Abstract
Background
The impact of COVID-19 on swallowing function is not well understood. Despite low hospital admission rates in Australia, the virus and subsequent treatment affects swallow function in those requiring intensive care unit (ICU) treatment. As such, the current pandemic provides a unique opportunity to describe swallowing function and outline dysphagia characteristics and trajectory of recovery for a series of cases across NSW.
Aim
The aims of this study were to describe (i) physiological characteristics of swallowing dysfunction and (ii) pattern of swallowing recovery and outcomes, in ICU patients with COVID-19.
Methods
All patients admitted to 17 participating NSW Health ICU sites over a 12-month period (March 2020–March 2021), diagnosed with COVID-19, treated with the aim for survival, and seen by a speech pathologist for clinical swallowing examination during hospital admission were considered for inclusion. Demographic, critical care airway management, speech pathology treatment, and swallowing outcome data were collected.
Results
Twenty-seven patients (22 male; 5 female) with a median age of 65 years (interquartile range [IQR] = 15.5) were recruited. All required mechanical ventilation. Almost 90% of the total cohort had pre-existing comorbidities, with the two most frequently observed being diabetes (63%, 95% confidence interval = 44%–78%) and cardiac disease (59%, 95% confidence interval = 40%–75%) in origin. Prevalence of dysphagia was 93%, with the majority (44%) exhibiting profound dysphagia at the initial assessment. Median duration to initiate oral feeding was 38.5 days (IQR = 31.25) from ICU admission, and 33% received dysphagia rehabilitation. Dysphagia recovery was observed in 81% with a median duration of 44 days (IQR = 29). Positive linear associations were identified between duration of intubation, mechanical ventilation, hospital and ICU length of stay, and the duration to speech pathology assessment (p < 0.005), dysphagia severity (p < 0.002), commencing oral intake (p < 0.02), dysphagia recovery (p < 0.004), and enteral feeding (p < 0.024).
Conclusion
COVID-19 considerably impacted swallowing function in the current study. Although many patients recovered within an acceptable timeframe, some experienced persistent severe dysphagia and a protracted recovery with dependence on enteral nutrition.
Keywords: COVID-19, SARS-CoV2, Dysphagia, Critically ill, Recovery
1. Introduction
The speech pathologist, as a core member of the multidisciplinary intensive care unit (ICU) team, is responsible for the timely and comprehensive management of complex swallowing and communication disorders. Such disorders may arise as a result of a patient's medical or surgical diagnosis and/or as a result of necessary ICU therapies including mechanical ventilation. Dysphagia in ICU patients has been associated with variables including duration of intubation,[1], [2], [3], [4], [5], [6], [7], [8], [9] presence of tracheostomy,10 advanced age,11 in addition to acute deconditioning.12 Presence of dysphagia in respiratory disease is also not uncommon, with incidence rates in conditions such as chronic obstructive pulmonary disease (COPD) documented as high as 85%.13 The pathophysiology behind dysphagia in both the ICU as well as respiratory patient appears to be multifactorial in nature, with evidence indicating anatomical changes,14 physiological deficits in laryngopharyngeal sensitivity,15 laryngeal and pharyngeal motor function,[16], [17], [18], [19], [20], [21] as well as altered breath–swallow synchrony.22 These physiological and anatomical changes translate into altered patterns of swallowing20 , 21 manifesting as a reduction in bolus flow efficiency, impaired airway closure, as well as diminished airway responsiveness. As such, patients in the ICU including those with respiratory disease are at a greater risk of dysphagia and aspiration and may take an increased duration to commence oral intake, thus increasing their dependence on enteral nutrition.
Given the complex and multifactorial profile of dysphagia in ICU patients, evidence suggests that application of instrumental swallowing assessment should be considered.23 , 24 Flexible endoscopic evaluation of swallowing (FEES) is frequently reported as the instrument of preference,16 , 25 , 26 given the high incidence of laryngeal impairment, medical fragility, and inability to easily transport these patients to the radiology department for other instrumental swallow assessments such as videofluoroscopy.23 Furthermore, although the role of the speech pathologist and application of FEES within the ICU is well recognised in the literature, referral protocols and degree of speech pathologist input along the care pathway remain largely site specific and at the discretion of the treating intensivist. A patient under the care of a speech pathologist within the ICU, as well as throughout acute and rehabilitative admission, receives assessment and treatment in line with national standards as outlined by the national peak body, Speech Pathology Australia. Dysphagia management includes regular clinical review of swallowing function and application of compensatory as well as therapeutic strategies with the aim to optimise safe diet and fluid consumption and enhance nutritional intake in addition to maximising quality of life.27
The impact of severe acute respiratory distress syndrome coronavirus 2 (SARS-COV2) on swallowing function is currently not well understood.28 , 29 Although the literature regarding dysphagia in patients with COVID-19 is currently limited, early evidence suggests a dysphagia prevalence of approximately 30% in those requiring hospitalisation.30 Looking specifically at the patient with COVID-19 in the ICU, data from international colleagues suggest that this dysphagia prevalence increases, with rates cited from 50%30 to 90%31 , 32 in those who require ICU treatment. Furthermore, authors have indicated that in this population, both the presence and duration of an endotracheal tube are positively correlated with dysphagia;30 , 33 however there are conflicting data regarding whether prone positioning for ventilation is associated with or predictive of dysphagia.32 , 33 Encouragingly, overall dysphagia recovery of the patient with COVID-19 in the ICU after extubation is rapid,31 with 70–90% regaining normal swallowing function during inpatient admission.30 , 32 , 33 Those who did experience persistent dysphagia frequently had pre-existing swallowing impairment or neurological diagnosis.33
Currently in Australia, we are fortunate to have much lower rates of COVID-19 than our international colleagues. Subsequently, we have had a relatively low rate of COVID-19 hospital admissions although we still see that the virus and its subsequent treatments can affect swallow function in those who require ICU treatment. As such, speech pathology assessment and management of COVID-19–related dysphagia has been necessary. With evidence defining the impact of COVID-19 on swallow function still emerging, the current pandemic provided us with the unique opportunity to describe swallowing function and outline clinical dysphagia characteristics and trajectory of recovery for a series of Australian cases treated in the ICU with confirmed COVID-19.
The aims of the study were to describe the (i) physiological characteristics of swallowing dysfunction and (ii) pattern of recovery and outcomes for swallowing, in ICU patients with COVID-19.
2. Methods
2.1. Design
This is a multisite prospective observational cohort study. This study has been conducted and reported in line with the STROBE statement.34
2.2. Participants & setting
A cohort of adult patients (aged 18–100 years) with confirmed COVID-19, admitted to ICUs across 17 participating NSW public hospitals (metropolitan and rural), and referred to speech pathology for face-to-face assessment of swallowing function during their acute hospital admission as per usual site-specific referral practices were considered for inclusion within the study. The study was conducted over a period of 12 months (1st March 2020 – 1st March 2021) with patients recruited only if the intention to treat was for survival.
2.3. Demographic data
Demographic data points collected on all participants from medical records included age, sex, hospital length of stay (LOS) in days, and past medical history including pre-existing dysphagia. ICU-specific data collected incorporated ICU LOS (days), APACHE-II35 score, duration of endotracheal intubation (days), duration of tracheostomy (days), duration of mechanical ventilation (days), number of intubations, complications, and discharge destination. All demographic endpoints relating to duration were calculated from the point of ICU admission.
2.4. Swallowing
Swallowing function was assessed via a clinical swallowing examination conducted by the speech pathologist with diagnosis and severity of impairment (dysphagia) defined by (i) the ability/inability to safely consume food and fluids and (ii) Functional Oral Intake Scale (FOIS),36 respectively. The clinical swallowing examination was conducted in accordance with individual patient needs and consistent with the clinical guidelines of Speech Pathology Australia.37 Specifically, this involved conducting a thorough medical and swallowing case history, cranial nerve assessment, oral trial of food and fluids, and potential trial of compensatory swallow strategies as clinically appropriate. The FOIS36 is a 7-point numerical scale where 1 = nothing by mouth; 2 = tube dependent with minimal attempts of food and fluid; 3 = tube dependent with consistent intake of food and fluid; 4 = total oral diet of a single consistency; 5 = total oral diet with multiple consistencies but requiring special preparation or compensations; 6 = total oral with multiple consistencies without special preparation, but with specific food limitations; 7 = total oral diet with no restriction.
Dysphagia management was deemed complete once either the patient had achieved a premorbid level of swallowing function ability or his/her swallow function had plateaued such that further improvement was deemed unlikely by the treating speech pathologist. Dysphagia resolution was defined by the ability to consume a full oral diet and fluids without modification or the aid of compensatory strategies.
Other specific swallowing endpoints included capturing information relevant to commencing oral intake, dysphagia rehabilitation, dysphagia resolution, instrumental assessment outcomes (if conducted), and non-oral (enteral) feeding. These endpoints are further detailed in Table 1 . All swallowing data relating to duration were calculated in days from the time of ICU admission. In the instance that the Videofluoroscopic Swallowing Study (VFSS) was conducted as part of standard of care, swallowing outcome measures applied to describe swallowing impairment were the Penetration–Aspiration Scale (PAS)39 and the Bolus Residue Scale (BRS).40 The PAS is an 8-point scale that describes the degree of food/fluid airway invasion and airway response, where 1 = no laryngeal penetration/aspiration and 8 = aspiration below the level of the vocal folds with nil airway response. The BRS is a 6-point scale which describes the degree of postswallow pharyngeal residue, where 1 = no residue and 6 = residue in the valleculae and posterior pharyngeal wall and piriform sinus. Each of these scales is a simple tool that can be efficiently applied to objectify VFSS interpretation specific to penetration/aspiration and pharyngeal clearance.
Table 1.
Swallowing endpoints (duration reported in days from ICU admission).
| Endpoint | Definition |
|---|---|
| DSPA | Duration to initial speech pathology assessment (telehealth/face-to-face) |
| DFSPA | Duration to face-to-face speech pathology assessment |
| Dysphagia presence & severity on CSE | Presence coded as dichotomous rating yes/no. Functional Oral Intake Scale: The FOIS is a 7-point scale where 1 = nothing by mouth and 7 = total oral diet with no restriction. (severe<4, moderate = 5, mild = 6, none = 7) |
| Dysphagia presence & severity on Videofluoroscopic Swallowing Study (VFSS) if conducted | Presence coded as dichotomous rating yes/no. Severity coded as follows:
|
| VFSS postponed or not completed due to COVID-19–specific considerations | Dichotomous rating yes/no, qualified by rationale for delay coded as follows:
|
| DCDR | Duration to commencing dysphagia rehabilitation Dysphagia rehabilitation defined as active therapeutic exercises beyond compensation with a targeted physiological outcome. |
| DIOF | Duration to initiate oral feeding |
| DROD | Duration to resolution of dysphagia |
| Persistent dysphagia after discharge | Patient discharged prior to treatment completion or dysphagia is considered permanent: Dichotomous rating yes/no |
| DOEF | Duration of enteral feeding |
| Persistent enteral feeding after discharge | Patient discharged prior to cessation of enteral feeding or enteral feeding is permanent: dichotomous rating yes/no |
| Speech pathology involvement in trachea weaning/decannulation | Dichotomous rating yes/no:
|
CSE, clinical swallowing examination.
2.5. Data collection
Individual site data were collected and entered by local site investigators and input into a purpose-built password-protected REDCap database38 via a secure survey link. A data dictionary defining each data point was provided to all sites to minimise bias.
To ensure completeness of data, the REDCap database38 was designed so that each data field (with the exception of APACHE-II35 score) was mandatory.
2.6. Data analysis
Anonymised data (deidentified at the point of data entry) were exported via an encrypted secure link generated by REDCap.38 This was subsequently downloaded for analysis in Excel and IBM SPSS Statistics for Windows version 27.0 (IBM Corp, Armonk, NY). Descriptive statistics were utilised to analyse all data. Normally distributed data are presented as means and standard deviation [mean (SD)], with non-normal data reported as medians and interquartile range [median (IQR)]. Categorical data are presented as a proportion of the sample [n (%)]. Correlation statistics between variables determined a priori was conducted using nonparametric assessments (Mann–Whitney U) between continuous and dichotomous variables, Spearman's rho between two continuous variables, and Fisher’s exact test between dichotomous variables, with statistical significance set at p < 0.05. Precisions of estimates were reported as 95% confidence intervals (CIs, with a normal approximation method).
This study received ethical approval (2020/ETH01301) from the CRGH Human Research & Ethics Committee. Written consent for the purposes of gathering outcomes was sought and obtained from all cases prior to data collection.
3. Results
3.1. Demographic & critical care outcomes
A total of 27 patients (22 male; 5 female) were recruited to participate in the study over a 12-month period (March 2020–March 2021). Seventeen NSW public hospitals contributed to participant recruitment. The median age of the cohort was 65 years (range = 38–81, IQR = 15.5), with all requiring mechanical ventilation and 59% (n = 16) tracheostomy as part of their ICU treatment. Almost 90% (n = 24) of the total cohort had pre-existing comorbidities, with the two most frequently observed being diabetes (63%, n = 17; 95% CI = 44%–78%) and cardiac disease (59%, n = 16; 95% CI = 41%–75%). Only one patient, who had multiple comorbidities including gastro-oesophageal reflux and recurrent oesophageal stenosis, had a known pre-existing dysphagia prior to admission (4%, n = 1; 95% CI = 1%–18%). The majority (74%, n = 20) of participants exhibited fewer than five comorbidities indexed prior to hospital admission. During the study period, a range of in-hospital complications were documented for participants. The most frequently occurring complications were ICU-acquired weakness (74%, n = 20; 95% CI = 55%–87%) and delirium (70%, n = 19; 95% CI = 51%–84%).
Participant discharge destination was most commonly home (70%, n = 19; 95% CI = 51%–84%) followed by another inpatient facility (26%, n = 7; 95% CI = 13%–45%). The in-hospital survival rate was 96% (n = 26; 95% CI = 82%–99%).
The detailed summary of demographic and critical care data is contained in Table 2 .
Table 2.
Demographic and critical care data.
| Population variable | Median (IQR) | Range |
|---|---|---|
| Age | 65 (15.5) | 38–81 |
| APACHE II score [n = 6] | 16.5 (27) | 13–53 |
| Total duration of MV (days) | 33 (27) | 8–72 |
| Total duration of ETT (days) | 21 (13.5) | 8–51 |
| Total duration of trachea (days) [n = 16] | 20 (9.5) | 7–46 |
| ICU LOS (days) | 37 (24) | 10–106 |
| Hospital LOS (days) | 50 (28) | 17–178 |
| Enteral feeding (days) | 40 (37) | 8–148 |
ETT, endotracheal tube; IQR, interquartile range; LOS, length of stay; MV, mechanical ventilation.
3.2. Swallowing outcomes
Prevalence of dysphagia on the initial assessment across the total cohort was 93% (n = 25), with the majority (n = 12, 44%) exhibiting profound dysphagia (FOIS 1) followed by those (n = 10, 37%) who were able to commence a modified diet (FOIS 5) at the point of the initial assessment. Of the remaining 11% (n = 3) who were dysphagic on the initial assessment, 4% (n = 1) were tube dependent with minimal attempts of food or fluid and 7% (n = 2) were able to tolerate a total oral diet but required specific food limitations. Calculated from the time of ICU admission, median duration to the initial speech pathology consultation (either face to face or via TeleHealth) was 29 days (IQR = 23), whereas median duration to face-to-face initial assessment was 33 days (IQR = 18). Duration to initiation of oral feeding was observed at a median of 38.5 days (IQR = 31.25) from the time of admission to the ICU, and for those who received therapeutic dysphagia rehabilitation (33%), the duration to commencing dysphagia rehabilitation occurred at a median of 39 days (IQR = 17). Dysphagia rehabilitation was not indicated in 63% (n = 17) of participants (owing to persistent delirium and/or favouring traditional compensatory strategies) and could not be provided owing to COVID considerations in 4% (n = 1). Resolution of dysphagia by the time of discharge from the acute care facility was achieved in 81% (n = 22) of all participants, with a median duration to resolution of dysphagiaof 44 days (IQR = 29). Enteral feeding was required in all cases, with a median duration of enteral feeding of 40 days (IQR = 37).
For the one-third of participants who received dysphagia rehabilitation (n = 9), the majority (n = 8, 89%; 95% CI = 57%–98%) had an FOIS score of 1 or 2 indicating profound dysphagia at the point of the initial assessment. These participants also took longer to commence oral intake (median = 48 days, IQR = 11) than those who did not require dysphagia rehabilitation (median = 34 days, IQR = 24). Of note, dysphagia rehabilitation was initiated prior to commencing oral intake in all but one of the nine cases, and dysphagia resolution took longer (median = 77 days, IQR = 27), with four (44.45%) unable to achieve dysphagia resolution. Furthermore, enteral feeding duration was longer in the rehabilitation group (median = 48 days, IQR = 18) than in the nonrehabilitation group (median = 35.5 days, IQR = 30).
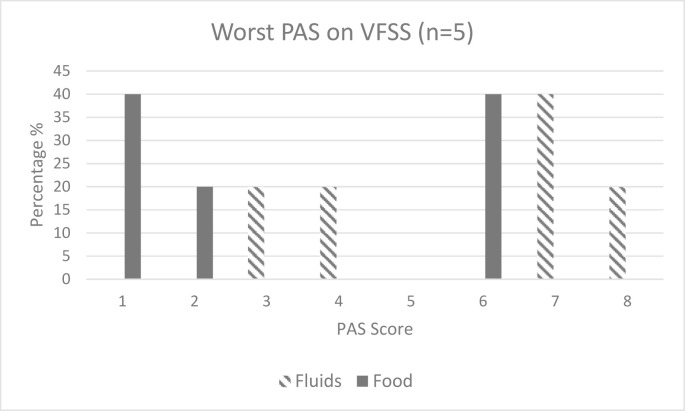
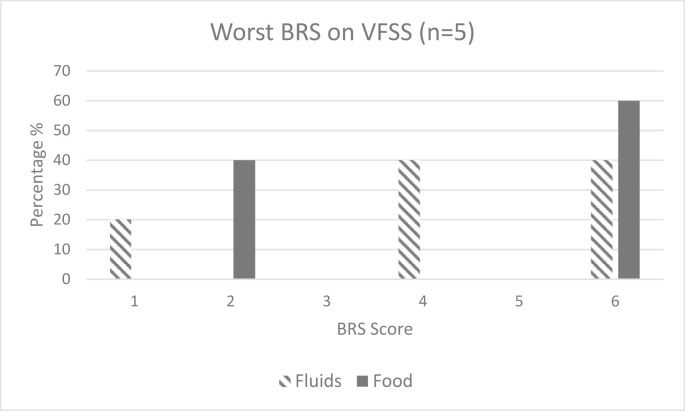
The VFSS was conducted by the speech pathologist and radiology staff members in 18.5% (n = 5) of the cohort, with two further cases not completed owing to COVID considerations or delayed owing to COVID-positive status. PAS39 scores (Fig. 1 ) were worse for fluids than for solids, but pharyngeal clearance as defined by the BRS40 scores (Fig. 2 ) was evenly distributed across both fluids and solids for those who underwent the VFSS. Those who underwent the VFSS took longer to initiate oral feeding (median = 47 days, IQR = 51) and commence dysphagia rehabilitation (median = 35.5 days, IQR = 31.75) and had a longer period of enteral feeding (median = 47 days, IQR = 59). Only two of the five who underwent the VFSS achieved dysphagia resolution (median = 104 days, IQR = 52).
Fig. 1.
Penetration–Aspiration Scale (PAS) scores on Videofluoroscopic Swallowing Study (VFSS); Penetration–Aspiration Scale: 1 = material does not enter airway; 2 = material enters airway above vocal cords & is ejected; 3 = material enters airway above vocal cords & is not ejected; 4 = material enters airway, contacts vocal cords & is ejected; 5 = material enters airway, contacts vocal cords & is not ejected; 6 = material enters airway, passes below vocal cords & is ejected; 7 = material enters airway, passes below vocal & is not ejected despite effort; 8 = material enters airway, passes below vocal cords with no effort made to eject.39
Fig. 2.
Bolus Residue Scale (BRS) scores on Videofluoroscopic Swallowing Study (VFSS), BRS: 1 = no residue; 2 = residue in valleculae; 3 = residue on posterior pharyngeal wall or piriform fossae; 4 = residue in valleculae and posterior pharyngeal wall or piriform fossae; 5 = residue in posterior pharyngeal wall and piriform fossae; 6 = residue in the valleculae and posterior pharyngeal wall and piriform sinus.40
3.3. Associations between demographic, critical care, and swallowing data
No association was identified between participant age and any swallowing outcomes reported, nor was there an association between duration of intubation or mechanical ventilation and duration to commence dysphagia rehabilitation. There were, however, significant positive linear associations identified between duration of intubation, mechanical ventilation, hospital and ICU LOS, the duration to speech pathology assessment, commencing oral intake, dysphagia recovery, and enteral feeding. Conversely, a negative linear association was identified between dysphagia severity and duration of intubation, mechanical ventilation, and hospital and ICU LOS. This is reflective of a lower FOIS score (indicating more severe swallowing impairment) being associated with longer duration of medical interventions and LOS. All swallow association results are summarised in Table 3 .
Table 3.
Association between critical care and swallowing data (n = 27).
| Swallowing variable | Median (IQR) | Age r (p-value) | ETT duration r (p-value) | MV duration r (p-value) | ICU LOS r (p-value) | Hospital LOS r (p-value) |
|---|---|---|---|---|---|---|
| Duration to SP assessment | 29 (23) | 0.022 (0.914) | 0.541 (0.004∗∗) | 0.840 (0.000∗∗) | 0.875 (0.000∗∗) | 0.727 (0.000∗∗) |
| Duration to face-to-face SP assessment | 33 (18) | 0.051 (0.799) | 0.733 (0.000∗∗) | 0.935 (0.000∗∗) | 0.930 (0.000∗∗) | 0.745 (0.000∗∗) |
| Dysphagia severity | 5 (4) | −0.169 (0.399) | −0.572 (0.002∗∗) | −0.637 (0.000∗∗) | −0.602 (0.001∗∗) | −0.633 (0.000∗∗) |
| Duration to initiate oral feeding | 38.5 (31.25) | 0.096 (0.642) | 0.454 (0.020∗) | 0.657 (0.000∗∗) | 0.867 (0.000∗∗) | 0.957 (0.000∗∗) |
| Duration to commencing dysphagia rehabilitation | 39 (17) | 0.288 (0.452) | −0.291 (0.447) | 0.156 (0.156) | 0.980 (0.000∗∗) | 0.872 (0.002∗∗) |
| Duration to resolution of dysphagia | 44 (29) | 0.109 (0.630) | 0.626 (0.002∗∗) | 0.586 (0.004∗∗) | 0.605 (0.003∗∗) | 0.918 (0.000∗∗) |
| Duration of enteral feeding | 40 (37) | 0.184 (0.378) | 0.451 (0.024∗) | 0.669 (0.000∗∗) | 0.872 (0.000∗∗) | 0.962 (0.000∗∗) |
ETT, endotracheal tube; LOS, length of stay; MV, mechanical ventilation; SP, speech pathology.
∗sig at 0.05; ∗∗ sig at 0.01.
More than half of participants within the cohort required tracheostomy as part of their ICU treatment (n = 16). Whilst the presence of tracheostomy was not associated with whether dysphagia recovery was achieved (p = 0.06), nor the duration to commence dysphagia rehabilitation (Z = −2.049, p = 0.056), it was associated with severity of dysphagia (Z = −2.100, p = 0.05), duration to initiation of oral feeding (Z = −2.934, p = 0.002), duration to resolution of dysphagia (Z = −3.056, p = 0.001) and duration of enteral feeding (Z = −3.112, p = 0.001).
4. Discussion
Dysphagia prevalence in the patient with COVID-19 requiring ICU treatment and referred to speech pathology is high. ICU-acquired weakness and delirium also commonly occur in these patients and are likely the sequelae of critical illness. Severity of dysphagia is found to often be profound on the initial assessment and is associated with increased duration of endotracheal intubation, mechanical ventilation, and ICU and hospital LOS. Prognosis for dysphagia recovery during the acute admission is relatively good with most patients discharged home or transferred to inpatient rehabilitation on separation from the acute care facility.
Participant demographic and critical care data within the current cohort were overall consistent with those existing published studies who also examined patients referred to speech pathology. Similar characteristics included elevated rates of delirium30 , 33 and mean age,30 , 32 although average duration of endotracheal intubation in the current study was longer (19 days compared to 14 days).32 , 33 Despite the cohort being much smaller than those of our international colleagues, dysphagia prevalence was comparable to those recently published studies.30 , 32 , 33 Furthermore, our observed severity of dysphagia was also comparable to the data presented by Dawson et al.30 and Regan et al.,32 although this differed from the findings presented by Lima et al.,31 who found a lesser degree of dysphagia in patients with COVID-19 than in patients with other critical illness. Optimistically and similar to existing literature,[30], [31], [32], [33] we observed encouraging rates of dysphagia resolution at the point of discharge from the acute care facility. Interestingly however, whilst our international colleagues have commented on adverse neurological comorbidities as the cause for persistent dysphagia,33 secondary neurological pathology was fortunately not observed in our cohort.
Penetration–aspiration results on instrumental assessment have only been documented in one other published study to date. Similar to the current study, Sandblom et al.41 observed worse PAS scores for fluids than for solids. Furthermore, impaired pharyngeal clearance41 (defined by the BRS in the current study) also appeared to be a feature consistent between to the two studies. The cause for worse PAS and BRS scores is postulated to be commensurate with the profile of swallowing impairment in critical illness which is further explored in the following part of the article.
We identified several associations between critical care data and swallowing outcomes. Specifically, presence and duration of endotracheal intubation, presence of tracheostomy, duration of mechanical ventilation, and ICU and hospital LOS were associated with poorer swallowing outcomes and increased periods of enteral feeding. These results are analogous with those already published.30 , 32 , 33 Contrary to our findings however, Regan et al.32 identified that age was also associated with poorer swallowing outcomes where we did not. Moreover, Regan et al.32 identified two other variables as predictors for dysphagia: proning and history of respiratory disease. Proning was not captured in the present study and history of respiratory disease, although present in some participants, was not observed at a high frequency. Finally, the duration to initiate oral intake calculated from the point of intubation could be compared with the study by Dawson et al. (2020)30 only, with an apparent increased delay in commencing oral intake for participants in the present study (40 days compared to 22 days).
Postulating the pathophysiology behind the mechanism of dysphagia specifically in the patient with COVID-19, there are several aspects that are consistent with existing literature on dysphagia in critical illness.29 Existing evidence indicates that dysphagia in critical illness may manifest as delay in swallow onset, impaired airway closure, high rates of laryngeal penetration and aspiration, and diminished efficiency and effectiveness of cough response with worse swallow outcomes observed when various forms of laryngeal injury were copresent.16 Further to this, presence of the endotracheal tube itself has been associated with a reduction in base of tongue muscle bulk and base of tongue strength, in addition to diminished laryngeal sensation, vocal cord damage and compression of the recurrent laryngeal nerve by the endotracheal tube cuff.14 In addition to critical illness, other co-occurring medical conditions including neurological8 , 42 and respiratory disease26 are known to further complicate the swallowing profile. Specifically, diseases of the respiratory system such as acute respiratory distress syndrome (ARDS) can disrupt normal coordination of the respiratory–swallow cycle.26 Although there is no evidence regarding breath–swallow synchrony in COVID-19, this is another factor that needs to be considered. To date, only one very recent study has documented the pathophysiology of dysphagia in the patient with COVID-19, suggesting in accordance with FEES outcomes that reduced pharyngeal muscle strength, impaired airway closure, and laryngeal sensation were at the core of dysphagia in this population.41 Interestingly however, both Lima et al.31 and Archer et al.33 describe that dysphagia resolves more rapidly in patients with COVID-19 than in non-COVID critically ill patients, highlighting the importance of assessment and compensatory strategies and suggesting that weakness alone may not be the primary origin for dysphagia. This indicates that more work is required to fully understand the mechanisms underpinning dysphagia in the patient with COVID-19. Further to this, knowledge gained will also guide methods of multidisciplinary team driven rehabilitation, to optimise and expedite dysphagia recovery which is clearly indicated in a proportion of these patients.43
This study is the first of its kind across Australia to describe the clinical profile of dysphagia in ICU patients with COVID-19, and the clinical applicability of results is strengthened by its multisite methodology and use of clearly defined outcome measures. There are, however, several limitations to the execution and interpretation of this study. First, routine instrumental assessment and specifically FEES, which is accessible in many tertiary ICUs and well accepted to be the gold standard for the provision of swallow kinematic information in ICU patients, was lacking. This was due to the universally and internationally accepted recommendations to avoid aerosol-generating procedures in the efforts to promote staff safety by minimising viral transmission.44 Other studies to date have also cited this aspect as a limitation to investigating the nature of dysphagia in the COVID-19–positive patient.30 , 32 , 33 Second, the absence of detail regarding dysphagia rehabilitation strategies has not allowed for in-depth understanding of the swallowing rehabilitation needs of this population. Third, the small sample size, although fortunate from a population health perspective, suggests that these results should be interpreted with caution. Fourth, the sample was limited by the inclusion criteria that not all those patients admitted to the ICU were seen by a speech pathologist, only those who were referred resulting in a possible under-reporting of dysphagia prevalence. However, the inclusion of only those who were referred to speech pathology for assessment may also lead to an overestimation of the severity of dysphagia in the cohort, as those with only mild issues may not have been referred. It would be challenging to assert that patients with COVID-19 undergo routine speech pathology examination, again owing to the highly transmissible nature of this virus. Furthermore, differences in individual site practices and infection control protocols may have resulted in delays in the provision of care. Finally, very recent studies from international colleagues indicate that laryngeal pathology may be an issue in the postextubation and tracheostomy patient with COVID-19.32 , 41 , 45 , 46 As such, future studies examining the ICU COVID-19 patient, detailing outcomes specific to voice, laryngeal pathology, as well as dysphagia rehabilitation are recommended.
5. Conclusion
Consistent with international evidence, diagnosis of COVID-19 in the current study had considerable impact on patients' swallowing function across NSW ICUs, with high prevalence and severity dysphagia apparent on initial assessment. Dysphagia was associated with increased duration of intubation, mechanical ventilation, and hospital and ICU LOS, with one-third requiring active dysphagia rehabilitation. Although many patients in the present study recovered within an acceptable timeframe, some experienced persistent severe dysphagia and a protracted recovery with dependence on enteral nutrition. Further work is required to investigate the pathophysiology underpinning dysphagia in this population and the appropriate multidisciplinary rehabilitative strategies that should be applied to optimise and expedite swallow recovery.
Conflict of interest
No conflicts of interest to declare.
CRediT authorship contribution statement
NA Clayton: Conceptualisation, Methodology, Formal analysis, Investigation, Writing - original draft, Visualisation; E Walker: Conceptualisation, Methodology, Investigation, Writing - review & editing, Visualisation; A Freeman–Sanderson: Methodology, Formal analysis, Writing - review & editing, Visualisation.
Acknowledgements
This work would not have been possible without the commitment and assistance from the principal investigators across all 17 NSW Health ICU facilities for the purposes of data collection. The authors convey special thanks to Natasha Absalom (Wollongong Hospital), Rebecca Black (St Vincents Hospital), Sara Bolt (Liverpool Hospital), Nicola Clayton (Concord Repatriation General Hospital), Georgia Donovan (Blacktown-Mount Druitt Hospital), Klint Goers (Nepean Hospital), Elise Hamilton–Foster (Westmead Hospital), Karen Kostal (Northern NSW LHD), Julia Maclean (St George Hospital), Penny Mogg (Prince of Wales Hospital), Deanna Rolfe (Hornsby Hospital), Elizabeth Walker (Royal Prince Alfred Hospital), and Alyssa Walter (Western NSW LHD).
References
- 1.Brodsky M.B., Gellar J.E., Dinglas V.D., Colantuoni E., Mendez-Tellez P.A., Shanholtz C., et al. Duration of oral endotracheal intubation is associated with dysphagia symptoms in acute lung injury patients. J Crit Care. 2014;29(4):574–579. doi: 10.1016/j.jcrc.2014.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Skoretz S.A., Yau T.M., Ivanov J., Granton J.T., Martino R. Dysphagia and associated risk factors following extubation in cardiovascular surgical patients. Dysphagia. 2014;29(6):647–654. doi: 10.1007/s00455-014-9555-4. [DOI] [PubMed] [Google Scholar]
- 3.Bordon A., Bokhari R., Sperry J., Testa D., 4th, Feinstein A., Ghaemmaghami V. Swallowing dysfunction after prolonged intubation: analysis of risk factors in trauma patients. Am J Surg. 2011;202(6):679–682. doi: 10.1016/j.amjsurg.2011.06.030. discussion 682-3. [DOI] [PubMed] [Google Scholar]
- 4.Skoretz S.A., Flowers H.L., Martino R. The incidence of dysphagia following endotracheal intubation: a systematic review. Chest. 2010;137(3):665–673. doi: 10.1378/chest.09-1823. [DOI] [PubMed] [Google Scholar]
- 5.Kim M.J., Park Y.H., Park Y.S., Song Y.H. Associations between prolonged intubation and developing post-extubation dysphagia and aspiration pneumonia in non-neurologic critically ill patients. Ann Rehabil Med. 2015;39(5):763–771. doi: 10.5535/arm.2015.39.5.763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Barker J., Martino R., Reichardt B., Hickey E.J., Ralph-Edwards A. Incidence and impact of dysphagia in patients receiving prolonged endotracheal intubation after cardiac surgery. Can J Surg. 2009;52(2):119–124. [PMC free article] [PubMed] [Google Scholar]
- 7.Macht M., King C.J., Wimbish T., Clark B.J., Benson A.B., Burnham E.L., et al. Post extubation dysphagia is persistent and associated with poor outcomes in survivors of critical illness. Crit Care. 2011;15(5):R231. doi: 10.1186/cc10472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Macht M., King C.J., Wimbish T., Clark B.J., Benson A.B., Burnham E.L., et al. Post-extubation dysphagia is associated with longer hospitalization in survivors of critical illness with neurologic impairment. Crit Care. 2013;17(3):R119. doi: 10.1186/cc12791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Malandraki G.A., Markaki V., Georgopoulos V.C., Psychogios L., Nanas S. Postextubation dysphagia in critical patients: a first report from the largest step-down intensive care unit in Greece. Am J Speech Lang Pathol. 2016;25(2):150–156. doi: 10.1044/2015_AJSLP-14-0069. 1. [DOI] [PubMed] [Google Scholar]
- 10.Skoretz S.A., Riopelle S.J., Wellman L., Dawson C. Investigating swallowing and tracheostomy following critical illness: a scoping review. Crit Care Med. 2020;48(2):e141–e151. doi: 10.1097/CCM.0000000000004098. [DOI] [PubMed] [Google Scholar]
- 11.Tsai M.H., Ku S.C., Wang T.G., Hsiao T.Y., Lee J.J., Chan D.C., et al. Swallowing dysfunction following endotracheal intubation: age matters. Medicine. 2016;95(24) doi: 10.1097/MD.0000000000003871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Clayton N.A., Ward E.C., Nicholls C., Giannone R., Skylas K., Maitz P.K. The addition of respiratory muscle strength training to facilitate swallow and pulmonary rehabilitation following massive tissue loss and severe deconditioning: a case series. Aust Crit Care. 2021;23(21):S1036–S7314. doi: 10.1016/j.aucc.2021.03.003. 00041-2. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 13.Ghannouchi I., Speyer R., Doma K., Cordier R., Verin E. Swallowing function and chronic respiratory diseases: systematic review. Respir Med. 2016;117:54–64. doi: 10.1016/j.rmed.2016.05.024. [DOI] [PubMed] [Google Scholar]
- 14.Brodsky M.B., Levy M.J., Jedlanek E., Pandian V., Blackford B., Price C., et al. Laryngeal injury and upper airway symptoms after oral endotracheal intubation with mechanical ventilation during critical care: a systematic review. Crit Care Med. 2018;46(12):2010–2017. doi: 10.1097/CCM.0000000000003368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Clayton N.A., Carnaby-Mann G.D., Peters M.J., Ing A.J. The effect of chronic obstructive pulmonary disease on laryngopharyngeal sensitivity. Ear Nose Throat J. 2012;91(9):370–372. 374. [PubMed] [Google Scholar]
- 16.Scheel R., Pisegna J.M., McNally E., Noordzij J.P., Langmore S.E. Endoscopic assessment of swallowing after prolonged intubation in the ICU setting. Anals of otology, Rhinology & Laryngology. 2016;125(1):43–52. doi: 10.1177/0003489415596755. [DOI] [PubMed] [Google Scholar]
- 17.Clayton N.A., Carnaby-Mann G.D., Peters M.J., Ing A.J. Impaired laryngopharyngeal sensitivity in patients with chronic obstructive pulmonary disease: the association swallow function. Int J Speech Lang Pathol. 2014;16(6):615–623. doi: 10.3109/17549507.2014.882987. [DOI] [PubMed] [Google Scholar]
- 18.Chaves R.D., Carvalho C.R.F., Cukier A., Stelmach R., Andrade C.R.F. Symptoms of dysphagia in patients with COPD. J Bras Pneumol. 2011;37(2):176–183. doi: 10.1590/s1806-37132011000200007. [DOI] [PubMed] [Google Scholar]
- 19.Coelho C.A. Preliminary findings on the nature of dysphagia in patients with chronic obstructive pulmonary disease. Dysphagia. 1987;2(1):28–31. doi: 10.1007/BF02406975. [DOI] [PubMed] [Google Scholar]
- 20.Cvejic L., Harding R., Churchward T., Turton A., Finlay P., Massey D., et al. Laryngeal penetration and aspiration in individuals with stable COPD. Respirology. 2011;16(2):269–275. doi: 10.1111/j.1440-1843.2010.01875.x. [DOI] [PubMed] [Google Scholar]
- 21.Reid K.L. University of Sydney; Sydney, Australia: 1998. The role of swallowing dysfunction in acute exacerbations of chronic airflow limitation. Unpublished masters thesis. [Google Scholar]
- 22.Shaker R., Qun L., Ren J., Townsend W.F., Dodds W.J., Martin B.J., et al. Coordination of deglutition and phases of respiration: effect of aging, tachypnea, bolus volume, and chronic obstructive pulmonary disease. Am J Physiol. 1992;263(5 Pt 1):G750–G755. doi: 10.1152/ajpgi.1992.263.5.G750. [DOI] [PubMed] [Google Scholar]
- 23.Hafner G., Neuhuber A., Hirtenfelder S., Schmedler B., Eckel H.E. Fiberoptic endoscopic evaluation of swallowing in intensive care unit patients. Eur Arch Oto-Rhino-Laryngol. 2008;265:441–446. doi: 10.1007/s00405-007-0507-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hales P.A., Drinnan M.J., Wilson J.A. The added value of fibreoptic endoscopic evaluation of swallowing in tracheostomy weaning. Clin Otolaryngol. 2008;33:319–324. doi: 10.1111/j.1749-4486.2008.01757.x. [DOI] [PubMed] [Google Scholar]
- 25.Ajemian M.S., Nirmul G.B., Anderson M.T., Zirlen D.M., Kwasnik E.M. Routine fiberoptic endoscopic evaluation of swallowing following prolonged intubation - implications for management. Arch Surg. 2001;136:434–437. doi: 10.1001/archsurg.136.4.434. [DOI] [PubMed] [Google Scholar]
- 26.Borders J.C., Fink D., Levitt J.E., McKeehan J., McNally E., Rubio A., et al. Relationship between laryngeal sensation, length of intubation, and aspiration in patients with acute respiratory failure. Dysphagia. 2019;34:521–528. doi: 10.1007/s00455-019-09980-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cardinal L.A., Freeman-Sanderson A., Togher L. The speech pathology workforce in intensive care units: results from a national survey. Aust Crit Care. 2020;33:250–258. doi: 10.1016/j.aucc.2020.02.003. [DOI] [PubMed] [Google Scholar]
- 28.Dziewas R., Warnecke T., Zürcher P., Schefold J.C. Dysphagia in COVID-19 –multilevel damage to the swallowing network? Eur J Neurol. Eur J Neurol. 2020;27(9):e46–e47. doi: 10.1111/ene.14367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Frajkova Z., Tedla M., Tedlova E., Suchankova M., Geneid A. Postintubation dysphagia during COVID-19 outbreak-contemporary review. Dysphagia. 2020;35(4):549–557. doi: 10.1007/s00455-020-10139-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dawson C., Capewell R., Ellis S., Matthews S., Adamson S., Wood M., et al. Dysphagia presentation and management following COVID-19: an acute care tertiary centre experience. J Laryngol Otol. 2020;10:1–6. doi: 10.1017/S0022215120002443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lima M.S., Chiarion Sassi F., Medeiros G.C., Ritto A.P., de Andrade C.R.F. Preliminary results of a clinical study to evaluate the performance and safety of swallowing in critical patients with COVID-19. Clinics. 2020;75 doi: 10.6061/clinics/2020/e2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Regan J., Walshe M., Lavan S., Horan E., Gillivan Murphy P., Healy A., et al. Post-extubation dysphagia and dysphonia amongst adults with COVID-19 in the Republic of Ireland: a prospective multi-site observational cohort study. Clin Otolaryngol. 2021 doi: 10.1111/coa.13832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Archer S.K., Iezzi C.M., Gilpin L. Swallowing and voice outcomes in patients hospitalized with COVID-19: an observational cohort study. Arch Phys Med Rehabil. 2021;102(6):1084–1090. doi: 10.1016/j.apmr.2021.01.063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.von Elm E., Altman D.G., Egger M., Pocock S.J., Gøtzsche P.C., Vandenbroucke J.P., STROBE Initiative The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol. 2008;61(4):344–349. doi: 10.1016/j.jclinepi.2007.11.008. [DOI] [PubMed] [Google Scholar]
- 35.Knaus W.A., Draper E.A., Wagner D.P., Zimmerman J.E. Apache II: a severity of disease classification system. Crit Care Med. 1985;13(10):818–829. [PubMed] [Google Scholar]
- 36.Crary M.A., Mann G.D., Groher M.E. Initial psychometric assessment of a functional oral intake scale for dysphagia in stroke patients. Arch Phys Med Rehabil. 2005;86(8):1516–1520. doi: 10.1016/j.apmr.2004.11.049. [DOI] [PubMed] [Google Scholar]
- 37.The Speech Pathology Association of Australia . 2012. Dysphagia clinical guidelines.https://www.speechpathologyaustralia.org.au/SPAweb/Members/Clinical_Guidelines/spaweb/Members/Clinical_Guidelines/Clinical_Guidelines.aspx?hkey=f66634e4-825a-4f1a-910d-644553f59140 Retrieved from. [Google Scholar]
- 38.Harris P.A., Taylor R., Thielke R., Payne J., Gonzalez N., Conde J.G. Research electronic data capture (REDCap) a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inf. 2009;42(2):377–381. doi: 10.1016/j.jbi.2008.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rosenbek J.C., Robbins J.A., Roecker E.B., Coyle J.L., Wood J.L. A penetration-aspiration scale. Dysphagia. 1996;11(2):93–98. doi: 10.1007/BF00417897. [DOI] [PubMed] [Google Scholar]
- 40.Rommel N., Borgers C., Van Beckevoort D., Goeleven A., Dejaeger E. Omari TI. Bolus residue scale: an easy-to-use and reliable videofluoroscopic analysis tool to score bolus residue in patients with dysphagia. Int J Otolaryngol. 2015;2015:1–7. doi: 10.1155/2015/780197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Osbeck Sandblom H., Dotevall H., Svennerholm K., Tuomi L., Finizia C. Characterization of dysphagia and laryngeal findings in COVID-19 patients treated in the ICU-An observational clinical study. PLoS One. 2021;16(6) doi: 10.1371/journal.pone.0252347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zuercher P., Schenk N.V., Moret C., Berger D., Abegglen R., Schefold J.C. Risk factors for dysphagia in ICU patients after invasive mechanical ventilation. Chest. 2020;158:1983–1991. doi: 10.1016/j.chest.2020.05.576. [DOI] [PubMed] [Google Scholar]
- 43.Parker A.M., Brigham E., Connolly B., McPeake J., Agranovich A.V., Kenes M.T., et al. Addressing the post-acute sequelae of SARS-CoV-2 infection: a multidisciplinary model of care. Lancet Respir Med. 2021;9(11):1328–1341. doi: 10.1016/S2213-2600(21)00385-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Clinical Excellence Commission . 2021. COVID-19 infection, prevention and control manual.https://www.cec.health.nsw.gov.au/__data/assets/pdf_file/0018/644004/COVID-19-IPAC-manual.pdf Retrieved from. (August 2021) [Google Scholar]
- 45.McGrath B.A., Wallace S., Goswamy J. Laryngeal oedema associated with COVID-19 complicating airway management. Anaesthesia. 2020;75(7):972. doi: 10.1111/anae.15092. [DOI] [PubMed] [Google Scholar]
- 46.Rouhani M.J., Clunie G., Thong G., Lovell L., Roe J., Ashcroft M., et al. A prospective study of voice, swallow, and airway outcomes following tracheostomy for COVID-19. Laryngoscope. 2021;131:E1918–E1925. doi: 10.1002/lary.29346. [DOI] [PubMed] [Google Scholar]