Abstract
Hybrid polymer/lipid vesicles are self-assembled structures that have been the subject of an increasing number of studies in recent years. They are particularly promising tools in the development of cell membrane models because they offer the possibility to fine-tune their membrane structure by adjusting the distribution of components (presence or absence of “raft-like” lipid domains), which is of prime importance to control their membrane properties. Line tension in multiphase membranes is known to be a key parameter on membrane structuration, but remains unexplored, either experimentally or by computer modeling for hybrid polymer/lipid vesicles. In this study, we were able to measure the line tension on different budded hybrid vesicles, using a micropipette aspiration technique, and show the influence of the molar mass and the architecture of block copolymers on line tension and its consequences for membrane structuration.
Graphical abstract

Significance
Hybrid polymer/lipid vesicles with a membrane composed of phospholipids and amphiphilic copolymers are extremely promising structures for various fields of applications (drug delivery, development of nano-/micro-reactors, artificial cells, etc). For all these applications, a perfect control and knowledge of the membrane structure is essential. The line tension (energy per length unit) at the boundaries of polymer and lipid is a key parameter governing the membrane structure. However, this parameter is so far totally unknown for these systems, hindering the optimization of their membrane properties. We report here the first experimental measurements of this parameter and clarify the role of the copolymer molar mass and architecture on the line tension and thus the membrane structure.
Introduction
Lipids are the major components of the cell membrane. In a bottom-up approach to understanding cell function and behavior, phospholipids were the first synthetic molecules used to develop vesicles as a basic model of the cell membrane. Although more complex structures have been developed since then, for example, by inserting membrane proteins or using different phospholipids to elaborate the membrane, these advanced structures remain short lived, not very stable, and without a natural mechanism of regeneration. In the late 1990s, polymersomes obtained by self-assembly of amphiphilic copolymers emerged as a potential replacement for phospholipids in the development of synthetic cells, due to their higher membrane toughness, chemical versatility and resistance (1). In particular, they also allow the insertion of membrane proteins and grant them an extended functional lifetime (2, 3, 4). More recently, hybrid polymer/lipid membranes have attracted great interest, because they can be considered as advanced vesicular structures compared with their forerunners (liposomes and polymersomes) as they harness the advantages of both components. The modulation of membrane properties between those of pure liposomes and polymersomes has indeed been observed, and, generally speaking, these systems are of great interest to go further in the reproduction of dynamic biological phenomena. In particular, hybrid polymer/lipid systems tend to promote membrane fission and fusion for more efficient molecular trafficking (2). In most cases, the phenomenon results from the presence of a phase separation in the membrane, leading to polymer/lipid boundaries that exhibit a line tension, unquantified until now for such systems.
Line tension is defined as energy per unit length at boundaries in multiphase planar systems. It can be considered as the 1-dimensional analogue of the interfacial tension in 2-dimensional coexisting phases. The concept of line tension has been introduced to understand complex phenomena such as lateral phase separation, fusion, budding, and fission in biological membranes (5), which are essential for biological functions, such as the production of transport vesicles, the presence of signaling domains in the membrane (existence of lipid raft), and so on. Research in this area is particularly driven by the lively debate surrounding functionally important lipid/protein membrane inhomogeneities in living cells (6,7), and some applications such as the development of biomimetic bilayer sensors. Line tension has been shown to be one of the key parameters to modulate the shape, size, and dynamics of the domains in a multiphase lipid bilayer membrane (8,9). Its experimental determination is tricky and relatively few values are reported in the literature for lipid bilayers. Baumgart et al. (10) determined the line tension through the shape of the domains observed by microscopy using a shape theory previously developed by Jülicher and Lipowsky (11). Esposito et al. (12) calculated the line tension from model boundary fluctuation in vesicles using domain flicker spectroscopy. Honerkamp-Smith et al. (13) used the same approach to determine line tension in DPPC/diPhyPc/cholesterol membranes. Micropipette aspiration has also been used to measure line tension in giant unilamellar vesicles with a liquid-ordered and a liquid-disordered phase (14). Overall, the typical value is about a few piconewtons for purely lipidic bilayers and can vary with the composition of the membrane. So far, these variations have not been rationalized. However, it has been shown that the line tension tends to decrease when the miscibility critical point is reached and increases when the thickness mismatch between the lipid phases increases (15).
Hybrid polymer/lipid vesicles have emerged as self-assembled structures that spark an increasing interest from different scientific communities as promising objects for numerous application areas such as controlled drug delivery, the development of bioinspired micro-/nano-reactors, and functional membranes for artificial cells (16,17). However, despite an increasing number of studies, there is a lack of a systematic approach that could decipher the molecular parameters necessary to control their membrane structuration and reach the desired membrane properties for a given application. Line tension has been discussed in direct analogy with a multicomponent lipid bilayer as a key parameter to control membrane structure. However, the experimental values of the line tension have been unknown until now. The determination of the line tension in these hybrid vesicles is of prime importance to go further in the development of more complex and functional artificial cells. In this work, we designed different hybrid polymer/lipid vesicles from a mixture of 1-palmitoyl-2-oleoyl-glycero-3-phosphocholine (POPC) and poly(dimethylsiloxane)-b-poly(ethylene oxide) diblock or triblock copolymers (PDMS-b-PEO or PEO-b-PDMS-b-PEO), in a given lipid composition range. Although these PDMS-based copolymers have already been used for the development of hybrid vesicles, they are also of great interest in synthetic biology for the development of artificial cells; they have been shown to allow the insertion of a membrane protein in a functional state with an extended functional lifetime. Giant hybrid unilamellar vesicles (GHUVs) obtained with triblock copolymers present stable budded lipid domains, as demonstrated in a previous study (18). These budded vesicles were also obtained with diblock copolymers (19), allowing line tension measurement by the micropipette technique. We will show and discuss the importance of block copolymer architecture and hydrophobic length mismatch at the polymer/lipid boundaries on the line tension and the consequences in terms of membrane structuration.
Materials and methods
All the copolymers used in this study were synthesized and fully characterized in previous work (18,20). The molecular characteristics of the copolymers as well as the membrane thicknesses of the polymersomes are indicated in Table 1. POPC, 1,2-dioleoyl-sn-glycero-3-phosphocholine (DOPC), and 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine-N-(lissamine rhodamine B sulfonyl) (DOPE-Rhod) were purchased from Sigma Aldrich and used without further purification.
Table 1.
Molecular characteristics of the copolymers used in this study
| Copolymer |
1H NMR in CDCl3 |
SEC in THF |
SANS |
|||
|---|---|---|---|---|---|---|
| Mn PDMS (g.mol−1) | Mn PEO (g.mol−1) | Mn copolymer (g.mol−1) | Hydrophilic fraction (%) | Dispersity Đ | Membrane Thicknessa (nm) | |
| PEO8-b-PDMS22-b-PEO8 (18) | 1628 | 350 | 2668 | 30 | 1.18 | 5.4 ± 0.4 |
| PDMS23-b-PEO13 (20) | 1700 | 600 | 2500 | 26 | 1.15 | 7.2 ± 0.8 |
| PDMS27-b-PEO17 (20) | 2000 | 700 | 2900 | 26 | 1.11 | 8.3 ± 1.1 |
GHUV preparation
Giant unilamellar vesicles were prepared at room temperature using the electro-formation protocol reported by Angelova and Dimitrov (22). Briefly, solutions containing the appropriate amount of copolymer and POPC were solubilized in chloroform at 1 mg · mL−1. Then, 50 μL of the solution were spread on indium tin oxide glass plates. Traces of chloroform were removed under vacuum over at least 3 h. Next, the indium tin oxide glass plates were sealed together to form the electro-formation chamber, connected to an AC voltage and filled with a 100-mM sucrose solution. A sinusoidal tension (2 V, 10 Hz) was applied during 75 min for all systems used. For the identification of polymer and lipid phases in GHUVs, fluorescent probes were used: PDMS-nitrobenzoxadiazole (1 mol.%) for polymer phases (synthesis protocol is available in (20)) and DOPE-Rhod at 0.1 mol.% for lipid phases.
The micropipette aspiration experiment
The micropipette aspiration experiment was carried out using a Leica TCS SP5 (Leica Microsystems CMS GmbH, Mannheim, Germany) inverted confocal microscope (DMI6000) equipped with an ×63 apochromatic water immersion objective with an NA of 1.2 (Zeiss, Jena, Germany). Micropipettes were obtained by stretching Borosilicate capillaries (1 mm outer diameter, 0.58 mm inner diameter) from WPI (Worcester, MA), using a pipette puller (Sutter Instrument P-97, Novato, CA). The pulled pipettes were then forged into the desired diameter using a Narishige MF-900 micro-forge (London, UK). The micropipettes were coated with bovine serum albumin to prevent vesicle adhesion. Giant unilamellar vesicles were injected and allowed to sediment in a glucose meniscus formed between 2 glass slides glued to a home-made aluminum microscope stage. Vesicle tension was controlled using a home-made hydraulic water-tight setup. The micropipette was controlled using a micromanipulator (Eppendorf, Patchman NP2, Montesson, France).
The suction pressure exerted over the membrane can be calculated from:
| (1) |
where ρ is the water density, g is the gravitational acceleration, h is the position of the water tank and h0 is the initial position where the pressure is zero.
The membrane tension was classically calculated using the Laplace equation:
| (2) |
where RP and RV are, respectively, the micropipette and the vesicle radii (outside the micropipette), and DP is the suction pressure.
The relative area change of the membrane α is defined as:
| (3) |
where A0 is the membrane area of the vesicle at the lower suction pressure. α can be estimated from the increase in projection length ΔL of the vesicle inside the capillary tip according to the following:
| (4) |
The surface area strain can be linked to the membrane tension through the following equation:
| (5) |
where k is the Boltzmann constant, T the temperature, and Kb and Ka, respectively the bending and stretching modulus. The Kb can be extracted from the slope of the curve Ln(σ) versus α, at a low tension regime (typically up to α = 1%), where the surface area increase is almost entirely owing to damping of thermal shape fluctuations, in other words, bending undulations in the bilayer (23, 24, 25, 26, 27, 28).
Results and discussion
In a previous study, we established an apparent phase diagram of GHUV membrane structuration obtained from the mixture of POPC and triblock copolymer PEO8-b-PDMS22-b-PEO8 (18). In a given compositional range, stable budded vesicles were obtained, with each hemisphere being formed by either the polymer phase or the lipid phase. Fig. 1 illustrates budded vesicles under micropipette suction.
Figure 1.
Example of line tension measurement on GHUVs made from PDMS23-b-PEO13/POPC at different suction pressure (values indicated in the top right of each picture). Lipid phases are tagged in red while polymer phases are tagged in green.
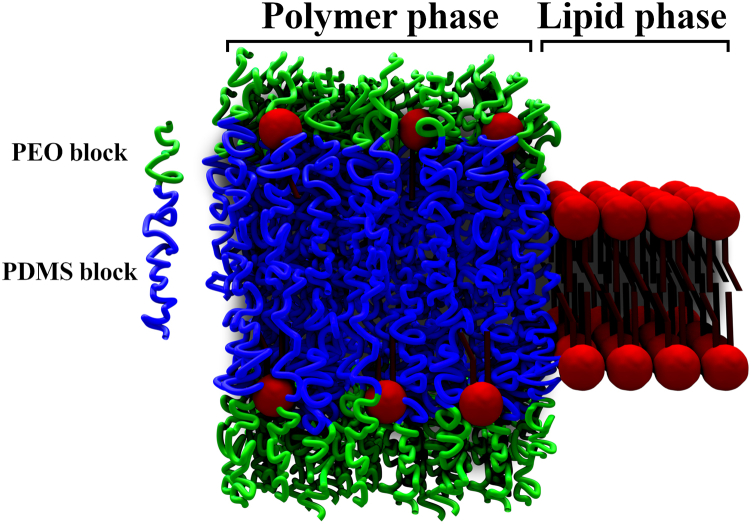
This morphology reflects a budding-fission process that was stopped through a balance between the boundary energy and the bending cost of energy. This phenomenon was also observed with diblock copolymers PDMS27-b-PEO17 and PDMS23-b-PEO13 when forming GHUVs (19). This particular shape of GHUVs can provide insight into the boundary energy between the polymer and the lipid phases, also known as line tension. In Fig. 2, a schematic of the hybrid polymer/lipid membrane with lateral phase separation is shown for clarification.
Figure 2.
Schematic view of the hybrid polymer/lipid membrane with a lateral phase separation.
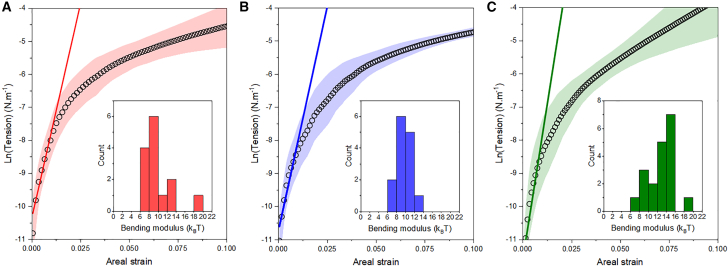
We used a model developed by Baumgart et al. (14) to extract the line tension from micropipette measurements performed on GHUVs made from diblock or triblock copolymers. First, the bending moduli Kb of the different polymersomes were evaluated to verify whether the bending energy can indeed be neglected compared to the boundary energy. The corresponding experimental curves are shown in Fig. 3. We obtained for the polymersomes made from the diblock copolymers PDMS27-b-PEO17 and PDMS23-b-PEO13 bending moduli of 10.0 ± 1.6 kT and 10.1 ± 3.4 kT, respectively. Bending modulus of polymersomes made from triblock copolymers PEO8-b-PDMS22-b-PEO8 was slightly higher, with an average value of 12.7 ± 2.9 kT. Because the measurement of bending moduli and line tension involves the use of very low suction pressure, we verified the accuracy of our experimental setup and procedure, by measuring bending modulus of giant unilamellar vesicles made from DOPC (data available in supporting material). We obtained a bending modulus of 10.9 ± 0.8 kT, in reasonable agreement with the value of 11.7 ± 1.5 kT obtained in literature (29,30) by micropipette aspiration for similar sucrose concentration. We can, therefore, consider our data to be reliable.
Figure 3.
Average measurements (black circles) and standard deviation (colored areas) of the membrane tension as a function of areal strain for the polymersomes obtained from PDMS23-b-PEO13 (A), PDMS27-b-PEO17, (B) and PEO8-b-PDMS22-b-PEO8 (C). Bending moduli are extracted from the slope of the curve (linear regression) in the low-tension regime.
The values obtained for the 3 copolymers are in the same range than DOPC (26,27) and lower than what is observed for POPC (26,28). They are also significantly lower than the bending moduli reported for polymersomes made from coil-coil block copolymers (23), with comparable membrane thicknesses (∼7–9 nm). As the PDMS chains in the membrane has also been shown to exhibit a coil conformation (membrane thickness scales with M∼0.52) (20), this may be related to the high fluidity for such molar mass owing to their low Tg (∼−120°C) and high critical entanglement molar mass (∼30,000 g · mol−1). Similar values of Kb were observed for both diblock copolymers, despite a slight but noticeable difference in membrane thickness. The relatively high uncertainty of such measurements probably prevent to see the expected effect of the membrane thickness. The variation in length of the hydrophobic block is not very high (only 4 units between the 2 diblock copolymers). As Kb scales as d2 (23), and d scales as M∼0.52, Kb should scale as M∼1.3. This may be the case, but we cannot confirm it regarding the size of the error bars. However, it is interesting to note that, in the literature, the Kb of large unilamellar vesicles of PDMS60-b-PMOXA21 has been measured at 25 kT (31). Considering the value of ∼10 kT for polymersomes made of PDMS27, a reasonable agreement is found with the literature value. Indeed, assuming a scaling law of Kb ∼ M1.3, a value of 28 kT should be obtained for the PDMS60, which is very close to what has been observed.
Line tension was determined by measuring the geometric factor of the vesicles at different suction pressures. We used a model developed by Baumgart et al. (14) based on the analysis of the shape of budded vesicles during their deformation. Basically, the 2 meridional tangent angles ϕ1 and ϕ2 of the lipid and polymer phases, as well as the boundary and pipette radius Rb and Rp are considered (see Fig. 1). The evolution of Rb, ϕ1, and ϕ2 with the suction pressure ΔP is measured. The thermomechanical equilibrium of the budded vesicle is assumed to depend mainly on the lateral tension, the suction pressure ΔP, the vesicle normal pressure difference outside the pipette and the line tension λ. The bending-stiffness contribution is neglected, assuming that the boundary energies are significantly higher than the bending energy (32). The line tension λ can be linearly related to ΔP and a geometric factor A as follows:
| (6) |
The boundary energy was estimated assuming a typical value found in a lipid bilayer membrane for the line tension λ (∼1 pN) and a radius of the circular boundary Rb of 10 μm. This gives a boundary energy of Eb ∼ 2πλRb ∼ 6 × 10−17 J, which is higher than the bending energy for a membrane (∼8πKb): ∼1 × 10−18 J. This confirms the validity of the assumption made in the model used. A more detailed comment about these calculations is available in the Supporting material.
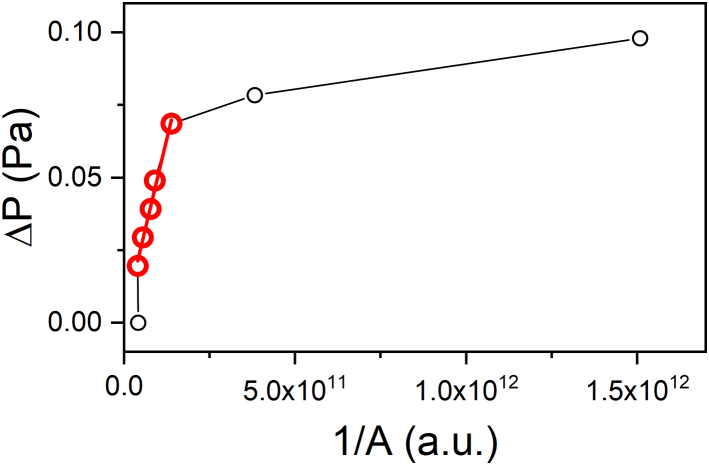
The line tension measurements were carried out using the micropipette set-up described previously. We injected 10 μL of the vesicle suspension in the glucose meniscus before surrounding the meniscus with mineral oil to prevent water evaporation. After 15 min of sedimentation, vesicles deposited at the bottom. The GHUVs showing budding with strong angles were selected. The pipette was gently lowered and approached the lipid-rich side of the membrane with low suction pressure. The pressure was increased to create a significant projection length inside the pipette until the budding disappears. From this position, the pressure was reduced to restore the budded structure. A series of 3–8 images was recorded at different suction steps. The zero pressure was corrected to correspond with the stage where the length projection was no longer visible. From the recorded images, some characteristic dimensions were measured such as the radius at the budding boundary Rb, the radius of the pipette Rp, and the meridional tangent angles Φ1 and Φ2. These parameters are linked to the line tension according to Equation (6). The inverse of the parameter A was plotted as a function of the suction pressure ΔP (Fig. 4), allowing the line tension to be calculated from the slope of the linear part.
Figure 4.
Example of the evolution of the suction pressure as a function of the geometrical parameter (1/A) for the hybrid system POPC/PEO8-b-PDMS22-b-PEO8. The red part corresponds with a linear evolution whose slope allow to determine the line tension.
This experiment was carried out on GHUVs with a POPC content of 50 wt.%. Measurements were performed on 10 budded vesicles for PDMS23-b-PEO13, 16 vesicles for PDMS27-b-PEO17 and 13 vesicles for PEO8-b-PDMS22-b-PEO8.
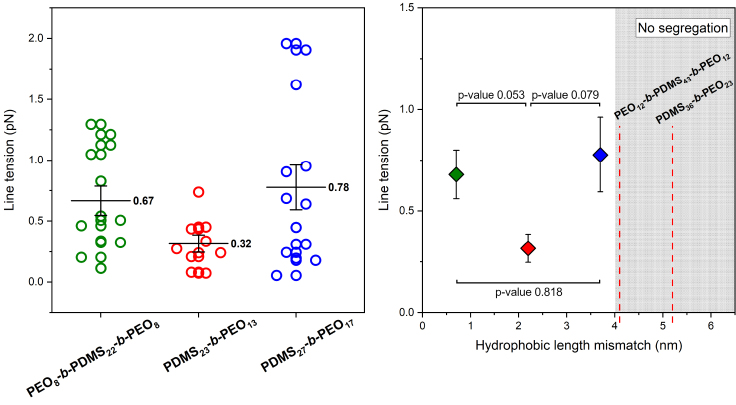
The line tension values are obtained with a relatively high uncertainty, as it is commonly observed for such measurements (Fig. 5). Calculation of the relative error inherent to the experimental procedure on the line tension, as well as statistical tests, was performed to found out significance of the line tension difference between all systems. All this information is available in the Supporting material. We can see that, for diblock copolymers, an increase in the hydrophobic length mismatch leads to an increase in the line tension. This result is in agreement with theoretical simulations (15) and observations in multiphase liposomes (33). The values are in the order of what is observed for multiphase lipid bilayer membranes (9,14), which at first sight can be quite surprising: the compatibility between dimethylsiloxane monomer and alkyl tail is lower than that between alkyl tails of different lipids. Indeed, the solubility parameter of PDMS is δ = 7.3 cal1/2/cm3/2 (34,35), while that of the fatty acid tails in phospholipids is δ = 9.1 cal1/2/cm3/2. It is worth mentioning that the lipid is able to diffuse into the polymer phase to some extent (21). This may contribute the decrease in the incompatibility between the 2 phases and thus the line tension.
Figure 5.
Left) Scatter plot of measurements obtained for PDMS23-b-PEO13/POPC (red dots), PDMS27-b-PEO17/POPC (blue dots) and PEO8-b-PDMS22-b-PEO8/POPC (green dots) (data: mean (value indicated) ± SE). (Right) Evolution of the line tension as a function of the hydrophobic length mismatch (data: mean ± SE; Brown-Forsythe and Welch test; Games-Howell's multiple comparisons post-test, p values are indicated). The red dotted line in the shaded area corresponds with the thickness mismatch for other copolymers, for which hybrid vesicles were obtained without lateral phase separation whatever the lipid composition.
Another interesting observation is that the line tension for PEO8-b-PDMS22-b-PEO8 is slightly higher than for PDMS23-b-PEO13, despite a slightly lower hydrophobic length mismatch. In fact, its line tension is closer to that of PDMS27-b-PEO17, whereas they have almost a 3-nm difference in hydrophobic length mismatch. This suggests that the adaptation between the polymer and the lipid at the boundaries depends on the architecture of the copolymer used. It has been established in the literature that diblock copolymers exhibit exclusively a loop conformation, ensuring a bilayer membrane, while a mixture of extended conformation and loop is observed in membrane composed of triblock copolymers (36,37). These conformational differences may be the cause of a less efficient chain adaptation at the polymer/lipid boundaries.
Finally, this could explain the differences in phase diagrams that have been established in previous studies for GHUVs obtained with a mixture of POPC and these diblock and triblock copolymers (18,19). For example, for a same lipid composition of 30 wt.% a majority of GHUVs made from triblock copolymers PEO8-b-PDMS22-b-PEO8 show micrometer-size domains, whereas only 10% of the GHUVs present a phase separation in the case of diblock PDMS23-b-PEO13 (18,19). Given the higher line tension for PEO8-b-PDMS22-b-PEO8/POPC, the system tends to form lipid microdomains more easily when increasing the lipid fraction. These domains are formed by the coalescence of nanodomains, in analogy to what has been observed with multiphase lipid vesicles (38), to limit the boundary energy cost. For higher hydrophobic length mismatch, both triblock and diblock copolymers do not form hybrid vesicles with micrometric lipid domains. The incompatibility and the line tension (although not measurable in this case) are probably too high. Above a given lipid fraction, only liposomes and polymersomes (containing a small amount of lipid) are formed, resulting from a fast budding fission process.
Conclusion
In this work, we report the first experimental measurements of line tension in hybrid polymer/lipid vesicles. The values are in the range of what is observed for multiphase lipid vesicles. This explains why such structures can be obtained relatively easily, which was not obvious until recently. In addition to the hydrophobic length mismatch effect, the effect of the block copolymer architecture is important on the line tension and, consequently, on the membrane structuration of these hybrid vesicles. This information is essential for the development of this type of synthetic cells where control of the transport of molecules across the membrane is a challenge. The determination of the line tension in polymer/lipid hybrid systems is an important step toward a better understanding of the structure and properties of hybrid membrane. This will make it possible in the future to design of functional artificial cells based on complex mixtures of polymers and lipids.
Author contributions
M.F.: Experiments and data analysis. Emmanuel Ibarboure: experiments and data analysis. J.F.L.M.: Funding acquisition, research orientation, methodology, writing manuscript.
Acknowledgments
M.F. gratefully acknowledges the Scientific Department of University of Bordeaux for a PhD fellowship.
Editor: Tommy Nylander.
Footnotes
Supporting material can be found online at https://doi.org/10.1016/j.bpj.2021.12.005.
Supporting material
References
- 1.Rideau E., Dimova R., et al. Landfester K. Liposomes and polymersomes: a comparative review towards cell mimicking. Chem. Soc. Rev. 2018;47:8572–8610. doi: 10.1039/c8cs00162f. [DOI] [PubMed] [Google Scholar]
- 2.Marušič N., Otrin L., et al. Dimova R. Constructing artificial respiratory chain in polymer compartments: insights into the interplay between bo3 oxidase and the membrane. Proc. Natl. Acad. Sci. U S A. 2020;117:15006. doi: 10.1073/pnas.1919306117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kleineberg C., Wolfer C., et al. Vidakovic-Koch T. Light-driven ATP regeneration in diblock/grafted hybrid vesicles. ChemBioChem. 2020 doi: 10.1002/cbic.201900774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Itel F., Najer A., et al. Meier W. Dynamics of membrane proteins within synthetic polymer membranes with large hydrophobic mismatch. Nano Lett. 2015;15:3871–3878. doi: 10.1021/acs.nanolett.5b00699. [DOI] [PubMed] [Google Scholar]
- 5.Yang S.-T., Kiessling V., Tamm L.K. Line tension at lipid phase boundaries as driving force for HIV fusion peptide-mediated fusion. Nat. Commun. 2016;7:11401. doi: 10.1038/ncomms11401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Munro S. Lipid rafts: elusive or illusive? Cell. 2003;115:377–388. doi: 10.1016/s0092-8674(03)00882-1. [DOI] [PubMed] [Google Scholar]
- 7.Levental I., Levental K.R., Heberle F.A. Lipid rafts: controversies resolved, mysteries remain. Trends Cell Biol. 2020;30:341–353. doi: 10.1016/j.tcb.2020.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Garcia-Saez A.J., Chiantia S., Schwille P. Effect of line tension on the lateral organization of lipid membranes. J. Biol. Chem. 2007;282:33537–33544. doi: 10.1074/jbc.M706162200. [DOI] [PubMed] [Google Scholar]
- 9.Usery R.D., Enoki T.A., et al. Feigenson G.W. Line tension controls liquid-disordered + liquid-ordered domain size transition in lipid bilayers. Biophys. J. 2017;112:1431–1443. doi: 10.1016/j.bpj.2017.02.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Baumgart T., Hess S.T., Webb W.W. Imaging coexisting fluid domains in biomembrane models coupling curvature and line tension. Nature. 2003;425:821–824. doi: 10.1038/nature02013. [DOI] [PubMed] [Google Scholar]
- 11.Jülicher F., Lipowsky R. Shape transformations of vesicles with intramembrane domains. Phys. Rev. E. 1996;53:2670–2683. doi: 10.1103/PhysRevE.53.2670. [DOI] [PubMed] [Google Scholar]
- 12.Esposito C., Tian A., et al. Baumgart T. Flicker spectroscopy of thermal lipid bilayer domain boundary fluctuations. Biophys. J. 2007;93:3169–3181. doi: 10.1529/biophysj.107.111922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Honerkamp-Smith A.R., Cicuta P., et al. Keller S.L. Line tensions, correlation lengths, and critical exponents in lipid membranes near critical points. Biophys. J. 2008;95:236–246. doi: 10.1529/biophysj.107.128421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tian A., Johnson C., et al. Baumgart T. Line tension at fluid membrane domain boundaries measured by micropipette aspiration. Phys. Rev. Lett. 2007;98:208102. doi: 10.1103/PhysRevLett.98.208102. [DOI] [PubMed] [Google Scholar]
- 15.Kuzmin P.I., Akimov S.A., et al. Cohen F.S. Line tension and interaction energies of membrane rafts calculated from lipid splay and tilt. Biophys. J. 2005;88:1120–1133. doi: 10.1529/biophysj.104.048223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Schulz M., Binder W.H. Mixed hybrid lipid/polymer vesicles as a novel membrane platform. Macromol. Rapid Comm. 2015;36:2031–2041. doi: 10.1002/marc.201500344. [DOI] [PubMed] [Google Scholar]
- 17.Le Meins J.F., Schatz C., et al. Sandre O. Hybrid polymer/lipid vesicles: state of the art and future perspectives. Mater. Today. 2013;16:397–402. [Google Scholar]
- 18.Dao T.P.T., Fernandes F., et al. Le Meins J.-F. Modulation of phase separation at the micron scale and nanoscale in giant polymer/lipid hybrid unilamellar vesicles (GHUVs) Soft Matter. 2017;13:627–637. doi: 10.1039/c6sm01625a. [DOI] [PubMed] [Google Scholar]
- 19.Fauquignon M., Ibarboure E., Le Meins J.-F. Membrane reinforcement in giant hybrid polymer lipid vesicles achieved by controlling the polymer architecture. Soft Matter. 2021;17:83–89. doi: 10.1039/d0sm01581d. [DOI] [PubMed] [Google Scholar]
- 20.Fauquignon M., Ibarboure E., et al. Le Meins J.-F. Large and giant unilamellar vesicle(s) obtained by self-assembly of poly(dimethylsiloxane)-b-poly(ethylene oxide) diblock copolymers, membrane properties and preliminary investigation of their ability to form hybrid polymer/lipid vesicles. Polymers. 2019;11:2013. doi: 10.3390/polym11122013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dao T.P.T., Brûlet A., et al. Le Meins J.F. Mixing block copolymers with phospholipids at the nanoscale: from hybrid polymer/lipid wormlike micelles to vesicles presenting lipid nanodomains. Langmuir. 2017;33:1705–1715. doi: 10.1021/acs.langmuir.6b04478. [DOI] [PubMed] [Google Scholar]
- 22.Angelova M.I., Dimitrov D.S. Liposome electroformation, Faraday discuss. Chem. Soc. 1986;81:303–311. [Google Scholar]
- 23.Bermudez H., Hammer D.A., Discher D.E. Effect of bilayer thickness on membrane bending rigidity. Langmuir. 2004;20:540–543. doi: 10.1021/la035497f. [DOI] [PubMed] [Google Scholar]
- 24.Evans E., Rawicz W. Entropy-driven tension and bending elasticity in condensed-fluid membranes. Phys. Rev. Lett. 1990;64:2094–2097. doi: 10.1103/PhysRevLett.64.2094. [DOI] [PubMed] [Google Scholar]
- 25.Fa N., Marques C., et al. Schröder A. Rheology of giant vesicles: a micropipette study. Phys. Rev. Lett. 2004;92:108103. doi: 10.1103/PhysRevLett.92.108103. [DOI] [PubMed] [Google Scholar]
- 26.Dimova R. Recent developments in the field of bending rigidity measurements on membranes. Adv. Colloid Interface Sci. 2014;208:225–234. doi: 10.1016/j.cis.2014.03.003. [DOI] [PubMed] [Google Scholar]
- 27.Ly H.V., Block D.E., Longo M.L. Interfacial tension effect of ethanol on lipid bilayer rigidity, stability, and area/molecule:A micropipet aspiration approach. Langmuir. 2002;18:8988–8995. doi: 10.1021/la026010q. [DOI] [Google Scholar]
- 28.Rawicz W., Olbrich K.C., et al. Evans E. Effect of chain length and unsaturation on elasticity of lipid bilayers. Biophys. J. 2000;79:328–339. doi: 10.1016/S0006-3495(00)76295-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nagle J.F., Jablin M.S., et al. Akabori K. What are the true values of the bending modulus of simple lipid bilayers? Chem. Phys. Lipids. 2015;185:3–10. doi: 10.1016/j.chemphyslip.2014.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Shchelokovskyy P., Tristram-Nagle S., Dimova R. Effect of the HIV-1 fusion peptide on the mechanical properties and leaflet coupling of lipid bilayers. New J. Phys. 2011;13:25004. doi: 10.1088/1367-2630/13/2/025004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Winzen S., Bernhardt M., et al. Kroeger A. Submicron hybrid vesicles consisting of polymer-lipid and polymer-cholesterol blends. Soft Matter. 2013;9:5883–5890. doi: 10.1039/c3sm50733e. [DOI] [Google Scholar]
- 32.Allain J.-M., Ben Amar M. Budding and fission of a multiphase vesicle. Eur. Phys. J. E Soft Matter. 2006;20:409–420. doi: 10.1140/epje/i2006-10030-4. [DOI] [PubMed] [Google Scholar]
- 33.Blanchette C.D., Lin W.-C., et al. Longo M.L. Domain nucleation rates and interfacial line tensions in supported bilayers of ternary mixtures containing galactosylceramide. Biophys. J. 2008;94:2691–2697. doi: 10.1529/biophysj.107.122572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Roth M. Solubility parameter of poly(dimethyl siloxane) as a function of temperature and chain length. J. Polym. Sci. B. 1990;28:2719. [Google Scholar]
- 35.King J.W. In: Lipid Biotechnology. Kuo T.M., Gardner H.W., editors. Marcel Dekker; 2002. Supercritical fluid technology for lipid extraction, fractionation and reactions; pp. 663–687. [Google Scholar]
- 36.Itel F., Chami M., et al. Meier W. Molecular organization and dynamics in polymersome membranes: a lateral diffusion study. Macromolecules. 2014;47:7588–7596. doi: 10.1021/ma5015403. [DOI] [Google Scholar]
- 37.Tsai H.-C., Yang Y.-L., et al. Tsao H.-K. Formation of asymmetric and symmetric hybrid membranes of lipids and triblock copolymers. Polymers. 2020;12:639. doi: 10.3390/polym12030639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Heberle F.A., Petruzielo R.S., et al. Katsaras J. Bilayer thickness mismatch controls domain size in model membranes. J. Am. Chem. Soc. 2013;135:6853–6859. doi: 10.1021/ja3113615. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.