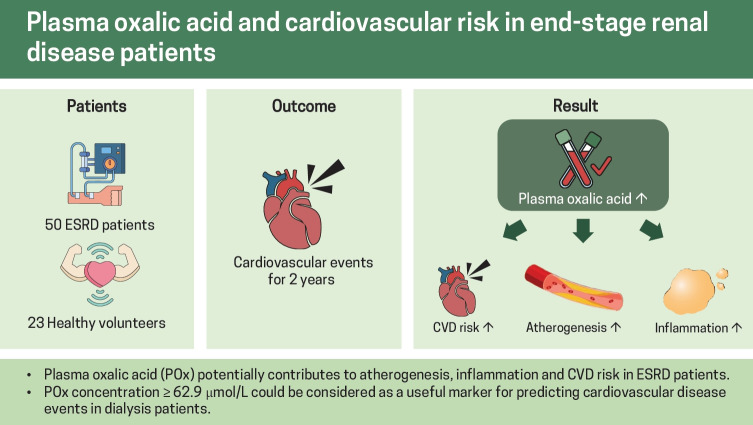
Abstract
Background/Aims
It was hypothesized that oxalate might be strongly involved in atherogenesis and the inflammatory pathway that could result in an increased risk of cardiovascular disease (CVD) in end-stage renal disease (ESRD) patients. Therefore, this study aimed to address two primary research questions: to characterize the lipid profile and the pattern of pro-inflammatory cytokines according to plasma oxalic acid (POx) concentration in ESRD patients; to evaluate the potential role of elevated POx concentration in the development of CVD risk.
Methods
A total of 73 participants were enrolled in this prospective, observational cohort pilot study. Among them, there were 50 ESRD patients and 23 healthy volunteers. The lipid profile and the pro-inflammatory cytokines were analyzed according to the distribution of POx concentration into tertiles. After the clinical examination, 29 hemodialysis patients and 21 peritoneal dialysis patients without prevalent CVD were observed for CVD events for 2 years. The Cox regression analysis and a set of different types of sensitivity analyses were used to determine whether elevated POx was associated with an increased risk of CVD.
Results
An increasing trend in the atherogenic lipoprotein fractions and the pro-inflammatory markers as well as a linear decrease in high-density lipoprotein was significantly associated with elevated POx. POx concentration ≥ 62.9 μmol/L was significantly associated with CVD events independently of other examined CVD risk factors.
Conclusions
This pilot study firstly demonstrated a potential contribution of POx to atherogenesis, inflammation and CVD risk in ESRD patients.
Keywords: Oxalic acid, End-stage kidney disease, Dyslipidemia, Atherosclerosis
Graphical abstract
INTRODUCTION
Cardiovascular disease (CVD) has consistently remained the leading cause of morbidity and mortality in end-stage renal disease (ESRD) patients [1–3]. Although many traditional and non-traditional risk factors have been identified [2], CVD prevalence still continues to be 20 times higher in ESRD patients than in the general population [1].
Atherogenic dyslipidemia and chronic inflammation have been considered major risk factors for cardiovascular (CV) morbidity and mortality in ESRD patients [1,4,5]. In turn, changes in the balance of oxalate have been reported to be associated with dyslipidemia [6], systemic inflammation [7–9], and thus a high risk of CVD independent of its cause [10,11]. It has been demonstrated that plasma oxalic acid (POx) concentration increases according to the progression of chronic kidney disease (CKD) and reaches its highest level in ESRD patients [12,13].
Moreover, recently, it has been speculated that oxalate is a uremic toxin originating from the colonic microbial metabolism, which indirectly suggests its contribution to systemic inflammation associated with CKD [14]. In addition, in a series of early in vitro studies, Levin et al. [15] and Recht et al. [16] have provided the first evidence that oxalate may be an atherogenic toxin in uremic conditions. Although hyperoxalemia is a well-known occurrence in ESRD patients, the clinical data on the association between POx and CV outcomes in the dialysis population has never been analysed. Considering the above, we therefore hypothesized that oxalate might be strongly involved in atherogenesis and the inflammatory pathway and could result in an increased risk of CVD in ESRD patients.
To address this issue, this prospective, observational, single-centre pilot study was primarily designed to characterize the lipid profile and the pattern of pro-inflammatory cytokines according to POx concentration and to evaluate the potential role of elevated POx in the development of CVD risk. We also aimed to test the feasibility of the study for future large-scale research.
METHODS
Study design
This prospective, observational cohort pilot study was part of an ongoing institute project titled “Effect of oxalate and urate metabolism on the evolution of kidney disease” (ClinicalTrials.gov Identifier: NCT04399915, Domestic Trial Registration Number 0119U000002). The study was carried out in accordance with the Declaration of Helsinki and was conducted between January 2018 and May 2020. The study protocol was confirmed by the Ethics Committee of the Institute (Protocol Number: 8/2017 from September 19, 2017). Written informed consent was obtained from all participants before enrolling in the study.
Sample size
Due to the exploratory nature of this study with several different aims and the fact that an association between POx concentration and CV events in ESRD patients has never been studied before, the estimation of the appropriate sample size was based only on some of the issues being explored. Previous studies on assessing the differences in POx concentration between ESRD patients and healthy controls and on the association between POx concentration and pro-inflammatory mediators in peritoneal dialysis effluent have reported effect sizes of 0.73 to 2.52 and correlation effect sizes of 0.7 to 0.83 based on sample sizes ranging from 11 to 30 [9,14,17]. Therefore, the required sample size was estimated based on the previously mentioned studies using MedCalc Statistical Software (MedCalc Software Ltd., Ostend, Belgium). A minimum sample size of 21 participants in each group would be required to achieve power of 0.80 and an alpha of 0.05 to detect the differences between the groups using the Student’s t test or the nonparametric Mann-Whitney test. Similarly, a minimum sample size of 23 participants would be required to achieve power of 0.80 and an alpha of 0.05 in the correlation analysis. Taking into account the recommended sample size of at least 50 participants for a pilot study [18], we decided to enroll 50 ESRD patients and 23 healthy volunteers in the study.
Participants
A total of 73 participants were enrolled in the study, including 50 ESRD patients without prevalent CVD and 23 healthy volunteers on a free-choice diet who served as a control reference group to evaluate POx concentration. Among the ESRD patients were 29 hemodialysis (HD) patients and 21 peritoneal dialysis (PD) patients.
Inclusion criteria
The criteria for enrollment in the study were age > 18 years, dialysis treatment for at least 3 months, being in a clinically stable condition and having adequately functioning arteriovenous fistula or peritoneal access. In addition, the target level of Kt/V was ≥ 1.4 for the HD patients and weekly Kt/V ≥ 1.7 for the PD patients. Moreover, the enrolled patients did not take antibiotics and/or probiotics for at least 3 months before enrolling in the study.
Exclusion criteria
The exclusion criteria were hospitalization for any reason in the preceding 3 months, prevalent CVD (myocardial infarction, stroke, heart failure or left ventricular ejection fraction ≤ 40%), kidney stones, actual peritonitis, systemic and malignant diseases, acute inflammation processes and immunosuppressive treatment.
Dialysis prescription
All participants underwent their routine prescribed dialysis treatment. The HD patients were routinely dialyzed with bicarbonate-based dialysate, volumetric ultrafiltration control, single-use synthetic (polysulphone) dialyzers at a median blood flow rate of 300 mL/min and a dialysate flow rate of 500 mL/min for 4 hours per session three times a week. Heparin was used as a standard anticoagulant. HD therapy was performed, setting the target single-pool Kt/Vurea ≥ 1.4 in accordance with the National Kidney Foundation Kidney Disease Outcomes Quality Initiative Clinical Practice Guidelines for Hemodialysis Adequacy.
The PD patients were treated with continuous ambulatory PD with usual dwell time (4 to 5 hours during the daytime and 8 to 10 hours at night). All patients received commercially available glucose-based Dianeal PD solution (Baxter Healthcare SA, Castlebar, Ireland) of various strengths (1.36%, 2.27%) and Icodextrine. Dialysis prescription was guided by the target to achieve a value of weekly Kt/V ≥ 1.7 in accordance with the National Kidney Foundation Kidney Disease Outcomes Quality Initiative Clinical Practice Guidelines for Peritoneal Dialysis Adequacy.
Clinical and routine laboratory measurements
Demographics and clinical and laboratory data were obtained during routine clinical practice immediately after enrolling the participants in the study. All blood samples were collected after an overnight fast during the longest dialytic interval for the HD patients and at the time of the routine outpatient visit for the PD patients. The blood samples were processed immediately after sampling.
Routine biochemical parameters, including blood urea nitrogen and creatinine, serum albumin, C-reactive protein (CRP), glucose, electrolytes and the lipid profile parameters, were determined using an automatic Flexor Junior analyser (Vital Scientific, Spankeren, the Netherlands). Hematological parameters of blood were determined using an ABX Micros-60 (Horiba Medical, Montpellier, France).
The lipid profile parameters included triglyceride (TG), total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), and very-low-density lipoprotein cholesterol (VLDL-C). The atherogenic index of plasma (AIP) was calculated from plasma TG and HDL-C (log [TG/HDL-C]). Atherogenic dyslipidemia was defined as a combination of low plasma levels of LDL-C ≥ 2.59 mmol/L and elevated TG ≥ 2.26 mmol/L according to Kidney Disease Outcomes Quality Initiative (KDOQI) Clinical Practice Guidelines for Managing Dyslipidemias in Chronic Kidney Disease [19]. Parathyroid hormone (PTH) was measured using an immunoradiometric assay, and electrolytes were measured using standard autoanalyzer techniques. Body mass index (BMI) was calculated as weight in kilograms divided by the square of the height in meters.
Measurement of POx concentration
POx concentration was measured spectrophotometrically using a commercially available kit (MAK315, Sigma, Barcelona, Spain). The plasma samples were immediately frozen and stored at −20°C in the freezer until further analysis, according to the manufacturers’ protocols. Predialysis plasma samples were collected from the HD patients.
Measurements of immune mediators
Interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α) and monocyte chemoattractant protein-1 (MCP-1) concentrations were detected in serum using STAT FAX-303 PLUS and commercially available enzyme-linked immunosorbent assay (ELISA) test kits (Diaclon, Besançon, France; DRG, Marburg, Germany; Ukr medservice, Kyiv, Ukraine), according to the manufacturers’ protocols.
Endpoint and definition of CV events
After routine clinical laboratory testing and the collection of blood samples, the patients were monitored and assessed for CV events for 2 years (until May 2020). The average duration of dialysis therapy at study entry (at the time of baseline data) was 30 months (20 to 78 months). CV events were defined as newly diagnosed angina, myocardial infarction, stroke, heart failure or peripheral artery diseases requiring hospitalization.
Statistical analysis
The statistical analysis and all graphs were done using MedCalc and the XLSTAT software (Addinsoft, New York, NY, USA). The average mean ± standard deviations or the median (interquartile range) were calculated according to the standard normal distribution. For the statistical analysis, we used the Student’s t test and nonparametric (U test) Mann-Whitney or Kruskal-Wallis tests. Categorical variables were expressed as proportions. Chi-square tests were used to compare two groups. A Spearman correlation test and the partial correlation coefficient adjusted for age, sex, diabetic status and BMI were used to evaluate the association between POx concentration, the lipid profile parameters and the pro-inflammatory biomarkers.
A Kaplan-Meier analysis was used to evaluate the differences in CV event rates according to POx concentration; the curves were compared using a log-rank test. In addition, Cox proportional hazard regression models were used to adjust for the confounding effects of numerous factors associated with CVD. The analysis was initially performed without adjustment and then performed again after adjusting for several covariates. The detailed justification of the covariates selected for the models is provided in Supplementary methods (Justification of the covariates selected for the Cox proportional hazard regression models) and Supplementary Table 1. Finally, two adjusted models were represented: (1) Model 1 adjusted for age, sex and dialysis modality and (2) Model 2 adjusted for CVD risk factors, including diabetic status, systolic blood pressure, BMI, serum uric acid, hemoglobin (Hb), TG, and IL-6 in addition to the covariates of Model 1. The models were censored at CV events, death or the end of the follow-up period. The covariates included in this data set had no missing data. Hazard ratios (HRs) and 95% confidence intervals (CIs) were obtained using the Cox proportional hazards regression models.
Different types of sensitivity analyses were performed to evaluate the validity of our findings due to the small sample size of the entire cohort (Supplementary methods, Sensitivity analysis). The authors confirm that all data underlying the findings are fully available without restriction from Mendeley Dataset at http://dx.doi.org/10.17632/d2fg9cpts3.1.
RESULTS
Baseline characteristics of the study participants
A total of 50 ESRD patients (29 HD and 21 PD patients) and 23 healthy volunteers were enrolled in the study at our centre. The participants comprised 39 (53.4%) males and 34 (46.6 %) females. The mean age of all participants was 48.4 ± 13.6 years. The baseline characteristics of the study participants are shown in Table 1.
Table 1.
Demographic and clinical characteristics of the healthy volunteers and the ESRD patients
| Parameter | Healthy volunteers (n = 23) | A total of dialysis patients (n = 50) | p value |
|---|---|---|---|
| Clinical parameters | |||
| Male sex | 9 (39.1) | 30 (60) | 0.092 |
| Age, yr | 45.9 ± 10.9 | 48.4 ± 13.6 | 0.181 |
| Dialysis vintage at this study entry, mo | - | 30 (20–78.5) | - |
| Diabetics | - | 10 (20) | - |
| Body mass index, kg/m2 | 27.1 ± 4.0 | 25.9 ± 4.4 | 0.672 |
| Anuria | - | 17 (34) | - |
| RRF, mL/min/1.73 m2 | 72.0 (63.0–77.8) | 5.0 (4.0–6.0) | < 0.001 |
| Serum albumin, g/L | 43.4 (41.1–45.1) | 40.0(36.6–40.9) | < 0.001 |
| Total blood protein, g/L | 69.6 ± 4.7 | 63.7 ± 7.53 | < 0.001 |
| Systolic blood pressure, mmHg | 115 (100–125) | 140 (134–145) | < 0.001 |
| Diastolic blood pressure, mmHg | 79.0 (66.2–82.0) | 90 (85–90) | < 0.001 |
| Hemoglobin, g/L | 126.3 ± 11.5 | 110.2 ± 16.9 | < 0.001 |
| Glucose, mmol/L | 5.3 (4.9–5.5) | 5.2 (4.2–5.7) | 0.377 |
| Calcium, mmol/L | 2.4 (2.3–2.5) | 2.3 (2.22–2.46) | 0.022 |
| Phosphorus, mmol/L | 1.1 ± 0.1 | 1.9 ± 0.7 | < 0.001 |
| iPTH, ng/L | - | 294.5 (184.0–437.0) | |
| Uric acid, mmol/L | 266 (162.8–375.6) | 326.5 (278.8–387.7) | 0.008 |
| Lipid profile parameters | |||
| Totalcholesterol, mmol/L | 4.9 ± 0.96 | 5.6 ± 1.6 | 0.083 |
| Triglyceride, mmol/L | 1.1 (0.8–1.6) | 1.6 (1.16–2.5) | 0.015 |
| LDL-C, mmol/L | 2.5 ± 1.0 | 3.18 ± 0.38 | 0.013 |
| VLDL-C, mmol/L | 0.55 (0.3–0.86) | 0.7 (0.45–1.2) | 0.021 |
| HDL-C, mmol/L | 1.55 (1.3–1.68) | 1.2 (1.03–1.4) | 0.001 |
| AIP | 2.6 ± 0.8 | 3.8 ± 1.4 | < 0.001 |
| Pro-inflammatory markers | |||
| CRP, mg/L | 4.9 (3.7–9.9) | 11.6 (7.6–15.5) | 0.001 |
| Interleukin 6, pg/mL | 0 | 2.17 (0.2–5.1) | - |
| TNF-α, pg/mL | 0.1 (0–2.1) | 1.2 (0.3–2.3) | 0.011 |
| MCP-1, pg/mL | 188 (114.5–215.5) | 318.7 (278–378.1) | < 0.001 |
Values are presented as number (%), mean ± standard deviation, or median (interquartile range). The values are compared between the groups using the chi-square tests, the Student’s t test and the Mann-Whitney U test as appropriate.
ESRD, end-stage renal disease; RRF, renal residual function; iPTH, intact parathyroid hormone; LDL-C, low-density lipoprotein cholesterol; VLDL-C, very-low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; AIP, atherogenic index of plasma; CRP, C-reactive protein; TNF-α, tumor necrosis factor-alpha; MCP-1, monocyte chemoattractant protein-1.
As presented in Table 1, no significant differences in age or sex distribution were observed between the control and the dialysis groups. However, as expected, significantly higher levels of all examined pro-inflammatory markers, TG and the atherogenic lipoprotein fractions were observed in the dialysis patients compared with the healthy volunteers. In particular, the blood concentration of IL-6 was undetectable in the healthy volunteers.
Association between POx concentration, the lipid profile and the pro-inflammatory markers
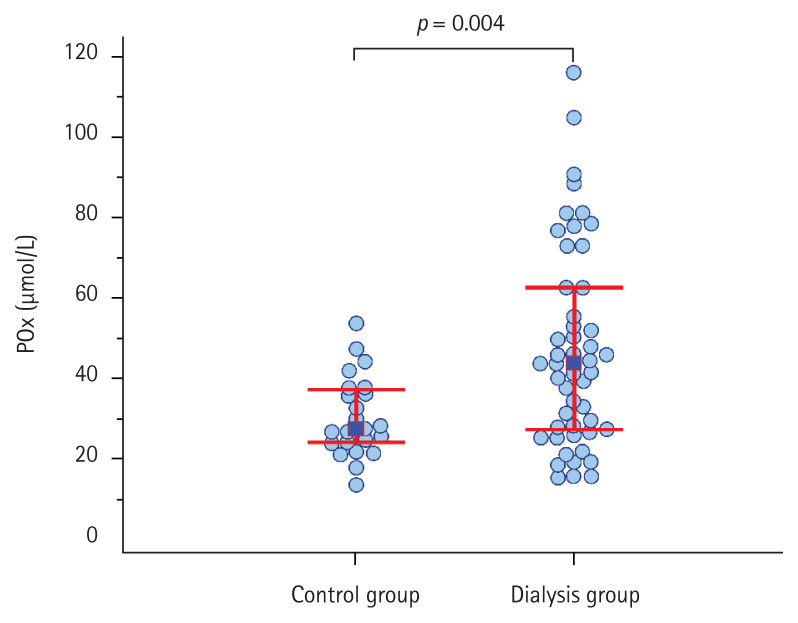
POx concentration ranged from 15.7 to 116.2 μmol/L and was significantly higher in the ESRD patients compared with the control group at the time of entering the study (Fig. 1). POx tended to be higher in the PD patients compared with the HD patients. However, there was no statistically significant difference between the groups (41.5 μmol/L [28.2 to 53.6] vs. 46.5 μmol/L [26.0 to 65.4], respectively; p = 0.73) (Supplementary Fig. 1). Moreover, POx concentration was directly associated with the serum calcium level (r = 0.28, p = 0.04) and had an inverse correlation with the residual renal function in the patients (r = −0.43, p = 0.004). However, no difference was found in POx between the anuric patients and the ESRD patients with preserved diuresis (44.6 μmol/L [32.1 to 73.9] vs. 42.8 μmol/L [26.8 to 52.2]; p = 0.48).
Figure 1.
Plasma oxalic acid (POx) concentration in the end-stage renal disease patients and the control group.
To assess the clinical significance of POx in atherogenesis and inflammation, we analysed the lipid profile parameters and the pro-inflammatory markers in the dialysis patients according to the distribution of POx into tertiles, as presented in Fig. 1.
The analysis demonstrated a gradually increasing trend in the majority of the examined parameters according to the tertiles of POx. The dialysis patients in the upper tertile of POx concentration had higher levels of TC, TG, VLDL-C, AIP, IL-6, TNF-α and MCP-1 with a corresponding decreasing trend in HDL-C compared with those in the middle and the tertiles of POx (Table 2).
Table 2.
Baseline blood lipid profile and the pro-inflammatory markers data according to the tertiles of POx in the ESRD patients
| Variable | POx concentration in the ESRD patients | p value | ||
|---|---|---|---|---|
| Tertile 1 (≤ 27.7 μmol/L) (n = 13) | Tertile 2 (27.8–62.9 μmol/L) (n = 25) | Tertile 3 (≥ 63 μmol/L) (n = 12) | ||
| Lipid profile parameters | ||||
| Total cholesterol, mmol/L | 5.2 (4.2–5.7) | 5.07 (4.4–5.5) | 6.6 (5.9–6.9) | 0.052 |
| Triglycerides, mmol/L | 1.28 (0.87–2.02)c | 1.42 (1.08–1.84)c | 2.8 (1.9–3.1)a,b | 0.002 |
| LDL-C, mmol/L | 3.1 (1.7–3.8) | 2.7 (2.2–3.3) | 3.5 (2.4–3.7) | 0.196 |
| VLDL-C, mmol/L | 0.51 (0.35–0.69)c | 0.64 (0.43–0.88)c | 0.94 (0.63–1.2)a,b | 0.022 |
| HDL-C, mmol/L | 1.44 (1.12–1.66)c | 1.31 (1.2–1.61)c | 1.01 (0.97–1.14)a,b | 0.007 |
| AIP | 2.9 (2.1–3.3)c | 3.1 (2.2–4.0)c | 4.1 (3.7–4.4)a,b | 0.026 |
| Pro-inflammatory markers | ||||
| CRP, mg/L | 7.6 (4.1–11.4) | 11.6 (4.8–15.1) | 11.7 (8.4–14.9) | 0.355 |
| Interleukin 6, pg/mL | 1.1 (0–5.8)c | 1.9 (0.5–4.7)c | 15.9 (8.8–21.1)a,b | 0.006 |
| TNF-α, pg/mL | 2.1 (1.1–4.3)b | 0.16 (0–0.8)a,c | 1.8 (0.5–2.8)b | < 0.001 |
| MCP-1, pg/mL | 254 (219.6–300)b,c | 331.2 (289.5–361.5)a,c | 402 (300–503.8)a,b | 0.001 |
Values are presented as median (interquartile range) and compared between the groups using the Kruskal-Wallis test.
POx, plasma oxalic acid; ESRD, end-stage renal disease; LDL-C, low-density lipoprotein cholesterol; VLDL-C, very-low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; AIP, atherogenic index of plasma; CRP, C-reactive protein; TNF-α, tumor necrosis factor-alpha; MCP-1, monocyte chemoattractant protein-1.
The groups of POx tertiles in the ESRD patients with the statistical difference p < 0.05.
The Spearman correlation analysis indicated a direct association between POx concentration and blood TG (r = 0.56, p <0.0001), AIP (r = 0.34, p < 0.0001), IL-6 (r = 0.49, p < 0.0001), and MCP-1 levels (r = 0.55, p < 0.0001). The Spearman correlation matrix is shown in Supplementary Table 2. Roughly similar results to those observed for the Spearman correlation analysis were obtained from the partial correlation analysis adjusted for age, sex, diabetic status and BMI; POx concentration remained directly associated with TG (r = 0.54, p = 0.0001), IL-6 (r = 0.51, p = 0.0003), and MCP-1 (r = 0.47, p = 0.0009). However, no association was observed between POx and AIP (r = 0.2, p = 0.19) in the partial correlation test.
POx concentration and CV events
A total of eight (16%) ESRD patients experienced a CV event during the 2-year follow-up period. Of these, seven (14 %) patients had a non-fatal CV event and one (2%) patient died due to a stroke. Non-fatal CV events included newly diagnosed angina (three cases, 42.8%), heart failure (two cases, 28.6%), acute coronary syndrome (one case, 14.3%) and cardiac arrhythmia (one case, 14.3%).
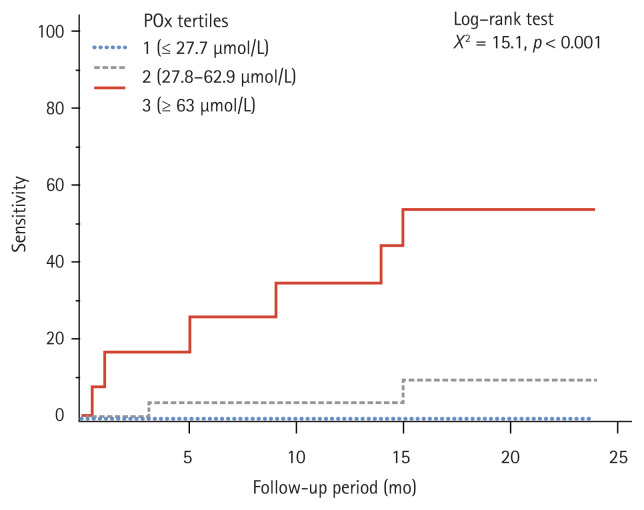
The comparison of the proportions of CV events across the tertiles of POx indicated that POx encompassed the highest number of CV events in the upper tertile compared with the middle and the low tertiles. The event rates across the tertiles of POx were six cases (50%) in the patients in tertile 3 and two cases (8%) in the patients in tertile 2. There were no cases of CV events in the patients in tertile 1 (χ2 = 13.9, p < 0.001).
In order to explore the association between POx and CV events, we further determined the average POx concentration levels depending on experienced CV events and plotted the Kaplan-Meier curves according to the tertiles of POx. POx concentration was significantly higher in the group with CV events compared with the group without CV events (74.9 μmol/L [59.5 to 78.4] vs. 40.9 μmol/L [26.2 to 50.7]; p = 0.007). POx stratified according to CV events in the ESRD patients during the 2-year follow-up period and the cumulative frequency distribution of POx are detailed in Supplementary Figs. 2 and 3.
Fig. 2 illustrates a significantly higher CV risk in the dialysis patients with POx concentration ≥ 63 μmol/L (tertile 3) compared with the patients in tertile 1 and tertile 2 (log-rank test p = 0.0005). The Cox proportional hazard regression model was used to control the confounding effects of the factors associated with CVD. After adjusting the model for the effects of the potential confounding factors previously described, POx remained a significant risk factor for CV events in the ESRD patients (Table 3). Subgroup analysis was not performed due to the small sample size of the entire cohort.
Figure 2.
The 2-year cumulative cardiovascular event rates stratified according to the tertiles of plasma oxalic acid (POx) concentration in the end-stage renal disease patients.
Table 3.
Association between POx concentration and CV events in the ESRD patients
| Variable | Value | SD | Wald χ2 | p value | HR (95% CI) |
|---|---|---|---|---|---|
| Unadjusted | 0.028 | 0.012 | 5.808 | 0.016 | 1.03 (1.005–1.05) |
| Model 1 | 0.074 | 0.023 | 10.732 | 0.001 | 1.1 (1.46–1.15) |
| Model 2 | 0.131 | 0.037 | 12.642 | < 0.001 | 1.14 (1.07–1.22) |
Model 1 was adjusted for age, sex, and dialysis modality; Model 2 was additionally adjusted for cardiovascular disease risk factors, including diabetic status, systolic blood pressure, body mass index, serum uric acid, and variables that were statistically significant in the univariate analysis (hemoglobin, triglycerides, interleukin-6).
POx, plasma oxalic acid; CV, cardiovascular; ESRD, end-stage renal disease; SD, standard error; HR, hazard ratio; CI, confidence interval.
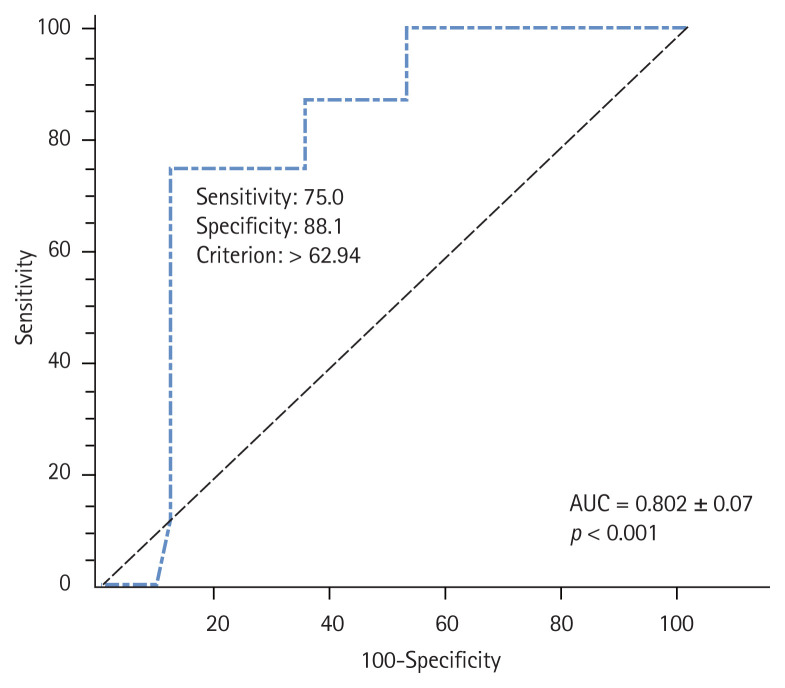
The receiver operating characteristic (ROC) analysis indicated that the most appropriate cut-off point for POx concentration as a predictor for CV events in the dialysis patients was 62.9 μmol/L with sensitivity of 75% and specificity of 88.1% (Fig. 3).
Figure 3.
Receiver operating characteristic curve for the cut-off value of plasma oxalic acid concentration to predict cardiovascular events in the end-stage renal disease patients. AUC, area under the curve.
The area under the ROC curve (AUROC) was 0.80 (95% CI, 0.86 to 0.90; p < 0.0001) (Supplementary Table 3). The AUROC for CV endpoint events was 0.94 (95% CI, 0.84 to 0.99; p = 0.0001) and 0.93 (95% CI, 0.83 to 0.98; p = 0.0007) in Model 1 and Model 2, respectively. The sensitivity analysis with a focus on the cumulative incidence function demonstrated that Gray’s test correctly detected the difference in the cumulative incidence of CV events according to POx concentration in the ESRD patients (≥ 62.9 μmol/L vs. < 62.9 μmol/L; Gray’s χ2 = 11.2; p = 0.001) (Supplementary Fig. 4). We examined the change in HR in the newly created dyslipidemia-adjusted model and found that the results were similar to those of our main analysis (Table 3). We also found a significant association between POx concentration and CV events (HR, 1.094; 95% CI, 1.04 to 1.15; p = 0.001). The sensitivity analysis excluding the diabetics produced HR of 1.09 (95% CI, 1.02 to 1.17; Wald χ2 = 6.8 ± 0.03; p = 0.009); this remained qualitatively unchanged from the results obtained from Model 2 in the primary analysis.
Finally, the effect size analysis for the Cox regression result demonstrated the risk of committing a type II error of 19.1% and power of 80.9% with an alpha of 0.05 and the sample size of 50 observations. However, a minimum of 41 CVD cases would be required for the analysis to achieve power of 0.90 and alpha of 0.05.
DISCUSSION
Oxalate is an ionic form of a potentially toxic oxalic acid that is primarily eliminated by the kidneys [8,13]. Loss of kidney function leads to a decrease in renal oxalate clearance and hyperoxalemia in ESRD patients [11]. The accumulation of oxalate is associated with inflammation [8,9,13], high risk factors for CVD [20,21] and an increased mortality rate [8,10] in kidney stone disease patients. However, oxalate has never been investigated as a potential contributor to atherosclerosis, systemic inflammation and CVD risk in ESRD patients. To our knowledge, this is the first study to prospectively evaluate the association between POx concentration and CVD events in the dialysis cohort during a 2-year follow-up period using multivariable Cox regression analysis and a set of sensitivity analyses. Nevertheless, atherogenic dyslipidemia and systemic inflammation are proven risk factors for CVD incidence in ESRD patients and can be important independent confounders. Therefore, in the present study we separately characterized the association between both the lipid profile and the pro-inflammatory markers and POx concentration, which has never been reported before.
Our study resulted in a number of new findings. First, an increasing trend in the atherogenic lipoprotein fractions and the pro-inflammatory markers and a linear decrease in HDL-C was significantly associated with elevated POx concentration. Second, POx concentration was directly associated with blood TG, AIP, IL-6, and MCP-1 levels. Third, POx concentration ≥ 62.9 μmol/L was significantly associated with experienced CVD events independent of other examined CVD risk factors and can be considered a useful marker for predicting CVD events in dialysis patients.
Numerous studies have demonstrated increased CVD morbidity in kidneys stone formers [10,20,21]. Recently, several hypotheses have been proposed to explain the association between kidney stones and CVD: (1) urinary stones and CVD have similar traditional atherosclerotic risk factors, including diabetes, hypertension, metabolic syndrome and obesity; (2) a common origin of both diseases is based on the response-to-injury mechanism, including oxidative stress and inflammation; and (3) kidney stone disease leads to renal function decline, which in turn could increase CV morbidity and mortality [6,8,11,20,22]. In our opinion, all hypotheses mentioned above could doubtless be extrapolated to indicate an association between elevated POx and CVD in ESRD patients.
A wide variety of scientific data indicate that there are common risk factors for calcium oxalate (CaOx) stone diseases and CVD. It has been demonstrated that both diseases are significantly associated with older age, diabetic status, smoking habits, hypertension, hypercholesterolemia, hyperlipidemia, and obesity in these patients compared with controls, and this association remains significant after adjusting for multiple potential confounders [10,23,24].
Apart from the traditional factors mentioned above, there are a number of other uremia-related CVD risk factors in ESRD patients, such as anemia, mineral and bone disorders, malnutrition, overhydration and electrolyte imbalance, that in turn aggravate the prognosis [1,25,26]. We did not focus on well-known risk factors of CVD in our study. However, except for POx concentration, roughly similar confounders for the risk of CVD events (age, diabetes, Hb, TG, BMI, iPTH, and IL-6) were found among baseline parameters in our sample (Supplementary Table 1).
In the present study, we focused on POx concentration and its potential role in dyslipidemia and systemic inflammation associated with CVD risk in ESRD patients. It is somewhat surprising that notwithstanding the fact that the comparative analysis clearly identified a gradually increasing trend in the atherogenic lipoprotein fractions associated with elevated POx, the partial correlation analysis indicated that POx was significantly associated only with TG. It should be emphasized that it was difficult to compare the results of our study with the results of previous findings, as most studies in this field investigated only the association between dyslipidemia and the presence of nephrolithiasis [24,27]. It is unfortunate that the authors did not take into account POx concentration. To the best of our knowledge, in contrast to our findings, a negative association between POx concentration and LDL-C in HD patients was reported in only one study [28].
The available data indicate that oxidative stress and inflammation are the major risk factors for accelerated atherosclerosis [6,29]. Reactive oxygen species oxidize LDL-C to minimally modified LDL-C (mm-LDL-C), which induces the secretion of various cytokines, including MCP-1 [6,30]. In turn, MCP-1 is directly involved in monocyte recruitment, resulting in transformation into foam cells due to modified lipoprotein. Therefore, MCP-1 is one of the earliest events in the pathogenesis of atherosclerosis [30]. In addition to MCP-1, oxidatively modified LDL-C induces the synthesis of pro-inflammatory cytokines, such as IL-6 and TNF-α [30,31]. On the other hand, these mediators are strongly involved in oxalate-induced inflammation [32–34]. A crucial role of MCP-1, IL-6, and TNF-α in the oxalate-induced inflammatory response of the kidneys has been demonstrated in early in vitro and experimental studies [33–35]. In a recent in vitro study, Dominguez-Gutierrez et al. [7] found that CaOx could activate human monocytes, which promoted M1 macrophage development and produced the inflammatory response. Monocytes or neutrophils exposed to oxalate crystals produce TNF-α, IL-6, and MCP-1. As a result, these identical inflammatory pathways in atherosclerosis and hyperoxalemia became the main reason for the initial selection of this cytokine panel in our study. It is unfortunate that clinical studies on the association between POx and cytokine concentrations in ESRD patients have never been performed. Therefore, the results obtained from our study could not be directly compared with the results of previous reports. Nevertheless, in support of the previously mentioned studies, our findings have provided preliminary clinical evidence that elevated POx concentration is associated with increased levels of IL-6, TNF-α, and MCP-1 in ESRD patients. However, it should be noted that the correlation analysis did not confirm the association between POx and serum TNF-α, while the univariate Cox regression analysis substantiated the association between POx and the only pro-inflammatory marker, IL-6. Moreover, we did not observe an association between POx and CRP. The obtained results contradict the existing data, including that for patients with nephrolithiasis [8], and require further clarification. Possible explanations for the contradictory results may be the clinically stable condition of the patients during the entire 2-year follow-up period and the small sample size.
Several studies have found that kidney stone disease is strongly associated with CKD progression and ESRD and that both diseases can lead to CVD [36,37]. POx has been defined as a significant predictor of kidney function decline not only in nephrolithiasis but also in primary hyperoxaluria and enteric hyperoxaluria [12,13]. Moreover, Mulay et al. [38] have identified numerous clinically important CKD complications, including hyperphosphatemia, hyperparathyroidism, hypertension and cardiac fibrosis, in the proposed experimental oxalate diet-induced model of CKD. Thus, both POx and uremia-related complications could lead to CVD and accelerate the process in ESRD patients. A statistically significant association between POx and CVD events during the 2-year follow-up period was demonstrated in the present study. It is important to note that this association remained significant after adjusting the model for the effects of the potential confounding factors previously described.
According to our results, POx concentration ≥ 62.9 μmol/L can predict CVD risk independent of CKD-related factors in dialysis patients. Our data are consistent with the findings of the following two studies. In a very early study, Tomson et al. [39] demonstrated a significant association between POx and vascular calcification in HD patients. More recently, Gulhan et al. [28] found that aortic pulse wave velocity was independently predicted by POx, glucose and TG. This finding undoubtedly confirms a significant role of POx in the pathogenesis of adverse CV outcomes in dialysis patients. However, notwithstanding the statistical significance and sufficient power of our findings in the Cox regression analysis, the sample size was relatively small, and a causal association between POx and CVD events could not be established, although the post hoc analysis showed that a minimum of 41 CVD cases would be required for the analysis to achieve power of 0.90 and alpha of 0.05 in further investigations.
There are several important limitations that should be acknowledged. First, this was an observational study, and the examined parameters (POx concentration and cytokines) were measured at only one time point. Therefore, causality could not be established. Second, the study sample size was relatively small, and the study cohort represented a select group of patients with a clinically stable condition with the target level of Kt/V. This selection criterion could lead to greatly exaggerated findings in our research. In addition, due to the sample size it was not possible to perform subgroup analysis, and we could not rule out the effects of other potential confounding variables (age, sex, dialysis modality, etc.) on the results. Third, we did not evaluate the effects of other factors that could be associated with elevated POx concentration (changes in Ca homeostasis or the administration of phosphate binders and other medications).
Notwithstanding these limitations, our study is the first to report a direct association between elevated POx concentration, atherogenesis, inflammation and CVD risk in ESRD patients. The results of our study have brought to light many questions that require further investigation. More research with a larger cohort is needed to confirm this preliminary evidence and to validate POx as a proposed biomarker for CVD risk in clinical practice. However, we believe our findings can be useful in determining sample size considerations and can serve as the basis for future research projects.
KEY MESSAGE
1. Plasma oxalic acid (POx) concentration was significantly associated with an increasing trend in the atherogenic lipoprotein fractions as well as a linear decreased high-density lipoprotein cholesterol in end-stage renal disease patients.
2. POx concentration was directly associated with interleukin-6 and monocyte chemoattractant protein-1 levels.
3. POx concentration ≥ 62.9 μmol/L could be considered as a useful marker for predicting cardiovascular disease events in dialysis patients.
Footnotes
No potential conflict of interest relevant to this article was reported.
Supplementary Material
REFERENCES
- 1.Cozzolino M, Mangano M, Stucchi A, Ciceri P, Conte F, Galassi A. Cardiovascular disease in dialysis patients. Nephrol Dial Transplant. 2018;33(suppl 3):iii28–iii34. doi: 10.1093/ndt/gfy174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Stenvinkel P, Carrero JJ, Axelsson J, Lindholm B, Heimburger O, Massy Z. Emerging biomarkers for evaluating cardiovascular risk in the chronic kidney disease patient: how do new pieces fit into the uremic puzzle? Clin J Am Soc Nephrol. 2008;3:505–521. doi: 10.2215/CJN.03670807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Demikhov O, Dehtyarova I, Rud O, et al. Arterial hypertension prevention as an actual medical and social problem. Bangladesh J Med Sci. 2020;19:722–729. [Google Scholar]
- 4.Shifris IM. Diabetic status, comorbidity and survival in patients with chronic kidney disease stage VD: a cohort study. Probl Endocr Pathol. 2020;2:95–103. [Google Scholar]
- 5.Kon V, Yang H, Fazio S. Residual cardiovascular risk in chronic kidney disease: role of high-density lipoprotein. Arch Med Res. 2015;46:379–391. doi: 10.1016/j.arcmed.2015.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Devarajan A. Cross-talk between renal lithogenesis and atherosclerosis: an unveiled link between kidney stone formation and cardiovascular diseases. Clin Sci (Lond) 2018;132:615–626. doi: 10.1042/CS20171574. [DOI] [PubMed] [Google Scholar]
- 7.Dominguez-Gutierrez PR, Kusmartsev S, Canales BK, Khan SR. Calcium oxalate differentiates human monocytes into inflammatory M1 macrophages. Front Immunol. 2018;9:1863. doi: 10.3389/fimmu.2018.01863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ermer T, Eckardt KU, Aronson PS, Knauf F. Oxalate, inflammasome, and progression of kidney disease. Curr Opin Nephrol Hypertens. 2016;25:363–371. doi: 10.1097/MNH.0000000000000229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Stepanova N, Driianska V, Kolesnyk M. Hyperoxaluria-associated cytokines dysregulation in women with recurrent pyelonephritis. Int Med J. 2020;25:783–790. [Google Scholar]
- 10.Aydin H, Yencilek F, Erihan IB, Okan B, Sarica K. Increased 10-year cardiovascular disease and mortality risk scores in asymptomatic patients with calcium oxalate urolithiasis. Urol Res. 2011;39:451–458. doi: 10.1007/s00240-011-0383-9. [DOI] [PubMed] [Google Scholar]
- 11.Arafa A, Eshak ES, Iso H. Oxalates, urinary stones and risk of cardiovascular diseases. Med Hypotheses. 2020;137:109570. doi: 10.1016/j.mehy.2020.109570. [DOI] [PubMed] [Google Scholar]
- 12.Perinpam M, Enders FT, Mara KC, et al. Plasma oxalate in relation to eGFR in patients with primary hyperoxaluria, enteric hyperoxaluria and urinary stone disease. Clin Biochem. 2017;50:1014–1019. doi: 10.1016/j.clinbiochem.2017.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Prenen JA, Dorhout Mees EJ, Boer P. Plasma oxalate concentration and oxalate distribution volume in patients with normal and decreased renal function. Eur J Clin Invest. 1985;15:45–49. doi: 10.1111/j.1365-2362.1985.tb00142.x. [DOI] [PubMed] [Google Scholar]
- 14.Mydlik M, Derzsiova K. Oxalic acid as a uremic toxin. J Ren Nutr. 2008;18:33–39. doi: 10.1053/j.jrn.2007.10.008. [DOI] [PubMed] [Google Scholar]
- 15.Levin RI, Moscatelli DA, Recht PA. Oxalate, a potential atherogenic toxin of uremia, inhibits endothelial proliferation induced by heparin-binding growth factors in vitro. Endothelium. 1993;1:179–192. [Google Scholar]
- 16.Recht PA, Tepedino GJ, Siecke NW, et al. Oxalic acid alters intracellular calcium in endothelial cells. Atherosclerosis. 2004;173:321–328. doi: 10.1016/j.atherosclerosis.2003.11.023. [DOI] [PubMed] [Google Scholar]
- 17.Marangella M, Petrarulo M, Mandolfo S, Vitale C, Cosseddu D, Linari F. Plasma profiles and dialysis kinetics of oxalate in patients receiving hemodialysis. Nephron. 1992;60:74–80. doi: 10.1159/000186708. [DOI] [PubMed] [Google Scholar]
- 18.Sim J, Lewis M. The size of a pilot study for a clinical trial should be calculated in relation to considerations of precision and efficiency. J Clin Epidemiol. 2012;65:301–308. doi: 10.1016/j.jclinepi.2011.07.011. [DOI] [PubMed] [Google Scholar]
- 19.Kidney disease: improving global outcomes (KDIGO) lipid work group. KDIGO clinical practice guideline for lipid management in chronic kidney disease. Kidney Int Suppl. 2013;3:259–305. [Google Scholar]
- 20.Ferraro PM, Taylor EN, Eisner BH, et al. History of kidney stones and the risk of coronary heart disease. JAMA. 2013;310:408–415. doi: 10.1001/jama.2013.8780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lin SY, Lin CL, Chang YJ, et al. Association between kidney stones and risk of stroke: a nationwide population-based cohort study. Medicine (Baltimore) 2016;95:e2847. doi: 10.1097/MD.0000000000002847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jeong IG, Kang T, Bang JK, et al. Association between metabolic syndrome and the presence of kidney stones in a screened population. Am J Kidney Dis. 2011;58:383–388. doi: 10.1053/j.ajkd.2011.03.021. [DOI] [PubMed] [Google Scholar]
- 23.Vale L, Ribeiro AM, Costa D, et al. Metabolic evaluation in urolithiasis: study of the prevalence of metabolic abnormalities in a tertiary centre. Cent European J Urol. 2020;73:55–61. doi: 10.5173/ceju.2020.0051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Masterson JH, Woo JR, Chang DC, et al. Dyslipidemia is associated with an increased risk of nephrolithiasis. Urolithiasis. 2015;43:49–53. doi: 10.1007/s00240-014-0719-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Di Lullo L, House A, Gorini A, Santoboni A, Russo D, Ronco C. Chronic kidney disease and cardiovascular complications. Heart Fail Rev. 2015;20:259–272. doi: 10.1007/s10741-014-9460-9. [DOI] [PubMed] [Google Scholar]
- 26.Yamamoto S, Kon V. Mechanisms for increased cardiovascular disease in chronic kidney dysfunction. Curr Opin Nephrol Hypertens. 2009;18:181–188. doi: 10.1097/MNH.0b013e328327b360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Torricelli FC, De SK, Gebreselassie S, Li I, Sarkissian C, Monga M. Dyslipidemia and kidney stone risk. J Urol. 2014;191:667–672. doi: 10.1016/j.juro.2013.09.022. [DOI] [PubMed] [Google Scholar]
- 28.Gulhan B, Turkmen K, Aydin M, Gunay M, Cikman A, Kara M. The relationship between serum oxalic acid, central hemodynamic parameters and colonization by oxalobacter formigenes in hemodialysis patients. Cardiorenal Med. 2015;5:164–174. doi: 10.1159/000381219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yang X, Li Y, Li Y, et al. Oxidative stress-mediated atherosclerosis: mechanisms and therapies. Front Physiol. 2017;8:600. doi: 10.3389/fphys.2017.00600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Navab M, Reddy ST, Van Lenten BJ, Anantharamaiah GM, Fogelman AM. The role of dysfunctional HDL in atherosclerosis. J Lipid Res. 2009;50(Suppl):S145–S149. doi: 10.1194/jlr.R800036-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ilhan F, Kalkanli ST. Atherosclerosis and the role of immune cells. World J Clin Cases. 2015;3:345–352. doi: 10.12998/wjcc.v3.i4.345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Stepanova N, Driianska V, Snisar L, Lebid L. Inverse association between dialysate oxalate removal and intraperitoneal inflammation in peritoneal dialysis patients. Nephrol Dial Transplant. 2020;35(Suppl 3):1215. [Google Scholar]
- 33.Stepanova N, Driianska V, Savchenko S. Dyslipidemia and intraperitoneal inflammation axis in peritoneal dialysis patients: a cross-sectional pilot study. Kidney Dis (Basel) 2020;6:35–42. doi: 10.1159/000503632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Khan SR. Reactive oxygen species, inflammation and calcium oxalate nephrolithiasis. Transl Androl Urol. 2014;3:256–276. doi: 10.3978/j.issn.2223-4683.2014.06.04. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Horuz R, Goktas C, Cetinel CA, et al. Role of TNF-associated cytokines in renal tubular cell apoptosis induced by hyperoxaluria. Urolithiasis. 2013;41:197–203. doi: 10.1007/s00240-013-0559-6. [DOI] [PubMed] [Google Scholar]
- 36.Dhondup T, Kittanamongkolchai W, Vaughan LE, et al. Risk of ESRD and mortality in kidney and bladder stone formers. Am J Kidney Dis. 2018;72:790–797. doi: 10.1053/j.ajkd.2018.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shah RJ, Vaughan LE, Enders FT, Milliner DS, Lieske JC. Plasma oxalate as a predictor of kidney function decline in a primary hyperoxaluria cohort. Int J Mol Sci. 2020;21:3608. doi: 10.3390/ijms21103608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mulay SR, Eberhard JN, Pfann V, et al. Oxalate-induced chronic kidney disease with its uremic and cardiovascular complications in C57BL/6 mice. Am J Physiol Renal Physiol. 2016;310:F785–F795. doi: 10.1152/ajprenal.00488.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tomson CR, Channon SM, Ward MK, Laker MF. Plasma oxalate concentration, oxalate clearance and cardiac function in patients receiving haemodialysis. Nephrol Dial Transplant. 1989;4:792–799. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.