Abstract
Context:
Carcinogenesis causes much human misery. It is a process involving multistage alterations. Medicinal plants are candidates for beneficial anticancer agents.
Objectives:
Investigation of anticancer proficiencies of the plant Dicliptera roxburghiana.
Material and methods:
Crude extract and derived fractions were inspected for their inhibitory potential against nuclear factor KB (NFκB), nitric oxide synthase inhibition, aromatase inhibition and induction of quinone reductase 1 (QR 1). Antiproliferative activity was determined by using various cancer cell lines for example hormone responsive breast cancer cell line MCF-7, estrogen receptor negative breast cancer cell line MDA-MB-231, murine hepatoma cells Hepa 1c1c7, human neuroblastoma cells SK-N-SH and neuroblastoma cells MYCN-2.
Results:
Ethyl acetate and n-butanol fractions of D. roxburghiana were strongly active against NFκB with IC50 of 16.6 ± 1.3 and 8.4 ± 0.7 µg/ml respectively with 100% survival. Chloroform fraction of the plant exhibited an induction ratio of 2.4 ± 0.09 with CD value of 17.7 µg/ml. Regarding the nitrite assay, the n-hexane fraction exhibited significant inhibition of NO activity with IC50 of 17.8 ± 1.25 µg/ml. The n-butanol fraction exhibited strong antiproliferative activity against IcIc-7 cell lines with IC50 values of 13.6 ± 1.91 µg/ml; against MYCN-2 a cytotoxic effect developed with dose dependence, with IC50 of 12.6 ± 1.24 µg/ml. In antiproliferative activity against SK-N-SH cell lines, chloroform, ethyl acetate and n-butanol fractions were efficiently active with IC50 values of 11.2 ± 0.84, 14.6 ± 1.71 and 16.3 ± 1.57 respectively.
Discussion and Conclusion:
It was demonstrated that various fractions of D. roxburghiana displayed appreciable anticancer characteristics and could be a potent source for the development of anticancer leads.
Keywords: Dicliptera roxburghiana, NFκB, quinone reductase 1, cytotoxicity, cancer chemoprevention
Introduction
Carcinogenesis is a deadly and miserable fact of the modern world and is regarded as a deleterious phenomenon. It involves multistage alterations in the genetic makeup of the normal cells provoked by carcinogens or by the mismanagement of DNA repair systems of the cell. Initially a single mutated cell undergoes the promotion stage which results in the production of a tumor bulk of extremely proliferative cells. 1 Further progression of these proliferative cells lead to the development of tumor cells which undergo nonstop division that outstrip their normal counterparts.2,3 A flurry of research data demonstrated the intense need to develop new strategies against these bleak realities and to investigate potential naturally derived candidates with the capability to halt these modifications and ultimately stop carcinogenesis. 4 In this context a number of publications that provide evidence for positive prospective of plant extracts and their derived compounds as anticarcinogenic agents with cancer chemopreventive characteristics, are readily available.1,5 -7
Many diversified lines of research have resulted in several approved anticancer drugs are plant based. Example includes paclitaxel from Taxus brevifolia; vinblastine and vincristine from Catharanthus roseus8,9 Two types of cancer screenings assays i.e. chemopreventive assays and antiproliferative assays are practiced widely. Cancer chemopreventive assessments are important to investigate the cancer retarding potential of botanicals. Cancer chemopreventive screenings monitor various outcomes for example TNF-α mediated nuclear factor kappa-B (NFκB) inhibitor, nitric oxide inhibitory, aromatase inhibitory screening, and induction of quinone reductase 1.
Nuclear factor kappa is a light chain enhancer of activated B cells (NFκB) and is basically confined to the cytoplasm but on activation can translocate to the nucleus where it mediates the expression of various growth, immune, and inflammatory system genes. 10 NFκB is thought to play a dual role, acting as a friend by regulating the normal functioning of the immune system or acting as a foe by its anomalous activation that mediates tumor progression, inflammation and sometime drug resistance in chemotherapy and radiotherapy. 11 Tumor necrosis factor (TNF) is considered a potent activator of NFκB. Blockage of NFκB can cause cell cycle arrest, halt proliferation and initiate apoptosis. It is proposed that agents with substantial potential to halt the activation of NFκB are beneficial against carcinogenesis. 12
Nitric oxide is reactive nitrogen radical that acts as an intercellular messenger and is present in diverse mammalian cells. It is manufactured from L-arginine by nitric oxide synthase (NOS) using NADPH and O2. NO is known to be involved in processes like DNA impairments and mutations, DNA repairing enzymes suppression along with production of carcinogenic moieties of N-nitroso compounds. 13 Nitric oxide is a potent signaling mediator of many physiological processes for example vasodilation, host defense, neurotransmission, iron metabolism, and platelet aggregation whereas elevated levels of NO may cause pathological manifestations for example cancer; hence inhibitory moieties that halt the NO generation may act as efficacious anticancer agents. 14
Aromatase is a complex enzyme that instigates production of estrogen (a potent estradiol) from androgen. In ovarian granulosa cells, testicular Leydig cells, placenta, adipose tissue, and skin, aromatase is expressed at normal levels where as anomalous elevated expression of aromatase is linked with breast cancer, endometrial cancer, endometriosis, and uterine fibroids. 15 Aromatase inhibitors may serve as strong therapeutic candidates for the control and management of these health malaises.
Quinone reductase (QR1) is a phase II metabolizing enzyme which converts quinones to hydroquinones ultimately diminishing oxidative cycling. 16 Quinone reductase can protect cells from quinone toxicity, stabilize the p53 anticancer protein and help to maintain the antioxidant potential of other endogenous antioxidants. 17 Therapeutic agents responsible for QR1 inductions are regarded as potential anticancer moieties.
Cancers are heterogeneous and diverse but all share the property of proliferation. Cancerous cells can be differentiated from their normal counterparts by increased proliferation and resistance to apoptosis or programed cell death. To develop tumors, deviations in the regulation of appropriate key pathways that control cell proliferation and cell survival are mandatory. 18 Antiproliferative agents are substantial and significant for the management of different type of cancers. 19 Using diversified cancer cell lines, these curative agents have been tested for their efficacy against cancerous cell proliferations. 20
Medicinal plants are potent candidates to overcome the carcinogenic progression.5,7 Dicliptera roxburghiana Nees, locally called “Marchak bootay”and “Churu” is a perennial herbaceous plant of the family Acanthaceae. A large collection of ethanobotanical data confirmed its different uses for various purposes in Pakistan. It is confirmed in an in vivo study that the plant is nontoxic. 21 Powder of the leaves and flower parts is used as general tonic in some areas of Pakistan 22 Sprinkling the powder of the roots 3 times a day is a potent remedy for wound healing in Pakistan. 23 Chemical characterization of the plant revealed that plant is rich in saturated fatty acids along with important flavonoids for example apigenin, kaempferol, luteolin and apigenin-7-O-glucoside. 24 In vitro antioxidant aptitude of D. roxburghiana was also demonstrated. 25
Methods
Plant Material
Dicliptera roxburghiana was collected at maturity from the campus of Quaid-i-Azam University. Identification of D. roxburghiana was certified by taxonomist Prof. Dr. Rizwana Aleem Qureshi, Department of Plant Sciences, Quaid-i-Azam University, Islamabad. Voucher specimen of D. roxburghiana (accession#125521) was placed in the Herbarium of Pakistan situated at Quaid-i-Azam University Islamabad, Pakistan. Plant material was shade dried (28 ± 2°C) and leaves were coarsely pulverized into dry powder using a Wiley mill (60-mesh size).
Preparation of Methanol Extracts
Powder of the plant (2.0 kg) was soaked in crude methanol (4.0 L) and was regularly shaken for 5 days at room temperature (28 ± 2°C). Filtration was carried out through Whatmann filter paper No. 45 and the re-extraction of the remainder was repeated twice. Plant filtrate was dried under rotary vacuum evaporator (Panchun Scientific Co, Kaohsiung, Taiwan) at 40°C to yield concentrated dry extract. The methanol extract of plant D. roxburghiana (DRME) yielded dark green viscous material (200 g).
Preparation of Fractions
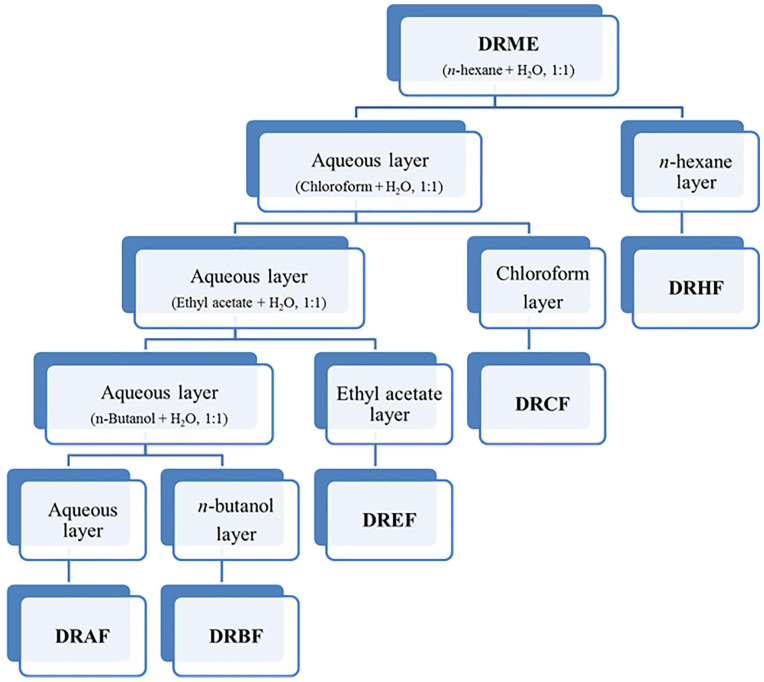
DRME aqueous solution was then successively partitioned with n-hexane, chloroform, ethyl acetate and n-butanol in a separating funnel in 1:1 ratio. Figure 1 describes the fractionation scheme of crude methanol extract of D. roxburghiana (DRME) which yielded n-hexane fraction (DRHF), chloroform fraction (DRCF), ethyl acetate fraction (DREF), n-butanol fraction (DRBF) along with residual aqueous fraction (DRAF). All fractions were collected, dried and stored at 4°C.
Figure 1.
Fractionation scheme of D. roxburghiana from crude methanol extract. D. roxburghiana methanol extract (DRME); D. roxburghiana n-hexane fraction (DRHF); D. roxburghiana chloroform fraction (DRCF); D. roxburghiana ethyl acetate fraction (DREF); D. roxburghiana n-butanol fraction (DRBF); D. roxburghiana aqueous fraction (DRAF).
GCMS Profile of DRME
Gas chromatography mass spectrometry was performed to determine the components present in the methanol extract of D. roxburghiana. For this purpose, analysis was carried out using thermo GC-trace ultra ver: 5.0, thermo MS DSQ 11 equipment fitted with a ZB 5-MS capillary standard non polar column with dimensions 30 Mts and 0.25 µm film. Helium was used as carrier gas with flow rate 1.0 ml/minutes. Oven temperature was adjusted in such a way that initially it was 70°C which was then raised to 260°C at 6°C/minutes. Injection volume was 1 µl.
Inhibition of TNF-α Induced NFκB
To demonstrate this activity, 293/NFκB-Luc HEK cells were maintained in Dulbecco’s Modified Eagle medium accompanied with antibiotic and fetal bovine serum (10%). On 96-well plate, cells were seeded in such a way that there were 2 × 104 cells in 200 µl. Following incubation at 37°C for 48 hours with 5% CO2, medium was changed subsequently and 20 µg/ml samples were loaded. Tenng/ml of TNF-α was added and incubated for 6 hours. Following washing of cells with PBS and addition of 50 µl of 1X reporter lysis buffer, cells were exposed to one freeze/thaw cycle (−80°C/37°C). Inhibition was recorded by luminometer using Luciferase Assay system. Percentage inhibitions were calculated and samples which were displaying % inhibition more than 70 at 20 µg/ml were selected to check their response in dose dependent manner to determine IC50. 26 To compare with positive control, N-tosyl-L-phenylalanyl chloromethyl ketone (TPCK) and (E)-3-(4-Methylphenylsulfonyl)-2-propenenenitrile (BAY-11) were used. Cytotoxic effects were determined by sulforhodamine B (SRB) procedure which is described later.
Assessment of Aromatase Inhibition
A protocol designed by Maiti et al 27 was followed to demonstrate aromatase inhibition. Samples were incubated at 37°C for 30 minutes with NADPH regenerating system. Following addition of substrate and enzyme mixture, plate was placed for incubation at 37°C for 10 minutes before quenching with NaOH. Subsequent to reaction termination and 5 minutes shaking, plate was incubated at 37°C for 2 hours to augment the ratio of signal to background. Fluorescence was analyzed at 485 nm and 530 nm for excitation and emission respectively. Naringenin was used as positive control (IC50 = 0.23 µM). IC50 and dose response curves of samples were measured in 2 independent experiments using 5 concentrations of sample tested.
LPS-instigated NO Production Inhibition (Nitrite Assessment)
For demonstration of nitrite assay, a protocol of Park et al 28 was adopted. Raw 264.7 cells (10 × 104 cells per well) were maintained in DMEM accompanied with FBS (10%). These cells were seeded onto 96-well plates and were kept for incubation for 24 hours. After that, media was replaced with 1% FBS (190 µl) comprised of phenol red free DMEM. About 10 µl sample in DMSO (10%) was used to treat the cells for 15 minutes, subsequently followed by LPS treatment (1 µg/ml) for 20 hours. Nitrites formed were quantified to monitor the effects of tested samples on biosynthesis of NO. The amount of nitrite, the major oxidized metabolite of NO, was measured to evaluate the effects of samples on NO biosynthesis. About 100 µl of incubation media was shifted to 96-well plate to gauge the reaction with Griess reagent [90 µl of sulfanilamide (1%) in phosphoric acid (5%) and 90 µl of N-(1-naphthyl) ethylenediamine] and absorbance was recorded at 540 nm. Test sample with inhibition more than 70% at 20 µg/ml were selected to screen IC50 at 3 fold serial dilutions. Cytotoxicity capabilities of samples were gauged by SRB assay.
Quinone Reductase Monitoring
For Quinone reductase monitoring, murine hepatoma (Hepa 1c1c7) cells were used. Cells (200 µl, 0.5 × 104 cells/ml) were plated using minimum essential medium (MEM-α) lacking ribonucleoside or deoxyribonuceosides and supplemented with FBS (10%) and antibiotic/antimycotics. Incubation was done using CO2 incubator for 24 hours, and medium was replaced with fresh medium (190 µl). Test samples (10 µl) were added with 20 µg/ml as final concentration and placed for incubation for 48 hours. Digitonin was taken to permeabilize cell membranes and enzyme activity was monitored by analyzing reduction of MMT [3-4,5-dimethylthiazo-2-yl)-2,5-diphenyltetrazolium bromide] to a blue formazan. Following absorbance at 595 nm, production was determined. 29 Using crystal violet staining, total protein determination was carried out at the same time. 30 To make comparison with positive control, 4’-bromoflavone (CD = 0.01 µM) was used. Samples with induction ratio more than 2 at 20 µg/ml were considered active and were monitored for their CD values determined in 5-fold serial dilutions.
Sulforhodamine B Assay (SRB)
According to You et al 31 SRB is a colorimetric assay used to demonstrate the cytotoxic capabilities of multiple cancer cell lines for example hormone responsive breast cancer cell lines MCF-7 (ATCC NO. is HTB-22), estrogen receptor negative breast cancer cell lines MDA-MB-231 (ATCC NO. is HTB-26), murine hepatoma cells Hepa 1c1c7 (ATCC NO. is CRL-2026), human neuroblastoma cells SK-N-SH (ATCC NO. is HTB 11) and neuroblastoma cells MYCN-2. Test sample (10 µl) in DMSO (10%) and PBS were shifted to 96-well plates along with 190 µl cells (5 × 104 cells/ml). Plates were incubated at 37°C for 72 hours in a CO2 incubator and the reaction was terminated with the addition of TCA (50 µl, 20%). Following washing, cells were dried and stained with 0.4% SRB in acetic acid (1%) at room temperature for 30 minutes. Following washing with 1% acetic acid for 4 times, plates were dried overnight. Bound dye was solubilized with Tris base (200 µl, 10 mM, pH 10) on a gyratory shaker for 10 minutes. At 515 nm, optical density was recorded at micro-plate reader and percent survival was calculated. A 0-day control was performed in each case following addition of equal quantity of cells in 16 wells, with subsequent incubation for 30 minutes at 37°C and was processed as mentioned earlier. Cell survival percentage was calculated.
Results
It is universally accepted that plants are a significant source of therapeutic and biologically active constituents. A number of phytochemicals are protectants against various serious health maladies. Keeping in view this undeniable worth of plants, the methanolic sample of D. roxburghiana was subjected to gas chromatography/mass spectrometry to inspect and unravel the phytoconstituents. The GCMS chromatogram of D. roxburghiana is shown in Figure 2. Compound details along with their molecular formulas, molecular weight and retention times are summarized in Table 1.
Figure 2.
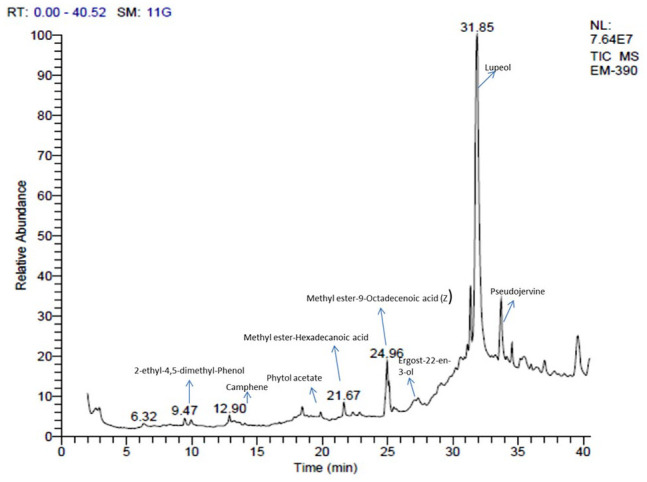
GCMS chromatogram of DRME.
Table 1.
GCMS Profile of DRME.
| Serial no. | Retention time | Molecular formula | Molecular weight | Name |
|---|---|---|---|---|
| 1 | 6.32 | C30H56 | 416 | 1-[1-(3-cyclohexylpropyl)undecyl] decahydro- Naphthalene |
| 2 | 7.53 | C12H18O | 178 | (1a,5a,6a,7a)-6,7-(Z,E)-Dipropenyl-3-oxabicyclo[3.2.0]heptane |
| 3 | 9.47 | C10H14O | 150 | 2-ethyl-4,5-dimethyl-Phenol |
| 4 | 9.55 | C10H14O | 150 | 2,3,4,6-tetramethyl-Phenol |
| 5 | 9.59 | C10H17Cl | 172 | 2-chloro-2,3,3-trimethyl-bicyclo [2.2.1] heptane |
| 6 | 9.61 | C14H22O | 206 | 2-tert-Butyl-4-isopropyl-5-methylphenol |
| 7 | 10.56 | C10H18O | 154 | Cis-sabinenehydrate |
| 8 | 10.93 | C10H16 | 136 | Camphene |
| 9 | 13.26 | C6H12O2 | 116 | Tetrahydro-2-methoxy-3-[13C]-pyran |
| 10 | 13.95 | C6H12S | 116 | 3-[(1-methylethyl)thio]-1-Propene |
| 11 | 14.77 | C13H17N | 187 | 1-(2-Methyl-propenyl)-indan-2-ylamine |
| 12 | 15.48 | C11H16O3 | 196 | Loliolide |
| 13 | 17.15 | C14H22O3 | 238 | [2-(2′,6′,6′-Trimethyl-1′,2′-epoxycy clohexyl-1-propen-1-yl] acetate |
| 14 | 18.27 | C13H24O | 196 | 2,2,6,7-tetramethyl-7-hydroxy- Bicyclo[4.3.0]nonane |
| 15 | 19.88 | C20H40O | 296 | 3,7,11,15-Tetramethyl-2-hexadecen-1-ol |
| 16 | 20.47 | C20H38 | 278 | Neophytadiene |
| 17 | 20.78 | C22H42O2 | 338 | Phytol acetate |
| 18 | 21.67 | C17H34O2 | 270 | Methyl ester-Hexadecanoic acid |
| 19 | 21.93 | C19H38O4 | 330 | 2,3-dihydroxypropyl ester-Hexadecanoic acid |
| 20 | 22.34 | C17H34O2 | 270 | 14-methyl-, methyl ester-Pentadecanoic acid |
| 21 | 22.61 | C15H30O2 | 242 | Pentadecanoic acid |
| 22 | 22.74 | C16H32O2 | 256 | Hexadecanoic acid |
| 23 | 24.17 | C8H9Br | 184 | (1-bromoethyl)-Benzene |
| 24 | 24.68 | CH8Si2 | 76 | Methylenebis-Silane |
| 25 | 24.83 | C19H36Cl2O2 | 366 | 9,10-dichloro-methyl ester- Octadecanoic acid |
| 26 | 24.96 | C19H36O2 | 296 | Methyl ester-9-Octadecenoic acid (Z) |
| 27 | 25.48 | C17H34O2 | 270 | 14-methyl-methyl ester-Pentadeca noic acid |
| 28 | 25.99 | C19H36D2O2 | 298 | Methyl 13,14-dideutero-octadecan oate |
| 29 | 26.01 | C5H10O2 | 102 | (2S)-Methylbutanoic Acid |
| 30 | 26.52 | C19H36O2 | 296 | Methyl ester-cis-13-Octadecenoic acid |
| 31 | 26.95 | C10H14D2O | 152 | 5,6-Dideuterio-8-trans-methylhydrin dan-2-one. |
| 32 | 27.51 | C16H32O3 | 272 | (E)-1-(Methoxymethoxy)-1-tetradecen-3-ol |
| 33 | 27.62 | C14H26 | 194 | 1-(cyclohexylmethyl)-3-methyl-, trans- Cyclohexane |
| 34 | 28.11 | C20H18 | 258 | 3b,4,5,6,7,7a-Hexahydrobenzo [b] fluoranthene |
| 35 | 28.05 | C19H14BrN3 OS | 411 | 2-amino-4-(4-Bromophenyl)-7,9-dim ethyl-4hhpyrano[2’,3’:4,5]thieno [2,3 -b]pyridine-3-carbonitrile |
| 36 | 28.62 | C28H48O | 400, | Ergost-22-en-3-ol |
| 37 | 28.94 | C21H42O2 | 326 | Methyl ester-Eicosanoic acid |
| 38 | 29.42 | C17H34O2 | 270 | 14-methyl-, methyl ester-Pentadecan oic acid |
| 39 | 29.73 | C25H50O2 | 382 | Methyl ester-Tetracosanoic acid |
| 40 | 30.28 | C20H28O3 | 316 | 16-Hydroxymethyleneandrost-5-en-3-ol-17-one |
| 41 | 30.35 | C22H19F2N 3OS |
411 | 2-amino-1-(2,6-diflouro-phenyl)-4-(-5 ethyl-thiophene-2-yl)-5-oxo-1,4,5,6,7, 8-hexahydro-quinoline-3-carbonitrile |
| 42 | 30.41 | C16H17F3N 2O4 |
358 | 2,2,2-trifluoroethyl ester -1,2,3,4-tetra hydro-4-(4-ethoxyphenyl)-6-methyl2- oxo- Pyrimidine-5-carboxylic acid |
| 43 | 30.57 | C8H20N2S2 | 208 | 2,2′-dithiobis(N,N-dimethyl) Ethanamine, |
| 44 | 31.10 | C19H38O4 | 330 | 2-hydroxy-1-(hydroxyl methyl)ethyl ester Hexadecanoic acid |
| 45 | 31.29 | C19H38O4 | 330 | Glycerol 1-palmitate |
| 46 | 31.55 | C24H38O4 | 390 | Di(2-propylpentyl) ester-Phthalic acid |
| 47 | 31.83 | C30H50O | 426 | Lupeol |
| 48 | 31.96 | C30H50O | 426 | (3a)-Lup-20(29)-en-3-ol |
| 49 | 32.11 | C32H52O2 | 468 | (3a)-Lup-20(29)-en-3-ol-acetate |
| 50 | 33.29 | C30H40O8 | 528 | 2,4-Octadienoic acid,9a-(acetyloxy)-1a,1b,4,4a,5,7a,7b,8,9,9a-decahydro-4a,7b-dihydroxy-3- (hydroxymethyl)-1,1,6,8-tetramethyl-5-oxo 1Hcyclopr opa [3,4]benz [1,2-e]azulen-9-ylester, [1aR [1aà,1bá, 4aá,7aà,7bà, 8à,9á (2Z,4E),9aà] |
| 51 | 33.35 | C16H18N4 | 298 | 2-benzyl-3-oxo-13-hydroxy-1,4,11,12 -tetraaza tricyclo[8.3.0.0 (4,8)] trideca -10(11),12-diene |
| 52 | 33.59 | C7H8O3 | 140 | (+-)-endo-7-Oxabicyclo[2.2.1]hept-5-ene-2-carboxylic acid |
| 53 | 33.68 | C28H32 | 368 | 1,1'-[4-(3-phenylpropyl)-3-heptene-1,7-diyl]bis-Benzene |
| 54 | 33.70 | C21H40O4 | 356 | 2,3-dihydroxypropyl ester-9-Octadecenoic acid (Z) |
| 55 | 33.89 | C21H40O3 | 340 | 3-hydroxypropyl ester-Oleic acid |
| 56 | 34.15 | C33H49NO8 | 587 | Pseudojervine |
| 57 | 34.22 | C32H49NO8 | 575 | Angeloylzygadenine |
| 58 | 34.41 | C19H35FO2 | 314 | Methyl-18-fluoro-octadec-9-enoate |
| 59 | 34.54 | C24H38O4 | 390 | bis(2-ethyl hexyl)ester-1,4-Benzenedi carboxylic acid |
| 60 | 34.88 | C24H38O4 | 390 | 2-ethylhexyl octyl ester Terephthalic acid |
| 61 | 35.46 | C29H50O | 414 | (3a,24S)-Stigmast-5-en-3-ol |
| 62 | 35.71 | C23H32N2O4 | 400 | 1-acetyl-5-ethyl-2-[3-(2hydroxyethyl) -1H-indol-2-yl]-a-methyl-methylester 4-Piperidineacetic acid |
| 63 | 35.92 | C28H38O9 | 518 | 3-methoxy-7,11,18-triacetoxy3,9-Epo xypregn-16-en-20-one |
| 64 | 36.01 | C15H26O | 222 | trans-Farnesol |
| 65 | 36.27 | C30H52O | 428 | 2,2,4-Trimethyl-3-(3,8,12,16-tetrame thyl-heptadeca-3,7,1 1,15-tetraenyl) -cyclohexanol |
| 66 | 36.41 | C22H34O4 | 362 | 4,4-dimethyl- (13a) 3a-Methoxy-3a,19-epoxyandr ost -5-en-7,17a-diol |
| 67 | 37.03 | C24H14Cl2N2 | 400 | 3,8-Dichloro-6-phenylindolo[2,3-a]carbazole |
| 68 | 37.22 | C26H18Cl2 | 400 | (E) and (Z)-1,2-Bis(p-chlorophenyl)-1,2-diphenylethene |
| 69 | 38.66 | C30H19NO2 | 425 | 2,3-Dibenzoyl-1H-phenanthro[9,10-b]pyrrole |
| 70 | 39.57 | C29H26O2S | 438 | Methylsulfoxyphenyldimethylcyclophanene |
| 71 | 39.62 | C23H20 | 296 | 1-(Methylphenyl)-2,7-dimethyl anthracene |
| 72 | 39.95 | C20H14N2O | 298 | Ethanone, 2-(5H-indeno[1,2 b]pyrid ineyli de ne)-1-phenyl-oxime |
Regarding antiproliferative assays tested on different cancer cell lines, different extracts displayed a very appreciable activity with very good survival % and IC50 values as described in Table 2. In these assays samples which were displaying a survival % ≤ 60 were considered as active against respective cell lines. DRBF exhibited strong antiproliferative activity against IcIc-7 cells with % survival of 47.6 ± 18.91 and IC50 values of 13.6 ± 1.91 µg/ml (Table 2).
Table 2.
Antiproliferative Activity of Various Extracts of D. roxburghiana Against Different Cancer Lines.
| Extract | IcIc-7 | MYCN-2 | SK-N-SH | MCF-7 | MDA-MB-231 | |||
|---|---|---|---|---|---|---|---|---|
| % Survival | IC50 (µg/ml) | % Survival | IC50 (µg/ml) | % Survival | IC50 (µg/ml) | % Survival | % Survival | |
| DRME | 126.2 ± 9.15 | — | 109.33 ± 18.90 | — | 82.7 ± 2.48 | — | 117.7 ± 8.22 | 91.7 ± 12.66 |
| DRHF | 127.6 ± 0.55 | — | 122.6 ± 3.77 | — | 96.3 ± 0.62 | — | 131.9 ± 27.21 | 89.16 ± 8.85 |
| DRCF | 121.1 ± 0.76 | — | 75.9 ± 3.63 | — | 51.4 ± 6.84 | 11.2 ± 0.84 | 101.7 ± 2.69 | 82.0 ± 5.75 |
| DREF | 121.6 ± 9.21 | — | 67.02 ± 1.56 | — | 57.6 ± 1.86 | 14.6 ± 1.71 | 114.9 ± 4.62 | 77.3 ± 2.83 |
| DRBF | 47.6 ± 18.91 | 13.6 ± 1.91 | 28.9 ± 14.41 | 12.6 ± 1.24 | 51.9 ± 2.48 | 16.3 ± 1.57 | 91.5 ± 14.16 | 83.2 ± 10.36 |
| DRAF | 130.7 ± 1.44 | — | 77.0 ± 4.86 | — | 112.2 ± 3.11 | — | 154.4 ± 36.6 | 80.0 ± 7.70 |
Values represent Mean ± SD (n = 3).
While evaluating antiproliferative potential against MYCN-2 cancer cell lines, DRBF was very active with % survival only 28.9 ± 14.41 and IC50 of 12.6 ± 1.24 µg/ml as depicted in Table 2. In antiproliferative activity against SK-N-SH cell lines, DRCF, DREF, and DRBF were efficiently active with survival percentage of 51.4 ± 6.84, 57.6 ± 1.86, and 51.9 ± 2.48 respectively. The IC50 values for DRCF, DREF and DRBF were measured as 11.2 ± 0.84, 14.6 ± 1.71, and 16.3 ± 1.57 respectively as shown in Table 2. In evaluating activity against MCF-7 and MDA-MB-231 cancer cell lines, none of the extracts exhibited good anticancer activity and their values exceeded the 60% (Table 2).
Anticancer activity of D. roxburghiana was evaluated by different regulatory factors involved in cell growth and division. In the Quinone reductase assay (QR), samples with IR value more than 2 were considered as efficient. DRCF was potently active and exhibited an induction ratio of 2.4 ± 0.09 with CD value 17.7 µg/ml as described in Table 3. The rest of the fractions were not so active against Quinone reductase. In TNF-α mediated NFκB inhibition analysis, samples with percentage inhibition more than 50 were taken as highly active. DREF and DRBF were strongly active with inhibition of 69.5% ± 0.64% and 81.2% ± 4.85% respectively. IC50 values for DREF and DRBF were calculated as 16.6 ± 1.3 and 8.4 ± 0.7 µg/ml respectively with 100% survival (Table 3).
Table 3.
QR1, NFκB, Nitrite, and Aromatase Activities of Different Fractions of D. roxburghiana.
| Extract | QR1 | NFκB | Nitrite | Aromatase | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| IR | CD (µg/ml) | % Inhibition | % Survival | IC50 (µg/ml) | % Inhibition | IC50 (µg/ml) | % Survival | % Inhibition | IC50 (µg/ml) | |
| DRME | 1.2 ± 0.16 | 5.1 ± 1.63 | 164.7 ± 7.79 | 13.7 ± 4.10 | — | 111.4 ± 1.27 | 41.6 ± 1.37 | — | ||
| DRHF | 1.4 ± 0.05 | –9.7 ± 6.01 | 124.9 ± 13.39 | 54.7 ± 2.37 | 17.8 ± 1.25 | 125.0 ± 2.23 | 43.4 ± 0.70 | — | ||
| DRCF | 2.4 ± 0.09 | 17.7 ± 0.8 | 13.5 ± 1.8 | 133.4 ± 17.24 | 32.1 ± 3.14 | — | 114.2 ± 1.23 | 24.0 ± 2.56 | — | |
| DREF | 0.9 ± 0.30 | 69.5 ± 0.64 | 138.1 ± 6.06 | 16.6 ± 1.3 | 4.1 ± 3.36 | — | 111.5 ± 2.10 | 37.3 ± 2.83 | — | |
| DRBF | 1.5 ± 0.39 | 81.2 ± 4.85 | 125.0 ± 4.46 | 8.4 ± 0.7 | 11.5 ± 5.55 | — | 111.1 ± 1.27 | 19.2 ± 2.40 | — | |
| DRAF | 0.7 ± 0.11 | 27.2 ± 2.69 | 166.4 ± 5.06 | –3.6 ± 0.14 | — | 110.1 ± 4.45 | 26.7 ± 3.33 | — | ||
Values represent Mean ± SD (n = 3).
In the LPS-initiated NO inhibition assay (nitrite assay) samples exhibiting % inhibition ≥ 50 were considered as highly potent. In the nitrite assay, DRHF exhibited significant activity against NO with 54.7% ± 2.37% inhibition and IC50 of 17.8 ± 1.25 µg/ml as depicted in Table 3. All the samples displayed good survival without any cytotoxicity. Regarding aromatase activity, sample displaying % inhibition ≥ 50 were considered as active against aromatase. It was observed that none of the sample showed inhibition against aromatase and so all were reflected as inactive (Table 3).
Discussion
Cancer is the leading cause of death in most countries of the world and is promoted by a number of factors including diet, life style, environmental pollutants and carcinogens. 32 A diversified range of cancer chemopreventive assays are attaining prominence for many researchers to unravel the active principles present in botanicals. Preliminary phytochemical screening of plant extracts is a good measure to get a clue about the secondary metabolites present in plants. A number of important phytochemicals were confirmed from the GCMS profile of D. roxburghiana. These phytochemicals could play important roles to control the onset and spread of diseases. This study also confirmed the phytochemical profile of D. roxburghiana and confirmed that the plant was rich source of flavonoids and phenolics which are strong antioxidant compounds. 25 Our findings are in accord with the results of another study in which it was concluded that the plant was richly supplied with the active biodynamic phytochemicals hence strengthening the medicinal worth of the plant. 33
A large body of research has shown that plants synthesize a wide range of secondary metabolites during the course of their normal metabolic pathways. These secondary metabolites are distributed in specific plant genera or species and are supposed to defend the plants from many hazards for example microbial infections etc. 34 The term secondary metabolites describes a wide array of compounds such as flavonoids, terpenoids, coumarins, alkaloids, tannins, phlobatannins, anthraquinones, saponins, and cardiac glycosides. These metabolites are observed to be endowed with a number of biological properties such as antioxidant, antimicrobial and anticancer. 35 For example, a research study showed the chemopreventive effect of ginger on hepatic cancer by inhibiting cell growth through induced apoptosis. 5
Regarding antiproliferative screenings, cytotoxic potential and antitumor characteristics of all fractions of D. roxburghiana were investigated against different cancer cell lines for example IcIc-7, MYCN-2, SK-N-SH, MCF-7 (estrogen receptor positive breast cancer cell line) and MDA-MB-231 (estrogen receptor negative breast cancer cell line) cancer cell lines. It was resolved that DRBF had highly significant antitumor activity with survival rates of < 60 in case of IcIc-7, MYCN-2, and SK-N-SH cell lines whereas DREF and DRCF showed antiproliferative activity against the SK-N-SH cell line. No positive results were displayed against MCF-7 and MDA-MB-231 cancer cell lines. These results indicate that the plant contains some active antiproliferative constituents that can play critical roles in selected cancer cell lines. Other researchers also reported the similar observations for example Haq et al 36 investigated and validated the positive consequences of various medicinal plants with regard to cancer chemopreventive (aromatase, QR1, nitric and TNF-a mediated NFκB) and antiproliferative proficiencies (MCF-7, MDA-MB-231 etc.). In another study Vernonia amygdalina extract exhibited good anticancer potential against various breast cancer cell line for example MCF-7, MDA-MB-231 etc. 37
Nitric oxide (NO) production in lipopolyscharides (LPS) activated murine macrophage cells inhibition assay was carried out to determine nitric oxide synthase inhibition by the plant samples. Nitric oxide is a very reactive nitrogen radical that leads to the onset of carcinogenesis and mutations in DNA. Nitric oxide over-expression results in a wide range of inflammatory manifestations and so by itself serves as an indicator for monitoring the extent of inflammation in affected cells. Nitric oxide synthase inhibitors (iNOS) are assumed to be competent moieties to regulate the NO induced stress and health hazards. Phytochemicals may inhibit NOS and in turn may halt the carcinogenic manifestations. All fractions of D. roxburghiana were tested in the iNOS assay and it was demonstrated that DRHF was behaving as a potent inhibitor of NOS, suggesting a positive role of D. roxburghiana in cancer prevention. It is worth stating here that all the samples were nontoxic to the cells and showed appreciable survival percentages. DRHF inhibited the NO production and at the same time was shown as nontoxic to cells, indicating that DRHF may control NO production without killing the cell.
QR1 is another noteworthy parameter to monitor the positive consequences of plant extracts in cancer chemoprevention assays. QR1 is a defensive enzyme used for the detoxification of deadly reactive species. It can easily transform quinones to hydroquinones with broad spectrum specificity for reduction of hydrophobic quinones. QR1 is widely distributed in mammalian cells and easily demonstrated by monitoring the induction responses. Among all fractions of D. roxburghiana, DRCF was most effective fraction with appreciable CD value. Secondary metabolites of plants for example flavonoids and triterpenoids are well known mediators of QR1 with appreciable roles in cancer chemoprevention.38,39 This activity in D. roxburghiana was also attributed to its flavonoids for example apigenin, kaempferol, luteolin and apigenin-7-O-glcoside and other phenolics as well. 24
TNF-α activated nuclear factor kappa-B (NFκB) inhibition was also assessed along with other parameters. Being a transcription factor, NFκB readily mediates the expression patterns of those genes which are of primary importance in cell proliferation, differentiation and inflammation responses. In this analysis all fractions displayed their ability to inhibit NFκB activity. It was also demonstrated that DREF and DRBF can very strongly inhibit NFκB at very low doses. This activity is also assigned to the high contents of active secondary metabolites present in D. roxburghiana. Similar activity was shown in research on dandelion extract, which was highly effective against TNF-α to control the inflammatory response in rats. 1 In another study, some Thai plant extracts were screened for their NFκB inhibition activity and it was concluded that those herbal extracts were influencing the expression patterns of NFκB for the prevention of psoriasis. 40
Aromatase inhibition was also monitored in chemoprevention assays. Aromatase is very significant for the production of estrogens therefore play a unique roles for cancer inductions in particular conditions for example post menopause. Constituents that inhibit aromatase are considered as valuable moieties for cancer chemoprevention. All fractions of D. roxburghiana were analyzed for the aromatase inhibition assessment but none of them displayed positive effects.
Conclusion
This experimental study concluded that D. roxburghiana was rich in anticancer compounds and active metabolites and may serve as a valuable therapeutic source to develop novel anticancer leads.
Footnotes
Author Contributions: BA and TPK made significant contribution to acquisition of data, analysis, drafting of the manuscript. BA, MRK, JMP, and NAS have made substantial contribution to conception and design, interpretation of data, drafting, and revising the manuscript for intellectual content. NA, AG, and AK participated in the design and collection of data and analysis. All authors read and approved the final manuscript.
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge Higher Education Commission (HEC) of Pakistan for providing financial support for the present work under grant No117-5638-BM7-028.
ORCID iD: Bushra Ahmad  https://orcid.org/0000-0002-7872-3255
https://orcid.org/0000-0002-7872-3255
References
- 1. Hamza AA, Mohamed MG, Lashin FM, Amin A. Dandelion prevents liver fibrosis, inflammatory response, and oxidative stress in rats. JOBAZ. 2020;81:43. [Google Scholar]
- 2. Amin A, Hamza AA, Bajbouj K, Ashraf SS, Daoud S. Saffron: a potential candidate for a novel anticancer drug against hepatocellular carcinoma. Hepatology. 2011;54:857-867. [DOI] [PubMed] [Google Scholar]
- 3. Pezzuto JM, Kosmeder JW, Park EJ, et al. Characterization of chemopreventive agents in natural products. In: Kelloff GJ, Hawk ET, Sigman CC, eds. Cancer Chemoprevention, 2005. 2: Strategies for Cancer Chemoprevention. Humana Press Inc; 2005;3-37. [Google Scholar]
- 4. Amin A, Mahmoud-Ghoneim D. Texture analysis of liver fibrosis microscopic images: a study on the effect of biomarkers. Acta Biochim Biophys Sin. 2011;43:193-203. [DOI] [PubMed] [Google Scholar]
- 5. Hamza AA, Heeba GH, Hamza S, Abdalla A, Amin A. Standardized extract of ginger ameliorates liver cancer by reducing proliferation and inducing apoptosis through inhibition oxidative stress/ inflammation pathway. Biomed Pharmacother. 2021;134:111102. [DOI] [PubMed] [Google Scholar]
- 6. El-Dakhly SM, Salama AAA, Hassanin SOM, Yassen NN, Hamza AA, Amin A. Aescin and diosmin each alone or in low dose- combination ameliorate liver damage induced by carbon tetrachloride in rats.. BMC Res Notes. 2020;13:259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Ashktorab H, Soleimani A, Singh G, et al. Saffron: the golden spice with therapeutic properties on digestive diseases. Nutrients. 2019;11:943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Wall ME, Wani MC. Camptothecin and taxol: discovery to clinic: thirteenth Bruce F. Cain Memorial Award lecture. Cancer Res. 1995;55:753-760. [PubMed] [Google Scholar]
- 9. Gupta MM, Singh DV, Tripathi AK, et al. Simultaneous determination of vincristine, vinblastine, catharanthine, and vindoline in leaves of catharanthus roseus by high-performance liquid chromatography. J Chromatogr Sci. 2005;43:450-453. [DOI] [PubMed] [Google Scholar]
- 10. Gilmore TD. Introduction to NF-kappaB: players, pathways, perspectives.. Oncogene. 2006;25:6680-6684. [DOI] [PubMed] [Google Scholar]
- 11. Balkwill F, Mantovani A. Inflammation and cancer: back to Virchow? Lancet. 2001;357:539-545. [DOI] [PubMed] [Google Scholar]
- 12. Karin M. Nuclear factor-kappaB in cancer development and progression.. Nature. 2006;441:431-436. [DOI] [PubMed] [Google Scholar]
- 13. Fukumura D, Kashiwagi S, Jain RK. The role of nitric oxide in tumour progression. Nat Rev Cancer. 2006;6:521-534. [DOI] [PubMed] [Google Scholar]
- 14. Hofseth LJ, Hussain SP, Wogan GN, Harris CC. Nitric oxide in cancer and chemoprevention. Free Radic Biol Med. 2003;34:955-968. [DOI] [PubMed] [Google Scholar]
- 15. Bulun SE, Lin Z, Imir G, et al. Regulation of aromatase expression in estrogen-responsive breast and uterine disease: from bench to treatment.. Pharmacol Rev. 2005;57:359-383. [DOI] [PubMed] [Google Scholar]
- 16. Cuendet M, Oteham CP, Moon RC, Pezzuto JM. Quinone reductase induction as a biomarker for cancer chemoprevention. J Nat Prod. 2006;69:460-463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Nioi P, Hayes JD. Contribution of NAD(P)H: quinone oxidoreductase 1 to protection against carcinogenesis, and regulation of its gene by the Nrf2 basic-region leucine zipper and the arylhydrocarbon receptor basic helix-loop-helix transcription factors. Mutat Res. 2004;555:149-171. [DOI] [PubMed] [Google Scholar]
- 18. Evan GI, Vousden KH. Proliferation, cell cycle and apoptosis in cancer. Nature. 2001;411:342-348. [DOI] [PubMed] [Google Scholar]
- 19. Mu C, Sheng Y, Wang Q, Amin A, Li X, Xie Y. Potential compound from herbal food of Rhizoma polygonati for treatment of COVID-19 analyzed by network pharmacology: viral and cancer signaling mechanisms. J Funct Foods. 2021;77:104149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Murali C, Mudgil P, Gan CY, et al. Camel whey protein hydrolysates induced G2/M cellcycle arrest in human colorectal carcinoma. Sci Rep. 2021;11:7062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Marles RJ, Farnsworth NR. Antidiabetic plants and their active constituents. Phytomedicine. 1995;2:137-189. [DOI] [PubMed] [Google Scholar]
- 22. Qureshi R, Waheed A, Arshad M, Umbreen T. Medico-ethnobotanical inventory of tahsil Chakwal. Pakistan. Pak J Bot. 2009;41:529-538. [Google Scholar]
- 23. Khan MZ, Khan MA, Hussain M. Medicinal plants used in folk recipes by the inhabitants of himalayan region Poonch Valley Azad Kashmir (Pakistan). J Basic Appl Sci. 2012;8:35-45. [Google Scholar]
- 24. Bahuguna RP, Jangwan JS, Kaiya T, Sakakibara J. Flavonoids and fatty acids of Dicliptera roxburghiana. Int J Crude Drug Res. 1987;25:177-178. [Google Scholar]
- 25. Ahmad B, Khan MR, Shah NA, Khan RA. In vitro antioxidant potential of Dicliptera roxburghiana. BMC Complement Altern Med. 2013;13:140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Hoshino J, Park EJ, Kondratyuk TP, et al. Selective synthesis and biological evaluation of sulfate-conjugated resveratrol metabolites. J Med Chem. 2010;53:5033-5043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Maiti A, Cuendet M, Croy VL, Endringer DC, Pezzuto JM, Cushman M. Synthesis and biological evaluation of (+/-)-abyssinone II and its analogues as aromatase inhibitors for chemoprevention of breast cancer. J Med Chem. 2007;50:2799-2806. [DOI] [PubMed] [Google Scholar]
- 28. Park EJ, Kondratyuk TP, Morrell A, et al. Induction of retinoid X receptor activity and consequent upregulation of p21WAF1/CIP1 by indenoisoquinolines in MCF7 cells. Cancer Prev Res. 2011;4:592-607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Song LL, Kosmeder JW, Lee SK, et al. Cancer chemopreventive activity mediated by 4’-bromoflavone, a potent inducer of phase II detoxification enzymes. Cancer Res. 1999;59:578-585. [PubMed] [Google Scholar]
- 30. Su BN, Gu JQ, Kang YH, Park EJ, Pezzuto JM, Kinghorn AD. Induction of the phase II enzyme, quinone reductase, by withanolides and norwithanolides from Solanaceous species. Mini-Rev Org Chem. 2004;1:115-123. [Google Scholar]
- 31. You M, Wickramaratne DB, Silva GL, et al. Roemerine, an aporphine alkaloid from Annona senegalensis that reverses the multidrug-resistance phenotype with cultured cells. J Nat Prod. 1995;58:598-604. [DOI] [PubMed] [Google Scholar]
- 32. Key TJ, Schatzkin A, Willett WC, Allen NE, Spencer EA, Travis RC. Diet, nutrition and the prevention of cancer. Public Health Nutr. 2004;7:187-200. [DOI] [PubMed] [Google Scholar]
- 33. Tripathi YC, Aggarwal S, Tewari D. Pharmacognostical investigations of Dicliptera roxburghiana grown in Doon valley of the foot hills of Himalaya. World J Pharm Sci. 2014;3:1090-1097. [Google Scholar]
- 34. Mazid M, Khan TA, Mohammad F. Role of secondary metabolites in defense mechanisms of plants. Source Code Biol. 2011;3:232-249. [Google Scholar]
- 35. Wadood A, Ghufran M, Jamal SB, et al. Phytochemical analysis of medicinal plants occurring in local area of Mardan. Biochem Anal Biochem. 2013;2:144. [Google Scholar]
- 36. Haq I, Mirza B, Kondratyuk TP, et al. Preliminary evaluation for cancer chemopreventive and cytotoxic potential of naturally growing ethnobotanically selected plants of Pakistan. Pharm Biol. 2013;51:316-328. [DOI] [PubMed] [Google Scholar]
- 37. Wong FC, Woo CC, Hsu A, Tan BKH. The anti-cancer activities of Vernonia amygdalina extract in human breast cancer cell lines are mediated through caspase-dependent and p53-independent pathways. PLoS One. 2013;8:e78021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Cheng L, Shen LM, Zhang M, et al. Eleven new triterpenes from Eurycorymbus cavaleriei. Helv Chim Acta. 2010;93:2263-2275. [Google Scholar]
- 39. Fahey JW, Stephenson KK. Pinostrobin from honey and Thai ginger (Boesenbergia pandurata): a potent flavonoid inducer of mammalian phase 2 chemoprotective and antioxidant enzymes. J Agric Food Chem. 2002;50:7472-7476. [DOI] [PubMed] [Google Scholar]
- 40. Saelee C, Thongrakard V, Tencomnao T. Effects of Thai medicinal herb extracts with antipsoriatic activity on the expression on NF-κB signaling biomarkers in HaCaT keratinocytes. Molecules. 2011;16:3908-3932. [DOI] [PMC free article] [PubMed] [Google Scholar]