Abstract
Soybeans [Glycine max (L.)] are a good source of isoflavones. The main isoflavone components of soybean are daidzein, genistein, and glycitein. World soybean production is very high. Because of its pharmacological activity, soy isoflavone intake over a long period of time may result in interactions with the drugs. This review summarizes soy isoflavone-drug interactions based on the pharmacokinetic parameters. Soy isoflavones have pharmacokinetic interactions with celecoxib, theophylline, paclitaxel, midazolam, imatinib, carbamazepine, valproic acid, repaglinide, omeprazole and danofloxacin. This is due to the changes in the area under the curve, maximum serum concentration, time that a drug is present at the maximum concentration in serum, clearance and half-life of the drugs when delivered together with soy isoflavones. The mechanisms of pharmacokinetic interactions occurs through the inhibition/induction of drug metabolizing cytochrome P450 (CYP450) enzymes such as CYP3A4, CYP2A1, and CYP2C9 or through the inhibition of drug transporters such as P-glycoprotein and breast cancer resistance protein. Thus, the consumption of soybean, soy isoflavones or soy products with drugs needs to be reconsidered.
Keywords: Soybean, isoflavones, pharmacokinetic ınteraction, drug metabolizing enzyme, drug transporter
INTRODUCTION
Soybeans [Glycine max (L.)] are a source of isoflavones in the daily meals. In 2016, global soybean production amounted to be 34,894,085 tons; with 293,414,006 tons from the Americas, 28,808,950 tons from Asia, 10,488,759 tons from Europe, and 2,119,814 tons from Africa. In 2016, total of 89.05% of soybean production was from five countries: India (4.18%), China (3.57%), Argentina (17.56%), Brazil (28.75%), and the USA (34.99%).1Soybeans contain non-steroidal polyphenol compounds2 with a chemical structure similar to that of oestradiol-17b, so these compounds may have a similar effect to that of the estrogen.3,4 The main isoflavone content of soybean is in aglycone form, including genistein, daidzein, and glycitein; the glycosidic forms are genistin, daidzin, and glycitin, which are precursors of the metabolic process which forms daidzein and genistein aglycones.5 The total glycitein and glycoside content in soybeans is only 5-10% of the total isoflavones, while the remaining is comprised of daidzein and genistein.6 Isoflavones have effects on postmenopausal nutrition,7 relief of postmenopausal vasomotor symptoms,8 osteoporosis,9 inflammation,10 and cardiovascular disease.11 The compounds also have antioxidant activity,12 increase the efficacy of cancer therapy,13 and inhibit the cancer cell proliferation.14
Based on this pharmacological activity, soy isoflavones could be used as a dietary nutrition over a long period of time. Soybean consumption continued to increase in 2011.15 Fonseca et al.16 showed that the amount of isoflavones taken in by the infants fed with soy-based formula is 0.8 mg/day/kg of body weight; this number is two-fold higher than the level of isoflavones consumed by the adults in Japan. The daily intake of isoflavones is related to how much soy is consumed and differs in each country, [i.e., it is much higher in east and south Asian countries (20-50 mg/day), than in Europe (0.49-1 mg/day)].17,18 To fulfill the daily nutrient needs, the Chinese government has recommended that every citizen consumes 50 mg of soy food daily. Simple processed soy foods from Asia usually contain 3.5 mg of isoflavones in every gram. Large studies performed in the United States showed that each adult there consumes 2.5 mg of isoflavones per day, but other research data shows different results where the consumption of isoflavones per day may reach the range of 30-50 mg. In China, the average daily consumption of isoflavones is 40.8±28.7 mg/day.19
Isoflavone consumption patterns in this community therefore raise the possibility of drug interactions when used together, so their use must be monitored. Drug interactions occur when other substances affect the activity of a drug.20 These interactions may occur with the soy isoflavones. Soy extracts, soy products, and soy isoflavones have interactions with the drugs such as: Warfarin,21 tamoxifen,22 levodopa,23 and ciprofloxacin.24 The mechanism of the drug-isoflavone interaction is by the inhibition or induction of drug metabolizing enzymes (DMEs) or drug transporters.25
Almost all the drug biotransformation reactions need a metabolic enzyme, and the enzymes most often used to process the drugs are the liver microsomal cytochrome P450 (CYP450) enzymes. The CYP enzymes involved in the drug metabolism are CYP2C9, CYP2C19, CYP2D6, CYP3A4, and CYP3A5.26 Drugs or bioactive compounds such as isoflavonoids interact with these enzymes, and change the efficacy and action of the drug.27 Soybean products (infusions) have an inhibitory effect on human CYP enzymes, including CYP2C9, CYP2C19, CYP3A4, and CYP2D6.28 It has also been reported that soy isoflavones reduce the hepatic CYP2E1 and CYP3A activities related to acetaminophen metabolism.29
Furthermore, drug transporters could be involved in the drug interactions, because drug transporters mediate the absorption, distribution, and excretion of the drugs in the transport process across the plasma membrane.30 There are two classifications of these drug transporters: The ATP-binding cassette (ABC) family and the solute carrier (SLC) family. P-glycoprotein (P-gp) is a member of the ABC family, and could be induced by various factors, including clinical drugs, environmental xenobiotics, and dietary compounds;31 which is known to be involved in the drug interactions. There are reports that genistein from soy inhibits the efflux of the P-gp substrates cimetidine32 and paclitaxel.33The efflux of vinblastine in KB-V1 cells highly expressing P-gp, and the P-gp substrate paclitaxel could be inhibited by genistein at some doses.34 In addition to the P-gp, interactions may occur through other drug transporters. So, DMEs and drug transporters play important roles in the absorption, distribution, metabolism, and the excretion (ADME) of the drugs, and are involved in the interactions that will affect the pharmacokinetics and pharmacodynamics of the drugs.
These pharmacokinetic interactions could be seen by assessing the pharmacokinetic parameters including the area under the curve (AUC), maximum concentration (Cmax), volume of distribution (Vd), half-life (t1/2), and clearance. Nagashima et al.23 found that soybean increases the AUC of levodopa. Soybean also reduces the AUC and Cmax of losartan.35 These differences in pharmacokinetic parameters depend on the mechanism. Until now, there has been no summary to explain how soy isoflavones could affect the pharmacokinetic profile of a drug and the mechanisms involved. This is needed as a reference regarding the safety of using soy isoflavones as daily nutrients with the co-administration of the drugs.
Methods
This review is based on the literature collected from the internet through Google Scholar, Elsevier, PubMed, and NCBI, using the keywords soybean, soy products, soy isoflavones, soy drug interaction, isoflavone content, daidzein, genistein, isoflavone interaction, pharmacokinetic parameter, and the pharmacokinetic interaction. In total, 181 articles were collected, but only 99 articles were included based on the inclusion criteria. The inclusion criteria were articles with a publication year before 2000, containing a description of pharmacokinetic parameter values, describing interactions with soybeans, containing isoflavone content data, or related to isoflavones, soybeans, and pharmacokinetic interactions. The flowchart of the search is illustrated in Figure 1.
Figure 1.
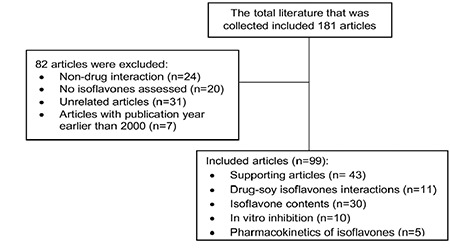
Flow chart of the literature review
Soy isoflavones
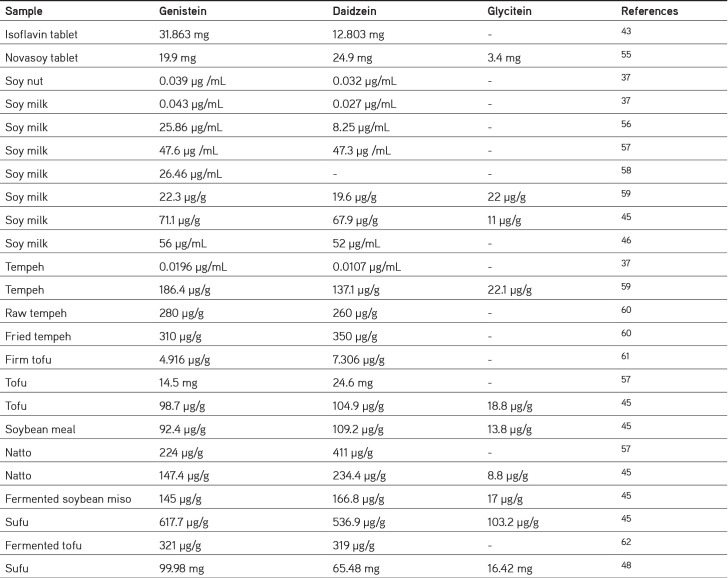
Isoflavones are bioactive metabolites and include a group of phytoestrogens. Isoflavones have structures like those of mammalian estrogens. The largest source of isoflavones is soybean. Soy isoflavones are present in 12 different isoforms, divided into four chemical forms: Acetylglucoside (acetylgenistin, acetylglycitin, acetyldaidzin), malonylglucoside (malonylgenistin, malonyldaidzin, malonylglycitin), glucoside (genistein, daidzin, and glycitin), and aglycone (genistein, daidzein, and glycitein).36 After the metabolism process in the human gut, glucoside isoflavones become aglycones through the effect of gastrointestinal enzymes.5 Genistein, daidzein, and glycitein comprise approximately 50%, 40%, and 10% of the isoflavones in soybean.37 The isoflavone content is influenced by several factors; in this article, we summarize the content of genistein, daidzein, and glycitein in soybeans, as seen in Table 1.37,38,39,40,41,42,43,44,45,46,47 The amount of isoflavones is in the order genistein > daidzein > glycitein, and the content of the glycoside form is lower than that of the aglycone form; differences arise based on the variety, location of the production, humidity etc.38 Sources of isoflavones include soy products such as traditional soy foods (such as tofu and soy milk), isolated soy protein, soybean paste, soy flakes, soy flour, fermented soybean products (such as tempeh, miso, and natto), and soy sauce.39
Table 1. Summary of isoflavone contents in soybean.
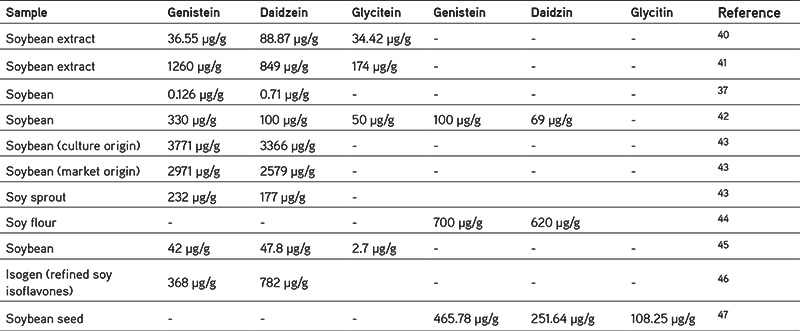
In each type of soybean product containing different soy isoflavones, we summarize the isoflavone content focusing only on the aglycone form, (i.e., genistein, daidzein, and glycitein in various soy products from the several studies, presented in Table 2).48,49,50,51,52,53,54,55,56,57,58,59,60,61,62 It appears that soy tablets commercially contain the highest levels of isoflavones, because soy tablets are usually used as additional nutrients so the soy isoflavone content is adjusted to nutritional requirements. Of the soy products shown Table 2, sufu has the highest content compared to others. Sufu is a traditional food from China, and it is made of fermented soybean curd.48 Other fermented foods that also have high soy isoflavones content are natto, tempeh, and miso. The fermentation process influences the isoflavone content. Fermentation can increase aglycone isoflavones from black soybean pulp49 in tempeh and tofu.50,51 Another study reported a 75% increase in aglycone isoflavones in soybean flour after fermentation.52 The fermentation process is also influenced by the several factors such as time and temperature.53,54
Table 2. Isoflavone contents of soybean products.
The differences in the isoflavone content of soybean products also leads to variations in the pharmacokinetic profile of isoflavones, as presented in Table 3. There are variations in the different levels, caused by many factors, [i.e., differences in the test subjects used (human, rats, or mice), variations in the age, the hydrolysis process of glycosides by the gut bacteria or gut wall enzymes, uptake, ethnicity, etc].44 The content of daidzein and genistein in soy products depends on the raw material and the conditions while processing, Faughnan et al.63 found that urinary recovery of equol from tempeh is higher than the soymilk, although the solid food matrix and fermentation may increase the production of equol. Equol is a metabolite of daidzein produced by the intestinal bacteria; the level of equol production has been linked to the consumption, and the content of the isoflavone daidzein. The solid food matrix of tempeh may protect isoflavones from degradation, so they could reach the large intestine and metabolized into equol by the gut bacteria. This indicates that tempeh contains more daidzein than soymilk. Information about pharmacokinetics is very important to evaluate the safety and understand the efficacy. For example, from the t1/2, we could predict that how long isoflavones are still present in the body, so that its consumption time could be regulated by the medication.
Table 3. Pharmacokinetics of isoflavones after oral administration in humans.
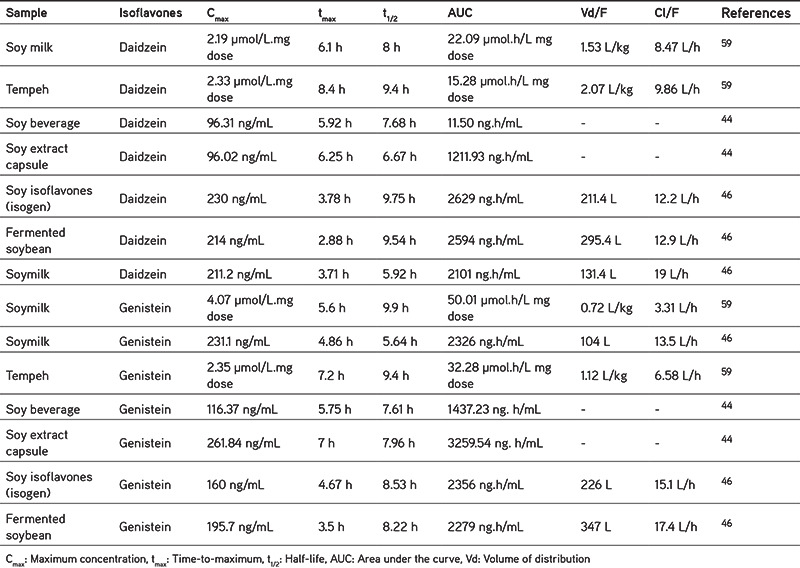
It turns out that not only isoflavone tablets are high in isoflavones, but daily food processed from the soy also contains quite high levels of isoflavones, and may interact if taken together with certain drugs. Thus, there is a need for careful monitoring. An assessment of the pharmacokinetic profile of several other processed soybean products needs to be done, for example tofu, to obtain more information.
The mechanism of drug-isoflavone pharmacokinetic interactions
Drug interactions not only occur between drugs, but also occur between drugs and herbal or natural compounds, such as isoflavones. Isoflavones are a component of dietary foods or herbal supplements, so there is a possibility of long-term exposure together with the drugs. This simultaneous use may lead to the drug-isoflavone interactions. This is supported by Laurenzana et al.64, who found that the content of natural materials such as flavones, isoflavones, and tangeretin affects the activity of human CYP enzymes when given orally together with the drugs. These changes in ADME will certainly affect the pharmacokinetic parameters of the drugs, because of the interactions with DMEs and interaction with the drug transporters.
In this article, the pharmacokinetic interactions between isoflavones and some drugs and their mechanisms of interaction have been summarized, focusing on the enzymes and drug transporters, as shown in Table 4. It has been reported that the co-administration of soy isoflavones (genistein or daidzein), soy tablets, or soybean extract with drugs results in changes in the pharmacokinetic parameters of the drug, which indicates an interaction. These effects include the changes in the AUC, Cmax, and the clearance. These changes may be either an increase or a decrease in the pharmacokinetic parameters, depending on the mechanism. The mechanisms that will be discussed here involve enzymes and drug transporters.
Table 4. Interaction of soy isoflavones with drugs based on pharmacokinetic parameters.
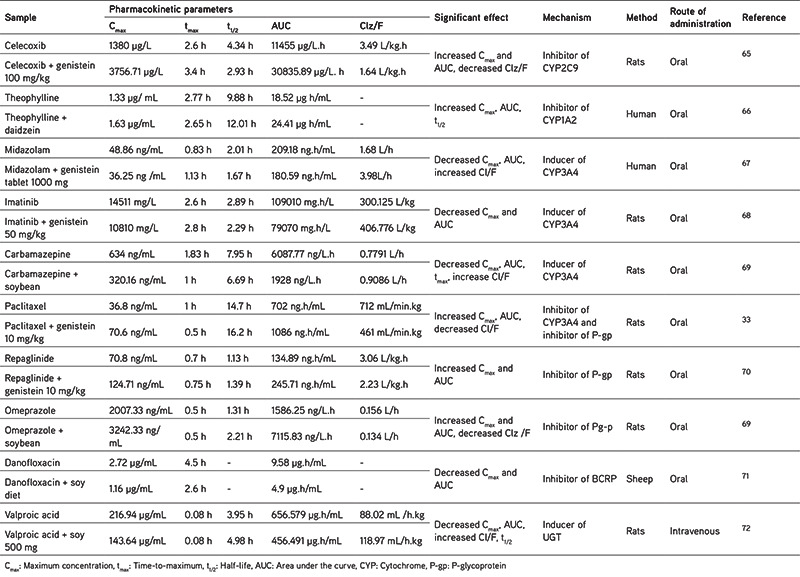
Effects of soy isoflavones on drug metabolizing enzymes
Soybeans influence the metabolism of the drugs, and affect ADME through the interactions with phase-I or phase-II DMEs. The enzymes involved in phase-I metabolism are the CYP450 families, while the enzymes involved in phase-II metabolism are sulfotransferases, uridine diphosphate glucuronosyltransferases (UDPGT/UGTs), N-acetyl transferases (UDPGT/UGTs), glutathione-S-transferases, and methyltransferases.25
Phase-I metabolism enzymes
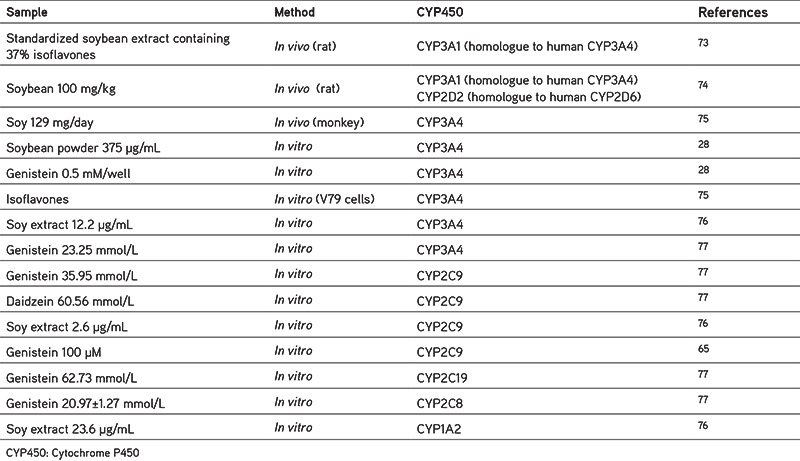
CYP450 are the main group of enzymes that catalyze the oxidative biotransformation of the drugs and other lipophilic xenobiotics.27 The enzymes involved in the drugs metabolism are CYP1A2, CYP2C9, CYP2C19, CYP2D6, and CYP3A4.26 The enzymes that are influenced by soy isoflavone are discussed below based on the pharmacokinetic interaction mechanism of some drugs (Table 4). To support the discussion, we have summarized the inhibitory effect of soy isoflavones on CYP enzymes in Table 5.
Table 5. Summary of the inhibitory effects of soy isoflavones on CYP enzymes.
CYP2C9
Pharmacokinetic interactions may occur through the inhibition or induction of DMEs. Around 15% of all drug biotransformation is metabolized by the CYP2C9.78 An interaction between celecoxib and genistein has been reported;65 as shown in Table 4, there is an increase in Cmax and AUC is almost 2.7 times higher than celecoxib alone, because of the inhibition of the CYP2C9 enzyme by genistein. Thus, the metabolism of celecoxib is reduced, clearance also decreases, and celecoxib accumulates in the body. This mechanism is also in line with the results of Kopecna-Zapletalova et al.77 based on in vitro studies (Table 5) showing that genistein may inhibit CYP2C9 at doses of 35.96 mmol/L and 100 µM.65 The flavone structure of genistein (4,5,5,7-trihydroxyisoflavone) may suppress CYP2C9 by interacting with the active site of CYP2C9.79
CYP1A2
The same effect was also seen by Peng et al.66 when theophylline was given with soy isoflavones such as daidzein at a dose of 200 mg twice a day to healthy volunteers. There was an increase in the AUC and Cmax. Theophylline is mainly excreted through the hepatic metabolism pathway, and CYP1A2 catalyzes all these pathways; thus, the inhibition of CYP1A2 will inhibit the metabolism of this drug. This is also related to the results of Anderson et al.76 who showed that soybean extract inhibits the CYP1A2 enzyme in vitro; one of the isoflavones contained in soy extract is daidzein.
CYP3A4
Inhibition of CYP3A4 will increase the drug levels, as shown by Li and Choi.33 In vivo, genistein may increase the value of AUC and Cmax of paclitaxel through the inhibition of CYP3A4; this is also supported by in vitro studies. It has been widely reported that genistein from soybean inhibits CYP3A4.28,76,77 In vitro studies have reported that genistein inhibits CYP3A4 at a concentration of 0.5 mM/well, supported by Kopecna-Zapletalova et al.77 from 2016 showing that genistein may inhibit CYP3A4 at a concentration of 23.25 mM. Another trial using different cells, namely V79 cells, showed the inhibitory activity of genistein on CYP3A4.75 The inhibitory effect of isoflavones on CYP3A4 is classified as moderate inhibition and is non-competitive.77
In addition to the inhibitory effect described above, some studies show that the mechanism of isoflavones also may alter the pharmacokinetics of drugs by the induction of enzymes. This may decrease the AUC and Cmax and increase clearance. Studies by Xiao et al.67 showed there is a change in the value of midazolam pharmacokinetic parameters after the patients were given genistein tablets (1000 mg) for 14 days; the same thing was also found for imatinib80 and carbamazepine.69 Midazolam and imatinib are primarily metabolized by CYP3A4 after oral administration.67 Imatinib is metabolized into N-desmethyl imatinib by CYP3A480,81 and is a prodrug. This means that there is a different mechanism for the prodrug. Prodrugs are activated by a CYP, so it is important to know if metabolism or the activation of enzymes may alter CYP activity.82 Genistein increases the Cmax and AUC of N-desmethyl imatinib by the induction of CYP3A4. In the future, to clarify the mechanism, it will be necessary to carry out a deeper investigation related to the effect of soy isoflavones on prodrugs.
Induction of xenobiotic-mediated CYP3A genes in humans is known to be regulated by pregnane X receptors (PXR), constitutive and immune receptors, glucocorticoid receptors, and other receptors.83 PXR is the main regulator of xenobiotic-induced CYP3A gene expression. Previous research has found that genistein may significantly activate the human PXR, and induce the human CYP3A4 luciferase reporter activity.84 According to this study, we consider that genistein acts as an inducer of CYP3A4 in humans. However, the CYP3A4 induction mechanism is contrary to in-vitro studies (Table 4) because many studies report that soy, and its isoflavones have an inhibitory effect rather than induction, so there is no in vivo/in vitro correlation related to the effect of soybean on CYP3A4.
According to Cheng et al.48 soybean contains 42 µg/g genistein and 4.78 µg/g daidzein, while some have reported extracts containing and 1260 µg/g genistein and 849 µg/g daidzein,41 or 36.55 µg/g genistein and 88.87 µg/g daidzein.40 These variations in the content may be caused by the differences in the soybean variety assessed, the location of growth, plant age, etc. Other than that, processed soybean foods such as tofu, tempeh, soy milk, natto, miso, and sufu also have variable contents, which could be seen in Table 3. For example, fried tempeh contains 310 µg/g genistein and 350 µg/g daidzein.60 In natto, the level of genistein is 224 µg/g and that of daidzein is 411 µg/g57, but soymilk has a lower content of 56 µg/mL and 52 µg/mL, respectively.46 When linked with experimental data in vitro from the various studies, it appears that soybean extract may inhibit the CYP3A4 enzyme at a concentration of 12.2 µg/mL, CYP2C9 at 2.6 µg/mL, and CYP1A2 at 23.6 µg/mL.76 This means that consuming 1 gram of soybean extract may influence these enzymes. The same thing is also the case with soybeans and soybean products, because the content of genistein and daidzein (shown in Table 3) in each gram exceeds the inhibitory dose reported by Kopecna-Zapletalova et al.77 However, further in vivo studies in human subjects need to be performed, as in vivo studies have only been conducted on mice with soybean doses that inhibit CYP3A1 (the homologue to CYP3A4 in humans), (i.e., 100 mg/kg).74 If simplified, the dose is equivalent to 100 µg/g, so based on this the consumption of soymilk, tofu, soybeans could be said to be safe, but again further research is needed to obtain more accurate results.
Phase-II metabolism enzymes
Uridine diphosphate glucuronosyltransferases
Soybean increases the phase-II metabolism of drugs to increase the detoxification and clearance of potentially carcinogenic intermediaries. The results of Marahatta et al.72 report that the administration of 500 mg for 5 days could affect valproic acid (VPA) in terms of its pharmacokinetic parameters. Specifically, the Cmax decreased by 65%, but time-to-maximum (tmax) was not significantly different. AUC decreased by 69%. There were significant differences in Cmax, t1/2, AUC, and clearance between the treatment and control groups.72 Soybean contributes to VPA excretion, which is very effective as it increases VPA glucuronidation. Valproate glucuronide is the main metabolite of VPA in urine and is metabolized by UGT1A3, UGT1A4, UGT1A6, UGT1A8, UGT1A9, UGT1A10, UGT2B7, and UGT2B15. The metabolism and elimination of VPA is affected by glucuronidation, especially by uridine 59-diphosphate-glucuronosyltransferase. Similarly, previous studies have shown that soy induces the UGT enzyme, an important component of glucuronidation.85 Daidzein may stimulate glucuronidation.86 Similarly, genistein has been reported to induce UGT activity.87 The inhibition or induction of important enzymes for drugs that require therapeutic drug monitoring and food-drug interactions depend on the therapeutic index of each drug.72
Effects of soy ısoflavones on drug transporters
Drug transporters have an important role in the ADME of drugs and xenobiotics.88 Drug transporters are also related to disposition of drug and drug interactions.89 Drug transporters are classified as uptake and efflux transporters. Uptake transporters play a role in facilitating the translocation of drugs into cells such as organic anion transporting polypeptides (OATP; SLCO)90, organic anion transporters (OAT; SLC22A)91, and organic cation transporters (OCT; SLC22A)92, while efflux transporters transfer or remove drugs from the intracell to the extra cell, for example the ABC group and SLC transporters. The ABC family includes transporters for the elimination of drugs likes P-gp [multidrug resistance protein 1 (MDR1); ABCB1], certain members of the multidrug resistance-associated protein (MRP; ABCC) family, and breast cancer resistance protein [(BCRP); ABCG]. These drug transporters are expressed in the intestine or liver, two main locations that affect how much drug will enter the body after the administration of an oral dose. Thus, the effect of isoflavones on drug transporters is important because it will affect the pharmacokinetic profile of a drug.93 As shown in Table 4, the pharmacokinetic interaction mechanisms of some drugs occur only through efflux transporters.
Efflux drug transporters
P-glycoprotein
P-gp is a product of the MDR1 gene, which is an efflux transporter that is widely studied and known for its ability to limit the entry of drugs into various organ compartments. P-gp functions as an efflux pump, such that it facilitates the transfer of intracellular drugs to the extracellular space.26 Genistein may influence the administration of the drugs by modulating efflux proteins such as MDR1 and P-gp. P-gp is expressed mainly in the apical membrane of the intestine. MDR1 has been reported to increase the elimination of drugs in the intestinal lumen.34 This mechanism is shown in Figure 2. Genistein inhibits P-gp and causes pharmacokinetic interactions with repaglinide at a genistein concentration of 10 mg/kg, characterized by an increase in the repaglinide AUC of 53% and Cmax by 36%.70 Genistein affects P-gp by increasing intestinal absorption. Li and Choi.33 found an increase in the paclitaxel plasma concentration with a mechanism of P-gp inhibition, similar to what was also found with midazolam.69 To confirm, Li et al.54 tested Caco-2 cells and IEC-6 cells to investigate further repaglinide absorption in human cells and in mice, resulting in significantly increased intracellular repaglinide accumulation with genistein administration.70 This means that P-gp transporters, which are supposed to carry drugs to the extracellular are blocked by genistein, resulting in the intracellular accumulation of repaglinide.
Figure 2.
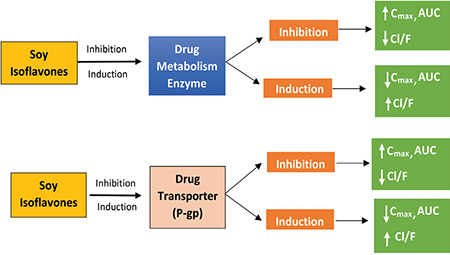
Mechanisms of pharmacokinetic interactions between drugs and soy isoflavones (based on Table 4)
Cmax: Maximum concentration, AUC: Area under the curve, P-gp: P-glycoprotein, CL/F: Oral clearance
The mechanism by which genistein inhibits P-gp was revealed by molecular docking studies. The basic structure of P-gp includes four main core regions, with two nucleotide-binding domains (NBD) located in the cytoplasm and two hydrophobic transmembrane domains (TMD).94 The TMD serve as a channel to facilitate drug transport, whereas the NBD located in the cytoplasm have binding sites for ATP, used as the energy supply for drug transport.95 6C0V was chosen as a P-gp molecule with a three-dimensional structure combined with NBD simulation; it was found that genistein has a certain binding affinity for NBD and shares several binding sites with ATP in the corresponding functional area, which affects the energy supply when the drug is transported by P-gp. This is what causes the inhibition of the efflux function of P-gp.70
BCRP
Drug interactions that lead to the inhibition of efflux transporters can cause changes in the pharmacokinetics of the drug. For example, in the case of BCRP, several drugs are secreted into milk, such as danofloxacin as shown in a study performed in sheep given a soy diet to see its effect on drug levels in milk. A change was observed in the pharmacokinetic parameters of danofloxacin, namely a 50% decrease in Cmax and AUC.71 BCRP inhibitors administered with drugs that are substrates of the transporter could have effects on in vivo ADME, as well as the presence of drugs in milk.96,97 A soy diet contains daidzein and genistein, which are BCRP inhibitors.98,99
From what has been discussed above, we could see that the pharmacokinetic interaction of soy isoflavones with drugs occurs through the several mechanisms, (i.e., through DMEs or drug transporters). These interactions will affect the bioavailability of drugs in the blood. The mechanisms are summarized and illustrated in Figure 2.
CONCLUSION
Soybeans are a good source of isoflavones. The isoflavone content of soybean is mainly in the aglycone form as daidzein, genistein, and glycitein. Soybean products also contain variable levels of isoflavones. Co-administration of soy isoflavones with the drugs may cause pharmacokinetic interactions. These interactions may cause changes in the AUC, Cmax, tmax, and t1/2 of the drugs. These interactions occur through mechanisms related to the inhibition/induction of DMEs, namely CYP3A4, CYP2C9, CYP1A2, and UGT or the inhibition/induction of drug transporters, such as P-gp and BCRP. Thus, the consumption of soy, soy isoflavones, or soy products together with the drugs needs to be considered because this diet may affect the efficacy of the drugs. Furthermore, the timing and consumption of soy isoflavones with the drugs should be monitored.
Footnotes
Conflict of interest: No conflict of interest was declared by the authors. The authors alone are responsible for the content and writing of this article.
References
- 1.Terzic D, Popovic V, Tatic M. Soybean area, yield and production. Ecol Mov Novi Sad. 2018:135–143. [Google Scholar]
- 2.Zaheer K, Humayoun Akhtar M. An updated review of dietary isoflavones: Nutrition, processing, bioavailability and impacts on human health. Crit Rev Food Sci Nutr. 2015;57:1280–1293. doi: 10.1080/10408398.2014.989958. [DOI] [PubMed] [Google Scholar]
- 3.Cheng PF, Chen JJ, Zhou XY, Ren YF, Huang W, Zhou JJ, Xie P. Do soy isoflavones improve cognitive function in postmenopausal women? A meta-analysis. Menopause. 2015;22:198–206. doi: 10.1097/GME.0000000000000290. [DOI] [PubMed] [Google Scholar]
- 4.Larkin T, Price WE, Astheimer L. The key importance of soy isoflavone bioavailability to understanding health benefits. Crit Rev Food Sci Nutr. 2008;48:538–552. doi: 10.1080/10408390701542716. [DOI] [PubMed] [Google Scholar]
- 5.Tsourounis C. Clinical effects of phytoestrogens. Clin Obstet Gynecol. 2001;44:836–842. doi: 10.1097/00003081-200112000-00021. [DOI] [PubMed] [Google Scholar]
- 6.Teekachunhatean S, Hanprasertpong N, Teekachunhatean T. Factors affecting isoflavone content in soybean seeds grown in Thailand. Int J Agron. 2013;2013:1–11. [Google Scholar]
- 7.Ahsan M, Mallick AK. The effect of soy isoflavones on the menopause rating scale scoring in perimenopausal and postmenopausal women: A pilot study. J Clin Diagnostic Res. 2017;11:13–16. doi: 10.7860/JCDR/2017/26034.10654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Agostoni C, Bresson J, Tait S, Flynn A, Golly I, Korhinen H. Scientific Opinion on the substantiation of health claims related to soy isoflavones and maintenance of bone mineral density (ID 1655) and reduction of vasomotor symptoms associated with menopause. EFSA J. 2012;10:2847. [Google Scholar]
- 9.Chi XX, Zhang T. Isoflavone intake inhibits the development of 7,12 dimethylbenz(a)anthracene(DMBA) induced mammary tumors in normal and ovariectomized rats. J Clin Biochem Nutr. 2013;54:31–38. doi: 10.3164/jcbn.13-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mace TA, Ware MB, King SA, Loftus S, Farren MR, McMichael E, Scoville S, Geraghty C, Young G, Carson WE 3rd, Clinton SK, Lesinski GB. Soy isoflavones and their metabolites modulate cytokine-induced natural killer cell function. Sci Rep. 2019;9:5068. doi: 10.1038/s41598-019-41687-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhang X, Gao YT, Yang G, Li H, Cai Q, Xiang YB, Ji BT, Franke AA, Zheng W, Shu XO. Urinary isoflavonoids and risk of coronary heart disease. Int J Epidemiol. 2012;41:1367–1375. doi: 10.1093/ije/dys130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yoon GA, Park S. Antioxidant action of soy isoflavones on oxidative stress and antioxidant enzyme activities in exercised rats. Nutr Res Pract. 2014;8:618–624. doi: 10.4162/nrp.2014.8.6.618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sahin I, Bilir B, Ali S, Sahin K, Kucuk O. Soy isoflavones in integrative oncology: increased efficacy and decreased toxicity of cancer therapy. Integr Cancer Ther. 2019:1–11. doi: 10.1177/1534735419835310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mizushina Y, Shiomi K, Kuriyama I, Takahashi Y, Yoshida H. Inhibitory effects of a major soy isoflavone, genistein, on human DNA topoisomerase II activity and cancer cell proliferation. Int J Oncol. 2013;43:1117–1124. doi: 10.3892/ijo.2013.2032. [DOI] [PubMed] [Google Scholar]
- 15.He F-J, Chen J-Q. Consumption of soybean, soy foods, soy isoflavones and breast cancer incidence: Differences between Chinese women and women in Western countries and possible mechanisms. Food Sci Hum Wellness. 2013;2:146–161. [Google Scholar]
- 16.Fonseca ND, Villar MPM, Donangelo CM, Perrone D. Isoflavones and soyasaponins in soy infant formulas in Brazil: Profile and estimated consumption. Food Chem. 2014;143:492–498. doi: 10.1016/j.foodchem.2013.07.126. [DOI] [PubMed] [Google Scholar]
- 17.Klein CB, King AA. Genistein genotoxicity: critical considerations of in vitro exposure dose. Toxicol Appl Pharmacol. 2007;224:1–11. doi: 10.1016/j.taap.2007.06.022. [DOI] [PubMed] [Google Scholar]
- 18.Rizzo G, Baroni L. Soy, soy foods and their role in vegetarian diets. Nutrients. 2018;10:1–51. doi: 10.3390/nu10010043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.U.S. Soybean Export Council. Recommended Soy Intakes. 2020:1-5. [Internet] www.ussec.org.
- 20.Varma M V, Pang SK, Isoherranen N, Zhao P. Dealing with the complex drug-drug interactions: Towards mechanistic models Manthena. Biopharm Drug Dispos. 2015;36:71–92. doi: 10.1002/bdd.1934. [DOI] [PubMed] [Google Scholar]
- 21.Cambria-Kiely JA. Effect of soy milk on warfarin efficacy. Ann Pharmacother. 2002;36:1893–1896. doi: 10.1345/aph.1C160. [DOI] [PubMed] [Google Scholar]
- 22.Chen J, Halls SC, Alfaro JF, Zhou Z, Hu M. Potential beneficial metabolic interactions between tamoxifen and isoflavones via cytochrome P450-mediated pathways in female rat liver microsomes. Pharm Res. 2004;21:2095–2104. doi: 10.1023/b:pham.0000048202.92930.61. [DOI] [PubMed] [Google Scholar]
- 23.Nagashima Y, Kondo T, Sakata M, Koh J, Ito H. Effects of soybean ingestion on pharmacokinetics of levodopa and motor symptoms of Parkinson’s disease - In relation to the effects of Mucuna pruriens. J Neurol Sci. 2016;361:229–234. doi: 10.1016/j.jns.2016.01.005. [DOI] [PubMed] [Google Scholar]
- 24.Temyingyong N, Koonrungsesomboon N, Hanprasertpong N, Na Takuathung M, Teekachunhatean S. Effect of short-course oral ciprofloxacin on isoflavone pharmacokinetics following soy milk ingestion in healthy postmenopausal women. Evidence-based Complement Altern Med. 2019;2019:1–10. doi: 10.1155/2019/7192326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Taneja I, Raju KSR, Wahajuddin M. Dietary Isoflavones as modulators of drug metabolizing enzymes and transporters: effect on prescription medicines. Crit Rev Food Sci Nutr. 2015;56:1–69. doi: 10.1080/10408398.2015.1045968. [DOI] [PubMed] [Google Scholar]
- 26.Tirona RG, Kim RB. Introduction to clinical pharmacology. Clin Transl Sci Princ Hum Res. 2017;20:365–388. [Google Scholar]
- 27.Zanger UM, Schwab M. Cytochrome P450 enzymes in drug metabolism: Regulation of gene expression, enzyme activities, and impact of genetic variation. Pharmacol Ther. 2013;138:103–141. doi: 10.1016/j.pharmthera.2012.12.007. [DOI] [PubMed] [Google Scholar]
- 28.Foster BC, Vandenhoek S, Hana J, Krantis A, Akhtar MH, Bryan M, Budzinski JW, Ramputh A, Arnason JT. In vitro inhibition of human cytochrome P450-mediated metabolism of marker substrates by natural products. Phytomedicine. 2003;10:334–342. doi: 10.1078/094471103322004839. [DOI] [PubMed] [Google Scholar]
- 29.Liu YT, Chen YH, Uramaru N, Lin AH, Yang HT, Lii CK, Yao HT. Soy isoflavones reduce acetaminophen-induced liver injury by inhibiting cytochrome P-450-mediated bioactivation and glutathione depletion and increasing urinary drug excretion in rats. J Funct Foods. 2016;26:135–143. [Google Scholar]
- 30.Liu L, and Liu X. Contributions of drug transporters to blood-brain barriers. Adv Exp Med Bio. 2019;114:407–466. doi: 10.1007/978-981-13-7647-4_9. [DOI] [PubMed] [Google Scholar]
- 31.Estudante M, Morais JG, Soveral G, Benet LZ. Intestinal drug transporters: An overview. Adv Drug Deliv Rev. 2013;65:1340–1356. doi: 10.1016/j.addr.2012.09.042. [DOI] [PubMed] [Google Scholar]
- 32.Taur JS, Rodriguez-Proteau R. Effects of dietary flavonoids on the transport of cimetidine via P-glycoprotein and cationic transporters in Caco-2 and LLC-PK1 cell models. Xenobiotica. 2008;38:1536–1550. doi: 10.1080/00498250802499467. [DOI] [PubMed] [Google Scholar]
- 33.Li X, Choi JS. Effect of genistein on the pharmacokinetics of paclitaxel administered orally or intravenously in rats. Int J Pharm. 2007;337:188–193. doi: 10.1016/j.ijpharm.2007.01.002. [DOI] [PubMed] [Google Scholar]
- 34.Limtrakul P, Khantamat O, Pintha K. Inhibition of P-glycoprotein function and expression by kaempferol and quercetin. J Chemother. 2005;17:86–95. doi: 10.1179/joc.2005.17.1.86. [DOI] [PubMed] [Google Scholar]
- 35.Wang G, Xiao CQ, Li Z, Guo D, Chen Y, Fan L, Qian RH, Peng XJ, Hu DL, Zhou HH. Effect of soy extract administration on losartan pharmacokinetics in healthy female volunteers. Ann Pharmacother. 2009;43:1045–1049. doi: 10.1345/aph.1L690. [DOI] [PubMed] [Google Scholar]
- 36.Wang Q, GE X, Tian X, Zhang Y, Zhang J, Zhang P. Soy isoflavone: The multipurpose phytochemical. Biomed Rep. 2013;1:697–701. doi: 10.3892/br.2013.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Setchell KDR, Brown NM, Zhao X, Lindley SL, Heubi JE, King EC, Messina MJ. Soy isoflavone phase II metabolism differs between rodents and humans: Implications for the effect on breast cancer risk. Am J Clin Nutr. 2011;94:1284–1294. doi: 10.3945/ajcn.111.019638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lozovaya VV, Lygin AV, Ulanov AV, Nelson RL, Daydé J, Widholm JM. Effect of temperature and soil moisture status during seed development on soybean seed isoflavone concentration and composition. Crop Sci. 2005;45:1934–1940. [Google Scholar]
- 39.Ho HM, Chen RY, Leung LK, Chan FL, Huang Y, Chen ZY. Difference in flavonoid and isoflavone profile between soybean and soy leaf. Biomed Pharmacother. 2002;56:289–295. doi: 10.1016/s0753-3322(02)00191-9. [DOI] [PubMed] [Google Scholar]
- 40.Mebrahtu T, Mohamed A, Wang CY, Andebrhan T. Analysis of isoflavone contents in vegetable soybeans. Plant Foods Hum Nutr. 2004;59:55–61. doi: 10.1007/s11130-004-0023-4. [DOI] [PubMed] [Google Scholar]
- 41.Andrade JE, Twaddle NC, Helferich WG, Doerge DR. Absolute bioavailability of isoflavones from soy protein isolate-containing food in female Balb/c mice. J Agric Food Chem. 2010;58:4529–4536. doi: 10.1021/jf9039843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Aresta A, Cotugno P, Massari F, Zambonin C. Determination of isoflavones in soybean flour by matrix solid-phase dispersion extraction and liquid chromatography with UV-diode array detection. J Food Qual. 2017;2017:1–5. [Google Scholar]
- 43.Orhan I, Ozcelik B, Kartal M, Aslan S, Sener B, Ozguven M. Quantification of daidzein, genistein and fatty acids in soybeans and soy sprouts, and some bioactivity studies. Acta Biol Cracoviensia Ser Bot. 2007;49:61–68. [Google Scholar]
- 44.Anupongsanugool E, Teekachunhatean S, Rojanasthien N, Pongsatha S, Sangdee C. Pharmacokinetics of isoflavones, daidzein and genistein, after ingestion of soy beverage compared with soy extract capsules in postmenopausal Thai women. BMC Clin Pharmacol. 2005;5:1–10. doi: 10.1186/1472-6904-5-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chen TR, Wei QK. Analysis of bioactive aglycone isoflavones in soybean and soybean products. Nutr Food Sci. 2008;38:540–547. [Google Scholar]
- 46.Chang Y, Choue R. Plasma pharmacokinetics and urinary excretion of isoflavones after ingestion of soy products with different aglycone/glucoside ratios in South Korean women. Nutr Res Pract. 2013;7:393–399. doi: 10.4162/nrp.2013.7.5.393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Zhang J, Ge Y, Han F, Li B, Yan S, Sun J, Wang L. Isoflavone content of soybean cultivars from maturity group 0 to VI grown in northern and southern China. J Am Oil Chem Soc. 2014;91:1019–1028. doi: 10.1007/s11746-014-2440-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Cheng YQ, Zhu YP, Hu Q, Li LT, Saito M, Zhang SX, Yin LJ. Transformation of isoflavones during sufu (a traditional Chinese fermented soybean curd) production by fermentation with Mucor flavus at low temperature. Int J Food Prop. 2011;14:629–639. [Google Scholar]
- 49.Hong GE, Mandal PK, Lim K won, Lee CH. Fermentation increases isoflavone aglycone contents in black soybean pulp. Asian J Anim Vet Adv. 2012:502–511. [Google Scholar]
- 50.Kuligowski M, Pawłowska K, Jasińska-Kuligowska I, Nowak J. Composición de isoflavonas, contenido de polifenoles y actividad antioxidante de las semillas de soja durante fermentación de tempeh. CYTA J Food. 2016;15:27–33. [Google Scholar]
- 51.Riciputi Y, Serrazanetti DI, Verardo V, Vannini L, Caboni MF, Lanciotti R. Effect of fermentation on the content of bioactive compounds in tofutype products. J Funct Foods. 2016;27:131–139. [Google Scholar]
- 52.Da Silva LH, Celeghini RMS, Chang YK. Effect of the fermentation of whole soybean flour on the conversion of isoflavones from glycosides to aglycones. Food Chem. 2011;128:640–644. [Google Scholar]
- 53.Huang YH, Lai YJ, Chou CC. Fermentation temperature affects the antioxidant activity of the enzyme-ripened sufu, an oriental traditional fermented product of soybean. J Biosci Bioeng. 2011;112:49–53. doi: 10.1016/j.jbiosc.2011.03.008. [DOI] [PubMed] [Google Scholar]
- 54.Li S, Jin Z, Hu D, Yang W, Yan Y, Nie X, Jing L, Qingyu Z, Gai D, Ji Y, Chen X. Effect of solid-state fermentation with Lactobacillus casei on the nutritional value, isoflavones, phenolic acids and antioxidant activity of whole soybean flour. Lebensm Wiss Technol. 2020;125:109264. [Google Scholar]
- 55.Gardner CD, Chatterjee LM, Franke AA. Effects of isoflavone supplements vs. soy foods on blood concentrations of genistein and daidzein in adults. J Nutr Biochem. 2009;20:227–234. doi: 10.1016/j.jnutbio.2008.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Golkhoo S, Ahmadi AR, Hanachi P, Barantalab F, Vaziri M. Determination of daidzein and genistein in soy milk in Iran by using HPLC analysis method. Pakistan J Biol Sci. 2008;11:2254–2258. doi: 10.3923/pjbs.2008.2254.2258. [DOI] [PubMed] [Google Scholar]
- 57.Miura A, Sugiyama C, Sakakibara H, Simoi K, Goda T. Bioavailability of isoflavones from soy products in equol producers and non-producers in Japanese women. J Nutr Intermed Metab. 2016;6:41–47. [Google Scholar]
- 58.Freddo N, Nardi J, Bertol CD, Dallegrave E, Leal MB, Barreto F. Isoflavone quantitation in soymilk: Genistein content and its biological effect. CYTA J Food. 2019;17:20–24. [Google Scholar]
- 59.Cassidy A, Brown JE, Hawdon A, Faughnan MS, King LJ, Millward J, Zimmer-Nechemias L, Wolfe B, Setchell KD. Factors affecting the bioavailability of soy isoflavones in humans after ingestion of physiologically relevant levels from different soy foods. J Nutr. 2006;136:45–51. doi: 10.1093/jn/136.1.45. [DOI] [PubMed] [Google Scholar]
- 60.Haron H, Ismail A, Azlan A, Shahar S, Peng LS. Daidzein and genestein contents in tempeh and selected soy products. Food Chem. 2009;115:1350–1356. [Google Scholar]
- 61.Prabhakaran MP, Perera CO, Valiyaveettil S. Quantification of isoflavones in soymilk and tofu from South East Asia. Int J Food Prop. 2005;8:113–123. [Google Scholar]
- 62.Lee MK, Kim JK, Lee SY. Effects of fermentation on SDS-PAGE patterns, total peptide, isoflavone contents and antioxidant activity of freeze-thawed tofu fermented with Bacillus subtilis. Food Chem. 2018;249:60–65. doi: 10.1016/j.foodchem.2017.12.045. [DOI] [PubMed] [Google Scholar]
- 63.Faughnan MS, Hawdon A, Ah-Singh E, Brown J, Millward DJ, Cassidy A. Urinary isoflavone kinetics: the effect of age, gender, food matrix and chemical composition. Br J Nutr. 2004;91:567–574. doi: 10.1079/BJN20041087. [DOI] [PubMed] [Google Scholar]
- 64.Laurenzana EM, Weis CC, Bryant CW, Newbold R, Delclos KB. Effect of dietary administration of genistein, nonylphenol or ethinyl estradiol on hepatic testosterone metabolism, cytochrome P-450 enzymes, and estrogen receptor alpha expression. Food Chem Toxicol. 2002;40:53–63. doi: 10.1016/s0278-6915(01)00095-3. [DOI] [PubMed] [Google Scholar]
- 65.Zheng X, Wen J, Liu TH, Ou-Yang QG, Cai JP, Zhou HY. Genistein exposure interferes with pharmacokinetics of celecoxib in SD male rats by UPLC-MS/MS. Biochem Res Int. 2017;2017:1–7. doi: 10.1155/2017/6510232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Peng WX, Li H De, Zhou HH. Effect of daidzein on CYP1A2 activity and pharmacokinetics of theophylline in healthy volunteers. Eur J Clin Pharmacol. 2003;59:237–241. doi: 10.1007/s00228-003-0596-0. [DOI] [PubMed] [Google Scholar]
- 67.Xiao CQ, Chen R, Lin J, Wang G, Chen Y, Tan ZR, Zhou HH. Effect of genistein on the activities of cytochrome P450 3A and P-glycoprotein in Chinese healthy participants. Xenobiotica. 2012;42:173–178. doi: 10.3109/00498254.2011.615954. [DOI] [PubMed] [Google Scholar]
- 68.Wang Z, Wang L, Xia MM, Sun W, Huang CK, Cui X, Hu GX, Lian QQ, Wang ZS. Pharmacokinetics interaction between imatinib and genistein in rats. Biomed Res Int. 2015;2015:368976. doi: 10.1155/2015/368976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Singh D, Asad M. Effect of soybean administration on the pharmacokinetics of carbamazepine and omeprazole in rats. Fundam Clin Pharmacol. 2010;24:351–355. doi: 10.1111/j.1472-8206.2009.00762.x. [DOI] [PubMed] [Google Scholar]
- 70.Jin H, Zhu Y, Wang C, Meng Q, Wu J, Sun P, Ma X, Sun H, Huo X, Liu K, Tan A. Molecular pharmacokinetic mechanism of the drug-drug interaction between genistein and repaglinide mediated by P-gp. Biomed Pharmacother. 2020;125:110032. doi: 10.1016/j.biopha.2020.110032. [DOI] [PubMed] [Google Scholar]
- 71.Perez M, Otero JA, Barrera B, Prieto JG, Merino G, Alvarez AI. Inhibition of ABCG2/BCRP transporter by soy isoflavones genistein and daidzein: Effect on plasma and milk levels of danofloxacin in sheep. Vet J. 2013;196:203–208. doi: 10.1016/j.tvjl.2012.09.012. [DOI] [PubMed] [Google Scholar]
- 72.Marahatta A, Bhandary B, Jeong SK, Kim HR, Chae HJ. Soybean greatly reduces valproic acid plasma concentrations: A food-drug interaction study. Sci Rep. 2014;4:1–7. doi: 10.1038/srep04362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Mrozikiewicz PM, Bogacz A, Czerny B, Karasiewicz M, Kujawski R, Mikolajczak PL, Seremak-Mrozikiewicz A, Grzeskowiak E, Bobkiewicz-Kozlowska T. The influence of a standardized soybean extract (Glycine max) on the expression level of cytochrome P450 genes in vivo. Ginekol Pol. 2010;81:516–520. [PubMed] [Google Scholar]
- 74.Bogacz A, Bartkowiak-Wieczorek J, Mikołajczak PŁ, Rakowska-Mrozikiewicz B, Grześkowiak E, Wolski H, Czerny B, Mrozikiewicz PM. The influence of soybean extract on the expression level of selected drug transporters, transcription factors and cytochrome P450 genes encoding phase I drug-metabolizing enzymes. Ginekol Pol. 2014;85:348–353. doi: 10.17772/gp/1741. [DOI] [PubMed] [Google Scholar]
- 75.Scott L, Durant P, Leone-Kabler S, Scott LM, Durant P, Leone-Kabler S, Wood CE, Register TC, Townsend A, Cline JM. Effects of prior oral contraceptive use and soy isoflavonoids on estrogen-metabolizing cytochrome P450 enzymes. J Steroid Biochem Mol Biol. 2008;112:179–185. doi: 10.1016/j.jsbmb.2008.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Anderson GD, Rosito G, Mohustsy MA, Elmer GW. Drug interaction potential of soy extract and Panax ginseng. J Clin Pharmacol. 2003;43:643–648. [PubMed] [Google Scholar]
- 77.Kopecna-Zapletalova M, Krasulova K, Anzenbacher P, Hodek P, Anzenbacherova E. Interaction of isoflavonoids with human liver microsomal cytochromes P450: inhibition of CYP enzyme activities. Xenobiotica. 2016;47:324–331. doi: 10.1080/00498254.2016.1195028. [DOI] [PubMed] [Google Scholar]
- 78.Isvoran A, Louet M, Vladoiu DL, Craciun D, Loriot MA, Villoutreix BO, Miteva MA. Pharmacogenomics of the cytochrome P450 2C family : impacts of amino acid variations on drug metabolism. Drug Discov Today. 2017;22:366–376. doi: 10.1016/j.drudis.2016.09.015. [DOI] [PubMed] [Google Scholar]
- 79.Shimada T, Tanaka K, Takenaka S, Murayama N, Martin MV, Foroozesh MK, Yamazaki H, Guengerich FP, Komori M. Structure-function relationships of inhibition of human cytochromes P450 1A1, 1A2, 1B1, 2C9, and 3A4 by 33 flavonoid derivatives. Chem Res Toxicol. 2010;23:1921–1935. doi: 10.1021/tx100286d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Nebot N, Crettol S, Esposito F, Tattam B. Participation of CYP2C8 and CYP3A4 in the N-demethylation of imatinib in human hepatic microsomes. Br J Pharmacol. 2010;161:1059–1069. doi: 10.1111/j.1476-5381.2010.00946.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Coutre P, Kreuzer KA, Pursche S, Bonin M, Leopold T, Baskaynak G, Dörken B, Ehninger G, Ottmann O, Jenke A, Bornhäuser M, Schleyer E. Pharmacokinetics and cellular uptake of imatinib and its main metabolite CGP74588. Cancer Chemother Pharmacol. 2004:53;:53;:53;313–323. doi: 10.1007/s00280-003-0741-6. [DOI] [PubMed] [Google Scholar]
- 82.Preissner S, Simmaco M, Gentile G, Preissner R. Personalized cancer therapy considering cytochrome P450 variability. Adv Pharmacol. 2015;74:113–130. doi: 10.1016/bs.apha.2015.03.004. [DOI] [PubMed] [Google Scholar]
- 83.Moore LB, Goodwin B, Jones SA, Wisely GB, Serabjit-Singh CJ, Willson TM, Collins JL, Kliewer SA. St. John’s wort induces hepatic drug metabolism through activation of the pregnane X receptor. Proc Natl Acad Sci U S A. 2000;97:7500–7502. doi: 10.1073/pnas.130155097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Li Y, Ross-Viola JS, Shay NF, Moore DD, Ricketts M. Human CYP3A4 and murine CYP3A11 are regulated by equol and genistein via the pregnane X receptor in a spesies-specific manner. J Nutr. 2009;139:898–904. doi: 10.3945/jn.108.103572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Froyen EB, Reeves JLR, Mitchell AE, Steinberg FM. Regulation of phase II enzymes by genistein and daidzein in male and female Swiss Webster mice. J Med Food. 2009;12:1227–1237. doi: 10.1089/jmf.2009.0084. [DOI] [PubMed] [Google Scholar]
- 86.Pfeiffer E, Treiling CR, Hoehle SI, Metzler M. Isoflavones modulate the glucuronidation of estradiol in human liver microsomes. Carcinogenesis. 2005;26:2172–2178. doi: 10.1093/carcin/bgi197. [DOI] [PubMed] [Google Scholar]
- 87.Galijatovic A, Walle UK, Walle T. Induction of UDP-glucuronosyltransferase by the flavonoids chrysin and quercetin in caco-2 cells. Pharm Res. 2000;17:21–26. doi: 10.1023/a:1007506222436. [DOI] [PubMed] [Google Scholar]
- 88.Mao Q, Lai Y, Wang J. Drug transporters in xenobiotic disposition and pharmacokinetic prediction. Drug Metab Dispos. 2018;46:561–566. doi: 10.1124/dmd.118.081356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Lee SC, Arya V, Yang X, Volpe DA, Zhang L. Evaluation of transporters in drug development: Current status and contemporary issues. Adv Drug Deliv Rev. 2017;116:100–118. doi: 10.1016/j.addr.2017.07.020. [DOI] [PubMed] [Google Scholar]
- 90.Hagenbuch B, Meier PJ. The superfamily of organic anion transporting polypeptides. Biochim Biophys Acta Biomembr. 2003;1609:1–18. doi: 10.1016/s0005-2736(02)00633-8. [DOI] [PubMed] [Google Scholar]
- 91.Russel FGM, Masereeuw R. Molecular aspects of renal anionic drug transport. Annu Rev Physiol. 2002;64:563–594. doi: 10.1146/annurev.physiol.64.081501.155913. [DOI] [PubMed] [Google Scholar]
- 92.Jonker JW, Schinkel AH. Pharmacological and physiological functions of the polyspecific organic cation transporters: OCT1, 2, and 3. J Pharmacol Exp Ther. 2004;308:2–9. doi: 10.1124/jpet.103.053298. [DOI] [PubMed] [Google Scholar]
- 93.Shugarts S, Benet LZ. The role of transporters in the pharmacokinetics of orally administered drugs. Pharm Res. 2009;26:2039–2054. doi: 10.1007/s11095-009-9924-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Robey RW, Pluchino KM, Hall MD, Fojo AT, Bates SE, Gottesman MM. Revisiting the role of ABC transporters in multidrug-resistant cancer. Nat Rev Cancer. 2018;18:452–464. doi: 10.1038/s41568-018-0005-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Liu W, Meng Q, Sun Y, Changyuan W, Huo X, Liu Z, Sun P, Sun H, Ma X, and Liu K. Targeting P-glycoprotein : nelfinavir reverses adriamycin resistance in K562/ADR cells. Cell Physiol Biochem. 2018;51:1616–1631. doi: 10.1159/000495650. [DOI] [PubMed] [Google Scholar]
- 96.Ballent M, Lifschitz A, Virkel G, Sallovitz J, Maté L, Lanusse C. In vivo and ex vivo assessment of the interaction between ivermectin and danofloxacin in sheep. Vet J. 2012;192:422–427. doi: 10.1016/j.tvjl.2011.09.006. [DOI] [PubMed] [Google Scholar]
- 97.Mealey KL. ABCG2 transporter: therapeutic and physiologic implications in veterinary species. J Vet Pharmacol Ther. 2011;35:105–112. doi: 10.1111/j.1365-2885.2011.01313.x. [DOI] [PubMed] [Google Scholar]
- 98.Naya M, Imai M. Recent advances on soybean isoflavone extraction and enzymatic modification of soybean oil. London: IntechOpen; 2013:1–24. [Google Scholar]
- 99.Merino G, Perez M, Real R, Egido E, Prieto JG, Alvarez AI. In vivo inhibition of BCRP/ABCG2 mediated transport of nitrofurantoin by the isoflavones genistein and daidzein: A comparative study in Bcrp1-/- mice. Pharm Res. 2010;27:2098–2105. doi: 10.1007/s11095-010-0208-5. [DOI] [PubMed] [Google Scholar]