Abstract
Background
Management of high-risk newborns should involve the use of standardized protocols and training, continuous and specialized brain monitoring with electroencephalography (EEG), amplitude integrated EEG, Near Infrared Spectroscopy, and neuroimaging. Brazil is a large country with disparities in health care assessment and some neonatal intensive care units (NICUs) are not well structured with trained personnel able to provide adequate neurocritical care. To reduce this existing gap, an advanced telemedicine model of neurocritical care called Protecting Brains and Saving Futures (PBSF) Guidelines was developed and implemented in a group of Brazilian NICUs.
Methods
A prospective, multicenter, and observational study will be conducted in all 20 Brazilian NICUs using the PBSF Guidelines as standard-of-care. All infants treated accordingly to the guidelines during Dec 2021 to Nov 2024 will be eligible. Ethical approval was obtained from participating centers. The primary objective is to describe adherence to the PBSF Guidelines and clinical outcomes, by center and over a 3-year period. Adherence will be measured by quantification of neuromonitoring, neuroimaging exams, sub-specialties consultation, and clinical case discussions and videoconference meetings. Clinical outcomes of interest are detection of seizures during hospitalization, use of anticonvulsants, inotropes, and fluid resuscitation, death before hospital discharge, length of hospital stay, and referral of patients to specialized follow-up.
Discussion
The study will provide evaluation of PBSF Guidelines adherence and its impact on clinical outcomes. Thus, data from this large prospective, multicenter, and observational study will help determine whether neonatal neurocritical care via telemedicine can be effective. Ultimately, it may offer the necessary framework for larger scale implementation and development of research projects using remote neuromonitoring.
Trial registration
NCT03786497, Registered 26 December 2018, https://www.clinicaltrials.gov/ct2/show/NCT03786497?term=protecting+brains+and+saving+futures&draw=2&rank=1.
Introduction
Neonates requiring intensive care are exposed to a variety of interventions and treatments that improves survival but can affect their neurological system. Epidemiological studies indicate that every year 1.15 million infants suffer a hypoxic-ischemic event at birth and 13 million are born prematurely [1–5]. Of these, a substantial number will develop moderate or severe neurological impairment. Furthermore, other disorders such as congenital heart disease, cerebral malformations, congenital and nosocomial infections, intraventricular hemorrhages, inborn errors of metabolism, seizures, and severe hemodynamic and ventilatory instability can compromise brain integrity and function [6–8]. Modern neonatal intensive care units (NICU) can provide strategies to prevent neurological impairment using standardized clinical protocols and training, neuromonitoring, and neuroimaging; the so called neonatal neurocritical care units or neuroNICUs.
Amplitude Integrated Electroencephalography (aEEG) is a non-invasive methodology for continuous monitoring of brain function at the bedside, specifically for determination of electrical background activity and sleep-wake cycle (SWC) patterns, and detection of seizures [9, 10]. Indeed, the use of a two-channel aEEG with raw EEG readings (aEEG/EEG) has showed improved accuracy for the detection of seizures on real-time [11, 12], an important effect as seizure burden is an independent risk factor for neurodevelopmental delay [13–16] and treatment of subclinical seizures is associated with improved neurodevelopmental outcomes [17]. Moreover, in infants with hypoxic ischemic encephalopathy (HIE), aEEG findings have good predictive value for neurological short- and long-term outcomes [18–21].
Another available methodology for brain monitoring at the bedside is the Near Infrared Spectroscopy (NIRS), a non-invasive tool that provides continuous measurements of cerebral tissue oxygenation and perfusion (rSO2). NIRS evaluates the balance between tissue oxygen delivery and consumption allowing the early identification of perfusion abnormalities and hemodynamic changes [22, 23]. Therefore, NIRS monitoring is important in several clinical scenarios including HIE, prematurity, shock, anemia, congenital heart disease and other cases that lead to hemodynamic and ventilatory instability [24–26].
According to the World Health Organization (WHO), telemedicine is defined as the delivery of health care services, where distance is a critical factor, by all health care professionals using information and communication technologies for the exchange of valid information for diagnosis, treatment and prevention of disease and injuries, research and evaluation, and for the continuing education of health care providers, all in the interest of advancing the health of individuals and their communities [25]. WHO also listed four important elements to telemedicine: provide clinical support; intention to overcome geographic barriers; use of several types of information and communication technologies; and improve health outcomes [26]. This model portrays the natural evolution of healthcare in the digital era, resulting in reduction of the distance between two or more locations, promoting access to specific methodologies in addition to reducing the cost of structure. It can also work through live interactive audiovisual links, live video transmission or online viewing of stored education material [27–29].
Brazil is the largest South America country with approximately 3 million live births per year and a significant proportion of infants at high-risk for brain injury [30]. Like any middle-income country, the available resources for neonatal care are unequal. Therefore, telemedicine can be used as a tool for education, consultation, patient care and research. This model allows low-cost access of remote and underserved areas to specific methodologies and expertise [27–29]. Considering that less than 5% of Brazilian NICUs provide well-structured neurocritical care [31], and the implementation of that requires specialized resources, adequate equipment, and qualified personnel, the use of an advanced and protocolized telemedicine model may be a better solution to decrease these disparities. For that, in order to provide neurocritical care to a broader range of Brazilian NICUs, a protocolized telemedicine system called Protecting Brains and Saving Futures (PBSF) Guidelines was developed and implemented in some NICUs. The goals of the PBSF Guidelines are to establish remote neurocritical care units by providing adequate equipment, specialized training, implementation of standardized protocols, continuous neuromonitoring and expert support, quickly and at low cost.
The aim of this prospective multicenter and observational study is to describe the centers’ adherence to the PBSF Guidelines and clinical outcomes over a 3-years period. We hypothesize that the adherence to the PBSF Guidelines will increase over time and this will be associated with improvement of clinical outcomes. Results of this study may provide the necessary background information on the use of telemedicine aiming to protect neonatal brains and save futures, and for the development of larger studies and initiatives.
Materials and methods
Study design and setting
This prospective, multicenter, and observational study will be performed in the 20 Brazilian NICU’s located at 4 different states, of which 15 are private and 5 are part of the Brazilian free health care system or phylantropic. All units have adopted the PBSF Guidelines as a standard-of-care for high-risk newborns and agreed to participate in the study. Data will be collected for a period of 3 years, from Dec 2021 to Nov 2024.
Ethics approval and consent to participate
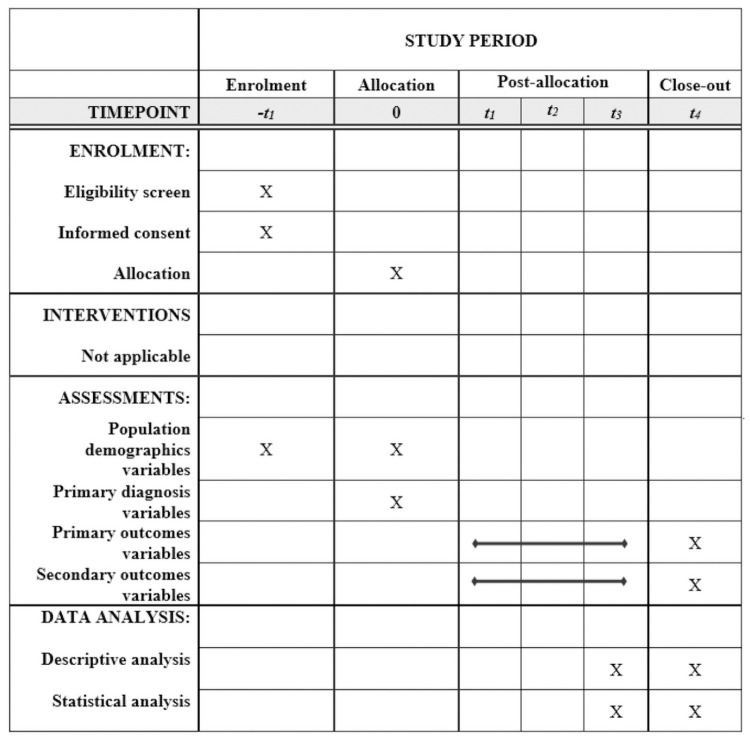
The study followed the precepts of good clinical practice and has been performed in accordance with the Declaration of Helsinki. The study proposal was approved by the Research Ethics Committee of the Irmandade da Santa Casa de Misericórdia de São Paulo under the following approval numbers and dates: 3.142.318 on February 12th, 2019 (S1 and S2 Files); 3.357.239 on May 30th, 2019 (S3 and S4 Files); and 3.506.106 on August 13th, 2019 (S5 and S6 Files). In addition, it received formal authorization from the Clinical and Administrative Board of each center. The original protocol approved by ethics is available at S7 File. The written informed consent from parents or guardians will be obtained by trained research investigators prior to inclusion in the study. The same care under the PBSF Guidelines will be provided to patients who decline study entry. All data will be treated anonymously and confidentially, and in no phase of the study any name, image, or data that allows participants identification will be disclosed. The study was registered in ClinicalTrials.gov (NCT03786497). It is reported using the Standard Protocol Items Recommendations for Interventional Trials (SPIRIT). The SPIRIT schedule is showed in Fig 1 and the completed SPIRIT checklist is available in S1 Appendix. Any proposed amendments will be discussed with the Research Ethics Committee and each institution’s Clinical and Administrative Board and communicated with all investigators, sponsor, trial participants and trial registry (Clinicaltrials.gov).
Fig 1. Schedule of enrollment, interventions, and assessments.
Eligibility criteria
In this study, the NICUs eligibility criteria are to have adopted the PBSF guidelines and agree to participate in the study (S2 Appendix). The characteristics of each NICU will be recorded and described. In those NICUs, all infants admitted from birth up to 3 months of age, and submitted to the PBSF Guidelines during the study period will be eligible. Indications for the use of the PBSF Guidelines are provided on Box 1. The PBSF Guidelines is the standard-of-care for high-risk newborns at all these hospitals. Patients with genetic syndromes or malformation incompatible with life will not be included. Details on the PBSF Guidelines are provided in (S3 Appendix). Briefly, it includes provision of equipment and resources, connection between the associated NICU and the remote monitoring center called the Central of Surveillance and Intelligence (CSI), training and teaching of all health care professionals of each NICU, and customized multiparametric recordings of biological signals from each patient. At the CSI, a team is available 24 hours a day all year around, allowing for case discussions and simplified reports with brain monitoring information at the patient monitor’s display every 6 hours.
Box 1. Indications for use of the PBSF Guidelines in the participating centers.
Indications
Extreme prematurity
Peri-intraventricular Hemorrhage
Hypoxic-ischemic encephalopathy (mild, moderate, or severe)
Congenital heart disease
Neonatal stroke
Congenital infections
Nosocomial infections
Inborn errors of metabolism
Severe hemodynamic/ventilatory instability
Seizures
Brain malformations
Central nervous system infection
Extracorporeal membrane oxygenation
PBSF, protecting brains and saving futures.
Variables
Population demographics: gender, type of delivery, gestational age at birth, gestational age ≤ 32 weeks and < 37 weeks, Apgar score at 1 min, 5 min and 10 min, birth weight, current weight, inborn or out born, use of antenatal steroids and magnesium sulfate. Primary diagnosis: seizure, mild HIE, moderate or severe HIE/cooling, neurologic abnormalities or congenital central nervous system (CNS) anomalies, grade III or IV intraventricular hemorrhage (IVH) or hydrocephalus, periventricular leukomalacia, meningitis, neural tube defects, stroke, cyanotic congenital heart defect (CHD), prematurity, gestational age ≤ 32 weeks, meconium aspiration syndrome, cardiorespiratory instability (anemia, shock and respiratory alkalosis/acidosis), necrotizing enterocolitis, metabolic disease, extracorporeal membrane oxygenation (ECMO)/pre-ECMO. For descriptive analysis patients will be divided into two groups accordingly to their primary diagnosis: neurological or clinical. A minimal anonymized data set necessary to replicate future study findings is provided as (S4 Appendix).
Outcomes
The primary outcomes are detailed on Box 2. Results will be described as n (%) or median [IQR] for the overall population and for each center. Then, we will compare changes over time for the 3 years of data collection within and between centers. Finally, those results will be compared between 2 major groups: neurologic vs clinical patients.
Box 2. Primary outcomes.
Adherence to the PBSF Guidelines
Use and duration of aEEG/EEG/NIRS monitoring
Use of neuroimaging exams
Number of Sub-specialties consultation (neurology and neurosurgery)
Number of primary neurologic or medical patients with aEEG, EEG and/or NIRS monitoring and the duration of the monitoring (hours)
Number of primary neurologic or medical patients with brain MRI, neurology consult, and neurosurgery consult
Number of clinical case discussions and videoconference meetings
Clinical outcomes
Number of electroencephalographic seizures during hospitalization
Use and types of anticonvulsants administered
Use and types of inotropes and fluid resuscitation administered during NICU stay
Death before hospital discharge
Length of hospital stay
Number of patients referred to high-risk infant follow-up*
PBSF, protecting brains and saving futures. *Every attempt will be made to obtain 18–24 months follow-up of these infants, with a Bayley III evaluation.
Secondary outcomes are outlined on Box 3. To address potential source of bias related to financial availability, the outcomes of interest will be also adjusted based on the unit primary profile: private, philanthropic, foundation or public.
Box 3. Secondary outcomes.
Descriptive usage of PBSF Guidelines
Number of remote communications between CSI and local team
Number of reports issued for aEEG / EEG exams with or without the use of NIRS
Number of patients who performed Therapeutic Hypothermia
Adverse effects of brain monitoring expressed by skin lesion due to electrode / sensor positioning
PBSF, protecting brains and saving futures; CSI, Central of Surveillance and Intelligence; NIRS, near infrared spectroscopy; aEEG, amplitude integrated electroencephalography; EEG, electroencephalography; MRI, magnetic resonance image; US, ultrasonography.
Sample size
Based on our current experience, an average of 5 patients per month with criteria for neurocritical care are admitted on each center, making a total of 3,240 eligible patients during the 3-year period. Considering a 30% loss due to refusal to consent, non-availability of the research team or missing data, we expect to recruit 2,268 infants from all study centers (around 756 patients per year).
Data collection and management
Patient demographics, diagnosis and clinical outcomes will be extracted from the medical charts of each patient and entered in the PBSF database. The adherence to the PBSF Guidelines will be measured with the data collected by the CSI center. Details of these recordings are provided on S3 Appendix.
A flow chart of all eligible and enrolled patients will be produced, and the total number of patients will be reported, overall and per center.
Statistical methods
Categorical variables will be analyzed by descriptive statistics and presented as number of valid cases and percentage (%). Numeric variables will be analyzed as mean and standard deviation, median and interquartile ranges, or confident intervals. Since, it is possible that the patient population is quite different across the sites, analyses for the overall population will account for site differences or for the possibility of correlated observations within a site.
To compare adherence and outcomes over time (i.e., between years 1, 2 and 3) the following statistical approach will be applied: a) a test for normality in each category using the Shapiro-Wilks (<50 observations) or Kolmogorov–Smirnov, b) if normality assumption holds, a Welch’s F test will be done, c) if normality assumption still holds the ANOVA with post hoc Bonferroni will be performed. If the normality assumption is violated, a non-parametric test (Kruskal–Wallis) will be used to analyze categorical variables and for significant differences between the two groups (neurological vs clinical). A final p value of <0.05 will be considered statistically significant. Analysis of variability (distribution) per site for each variable of the 2 main outcomes will also be performed and Run Chart will be generated to follow the evolution of the outcomes over time for the 2 groups of patients: neurologic and clinical. All analysis will be done using the StataCorp. 2021. Stata Statistical Software: Release 17. College Station, TX: StataCorp LLC.
Missing data
As missing data can impact study validity by reducing sample size and statistical power of subgroup analyses, a comprehensive strategy was developed. First, missing data will be defined as values that are not available and that would be meaningful for analysis if they were observed. Second, the proportion of missing data for each variable will be presented. Furthermore, analysis to assess if patients with missing data differ from those with complete information for a given variable will be performed. Finally, information on why data are missing will be collected.
All efforts will be made to prevent missing data by a) collecting only critical data elements, b) focus on routinely collected data in real-world practice, c) develop completion guidelines, and d) perform pilot-test forms for the first 10 patients. Also, an ongoing data review in the CSI will be conducted for data completeness and quality. Moreover, investigators of each site will be trained on data collection and the importance of core data.
For analysis, we will understand the missing data, select the appropriate methodology (ies) for handling it and examine the sensitivity of different approaches. Thus, for each core variable we will generate frequency distributions and graphical displays. Two major methods will be used during analysis to handle missing data. For the primary analysis we are planning to perform a complete case analysis (use only cases with complete data) and them apply likelihood methods, i.e. mixed effect models to find the values of parameters which maximize the known likelihood value.
Discussion
This large prospective, multicenter, and observational study will describe the use and adherence to a unique telemedicine neonatal neurocritical care guidelines and clinical outcomes. As a multidisciplinary approach to care for newborns with high-risk of brain injury has increased over the last 10 years, the implementation of an organized, low-cost, and high-expertise telemedicine guidelines has the potential to streamline clinical care improving outcomes.
The PBSF Guidelines defines high-risk neonates, with clear indications for neonatal neurocritical care. Among the etiologies associated with impaired neurodevelopmental outcomes, neonatal asphyxia represents the third cause of neonatal death worldwide (23%) [2, 3, 32–34]. In newborns with moderate or severe HIE the risk of death, cerebral palsy and/or disability among survivors is high. In addition, some disability might also occur in mild HIE infants [4, 6, 7, 35]. Neonatal seizures have been reported in approximately 3.5 per 1000 live births of term neonates [17] and several conditions are associated with increased risks for developing seizures such as perinatal asphyxia and ischemic stroke. Indeed, it is estimated that perinatal asphyxia is the leading cause of seizures in the neonatal period, accounting for 40 to 60% of all seizures in term newborns [36, 37]. Seizure burden is associated with more severe brain injury and worse neurodevelopmental outcomes after adjusting for underlying severity of brain injury. Status epilepticus is also associated with post neonatal epilepsy [17]. Since seizures are difficult to diagnose clinically, continuous monitoring by using aEEG, EEG or video aEEG/EEG will help the identification of sub-clinical events allowing immediate recognition and treatment, which can reduce seizure burden and improve neurodevelopmental outcomes [13–16].
Preterm infants are also at high risk for neurodevelopmental problems, and different areas may be affected such as neurosensory, cognitive, and behavioral functioning. The more immature, the greater the risks [8, 38] and extreme prematurity has been associated with moderate or severe neurocognitive deficits during childhood [39]. Moderate and late preterm infants (MLPT) can also have worse cognitive, language, motor development, social-emotional competence at 2 years of age when compared to term infants [40]. Other disorders such as cerebral malformations, infections, inborn errors of metabolism, and severe hemodynamic and ventilatory instability also carry risks of brain injury and seizures. Moreover, CHD affects from 4 to 75 per 1,000 live births and has been recognized as high risk for neurodevelopmental problems [41, 42]. In CHD, infants brain injury may follow episodes of cerebral hypoxia or hypoperfusion that can occur during the pre-, intra- and/or postoperative periods, in addition to biological and environmental factors [43–45]. Indeed, more than 50% of CHD patients have been reported with neurodevelopmental deficiencies [44–47] that may persist into adolescence [47]. All these situations are included as indications for the use of the PBSF Guidelines.
In recent decades, brain monitoring has become increasingly common in NICUs [48] but not yet a consolidated practice in all services, especially in low- and middle- income countries (LMIC), where the lack of structure, resources, and professional training is a major problem. Brazil is a large country with a population of > 200 million people and approximately 3 million births per year, with great financial disparity. This discrepancy directly affects the Brazilian health system, with differences in availability of financial, material, and personal resources, including adequate training of professionals. A prospective cross-sectional national survey conducted by our group evaluated the assessment of HIE and TH practices in Brazil. A total of 1,092 professionals answered and 62% reported using TH in their units. However only 13% received specific training for HIE assessment, 22% did not use any neurologic score, and only 12% had brain monitoring readily available, showing the need for well-established neurocritical care guidelines [31].
There are a variety of clinical applications of video aEEG/EEG monitoring and NIRS in neonates at high-risk for brain injury:
Seizures: clinical signs of seizures are easy to evaluate and overdiagnosis occurs frequently, resulting in unnecessary treatment [21, 33, 36]. Neonatal seizures are frequently underdiagnosed since more than 80–90% of seizures are subclinical, resulting in undertreatment. Indeed, the accuracy for seizure detection with clinical evaluation alone is < 9% [37]. New devices associate aEEG with raw EEG readings and even video imaging (video aEEG/EEG), providing higher accuracy for the detection of seizures [12, 36, 48, 49]. Shah et al. evaluated term newborns with clinical seizures monitored with aEEG with simultaneous continuous conventional EEG for seizure detection. In 41 non-status epilepticus seizures, 31 were correctly identified (sensitivity, 76%; specificity, 78%; PPV, 78%; NPV, 78%). However, they found a lower sensitivity (27–56%) when aEEG was assessed without the conventional EEG findings [11].
Hypoxic Ischemic Encephalopathy: Three of the six major clinical trials of TH used an abnormal aEEG background activity as entry criteria [50–52]. Indeed, an abnormal aEEG may improve selection of neonates to be treated [53]. However, the presence of a normal early aEEG should not exclude indication of TH in infants with neurological evidence of moderate to severe HIE [54]. After the implementation of TH, the predictive value of aEEG has been evaluated. Chandrasekaran et al. found that persistently abnormal trace on aEEG at ≥ 48 hours of life was associated with adverse long-term outcomes and a systematic review [55] found that maximum predictive reliability was achieved at 72 h of life with a post-test probability of 95.7% (95% CI: 84.4 to 98.5%). Therefore, aEEG is a valuable tool in infants with HIE to be used for the classification of the level of encephalopathy and as a predictor of long-term outcomes. The persistence of elevated values of rScO2 was also associated with abnormal neuroimaging and adverse neurodevelopmental outcomes as low cerebral metabolism indicates low oxygen use, cerebral hyperperfusion and impaired autorregulation [56–58].
Preterm infants: the incidence of seizures in preterm infants has been reported between 4 to 48%, and seizures were associated with adverse outcomes including IVH, white matter injury (WMI), cognitive impairment and neonatal death [13, 16, 59, 60]. The presence of SWC and continuous background pattern in the first week of life were associated to good neurodevelopmental outcomes, while absence of SWC and abnormal background activity were associated with neonatal death, severe IVH, WMI and neurological impairment at 2 years of age [61–64]. In extremely premature newborns during the first days of life, the occurrence of episodes of hypoxia/hyperoxia, hemodynamic and ventilatory instability, and systemic hypotension with decreased cerebral flow are common. For that, NIRS monitoring has been used extensively used in this population. A phase II randomized controlled trial conducted by the SafeBoosC Consortium evaluated newborns ≤ 72 h of life to demonstrate the feasibility and effectiveness of implementing continuous NIRS monitoring with a clinical management protocol. Neonates monitored with NIRS had lower burden of cerebral hypoxia or hyperoxia compared to controls and a trend towards lower IVH and mortality rates [65]. A hemodynamically significant patent ductus arteriosus (hsPDA) may be associated with increased pulmonary blood flow and decreased systemic blood flow. Chock et. al found that low renal oxygenation values (rSrO2), with rSrO2 < 66%, were associated with the presence of hemodynamically significant PDA [24]. Other studies have successfully demonstrated association between PDA closure and significant increases on cerebral and renal saturation [66, 67].
-
Other Applications: ventilatory instability (respiratory acidosis/alkalosis), metabolic disorders, inborn errors of metabolism, cerebral malformations, stroke, congenital infections, sepsis, meningitis and need of extracorporeal circulation (ECMO) [48, 68, 69]. In 20% of infants with CHD electrographic seizures were detected, including repetitive seizures and status epilepticus [70]. Early recovery to normal aEEG background activity and presence of SWC was associated with good neurological outcomes after cardiac surgery [46, 71]. Perioperative electrical seizures were found in 30% of infants, of which only ¼ showed any clinical correlation. Failure to recover the background activity to a continuous pattern ≥ 48 h postoperatively was associated with neurodevelopmental delay. The persistence of abnormal aEEG for seven days after surgery was also highly associated with death [46].
NIRS monitoring may help to evaluate efficacy or need for additional interventions such as ventilator changes, use of diuretics, change in prostaglandin (PGE) dose, need for blood transfusion or early surgical intervention. A rSrO2 < 30% was correlated with increased Intensive Care Units (ICU) stay, and rScO2 and rSrO2 < 45% and < 40% respectively, were correlated with need of ECMO and death [72]. NIRS monitoring was also useful to diagnose early renal insufficiency after surgical intervention [73–75].
Anemia and shock: tissue oxygen delivery, somatic and cerebral saturations are directly affected by hemoglobin levels, especially in scenarios of important anemia, suggesting that NIRS monitoring may play a role in the indication of blood transfusion. A significant increase in rScO2 values was reported in children with rScO2 <55% treated with blood transfusion [76]. NIRS has also been demonstrated as a good early indicator of abnormal somatic perfusion and shock in different populations including newborns [77–79]. In the setting of shock, two site NIRS monitoring (cerebral and somatic) may be particularly useful in cases where autoregulation remains intact and somatic tissue oxygenation may be compromised earlier [80].
Implementation of a NeuroNICU requires specialized education, training, use of validated protocols and implementation of specific methodologies. Financial resources are needed for equipment, such as servo-controlled temperature and brain monitoring devices. Lack of specialized assistance and resources may result in inadequate interventions with possible lack of benefit or even harm to the patients. A systematic review found no reduction in mortality with the use of TH in LMIC countries [81] and possible reasons to justify this finding include inefficiency of the low technology cooling devices and lack of optimal care. Brain monitoring using conventional EEG requires skilled technologists, experienced neurologist interpretation which may not be readily available in many centers, especially for continuous bedside monitoring. aEEG and NIRS monitoring can be interpreted at the bedside but also requires training and experienced users [82, 83]. Unfortunately, the majority of neonatal units lack adequate longitudinal training, specialized staff, and access to all these resources, especially in LMIC. This is what motivated our group to consider telemedicine as a solution in this scenario.
Differently from the previous studies, the PBSF Guidelines (S3 Appendix) is unique for the use of an advanced telemedicine system to provide neurocritical care. The guidelines include continuous neuromonitoring with data recording and real time feedback from experts, provision of equipment and resources to the NICUs, and short- and long-term teaching to the professionals working at the units (S2 Appendix). Telemedicine has several applications including education, consultation, practice, and research [28]. This type of communication has shown results in continuing education among physicians and nurses, and has assisted healthcare professionals to improve health disparities, promoting access to specialized care [27, 29]. Live interactive audiovisual materials, such as live streaming video and viewing stored educational material, are important to deliver tele-education; it promotes opportunities for continuous learning and development by dissemination of information and training health-care professionals [26, 28]. Telemedicine may also be helpful in case discussions for decision making which is useful to promote interaction between physicians, nurses, and patients. Udeh et al. exemplified some benefits of telemedicine including [84]: a) promote evidence-based best practices through checklists and prompting; b) enhance monitoring, early identification, and treatment of critical illness; c) improve coordination of care; and d) increase night-time vigilance. The use of telemedicine in ICUs is associated with decreased mortality and length of stay [84, 85]. Surprisingly, telemedicine has not been used on a significant scale worldwide, although this methodology is low-cost, feasible, clinically useful, and sustainable. It could be even more useful in LMIC countries due to the limited structure and resources.
Brazil allows several opportunities for telemedicine implementation, expanding the actions of healthcare professionals and integrating healthcare services [86]. We developed the PBSF Guidelines with a surveillance and intelligence center to support clinical decisions across the country. Telemedicine has been used by our group in the past few years to promote educational training and remote neuromonitoring but its impact on clinical outcomes needs to be formally evaluated. Thus, results of this 3 years prospective, multicenter, and observational study will help to determine the incidence of neonatal problems associated with brain injury and disabilities, the incidence of clinical and subclinical seizures, detection of brain hemodynamic/oxygenation instability and associated interventions. The study will be limited to the 20 NICUs using the PBSF Guidelines and variability in clinical practices, experience of health care providers and staffing of the units, will be analyzed as it may affect some of the outcomes. Enrollment of such large number of patients will require a strong coordination between the central unit and all 20 centers, which has already been established by the CSI. Practical or operational issues will be to assure that all eligible patients are identified in a daily basis and approached for consent. Data collection requires careful training of data extractors and timely and precise data entry. A computerized and safe double-checking system has been developed. Biological signals will be continuously collected and stored at the CSI and multimodal signal analysis has been developed. Although low, there are costs for the implementation and use of the PBSF Guidelines. Thus, an adjusted analysis based on the profile of the units will be performed as costs might represent a limitation on the generalizability of the study findings. A future cost/benefit analysis is under planning.
Supporting information
(PDF)
(PDF)
(PDF)
(PDF)
(PDF)
(PDF)
(PDF)
(PDF)
(PDF)
(PDF)
(PDF)
Abbreviations
- aEEG
Amplitude Integrated Electroencephalography
- CBF
Cerebral Blood Flow
- CHD
Congenital Heart Defect
- cMRI
Cranial Magnetic Resonance Imaging
- CNS
Central Nervous System
- CSI
Central of Surveillance and Intelligence
- cUS
Cranial Ultrasonography
- ECMO
Extracorporeal Membrane Oxygenation
- EEG
Electroencephalography
- HIE
Hypoxic-Ischemic Encephalopathy
- HR
Heart Rate
- hsPDA
Hemodynamically Significant Patent Ductus Arteriosus
- ICU
Intensive Care Units
- IVH
Intraventricular Hemorrhage
- MLPT
Moderate and Late Preterm Infants
- NIBP
Non-invasive Blood Pressure
- NICU
Neonatal Intensive Care Unit
- NIRS
Near Infrared Spectroscopy
- PBSF
Protecting Brains & Saving Futures
- pCO2
Partial Pressure of Carbon Dioxide
- PGE
Prostaglandin
- RCT
Randomized Controlled Trials
- rScO2
Cerebral Tissue Oxygenation
- rSO2
Cerebral and Somatic Tissue Oxygenation
- rSrO2
Renal Tissue Oxygenation
- SpO2
Pulse Oximetry
- SWC
Sleep-Wake Cycle
- TH
Therapeutic Hypothermia
- WHO
World Health Organization
- WMI
White Matter Injury
Funding Statement
GFTV and AN received funds from the Protecting Brain and Saving Futures (PBSF) Organization (www.pbsf.com.br). Both authors were involved in the design of the study, data collection and analysis, writing of the final manuscript, and decision to publish. PBSF Organization is registered with CNPJ 28.905.143/0001-61, headquartered at Av. Nove de Julho, 3229 – 13rd floor, room 1301 – Jardim Paulista, São Paulo, SP, Brazil – 01407-000, E-mail: contato@pbsf.com.br; Phone: + 55 (11) 3044-2557.
References
- 1.Kurinczuk JJ, White-Koning M, Badawi N. Epidemiology of neonatal encephalopathy and hypoxic-ischaemic encephalopathy. Early Hum Dev. 2010;86(6): 329–338. doi: 10.1016/j.earlhumdev.2010.05.010 [DOI] [PubMed] [Google Scholar]
- 2.Lawn JE, Cousens S, Zupan J, Lancet Neonatal Survival Steering Team. 4 million neonatal deaths: When? Where? Why? Lancet. 2005;365(9462): 891–900. doi: 10.1016/S0140-6736(05)71048-5 [DOI] [PubMed] [Google Scholar]
- 3.Lawn JE, Wilczynska-Ketende K, Cousens SN. Estimating the causes of 4 million neonatal deaths in the year 2000. Int J Epidemiol. 2006;35(3): 706–718. doi: 10.1093/ije/dyl043 [DOI] [PubMed] [Google Scholar]
- 4.Shankaran S, Woldt E, Koepke T, Bedard MP, Nandyal R. Acute neonatal morbidity and long-term central nervous system sequelae of perinatal asphyxia in term infants. Early Hum Dev. 1991;25(2): 135–148. doi: 10.1016/0378-3782(91)90191-5 [DOI] [PubMed] [Google Scholar]
- 5.Robertson CMT. Long-term follow-up of term infants with perinatal asphyxia. In: Stevenson DK, Benitz WE, Sunshine P. Fetal and neonatal brain injury. 3. Ed. New York: Cambridge University; 2003. p. 829–858. [Google Scholar]
- 6.de Vries LS, Jongmans MJ. Long-term outcome after neonatal hypoxic-ischemic encephalopathy. Arch Dis Child Fetal Neonatal Ed. 2010;95(3): F220–F224. doi: 10.1136/adc.2008.148205 [DOI] [PubMed] [Google Scholar]
- 7.Marlow N, Rose AS, Rands CE, Draper ES. Neuropsychological and educational problems at school age associated with neonatal encephalopathy. Arch Dis Child Fetal Neonatal Ed. 2005;90(5): F380–F387. doi: 10.1136/adc.2004.067520 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Blencowe H, Lee ACC, Cousens S, Bahalim A, Narwal R, Zhong N, et al. Preterm birth associated neurodevelopmental impairment estimates at regional and global levels for 2010. Pediatr Res. 2013;74(Suppl 1): 17–34. doi: 10.1038/pr.2013.204 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hellström-Westas L, Rosén I. Continuous brain-function monitoring: state of the art in clinical practice. Semin Fetal Neonatal Med. 2006;11(6): 503–511. doi: 10.1016/j.siny.2006.07.011 [DOI] [PubMed] [Google Scholar]
- 10.Hellström-Westas L, Rosén I, de Vries LS, Greisen G. Amplitude-integrated EEG Classification and Interpretation in Preterm and Term Infants. NeoReviews. 2006;7(2): e76–e87. [Google Scholar]
- 11.Shah DK, Mackay MT, Lavery S, Watson S, Harvey AS, Zempel J, et al. Accuracy of bedside electroencephalographic monitoring in comparison with simultaneous continuous conventional electroencephalography for detection of neonatal seizures in term infants. Pediatrics. 2008;121(6): 1146–1154. doi: 10.1542/peds.2007-1839 [DOI] [PubMed] [Google Scholar]
- 12.Mastrangelo M, Fiocchi I, Fontana P, Gorgone G, Lista G, Belcastro V. Acute neonatal encephalopathy and seizures recurrence: a combined aEEG/EEG study. Seizure. 2013;22(9): 703–707. doi: 10.1016/j.seizure.2013.05.006 [DOI] [PubMed] [Google Scholar]
- 13.Shah DK, Zempel J, Barton T, Lukas K, Inder TE. Electrographic seizures in preterm infants during the first week of life are associated with cerebral injury. Pediatr Res. 2010;67(1): 102–106. doi: 10.1203/PDR.0b013e3181bf5914 [DOI] [PubMed] [Google Scholar]
- 14.Payne ET, Zhao XY, Frndova H, McBain K, Sharma R, Hutchison JS, et al. Seizure burden is independently associated with short term outcome in critically ill children. Brain. 2014;137(5): 1429–1438. doi: 10.1093/brain/awu042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.van Rooij LG, Toet MC, van Huffelen AC, Groenendaal F, Laan W, Zecic A, et al. Effect of treatment of subclinical neonatal seizures detected with aEEG: randomized, controlled trial. Pediatrics 2010;125(2): e358–e366. doi: 10.1542/peds.2009-0136 [DOI] [PubMed] [Google Scholar]
- 16.Vesoulis ZA, Inder TE, Woodward LJ, Buse B, Vavasseur C, Mathur AM. Early electrographic seizures, brain injury, and neurodevelopmental risk in the very preterm infant. Pediatr Res 2014;75(4): 564–569. doi: 10.1038/pr.2013.245 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Srinivasakumar P, Zempel J, Trivedi S, Wallendorf M, Rao R, Smith B, et al. Treating EEG Seizures in Hypoxic Ischemic Encephalopathy: A Randomized Controlled Trial. Pediatrics. 2015;136(5): e1302–e1309. doi: 10.1542/peds.2014-3777 [DOI] [PubMed] [Google Scholar]
- 18.Hellström-Westas L, Rosen I, Svenningsen NW. Predictive value of early continuous amplitude integrated EEG recordings on outcome after severe birth asphyxia in full term infants. Arch Dis Child Fetal Neonatal Ed. 1995;72(1): F34–F38. doi: 10.1136/fn.72.1.f34 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Thoresen M, Hellström-Westas L, Liu X, de Vries LS. Effect of hypothermia on amplitude-integrated electroencephalogram in infants with asphyxia. Pediatrics. 2010;126(1): e131–e139. doi: 10.1542/peds.2009-2938 [DOI] [PubMed] [Google Scholar]
- 20.van Rooij LG, Toet MC, Osredkar D, van Huffelen AC, Groenendaal F, de Vries LD. Recovery of amplitude electroencephalographic background patterns within 24 hours of perinatal asphyxia. Arch Dis Child Fetal Neonatal Ed. 2005;90(3): F245–F251. doi: 10.1136/adc.2004.064964 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Del Río R, Ochoa C, Alarcon A, Arnáez J, Blanco D, García-Alix A. Amplitude integrated electroencephalogram as a prognostic tool in neonates with hypoxic- ischemic encephalopathy: a systematic review. PLoS One. 2016;11(11): e0165744. doi: 10.1371/journal.pone.0165744 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.van Bel F, Lemmers PMA, Naulaers G. Monitoring neonatal regional cerebral oxygen saturation in clinical practice: value and pitfalls. Neonatology. 2008;94(4): 237–244. doi: 10.1159/000151642 [DOI] [PubMed] [Google Scholar]
- 23.Alderliesten T, De Vis JB, Lemmers PMA, Hendrikse J, Groenendaal F, van Bel F, et al. Brain oxygen saturation assessment in neonates using T2-prepared blood imaging of oxygen saturation and near-infrared spectroscopy. J Cereb Blood Flow Metab. 2017;37(3): 902–913. doi: 10.1177/0271678X16647737 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chock VY, Rose LA, Mante JV, Punn R. Near-infrared spectroscopy for detection of a significant patent ductus arteriosus. Pediatr Res. 2016;80(5): 675–680. doi: 10.1038/pr.2016.148 [DOI] [PubMed] [Google Scholar]
- 25.World Health Organization. A health telematics policy in support of WHO’s Health-for-All Strategy for Global Health Development. Geneva (Switzerland); 1998. [Google Scholar]
- 26.World Health Organization. Telemedicine: Opportunities and Development in Member States: Report on the second global survey on eHealth. Geneva (Switzerland); 2010. [Google Scholar]
- 27.Hall RW, Hall-Barrow J, Garcia-Rill E. Neonatal Regionalization Through Telemedicine Using a Community Based Research and Education Core Facility. Ethn Dis. 2010;20(1Suppl 1): S1-136-140. [PMC free article] [PubMed] [Google Scholar]
- 28.Burke BL Jr, Hall RW, Section on Telehealth Care. Telemedicine: Pediatric Applications. Pediatrics. 2015;136(1): e293–e308. doi: 10.1542/peds.2015-1517 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.McConnochie K, Wood N, Herendeen N, ten Hoopen C, Denk L, Neuderfer J. Integrating telemedicine in urban pediatric primary care: provider perspectives and performance. Telemed J E Health. 2010;16(3): 280–2888. doi: 10.1089/tmj.2009.0112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ministério da Saúde. DATASUS. [online] Tabnet. [accessed on Jan 10th 2018] https://datasus.saude.gov.br/informacoes-de-saude-tabnet/.
- 31.Variane GFT, Cunha LM, Pinto P, Brandao P, Mascaretti RS, Magalhães M, et al. Therapeutic Hypothermia in Brazil: A MultiProfessional National Survey. Am J Perinatol. 2019;36(11): 1150–1156. doi: 10.1055/s-0038-1676052 [DOI] [PubMed] [Google Scholar]
- 32.Triulzi D, Parazzini C, Righini A. Patterns of damage in the mature neonatal brain. Pediatric Radiol. 2006;36(7): 608–620. doi: 10.1007/s00247-006-0203-5 [DOI] [PubMed] [Google Scholar]
- 33.Ferriero DM. Neonatal brain injury. N Engl J Med. 2004;351(19): 1985–1995. doi: 10.1056/NEJMra041996 [DOI] [PubMed] [Google Scholar]
- 34.World Health Organization. [online] The World Health Report 2005—make every mother and child count; 2005. [accessed on Oct 14th 2019] https://www.who.int/whr/2005/en/.
- 35.Prempunpong C, Chalak LF, Garfinkle J, Shah B, Kalra V, Rollins N, et al. Prospective research on infants with mild encephalopathy: the PRIME study. J Perinatol. 2018;38(1): 80–85. doi: 10.1038/jp.2017.164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Abend NS, Wusthoff CJ, Goldberg EM, Dlugos DJ. Electrographic seizures and status epilepticus in critically ill children and neonates with encephalopathy. Lancet Neurol. 2013;12(12): 1170–1179. doi: 10.1016/S1474-4422(13)70246-1 [DOI] [PubMed] [Google Scholar]
- 37.Murray DM, Boylan GB, Ali I, Ryan CA, Murphy BP, Connolly S. Defining the gap between electrographic seizure burden, clinical expression and staff recognition of neonatal seizures. Arch Dis Child Fetal Neonatal Ed. 2008;93(3): F187–F191. doi: 10.1136/adc.2005.086314 [DOI] [PubMed] [Google Scholar]
- 38.Burnett AC, Cheong JLY, Doyle LW. Biological and Social Influences on the Neurodevelopmental Outcomes of Preterm Infants. Clin Perinatol. 2018;45(3): 485–500. doi: 10.1016/j.clp.2018.05.005 [DOI] [PubMed] [Google Scholar]
- 39.Joseph RM, O’Shea TM, Allred EN, Heeren T, Hirtz D, Jara H, et al. Neurocognitive and Academic Outcomes at Age 10 years of Extremely Preterm Newborns. Pediatrics. 2016;137(4): e20154343. doi: 10.1542/peds.2015-4343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cheong JL, Doyle LW, Burnett AC, Lee KJ, Walsh JM, Potter CR, et al. Association Between Moderate and Late Preterm Birth and Neurodevelopmental and Social-Emotional Development at Age 2 Years. JAMA Pediatr. 2017;171(4): e164805. doi: 10.1001/jamapediatrics.2016.4805 [DOI] [PubMed] [Google Scholar]
- 41.Bosi G, Garani G, Scorrano M, Calzolari E, IMER Working Party. Temporal variability in birth prevalence of congenital heart defects as recorded by a general birth defects registry. J Pediatr. 2003;142(6): 690–698. doi: 10.1067/mpd.2003.243 [DOI] [PubMed] [Google Scholar]
- 42.Hoffman JIE, Kaplan S. The incidence of congenital heart disease. J Am Coll Cardiol. 2002;39(12): 1890–1900. doi: 10.1016/s0735-1097(02)01886-7 [DOI] [PubMed] [Google Scholar]
- 43.Marino B, Lipkin PH, Newburger JW, Peacock G, Gerdes M, Gaynor JW, et al. Neurodevelopmental outcomes in children with congenital heart disease: Evaluation and management: A scientific statement from the American heart association. Circulation. 2012;126(9): 1143–1172. doi: 10.1161/CIR.0b013e318265ee8a [DOI] [PubMed] [Google Scholar]
- 44.Dimitropoulos A, McQuillen PS, Sethi V, Moosa A, Chau V, Xu D, et al. Brain injury and development in newborns with critical congenital heart disease. Neurology. 2013;81(3): 241–248. doi: 10.1212/WNL.0b013e31829bfdcf [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Limperopoulos C, Majnemer A, Shevell MI, Resenblatt B, Rohlicek C, Tchervenkov C. Neurologic status of newborns with congenital heart defects before open heart surgery. Pediatrics. 1999;103(2): 402–408. doi: 10.1542/peds.103.2.402 [DOI] [PubMed] [Google Scholar]
- 46.Gunn JK, Beca J, Hunt RW, Olischar M, Shekerdemian LS. Perioperative amplitude-integrated EEG and neurodevelopment in infants with congenital heart disease. Intensive Care Med. 2012;38(9): 1539–1547. doi: 10.1007/s00134-012-2608-y [DOI] [PubMed] [Google Scholar]
- 47.Gunn JK, Beca J, Penny DJ, Horton SB, d’Udekem YA, Brizard CP, et al. Amplitude-integrated electroencephalography and brain injury in infants undergoing Norwood-Type operations. Ann Thorac Surg. 2012;93(1): 170–176. doi: 10.1016/j.athoracsur.2011.08.014 [DOI] [PubMed] [Google Scholar]
- 48.Toso PA, González AJ, Pérez ME, Kattan J, Fabres JG, Tapia JL, et al. Clinical utility of early amplitude integrated EEG in monitoring term newborns at risk of neurological injury. J Pediatr (Rio J). 2014;90(2): 143–148. doi: 10.1016/j.jped.2013.07.004 [DOI] [PubMed] [Google Scholar]
- 49.Shah NA, Wusthoff CJ. How to use: amplitude-integrated EEG (aEEG). Arch Dis Child Educ Pract Ed. 2015;100(2): 75–81. doi: 10.1136/archdischild-2013-305676 [DOI] [PubMed] [Google Scholar]
- 50.Azzopardi DV, Strohm B, Edwards AD, Dyet L, Halliday HL, Juszczak E, et al. Moderate hypothermia to treat perinatal asphyxial encephalopathy. N Engl J Med. 2009;361(14): 1349–1358. doi: 10.1056/NEJMoa0900854 [DOI] [PubMed] [Google Scholar]
- 51.Gluckman PD, Wyatt JS, Azzopardi D, Ballard R, Edwards AD, Ferriero DM, et al. Selective head cooling with mild systemic hypothermia after neonatal encephalopathy: multicentre randomized trial. Lancet. 2005;365(9460): 663–670. doi: 10.1016/S0140-6736(05)17946-X [DOI] [PubMed] [Google Scholar]
- 52.Simbruner G, Mittal RA, Rohlmann F, Muche R, neo.nEURO.network Trial Participants. Systemic hypothermia after neonatal encephalopathy: outcomes of neo.nEURO.network RCT. Pediatrics. 2010;126(4): e771–e778. doi: 10.1542/peds.2009-2441 [DOI] [PubMed] [Google Scholar]
- 53.Skranes JH, Løhaugen G, Schumacher EM, Osredkar D, Server A, Cowan FM, et al. Amplitude-Integrated Electroencephalography Improves the Identification of Infants with Encephalopathy for Therapeutic Hypothermia and Predicts Neurodevelopmental Outcomes at 2 Years of Age. J Pediatr. 2017;187: 34–42. doi: 10.1016/j.jpeds.2017.04.041 [DOI] [PubMed] [Google Scholar]
- 54.Sarkar D, Barks JD, Donn SM. Should amplitude-integrated electroencephalography be used to identify infants suitable for hypothermic neuroprotection? J Perinatol. 2008;28(2): 117–122. doi: 10.1038/sj.jp.7211882 [DOI] [PubMed] [Google Scholar]
- 55.Chandrasekaran M, Chaban B, Montaldo P, Thayyil S. Predictive value of amplitude-integrated EEG (aEEG) after rescue hypothermia neuroprotection for hypoxic ischemic encephalopathy: a meta-analysis. J Perinatol. 2017;37(6): 684–689. doi: 10.1038/jp.2017.14 [DOI] [PubMed] [Google Scholar]
- 56.Ancora G, Maranella E, Grandi S, Sbravati F, Coccolini E, Savini S, et al. Early predictors of short term neurodevelopmental outcome in asphyxiated cooled infants. A combined brain amplitude integrated electroencephalography and near infrared spectroscopy study. Brain Dev. 2013;35(1): 26–31. doi: 10.1016/j.braindev.2011.09.008 [DOI] [PubMed] [Google Scholar]
- 57.Jain SV, Pagano L, Gilliam-Krakauer M, Slaughter JC, Pruthi S, Engelhardt B. Cerebral regional oxygen saturation trends in infants with hypoxic-ischemic encephalopathy. Early Hum Dev. 2017;113: 55–61. doi: 10.1016/j.earlhumdev.2017.07.008 [DOI] [PubMed] [Google Scholar]
- 58.Lemmers PM, Zwanenburg RJ, Benders MJNL, de Vries LS, Groenendaal F, van Bel F, et al. Cerebral oxygenation and brain activity after perinatal asphyxia: does hypothermia change their prognostic value? Pediatr Res. 2013;74(2): 180–185. doi: 10.1038/pr.2013.84 [DOI] [PubMed] [Google Scholar]
- 59.Tekgul H, Gauvreau K, Soul J, Murphy L, Robertson E, Stewart J, et al. The current etiologic profile and neurodevelopmental outcome of seizures in term newborn infants. Pediatrics. 2006;117(4): 1270–1280. doi: 10.1542/peds.2005-1178 [DOI] [PubMed] [Google Scholar]
- 60.Hellström-Westas L, Rosén I, Svenningsen NW. Cerebral function monitoring during the first week of life in extremely small low birthweight (ESLBW) infants. Neuropediatrics. 1991;22(1): 27–32. doi: 10.1055/s-2008-1071411 [DOI] [PubMed] [Google Scholar]
- 61.Wikström S, Pupp IH, Rosén I, Norman E, Fellman V, Ley D, et al. Early single-channel aEEG/EEG predicts outcome in very preterm infants. Acta Paediatr. 2012;101(7): 719–726. doi: 10.1111/j.1651-2227.2012.02677.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Soubasi V, Mitsakis K, Sarafidis K, Griva M, Nakas CT, Drossou V. Early abnormal amplitude-integrated electroencephalography (aEEG) is associated with adverse short-term outcome in premature infants. Eur J Paediatric Neurol. 2012;16(6): 625–630. doi: 10.1016/j.ejpn.2012.02.008 [DOI] [PubMed] [Google Scholar]
- 63.Klebermass K, Olischar M, Waldhoer T, Fuiko R, Pollak A, Weninger M. Amplitude-integrated EEG pattern predicts further outcome in preterm infants. Pediatr Res. 2011;70(1): 102–108. doi: 10.1203/PDR.0b013e31821ba200 [DOI] [PubMed] [Google Scholar]
- 64.Variane GFT, Magalhães M, Gasperine R, Alves HCBR, Scoppetta TLPD, Figueredo RJG, et al. Early amplitude-integrated electroencephalography for monitoring neonates at high risk for brain injury. J Pediatr. 2017;93(5): 460–466. doi: 10.1016/j.jped.2016.12.003 [DOI] [PubMed] [Google Scholar]
- 65.Hyttel-Sorensen S, Pellicer A, Alderliesten T, Austin T, van Bel F, Benders M, et al. Cerebral near infrared spectroscopy oximetry in extremely preterm infants: phase II randomised clinical trial. BMJ. 2015;350: g7635. doi: 10.1136/bmj.g7635 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lemmers PMA, Toet MC, van Bel F. Impact of patent ductus arteriosus and subsequent therapy with indomethacin on cerebral oxygenation in preterm infants. Pediatrics. 2008;121(1): 142–147. doi: 10.1542/peds.2007-0925 [DOI] [PubMed] [Google Scholar]
- 67.Underwood MA, Milstein JM, Sherman MP. Near-infrared spectroscopy as a screening tool for patent ductus arteriosus in extremely low birth weight infants. Neonatology. 2007;91(1): 134–139. doi: 10.1159/000097131 [DOI] [PubMed] [Google Scholar]
- 68.Olischar M, Shany E, Aygün C, Azzopardi D, Hunt RW, Toet MC, et al. Amplitude-integrated electroencephalography in newborns with inborn errors of metabolism. Neonatology. 2012;102(3): 203–211. doi: 10.1159/000339567 [DOI] [PubMed] [Google Scholar]
- 69.Helderman JB, Welch CD, Leng X, O’Shea TM. Sepsis-associated electroencephalographic changes in extremely low gestational age neonates. Early Hum Dev. 2010;86(8): 509–513. doi: 10.1016/j.earlhumdev.2010.06.006 [DOI] [PubMed] [Google Scholar]
- 70.ter Horst HJ, Mud M, Roofhooft MTR, Bos AF. Amplitude integrated electroencephalographic activity in infants with congenital heart disease before surgery. Early Hum Dev. 2010;86(12): 759–764. doi: 10.1016/j.earlhumdev.2010.08.028 [DOI] [PubMed] [Google Scholar]
- 71.Latal B, Wohlrab G, Brotschi B, Beck I, Knirsch W, Bernet V. Postoperative amplitude-integrated electroencephalography predicts four-year neurodevelopmental outcome in children with complex congenital heart disease. J Pediatr. 2016;178: 55–60. doi: 10.1016/j.jpeds.2016.06.050 [DOI] [PubMed] [Google Scholar]
- 72.Dodge-Khatami J, Gottschalk U, Eulenburg C, Wendt U, Schnegg C, Rebel M, et al. Prognostic value of perioperative near-infrared spectroscopy during neonatal and infant congenital heart surgery for adverse in-hospital clinical events. World J Pediatr Congenit Heart Surg. 2012;3(2): 221–228. doi: 10.1177/2150135111426298 [DOI] [PubMed] [Google Scholar]
- 73.Colasacco C, Worthen M, Peterson B, Lamberti J, Spear R. Near-Infrared Spectroscopy Monitoring to Predict Postoperative Renal Insufficiency Following repair of Congenital Heart Disease. World J Pediatr Congenit Heart Surg. 2011;2(4): 536–540. doi: 10.1177/2150135111411932 [DOI] [PubMed] [Google Scholar]
- 74.Hoffman GM, Guanayem NS, Scott JP, Tweddell JS, Mitchell ME, Mussatto KA. Postoperative Cerebral and Somatic Near-Infrared Spectroscopy Saturations and Outcome in Hypoplastic Left Heart Syndrome. Ann Thorac Surg. 2017;103(5): 1527–1535. doi: 10.1016/j.athoracsur.2016.09.100 [DOI] [PubMed] [Google Scholar]
- 75.Clair MP, Rambaud J, Flahault A, Guedj R, Guilbert J, Guellec I, et al. Prognostic value of cerebral tissue oxygen saturation during neonatal extracorporeal membrane oxygenation. PLoS One. 2017;12(3): e0172991. doi: 10.1371/journal.pone.0172991 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Sood BG, McLaughlin K, Cortez J. Near-infrared spectroscopy: applications in neonates. Semin Fetal Neonatal Med. 2015;20(3): 164–172. doi: 10.1016/j.siny.2015.03.008 [DOI] [PubMed] [Google Scholar]
- 77.Johnson BA, Hoffman GM, Tweddell JS, Cava JR, Basir M, Mitchell ME, et al. Near-infrared spectroscopy in neonates before palliation of hypoplastic left heart syndrome. Ann Thorac Surg. 2009;87(2): 571–577. doi: 10.1016/j.athoracsur.2008.10.043 [DOI] [PubMed] [Google Scholar]
- 78.Hanson SJ, Berens RJ, Havens PL, Kim MK, Hoffman GM. Effect of volume resuscitation on regional perfusion in dehydrated pediatric patients as measured by two-site near-infrared spectroscopy. Pediatr Emerg Care. 2009;25(3): 150–153. doi: 10.1097/PEC.0b013e31819a7f60 [DOI] [PubMed] [Google Scholar]
- 79.Pichler G, Höller N, Baik-Schneditz N, Schwaberger B, Mileder L, Stadler J, et al. Avoiding arterial hypotension in preterm neonates (AHIP)–a single center randomised controlled study investigating simultaneous near infrared spectroscopy measurements of cerebral and peripheral regional tissue oxygenation and dedicated interventions. Front Pediatr. 2018;6: 15. doi: 10.3389/fped.2018.00015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Variane GFT, Chock VY, Netto A, Pietrobom RFR, Van Meurs KP. Simultaneous Near-Infrared Spectroscopy (NIRS) and Amplitude-Integrated Electroencephalography (aEEG): Dual Use of Brain Monitoring Techniques Improves Our Understanding of Physiology. Front Pediatr. 2020;7: 560. doi: 10.3389/fped.2019.00560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Pauliah SS, Shankaran S, Wade A, Cady EB, Thayyil. Therapeutic hypothermia for neonatal encephalopathy in low-and middle-income countries: a systematic review and meta-analysis. PLoS One. 2013;8(3): e58834. doi: 10.1371/journal.pone.0058834 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Frenkel N, Friger M, Meledin I, Berger I, Marks K, Bassan H, et al. Neonatal seizure recognition–comparative study of continuous-amplitude integrated EEG versus short conventional EEG recordings. Clin Neurophysiol. 2011;122(6): 1091–1097. doi: 10.1016/j.clinph.2010.09.028 [DOI] [PubMed] [Google Scholar]
- 83.Rakshasbhuvankar A, Paul S, Nagarajan L, Ghosh S, Rao S. Amplitude-integrated EEG for detection of neonatal seizures: a systematic review. Seizure. 2015;33: 90–98. doi: 10.1016/j.seizure.2015.09.014 [DOI] [PubMed] [Google Scholar]
- 84.Udeh C, Udeh B, Rahman N, Canfield C, Campbell J, Hata JS. Telemedicine/Virtual ICU: Where Are We and Where Are We Going? Methodist Debakey Cardiovasc J. 2018;14(2): 126–133. doi: 10.14797/mdcj-14-2-126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Kahn JM, Le TQ, Barnato AE, Hravnak M, Kuza CC, Pike F, et al. ICU telemedicine and critical care mortality: a national effectiveness study. Med Care. 2016;54(3): 319–325. doi: 10.1097/MLR.0000000000000485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Maldonado JM, Marque AB, Cruz A. Telemedicine: challenges to dissemination in Brazil. Cad Saúde Pública. 2016;32(Suppl 2): e00155615. doi: 10.1590/0102-311X00155615 [DOI] [PubMed] [Google Scholar]