Abstract:
The intestinal epithelial layer acts as a mechanical and functional barrier between the intraluminal microbiota and the immunologically active submucosa. A progressive loss of gut barrier function (leaky gut) leads to enhanced translocation of microbial products, which in turn contributes as endotoxins to inflammaging. Th17 T cell represents the main immune sentinels in the gut epithelium, preventing aggression from commensal and pathogenic microbes. As HIV infection deeply affects gut Th17 function and increases gut permeability, microbial translocation occurs at high level in people living with HIV (PLWH) and has been associated with the development of non-AIDS comorbidities. Although the inflammatory role of endotoxins like lipopolysaccharide produced by Gram-negative bacteria is well-established, fungal products such as β-D-glucan emerge as new contributors. In addition, PLWH are more frequently infected with cytomegalovirus (CMV) than the general population. CMV infection is a well-described accelerator of immune aging, through the induction of expansion of dysfunctional CD8 T-cells as well as through enhancement of gut microbial translocation. We critically review immune mechanisms related to bacterial and fungal translocation, with a focus on the contribution of CMV coinfection in PLWH. Improving gut barrier dysfunction, microbial composition, and reducing microbial translocation constitute emerging strategies for the prevention and treatment of HIV-associated inflammation and may be relevant for age-related inflammatory conditions.
Key Words: HIV, aging, CMV, leaky gut, LPS, BDG
INTRODUCTION
First let's quote Andrew Aitken Rooney, an acclaimed journalist at CBS News program 60 Minutes who worked until the age of 92: “It's paradoxical that the idea of living a long life appeals to everyone, but the idea of getting old doesn't appeal to anyone.” Such paradox may particularly be relevant for people living with HIV (PLWH), as some age-related diseases appear to be overrepresented and emerging 5–10 years earlier than in the general population.1 Several factors contribute to the accelerated and/or earlier aging, including a high prevalence of traditional risk factors such as substance abuse, obesity, liver steatosis and hypertension, antiretroviral drug long-term toxicity, and persistent inflammation. In addition to HIV persistence and coinfections with viruses such as cytomegalovirus (CMV), mucosal gut damage through microbial translocation is a significant factor contributing to inflammation even in long-term–treated PLWH.
MICROBIAL TRANSLOCATION: WHEN FUNGI JOIN FORCE WITH BACTERIA
The gut barrier plays a complex role in allowing nutrient absorption while preventing microbe translocation. Gut epithelial damage and its consequential increased gut permeability remain the Achilles heel of treated HIV infection.2,3 On HIV infection, the interleukin (IL)-17-producing CD4-helper cells (Th17) loss disrupts epithelial integrity and contributes to the elevated passage of microbial byproducts from the gut lumen into the systemic circulation, referred to as microbial translocation.4 Persistent elevation of Gram-negative bacterial cell wall antigen lipopolysaccharide (LPS) in the circulation is a well-established trigger for innate immune activation contributing to the “inflammaging” and the development of non-AIDS comorbidity.5–9 First demonstrated in monkey models infected with the simian immunodeficiency virus (SIV), further studies showed that SIV/HIV-associated gut damage was associated with microbial translocation of LPS in PLWH.3,10–12
Microbial translocation research mainly focuses on bacteria community. However, fungi constitute the second microbial population in the composition of stool microbiota. Fungi and bacteria encounter each other in various niches of the human body, including in the gut. Locally they can interact with 1 another and influence directly or indirectly the host response.
Fungi such as Saccharomyces cerevisiae, Malassezia restricta, and Candida albicans represent the second largest community in stools in the general population, including PLWH.13,14 Like LPS for bacteria, (1→3)-β- D-glucan (BDG) is considered as the main cell wall component of most fungi and used as a biomarker for diagnosing and managing invasive fungal infection.15 The Fungitell assay, a modified version of the limulus amoebocyte lysate assay specifically detecting BDG is used in clinics and research to quantify microbial translocation of fungal products.16 In absence of invasive fungal infection, BDG plasma levels have been evaluated as a possible fungal translocation marker in PLWH.16
We and others have reported an association between epithelial gut damage and elevation of plasma BDG levels in PLWH. In addition to correlations with LPS levels, plasma BDG levels were associated with marker of gut damage intestinal fatty acid binding protein and marker of gut permeability regenerating islet-derived protein 3 α.5,17,18 These findings suggest that BDG is originating from the gut.16 Moreover, BDG levels were associated with inflammation markers, including proinflammatory cytokines, markers of myeloid cell activation, neopterin levels, and tryptophan metabolism in PLWH.5,16,19,20 Although the association of BDG with gut damage and microbial translocation–associated inflammation were found by several studies in PLWH, the gut origin of these fungal products remain correlative. Strategies used in animal models to confirm the gut origin of translocation LPS in SIV-infected macaques could be used in future studies.3,11
BDG AS A MARKER OF FUNGAL TRANSLOCATION, NON-AIDS COMORBIDITIES, AND AGING IN PLWH
To validate BDG as a marker of fungal translocation, we have assessed whether oral uptake of BDG present in food such as oatmeal, mushrooms, and seaweed can influence BDG plasma levels. In collaboration with Hoenigl et al,21 a clinical trial was implemented to evaluate the effect of oral intake of BDG-rich food in a controlled environment. This study included participants with advanced hepatitis C virus–associated liver cirrhosis, a condition known to be linked with severe epithelial gut damage, antiretroviral therapy (ART) treated or not PLWH, and uninfected age-matched controls. Up to 8 hours after a standardized BDG-rich food intake, no changes in plasma BDG levels could be detected.
To further assess food intake and diurnal influence on BDG plasma levels, we assessed their variation every 4 hours over 24 hours in 11 male ART-treated PLWH.22 BDG levels remained stable over 24 hours. However, as observed in mice models,23 LPS plasma levels increased after lunch and dinner and decreased during the night.
These 2 experiments support the premise that translocated BDG originates only from gastrointestinal fungal communities rather than from food intake.
Association of microbial translocation with risk of developing non-AIDS comorbidities has been linked with induction of inflammation. Although LPS levels are rarely used as markers of non-AIDS comorbidities, we and others have established that BDG levels are associated with non-AIDS comorbidities including cognitive function, metabolic and cardiovascular diseases, as well as all-cause mortality.5,16,19,24–29
Hoenigl et al performed a large cross-sectional analysis of 451 PLWH receiving ART for more than a decade, to assess the merit of several biomarkers associated with non-AIDS events and death. Among several markers of inflammations and coagulation, BDG mean plasma level was the second-best marker after soluble urokinase plasminogen activator receptor, a marker of T cell and monocyte activation in association with risks of non-AIDS comorbidity.30
We also cross-sectionally evaluated BDG and LPS plasma levels in 93 ART-treated PLWH and 52 uninfected controls with similar age and no apparent risk factors for cardiovascular disease.29 All participants underwent a cardiac computed tomography to measure total coronary atherosclerotic plaque volume. We showed an association between plasma BDG levels, but not LPS, and the size of coronary atherosclerosis plaque, independently of age, sex, and other typical cardiovascular risk factors. Interestingly, although associated with plaque volume, BDG levels were similar between ART-treated PLWH and uninfected controls. Importantly, BDG levels were more strongly associated with coronary plaque prevalence than age, smoking habits, hypertension, obesity, and statin usage.29
Another study showed that 2 years after ART initiation, BDG levels were associated with total body fat accumulation, a potential marker of metabolic syndrome.24
Moreover, association between fungal translocation and neurocognition was assessed by the Global Deficit Score in 61 ART-treated PLWH. Investigators observed that BDG elevation was associated with lower neurocognition function, independently of CD4 T-cell count.25
Microbial translocation markers, including LPS, have been shown to be elevated in older PLWH.31 Although several confounding factors could exist such as difference in treatment history and comorbidities, these findings suggest that microbial translocation and inflammation may be linked with aging in PLWH.32 Fungal translocation measured by the BDG plasma level is now a validated marker of inflammation and non-AIDS comorbidities, likely contributing to accelerated aging of PLWH. However, current observations rely only on cross-sectional studies indicating associations not causation between microbial translocation and aging. Moreover, studies are warranted to identify the mechanism by which microbial translocation of BDG triggers non-AIDS comorbidity development. Improving gut epithelial damage and targeted strategies to reduce levels or inflammatory effect of microbial translocation in PLWH and in persons having colitis or diabetes with gut epithelial damage represent an important research agenda.33
LATENT BY NOT SILENT CMV COINFECTION IN PLWH RECEIVING ART
Human CMV, a member of the Herpesviridae family, is ubiquitous worldwide and persists forever in its host. In elderly persons, low CD4/CD8 T-cell ratio, elevated CMV-specific T-cell response, and anti-CMV immunoglobulin G (IgG) levels have been largely linked with adverse health outcomes in large epidemiological studies.34 However, a meta-analysis of 5 studies could not associate CMV infection with all-cause and cardiovascular mortality in more than 10,000 elderly people in Northern Europe.35 Nevertheless, CMV infection, through induction of inflammation, is believed to participate in immune aging, including decreased response to vaccine.36,37
In immunocompetent individuals, CMV infects monocytes and establishes latency in a small subset of pluripotent CD34+ hematopoietic stem cells in the bone marrow.38 In parallel to a true latent stage, asymptomatic CMV reactivation also occurs and spreads in various tissues, including in epithelial cells from the gastrointestinal tract. Those frequent episodes of asymptomatic reactivation, triggered by biological stress, bias the immune response toward a control of CMV replication, a process referred as memory inflation.39 As such, decades after CMV primary infection, up to half of the total pool of effector memory CD4 and CD8 T cells targets CMV antigens.40 These CMV-specific T cells exhibit signs of hypofunction including senescence markers, such as shortened telomeres, decreased expression of fitness markers CD27 and CD28, and increased expression of senescence marker CD57.41 As in other chronic viral infections, higher levels of anti-CMV–specific IgG reflect the B-cell response to episodes of reactivation.42,43 By skewing the immune system and persistently stimulating immune cells, CMV contributes to the inflammaging and notably a decreased response to vaccines.36
In PLWH, CMV seropositivity is almost universal and enhances HIV-induced immune activation, further contributing to non-AIDS comorbidities. While remaining asymptomatic, CMV shedding is frequently detected in the blood and the genital tract of PLWH and has been associated with T-cell immune activation and a decreased CD4/CD8 T-cell ratio, a marker associated with non-AIDS comorbidities and decreased response to vaccines in this population.36,43,44 Moreover, anti-CMV IgG levels were associated with CD4 T-cell count decay in HIV elite controllers, further suggesting a role for CMV in HIV disease progression.45,46 Interestingly, Hunt et al showed that valganciclovir treatment decreased CD4 and CD8 T-cell activation in PLWH. Although valganciclovir is not a CMV-specific inhibitor, these results highlight the inflammatory role of CMV in PLWH.
As CMV coinfection has been linked with persisting inflammation, 1 can hypothesize that CMV would be associated with increase non-AIDS comorbidities in ART-treated PLWH. Indeed, several studies found that CMV-seropositive PLWH, or those with CMV DNA detectable in blood or bodily fluids, had increased risk of inflammatory, metabolic, or cardiovascular diseases.44,47–55 As observed in the general population, CMV seems associated with lower response to vaccines in PLWH, although large randomized controlled trials are required to confirm such observation.36
In parallel, De Francesco et al56 showed that CMV coinfection is 1 of the factors associated with advanced aging in ART-treated PLWH compared with uninfected people with similar lifestyle. Such findings rely on cross-sectional observations and should be confirmed in large prospective studies. CMV infection is believed to promote aging through induction of inflammation in PLWH.
CMV AS A NEW PLAYER IN GUT MICROBIAL TRANSLOCATION
As discussed above, damages to the gastrointestinal epithelial gut barrier and subsequent translocation of microbial products into the circulation constitute hallmarks of HIV infection and are partially improved with ART.3,9
The gastrointestinal tract constitutes both a major site of CMV disease (esophageal and colon ulcers) in immunocompromised hosts and a site of viral persistence.57,58 Damage to the gut epithelium is a hallmark of HIV infection leading to increased gut permeability, microbial translocation, and inflammation.3,59 Precise mechanisms responsible for elevated epithelial permeability are not fully understood, and these damages only partially improve with ART, raising the possible role of CMV.60,61 Indeed, CMV infection has been shown to further contribute to inflammation through epithelial gut damage.17,43,57 In ART-treated PLWH, CMV detection in gut biopsies was associated with a disrupted epithelial barrier and decreased zonula occludens-1 (ZO-1) expression, a marker of tight junctions.57 Moreover, in vitro, CMV infection of gut epithelial cell lines led to a decrease in tight junction protein expression. Importantly, when CMV-infected tissues were treated in vitro with letermovir, a novel anti-CMV agent, tissue integrity was restored.57 These findings were confirmed in PLWH receiving valganciclovir, an antiherpes treatment blocking CMV replication: After treatment, CMV protein expression was decreased and tight junction expression restored in gut biopsies of ART-treated participants.57
We provided clinical evidence of the detrimental effect of CMV on the gut by showing in a cross-sectional study that CMV seropositivity was associated with elevated CD8 T-cell counts and lower CD4/CD8 ratio, higher plasma levels of gut damage markers (intestinal fatty-acid binding protein), and microbial translocation (LPS and BDG) in both PLWH and also in HIV-uninfected participants.43 In addition, markers of gut integrity were linked to proinflammatory cytokines (CXCL13, IL-6, and IL-8) in PLWH. Conversely to levels of anti-EBV IgG, only anti-CMV IgG levels correlated with gut damage/microbial translocation markers, suggesting that CMV is an additional driver of the gut epithelial damage in PLWH.
The influence of CMV-related microbial translocation on aging and risk of non-AIDS comorbidity is poorly studied. Nevertheless, as CMV-related microbial translocation plays a role in persisting inflammation, we can infer that it plays a role in aging and increased risks of non-AIDS comorbidity and aging in ART-treated PLWH (Fig. 1).62 Studies aiming at inhibiting CMV replication with novel drugs such as letermovir (NCT04840199) will help addressing this hypothesis.
FIGURE 1.
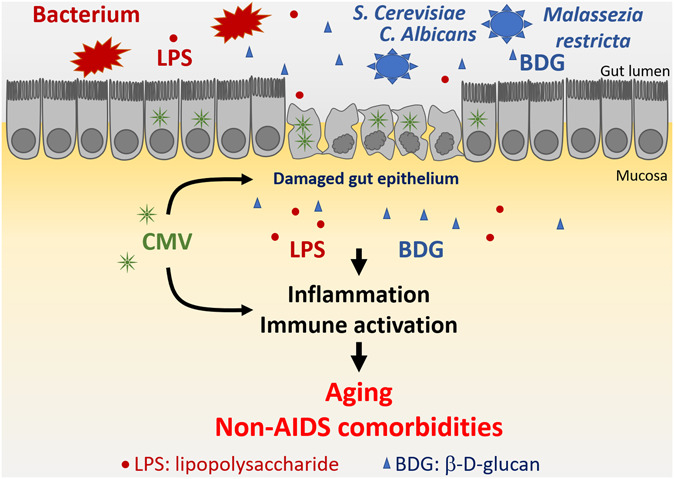
In addition to a direct proinflammatory effect, CMV potentiates microbial translocation of both bacterial (LPS) and fungal (BDG) products in ART-treated PLWH by inducing gut epithelial damage. Persistent inflammation and immune activation increase the risk of developing non-AIDS comorbidities and promote aging in ART-treated PLWH.
GUT MICROBIOTA COMPOSITION: POSSIBLE INFLUENCE OF LATENT CMV
The gut microbiota composition contributes to the development and modulation of the local and systemic immune response, either directly or through microbial byproducts like small-chain fatty acids such as acetate, propionate, and butyrate.63 Conversely, systemic immune response can also influence composition of gut microbiota. The gut microbiota composition has been shown to differ in PLWH, even those on stable ART, compared with control, independently of age, sex, or sexual practice. Moreover, gut microbiota composition has been associated with increased risk of non-AIDS comorbidities.64 As CMV predominantly infects the gut mucosa and induces local inflammation, CMV infection may participate in modifications of the gut microbial composition. Conversely, disruption of the gut microbiota composition could play a role in promoting CMV reactivation in PLWH. Gianella et al65 examined colon biopsies from both PLWH CMV-seropositive and CMV-seronegative individuals and reported on a lower relative abundance of Actinobacteria in those coinfected with HIV and CMV. The clinical significance on inflammation, aging, and risk of non-AIDS comorbidity of such early findings should be further explored.66,67 Fecal microbiota transplant studies could also inform on the influence of the gut microbiota on CMV replication in the gut.33
CONCLUSIONS
Mechanistic, epidemiologic, and clinical evidence indicate that CMV infection contributes to epithelial gut damage and enhanced bacterial and fungal translocations. For PLWH and HIV-uninfected people, subclinical CMV replication in the gut and its influence on microbial translocation is increasingly recognized. As CMV infection is associated with higher levels of inflammatory cytokines, the long-term companionship between CMV infection and treated HIV infection represents a double jeopardy to the health of PLWH. Such CMV-HIV coinfection mimics several aspects of the immune response observed in aging, probably contributing to an accelerated or earlier aging in PLWH. Such important observations constitute the rationale for the implementation of clinical trials using anti-CMV medication such as letermovir and anti-CMV vaccines to prevent infection and reduce gut damage and microbial translocation in PLWH, aiming at improving their long-term outcomes.
ACKNOWLEDGMENTS
Authors are grateful to Angie Massicotte, Josée Girouard, and Cezar Iovi for coordination and support.
Footnotes
Supported by the Fonds de la Recherche Québec-Santé (FRQ-S): Réseau SIDA/Maladies infectieuses and Thérapie cellulaire; the Canadian Institutes of Health Research (CIHR; grant numbers MOP 103230 and PTJ 166049); and the CIHR-funded Canadian HIV Cure Enterprise (team grant number HB2-164064). Jean-Pierre Routy is the holder of the Louis Lowenstein Chair in Hematology and Oncology, McGill University. Léna Royston is a post-doctoral fellow supported by the “Fonds de perfectionnement” of the Geneva University Hospitals, Switzerland, the Swiss National Science Foundation, Switzerland, and CIHR-CTN, Canada. Stéphane Isnard is a postdoctoral fellow supported by the FRQ-S and CIHR-CTN, Canada.
Presented at the HIV and Aging in the era of ART and COVID-19; February 2021, Virtually.
The authors have no conflicts of interest to disclose.
Contributor Information
Jean-Pierre Routy, Email: jean-pierre.routy@mcgill.ca.
Léna Royston, Email: jena.royston@mail.mcgill.ca.
Stéphane Isnard, Email: stephane.isnard@mail.mcgill.ca.
REFERENCES
- 1.Hsu DC, Sereti I. Serious non-AIDS events: therapeutic targets of immune activation and chronic inflammation in HIV infection. Drugs. 2016;76:533–549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Brenchley JM, Douek DC. Microbial translocation across the GI tract. Annu Rev Immunol. 2012;30:149–173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brenchley JM, Price DA, Schacker TW, et al. Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nat Med. 2006;12:1365–1371. [DOI] [PubMed] [Google Scholar]
- 4.Ghosn J, Taiwo B, Seedat S, et al. HIV. Lancet. 2018;392:685–697. [DOI] [PubMed] [Google Scholar]
- 5.Mehraj V, Ramendra R, Isnard S, et al. Circulating (1-->3)-beta-D-glucan is associated with immune activation during human immunodeficiency virus infection. Clin Infect Dis. 2020;70:232–241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tudesq JJ, Dunyach-Remy C, Combescure C, et al. Microbial translocation is correlated with HIV evolution in HIV-HCV co-infected patients. PLoS One. 2017;12:e0183372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Epeldegui M, Magpantay L, Guo Y, et al. A prospective study of serum microbial translocation biomarkers and risk of AIDS-related non-Hodgkin lymphoma. AIDS. 2018;32:945–954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Marchetti G, Cozzi-Lepri A, Merlini E, et al. Microbial translocation predicts disease progression of HIV-infected antiretroviral-naive patients with high CD4+ cell count. AIDS. 2011;25:1385–1394. [DOI] [PubMed] [Google Scholar]
- 9.Marchetti G, Tincati C, Silvestri G. Microbial translocation in the pathogenesis of HIV infection and AIDS. Clin Microbiol Rev. 2013;26:2–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ericsen AJ, Lauck M, Mohns MS, et al. Microbial translocation and inflammation occur in hyperacute immunodeficiency virus infection and compromise host control of virus replication. Plos Pathog. 2016;12:e1006048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Estes JD, Harris LD, Klatt NR, et al. Damaged intestinal epithelial integrity linked to microbial translocation in pathogenic simian immunodeficiency virus infections. Plos Pathog. 2010;6:e1001052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Evans TI, Li H, Schafer JL, et al. SIV-induced translocation of bacterial products in the liver mobilizes myeloid dendritic and natural killer cells associated with liver damage. J Infect Dis. 2016;213:361–369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hager CL, Ghannoum MA. The mycobiome in HIV. Curr Opin HIV AIDS. 2018;13:69–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nash AK, Auchtung TA, Wong MC, et al. The gut mycobiome of the Human Microbiome Project healthy cohort. Microbiome. 2017;5:153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Farhour Z, Mehraj V, Chen J, et al. Use of (1-->3)-beta-d-glucan for diagnosis and management of invasive mycoses in HIV-infected patients. Mycoses. 2018;61:718–722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Isnard S, Lin J, Simeng B, et al. Gut Leakage of fungal-related products: turning up the heat for HIV infection. Front Immunol. 2021:12:656414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ramendra R, Isnard S, Mehraj V, et al. Circulating LPS and (1-->3)-beta-D-Glucan: A folie a deux contributing to HIV-associated immune activation. Front Immunol. 2019;10:465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Isnard S, Ramendra R, Dupuy FP, et al. Plasma levels of C-type lectin REG3alpha and gut damage in people with human immunodeficiency virus. J Infect Dis. 2020;221:110–121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Weiner LD, Retuerto M, Hager CL, et al. Fungal translocation is associated with immune activation and systemic inflammation in treated HIV. AIDS Res Hum Retroviruses. 2019;35:461–472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mehraj V, Ramendra R, Isnard S, et al. CXCL13 as a biomarker of immune activation during early and chronic HIV. Infect Front Immunol. 2019;10:289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hoenigl M, Lin J, Finkelman M, et al. Glucan rich nutrition does not increase gut translocation of beta-glucan. Mycoses. 2021;64:24–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ouyang J, Isnard S, Lin J, et al. Daily variations of gut microbial translocation markers in ART-treated HIV-infected people. AIDS Res Ther. 2020;17:15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cani PD, Amar J, Iglesias MA, et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes. 2007;56:1761–1772. [DOI] [PubMed] [Google Scholar]
- 24.Dirajlal-Fargo S, Moser C, Rodriguez K, et al. Changes in the fungal marker β-D-glucan after antiretroviral therapy and association with adiposity. Open Forum Infect Dis. 2019:6:434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gianella S, Letendre SL, Iudicello J, et al. Plasma (1--> 3)-beta-D-glucan and suPAR levels correlate with neurocognitive performance in people living with HIV on antiretroviral therapya CHARTER analysis. J Neurovirol. 2019;25:837–843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hoenigl M, de Oliveira MF, Perez-Santiago J, et al. 1-->3)-beta-D-Glucan levels correlate with neurocognitive functioning in HIV-infected persons on suppressive antiretroviral therapy.A cohort study. Medicine (Baltimore). 2016;95:e3162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hoenigl M, de Oliveira MF, Perez-Santiago J, et al. Correlation of (1-->3)-beta-D-glucan with other inflammation markers in chronically HIV infected persons on suppressive antiretroviral therapy. GMS Infect Dis. 2015;3;Doc3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hoenigl M, Perez-Santiago J, Nakazawa M, et al. 1-->3)-beta-d-Glucan.A biomarker for microbial translocation in individuals with acute or early HIV infection? Front Immunol. 2016;7:404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Isnard S, Fombuena B, Sadouni M, et al. Circulating β-D-Glucan as a marker of subclinical coronary plaque in ART-treated people living with HIV. Open Forum Infect Dis. 2021:8:ofab109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hoenigl M, Moser CB, Funderburg N, et al. Soluble urokinase plasminogen activator receptor is predictive of non-AIDS events during antiretroviral therapy-mediated viral suppression. Clin Infect Dis. 2019;69:676–686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Scully E, Lockhart A, Huang L, et al. Elevated levels of microbial translocation markers and CCL2 among older HIV-1-Infected men. J Infect Dis. 2016;213:771–775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang H, Kotler DP. HIV enteropathy and aging: gastrointestinal immunity, mucosal epithelial barrier, and microbial translocation. Curr Opin HIV AIDS. 2014;9:309–316. [DOI] [PubMed] [Google Scholar]
- 33.Ouyang J, Isnard S, Lin J, et al. Treating from the inside out: relevance of fecal microbiota transplantation to counteract gut damage in GVHD and HIV infection. Front Med (Lausanne). 2020;7:421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fakhreddine AY, Frenette CT, Konijeti GG. A practical review of cytomegalovirus in gastroenterology and hepatology. Gastroenterol Res Pract. 2019;2019:6156581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chen S, Pawelec G, Trompet S, et al. Associations of cytomegalovirus infection with all-cause and cardiovascular mortality in multiple observational cohort studies of older adults. J Infect Dis. 2021;223:238–246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Royston L, Isnard S, Lin J, et al. Cytomegalovirus as an uninvited guest in the response to vaccines in people living with HIV. Viruses. 2021;13:1266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cicin-Sain L, Brien JD, Uhrlaub JL, et al. Cytomegalovirus infection impairs immune responses and accentuates T-cell pool changes observed in mice with aging. PLoS Pathog. 2012;8:e1002849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Min CK, Shakya AK, Lee BJ, et al. The differentiation of human cytomegalovirus infected-monocytes is required for viral replication. Front Cel Infect Microbiol. 2020;10:368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jergović M, Contreras NA, Nikolich-Žugich J. Impact of CMV upon immune aging: facts and fiction. Med Microbiol Immunol. 2019:208:263–269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sylwester AW, Mitchell BL, Edgar JB, et al. Broadly targeted human cytomegalovirus-specific CD4+ and CD8+ T cells dominate the memory compartments of exposed subjects. J Exp Med. 2005;202:673–685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bellon M, Nicot C. Telomere dynamics in immune senescence and exhaustion triggered by chronic viral infection. Viruses. 2017;9:9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chanouzas D, Sagmeister M, Faustini S, et al. Subclinical reactivation of cytomegalovirus drives CD4+CD28null T-cell expansion and impaired immune response to pneumococcal vaccination in antineutrophil cytoplasmic antibody-associated vasculitis. J Infect Dis. 2019;219:234–244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ramendra R, Isnard S, Lin J, et al. Cytomegalovirus seropositivity is associated with increased microbial translocation in people living with human immunodeficiency virus and uninfected controls. Clin Infect Dis. 2020;71:1438–1446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gianella S, Moser C, Vitomirov A, et al. Presence of asymptomatic cytomegalovirus and Epstein–Barr virus DNA in blood of persons with HIV starting antiretroviral therapy is associated with non-AIDS clinical events. AIDS. 2020;34:849–857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Isnard S, Ramendra R, Lin J, et al. Anti-CMV IgG is linked to CD4 T-cell count decay in HIV elite controllers. Clin Infect Dis. 2020:73:144–147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Patel EU, Gianella S, Newell K, et al. Elevated cytomegalovirus IgG antibody levels are associated with HIV-1 disease progression and immune activation. AIDS. 2017;31:807–813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gianella S, Morris SR, Tatro E, et al. Virologic correlates of anti-CMV IgG levels in HIV-1–Infected men. J Infect Dis. 2014;209:452–456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Cevik M, Russell CD, Ong DSY. Challenges of interpreting CMV DNAemia and its potential association with chronic lung disease in children and adolescents with perinatally acquired HIV infection. Clin Infect Dis. 2019:70:989–990. [DOI] [PubMed] [Google Scholar]
- 49.Lichtner M, Cicconi P, Vita S, et al. Cytomegalovirus coinfection is associated with an increased risk of severe non-AIDS-defining events in a large cohort of HIV-infected patients. J Infect Dis. 2015;211:178–186. [DOI] [PubMed] [Google Scholar]
- 50.Knudsen A, Kristoffersen US, Panum I, et al. Coronary artery calcium and intima-media thickness are associated with level of cytomegalovirus immunoglobulin G in HIV-infected patients. HIV Med. 2019;20:60–62. [DOI] [PubMed] [Google Scholar]
- 51.Letendre S, Bharti A, Perez-Valero I, et al. Higher anti-cytomegalovirus immunoglobulin G concentrations are associated with worse neurocognitive performance during suppressive antiretroviral therapy. Clin Infect Dis. 2018;67:770–777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ballegaard V, Brændstrup P, Pedersen KK, et al. Cytomegalovirus-specific T-cells are associated with immune senescence, but not with systemic inflammation, in people living with HIV. Sci Rep. 2018;8:3778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Yindom LM, Simms V, Majonga ED, et al. Unexpectedly high prevalence of cytomegalovirus DNAemia in older children and adolescents with perinatally acquired human immunodeficiency virus infection. Clin Infect Dis. 2019;69:580–587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Christensen-Quick A, Vanpouille C, Lisco A, et al. Pouring gas on the fire. AIDS Res Hum Retroviruses. 2017;33:S23–S30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Gianella S, Anderson CM, Vargas MV, et al. Cytomegalovirus DNA in semen and blood is associated with higher levels of proviral HIV DNA. J Infect Dis. 2013;207:898–902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.De Francesco D, Wit FW, Burkle A, et al. Do people living with HIV experience greater age advancement than their HIV-negative counterparts? AIDS. 2019;33:259–268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Maidji E, Somsouk M, Rivera JM, et al. Replication of CMV in the gut of HIV-infected individuals and epithelial barrier dysfunction. PLoS Pathog. 2017;13:e1006202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Gordon CL, Miron M, Thome JJ, et al. Tissue reservoirs of antiviral T cell immunity in persistent human CMV infection. J Exp Med. 2017;214:651–667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Wacleche VS, Landay A, Routy JP, et al. The Th17 lineage: from barrier surfaces homeostasis to autoimmunity, cancer, and HIV-1 pathogenesis. Viruses. 2017;9:9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Fletcher CV, Staskus K, Wietgrefe SW, et al. Persistent HIV-1 replication is associated with lower antiretroviral drug concentrations in lymphatic tissues. Proc Natl Acad Sci U S A. 2014;111:2307–2312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Gosselin A, Wiche Salinas TR, Planas D, et al. HIV persists in CCR6+CD4+ T cells from colon and blood during antiretroviral therapy. AIDS. 2017;31:35–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Steele AK, Lee EJ, Vestal B, et al. Contribution of intestinal barrier damage, microbial translocation and HIV-1 infection status to an inflammaging signature. PLoS One. 2014;9:e97171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ouyang J, Lin J, Isnard S, et al. The bacterium akkermansia muciniphila: a sentinel for gut permeability and its relevance to HIV-related inflammation. Front Immunol. 2020;11:645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Vujkovic-Cvijin I, Sortino O, Verheij E, et al. HIV-associated gut dysbiosis is independent of sexual practice and correlates with noncommunicable diseases. Nat Commun. 2020;11:2448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Gianella S, Chaillon A, Mutlu EA, et al. Effect of cytomegalovirus and Epstein-Barr virus replication on intestinal mucosal gene expression and microbiome composition of HIV-infected and uninfected individuals. AIDS. 2017;31:2059–2067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Deeks SG. HIV infection, inflammation, immunosenescence, and aging. Annu Rev Med. 2011;62:141–155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Desai SN, Landay AL. HIV and aging: role of the microbiome. Curr Opin HIV AIDS. 2018;13:22–27. [DOI] [PubMed] [Google Scholar]


