Abstract
Inflammation is an essential protective response against harmful stimuli, such as invading pathogens, damaged cells, or irritants. Physiological inflammation eliminates pathogens and promotes tissue repair and healing. Effective immune response in humans depends on a tightly regulated balance among inflammatory and anti-inflammatory mechanisms involving both innate and adaptive arms of the immune system. Excessive inflammation can become pathological and induce detrimental effects. If this process is not self-limited, an inappropriate remodeling of the tissues and organs can occur and lead to the onset of chronic degenerative diseases. A wide spectrum of infectious and non-infectious agents may activate the inflammation, via the release of mediators and cytokines by distinct subtypes of lymphocytes and macrophages. Several molecular mechanisms regulate the onset, progression, and resolution of inflammation. All these steps, even the termination of this process, are active and not passive events. In particular, a complex interplay exists between mediators (belonging to the group of Eicosanoids), which induce the beginning of inflammation, such as Prostaglandins (PGE2), Leukotrienes (LT), and thromboxane A2 (TXA2), and molecules which display a key role in counteracting this process and in promoting its proper resolution. The latter group of mediators includes: ω-6 arachidonic acid (AA)-derived metabolites, such as Lipoxins (LXs), ω -3 eicosapentaenoic acid (EPA)-derived mediators, such as E-series Resolvins (RvEs), and ω -3 docosahexaenoic (DHA)-derived mediators, such as D-series Resolvins (RvDs), Protectins (PDs) and Maresins (MaRs). Overall, these mediators are defined as specialized pro-resolving mediators (SPMs). Reduced synthesis of these molecules may lead to uncontrolled inflammation with possible harmful effects. ω-3 fatty acids are widely used in clinical practice as rather inexpensive, safe, readily available supplemental therapy. Taking advantage of this evidence, several researchers are suggesting that SPMs may have beneficial effects in the complementary treatment of patients with severe forms of SARS-CoV-2 related infection, to counteract the “cytokine storm” observed in these individuals. Well-designed and sized trials in patients suffering from COVID-19 with different degrees of severity are needed to investigate the real impact in the clinical practice of this promising therapeutic approach.
Keywords: Inflammation, Cytokine storm, ω-3, ω-6, SPMs, Resolvins, Covid-19, CoV-2
1. Introduction
The immune system counteracts harmful stimuli, such as invading pathogens, damaged cells, or irritants by the activation of a protective response which is defined as inflammation [1]. Each biological process is characterized by an earlier phase of stimulation and self-amplification, which promotes its onset and course, and by a later stage of down-regulation, which leads to a progressive decrease in intensity and strength of the process and its end. Several control mechanisms are involved in modulating the development of all these events and in their following inhibition [2]. This type of control helps to prevent the development of deleterious effects on the body. Acute inflammation also is a dynamic process and it is characterized by this type of organization with a phase of onset, progression, and resolution [3]. It has evolved in the higher organisms to protect them from both infectious and non-infectious harmful stimuli. Inflammation represents a crucial mechanism for the induction and the development of innate immune response and orchestrates the stimulation of long-term adaptive immunity toward specific pathogens. This process depends on the tightly regulated and sequential activation as well as on a proper balance among inflammatory and anti-inflammatory factors. A wide spectrum of distinct mediators is involved in modulating the different phases of this protective response and in regulating innate immunity [1,4], such as chemokines, cytokines, pro-oxidant and anti-oxidant species, pro-inflammatory metabolites, molecules with the capability to actively counteract inflammation and promote its proper resolution, defined specialized pro-resolving mediators (SPMs) and several costimulatory proteins [1]. The mechanisms regulating the development of inflammation cause the change in the physical location of different blood components. Under the action of the above-mentioned chemical mediators, immune cells (monocytes, basophils, eosinophils, and neutrophils), plasma, and fluids, extravasate from blood vessels and are recruited at the inflamed site, where they exert their effector function to counteract the harmful stimulus. If this process occurs properly, the dangerous element is eliminated, the acute defense response self-limits, turns off and complete recovery of the involved tissues or organs arises. On the other hand, if the cause of the injury is not removed, the inflammation persists and may result in different clinical outcomes. This process may become quickly uncontrolled, generating potentially life-threatening clinical conditions, such as sepsis, acute respiratory distress syndrome (ARDS), and multi-organ failure (MOF) [[5], [6], [7], [8], [9], [10], [11]]. These very serious syndromes may require hospitalization in the Intensive Care Units (ICUs) and are burdened with high mortality rates. On the other hand, the persistence of inflammation may lead to the development of diseases with a slower and prolonged clinical course, causing chronic pathological conditions with different grades of severity and mortality over time [12]. Therefore, all these reasons continue to stimulate Researchers to improve the understanding of the biological events occurring in the different steps of this defensive response as well as of complex interplay among cells and mediators involved in this process. As a result of these efforts, our knowledge about inflammation has progressively increased in the last years and new concepts concerning these topics have emerged from the most recent studies in comparison with the earlier ones. Several investigations have been carried out to better clarify the different phases of this process. The results of these studies have changed, even deeply, the previously accepted notions and the traditional views. In particular, although for a long time in the past the resolution phase of inflammation has been considered to originate from the downregulation or inactivation of proinflammatory mediators, current evidence suggests that the termination of this process is an active and not a passive event and it is mediated and regulated by the generation of pro-resolving molecules [13]. Mediators and bioactive molecules, mainly derived from polyunsaturated fatty acids (PUFA) and known as specialized pro-resolving mediators (SPMs), modulate the action of several signaling pathways and control the self-resolution of inflammation [14]. In particular, several years ago, Serhan and his Team were the first Researchers to focus their investigations on these molecules, and afterward, they have contributed to increasing and updating our knowledge about the cells, molecules, and events involved in this process. They have detected SPMs in self-limited exudates formed in in vivo animal models and human cells, using a liquid chromatography-tandem mass spectrometry-based analysis [13,15]. Our paper aims to summarize in brief the following points:
-
1)
main events involved in the physiological activation, development, and termination of the inflammation in humans as well as factors mediating an uncontrolled or unfavorable course;
-
2)
the resolution phase of inflammation and the events and molecules mediating it.
-
3)
the occurrences and the mechanisms which develop during the activation of an uncontrolled inflammatory response in patients with severe forms of SARS-CoV-2 related infection.
1.1. Mechanisms and mediators involved in the onset of inflammation
Inflammation is triggered when different dangerous stimuli act on the host’s tissues causing their damage. In response to this event, some molecules, known as pathogen-associated molecular patterns (PAMPs) or danger-associated molecular patterns (DAMPs), become detectable in injured cells and are sensed by germline-encoded pattern recognition receptors (PRRs) [16]. The stimulation of PRRs represents a crucial step in the induction of innate immune responses as it triggers a series of downstream signaling pathways, leading to the synthesis and release of inflammatory cytokines, type I interferon (IFN), and other mediators. Several classes of PRRs have been identified in mammalian cells, including Toll-like receptors (TLRs), Retinoic acid-inducible gene-I-like receptors (RIG-I-like receptors or RLRs), Nod-like receptors (NLRs), AIM2-like receptors (ALRs), C-type lectin receptors (CLRs), and intracellular DNA sensors such as cyclic-GMP-AMP (cGAMP) synthase (cGAS) [17]. Among PRRs, TLRs and CLRs are transmembrane receptors, recognizing extracellular and endosomal-derived PAMPs, whereas RLRs and NLRs proteins are cytosolic receptors, involved in the intracellular surveillance toward infections and the recognition of DAMPs. In particular, in response to tissue damage, NLRs and RLRs contribute to generating supramolecular complexes, known as inflammasomes [10,11]. The classical form of these complexes consists of three elements: a) a cytosolic-sensor, represented either by a nucleotide-binding oligomerization domain and leucine-rich-repeat-containing (NLR) protein or by a member of the AIM2 like receptor (ALR) family, b) an adaptor protein apoptosis-associated speck-like protein, including a domain with caspase activation and recruitment site (CARD; ASC) and c) a pro-caspase-1. The assembly of inflammasomes stimulates the conversion of this pro-enzyme into the active caspase. This catalytic protein promotes the maturation and secretion of two key pro-inflammatory cytokines, such as IL-1β and pro-IL-18 [18]. Both these interleukins contribute to orchestrate the generation of the complex inflammatory network in the damaged tissue microenvironment, including cells, danger-sensing receptors, chemokines, growth factors, reactive oxygen species, enzymes, and bioactive lipid-mediators. The next paragraph focuses on the genesis, structure, and role of these latter compounds in the initiation, course, and resolution of inflammation.
1.2. Time course of the inflammatory response: onset, development, resolution and class switch of the lipid-mediators
In the early phases of the host’s response to pathogens, causing tissue damage, polyunsaturated fatty acids are released from the membrane phospholipids of some cells, such as neutrophils, dendritic cells, and macrophages, or of other blood components, such as platelets. These molecules are converted by a large series of enzymes into specialized pro-inflammatory mediators, including PGs, LTs, and TXs [19,20]. Among these metabolites, LTB4 displays a key role in orchestrating the onset of inflammation, as it acts as a chemotactic factor for the recruitment and the influx of further neutrophils to the site of infection [19,21], LTB4 works in a coordinated manner with a large series of interleukins, oxygen-species and other molecules, including PGs. In particular, PGE2 and PGD2 amplify and accelerate the development of inflammatory response. PGE2 and PGI2 regulate blood flow, LTC4 and LTD4 modulate vascular permeability [19,21]. Therefore, Eicosanoids orchestrate the onset of the inflammatory process and the activation of the innate immune response, modulating its early and later phases [22]. Furthermore, these mediators connect innate with adaptive arms of immunity, modulating the functions and the activity of the cells belonging to both systems [23]. In particular, PGE2 regulates several functions of immune cells, modulating the activities of both innate and adaptive arms of immunity. Therefore, this mediator exerts a crucial role in the connection of the two systems, involving antigen-presenting cells (APCs) and T lymphocytes [23,24]. These molecules act together with pro-inflammatory cytokines, which are produced in part from the cells of the adaptive arm of the immune system, with some chemokines and with complement factors (C5a and C3b), inducing the accumulation of neutrophils at the site, where the damage occurs. Both infectious and non-infectious stimuli may induce the onset of inflammation [19,21]. In the first case, neutrophils, antigen-presenting cells, and inflammatory macrophages cells engulf and kill pathogens [8,25]. Furthermore, these immune cells are activated, when a non-infectious injury occurs, with a similar pattern of cells and mediators involved. The resolution of the inflammation is also an active temporal and spatial independent physiological action in the process of non-sterile and sterile infections [14,26,27] and modulatory mechanisms also exist in the host. They are activated immediately and after the onset of this response and consist of regulatory cells and molecules leading to its resolution. All these elements cooperate in a coordinated way to prevent an excessive and exuberant inflammatory response, potentially leading to severe tissue damage with harmful clinical effects [14,26,27]. This event occurs when the immune cells progressively down-regulate the synthesis of pro-inflammatory mediators, such as leukotrienes, and switch the immune response from a pro-inflammatory phenotype towards a pro-resolving one, with the production of other mediators, known as pro-resolving lipoxins [28] at the site of infection. This crucial phase is marked by critical modifications in the synthesis and release of lipid-mediators by immune cells involved. This process is active and is defined as class switching of lipid mediators [29]. In particular, the synthesis of PGE2 and PGD2, occurring in the early stage of inflammation, is not only associated with the amplification of this response in its initial step, but they also induce the enzyme 15-lipoxygenase (15-LOX) [28,30]. This event results in the switching from the PMN phenotype, producing LTB4, to the PMN phenotype, producing LXA4. Indeed, LXA4 is a stop signal to the recruitment of PMNs and is associated with the sequential synthesis of a large spectrum of pro-resolving mediators (SPMs), including Resolvins, Protectins, and Maresins. Overall, these metabolites promote the onset of the resolution phase in the context of the inflammatory response [28,30,31]. The mechanisms by which all these mediators stimulate the end of inflammation are the following: a) block of neutrophils recruitment; b) promotion of the apoptotic cells efferocytosis and clearance of the cellular debris (the persistence of these cell components contributes to maintaining inflammation) by macrophages; c) increase in phagocytosis of microbes; d) down-regulation or sequester of pro-inflammatory cytokines, by decreasing concentration of prostanoids [[32], [33], [34], [35], [36]]. SPMs also display a crucial role in lymphocyte maturation, T cell differentiation, and IgG switching and increase antimicrobial responses against bacterial and viral pneumonia, by counteracting cytokine storm and by protecting inflamed organs from injury [[37], [38], [39], [40], [41]].
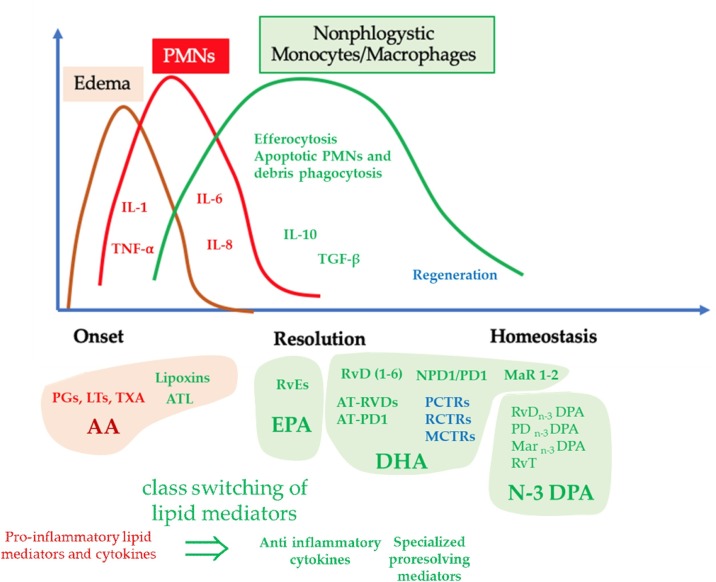
Classically, activated M1 macrophage populations with pro-inflammatory activities and with pathogen-killing abilities are initially predominant, during the onset of inflammation, but they are later replaced by macrophages with an M2 phenotype. Following their stimulation, these cells play important roles in the resolution of inflammation, by promoting tissue regeneration [42,43]. Pro-resolving M2 macrophages and also apoptotic neutrophils produce SPMs [44] (Fig. 1 ). In particular, these mediators stimulate phagocytosis by M2 macrophages without stimulation of phlogosis and promote cell proliferation and tissue repair [33,45].
Fig. 1.
Chronobiology of cellular and molecular events which characterize the evolution of the self-limiting inflammatory process. The molecular and cellular events associated with classic signs of inflammation onset (Calor: heat; Rubor: redness; Tumor: swelling; Dolor: pain); include: a) the synthesis and release of molecular mediators (Complement, Cytokines, Chemokines, Prostaglandins, Leukotrienes), b) the recruitment of leukocytes (PMNs, eosinophils) and their extravasation from blood vessels into the inflamed tissues. Harmful stimuli activate phospholipase (FLP) A2 that produces free AA. This PUFA is the precursor of eicosanoids, such as prostaglandins (PGs) and leukotrienes (LTs), as well as prostacyclins, thromboxanes, lipoxins, hydroxy-, and hydroperoxyl-fatty acids. LTs and in particular LTB4 induce the onset of the inflammatory response, whereas PGD2 and PGE2 not only amplify this process but also stimulate the activity of 15-lipoxygenase (15-LOX). It is a crucial enzyme in initiating the resolution of inflammation, as it catalyzes the production of mediators with pro-resolving capabilities. During this phase, critical modifications in the synthesis and release of lipid-mediators by immune cells occur, a process known as class switching of lipid mediators. These metabolites are defined as specialized pro-resolving mediators (SPMs) and regulate the final stage of inflammation, which is now known as an active process. Cessation of PMN recruitment, modulation of leukocyte trafficking, macrophage reprogramming from M1 to M2 phenotype, macrophage efferocytosis, and increased phagocytosis of apoptotic neutrophils by non-inflammatory macrophages with an M2 phenotype represent critical events during this phase. Whether all these events develop in a proper qualitative and quantitative way, the acute inflammation will resolve completely, with restoration of tissue homeostasis and without their excessive damage, whereas the activation of excessive or uncontrolled inflammatory responses or the presence of defective pro-resolving mechanisms may cause the development of severe pathological conditions in humans, such as sepsis, ARDS, and MOF. AA: arachidonic acid, DHA: docosahexaenoic-acid, EPA: eicosapentaenoic acid, N3 DPA: Docosapentaenoic acid;IL-1: Interleukin 1; RvDs: D-series Resolvin; RvEs: E-series Resolvins; Leukotriene (LT), Thromboxane (TX), Thromboxane A2 (TXA2), Protectin D: (PD).
The activation of an uncontrolled inflammatory process leads to severe pathological conditions in humans, such as sepsis, ARDS, and MOF. Several harmful stimuli may be involved in the development of these syndromes. Taking advantage of the current knowledge, in the following paragraph, we briefly consider the pathogenetic mechanisms, involved in the onset of inflammatory events associated with SARS-CoV-2 related infection and with COVID-19.
1.3. Inflammatory response in patients with severe forms of SARS-CoV-2 infection
In December of 2019, a novel single-stranded ribonucleic acid (RNA) human-infecting coronavirus, defined SARS-CoV-2, has originated in Wuhan, Hubei Province in China, and from there it has spread worldwide, causing a severe pandemic. This pathological condition is now known as Coronavirus Disease 19 (COVID-19) [46] and it is characterized by a wide spectrum of signs and symptoms, such as cough, sore throat, fever, shortness of breath, sudden onset of anosmia, ageusia, or dysgeusia, nausea or vomiting and diarrhea with different degrees of severity [46], The World Health Organization (WHO) has declared this epidemic “public health emergency of international concern” [47]. This pandemic has already caused near 5 million deaths around the globe. SARS-CoV-2 infection is associated with elevated morbidity and mortality in the human population, due to the high risk of developing some life-threatening pathological conditions, such as sepsis, ARDS, and MOF [48]. The above-mentioned syndromes are characterized by the induction of a dysregulated host immune response, resulting in an uncontrolled self-amplifying proinflammatory cascade through the production of a large spectrum of mediators and cytokines. A recent review has summarized structure, mechanisms of pathogenicity, immune response, clinical course, and potential short- and long-term effects in the affected patients of SARS-CoV-2 and has compared its biological behavior with that of other well-known RNA-viruses, such as Hepatitis C Virus (HCV), Human Immunodeficiency Virus (HIV) and Influenza Viruses (IVs). This paper has aimed to search common biological characteristics among all these viruses so that to draw useful lessons and insights about SARS-CoV-2 and its related disease (Coronavirus Disease or COVID-19). This methodological approach may increase our knowledge of this virus and contribute to improving the management and treatment of the patients affected by this pathogen [49]. This complex process is triggered by the hyperactivation of the inflammasome, leading to the rapid synthesis and release of lipid molecules. This event has been defined as an “eicosanoid storm” and it is characterized by increased production of pro-inflammatory mediators [19,[50], [51], [52]]. The stimulation of eicosanoid release, in turn, is associated with an uncontrolled and rapid production and activation of cytokine self-amplifying cascade by cells belonging to the adaptive arm of the immune system [50]. This process is known as “cytokine storm” (CS), according to Cron’s and Behrens’ definition [53], although some studies have suggested that COVID-19 is not properly characterized by the development of this pathological condition, as observed in other syndromes, such as Sepsis, ARDS unrelated-COVID, Cytokine Release Syndrome and Severe Acute Pancreatitis, but rather it is associated with a “quasi-cytokine storm”. This conclusion seems to be confirmed even in individuals, suffering from ARDS associated with COVID-19, requiring mechanical ventilation. Serum median levels of IL-6 and IL-8 in subjects with severe COVID-19 are about 10 until 100 times lower than those of patients with the above-mentioned syndromes [42,54]. The group of cytokines involved in the development of these syndromes includes IL-2, IL-6, IL-8, IL-17, IFN-γ, TNF-α. They are released by macrophages and some subclasses of lymphocytes. Cytokine Release Syndrome. Sepsis, ARDS, and MOF are characterized by an exuberant and excessive systemic inflammation and are also associated with a status of immunosuppression [55], blood hypercoagulability, and thrombophilia [42,56]. The action of inflammatory mediators, which are released following strong harmful stimuli, is systemic [57]. A wide spectrum of infectious and non-infectious agents may induce CS, including viral respiratory infections such as H5N1 influenza, Epstein-Barr virus, cytomegalovirus, and group A streptococcus and non-infectious conditions such as graft-versus-host disease and chimeric antigen receptor (CAR)-T cell therapy. Although several reports have suggested that severe forms of SARS-CoV-2 infection are also associated with the development of CS [9,[58], [59], [60], [61]], further studies are needed to clarify this point [54]. Based on the results emerging from current evidence, such as autopsy studies, the most important causes of deaths associated with SARS-CoV-2 infection are represented by respiratory failure, thrombosis, and sepsis [62]. In particular, two major pathogenic mechanisms have been described in patients with COVID-19, including vessel endothelium and dysfunction and immunothrombosis [63,64]. The latter term indicates an intrinsic effector cascade of the innate immunity, which is activated by infectious agents to counteract their diffusion and their survival in the host [65]. According to available data, COVID-19 is characterized by a prothrombotic state and microthrombosis is one of the key signs of this syndrome [66]. In general, a large series of systems regulates crucial defensive functions in the human body, such as inflammation-, complement- and coagulation-cascades as well as platelet-activation. Overall, these pathways form a closely interconnected network. The activation of the elements belonging to each of these systems may lead to the stimulation of other components involved in this complex axis [67]. In particular, some elements of the complement cascade, such as C3a and C5a, may stimulate the coagulation process [68]. On the other hand, Thrombin may activate C3 and C5 as well as von Willebrand Factor (vWF) modulates the function of the complement system [69]. Small vWF components induce the complement inhibitor factor I and the inactivation of C3b, whereas large vWF ones stimulate the C3b and promote the action of several elements, belonging to the complement system [70]. Some activated components of the coagulation pathway (FIXa, FXa, FXIa as well as plasmin) may generate C3a and C5a from C3 and C5, respectively [71]. C5a displays a prothrombotic activity, inducing the upregulation of the Tissue Factor (TF) [72]. Endothelial cells, neutrophils, monocytes, and interleukins/cytokines, originating a cross-talk between inflammation and coagulation pathways are involved in this process, leading to the inflammation of microvessels. Some interleukins exert crucial roles in these events. In particular, 1) IL-6 is able to increase both the synthesis and activity of the platelets and the expression of tissue factor (TF) 2) interferon-γ (IFN-γ), enhances platelets production; 3) IL-2 upregulates plasminogen-activator inhibitor-1 (PAI-1) decreasing the fibrinolysis; 4) IL-8 predisposes to the formation of neutrophil extracellular traps (NETs) by attracting the neutrophils to the site of infection [73,74]. Viral proteins, produced by SARS-CoV2, may perturb the function of the host’s complement- and coagulation systems. A recent study has reported that the Spike 1 protein of this virus may damage the endothelium of microvessels, causing endotheliitis and cell injury [75]. This event leads to the generation of vWF and the release of the plasminogen activator inhibitor-1 (PAI-1), promoting or exacerbating thrombosis. Coagulopathy detectable in COVID-19 patients is generally characterized by high levels of fibrinogen, von Willebrand factor (VWF) and D-dimer in the blood. On the other hand, these individuals generally present no or only small modifications in prothrombin time, activated partial thromboplastin time (coagulation time), antithrombin levels, and activated protein C values [76,77]. SARS-CoV-2-induced alteration in the function of vessel endothelium and of platelets in the infected patients causes the development of a pro-adhesive phenotype of endothelial cells [78] and these events activate the inflammatory-, complement- and neutrophil-cascades. LTB4, C3 and C5 act as potent chemoattractant factors for neutrophils and monocytes that are recruited into different organs, such as at pulmonary level. The activation of these mediators generates neutrophil extracellular traps (NETs), which activate the alternative complement pathway. This process triggers a feedback loop and induces a state of hypercoagulability, endotheliopathy, thrombotic microangiopathy, leading to severe clinical manifestations, such as ARDS in patients with severe forms of COVID-19 [79]. However, all these events occur not only in lung tissue, but also in other organs, including heart, kidney, brain, and skin in patients with SARS-CoV-2 infection [80,81].
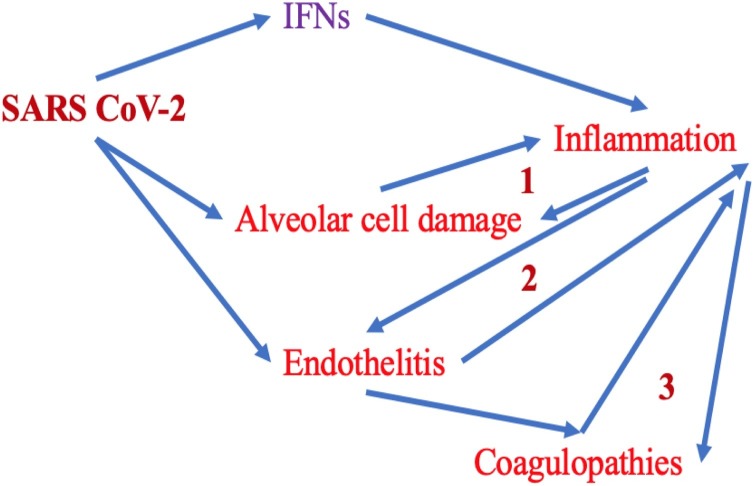
A schematic representation of the pathogenetic events occurring in the course of severe COVID-19 forms as well as the mechanisms involved are shown in Fig. 2 [82,83].
Fig. 2.
The first line of defense against viruses is represented by the interferon response (IFNs I and III). Interferons trigger an inflammatory response. SARS CoV-2 can infect the alveolar- and endothelial cells of the lung, via the binding with the membrane receptor ACE2. Virus entry activates the inflammasome and the transcription factor NF-kB, inducing the onset of the inflammation. Several pathogenetic mechanisms are involved in this complex process. Activated endothelial cells support inflammation (endotheliitis) and promote coagulation disorders (thrombosis). These events, in turn, maintain and amplify the inflammatory process in several tissues and organs, mainly in the lungs. The resulting damage in the pulmonary alveolar cells contributes to the progressive worsening of endotheliitis and coagulation disorders (thrombosis and disseminated intravascular coagulation). At least 3 mechanisms of self-amplification of inflammation are involved (in the figure they are numbered from 1 to 3). They can cause the production and release of pro-inflammatory cytokines into the systemic circulation (cytokine storm). The anti-inflammatory therapies tested up to now have been directed against the inflammation effectors (tocilizumab: anti-IL-6 receptor or Anakinra: anti-IL-1 receptor). Their use is associated with a real risk of causing immunosuppression. However, regulatory anti-inflammatory innate mechanisms exist in the host and they are generally activated immediately after the onset of the inflammation to counteract an excessive response, potentially leading to severe tissue damage with harmful clinical effects. A large series of endogenous molecules, derived from the omega-3 eicosanoids (resolvins, maresins, protectins), with pro-resolving functions of inflammation have been detected and studied. These mediators can limit the triggering of inflammation, but without inducing immunosuppression. Resolvins, maresins, protectins exhibit unique and very interesting mechanisms of action. They turn off the inflammatory process and promote the elimination of cellular debris and dead cells by macrophages, also counteracting coagulation disorders, but in the absence of inflammation. Therefore, resolvins, maresins, and protectins attenuate or block tissue injury. On the other hand, the anti-inflammatory and anti-COX-2 drugs block not only the production of pro-inflammatory eicosanoids but also the synthesis and release of these very important molecules with anti-inflammatory activities.
In recent years, several studies have reported an interesting role of SPMs as a complementary treatment of several infectious pathogens, including bacteria, viruses, and fungi [84]. The mechanisms of actions of these lipid mediators have been investigated in patients affected by these microorganisms. In particular, the biological activities of SPMs have been examined in some viral infections, caused by Influenza-, Respiratory Syncytial- and Herpes Simplex Viruses [82]. The results from available experimental and clinical studies suggest that these mediators contribute to attenuating or inhibiting the inflammatory process. Furthermore, the biological activities of SPMs may modulate and regulate viral pathogenicity and its ability to disseminate within the host [83]. A complex interplay exists between some viruses and these lipid molecules. In particular, a study carried out in a mouse model of infection has suggested that systemic spread of H5N1 influenza virus results both in down-regulation of genes codifying molecules, such as SPMs, with resolving ability of inflammatory process and in up-regulation of mediators, such as cytokines and eicosanoids, with pro-inflammatory properties. This unbalanced inflammatory response is associated with increased virulence of the influenza A virus [85]. The levels of these mediators may be decreased by more virulent viral strains as well as distinct SPMs may exert antiviral activity [86]. A further and important point must be considered. The use of non-steroidal anti-inflammatory-drugs (NSAIDs) and anti-COX-2 ones may have harmful effects in patients with various types of viral infection, as these substances inhibit or block not only the production of pro-inflammatory eicosanoids but also the synthesis and release of these lipid mediators, therefore reducing their anti-inflammatory activities [87,88]. Taking advantage of all these data, the strategies aimed at stimulation of SPMs production and release may be a useful approach to prevent or to counteract the most severe forms of SARS-CoV-2 related infections. In particular, some interesting experimental studies in animal models have investigated the possible ability of these lipid mediators to promote the resolution of the inflammatory process in the lung, decreasing tissue injury [37]. A review article has summarized the current evidence for the use of omega-3 fatty acids supplementation in the prevention and therapy of a wide spectrum of human diseases, including coronary artery disease, diabetes, hypertension, arthritis, and other inflammatory and autoimmune conditions. Current evidence suggests that omega-3 fatty acids exert a key therapeutic role in these pathologies. They actively modulate the inflammatory process by inducing the synthesis of SPMs. These mediators down-regulate the excessive synthesis of pro-inflammatory lipid-derived molecules and cytokines and decrease the extent of the inflammatory process, promoting its active resolution [89]. Some papers have suggested that systemic dysregulation of metabolism and widespread changes in the lipidome and lipid-mediators synthesis may occur and it may contribute to the inflammatory progression to severe forms of COVID-19 [90,91]. These results are in accordance with the current epidemiological studies, reporting that SARS-CoV2 positive patients with advanced age, hypertension, diabetes, and obesity are at higher risk of both more severe forms of disease and mortality [92,93].
A recent study has analyzed the serum pattern of PUFAs and precursors of lipid-mediators in healthy subjects, in hospitalized COVID-19 positive patients with respiratory symptoms not requiring Intensive Care Unit (ICU) admission as well as in COVID-19 positive individuals with respiratory symptoms requiring ICU treatment. In particular, it has been reported a dysregulation in the pattern of PUFA containing lipids between patients with SARS-CoV-2 infection and controls. Decreased levels of PUFA-containing phosphatidylcholine, PUFA-containing phosphatidylserine, and PUFA-containing phosphatidylethanolamine-plasmalogen and increased levels of PUFA free fatty acids, as well as PUFA-containing phosphatidylethanolamine, PUFA-containing lysophospholipids, and PUFA-containing triacylglycerols, were observed in patients with COVID-19 in comparison with healthy individuals and this change was more evident in individuals with severe forms of the disease. Furthermore, subjects with moderate clinical manifestations of SARS-CoV-2 related infection presented significantly more elevated serum amounts of lipid metabolites with pro-resolving activities, such as resolvin (Rv) E3 as well as with a trend toward higher levels of some PG family members, mainly PGE2, PGD2, and PGF2a. On the other hand, in patients with severe forms of COVID-19, the pattern of lipid-mediators mainly consisted of free PUFAs levels, monohydroxylated species, and AA-derived dihydroxylation elements. All these metabolites are the expression of the increased Lypoxygenases 5 and cytochrome p450 activities and the down-regulated production of compounds, mediated by COX2 and Lypoxygenases 12 [94]. Furthermore, some studies are investigating the type and magnitude of the host’s immune responses against the proteins of SARS-CoV-2 and their capability to trigger SPMs biosynthesis. A recent article has shown the efficacy of RvD1 and RvD2 in modulating the inflammatory and antimicrobial responses, which were elicited by the viral spike 1 glycoprotein (S1) in macrophages obtained from patients with cystic fibrosis (CF) or healthy controls. In particular, this viral protein has been able to activate SPMs biosynthesis as well as it was able to cause a reduction in FPR2/ALX (formyl peptide receptor 2/lipoxin A4 receptor), RvD1 receptor, and RvD2 receptor genes expression in macrophages of both groups of individuals. In macrophages from volunteers with and without CF stimulated by S1, RvD1 and RvD2 have also restored the functionality of these receptors, suppressed by S1, and have decreased the production of IL-8, Monocyte chemoattractant protein 1 (MCP-1) and Macrophage inflammatory protein-1β (MIP-1β). Overall, this study has demonstrated the regulatory activity of RvD1 and RvD2 on SARS-CoV-2-caused inflammation [95]. Taking advantage of all the available results and data, it may be hypothesized that, in patients suffering from severe forms of SARS-CoV-2 infection, the use of SPMs in clinical practice as a complementary treatment of COVID-19 may provide possible beneficial effects [96,97].
The promising heart and lung-protective mechanisms of ω-3 polyunsaturated fatty acids reported in the literature include 1) the change in the membrane cell composition with the replacement of ω-6 polyunsaturated fatty acids with ω-3 polyunsaturated fatty acids. This event produces a modification in physical properties of the cell membrane bilayer, such as its fluidity, and modulates the structure and the activity of cell membrane and very important complexes, known as lipid rafts. These specialized microdomains function as platforms, enriched in cholesterol, sphingolipid, and proteins regulating the signaling cascade pathways. Overall, these modifications affect protein trafficking, synthesis of cytokines, production of lipid mediators, and gene transcription, inducing a switching from proinflammatory to antiinflammatory response of immune cells [[98], [99], [100]].
2) Modulation of mitochondrial function. The excessive production of proinflammatory metabolites caused by viral infections causes harmful effects in the activities of mitochondria, by inducing oxidative stress with different degrees of severity in these micro-organelles and cell cytoplasm. The decrease in amounts of these molecules contributes to restoring the normal function of mitochondria. The detrimental effects induced by SARS-CoV-2 infection may be attenuated by ω-3 polyunsaturated fatty acids similarly [101].
3) Possible decrease in viral load via a direct and/or indirect interaction. ω-3 polyunsaturated fatty acids may induce the damage or lysis of viral envelopes, causing their destruction and inactivation as well as may promote the production of antibodies to counteract the SARS-CoV-2 related infection.
4) Potential role in improving disorders in the system of blood coagulation. SARS-CoV-2 infection is associated with the development of coagulopathy with different degrees of severity. Various alterations have been described, consisting of a widespread micro-vessel thrombosis, elevated circulatory levels of prothrombin, D-dimer and its products of degradation, prolongation of the prothrombin time, thrombocytopenia, and reduction in fibrinogen amounts. ω-3 polyunsaturated fatty acids are able to modify the composition and structure of phospholipids incorporated into platelet membranes, therefore causing a modification in their physical properties, such as their stiffness and fluidity. This event is associated with a change in the biological characteristics of platelet plasmatic membranes and of the mediators, deriving from the metabolism of ω-3 polyunsaturated fatty acids. According to available data, these lipid molecules display antithrombotic activities, decreasing the stimulation of platelets and the process of their aggregation as well as modulating the process of coagulation by regulating the function of the molecules and factors involved in the activation of the thrombin, collagen, and adenosine diphosphate pathways [102]. 5) regulatory activity on renin/angiotensin/aldosterone system. In particular, SARS-CoV-2 can bind to the angiotensin-converting enzyme-2 (ACE2) receptor, causing its entry into the cells with its consequent down-regulation on the cell membrane surface. This event is associated with elevated levels of circulatory Angiotensin II. The rise in the amounts of this mediator causes harmful effects, including the release of pro-inflammatory cytokines, the enrolment of inflammatory cells, the stimulation of arterial vessel vasoconstriction, and the increase in ACE and blood pressure [[103], [104], [105]]. A recent review has summarized the potential pathogenetic role of an unbalanced stimulation, concerning the pathway ACE2/Angiotensin and derived (1–7) peptides as well as of the inhibition in the activity of renin and ACE/Ang II/AT1R axis in the development of some severe human pathological conditions, such as sepsis, severe acute pancreatitis, and COVID-19. The researchers have suggested that these distinct and different syndromes may display common pathogenetic mechanisms [42]. Therefore, the modulation of this cascade activity may contribute to preventing the arising of severe COVID-19 forms. A recent paper has suggested that spike protein impairs endothelial function via down-regulation of ACE-2 [75]. Based on these concepts, the adoption of diets including not only ω-3 polyunsaturated fatty acids, but also other nutrients, such as vitamin D, vitamin A, vitamin E, vitamin B1, vitamin B6, vitamin B12, vitamin C, Iron, Zinc, and Selenium may exert beneficial effects, mainly in some types of patients with Sars-CoV-2 infection [11,106,107] as well as some trials designed to investigate these perspectives are currently in progress [108]. Obese individuals are at high risk to develop severe forms of COVID-19. These individuals have a persistent pro-inflammatory status associated with a major probability of death due to cardiovascular-, respiratory-, neoplastic- causes. Moreover, they have a higher risk to develop more severe Sars-CoV-2 infections. These individuals have a persistent pro-inflammatory status associated with a major probability of death due to cardiovascular-, respiratory-, neoplastic- causes. Furthermore, they have a higher risk to develop more severe SARS-CoV-2 infections with a more elevated mortality rate in comparison with normal-weight people [109]. Some studies have demonstrated the beneficial role of parenteral nutrition enriched with ω-3 polyunsaturated fatty acids on survival outcomes in critically ill patients with sepsis. Taking advantage of these current data, it is conceivable that the supplementation of patients with severe forms of COVID-19, using the prior reported nutrients may have a real benefit. A large series of experimental studies in vitro, in animal models, and in humans, are suggesting that SPMs have a beneficial role in restoring homeostasis in tissues damaged by several infectious (deriving from both viral- or bacterial-pathogens) or non-infectious insults [110]. Several mechanisms are involved in these effects. These lipid-mediators may act by decreasing viral load and lung inflammation in mice, during Respiratory Syncytial Virus Infection [111], by reverting lipopolysaccharide-induced pulmonary endothelial damage in bacterial infections [112], by improving pulmonary edema fluid clearance via activation of the sodium channel and lymphatic drainage in lipopolysaccharide-induced ARDS [113]. SPMs may exert a protective or restoring function in endothelial dysfunction and endothelitis in patients with COVID-19, although no trials have investigated this topic and definitive conclusions are not available [75].
Furthermore, the possible cooperative activities of SPMs and fat-soluble vitamins, such as vitamin D have not been investigated, but it may represent an interesting research field. To date, only a small study has studied the effects of 1,25D3 and RvD1 on phagocytosis and inflammation in Alzheimer's disease patients. Both these compounds have been able to retune the imbalance between phagocytosis and inflammation in these individuals [114]. Based on the trials reporting beneficial effects of supplemental intravenous n-3 fatty acids administration and n-3 fatty acid status on the outcome of critically ill elderly patients in the ICU, some researchers have proposed the use of this therapeutic strategy also in patients with severe forms of SARS-CoV-2 infections [115]. However, although the data concerning this approach are promising, no definitive results are available [116]. Therefore, well-designed and sized trials evaluating this type of treatment in clinical practice are needed [[117], [118], [119]].
2. Conclusion
Inflammation is an essential protective response against infectious and non-infectious stimuli. Physiological inflammation eliminates pathogens, promotes repair and healing of tissues [1]. Some syndromes such as sepsis, ARDS [120], and MOF are caused by different pathological conditions, including polytrauma, burns, graft-versus-host disease, and chimeric antigen receptor (CAR)-T cell therapy as well as severe forms of infections due to bacteria, fungi, and viruses including SARS-CoV-2 [9,[58], [59], [60], [61]].
All above-mentioned syndromes are characterized by a condition of uncontrolled hyperinflammation associated with a condition of immunosuppression. If the phlogosis course is not controlled, the remodeling of the tissues can lead to the onset of severe pathological conditions, like sepsis and MOF [48]. The resolution of inflammation is an active process that occurs when the class switching of the lipid mediators of inflammation promotes the release of Lipoxins, Resolvins, Protectins, and Maresins [121]. This phase is characterized by the cessation of neutrophil recruitment and the enrolment and differentiation of macrophages. These cells clear apoptotic cells and tissue debris to restore tissue homeostasis. The resolving lipid mediators stimulate the resolution of inflammation by interrupting the recruitment of neutrophils, promote the efferocytosis of the apoptotic cells and the clearance of the cellular debris (which would maintain inflammation) by macrophages, increase the phagocytosis of microbes, down-regulate the synthesis of pro-inflammatory cytokines and prostanoids [121]. Based on the above mentioned pathogenetic mechanisms detectable in inflammatory processes, it is now known that some compounds, such as ω-3 polyunsaturated fatty acids, fat-soluble and water-soluble vitamins as well as some micronutrients such as iron, selenium, and zinc may have beneficial effects in critically ill patients with severe forms of sepsis and they have been proposed in clinical practice [122]. Taking advantage of these results and from the current knowledge about COVID-19, it has been suggested that some similarities may exist in the pathogenesis of these pathological conditions. Therefore, it has been suggested that a wide spectrum of nutrients may have a potentially useful role in the complementary treatment of patients with SARS-CoV-2 related infections [123]. These compounds might effectively counteract both the “eicosanoid and cytokine storm” which characterizes the inflammatory conditions, and which is also observed in individuals, suffering from COVID-19 with different degrees of severity [89,124,125].
Furthermore, it has been shown that SPMs may effectively down-regulate and attenuate the phlogistic process arising in ARDS [120]. Currently, no definitive data about this interesting therapeutic approach are available for the treatment of patients with COVID-19. Therefore, well-designed and well-sized trials are needed to investigate the real impact in the clinical practice of this promising strategy in individuals suffering from SARS-CoV-2 infection with different degrees of severity.
Declaration of Competing Interest
The authors declare no conflicts of interest regarding the publication of this paper.
References
- 1.Chen L., Deng H., Cui H., Fang J., Zuo Z., Deng J., Li Y., Wang X., Zhao L. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget. 2018;9:7204–7218. doi: 10.18632/oncotarget.23208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Medzhitov R. Origin and physiological roles of inflammation. Nature. 2008;454:428–435. doi: 10.1038/nature07201. [DOI] [PubMed] [Google Scholar]
- 3.Medzhitov R. Inflammation 2010: new adventures of an old flame. Cell. 2010;140:771–776. doi: 10.1016/j.cell.2010.03.006. [DOI] [PubMed] [Google Scholar]
- 4.Williams M.A., Bevan M.J. Effector and memory CTL differentiation. Annu. Rev. Immunol. 2007;25:171–192. doi: 10.1146/annurev.immunol.25.022106.141548. [DOI] [PubMed] [Google Scholar]
- 5.Chaudhry H., Zhou J., Zhong Y., Ali M.M., McGuire F., Nagarkatti P.S., Nagarkatti M. Role of cytokines as a double-edged sword in sepsis. In Vivo. 2013;27:669–684. [PMC free article] [PubMed] [Google Scholar]
- 6.Chousterman B.G., Swirski F.K., Weber G.F. Cytokine storm and sepsis disease pathogenesis. Semin. Immunopathol. 2017;39:517–528. doi: 10.1007/s00281-017-0639-8. [DOI] [PubMed] [Google Scholar]
- 7.Harrison C. Sepsis: calming the cytokine storm. Nat. Rev. Drug Discov. 2010;9:360–361. doi: 10.1038/nrd3162. [DOI] [PubMed] [Google Scholar]
- 8.Mizgerd J.P. Acute lower respiratory tract infection. N. Engl. J. Med. 2008;358:716–727. doi: 10.1056/NEJMra074111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tisoncik J.R., Korth M.J., Simmons C.P., Farrar J., Martin T.R., Katze M.G. Into the eye of the cytokine storm. Microbiol. Mol. Biol. Rev. 2012;76:16–32. doi: 10.1128/MMBR.05015-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ye Q., Wang B., Mao J. The pathogenesis and treatment of the `Cytokine Storm’ in COVID-19. J. Infect. 2020;80:607–613. doi: 10.1016/j.jinf.2020.03.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fiorino S., Gallo C., Zippi M., Sabbatani S., Manfredi R., Moretti R., Fogacci E., Maggioli C., Travasoni Loffredo F., Giampieri E., et al. Cytokine storm in aged people with CoV-2: possible role of vitamins as therapy or preventive strategy. Aging Clin. Exp. Res. 2020;32:2115–2131. doi: 10.1007/s40520-020-01669-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Panigrahy D., Gilligan M.M., Serhan C.N., Kashfi K. Resolution of inflammation: an organizing principle in biology and medicine. Pharmacol. Ther. 2021;227 doi: 10.1016/j.pharmthera.2021.107879. [DOI] [PubMed] [Google Scholar]
- 13.Serhan C.N., Clish C.B., Brannon J., Colgan S.P., Chiang N., Gronert K. Novel functional sets of lipid-derived mediators with antiinflammatory actions generated from omega-3 fatty acids via cyclooxygenase 2-nonsteroidal antiinflammatory drugs and transcellular processing. J. Exp. Med. 2000;192:1197–1204. doi: 10.1084/jem.192.8.1197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Serhan C.N., Brain S.D., Buckley C.D., Gilroy D.W., Haslett C., O’Neill L.A., Perretti M., Rossi A.G., Wallace J.L. Resolution of inflammation: state of the art, definitions and terms. FASEB J. 2007;21:325–332. doi: 10.1096/fj.06-7227rev. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Serhan C.N., Petasis N.A. Resolvins and protectins in inflammation resolution. Chem. Rev. 2011;111:5922–5943. doi: 10.1021/cr100396c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kawasaki T., Kawai T. Toll-like receptor signaling pathways. Front. Immunol. 2014;5:461. doi: 10.3389/fimmu.2014.00461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Akira S., Uematsu S., Takeuchi O. Pathogen recognition and innate immunity. Cell. 2006;124:783–801. doi: 10.1016/j.cell.2006.02.015. [DOI] [PubMed] [Google Scholar]
- 18.von Moltke J., Ayres J.S., Kofoed E.M., Chavarria-Smith J., Vance R.E. Recognition of bacteria by inflammasomes. Annu. Rev. Immunol. 2013;31:73–106. doi: 10.1146/annurev-immunol-032712-095944. [DOI] [PubMed] [Google Scholar]
- 19.Dennis E.A., Norris P.C. Eicosanoid storm in infection and inflammation. Nat. Rev. Immunol. 2015;15:511–523. doi: 10.1038/nri3859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kasuga K., Yang R., Porter T.F., Agrawal N., Petasis N.A., Irimia D., Toner M., Serhan C.N. Rapid appearance of resolvin precursors in inflammatory exudates: novel mechanisms in resolution. J. Immunol. 2008;181:8677–8687. doi: 10.4049/jimmunol.181.12.8677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Plagge M., Laskay T. Early production of the neutrophil-derived lipid mediators LTB4 and LXA4 is modulated by intracellular infection with leishmania major. Biomed Res. Int. 2017;2017 doi: 10.1155/2017/2014583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Martinez-Colon G.J., Moore B.B. Prostaglandin E2 as a regulator of immunity to pathogens. Pharmacol. Ther. 2018;185:135–146. doi: 10.1016/j.pharmthera.2017.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Harizi H. Reciprocal crosstalk between dendritic cells and natural killer cells under the effects of PGE2 in immunity and immunopathology. Cell. Mol. Immunol. 2013;10:213–221. doi: 10.1038/cmi.2013.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kalinski P. Regulation of immune responses by prostaglandin E2. J. Immunol. 2012;188:21–28. doi: 10.4049/jimmunol.1101029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dinarello C.A., Simon A., van der Meer J.W. Treating inflammation by blocking interleukin-1 in a broad spectrum of diseases. Nat. Rev. Drug Discov. 2012;11:633–652. doi: 10.1038/nrd3800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ortega-Gomez A., Perretti M., Soehnlein O. Resolution of inflammation: an integrated view. EMBO Mol. Med. 2013;5:661–674. doi: 10.1002/emmm.201202382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sugimoto M.A., Sousa L.P., Pinho V., Perretti M., Teixeira M.M. Resolution of inflammation: what controls its onset? Front. Immunol. 2016;7:160. doi: 10.3389/fimmu.2016.00160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Levy B.D., Clish C.B., Schmidt B., Gronert K., Serhan C.N. Lipid mediator class switching during acute inflammation: signals in resolution. Nat. Immunol. 2001;2:612–619. doi: 10.1038/89759. [DOI] [PubMed] [Google Scholar]
- 29.Serhan C.N. Pro-resolving lipid mediators are leads for resolution physiology. Nature. 2014;510:92–101. doi: 10.1038/nature13479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Serhan C.N. Lipoxins and novel aspirin-triggered 15-epi-lipoxins (ATL): a jungle of cell-cell interactions or a therapeutic opportunity? Prostaglandins. 1997;53:107–137. doi: 10.1016/s0090-6980(97)00001-4. [DOI] [PubMed] [Google Scholar]
- 31.Buckley C.D., Gilroy D.W., Serhan C.N. Proresolving lipid mediators and mechanisms in the resolution of acute inflammation. Immunity. 2014;40:315–327. doi: 10.1016/j.immuni.2014.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Dalli J., Serhan C. Macrophage proresolving mediators-the when and where. Microbiol. Spectr. 2016;4 doi: 10.1128/microbiolspec.MCHD-0001-2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Dalli J., Serhan C.N. Pro-resolving mediators in regulating and conferring macrophage function. Front. Immunol. 2017;8:1400. doi: 10.3389/fimmu.2017.01400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fredman G., Tabas I. Boosting inflammation resolution in atherosclerosis: the next frontier for therapy. Am. J. Pathol. 2017;187:1211–1221. doi: 10.1016/j.ajpath.2017.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Norris P.C., Skulas-Ray A.C., Riley I., Richter C.K., Kris-Etherton P.M., Jensen G.L., Serhan C.N., Maddipati K.R. Identification of specialized pro-resolving mediator clusters from healthy adults after intravenous low-dose endotoxin and omega-3 supplementation: a methodological validation. Sci. Rep. 2018;8:18050. doi: 10.1038/s41598-018-36679-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Godson C., Mitchell S., Harvey K., Petasis N.A., Hogg N., Brady H.R. Cutting edge: lipoxins rapidly stimulate nonphlogistic phagocytosis of apoptotic neutrophils by monocyte-derived macrophages. J. Immunol. 2000;164:1663–1667. doi: 10.4049/jimmunol.164.4.1663. [DOI] [PubMed] [Google Scholar]
- 37.Morita M., Kuba K., Ichikawa A., Nakayama M., Katahira J., Iwamoto R., Watanebe T., Sakabe S., Daidoji T., Nakamura S., et al. The lipid mediator protectin D1 inhibits influenza virus replication and improves severe influenza. Cell. 2013;153:112–125. doi: 10.1016/j.cell.2013.02.027. [DOI] [PubMed] [Google Scholar]
- 38.Chiang N., Fredman G., Backhed F., Oh S.F., Vickery T., Schmidt B.A., Serhan C.N. Infection regulates pro-resolving mediators that lower antibiotic requirements. Nature. 2012;484:524–528. doi: 10.1038/nature11042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ramon S., Baker S.F., Sahler J.M., Kim N., Feldsott E.A., Serhan C.N., Martinez-Sobrido L., Topham D.J., Phipps R.P. The specialized proresolving mediator 17-HDHA enhances the antibody-mediated immune response against influenza virus: a new class of adjuvant? J. Immunol. 2014;193:6031–6040. doi: 10.4049/jimmunol.1302795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Recchiuti A., Mattoscio D., Isopi E. Roles, actions, and therapeutic potential of specialized pro-resolving lipid mediators for the treatment of inflammation in cystic fibrosis. Front. Pharmacol. 2019;10:252. doi: 10.3389/fphar.2019.00252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Spite M., Norling L.V., Summers L., Yang R., Cooper D., Petasis N.A., Flower R.J., Perretti M., Serhan C.N. Resolvin D2 is a potent regulator of leukocytes and controls microbial sepsis. Nature. 2009;461:1287–1291. doi: 10.1038/nature08541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Gallo C.G., Fiorino S., Posabella G., Antonacci D., Tropeano A., Pausini E., Pausini C., Guarniero T., Hong W., Giampieri E., et al. COVID-19, what could sepsis, severe acute pancreatitis, gender differences, and aging teach us? Cytokine. 2021;148 doi: 10.1016/j.cyto.2021.155628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Italiani P., Boraschi D. From monocytes to M1/M2 macrophages: phenotypical vs. functional differentiation. Front. Immunol. 2014;5:514. doi: 10.3389/fimmu.2014.00514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Serhan C.N. Treating inflammation and infection in the 21st century: new hints from decoding resolution mediators and mechanisms. FASEB J. 2017;31:1273–1288. doi: 10.1096/fj.201601222R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Gudernatsch V., Stefanczyk S.A., Mirakaj V. Novel resolution mediators of severe systemic inflammation. Immunotargets Ther. 2020;9:31–41. doi: 10.2147/ITT.S243238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Seven days in medicine: 8-14 Jan 2020. BMJ. 2020;368:m132. doi: 10.1136/bmj.m132. [DOI] [Google Scholar]
- 47.Zheng M., Gao Y., Wang G., Song G., Liu S., Sun D., Xu Y., Tian Z. Functional exhaustion of antiviral lymphocytes in COVID-19 patients. Cell. Mol. Immunol. 2020;17:533–535. doi: 10.1038/s41423-020-0402-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zaim S., Chong J.H., Sankaranarayanan V., Harky A. COVID-19 and multiorgan response. Curr. Probl. Cardiol. 2020;45 doi: 10.1016/j.cpcardiol.2020.100618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Fiorino S., Tateo F., Biase D., Gallo C.G., Orlandi P.E., Corazza I., Budriesi R., Micucci M., Visani M., Loggi E., et al. SARS-CoV-2: lessons from both the history of medicine and from the biological behavior of other well-known viruses. Future Microbiol. 2021;16:1105–1133. doi: 10.2217/fmb-2021-0064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hammock B.D., Wang W., Gilligan M.M., Panigrahy D. Eicosanoids: the overlooked storm in coronavirus disease 2019 (COVID-19)? Am. J. Pathol. 2020;190:1782–1788. doi: 10.1016/j.ajpath.2020.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.von Moltke J., Trinidad N.J., Moayeri M., Kintzer A.F., Wang S.B., van Rooijen N., Brown C.R., Krantz B.A., Leppla S.H., Gronert K., et al. Rapid induction of inflammatory lipid mediators by the inflammasome in vivo. Nature. 2012;490:107–111. doi: 10.1038/nature11351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Islam A.B., Dave M., Amin S., Jensen R.V., Amin A.R. Genomic, lipidomic and metabolomic analysis of cyclooxygenase-null cells: eicosanoid storm, cross talk, and compensation by COX-1. Genomics Proteomics Bioinformatics. 2016;14:81–93. doi: 10.1016/j.gpb.2014.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Cron R., Behrens E.M. In: Cytokine Storm Syndrome. Publishing, C.S.N.S.A.S.I, editor. 2019. [Google Scholar]
- 54.Leisman D.E., Ronner L., Pinotti R., Taylor M.D., Sinha P., Calfee C.S., Hirayama A.V., Mastroiani F., Turtle C.J., Harhay M.O., et al. Cytokine elevation in severe and critical COVID-19: a rapid systematic review, meta-analysis, and comparison with other inflammatory syndromes. Lancet Respir. Med. 2020;8:1233–1244. doi: 10.1016/S2213-2600(20)30404-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J., Hlh Across Speciality Collaboration, U.K COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395:1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Jose R.J., Manuel A. COVID-19 cytokine storm: the interplay between inflammation and coagulation. Lancet Respir. Med. 2020;8:e46–e47. doi: 10.1016/S2213-2600(20)30216-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Lopez-Collazo E., Avendano-Ortiz J., Martin-Quiros A., Aguirre L.A. Immune response and COVID-19: a mirror image of Sepsis. Int. J. Biol. Sci. 2020;16:2479–2489. doi: 10.7150/ijbs.48400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Fitzgerald J.C., Weiss S.L., Maude S.L., Barrett D.M., Lacey S.F., Melenhorst J.J., Shaw P., Berg R.A., June C.H., Porter D.L., et al. Cytokine release syndrome after chimeric antigen receptor T cell therapy for acute lymphoblastic leukemia. Crit. Care Med. 2017;45:e124–e131. doi: 10.1097/CCM.0000000000002053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Song F., Hou J., Chen Z., Cheng B., Lei R., Cui P., Sun Y., Wang H., Fang X. Sphingosine-1-phosphate receptor 2 signaling promotes caspase-11-dependent macrophage pyroptosis and worsens Escherichia coli sepsis outcome. Anesthesiology. 2018;129:311–320. doi: 10.1097/ALN.0000000000002196. [DOI] [PubMed] [Google Scholar]
- 60.Tang Y., Liu J., Zhang D., Xu Z., Ji J., Wen C. Cytokine storm in COVID-19: the current evidence and treatment strategies. Front. Immunol. 2020;11:1708. doi: 10.3389/fimmu.2020.01708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wong J.P., Viswanathan S., Wang M., Sun L.Q., Clark G.C., D’Elia R.V. Current and future developments in the treatment of virus-induced hypercytokinemia. Future Med. Chem. 2017;9:169–178. doi: 10.4155/fmc-2016-0181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Li H., Liu L., Zhang D., Xu J., Dai H., Tang N., Su X., Cao B. SARS-CoV-2 and viral sepsis: observations and hypotheses. Lancet. 2020;395:1517–1520. doi: 10.1016/S0140-6736(20)30920-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Bonaventura A., Vecchie A., Dagna L., Martinod K., Dixon D.L., Van Tassell B.W., Dentali F., Montecucco F., Massberg S., Levi M., et al. Endothelial dysfunction and immunothrombosis as key pathogenic mechanisms in COVID-19. Nat. Rev. Immunol. 2021;21:319–329. doi: 10.1038/s41577-021-00536-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Loo J., Spittle D.A., Newnham M. COVID-19, immunothrombosis and venous thromboembolism: biological mechanisms. Thorax. 2021;76:412–420. doi: 10.1136/thoraxjnl-2020-216243. [DOI] [PubMed] [Google Scholar]
- 65.Engelmann B., Massberg S. Thrombosis as an intravascular effector of innate immunity. Nat. Rev. Immunol. 2013;13:34–45. doi: 10.1038/nri3345. [DOI] [PubMed] [Google Scholar]
- 66.McFadyen J.D., Stevens H., Peter K. The emerging threat of (micro)thrombosis in COVID-19 and its therapeutic implications. Circ. Res. 2020;127:571–587. doi: 10.1161/CIRCRESAHA.120.317447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Amara U., Rittirsch D., Flierl M., Bruckner U., Klos A., Gebhard F., Lambris J.D., Huber-Lang M. Interaction between the coagulation and complement system. Adv. Exp. Med. Biol. 2008;632:71–79. doi: 10.1007/978-0-387-78952-1_6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ganter M.T., Brohi K., Cohen M.J., Shaffer L.A., Walsh M.C., Stahl G.L., Pittet J.F. Role of the alternative pathway in the early complement activation following major trauma. Shock. 2007;28:29–34. doi: 10.1097/shk.0b013e3180342439. [DOI] [PubMed] [Google Scholar]
- 69.Huber-Lang M., Sarma J.V., Zetoune F.S., Rittirsch D., Neff T.A., McGuire S.R., Lambris J.D., Warner R.L., Flierl M.A., Hoesel L.M., et al. Generation of C5a in the absence of C3: a new complement activation pathway. Nat. Med. 2006;12:682–687. doi: 10.1038/nm1419. [DOI] [PubMed] [Google Scholar]
- 70.Feng S., Liang X., Kroll M.H., Chung D.W., Afshar-Kharghan V. Von Willebrand factor is a cofactor in complement regulation. Blood. 2015;125:1034–1037. doi: 10.1182/blood-2014-06-585430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Wojta J., Kaun C., Zorn G., Ghannadan M., Hauswirth A.W., Sperr W.R., Fritsch G., Printz D., Binder B.R., Schatzl G., et al. C5a stimulates production of plasminogen activator inhibitor-1 in human mast cells and basophils. Blood. 2002;100:517–523. doi: 10.1182/blood.v100.2.517. [DOI] [PubMed] [Google Scholar]
- 72.Lee C.H. Role of specialized pro-resolving lipid mediators and their receptors in virus infection: a promising therapeutic strategy for SARS-CoV-2 cytokine storm. Arch. Pharm. Res. 2021;44:84–98. doi: 10.1007/s12272-020-01299-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Foley J.H., Conway E.M. Cross talk pathways between coagulation and inflammation. Circ. Res. 2016;118:1392–1408. doi: 10.1161/CIRCRESAHA.116.306853. [DOI] [PubMed] [Google Scholar]
- 74.Ritis K., Doumas M., Mastellos D., Micheli A., Giaglis S., Magotti P., Rafail S., Kartalis G., Sideras P., Lambris J.D.A. Novel C5a receptor-tissue factor cross-talk in neutrophils links innate immunity to coagulation pathways. J. Immunol. 2006;177:4794–4802. doi: 10.4049/jimmunol.177.7.4794. [DOI] [PubMed] [Google Scholar]
- 75.Lei Y., Zhang J., Schiavon C.R., He M., Chen L., Shen H., Zhang Y., Yin Q., Cho Y., Andrade L., et al. SARS-CoV-2 spike protein impairs endothelial function via downregulation of ACE 2. Circ. Res. 2021;128:1323–1326. doi: 10.1161/CIRCRESAHA.121.318902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Iba T., Levy J.H., Levi M., Connors J.M., Thachil J. Coagulopathy of coronavirus disease 2019. Crit. Care Med. 2020;48:1358–1364. doi: 10.1097/CCM.0000000000004458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Tang N., Li D., Wang X., Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thromb. Haemost. 2020;18:844–847. doi: 10.1111/jth.14768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Massberg S., Grahl L., von Bruehl M.L., Manukyan D., Pfeiler S., Goosmann C., Brinkmann V., Lorenz M., Bidzhekov K., Khandagale A.B., et al. Reciprocal coupling of coagulation and innate immunity via neutrophil serine proteases. Nat. Med. 2010;16:887–896. doi: 10.1038/nm.2184. [DOI] [PubMed] [Google Scholar]
- 79.Carvelli J., Demaria O., Vely F., Batista L., Chouaki Benmansour N., Fares J., Carpentier S., Thibult M.L., Morel A., Remark R., et al. Association of COVID-19 inflammation with activation of the C5a-C5aR1 axis. Nature. 2020;588:146–150. doi: 10.1038/s41586-020-2600-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Noris M., Benigni A., Remuzzi G. The case of complement activation in COVID-19 multiorgan impact. Kidney Int. 2020;98:314–322. doi: 10.1016/j.kint.2020.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Skendros P., Mitsios A., Chrysanthopoulou A., Mastellos D.C., Metallidis S., Rafailidis P., Ntinopoulou M., Sertaridou E., Tsironidou V., Tsigalou C., et al. Complement and tissue factor-enriched neutrophil extracellular traps are key drivers in COVID-19 immunothrombosis. J. Clin. Invest. 2020;130:6151–6157. doi: 10.1172/JCI141374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Dalli J. Does promoting resolution instead of inhibiting inflammation represent the new paradigm in treating infections? Mol. Aspects Med. 2017;58:12–20. doi: 10.1016/j.mam.2017.03.007. [DOI] [PubMed] [Google Scholar]
- 83.Sandhaus S., Swick A.G. Specialized proresolving mediators in infection and lung injury. Biofactors. 2021;47:6–18. doi: 10.1002/biof.1691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Basil M.C., Levy B.D. Specialized pro-resolving mediators: endogenous regulators of infection and inflammation. Nat. Rev. Immunol. 2016;16:51–67. doi: 10.1038/nri.2015.4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Cilloniz C., Pantin-Jackwood M.J., Ni C., Goodman A.G., Peng X., Proll S.C., Carter V.S., Rosenzweig E.R., Szretter K.J., Katz J.M., et al. Lethal dissemination of H5N1 influenza virus is associated with dysregulation of inflammation and lipoxin signaling in a mouse model of infection. J. Virol. 2010;84:7613–7624. doi: 10.1128/JVI.00553-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Tam V.C., Quehenberger O., Oshansky C.M., Suen R., Armando A.M., Treuting P.M., Thomas P.G., Dennis E.A., Aderem A. Lipidomic profiling of influenza infection identifies mediators that induce and resolve inflammation. Cell. 2013;154:213–227. doi: 10.1016/j.cell.2013.05.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Chiang N., Serhan C.N. Structural elucidation and physiologic functions of specialized pro-resolving mediators and their receptors. Mol. Aspects Med. 2017;58:114–129. doi: 10.1016/j.mam.2017.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Levy B.D., Serhan C.N. Resolution of acute inflammation in the lung. Annu. Rev. Physiol. 2014;76:467–492. doi: 10.1146/annurev-physiol-021113-170408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Rogero M.M., Leao M.C., Santana T.M., Pimentel M., Carlini G.C.G., da Silveira T.F.F., Goncalves R.C., Castro I.A. Potential benefits and risks of omega-3 fatty acids supplementation to patients with COVID-19. Free Radic. Biol. Med. 2020;156:190–199. doi: 10.1016/j.freeradbiomed.2020.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Shen B., Yi X., Sun Y., Bi X., Du J., Zhang C., Quan S., Zhang F., Sun R., Qian L., et al. Proteomic and metabolomic characterization of COVID-19 patient sera. Cell. 2020;182:59–72. doi: 10.1016/j.cell.2020.05.032. e15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Song J.W., Lam S.M., Fan X., Cao W.J., Wang S.Y., Tian H., Chua G.H., Zhang C., Meng F.P., Xu Z., et al. Omics-driven systems interrogation of metabolic dysregulation in COVID-19 pathogenesis. Cell Metab. 2020;32:188–202. doi: 10.1016/j.cmet.2020.06.016. e185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Lopez-Reyes A., Martinez-Armenta C., Espinosa-Velazquez R., Vazquez-Cardenas P., Cruz-Ramos M., Palacios-Gonzalez B., Gomez-Quiroz L.E., Martinez-Nava G.A. NLRP3 inflammasome: the stormy link between obesity and COVID-19. Front. Immunol. 2020;11 doi: 10.3389/fimmu.2020.570251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Sharifi Y., Payab M., Mohammadi-Vajari E., Aghili S.M.M., Sharifi F., Mehrdad N., Kashani E., Shadman Z., Larijani B., Ebrahimpur M. Association between cardiometabolic risk factors and COVID-19 susceptibility, severity and mortality: a review. J. Diabetes Metab. Disord. 2021:1–23. doi: 10.1007/s40200-021-00822-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Schwarz B., Sharma L., Roberts L., Peng X., Bermejo S., Leighton I., Casanovas-Massana A., Minasyan M., Farhadian S., Ko A.I., et al. Cutting edge: severe SARS-CoV-2 infection in humans is defined by a shift in the serum lipidome, resulting in dysregulation of eicosanoid immune mediators. J. Immunol. 2021;206:329–334. doi: 10.4049/jimmunol.2001025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Recchiuti A., Patruno S., Mattoscio D., Isopi E., Pomilio A., Lamolinara A., Iezzi M., Pecce R., Romano M. Resolvin D1 and D2 reduce SARS-CoV-2-induced inflammatory responses in cystic fibrosis macrophages. FASEB J. 2021;35:e21441. doi: 10.1096/fj.202001952R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Regidor P.A.C. ovid-19 management with inflammation resolving mediators? Perspectives and potential. Med. Hypotheses. 2020;142 doi: 10.1016/j.mehy.2020.109813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Torrinhas R.S., Calder P.C., Lemos G.O., Waitzberg D.L. Parenteral fish oil: an adjuvant pharmacotherapy for coronavirus disease 2019? Nutrition. 2020;81 doi: 10.1016/j.nut.2020.110900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Baglivo M., Baronio M., Natalini G., Beccari T., Chiurazzi P., Fulcheri E., Petralia P.P., Michelini S., Fiorentini G., Miggiano G.A., et al. Natural small molecules as inhibitors of coronavirus lipid-dependent attachment to host cells: a possible strategy for reducing SARS-COV-2 infectivity? Acta Biomed. 2020;91:161–164. doi: 10.23750/abm.v91i1.9402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Guo H., Huang M., Yuan Q., Wei Y., Gao Y., Mao L., Gu L., Tan Y.W., Zhong Y., Liu D., et al. The important role of lipid raft-mediated attachment in the infection of cultured cells by coronavirus infectious bronchitis virus beaudette strain. PLoS One. 2017;12 doi: 10.1371/journal.pone.0170123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Lajoie P., Nabi I.R. Regulation of raft-dependent endocytosis. J. Cell. Mol. Med. 2007;11:644–653. doi: 10.1111/j.1582-4934.2007.00083.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Das U.N. Essential fatty acids and their metabolites in the pathobiology of COVID-19. Nutrition. 2020 doi: 10.1016/j.nut.2020.111052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Tsoupras A., Lordan R., Zabetakis I. Thrombosis and COVID-19: the potential role of nutrition. Front. Nutr. 2020;7 doi: 10.3389/fnut.2020.583080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Ben-Eltriki M., Hopefl R., Wright J.M., Deb S. Association between vitamin D status and risk of developing severe COVID-19 infection: a meta-analysis of observational studies. J. Am. Coll. Nutr. 2021:1–11. doi: 10.1080/07315724.2021.1951891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Nikniaz L., Akbarzadeh M.A., Hosseinifard H., Mohammad-Salar Hosseini M.S. The impact of vitamin D supplementation on mortality rate and clinical outcomes of COVID-19 patients: a systematic review and meta-analysis. BMJ. 2021 doi: 10.1101/2021.01.04.21249219. In medrxiv. [DOI] [Google Scholar]
- 105.Zaloga G.P. Narrative review of n-3 polyunsaturated fatty acid supplementation upon immune functions, resolution molecules and lipid peroxidation. Nutrients. 2021;13 doi: 10.3390/nu13020662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Galmes S., Serra F., Palou A. Current state of evidence: influence of nutritional and nutrigenetic factors on immunity in the COVID-19 pandemic framework. Nutrients. 2020;12 doi: 10.3390/nu12092738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Name J.J., Souza A.C.R., Vasconcelos A.R., Prado P.S., Pereira C.P.M. Zinc, vitamin D. and vitamin C: perspectives for COVID-19 with a focus on physical tissue barrier integrity. Front. Nutr. 2020;7 doi: 10.3389/fnut.2020.606398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Vitamin D., Omega-3, and Combination Vitamins B., C and Zinc Supplementation for the Treatment and Prevention of COVID-19. Availabe online: https://clinicaltrials.gov/ct2/show/NCT04828538 (accessed on September 23, 2021).
- 109.Sahin E., Orhan C., Uckun F.M., Sahin K. Clinical impact potential of supplemental nutrients as adjuncts of therapy in high-risk COVID-19 for obese patients. Front. Nutr. 2020;7 doi: 10.3389/fnut.2020.580504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.de la Rosa X., Norris P.C., Chiang N., Rodriguez A.R., Spur B.W., Serhan C.N. Identification and complete stereochemical assignments of the new resolvin conjugates in tissue regeneration in human tissues that stimulate proresolving phagocyte functions and tissue regeneration. Am. J. Pathol. 2018;188:950–966. doi: 10.1016/j.ajpath.2018.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Walker K.H., Krishnamoorthy N., Bruggemann T.R., Shay A.E., Serhan C.N., Levy B.D. Protectins PCTR1 and PD1 reduce viral load and lung inflammation during respiratory syncytial virus infection in mice. Front. Immunol. 2021;12 doi: 10.3389/fimmu.2021.704427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Wang X.Y., Li X.Y., Wu C.H., Hao Y., Fu P.H., Mei H.X., Chen F., Gong Y.Q., Jin S.W., Li H. Protectin conjugates in tissue regeneration 1 restores lipopolysaccharide-induced pulmonary endothelial glycocalyx loss via ALX/SIRT1/NF-kappa B axis. Respir. Res. 2021;22:193. doi: 10.1186/s12931-021-01793-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Zhang P.H., Han J., Cao F., Liu Y.J., Tian C., Wu C.H., Smith F.G., Hao Y., Jin S.W. PCTR1 improves pulmonary edema fluid clearance through activating the sodium channel and lymphatic drainage in lipopolysaccharide-induced ARDS. J. Cell. Physiol. 2020;235:9510–9523. doi: 10.1002/jcp.29758. [DOI] [PubMed] [Google Scholar]
- 114.Mizwicki M.T., Liu G., Fiala M., Magpantay L., Sayre J., Siani A., Mahanian M., Weitzman R., Hayden E.Y., Rosenthal M.J., et al. 1alpha,25-dihydroxyvitamin D3 and resolvin D1 retune the balance between amyloid-beta phagocytosis and inflammation in Alzheimer’s disease patients. J. Alzheimers Dis. 2013;34:155–170. doi: 10.3233/JAD-121735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Doaei S., Gholami S., Rastgoo S., Gholamalizadeh M., Bourbour F., Bagheri S.E., Samipoor F., Akbari M.E., Shadnoush M., Ghorat F., et al. The effect of omega-3 fatty acid supplementation on clinical and biochemical parameters of critically ill patients with COVID-19: a randomized clinical trial. J. Transl. Med. 2021;19:128. doi: 10.1186/s12967-021-02795-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Darwesh A.M., Bassiouni W., Sosnowski D.K., Seubert J.M. Can N-3 polyunsaturated fatty acids be considered a potential adjuvant therapy for COVID-19-associated cardiovascular complications? Pharmacol. Ther. 2021;219 doi: 10.1016/j.pharmthera.2020.107703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Barros K.V., Cassulino A.P., Schalch L., Della Valle Munhoz E., Manetta J.A., Noakes P.S., Miles E.A., Calder P.C., Flor Silveira V.L. Supplemental intravenous n-3 fatty acids and n-3 fatty acid status and outcome in critically ill elderly patients in the ICU receiving enteral nutrition. Clin. Nutr. 2013;32:599–605. doi: 10.1016/j.clnu.2012.10.016. [DOI] [PubMed] [Google Scholar]
- 118.Hall T.C., Bilku D.K., Al-Leswas D., Neal C.P., Horst C., Cooke J., Metcalfe M.S., Dennison A.R.A. Randomized controlled trial investigating the effects of parenteral fish oil on survival outcomes in critically ill patients with sepsis: a pilot study. JPEN J. Parenter. Enteral Nutr. 2015;39:301–312. doi: 10.1177/0148607113518945. [DOI] [PubMed] [Google Scholar]
- 119.Torrinhas R.S., Calder P.C., Waitzberg D.L. Response to Bistrian BR. Parenteral fish-oil emulsions in critically ill COVID-19 emulsions. JPEN J. Parenter. Enteral Nutr. 2020 doi: 10.1002/jpen.1933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Krishnamoorthy N., Abdulnour R.E., Walker K.H., Engstrom B.D., Levy B.D. Specialized proresolving mediators in innate and adaptive immune responses in airway diseases. Physiol. Rev. 2018;98:1335–1370. doi: 10.1152/physrev.00026.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Serhan C.N., Levy B.D. Resolvins in inflammation: emergence of the pro-resolving superfamily of mediators. J. Clin. Invest. 2018;128:2657–2669. doi: 10.1172/JCI97943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Garcia de Acilu M., Leal S., Caralt B., Roca O., Sabater J., Masclans J.R. The role of Omega-3 polyunsaturated fatty acids in the treatment of patients with acute respiratory distress syndrome: a clinical review. Biomed Res. Int. 2015;2015 doi: 10.1155/2015/653750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Fiorino S., Zippi M., Gallo C., Sifo D., Sabbatani S., Manfredi R., Rasciti E., Rasciti L., Giampieri E., Corazza I., et al. The rationale for a multi-step therapeutic approach based on antivirals, drugs and nutrients with immunomodulatory activity in patients with coronavirus-SARS2-induced disease of different severities. Br. J. Nutr. 2020:1–19. doi: 10.1017/S0007114520002913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Pal A., Gowdy K.M., Oestreich K.J., Beck M., Shaikh S.R. Obesity-driven deficiencies of specialized pro-resolving mediators may drive adverse outcomes during SARS-CoV-2 infection. Front. Immunol. 2020;11:1997. doi: 10.3389/fimmu.2020.01997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Panigrahy D., Gilligan M.M., Huang S., Gartung A., Cortes-Puch I., Sime P.J., Phipps R.P., Serhan C.N., Hammock B.D. Inflammation resolution: a dual-pronged approach to averting cytokine storms in COVID-19? Cancer Metastasis Rev. 2020;39:337–340. doi: 10.1007/s10555-020-09889-4. [DOI] [PMC free article] [PubMed] [Google Scholar]