J Clin Pharmacol. 2018;58:823‐833. [DOI] [PubMed] [Google Scholar]
- 12. Roszkiewicz J, Michałek D, Ryk A, Swacha Z, Szmyd B, Smolewska E. SLCO1B1 variants as predictors of methotrexate‐related toxicity in children with juvenile idiopathic arthritis. Scand J Rheumatol. 2021;50:213‐217. [DOI] [PubMed] [Google Scholar]
- 13. Nies AT, Niemi M, Burk O, et al. Genetics is a major determinant of expression of the human hepatic uptake transporter OATP1B1, but not of OATP1B3 and OATP2B1. Genome Med. 2013;5(1):1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Ramsey LB, Moncrieffe H, Smith CN, et al. Association of SLCO1B1*14 allele with poor response to methotrexate in juvenile idiopathic arthritis patients. ACR Open Rheumatol. 2019;1:58‐62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Ćalasan MB, van den Bosch OF, Creemers MC, et al. Prevalence of methotrexate intolerance in rheumatoid arthritis and psoriatic arthritis. Arthritis Res Ther. 2013;15:1‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Dupont‐Lucas C, Grandjean‐Blanchet C, Leduc B, et al. Prevalence and risk factors for symptoms of methotrexate intolerance in pediatric inflammatory bowel disease. Inflamm Bowel Dis. 2017;23:298‐303. [DOI] [PubMed] [Google Scholar]
- 17. Egan LJ, Sandborn WJ. Methotrexate for inflammatory bowel disease: Pharmacology and preliminary results. Mayo Clin Proc. 1996;71:69‐80. [DOI] [PubMed] [Google Scholar]
- 18. Colman RJ, Rubin DT. Optimal doses of methotrexate combined with anti‐TNF therapy to maintain clinical remission in inflammatory bowel disease. J. Crohn’s Colitis. 2015;9:312‐317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Schiff MH, Sadowski P. Oral to subcutaneous methotrexate dose‐conversion strategy in the treatment of rheumatoid arthritis. Rheumatol Int. 2017;37:213‐218. [DOI] [PubMed] [Google Scholar]
- 20. Kempinska A, Benchimol EI, Mack A, Barkey J, Boland M, Mack DR. Short‐course ondansetron for the prevention of methotrexate‐induced nausea in children with Crohn disease. J Pediatr Gastroenterol Nutr. 2011;53:389‐393. [DOI] [PubMed] [Google Scholar]
- 21. Falvey S, Shipman L, Ilowite N, Beukelman T. Methotrexate‐induced nausea in the treatment of juvenile idiopathic arthritis. Pediatr. Rheumatol. 2017;15:52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Hawwa AF, AlBawab A, Rooney M, Wedderburn LR, Beresford MW, McElnay JC. Methotrexate polyglutamates as a potential marker of adherence to long‐term therapy in children with juvenile idiopathic arthritis and juvenile dermatomyositis: An observational, cross‐sectional study. Arthritis Res Ther. 2015;17:295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Kyvsgaard N, Mikkelsen TS, Als TD, Christensen AE, Corydon TJ, Herlin T. Single nucleotide polymorphisms associated with methotrexate‐induced nausea in juvenile idiopathic arthritis. Pediatr. Rheumatol. 2021;19:51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Shea B, Swinden MV, Tanjong Ghogomu E, et al. Folic acid and folinic acid for reducing side effects in patients receiving methotrexate for rheumatoid arthritis. Cochrane Database Syst Rev. 2013;41(6):1049‐1060. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Material


